Abstract
The pattern of chronic kidney disease–mineral bone disease has changed following the increase in elderly patients receiving dialysis, with escalating likelihood of osteoporosis, with associated increased fracture risk and mortality. Thus, we wished to determine the prevalence of osteoporosis in our peritoneal dialysis (PD) cohort. Lumbar spine (LS) and femoral neck (FN) bone mineral density (BMD) measured by dual-energy X-ray absorptiometry (DXA), and low BMD (osteoporosis) and reduced BMD (osteopenia) defined according to the World Health Organisation T scores. DXA scans from 734 patients, 57.2% male, mean age 61.0 ±16.0 years, 34.6% diabetic, with a median 8.0 (2–24) months of treatment with PD were reviewed. Fewer patients had normal BMD at the FN vs. LS (24.1 vs. 55%), and more classified as reduced (FN 55.9 vs. 34% LS) and low BMD (FN 17 vs. 11% LS), χ 2 112.2, p < 0.001. Agreement between FN and LS T scores was r = 0.36 with Spearman rank correlation, and 0.34 by Kendall’s tau b, Cohen’s kappa score 0.17, and Bland–Altman bias −0.37 (95% limits of agreement −1.8 to 1.08). The prevalence of reduced and low BMD was much greater when using FN T scores compared to LS, with LS T scores a mean of 0.34 higher than FN. DXA scanning at the LS can be affected by overlying vascular calcification and electron densities in the bowel, and as such we suggest that FN T scores are used to report BMD in PD patients.
Introduction
Chronic kidney disease (CKD) patients are at risk of bone mineral disease (CKD-BMD). 1 –3 Dual-energy X-ray absorptiometry (DXA) is most widely used to assess bone mineral density (BMD), due to its high accuracy, short scan duration and low radiation dosage. 4 The World Health Organisation (WHO) defined osteopenia and osteoporosis according to DXA measurements. 5 Osteoporotic and CKD patients have an increased risk of bone fracture and mortality. 1 –6
World-wide, most dialysis patients receive hemodialysis, so there are few reports of osteoporosis in peritoneal dialysis (PD) patients. 7,8 In Western Europe, increasing numbers of older patients are receiving PD, so we wished to review the prevalence of osteoporosis in a contemporary cohort.
Methods
DXA scans of adult PD patients attending a United Kingdom (UK) university hospital, between March 2000 and March 2021, were reviewed. Scans were performed using a standardised protocol after drainage of PD dialysate (Hologic Discovery A (S/N87402.1), software version 13.5.2.1, Hologic, USA). 9 DXA scans measured body composition, and BMD in the lumbar spine (LS, L1–L5) and femoral neck (FN) were reported as T scores (bone density comparison to that of a 30-year-old healthy gender matched person) and Z scores (comparing bone density to the average values of age and gender matched persons). 10 According to WHO criteria, patients were categorized into three groups: normal BMD with a T score no less than −1.0, reduced BMD (osteopenia) with a T score between −1.0 and −2.5 and low BMD (osteoporosis) for patients with a T score less than −2.5. 5,10
Hospital computerised records were reviewed to retrieve patient demographics, relevant medical history and laboratory investigations. Previous transplantation was recorded along with medical kidney diseases potentially treated by steroids and other immunosuppressants (membranous and mesangio-capillary glomerulonephritis, focal segmental glomerular sclerosis, minimal change, Henoch-Schonlein purpura, idiopathic rapidly progressive glomerulonephritis, vasculitis, anti-glomerular basement disease, haemolytic uraemic syndrome, systemic lupus erythematosus, scleroderma, and myeloma, lymphoma, and amyloid). All glucose containing dialysates were low calcium (1.25 mmol/L) and all patients achieved current UK PD targets for small solute clearances, 11 with estimates of dietary protein intake calculated using the normalised nitrogen appearance (nPNA) rate. 12
Statistical analysis
Normally distributed continuous variables were expressed by mean values ± standard deviation (SD), non-parametric continuous variables were reported as median (25 and 75 percentile). Categorical variables were expressed by frequencies and percentages. Standard analyses included t-test, ANOVA, Mann–Whitney U test, Kruskal–Wallis and the Chi-square test, with appropriate adjustment for multiple testing. Spearman and Kendall’s tau b rank correlation were used to compare WHO categories, and Bland–Altman and Cohen’s kappa score for levels of agreement. Analyses were performed using Statistical Package for Social Sciences (SPSS Version 27.0 software, IBM Corp., Armonk, NY, USA), Prism 9.4 (Graph Pad, San Diego, USA). A two-tailed p value <0.05 was considered statistically significant.
Ethics
This retrospective audit was conducted according to the National Research Ethics guidelines and did not require additional local ethical approvals or individual patient consent. The audit was registered with the University hospital, and all data were anonymised.
Results
DXA scans of the LS and FN were available for review in 734 patients out of 909 (80.7%) adult PD patients attending a university hospital PD program. Patients were excluded if they had not had a DXA scan (awaiting living donor transplant, or transplanted before DXA scan appointment, unable to lie on the DXA scanning table), if they did not have results of both LS and FN BMD measurements (hip replacements) or patients who did not attend for scanning.
Patient demographics are displayed in Table 1. The average age was 61 years, with 420 male patients (57.2%) and 254 (34.6%) diabetic. BMD was greater for male patients (LS median 1.03 (0.93–1.17) vs. 0.97 (0.85–1.08 g/cm3), p < 0.001, and FN (0.71 (0.62–0.80) vs. 0.69 (0.58–0.80 g/cm3), p < 0.05)). Although the LS T scores were greater for males (mean −0.28 ± 1.93 vs. −0.97 ± 1.58, p < 0.001), Z-scores at both LS and FN and also T scores at the FN were not different between genders.
Patients divided according to dual-energy X-ray absorptiometry (DXA) bone densitometry lumbar spine and femoral neck T scores, as to normal bone density (T score no less than −1.0), reduced (T score −1.0 to −2.0) and low (T score greater than −2.0).a,5,10
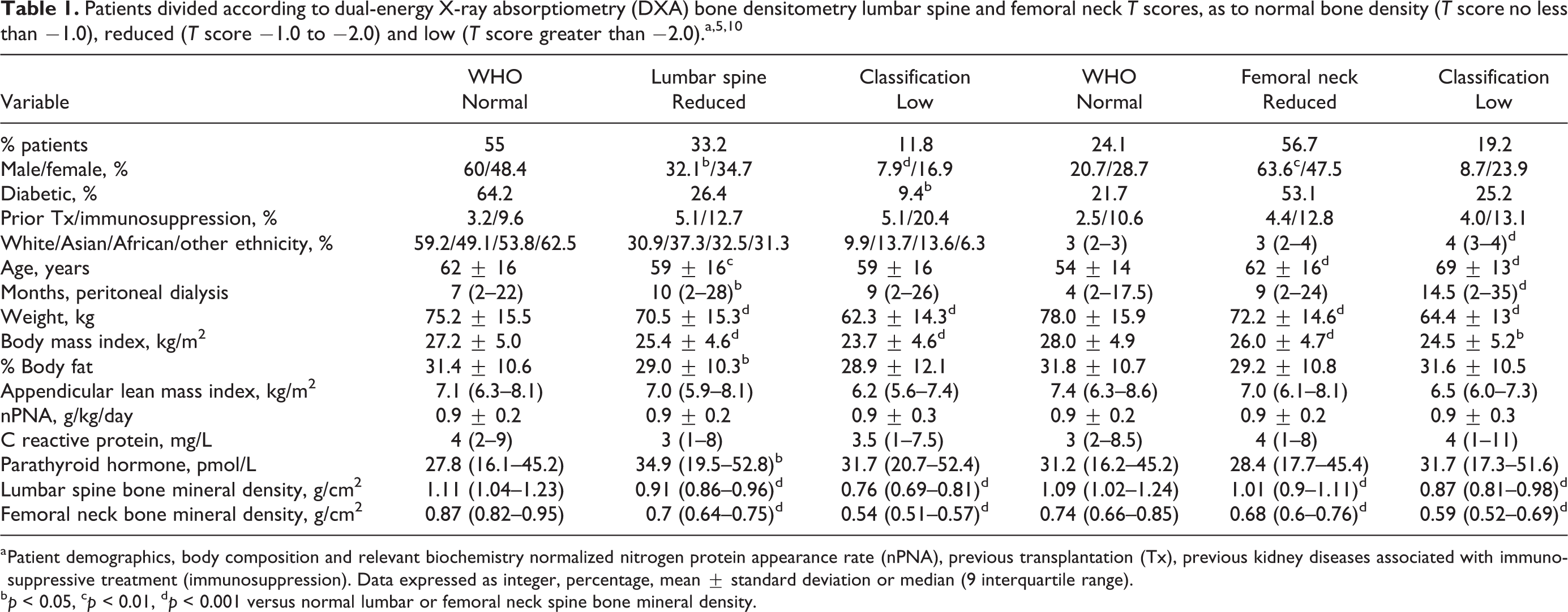
a Patient demographics, body composition and relevant biochemistry normalized nitrogen protein appearance rate (nPNA), previous transplantation (Tx), previous kidney diseases associated with immunosuppressive treatment (immunosuppression). Data expressed as integer, percentage, mean ± standard deviation or median (9 interquartile range).
b p < 0.05, c p < 0.01, d p < 0.001 versus normal lumbar or femoral neck spine bone mineral density.
Patients were classified according to WHO grading. 5,10 Most patients had normal BMD according to LS T scores but reduced when using FN T scores (Figure 1). Dividing patients according to LS BMD, then more women were classified as having low BMD (Table 1). Fewer patients with low BMD had diabetes, and weighed less and had lower body mass index (BMI). Comparing patients according to FN T scores, then patients with low BMD were older, weighed less, with lower BMI and longer treatment with PD. Only a relatively few patients had previously been transplanted, and there were no statistical differences in those with normal, reduced or low BMD T scores at the LS or FN. Similarly, there were no differences in the numbers of patients who may have had steroids or other immunosuppressants.
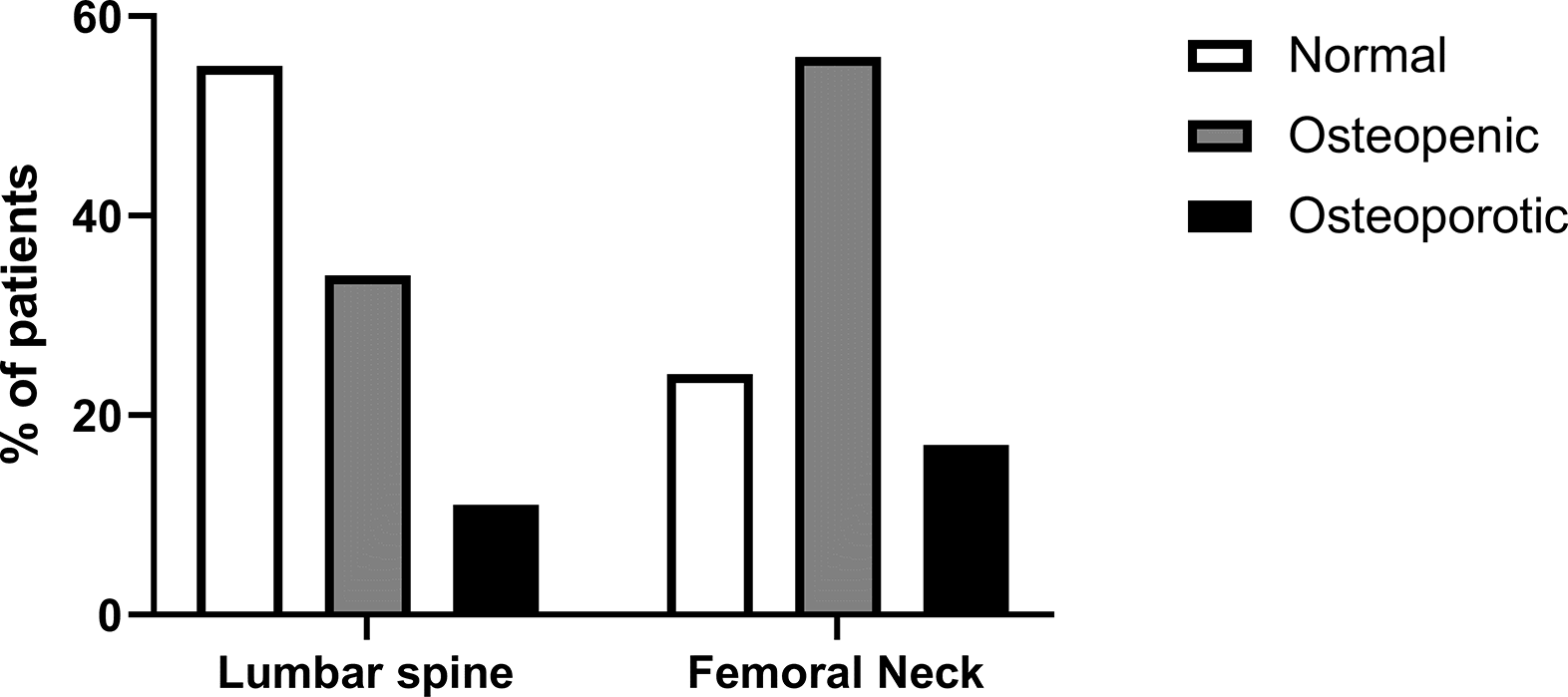
Percentage of patients classified according to dual-energy X-ray absorptiometry (DXA) bone densitometry lumbar spine and femoral neck T scores, as to normal bone mineral density (BMD), reduced BMD (osteopenia) and low BMD (osteoporosis) according to the World Health Organisation definitions.5,10 χ 2 112.2, p < 0.001.
Due to the differences between WHO grading using T scores at the LS and FN sites, rank correlation was performed using Spearman rho, r = 0.36, and 0.34 by Kendall’s tau b. Levels of agreement were assessed by Cohen’s kappa score of 0.17, and Bland–Altman bias of FN compared to LS was −0.37 (95% limits of agreement −1.8 to 1.08).
Discussion
The pattern of CKD-MBD disease is changing with increasing rates of osteoporosis reported. 13 DXA scanning is recommended by KDOQI, 1 with reports of osteoporosis and increased risk of bone fracture and mortality. 13 –15 Using WHO guidelines, the prevalence of low BMD in our PD cohort was much higher at the FN compared to the LS (17 vs. 11%). This is keeping with a previous smaller study comparing DXA scanning and high-resolution computerised scanning (hr-CT), which reported strong correlation between both techniques at the FN, but higher BMD values at the LS with DXA. 16 DXA scanning calculates BM based on the number of electrons counted compared to those emitted, so the denser the bone, the fewer electrons transmitted. However, at the LS, the DXA calculated density can be increased by crush vertebral micro- and macro-fractures which increase the apparent bone density as bone becomes impacted, and also calcification outside bones such as aortic calcification, and even electron dense materials in the bowel. 17,18 These and other confounders equally apply to measurements of whole body and pelvic bone density, and other potential confounders could include cardiac pacemakers, other implantable cardiac devices, breast implants, and artificial joints and cardiac valves and surgical metal work. As such more patients had higher LS T scores with most of our patients classified as having normal bone density by lumbar T scores, whereas most patients were classified as having reduced BMD according to FN T scores, and more patients would be classified as low BMD (osteoporotic) at the FN using WHO definitions.
Relatively, more women were classified as having low BMD using the LS T scores, but there was no gender difference with FN T scores. Patients with low BMD were older, weighed less, with a lower BMI. The differences between BMD measured at the LS and FN were probably due to confounding by micro- or macro-crush vertebral fractures particularly in older patients, and possibly the effects of overlying vascular calcification especially with the diabetic patients. Whereas at the FN, there were no confounding effects of overlying calcified blood vessels or electron dense material in the bowel.
We observed that BMD at the FN was lower with longer duration of PD treatment. There have been very few studies which have reviewed serial DXA scans in PD patients, 1 one reported no major changes in whole body and LS DXA BMD after 12 months, 19 whereas a larger Asian study noted a decline in a substantial proportion of patients over time, 8 but neither study reported on BMD at the FN.
Although the prevalence of osteoporosis in PD patients may be increasing with the greater number of older patients now treated, 20 dialysis patients may have an additional bone mineral disease. The optimum parathyroid hormone (PTH) range has not been elucidated, with KDIGO recommending a wide range of 130–600 pg/mL (14–64 pmol/L).1 As high levels of PTH lead to a fall in BMD and low levels an increase, then DXA scans allow for a more individualised PTH target, and in cases of low BMD referral for consideration of hormone replacement therapy, and other agents to combat osteoporosis and exercise programs. 1
We report a cross-sectional observational study classifying patients according to WHO definitions of reduced and low BMD. Our study highlights the difference between DXA BMD LS and FN measurements, in keeping with earlier studies comparing DXA and hr-CT. Measurements at the LS may not be as accurate as FN due to potential confounding from micro- or macro-crush fractures, overlying vascular calcification or electron dense material in the bowel, although this would require histological confirmation.
Footnotes
Acknowledgements
I wish to thank Jieying Zheng for the statistical analysis of the data.
Author contributions
A Davenport conceived audit, analyzed data and wrote drafts.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This retrospective audit was conducted according to the National Research Ethics guidelines and did not require additional local ethical approvals or individual patient consent. The audit was registered with the University hospital, and all data were anonymised.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Not applicable.
