Abstract
Background
Technique survival, also reported with negative connotations as technique failure or transfer from peritoneal dialysis to haemodialysis, has been identified by patients, caregivers and health professionals as a critically important outcome to be reported in all trials. However, there is wide variation in how peritoneal dialysis technique survival is defined, measured and reported, leading to difficulty in comparing or consolidating results.
Methods
We conducted an online international consensus workshop to establish a core outcome measure of technique survival. Discussions were analysed thematically.
Results
Fifty-five participants including 14 patients and caregivers from 13 countries took part in facilitated breakout discussions using video-conferencing. The following themes were identified: capturing important aspects of the outcome (requiring a core event to define the outcome, distinguishing temporary from permanent events, recognising heterogeneous experiences of transfers), adopting appropriate neutral nomenclature (conveying with clarity, avoiding negative connotations), and ensuring feasibility and applicability (capturing data relevant to clinical and research settings, ease of adoption). The suggested definitions for the core outcome measure were ‘the event of a transfer to haemodialysis’, or ‘discontinuation of peritoneal dialysis’. Applying the principles described within the workshop, defining the outcome measure as a ‘transfer to haemodialysis’ was preferable.
Conclusions
It is proposed that the core outcome of technique survival is redefined as ‘transfer to haemodialysis’ and that its components are standardised using simple, neutral terminology Components considered important by stakeholders included recording the reasons for transfer from peritoneal dialysis, and focussing on permanent events whilst ensuring the outcome remains easy to implement.
This is a visual representation of the abstract.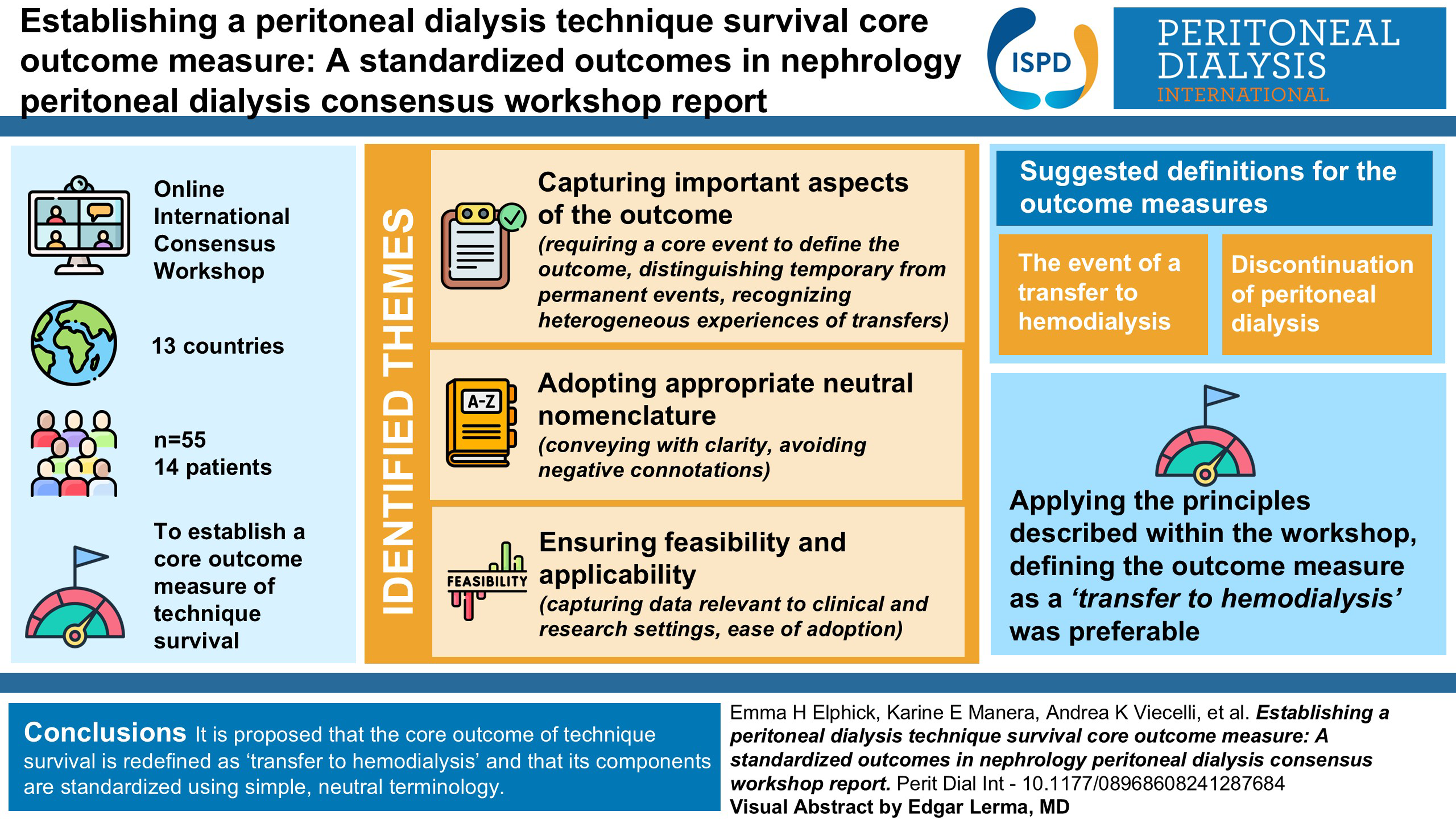
Background
The terms technique failure or technique survival are often used to describe patients who, for a variety of reasons, have to discontinue peritoneal dialysis (PD) and most frequently transfer to haemodialysis (HD), but there is a lack of consistency in current definitions, which may or may not include temporary discontinuations, death or stopping PD to palliate. Nevertheless this is a large problem for patients and carers, illustrated both by the high rate of this outcome,1–6 and by the widespread reporting of rates of transfer to HD by national registries. Transferring a patient from their preferred modality has adverse impacts upon healthcare expenditure, 7 mortality, 8 morbidity and quality of life, with patients frequently reporting negative experiences in the process. 9 Furthermore, there is significant variability between centres both nationally and internationally in rates of transfer to HD.4,10 This is an example of unwarranted variation, strongly indicating although not definitively proving that differences in centre-level practices are directly leading to worse outcomes for patients and their caregivers, with scope for improvement.
Despite the importance of technique survival, there has been no attempt to standardise the definition of this outcome internationally. This has resulted in registries reporting the outcome according to different or unclear definitions, preventing meaningful international comparisons, although the Peritoneal Dialysis Outcomes and Practice Patterns Study has now demonstrated between-country variability in rates of transfer to HD with reasonable standardization of criteria. 2 Lack of standardisation is also an issue in trials, with a systematic review of all PD trials identifying a highly heterogeneous approach in measuring and reporting this outcome, including the use of different terms (technique failure, technique survival and transfer to HD), variable inclusion/exclusion of death, and variation in the minimum duration required before transfer to HD is considered permanent. 11
The Standardised Outcome in Nephrology – Peritoneal Dialysis (SONG-PD) group, representing over 900 patients, family members and health professionals from 68 countries, identified technique survival to be a critically important outcome that must be reported in all future trials. 12 To reduce the variability in the recording and reporting of this core outcome, trials need a standardised definition and a stakeholder workshop to establish the essential features was convened. This workshop report summarises the perspectives of the patients, caregivers, and healthcare professionals on defining a core outcome measure for technique survival to be reported in all future trials involving PD patients.
SONG PD technique survival workshop
Context and scope
The SONG PD technique survival consensus workshop was held online on 15 September 2020. Patients, caregivers, healthcare professionals, researchers, policy makers and epidemiologists were invited to participate. This was held over Zoom, a modern video communication platform, to allow for global participation from the PD community.
Attendees and contributors
55 participants from 13 countries including the UK (9), Australia (17), France (3), Lebanon (1), Canada (5), South Korea (1), USA (6), Brazil (2), Singapore (3), Poland (1), Hong Kong (3), South Africa (1) and Belgium (1) attended the workshop. Thirteen participants were patients, and one was a caregiver, all with experience of PD. Other participants included paediatric and adult nephrologists, researchers and renal nurses. Healthcare professionals were invited if they had an interest in PD and/or PD expertise or leadership roles in relevant professional societies (including the International Society for Peritoneal Dialysis [ISPD]), regulatory agencies, funding organisations, patient charities (Kidney Care UK) and registries (Australia and New Zealand Dialysis and Transplant Registry [ANZDATA], UK Renal Registry). Patients and caregivers were invited by workshop conveners.
Workshop overview
Prior to the workshop, background materials were sent out to all participants. First, we provided a brief overview of the SONG-PD initiative with preliminary results of a systematic review on the outcome measures for technique survival. Following this, participants were allocated into break-out rooms for 35 min to discuss the core outcome measure. Each group consisted of eight to 10 participants, which included professionals and patients/caregivers. A facilitator in each group followed a standard run sheet including questions to bring to the group.
Each group was asked to discuss the pro and cons of the terms ‘technique survival/failure’ and ‘transfer to haemodialysis’. They were then asked to focus on what is the best/most meaningful definition for the core outcome measure, what aspects are important to consider for the definition, and what duration of haemodialysis would be regarded as a ‘transfer or technique failure’. Following the break-out sessions, a representative from each group was allocated to feedback to the large consensus workshop group. This involved a brief summary of the discussion from every group. The session was concluded with a final wrap-up from the chair (ML).
All breakout discussions were recorded and transcribed verbatim. Transcripts were entered into HyperRESEARCH (ResearchWare Inc, Version 3.0) to facilitate coding and analysis of the data. Investigators EE and ML read the de-identified transcripts line-by-line and inductively identified and coded concepts into themes reflecting participants’ perspectives and suggestions regarding the development and implementation of a core outcome measure for technique survival. Preliminary themes were discussed and revised with YC, AJ and KM who independently read the transcripts. The agreed findings were then sent to all workshop attendees for review and feedback to ensure that it reflected the full range of perspectives and additional comments integrated into the final report.
Summary of the workshop discussion
We identified three main themes that encompassed the discussions across the breakout groups: (1) capturing important aspects of the outcome; (2) adopting appropriate, neutral nomenclature; and, 3) ensuring feasibility and applicability.
Capturing important aspects of the outcome
Recognising heterogeneous experiences of technique survival
Requiring a core event to define the outcome. Participants agreed that technique survival should be defined clearly by an event. Some suggested that technique survival should be defined as stopping PD to be able to identify all problems leading to discontinuation of PD, irrespective of whether patients then transferred to HD. It was uncertain whether kidney recovery, withdrawal of dialysis, temporary stoppages, death either due to complications of PD or unrelated deaths, or kidney transplantation should be included and how it should be reported. A ‘global’ outcome measure of stopping PD, with specific events recorded was suggested.
Some asserted that transferring from PD to HD was the key event that should define the core outcome measure. Other events (e.g. kidney recovery, death) were assumed to be recorded as separate outcomes. Transfer to HD was regarded as a clear and simple definition that would remove causes that might cause confusion, such as stopping PD as part of end-of-life care, that was easily interpretable by healthcare professionals and patients and could be easily captured in trials and registries.
Distinguishing temporary from permanent events. The reasons for transferring to HD or stopping PD were considered by participants in relation to the permanency of the event. An intended permanent change (e.g. patient choice relating to burnout) was viewed as having different implications compared with an unintentional temporary change (e.g. acute severe peritonitis), but it was noted that the intention could be difficult to collect accurately. The duration of the transfer to HD (i.e. time off PD) was regarded to be the optimum method to determine whether an event was ‘permanent’. However, it was recognised that the duration of qualifying permanence would vary between patients, centres and countries requiring an individualised approach.
Healthcare professionals and patients agreed that different time frames would provide data on different patient populations. Shorter timeframes, such as 30 days, would likely include more patients with temporary problems, for example post peritonitis. Longer time frames (e.g. 180 days) would generally identify problems that required permanent cessation of PD. Two patients/caregivers commented that even a very short period on HD was relevant and meaningful, and some healthcare professionals felt that identifying short periods on HD/off PD would help to identify problems as part of a governance process. However, others viewed the permanent transfers as more relevant, thereby justifying a longer period to define a transfer to HD. ‘The definition doesn’t really interpret the groups that we really want to focus on like the group where they permanently fail to go back on PD’. (Caregiver)
This included a suggestion by a healthcare professional that in some situations a short period of PD, such as for a hernia repair, should not be viewed as a problem, but as part of the therapy. There was a concern that shorter durations would reflect differences in the speed of catheter re-insertion for temporary transfers, and also inflate the reported rate, potentially discouraging prospective PD patients due to concerns about permanent transfers to HD. The main concern about longer durations on HD/off PD is related to problems with trials (see below). Specific durations discussed were 30, 90 and 180 days, and where a preference was expressed it was for 30 or 90 days
Adopting appropriate, neutral nomenclature
Conveying with clarity. With regards to potentially adopting the definition of technique survival as ‘transfer to HD’, the alternative terms to ‘transfer’ were ‘switch’ or ‘change’. For stopping PD, suggestions for nomenclature were more variable, including ‘cessation of PD’, ‘termination of PD’ and ‘discontinuation of PD’.
The use of the word ‘technique’ (for technique failure) was considered to be ambiguous as it was not clear if death, transplantation, recovery of kidney function, and dialysis withdrawal were captured in this term. Patients and health care professionals felt it was important to report the death, which could be viewed as a failure of the treatment, and the discussion included one proposal to specify death as related to complications of PD or unrelated causes, although how this could be applied in practice was not discussed.
Use of the term ‘technique survival’ was considered but this was thought to imply the lack of an event, introducing a lack of clarity given that defining an outcome measure as an event would be clearer, and easier to record and analyse. Similarly, the word ‘failure’ was potentially inaccurate, in that this did not capture situations where there was not a failure, but a planned integrated care strategy or patient choice.
Avoiding negative connotations. The use of the word ‘failure’ was felt to have negative connotations, with patients expressing the perception that this may be attributing blame to them, and that it was their fault PD had ‘failed’. As one patient highlighted, ‘I know this is for trial purposes and studies, but inevitably that creeps into clinical language as well’. Some patients felt the term ‘failure’ was ‘… putting the blame on us for it failing, whereas it could just be the body doing it … (patient)’. Similarly, when considering stopping PD, ‘termination’ was viewed as having negative connotations that should be avoided. Instead of the term ‘failure’, others wanted more clarity in the definition to describe the event of interest, ‘I … prefer the transfer to hemo[dialysis]. When someone chooses to be transferred to hemo[dialysis], that's not a failure. That's a choice (patient)’.
Ensuring feasibility and applicability
Capturing data relevant to clinical and research settings. As a core outcome for studies, participants suggested to limit the duration of follow-up for feasibility. For example, to define an event requiring 90 days of follow-up could exceed the duration and resources of some trials, ‘Let's say for a one year follow up trial, definition that includes 90 days means that you extend the trial for 30% of the time, and I think that's complicated from a trial logistic perspective (Health Professional)’. This need to minimise the duration of follow-up required was recognised as conflicting with the need to emphasise permanent events ‘30 days in terms of length of time for either technique survival or transfer to hemodialysis should be meaningful because from the patient's point of view, 30 days there's a fair chance that it would be a permanent transfer … … seems to be a bit of a compromise in terms of feasibility as well, not having to wait for example 180 days doing a trial (Health Professional)’.
Ease of adoption. While the core outcome measure of ‘technique survival’ was primarily intended for clinical trials, participants felt that reporting of technique survival by registries was of similar importance. Thus, the definition of technique survival had to be applicable to and feasible to implement in registries. Recording of the reason for transfer to HD was potentially important in the interpretation of international differences in this outcome, particularly when comparing countries with restricted availability of HD. The burden of data collection was cited as the biggest issue when considering adoption by registries, as there could be a need to capture whether a transfer was intended to be temporary or permanent at the point of transfer, reasons for a transfer to HD and all episodes where PD was temporarily stopped.
Discussion
Participants in the consensus workshop for defining a core outcome for ‘technique survival’ in PD were in agreement that it should be defined as a single, clear event, with a majority view that the outcome measure should be defined as the event: ‘transfer to HD’. However, there was a minority view that the definition should be the event of ‘stopping PD’, whether or not the patient had HD subsequently. There was uniform support for changing the nomenclature describing the core outcome measure, avoiding the terms ‘technique’, ‘failure’ and ‘survival’ and instead using simple language reflecting the event defining the outcome measure without any positive or negative connotations. Other principles that should determine aspects of the core outcome were also agreed upon, particularly the competing priorities to be balanced when considering the reason for the event, and how long the change must have been sustained for (Table 1).
Summary table of thematic analysis with illustrative quotes.

P = patient; HP = health professional; C = caregiver; HD = haemodialysis.
The distinction between transfer to HD and stopping PD as the outcome measure was the main point of discussion raised in the workshop, suggesting that this was viewed as the key issue. The event of transferring to HD is unambiguous and clearly related to technique survival, but whether transplantation, dialysis withdrawal, renal recovery and temporary PD stoppages without any form of dialysis should also be included in an outcome measure of technique survival is less clear. Furthermore, the implications of including temporary PD stoppages without any dialysis in the outcome measure are not clear because, although there is considerable published research on transfers to HD, there is little reported on rates of, and relative importance of different causes of, temporary PD stoppages. PD stoppages are an important problem, often reflecting problems with the technique, but many reasons for PD stoppages do not reflect a problem with the technique, making this a suboptimal measure of technique survival. Using the principles identified within the workshop, a comparison of using transfer to HD or stopping PD is shown in Table 2, supporting the majority view within the workshop that the core outcome measure should be defined as transfer to HD. PD stoppages will need to be captured better to holistically address technique issues in PD, but the complexities and details of this were beyond the scope of this workshop.
Comparison of options for core event defining the outcome according to principles identified during the workshop.

HD = haemodialysis; PD = peritoneal dialysis.
Competing arguments were identified for the duration of the event recorded (i.e. time on HD/off PD). Shorter periods would identify more clinical problems thereby potentially improving governance, 13 and they would be easier to implement in clinical trials. Longer periods were felt to be focussing on the more significant permanent events. The duration of time on HD used to define the event of transfer to HD also affects the relative contribution of different reasons.2,14
Capturing more detail around reasons for the event was considered important to interpret the event defining the core outcome, and to better reflect the patient experience, at the expense of increasing the data collection burden, thereby reducing the likelihood of widespread uptake of the core outcome. When the core outcome is being defined, these competing principles will have to be balanced against each other. One option would be to incorporate more detail such as reasons for the event where it is directly relevant to a trial (e.g. where technique survival is a key outcome that is likely influenced by the intervention), and use the simpler measure by recording the event without further detail in all trials.
One of the drivers underpinning the interest in technique survival is seeking to understand differences in the longevity of PD as a therapy, although PDOPPS has now demonstrated that international differences in the duration of PD mostly reflect differences in transplantation, infection and death. 2 It is also now clear that burnout occurs in people undertaking PD, 15 but neither of these results were available at the time of the workshop and might have influenced the discussions had they been. There is no standardisation in when PD is considered to have started, although the International Society for Peritoneal Dialysis (ISPD) have now convened a group to produce a standardised approach to the metric of time on PD. We also note that, despite the wide geographical representation in the group, this cannot be considered a perfect representation of all perspectives.
In conclusion, technique survival should be defined as transfer to HD. Further agreement on the detail required for a working definition was not possible, but the workshop did establish the principles necessary for this (Table 3). The ISPD working group for time on PD will apply these principles to produce a working definition, including appropriate international collaboration to ensure the definition is widely applicable, and relevant to all healthcare settings. Given that routine reporting of PD outcomes for meaningful quality assurance was a widely agreed principle in the workshop, subsequent endorsement by international societies and national registries will be necessary. To ensure that the outcome measure is reported within trials as intended, liaison with funding and research organisations may be necessary to compel researchers to include core outcome measures in future research, and journal editors to encourage routine reporting of the results (already required by some).
Summary of workshop recommendations for establishing a core outcome measure for technique survival.

HD = haemodialysis; PD = peritoneal dialysis.
Footnotes
Acknowledgements
None.
Author contributions
All authors participated in the workshop. EE and ML coded concepts into themes, YC, KM and AJ independently reviewed the themes. EE wrote the first draft, all authors reviewed and contributed to subsequent drafts.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: ML and SD have received speaker's honoraria from Baxter Healthcare and Fresenius Medical Care, and an unrestricted research grant from Baxter Healthcare. JSFC has received speaker's honoraria and research funds from Baxter Healthcare, Fresenius Medical Care, Roche Pharmaceuticals and Amgen. NB has received speaking honoraria from Baxter Healthcare and Otsuka and is on the Advisory Board for Astra Zeneca, Fresenius Medical Care, Vifor, and GSK. DWJ has received consultancy fees, research grants, speaker's honoraria and travel sponsorships from Baxter Healthcare and Fresenius Medical Care, consultancy fees from Astra Zeneca, Bayer, and AWAK, speaker's honoraria from ONO and Boehringer Ingelheim & Lilly, and travel sponsorships from Ono and Amgen. He is a current recipient of an Australian National Health and Medical Research Council (NHMRC) Leadership Investigator Grant. AL has served as a consultant and member of advisory boards for Alnylam Pharmaceuticals, AstraZeneca, Baxter Healthcare, Bayer AG, Boehringer-Ingelheim, Chinook Therapeutics, Dimerix Limited, Eledon Pharmaceuticals, George Clinical, GlaxoSmithKline, Kira Pharmaceuticals, Prokidney, Otsuka Pharmaceuticals and Visterra Inc, Zai Lab Co. Ltd; has received Speaker's honorarium from AstraZeneca, Baxter Healthcare, Boehringer-Ingelheim, Chinook Therapeutics and Otsuka Pharmaceuticals; and has served as a member of Data Safety and Monitoring Committee for Dimerix Limited and Zai Lab Co. Ltd. AKJ has received research and education grants from Baxter Healthcare, Otsuka Pharmaceuticals and serves as an advisory board member for AWAK Technologies. JIS has served as a consultant and member of advisory boards for Healthmap Solutions and Dialco Medical, grants from the Canadian Institutes of Health Research, and speaker's honorarium from Outset Medical. AKV receives grant support from a Queensland Advancing Clinical Research Fellowship and an NHMRC Emerging Leader Grant. MW has received speakers honoraria from Baxter Healthcare and Fresenius Medical Care, consulting fees from Triomed AB, and an unrestricted research grant from Baxter Healthcare. RQ is the co-inventor of the Dialysis Information Management System™, Canada, and is the co-owner of the intellectual property associated with it, and has received honoraria and attended advisory boards for Baxter Healthcare. YC is a current recipient of the Australian NHMRC Emerging Leadership Investigator Grant (2022–2026) and Queensland Advancing Clinical Fellowship (2022–2025; mid-career). She has received honoraria and research grants from Baxter Healthcare and Fresenius Medical Care, and a consultancy fee from Bupa Health Insurance. ACNF has received honoraria for presentations from Baxter Healthcare and a Scholarship from Fond de Recherche du Québec – Santé. NA is supported by an NHMRC Postgraduate Scholarship. SQ is on the board of the Renal Physician's Association (unpaid) and a medical director at the Davita home dialysis unit. PG, KEM, TH, both AJ's, AMG, DP, AYW, JCC and EE have no disclosures.
Ethical approval
Not applicable.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Health and Medical Research Council (grant number 1197324).
Informed consent to participate
Not applicable.
Informed consent to publish
Not applicable.
ORCID iDs
Trial registration
Not applicable.
Appendix
The collaborators who attended the workshop, in addition to the co-authors, were:
Healthcare professionals: Ali Abu-Alfa, Christoph Aufricht, Amanda Baumgart, Clemence Bechade, Edwina Brown, Gillian Brunier, Indranil das Gupta, Marjorie Foo, Eric Goffin, Elyssa Hannan, Htay Htay, Talerngsak Kanjanabuch, Yong Lim Kim, John Knight, Monica Lichodziejewska-Niemierko, Thierry Lobbedez, Sarala Naicker, Roberto Pecoits-Filho, Benedicte Sautenet, CC Szeto and Eric Wallace. Patients and family members: Gary Braddock, Shelley Downs, Derek Forfang, Pamela Griffiths, Danielle Horton, Nichole Jefferson, Sharon Kidwell, Agnes Lau, Nigel Mulvey, Raymond Phang, Lisa Richardson, Nancy Verdin.
