Abstract

Herein we describe a case of a patient successfully performing peritoneal dialysis post-appendicovesicostomy (Mitrofanoff procedure) for congenital lower urinary tract disease. Originally developed in 1980, the appendicovesicostomy involves utilisation of the appendix to create a conduit between the urinary bladder and skin surface. 1,2 This procedure provides an alternative channel for bladder catheterisation for patients with difficult or painful urethral catheterisation in the context of neurogenic bladder dysfunction, spinal cord injury, congenital urethral defects, urethral trauma or urethral malignancy. The stoma site is typically created at the umbilicus or lower abdominal wall, through which patients catheterise their bladder every 3–4 h.
Our patient is a 26-year-old man with a history of left solitary kidney and posterior urethral valves who underwent Mitrofanoff appendicovesicostomy at the age of 3. He required bladder augmentation (ileocystoplasty) prior to his Mitrofanoff procedure to allow for greater urine storage. Several years following this surgery, he developed vesicoureteral reflux and experienced recurrent episodes of acute kidney injury secondary to urinary tract infections and ureteric obstruction. He subsequently developed chronic kidney disease and progressed to end-stage kidney disease at the age of 25, at which point he was initiated on haemodialysis with a goal to transition to peritoneal dialysis. His peritoneal dialysis catheter was inserted laparoscopically, revealing numerous lower abdominal adhesions that were not amenable for lysis, as they were a part of his appendicovesicostomy. His initial procedure was unfortunately complicated by gallbladder perforation and consequent cholecystectomy; however, a peritoneal dialysis catheter was subsequently successfully inserted in the right upper abdominal quadrant. Figure 1 displays his peritoneal dialysis catheter exit site in the right upper quadrant (label A) and his Mitrofanoff umbilical stoma through which he catheterises his bladder (label B).
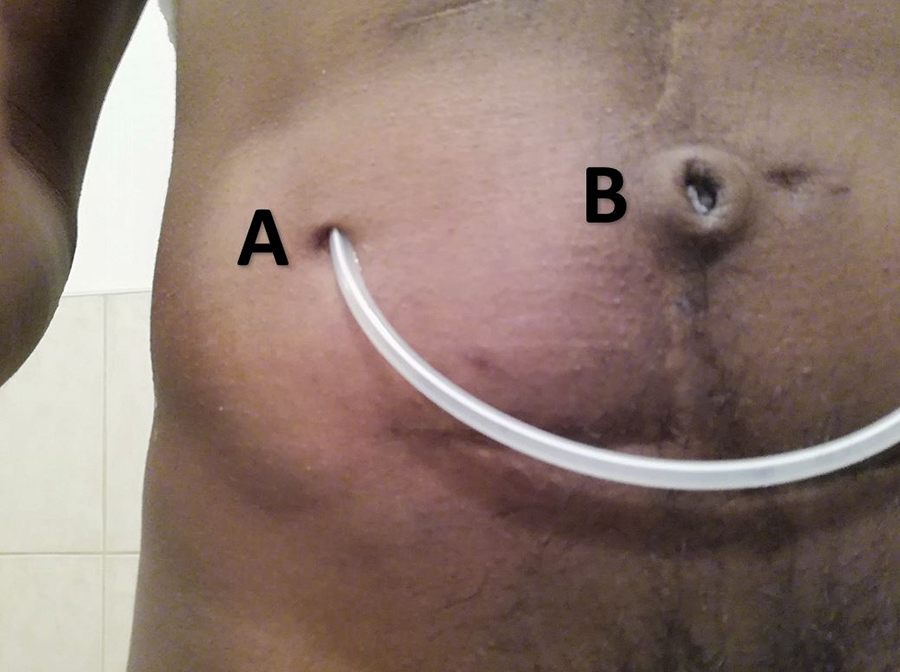
Patient post-appendicovesicostomy (Mitrofanoff) procedure for posterior urethral valves. Label A illustrates the peritoneal dialysis catheter exit site. Label B illustrates the Mitrofanoff umbilical stoma through which the patient catheterises his bladder.
At this juncture, our patient has completed 7 months of peritoneal dialysis therapy with minimal complications. He was transitioned from automated to ambulatory peritoneal dialysis to address frequent cycler low drain alarms, likely secondary to fluid pocketing caused by his intrabdominal adhesions. Our patient is able to comfortably dialyse with a fill volume of 1.5 L (body weight 60 kg). He continues to have residual kidney function and can catheterise his bladder through his Mitrofanoff conduit when his peritoneum is both full and empty. While there may be theoretically an increased risk of peritonitis from repeated instrumentation of the appendiceal conduit, this risk has not transpired. Two months into treatment, our patient had an episode of culture-negative peritonitis, which responded to a 2-week course of intraperitoneal antimicrobial treatment. Given the early time course of this peritonitis, it is possible that technique error– rather than Mitrofanoff catheterisation– contributed to this infection.
Posterior urethral valves represent the most common etiology of congenital urethral obstruction with 8–22% of patients progressing to end-stage kidney disease. 3 –6 Our case illustrates that peritoneal dialysis can be considered as an option of kidney replacement therapy in this rare patient population post-Mitrofanoff appendicovesicostomy.
Footnotes
Acknowledgement
None
Author contributions
ZA reviewed the literature and wrote the first draft of the manuscript. JMB reviewed and edited the manuscript. Both authors approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical approval
The University Health Network Research Ethics Board does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Informed consent to participate
Written informed consent was obtained from the patient before the study.
Informed consent to publish
Written informed consent was obtained from the patient for their anonymised information to be published in this article.
