Abstract
There is growing emphasis on increasing utilization of peritoneal dialysis (PD) in patients with end stage kidney disease (ESKD); however, use in patients with severe obesity has still been fraught for various reasons. We aim to assess the viability of PD in patients with severe obesity (BMI > 40 Kg/m2). We conducted a retrospective chart review of patients admitted at the home dialysis center of an academic center between 2014 and 2020 (n = 99). Patients with a BMI>40 kg/m2 at the time of PD initiation (n = 9) were selected. We extracted and examined the data for these nine patients till March 2023. The mean age at baseline was 47.8 ± 12.6 year, 56% were males, 67% were Hispanic, 33% were white, and mean BMI was 43.3 ± 3.4 kg/m2. By the end of the follow-up of this report, five (56%) patients were still active on PD (mean duration 27.8 ± 4.5 months). PD therapy was terminated in 3 (33%) patients (17.7 ± 6.8 months) due to refractory peritonitis, burnout, transfer to rehabilitation facility, respectively. One patient transferred out to another facility after 10.6 months. Rates of access and mechanical complications as well as peritonitis in these nine patients were similar to center's overall rates. All the patients had elimination of uremic symptoms using incremental prescription and met weekly Kt/V targets of >1.7 using adjusted weight. Overall, patients’ weight and glycemic control remained stable. In conclusion, PD can be an effective long-term high-quality dialysis option for patients with ESKD and severe obesity. Further studies in a larger population are required to confirm our findings.
Introduction
Like the trend in the general population, rates of obesity have increased in the end stage kidney disease (ESKD) patients. 1 Previously, obesity was viewed as a relative contraindication for peritoneal dialysis (PD) due to concerns related to poor healing post-catheter placement, mechanical complications (leak, abdominal hernia), decreased solute clearance, increased risk of PD-related infections, and dialysate-related metabolic effects such as weight gain and poor glycemic control.2–4 More recent studies have shown successful performance of PD in those with a body mass index (BMI) of 30–35 kg/m;2,5,6 however, data in patients with severe obesity (BMI > 40 kg/m2) is limited. We aim to assess the viability of PD as a long-term therapy option in patients with severe obesity.
Methods
Study design and population
In a retrospective cohort design, we reviewed the charts of patients who initiated PD between 2014 and 2020 at a free-standing adult home dialysis facility affiliated with a large academic hospital system (n = 99). For this analysis, we identified patients who had a BMI > 40 kg/m2 at the time of PD initiation. Nine patients (10%) met the selection criteria whose data was examined till March 2023. The study was approved by the Institutional Review Board with an exempt status.
We assessed demographic, clinical and laboratory parameters and information of PD prescriptions at the baseline and during follow-up period. The operator and the technique used to place the PD catheter were recorded. At our center, PD catheters are placed percutaneously unless patients have history of hernia or prior refractory peritonitis or extensive abdominal surgery, in those cases, catheters are placed laparoscopically along with as needed interventions. We follow the best practices for PD catheter placement.
Outcomes
The main outcomes were duration on PD and reasons for PD termination. Secondarily, we evaluated mechanical complications; short-term including wound healing, peri-catheter leak, and periprocedural infections; and long-term including slow drains, leak, migration, and hernia. We assessed the sufficiency of dialysis clinically by reviewing monthly provider's notes for symptoms, and by noting total small solute clearance (weekly renal and peritoneal Kt/Vurea) at 6-month intervals. We used adjusted body weight (AdjBW) to calculate the volume of distribution (Vd). AdjBW was calculated as Ideal body weight (IBW) + (0.4*[Actual weight - IBW]). 7 Lastly, we gathered information about PD-related infections. The peritonitis rate was calculated by dividing the total number of peritonitis episodes over the aggregate duration of time (in years) in these nine patients.
Results
Baseline characteristics
The mean age of nine patients was 47.8 ± 12.6 year, 56% were males, 67% were Hispanic, 33% were white, mean BMI was 43.3 ± 3.4 kg/m2 and mean body weight was 130.2 ± 22 kg at the time of PD initiation. The cause for ESKD was diabetic nephropathy in 8 (89%) and IgA nephropathy in 1 (11%) patient. Two patients transferred from in-center hemodialysis (HD) and the rest were new start. The PD catheter was placed laparoscopically by general surgeons in 2 (22%) and a vascular surgeon in 1 (11%), percutaneously using ultrasound or fluoroscopic guidance by interventional radiologists in 4 (44%) and an interventional nephrologist in 2 (22%) patients. The indication for laparoscopy was the history of abdominal surgery in one patient and logistic issues in the other two patients without any pertinent medical indication. No additional adaptive approaches like pre-sternal location or catheter extenders were used in these patients to allow an easily visible exit site location. Figure 1 demonstrates the exit site location in two separate patients. Information regarding length and type of tip of the catheters could not be ascertained for every patient; however, all our operators usually stock 62-cm length catheter with coiled tip. No deviation in the catheter design was noted during the chart review. The mean duration between catheter placement and initiation of PD training was 7.4 ± 11.4 days.
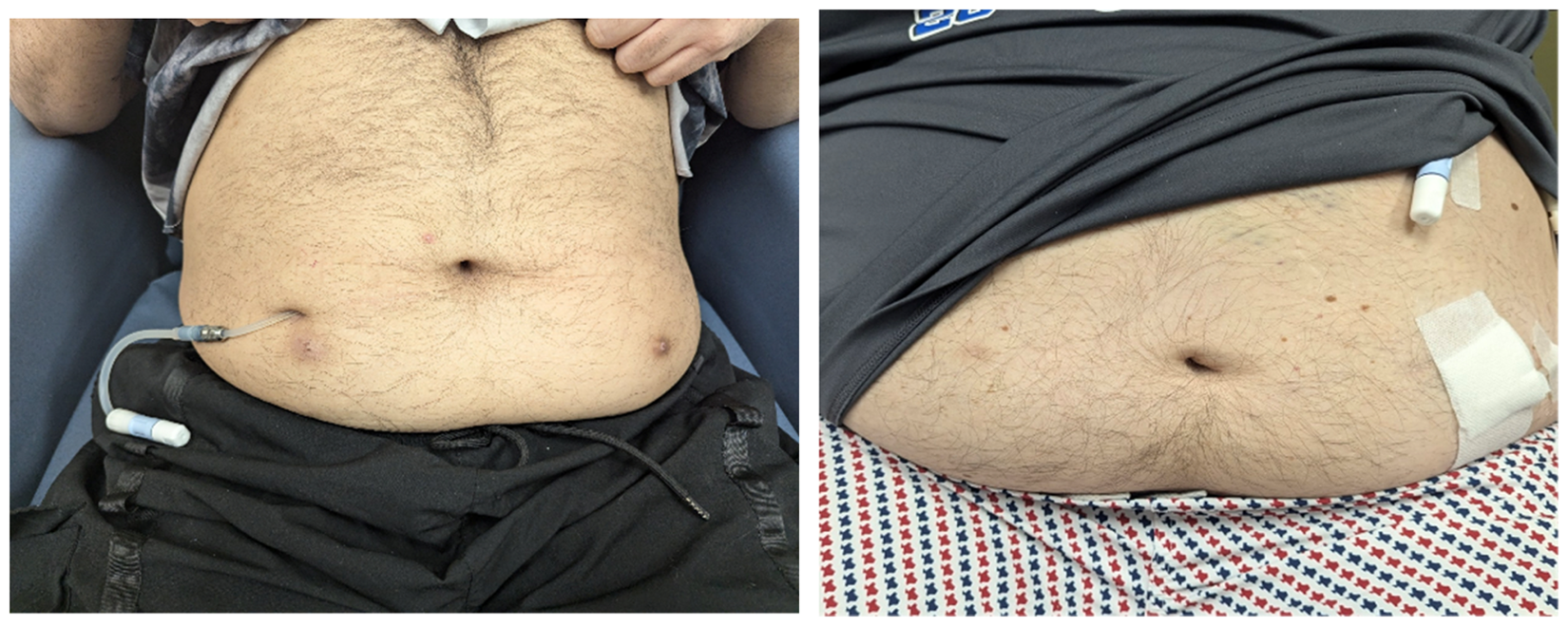
Location of exit site in two separate patients with severe obesity. No catheter extender was used.
Time on PD
By the end of the follow-up, five (56%) patients were still on PD with a mean duration of 27.8 ± 4.5 months. PD therapy was terminated in 3 (33%) patients after a mean duration of 17.7 ± 6.8 months (Figure 2). Overall, duration on PD for these nine patients was 23.4 ± 7.5 months or 1.94 ± 0.6 years. The causes for termination were refractory peritonitis, burnout, transfer to rehabilitation facility after a prolonged hospitalization in three patients, respectively. One patient transferred out to another facility after 10.6 months with no follow-up available.
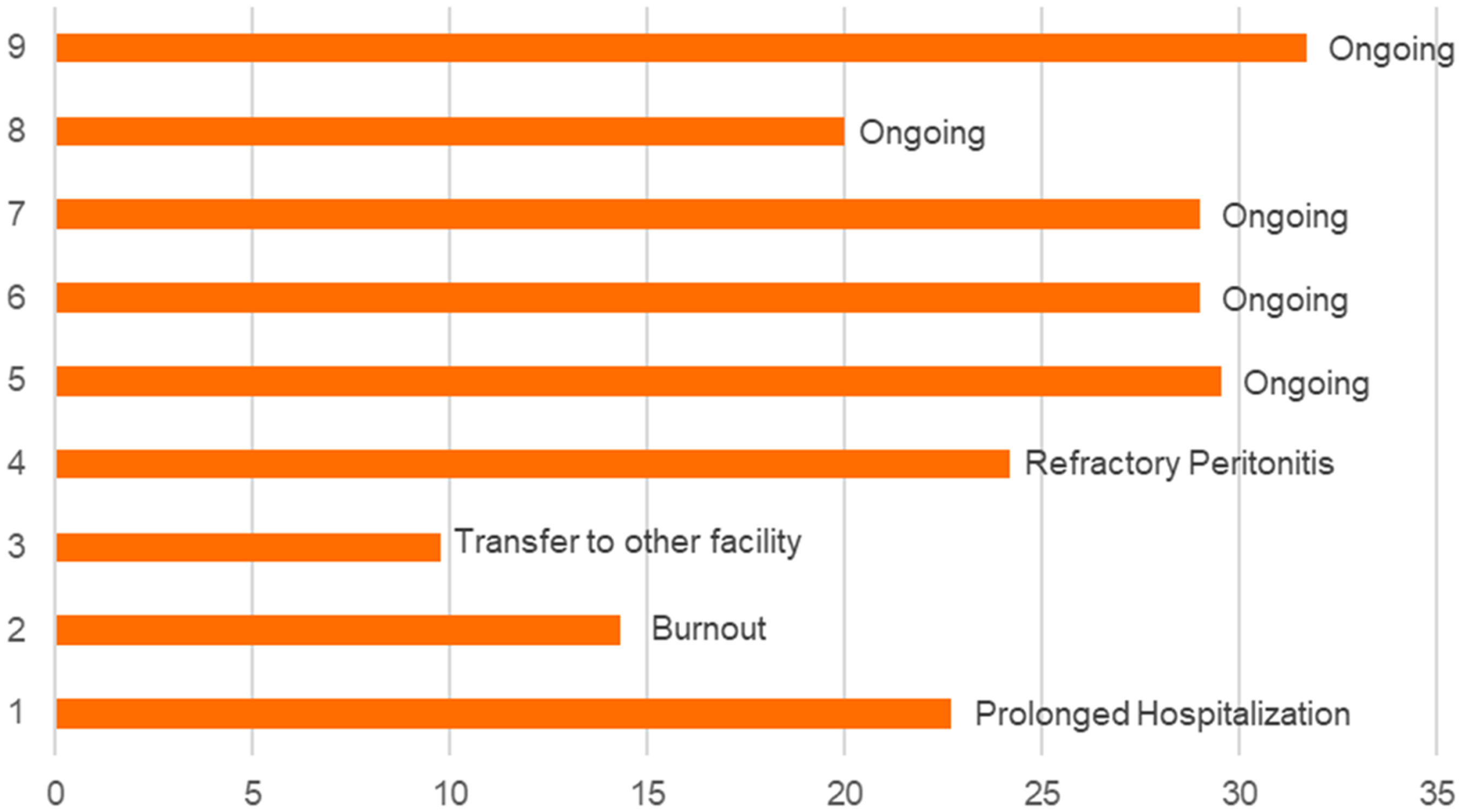
Duration on pd therapy and causes of termination.
Early- and long-term outcomes
None of the nine patients had delayed wound healing or peri-catheter leak after PD catheter placement. One patient was found to have omental wrapping at the beginning of treatment which was repaired surgically with no further complications. This patient had the initial catheter placed by the surgeon. All the nine patients with BMI > 40 kg/m2 successfully begin PD without any futile catheter placement. During a later period of follow-up, three patients encountered slow drains related to constipation and one patient's PD catheter migrated high in the pelvis with high stool burden requiring repositioning by an interventional nephrologist 17 months into treatment. None of the patients had hernia development.
Dialysis prescription and achieved KT/V
All the nine patients chose cyclic PD as the modality. Eight patients had residual kidney function (RKF) at the start of PD, and seven of these patients maintained the kidney function by the end of follow-up. The initial prescription utilized an average of 4.2 ± 0.6 cycles over 8.7 ± 0.6 h a night, a last fill of 0.8 ± 0.9 lt (N = 6 of 9), total volume 10.01 ± 2.0 lt. One patient with no RKF required a mid-day exchange. Table 1 demonstrates the trajectory of incremental PD prescription over time. Every patient attained solute clearance (both clinically and current CMS targets of weekly Kt/Vurea > 1.7) (Table 1).
Peritoneal dialysis prescription and adequacy over follow-up time.
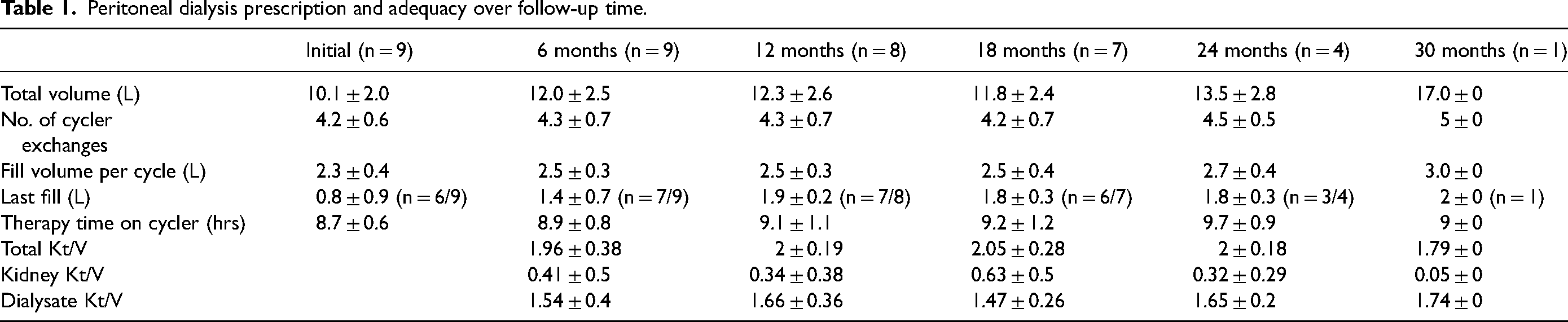
Metabolic and infectious outcomes
Overall, six patients gained weight (6.1 ± 3.5 kg) and three patients had intentional weight loss (15 ± 12.8 kg) without glucagon-like-peptide-1 agonist therapy. The mean hemoglobin A1C was 7.8 ± 2.3% at baseline and 7.1 ± 1.3% at the end of follow-up. A total of six episodes of peritonitis occurred in five patients, a peritonitis rate of 0.3 episodes/pt-year for these nine patients. One patient required PD catheter removal for refractory peritonitis, the rest of episodes were treated successfully using intraperitoneal antibiotic/s as outpatient. Two patients sustained exit site infection, one episode in each and both had successful outcomes with the administration of oral antibiotics. No tunnel infection was observed.
Discussion
We report our experience of successful PD therapy in nine patients with severe obesity. Four patients terminated PD therapy after a mean of about 18 months; however, for reasons unrelated to severe obesity. The rest of patients were active on PD by the end of this study with an average vintage of 27 months. We did not observe any occurrence of short- or long-term complications above ordinary.
There has been growing emphasis on increasing utilization of home dialysis as the modality of choice for ESKD treatment.8,9 However, there have been reservations against the use of PD in patients with obesity.2–4 Although the evidence is varied across the literature, more recent studies have shown no difference in survival or hospitalization in patients with a BMI > 30 kg/m2 undergoing PD therapy when compared to patients with similar BMI on HD.6,10 Similarly, in a recent series of 15 patients with BMI > 35 kg/m2, no difference in dialysate volume, Kt/V or metabolic parameters was observed between obese and lean patients. 11 Now, we report the success and longevity of PD in the patients with BMI > 40 kg/m2, which was comparable to what has been reported for overall PD population in contemporary data (2 ± 3.2 years). 12 This PD duration can be considered better given that five of nine patients were still active on PD therapy by the of end of this study.
One of the major concerns for PD in patients with obesity has been failure to meet small solute clearance targets. Although, the focus has moved away from an over emphasis on achieving small solute clearance targets to a more patient-centered approach, Kt/V is one of the few objective measures which assess the amount of delivered dialysis and remains a quality-metric for dialysis units. In patients with obesity, use of actual weight can result in a very high Vd for urea resulting in low Kt/V despite patients clinically doing well. In this calculation, it is assumed that the metabolic activity and water distribution of excess weight, mostly consisting of adipocytes, is similar to the ideal weight. However, this assumption is not true and use of AdjBW has been proposed that accounts for the fact that water distribution in adipocyte is much lower than the other tissues. 7 Using the AdjBW, all the nine patients, attained weekly small solute clearance targets and none required transition to HD due to inadequate solute clearance.
We also report successful PD catheter placement using percutaneous technique in these patients. Six of nine patients had their catheter placed by either an interventional radiologist or a nephrologist and did not encounter any short- or long-term complications related to PD catheter placement above the ordinary. Percutaneously placed PD catheters are shown to have similar outcomes compared to surgically placed catheters. These catheters offer advantages of quicker access, more suitability for urgent start, no need for general anesthesia, etc. and have been used increasingly. 13 However, there are still hesitations about their use in patients with severe obesity. Our results are encouraging for the percutaneously placed PD catheters, which paves a rapid pathway to increase the access to PD.
There was no significant dialysate related metabolic effects such as weight gain or worsening glycemic control. We believe that an incremental PD approach 14 and use of AdjBW to calculate Vd allow the total dialysate volume to be contained in a reasonable limit, increase patients’ acceptance, maintain small solute targets and limit glucose exposure while at the same time provide sufficient dialysis. Furthermore, the peritonitis rates were lower than recommended by International Society of PD guidelines (<0.4 episodes/pt-yr). Importantly, most patients maintained RKF by the end of follow up.
The main limitations of our study are a small sample size, retrospective review and short follow-up period; however, a successful experience in each patient strengthens the impact of this study.
Conclusion
Our results demonstrate that PD can be an effective long-term dialysis option for patients with ESKD and severe obesity (BMI >40Kg/m2), the prevalence of both is increasing with rising rates of obesity worldwide. PD therapy was able to provide a high-quality dialysis and meet small solute clearance targets, without undue burden of dialysate volume, worsening of metabolic parameters, and other complications. We recommend the use of AdjBW to calculate Vd in these patients. Given the small sample size, further studies in a larger population are required to confirm our findings.
Footnotes
Authorship
SB and AN researched literature and conceived the study. AN and AL were involved in protocol development, gaining ethical approval, and data analysis. OY and GD performed data collection and data analysis. AM conducted the literature review and data analysis. AN wrote the first draft of the manuscript and SB revised it critically. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
S. Bansal reports consultancy for Calliditas NA Enterprises Inc., Daxor Corporation, Vera therapeutics, and PD Excellence Academy, a branch of Baxter Healthcare Corporation; research funding from Bayer, Boehringer Ingelheim, NIH, and Novo Nordisk; royalties from UpToDate; speakers bureau and honoraria from Home Dialysis University; advisory or leadership role on the Editorial Boards of CJASN and Kidney360, and Section Editor for Clinical Nephrology Journal.
Ethical approval
Ethical approval for this study was obtained from University of Texas Health San Antonio Institutional Review Board with an exempt status. IRB# 2022-0431E.
Funding
Research reported in this publication was supported by the National Center for Advancing Translational Sciences of the National Institutes of Health through Grant UM1 TR004538 to S. Bansal. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
National Center for Advancing Translational Sciences of the National Institutes of Health, (grant number UM1 TR004538).
New affiliations
Akwe Nyabera: Division of Nephrology, Department of Medicine, Columbia University Irving Medical Center, New York, NY, USA.
Omar Ayah: Department of Transplant Nephrology, Houston Methodist Hospital, Houston, TX, USA.
Gabriela Dande: Division of Nephrology and Hypertension. University of North Carolina School of Medicine, Chapel Hill, NC, USA.
Alexis Lorio: Department of Neurology, Duke University School of Medicine, Durham, NC, USA.
