Abstract
Background:
This retrospective cohort study investigated the characteristics and outcomes of the end-stage kidney disease (ESKD) patients treated with incremental peritoneal dialysis (PD) at a large academic centre.
Methods:
ESKD patients initiating PD with a dialysate volume ≤6 L/day were analysed.
Results:
One hundred and seventy-five patients were included and were followed up for 352.6 patient-years. The baseline residual kidney function (RKF) was 8.3 ± 3.4 mL/min/1.73 m2. The unadjusted 1- to 5-year patient survival rate was 89.6%, 80.4%, 65.4%, 62.7% and 48.8%, respectively, and the corresponding time on PD therapy rate was 95.1%, 89.1%, 89.1%, 82.4% and 77.6%. Greater initial PD dose (hazard ratio = 1.608, 95% confidence interval 1.089–2.375) was associated with death after adjusting for age, Charlson comorbidity index (CCI), haemodialysis prior to PD, assisted PD and baseline RKF, likely as a result of residual confounding. There was no association with PD discontinuation. The average peritonitis rate and hospitalisation rate were 0.122 and 0.645 episodes per patient-year, respectively. The dialysate volume increased from 4.5 (4.3–5.7) L/day to 8.0 (6.0–9.8) L/day at 5 years. Fifty-seven (32.6%) patients graduated to full-dose PD at a median time of 10.3 (6.2, 15.7) months. Male sex, greater body mass index and lower baseline serum albumin were risk factors for increasing PD dose to over 6 L/day within 1 year.
Conclusions:
Incremental PD is a safe approach to initiate dialysis, and it offers satisfactory outcomes. Close monitoring, comprehensive evaluation of clinical responses and prompt adjustment of the prescription as needed play a crucial role in this patient-centred treatment.
Introduction
Incremental dialysis refers to the strategy to start dialysis in incident end-stage kidney disease (ESKD) patients with residual kidney function (RKF) at a lower dose than is typically prescribed for anuric patients and then increase the dose as RKF deteriorates over time. This has been practiced in both peritoneal dialysis (PD) and haemodialysis (HD). 1
Incremental PD has now attracted widespread interest. In Italy, the proportion of incremental PD patients increased from 11.5% to 27.5% over a decade. 2 Recent single centre studies from Canada and Korea both reported the use of incremental PD accounting for half of the incident patients. 3,4 It may reduce glucose exposure, cost and burden to the patients and caregivers, increase PD adoption and patient adherence and improve quality of life. Current evidence suggests that incremental PD is associated with a lower peritonitis rate, attenuated RKF loss and at least similar patient survival and time on PD therapy relative to full-dose PD. 3 –6 However, most of the studies were observations with limited sample size.
Incremental PD has been implemented at the Home Peritoneal Dialysis Unit (HPDU) of Toronto General Hospital as an important part of patient-centred care, and it has involved a large number of ESKD patients. This study was designed to analyse the characteristics of the patients and their outcomes.
Methods
Study design
This was a single centre, retrospective, cohort study. Incident ESKD patients who initiated home PD at the HPDU between 1 January 2013 and 31 December 2017 were reviewed, and those starting with incremental PD were identified. Patients were excluded from the analysis if they were treated with PD for acute kidney injury, non-kidney indications or as an approach of palliative therapy. The study was approved by the Research Ethics Board of University Health Network (approval number: 18-5320) and was performed in accordance with the Helsinki Declaration.
Incremental PD was defined regardless of continuous ambulatory PD (CAPD) or automated PD (APD) if any of the following criteria were met: (1) the daily dialysate volume, literally the daily amount of PD fluid dwelled in a patient’s peritoneal cavity, was no more than 6 L/day; (2) PD for fewer than 7 days a week. In contrast, full-dose PD was termed as regimens performed 7 days a week with daily dialysate volume over 6 L.
There were no preset criteria for the assignment of incremental prescriptions, although residual glomerular filtration rate (GFR) and urine volume were approximate guides. There was also increasing comfort over time with initiating dialysis with an incremental regimen.
Demographic and anthropometric features, causes of ESKD, comorbidity status, laboratory parameters, estimated GFR (eGFR) calculated by chronic kidney disease epidemiology collaboration equation and urine output before dialysis, and PD prescriptions at initiation of home PD were collected. PD modality was categorised as CAPD, nocturnal intermittent PD (NIPD), continuous cyclic PD (CCPD) and enhanced CCPD (E-CCPD). Assisted PD was defined as PD implemented by a visiting nurse. The incremental PD patients were compared with the counterparts on full-dose PD in respect to the baseline characteristics.
The incremental patients were followed up until death, switching to permanent HD, kidney transplant, loss of follow-up or to 31 December 2018. The primary outcome was patient survival. The secondary outcomes included time on PD therapy, peritonitis rate and hospitalisation rate. PD discontinuation was defined as PD cessation for more than 3 months due to PD-related complications. Changes of PD dose and time before transition to full dose were recorded.
Statistical analysis
Data are reported as absolute number (percentage) for categorical variables, mean ± SD for normally distributed continuous variables and median (interquartile range) for nonnormally distributed continuous variables. Chi-squared test, Student’s t-test or Mann–Whitney U-test were conducted to compare the incremental PD patients with the full-dose PD patients where appropriate. Pearson correlation analysis was used to investigate the correlation between variables. Multivariate Cox regression analysis was employed to explore risk factors of death and PD discontinuation in incremental PD patients. The patients were dichotomised according to whether they increased dialysate volume to over 6 L/day at 1 year after PD initiation, and the baseline characteristics of the subgroups were compared. Multivariate logistic regression was used to identify determinants of increasing PD dose to over 6 L/day within 1 year. The curve showing the proportion of patients stepping up to full-dose PD was generated by the Kaplan–Meier method. Statistical analyses were performed using SPSS, version 25 (IBM Corp., Armonk, New York, USA). A two-tailed p-value <0.05 was considered statistically significant.
Results
Baseline characteristics of the incremental PD patients
A total of 290 patients commenced home PD during the period investigated, amongst whom 205 patients were eligible for analysis, including 175 commencing with incremental PD and 30 with full dose (Figure 1).
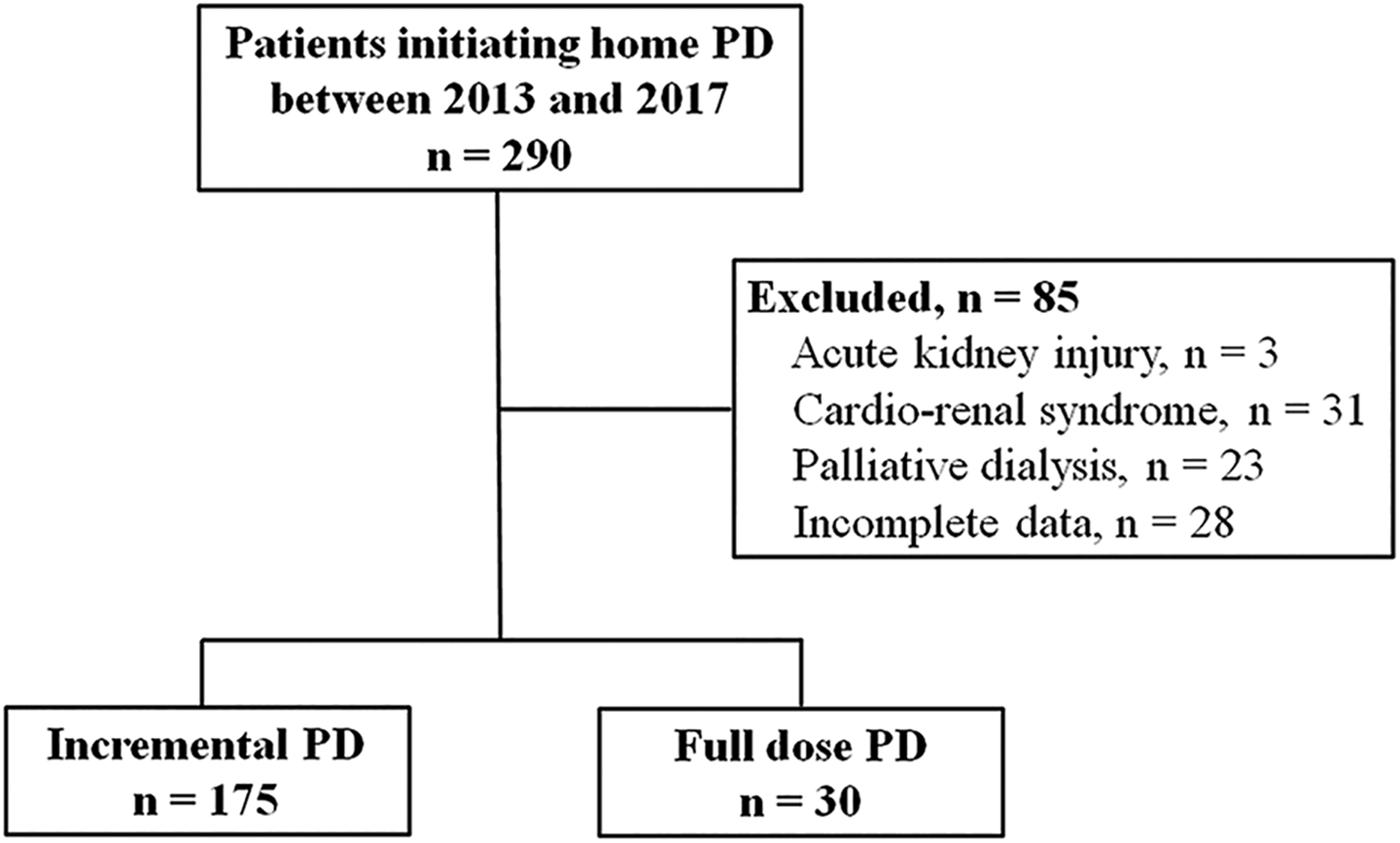
Inclusion of the study subjects.
Compared to full-dose patients, there were more Asians (24.0% vs. 6.7%, p = 0.033) but fewer of African descent (13.7% vs. 36.7%, p = 0.002) among the incremental patients (Table 1). They were comparable in age, sex, body weight, body mass index (BMI), underlying ESKD causes and comorbidity status except peripheral vascular disease (PVD) which was more common in the incremental cohort (14.3% vs. 0.0%, p = 0.030; Table 1).
Characteristics of incremental and full-dose PD patients.
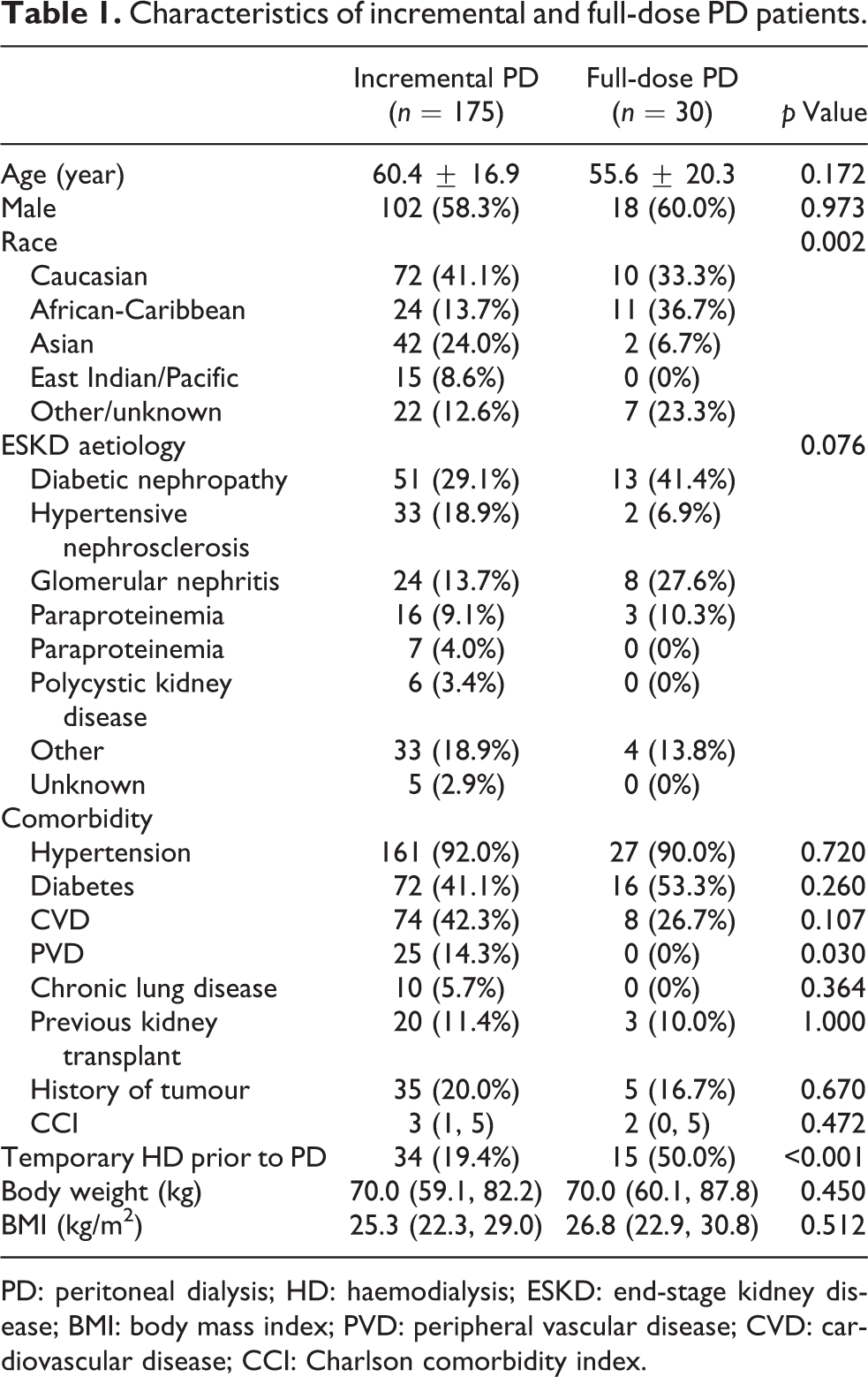
PD: peritoneal dialysis; HD: haemodialysis; ESKD: end-stage kidney disease; BMI: body mass index; PVD: peripheral vascular disease; CVD: cardiovascular disease; CCI: Charlson comorbidity index.
The incremental patients showed lower serum creatinine (613 ± 227 vs. 988 ± 421 µmol/L, p < 0.001) and phosphate (1.66 ± 0.45 vs. 2.02 ± 0.66 mmol/L, p = 0.009), superior eGFR (8.3 ± 3.4 vs. 5.5 ± 3.2 mL/min/1.73 m2, p < 0.001) and greater urine volume (1.5 ± 0.7 vs. 1.0 ± 0.9 L/day, p = 0.001) at PD commencement and they were less likely to undergo HD prior to PD (19.4% vs. 50.0%, p < 0.001). However, the groups shared similar haemoglobin, serum albumin, potassium and bicarbonate (Table 2).
Laboratory parameters and RKF of incremental and full-dose PD patients at initiation of dialysis.
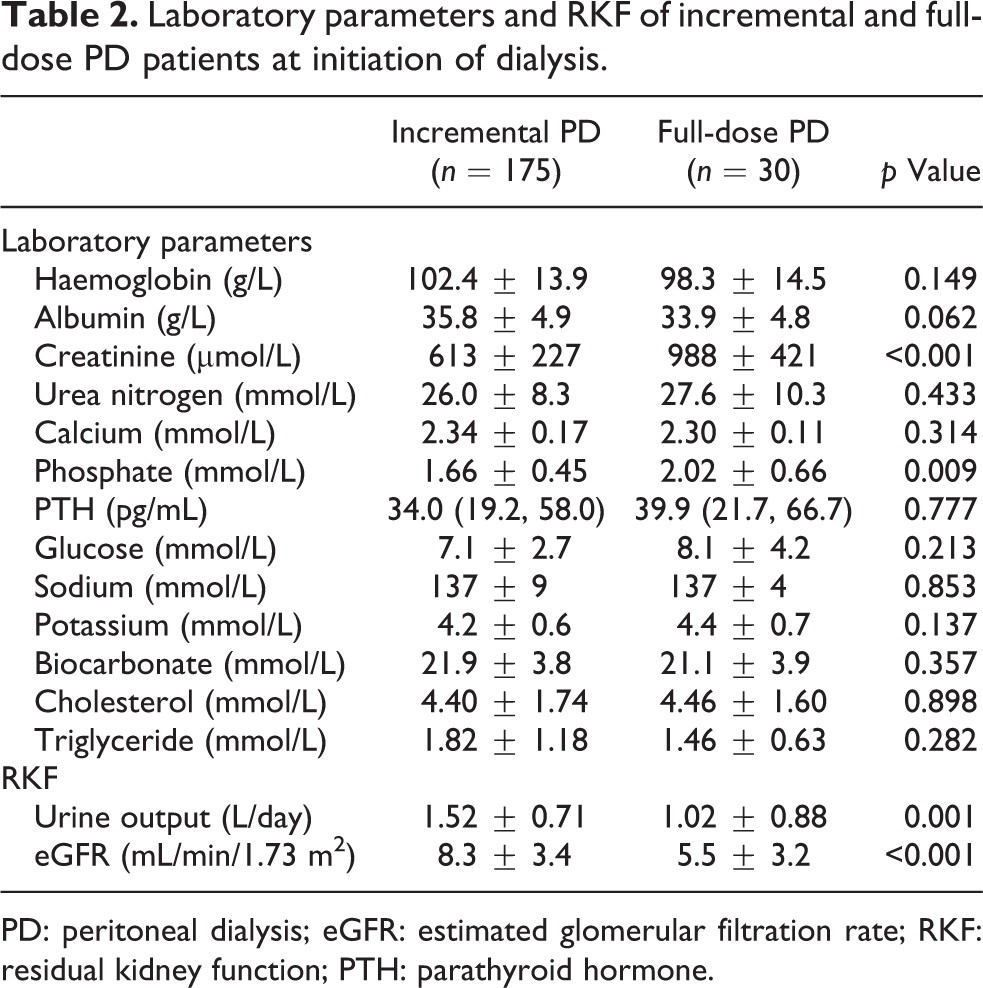
PD: peritoneal dialysis; eGFR: estimated glomerular filtration rate; RKF: residual kidney function; PTH: parathyroid hormone.
Initial home PD prescriptions are summarised in Table 3. For incremental patients, the median dialysate volume at initiation was 4.5 (4.3–5.7) L/day. Among those undergoing CAPD, 4 patients had two manual exchanges, 48 had three and 17 had four. For APD, the number of cycles per session was three in 85 patients and four in 21 patients, respectively. There were two NIPD patients who underwent dialysis fewer than 7 days a week.
Initial home PD prescriptions of incremental and full-dose PD patients.
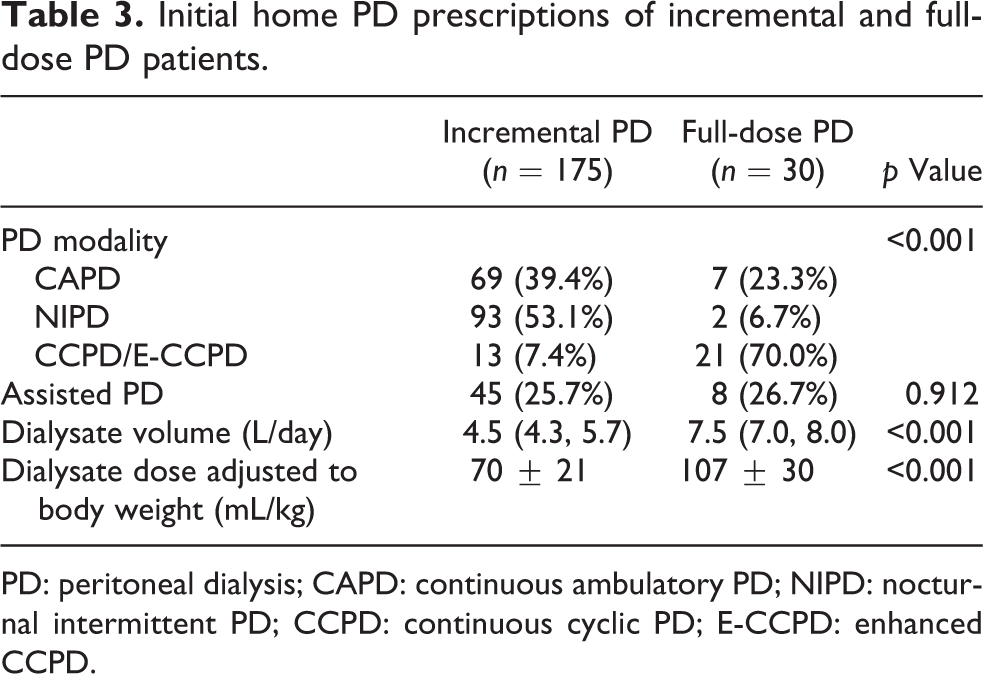
PD: peritoneal dialysis; CAPD: continuous ambulatory PD; NIPD: nocturnal intermittent PD; CCPD: continuous cyclic PD; E-CCPD: enhanced CCPD.
Primary and secondary outcomes
The incremental cohort was followed up for 352.6 patient-years. During the follow-up, 42 patients deceased, 19 patients encountered PD discontinuation, 20 patients had kidney transplantation and 4 patients transferred to other centres.
The unadjusted patient survival rates through 1–5 years on PD were 89.6% (95% confidence interval (CI) 84.9–94.3%), 80.4% (95% CI 73.7–87.1%), 65.4% (95% CI 55.2–75.6%), 62.7% (95% CI 51.5–73.9%) and 48.8% (95% CI 31.9–65.7%), respectively, and the corresponding time on PD therapy rates were 95.1% (95% CI 91.8–98.4%), 89.1% (95% CI 83.4–94.8%), 89.1% (95% CI 83.4–94.8%), 82.4% (95% CI 72.0–92.8%) and 77.6% (95% CI 64.3–90.9%). The overall peritonitis rate was 0.122 (95% CI 0.066–0.180) episodes per year. The average hospitalisation rate was 0.645 (95% CI 0.476–0.813) episodes per year, and the mean duration of hospitalisation was 5.9 (95% CI 4.7–7.1) days/year.
Amongst the deceased, cardiovascular death and infection-related death occurred in 20 and 8 patients, respectively. Another seven patients terminated PD to receive palliative treatment and died shortly. The remaining seven patients died from other or unknown causes. Twenty-seven patients died before their PD dose could exceed 6 L/day. Older age (1 year, hazard ratio (HR) 1.046, 95% CI 1.012–1.080), assisted PD (HR 2.563, 95% CI 1.212–5.421) and greater initial PD dose (1 L/day, HR 1.628, 95% CI 1.071–2.474) were associated with mortality after adjustment for sex, baseline RKF, cardiovascular disease, PVD, chronic lung disease, CCI, HD prior to PD and baseline serum albumin.
The causes of PD discontinuation included peritonitis (n = 4), non-compliance to PD treatment (n = 4), inadequate solute removal (n = 3), dialysate leak (n = 2), intraabdominal complications (n = 2), catheter dysfunction (n = 1), ultrafiltration failure (n = 1) and other causes (n = 2). Among these patients, four developed PD discontinuation after their PD dose was increased to more than 6 L/day, including two cases due to underdialysis, one case due to catheter dysfunction and one case of rectal tumour. Serum albumin was the only factor associated with PD discontinuation (1 g/L, HR 0.890, 95% CI 0.809–0.979) after adjustment for sex, age, baseline serum creatinine and RKF.
Changes of PD dose
At PD initiation, there was no significant correlation between PD volume and body weight when the incremental and full-dose groups were pooled together (p = 0.216). The duration before transition to full dose of the incremental cohort was 15.0 (8.1, 26.2) months. The daily dialysate volume was 5.4 (4.5–6.0) L, 6.0 (4.5–7.0) L, 6.0 (4.8–8.0) L, 7.0 (5.7–9.8) L and 8.0 (6.0–9.8) L at 1–5 years on PD, respectively (Figure 2). Fifty-seven (32.6%) patients increased their PD dose to over 6 L/day (Online Supplemental Figure S1) with a time of 10.3 (6.2, 15.7) months, among which 34 (19.4%) patients did this within 1 year after PD initiation. Those transitioning to full-dose PD within 1 year were more likely to be male (79% vs. 53%, p = 0.005), of greater BMI (28.6 ± 6.9 vs. 25.6 ± 5.6 kg/m2, p = 0.007), with lower serum albumin (34.0 ± 4.4 vs. 36.2 ± 4.9 g/L, p = 0.023) but higher serum creatinine (706 ± 274 vs. 591 ± 209 µmol/L, p = 0.018), glucose (8.0 ± 3.4 vs. 6.9 ± 2.6 mmol/L, p = 0.045) and initial daily dialysate volume (4.7 (4.5, 6.0) vs. 4.5 (4.3, 5.4) L/day, p = 0.019; Online Supplemental Table S1). Multivariate analysis showed that male sex (OR = 3.338, 95% CI 1.240–8.984), BMI (1 kg/m2 increase, OR = 1.117, 95% CI 1.039–1.202) and serum albumin (1 g/L increase, OR 0.902, 95% CI 0.829–0.983) at PD initiation were independently associated with PD dose increased to over 6 L/day within 1 year (Online Supplemental Table S2).
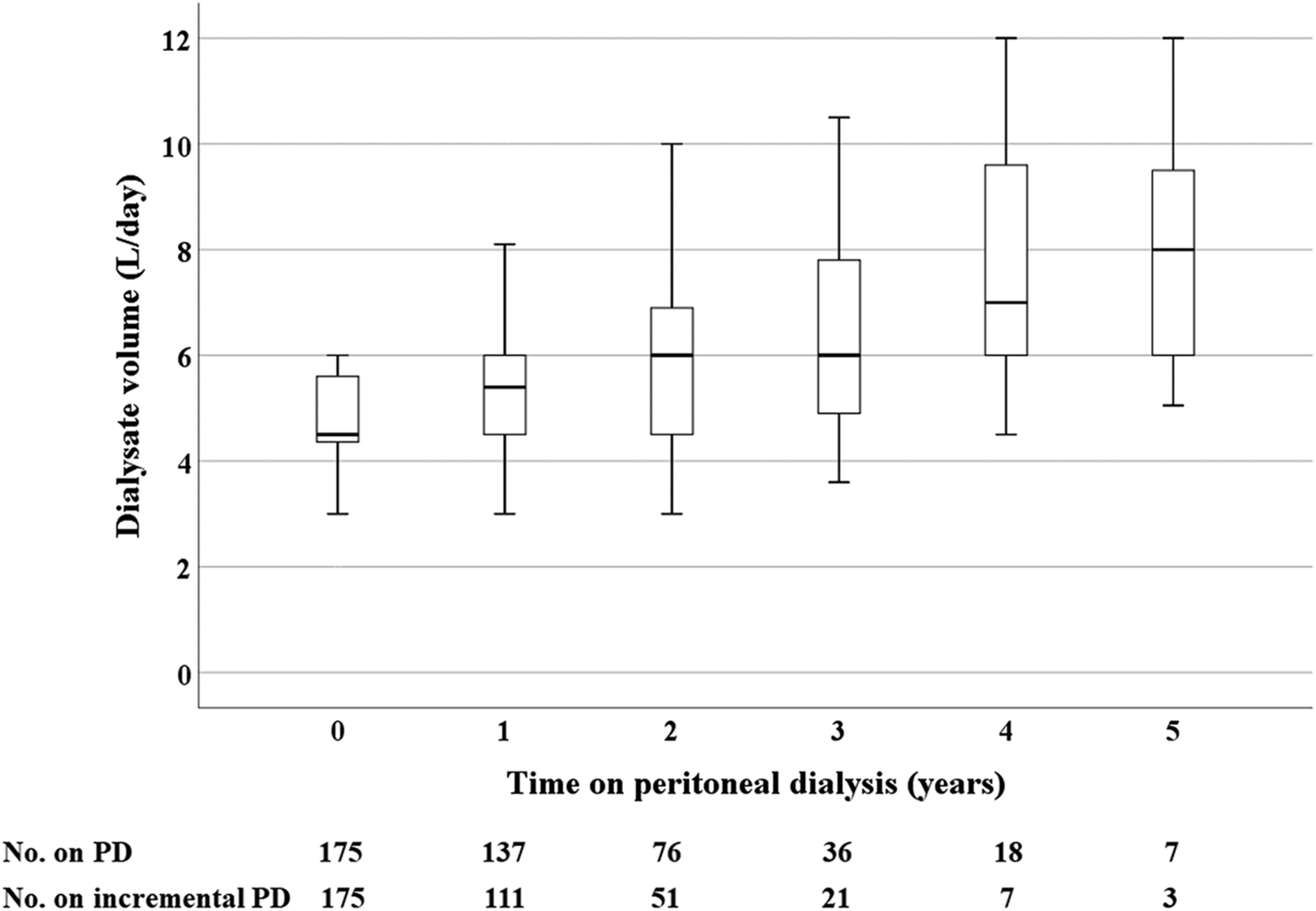
Changes of dialysis dose over time.
Discussion
The present study included one of the largest incremental PD cohorts up to date. This strategy has become a predominant modality to treat incident ESKD patients at our centre, and it is capable to confer excellent outcomes. It is also of particular relevance given the multi-ethnic background of the study.
The optimal timing to start chronic dialysis for ESKD patients remains unaddressed. The Initiating Dialysis Early and Late study has justified initiating dialysis at GFR levels of 5–9 mL/min/1.73 m2 with uraemia or overhydration symptoms. 7 –9 A prospective cohort study from Korea suggests that it is reasonable to start PD when GFR is between 5 mL/min/1.73 m2 and 10 mL/min/1.73 m2. 10 Previous studies also take RKF into account to determine eligibility for incremental PD, and there is great overlap between the RKF permitting incremental PD and that for full-dose PD. 5,11,12 Similarly, our cohort presented with an average eGFR of 8.3 mL/min at PD commencement. This disagrees with the assumption that the increasing trend of incremental dialysis is a result of early initiation of chronic dialysis. Therefore, the incremental strategy is feasible for a considerable proportion of the typical incident ESKD patients in current clinical practice.
Incremental PD protocols vary among reports, 2 –6,11 –16 and the recent International Society for Peritoneal Dialysis (ISPD) guideline establishes the definition of incremental PD and summarises the common prescriptions. 17 The significant heterogeneity leads to difficulties in comparison among studies, and demarcation merely based on the number of manual exchanges or the frequency of cycler use may confuse the PD dose. We define incremental PD essentially based on dialysate volume, which is more easily measured regardless of PD modality. For instance, three 1.5-L exchanges CAPD and three 1.5-L dwells NIPD were the most typical initial prescriptions, both providing a dialysate volume of 4.5 L/day; in a minority of patients undergoing CCPD, three nocturnal exchanges and a day dwell of 0.5–1.5 L were prescribed, and the dialysate volume ranged from 3.5 L to 6 L. Surprisingly, we observed that a significant number of patients were stable for years with dose no more than 6 L/day.
Previous small-scale studies reported that incremental PD could postpone the use of full dose for 6–24.5 months. 1,15 The vintage in the present study was moderate. Nearly one-third of the patients increased the dose to over 6 L/day during the follow-up. Decline rate of RKF is an essentially unmodifiable factor associated with the time before transition to full dose. Although we did not measure RKF deterioration over time, to the best of our knowledge, we are the first to report the independent predictors of transition to full dose within 1 year after PD initiation including male sex, higher BMI and lower serum albumin. This implies that close monitoring is particularly important when incremental PD is prescribed to certain subgroups of patients.
In contrast with a historical cohort from our centre before the incremental era, 18 the present cohort had more favourable time on PD therapy and achieved a much lower peritonitis rate although this likely reflects a secular trend, that is, these metrics were improving over time. According to the case-by-case analysis in the present cohort, PD discontinuation that occurred during incremental PD were rarely related to low PD dose, except in a polycystic kidney disease patient who could not tolerate a larger dwell volume and experienced underdialysis as his RKF declined. Similarly, a recent study from the UK reported that reduced PD intensity was independently associated with superior time on PD therapy through the first 3 years on PD. 19
Patient survival within 2 years was almost identical between the present and prior cohorts 18 ; however, the 5-year patient survival was numerically lower for the incremental cohort (49% vs. 61%). Previously, comparable long-term survival among incremental and full-dose PD patients were reported. 4,5 We consider a heavier comorbidity burden carried by the incremental cohort, with more prevalent diabetes and history of tumors, might be an explanation to the suboptimal long-term survival in the present study, and our results need further study to be validated.
Conventionally, PD dose is adapted based on small solute clearance or in proportion to the reduction in GFR. 14,15 Kt/V has been abandoned at the HPDU because of its obscure role in validating dialysis adequacy and predicting outcomes, 20 and RKF assessment based on 24-h urine collection is cumbersome for patients. Instead, we monitor the patients closely and determine whether to increase dialysis dose primarily according to a comprehensive evaluation focusing on patient-reported outcome measures regarding clinical well-being, correction of anaemia, malnutrition, mineral disorders and metabolic acidosis as well as diuresis and volume status, a way that has been advocated by the recent ISPD guideline. 21 All the patients are well-informed of the principles of incremental dialysis, and prescription is adjusted accordingly. As we demonstrated, older age and assisted PD independently predicted death. These patients tended to be frail, with multiple comorbidities as well as inferior socio-economic status and family support, which are related to adverse outcomes. Although greater initial daily contact PD volume was independently associated with risk of death, it is likely a result of residual confounding.
The present study has several limitations. Firstly, this was a retrospective, single-centre, observational study with the absence of a control arm, and there were drawbacks inherent in the design. It is difficult to conduct randomised controlled trials to compare incremental and full-dose PD, and relevant evidence is mainly obtained from observations. There was a contemporaneous group of patients initiating PD with full dose; however, they were highly different from the study cohort and the patient number was quite small. Consequently, we compared only the characteristics of the groups at the beginning of PD but not the outcomes. Secondly, there could be significant confounding in the study. Although we intended to minimise the discrepancy in dialysis prescription, the decision of initial PD prescription depended on a number of clinical factors and it was finally at the discretion of different physicians. Some factors potentially associated with outcomes, for example, inflammation status, were not assessed. RKF was estimated according to the approximate urine output and serum creatinine during follow-up, therefore the longitudinal profile of RKF remained indistinct. Finally, although we suspect that a reduction in PD prescription would be associated with improvement in some spheres of quality of life, it was not formally evaluated in this study.
In conclusion, this relatively large cohort study demonstrates that incremental PD is applicable to many, if not the majority, of incident ESKD patients with RKF. It allows individualised PD modality and prescription, eases the workload on patients or the caregivers, possibly improves the quality of life and offers satisfactory prognosis. Combining with advances proved by other studies, for example increasing PD adoption, preserving RKF, reducing glucose exposure and cost-effectiveness, it is a safe scheme to initiate dialysis. A new approach to incremental PD that focuses on clinical responses rather than achieving a specific Kt/V urea target can be successful, and close monitoring of a patient’s general well-being, RKF, overall laboratory assessments and prompt adjustment as indicated take a crucial role in this patient-centred treatment.
Supplemental material
Supplemental Material, sj-pdf-1-ptd-10.1177_08968608211036796 - Incremental peritoneal dialysis in incident end-stage kidney disease patients
Supplemental Material, sj-pdf-1-ptd-10.1177_08968608211036796 for Incremental peritoneal dialysis in incident end-stage kidney disease patients by Hao Yan, Zita Abreu and Joanne M Bargman in Peritoneal Dialysis International
Footnotes
Acknowledgement
We would like to thank PD nursing staff of Home Peritoneal Dialysis Unit of Toronto General Hospital for their work.
Author contributions
JB conceived the study. JB and YH participated in protocol development, gaining ethical approval, and data collection and analysis. ZA was involved in data maintenance. YH wrote the first draft of the manuscript. All authors reviewed the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Joanne Bargman has served as a consultant for DaVita Healthcare Partners and Baxter Healthcare Canada.
Ethical approval
Ethical approval for this study was obtained from the Research Ethics Board of University Health Network (approval number: 18-5320). The study was performed in accordance with the ethical standards of the responsible committee and with the Helsinki Declaration.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
