Abstract
There is limited data on the standards of diabetes care in people on peritoneal dialysis (PD). Our aim was to assess the standards of diabetes care and the burden of hypoglycaemia in people with diabetes on PD. We performed a retrospective study at three university hospitals from December 2021 to January 2022. Clinical data were extracted from electronic health records. Diabetes care of people on PD was compared against recommended standards for people with diabetes on haemodialysis (as there are no agreed standards for PD). The degree of hypoglycaemia awareness was assessed by validated questionnaires. A total of 65 adults (15 type 1, 49 type 2 and 1 monogenic-diabetes) with a mean age of 63 (range 29–88) years were evaluated. Of them, 92% had diabetes retinal screening with annual review. In contrast, in this high-risk group for foot disease, only 77% had annual foot reviews. The rates of diabetes specialist reviews were variable between hospitals at 63–94% and 10 (15%) had impaired hypoglycaemia awareness. Of the cohort, 32% had HbA1c within the acceptable range of 58–80 mmol/mol (7.5–8.5%), 21% had HbA1c below 58 mmol/mol (7.5%) and 21% (n = 14) reported at least one hypoglycaemic event per month. Our results indicate variation of care within and between different centres, and the need for improved diabetes care in people on PD. Further work is required to establish agreed standards/recommendations of diabetes care in this population. Our findings highlight the necessity of an integrated multidisciplinary approach to improve the standard of diabetes care for people on PD.
This is a visual representation of the abstract.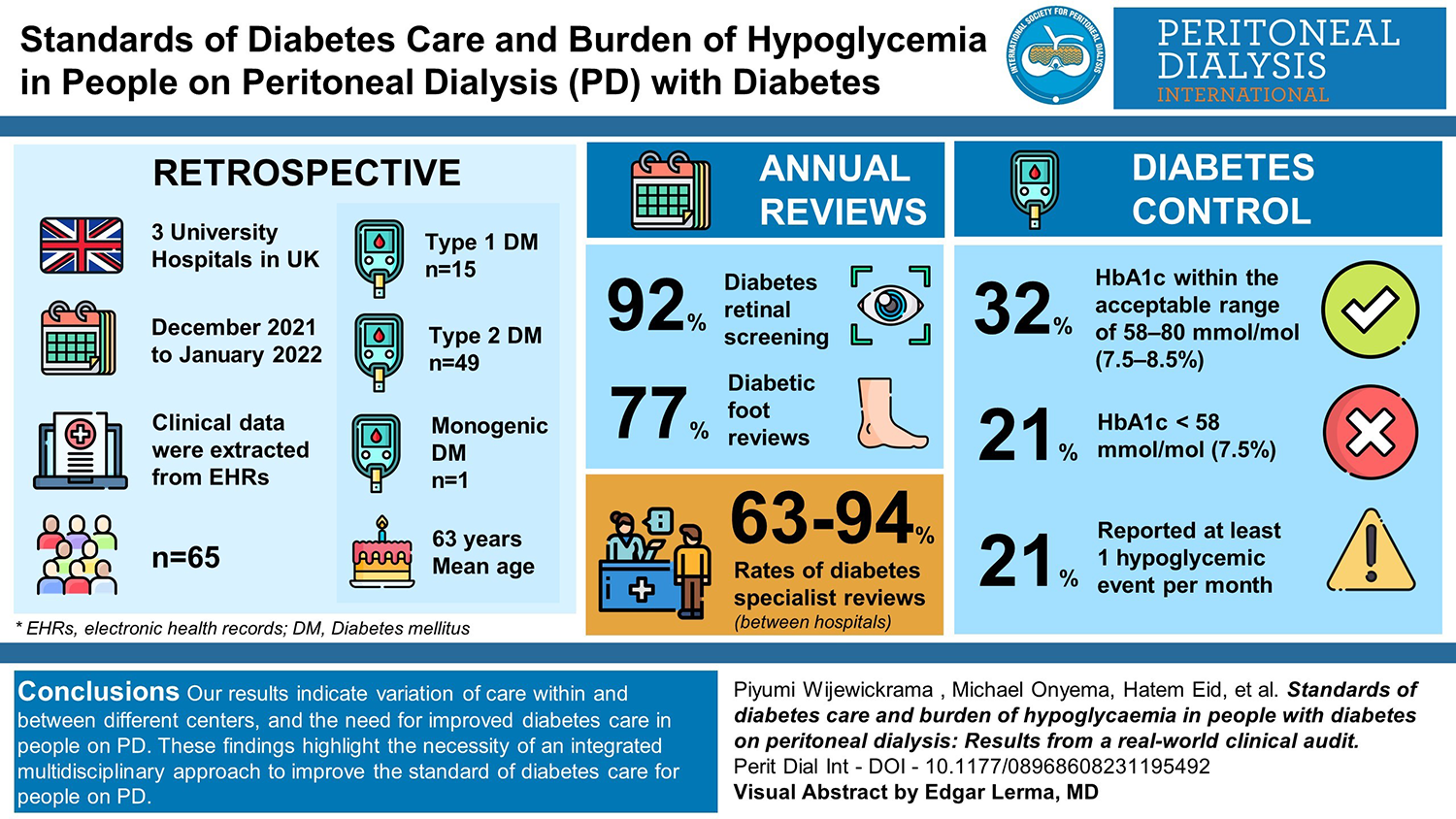
Introduction
Diabetes is one of the commonest causes of kidney failure (KF) worldwide. 1 Nearly 10% of all people receiving kidney replacement therapy are on peritoneal dialysis (PD). 2 People with diabetes on PD may have challenging diabetes control due to factors including dialysate glucose burden, risk of hypoglycaemia and challenges of glycaemic monitoring in the setting of KF. There is scarce clinical information on diabetes care processes and risks of hypoglycaemia in this group of people.
Our aim was to assess the standard of diabetes care and burden of hypoglycaemia in people with diabetes on PD at three university hospitals in the United Kingdom against the standards of care for diabetes for people on haemodialysis, as no specific guidelines/standards are available for PD.
Methods
All people with diabetes on PD at each hospital were identified using dialysis registry records and clinical health records. Clinical information and results were extracted from electronic health records between December 2021 and January 2022. This work was approved by the local clinical governance committees as quality improvement clinical audit work. The data and records collected for each person were compared against the minimum standards set by the Diabetes in Haemodialysis programme in the United Kingdom, 3 which specifies 100% of people with diabetes should have documented annual review of glycaemic control by a diabetes specialist, annual eye screening and annual foot risk assessment.
These recommendations also set target HbA1c as 58–68 mmol/mol (7.5–8.5%), with poor glycaemic control defined as HbA1c above 80 mmol/mol (9.5%). If HbA1c is below 58 mmol/mol (7.5%), a reduction in treatment should be considered due to the increased risk of hypoglycaemia. 3
Hypoglycaemia was defined as blood glucose below 3.9 mmol/L (70 mg/dL) at any point in the day. All individual records were checked and confirmed by two clinicians.
We assessed the degree of hypoglycaemia awareness using Gold and Clarke questionnaires, which are validated for this purpose. 4,5 Impaired hypoglycaemia awareness was defined as a score of ≥4 in the Gold or Clarke score. 4,5
Results
The baseline characteristics of the cohort and main findings are summarised in Table 1. A total of 65 records of adults with diabetes on PD were examined. Mean age was 63 (29–88) years. Fifteen (23%) had type 1 diabetes mellitus, while 49 (75%) had type 2 diabetes mellitus and 1 had maturity-onset diabetes of the young. Mean duration of diabetes was 20.9 years, ranging from 2 to 70 years. Mean time on PD was 28.5 months, ranging from 2 to 144 months. All participants except one were on glucose-containing dialysis regimens. Forty-four (67.6%) were on insulin, while 13 (20%) were only on oral hypoglycaemics, and 8 (12.3%) were not on any diabetes medications. Nineteen (29%) had established cardiovascular disease.
Baseline characteristics and the summary of audit standards assessed in a cohort of 65 people with diabetes on PD.a
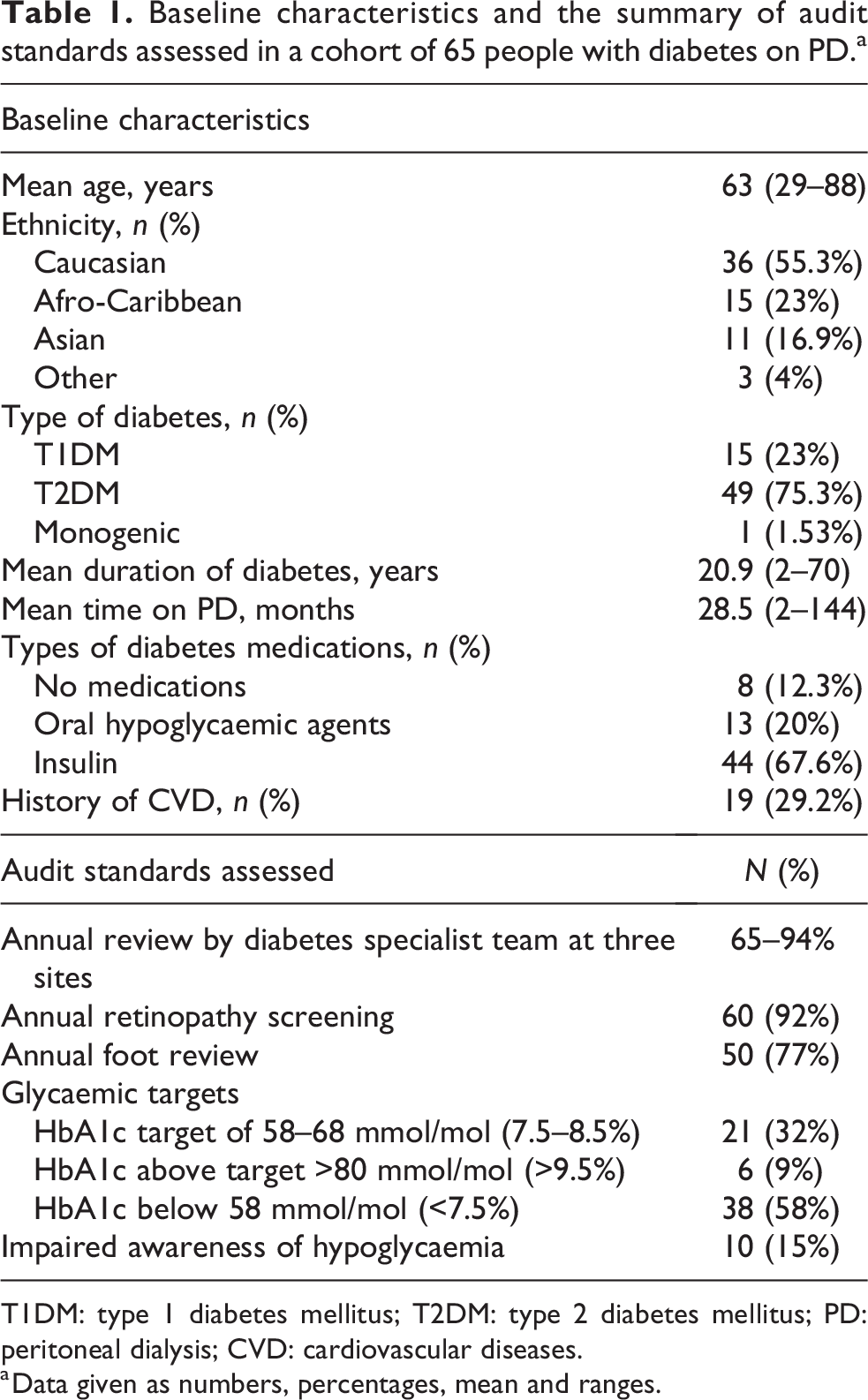
T1DM: type 1 diabetes mellitus; T2DM: type 2 diabetes mellitus; PD: peritoneal dialysis; CVD: cardiovascular diseases.
a Data given as numbers, percentages, mean and ranges.
Annual review by a diabetes specialist was variable among different hospitals with 63–94% receiving at least annual follow-up, which may reflect the lack of agreed standard of care for diabetes review for people on PD. Among the 18 people seen less than annually, 58% were on insulin. With regard to screening for diabetes complications, 92% (n = 60) were on a retinal screening programme with at least annual review. Only 77% (n = 50) had at least an annual documented foot review, with 35% of the whole cohort (n = 18) having a diabetes-related foot complication in the past (Table 1). The frequency of patient-reported foot reviews was variable from never (10 patients) to weekly (2 patients), the mode being every 12 months. Foot reviews reported were those undertaken by the hospital-based diabetes team and/or primary care review of diabetes care processes.
Twenty-one people (32%) had HbA1c within the target range of 58–68 mmol/mol (7.5–8.5%), while 9% (n = 6) had poor control (>80 mmol/mol, >9.5%). Among the 58% (n = 38) who had HbA1c below 58 mmol/mol (7.5%), 66% (n = 25) were on a diabetes medication which could cause hypoglycaemia (sulphonylurea or insulin). While impaired hypoglycaemia awareness was observed in 15% (n = 10) of the total cohort.
At least one episode of self-reported hypoglycaemia per month was reported by 21% (n = 8) in the subgroup who had HbA1c below target [58 mmol/mol (7.5%)].
In our cohort of 65 people, 2 had diabetes-related admissions in the preceding 12 months. Of the 44 people on insulin, only 7 were on continuous or intermittently scanned glucose monitoring systems.
Discussion and conclusion
Our results from a real-world clinical audit of diabetes care in people on PD demonstrate the need for improved and standardised diabetes care in this group of people.
The frequency of diabetes specialist team review varied both within and between centres and needs further improvement. Adherence to annual retinopathy screening was >90%. People with KF are at very high risk for diabetic foot disease, and recent recommendations suggest at least monthly foot checks in people on haemodialysis. 6 In our study, the frequency of at least annual foot checks was only 77% with most people having annual foot reviews rather than the recommended higher intensity foot surveillance. A study involving people with KF on haemodialysis or PD demonstrated five times higher rate of foot ulceration in dialysis compared to pre-dialysis populations. 7 Further education is required to ensure awareness of the need for frequent foot checks in this very high-risk group to reduce unwarranted variation in foot care and related outcomes.
With regard to glycaemic targets, we found that only 32% in our cohort achieved acceptable HbA1c target (HbA1c 7.5% to 8.5%) while 9% had poor control (defined as HbA1c> 9.5 %). There are conflicting data on the impact of HbA1c on mortality in PD. Observational studies have shown higher all-cause mortality in people having raised HbA1c ≥64 mmol/mol (≥8%), particularly due to infections, 8,9 while other studies have not. 10
There are no previous data on the burden of hypoglycaemia in people with diabetes on PD. We observed that more than half of our cohort had HbA1c below target, hence at high risk of hypoglycaemia, and of more concern, two-thirds of this group were on medications that can cause hypoglycaemia. Moreover, 15% of the cohort had impaired awareness of hypoglycaemia and 21% reported at least one hypoglycaemic event per month. These findings highlight the need for diabetes specialist review to ensure appropriate de-escalation of diabetes treatments and provide support with strategies to mitigate hypoglycaemia.
Previous studies in haemodialysis populations have demonstrated a high hypoglycaemia burden with a need to reduce insulin doses by 25% on days of haemodialysis. 11,12 There are no similar data in PD, and indeed the impact of glucose loads in PD fluids and related glycaemic variability can make optimising glucose levels challenging in people on PD. Our results and a recent review highlight the importance of individualised diabetes care to minimise glycaemic variability and hypoglycaemia in people with KF. 13,14 A greater use of diabetes technology for glucose monitoring for people on PD treated with insulin can help reduce the burden of hypoglycaemia and aid self-management. Studies in people on PD have demonstrated the accuracy of such continuous glucose monitoring but further studies are warranted as there are limited large long term clinical studies and evidence in this area. 15 Several studies in people with diabetes on haemodialysis have demonstrated the benefits of continuous glucose monitoring technology in terms of minimising hypoglycaemia and reducing glycaemic variability. 16,17
The importance of diabetes education and care has been shown to significantly improve outcomes including foot complications, (rate of amputations), diabetes/vascular-related hospital admissions, HbA1c and overall quality of life in people on haemodialysis. 18 Such interventions may be easier to implement in haemodialysis centres/units where people are attending at regular intervals for their care and where healthcare professionals present can be trained to deliver enhanced diabetes surveillance/care and education. Similar albeit less comprehensive strategies to optimise care of people on PD through specific insulin regimes and education have demonstrated reductions in both HbA1c and hypoglycaemia burden. 13
We observed a significant variation of diabetes care for people on PD within and in between the centres, suggesting the need for updated guidelines. A recent review highlighted the need for an integrated approach to managing diabetes in people with PD and the need for specific standards of care for this unique cohort of people at high risk of glycaemic variability, hypoglycaemia and fragmented diabetes care. 19
When considering the follow-up and screening of people with diabetes who did not have KF, a meta-analysis in 2018 reported a diabetic retinopathy screening uptake of between 61% and 89%. 20 Authors from the United States report that 63% of people with diabetes had received an annual foot check, while more recent Canadian data from 2022 showed that 53% had undergone an annual foot examination by a healthcare provider. 21,22 In people with diabetes on haemodialysis, recent data from the United Kingdom reported that between 19% and 44% had annual reviews of their diabetes control by a diabetes specialist, between 74% and 92% had at least annual eye screening, and between 20% and 77.8% had documented annual foot screening. 23
These results in people undergoing haemodialysis are comparable to our findings among people undergoing PD. National and international guidelines on diabetes management and standards of care in people with kidney disease also document the need for regular surveillance and care in people with KF 24 and emphasise the importance of enhanced surveillance in this high-risk group.
In conclusion, a multidisciplinary team (MDT) approach between kidney and diabetes teams is required to improve standards of care, optimise diabetes medications, minimise hypoglycaemia and ensure better screening for other diabetes-related complications. This can be implemented via joint kidney and diabetes clinics or MDT meetings. Subsequent to the results of this audit the centres involved are planning to pilot a virtual MDT case review of all people with diabetes on PD to ensure diabetes care processes are reviewed and followed. Similar models of care should be considered taking into account patient preferences, and the unique challenges of fragmented care processes which can occur in this cohort who have often chosen home-based kidney replacement treatment.
We propose that national and international working groups consisting of diabetes and kidney care clinical experts and people with diabetes on PD work together to share examples of best practice and define standards of care for diabetes managament in this high risk group of people on PD.
Footnotes
Acknowledgements
None.
Author contributions
JK conceptualised the study. JK, PW, JW and MO researched the literature. JK, MO, HE, NP, JD, DM, ML, PV and JW collected data and analysed data. PW and JK wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The audit was approved by the local clinical governance committees of the hospitals involved. Ethical approval was not required as this was a retrospective data collection.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent to participate
Informed consent to participate was not sought because this was a retrospective database study.
Informed consent to publish
Informed consent to publish was not sought as this contained no patient identification data.
