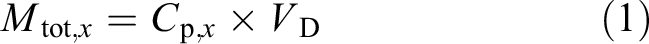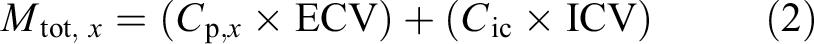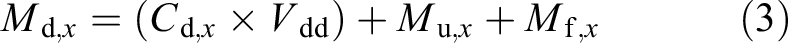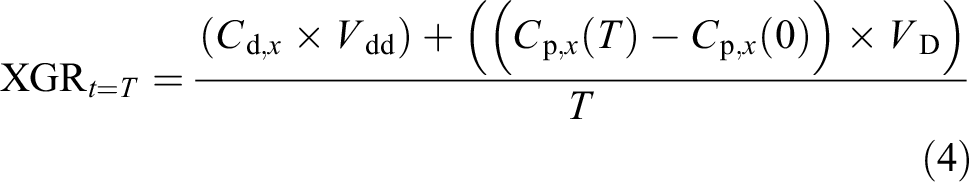Abstract
Background:
Patients with end-stage kidney disease (ESKD) require dialysis or transplantation for their survival. There are few experimental animal models mimicking the human situation in which the animals are dependent on dialysis for their survival. We developed a peritoneal dialysis (PD) system for rats to enable long-term treatment under controlled conditions.
Method:
Rats were chemically nephrectomised using orellanine to render them uremic. Two studies were performed, the first with highly uremic rats on PD for 5 days, and the other with moderately uremic rats on PD for 21 days. Blood and dialysate samples were collected repeatedly from the first study and solute concentrations analysed. Based on these values, dialysis parameters were calculated together with generation rates allowing for kinetic modelling of the effects of PD. In the second study, the general conditions of the rats were evaluated during a longer dialysis period.
Results:
For rats with estimated glomerular filtration rate (GFR) 5–10% of normal (moderately uremic rats), five daily PD cycles kept the rats in good condition for 3 weeks. For highly uremic rats (GFR below 3% of normal), more extensive dialysis is needed to maintain homeostasis and our simulations show that a six daily and four nightly PD cycles should be needed to keep the rats in good condition.
Conclusion:
In conclusion, the PD system described in this study can be used for long-term studies of PD on uremic dialysis-dependent rats mimicking the human setting. To maintain whole body homeostasis of highly uremic rats, intense PD is needed during both day and night.
Introduction
Chronic kidney disease (CKD) has been recognised by the World Health Organization as one of the main threats to global health with large challenges to the medical community. 1,2 Patients that develop end-stage kidney disease (ESKD) require kidney replacement therapy for their survival with the options of transplantation or dialysis. 3,4 Haemodialysis (HD) is the most common form of dialysis. The other form of dialysis is peritoneal dialysis (PD) and approximately 200,000 patients are treated with PD 5 and of those 20,000 reside in Europe. 6 The major challenge with kidney replacement therapy is that large patient groups do not have access to dialysis leading to large numbers of deaths annually worldwide from kidney failure. 7 There is an urgent need to improve dialysis and reduce the cost making it accessible for patients worldwide. Compared to HD, PD is less expensive 8 and provides a more flexible and better quality of life 9 while having similar therapeutic outcomes. 10 One of the challenges with PD is that long time treatment with PD may induce changes in the peritoneal membrane leading to peritoneal ultrafiltration failure and the need to switch from PD to HD. To improve PD in humans there is a need for a good experimental animal model of PD that resembles the human setting as close as possible. Rats are considered a suitable animal for PD modelling and two approaches are mainly used, either on non-uremic or uremic animals. The first approach uses only intraperitoneal (ip) injection of PD fluid, without draining the fluid, and the second involves implanting a peritoneal catheter enabling drainage of the PD fluid. 11 The most common method for achieving a state similar to that of clinical uremia in these PD rat models is 5/6 nephrectomy, 6 , 12 – 16 which requires surgical trauma. However, these rats do not develop clinically relevant dialysis-dependent uremia. 14,17 One reason for limited innovation in the PD field might be lack of clinically relevant animal models. 6 , 14 , 15 , 18 , 19
In this study, to enable adequate PD dialysis of uremic rats we used an automated PD model previously developed in our laboratory for studies of kidney cancer. 20 To render the rats uremic, the rats were chemically nephrectomised using the nephrotoxin orellanine. 21 Two separate studies were conducted, one with moderately uremic rats on long-term PD (21 days) and one with highly uremic rats on short-term PD (5 days).
Methods
Animals and experimental design
Ethical approval for this study was obtained from the Gothenburg Ethical Board for Animal Experiments: approval number 144-12, 44-15 and 1451 and performed according to the NIH guidelines. During the study, all rats had free access to standard food and water and were housed in a specially adapted Scantainer (Scanbur Technology A/S, Denmark) that ensured a pathogen-reduced environment with 12-h dark–light cycles and optimised humidity, temperature and ventilation. Video cameras (KGUARD QRT-301, Taiwan) enabled monitoring of the animals.
Highly uremic rats on short-term PD: 23 female Wistar rats weighing 175–200 g upon arrival were used. The rats were randomly assigned to four different groups, one control group consisting of healthy rats (n = 5) that received PD with five dwells a day and three groups that were chemically nephrectomised using orellanine (7 or 12 mg/kg ip) producing dialysis-dependent ESKD. The 5 days PD schedules for the three groups of uremic rats were: one with five cycles per day (5D/0N, n = 6), one with PD with five cycles per day and one long exchange with icodextrin during the night (5D/icodextrin, n = 6) and one group that received five cycles per day and then two cycles during the night after the two first nights had passed (5D/2N, n = 6), see Figure 1(a). As mandated by the experimental protocol, all rats were euthanised after 5 days of dialysis.
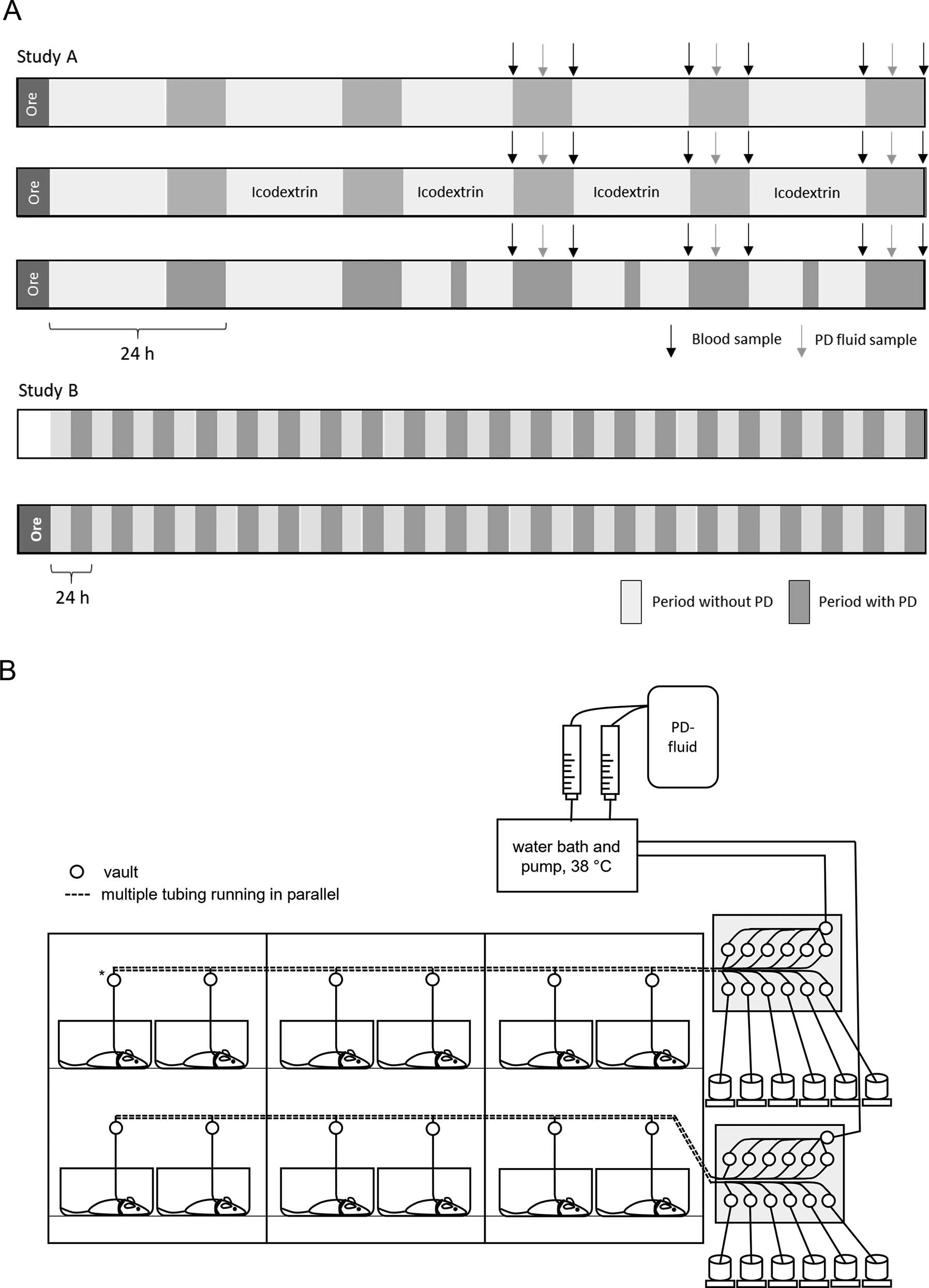
Illustration of the experimental set-up and PD system. (a) Schematic illustration of the experimental set-up for the different PD prescriptions in study A and B. Orellanine was administered the day before PD was initiated, 7 or 12 mg/kg for study A and 5 mg/kg for study B. (b) Drawing illustrating the PD system. Rats were housed in a Scantainer allowing a controlled and sterile environment with two systems for PD installed. The rats had a harness attached to the two PD catheters and connected to a swivel allowing the rats to move freely. The PD machines controlled in- and outflow of each rat and the fill and drain of the dosing syringe. A water bath ensured a constant temperature of 38°C. The syringe was equipped with fluid detectors and air supply through a sterile filter. The drained dialysate was collected from each animal in a container and the volume monitored. *Three-way valve connected to a sterile filter and syringe in case tubing needs to be flushed with sterile saline.
Moderately uremic rats on long-term PD: 16 female Wistar rats weighing approximately 150 g upon arrival. The rats were randomly assigned to two groups: a control group or a group that was rendered moderately uremic with a lower dose of orellanine (5 mg/kg ip) than in the study of highly uremic rats to avoid severe uremia. Both groups received daytime PD with five cycles per day for 21 days (n = 8 respectively). As mandated by the experimental protocol, all rats were euthanised after 21 days of dialysis.
PD-catheter implantation
After arrival, the rats were allowed a minimum of 1 week to adapt. Before surgical implantation of a PD catheter, the rats were washed with Hibiscrub (Regent Medical Overseas Limited, London, UK) and chlorhexidine. Customised sterile rat heparin-coated polyurethane PD catheters (CBAS-50; Instech Laboratories Inc., Pennsylvania, USA) were used. Inhalation of isoflurane (2–3% v/v; Schering-Plough, Stockholm, Sweden) mixed with air (approximately 1 L/min) in an isoflurane vaporiser (AngThos, Lidingö, Sweden) was used for anaesthesia. Before the surgery, the abdomen and a small section of the back of the neck were shaved and covered with a sterile barrier. The catheters were implanted in the peritoneal cavity and secured in place with stitches and passed subcutaneously via a steel guide to the back of the neck where the ends were connected to a dual Luer Lock harness (SAI-Infusion Technologies, Lake Villa, Illinois, USA). The abdominal surgical incision was closed with skin staples. A subcutaneous dose of 0.03 mg/kg buprenorphine (Temgesic®; RB Pharmaceuticals Limited, Berkshire, UK) was used to ensure minimal pain sensation after the operation. A subcutaneous dose of 2 mg/kg ciprofloxacin (Hospira UK Limited, Maidenhead, UK) was given as a prophylactic dose for infection. One day after surgery, the harness was connected to the PD system through a swivel tubing system.
Introduction of dialysis-dependent ESKD
Dialysis-dependent ESKD was induced using orellanine (Ramidus AB, Lund, Sweden), a kidney-specific mushroom toxin. 20 –23 The orellanine (5, 7 or 12 mg/kg body weight) was administered directly into the peritoneal cavity, through the connected harness the day after the surgery.
Peritoneal dialysis
The day after the orellanine dose, dialysis was initiated and continued for 5 days (highly uremic rats) or 3 weeks (moderately uremic rats). The dialysis fluid (Physioneal 40 Baxter, Canada) was supplemented with vancomycin, 20 mg/L, ceftazidime 30 mg/L and heparin (2.5 IU/mL). The tubing system was pre-warmed (38°C) in a circulating water system and a syringe was used for precise dosing. Each dialysis exchange was performed with a dwell time of 45 min and 15 min of emptying by gravity. The nightly icodextrin dwell was left over night (from 18.00 to 8.00). The PD system was controlled by a computer software acting on the in- and outflow valves. Drained dialysate was collected, and volume measured. Two PD systems were used allowing dialysis of 12 rats simultaneously, see Figure 1(b) and Supplementary Figure 1. The PD fill volume was 15 mL/dwell for all rats in both sub-studies. All PD cycles were supervised for animal safety and the PD catheter could be flushed manually to ensure complete drainage.
Highly uremic rats on short time PD, sample collection and analysis of small solutes
In the study of highly uremic rats, blood samples were collected before and after daytime dialysis on days 3–5 from the tail vein. Dialysis fluid from the second cycle of the daytime PD was collected on days 3–5. Serum and dialysis concentrations of creatinine, blood urea nitrogen (BUN), phosphate, glucose and potassium were analysed by Adlego Biomedical AB (Solna, Sweden).
Highly uremic rats on short time PD, kinetic modelling
All highly uremic rats from this study were included in the kinetic modelling. The total body water (TBW) in rats weighing about 250 g is 170 mL, and by calculations the estimated distribution is 113.3 mL intracellularly (ICV) and 56.7 mL extracellularly (ECV). The plasma volume is 7.8 mL (total blood volume is 15.8 mL). 24 Volumes of distribution (V D) for solutes vary as not all solutes are equally distributed in the body.
The kinetic modelling was done assuming kidney and faecal elimination to be negligible, and metabolites and solutes only eliminated by dialysis. Metabolites that are not eliminated accumulate in the body fluids. Thus, the metabolic generation rate (GR) of a solute equals the amount eliminated via dialysis and the amount accumulated in the body during the observation period (t). The total amount of a solute X in the body at any given time is calculated by
C p, x is the concentration of solute X in the plasma. For solutes mainly distributed in extracellular compartments V D equals ECV. For metabolites with similar concentrations in ECV and ICV, V D equals TBW. Solutes with much higher concentration in the ICV than ECV is calculated by
where C ic is the concentration of solute X in the ICV.
The amount of eliminated solute X equals
where C d, x is the concentration of solute X in the dialysate and V dd is the dialysate drain volume. M u, x and M f, x are the amount of solute in the urine and faeces and they are considered negligible in our experimental set-up.
In nephrectomised rats, the GR equals the elimination rate (ER) by dialysis corrected for potential changes in the total mass of the metabolite in the body fluids. The generation rate of solute X at any given time T (XGR t = T , in mmol/min) is then given by
Thus, if the plasma concentration is the same at time 0 and time T, the XGR equals the amount eliminated in the dialysate during the time period. If C p, x (T) > C p, x (0), more metabolites have been generated than eliminated. The GR is assumed to be constant during the time period between the blood samples for all metabolites. Due to the limited number of blood samples and blood sample volume this assumption is necessary for further calculations, even though food intake and activity level may vary between two blood samples. The calculations of PD clearance, Kt/V, and the GR of each solute are described in the supplement.
Statistical analyses
All results are presented as a mean ± standard error of the mean if nothing other is stated. Statistically significant differences were determined using Student’s t test or Kruskal–Wallis with Tukey’s multiple comparisons test. Statistics were calculated using the software GraphPad Prism version 7.02 (GraphPad Software, La Jolla, California, USA). A p value of <0.05 was considered significant. Information about missing samples and samples out of the detection ranges is found in the supplement.
Results
Two studies were conducted: one study with highly uremic rats on short time PD for 5 days and one with moderately uremic rats with long time PD for 21 days. For the highly uremic rats three different dialysis schedules were used: 5D/0N, 5D/icodextrin and 5D/2N.
Highly uremic rats on short-term PD: The general condition of rats
The rat’s body weight at the time of surgery was 249 ± 4 g for the 5D/0N, 254 ± 5 g for the 5D/icodextrin and 280 ± 9 g for the 5D/2N group. Orellanine induced kidney failure in all rats as reflected by the increased creatinine in the blood compared to the control group of healthy rats on PD (healthy rats on PD 44.4 ± 2.6 µmol/L and highly uremic rats, pre-dialysis day 3, 435.2 ± 27.6 µmol/L, p < 0.001). Estimated glomerular filtration rates (GFRs) for the rats were calculated using the equation developed by Besseling et al. 25 which gave a total eGFR (kidney and PD clearance components) of 3.9% ± 1.95% (error assumed to be 50% of mean) of normal GFR. The PD clearance was measured to be around 6.3% ± 3.15% (error assumed to be 50% of mean) of normal GFR. This would imply a negative GFR of −2.4% but with the uncertainty in the estimations the range is likely to be 0–2.7% of normal GFR. On average the ultrafiltration was +15 mL during the day and +5 mL for the rats getting icodextrin PD during the night.
Highly uremic rats on short time PD: Electrolytes in blood and dialysate
After the daytime PD, the concentrations of BUN, creatinine, potassium, phosphate and glucose in the plasma of uremic rats were reduced, see Table 1. As the rats were highly uremic, the concentrations were higher than for healthy rats on PD (Table 1). Supplementary Table 1 contains the concentrations of solutes and glucose in the drained dialysate on days 3–5 for the different groups.
Electrolytes and glucose in plasma before and after daytime peritoneal dialysis.
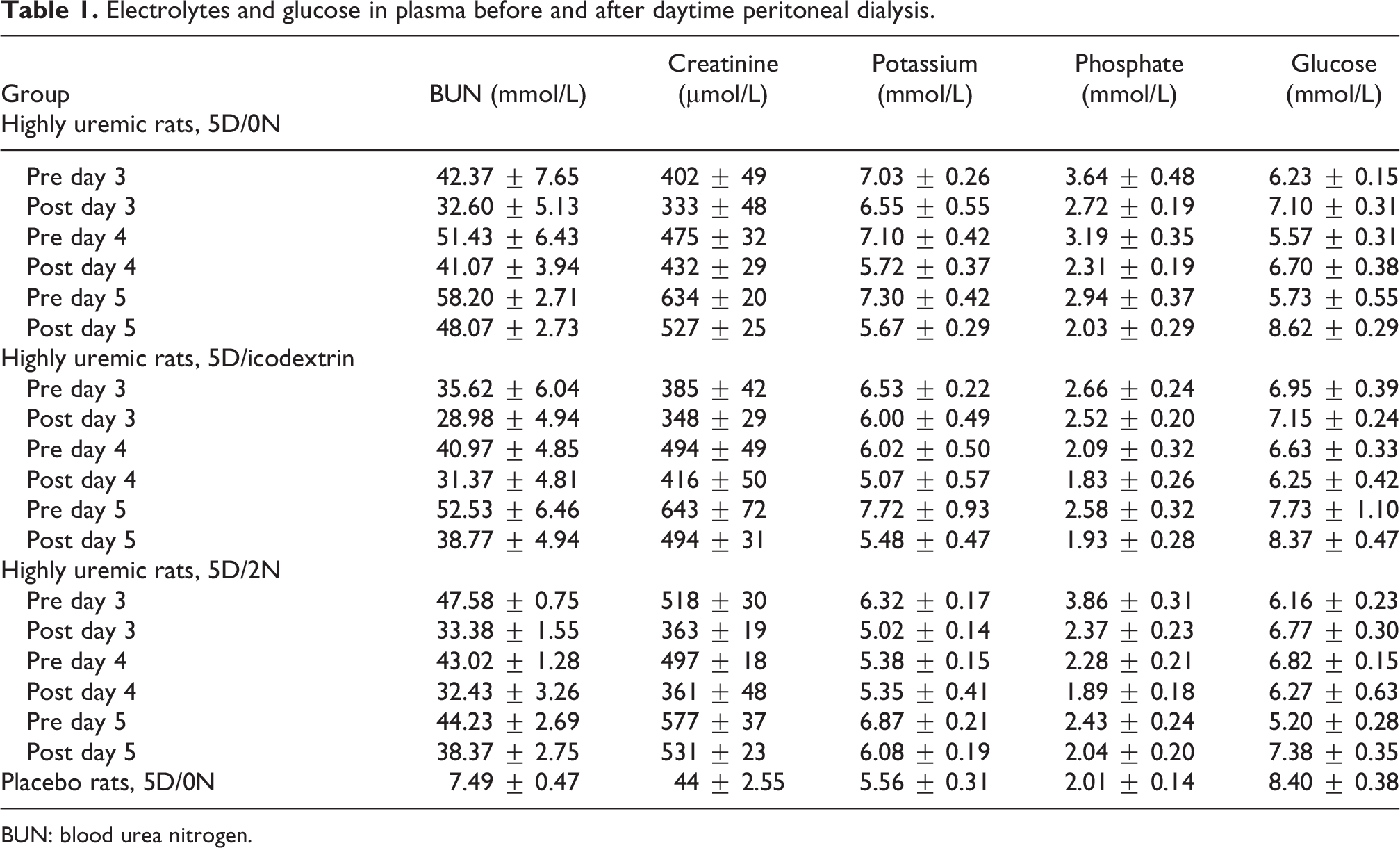
BUN: blood urea nitrogen.
Highly uremic rats on short time PD: Dialysate/plasma ratio, ER, clearance and Kt/V
Results from calculations of dialysis parameters for BUN, creatinine, potassium, phosphate and glucose are presented in Table 2. For BUN and creatinine there was no significant differences between the groups for any of the parameters calculated (D/P, PD ER constant, clearance, Kt/V per day as well as Kt/V per week). The D/P for potassium was significantly higher for the 5D/2N than the 5D/0N, p < 0.05. The PD ER and clearance of potassium was significantly higher for the 5D/2N than the 5D/0N, p < 0.01 and p < 0.001 respectively. The potassium Kt/V per day and per week was significantly higher for the 5D/2N, p < 0.01 compared to the 5D/0N group. There was no significant difference in phosphate concentrations regarding D/P and the PD ER. The clearance of phosphate was significantly higher in the 5D/icodextrin (p < 0.05) and 5D/2N (p < 0.001) compared to the 5D/0N. The same was true for the Kt/V per day and week for phosphate (5D/icodextrin, p < 0.5, 5D/2N p < 0.01). For glucose there was no significant difference between the groups for D/D0. The PD glucose uptake rate was significantly higher for the 5D/2N than the 5D/0N rats, p < 0.01 and the same was true for glucose clearance, p < 0.01. For Kt/V per day and week there was no significant difference between the groups.
D/P, elimination rate, clearance, Kt/V for the three different peritoneal dialysis prescriptions of uremic rats.a
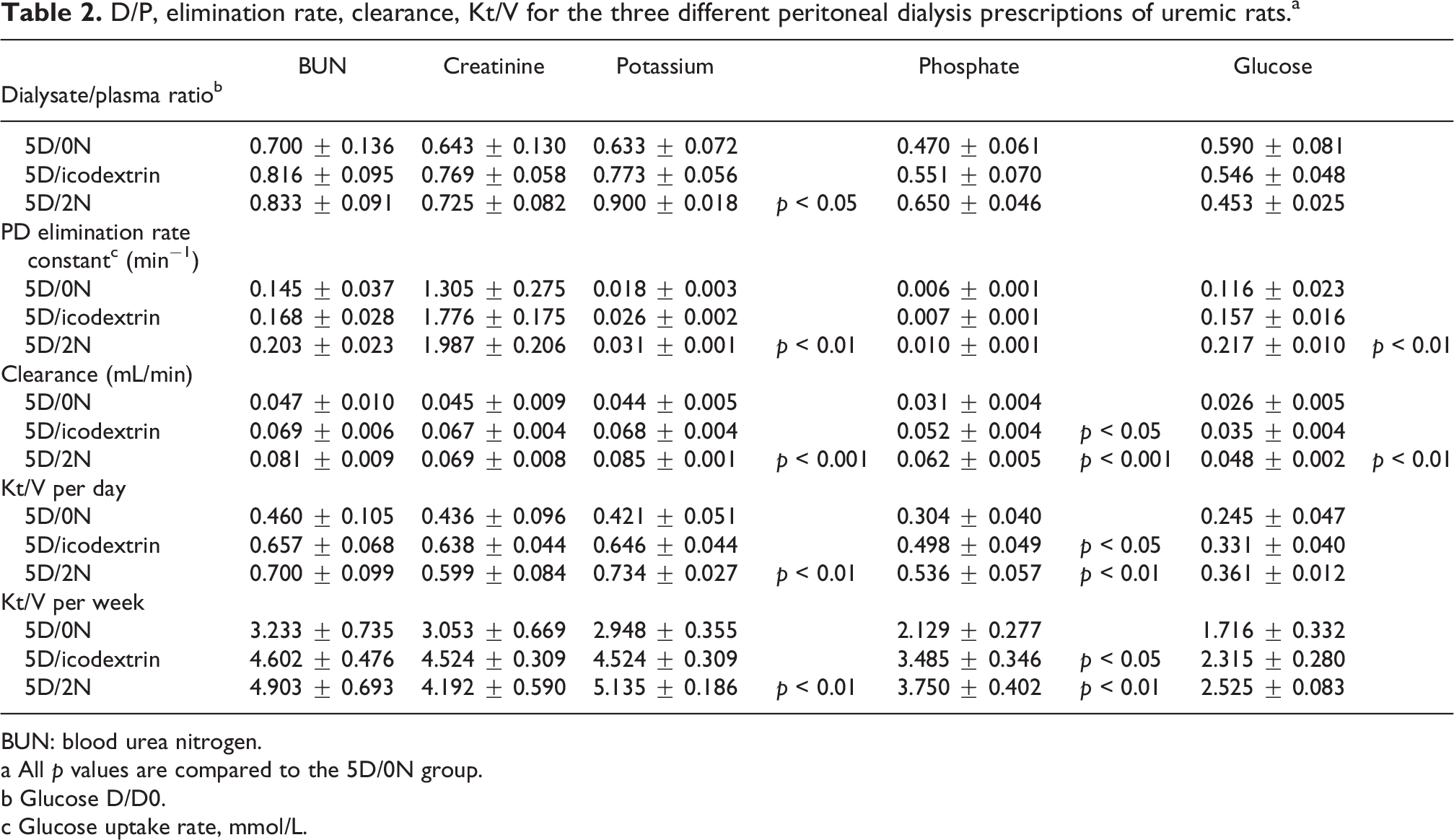
BUN: blood urea nitrogen.
a All p values are compared to the 5D/0N group.
b Glucose D/D0.
c Glucose uptake rate, mmol/L.
The exponential constant was calculated for the D/P ratio (D/D0 for glucose) and used to calculate and to illustrate the D/P concentration ratios during a dwell, see Figure 2(a). D/P ratios for potassium, phosphate, BUN and creatinine increased over time while D/P concentration ratio for glucose decreased over time.
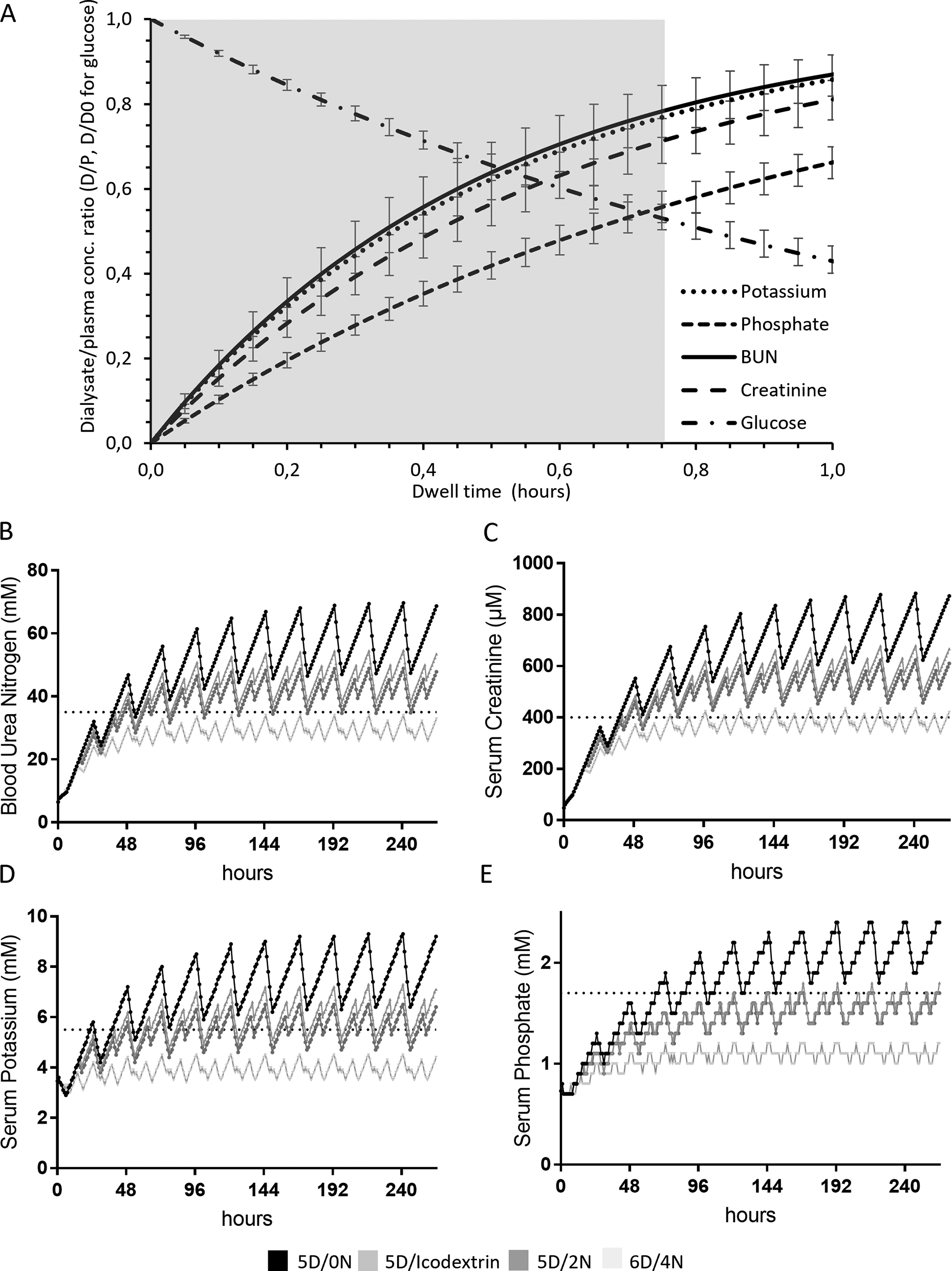
Kinetic modelling. (a) Dialysate/plasma concentration ratios during a dwell with 15 mL of fill volume in 250 g rats. For glucose the D/D0 is illustrated instead of D/P. Grey area indicates dwell period. Error bars represent SEM. (b) to (e) Kinetic modelling of small solutes concentration for four different PD prescriptions over 10 days; five daytime cycles only, five daytime cycles in combination with a long night-time exchange with icodextrin, five daily cycles and two night-time cycles and six daytime cycles and four night-time cycles. The dotted line represents the concentration that the small solutes should be below to maintain an adequate homeostasis while on PD.
Highly uremic rats on short time PD: PD ERs
Based on the experimental data, the PD ERs were assessed for each experimental group (Table 3). The average for all rats were 0.121 ± 0.002 mmol/h for BUN (UER), 1.258 ± 0.0.064 µmol/h for creatinine (CER), 0.018 ± 0.001 mmol/h for potassium (KER) and 0.0053 ± 0.0004 mmol/h for phosphate (PER).
GRs and PD ERs for small solutes.
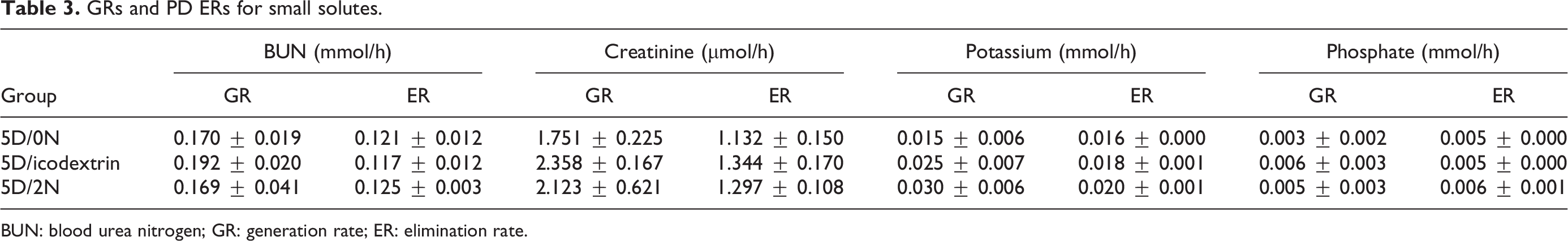
BUN: blood urea nitrogen; GR: generation rate; ER: elimination rate.
Highly uremic rats on short time PD: GRs of metabolites
The GRs for each of the metabolites were calculated using the experimental data and the assumption that faecal and kidney excretion was close to negligible. The GR values for each of the three experimental groups are seen in Table 3. For the kinetic modelling, the average GR values based on all data from all animals were used. Thus, the GRs were 0.177 ± 0.007 mmol/h for BUN (UGR), 2.077 ± 0.177 µmol/h for creatinine (CGR), 0.0233 ± 0.0044 mmol/h for potassium (KGR) and 0.0047 ± 0.0010 mmol/h for phosphate (PGR).
Highly uremic rats on short time PD: Kinetic modelling
The results from the kinetic modelling show that none of the PD prescriptions used in this study are sufficient to achieve a serum BUN below 40 mM (Figure 2(b)) during extended PD of severely uremic rats. Serum creatinine for all groups in the simulation was above the set limit of 400 μM (Figure 2(c)). As for humans, serum creatinine is expected to be elevated in uremic animals on dialysis. For the dialysis to be of acceptable quality in rats, we propose the creatinine levels to be less than 10 times that of control rats, that is, <400 µM. The same results were seen for potassium, although the results with 5D/icodextrin and 5D/2N are below the serum potassium limit of 5.5 mM during parts of the day (Figure 2(d)). The simulation showed that the serum levels of phosphate were nearly adequate for 5D/icodextrin as well as 5D/2N (Figure 2(e)). Simulation showed that a PD prescription of 6D/4N is required to keep the levels of BUN, potassium and phosphate below the set threshold. Creatinine would be under the set threshold for most parts of the day.
Moderately uremic rats on long time PD: Three-week dialysis of uremic rats
To confirm that the PD systems could be used for long-duration dialysis of uremic rats a study was conducted with rats receiving 5 mg/kg of orellanine with an estimated GFR 5–10% of normal. The rats received daytime dialysis for 3 weeks with five daytime cycles per day. The moderately uremic rats were in good condition during the PD period as assessed by visual inspection conducted several times per day. The moderately uremic rats behaved similarly to the healthy rats, were curious and appeared to have a normal appetite. However, the moderately uremic rats gained less in weight than the healthy controls treated with PD. At the start of experiment, control rats weighed 160 ± 4 g, and moderately uremic rats 141 ± 3 g. After 3 weeks, control rats weighed 218 ± 6 g, and moderately uremic rats 177 ± 8 g, showing a significant difference in the weight gain between the groups, 58 g in control rats and 16 g in moderately uremic rats, p < 0.05 The daily ultrafiltration at the end of the 3 weeks was 18.65 ± 3.39 mL/24 h for the control rats and 23.19 ± 1.89 mL/24 h for the moderately uremic rats, with no significant difference between the groups.
Discussion
We have evaluated different PD prescriptions to understand the amount of dialysis required to control selected metabolic parameters in uremic rats. Uremia was induced via chemical nephrectomy using orellanine. 21 By collecting data from the rats treated with different PD prescriptions the effects of PD could be evaluated and modelled. Our study shows that to maintain homeostasis in rats without kidney function, extensive dialysis is required both day and night. We show that a PD prescription of six daytime and four night-time dwells would be sufficient to avoid hyperkalaemia and ensure long-term survival for highly uremic rats. For rats with moderate uremia PD with five daily exchanges was sufficient and uremic rats behaved like rats with intact kidney function during 3 weeks of PD, albeit with reduced growth rate.
Previous experimental studies of PD have either been on rats with normal kidney function, uremic rats after nephrectomy or uremia induced by drugs (adenine or adriamycin). 11 In this study, to avoid surgical trauma, dialysis-dependent kidney disease was introduced by the nephrotoxin orellanine. Orellanine is a compound found in the Cortinarius family of mushrooms that specifically targets the tubular epithelium, where it causes irreversible damage. 21 , 26 – 28 In humans, no organs other than the kidneys have been reported to be affected after accidental intake of orellanine. 23 A study by Nilsson et al. showed that a dose of 5 mg/kg orellanine induced fatal acute kidney failure in the absence of dialysis in rats. 21 Orellanine doses of 7 or 12 mg/kg ip (highly uremic rats) and 5 mg/kg ip (moderately uremic rats) resulted in estimated GFR of <3% and 5–10% of normal, respectively.
Thus, by adjusting the dose of orellanine different levels of uremia can be produced. This makes orellanine an ideal compound for introducing ESKD in rats compared to adriamycin and adenine-induced CKD as these compounds affect organs other than the kidney. 29,30 The surgical methods often used to produce uremia in rats, for example, 5/6 nephrectomy, not only has the drawback of introducing surgical trauma but may produce a variable result. In the 5/6 nephrectomy rat model, GFR is normally reduced to a range of 5–45% of normal 31 –34 and in some cases even less 35 making it a highly variable model for uremia. Recently, Gyuraszova et al. presented serum creatinine and urea data following 5/6 nephrectomy or surgical removal of both kidneys under otherwise identical conditions where the 5/6 nephrectomy rats only had an increase in plasma creatinine concentration with 48% and plasma urea concentration was increased twofold. 17 Therefore the 5/6 nephrectomy models seem to be an unreliable choice for the production of a stable uremic state appropriate for PD studies in uremic rats.
Our data show that for a GFR of 5–10% of normal, daytime PD with five exchanges of 15 mL seems adequate, as seen in the study of moderately uremic rats. The three different PD prescriptions tested in the short time study of highly uremic rats, five daytime cycles (5D/0N), five daytime cycles in combination with a long PD exchange with icodextrin over night (5D/icodextrin) and five daytime cycles combined with two cycles during the night (5D/2N), were not sufficient to keep the concentrations of small solutes low. The clearance, Kt/V and D/P values of BUN and creatinine in all groups indicate that dialysis was effective. The best effect was achieved with the 5D/2N prescription. The D/P and PD ER, clearance and Kt/V for potassium were significantly higher for the 5D/2N than the 5D/0N group indicating that this prescription was the best, even though not enough to maintain potassium levels within the normal range.
Pedersen et al. developed a bilateral nephrectomy PD model where the rats were continuously dialysed for 48 h with 12 exchanges per day. 36 Each dwell was 30 min with 10 mL of infusion. The potassium level in their study was around 3.6 mmol/L at the end of the experiment. In our study of highly uremic rats on short time PD, the most intense dialysis prescription (5D/2N) resulted in a potassium level of around 7.4 mmol/L at the end of the experiment. The kinetic simulations showed that a dialysis prescription of 6D/4N would be needed to keep the potassium within an acceptable range. Fujii et al. presented data on the effect of PD in rats after bilateral nephrectomy. In this study, PD with 40 mL/h for 6 h/day kept the rats alive for 54 h 18 which is consistent with our data on the effect of long-term dialysis on plasma urea and creatinine concentrations. However, our kinetic modelling suggests the development of rapidly developing hyperkalaemia unless more intense dialysis prescriptions are used. For totally anuric rats, that is, GFR = 0, we speculate that exchanges every second hour (12 PD dwells per 24 h) would be required to maintain normal homeostasis and allow for long-term survival on dialysis. Note, however, that for long-term dialysis using such intense PD, there is a possibility that potassium might need to be added to the dialysis fluid to avoid hypokalaemia.
In conclusion, a single dose of orellanine (7 or 12 mg/kg ip) can be used to induce severe uremia in rats while avoiding the surgical trauma of bilateral nephrectomy, and without the limitations of 5/6 nephrectomy. Our PD system can be used for long-term studies of PD on uremic dialysis-dependent rats that mimic the human setting.
Supplemental material
Supplemental Material, sj-docx-1-ptd-10.1177_08968608231191054 - Evaluation of peritoneal dialysis prescriptions in uremic rats
Supplemental Material, sj-docx-1-ptd-10.1177_08968608231191054 for Evaluation of peritoneal dialysis prescriptions in uremic rats by Esther Nuñez-Durán, Jessica Westlund, Deman Najar and Kerstin Ebefors in Peritoneal Dialysis International
Footnotes
Authors’ note
END and JW contributed equally to this work.
Acknowledgements
The authors are grateful for the support by Prof Börje Haraldsson in developing the dialysis system, and for his expertise in kinetic modelling and to Prof Gerald F DiBona for careful editing of the manuscript.
Author contributions
END, JW and DN performed the experiments. KE analysed the data and wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from the Gothenburg Ethical Board for Animal Experiments: approval number 144-12, 44-15 and 1451 and performed according to the NIH guidelines.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Swedish research council, grant number 09898, The Swedish kidney foundation, Inga and Arne Lundberg research foundation.
Informed consent
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.