Abstract
Effective treatment of infections is a growing challenge owing to antimicrobial resistance. Peritoneal dialysis (PD) patients experience more frequent hospitalisations than the general population and have greater exposure to antibiotics, making them particularly vulnerable to this threat. Over the last decade, we have noted a surge in cases of complicated peritoneal dialysis–associated peritonitis (PD peritonitis) caused by antimicrobial-resistant organisms, including extended-spectrum beta-lactamase (ESBL), AmpC beta-lactamase-producing Enterobacterales, Pseudomonas aeruginosa and fungi. Practitioners must be alert to these organisms, seek early recognition of these resistance patterns and make timely adjustments in order to avoid delay in treatment that may increase risk of PD catheter removal and technique failure. We present a case of successful treatment of ESBL peritonitis, highlight its challenges, while providing guidance on management of other unusual and complicated PD peritonitis.
Keywords
Take home points
Peritoneal dialysis (PD) patients have a high risk of acquiring antimicrobial-resistant infections. Clinicians should tailor empiric peritonitis treatment to their local sensitivity patterns and have a low threshold to screen for ESBL.
While pseudomonal peritonitis has become increasingly virulent, and should be treated with dual antibiotics, it does not warrant immediate PD catheter removal, unless there is concurrent exit site or tunnel infection with the same organism.
Detection of multiple gram-negative peritonitis should always raise the concern for bowel perforation. Choice of antibiotics should include anaerobic coverage, and surgery consultation should be sought urgently in the case of an acute abdomen or sepsis.
Fungal peritonitis carries a high risk of PD failure and mortality. Prompt catheter removal and initiation of antifungal therapy are key to treatment success. While the PD catheter needs to be removed immediately, catheter re-insertion is feasible in the future.
Simultaneous catheter removal and replacement has emerged as a safe and effective alternative to PD catheter removal with interim haemodialysis in select cases of tunnel infection and relapsing peritonitis.
Case vignette
A 62-year-old man with end-stage kidney disease due to type 2 diabetes mellitus on automated peritoneal dialysis (APD) for over a year presented to the emergency department with a 1-day history of diffuse abdominal pain, nausea, vomiting, lethargy and cloudy effluent. His medical history was significant for long-standing hypertension, peripheral vascular disease, an episode of urinary tract infection (UTI) 2 years earlier, as well as previous severe community acquired pneumonia treated in India, his native country. He had no prior history of peritoneal dialysis (PD) peritonitis.
In the emergency department, his PD effluent was sent for cell count and culture, and he received 2 g of intravenous (IV) ceftriaxone empirically. He was referred to the nephrology consultation service, which, on reviewing his cell count of 16,324 cells/µL with neutrophilic predominance, diagnosed PD peritonitis and initiated him on intraperitoneal (IP) cefazolin and ceftazidime, along with oral prophylactic nystatin. Another sample collected after a 2-h dwell time confirmed the suspicion (Figure 1). He was admitted to the nephrology in-patient service. On day 2 of antibiotic therapy, his cell count nearly halved, yet there was no clinical improvement. His PD effluent stained for gram-negative bacilli, and a covering physician stopped his IP cefazolin. On day 3, Escherichia coli (E. coli) was isolated. While it demonstrated in vitro susceptibility to ceftazidime, tobramycin and piperacillin/tazobactam, it was resistant to ciprofloxacin and trimethoprim/sulfamethoxazole (TMP-SMX). On careful review, it was found that his previous E. coli UTI exhibited extended-spectrum beta-lactamase (ESBL) properties (resistant to ceftriaxone, gentamicin, piperacillin/tazobactam, tobramycin, TMP-SMX; sensitive to ciprofloxacin), so out of caution, a second agent, IP tobramycin, was added, and an ESBL screen was sent. Accounting for weight and some residual kidney function, a dose of 0.6 mg/kg rounded to 40 mg was chosen to be administered daily over a 6-h dwell. On day 4, his abdominal pain resolved, coinciding with further improvement of PD cell counts. His ESBL screen returned positive: he carried ESBL E. coli resistant to ceftriaxone, ciprofloxacin, piperacillin/tazobactam, only sensitive to tobramycin. He was discharged home on IP tobramycin with close outpatient follow-up. In the days following his discharge, his cell counts continued to improve, and he remained symptom-free. Two weeks later, he developed worsening gait instability with supratherapeutic tobramycin levels, up to 4.1 mg/L. Vestibular ototoxicity and neurotoxicity could not be excluded, so his IP tobramycin was changed to IP meropenem. He successfully completed a 3-week course of antibiotics.
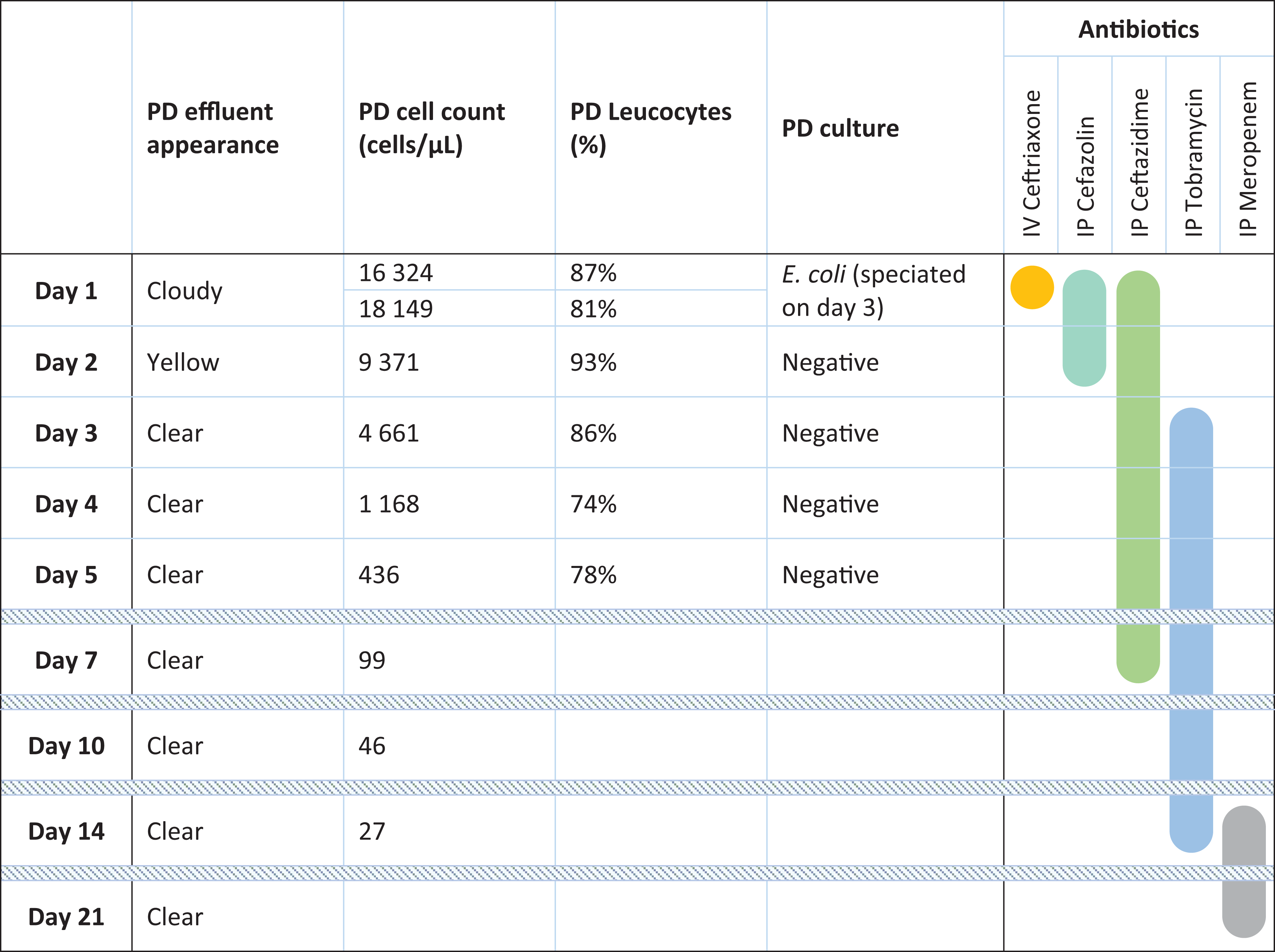
Evolution of PD peritonitis parameters in relation to antibiotic therapy.
In the subsequent six months, he unfortunately experienced an inferior ST-elevation myocardial infarction, followed by Salmonella enterocolitis, and acute subdural haematoma requiring hemicraniotomy. In spite of these complications, he continued to be successfully managed on APD without subsequent relapse, loss of PD catheter, ultrafiltration failure or dialysis inadequacy.
What are risk factors for ESBL peritonitis and when should it be suspected?
ESBL is a plasmid-mediated enzyme that confers some Enterobacterales resistance against most beta-lactam antibiotics except carbapenems and cephamycins. 1 These ESBL-encoding bacteria often carry resistance genes to additional antibiotic classes, such as sulphonamides, aminoglycosides and fluoroquinolones, 2 making their treatment especially challenging. Frequently eluding empiric therapy, they are associated with longer hospital stays, increased morbidity and mortality and pose a growing threat to public health. 3,4 Incidence of ESBL infections is on the rise globally. In the United States alone, the incidence rate increased by 53% from 2012 to 2017. 5,6 E. coli has become the leading pathogen, followed by Klebsiella species and Proteus mirabilis. 2 –5 These bacteria acquire their multi-drug resistance properties either through healthcare exposure or community-acquired infections. Risk factors include prior infection or colonisation with ESBL organisms, recent hospitalisation, residence in a long-term care facility, haemodialysis, presence of an intravascular catheter, recent antibiotic therapy, corticosteroids and immunosuppression (Figure 2). 2,7,8 In recent years, international travel, particularly to Asia, which has the highest burden of ESBL across the globe, has become a major risk factor for ESBL acquisition. 4,9 –13 Practitioners must choose empiric therapy judiciously based on previous susceptibility patterns, have a low threshold to send ESBL screening and adjust antimicrobials promptly at the first sign of resistance.
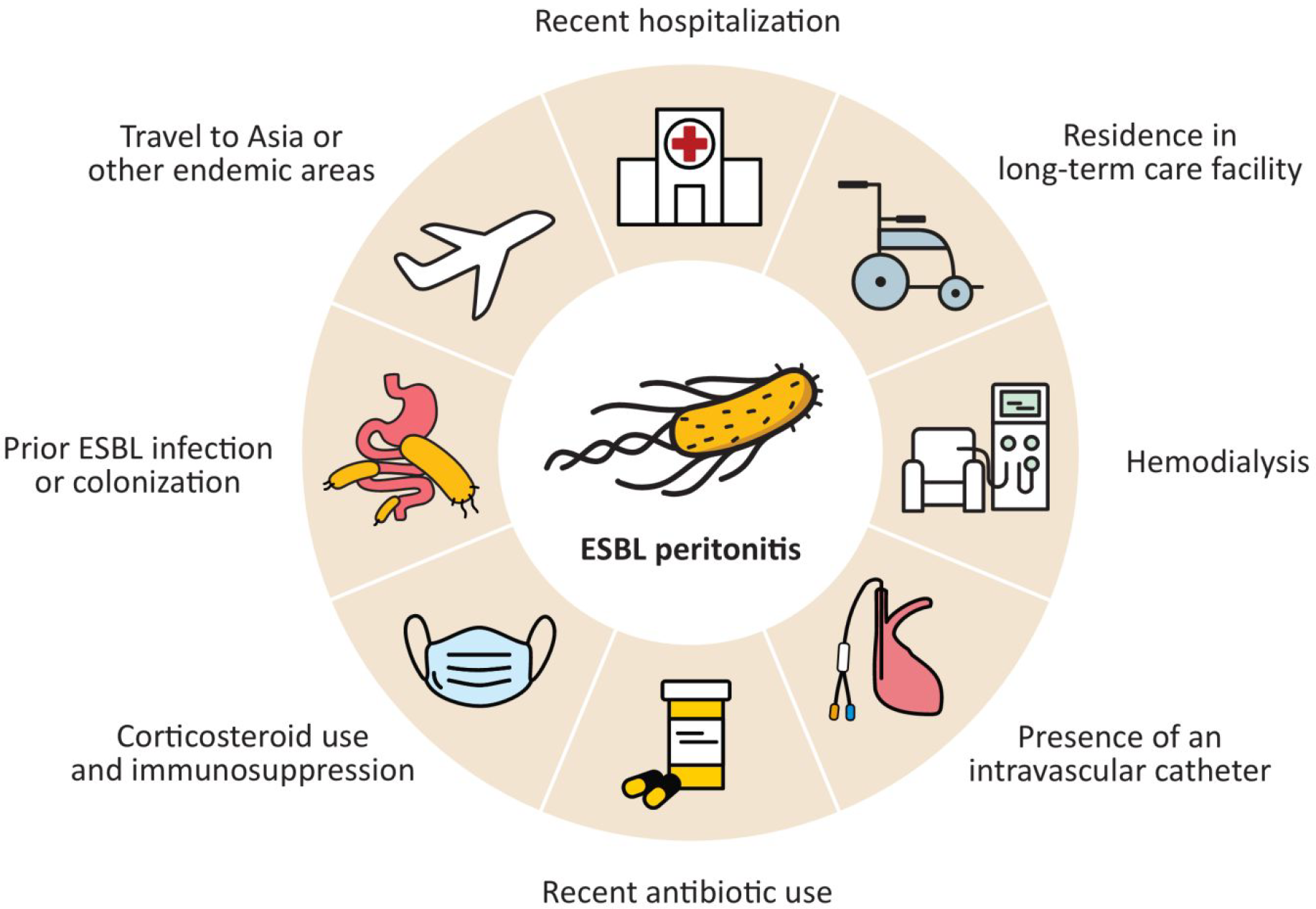
Risk factors for ESBL peritonitis
What are preferred antibiotics for the treatment of ESBL peritonitis?
Carbapenems are recommended as first-line agents to treat ESBL peritonitis. 5,14 Regimens such as once daily IP meropenem are easy to administer and require no drug level monitoring. Alternatively, once daily oral ciprofloxacin is also acceptable if the ESBL in question is susceptible. Table 1 provides a list of reasonable antibiotic options in treating ESBL peritonitis, along with their recommended doses. Selection of antibiotics should be tailored to the specific sensitivity profile. Similar classes are recommended in the treatment of AmpC beta-lactamase-producing Enterobacterales, classically denoted as the ‘SPICE-HAM’ organisms, namely, Serratia, Providencia, indole-positive Proteus, Citrobacter, Enterobacter, Hafnia, Acinetobacter and Morganella species. 5,14
Systemic antibiotic dosing recommendations for treatment of ESBL peritonitis.a

APD: automated peritoneal dialysis; BID: twice daily; CAPD: continuous ambulatory peritoneal dialysis; IP: intraperitoneal; LD: loading dose; MD: maintenance dose; CCPD: continuous cyclic peritoneal dialysis.
a Adapted from International Society for Peritoneal Dialysis (ISPD) and Infectious Diseases Society of America (IDSA).
In the case illustrated above, empiric PD peritonitis treatment with IP cefazolin and ceftazidime was selected, as this was our centre’s standard of practice. Had the history of ESBL-producing E. coli infection been recognised earlier, a different gram-negative coverage should have been considered. In centres with high prevalence of ESBL-producing organisms, empirical aminoglycoside may offer superior gram-negative coverage. 15 One should, however, be mindful of the need for frequent monitoring, and potential ototoxicity and nephrotoxicity. To mitigate risks of aminoglycoside-associated ototoxicity, the use of adjunctive oral N-acetylcysteine has been endorsed by the latest ISPD guidelines on the basis of promising results from three small-sized randomised controlled trials. 14,16 –18 All programmes should adopt continuous quality improvement strategies to review and adapt their individual practice based on evolving susceptibility patterns, and local safety and treatment outcomes.
How should Pseudomonas and Stenotrophomonas peritonitis be treated?
Pseudomonas peritonitis has a cure rate of less than 50% and is thus associated with increased likelihood of catheter removal and permanent haemodialysis transfer. 19 –21 Its virulence stems from both its propensity to produce biofilm 22 –25 and increasing prevalence of multi-drug-resistant-Pseudomonas (MDR-Pseudomonas), defined as resistance to at least one antibiotic from three out of the five classic antipseudomonal classes: cephalosporins, fluoroquinolones, aminoglycosides, monobactams or carbapenems.5 Since 2018, a newer concept of ‘difficult-to-treat resistant’ Pseudomonas, abbreviated as DTR-Pseudomonas, has also emerged, referring to those strains demonstrating non-susceptibility to all antipseudomonal therapies aside from aminoglycosides. 5
In 2009, a large observational study from Australia and New Zealand demonstrated that the need for catheter removal and permanent transfer to haemodialysis reduced from 38% to 10% on dual antipseudomonal coverage. 19 The ISPD peritonitis guidelines have since adopted this practice, recommending that episodes of pseudomonal peritonitis be treated with two antibiotics with different mechanisms of action and to which the organism is sensitive. 14 Despite mounting therapeutic challenges, Pseudomonas peritonitis without concomitant pseudomonal exit site or tunnel infection should be given a therapeutic trial, as a significant proportion of these infections will resolve on antibiotics alone. Table 2 outlines a list of systemic antipseudomonal drugs along with their recommended dosages. As stated above, only peritonitis cases with concurrent exit site or tunnel infection with the same organism warrant PD catheter removal. Lastly, emerging data suggest that topical acetic acid can supplement oral systemic antibiotics when treating pseudomonal exit site infections to achieve synergistic activity and improved outcomes. 26,27
Systemic antibiotic dosing recommendations for treatment of Pseudomonas peritonitis.a

APD: automated peritoneal dialysis; CAPD: continuous ambulatory peritoneal dialysis; IP: intraperitoneal; LD: loading dose; MD: maintenance dose; CCPD: continuous cyclic peritoneal dialysis.
a Adapted from ISPD and IDSA.
Another opportunistic pathogen closely related to pseudomonal species is Stenotrophomonas maltophilia. This microbe was previously named Pseudomonas maltophilia and Xanthomonas maltophilia, prior to acquiring its current designation in 1993. 28 Though it constitutes a less frequent cause of peritonitis than Pseudomonas spp., it tends to be equally multi-drug resistant and prone to biofilm formation. As such, it also requires dual empiric coverage, typically with an oral combination of TMP-SMX and a fluoroquinolone like levofloxacin or ciprofloxacin. 14,29 –31
What should be done in case of multiple gram-negative peritonitis?
Polymicrobial gram-negative peritonitis should raise the concern for intra-abdominal pathology such as perforated viscus. The enteric organisms could enter the peritoneal cavity through a major perforation or through more microscopic perforations, such as across thin-walled colonic diverticula. Urgent surgical consultation must be sought if patient demonstrates signs of sepsis or haemodynamic instability. Treatment must be broadened to target all enteric organisms, including oral metronidazole for anaerobic coverage. Once an enteric source is confirmed, consider holding PD for a period of ‘dry treatment’. The absence of dialysis fluid in the peritoneal cavity may increase the effectiveness of the peritoneal defences to eliminate the infection, but data to support this practice are lacking. Antibiotics can be given intravenously during this dry period or in an isolated 6-h dwell. If laparotomy is needed, most will need PD catheter removal for definite source control. Such decisions should be made in consultation with the surgical team.
How should culture-negative peritonitis be treated?
Culture-negative peritonitis generally carries favourable outcomes, with a cure rate estimated at 67.5–82.3%, exceeding that of culture-positive peritonitis. 32 –35 It typically results from problems with effluent sampling, culture technique or recent antibiotic exposure. 32,33 To address the negative impact of recent antibiotic use on pathogen recovery, most microbiology laboratories in North America now employ culture systems with antimicrobial-neutralising-resins or activated charcoal added to their culture media. 36,37 These resin beads or activated charcoal partially adsorb antimicrobials present in aerobic cultures to enhance microorganism growth. 36 Their adsorption performance, however, is highly variable and directly linked to the specific organisms and the minimum inhibitory concentrations (MIC) of involved antimicrobials. In case of doubt, consultation with a microbiologist familiar with local culture media and laboratory practice is advised to optimise pathogen recovery and reduce false negatives. Persistent infectious symptoms or elevated effluent leukocyte count despite empiric therapy should prompt investigations for fastidious bacteria, mycobacteria and fungi. The current ISPD guidelines suggest that a given PD centre should have no more than 15% of their peritonitis cultures being negative, failing which there needs to be careful re-examination of the sampling and culture techniques. 14 Finally, it is important to recall that a series of non-infectious mimickers, including chemical peritonitis, eosinophilic peritonitis, malignancy, chylous ascites and samples taken from ‘dry abdomen’, can present with cloudy effluent. 38 The lack of neutrophilic predominance is the key feature that distinguishes them from true infectious culture-negative peritonitis.
How should we treat mycobacterial peritonitis (tuberculous and non-tuberculous)?
Tuberculous peritonitis is a rare complication of Mycobacterium tuberculosis (TB) and mimics bacterial peritonitis both clinically and biochemically. It presents as abdominal pain (89%), fever (81%) and cloudy effluent (77%). 39 Although the peritoneal cell count has been reported to show lymphocytic predominance, the majority of cases will exhibit a neutrophilic predominance (65–78%). 39 Owing to its striking similarity to bacterial peritonitis, it poses a diagnostic challenge to clinicians, who often mislabel it as culture-negative peritonitis or refractory culture-positive peritonitis. In a review by Talwani and Horvath, the mean time from presentation to initial treatment was 6.7 weeks. 40 Currently, acid-fast bacilli culture constitutes the gold standard for diagnosing tuberculous peritonitis. While waiting for culture results, adenosine deaminase and polymerase chain reaction (PCR) for M. tuberculosis DNA can serve as adjunct diagnostic tests, with the former having high sensitivity, but low specificity, and the latter highly specific while lacking in sensitivity. 39 Current data do not support PD catheter removal as part of TB peritonitis treatment. Early recognition and timely initiation of antituberculosis therapy remain the cornerstone of management. A comprehensive list of antituberculosis drugs and their dosage recommendations can be found in the ISPD guidelines. 14
In contrast to TB peritonitis, non-tuberculous mycobacterial (NTM) peritonitis requires both prolonged antimicrobial treatment and timely catheter removal for source control. 41,42 Consultation with local NTM expert is advised for optimal drug selection.
How should fungal peritonitis be managed?
Fungal peritonitis presents similarly to bacterial peritonitis with abdominal pain, cloudy effluent and effluent leucocytosis. Intestinal fungal overgrowth following PD peritonitis treatment or indeed any antibiotic exposure is thought to be the major risk factor. 43,44 The mainstay of therapy is catheter removal and prompt administration of an antifungal agent. With treatment failure and mortality as high as 50–91%, 43,45 the focus should be on preventive measures. The ISPD recommends antifungal prophylaxis whenever PD patients are on antibiotics. 14 Our preferred agent is nystatin at 500,000 units orally four times daily (QID) for the duration of antibiotic course, plus 1 week afterward, to allow time for repopulation of the normal bowel flora. It is widely available, is low cost and has an excellent safety profile. Alternative regimens such as oral ketoconazole or fluconazole have also been used, leading to similar success rates, albeit with more adverse effects. 46,47 There is no established ‘minimum duration’ of systemic antibiotics that triggers the need for fungal prophylaxis. At our centre, an antibiotic course beyond 5 days will generally be co-administered with oral nystatin; however, this number is arbitrary. We advise making individualised decisions based on patient risk factors and clinical circumstance.
When it comes to a re-trial of PD following fungal peritonitis, there are no robust data on outcomes. Based on two retrospective studies, 33–37% of patients were able to successfully resume PD after catheter removal for fungal peritonitis. 43,48 As for the optimal time for catheter reinsertion, observational studies suggest 4 weeks to few months. 48,49 The ultimate decision should be arrived at through shared decision-making, in consultation with patients and their caregivers. Patients should be warned that there is a possibility that extensive adhesions can hinder return to PD. 48–50
Of note, in the case vignette, a covering physician altered the antibiotic prescription on day 2 based on gram stain before cultures and sensitivities became available. This practice is generally not advisable and is discouraged by the ISPD guidelines. The main purpose of the gram stain is aimed at early detection of fungal elements, facilitating timely fungal peritonitis management. 14
How should we interpret and manage ongoing culture positivity with improving cell count?
Peritonitis due to more virulent species, such as Staphylococcus aureus or Pseudomonas aeruginosa, may take longer to resolve. In the face of a decreasing leucocyte trajectory and improving symptoms, providers should allow a reasonable trial of antibiotics prior to declaring failure of therapy. The updated 2022 ISPD guidelines reinforce this principle by revising their previous recommendation, changing from mandatory PD catheter removal if effluent does not clear by day 5, to allowing an observation period of longer than 5 days, as long as the effluent cell counts are declining on appropriate antibiotic treatment. During this interval, it is crucial to look for concurrent exit site or tunnel infection, which would be an indication for catheter removal. Even in the absence of clinically detectable exit site or tunnel infection, ongoing culture positivity in the face of improving or resolved peritoneal fluid leucocytosis implies that the IP portion of the PD catheter has been colonised by the bacterium. It is likely that the peritonitis will relapse with discontinuation of antibiotics. Because of the lack of source control, the PD catheter should be removed.
In contrast, a positive culture with a negative cell count suggests a false positive, likely arising from sample contamination. If this happens often, it is suggested to discuss the culture chain with the PD unit and the microbiology lab. If the patient presents with abdominal symptoms that prompted the culture, clinicians should repeat the cell count and, if it remains normal, investigate alternative aetiologies for abdominal pain or cloudy effluent, where appropriate.
There is emerging evidence that simultaneous catheter removal and replacement (SCR) under antibiotic coverage may be a safe alternative to PD catheter removal and interim bridging with haemodialysis. Whether for relapsing peritonitis or refractory tunnel infections, several pilot studies have shown a 72.7%–100% success rate of SCR at maintaining patients on PD without subsequent peritonitis relapse or perioperative transfer to haemodialysis. 51 –53 The effluent needs to be clear and free of organisms at the time of SCR, and the procedure needs to be done with peri-operative antibiotic coverage. In our programme, we have used SCR for coagulase-negative staphylococci (CoNS), but not for Pseudomonas spp. or S. aureus catheter colonisation. Larger-scale studies are needed to confirm the effectiveness of this promising technique.
What if symptoms are improving on empiric therapy but sensitivity analysis shows resistance?
Heywood and Bargman reviewed a series of CoNS peritonitis treated with cephalosporin despite resistance on the sensitivity report. While there was an initial response, these CoNS were more prone to relapse compared to those treated with vancomycin. 54 Extrapolating from these data, when facing resistant or intermediate sensitivity, our general approach is thus to tailor antibiotics to susceptibility patterns, as cell count and culture can transiently improve prior to relapsing. Targeted therapy also reduces risks of catheter removal, transfer to haemodialysis and antibiotic resistance. 55 Conversely, if initial sensitivity testing shows susceptibility to empiric antimicrobials, but symptoms persist or deteriorate, practitioners should have a low threshold to repeat effluent cell count and culture as many pathogens illustrated above develop inducible in vivo resistance which might not be reflected in the results of the first culture and sensitivity.
The remote patient – Should they have home antibiotics?
Delays in initiating antimicrobial therapy for the treatment of peritonitis increase the risk of PD failure considerably. 56 Because IP antibiotic treatment is not always available after hours, some dialysis centres supply patients with oral or IP antibiotics to keep at home for prompt administration when symptoms occur. While this approach allows for early treatment of apparent peritonitis, it should be weighed against the increased risk of culture-negative peritonitis from antibiotic use prior to adequate effluent sampling. To avert such mistakes, patients should be reminded of draining and refrigerating their PD effluent samples before antibiotic administration.
Footnotes
Acknowledgement
None.
Author contributions
SZ wrote the first draft of the manuscript. Both authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JMB is speaker and consultant at Baxter Healthcare and DaVita Healthcare Partners. This paper only addresses PD peritonitis management in adults; the approach in children may differ.
Ethical Approval
None.
Informed Consent
None.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
