Abstract
Introduction
This study evaluated the usability and patient experience of the Vivatum Alba Continuous Ambulatory Peritoneal Dialysis (CAPD)-System™, an integrated Bluetooth-enabled platform designed to enhance peritoneal dialysis (PD) delivery. Objectives included assessment of patient satisfaction, procedural competence, data accuracy, and reliability of transmission to the remote patient care (RPC™) platform. Such technology may help strengthen the safety and consistency of CAPD delivery, particularly where in-person follow-up is limited.
Methods
A prospective, two-phase, multi-centre cohort study was conducted at two South African sites. Adults on stable PD for ≥3 months were enrolled. In Phase-1, participants received education and completed a usability questionnaire. In Phase-2, they were trained on the Alba system and performed supervised simulated and live PD exchanges. Outcomes included structured observations of procedural competence, patient satisfaction, and accuracy of data capture.
Results
Of the 41 participants enrolled, 30 completed both phases. The median age was 49 years; 52% were male. Hypertension was the most prevalent comorbidity (88%), while fluid overload, defined by bioimpedance or clinical assessment, was present in 58%. Structured observations showed strong procedural competence with consistent adherence to aseptic technique. Patient satisfaction was high (97.5%), and participants valued the system's intuitive interface, infection control prompts, and integrated vital sign monitoring. Vital sign and ultrafiltration measurements obtained via the Alba system correlated closely with manual values, and data transfer to the RPC™ platform was reliable.
Conclusions
The Alba CAPD System™ demonstrated high usability, strong satisfaction, ultrafiltration accuracy, and reliable data transmission, offering a feasible approach to enhance PD delivery. Further evaluation in home environments is warranted.
Keywords
Introduction
By the year 2030, it is estimated that around 14.5 million individuals worldwide with end-stage kidney disease will require kidney replacement therapy (KRT). 1 Peritoneal dialysis (PD) remains an integral component of KRT, offering flexibility and adaptability to meet diverse patient needs. Unlike facility-based haemodialysis[HD], PD enables home-based care and is a practical option when healthcare resources are limited.2,3 PD is delivered via two main modalities: continuous ambulatory peritoneal dialysis (CAPD), involving multiple manual exchanges daily (average 4 times daily), and automated peritoneal dialysis (APD), which typically uses a machine to perform exchanges overnight. These approaches facilitate individualised care, improve accessibility, and enhance patient convenience.4,5 PD is cost-effective in resource-limited settings and contributes to equitable dialysis access while demonstrating comparable or improved survival rates during the early years of therapy.6–10 There is also evidence to support better quality of life and patient satisfaction with PD compared to HD.11–14
The clinical effectiveness of PD is traditionally assessed using parameters such as peritonitis rates, technique survival, mechanical complications, hospitalisations, and PD-related mortality.15,16 Patient-reported outcomes, which reflect quality of life, symptom burden, and life participation, are equally critical in understanding the overall impact of PD. 17 Despite importance, patient-reported outcomes remain underutilised in routine care and are often overshadowed by clinical measures.
Incorporating these outcomes into clinical practice is crucial for improving the overall patient experience and optimising treatment.
Despite its benefits, PD is not without limitations. For 50 years, CAPD has depended on manual exchanges, 18 increasing the risk of touch contamination, which leads to infectious complications such as peritonitis, exit-site and/or tunnel-infections. Additionally, CAPD lacks integrated systems for accurately recording treatment data and/or monitoring therapy compliance. The absence of remote patient care (RPC) systems further complicates timely interventions and efficient management. Mechanical complications, such as catheter malfunctions, are often underreported or diagnosed only after significant delays, resulting in increased reliance on unplanned and urgent care. These challenges adversely impact the long-term sustainability and effectiveness of CAPD, particularly in low-resource settings where healthcare systems are already burdened.
Recent advancements in remote-enabled technologies for dialysis aim to address some of these challenges. For instance, the Claria® system for APD integrates remote therapy monitoring, facilitating earlier detection of complications and improving treatment adherence. 19 However, such systems are often inaccessible in low- and middle-income countries due to high costs and technical requirements, highlighting a pressing need for innovations in CAPD systems. An ideal solution would be to integrate remote-monitoring capabilities into CAPD, providing real-time treatment data while preserving the principles of manual, home-based dialysis.
The Vivatum Alba CAPD System™ (Figure 1) incorporates an integrated Bluetooth-enabled medical device that guides patients through a structured sequence of preparatory tasks during manual exchanges. The system sequentially records with date/time stamps, preparation time, dwell time, exchange duration, and total exchange time, providing an objective measure of treatment adherence and procedural efficiency. It also captures vital signs and drainage volumes, ensuring accurate monitoring and reliable clinical data capture. Key infection control prompts (ICPs) are embedded to assess procedural adherence (Supplement-A). All data are securely transferred to the RPC™ platform, a cloud-based interface that receives encrypted data via Wi-Fi or mobile network from the Alba-System, enabling authorised healthcare providers to remotely review treatment performance in real time thus supporting remote patient monitoring and continuity of care. The system is compatible with major CAPD systems in use and has been successfully evaluated with the TwinBag (Extraneal Baxter/Vantive), UltraBag (Adcock-Ingram), and Stay-Safe (Fresenius Medical Care) technologies.
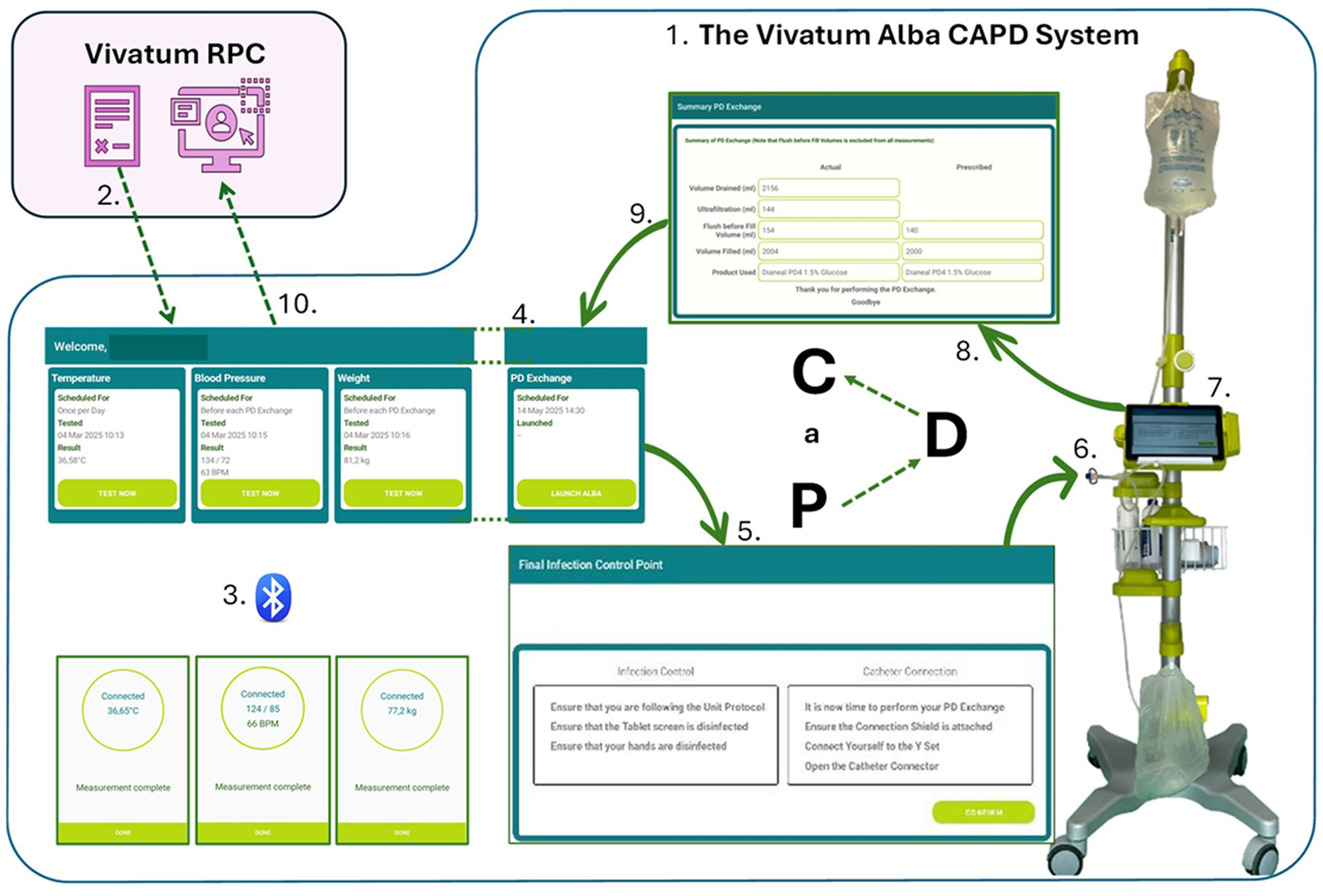
The Alba-CAPD-system. CAPD: Continuous Ambulatory Peritoneal Dialysis.
The primary objective of this study was to assess the usability of the Alba-CAPD-System™ and the overall patient experience. Secondary objectives included evaluating user satisfaction, the accuracy of vital sign and drainage measurements, and the reliability of data transfer to the RPC™ platform. By addressing key challenges in CAPD delivery, particularly in low-resource settings, this study contributes to the development of strategies aimed at enhancing home-based dialysis care and improving long-term patient outcomes.
Methods
We conducted a two-phase, multi-centre cohort study that took place in South Africa at Groote Schuur Hospital, Cape Town (Site-1), and the Hilton Dialysis Unit, Pietermaritzburg (Site-2). Participants were enrolled during routine clinical visits after providing informed consent. Routine laboratory tests were conducted as per Kidney Disease Improving Global Outcomes recommendations.
20
The study was approved by the University of Cape Town Human-Research-Ethics-Committee (HREC-218/2023
Participant selection
Inclusion criteria comprised of adult patients (≥18 years) on CAPD or APD who had been stable on their respective modality for at least 3 months. Exclusion criteria included patients with more than two peritonitis episodes in the preceding 12 months, those undergoing vascular procedures in preparation for HD, individuals younger than 18 years, and patients with cognitive impairment, terminal illness, or critical illness.
Phase-1: initial evaluation and Introduction
At the initial clinic visit, patients were enrolled, provided with study information (Supplement-B), and gave informed consent (Supplement-C). They underwent a comprehensive clinical assessment, viewed an educational video on the Alba-CAPD-System™, and were introduced to the Vivatum-Kiosk™ medical devices and software (Figure 1).
Demographic data, including primary language and comorbidities (hypertension, diabetes mellitus, human immunodeficiency virus [HIV], dyslipidaemia, and cardiac failure), were recorded in conjunction with prescribed medications. Additionally, the distance to the PD centre, mode of transport (public vs. private), and whether the patient resided in a rural or urban area, defined as the home address within a city boundary, were recorded.
Patients underwent a clinical assessment by the treating nephrologist to evaluate fluid status and overall condition. This was followed by a review of their PD prescription, with clinical adjustments made as necessary. Bioelectrical impedance spectroscopy (BIS) using the Body-Composition-Monitor was used to evaluate fluid status (only available at Site-1). Additional laboratory tests were conducted to monitor kidney function, including haemoglobin (Hb)(gm/dl), albumin (gm/L), and urea (mmol/L) and, where feasible, a peritoneal-equilibration-test was performed. The type of PD system used by the patient was also noted.
Patients viewed an educational video explaining the Alba-CAPD-System™, its functionality, and potential benefits. An introduction to the Vivatum-Kiosk medical device and system software and integrated Bluetooth-connected medical devices followed and, after training by the PD nurse investigator, vital signs measurements (blood pressure, heart rate, body weight, and temperature) were performed. The data was sent to the RPC (Figure 1).
Participants completed a structured usability questionnaire based on the validated Integrated Palliative care Outcome Scale–Renal (IPOS-Renal) tool, adapted to assess clinical status and initial impressions of the Alba-CAPD-System™. Modifications included the addition of self-reported symptoms of swollen legs and feet to suggest possible fluid overload (Question-2) and a specific question to capture patients’ initial opinions on the Alba-CAPD-System™ (Question-8). (Supplement-D) Interpreters assisted in questionnaire completion if required.
Phase-2: training and supervised use
At the second monthly clinic visit, patients entered Phase-2, which focused on hands-on training and supervised observation of the Alba-CAPD-System™. Patients were trained on the Alba-CAPD-System™ to perform PD-exchanges, using an abdominal simulator, under the supervision of the nurse investigator. After completing at least two training exchanges, each patient performed a nurse investigator observed PD exchange using the simulator (Supplement-E), which included direct observation of behaviour, recording of comments made by patients and notes made by the investigator. A live PD exchange using the Alba-CAPD-System™, under direct supervision of the nurse investigator, was then conducted. Before each PD exchange, patients used the Kiosk as introduced in Phase-1 to record vital signs (blood pressure, heart rate, body weight and temperature).
The Alba-CAPD-System™ PD exchange procedure followed a structured protocol as outlined in Figure 1. Data from the supervised exchanges included ultrafiltration, drain and fill volumes, and their respective times, previous fill dwell time, preparation and total PD exchange times, and infection control adherence. These parameters were then securely transmitted to the RPC™ platform for evaluation. Minor procedural variations were noted across training centres (Supplement-F).
After completing the PD exchange, patients completed a second structured usability questionnaire (IPOS-Renal) to assess their experience with the Alba-CAPD-System. Additional modifications were introduced to evaluate patient satisfaction and anticipated challenges with home use (Supplement-G).
Modifications to the second-visit questionnaire
Question-2: Retained the addition of Swollen Legs and Feet as symptom indicators.
Question-6: Modified to ‘Have you received as much information as you wanted?’
Question-8: Remained unchanged from the first visit.
Question-9: Revised to ‘Do you anticipate any difficulties using the CAPD device at home?’
These modifications aimed to enhance the assessment of patient satisfaction and usability of the Alba-CAPD-System™ in a home setting.
Statistical methods
Descriptive statistics were used to summarise baseline characteristics, travel data, device usage, and patient-reported outcomes. Continuous variables were reported as means with standard deviations for normally distributed data, and as medians with interquartile ranges (IQR) where appropriate. Categorical variables were summarised using frequencies and percentages and graphs generated using power pivot tables. Patient competence during PD-exchange steps was evaluated using structured observation tools adapted from human factors validation guidelines 21 and JASP analytics. Satisfaction and patient-reported outcomes, including responses from the IPOS-Renal questionnaire, were analysed descriptively using JASP analytics. Comparative performance between patients with and without prior peritonitis was assessed to explore potential associations with procedural adherence.
Results
Study population and baseline characteristics
A total of 41 participants were enrolled in the study, with 30 successfully completing both phases. The median age at enrolment was 49 years (IQR 39–59), 52% of participants were male, with a median PD duration of 34 months (IQR 16–44). The study cohort reflected a diverse linguistic background, Zulu (27%), isiXhosa (27%), Afrikaans (15%), and English (32%). Comorbid conditions included hypertension (88%), diabetes mellitus (19%), HIV (22%), and cardiac failure (2%).
Regarding PD-product technologies, 64% of participants used the Ultrabag system (Adcock-Ingram Twinbag®), 36% used the Dialler technology (Fresenius Stay-Safe®). BIS indicated fluid overload exceeding 0.5 litres in 58% of participants (n = 14/24), with a median value of 1.1L (Table 1).
Baseline characteristics and travel.
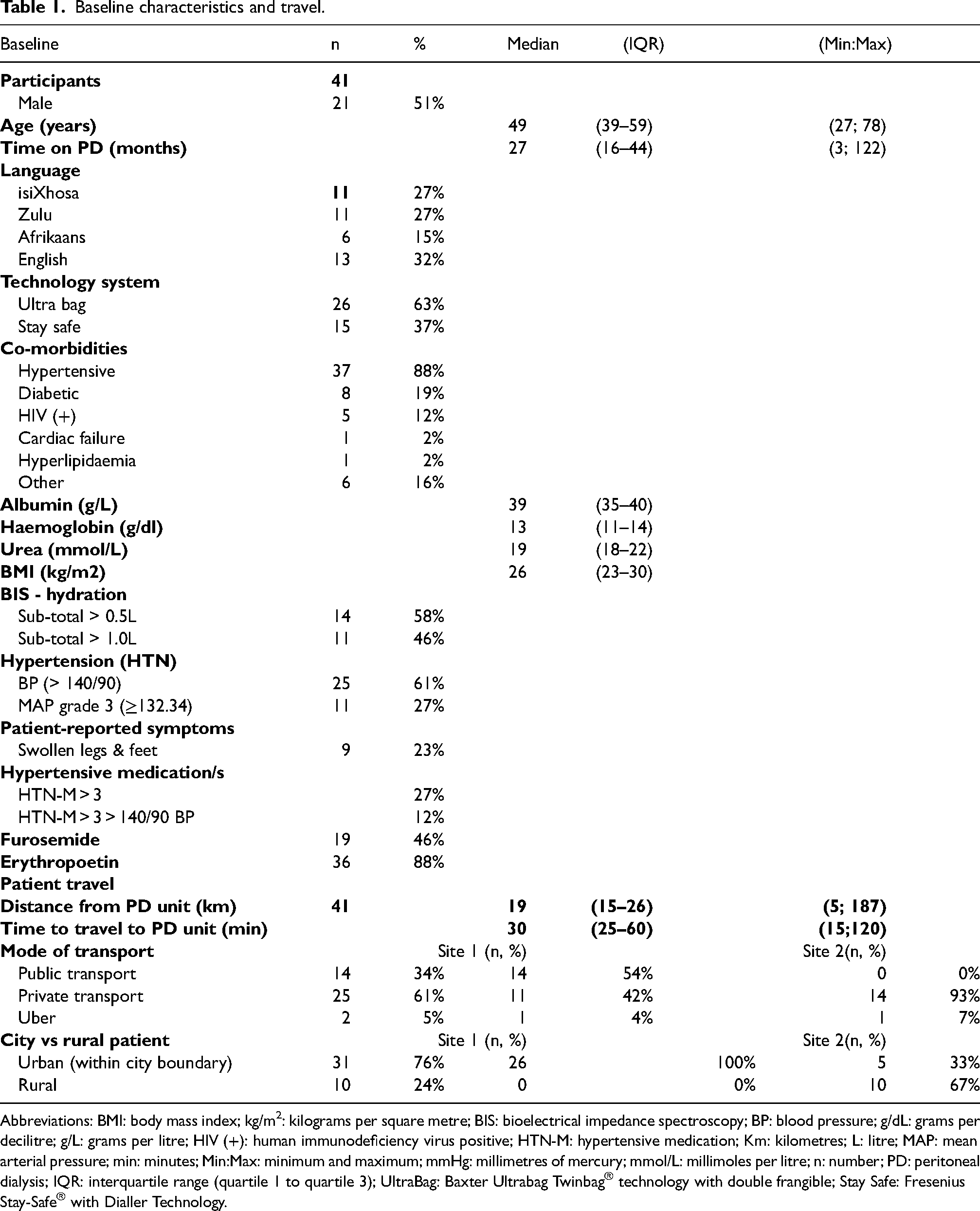
Abbreviations: BMI: body mass index; kg/m2: kilograms per square metre; BIS: bioelectrical impedance spectroscopy; BP: blood pressure; g/dL: grams per decilitre; g/L: grams per litre; HIV (+): human immunodeficiency virus positive; HTN-M: hypertensive medication; Km: kilometres; L: litre; MAP: mean arterial pressure; min: minutes; Min:Max: minimum and maximum; mmHg: millimetres of mercury; mmol/L: millimoles per litre; n: number; PD: peritoneal dialysis; IQR: interquartile range (quartile 1 to quartile 3); UltraBag: Baxter Ultrabag Twinbag® technology with double frangible; Stay Safe: Fresenius Stay-Safe® with Dialler Technology.
Distance travelled
Travel distance to the PD unit ranged from 5 to 187 km, with a median of 19 km (IQR 15–26). Travel time ranged from 15 to 120 min, with a median of 30 min (IQR 25–60) (Table 1). At Site-1 (Groote Schuur Hospital), the median (IQR) travel distance was 18 km (13–25 km) with over half of the patients (54%) using public transport. Patients at Site-2 (Hilton), where 67% lived in rural areas, travelled a median of 21 km (IQR 17–31 km), with private transport being more common (93%) (Table 1 and Figure 2). Importantly, some urban patients residing within 40 km of the unit reported travel times exceeding 60 min, underscoring barriers to access that extend beyond distance alone (Figure 2(b)).
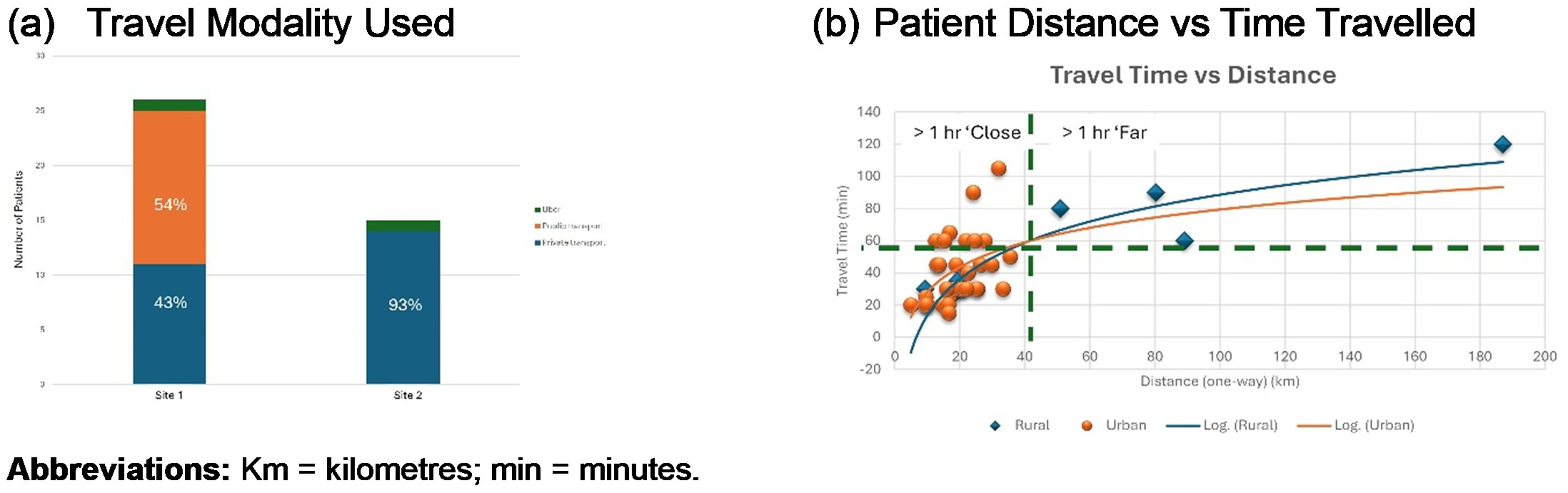
Patient travel details (a) modality (b) distance in kmvs time (min).
System usability and patient satisfaction
A total of 41 patients participated in the usability evaluation of the Alba-CAPD-System™. In Phase-1, after initial exposure to the system, 97.5% of patients (40/41) reported overall satisfaction with the device (Figure 3).
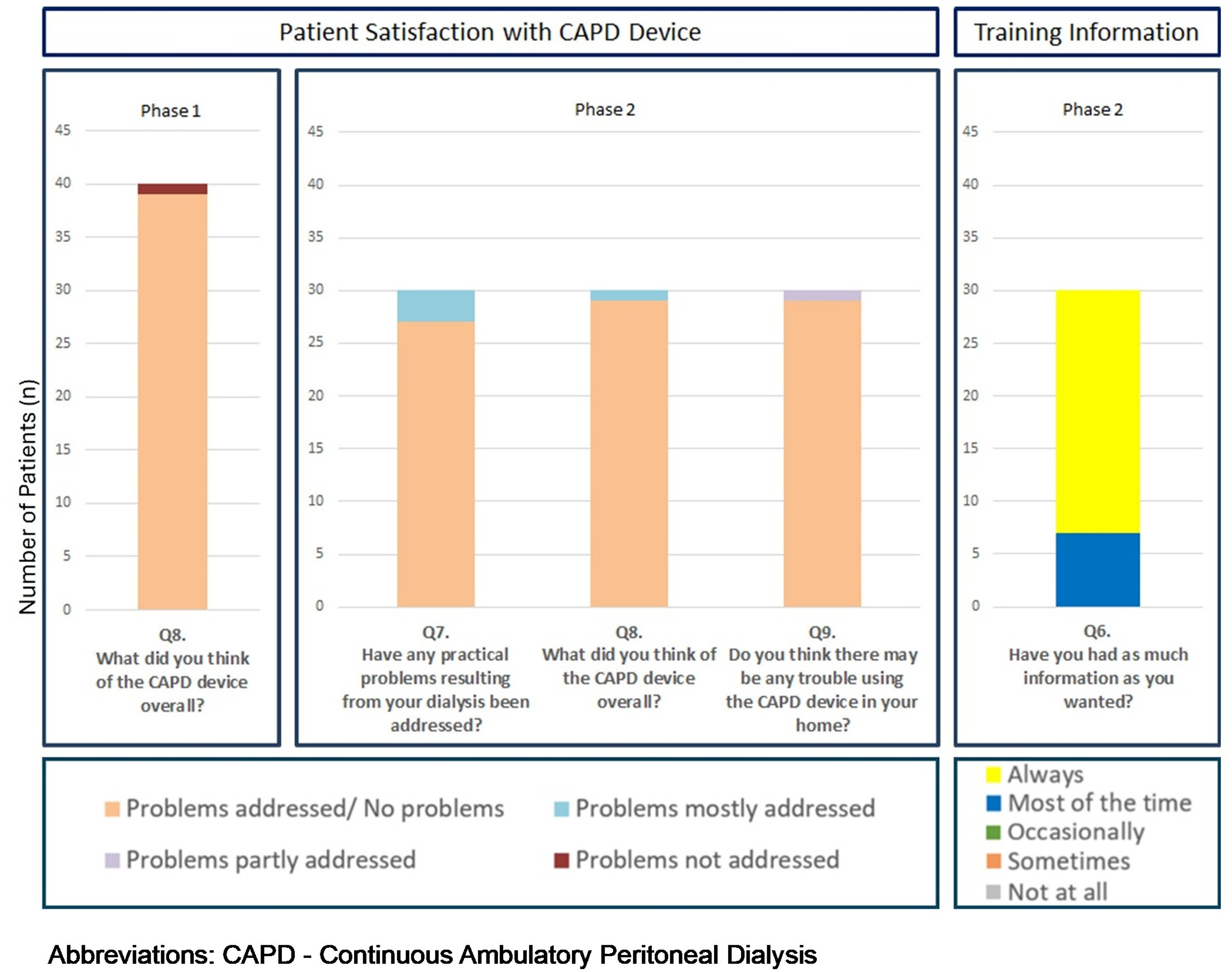
Patient satisfaction with the Alba system and information provided.
In Phase-2, following training and use on the Alba-CAPD-System™, the iPOS-Renal questionnaire was re-administered. Satisfaction remained high, with 97.5% of patients finding the system suitable for home use. One patient expressed concern regarding the adjustment period required to become accustomed to the new device. All patients were satisfied with the training.
Patient perspective and feedback
Patient feedback emphasised the system's intuitive design, step-by-step guidance, and infection prevention reminders. Many patients expressed positive feedback for its user-friendly prompts and the reassurance it provided:
‘This is so good because it reminds me of steps I need to do because sometimes I don't remember’. ‘It's not easy to get infection because the device keeps reminding me to wash my hands’. ‘I would really like to have this device if it was given to me, as it would make it much easier’. ‘It can help a lot, because it repeats the important steps and sends everything through when we have done it’.
Patients also highlighted the benefits of Bluetooth-enabled medical devices and expressed a strong preference for the Alba-CAPD-System™ over other devices. Overall, feedback underscored the system's role in enhancing patient confidence, adherence, and ease of use.
The study also assessed procedural memory retention by evaluating patient performance of a PD-exchange following initial training. At Site-1 (urban), patients were trained on Day-1 and returned within 0–9 days for supervised exchanges. At Site-2 (rural), training and observation were completed on the same day due to logistical constraints. Despite these differences, no variation in procedural adherence or ability to perform an exchange was observed between sites.
A total of 29 patients performed live PD exchanges, with a total of 53 PD-exchanges being performed, using the Alba-CAPD-System™ (Supplement-I). Both technologies commercially available were used (30 with Ultrabag & 23 Dialler, respectively) (Table 1). Three patients were unable to complete an exchange due to issues detected by the Alba-CAPD-System™; one a leaking drain bag, one dislodged the tubing after the flush, and one a leaking Y-set tube.
PD exchanges had a median duration of 35 min (IQR 31–38), with a median preparation time of 13 min (IQR 11–14). ICP times were consistent, reflecting strong protocol adherence, likely due to supervision by nurse investigators. The median treatment time was 21 min (IQR 17–24), with drain time contributing the largest component at 10 min (IQR 8–11). Ultrafiltration measurements based on actual prior fill volumes, excluding flush and priming volumes, differed notably from estimates based on the standard 2000 ml fill volume (Supplement-I).
The standardised protocol, which required patients to record vital signs (blood pressure, heart rate, body weight and temperature) prior to initiating PD-exchanges, was well received and smoothly incorporated into their procedure. Patients were not inconvenienced by the process and expressed appreciation for the added integrated devices. Many valued their own role in the monitoring process, reporting a greater sense of engagement and ownership of their treatment (Figure 4 and Supplement-H).
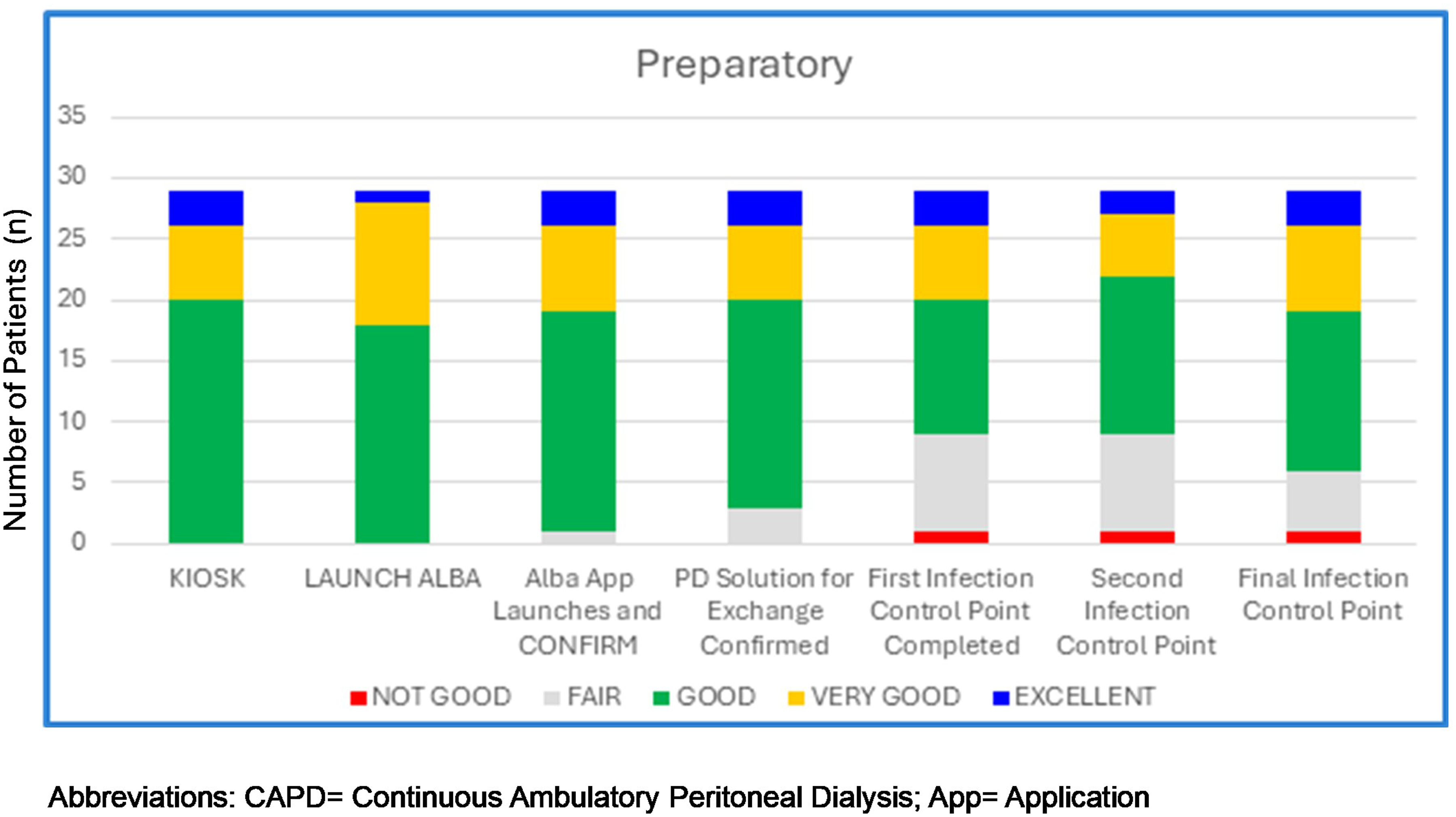
Investigator observed patient competence during preparatory phase.
Kiosk use by patients was consistently strong, considering they had been introduced to the procedure in Phase-1 reinforcing ease-of-use (Figure 4). Minor discrepancies were observed between Kiosk™ recorded and manually observed readings, supporting the accuracy and reliability of the system (Supplement-I). Notably, patients with a prior history of peritonitis exhibited lower overall performance during the ICPs. In contrast, those rated as Excellent or Very Good had no documented episodes of peritonitis.
Participants who successfully completed their PD exchanges reported that the Drain, Flush, and Fill sequences were straightforward and easy-to-follow (Supplement-H). The accuracy of therapy duration measurements, including dwell time, was further validated during consecutive control exchanges using the Alba-CAPD-System™ (Supplement-I). After the PD exchange, patients completed a Drain Bag Control check-list, and viewed the PD exchange outcomes summary (Supplement-L). Data was seamlessly transmitted to the RPC.
Eleven patients did not complete the study for various reasons, including transfer to HD (n = 2), transplantation (n = 3), meeting exclusion criteria, (dementia diagnosed after phase-I) (n = 1), personal decision (n = 4), and death (n = 1). All the participants successfully completed training and were able to perform simulated exchanges prior to withdrawal. Therefore, participant withdrawal reflected external clinical or personal factors rather than challenges with the Alba-CAPD-System™ and is unlikely to have introduced bias in the usability or satisfaction outcomes. While the study acknowledged anxiety during training and initiation (Supplement-J), the emotional impact of home-based PD remains unexplored and warrants further investigation.
Discussion
This prospective, two-phase, multi-centre cohort study evaluated the usability, safety, and reliability of the Alba-CAPD-System™ in 30 patients. The system demonstrated high patient satisfaction (97.5%), strong confidence in the structured training approach, and effective integration with the RPC™ platform. Key advantages included real-time remote monitoring, accurate data capture, and improved patient engagement. These findings underscore the potential of the Alba-CAPD-System™ to enhance PD management by promoting patient-centred care, supporting timely clinical decision-making, and ultimately contributing to improved patient outcomes.
Despite its clinical and logistical advantages, PD continues to yield variable outcomes globally, reflecting inconsistencies in clinical practice, limited monitoring capabilities, and the absence of standardised care protocols. 22 Traditional CAPD care often relies on manual data collection and infrequent clinical assessments, which can delay the recognition and management of complications such as fluid overload, peritonitis, or ultrafiltration failure. Broader systemic challenges, including workforce shortages, inadequate health information systems, and transport barriers further hinder the effective delivery of CAPD. 23 Addressing these challenges is critical to improving patient outcomes and ensuring more consistent, high-quality CAPD care.
The study cohort was linguistically diverse, highlighting the need for communication and education strategies tailored to multiple languages to ensure effective engagement with PD care. The Alba-CAPD-System™ supports this through its structured, patient-centred interface, and integrated remote-monitoring functionality. Remote monitoring is especially valuable in resource-constrained settings, enabling real-time oversight of treatment adherence and early identification of complications. This functionality helps bridge communication gaps, supports consistent care delivery, and enhances patient safety across diverse populations.
The cohort exhibited a high burden of comorbidity, with hypertension present in 88% of participants, and fluid overload of greater than 1.1 litres affecting 58% of patients. These clinical characteristics underscore the importance of precise fluid management. While the Alba-CAPD-System™ accurately records ultrafiltration, drain and fill volumes, and body weight, these parameters should be interpreted alongside residual kidney function, dietary intake, and bioimpedance assessments for a comprehensive evaluation of volume status. In this context, the system's remote-monitoring capability may support early detection of fluid imbalance and timely clinical intervention, thereby helping to optimise volume control and long-term outcomes. Supporting this approach, prior studies, including a cluster-randomized controlled trial by Paniagua et al., have demonstrated that remote automated PD can improve outcomes, including reductions in mortality and hospitalisation. 24
Across both study phases, the Alba-CAPD-System™ received high usability scores and patient satisfaction ratings. All participants in Phase-2 expressed satisfaction with the hands-on training and supervision provided, regardless of the delay between training and assessment. The structured training materials and step-by-step instructions supported procedural memory and promoted confidence in independent PD exchanges.
The system accurately captured vital signs including blood pressure, heart rate, temperature, and body weight before each exchange. This data aligned well with manual readings and enabled proactive clinical oversight via the RPC™ platform. Features such as automated alerts for data transmission gaps and offline tracking helped identify non-adherence or patient drop-out, supporting continuous care.
Peritonitis remains the most common preventable cause of technique failure in PD, with touch contamination recognised as a leading contributor. 25 The ICPs embedded within the Alba-CAPD-System™ are designed to minimise this risk by reinforcing aseptic behaviour and procedural consistency, although the system itself does not directly detect peritonitis. The intuitive design and integrated ICPs supported correct technique and helped identify users at higher risk for error, underscoring the value of structured, standardised processes. As one participant observed, ‘we don’t all perform the procedure as our nurse trains us’, highlighting variability in practice and the importance of guided protocols. These findings suggest that design led infection control strategies may help reduce peritonitis rates and improve PD technique longevity. Future studies should determine whether these integrated prompts and monitoring tools translate into measurable reductions in peritonitis incidence.
The system's structured protocol revealed variability in actual fill volumes due to retained priming volume and evaporation, which challenges current assumptions in ultrafiltration calculations. This insight suggests that standard assumptions of a 2000 mL fill may lead to overestimated ultrafiltration volumes, emphasising the need for precise measurements in PD management.
Long commute times, even for patients living within 40 km of a PD unit, posed significant barriers. The RPC™ platform may assist with this challenge by reducing the frequency of required in-person visits, enhancing care accessibility, and improving the patient experience.
Limitations
This study was conducted in a controlled setting and did not assess long-term outcomes in a home environment. The sample size while small, limiting generalizability, was sufficiently weighted according to human factors validation testing guidelines. 21 Patient performance was also influenced by previous experience and comorbidities, which may have introduced variability in outcomes.
Future research should explore whether integration of symptom reporting or effluent monitoring features could enhance early recognition of peritonitis and other infection-related complications. Additionally, evaluating the Alba-CAPD-System™ in the home setting assessing it's impact on clinical outcomes, complication rates, and healthcare utilisation are required. Comparative studies are needed to benchmark its performance against standard CAPD techniques. Tailoring implementation strategies to individual PD units particularly in linguistically and logistically diverse settings will be critical to ensure effectiveness.
Conclusion
The Alba-CAPD-System™ represents a meaningful advancement in PD care, addressing critical gaps in monitoring, training, and patient support. It's combination of accurate data collection, real-time clinical oversight, and high patient satisfaction suggests that it may significantly enhance PD outcomes. Broader adoption, supported by further evaluation in home-based settings, has the potential to transform CAPD treatment.
Supplemental Material
sj-pdf-1-ptd-10.1177_08968608261420157 - Supplemental material for Enhancing the peritoneal dialysis patient experience: Usability and satisfaction with the New Vivatum Alba Continuous Ambulatory Peritoneal Dialysis system
Supplemental material, sj-pdf-1-ptd-10.1177_08968608261420157 for Enhancing the peritoneal dialysis patient experience: Usability and satisfaction with the New Vivatum Alba Continuous Ambulatory Peritoneal Dialysis system by Nicola Wearne, Hamze Rage, Brett Cullis, Eleanor de Reuke, Grace Dlamini, Petronella Ramjiet, David Thorne and Jose Carolino Divino-Filho in Peritoneal Dialysis International
Supplemental Material
sj-pdf-2-ptd-10.1177_08968608261420157 - Supplemental material for Enhancing the peritoneal dialysis patient experience: Usability and satisfaction with the New Vivatum Alba Continuous Ambulatory Peritoneal Dialysis system
Supplemental material, sj-pdf-2-ptd-10.1177_08968608261420157 for Enhancing the peritoneal dialysis patient experience: Usability and satisfaction with the New Vivatum Alba Continuous Ambulatory Peritoneal Dialysis system by Nicola Wearne, Hamze Rage, Brett Cullis, Eleanor de Reuke, Grace Dlamini, Petronella Ramjiet, David Thorne and Jose Carolino Divino-Filho in Peritoneal Dialysis International
Supplemental Material
sj-docx-3-ptd-10.1177_08968608261420157 - Supplemental material for Enhancing the peritoneal dialysis patient experience: Usability and satisfaction with the New Vivatum Alba Continuous Ambulatory Peritoneal Dialysis system
Supplemental material, sj-docx-3-ptd-10.1177_08968608261420157 for Enhancing the peritoneal dialysis patient experience: Usability and satisfaction with the New Vivatum Alba Continuous Ambulatory Peritoneal Dialysis system by Nicola Wearne, Hamze Rage, Brett Cullis, Eleanor de Reuke, Grace Dlamini, Petronella Ramjiet, David Thorne and Jose Carolino Divino-Filho in Peritoneal Dialysis International
Supplemental Material
sj-docx-4-ptd-10.1177_08968608261420157 - Supplemental material for Enhancing the peritoneal dialysis patient experience: Usability and satisfaction with the New Vivatum Alba Continuous Ambulatory Peritoneal Dialysis system
Supplemental material, sj-docx-4-ptd-10.1177_08968608261420157 for Enhancing the peritoneal dialysis patient experience: Usability and satisfaction with the New Vivatum Alba Continuous Ambulatory Peritoneal Dialysis system by Nicola Wearne, Hamze Rage, Brett Cullis, Eleanor de Reuke, Grace Dlamini, Petronella Ramjiet, David Thorne and Jose Carolino Divino-Filho in Peritoneal Dialysis International
Footnotes
Acknowledgements
Prof Bianca Davidson and all the Peritoneal Dialysis patients who participated in the study
Author contributions
NW, JD, BC, and DT contributed to the development of the study protocol, preparation of the ethics application, and the writing and critical revision of the original manuscript. DT provided statistical support and led data management. HR was responsible for project administration and contributed to manuscript drafting. ER, PR, and GD provided essential support with project administration.
Declaration of conflicting interest
DT declares a conflict of interest as Product Development Consultant, Vivatum PD AG, Switzerland. JD declares a conflict of interest as Chief Medical Advisor, Vivatum PD AG, Switzerland. All other author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Vivatum, (grant number Private Funder).
Ethics approval and informed consent
This study was approved by the University of Cape Town, Faculty of Health Sciences, Human Research Ethics Committee (approval no. HREC REF 218/2023). All participants provided written informed consent prior to participating.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
