Abstract
Objective:
Peritoneal dialysis (PD)-related peritonitis is independently associated with low serum 25-hydroxy vitamin D [25(OH)D] levels. Our objective is to examine the feasibility of conducting a large, randomised controlled trial to determine the effects of vitamin D supplementation on the risk of PD-related peritonitis.
Design:
Pilot, prospective, open-label randomised controlled trial.
Setting:
Peking University First Hospital, China.
Participants:
Patients receiving PD who had recovered from a recent episode of peritonitis between 30 September 2017 and 28 May 2020.
Interventions:
Oral natural vitamin D supplementation (2000 IU per day) versus no vitamin D supplementation for 12 months.
Primary and secondary outcome measures:
Primary outcomes were feasibility (recruitment success, retention, adherence, safety) and fidelity (change in serum 25(OH)D level during follow-up) for a large, randomised controlled trial in the future to determine the effects of vitamin D on PD-related peritonitis. Secondary outcomes were time to peritonitis occurrence and outcome of subsequent peritonitis.
Results:
Overall, 60 among 151 patients were recruited (recruitment rate was 39.7%, 95% CI 31.9–47.5%, recruitment rate among eligible patients was 61.9%, 95% CI 52.2–71.5%). Retention and adherence rates were 100.0% (95% CI 100.0–100.0%) and 81.5% (95% CI 66.8–96.1%), respectively. During follow-up, serum 25(OH)D levels increased in the vitamin D (VD) group (from 19.25 ± 10.11 nmol/L to 60.27 ± 23.29 nmol/L after 6 months, p < 0.001, n = 31), and remained higher (p < 0.001) than those in the control group (n = 29). No differences were observed between the two groups with respect to time to subsequent peritonitis (hazard ratio 0.85, 95% CI 0.33–2.17) or any of the peritonitis outcomes. Adverse events were uncommon.
Conclusions:
A randomised controlled trial of the effect of vitamin D supplementation on peritonitis occurrence in patients receiving PD is feasible, safe and results in adequate serum 25(OH)D levels.
This is a visual representation of the abstract.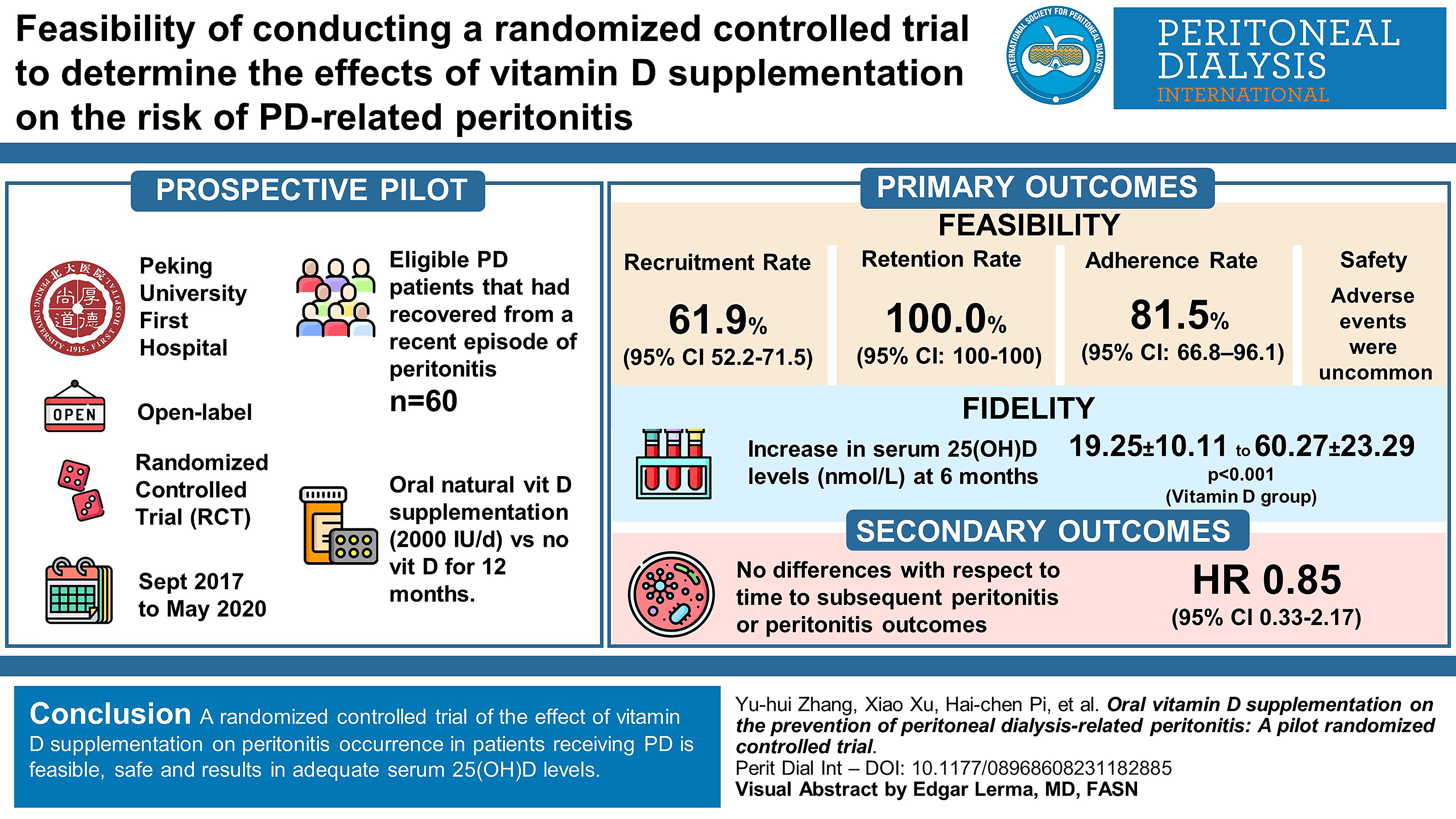
Keywords
Introduction
Peritoneal dialysis (PD)-related peritonitis is the most common complication among patients on PD. It is also considered as the top, most critical research priority for patients, caregivers and clinicians. 1,2 According to data from our cohort and the Peritoneal Dialysis Outcomes and Practice Patterns Study (PDOPPS), almost 30–40% of patients on PD experience one or more episodes of peritonitis. 3,4 Patients with an initial episode of peritonitis are at increased risk of a subsequent peritonitis for up to 8 months. 5 Peritonitis is also associated with increased risks of hospitalisation, transition to haemodialysis (HD), cardiovascular events and death. 6 –8 Therefore, interventional strategies for the prevention and management of peritonitis are urgently needed.
Recent investigations into the potential mechanisms underpinning PD-related peritonitis have focused on immune dysfunction, inflammation and malnutrition. 9 –12 However, studies of the effects of interventions targeting these mechanisms on peritonitis risk are limited.
Vitamin D exerts regulatory effects on both the innate and adaptive immune systems and may mitigate infection risk. 13 In the general population, vitamin D deficiency is associated with many infectious diseases, such as respiratory infection, 14,15 tuberculosis 16 and urinary tract infection. 17 Interventional studies have shown that supplementation of vitamin D2/D3 raises serum 25-hydroxy vitamin D (25(OH)D) levels efficiently and safely, 18 as well as reducing the risk of acute respiratory infection 19 and urinary infection, 20 while supplementation of active vitamin D did not effectively increase serum 25(OH)D levels. 21 Of note, vitamin D deficiency is highly prevalent among dialysis patients. 22 Using a serum level of 25(OH)D less than 75 nmol/L (30 ng/mL) as a diagnostic criterion, the prevalence of vitamin D deficiency among PD patients is 86–100%. 22 Moreover, in an observational cohort study of 346 patients on PD followed for more than 2 years at our centre, serum 25(OH)D levels were significantly and inversely associated with peritonitis risk. 12 However, prospective interventional studies evaluating a possible preventive effect of vitamin D against peritonitis have been limited to date.
We conducted a pilot, randomised controlled trial examining both the feasibility of conducting a trial of vitamin D supplementation to prevent PD-related peritonitis and the fidelity of the intervention to help inform the design and conduct of a larger randomised controlled trial that is adequately powered for the outcome of peritonitis.
Materials and methods
Trial design
This investigator-initiated, pilot, single-centre, prospective, parallel-arm, open-label, randomised controlled trial was conducted at the PD Center of Peking University First Hospital. The protocol for the trial has been published previously. 23 The study was approved by the Peking University First Hospital Research Ethics Committee (Approval number 2016[1149]) and registered at Clinicaltrials.gov (NCT03264625 24 ). The study was conducted and reported according to the Consolidated Standards of Reporting Trials (CONSORT statement). 25
Participants
All adult patients who had recovered from an episode of PD-related peritonitis between 30 September 2017 and 28 May 2020 were screened for eligibility at 1 month after the event. The inclusion criteria were age at least 18 years, undergoing PD for ≥1 month and clinically stable, weekly Kt/V ≥ 1.5 without clinical uremic symptoms and serum 25(OH)D < 75 nmol/L (30 ng/mL). Patients were excluded if they had any of the following: received vitamin D2/D3 or drugs known to alter serum 25(OH)D levels during the previous 12 months; a history of allergic reaction to cholecalciferol; current or past malignant disease, active hepatitis or hepatic failure, acute systemic infection, active autoimmune diseases, severe digestive malabsorption or eating disorder, or human immunodeficiency virus infection or acquired immune deficiency syndrome (HIV/AIDS); a high probability (assessed by the recruiting physician) of receiving a kidney transplant or transition to HD or dropout due to socio-economic causes within 6 months; or women who were pregnant or lactating.
Peritonitis was diagnosed when at least two of the following conditions were present: (1) clinical symptoms of peritonitis, such as abdominal pain, cloudy dialysis effluent; (2) presence of white blood cells (>100/uL) in dialysis effluent after a dwell time of at least 2 hours, with >50% polymorphonuclear leucocytes and (3) positive culture of dialysis effluent. 6
The sample size of our study was 60, which was in line with the median sample sizes for pilot and feasibility studies found in an audit of such trials registered with the UK Clinical Research Network Database (36 per arm for dichotomous end points and 30 per arm for continuous end points). 26
Randomisation and intervention
All consenting patients were registered in the trial and randomised 1:1 to either vitamin D supplementation (intervention group) or no vitamin D supplementation (control group) by a computer-generated random number list in sequence. An independent medical staff member was responsible for participant enrolment and allocation assignment.
Participants in the VD group received additional vitamin D (Liquid Natural Vitamin D3, Cholecalciferol) in a dose of 2000 IU orally per day for 12 months following randomisation. Participants in the control group did not receive any study drug or any form of vitamin D2/D3 or drugs known to alter serum 25(OH)D levels during the study period. The medication list for all participants was verified regularly by our staff. For both the intervention and control groups, all dialysis and other medication prescriptions were in accordance with routine clinical care and International Society for Peritoneal Dialysis (ISPD) guideline recommendations.
Due to the nature of the medication intervention, neither participants nor researchers were blinded. However, both the primary and secondary outcomes were evaluated by independent medical staff who were blinded to participants’ allocations. In addition, laboratory parameters for all participants were measured by laboratory staff who were also blinded to treatment assignment.
Baseline evaluation and follow-up
For all eligible and consenting patients, information regarding demographic data (age, gender, body mass index, education, marital status, occupation, income, health insurance), dialysis duration, primary disease, co-morbidities, current medication and history of PD-related peritonitis were recorded. Baseline biochemistry indices (including full blood, serum creatinine, albumin, lipids, electrolytes, parathyroid hormone (PTH) and serum 25(OH)D levels), dialysis small-solute clearance (including urea clearance index [Kt/V] and creatinine clearance rate) were evaluated. After randomisation, the assigned treatment was recorded in detail.
All participants were followed for 12 months, with clinic visits every month. During the follow-up period, clinical information including self-reported symptoms and physical examination were gathered. Biochemistry indices (including full blood count, serum creatinine, albumin, alanine transaminase, lipids, electrolytes, PTH and serum 25(OH)D levels) and dialysis small-solute clearance were evaluated every 3 months. Participants were reminded of vitamin D use monthly through telephone calls and evaluated for compliance by weighing residual liquid vitamin D every month. All outcomes and adverse events were recorded.
Primary and secondary outcomes
The primary outcomes were feasibility (recruitment rate, retention rate of participants for 12 months, adherence rate and safety) and fidelity (change in serum 25(OH)D level between baseline and 12 months). Recruitment rate was determined as the proportion of patients recruited into the trial among all potentially eligible patients during the recruitment period. Retention rate was calculated as the percentage of patients who completed the trial among patients recruited. Adherence rate was calculated as the percentage of patients who were adherent to the dosing regimen among patients in the VD group. Fidelity was measured by the difference of change in serum 25(OH)D level over time between the VD and control groups. The detailed definitions of outcomes are listed in our previously published protocol paper. 23
Secondary outcomes included time to peritonitis occurrence, recovery of peritonitis, peritonitis-related transition to HD and peritonitis-related death (defined as death within 30 days of peritonitis onset). Death not associated with peritonitis, transition to HD not associated with peritonitis and receiving a kidney transplant were recorded as competing outcomes. For the assessment of safety, both severe and nonsevere pre-specified adverse events during the study course, including allergic reaction to vitamin D, hypercalcaemia and hyperphosphataemia, were recorded. The detailed definitions of outcomes are listed in our previously published protocol paper. 23
Statistical analysis
Statistical analysis was conducted using SPSS for Windows (version 24.0, IBM, Armonk, New York, USA) and SAS software (version 9.4, SAS Institute, Cary, North Carolina, USA). Continuous normally distributed variables were expressed as mean ± standard deviation, continuous variables that were not normally distributed were expressed as median with interquartile range and categorical variables were expressed as numbers and percentages. Student’s t test, Mann–Whitney U test and the chi-square test were used to compare differences in baseline characteristics between intervention and control groups. Log rank test was used to compare causes of withdrawals from PD between the two groups.
Recruitment rate, retention rate and adherence rate were reported as percentages and associated 95% CI. Adverse events were recorded as event rate per patient-year. Analysis of variance for repeated measurements was used to analyse change in serum 25(OH)D levels for both groups and difference in change between the two groups (The analysis of variance for repeated measurements is more suitable here than linear mixed-effects regression models mentioned in the published protocol.). Competing risks Cox regression models and Kaplan Meier survival analysis were used to compare time to subsequent peritonitis between the two groups. Competing risks Cox regression models were also used to compare risk of subsequent peritonitis by different pathogenic bacteria between the two groups. Logistic regression models were applied to compare the short-term outcome of subsequent peritonitis. All analyses were ‘intention-to-treat’ analysis.
For all statistical analyses, the level of significance was set at 0.05.
Patient and public involvement
Patients or the public were not involved in the design, or conduct, or reporting, or dissemination plans of our research.
Results
Between 30 September 2017 and 28 May 2020, 151 patients with episodes of peritonitis were screened, 97 patients were eligible, of whom 60 were enrolled and randomised to the VD group (n = 31) or the control group (n = 29). Figure 1 shows the flow diagram of the trial. Patients in the VD and control groups were well matched for all baseline characteristics (Table 1). The mean duration of follow-up was 11.12 ± 2.15 months in the VD group, 11.55 ± 1.72 months in the control group. By the end of the study, 6 (19.4%) patients in the VD group discontinued PD due to transition to HD (n = 3), kidney transplantation (n = 1) or death (n = 2). In the control group, 2 (6.9%) patients discontinued PD due to transition to HD (n = 1) or kidney transplantation (n = 1) (Table S1).
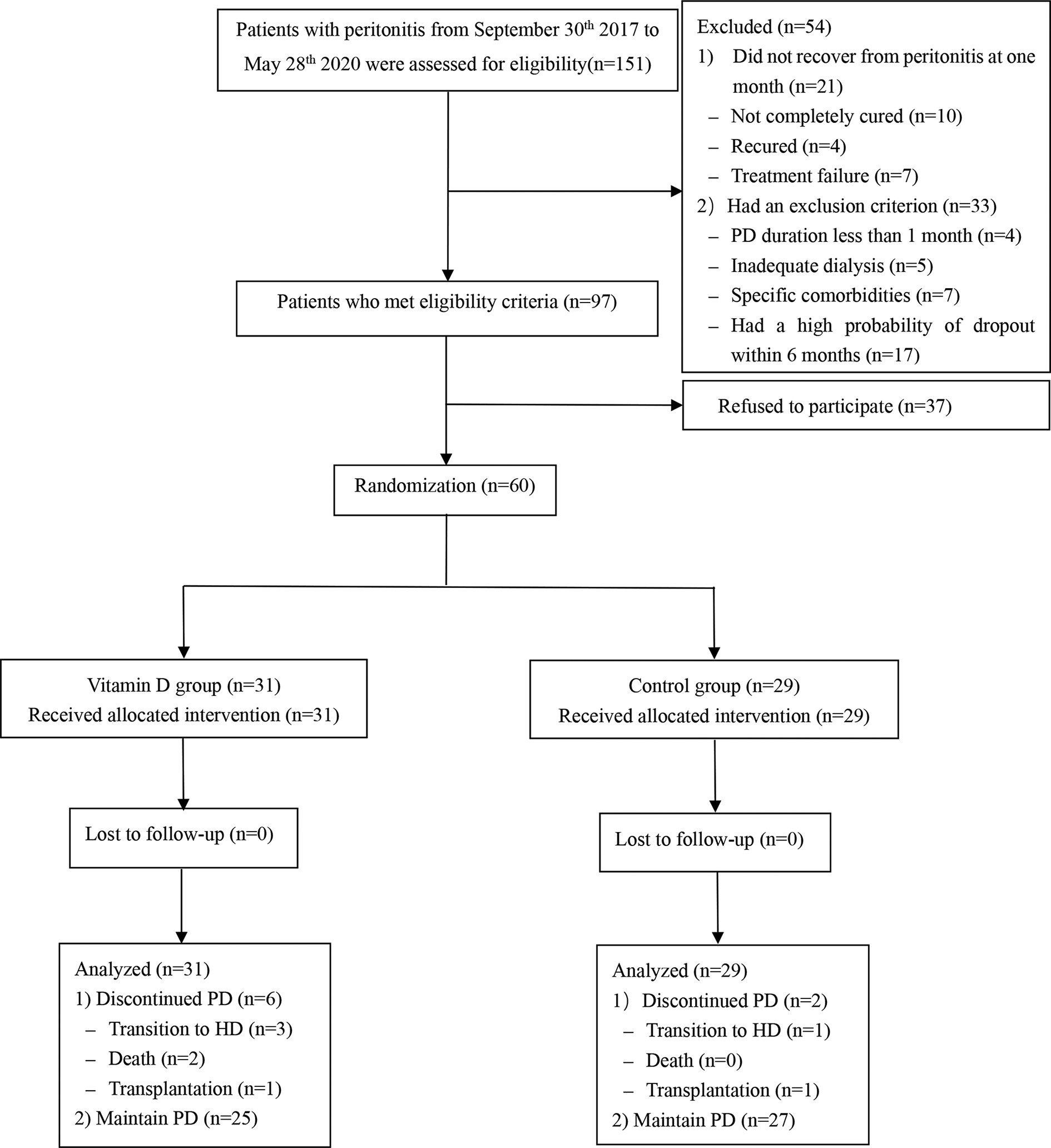
Flow diagram.
Baseline characteristics of the vitamin D and control groups.

25(OH)D: 25-hydroxy vitamin D; PTH: parathyroid hormone; Kt/V: urea clearance per week; Ccr: creatinine clearance per week.
Primary outcomesc
Recruitment
Among 151 patients with peritonitis, 97 met eligibility criteria, of whom 60 were recruited into the trial during the study period, with the remainder declining to participate. Thus, recruitment rate was 39.7% (95% CI 31.9–47.5%) among all potentially eligible patients, while the proportion recruited among eligible patients was 61.9% (95% CI 52.2–71.5%). Both of these rates compared favourably to the recruitment target of 40% (Table 2). Patients recruited in the trial had shorter PD duration and lower C-reactive protein levels than patients who refused to participate (Table S2).
Feasibility parameters.

a Recruitment was determined as the proportion of patients recruited into the trial among all potentially eligible patients during the recruitment period.
b Retention rate was calculated as the percentage of patients who completed the trial among patients recruited.
c Adherence rate was calculated as the percentage of patients who were adherent to the dosing regimen among patients in the vitamin D group.
Retention
There was no loss to follow-up in either group. Thus, the overall retention rate was 100.0% (95% CI 100.0–100.0%), which was above the target of 80% (Table 2).
Adherence rate
The overall adherence rate of the VD group was 81.5% (95% CI 66.8–96.1%). Four patients who were unable to take liquid vitamin D to the hospital for weighing during the COVID-19 pandemic were not included in the calculation of overall adherence rate. The adherence rates during the follow-up periods of 0–3 months, 3–6 months, 6–9 months and 9–12 months were 63.0%, 78.3%, 94.1% and 93.3%, respectively (Figure S1). The overall adherence rate (81.5%) was higher than the target level (70%) (Table 2).
Fidelity
During follow-up, serum 25(OH)D levels gradually increased in the VD group (p < 0.001) and remained significantly higher (p < 0.001) than those in the control group, which did not significantly change compared to the baseline during the follow-up period (p = 0.49), as shown in Figure S2 (Table 3).
Comparison of biochemical parameters between the VD group and the control group.
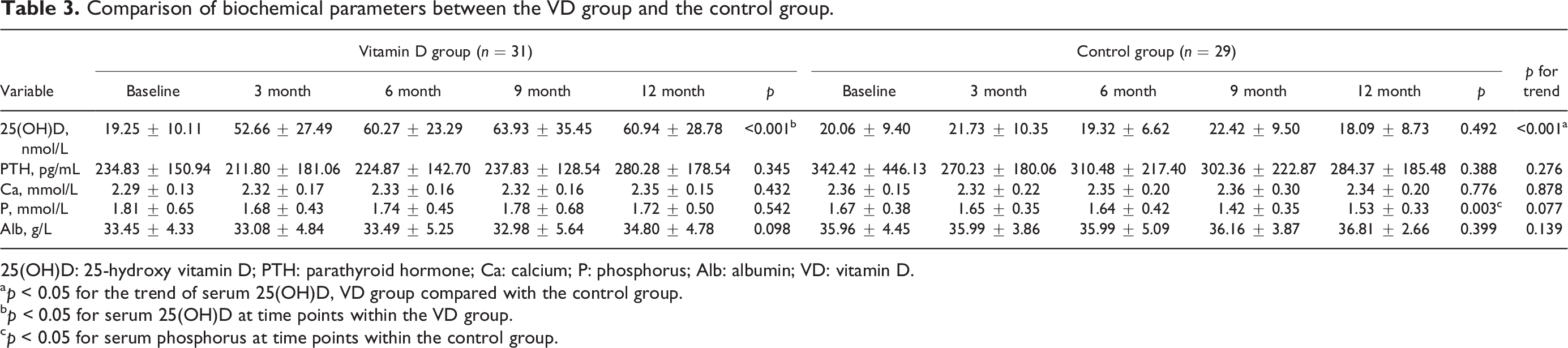
25(OH)D: 25-hydroxy vitamin D; PTH: parathyroid hormone; Ca: calcium; P: phosphorus; Alb: albumin; VD: vitamin D.
a p < 0.05 for the trend of serum 25(OH)D, VD group compared with the control group.
b p < 0.05 for serum 25(OH)D at time points within the VD group.
c p < 0.05 for serum phosphorus at time points within the control group.
Peritonitis occurrence
As shown in Figure S3, time to subsequent peritonitis was comparable between the two groups (hazard ratio [HR] 0.85, 95% CI 0.33–2.17). There were also no significant differences observed between the VD and control groups with respect to risk of first subsequent Gram-positive, Gram-negative and culture-negative peritonitis (Table 4). The rate for all subsequent peritonitis was 0.56/patient-year in the VD group and 0.82/patient-year in the control group.
Comparison of occurrence and outcomes of first peritonitis episodes between the two groups.
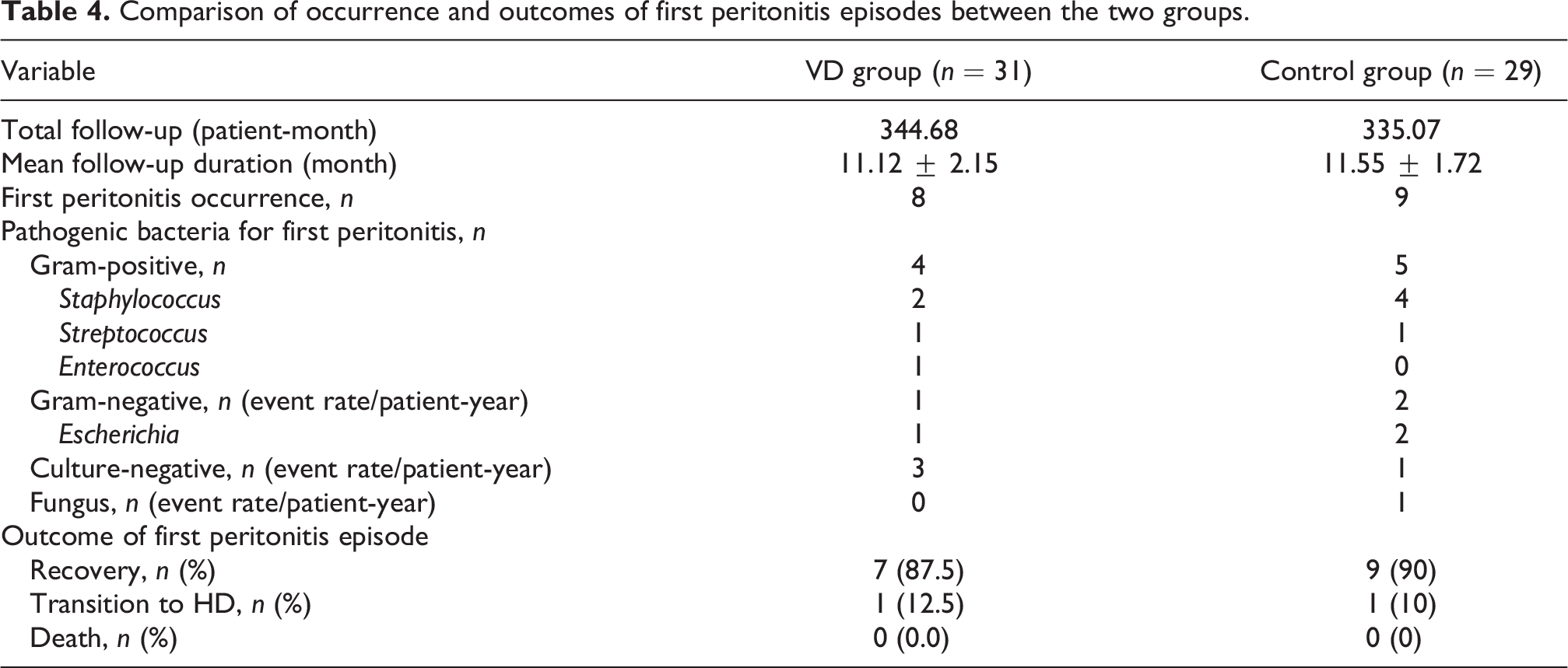
Peritonitis outcomes
No differences were observed between the two groups with respect to peritonitis recovery, peritonitis-related transition to HD or peritonitis-related death (Table 4).
Biochemical parameters
The serum level of phosphorus was stable in the VD group but decreased slightly in the control group (p = 0.003). There were no significant changes in serum levels of PTH, calcium or albumin in both groups during the follow-up (Table 3).
Safety assessment
No cases of pre-specified drug-related adverse event, including allergic reaction to vitamin D, hypercalcaemia and hyperphosphataemia, were observed during the study course. One patient had elevated transaminases 2 months after intervention. However, after discontinuation of vitamin D, the patient’s transaminase level was still elevated. Another patient experienced eczema onset during the intervention, which was cured after topical medication. No adverse event was considered to be associated with vitamin D (Table 2).
Discussion
The present study demonstrated that vitamin D supplementation significantly increased serum 25(OH)D levels and was safe, and that conducting a trial on the effects of oral vitamin D administration on peritonitis occurrence is feasible, thereby both supporting and informing the development of a future, large-scale randomised controlled trial.
The present study demonstrated that performing a vitamin D supplementation trial is feasible in terms of recruitment, retention and treatment adherence. The recruitment rate among all potentially eligible patients was 39.7% (95% CI 31.9–47.5%), which was comparable with those of previous randomised controlled trials regarding PD-related peritonitis. 27 –29 The definition of recruitment rate in our study was relatively strict. If the rate was alternatively calculated as the proportion of recruited patients among eligible patients, the rate was 61.9% (95% CI 52.2–71.5%). Recruiting patients who had recovered from a peritonitis episode provided an enriched cohort of individuals at high risk for subsequent peritonitis events, which will appreciably assist with powering a larger trial for a peritonitis primary outcome. Using the observed peritonitis rate of 0.82 episodes/patient-year in the control group in the present study, an interventional trial of the effect of vitamin D supplementation on peritonitis rate will need a sample size of 1173 overall to have 90% power to detect a 20% reduction in peritonitis rate in the intervention group, assuming a recruitment period of 2 years from 25 PD centres with 500 PD patients each, a follow-up period of 1 year and an alpha level of 0.05.
The adherence rate in the VD group was 77.8%, which was comparable with those of previous interventional studies using vitamin D supplementation as an intervention (61–96%). 30 –33 Our rigorous study procedure, including monthly reminders on vitamin D intake by our staff via telephone, helped to encourage and sustain treatment adherence. By assessing the adherence rate at different stages during the follow-up, we found that adherence increased as the time on the intervention increased; patients tended to miss doses during the initial period but gradually habituated to taking vitamin D regularly afterwards with support from staff.
In terms of fidelity, the present study confirmed that oral supplementation of vitamin D 2000 IU per day could significantly raise serum levels of 25(OH)D of PD patients. After continuous supplementation for 6–9 months, serum levels were close to the target for vitamin D sufficiency 34 and remained stable without significant changes in serum calcium and phosphorous levels. Our dosing strategy was formulated based on experience from previous research. A systemic review indicated that taking vitamin D in dosages of at least 2000 IU per day was necessary to ameliorate vitamin D deficiency in patients with non-dialysis CKD. 35 A randomised controlled trial among maintenance HD patients showed that a weekly dose of 10,333 IU cholecalciferol for 15 weeks raised vitamin D levels from 33.3 nmol/L (13.3ng/mL) to 59.0 nmol/L (23.6ng/mL). 36 Considering safety, efficacy and feasibility, the dose of vitamin D was set at 2000 IU per day. The mean serum level of 25(OH)D was increased from 19.25 nmol/L to 60.94 nmol/L in the present study, which seems more effective compared with another trial among patients receiving PD, in which oral supplementation of cholecalciferol 50,000 U per week for 8 weeks followed by 10,000 U per week for 44 weeks increased serum 25(OH)D levels by a mean of 17.2 ± 30.8 nmol/L. 37 However, it should be noted that 62.5–73.3% of patients in the VD group in our study still had serum 25(OH)D levels lower than 75 nmol/L at different follow-up time points. Future studies investigating individualised dose titration strategies to achieve the target level are warranted.
Previous data from our centre have shown that lower serum 25(OH)D level is independently associated with an increased risk of PD-related peritonitis. 12 During a follow-up period of more than 2 years, patients with middle (13.51–19.13 nmol/L) and higher tertiles of serum 25(OH)D (≥19.13 nmol/L) had significantly decreased peritonitis risk compared with those in the lowest tertile group (≤13.51 nmol/L), with HRs of 0.54 (95% CI 0.31–0.94) and 0.39 (95% CI 0.20–0.75), respectively. 12 The mechanism of the preventive effect of vitamin D on peritonitis lies in its role in strengthening the innate immune system via multiple pathways. 13 Serum 25(OH)D could be transformed to 1,25(OH)D and combine with vitamin D receptors on the surface of immune cells, promoting maturation of dendritic cells and macrophages, inducing the production of antimicrobial peptides and reducing the release of inflammatory factors. Increasing serum level of 25(OH)D could enhance the anti-infectious ability of the human body and limit the potential inflammatory damage. 13 Another study from Austria also indicated that patients receiving oral active vitamin D had a 57% decreased risk of peritonitis during a 10-year follow-up period. 38 However, oral active vitamin D did not effectively increase serum 25 (OH)D levels. 21 Moreover, those with higher intact PTH values who require active vitamin D therapy possibly represent individuals with good nutritional condition. 39 Therefore, these observational findings did not prove a cause–effect relationship between vitamin D and the prevention of peritonitis. The ISPD guidelines in 2016 listed vitamin D deficiency as one of the modifiable risk factors for PD-related peritonitis and recommended that interventional trials be performed to evaluate the effect of vitamin D supplementation on peritonitis. 40 Due to limited sample size, the present study was not adequately powered for the secondary outcomes, a larger study exploring the effects of oral vitamin D supplementation on PD-related peritonitis is warranted. Our pilot study lays the groundwork for such a definitive trial to be appropriately designed and conducted.
Our study has several strengths. First, as an innovative study, we explored the effects of vitamin D supplementation on PD-related peritonitis through an interventional design, which provided a foundation for future definitive studies and is of important clinical significance. Second, we recruited patients who had just recovered from peritonitis, providing an enriched cohort of patients at risk of peritonitis. Third, our rigorous study procedure ensured good retention and adherence rates, thereby leading to a significant increase in serum 25(OH)D levels among participants. Moreover, the liquid vitamin D formulation offered convenience for participants and facilitated their treatment adherence.
However, there were also several limitations in this study. Due to limited sample size and short observation period, we could not draw definitive conclusions regarding the effects of vitamin D supplementation on secondary outcomes, such as peritonitis occurrence. However, as for the primary outcome, the sample size of our study is in line with the median sample sizes for pilot and feasibility studies found in an audit of such trials registered with the UK Clinical Research Network Database (36 for dichotomous end points and 30 for continuous end points). 26 Second, the open-label nature of the trial may contribute to potential bias by making patients in the two groups open to different co-interventions that may impact risk of peritonitis. In addition, we did not use random envelopes to conceal allocation, such that the possibility of selection bias cannot be excluded. Finally, given the difficulty in producing liquid placebo with a thick taste, we did not administer a placebo to participants in the control group, leading to the possibilities of detection and attrition biases.
In conclusion, the present study achieved target levels for feasibility and fidelity parameters, thereby demonstrating that a future large-scale, powered randomised controlled trial of the effect of vitamin D supplementation on peritonitis is feasible. As the sample size and follow-up period of our pilot study were insufficient to meaningfully analyse the secondary outcome of peritonitis, a large, adequately powered study, whose design is informed by our pilot trial, is warranted to further explore this issue in the future.
Supplemental material
Supplemental Material, sj-docx-1-ptd-10.1177_08968608231182885 - Oral vitamin D supplementation on the prevention of peritoneal dialysis-related peritonitis: A pilot randomised controlled trial
Supplemental Material, sj-docx-1-ptd-10.1177_08968608231182885 for Oral vitamin D supplementation on the prevention of peritoneal dialysis-related peritonitis: A pilot randomised controlled trial by Yu-Hui Zhang, Xiao Xu, Hai-Chen Pi, Zhi-Kai Yang, David W Johnson and Jie Dong in Peritoneal Dialysis International
Footnotes
Authors’ note
Y-HZ and XX contributed equally to this work.
Acknowledgements
The authors express their deep appreciation to the patients, doctors and nursing staff of the PD Center of Peking University First Hospital. Their special thanks also go to the three cooperative hospitals that referred patients with PD-related peritonitis to our centre, including Beijing Chao-yang Hospital, Beijing Traditional Chinese Medicine Hospital and Beijing Haidian Hospital.
Author contributions
Research idea and study design: JD, Y-HZ, H-CP, DWJ; data acquisition: Y-HZ, XX, Z-KY, JD; supervision or mentorship: JD. Each author contributed important intellectual content during manuscript drafting or revision and accepts accountability for the overall work by ensuring that questions on the accuracy or integrity of any portion of the work are appropriately investigated and resolved.
Data availability statement
The data supporting the findings of this study are available on the request to the corresponding author.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: David Johnson has received consultancy fees, research grants, speaker’s honoraria and travel sponsorships from Baxter Healthcare and Fresenius Medical Care, consultancy fees from Astra Zeneca, Bayer and AWAK, speaker’s honoraria from ONO and BI & Lilly and travel sponsorships from Ono and Amgen. He is a current recipient of an Australian National Health and Medical Research Council Leadership Investigator Grant. The above organisations had no role in the design or conduction of this study. The remaining authors declare that they have no relevant financial interests.
Ethical approval
The study was reviewed and approved by the Peking University First Hospital Research Ethics Committee, approval number 2016[1149].
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National High Level Hospital Clinical Research Funding (Scientific and Technological Achievements Transformation Incubation Guidance Fund Project of Peking University First Hospital 2022CR82) and National High Level Hospital Clinical Research Funding (High Quality Clinical Research Project of Peking University First Hospital 2022CX09).
Informed consent
Yes.
Trial registration
Registered at Clinicaltrials.gov (NCT03264625).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
