Abstract
Background
Lack of data on the accuracy of continuous glucose monitoring systems currently limits their usage in people with diabetes on peritoneal dialysis (PD).
Aim
We aimed to assess the analytical and clinical accuracy of the FreeStyle Libre (FSL) continuous glucose monitoring system in people with diabetes on PD.
Methods
12 participants using Icodextrin in their PD regime were recruited to a single-centre observational study. They wore a blinded research model of the FSL for 10 days. Results from the FSL were compared with venous glucose measured on a Yellow Springs Instrument (YSI) and self-monitored capillary blood glucose (SMBG) recorded five times per day over the 10-day study.
Results
The mean absolute relative difference from 84 FSL-YSI matched pairs was 9.8% (95% CI 8.6–11.1) and from 416 FSL-SMBG matched pairs, it was 17.3% (95% CI 16.24–18.43). The systematic error for the FSL as determined by Bland–Altman analysis was −0.6 ± 1.0 mmol/l compared with YSI and −1.4 ± 1.9 mmol/l compared with SMBG. With regard clinical accuracy, compared with YSI and SMBG, respectively, 100% and 99.9% of sensor values were in clinically acceptable zones A and B of Parkes consensus error grid.
Conclusion
We demonstrated satisfactory performance of the FSL monitoring system by both analytical and clinical metrics in this cohort of PD patients using treatment prescriptions including Icodextrin-based fluids. Larger studies are now needed to provide clinicians with appropriate reassurance if this technology is to be used with confidence in people on PD.
This is a visual representation of the abstract.
Introduction
Up to 50% of patients undergoing peritoneal dialysis (PD) also have a diagnosis of diabetes. 1 These patients are at significant risk of both micro and macrovascular complications including heart attacks, strokes, amputations and sudden cardiac death. 2 The use of hypertonic glucose dialysate solutions also results in increased glucose exposure. 3 Reliably monitoring glycaemic control is challenging in this population as traditional markers of glycaemia such as HbA1c are much less accurate due to a comparatively limited and more variable lifespan of red blood cells 4 and the utility of alternatives such as glycated albumin and fructosamine remains unclear. Continuous interstitial glucose monitors (CGM) are now being widely used as a replacement for capillary glucose measurements for people with Type 1 and Type 2 diabetes.5,6 People using CGM benefit from significant additional information including glycaemic trends, glycaemic variability, time in range and detection of asymptomatic episodes of hypoglycaemia.
There is little evidence surrounding CGM use in dialysis patients especially those on PD 7 and their accuracy in this population remains largely untested. Several validation studies are ongoing in haemodialysis (HD) patients to assess whether the significant interstitial fluid shifts associated with that treatment affect the accuracy of these sensors. Although PD is not associated with the same degree of fluid shifts seen in HD, concern has been raised around possible interference with the enzymatic electrochemical reaction central to the CGM's function, from the dialysate itself. Glucometers are composed of a detector and one of three enzymatic reactions: glucose oxidase, glucose dehydrogenase (GDH) or hexokinase. GDH-based glucometers utilise a co-enzyme; pyrroloquinoline (PQQ), nicotine adenine dinucleotide or flavin adenine dinucleotide (FAD). These can be subject to interference from other molecules. Icodextrin, a glucose polymer which is used in some PD solutions as a glucose sparing osmotic agent, is metabolised to maltose which has previously been shown to interfere with GDH-PQQ-based glucose meters. Use of these non-specific glucose monitors in PD patients generated spurious normoglycaemic and hyperglycaemic readings resulting in serious adverse events including unrecognised hypoglycaemia and death.8,9 CGMs such as FreeStyle Libre (FSL) measure glucose in interstitial fluid using the glucose oxidase reaction. This has previously been shown to be highly selective for glucose in other glucometers; however, accuracy can vary depending on the source of the glucose oxidase. The FSL is not currently recommended by its manufacturer for use in dialysis patients as despite its GO-based chemistry there is not definitive scientific evidence for Icodextrin compatibility. 10
Consequently, their use in both clinical and research settings is limited.
We aimed to study the analytical and clinical accuracy of the FSL continuous glucose monitoring system in patients on PD in a real-world setting against a laboratory gold standard (Yellow Springs Instrument (YSI)) and the current standard of clinical care (self-monitored capillary blood glucose (SMBG)).
Research design and methods
Study design and population
This was a single-centre, prospective, open-label study of the continuous glucose monitor FSL ProiQ compared to laboratory gold standard and clinical standard of care measures of glucose. Patients with diabetes on PD were recruited at the Royal Devon University Healthcare NHS Foundation Trust. Participants were recruited from inpatient and outpatient settings.
Participants were eligible for inclusion if they were aged over 18 years, had a diagnosis of type 1 or type 2 diabetes and had been on PD for at least 2 months. Participants were excluded if they had a known allergy to any component of the CGM, they had a planned MRI or CT scan within the study period or they were on hydroxyurea (a medication known to interfere with the enzymatic reactions within the sensor).
Full-written, informed consent was obtained from all participants.
Baseline bloods were collected which included full blood count, renal function, liver function, CRP, lipid profile and HbA1c.
For those patients recruited in an outpatient setting, participants were fitted with the FSL ProiQ (Abbott Diabetes Care, Alameda, CA), a blinded research model of the FSL sensor, on their upper arm as per manufacturer's instructions. This model is factory calibrated and does not require further calibration once fitted. This model uses a glucose oxidase enzymatic reaction; this has previously been shown to be highly selective for glucose in other glucometers; however, accuracy can vary depending on the source of the glucose oxidase. We were unable to obtain information on sourcing of the glucose oxidase from the manufacturer. They wore the sensor continuously for the duration of the study which was 10 days. All participants were given a calibrated point of care testing glucose meter (GlucoRx HCT with GlucoRx HCT test strips) and re-education on accurately performing a SMBG test. The GlucoRx uses GDH-FAD enzymatic reaction and has previously been tested by the manufacturer for compatibility with maltose and Icodextrin to approved limits. Over the 10-day study period participants were asked to perform SMBG measurements five times per day which they recorded in a diary (with corresponding date and time of the sample); the timing of these measurements was by patient choice and not specified by the research team. They also recorded details of their PD prescription. Participants attended for a single-study visit at the NIHR Exeter Clinical Research Facility during which up to 10 venous blood samples were taken over a 4-h period via an intra-venous cannula and analysed immediately at the bedside on a YSI (YSI 2300 STAT Plus, YSI UK Ltd, Hampshire), which was calibrated before each study visit as per manufacturer's instructions. The YSI uses the glucose oxidase reaction; no interference by maltose has been demonstrated up to concentrations of 480 mg/dL. YSI is accepted as the reference method for testing accuracy of blood glucose monitors by the FDA and the majority of glucose meters are factory calibrated using this device as the standard.
Patients recruited in the inpatient setting were fitted with the same FSL ProiQ device as those recruited in the outpatient setting; however, the five times per day SMBG measurements were performed by nursing staff on the ward.
At the end of the study period, the FSL was removed; data were downloaded using LibrePro software. Each recorded FSL measurement is time stamped. This time stamp was used to retrospectively pair data points with the closest matched venous or capillary glucose readings for comparison.
All participants continued their PD prescription and diabetic treatment as prescribed by their responsible clinical team throughout the whole period of the study.
Ethical approval was obtained from the West London and GTAC Research Ethics Committee (Reference 22/PR/0723) and the study was conducted as per the Declaration of Helsinki.
Outcome measures
Aggregate mean absolute relative difference
Mean absolute relative difference (MARD) is a comparison between measurements from the investigated system and the reference method. Aggregate MARD was calculated as the mean value of the absolute relative differences for all temporally paired glucose measurements.
Absolute relative difference = 100% x [yFSL(tk) – yref(tk)]/yref(tk)
Where yFSL = value measured by FSL, yref = value measured by reference method at time k (tk).
MARD was calculated as an aggregate value from the total number of paired measurements for (1) FSL versus YSI and (2) FSL versus SMBG, over the whole glycaemic range.
MARD is reported as a percentage, the lower the value of MARD the closer the FSL readings are to the reference method. The Advanced Technologies and Treatments for Diabetes Congress guidelines recommend a MARD of ≤10% for continuous glucose monitors.11,12
Spearman's correlation coefficient
The data were found to be non-normally distributed, and therefore the differences between FSL readings and the paired comparators were also represented by Spearman's correlation coefficient.
Bland–Altman plot
A Bland–Altman plot is a plot of individual differences between results of the investigated system and the comparison method against their mean value. These plots allow the measurement of systematic bias and calculation of 95% limits of agreement within which 95% of differences are expected to lie for different systems to be considered similar in terms of quantitative performance.
Parkes Error grid
The Parkes Error grid originally devised in 2000 13 and revised in 2013 14 demonstrates the clinical risk associated with the distribution of results between the investigated system and the comparison method. Pairs of glucose measurements are categorised by 5 risk zones graded from zone A ‘no effect on clinical action’ to zone E ‘altered clinical action that could have dangerous consequences’. The error grid was incorporated into the guidelines produced by the International Organization and Standardization in 2013 (ISO 15197:2013), which stipulates that to be an acceptable method of blood glucose monitoring 99% of measurement results should fall within zones A and B, limiting the number of clinically unacceptable results to 1%. 15
Secondary outcomes
The glucose management indicator (GMI), a metric generated by the LibreView software, converts mean glucose readings derived from the CGM into an estimate of HbA1c using a population-based formula. 16 The relationship between GMI and HbA1c and Hba1c and average glucose was described using Spearman's correlation coefficient.
We also aimed to describe the 24-h glycaemic profiles of these patients with diabetes on PD with regard time in range and the influence of different PD prescriptions.
Statistical analyses were performed using StataSE version18.
Results
A total of 416 paired FSL-SMBG and 84 paired FSL-YSI were obtained from 12 participants who were recruited into the study between August 2022 and March 2024. Paired data points were taken as the reference value and the FSL value which was most closely temporally matched. The mean and median time between the two readings was 3.6minutes and 3 min, respectively. Two participants were recruited from an inpatient setting and the remainder from a community setting. One participant withdrew from the study after the FSL sensor fell off on the second day, no other adverse events related to the study occurred. The remaining 11 participants completed the entire study protocol with 100% valid FSL data. The demographic characteristics of the study participants are represented in Table 1. Nine participants were on automated peritoneal dialysis (APD) and two on continuous ambulatory peritoneal dialysis (CAPD); all participants had been on a stable prescription that included one daily Icodextrin dwell for at least 1 month prior to the study. The average residual urine output was 1127mls/24hours; this ranged widely from 100mls to 2200mls. The mean residual renal contribution to kt/v, measured using a 24 h collection, was 0.96 ± 0.54. We found no correlation between MARD for CGM-SMBG or CGM-YSI and residual kidney function, as measured by renal kt/v.
Participant characteristics.

Data are presented as mean ± SD, median (range) or n (%). PD: peritoneal dialysis; CAPD: continuous ambulatory peritoneal dialysis.
Accuracy of FreeStyle Libre by analytical and clinical measures
Aggregate MARD for FSL versus YSI from 84 paired FSI-YSI samples was 9.8% (95% CI 8.6–11.1). Aggregate MARD for FSL versus SMBG from 416 paired FSL-SMBG samples was higher at 17.3% (95% CI 16.24–18.43).
Zones A and B of the Parkes Error Grid are defined as the clinically acceptable zones, wherein any differences would have no significant impact on clinical outcome. Over the full glycaemic range, the percentage of FSL-YSI pairs in zone A of Parkes Error grid was 96.4% (n = 81) and in zone B was 3.6% (n = 3). For FSL-SMBG pairs, this was 80.5% (n = 335) in Zone A, 19% (n = 79) in Zone B and 0.5% (n = 2) in Zone C (Figure 1).
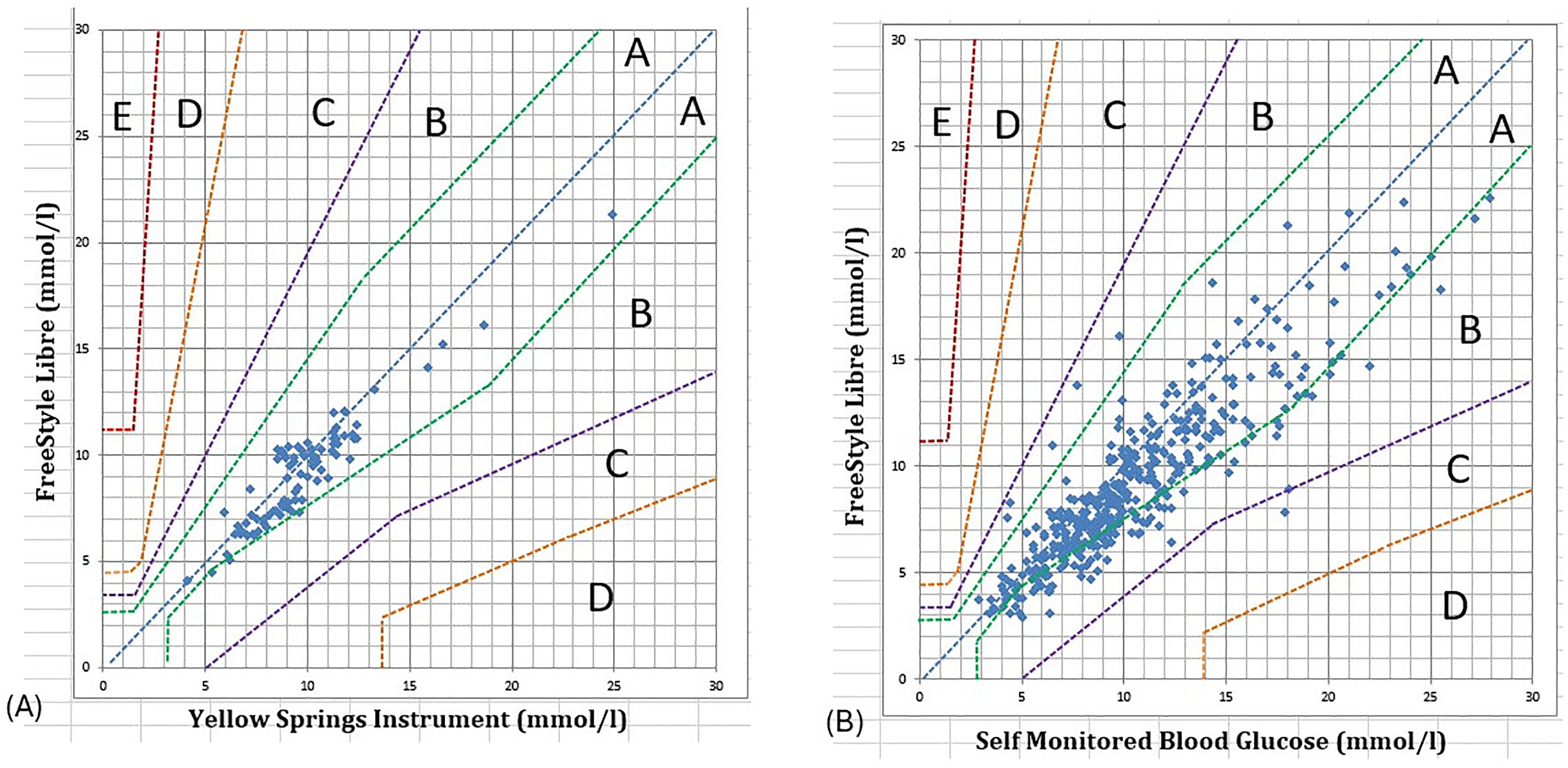
Parke's consensus error grid analysis of (A) FSL values vs. YSI (n = 84) and (B) vs. SMBG (n = 416). 5 clinical risk zones – Zone A no effect on clinical action, Zone B little or no effect on clinical outcome, Zone C likely to affect clinical outcome, Zone D could have significant medical risk, and Zone E could have dangerous consequences. FSL: FreeStyle Libre; YSI: Yellow Springs Instrument; SMBG: self-monitored capillary blood glucose.
There was a strong correlation between FSL and YSI (rs = 0.94) and FSL and SMBG (rs = 0.92).
The systematic difference between FSL and YSI was −0.6 ± 1.0 mmol/l (−10.8 ± 18.0 mg/dL) and −1.4 ± 1.9 mmol/l (−25.2 ± 34.2 mg/dL) for SMBG (Figure 2).
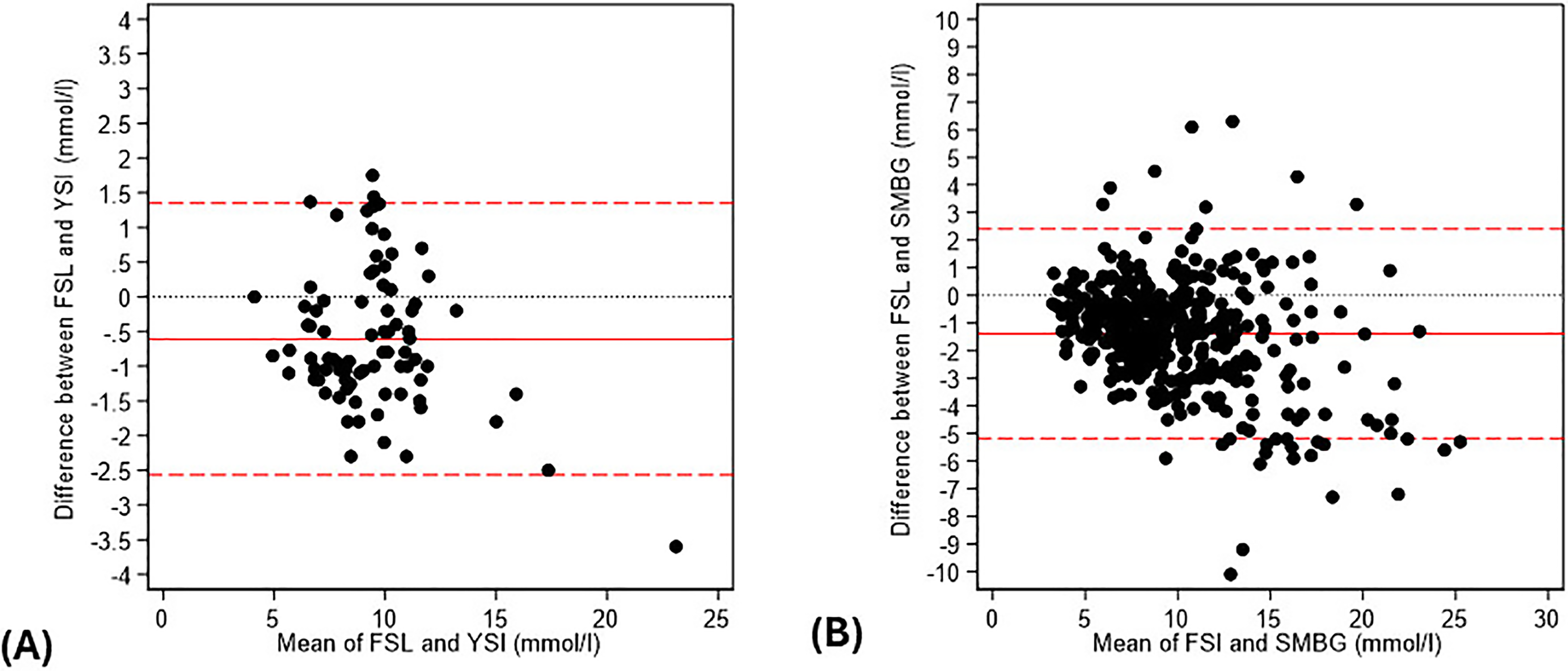
Bland–Altman plots for FSL compared with (A) YSI (n = 84) and (B) SMBG (n = 416). There was a negative systematic bias between the FSL and the reference measure (solid red line) of −0.6 mmol/l for YSI (A) and −1.4 mmol/l for SMBG (B). Dotted red lines = 95% CI. FSL: FreeStyle Libre; YSI: Yellow Springs Instrument; SMBG: self-monitored capillary blood glucose.
Glycaemic profiles in patients with diabetes on peritoneal dialysis
Average time in range (3.9–10 mmol/L or 70.2–180 mg/dL) was 63.3 ± 21.9%. Average time spent above 10 mmol/L (180 mg/dL) was 33.7 ± 22.9% and average time spent <3.9 mmol/l (70.2 mg/dL) was 3 ± 3.7%. There were only three pairs of readings (obtained from two separate patients) with a reference glucose <3.9 mmol/l (<70.2 mg/dL) but all 3 were correctly recorded as <3.9 mmol/l by the FSL and all were within 0.4 mmol/L (7.2 mg/dL) of the reference.
Average HbA1c was 7.3 ± 1.1% (55.7 ± 12.4 mmol/mol); average GMI was 7.2 ± 0.86% (55.4 ± 9.4 mmol/mol). There was a strong correlation between both HbA1c and GMI (rs = 0.94 p = 0.0002) and HbA1c and average glucose (rs = 0.92 p = 0.0003). On average, the GMI was 0.1 ± 0.5% (1.2 ± 5.6 mmol/mol) lower than HbA1c. 45% of patients (5/11) had an absolute discordance of ≥0.5% (≥6 mmol/mol) between laboratory-measured HbA1c and FSL-calculated GMI. See supplementary Figure 1 for Bland–Altman distribution.
Visual inspection of the 10-day glycaemic profiles demonstrated different patterns of glycaemic exposure for those on CAPD compared with APD (example traces from two participants can be seen in Figure 3). Patients on CAPD exhibited a traditional pattern of glycaemia with higher glucose levels during the day, peaks associated with meals and PD exchanges and lowest glucose levels overnight. In contrast, for those on APD where the majority of the glucose containing solution is administered overnight, daytime and nighttime average glucose levels were largely similar.
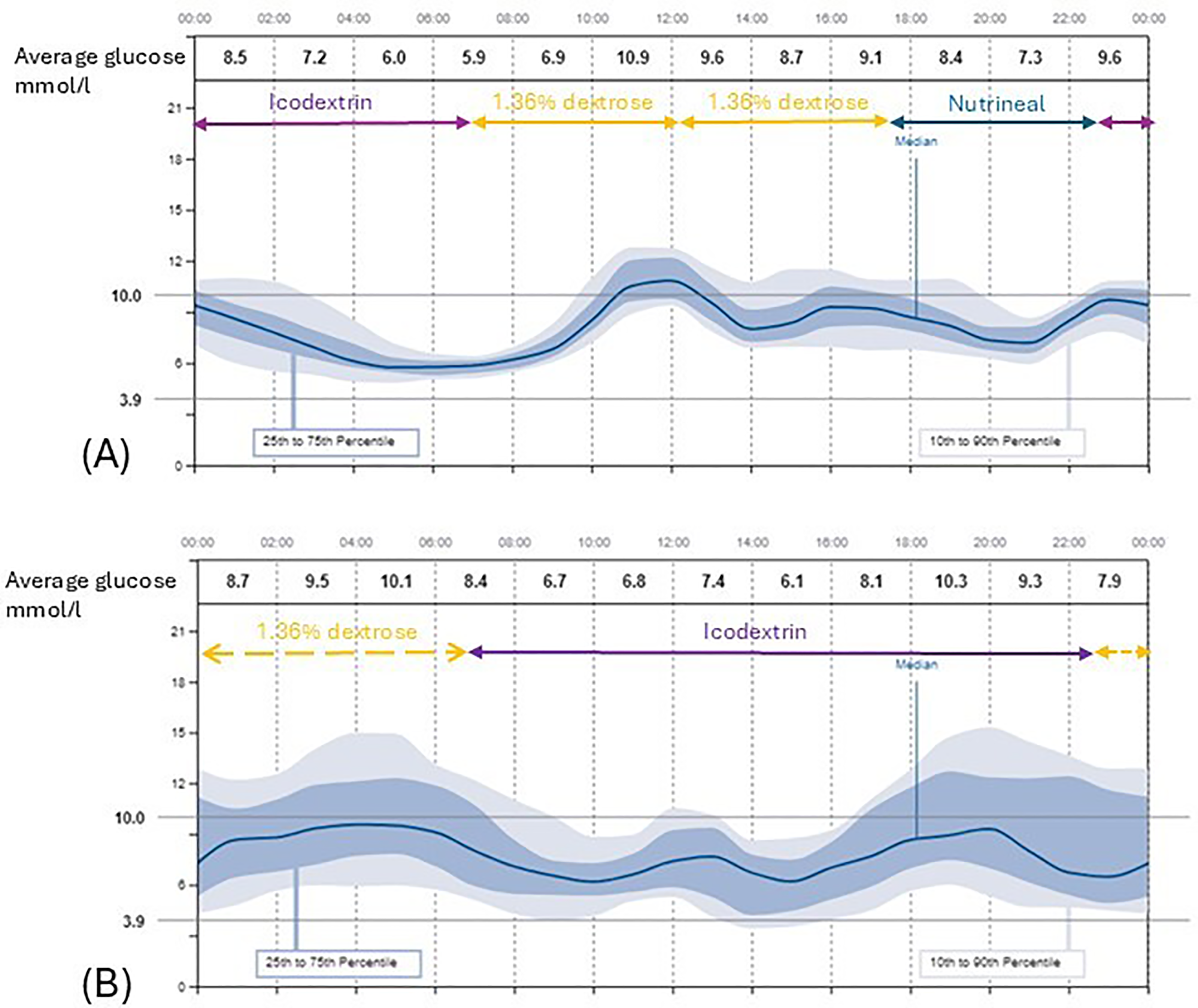
Examples of 10 day ambulatory glucose profiles from a patient on CAPD (A) and APD (B). The individuals PD regimen is demonstrated by the arrows – solid arrows = CAPD fill, broken arrows = APD. PD: peritoneal dialysis; APD: automated peritoneal dialysis; CAPD: continuous ambulatory peritoneal dialysis.
Discussion
In this real-world study, we report the performance of the FSL proIQ glucose monitor in patients on PD using multiple analytical and clinical metrics. Advanced Technologies and Treatments for Diabetes Congress guidelines recommend a MARD of ≤10% for continuous glucose monitors.11,12 Compared with YSI which is the gold standard measure we report an aggregate MARD of 9.8%. Compared with SMBG, the MARD was higher at 17.3%.
These results are comparable with the only other study of CGM accuracy (Medtronic Guardian 3 sensor) solely in PD patients who reported an aggregate MARD of 10.4% compared with YSI. 7 They report a better MARD of 9.3% for CGM versus SMBG. Notably patients using Icodextrin were excluded from this study and to our knowledge ours is the first study to test accuracy of a continuous glucose monitor in PD patients using Icodextrin containing fluids. With a plasma half-life of 14.7 h, plasma levels of Icodextrin and its metabolites return to baseline 3 to 7 days after the last dwell, 17 and therefore any potential interference would affect the whole 24-h period. Plasma levels of Icodextrin metabolites are thought to reach steady state after 2–4weeks of daily exposure; 18 all the patients in this study had been using daily Icodextrin exchanges for at least 1 month at the time of entry into the study. The prescription of Icodextrin containing fluids varies widely between different countries. In the Peritoneal Dialysis Outcomes and Practice Patterns Study (PDOPPS), the proportion of CAPD patients prescribed Icodextrin varied from 0% (Thailand) to 78% (UK). 19 In the majority of countries at least 50% of the CAPD population had Icodextrin included in their regime. Interference by maltose or other Icodextrin metabolites may explain the increased MARD in this study compared with the previously reported results. However, interference by Icodextrin metabolites has previous been shown to result in erroneous readings by glucometers that were systematically higher than the reference methods; 8 in this study, comprised entirely of patients using Icodextrin, we found that the systematic error (bias) of the CGM was lower than both reference methods.
Overall, MARD in HD populations is reported to be higher and more variable, ranging from 11.3% to 21.4%.20–24 This can likely be explained by acute fluid and insulin shifts during HD sessions leading to increased glycaemic variability; the accuracy of CGM systems is lower when glucose levels are rapidly changing.25–27 Additionally, the majority of these studies use SMBG as their sole comparator. The reported MARD in non-CKD populations is slightly better, 7.8–8.9% compared with YSI.28,29
Clinical point accuracy as assessed using Parke's Error Grid was reassuring, compared with YSI 100% of the CGM pairs were in zones A and B, for SMBG 99.5% of pairs were in zones A and B. Therefore, in this study, any discrepancy between the CGM and the standard measures of glycaemia would not have led to any meaningful change in clinical outcome.
Although not the primary purpose of this study it is of interest to look at the relationship between CGM-generated outcomes and traditional markers of glycaemic exposure. In our study, HbA1c and GMI were strongly correlated however nearly half of our patients had a 0.5% (6 mmol/mol) or greater discrepancy between laboratory-measured HbA1c and CGM-generated GMI. Another real-world study including both CKD and non-CKD patients 30 also reported a difference of >5% between HbA1c and GMI in 50% of their participants. GMI which is a relatively short-term measure of glycaemia is expected to differ from long-term exposure as measured by HbA1c. It has been proposed that the difference between the two would be stable over time within most individuals; 31 however, the effect of reduced red cell life span and exogenous erythropoietin administration on this has not been studied. HbA1c has well documented limitations in the CKD population4,32 and GMI has not been adequately studied in CKD therefore neither should be relied upon in isolation to determine individual patients glycaemic control. 33
CGMs provide a complete 24-h ambulatory glycaemic profile; however, there remains relatively little literature around these profiles in PD patients and particularly rates of hypoglycaemia. We observed very little hypoglycaemia during this study with on average 3% of time spent with glucose less than 3.9 mmol/l (70.2 mg/dL). This is in keeping with other CGM studies in PD 34 and considerably less than has been reported in HD studies where HD-induced hypoglycaemia is frequently observed. 35
This study has a number of strengths, notably the inclusion of patients on Icodextrin containing prescriptions and the inclusion of two comparators, YSI as the gold standard and SMBG as the clinical standard of care. It's real-world setting with the majority of SMBG comparators performed by the patient is helpful for clinicians who wish to translate this into their clinical practice.
There are also limitations. Although our data is based on a large number of paired glucose readings, these were collected from a relatively small number of patients. For the SMBG comparison data was collected over a 10 day period and the examined glucose range was 3.0 to 28 mmol/l (54.0 to 504 mg/dL). The YSI values were obtained over a 4 h period on a single day and the range was narrower; with very few points in the hypoglycaemic range, it was considered that attempts to manipulate glucose into the hypoglycaemic range would be unethical. This limits the study's ability to comment on FSL performance at extremes of glycaemia. Participants in this study had a mean residual urine volume of 1127mls/24hrs. As the metabolites of Icodextrin that have been demonstrated to interfere with glucose assays are renally excreted, it should be acknowledged that anuric patients or those with significantly reduced urine volumes may suffer more interference from these metabolites than has been demonstrated in this study.
Here we report the real-world performance of the FSL glucose monitoring system using multiple analytical and clinical metrics in a cohort of PD patients using treatment prescriptions including Icodextrin-based fluids. Point analytical accuracy was satisfactory compared to the gold standard YSI over a 4 h period but more variation was seen in comparison to SMBG. The systematic error did not suggest any significant interference from Icodextrin metabolites. Although we have demonstrated satisfactory performance by clinical metrics clinicians should be aware of the potential for deviation from the true blood glucose. Larger studies are now needed to provide clinicians and regulatory bodies with sufficient data to allow these technologies to be used with confidence in people on PD.
Supplemental Material
sj-jpg-1-ptd-10.1177_08968608251406514 - Supplemental material for Accuracy of a continuous glucose monitoring system in people with diabetes on peritoneal dialysis
Supplemental material, sj-jpg-1-ptd-10.1177_08968608251406514 for Accuracy of a continuous glucose monitoring system in people with diabetes on peritoneal dialysis by Jennifer K Williams, Mark Gilchrist and Angela C Shore in Peritoneal Dialysis International
Footnotes
Declaration of conflicting interest
J.K.W and A.C.S declare no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
M.G has received speaker fees from Astra Zeneca.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by project grants from the Northcott Devon Medical Foundation (TB/MG/NO5002) and Royal Devon University Hospital Foundation Trust Research Capacity Building fund.
Ethical considerations
Ethical approval was obtained from the West London and GTAC Research Ethics Committee (Reference 22/PR/0723).
Consent to participate
Written informed consent was obtained from all participants.
Consent for publication
All participants gave written consent for publication of study results in an anonymised format.
Author contributions
JKW, MG and ACS all conceived the project. JKW conducted all research visits, collated and analysed the data and wrote the first draft of the manuscript. MG and ACS reviewed and edited the manuscript. All authors approved the final version of the manuscript.
Data availability
The datasets generated and analysed in the current study are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
