Abstract
Background
Peritoneal dialysis (PD) is being promoted because it is cost-effective and has equivalent outcomes to facility-based hemodialysis (HD). Determining PD eligibility is critical but subjective, with high variability among renal programs. This study aimed to establish a predictive model for PD eligibility among individuals who started treatment with HD. A secondary objective was to identify predictors of PD eligibility and determine if eligible patients went on to receive PD.
Methods
This retrospective cohort study included individuals starting HD at multiple hospitals in Alberta, Canada, as part of the START program between 1 October 2016 and 31 March 2018. Twenty-seven predictors, including patient characteristics, laboratory values, and comorbidities, were considered in logistic regression modeling. The outcome variable was PD eligibility, as determined by a standardized interdisciplinary assessment. The model selection was based on the Akaike information criterion. The confusion matrix was used for each model to compare the predicted versus observed eligibility. The final model was calibrated and presented.
Results
Among the 598 participants, 391 (65.4%) were considered eligible for PD. The logistic regression model achieved a modest performance in discriminating patients who were eligible for PD, with a high sensitivity of 91.3%, an accuracy of 0.68 (95% CI, 0.65–0.72), and an area under the receiver operating characteristic curve ranging from 0.69 to 0.71. Age (OR = 0.98; 95% CI, 0.97–0.99), body mass index (OR = 0.95; 95% CI, 0.93–0.97), starting dialysis in intensive care unit (OR = 0.53; 95% CI, 0.31–0.92), and polycystic kidney disease (OR = 0.37; 95% CI, 0.13–0.99) were statistically significant factors associated with a lower likelihood of being considered eligible for PD. Out of the 391 eligible PD patients, 87 (22.3%) received PD treatment within 6 months of starting HD.
Conclusions
The majority of patients starting HD were considered eligible for PD. Our model exhibits a high level of sensitivity and could serve as a valuable tool for screening potential candidates following the commencement of HD.
Introduction
Peritoneal dialysis (PD) and hemodialysis (HD) are both established modalities for renal replacement therapy in patients with kidney failure. While both methods have shown similar overall survival rates, PD has been promoted as a dialysis treatment option for end-stage kidney disease (ESKD) patients with kidney failure because it maintains patient independence and is cost-effective.1–6
The utilization rate of PD among patients with ESKD kidney failure is highly variable among different regions and populations. 7 Studies have shown that factors such as patient eligibility, referral patterns, and physician preference can play a significant role in determining the rate of PD use and should be carefully evaluated. 8 Studies carried out in North American nephrology practices indicated that 87% of patients were deemed eligible for PD, and psychosocial evaluations suggested that 83% met the eligibility criteria. 9 Enhanced patient education, 10 promotion of home dialysis, and empowering patient choice can increase the utilization of home-based modalities. Early identification of potential candidates for PD can improve treatment outcomes, as patients may be resistant to changing their modality over time.
Determining PD eligibility can be subjective and variable. 9 Ideally, an experienced multidisciplinary team would determine a patient's eligibility for PD and offer it to all potential candidates. Using objective criteria to screen for eligible candidates can be beneficial, as determining eligibility in some patients can be time-consuming and labor-intensive. Developing predictive models may be useful to address this issue. This study aims to investigate whether a standardized predictive model for PD eligibility among individuals who initiate HD can be developed and to examine the factors associated with PD eligibility. Our goal was to predict eligibility for PD as the initial modality choice for patients starting dialysis.
Materials and methods
Study design and population
The data of this retrospective cohort study was collected prospectively from renal programs at multiple hospitals in Alberta, Canada, as part of the START program. 11 The study population was restricted to adults aged 18 years and older. Incident patients with end-stage kidney failure between 1 October 2016 and 31 March 2018, were included if their first modality was HD or continuous renal replacement therapy. We excluded patients who had a pre-emptive transplant or were transferred into the dialysis program already established on HD (prevalent patients). Our tool was specifically designed to assess patients who have commenced HD but may be eligible for PD, and thus, it should be ideally utilized within that population.
Data source and measurements
Baseline variables
The dataset used in this study consists of 598 patients with 25 variables. The baseline variables included patient age, sex, body mass index (BMI), and the province of residence. Baseline comorbidities included diabetes, coronary artery disease, congestive heart failure, other cardiac diseases (e.g., arrhythmias), polycystic kidney disease, history of gastrointestinal bleeding, cancer, cerebrovascular disease, peripheral vascular disease, and chronic obstructive lung disease. We also determined if and whether the patient received predialysis care of 4 months or greater, whether they started dialysis in the hospital or the intensive care unit (ICU), and the date of their first outpatient dialysis treatment (if it they started dialysis in the hospital); (ii) laboratory values included serum creatinine, albumin, hemoglobin, parathyroid hormone phosphate, calcium, and bicarbonate. Laboratory values were collected immediately before the start of dialysis and were therefore not affected by the dialysis treatment. For example, serum creatinine prior to the dialysis treatment reflects the kidney function at dialysis initiation.
Outcome variable
The outcome variable, PD eligibility (yes/no) assessment, was determined through an interdisciplinary assessment, adhering to standard clinical practices. This assessment involved completing a standardized form listing empirically developed contraindications and barriers to PD, as referenced in previous work. 12 The interdisciplinary team comprised nephrologists, modality nurses, PD nurses, and social workers, who collectively assessed each patient's eligibility for PD. A list of contraindications and barriers to PD can be seen in Supplementary Table S1. Data entered via a web form by the teams underwent review by investigators (MO, RQ) to ensure completion and consistency in decision-making. Patients were followed until a clear decision for PD eligibility was made. All eligible patients received modality education and were PD offered. This process was documented in the Dialysis Measurement Analysis and Reporting (DMAR) system.
Statistical analysis
Missing values were handled using the MICE (Multivariate Imputation via Chained Equations) package,
13
which involved generating 20 imputed datasets, each subjected to 100 iterations. The Shapiro–Wilk test and visual inspection of histograms were conducted to assess the normality of continuous variables. All continuous variables were found to be non-normally distributed. We tested various data transformation techniques, including log transformation, but these did not significantly improve the normality or model performance. Consequently, we reported the median and interquartile ranges (IQR, 25th–75th percentiles) for skewed continuous variables. For categorical data, numbers and percentages were presented. To compare baseline characteristics between eligible and non-eligible patients, we used non-parametric tests, specifically the Mann–Whitney
We modeled PD eligibility using logistic regression. All baseline variables were initially included. We screened variables for multicollinearity. For model development, an initial full model was employed, followed by an automated backward selection process across all 20 imputed datasets. The frequency with which predictors appeared in these iterations was then examined to refine the model. A priori, we determined that two clinical variables should be retained in all models, “started dialysis in the ICU” and “pre-dialysis care ≥4 months,” because of their prior associations with PD use.14–17 The model selection was based on the Akaike information criterion. To evaluate if the final backward elimination model provides a better fit to the data, we performed the likelihood ratio test, comparing the likelihood of the data under the full model against the backward elimination model. We utilized backward stepwise elimination primarily to simplify the model and enhance its practical utility by focusing on the most impactful predictors of PD eligibility.
Receiver operating characteristic (ROC) curves were plotted to evaluate the classification performance by comparing the predicted versus observed probabilities of eligibility for PD. The area under the ROC curves (AUC) was measured for both models. ROC curves were plotted to assess how well each model (full and backward elimination) performs on the classification and prediction of PD eligibility.
To measure calibration, we divided the total samples into 10 decile groups based on the predicted probabilities of being eligible for PD. The mean predicted probability and the proportion of the observed prevalence of being eligible for PD were plotted within each decile.
All statistical analyses were performed using R 4.1.0,
18
open-source software for statistical computing and graphics. All
Results
Descriptive statistics
Among the total number of participants (
Medical eligibility and ineligibility to PD based on laboratory and other secondary results (
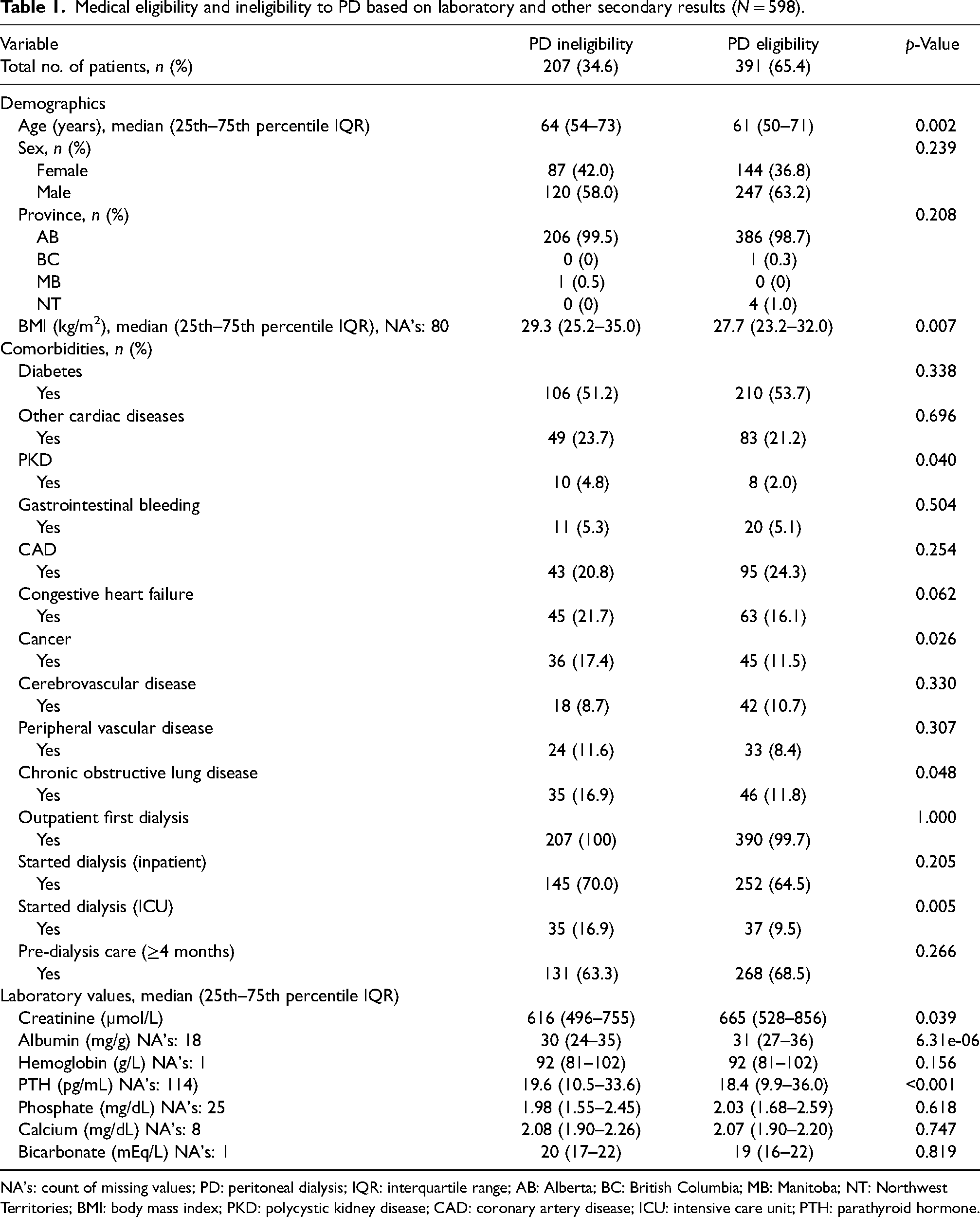
NA's: count of missing values; PD: peritoneal dialysis; IQR: interquartile range; AB: Alberta; BC: British Columbia; MB: Manitoba; NT: Northwest Territories; BMI: body mass index; PKD: polycystic kidney disease; CAD: coronary artery disease; ICU: intensive care unit; PTH: parathyroid hormone.
The results from the Mann–Whitney
Predictors of PD eligibility
The parsimonious backward elimination model revealed several significant predictors associated with PD eligibility. Age (OR = 0.98, 95% CI: 0.97–0.99,
Parameter estimates of backward elimination model.
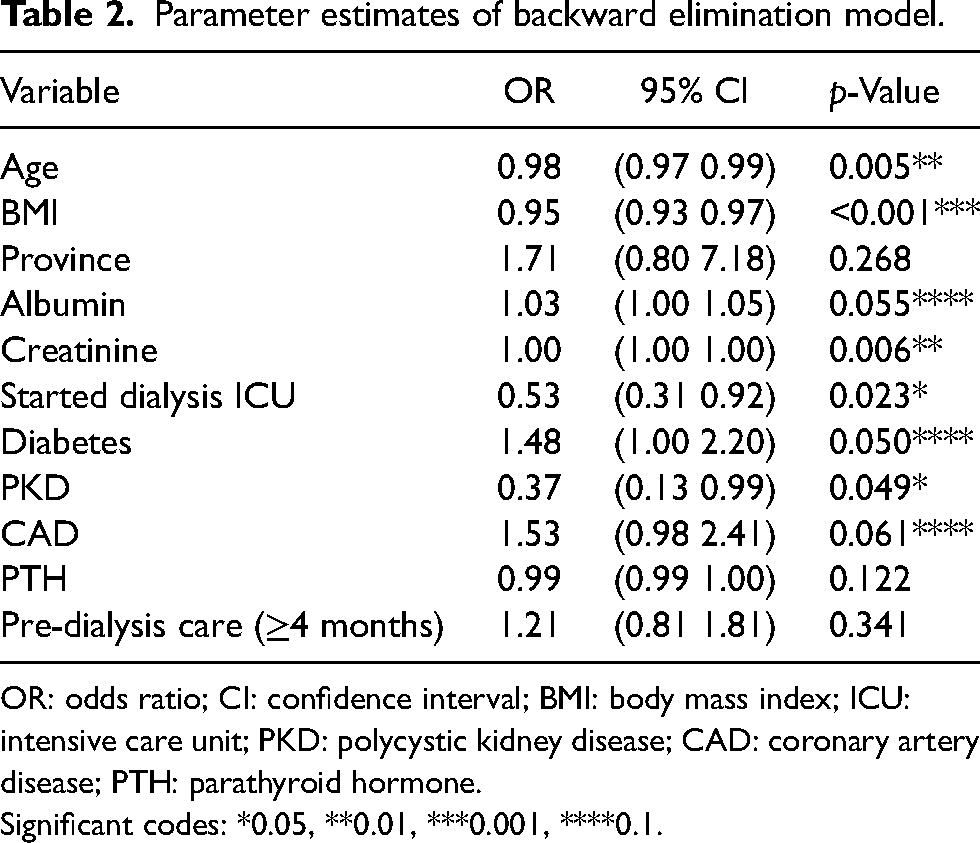
OR: odds ratio; CI: confidence interval; BMI: body mass index; ICU: intensive care unit; PKD: polycystic kidney disease; CAD: coronary artery disease; PTH: parathyroid hormone.
Significant codes: *0.05, **0.01, ***0.001, ****0.1.
Classification performance
In general, the parsimonious backward elimination model achieved adequate performance in discriminating patients eligible for PD. The overall accuracy was 0.68 (95% CI, 0.64–0.72). In this study, we used a 50% threshold to determine PD eligibility based on predicted probabilities from the logistic model. Patients with a predicted probability greater than 50% were classified as PD eligible. A 2 × 2 table was constructed below comparing model predictions with panel-determined eligibility, allowing calculation of sensitivity and specificity. We calculated sensitivity as the percentage of patients the model correctly identified as PD eligible, matching the panel's assessment. This resulted in a high sensitivity of 0.91 (355 out of 391). Specificity was determined by the percentage of patients the model accurately classified as not PD eligible, aligning with the panel's decision. The specificity was 0.26 (54 out of 207).
The
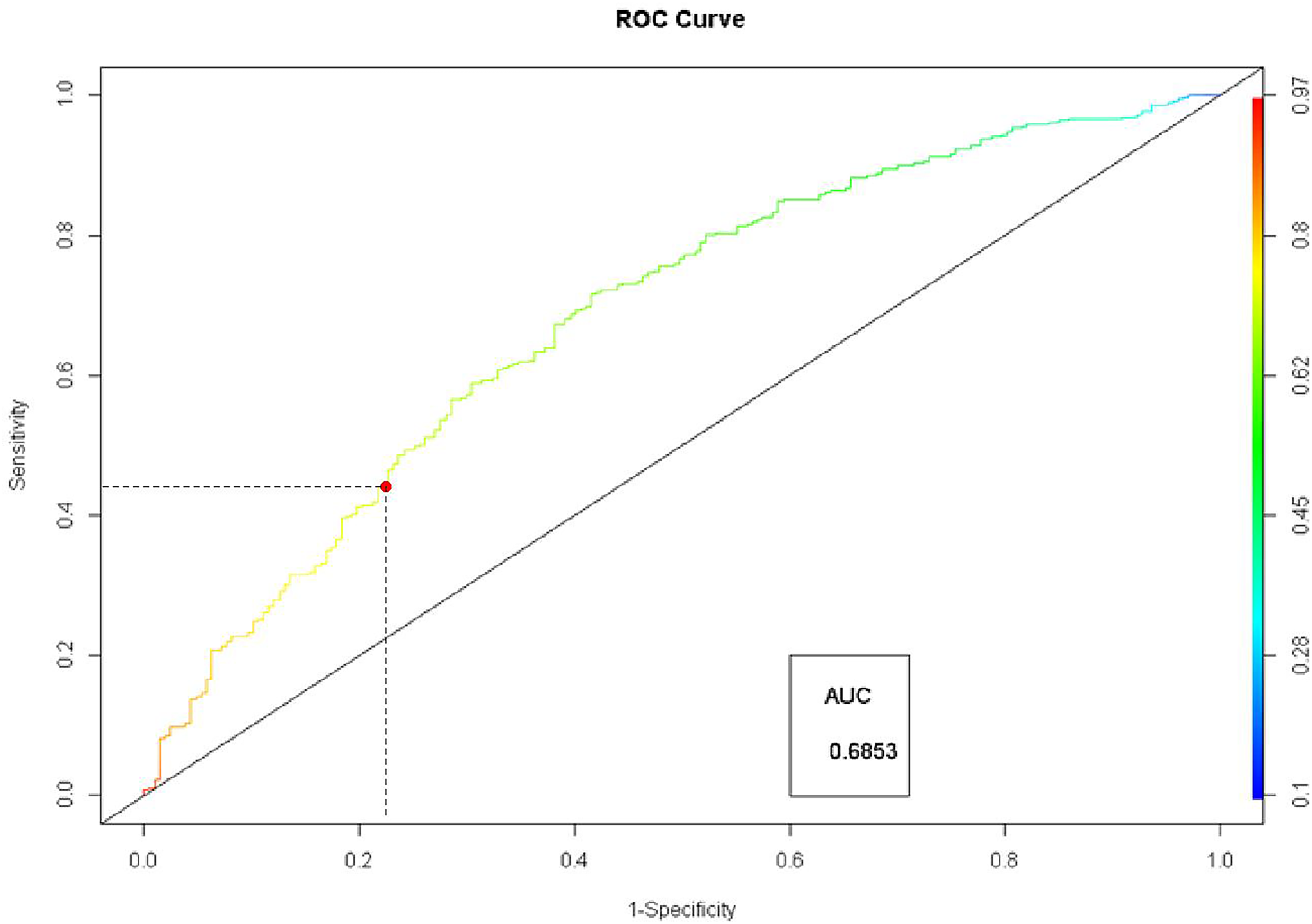
ROC curve for backward elimination model.
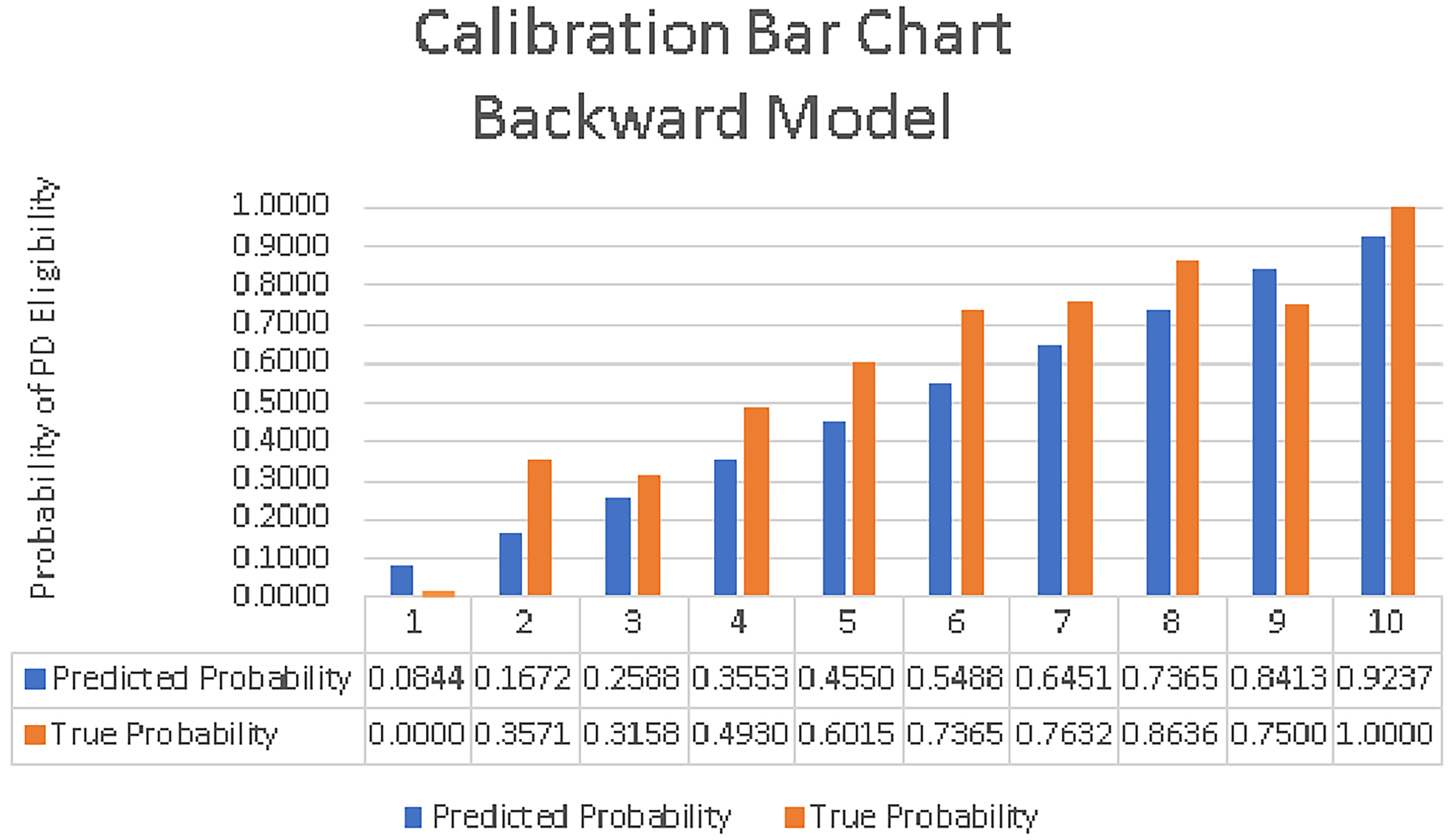
Calibration bar chart for backward elimination model.
Performance matrices of full model and backward elimination model.
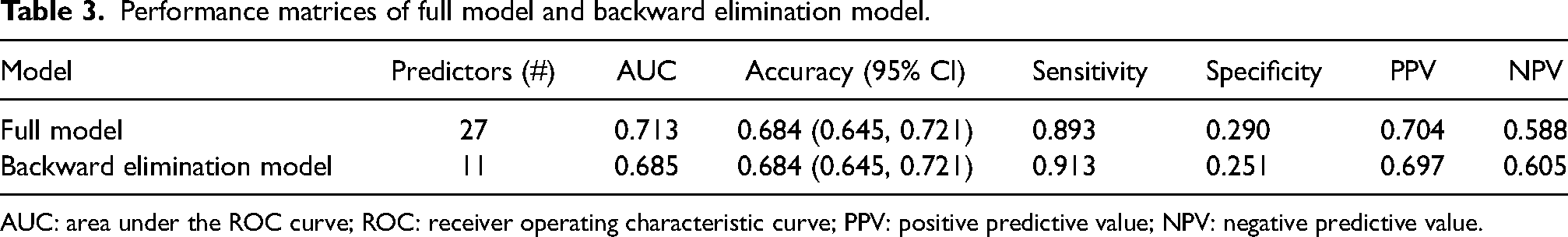
AUC: area under the ROC curve; ROC: receiver operating characteristic curve; PPV: positive predictive value; NPV: negative predictive value.
Discussion
In this study, we utilized logistic regression modeling techniques to create a screening tool that evaluates a patient's eligibility for PD treatment based on information commonly available at the initiation of HD. This tool offers an objective approach to prioritize patients for PD modality assessment. Our results unveiled that certain factors, namely older age, higher BMI, starting dialysis in an ICU, and the presence of polycystic kidney disease, significantly reduce PD eligibility. In contrast, patients with diabetes, coronary artery disease, and higher levels of creatinine or albumin are more likely to be eligible for PD treatment.
Our findings are consistent with established evidence, confirming the significant influence of various factors on PD eligibility. Previous research has identified medical contraindications to PD, including factors like previous major abdominal surgeries, 19 morbid obesity, 20 and special conditions such as polycystic kidney disease21,22 all of which can pose risks when considering PD as a treatment option. 9 Furthermore, the literature extensively reports that older patients and those with multiple comorbidities typically exhibit reduced PD eligibility.9,23,24 This reduced eligibility is often linked to the increased presence of contraindications or barriers to self-care in these individuals.
It's crucial to recognize that PD eligibility criteria are not universally applicable but rather depend on individual patient circumstances and evolving medical practices. Our findings emphasize the impact of various factors, including age, polycystic kidney disease, BMI, and the initiation of dialysis in an ICU, which aligns with established literature.9,20–24 This underscores the necessity for a comprehensive assessment when evaluating PD as a treatment option for ESKD patients.
In contrast, certain medical conditions, such as diabetes and heart failure, may benefit from PD. Cotovio et al. highlighted the advantages of PD for diabetic patients with kidney failure, including home therapy, preserved vascular access, and residual renal function. 25 For these patients, predialysis intervention, patient education, and multidisciplinary support are recommended to enhance diabetic care during PD. Additionally, PD is well tolerated in patients with heart failure due to its slow fluid removal rate. Studies have shown that initiating PD in patients with cardiorenal syndrome type 2 leads to reduced hospitalization rates, improved functional status, and an overall improvement in the quality of life.26,27 It is imperative to distinguish between barriers and contraindications. Unlike barriers, which may be amenable to interventions and support, contraindications do not permit PD regardless of support. While our model is geared towards identifying patients more likely to be eligible for PD, it doesn't explicitly address contraindications. Addressing these contraindications requires comprehensive evaluations integrating medical expertise and multidisciplinary collaboration to optimize dialysis modality selection and enhance patient outcomes. In this study, we provided a standardized list of contraindications, yet the interdisciplinary teams had the final say on whether a condition warranted PD contraindication. The present research did not interfere with the team's decision.
Our study presents a modeling approach for predicting PD eligibility using variables available at the initiation of HD, establishing an objective screening tool. While we acknowledge the potential for further refinement by eliminating additional variables that may not substantially contribute to the predictive value, we stopped at the resulting model to maintain a balance between simplicity and predictive accuracy. While the model's overall accuracy and AUC aren't superior, it demonstrates high sensitivity (0.91) and relatively low specificity (0.26). Our predictive model serves as a valuable tool for healthcare practitioners, streamlining and enhancing the prioritization of potential PD beneficiaries. While the model may flag some false positives, it excels in capturing those who could benefit from PD. In practice, this means that when the model suggests a patient as potentially eligible for PD, further clinical assessment should be conducted to confirm their suitability. This includes more in-depth evaluations, discussions with patients and their families, and medical examinations. By utilizing the model as an initial screening tool, healthcare providers can efficiently identify a pool of potential PD candidates, enabling them to focus their resources and attention on these individuals. This process streamlines the patient prioritization process, ensuring that those who might benefit from PD aren't missed. Ultimately, the model serves as a crucial first step in clinical decision-making, guiding healthcare practitioners to prioritize candidates efficiently and effectively, thus improving the overall process of modality selection for patients in need of urgent HD.
The model does not specifically identify patients who have started PD successfully. Instead, the model was developed to identify patients who could be offered PD based on their characteristics at the initiation of HD. The model's ability to identify individuals who may have a high likelihood of being eligible for PD, thus potentially benefiting from further evaluation and potential consideration for PD as a viable treatment option. However, 22.3% of patients deemed eligible for PD transitioned which is a meaningful rate of PD use among HD starts. 28
Our study represents a promising initial step toward developing PD eligibility screening tools, supporting a systematic and objective approach for patient prioritization. By utilizing baseline variables at HD initiation, this model simplifies decision-making and improves the efficiency of modality education for individuals requiring urgent HD. This study marks an initial but significant step towards the development of more refined predictive models for predicting PD eligibility. These forthcoming models will continue to enhance the screening process, ultimately contributing to more effective and personalized healthcare decision-making in the context of renal replacement therapy. The focus of this tool was on patients who have initiated HD, with the goal of identifying those who may transition to PD. In the future, we aim to develop tools for application in the predialysis setting.
Our study has certain limitations. Firstly, one notable omission is the absence of socioeconomic factors such as income, marital status, or size of residence, which can play a pivotal role in PD eligibility. Some social factors can be regarded as contraindications to PD, for example, residing in a facility that doesn't permit PD. The availability of support, such as spousal support, can significantly influence PD eligibility.12,29 Future models should incorporate these factors, enriching the predictive performance. Secondly, our analysis solely focused on patients at the onset of HD, omitting the prediction of PD eligibility in the predialysis phase. This is particularly relevant, as most patients transitioning to PD commence in the predialysis phase rather than starting on HD. Subsequent studies should also contemplate additional outcomes, such as patients' preferences for PD modality and the actual receipt of PD treatment. In our study, while we included patients after their initial dialysis session, we understand the necessity of incorporating data collection before dialysis initiation to optimize patient preparation and decision-making process. Assessing the duration of predialysis care becomes feasible only when the tool is applied post the commencement of dialysis. For future endeavors, the tool could be deployed during the predialysis phase, necessitating a modification in the assessment of predialysis care duration. Thirdly, while efforts were made to minimize subjectivity, PD eligibility assessment inherently involves some level of it. Despite following standardized procedures and utilizing interdisciplinary assessments, subjective elements may persist in the decision-making process. Despite these limitations, the process aimed to reduce subjectivity and align with real-world clinical practice. Lastly, separating model development and internal validation on distinct datasets is optimal for avoiding overfitting and evaluating generalizability. However, due to the retrospective data and sample size constraints, external validation methods were not included. We acknowledge this limitation and stress the need for future studies to validate externally, considering techniques like cross-validation or bootstrapping for robustness.
In conclusion, this study introduces a screening tool that assesses a patient's eligibility for PD treatment using objective variables available at the initiation of HD. This tool may facilitate identifying eligible patients early and prioritizing them for PD assessment. By doing so, we empower healthcare professionals to employ these models as objective screening tools, thereby standardizing patient assessment for PD treatment. This study marks a significant step in the ongoing development of systematic approaches for PD selection.
Supplemental Material
sj-docx-1-ptd-10.1177_08968608251317463 - Supplemental material for Predictive models on patients’ eligibility for peritoneal dialysis
Supplemental material, sj-docx-1-ptd-10.1177_08968608251317463 for Predictive models on patients’ eligibility for peritoneal dialysis by Yang Yang, Helen H. Chen, Robert R. Quinn, Joel A. Dubin and Matthew J. Oliver in Peritoneal Dialysis International
Supplemental Material
sj-docx-2-ptd-10.1177_08968608251317463 - Supplemental material for Predictive models on patients’ eligibility for peritoneal dialysis
Supplemental material, sj-docx-2-ptd-10.1177_08968608251317463 for Predictive models on patients’ eligibility for peritoneal dialysis by Yang Yang, Helen H. Chen, Robert R. Quinn, Joel A. Dubin and Matthew J. Oliver in Peritoneal Dialysis International
Footnotes
Acknowledgements
The authors would like to thank Kam Shamar for helping with the literature search.
Author contributions
YY, HHC, RRQ, and MJO conceived the study and were involved in protocol development. YY researched the literature, performed the data analysis, and wrote the first draft of the manuscript. RRQ and MJO were involved in gaining ethical approval and patient recruitment. All authors were involved in the data analysis. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The study was approved by the local ethical committee and performed according to the principles of the Declaration of Helsinki.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: YY received the Mitacs Accelerate Fellowship for supporting this research.
Informed consent to participate
Written informed consent was obtained from all subjects before the study.
Informed consent to publish
None.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
