Abstract

A diagnosis of filamentous mould peritonitis is challenging and may be misclassified as culture-negative peritonitis with routine diagnostic tests. Intraluminal colonisation of the peritoneal dialysis (PD) catheter might be the only clue for establishing the microbiologic diagnosis as presented here.
A 77-year-old woman on continuous ambulatory PD for 10 years presented with acute abdominal pain, diarrhoea and cloudy PD effluent (PDE) for 1 day. She denied having a prior recent history of peritonitis. The initial PDE leucocyte count was 270 cells/µL with neutrophil predominance (92%). Peritonitis was diagnosed, and daily intraperitoneal cefazolin and ceftazidime were administrated without antifungal prophylaxis. The PDE and blood cultures for bacteria and fungi demonstrated no growth. However, on day 7 after the initial presentation, she developed worsening abdominal pain. PDE leucocytes were also increased from 150 cells/µL (day 5) to 280 cells/µL (day 7) and 1660 cells/µL (day 8). Moreover, dark material was observed inside the PD catheter (Figure 1(a)), and a positive day-7 serum galactomannan index was reported on day 9 (6.2, cut-off 0.56); therefore, fungal peritonitis was suspected. Empiric intravenous amphotericin B was initiated, antibiotics were changed to intravenous meropenem (day 7) and the PD catheter was promptly removed (day 9). Dense and scattered particles were found lodging inside the distal and proximal ends of the PD catheter’s lumen, respectively (Figure 1(a)). Potassium hydroxide stain of the particles depicted numerous dark filamentous moulds (Figure 1(b)). Verruconis gallopava was identified by fungal culture from the catheter with proven pan-drug resistance against amphotericin, voriconazole, itraconazole, fluconazole and caspofungin (Figure 1(c) to (e)). On day 18, after the initial presentation, the patient passed away despite the initiation of antifungal therapy. 1
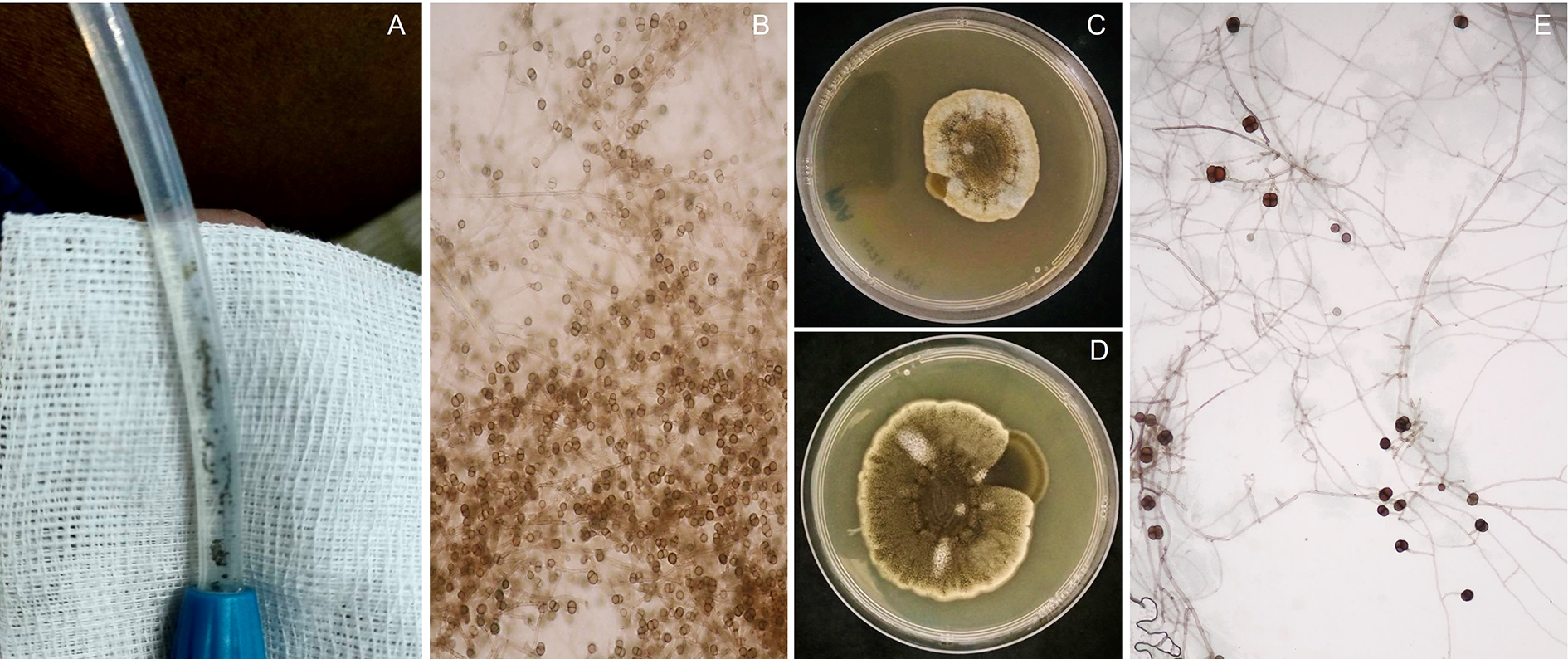
(a) Dark dense-packed materials were recognised inside the PD catheter. (b) KOH smear depicts numerous black moulds with global spores. (c) Colony on potato dextrose agar demonstrates velvety, cream colour with central brown to olivaceous on day 7 and (d) becomes darker and woolly appearance on day 17. (e) Long septate hyphae with sub-globose-shaped conidia attachment are observed in lactophenol blue (inset with 1000×).
Fungal peritonitis is associated with high morbidity and mortality. 2 Leaving the catheter in situ or delay in catheter removal after diagnosis of fungal peritonitis is independently associated with higher mortality. 3,4 Ram et al. 4 demonstrated that mortality rates increase exponentially with a delay in catheter removal. Thus, the 2022 International Society for PD (ISPD) peritonitis guidelines recommend ‘immediate catheter removal when fungi are identified in PDE (1C)’ 1 ; however, PDE culture has low sensitivity in detecting environmental moulds, as demonstrated in our case, and is frequently time-consuming. 5,6 Detection of dark particles inside the PD catheter was the primary clue for establishing the microbiologic diagnosis and management in our case.
V. gallopava, formerly known as Ochroconis gallopava, is a thermophilic dematiaceous mould, commonly found in soil, compost piles and wasted environment. 7 While it is recognised increasingly as a cause of pulmonary and central nervous system infections among immunocompromised and solid organ transplant recipients. 8 PD-related peritonitis caused by V. gallopava was described once in a cardiac transplant patient. 9 The presence of infectious particles led to the clinical suspicion of fungal peritonitis in both cases receiving PD. The location of visible fungal materials is different. The former found them around the PD catheter and in the drained PD bag, while in the present case, particles were found inside the catheter lumen. Since both patients shared the same occupation as agricultural workers and had no chest symptoms with normal chest radiographs, the environmental contaminations while performing PD bag exchanges could be suspected as the primary route of pathogen transmission.
Despite no history of an immunocompromise host and prompt initiation of the empirical antifungal agent and the PD catheter removal, the patient’s outcome was unfavourable. The antifungal susceptibility pattern turned out that the pathogen had broad resistance to antifungal medications. This led to concern about emerging drug-resistant pathogens in the environment.
In conclusion, a careful inspection of the PD catheter lumen should be integrated into routine clinical practice, particularly when the infection is refractory to empirical antimicrobials and PDE cultures yield negative microorganisms. We describe a fatal pan-drug resistant mould V. gallopava in an immunocompetent patient receiving PD. This reinforces the importance of hand hygiene and aseptic technique in all PD patients.
Footnotes
Acknowledgements
The authors would like to acknowledge the contributions of physicians, nurses, technicians and social workers who are not listed as authors in this work and take care of the patient in the Maharat Nakhon Ratchasima Hospital and the samples in the King Chulalongkorn Memorial Hospital Microbiological Laboratory. The authors also like to thank our research assistants, Ms Thunvarat Saejew and Ms Preeyarat Pavatung, Center of Excellence in Kidney Metabolic Disorders, for their fantastic works.
Author contributions
Watthikorn Pichitporn, Watanyu Parapiboon, Laddaporn Wongluechai, Suppachok Kirdlarp and Talerngsak Kanjanabuch collected the data. Kanin Thammavaranucupt and Talerngsak Kanjanabuch concentualized and drafted the manuscript, as well as responded to the reviewers. All authors reviewed and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TK has received consultancy fees from VISTERA as a country investigator and current recipient of the National Research Council of Thailand and received speaking honoraria from Astra Zeneca and Baxter Healthcare. All other authors declare that they have none.
Ethical approval
Chulalongkorn University does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Thailand Science research and Innovation Fund Chulalongkorn University CU_FRB65_hea (19)_026_30_07 and the Ratchadapiseksompotch Fund Chulalongkorn University, Chulalongkorn University, Thailand (HEA663000115 and HEA663000116).
Informed consent to participate
Informed consent, in accordance with the Declaration of Helsinki, was obtained from the patient’s next of kin.
Informed consent to publish
Written informed consent was obtained from the patient’s next of kin for their anonymised information to be published in this article.
