Abstract
Background
Peritoneal dialysis (PD) is a home-based treatment for kidney failure, offering significant social, economic and environmental advantages over haemodialysis (HD). It allows patients greater independence and flexibility, can reduce healthcare costs in some settings, and is reported to generate a smaller environmental footprint compared to HD. However, despite this, its environmental impact, particularly in terms of resource use and waste generation, is an area of growing concern. Evaluating PD through a sustainability lens, considering environmental, social, health and economic dimensions is crucial to improving its long-term viability and reducing the healthcare sector's environmental footprint.
Methods
An LCA of the PD treatment pathway was conducted at the Nephrology Dialysis and Kidney Transplantation Unit, ‘Azienda Ospedaliero-Universitaria (AOU), Policlinico di Modena, Italy. Data were collected between April and July 2024 and was categorised into 17 PD-related procedures. Key activities assessed were electricity use, resource consumption (procurement), water consumption, patient and staff travel and waste disposal. Flow diagrams were created to model the PD pathway and product components were dismantled and weighed to determine material contributions. Environmental impacts were analysed using OpenLCA and the Ecoinvent database.
Results
Automated peritoneal dialysis (APD) treatments produced greater emissions than continuous ambulatory peritoneal dialysis (CAPD), primarily due to higher electricity consumption and the use of polyvinylchloride. APD generated 3267 kg CO2e annually compared to 2975 kg CO2e for CAPD. Monthly patient examinations contributed 250 kg CO2e per year, largely from travel. CAPD produced more healthcare waste (320 kg), leading to 810 kg CO2e emissions, while APD waste emissions were lower at 374 kg CO2e. Incremental dialysis approaches showed substantial reductions, with emissions falling to 1642 kg CO2e (APD) and 1517 kg CO2e (CAPD). The most impactful materials included polyethylene and PVC, with plastic components contributing significantly to overall emissions.
Conclusion
This comprehensive life cycle assessment (LCA) highlights the environmental challenges associated with PD, particularly in waste generation and resource use. Key areas for improvement include reducing single-use plastic reliance, increasing recycling efforts and integrating energy-efficient solutions. This study provides a foundation for targeted sustainability interventions in PD and contributes to the broader goals of the KitNewCare project, which aims to optimise health outcomes while minimising environmental impacts in kidney care.
Keywords
Introduction
Kidney disease is a significant health burden, affecting over 850 million people worldwide. 1 Kidney failure, the most advanced stage of disease, may be treated with renal replacement therapy such as dialysis or kidney transplantation, though not all patients access or choose these options. 2 Dialysis remains the most common therapy, but its provision generates substantial environmental, social and economic burdens.
Peritoneal dialysis (PD) is a home-based therapy that provides greater flexibility and independence compared with haemodialysis (HD).3,4 It reduces patient travel to hospital and is associated with lower infrastructure and staffing needs. Despite these advantages, PD is highly dependent on single-use plastics and packaging. Fluid bags, tubing and catheters contribute large volumes of non-biodegradable waste. 5 Delivering treatment at home also shifts some electricity and water consumption from hospital to household settings, as the APD cycler operates using domestic electricity and additional water is required for hygiene and waste disposal, even though the dialysate fluid itself is manufactured remotely.
The healthcare sector contributes around 5% of global greenhouse gas emissions and nephrology services are recognised hotspots within this footprint. 6 Life cycle assessment (LCA) is the established method for evaluating environmental impacts across the full life span of a product or process, from raw material extraction through disposal. 7 While LCA studies have been carried out for HD in Europe and Australia, no comprehensive LCA of the entire PD pathway in a European context has been published. Existing research has focused either on selected products, waste volumes, or partial system boundaries, leaving a significant evidence gap.
This study forms part of the KitNewCare project, an EU-funded initiative that aims to reduce the carbon footprint of kidney care while optimising health outcomes, equity and costs. 8 By mapping the full PD treatment pathway at a large Italian centre, this work addresses the lack of detailed environmental evidence for PD in Europe. It also provides data to inform targeted interventions in procurement, product design and service delivery.
Aim of study
This article maps the environmental footprint of the full PD treatment pathway using LCA. It aims to identify high-impact components and processes, supporting strategies to reduce environmental burden without compromising patient care. Data were collected at the Nephrology Dialysis and Kidney Transplant Unit, AOU Policlinico di Modena, Italy.
Methods
LCA was conducted between April and July 2024, following ISO 14040/14044 standards.9,10
Patient pathways
Identification of procedures and pathways
The LCA included all stages of PD care, including education, medical evaluations, treatment and waste disposal. Key data categories included energy and water use, travel, procurement and waste. It covered both automated peritoneal dialysis (APD) and continuous ambulatory peritoneal dialysis (CAPD), including incremental approaches that start with reduced exchanges and increase as needed based on residual kidney function or as clinically indicated. 11
The main stages of the PD pathway evaluated are as shown in Supplementary Table S1 in the appendices.
Flow diagrams
Flow diagrams for each step were developed using Visio 10 and verified with clinical staff Supplementary Figure S1. These diagrams captured all activities and material usage. All data were compiled into a dataset published in the study appendices.
Dismantling products and weighing components
To estimate the environmental impact of the healthcare products used in PD, the products were dismantled into their individual components, the materials of each component identified and their weights measured.
A list of all products used in PD was collected. This included dialysis solutions, plastic tubing, connectors, bags and other consumables.
Each product was carefully dismantled to separate its components. For example, Dialysis bags were separated into plastic bags, caps and any other distinct parts.
Each component was weighed using a precision scale. 12 The weights were recorded and categorised based on component and material type (e.g., polyvinylchloride), polyethylene, polypropylene).
The recorded weights and material types were recorded for analysis.
APD machines were modelled using the Fresenius SleepSafe and Baxter HomeChoice systems, representing the main devices in clinical use at Modena. Inventories were built from prescriptions and consumables: CAPD involved three 2 L manual exchanges daily; APD used a cycler with two 5 L bags. Associated items included connectors, dressing kits and transfer sets. Brand-specific products from Baxter and Fresenius were modelled where data were available. A complete product inventory, including dressings, packaging and consumables, is provided in the supplementary dataset.
Incremental PD
Incremental prescriptions were defined as approximately half of the full dose prescription for each modality. For CAPD, the incremental regimen used around 50% of the exchanges and dialysate volume of the full dose (three 2 L manual exchanges per day; 6 L total dialysate for full prescription). For APD, incremental prescriptions similarly used roughly 50% of the dialysate volume compared with the full dose APD regimen of two 5 L automated exchanges per night (10 L total dialysate).
Inputting data into OpenLCA
Material weights and types, alongside the flow diagrams, were input into OpenLCA. 10 Processes for each step were modelled and linked to the flow diagrams. Environmental impacts were then calculated using the Ecoinvent v3.10 database. 13
To aid interpretation, results were normalised by scaling them relative to average environmental impacts per person, which indicates whether a process exceeds or falls below population-level benchmarks.
The incremental factors (0.5 for APD and CAPD) were applied as a pragmatic scaling method based on the observed reduction in annual dialysate volume and treatment intensity between full dose and incremental prescriptions. Where component level differences (for example specific consumables or materials) were directly quantified in the inventory, those measured values were used. For components that were not separately quantified, the same scaling factors were applied to the full dose data to ensure a consistent and transparent approach across all 17 procedures in the pathway.
System boundaries
The system boundaries (Figure 1) cover raw material extraction, manufacturing, transport, use and disposal. This includes materials for PD equipment such as dialysis solutions, tubing, connectors and disposal bags, as well as their processing into final products. Transport accounts for all freight and passenger journeys, including delivery from manufacturers to clinics or patients and waste disposal logistics. Packaging materials were included in procurement impacts. The use phase reflects solution preparation, administration, exchanges and drainage in the home setting. Disposal covers the treatment of medical waste, including used solutions, components and other consumables. Supplies originated mainly from European manufacturers and were delivered to Modena. Waste was segregated into clinical, recyclable and general streams.
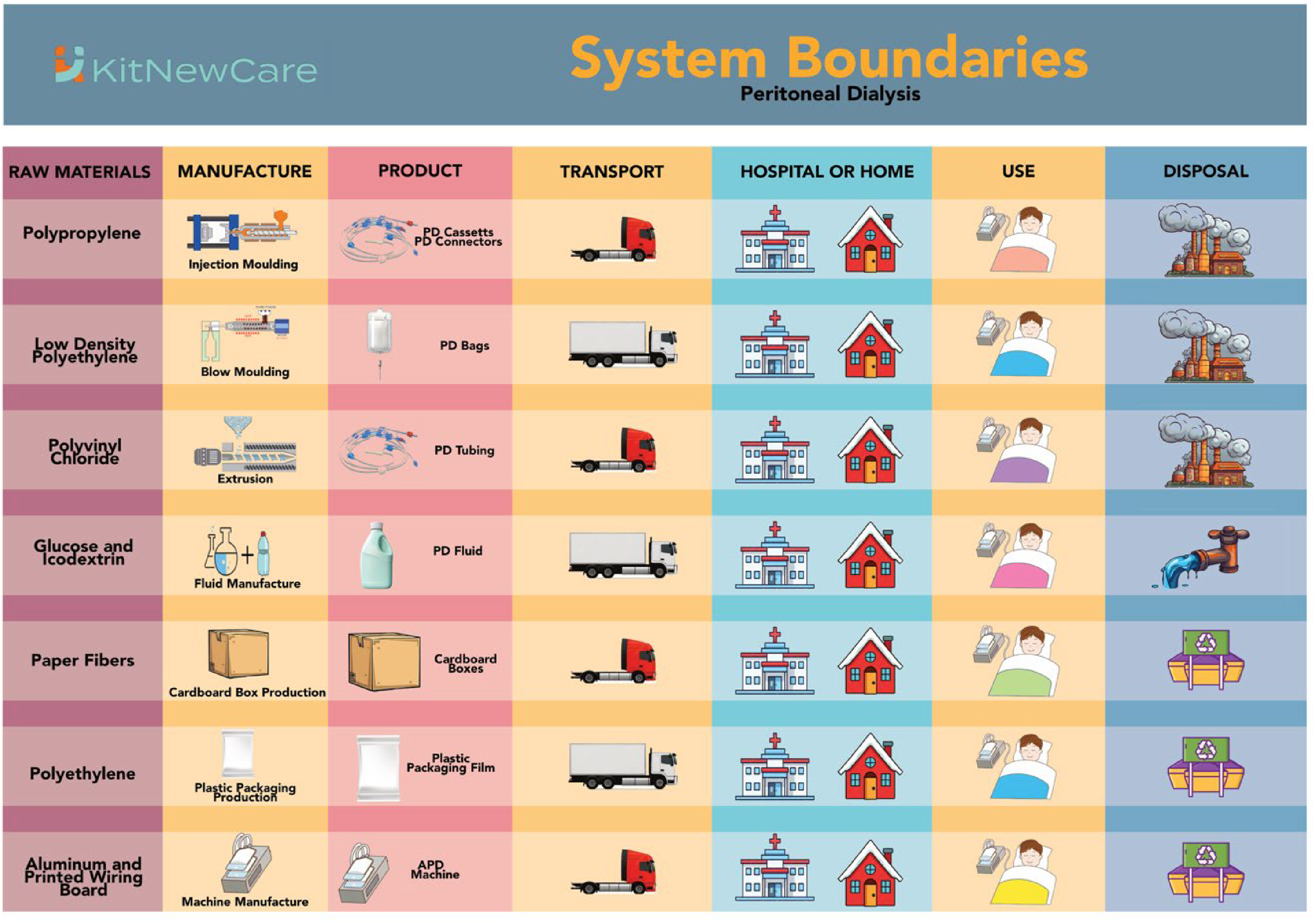
System boundaries for peritoneal dialysis. APD and CAPD are both included. The system boundary shows the ‘cradle to grave’ analysis of the LCA.
Processes
Product manufacture
Component weights were measured in April 2024. Manufacturing processes were identified through product safety data sheets and informed assumptions. Machinery energy use was sourced from Eco Invent. 13
Transportation
Transportation impacts included both freight of medical products and travel by patients and staff. All relevant data were modelled under a single process category for clarity.
Staff and patient travel
To avoid delays related to General data protection regulation (GDPR), staff and patient travel data were sourced from an Irish study on PD travel patterns.14, 15 This served as a proxy to estimate transportation-related environmental impacts, including average distances for PD sessions and staff commutes. Patients travelled an average 29 km round trip, with similar distances for staff. These figures were reviewed and agreed upon by Modena staff as representative for the region.
Freight transport for supplies
All freight transport of consumables, machines, and packaging materials was modelled within the same transportation process. Distances were calculated using Google Maps for road freight and Fluentcargo.com for shipping. Modes included truck and ship transport, based on Italian and European datasets.16,17 Where specific manufacturer data were unavailable, average European freight datasets from Ecoinvent v3.10 13 were used. Transport impacts covered the full journey from manufacturer to AOU Policlinico di Modena and included return of medical waste to disposal facilities.
Disposal
Waste management was modelled based on the segregation practices reported by hospital staff. Waste was categorised into clinical, recyclable and general streams. For home-based treatment, it was assumed that patients separated recyclable and general waste in line with hospital protocols, while clinical waste was collected for incineration.
Ethics
No ethical approval was required as the study involved only environmental assessment of devices and processes, with no personal data collected.
Functional unit
The functional unit for this study is defined as the complete PD treatment for a single patient over an average one-year period, allowing standardised comparison between treatment modalities. One-off activities such as education, surgery, catheter placement, peritonitis management and training were annualised by dividing their total footprint across an expected five-year treatment duration. This approach reduces their apparent contribution compared with recurrent activities such as monthly examinations or regular education sessions.
Results
The results have been analysed overall, by patient pathway and by process (e.g., transportation, procurement etc).
Results by patient pathway
The results by patient pathway can be found in Table 1. Results have been divided to show the difference in APD and CAPD. For simplicity the patient pathways are shown for carbon only PD treatment, monthly exam (the two highest patient pathways can be seen in (Table 2 and Supplementary Table 2).
Carbon footprint patient pathways CO2e emissions.

Pathway 11: PD treatment by process (including 30% APD, 4% CAPD, 30% incremental APD and 36% incremental CAPD).
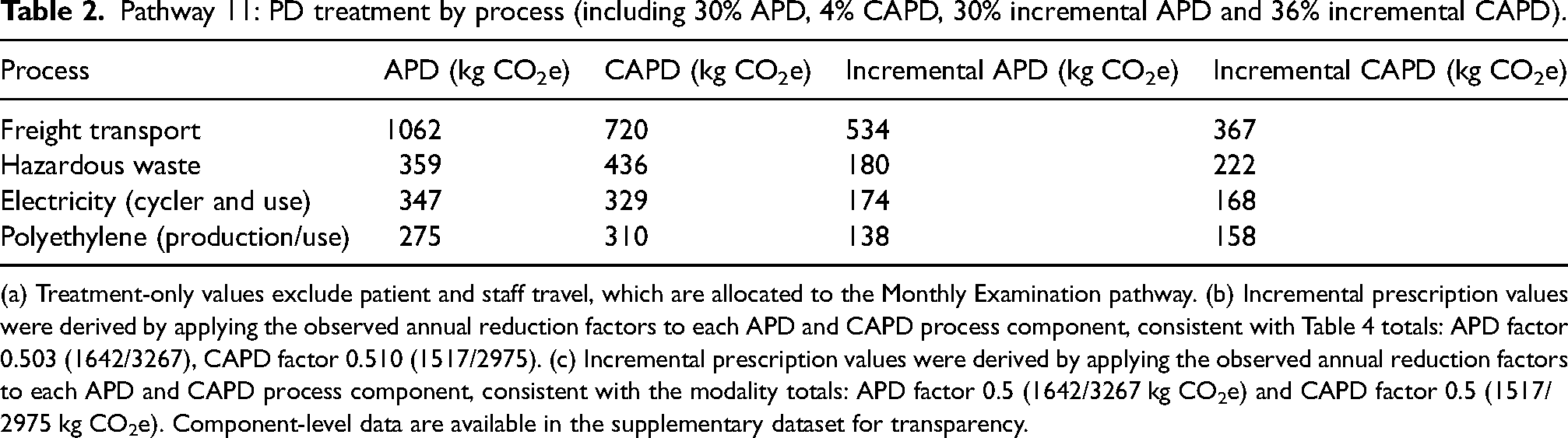
(a) Treatment-only values exclude patient and staff travel, which are allocated to the Monthly Examination pathway. (b) Incremental prescription values were derived by applying the observed annual reduction factors to each APD and CAPD process component, consistent with Table 4 totals: APD factor 0.503 (1642/3267), CAPD factor 0.510 (1517/2975). (c) Incremental prescription values were derived by applying the observed annual reduction factors to each APD and CAPD process component, consistent with the modality totals: APD factor 0.5 (1642/3267 kg CO2e) and CAPD factor 0.5 (1517/2975 kg CO2e). Component-level data are available in the supplementary dataset for transparency.
Table 2 reflects a case mix of 30% APD, 4% CAPD, 30% incremental APD and 36% incremental CAPD, representing the treatment distribution in Modena. For transparency, results are also presented disaggregated by modality.
Laparoscopic insertion shows a lower annualised value than local insertion because it was less frequent in the cohort, whereas post-operative evaluation applies to all patients and therefore has a higher annualised contribution.
Catheter-related procedures contribute a small share of the annual footprint in the one-year functional unit. Laparoscopic insertion produces higher emissions per procedure due to theatre energy use, sterilisation and surgical instruments. However, it was performed less frequently than local anaesthetic insertion in the Modena cohort, so its impact appears smaller once annualised across all patients. The annualised values therefore reflect frequency of occurrence rather than per-event intensity.
Table 2 pathway 11 values represent a case mix of full dose and incremental prescriptions (30% APD, 4% CAPD, 30% incremental APD and 36% incremental CAPD), reflecting the treatment distribution in Modena. In contrast, Table 3 presents disaggregated modality-specific values for each modality (full dose APD, full dose CAPD, incremental APD and incremental CAPD) modelled separately. The APD value in Table 2 is therefore higher than the incremental APD value in Table 3 because it reflects a mixture of full dose and incremental treatment, rather than incremental APD alone.
APD versus CAPD versus incremental approach (1 year).

PD treatment APD and CAPD
Figure 2 details the CO2e contributions of key processes and materials involved in APD and CAPD. The top contributors are freight transport (1062 kg CO2e for APD and 720 kg CO2e for CAPD), followed by hazardous waste (359 kg CO2e for APD and 436 kg CO2e for CAPD), electricity (347 kg CO2e for APD and 329 kg CO2e for CAPD), polyethylene production (275 kg CO2e for APD and 310 kg CO2e for CAPD) and passenger car travel (239 kg CO2e for APD and CAPD). Travel emissions associated with other pathways such as Monthly Examination and excluded from the treatment itself which is home based.
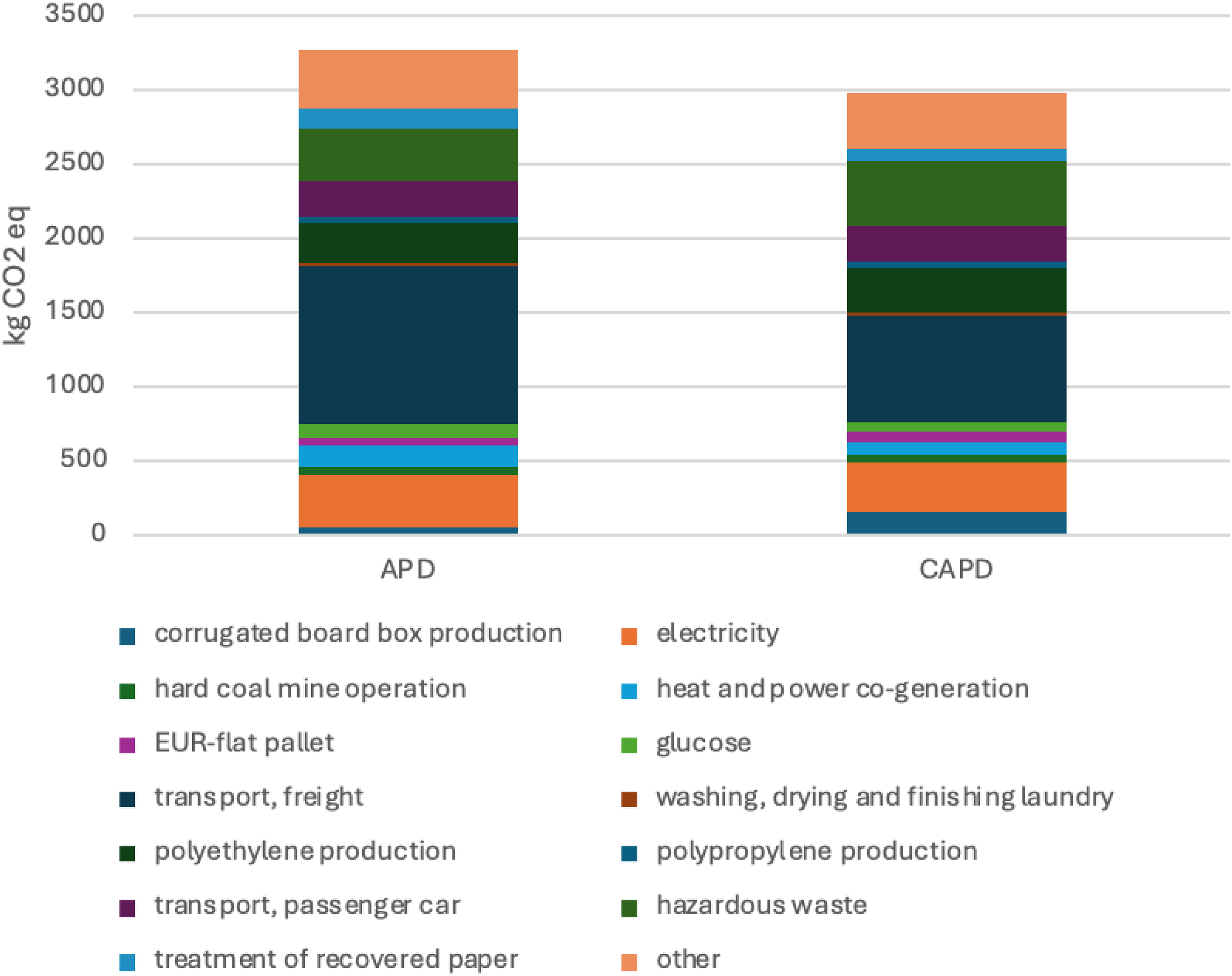
Contribution analysis of APD and CAPD.
Monthly examination
Supplementary Table S2 presents the CO2e emission associated with a single Monthly Examination. Patient travel contributes the largest share at 208 kg CO2, followed by staff travel at 28 kg CO2e. Other emissions include waste (4 kg CO2), plastic and other materials (7 kg CO2e combined), electricity for medical equipment (2 kg CO2) and manufacturing impacts (2 kg CO2).
Incremental approach
Table 3 compares the annual carbon footprint of different dialysis modalities. Standard APD results in the highest emissions at 3267 kg CO2e, followed by CAPD at 2975 kg CO2. Incremental approaches significantly lower emissions: Incremental APD emits 1642 kg CO2 and Incremental CAPD 1517 kg CO2e.
Results by process
Staff and patient travel (the same among APD and CAPD)
Supplementary Figure S2 shows that patient journeys contribute 311 kg CO2e per year while staff travel contributes 80 kg CO2.
Plastic material for APD and CAPD
Supplementary Figure S3 compares the amount of plastic materials used between APD and CAPD. CAPD involves more polyethylene (310 g vs. 275 g in APD), whereas APD uses more polypropylene (43 g vs. 30 g) and significantly more PVC (44 g vs. 1 g).
Disposal for APD and CAPD consumables
Supplementary Tables S3 and S4 present a comparison of waste profiles for APD and CAPD. APD generates a total of 145 kg of healthcare waste (general + clinical as PD takes place at home) resulting in 374 kg CO2, while CAPD generates a combined 320 kg healthcare waste with a higher impact of 810 kg CO2. CAPD also produces more cardboard and paper waste.
Results by environmental impacts
The results of the LCA are shown in Table 4. All results are reported as annual impacts per patient. The results include all 16 impact categories of PD and emphasise its environmental impact. The analysis included 16 distinct impact categories, reflecting a wide range of environmental concerns. These results highlight the environmental burden of PD and where improvements could be targeted.
LCA results across 16 impacts.

The results reveal that the environmental impacts of PD are significant across various categories, particularly climate change, acidification, ecotoxicity, eutrophication, resource use and human health impacts.
The results were normalised as recorded in Figure 3. Normalisation is the process of comparing the environmental impacts of the system to a broader reference for comparison.
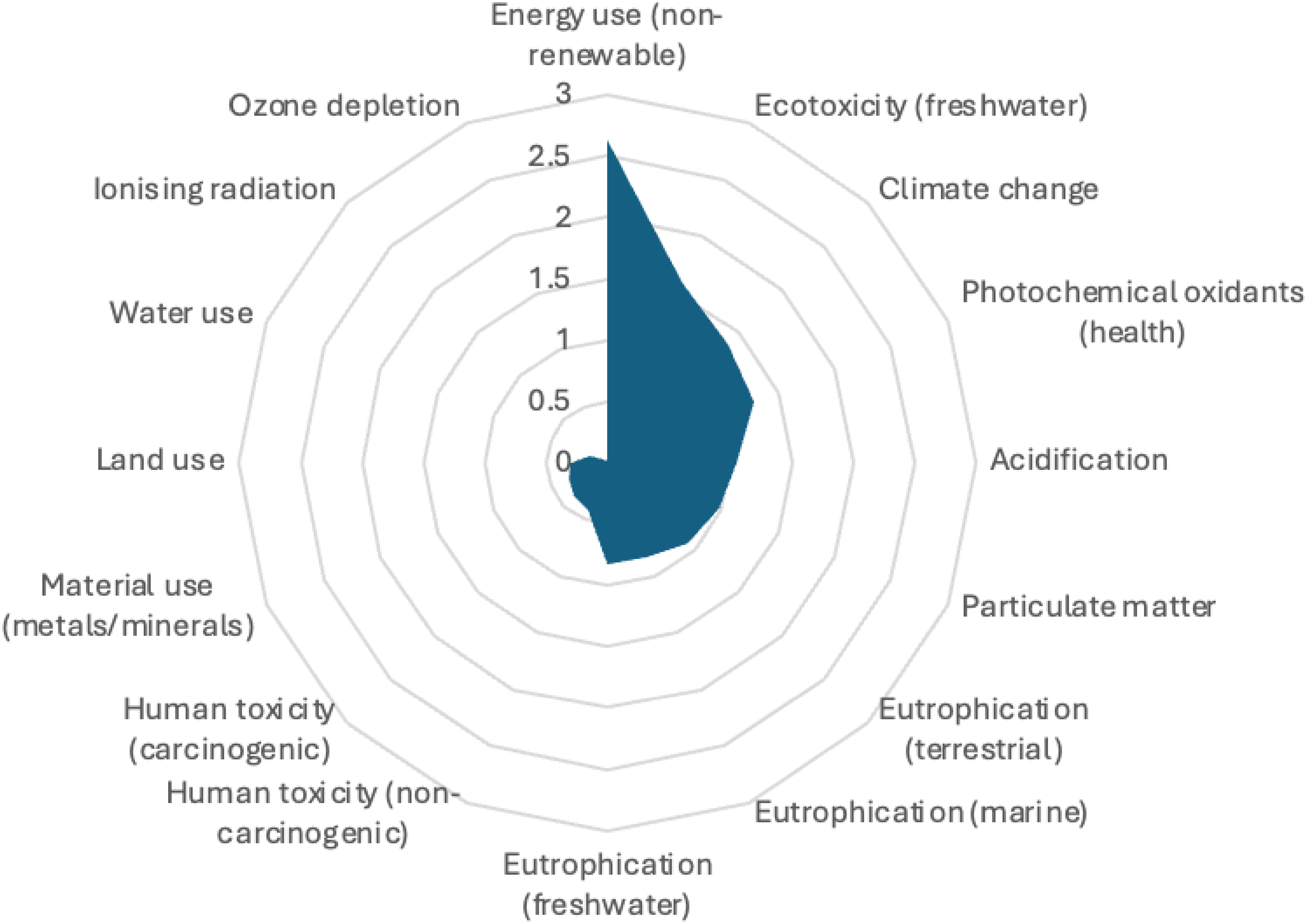
Normalised scores across 16 impacts.
The highest environmental burdens were associated with non-renewable energy use (2.63), freshwater ecotoxicity (1.59) and climate change (1.38). Other notable impacts included photochemical oxidants (1.29), acidification (1.05) and particulate matter (0.99). These categories align with KitNewCare key performance indicators, highlighting them as priority areas for intervention.
Supplementary Figures S4–S8 show the comparison between the APD and CAPD treatments across the main environmental categories.
Acidification: APD results in higher total emissions across freight (6.97 vs. 2.28 AE), electricity (0.28 vs. 0.07 AE) and PVC-related processes (0.16 AE in APD only). CAPD shows higher values for corrugated box use (0.52 AE) and hazardous waste (0.37 vs. 0.20 AE). Ecotoxicity (Freshwater): CAPD is more affected by hazardous waste (4172.28 CTUe vs. 3432.84 CTUe), while APD contributes more via transport (4470.99 vs. 3110.75 CTUe) and glucose (1345.14 vs. 807.08 CTUe). Energy Resources: APD has larger footprints for freight (9062.07 MJ vs. 7246.36 MJ), polyethylene (8041.63 vs. 5993.98 MJ) and PVC (795.23 MJ, not used in CAPD). CAPD's other significant contributors include uranium (387.40 MJ) and lignite (715.14 MJ). Photochemical Oxidants: APD sees larger impacts from freight (6.86 vs. 3.14 kg NMVOC-Eq) and PVC (0.13, not used in CAPD), while CAPD has higher contributions from hazardous waste (0.31 vs. 0.17). Water Use: APD’s glucose use (178.33 m3 vs. 107.00 m3), polyethylene (27.44 m3) and extrusion (8.09 m3, not found in CAPD) dominate. CAPD leads in corrugated board (29.76 m3) and hazardous waste (38.47 m3 vs. 10.87 m3).
Supplementary Figures S9 through S20 provide detailed breakdowns of environmental impacts by the top five more impactful procedure types (Monthly Examination, PET, APD Training and Changing Catheter) across the five highest environmental impacts from the normalisation and also including water use.
Climate Change: Monthly Examination contributes the most to climate change at 135.65 kg CO2-Eq, primarily from passenger car travel. PET produces 17.92 kg CO2-Eq, APD Training 16.59 kg CO2-Eq and Changing Catheter 15.03 kg CO2-Eq. Acidification: Monthly Examination has the highest impact at 0.299 mol H+-Eq, primarily from freight and passenger car transport and natural gas. PET contributes 0.063 mol H+-Eq, while APD Training and Changing Catheter have lower impacts at 0.050 and 0.005 mol H+-Eq respectively. Ecotoxicity, Freshwater: The largest contributor is Monthly Examination at 259.39 CTUe, primarily due to benzene and petroleum emissions. PET follows at 34.27 CTUe, with APD Training at 31.73 CTUe and Changing Catheter at 27.53 CTUe. Energy Resource Use: Monthly Examination again shows the highest impact at 2532.70 MJ, followed by PET at 334.96 MJ, APD Training at 308.66 MJ and Changing Catheter at 271.02 MJ. Petroleum dominates the energy resource category. Photochemical Oxidants: Monthly Examination leads with 0.281 kg NMVOC-Eq, driven by passenger car and freight transport. PET and APD Training follow at 0.042 and 0.033 kg respectively, while Changing Catheter contributes 0.036 kg. Water Use: Monthly Examination has the highest water use at 49.05 m3 world eq. deprived, predominantly from textile usage. PET, APD Training and Changing Catheter result in 1.60, 0.44 and 1.22 m3 respectively.
Discussion
This European LCA of the full PD pathway identifies the main contributors to environmental burden across materials, energy, waste, transport and clinical workflows. Aggregate results across 16 categories are in Table 1. Normalised scores in Figure 3 highlight non-renewable energy use, freshwater ecotoxicity and climate change as dominant categories, consistent with LCA method guidance and prior multi-category healthcare LCAs.7,18,19 Process contributions for APD and CAPD are in Figure 2.
APD showed higher total impacts than CAPD, driven by electricity for the cycler and PVC-containing disposables, while CAPD generated larger packaging and clinical waste streams. These treatment differences appear in Table 2 and in the contribution profiles in Figure 3. Plastic materials, in particular polyethylene and PVC, were consistent hotspots across categories. Within PD treatment, plastics account for about 41% of total CO2e in Pathway 11 based on the contribution analysis in Table 3. These patterns align with recent PD carbon studies in Australia and China that reported large contributions from dialysate, plastics and logistics, noting product and supply-chain differences between settings.17,20–22 The Australian study 21 analysed Baxter products, which contain higher proportions of PVC in dialysis bags and tubing, leading to PVC dominating the material footprint. The Modena centre used a mixed product supply that included Fresenius components with lower PVC content and a greater proportion of polyethylene, resulting in a smaller PVC contribution in this study. Chinese data 22 reported higher emissions per exchange, likely due to differences in supply chain energy sources and manufacturing processes.
Incremental prescriptions reduced impacts by lowering dialysate volume and consumable use during earlier treatment phases. Annual carbon footprints for the four modalities are in Table 4, with incremental APD and incremental CAPD showing the lowest CO2e. This is consistent with evidence that staged PD intensity can reduce plastic use and glucose exposure while preserving adequacy as residual kidney function declines.6,11
Normalisation results in Figure 2 show that focusing only on climate change misses other pressures. High freshwater ecotoxicity relates to upstream plastics production and end-of-life management. Energy resource use reflects fossil energy across manufacturing and freight. Photochemical oxidants are linked to transport and combustion. These findings are consistent with harmonisation work on toxicity in life cycle impact assessment and with guidance on water use indicators.18,19 Ozone depletion remains low, but related photochemical oxidants still warrant attention in transport and manufacturing. 23
Travel was a recurring hotspot for monthly in-centre reviews rather than for home treatment. Table 2 reports 250 kg CO2e per patient per year for monthly examinations, with patient travel the largest contributor as shown in Supplementary Table S2. Shifting routine reviews toward telehealth, community phlebotomy and validated home sampling can reduce travel emissions while maintaining clinical oversight.24–26
Waste and product design offer near-term improvement opportunities. CAPD generated more healthcare waste and higher disposal impacts than APD, as shown in Supplementary Tables S3 and S4. Material substitution away from PVC, design for disassembly and better segregation increase recovery of non-contaminated components, supporting lower toxicity and lower end-of-life burdens. 18 Freight and packaging also contribute, as shown in Figure 3, which supports local sourcing, packaging optimisation and lower-carbon logistics.17,21,24
Key take-away points for practice
Prioritise incremental PD where clinically appropriate to reduce dialysate volume and consumables.6,11 Reduce electricity impacts in APD through higher-efficiency cyclers and renewable household electricity, supported by energy supplier programmes and hospital facilitation.1,21 Substitute PVC and redesign components for recyclability to address toxicity and disposal burdens, using supplier standards aligned with LCIA characterisation guidance.
18
Consider incorporating telehealth and home or community testing approaches, where clinically appropriate and evidence-supported, to potentially reduce travel emissions while maintaining clinical oversight and patient safety.25,26 Optimise freight and packaging to lower upstream energy and transport impacts, building on lessons from recent PD carbon footprints in other regions.17,21,22
These findings provide a practical basis for the improvement opportunities summarised in Table 5 and align with the goals of KitNewCare to reduce environmental burdens while supporting high-quality care. 8
Summary of key improvement opportunities across the PD pathway.
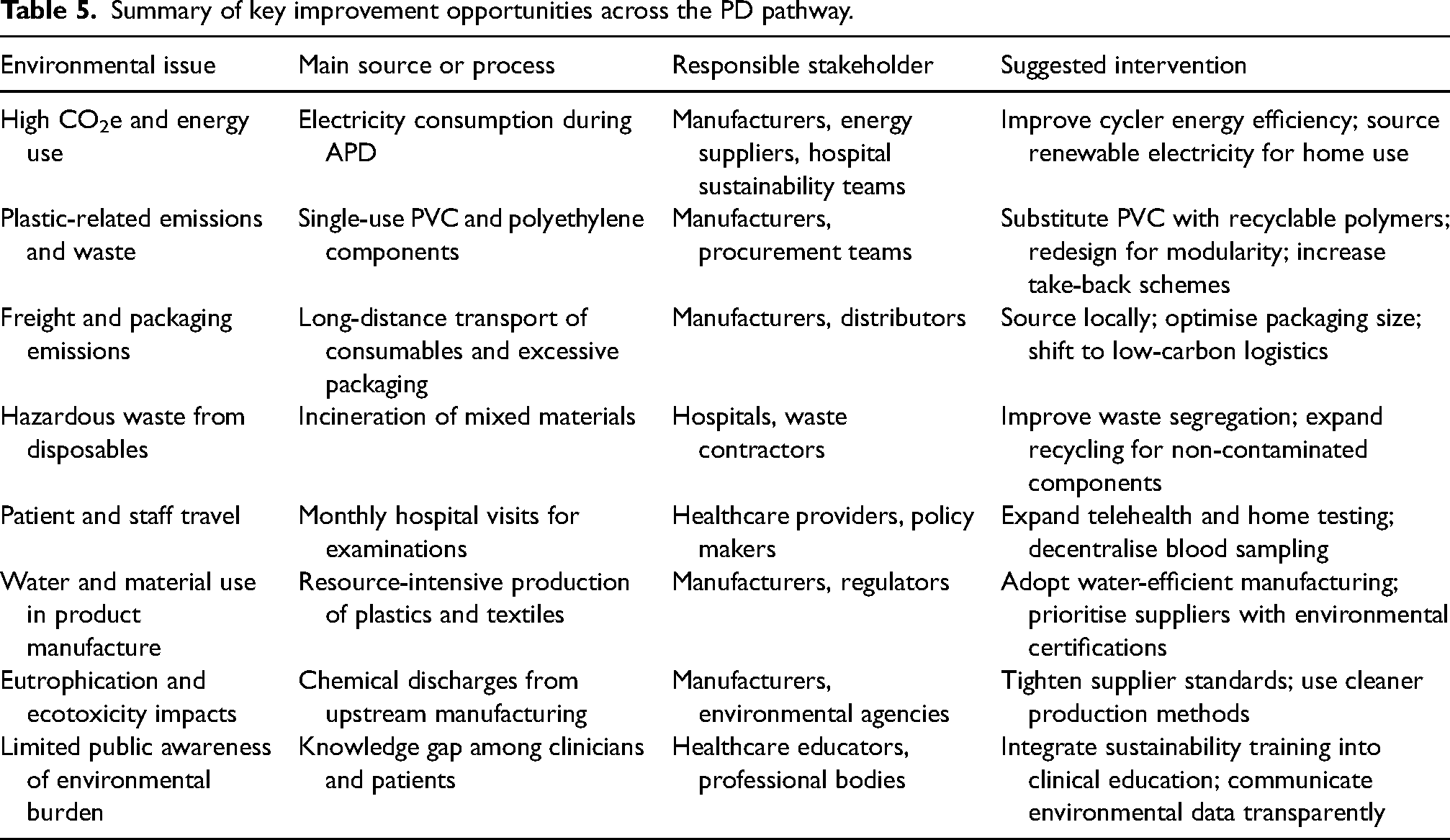
Limitations
This study has several limitations. Data collection was confined to a single site (AOU Policlinico di Modena) and a limited timeframe (April–July 2024), which may not reflect broader PD practices. Pharmaceuticals were excluded due to lack of reliable datasets, which may underestimate total environmental impacts. However, the KitNewCare project plans to replicate the study across three additional sites, enabling comparative analysis.
The LCA depends on the accuracy of input data, including material weights and types, which are available in the ‘Life Cycle Assessment Dataset for Peritoneal Dialysis in Modena’. Environmental modelling was limited by the available impact categories in OpenLCA and the Ecoinvent v3.10 database. While some healthcare-specific impacts (e.g., pharmaceutical emissions) are not fully represented, we selected 16 robust indicators to ensure methodological consistency and international relevance. Assumptions regarding patient travel mode, supply origins and household waste segregation may not fully reflect real-world variation and could influence results.
Conclusion
This article provides a comprehensive analysis of the environmental impacts of the PD treatment pathway over one year. The findings highlight product production, waste disposal and procurement as key contributors to the overall environmental burden. By addressing these areas, healthcare systems can reduce the environmental footprint of PD and promote more sustainable practices in kidney care.
Supplemental Material
sj-docx-1-ptd-10.1177_08968608251415439 - Supplemental material for Sustainable kidney care: A life cycle assessment of the peritoneal dialysis pathways
Supplemental material, sj-docx-1-ptd-10.1177_08968608251415439 for Sustainable kidney care: A life cycle assessment of the peritoneal dialysis pathways by James Larkin, Giulia Ligabue, Gaetano Alfano, Rodrigo Martínez Cadenas, Abass Fehintola, Ingeborg Steinbach, Aycan Yasar, Niccolo Morisi, Marta Arias-Guillen, Marialuisa Caiazzo, Gabriele Donati and Brett Duane in Peritoneal Dialysis International
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All data generated or analysed during this study are publicly available in the Zenodo repositories: Larkin, J. (2024) ‘Life Cycle Assessment Dataset For Peritoneal Dialysis in Modena’. Brett Duane. doi: 10.5281/zenodo.14258866; Larkin, J. (2024) ‘Product Images for Life Cycle Assessment Dataset For Peritoneal Dialysis in Modena’. Brett Duane. doi: 10.5281/zenodo.14258920.
Ethical approval
Ethical approval was not required for this study as it did not involve human participants or animal research. The study focused solely on environmental data collection and analysis related to peritoneal dialysis systems.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the EU Horizon Grant Funding Kitnewcare [grant number 101137054] and UKRI [grant number 10110449].
Informed consent to participate
Not applicable. This research did not involve any individual participants, patients, or human data.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
