Abstract
This is a visual representation of the abstract.
Introduction
Peritoneal dialysis (PD) should be offered to every eligible individual with kidney failure who is considering maintenance dialysis. Many individuals prefer PD because it can be provided in their homes and offers them more independence than in-centre haemodialysis (HD). PD is prioritised in many regions because it has similar health outcomes and is often less costly than in-centre HD. 1 –3 However, a significant number of individuals with kidney failure are elderly, frail or have other physical or cognitive disabilities, which may limit their ability to perform self-care PD. Individuals may also lack family support for PD. Providing assistance may overcome these barriers, permitting more individuals to receive PD, so assisted PD is a crucial strategy that increases patient choice and provides more equitable access to home dialysis. The objectives of this review are to define the scope of assisted PD for this statement, describe major aspects of assisted PD and provide recommendations to expand its availability internationally.
Defining the scope of assisted PD for this position statement
The inability to perform tasks required for self-care PD and a lack of family, partner or spousal support are common barriers to PD. For these patients, providing assistance with PD in the patient’s home can overcome these barriers and allow them to receive PD in their residence. Assistance with PD has been provided by family members, friends, domestic helpers, personal support workers, healthcare aids, practical nurses or registered nurses. 4 –6 While the working group acknowledges all forms of assistance are valuable, this position statement will focus on assisted PD that is funded by the healthcare system including governments, health insurance, charities or other organisations. This scope includes assistance provided by healthcare professionals, trained laypersons and paid family members. This position statement will exclude support provided by unpaid family members, friends, private caregivers or domestic workers paid by families. The position statement also excludes PD provided by long-term care facilities or other residential institution staff. If external government-funded assistants are visiting these institutions, it is included in the scope of this statement.
Justification of assisted PD
There are many potential barriers to self-care PD (Table 1). These barriers are common in older and often frail people with kidney failure. In a previous study by Oliver et al., PD nurses prospectively recorded barriers to PD during modality education and found that 63% of patients without a contraindication to PD had a physical or cognitive barrier to self-care PD. 7 In a separate study by the same research group, comprehensive geriatric assessments performed on individuals 50 years or older during PD training found that frailty, functional dependency and impaired cognition were very common, leading to a frequent need for assistance in the first 6 months of PD. 8 Experts recommend routine frailty screening in older patients considering PD, which could be done using tools such as the Clinical Frailty Scale. 9,10 If frailty is present, a comprehensive geriatric assessment can be conducted.
Common barriers to self-care PD that can be overcome with assisted PD.a
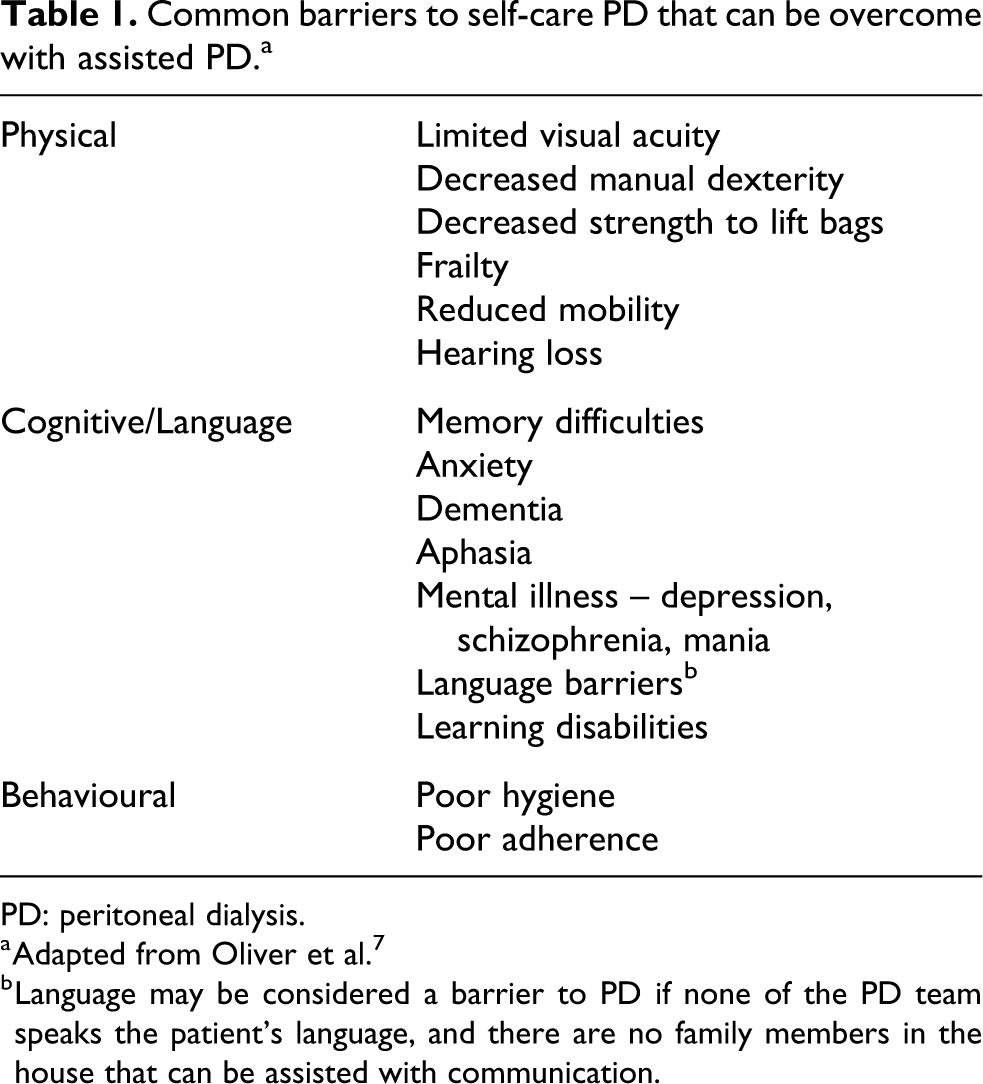
PD: peritoneal dialysis.
a Adapted from Oliver et al. 7
b Language may be considered a barrier to PD if none of the PD team speaks the patient’s language, and there are no family members in the house that can be assisted with communication.
Of particular importance is the issue of cognitive decline. Cognitive decline is a major concern for older people and their families. There is evidence of a higher prevalence of cognitive decline in the HD population compared to the general population with the potentially rapid decline associated with drops in cerebral blood flow and evidence of acute brain injury during HD sessions. 11 –14 People with cognitive decline may be more tolerant of receiving dialysis in a familiar home setting rather than a busy and often noisy HD centre but will require assistance to do so.
Patients who initiate self-care PD may experience a decline in physical or cognitive function over time. 9,15 Other patients may experience a new illness, which temporarily limits their ability to perform self-care. Changes in the health of a caregiver (family, friends) may also reduce support for PD in the home. Assisted PD can provide timely respite care and allow patients to stay on home dialysis – reducing the risk of transfer to HD. If patients recover from their illness, they can often return to self-care PD.
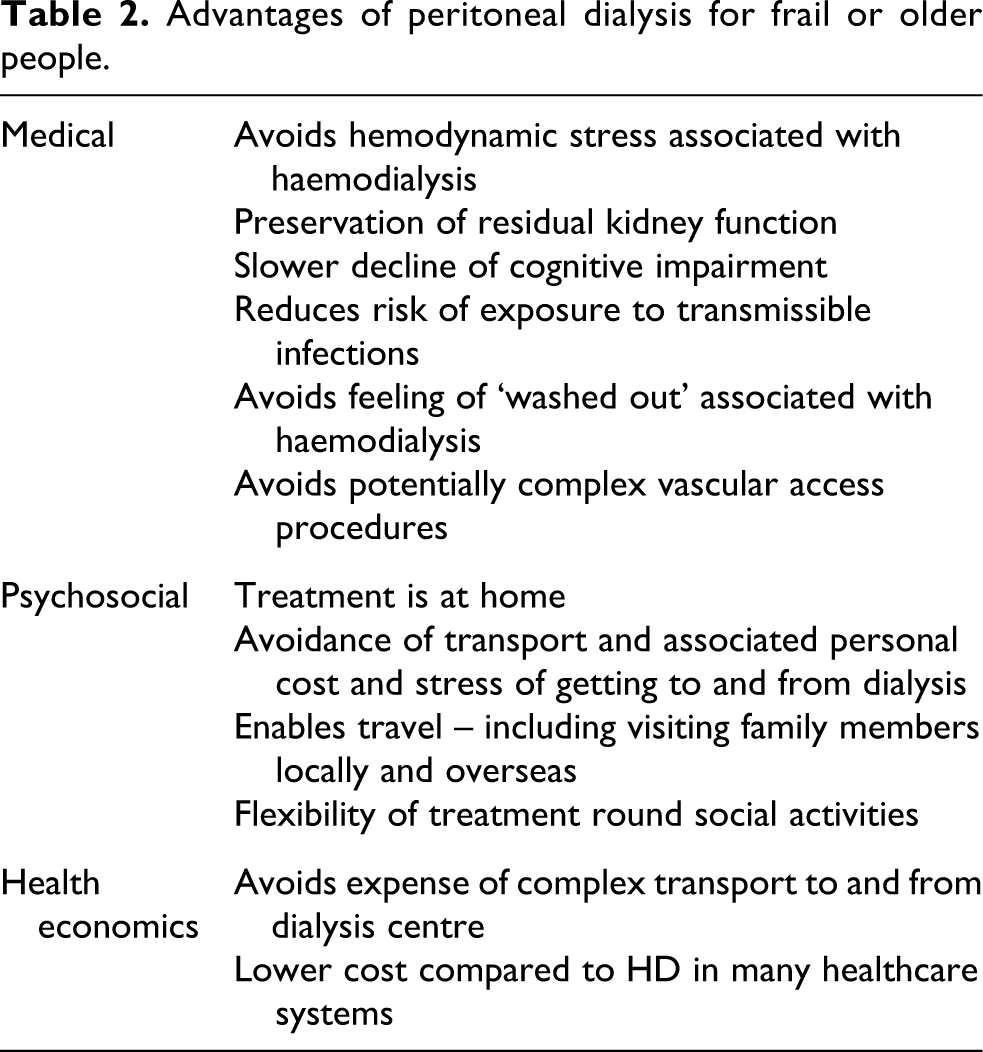
Age is not the only factor leading to the need for assistance. Children and younger adults with disabilities who rely on family-supported PD can benefit from assisted PD to reduce caregiver burden. Many of these patients prefer to receive dialysis in the familiar environment of their home, require complex transport arrangements and cope poorly with in-centre HD. Indeed, dialysis treatment satisfaction has been shown to be better for patients on assisted PD compared to in-centre HD requiring transport. 16 The value of home-based therapies was particularly seen during the COVID-19 pandemic, where infection rates were lower in PD patients and outbreaks in congregate settings occurred. 17 PD was associated with a lower risk of acquiring SARS-CoV-2 infection and related hospitalisation, so assisted PD may prevent vulnerable patients from acquiring transmissible diseases. 17 –19 The advantages of dialysis at home using PD compared to in-centre HD for older patients are shown in Table 2.
Advantages of peritoneal dialysis for frail or older people.
Caregiver burden is common in PD. Studies find that 60% of caregivers of PD patients experience mild to moderate burnout, while 13% experience moderate to severe burnout when measured by the Zarit Burden Interview tool. 20,21 Griva et al. compared the burden of care in family members assisting with PD, usually spouses or adult children, to family members of self-care PD patients also using the Zarit Burden Interview tool. 22 Family members assisting with PD reported more task oriented duties but the burden of care was similar between assisted and self-care PD patients.
Common indications for providing assistance include: during PD initiation to ensure safety and provide reassurance to patients and caregivers long-term assistance for patients with permanent barriers to self-care without caregivers short-term assistance for patients with temporary barriers to self-care without caregivers short-term assistance for patients receiving caregiver support during periods of caregiver illness or burnout short-term assistance for patients during acute illness (e.g. peritonitis)
Recommendations
Individuals considering or choosing PD should undergo an interdisciplinary assessment to identify physical, cognitive, emotional and social barriers to PD that may be overcome by assisted PD
Caregivers of individuals considering or choosing PD should be assessed for burden of care.
Self-care PD or family-assisted PD patients should be monitored regularly for the development of new barriers so that assisted PD can be implemented in a timely manner.
Caregivers of PD patients should be monitored regularly for the burden of care so that assisted PD can be implemented as required.
Assisted PD can be promoted as a strategy to reduce transmissible infection by allowing more patients to be treated at home instead of in a congregate setting.
Models of assisted PD
There is great heterogeneity in the models of assisted PD employed in different regions of the world related to the local culture, health policies, reimbursement systems and penetration of each dialysis modality. 23 –25 Models of care published in the literature differ by geographic scope, funding mechanisms, type of assistants, whether assistants perform cycle connections or manual exchanges, location of assistance and duration of assistance (Table 3).
Key characteristics differentiating models of assisted PD from published studies.

PD: peritoneal dialysis.
The largest geographic scope of assisted programs described in the literature is from France, which is a national program, and in the provinces of British Columbia and Ontario, Canada. Government health insurance funds these programs so they are sustainable. Short-term or respite care is provided along with chronic long-term support. Other assisted programs are often smaller in scope, single centres that are funded by research funds, private dialysis vendors or local dialysis providers and may be less sustainable than programs funded by governments.
The assistants in the French and Ontario programs include registered nurses, while the British Columbia model used licensed practical nurses provided by a private company. The skill set of other assisted models described in the literature includes registered nurses, licensed practical nurses, nurse assistants, healthcare assistants and personal support workers. Paid family members may also provide assistance through state funding in California in the United States, but no studies of this model have been published to date.
Another factor that differentiates assisted PD models is whether assistants provide connections or disconnections from PD cyclers or perform manual PD exchanges. Models that provide this service exist in France, Denmark, and Ontario, Canada, and were provided in a pilot study in Quebec, Canada. Most other programs restrict the assistance, which may allow PD cycler set-up but not connections or manual exchanges. This limited form of assistance may be helpful to patients and families but does not directly assist with PD treatments. Models employing registered or practical nurses can also conduct patient assessments (weight, blood pressure, volume status), provide PD catheter care and administer medications, including intraperitoneal antibiotics if required. A summary of PD related tasks that may require assistance is presented in Table 4.
PD related tasks that may require assistance.a

PD: peritoneal dialysis; BP: blood pressure.
The number of visits depends on whether assistants perform connections or manual exchanges and the patient or family’s capacity to support. In France, up to four visits per day are provided to fully support continuous ambulatory peritioneal dialysis (CAPD). 34 In Ontario, Canada, up to two visits per day are provided to support machine set-up, connections and disconnections. In some cases, patients or families can still perform disconnections, so the first visit is for machine set-up and can be provided throughout the day, while the second visit is timed for the evening connection. Other programs provide daily visits to support machine set-up but not connections, disconnections or manual exchanges. The number of visits may also be limited by cost. Some funding mechanisms may reimburse per visit, while others, such as those in Ontario, Canada, provide a bundled payment with more flexibility.
The duration of service also varies. Established, government-funded programs provide long-term assistance but often have separate funding for short-term or respite care. Assisted programs funded as research or pilot programs may not provide long-term assistance.
Finally, models vary by the location of the service. The majority of assistants visit private residences, but assistants can also visit retirement homes, nursing homes and other institutions other than acute care hospitals to facilitate PD while avoiding the need to train staff in these institutions. Maintaining the skill set of staff in institutions can be challenging if a PD census is not consistent or there is high staff turnover.
Recommendations
Models of assisted PD vary considerably, so their key characteristics should be explicitly considered prior to implementation and described when reporting their outcomes, including their geographic scope, funding mechanisms, type of assistants, whether assistants perform cycle connections or manual exchanges, and duration and location of assistance.
Training and communication with PD assistants
It is necessary to train assistants on PD procedures appropriate to their skill set. In most cases, training will include most tasks performed by self-care patients. The training may be provided by a nurse educator or an experienced PD nurse with knowledge of all PD modalities but can be done in partnership with private corporations or dialysis providers. Training is usually provided at the PD center, but it can also be provided at home care agencies or other educational institutions.
An adequate number of assistants should be trained to provide the required visits in an unrushed manner. The number of assistants will depend on the number of visits needed per patient, the task performed, the number of patients served and the time (distance) to travel between patients. Staff absences should be accounted for with redundant systems in place. Missed visits may have clinical consequences and be distressing to patients and families who are reliant on assistance to perform PD.
PD programs should have a robust communication system in place with assistants. This system can include regular phone calls, emails or rounds to review assisted patients. Virtual telephone or video visits from the home, with the assistant present, can reduce the need for clinic visits for frail, older patients. Remote patient monitoring for the general PD population is associated with reduced transfer to HD, hospitalisation, improved adherence and reduction in patient and caregiver burnout, but its specific role for assisted PD has not been determined. 35
Recommendations
Assistants should be trained by nurse educators or experienced nurses from the PD program using a standardised curriculum.
Assistants should be trained for tasks appropriate to their skill set.
An adequate number of assistants should be available to serve the expected size of the assisted population, accounting for their geographic area with redundant systems in place to account for staff absences.
A robust communication system should be in place between the PD program and the assistant.
Quality control
Continuous quality improvement (CQI), defined as a structured organisational process for involving people in planning and executing a continuous flow of improvement to provide quality healthcare that meets expectations, must be implemented in PD programs. 36,37 International Society for Peritoneal Dialysis (ISPD) guidelines recommend that the PD program monitors peritonitis rates, PD catheter-related complications and rates of transfer to HD in all PD patients, including those on assisted PD. The Standardized Outcomes in Nephrology (SONG) initiative has developed a core outcome set for PD based on the shared priorities of patients, caregivers, clinicians, researchers, policymakers and relevant stakeholders, which included PD-related infection, cardiovascular disease, mortality, time on PD and life participation. 38 Patient-reported outcome measures (PROMs) may be particularly relevant for patients receiving assisted PD. PROMS can be defined as a measure of how a person doing PD is experiencing life and their well-being. It should consider the person’s symptoms and the impact of the dialysis regimen on the person’s life, mental health and social circumstances. 39 Examples of PROMs include the Kidney Disease Quality of Life Instrument, iPOS-Renal, PROMIS Preference Scoring System and SONG Life Participation Questionnaire. 38,40,41 However, older, frail people with cognitive impairment are often unlikely to engage with formal PROMS. ISPD guidelines do not currently recommend measuring PROMS in routine practice. 39
CQI programs have been particularly effective in reducing peritonitis rates. 42 –44 Fang et al. also reported improvements in transfer to HD and survival over time. 45 The working group did not find published quality improvement projects specifically for assisted PD. Oliver et al. found that assisted PD patients have similar rates of hospitalisation as in-centre HD patients. 27 Assisted patient also have lower rates of transfer to HD and similar rates of peritonitis. 26,46 Assisted PD patients are substantially older than self-care PD patients, so they have lower rates of transplantation and higher mortality rates. 26
Transition from assisted PD to either self-care or unpaid family assistance, which can be termed graduation from assisted PD, may be another important indicator. In Ontario, Canada, 48 (38%) of 203 assisted patients graduated from nurse assistance to either self-care PD or family-assisted PD. 27 Graduation rates likely vary on how programs select patients for assistance. For example, some programs may provide assisted PD to nearly all individuals starting PD to ensure safety, provide additional support and then withdraw it. Other programs may take a more restrictive approach, reducing graduation rates.
There was general concordance among the working group that the achievement of sustainable quality control should use standard definitions, data collection methods, analysis and reporting, which a central organisation may facilitate. Assisted PD programs can develop processes and outcome measures to measure patient selection criteria, staff and assistant training, and monitoring of key performance indicators, but these should be formally evaluated and validated prior to widespread use. Specific areas of interest for assisted PD could include missed visits, improperly performed treatments (deviation from practice), predictors of graduation to self-care and patient/family satisfaction.
Recommendations
PD programs should monitor assisted PD populations for outcomes similar to the non-assisted population. These outcomes should take into account local requirements and harmonise with ISPD guidelines.
Reporting of outcomes should be stratified by the use of assistance.
Reporting of outcomes should account for the differences in the case mix of assisted patients (e.g. older age, higher co-morbidity).
Funding
The availability and source of funding for assisted PD varies considerably. Established programs receive sustainable funding provided by the government to support both short- and long-term assistance. Assisted programs, often on a pilot or limited basis, have received funding from research grants, private dialysis vendors or local clinics. Studies based on both real and modelled data have illustrated cost savings associated with assisted PD. 6,47 For example, in British Columbia, Canada, assisted PD added CAD $15,000 annually to the cost of PD, which was less expensive than providing PD in a long-term care facility or providing in-centre HD. The assisted model used licensed practical nurses rather than registered nurses, did not provide PD connections or manual exchanges and provided only one visit per day. In Ontario, Canada, the Ontario Renal Network receives funds from the Provincial Ministry of Health, which are directed to home care agencies or kidney programs to fund assisted PD. Funding is provided as either a short-term or long-term bundle per patient (Table 5). The annual funding for assistance is CAD $20,566 to support nurses who provide PD connections or manual exchanges, up to two visits per day. If dialysis programs receive the funds, they can contract with a home care provider or provide assistance using their staff. The latter model is employed in Ottawa, Canada, with the goal of increasing retention of PD and moving patients towards independence (i.e. self-care or family support), with only 10% of patients supported long term. Case managers are assigned to each patient, and virtual nursing support and physician assessments are also utilised to improve the programs cost-effectiveness.
Annual costs of assistance, peritoneal dialysis and in-centre haemodialysis.
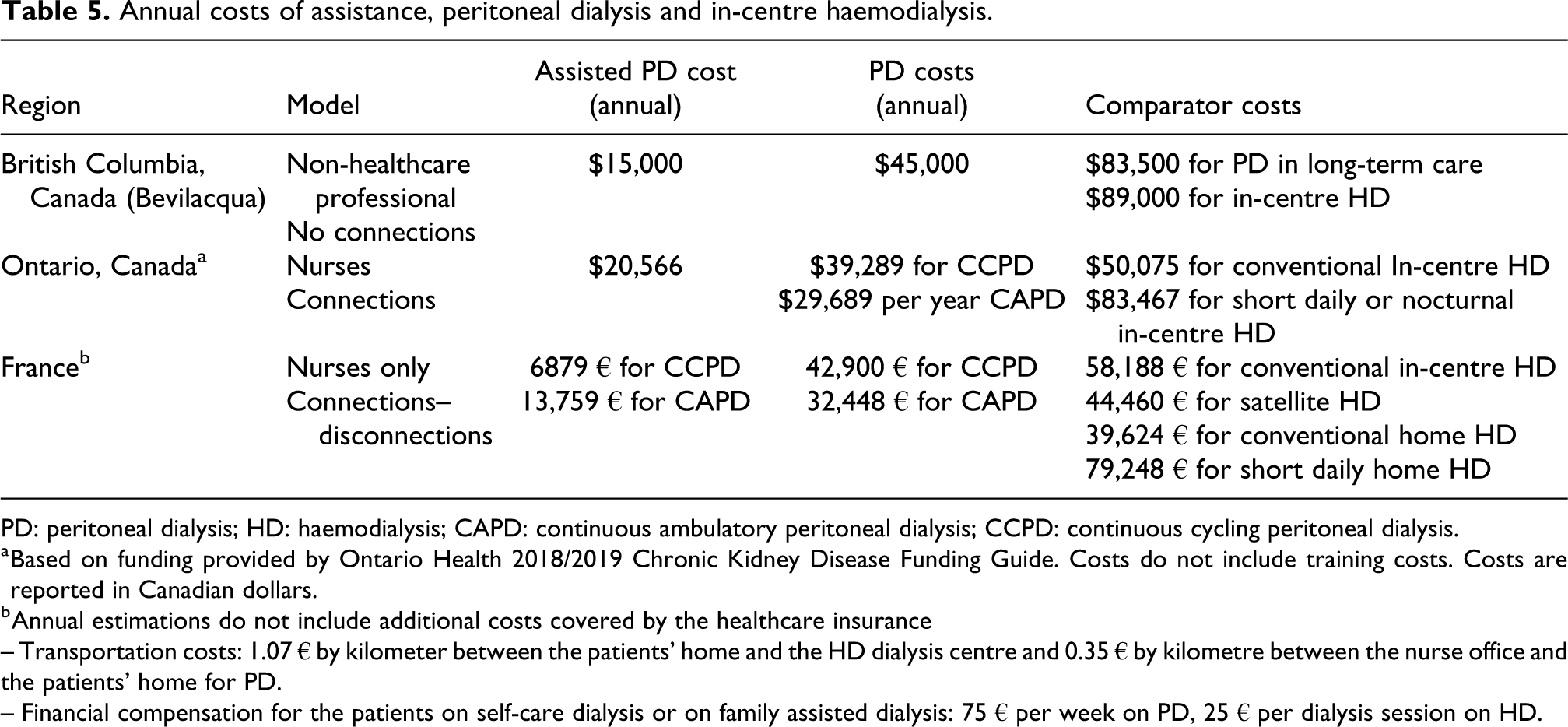
PD: peritoneal dialysis; HD: haemodialysis; CAPD: continuous ambulatory peritoneal dialysis; CCPD: continuous cycling peritoneal dialysis.
a Based on funding provided by Ontario Health 2018/2019 Chronic Kidney Disease Funding Guide. Costs do not include training costs. Costs are reported in Canadian dollars.
b Annual estimations do not include additional costs covered by the healthcare insurance
– Transportation costs: 1.07 € by kilometer between the patients’ home and the HD dialysis centre and 0.35 € by kilometre between the nurse office and the patients’ home for PD.
– Financial compensation for the patients on self-care dialysis or on family assisted dialysis: 75 € per week on PD, 25 € per dialysis session on HD.
In France, the yearly average per-patient cost for providing assistance to patients on PD, including nurse salary, transportation and training, was around 23,400 euros for CAPD and 18,200 euros for APD. 6 Nurses are paid for a maximum of four visits a day in assisted CAPD and two visits a day for APD. In addition, nurses are paid to provide care not directly related to PD and to follow the patient.
In France and Denmark, the operational cost of assisted PD was equal to or lower than that of in-centre HD. 23 Another unpublished study from France, showed that nurse assistance was less expensive for the healthcare system than providing in-centre HD. 48
In Sweden, although healthcare is government-funded, assisted PD differs in various regions. Staff-assisted PD is provided either through primary care teams or advanced home care teams. Approximately 25% of individuals on maintenance dialysis are performing PD, of whom 8% are family-supported, and 8% are receive assisted PD in the home or in nursing homes. Both short- and long-term assistance are provided. 6
Many of these models did not take into account the cost savings achieved with decreased transportation to and from dialysis units, as well as potentially reduced or shortened hospitalisations and emergency department visits. 32 The indirect effect of nurse assistance on social expenditure, such as the influence of nurse visits on the admission of the older dialysis population in nursing homes, has not been evaluated.
These examples illustrate pathways by which government funding effectively supports assisted PD. However, many jurisdictions have no governmental or public funding for assisted PD. 49 This leaves only options such as private insurance (which offers variable coverage) or private insurance paid by patients or families. Both of these options likely increase inequity by restricting PD assistance to patients of higher socio-economic status.
Recommendations
Healthcare systems should fund models of assisted PD so all patients who wish to receive PD but require assistance can have equitable access to PD.
Funding should support short-term and long-term assisted PD.
New programs should have an a priori analysis plan to measure the cost benefits of assisted PD, including indirect savings from reduced transportation, hospitalizations, nursing home admission and avoidance of complications. Costing should account for start-up costs, skill of the assistance, number of visits provided and duration of support.
Cost and availability of assistants can be limited in regions by restricting the number of visits or capping the number of patients in the assisted PD program.
Impact on PD use
There are no definitive studies demonstrating that assisted PD increases prevalent PD use, in part because PD programs usually offer assistance without a control group or randomisation, so the effect on PD prevalence is unknown. However, there is supporting evidence that assisted PD is associated with greater incident use of PD and reduced transfer to HD.
A non-randomised but controlled study from Canada showed that assistance was associated with a higher likelihood of patients being considered eligible for PD by their multidisciplinary team. 50 Forty-seven per cent of patients initiated PD when assistance was available, which was 10% higher in the control region without assistance. A recent study from the UK showed that introducing an assisted PD program was significantly associated with an increased rate of PD initiation (HR: 1.78, 95% CI: 1.21–2.61). 31
In France, Boyer et al. found that assisted PD increased from 2006 to 2015 and helped to maintain PD uptake when family assistance declined over the same time period. 31 Lobbedez et al. from France demonstrated that assisted PD was associated with a lower risk of transfer to HD. 26 Studies of peritonitis, which is a leading cause of transfer to HD, show variable results. Verger et al. found that assisted PD patients had a higher rate of peritonitis compared to family-supported PD, but this difference was mitigated by regular home visits. 46 In Quebec, Canada, where assisted PD was recently implemented, peritonitis rates were similar between assisted and self-care PD. 28 In two studies from France, nurse-assisted PD had a protective effect on peritonitis in diabetic and elderly patients. 51,52 Finally, although the utilisation of assisted PD ranges widely from 8% (Sweden) to 45% (France) of the PD population, it would be expected, although not yet proven, that many of these assisted patients would not remain on PD if assistance was not available.
It should be acknowledged that assisted PD patients are generally older than self-care PD patients, so their time on PD is expected to be less, thereby reducing the impact on PD prevalence. In Italy, when assisted PD was introduced in 2014, a significant increase in PD penetration was not observed. 53 It should also be acknowledged that assisted PD is just one intervention of many that can be used to grow PD, and other system-level factors may influence dialysis modality distributions, which may not be overcome by assisted PD. 54
Recommendations
Assisted PD can be promoted because it is associated with higher incident PD use and reduced rates of transfer to HD.
Prior to implementing an assisted PD programme in a region, historical measures of PD incidence use, time on PD and prevalent use should be reported. If possible, a control population without access to assisted PD should be identified to estimate the impact of assisted PD on penetration.
Future research
Numerous observations studies describe the feasibility, models of care and outcomes associated with assisted PD, but assisted PD is rich area for further investigation. The working group recommends the following areas for future research: Developing standardised methods of assessing patients and caregivers for assisted PD. Comparing low-skill to high-skill assisted PD models. Describing models of paid family assistance, including patient outcomes and costs. Assessing the impact of remote monitoring on assisted PD models of care. Developing and validating quality measures for assisted PD, including the reliability of the assistant visits, treatment errors and rates of adverse events. Assessing the feasibility of measuring PROMS in assisted PD patients. Comparing PROMS between assisted PD and alternative treatment options including in-centre HD and conservative care. Measuring rates and predictors of graduating from assisted PD to self-care or family supported PD. Determining the impact of assisted PD on PD utilisation. Determining the cost-effectiveness of assisted PD programs. Studies measuring the impact of assisted PD on caregiver burnout. Examining whether the impact of assisted PD changes over time on outcomes such as incident PD use, transfer to HD and prevalent PD use.
Summary
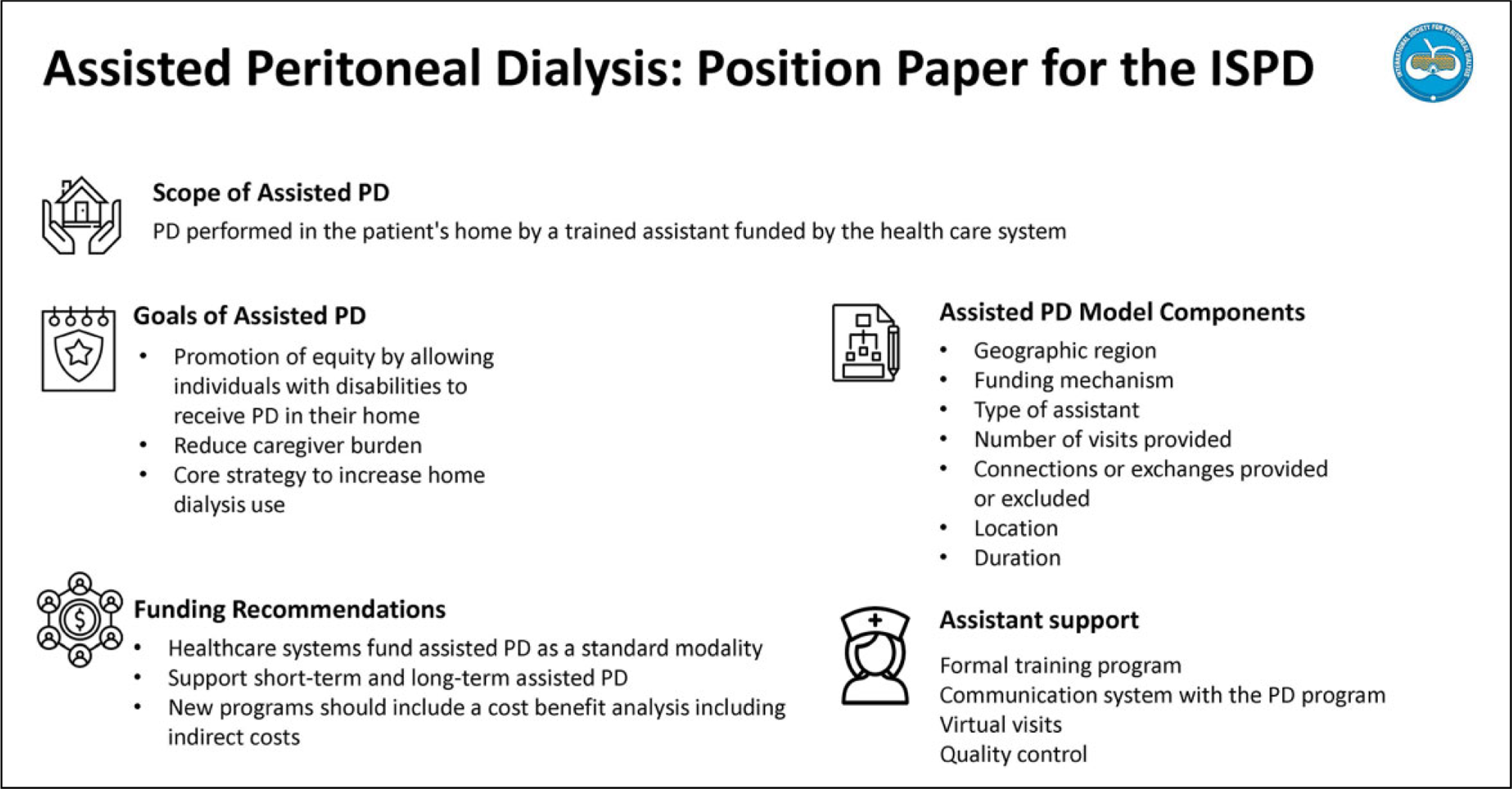
Assisted PD funded by the government is a well-established strategy to grow home dialysis in many regions of the world. Barriers to self-care PD are common in the elderly population and younger patients with physical or cognitive disabilities, so assisted PD increases equity by providing greater access to home dialysis. Models of assisted PD vary greatly, and these key characteristics determine who may receive assistance, their associated outcomes and cost. PD programs should apply the same quality of care metrics measured in the general PD population, but analyses should be stratified and adjusted for case-mix differences between self-care and assisted PD patients. Government healthcare programs or private insurers should adequately fund assisted PD to provide sustainable, long-term support. Assisted PD is associated with increased PD incidence and reduced transfer to HD. The increased costs of delivering assisted PD have generally been less than in-centre HD or providing PD in long-term care. Figure 1 summarizes the main themes of this position statement.
Working group methods and authors’ contributions
The leadership of the ISPD contacted TL and MJO to co-chair an ISPD position statement on assisted PD. The co-chairs assembled a list of experts on PD to join the working group. The co-chairs selected members to provide an international perspective from both low and high-income countries with input from ISPD leadership. The working group met in January 2023 to identify key sections for the position statement. Smaller worker groups were formed for each section including Scope of the position statement (MJO and TL); Justification (CB and EAB); Funding (UHL, JG and PS); Models of assistance; Training and communications with assistants (ASE, JG, EAB, NF and TK); Quality control (CB, DWJ and PL); and Impact on PD use (VJ, GA and AMG). Each group wrote a background summary with key references and provided recommendations based on their expert opinion. The sections were compiled and edited by the co-chairs (MJO and TL).
The working group did not conduct a formal literature review.
Additional excellent reviews of assisted PD are available. 4,6,49,55
Supplemental material
Supplemental Material, sj-pdf-1-ptd-10.1177_08968608241246447 - Assisted peritoneal dialysis: Position paper for the ISPD
Supplemental Material, sj-pdf-1-ptd-10.1177_08968608241246447 for Assisted peritoneal dialysis: Position paper for the ISPD by Matthew J Oliver, Graham Abra, Clémence Béchade, Edwina A Brown, Ana Sanchez-Escuredo, David W Johnson, Anabela Malho Guedes, Janet Graham, Natalia Fernandes, Vivekanand Jha, Nadia Kabbali, Talerngsak Kanjanabuch , Philip Kam-Tao Li, Ulrika Hahn Lundström, Page Salenger and Thierry Lobbedez in Peritoneal Dialysis International
Footnotes
Acknowledgement
None.
Author contributions
Drs MJO and TL were the primary and senior author, respectively. All co-authors contributed equally to the content and writing of the manuscript
Declaration of conflicting interests
MJO is an owner of DMAR systems; Honoraria: Baxter Healthcare; Contracted Medical; employed as a Medical Lead at Ontario Renal Network, Ontario Health. GA has received consulting fees from Baxter Healthcare. EAB had received speaker and consulting fees from Baxter Healthcare, Fresenius Medical Care, Vifor, AWAK and liberDi; DWJ has received consultancy fees, research grants, speaker’s honoraria and travel sponsorships from Baxter Healthcare and Fresenius Medical Care, consultancy fees from Astra Zeneca, Bayer and AWAK, speaker’s honoraria from ONO and Boehringer Ingelheim & Lilly and travel sponsorships from Ono and Amgen. He is a current recipient of an Australian National Health and Medical Research Council Leadership Investigator Grant. AMG has received speaker and consulting fees from Baxter Healthcare. VJ has received grant funding consultancy Fees and honoraria from Bayer, AstraZeneca, Boehringer Ingelheim, NephroPlus, Biocryst, Vera, Visterra, Otsuka, Chinook and Zydus Cadilla; TK has received consultancy fees from VISTERRA, ELEDON, Otsuka OLE and Otsuka VISIONARY as country investigators and is current recipients of the National Research Council of Thailand and the Royal College of Thailand Physician and received speaking honoraria from Astra Zeneca and Baxter Healthcare. PL received speaker Honoraria from AstraZeneca, Baxter Healthcare and Fibrogen; UHL has received speaker and consultancy engagements for Baxter Healthcare and Fresnenius Medical Care. TL received speaker Honoraria from Astellas, Baxter.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs
Informed consent to publish
Yes.
Correction (June 2024):
Article updated to correct the author’s name to “Talerngsak Kanjanabuch”.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
