Abstract
Background:
While numerous studies have explored the patient experience of dialysis or other end-stage kidney disease (ESKD) treatments, few have explored the process of transitioning between dialysis modalities. This study aimed to develop an in-depth understanding of patient and caregiver perceptions and experiences of the transition from peritoneal to haemodialysis (HD) and to identify ways in which transitions can be optimised.
Methods:
Fifty-four in-depth, semi-structured interviews were undertaken at six study sites across the West Midlands, UK (n = 23), and Queensland, Australia (n = 31). Thirty-nine participants were patients with ESKD; the remainder were family members. An inductive analytical approach was employed, with findings synthesised across sites to identify themes that transcended country differences.
Results:
Of the 39 patient transitions, only 4 patients reported a wholly negative transition experience. Three cross-cutting themes identified common transition experiences and areas perceived to make a difference to the treatment transition: resistance to change and fear of HD; transition experience shared with family; and bodily adjustment and sense of self.
Conclusion:
Although each transition is unique to the individual and their circumstances, kidney care services could optimise the process by recognising these patient-led themes and developing strategies that engage with them. Kidney care services should consider ways to keep patients aware of potential future treatment options and present them objectively. There is potential value in integrating expert support before and during treatment transitions to identify and address patient and family concerns.
Keywords
Introduction
Patients with end-stage kidney disease (ESKD) may be treated using home therapies (home haemodialysis (HD) or peritoneal dialysis (PD)), incentre HD, transplantation or conservative care. Many patients with ESKD progress through a series of kidney replacement therapies due to changing clinical needs, treatment complications or patient preferences. 1,2 The change from PD to other modalities is particularly common, 3 with a third of patients moving to another form of dialysis (usually HD) within their first 3 years on PD. 4 –6 Transitions from PD may be characterised broadly as planned, where PD effectiveness declines over time until it is no longer viable, or unplanned, where – usually due to infection or peritoneal membrane failure – patients without a stable fistula require emergency transfer to HD. 7,8 Changing dialysis modality requires physical and psychological adaptations which may be associated with substantial distress, 9,10 and disruption to lifestyles and relationships. 11 Evidence suggests the transitional period (weeks before to months afterwards) may also entail heightened morbidity and mortality risk compared with other points in the ESKD pathway. 12 While numerous studies have explored the patient experience of dialysis or other ESKD treatments, 13 few have explored the process of transitioning between dialysis modalities. Understanding patient and caregiver perspectives about treatment transitions is essential towards ensuring such transitions are effective and patient care is optimised. 4,14,15
This study is part of the INTEGRATED consortium 12 and aimed to develop in-depth understandings of patient and caregiver experiences of transitions from PD to HD, barriers and facilitators to successful transitions and views about how clinical practice could improve.
Methods
The published protocol summarises the methods. 12 Briefly, in-depth, semi-structured interviews were undertaken at three study sites in the West Midlands, UK, and three in Queensland, Australia. Incorporating perspectives across two continents allowed identification of treatment transition experiences likely to have relevance across different national contexts. Ethical approval was obtained from the Health Research Authority (Ref: 237901) and London Bridge Research Ethics Committee on 31 May 2018 (Ref: 18/LO/0974). Research Governance approval (Ref: RG_17-252) was obtained from each participating study site in England and Australia.
Participants and recruitment
Eligible patients were identified by clinical staff and were aged 18 and over, had transitioned from PD to incentre HD for at least 30 consecutive days between 2 and 18 months previously, and were clinically stable. Convenience sampling was used, with potential participants approached sequentially as they became eligible. Study packs containing an invitation letter, Participant Information Sheet and consent form were posted to eligible patients 7–14 days before their next outpatient appointment. Those who wished to participate gave permission for their details to be passed to the research team who then arranged an interview. Where possible, adult caregivers (all of whom self-identified as ‘family members’ – spouses, parents or children) providing support to a transitioning patient were also recruited, either at the patient’s clinic appointment if accompanying them or via a carer pack given to the patient. One participant identified did not speak English. This participant was approached and interviewed through a translation service.
Data collection
Interviews were conducted face-to-face at a participant’s home or their dialysis unit, or via telephone. Detailed interview topic guides for patients and caregivers are included as supplementary material. Patients and family members were interviewed separately if possible, although four interviews were undertaken jointly. The interview topic guide sought to understand participants’ experience of the transition from PD to HD, including what they knew about the change, why it was happening and the discussions they had before and after the transition and with whom. Interviews focused on patients’ most recent transition, but those who had experienced previous transitions were encouraged to reflect on how those experiences were similar to or different from their most recent transition. All interviews were audio-recorded (range 15–60 min) and independently transcribed verbatim, and transcripts were proofread against original recordings to ensure accuracy. The same researchers (KA and KS) undertook the England and Australia interviews. All participants provided written informed consent.
Analysis
An inductive analytical approach was employed, without a predetermined coding framework or a priori expectations about study findings. 16 Two researchers (KA and KS) independently analysed 10–15% of the transcripts to create an initial coding framework, using thematic analysis. 17 Data that did not fit existing codes or themes were discussed by the research team and amendments made or new codes added until all data had been analysed. Analysis was initially undertaken separately for England and Australia by two researchers (KA and KS). Themes were then compared across the two nations and discussed in the research team in order to identify themes that transcended country differences.
Results
Fifty-four individuals participated in total (23 in England and 31 in Australia). Fifteen were family members; the remaining 39 were patients with ESKD (Table 1). Clinical information such as the cause of patients’ kidney disease, comorbidities or diabetic status was not available unless patients described this during their interview.
Characteristics of study participants.

a Age group and ethnicity were not recorded for family members who participated in the study.
b‘Other’ ethnic group in England = Hispanic.
c‘Other’ ethnic group in Australia = Hispanic (n = 1); Indigenous Australian (n = 1); Middle Eastern (n = 1).
Transition type
Four discrete transition types were identified: (i) planned transition with long lead-in time, allowing opportunity for patient–healthcare professional (HCP) discussion (n = 7); (ii) ‘hybrid transition’ where the transfer between therapies was planned, but the point of transition itself was unplanned/emergency (n = 8); (iii) patient-led transition which could have been planned or unplanned but where the patient requested the treatment change (n = 7), and (iv) unplanned/emergency transition to HD, usually due to peritonitis (n = 17). Despite the different transition experiences observed, of the 39 ‘patient journeys’ assessed, most participants (n = 35) described their transition as positive, and only 4 patients reported a wholly negative experience. A number of patients with broadly positive transitions also described some negative elements to their treatment change.
Interview themes
Interview data illustrate that each experience of transition is unique to each patient’s individual situation. Despite this, three themes identified common transition experiences and areas that interviewees believed affected the treatment transition (Table 2). Themes were generated inductively, prioritising the most relevant findings evidenced in both national contexts and across transition types.
Summary of themes and subthemes.
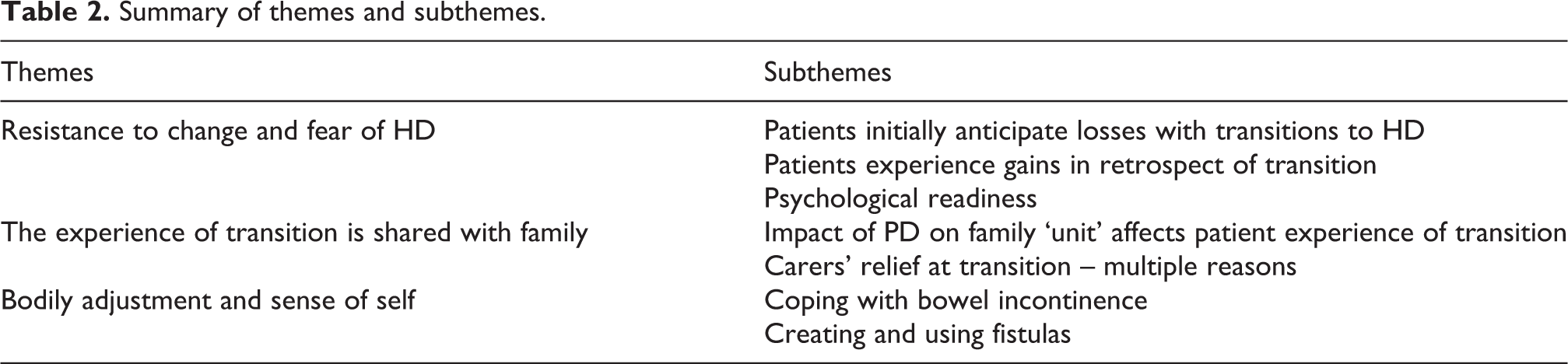
Resistance to change and fear of HD
For most patients, accepting HD was a gradual process. Patients initially anticipated losses of freedom with a move to HD, yet these losses were not borne out for most in practice.
Anticipatory losses
Many patients initially associated HD with negative impacts on their lives, affecting control over treatment (self-care), ability to work, maintaining current lifestyle and entailing an unwanted move away from the privacy of their home environment. Because it’s three days, and they told me this, three days at the hospital every week you know and that’s why and I didn’t want hospital at all, not at all…. (Site 2, patient) …my main concern throughout all of this process is still to be able to go to work and like bring money in. (Site 4, patient) I feel well in myself and that but when I first come down here I wished I was dead. I’d had it. Fair enough…I just wished I wasn’t here. I was…didn’t want to do nothing, I couldn’t do nothing, just felt “Why live like this?” (Site 6, patient)
Gains in retrospect
Many patients found HD less challenging than anticipated. Some expressed surprise about this, and even found the move to incentre HD more enabling and less stressful than home treatment. …the fear of having to go out to a unit and thinking I wouldn’t be able to do things my way, but dialysis fits in perfectly with my life the way it is, it’s better for me now than it was when I was doing PD. (Site 1, patient) It’s good (HD) because it gives me some freedom. I do 4 ½ hours three days a week, so that’s 13 hours a week therapy as opposed to 70 or 84 hours a week (on PD). (Site 2, patient) …they (PD staff) seemed satisfied that you’re doing alright when I just wasn’t you know…I remember going down by the river for something to eat and there was this short bank to get back onto like the main drag and I honestly thought I was going to pass out at some stage I’d gone all dizzy and I grabbed hold of [wife] I said I think I’m going to go here. (Site 2, patient) I just don’t see the point in having PD, you have to go in to surgery to get it done and then if you get infected you have get surgery to get it taken out. You get constipated all the time. There’s a lot of things that just don’t make it as good, whereas haemo, I don’t think I’ve had a problem. (Site 5, patient) At first I thought it was my fault with my fluid and when I went on to haemo they went “no you can’t help it it’s because of this, that and the other”. They helped me relax about it all and I think with being so stressed and tense thinking it was me, that wasn’t helping either. (Site 3, patient)
Psychological readiness
Resistance to HD was often linked to phobia of needles. Some patients recounted going against recommendations to transition to HD and extending the duration of their PD to try to avoid it: He [consultant] suggested moving over to HD because he didn’t think the dialysis that I was doing was efficient enough for me, but because of my fear of needles I point blank refused. I’d rather just carry on the way I’m going and increase my hours of dialysis. (Site 3, patient) It was really a shock and psychologically, I reckon they should start talking to you, prepare you for what they’re going to show you, what they’re going to do to you. I don’t know, I felt somebody not from this world, it was just terrible. (Site 5, patient)
The experience of transition is shared with family
During transitions, the experiences of patients and close family members were strongly interconnected. This partner describes how watching her husband’s physical and emotional decline when using PD and subsequent recovery after transition to HD affected her: I notice there’s a big change in him, when he was doing peritoneal he was angry with everybody. But now that he’s doing haemo that has slowly dissipated. That makes me happy because that’s what I strive for, to see him accomplishing things that he wants to do with his life. (Site 5, family member)
Impact of PD on the family ‘unit’ affects patient experience of transition
Where family had felt uncomfortable with home treatment, this mediated how patients felt about a move to HD. One patient described the additional caring responsibility his wife took on after he had a leg amputated, which made PD stressful for her: But when it comes to all the work that she had to do and me, only on one leg she had to help do everything. For her it was too much work and I didn’t feel good about it. (Site 4, patient) It [HD] gives me that space and the break and the times I need to, you know, me and the [0:07:06] on my own. No, it’s been good. He feels a lot better in himself, he looks a lot better in himself too since the change. (Site 4, family member)
Carers’ relief at transition
Many family members were relieved when dialysis moved incentre. For many families, having PD at home medicalised their living environment and they were glad to reclaim their personal space: Once that machine went the thought of it coming back it filled me with dread. I used to do the order and stock take, having it there every single time we pulled on the drive the boxes were there so you’re reminded, every single day of what’s happening. (Site 2, family member)
Bodily adjustment and sense of self
Coping with bowel incontinence and creating and using the fistula for HD were frequently described as problematic, and patients described a negative emotional impact on their sense of self, which in some cases caused disengagement with their dialysis treatment.
Coping with bowel incontinence
Patients described anxiety and shame, and a lack of control in connection to bowel incontinence, especially in public settings like the HD unit or when travelling to these settings in ambulances with others. Although for some patients this issue predated their treatment change, the incidence of bowel incontinence as a new symptom was particularly pertinent during transitions due to changes in medication regimens and had a strong impact for several participants on their perception of their transition: When the diarrhoea gets really bad, it’s just out of control when it happens. Every time I’m nervous, and I’m thinking oh please don’t let it happen…and it worries me so much before I leave home. It’s not a nice thing if you’re putting people off. So that’s why sometimes I don’t come [to HD], because of that reason. (Site 5, patient) At first, the treatment created havoc with my plumbing. I had permanent diarrhoea which was…well occasionally I was coming home soiled because just not able to stop it, just not able to do anything about it. (Site 2, patient)
Creating and using fistulas
Patients who found the creation of fistulas the most traumatic often felt they had little prior warning or education about the need and purpose of the fistula. Some were shocked and experienced additional anxiety because of this. Well I actually had no idea what a fistula was. I thought I was going to have toggles coming out of my arm. So I was a bit, yeah, shocked. (Site 2, patient) Oh my fistula. I had two operations for that, but then it stopped the blood flow to my fingers and my fingers were going really numb and white and everything. I think they’re thinking of putting one in this [other] arm and trying again. (Site 1, patient) Like the changeover [to HD] was just the fistula didn’t work, my arm went black from that from where it blew out. So yeah, I went back on the PD. (Site 4, patient)
Discussion
To our knowledge, this study represents the second, multisite qualitative investigation of dialysis modality transitions and the first to include the perspectives of family members. The broad aim of the programme of research, to which this study contributes, is to generate evidence that can enhance consideration of integrated dialysis pathways. 3 To this end, this study found successful PD to HD transitions require an awareness that treatment change is required, support for the change, sufficient knowledge of what the change will entail and the willingness and capacity to implement the change. This implies that patients should be made aware when beginning PD, that a future change may be required, what the alternatives are, and potential underlying reasons for the transition. 18 In particular, this research shows patients may anticipate losses of freedom and comfort with a move to HD, even when their overall experience of treatment transition was described as positive. Transition experiences were complex, with anticipatory losses often offset by perceived freedom from responsibility for self-care when moving to incentre dialysis. Recent work undertaken in Belgium as part of the INTEGRATED consortium reported similar findings regarding this ‘paradox of control’ by patients during transitions from PD to HD. 19 Another commonality between these two studies is the importance drawn to how previous ‘frames of reference’ can influence the transition experience. 19 In this study, the initial ‘resistence to change’ was often mentioned in relation to previous negative ideas about HD, either gained through direct experience or implied by being guided towards PD initially by HCPs.
Planned transitions and those where patients initiated discussions about changing dialysis treatment were more likely to be viewed positively than those where transitions were unplanned. Expert support during the transition phase may be valuable, either to facilitate timely decisions about transition in planned cases or to help patients and families cope psychologically in emergency transitions. Such support may focus on acceptance of deterioration in their condition; identifying and working with patients with needle phobias, and support for emotionally challenging aspects such as fistula issues. Peer support could also be used to help patients build realistic expectations of the physical and emotional impact of their treatment transition and identify important non-medical issues.
A recent systematic review of dialysis decision-making found that HCPs often focus on biomedical issues and prolonging life. 20 This study captured the anxiety experienced by family carers and subsequent relief at the transition to incentre HD. In many cases, relief was strongly influenced by negative impacts on patient health and well-being caused by the progressive failure of PD which were alleviated by the change to a more clinically effective treatment. Yet impacts on patients’ sense of self were particularly pertinent, and regular discussions between HCPs and patients/family members may help clinicians understand the impact of transitioning between treatments on the day-to-day experience of patients and family members. The example of patient anxiety about bowel incontinence around the time of transition is particularly important – if this may drive disengagement with dialysis, it is fundamental that clinicians are aware of this symptom and its consequences for patients so that information provision and HCP-patient communications can address these issues openly. Treatment modalities should also be discussed against an assessment of patient and family member capacity, 21,22 and the decision to start incentre HD should incorporate shared decision-making that helps patients make informed treatment decisions. 23
Issues raised in this study around the burden of home dialysis for patients and caregivers reflect recent review findings. 24 In particular, patients’ appreciation of gains in retrospect of moving on to HD often displayed a relative lack of confidence of how they were managing PD. The potential for ‘burnout’ is also evident, especially in the narratives of relief expressed by caregivers after their relatives transitioned to HD. It is important to note that the participants reporting their experience in this study were not a representative sample of people using PD. Participants had all transitioned from PD to HD and as such they had either experienced an acute medical emergency or declining clinical outcomes and related symptoms when using PD.
Limitations
Interviewing participants at one time point poses a limitation, as it prevents consideration of the longer-term reflections of patients and caregivers. However, this strategy allowed us to capture a greater number of individual experiences overall, maximising the range of experience within the sample. Recruiting patients who had transitioned from PD to HD over the previous 2–18 months gave a relatively small sampling frame and all patients who gave consent were selected to participate. Thus, while the study captured diversity in individual characteristics, it did not recruit a specific cross-section of participants that could be stratified by age, ethnicity or sex. Within both countries just one region was used for sampling, this may impact on the transferability of findings to other settings. There are limits to the transferability of these findings as all participants, except for one, were English speaking and the study was conducted in high-income countries where PD is practiced by a minority of patients.
Practice implications
To improve treatment transitions, renal care providers should consider ways to improve patient awareness of possible future treatment options and present them objectively by tailoring information and communications accordingly. Patients yet to begin dialysis should be given information about the positive and negative aspects of different treatments to facilitate informed choice about dialysis modality and the knowledge that this may need to change as their ESKD progresses. Patients already receiving PD who are developing problems that might require treatment change in the near future should be offered information about HD and the different models of care that are available. This might involve incentre self-care, incentre shared care, total dependence incentre, home-based self-care such as HD at home or a more intensive search for a kidney donor when transplantation is a viable option. There is also potential value in integrating greater expert support during the transitional period that identifies the concerns of patients and their families.
Conclusions
Patient and carer experiences of transitions from PD to HD are often marked by initial resistance to treatment change; the way the transition experience is shared with family members; and bodily adjustments to treatment transition which can be traumatic. Although each transition is unique to an individual’s own circumstances, renal care providers could optimise the transition process by recognising and developing strategies that engage with these patient-led themes.
Supplemental material
Supplemental Material, sj-docx-1-ptd-10.1177_0896860820975596 - How do patients and their family members experience the transition from peritoneal dialysis to incentre haemodialysis? A multisite qualitative study in England and Australia
Supplemental Material, sj-docx-1-ptd-10.1177_0896860820975596 for How do patients and their family members experience the transition from peritoneal dialysis to incentre haemodialysis? A multisite qualitative study in England and Australia by Kerry Allen, Sarah L Damery, Kim Sein, David W Johnson, Simon J Davies, Mark Lambie, Els Holvoet and Gill M Combes in Peritoneal Dialysis International
Footnotes
Authors’ note
This study is part of the international INTEGRATED consortium with (in alphabetical order): C Chan, G Combes, S Davies, F Finkelstein, C Firanek, R Gomez, K Jager, V George, D Johnson, M Lambie, M Madero, I Masakane, D McDonald, M Misra, S Mitra, T Moraes, A Nadeau-Fredette, P Mukhopadhyay, J Perl, R Pisoni, B Robinson, D Ryu, R Saran, J Sloand, N Sukul, A Tong, C Szeto and W van Biesen.
Acknowledgements
The authors would like to acknowledge those who facilitated data collection in our study sites, particularly Sridevi Govindarajulu and Kenn-Soon Tan in Queensland, Australia.
Author contributions
KA, SLD, KS and GMC made a substantial contribution to the design of the research study and data analysis. KA led the draft with significant input from SD, KS and GC. They have approved the final version for publication. DWJ, SJD, ML and EH made a substantial contribution to the concept or design of the study and revised it critically for important intellectual content. They have all approved the version to be published.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical approval
Ethical approval was obtained from the Health Research Authority (Ref: 237901) and London Bridge Research Ethics Committee on 31 May 2018 (Ref: 18/LO/0974). Research Governance approval (Ref: RG_17-252) was obtained from each participating study site in England and Australia.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Baxter Global Grant Program. The funder had no role in the collection of data, its analysis and interpretation or in the right to approve/disapprove publication of the finished manuscript. SLD and GC were supported by the National Institute for Health Research Collaboration for Leadership in Applied Health Research and Care West Midlands (NIHR CLAHRCWM), now recommissioned as NIHR Applied Research Collaboration West Midlands. The views expressed are those of the authors and not necessarily those of the NIHR or the Department of Health and Social Care. The study was sponsored by University of Birmingham. The study sponsor had no role in study design, in the collection, analysis and interpretation of data, in the writing of the report and in the decision to submit the article for publication.
Informed consent to participate
All participants received an information sheet and gave their informed consent to take part in the study. The signed consent forms are held at the Trust and with the research leads at the University of Birmingham.
Informed consent to publish
All participant quotes used in this article are unidentifiable. Participants have agreed (in the consent form) for their interview data to be published in this non-identifiable way.
Trial registration
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
