Abstract
Background:
Providing support is important to maintain a patient on peritoneal dialysis (PD), though its impact on outcomes has not been investigated thoroughly. We examined the association between having support and risk of a transfer to hemodialysis.
Methods:
In this retrospective observational cohort study, we used data captured in the Dialysis Measurement Analysis and Reporting system about patients who started PD in Alberta, Canada, between 1 January 2013 and 30 September 2018. Support was defined as the availability of a support person in the home who was able, willing and available to provide support for PD in the patient’s residence. The outcome of interest was a transfer to hemodialysis for at least 90 days. We estimated the cumulative incidence of a transfer over time accounting for competing risks and hazard ratios to summarise the association between support and a transfer. We split follow-up time as hazard ratios varied over time.
Results:
Six hundred and eighty-three incident PD patients, median age 58 years (IQR: 47–68) and 35% female, were followed for a median of 15 months. The cumulative incidence of a transfer to hemodialysis at 24 months was 26%. Having support was associated with a reduced risk of a transfer between 3 and 12 months after the start of dialysis (HR3-12mo: 0.44; 95% CI: 0.25–0.78), but not earlier (hazard ratio (HR)<3mo: 0.96; 95% confidence interval (CI): 0.55–1.69) or later (HR>12mo: 1.19; 95% CI: 0.65–2.17).
Conclusions:
A transfer to hemodialysis is common. Having a support person at home is associated with a short-term protective effect after the initiation of PD.
This is a visual representation of the abstract.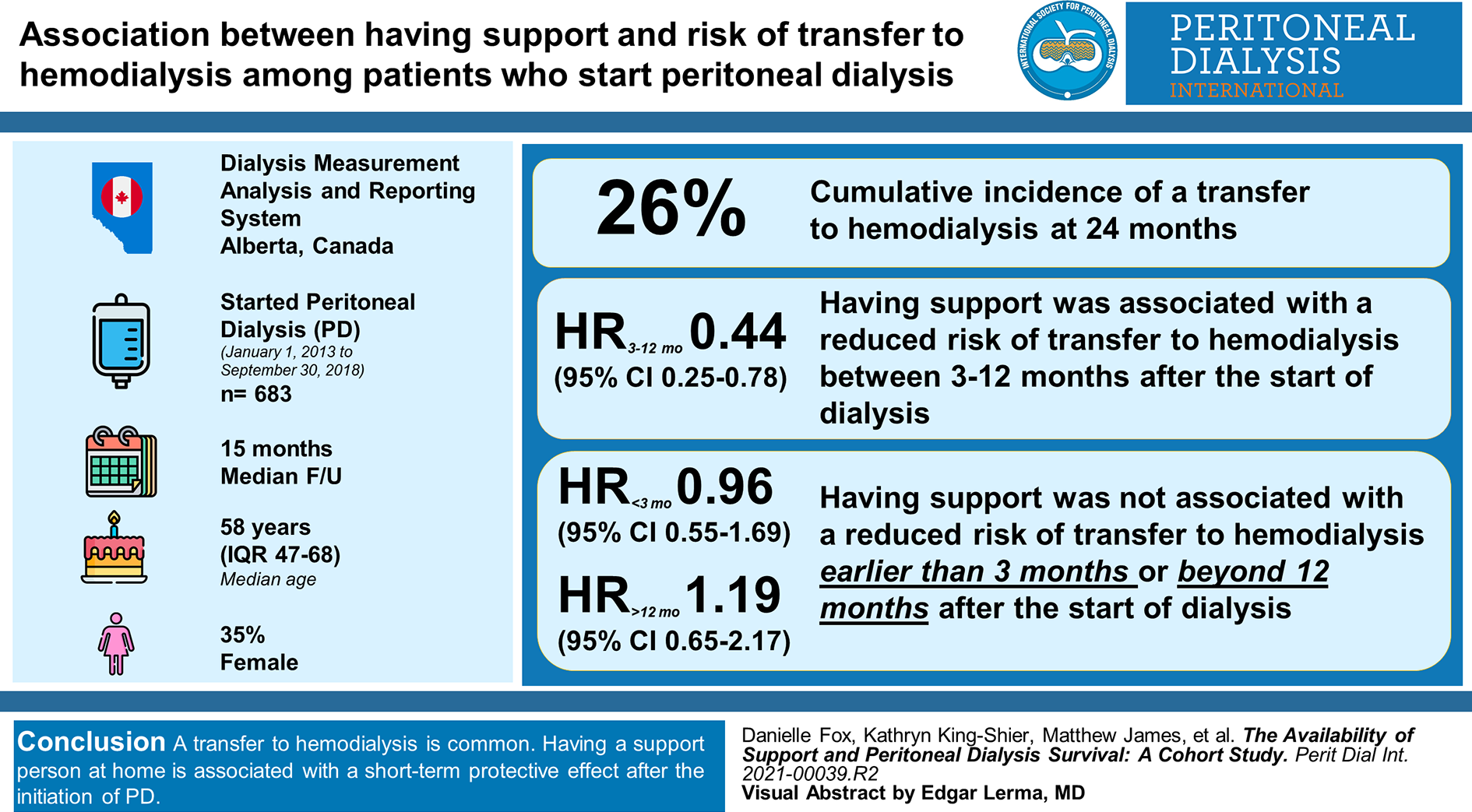
Keywords
Introduction
As the prevalence of end stage kidney disease (ESKD) increases, 1 patient appropriate, cost-effective therapies are necessary to contain healthcare spending. Patients on peritoneal dialysis (PD) have similar clinical outcomes with respect to survival, hospitalisations, infectious complications and quality of life compared to other dialysis therapies, but PD is significantly less costly to provide in most developed countries. 2 –10 As a result, PD is being promoted in many jurisdictions. 11 –13
Transfers from PD to hemodialysis (HD) are a common occurrence, and it is possible that by promoting PD, there is a risk of putting more marginal candidates on the therapy which may increase the risk of transfers to HD. To successfully sustain patients on therapy and increase PD prevalence, it is important to identify modifiable reasons for transfers to HD in order to implement strategies to mitigate them. Potentially modifiable social factors, including the availability of support, are largely underexplored, though there is evidence to suggest that the availability of support may increase PD eligibility and choice. 14 Further, the impact of support on outcomes has been primarily investigated through an instrumental support lens (e.g. performing the dialysis procedure), 15,16 which may not capture the true impact of having a support person in the home and the additional types of support they might provide. 17,18
The objective of this study was to examine the association between the availability of a support person in the home and the risk of a transfer to HD in patients starting PD in Alberta, Canada. We hypothesised that the availability of support from family or friends, regardless of who actually performs the dialysis procedure, would be associated with a reduced risk of a transfer to HD.
Materials and methods
Study design and data source
This retrospective, observational cohort study utilised data from the Dialysis Measurement Analysis and Reporting (DMAR) system. DMAR was a web-based platform that captured high-quality, expert-reviewed data on patients initiating dialysis in Alberta, Canada. Data were entered by trained front line staff following clear definitions and a standardised coding scheme. Data elements captured in DMAR include baseline information such as demographic variables, comorbidities, lab values, a structured assessment for PD eligibility, the availability of support and choice of treatment modality. Outcomes are tracked every 3 months and include changes to treatment modality and the occurrence of death, transplant, access procedures and hospitalisations. All data elements were reviewed by an expert to maintain data quality and to ensure consistency in coding. Queries are communicated back to the user and must be addressed prior to sign-off and data analysis.
Participants and setting
Alberta is a province of over four million people, 19 with regional PD programmes located in Calgary, Edmonton, Red Deer, Medicine Hat and Lethbridge. DMAR data was available for the Calgary Zone from January 2013 until 31 December 2018 and in the other centres from 1 October 2016 to 31 December 2018. To be eligible for inclusion in the DMAR database, patients must have (1) received at least one outpatient dialysis therapy; (2) had a diagnosis of ESKD documented by a nephrologist; or (3) had an acute kidney injury that required at least 28 days of dialysis. The cohort was restricted to adults 18 years of age, or older, who had started outpatient PD (i.e., received at least one outpatient PD treatment) in Alberta, Canada between 1 January 2013 and 30 September 2018. New starts were only included until 30th September 2018 to ensure at least 3 months of potential follow-up. Participants were followed with outcomes updated every 3 months until the first of: death, transplant, recovery of kidney function, transfer out of the programme, loss to follow-up or until 31st December 2018.
Availability of support
The availability of a support person in the home was captured at baseline and was defined as the presence of ‘an individual(s) who [was] able, willing, and available to provide support for PD in the patient’s residence’. A support person could include a spouse, other family member, paid caregiver or another support person. This assessment was conducted by the trained front line staff who entered the data obtained from both chart review and through their interaction with patients and other staff in the clinic. This study focused on the availability of a support person in the home and so receipt of home care assisted PD did not qualify. Only one centre in Alberta had a home care–assisted PD programme during the study and only 18 people were receiving home care–assisted PD at any one point in time (Hermann, L, personal communication, 13 May 2019). A sensitivity analysis was conducted excluding patients who had received home care–assisted PD or PD in a chronic care facility at any point during PD therapy.
Primary outcome: Transfers to HD
The primary outcomes was defined as a transfer to hemodialysis for a period of at least 90 days. In previous studies, only 11% of participants meeting this criterion returned to PD after 12 months, compared to 24% using the 30-day definition that is commonly employed. 20 If a patient transferred to HD, but had another event occur before 90 days (e.g., transplant, death, transfer out of the programme), they were not considered as a transfer to HD. Eighteen reasons for transferring to HD are reported in the DMAR database and they were grouped into eight categories for the purpose of this study: PD infectious complications, inadequate dialysis, PD catheter-related problems, other PD complications, social reasons, medical reasons, other and unknown.
Baseline characteristics
Baseline characteristics that were potentially relevant confounding variables in the clinical opinions of the authors were retrieved from the DMAR database and considered for inclusion in the model. These were captured at the time of dialysis initiation and included age, gender, body mass index (BMI), diabetes mellitus, cardiovascular disease (congestive heart failure, coronary artery disease and other cardiac disease), peripheral vascular disease, polycystic kidney disease, albumin, estimated glomerular filtration rate, started dialysis as an inpatient, receipt of HD before PD, receipt of at least 4 months of pre-dialysis care, the presence of a medical, physical, cognitive or social barrier to PD.
Statistical analysis
For continuous variables, differences in baseline characteristics between groups were calculated using the Mann–Whitney U test, and we reported the median and interquartile ranges, as they were not normally distributed. Categorical variables were analysed using Pearson’s chi-squared test, and we reported frequencies and percentages. All variables had less than 1% missing values, with the exception of BMI and albumin, which were missing in 5% and 2% of patients, respectively. For these cases, imputation was used. BMI was estimated and imputed using the probable value based on weight and gender, while albumin was replaced with the mean for a patient’s age group (18–44, 45–64, 65–75 and >75). We screened for multicollinearity and used a variance inflation factor of greater than 5 to represent the presence of significant collinearity.
The method of Fine and Gray 21 was used to study the effect of support on the prognostic of PD (i.e. the cumulative risk of a transfer to HD). Transplant, death (including a transfer to palliative care) and recovery of kidney function were considered competing events. The crude cumulative risk of a transfer to HD, death (competing risk: HD transfers, transplant and recovered kidney function) and transplant (competing risks: HD transfers, death and recovered kidney function) at 2 years was reported.
A Cox proportional hazards model was used to evaluate the etiological role of the availability of a support person in the home at baseline and a transfer to HD. Death (which included a transfer to palliative care), transplant, recovery of kidney function, transfer out of the programme, loss to follow-up and the end of follow-up were included as censoring events. We tested the model assumptions by means of graphical and formal tests based on residuals. Our exposure variable was screened for a violation of the proportional hazards assumption using graphical and statistical techniques.
The final model was adjusted for age, sex, diabetes, cardiac disease, peripheral vascular disease, polycystic kidney disease, albumin, inpatient dialysis starts, received HD before PD, having at least 4 months of pre dialysis care and having a medical, physical, cognitive or social/residential barrier to self-care PD. Of the baseline characteristics, BMI and estimated glomerular filtration rate were excluded from the model as they did not impact the magnitude of the association between support and a transfer to HD. All analyses were conducted using STATA statistical software, Version 15. 22
Results
Baseline characteristics
Seven hundred and seven patients started PD between 1 January 2013 and 30 September 2018 and were eligible for inclusion in the DMAR database. We included 683 in the study, excluding 8 who never received an outpatient PD treatment, and 16 who did not have at least three months of follow-up. The median age of patients was 58 years (IQR: 47–68) and 35% were female. Fifty-nine percent had a support person available in the home (spouse (70.5%); other family member (25.7%); other support person (3.8%)). Patient characteristics at dialysis initiation are shown in Table 1. Individuals with a support person in the home at baseline were more likely to be older, have cardiovascular disease, a lower albumin level and at least one physical or cognitive barrier to self-care PD. The median follow-up time for study participants was 15 months (IQR: 7–26).
Patient characteristics at start of peritoneal dialysis.
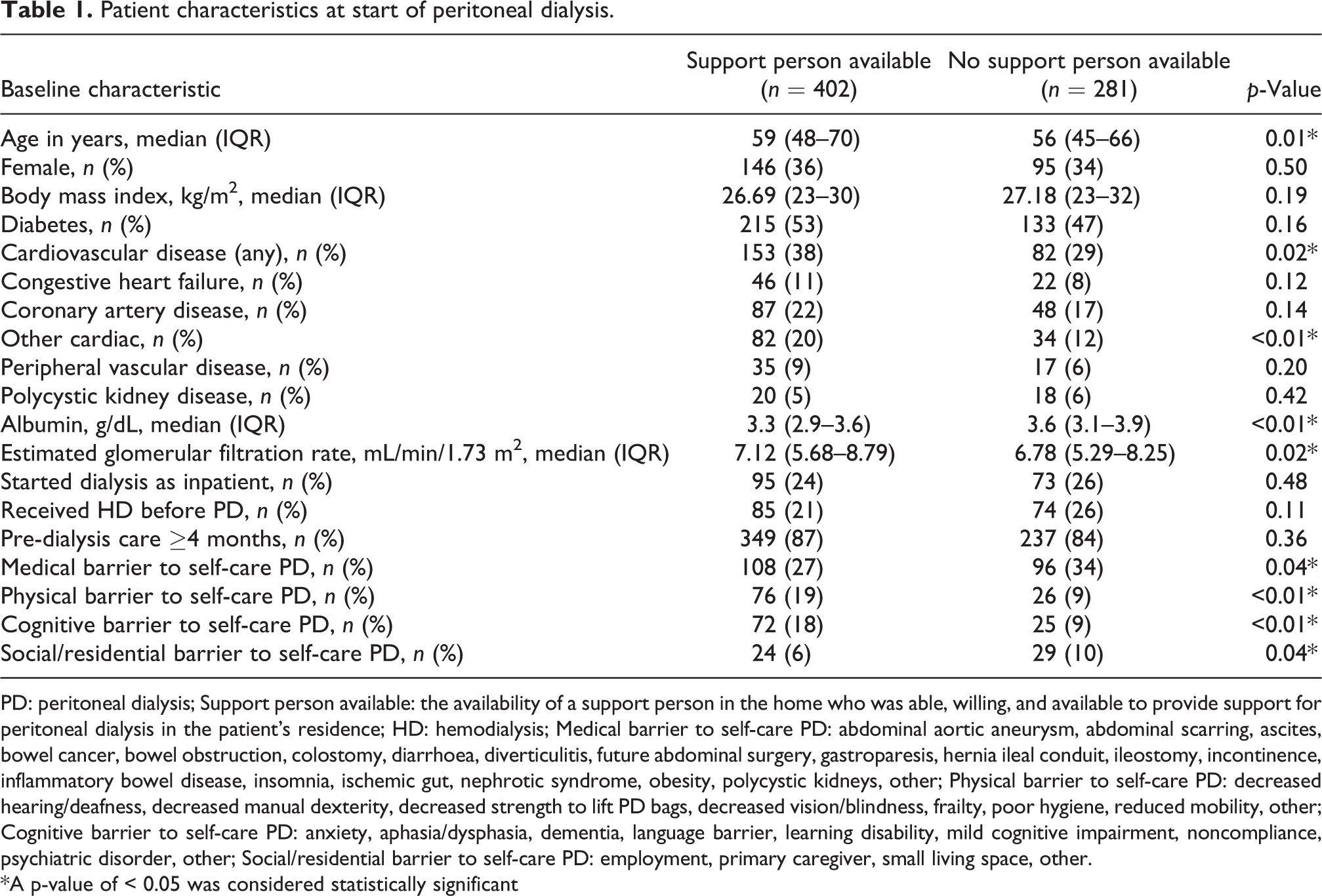
PD: peritoneal dialysis; Support person available: the availability of a support person in the home who was able, willing, and available to provide support for peritoneal dialysis in the patient’s residence; HD: hemodialysis; Medical barrier to self-care PD: abdominal aortic aneurysm, abdominal scarring, ascites, bowel cancer, bowel obstruction, colostomy, diarrhoea, diverticulitis, future abdominal surgery, gastroparesis, hernia ileal conduit, ileostomy, incontinence, inflammatory bowel disease, insomnia, ischemic gut, nephrotic syndrome, obesity, polycystic kidneys, other; Physical barrier to self-care PD: decreased hearing/deafness, decreased manual dexterity, decreased strength to lift PD bags, decreased vision/blindness, frailty, poor hygiene, reduced mobility, other; Cognitive barrier to self-care PD: anxiety, aphasia/dysphasia, dementia, language barrier, learning disability, mild cognitive impairment, noncompliance, psychiatric disorder, other; Social/residential barrier to self-care PD: employment, primary caregiver, small living space, other.
*A p-value of < 0.05 was considered statistically significant
Reasons for loss from therapy
One hundred and sixty patients (23.4%) experienced a transfer to HD at a median time of 6 months (IQR: 2–18). Other causes of loss from PD included transplant (10.1%), death (8.9%), a transfer to palliative care (1.6%), recovery of kidney function (1.8%) and transfer out of the programme (1.9%) (Table 2).
Reasons for loss from peritoneal dialysis therapy.
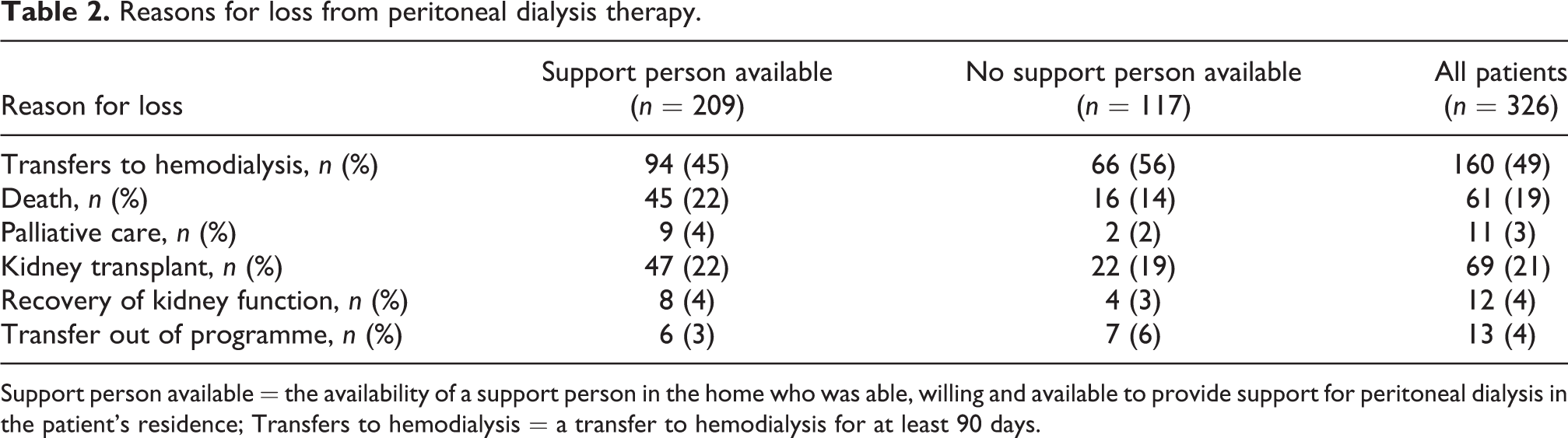
Support person available = the availability of a support person in the home who was able, willing and available to provide support for peritoneal dialysis in the patient’s residence; Transfers to hemodialysis = a transfer to hemodialysis for at least 90 days.
The most common reasons patients transferred to HD were inadequate dialysis (30%) and infectious complications (18%) (Table 3). Social reasons accounted for 12% of events and included patient-initiated switches to HD, the inability to cope, the need for increased care and psychosocial, social and financial reasons. Patients were more likely to transfer to HD within the first 3 months on PD due to catheter complications (n = 10) and other PD complication (n = 15), with transfers due to infections complications (n = 15) and inadequate dialysis (n = 29) being more common after 12 months on therapy.
Reasons for transfers from peritoneal dialysis to hemodialysis, by time period.
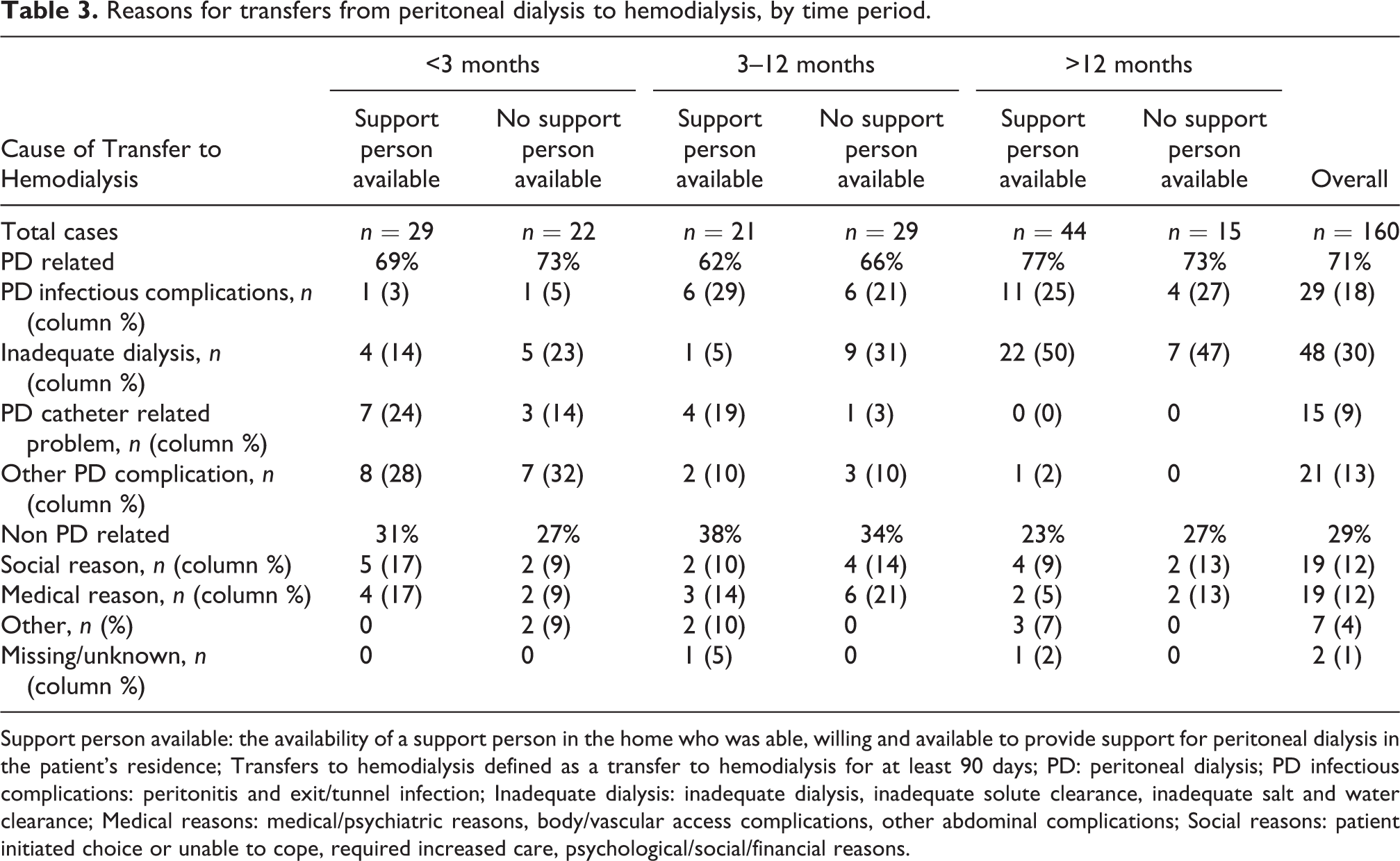
Support person available: the availability of a support person in the home who was able, willing and available to provide support for peritoneal dialysis in the patient’s residence; Transfers to hemodialysis defined as a transfer to hemodialysis for at least 90 days; PD: peritoneal dialysis; PD infectious complications: peritonitis and exit/tunnel infection; Inadequate dialysis: inadequate dialysis, inadequate solute clearance, inadequate salt and water clearance; Medical reasons: medical/psychiatric reasons, body/vascular access complications, other abdominal complications; Social reasons: patient initiated choice or unable to cope, required increased care, psychological/social/financial reasons.
Cumulative incidence of transfers to HD
The cumulative incidence of transferring to HD was 12%, 16% and 26% at 6, 12 and 24 months, respectively. The cumulative incidence curves for a transfer to HD, transplant and death (including transfer to palliative care), according to the presence or absence of the availability of a support person in the home, are reported in Figure 1 (transfer to HD) and Supplementary Figures 1 and 2 (transplant and death).
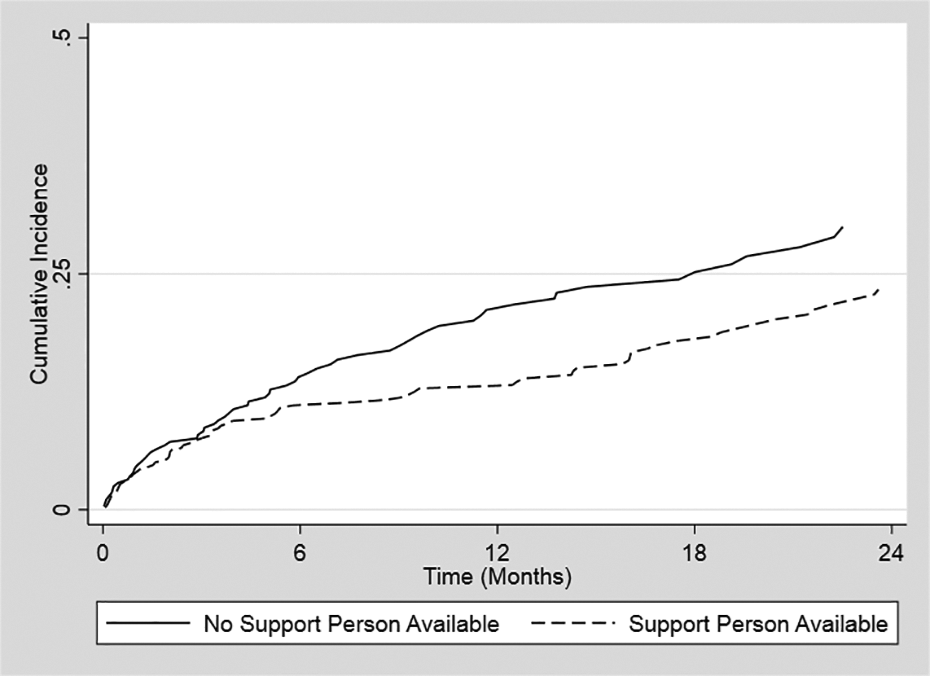
Cumulative incidence of a transfer to hemodialysis over time. The unadjusted, cumulative incidence of a transfer to hemodialysis over time for those with and without the availability of a support person in the home.
Availability of support person and the risk of a transfer to HD
Overall, the availability of a support person in the home was not associated with a transfer to HD (adjusted hazard ratio (HR): 0.79, 95% confidence interval (CI): 0.57–1.11). However, our exposure variable violated the proportional hazards assumption based on the Schoenfeld Residuals (p < 0.01) and non-parallel curves on the log(-log) survival versus time plot. Visual inspection of the plot suggested that the hazards diverged at 3 months, and then were constant until approximately 12 months. To address the violation of the proportional hazards assumption, we created a time dependent model, stratifying time into three periods (<3 months, 3–12 months and >12 months). 23 The availability of a support person in the home was not associated with a transfer to HD during the first 3 months or after 1 year of PD, however was associated with a lower probability of experiencing a transfer to HD between 3 and 12 months (Figure 2, Supplementary Table 1) (adjusted HR<3mo: 0.96, 95% CI: 0.55–1.69; adjusted HR3-12mo:0.44, 95% CI: 0.25–0.78; adjusted HR>12mo:1.19, 95% CI: 0.65–2.17). A sensitivity analysis excluding participants who had received home care assisted PD or PD in a chronic care facility at any point during PD therapy (n = 44) revealed similar results. No characteristics other than support were associated with a transfer to HD.
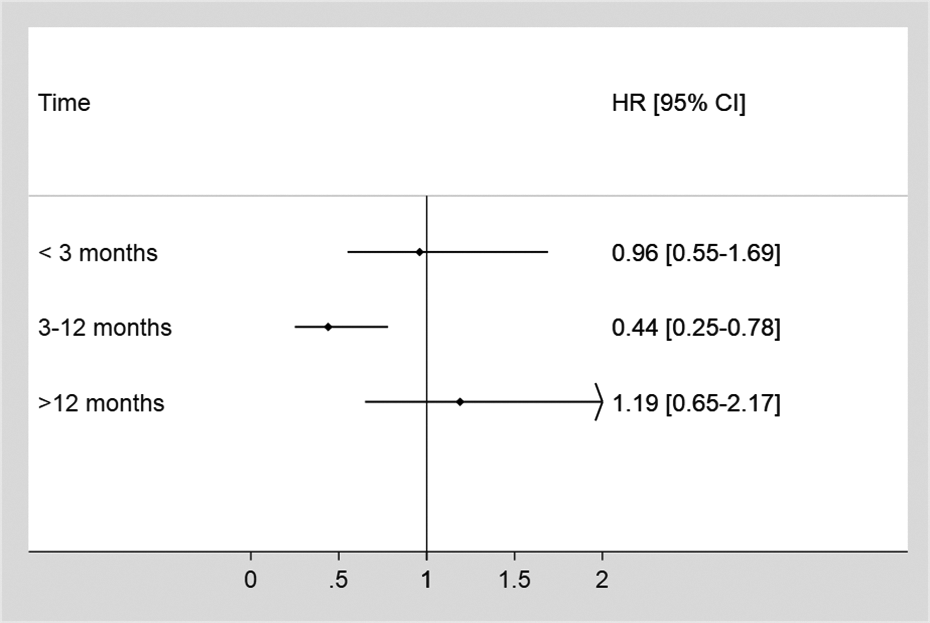
Hazard ratio of the effect of support on a transfer to hemodialysis. The adjusted hazard ratio of the effect of the availability of support person in the home on a transfer from PD to hemodialysis. The availability of a support person in the home between 3 and 12 months was associated with a lower probability of experiencing a transfer to hemodialysis. Note: Time: time from PD start to a transfer to hemodialysis; PD: peritoneal dialysis; HR: hazard ratio; CI: confidence interval; Model adjusted for: age, sex, diabetes, cardiac disease, peripheral vascular disease, polycystic kidney disease, albumin, inpatient dialysis starts, received HD before PD, having at least 4 months of pre dialysis care and having a medical, physical, cognitive or social/residential barrier to self-care PD.
Discussion
The association between the availability of a support person in the home and a transfer to HD in patients starting PD therapy in Alberta, Canada, was examined in this study. We found that having a support person who was able, willing and available to provide support for PD in the patient’s residence was associated with a 56% reduction in the risk of transferring to HD between 3 and 12 months, despite the fact that patients with support were older, had more comorbidities and more physical and cognitive barriers to self-care PD.
Support is associated with an increased likelihood of being eligible for and choosing PD, and positively impacts psychosocial outcomes. 14,18 Our study extended these findings and showed that support is also associated with a reduction in the risk of a transfers to HD. Lobbedez et al. found home care–assisted PD (assistance provided by the programme or family) also reduced the risk of transferring to HD in France (cause-specific relative hazard: 0.85; 95% CI (0.77, 0.95)). 15 Szeto et al. also reported that social support independently predicted a transfer to HD in prevalent Chinese patients on PD that were followed for 1 year. 16 Although statistically significant, the effect size was small (HR close to 1). Although findings from both studies suggested that support is important in reducing the risk of transferring to HD, the magnitude of the observed benefit was lower than in our study. This may be attributed to the more inclusive definition of support used in our study.
The reason for this survival benefit is important to consider. Family support has been shown to alleviate some of the burden that comes with PD therapy. 18 The type of support, the support needs of a patient, the availability of support resources and caregiver factors may all influence the impact of support on the outcomes of patients treated with PD over time. Support people do more than instrumental tasks for people on PD including developing confidence with self-care, 24 improving treatment- and medication-adherence, as well as providing nutritional support. 25,26 It is also possible that our findings are not reflective of support ‘tasks’, but rather reflect the impact of isolation on PD survival.
Enhancing social support may improve clinical outcomes for people on PD. 16,18 As family assistance in PD has been declining over the last decade, 27 it would be important for future work to understand how holistic supportive care can be optimised within PD programmes. In Alberta, people on PD have the support of a multidisciplinary healthcare team, and Calgary has a home care–assisted PD programme. Psychosocial resources (e.g. psychosocial educational programs, addressing family burden and patient self-esteem) and other support interventions are important parts of routine care in PD programs. 18 It is therefore important to have someone available to perform the PD procedure (i.e. instrumental support) when needed, but also to identify other support needs and provide individualised support in other forms.
We found that the availability of support was protective between 3 and 12 months, but not at earlier or later time points after the initiation of PD. This is in contrast to other studies which found that this relationship was consistent throughout the study period. 15,16 Support needs for people on PD are greater when starting therapy, when PD begins to fail, or as patients develop more complex medical needs. It is possible that during these times, support needs become greater and may explain the lack of a protective effect of support in the first 3 months of PD and after 12 months. The magnitude of effect support has on PD survival is known to vary by specific reasons for HD transfers. 28 Many reasons that patients experience transfers to HD also vary over time. Mechanical PD catheter complications are a common cause of early failure and inadequate dialysis becomes more likely later in the treatment course. It may be that certain types of complications were less likely to be prevented or overcome with support and because of their timing, may explain why an available support person in the home was not associated with an impact on the risk of transferring to HD in the first 3 months or after 1 year on PD.
No baseline characteristics were associated with a transfer to HD other than support. This remained true in a sensitivity analysis where support was removed from the model. This differs from other studies in which transfers to HD were associated with increased age, 29,30 diabetes mellitus 30,31 coronary artery disease 31 ; high BMI, 32 lower residual kidney function, 32 previous HD treatment, 29,30 male sex 29 and being widowed, divorced or separated. 31 It is possible that our study was underpowered to detect further associations. We also lacked data on certain baseline characteristics (e.g. residual urine output) which may have impacted our results. However, there is high variability in data quality and the definitions used to define a transfer to HD across studies. 20,33 This may have influenced the observed risk and factors that are associated with it. Though it is recognised that these limitations may reduce generalisability to areas with different support structures, this was a relatively large study (compared to other non-registry-based PD studies) and we found the availability of a support person in the home to be important.
This study was conducted using high quality data from a large, primary clinical data set. This enabled us to understand reasons patients transferred to HD on a granular level, gave us the ability to identify the availability of a support person in the home and incorporate other important variables into our model. However, this study is not without limitations. First, support was only captured at baseline and not tracked throughout the course of PD therapy. Support people may no longer be able or willing to provide support at different points throughout therapy, which would have been important to capture and may have impacted the measure of effect, especially at later time periods. We were unable to capture support needs over time and what type of support was being provided at different points in therapy. We were also unable to analyse the relationship between support and specific reasons for HD transfers in separate models, as the number of patients experiencing the individual reasons for transferring to HD were small. Although we know support is essential for people on PD, the breadth and nature of support that support people in the home provide would have been important to capture to begin to identify what elements of support have the most impact. This would be important to understand as they pertain to the specific reasons patients transfer to HD as well as the root causes of these transfers.
In summary, transfers to HD remain a concern for many PD programs. Support is associated with a reduced risk of a transfer to HD, but this effect may be restricted to the time period between 3 and 12 months. A more nuanced understanding of support is required to better understand this issue, specifically clarifying the reasons for lack of earlier and later benefit. Future work should also focus on the development of a standardised definition of support as part of the PD assessment, effective tracking of support needs and interventions overtime, and an understanding of their impact on specific reasons for HD transfers.
Supplemental material
Supplemental Material, sj-docx-1-ptd-10.1177_08968608221086752 - The availability of support and peritoneal dialysis survival: A cohort study
Supplemental Material, sj-docx-1-ptd-10.1177_08968608221086752 for The availability of support and peritoneal dialysis survival: A cohort study by Danielle E Fox, Kathryn M King-Shier, Matthew T James, Lorraine Venturato, Alix Clarke, Pietro Ravani, Matthew J Oliver and Robert R Quinn in Peritoneal Dialysis International
Footnotes
Authors’ note
RRQ and MJO are co-inventors of the Dialysis Measurement and Analysis System and receive speaking fees from Baxter Healthcare. MTJ was the primary investigator of an investigator-initiated research grant from Amgen Canada outside the submitted work.
Acknowledgement
The authors are grateful to the front line staff in the Alberta kidney programs who conducted data collection.
Ethical approval
This study received approval from the Conjoint Health Research Ethics Board of the University of Calgary (REB17-1247).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
