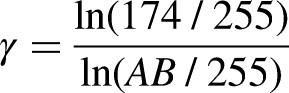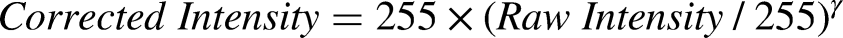Abstract
Background
Peritonitis is a severe complication in peritoneal dialysis (PD), necessitating prompt diagnosis and treatment. A key diagnostic indicator is elevated white blood cells (WBC) in spent PD effluent. Laboratory-based WBC testing can be slow, costly, and infrastructure-dependent, potentially delaying treatment. Urine dipsticks, commonly used for urinary tract infections, include a leukocyte esterase (LE) test that may serve as a rapid and low-cost point-of-care alternative. We evaluated the effectiveness of using an LE test of urine dipsticks to detect WBC in PD effluent for diagnosing PD-associated peritonitis.
Methods
Our research comprised a clinical and a laboratory part. Clinically, we analyzed data from Khartoum, Sudan, where LE dipsticks are routinely given to PD patients for point-of-care testing of PD effluent. In a laboratory, we compared dipstick results with WBC counts in stored, freeze–thawed PD effluents from Australia and the United States, and evaluated smartphone imaging of dipstick coloration.
Results
The clinical evaluation comprised 56 pediatric PD patients providing 530 PD effluent samples. Dipstick LE testing of freshly collected effluents demonstrated 100% sensitivity and 100% specificity for peritonitis diagnosis. In the laboratory study, testing of 352 freeze–thawed PD effluents showed reduced sensitivity (78%) and specificity (95%) after 1 month of refrigerator storage. Smartphone imaging reliably supported visual inspection and enabled semi-quantitative color assessment.
Conclusions
When used on freshly collected PD effluents, the LE test on urine dipsticks is a rapid, reliable, and affordable tool for diagnosing PD-associated peritonitis. This approach is particularly valuable in low-resource settings, home-based care, and telehealth.
Introduction
Peritonitis is a severe complication in peritoneal dialysis (PD) patients, caused by bacterial or fungal infections.1,2 Early diagnosis and treatment of peritonitis is critically important to prevent morbidity and mortality. 3 Per International Society for Peritoneal Dialysis (ISPD) guidelines, the diagnosis of peritonitis requires the presence of at least two of the following three criteria: (1) clinical signs or symptoms indicative of peritonitis (e.g. abdominal pain, cloudy dialysis effluent); (2) dialysis effluent white blood cell (WBC) count >100 cells/μL (after a dwell time of at least 2 h), with >50% polymorphonuclear leukocytes (PMN); and (3) a positive effluent culture. 4 However, laboratory tests can be time-consuming, costly, and require specialized facilities, potentially delaying diagnosis and treatment, and thus compromising patient outcomes.
Urine dipsticks are widely used as point-of-care screening tools for urinary tract infections (UTIs).5,6 These dipsticks typically include a leukocyte esterase (LE) colorimetric test, which primarily detects granulocytic esterases, specifically those released by activated neutrophils. In detail, an LE test pad contains an indoxylcarbonic acid ester or an amino acid ester that is rapidly hydrolyzed by LE, producing a compound that reacts with a diazonium salt on the test pad to generate a distinct color within seconds to minutes. 7 A positive test result indicates elevated urine PMN, suggesting UTI.
In recent years, LE tests have been demonstrated to be effective in detecting infections beyond UTIs, including periprosthetic joint infections, septic arthritis, and peritonitis.8–11 Additionally, six studies have specifically explored their utility in PD-associated peritonitis.12–17 In this study, we conducted a broader investigation to evaluate and quantify the effectiveness of LE dipsticks for PD-associated peritonitis diagnosis. First, we analyzed hitherto unreported real-world data from Soba University Hospital in Sudan, a low-resource setting where LE testing guides early antibiotic treatment decisions. Second, to further assess performance under controlled conditions, we conducted a systematic laboratory study of LE tests using freeze–thawed PD effluent samples from patients in Australia and the United States. In addition to visual inspection of LE test pads, we evaluated the use of smartphone imaging to quantify WBC levels based on LE pad coloration.
Methods
Studies and patient cohorts
Cohort 1 (Sudan)
The study was approved by the Soba University Hospital (SUH) Ethics Committee (protocol PD/002-2/2015) in Khartoum, Sudan. Verbal or written informed consent was obtained from all participants or their parents/guardians. All patients aged 18 years or younger who were admitted to, or underwent continuous ambulatory peritoneal dialysis at the Pediatric Nephrology Unit of SUH between January 2015 and December 2020 were approached and invited to participate. Patients for whom informed consent could not be obtained were excluded from the study. A total of 56 pediatric patients participated in the study.
Cohort 2 (United States)
The study was approved by the Western Institutional Review Board (RRI-19-002) in the United States. Eligible participants need to understand and provide written informed consents, be at least 18 years of age, and be receiving PD. Individuals who were unable to communicate in English or Spanish were excluded from participation. A total of 30 patients participated in the study.
Cohort 3 (Australia)
This IRB-approved study (WA34201) was conducted at the University of Western Australia between 2021 and 2024. PD effluent samples with sufficient volume were included if they were obtained from adult patients undergoing PD, who presented with clinical suspicion of PD-associated peritonitis and whose specimens were submitted to the clinical microbiology laboratory for diagnostic evaluation. Samples from patients who had already received antimicrobial therapy prior to collection were excluded. A total of 218 patients participated in the study.
Sample collection and testing
Cohort 1
In total, 530 PD effluent samples were collected after a 2-h dwell, including 56 initial samples and 474 samples during an up-to-3-week follow-up period. Immediately after sample collection, a urine dipstick (Multistix® 10 SG, Siemens Ltd, USA) was dipped into the PD effluent for 1–2 s. After 90 to 120 s, the color of the LE test pad was compared with reference color fields on the dipstick container. Results matching or darker than the “MODERATE++” reference were considered positive. Within 2 h of collection, the clinical laboratory quantified WBC and PMN counts and plated microbiological cultures for all 56 initial and 53 follow-up samples. The remaining 421 follow-up samples had dipstick testing only, without WBC counts.
Cohort 2
In total, 154 spent PD effluent samples were collected between 2020 and 2024. Each patient contributed 1 to 25 samples (median: 2). Fifteen samples with short dwells (<2 h) were excluded. The remaining 139 samples had dwell times between 120 min and 615 min (median: 120 min). Three patients with peritonitis each contributed one initial sample; two of these patients also provided a follow-up sample collected 2 days later. The remaining 134 samples were obtained from patients without peritonitis, with at least 1 month between samples collected from the same individual.
Samples were transported on ice pack to our New York research laboratory within 24 h of collection. WBC and PMN were counted using an ABX Pentra XL80 hematology analyzer (Horiba Medical, Irvine, California, USA), after concentrating the cells by a factor of 1 to 10,000. Samples with more than 100 WBC/μL were plated for bacterial culture. All samples were aliquoted and stored at −80 °C upon arrival. On the test day, samples were thawed and tested together in the New York laboratory using LotFancy urine dipsticks (11J-2136-D.1, California, USA).
Cohort 3
Each patient provided one PD effluent sample, resulting in a total of 218 samples. All samples were collected prior to any antimicrobial exposure. No follow-up samples were obtained. WBC, PMN count, and microbiological culture were analyzed in a local clinical laboratory within 24 h of sample collections. Five samples clotted before the cell counts were excluded from the study. All samples were stored at 4 °C for approximately 1 month and then frozen at −80 °C. In June 2024, all samples were thawed, aliquoted, refrozen, and shipped on dry ice to our New York research laboratory, where they were stored at −80 °C. On the test day, samples were thawed and tested together in the New York laboratory using LotFancy urine dipsticks (11J-2136-D.1, California, USA).
WBC purification from whole blood for preparation of WBC standards
Whole blood was collected from healthy donors in ethylenediaminetetraacetic acid tubes and subjected to red blood cell (RBC) lysis using RBC lysis buffer (Thermo Fisher J62990, USA) at 1:10 ratio (blood:buffer) for 15 min at room temperature. The suspension was centrifuged at 500×g for 5 min. The pellet was washed once with 1× phosphate-buffered saline (PBS, pH 7.2) and resuspended in 1× PBS. WBC and PMN were counted by the Pentra XL80 hematology analyzer. PMN percentages ranged from 48% to 65% across different batches.
Quantitative evaluation of LE tests using freeze–thawed PD effluent samples
Positive and negative controls
For the negative control (NC), a buffer containing 1.06% dextrose was prepared by mixing 4.25% dextrose DELFLEX® (Fresenius Medical Care 054-20224, USA) and 1× PBS (pH 7.2) in a 1:3 ratio. For the positive control (PC), purified WBC were diluted to 100 cells/µL (i.e. the WBC threshold per ISPD guidelines) using the NC buffer, aliquoted, and stored at −80 °C until analysis.
Test setup
Aliquots of samples and PCs were thawed right before testing. One PC, one NC, and six samples (250 µL each) in Cryo biobanking tubes (Greiner 976561, Germany) were arranged in a row and thoroughly mixed by pipetting. The LE pads of eight urine dipsticks (LotFancy, 11J-2136-D.1, California, USA) were simultaneously dipped into samples for 2 s (Supplemental Figure 1A), then laid flat while a video of the LE pads was recorded for 10 min with a Samsung Galaxy S9 smartphone camera app (Supplemental Figure 1B).
Data analysis
Still images were extracted from videos at specific time points. The mean 8-bit grayscale pixel intensity of each LE pad was measured using ImageJ (https://imagej.net/ij/), and the maximum brightness (white) corresponds to value 255. The blank area above each dipstick was designated as the ambient background (AB) for that dipstick.
To minimize the effect of ambient light, all measured intensities were subject to gamma correction. Specifically, the background intensity of all dipsticks was standardized to 174 (medium AB intensity). The gamma correction factor (γ) for each dipstick was calculated as:
The gamma-corrected LE pad intensity was calculated as:
Supplemental Figure 1C shows an example image extracted from a video at the 5-min time point, with dipstick 1 representing a bacterial peritonitis case. The relative intensity of each LE test was calculated as:
Statistical analysis
All statistical analyses were performed using R Statistical Software v4.3.0. 18 Figure was generated in R using the ggplot package. Paired t-tests were used to compare WBC counts across sequential time points in non-peritonitis patients (day 0 vs. day 1; day 1 vs. day 2) and in peritonitis patients (day 0 vs. days 3–5). Welch's t-test was used to compare day-0 WBC counts between peritonitis and non-peritonitis groups. Statistical significance was defined as p < 0.05.
Results
Clinical assessment in cohort 1 (Sudan)
The 56 pediatric PD patients were categorized into two groups per ISPD guidelines, 20 with peritonitis and 36 without. Patient characteristics are summarized in Table 1. Because of sepsis, 24 patients (6 in peritonitis and 18 in non-peritonitis group) had received antibiotics (including vancomycin, gentamycin, ampicillin, ceftriaxone, artesunate, meropenem) 1 day to 4 days prior to the initial dipstick test and WBC counts. Groups did not differ regarding age, sex, cause of dialysis, and prior antibiotic use. Intermittent peritoneal dialysis was significantly more common in the non-peritonitis group (Table 1).
Patient characteristics of cohort 1 (Sudan).
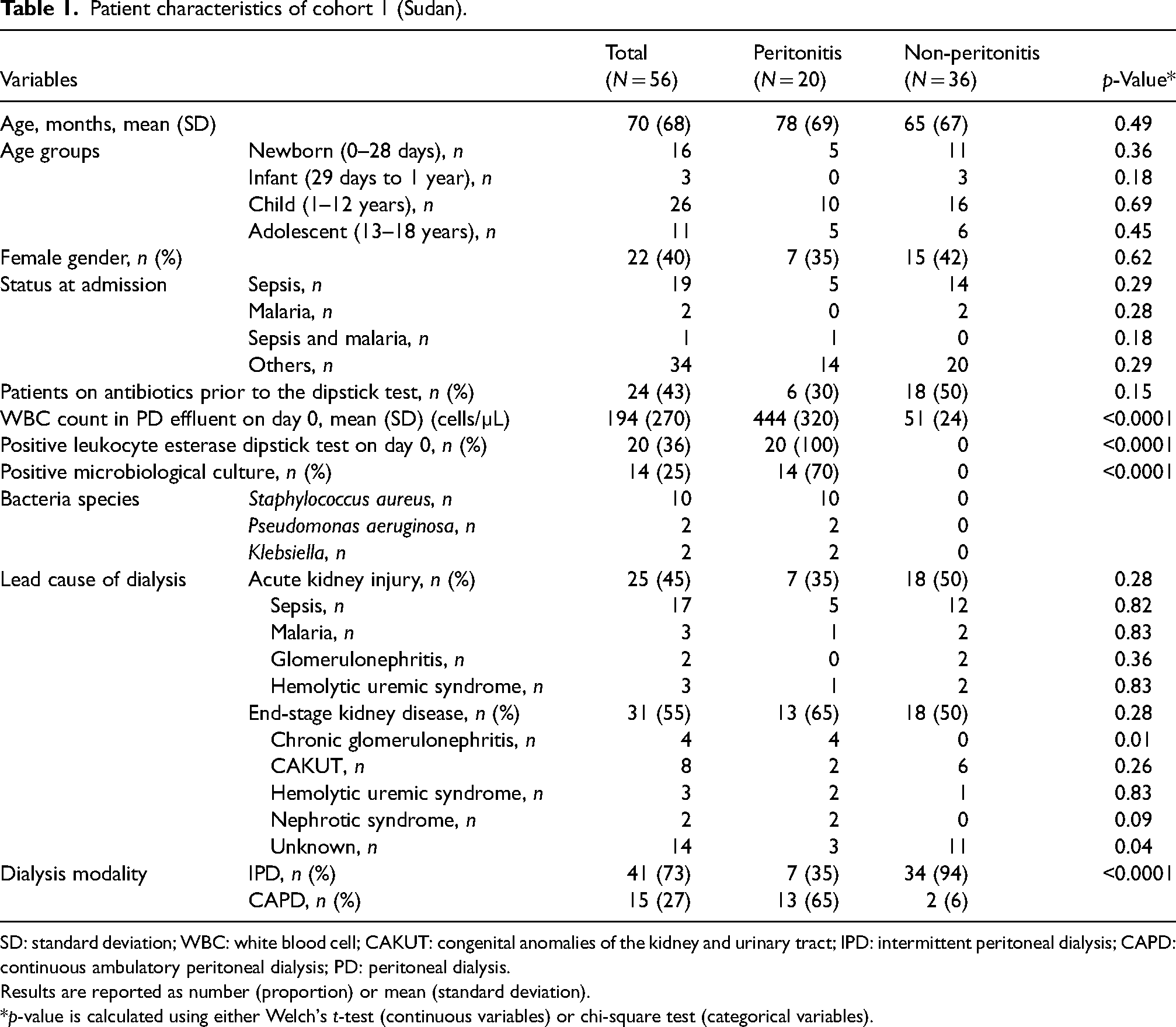
SD: standard deviation; WBC: white blood cell; CAKUT: congenital anomalies of the kidney and urinary tract; IPD: intermittent peritoneal dialysis; CAPD: continuous ambulatory peritoneal dialysis; PD: peritoneal dialysis.
Results are reported as number (proportion) or mean (standard deviation).
*p-value is calculated using either Welch's t-test (continuous variables) or chi-square test (categorical variables).
Peritonitis group (N = 20)
The day of the first dipstick test for peritonitis screening was defined as day 0. All 20 PD effluent samples from the peritonitis group were LE-positive on day 0, indicating 100% sensitivity for fresh samples (Table 2, row 1). Their WBC count (mean ± SD) on day 0 was 440 ± 320 cells/µL (median: 432, range: 98–1018). Fifteen follow-up samples with WBC counts were available from 15 patients between day 3 and day 5. Among these, six tested LE-negative and had WBC counts below 100 cells/µL (median: 70; range: 15–86); nine tested LE-positive, with all WBC counts exceeding 100 cells/µL (median: 388; range: 101–513). WBC counts significantly declined between day 0 (median: 365 cells/µL) and follow-up samples (median: 113 cells/µL; p = 0.0003, paired t-test) (Figure 1, peritonitis group). All peritonitis patients received either vancomycin + gentamycin or cefazolin + ceftazidime for 14 days to 21 days, except for one patient who received fewer than 5 days of antibiotics before leaving the hospital. During treatment period, an additional 253 PD effluent samples were collected and tested only by dipsticks, without WBC counts. Each patient contributed a median of 10.5 samples (range: 5–30), consisting of a median of 6 LE-positive samples (range: 3–18) and 4 LE-negative samples (range: 0–12). Nineteen patients eventually converted to LE-negative and remained LE-negative for at least two consecutive tests. They were considered recovered. The patient who left early had five consecutive LE-positive results and no follow-up was available.
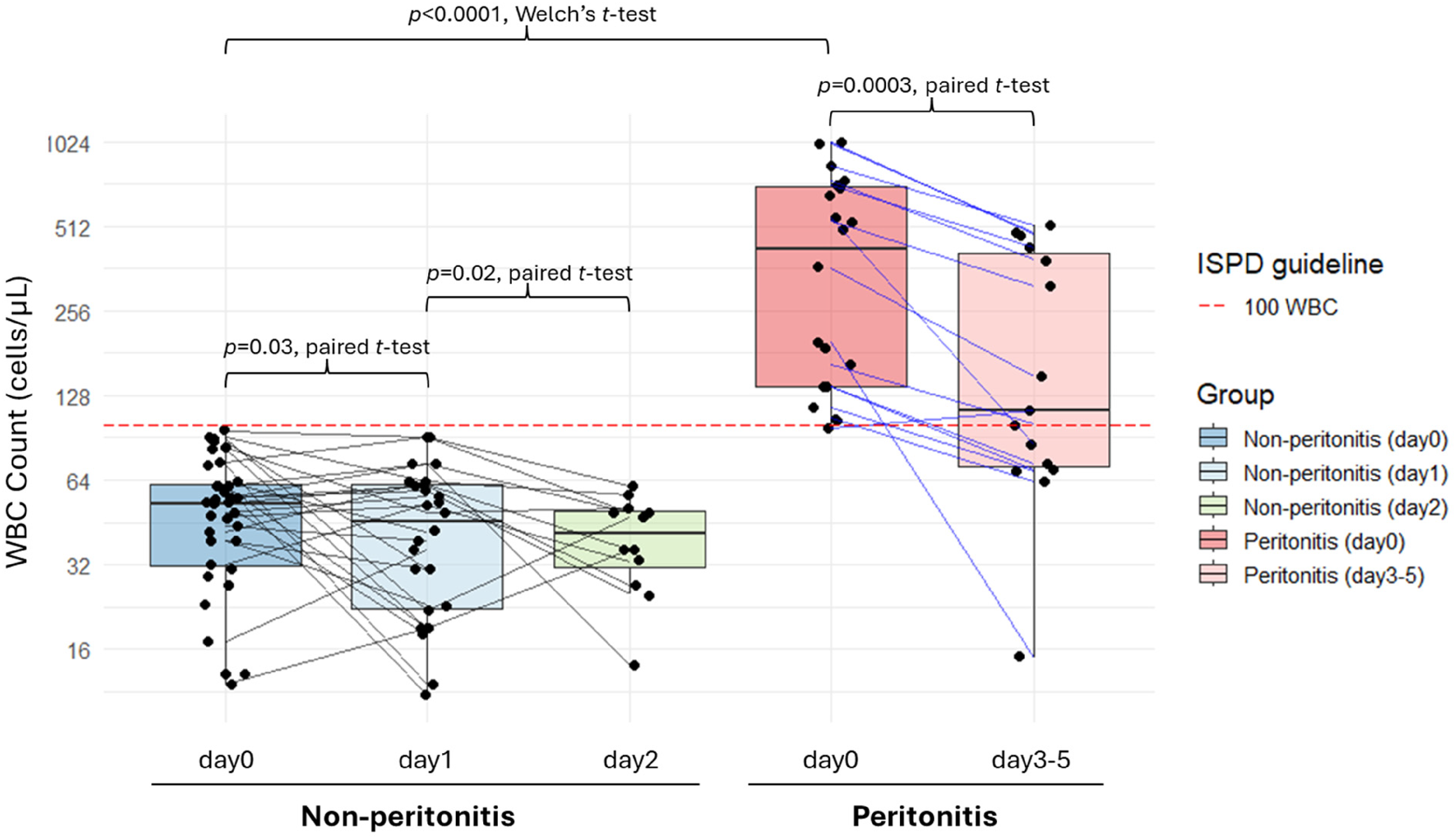
WBC counts in peritonitis and non-peritonitis patients from cohort 1. The red dashed line marks the ISPD-recommended diagnostic threshold of 100 WBC/μL. On the left, three box plots show WBC counts from non-peritonitis patients at day 0 (initial LE dipstick testing), day 1, and day 2; individual patient trajectories are connected by solid black lines. On the right, two box plots show WBC counts from peritonitis patients at day 0 and days 3–5; individual patient trajectories are connected by solid blue lines. Note that the y-axis is displayed on a log2 scale. WBC: white blood cell; LE: leukocyte esterase.
Evaluation of leukocyte esterase dipstick performance for diagnosing PD-associated peritonitis in three cohorts.
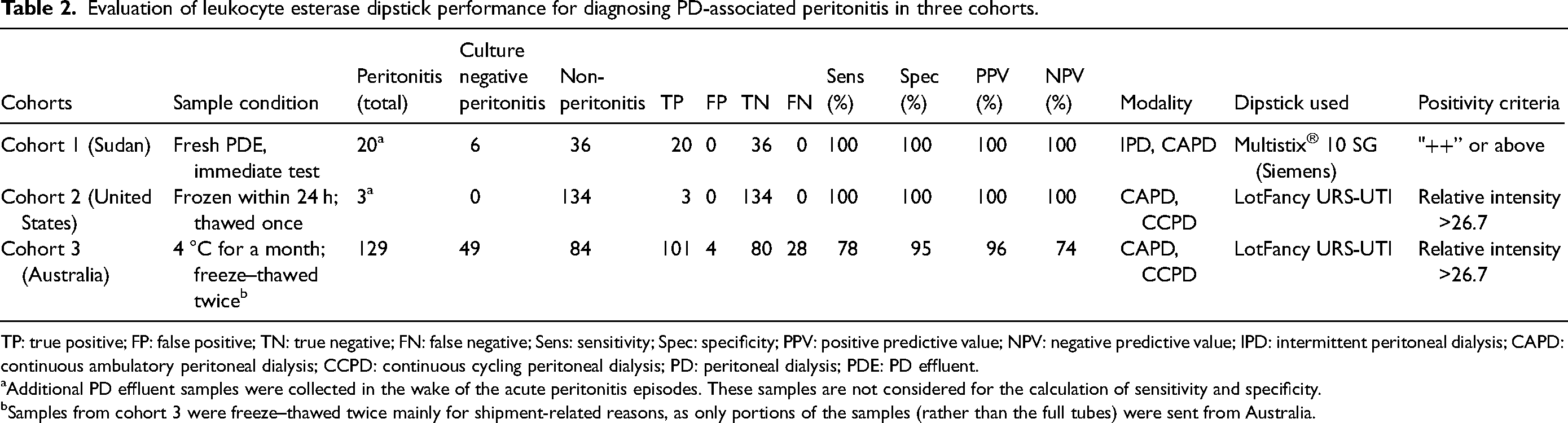
TP: true positive; FP: false positive; TN: true negative; FN: false negative; Sens: sensitivity; Spec: specificity; PPV: positive predictive value; NPV: negative predictive value; IPD: intermittent peritoneal dialysis; CAPD: continuous ambulatory peritoneal dialysis; CCPD: continuous cycling peritoneal dialysis; PD: peritoneal dialysis; PDE: PD effluent.
Additional PD effluent samples were collected in the wake of the acute peritonitis episodes. These samples are not considered for the calculation of sensitivity and specificity.
Samples from cohort 3 were freeze–thawed twice mainly for shipment-related reasons, as only portions of the samples (rather than the full tubes) were sent from Australia.
Non-peritonitis group (N = 36)
In this group, on day 0, PD effluent samples from all 36 patients tested LE-negative, indicating 100% specificity (Table 2, row 1). The WBC count was 51 ± 24 cells/µL (median: 53, range: 12–96). On day 1, LE tests and WBC counts were available in 26 patients. All were LE-negative and showed WBC counts <100 cells/µL (median: 46, range: 11–91). Twelve patients continued testing on day 2; all samples were LE-negative and had WBC counts <100 cells/µL (median: 42; range: 14–61) (Figure 1, non-peritonitis group). Additional 168 samples were collected every 1–2 days from all 36 non-peritonitis patients for 7 days. All these samples tested LE-negative; WBC counts were not measured.
WBC counts in PD effluent samples on day 0 were significantly higher in the peritonitis versus the non-peritonitis groups (median 431 vs. 53 WBC/µL; p < 0.0001, Welch's t-test) (Figure 1).
LE pad intensity in relation to WBC concentration and time
To explore the quantitative assessment of LE dipstick results by a smartphone camera and imaging software that quantitates LE test pad coloration, we compared LE pad intensities using fresh WBC at concentration ranging from 25 to 1600 WBC/µL, alongside a PC (100 WBC/µL, freeze–thawed once) (Figure 2). The results showed a dose- and time-dependent increase in signal intensity. As expected, lower WBC concentrations exhibited a more linear increase in signal intensity over time, whereas higher WBC concentrations reached signal saturation sooner. Notably, the signal intensity of freeze–thawed samples (100 WBC/µL) was consistently higher than that of fresh WBC from the same preparation (Figure 2, blue dotted line vs. blue solid line).
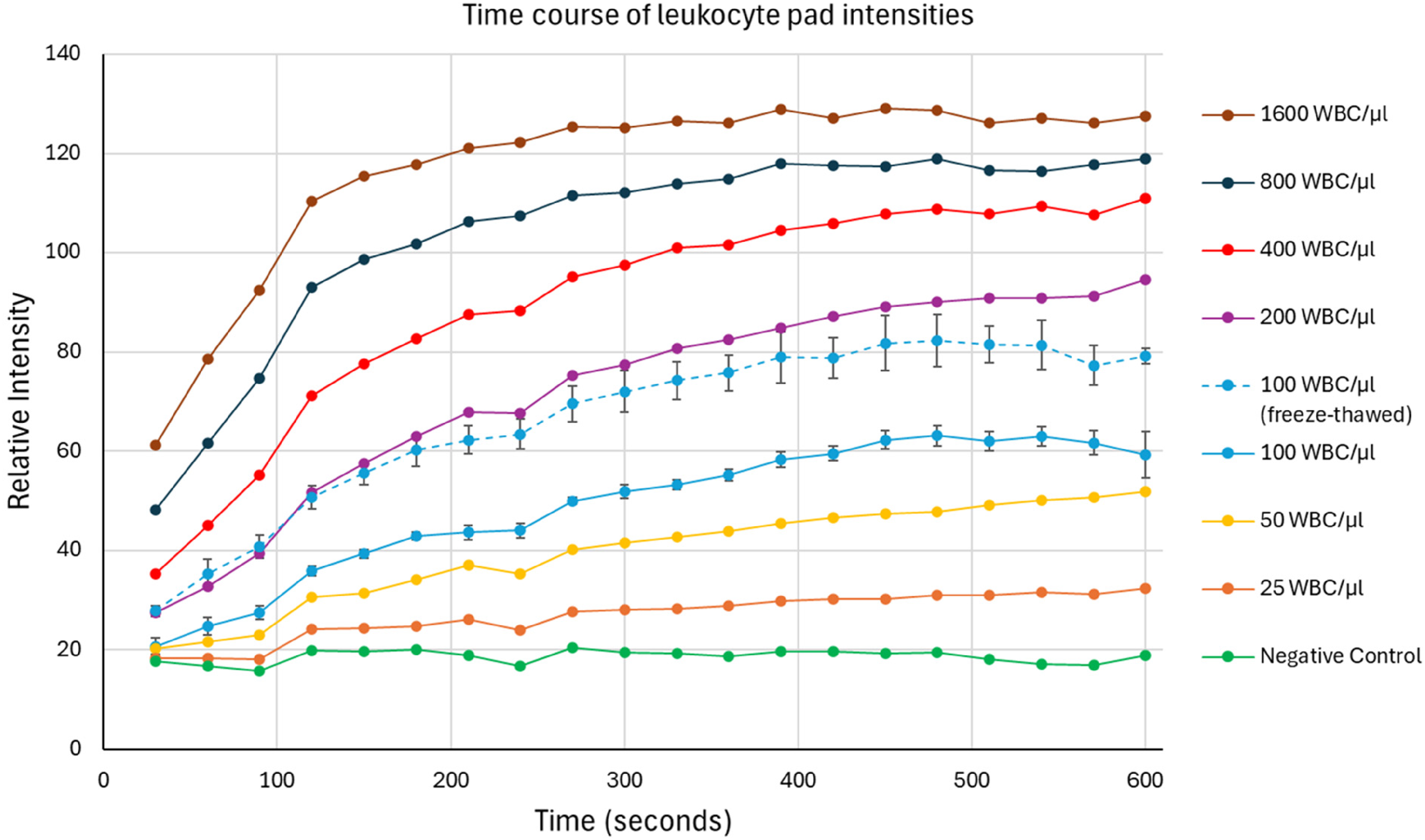
Quantitative leukocyte esterase dipstick results. Fresh WBC ranging from 25/µL to 1600/µL, along with a negative control (1.06% dextrose, pH 7.2), were tested over a 10-min development period. In addition, four positive controls (100 WBC/µL, freeze–thawed once) and four fresh WBC at 100 cells/µL were tested separately. Data for 100 cells/μL represent the average of 4–5 replicate dipsticks, while results for all other concentrations are based on single-dipstick tests. WBC: white blood cell.
Evaluation of PD effluent samples that were frozen within 24 h
Positivity threshold for frozen PD effluent
A total of 86 dipstick tests were obtained for both the PC (freeze–thawed once) and NC. LE pad intensities were analyzed at the 5-min time point for all tests. The mean relative intensity of the LE pad was 29.3 ± 2.6 for the PC and 19.2 ± 2.3 for the NC. A positivity threshold was set at 26.7, corresponding to the PC's mean relative intensity minus one standard deviation. PD effluent samples with LE pad intensities above this threshold were considered positive.
All 139 PD effluent samples from cohort 2 (United States) were frozen within 24 h of collection and thawed on the day of LE dipstick testing. One hundred and thirty-four samples came from patients without peritonitis; they all tested LE-negative (relative intensity: 21.6 ± 1.9), indicating 100% specificity (Table 2, row 2).
Three patients suffered from PD-associated peritonitis. During the acute phase, they provided one sample each. These were culture-positive for bacteria and had WBC counts of 17,800, 900, and 248 cells/µL, respectively. These samples tested LE-positive, with relative intensities of 148.0, 52.9, and 38.1, resulting in 100% sensitivity (Table 2, row 2). The LE signal was saturated after a few seconds in the sample with 17,800 WBC/µL. Two patients provided one follow-up sample each, one with 17 WBC/µL that tested LE-negative (relative intensity: 22.6), and one with 180 WBC/µL that tested LE-positive (relative intensity: 35.6).
Across both groups, seven samples had WBC of 25 to 1600 cells/µL (i.e. the concentration range we evaluated in Figure 2). A significant linear correlation was observed between WBC concentration and LE pad intensity at 5 min (R2 = 0.87; p = 0.002).
Evaluation of PD effluent samples that were frozen after a month
All 213 PD effluent samples from cohort 3 (Australia) were stored at 4 °C for approximately 1 month prior to freezing. Each sample underwent two freeze–thaw cycles before LE dipstick testing. Of the samples, 129 were classified as peritonitis samples, including 74 that were culture-positive for bacteria, 3 culture-positive for fungi (Candida parapsilosis), 3 culture-positive for both bacteria and fungi (Candida glabrata), and 49 that were culture-negative. Among peritonitis samples, 101 samples tested LE-positive, yielding a test sensitivity of 78% (Table 2, row 3). Sensitivity did not differ between culture-positive and culture-negative peritonitis cases.
Of the 28 false-negative samples, 25 had WBC counts exceeding 100 cells/µL (tested within 24 h of collection). Of note, at the time of LE dipstick testing, all 28 samples appeared clear on visual inspection, and neither cells nor debris were observed by microscopy. We consider it likely that prolonged storage at 4 °C led to cell aggregation and sedimentation, leaving few or no cells in the aliquots subsequently frozen, thereby reducing LE detection sensitivity.
Among the 84 non-peritonitis samples, four showed intensities above the threshold, resulting in a test specificity of 95% (Table 2, row 3). In all four false-positive cases, the LE pad displayed a yellowish hue rather than expected purple. This color difference was distinguishable by eye but not by ImageJ, as it relies on grayscale intensity. This yellowish coloration appeared to originate from the sample itself.
Across both groups, 67 samples had WBC counts between 25 cells/µL and 1600 cells/µL. Unlike cohort 2 samples, they showed a weaker linear correlation between WBC concentration and LE pad intensity (R2 = 0.16, p = 0.001), consistent with the assumption that prolonged storage led to WBC loss or skewed distribution of cells in test aliquots.
Discussion
Each year, millions of patients with end-stage kidney disease die prematurely due to a lack of access to affordable kidney replacement therapy, primarily driven by socio-economic barriers. 19 Reducing the cost of such therapies is urgently needed. We believe that innovations which lower the cost of diagnosing PD-associated peritonitis in a timely and objective way are meaningful to this effort. To this end, we evaluated the use of commercially available urine dipsticks—commonly used for diagnosing UTI—as a tool to detect elevated WBC counts in spent PD effluents. Our study validates the effectiveness of LE dipsticks for diagnosing PD-associated peritonitis. By integrating real-world clinical data with stored PD effluent samples, we provide a robust evaluation of LE dipstick performance in both fresh and stored PD effluent.
Review of studies on LE dipstick for PD peritonitis
Our literature review identified six studies evaluating the diagnostic accuracy of LE dipstick testing for PD-associated peritonitis, comprising 181 peritonitis episodes and 545 non-peritonitis controls (Table 3).12–17 Across these studies, either Multistix (Siemens) or Guardian™ PeriScreen (Serim) reagents were used. Three studies defined a positive result as “trace” or higher, whereas the remaining three applied more stringent thresholds (≥ “+” or ≥ “+++”). Despite these differences, the LE dipstick demonstrated consistently high diagnostic performance. Reported sensitivity ranged from 95% to 100%, with five of six studies showing 100% sensitivity and only a single false-negative result overall. Specificity ranged from 95% to 98%, except for one study that could not be evaluated due to the absence of non-peritonitis cases. When pooled, LE dipstick testing yielded an overall sensitivity of 99%, specificity of 96%, positive predictive value of 89%, and negative predictive value of 100%.
Summary of published studies on leukocyte esterase dipstick performance for diagnosing PD-associated peritonitis.
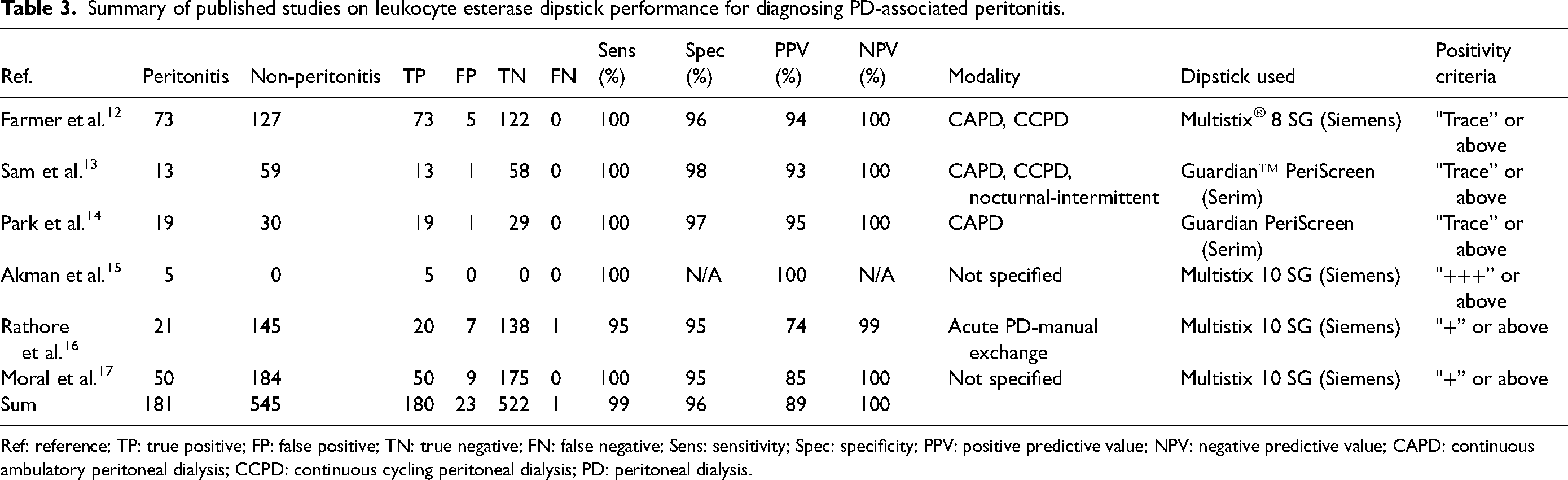
Ref: reference; TP: true positive; FP: false positive; TN: true negative; FN: false negative; Sens: sensitivity; Spec: specificity; PPV: positive predictive value; NPV: negative predictive value; CAPD: continuous ambulatory peritoneal dialysis; CCPD: continuous cycling peritoneal dialysis; PD: peritoneal dialysis.
Clinical experience
When used as a routine point-of-care test at Soba University Hospital in Khartoum, Sudan, LE dipsticks demonstrated 100% sensitivity and 100% specificity for detecting peritonitis in pediatric PD patients. The strong correlation between LE positivity and elevated effluent WBC counts, combined with treatment response, underscores the reliability of LE dipsticks in point-of-care, real-time clinical decision-making. Diagnosing peritonitis in pediatric patients can be particularly challenging, as clinical symptoms are often difficult to interpret or may be absent. For instance, two infants in the cohort showed no signs or symptoms of peritonitis at the time of routine LE screening for peritonitis. Both tested LE-positive, and bacterial cultures confirmed infection the following day. Prompt antibiotic administration on day 0 (i.e. prior to culture confirmation) successfully controlled the infection. By day 4, WBC counts in their PD effluents dropped from 138 cells/µL and 198 cells/µL (day 0) to 73 cells/µL and 15 cells/µL, respectively, and cultures turned negative. Across the entire cohort 1, all peritonitis patients with known follow-up recovered without complications. These outcomes highlight the clinical value of using common urine dipstick in managing PD-associated peritonitis.
In fact, using urine dipsticks is now a common practice for PD pediatric patients in Sudan, especially in rural areas with limited access to healthcare facilities. In these settings, physicians provide patients with urine dipsticks and a 14-day supply of antibiotics, along with training of self-administering intraperitoneal antibiotics. Patients are advised to initiate antibiotic therapy immediately upon experiencing symptoms suggestive of peritonitis combined with a positive LE test result, and to seek prompt evaluation at the nearest healthcare facility. Conversely, if the LE test is negative, patients are advised not to initiate antibiotic therapy without evaluation by a health care professional. This approach supports antimicrobial stewardship by avoiding unnecessary medication, thereby reducing the risk of antimicrobial resistance and minimizing potential adverse effects associated with broad-spectrum antibiotics.
Urine dipsticks have also been implemented in a high-income country. At the Department of Nephrology at Skåne University Hospital, Sweden, dipsticks support patient self-assessment (Dr Giedre Martus, personal communication in 2025). Home PD patients are trained and encouraged to test spent PD dialysate every morning, and whenever they experience signs or symptoms of peritonitis, using the Combur-Test® (Roche Diagnostics, Germany) or the Multistix 5 (Siemens) dipsticks. If the result is LE-positive (“+” or above), patients are advised to contact the clinic promptly for further evaluation. Additionally, the test is used as a tool to monitor the effectiveness of peritonitis treatment. The integration of LE dipsticks into care pathways in both high- and low-resource settings illustrates its wide applicability.
Laboratory testing and smartphone camera use
Automated urine dipstick analyzers, such as the CLINITEK Status®+ urine analyzer (Siemens) for Multistix SG strips, can aid result interpretation. 20 However, these devices are expensive and designed for specific test strips. Given the widespread availability of smartphones and tablets, we also assessed the feasibility of using smartphone cameras to semi-quantitatively analyze LE pad color changes. We observed a strong correlation between LE pad color intensity and WBC concentration in both purified WBC preparations and PD effluent samples. These findings support the development of a dipstick-agnostic mobile application to standardize test pad interpretation and reduce subjectivity—an especially relevant feature since over half of patients with end-stage kidney disease experience visual impairment. 21 This approach could also estimate WBC counts at a certain range (i.e. 25–1600 cells/µL). A standardization is particularly valuable given the variability in positivity thresholds reported across studies (Tables 2 and 3), ranging from “trace” to “+++.” By linking LE test results to WBC counts and contextual factors (such as lapsed reaction time), an app-supported method could enhance clinical decision-making for both physicians and patients. Additionally, the choice of cut-off values may be tailored to local clinical practices, patient-specific conditions, and dialysis variables (i.e. dwell time, fill volume, and PD modality).
To systematically bench test the use of smartphone cameras for LE test pad interpretation, we used previously collected PD effluent samples that had been stored under varying conditions. These samples were not originally collected for the purpose of LE testing, but rather for unrelated research activities, and were therefore processed and stored according to the requirements of their initial studies rather than being tested immediately.
In cohort 2, where samples were frozen within 24 h, the LE test demonstrated 100% sensitivity and 100% specificity. However, in cohort 3, where samples were stored at 4 °C for a month prior to freezing, LE sensitivity declined to 78%, likely due to cell aggregation and sedimentation, resulting in reduced WBC counts in the analyzed aliquots. We also observed that the LE intensity from freeze–thawed isolated WBC at 100/µL was consistently higher than that of fresh WBC, further highlighting the complex effects of sample handling and storage on testing performance.
These observations underscore an important limitation of our study. Because the samples were not tested freshly, the gold-standard comparator (WBC count) may have been affected by cell sedimentation or degradation. The small number of peritonitis cases in cohort 2 also limits the strength of conclusions regarding optimal storage duration. Taking together, these findings indicate that results obtained from stored samples, especially those subjected to prolonged refrigeration, should be interpreted with caution. Nonetheless, these bench-testing data remain informative for the development of a smartphone-based LE application, as they emphasize the need for standardized sample handling in any future prospective validation.
It is worth noting that most common leukocyte dipsticks primarily reflect neutrophil activity, as the specific esters embedded in the pad are preferentially hydrolyzed with granulocytic esterase enzymes, which are most highly concentrated in neutrophil granules. 22 Other leukocyte types, such as monocytes and eosinophils, contain lower levels or different isozymes of these esterases, while lymphocytes likely lack them, often resulting in weak or negative reactions. Therefore, non-neutrophil-predominant infections, such as very early peritonitis, or infections caused by low-virulence organisms, may present with less neutrophils and yield a false-negative LE result.
In our US laboratory, a urine dipstick costs just 6 US cents. Most commercially available dipsticks include multiple reagent pads (e.g. for nitrite, glucose, pH) in addition to LE. However, since only the LE pad is necessary to detect elevated WBC counts in spent PD effluents, a simplified dipstick containing only the LE test could further reduce manufacturing and diagnostic costs.
Other rapid strip tests targeting inflammatory protein markers such as interleukin-6, matrix metalloproteinase-8, and neutrophil gelatinase-associated lipocalin have also been investigated by various research groups for the detection of PD-associated peritonitis.23,24 Whether combining LE dipstick testing with one or more of these biomarkers is cost-efficient, improves diagnostic accuracy, and offers a more robust tool for early detection of PD-associate peritonitis warrants prospective clinical studies.
In summary, our findings indicate that inexpensive, common urine dipsticks are a reliable and rapid method for detecting elevated WBC counts indicative of PD-associated peritonitis, particularly when used on freshly collected PD effluent. While PD effluent with 100 WBC/µL (the threshold for peritonitis per ISPD guidelines) can appear unremarkable at visual inspection, the LE test pad displays a noticeable color change within 2 min. The integration of mobile phone-based WBC quantification offers a promising advancement for point-of-care diagnostics, with potential applications in remote and low-resource settings, telehealth, and home-based care.
Supplemental Material
sj-docx-1-ptd-10.1177_08968608261434277 - Supplemental material for Evaluation of common urine leukocyte dipsticks for diagnosing peritoneal dialysis (PD)-associated peritonitis
Supplemental material, sj-docx-1-ptd-10.1177_08968608261434277 for Evaluation of common urine leukocyte dipsticks for diagnosing peritoneal dialysis (PD)-associated peritonitis by Rasha Hussein, Nadja Grobe, Nahla Allam, Mohamed EO Yousif, Asmaa Abdelkareem, Amna Ahmed, Rashid A Ellidir, Yassir Bakhiet, Mohamed BA Elfakky, El Tigani M Ali, Lisa Haizel Watson, Naomi Taylor, Kieran Trelawn Mulroney, Aron Chakera, Peter Kotanko and Xiaoling Wang in Peritoneal Dialysis International
Footnotes
Acknowledgments
We thank Dr Giedre Martus for sharing her insights on clinical practice guidelines at the home dialysis clinic of Skåne University Hospital, Sweden, and Dr Karin Bergling (Renal Research Institute) for her valuable feedback based on her practical experience while working in a Swedish hospital. Parts of this manuscript were presented at the 2025 World Congress of Nephrology and the 2025 European Renal Association-European Dialysis and Transplant Association (ERA-EDTA) conferences. The authors acknowledge the conference for publishing the abstract in the conference proceedings.
Author contributions
RH, AC, PK, and XW devised the project. RH, NA, MEOY, Asmaa A, Amna A, RAE, YB, MBAE, and ETMA performed the clinical assessment in Sudan; KTM collected patient samples and their associated clinical data in Australia; NG and XW collected patient samples in the United States. LHW, NT, and XW conducted the strip test in the US laboratory. XW analyzed the data. XW wrote the manuscript with input from all authors.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RH, NG, and XW are employees of Fresenius Medical Care. PK holds stock in Fresenius Medical Care and is a former employee of the Renal Research Institute, a wholly owned subsidiary of Fresenius Medical Care. KTM is a major shareholder and has an operational role in Cytophenix—an early-stage Australian spinout company working on antimicrobial susceptibility testing. The other authors declare no competing interests.
Ethical considerations
Ethics approval details are provided in the Methods section.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Fresenius Medical Care North America.
Data availability
The authors confirm that the data supporting the findings of this study are available within the article.
Consent to participate
Verbal or written consent was obtained for clinical assessments conducted in Sudan. Written consent was obtained for studies conducted in Australia and the United States.
Consent for publication
Not applicable. No individual person's data are included.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.