Abstract
Summary statements
(1) Peritoneal dialysis (PD) should be considered a suitable modality for treatment of acute kidney injury (AKI) in all settings
Guideline 2: Access and fluid delivery for acute PD in adults
(2.1) Flexible peritoneal catheters should be used where resources and expertise exist (2.2) Rigid catheters and improvised catheters using nasogastric tubes and other cavity drainage catheters may be used in resource-poor environments where they may still be life-saving (2.3) We recommend catheters should be tunnelled to reduce peritonitis and peri-catheter leak (2.4) We recommend that the method of catheter implantation should be based on patient factors and locally available skills (2.5) PD catheter implantation by appropriately trained nephrologists in patients without contraindications is safe and functional results equate to those inserted surgically (2.6) Nephrologists should receive training and be permitted to insert PD catheters to ensure timely dialysis in the emergency setting (2.7) We recommend, when available, percutaneous catheter insertion by a nephrologist should include assessment with ultrasonography (2.8) Insertion of PD catheter should take place under complete aseptic conditions using sterile technique (2.9) We recommend the use of prophylactic antibiotics prior to PD catheter implantation (2.10) A closed delivery system with a Y connection should be used (2.11) The use of automated or manual PD exchanges are acceptable and this will be dependent on local availability and practices
Guideline 3: Peritoneal dialysis solutions for acute PD
(3.1) In patients who are critically ill, especially those with significant liver dysfunction and marked elevation of lactate levels, bicarbonate containing solutions should be used ( (3.2) Commercially prepared solutions should be used (3.3) Once potassium levels in the serum fall below 4 mmol/L, potassium should be added to dialysate (using strict sterile technique to prevent infection) or alternatively oral or intravenous potassium should be given to maintain potassium levels at 4 mmol/L or above (3.4) Potassium levels should be measured daily
Guideline 4: Prescribing and achieving adequate clearance in acute PD
(4.1) Targeting a weekly (4.2) Cycle times should be dictated by the clinical circumstances. Short cycle times (1–2 h) are likely to more rapidly correct uraemia, hyperkalaemia, fluid overload and/or metabolic acidosis; however, they may be increased to 4–6 hourly once the above are controlled to reduce costs and facilitate clearance of larger sized solutes (4.3) The concentration of dextrose should be increased and cycle time reduced to 2 hourly when fluid overload is evident. Once the patient is euvolemic, the dextrose concentration and cycle time should be adjusted to ensure a neutral fluid balance (4.4) Where resources permit, creatinine, urea, potassium and bicarbonate levels should be measured daily; 24 h (4.5) Interruption of dialysis should be considered once the patient is passing >1 L of urine/24 h and there is a spontaneous reduction in creatinine
The use of peritoneal dialysis (PD) to treat patients with acute kidney injury (AKI) has become more popular among clinicians following evidence of similar outcomes when compared with other extracorporeal therapies. Although it has been extensively used in low-resource environments for many years, there is now a renewed interest in the use of PD to manage patients with AKI (including patients in intensive care units) in higher income countries. Here we present the update of the International Society for Peritoneal Dialysis guidelines for PD in AKI. These guidelines extensively review the available literature and present updated recommendations regarding peritoneal access, dialysis solutions and prescription of dialysis with revised targets of solute clearance.
Introduction
Peritoneal dialysis (PD) has been effectively used to treat acute kidney injury (AKI) since 1946. 1 Initial problems relating to access, overhydration and hyperchloremic acidosis were overcome and with improved outcomes PD became a well-respected dialysis modality for AKI. 2,3 The introduction of extracorporeal continuous renal replacement therapy (CRRT) in intensive care units (ICUs) led to a rapid decline in the use of PD despite no evidence of superiority of this modality in terms of outcomes, even when compared with intermittent haemodialysis (IHD). 3 –5 Despite this decline in the use of PD, many clinicians feel that acute PD is still a suitable modality for treating patients with AKI both in the intensive care and in the ward environment. 6 Recent studies have demonstrated that there is equivalent survival (and perhaps a shorter need for renal replacement therapy (RRT)) using PD compared with other extracorporeal modalities, and as a result, there has been renewed interest in the use of PD for AKI. 7,8 Acute PD is largely practiced in Low/Low Middle Income Countries (LLMICs) due to a number of significant benefits over CRRT/IHD: the lack of requirements for water and electricity, significantly less training for nursing staff, cardiovascular stability in hypotensive patients and most significantly costs. Kilonzo et al. showed that it costs approximately US$400 per life saved with PD and George et al. noted that acute PD costs half that of haemofiltration. 9,10 These benefits and the simplicity of acute PD is why the Saving Young Lives (SYL) program has dedicated its efforts into developing acute PD programs in LLMIC’s and it has become the effector arm of the International Society of Nephrology’s 0 × 25 initiative for delivery of dialysis. Thus far over 500 patients in SYL centres have been treated with a >60% survival along with recovery of renal function. 11 –15 Finally, the recent Covid-19 pandemic has highlighted the benefits of acute PD in patients who have a hypercoagulable state, especially when extracorporeal therapy options are limited due to demands on machines, supplies and staffing. 16
The previous version of these guidelines evaluated the evidence for acute PD as well offering standardised prescriptions and options for access. 17 Subsequent studies have tested some of the recommendations and validated them. This current guideline is an update on the 2014 version and as such contains less discussion on the evidence base which can be referred to in the earlier guidelines, but rather focus on what is new in the literature with revised recommendations.
Methods
These guidelines have been developed under the auspices of the International Society for Peritoneal Dialysis. The committee has been carefully selected to include adult and paediatric nephrologists as well as intensive care specialists from around the world with a bias towards including practitioners from those countries where PD for AKI is practiced as a routine. Each section was written by at least two authors who performed a review of the literature in that area. The section was reviewed by the co-chairs (BC, FF), and finally, the recommendations and their grading were made by consensus of the whole committee. The final guidelines were then subsequently reviewed by all authors. The authors of each section can be found in Appendix 2. The recommendations are based on the GRADE system, a well validated structure which matches the strength of the recommendation to the level of evidence. 18 Grade 1 is a strong and 2 is a weak recommendation. The letters (A–D) indicate the level of evidence used to make the recommendations. Where no evidence exists, but there is enough clinical experience for the committee to make a recommendation, this will be categorised as opinion or practice point.
These guidelines have been developed for practitioners working in very different conditions. In some cases, what is felt to be optimal care may not be practical due to resource limitations. It is, therefore, important to define a minimum standard which needs to be achieved to ensure that the benefits of PD treatment for AKI outweigh the risks; this minimum standard may not, however, be deemed optimal treatment. There will, therefore, be recommendations made for ‘minimum standard’ or ‘optimal’, but practitioners should always strive to achieve the latter. There is no validated method of defining these two standards, and they are based on consensus by the Authors, using the best available evidence.
These are guidelines and as such should be used to direct practice patterns. It is important to keep in mind, however, that the guidelines may not be applicable to all clinical situations; clinicians should use the information to offer the best care to patients understanding that deviation from the guidelines may be necessary.
Guideline 1: Is PD a suitable modality for treating AKI?
PD should be considered a suitable modality for treatment of AKI in all settings
There are a number of advantages of PD over extracorporeal therapy in the treatment of AKI. However, there has been a long-held belief that PD is unable to achieve adequate clearances to make it comparable with extracorporeal therapies. As a result, there have been concerns that outcomes would be suboptimal in those patients treated with PD. The previous guidelines reviewed this question in depth and came up with a 1B recommendation that acute PD is a suitable modality for treating AKI.
17
Subsequent to these guidelines, a further single-centre randomised controlled trial comparing PD with continuous venovenous haemodiafiltration in critically ill patients was reported and showed a trend towards improved survival with PD.
8
A Cochrane review published at a similar time made the following conclusion: ‘Based on moderate (mortality, recovery of kidney function), low (infectious complications), or very low certainty evidence (correction of acidosis) there is probably little or no difference between PD and extracorporeal therapy for treating AKI. Fluid removal (low certainty) and weekly delivered
Guideline 2: Access and fluid delivery for acute PD in adults
(2.1) Flexible peritoneal catheters should be used where resources and expertise exist
(2.2) Rigid catheters and improvised catheters using nasogastric tubes and other cavity drainage catheters may be used in resource-poor environments where they may still be life-saving
(2.3) We recommend catheters should be tunnelled to reduce peritonitis and peri-catheter leak
(2.4) We recommend that the method of catheter implantation should be based on patient factors and locally available skills
(2.5) PD catheter implantation by appropriately trained nephrologists in patients without contraindications is safe and functional results equate to those inserted surgically
(2.6) Nephrologists should receive training and be permitted to insert PD catheters to ensure timely dialysis in the emergency setting
(2.7) We recommend, when available, percutaneous catheter insertion by a nephrologist should include assessment with ultrasonography
(2.8) Insertion of PD catheter should take place under complete aseptic conditions using sterile technique
(2.9) We recommend the use of prophylactic antibiotics prior to PD catheter implantation
(2.10) A closed delivery system with a Y connection should be used
(2.11) The use of automated or manual PD exchanges are acceptable and this will be dependent on local availability and practices
Rationale
Guideline 2.1–2.4 – Catheter type
Many PD catheters have been developed over the years to address the most common complications associated with PD access viz. catheter tip migration, peritoneal leak, peritonitis, exit site infection and catheter entrapment. Despite many innovative designs, no catheter has consistently proven superiority to the double-cuff Tenckhoff catheter. The most appropriate PD catheter is the one that can be positioned deep in pelvis, can be kept out of reach of the omentum and can provide an exit site that is easily visible and free of belt line.
Tenckhoff catheters are preferred over rigid catheters as they have a larger diameter lumen and side holes, resulting in better dialysate flow rates and less obstruction, which is imperative in acute PD to achieve adequate clearances. They are also less prone to leakage and have a lower incidence of peritonitis. 20,21 If the patient does not recover renal function, a Tenkhoff catheter may be used for chronic dialysis without the need for a new access procedure.
All catheters can be inserted under local anaesthesia at the bedside or in a surgical theatre. The bedside insertion utilises a modified Seldinger approach using a guidewire and peel-away sheath. This is a blind procedure and therefore contraindicated in those who have a midline surgical scar or history to suggest intra-abdominal adhesions. A study by Shanmugalingam et al. from Australia has challenged this rule demonstrating that the use of ultrasound assessment prior to insertion using the ‘slide test’ can identify those with previous abdominal surgery who may be suitable for a blind percutaneous insertion. 22 Where death from kidney failure is imminent and no options for ultrasound or direct visualisation exist, and PD is the only option, prior surgery could be considered a relative contraindication. (Step-by-step insertion guidelines are available on the SYL Website – https://www.theisn.org/programs/saving-young-lives-project).
Alternative catheters
Rigid catheter
These catheters are inserted using a sharp removable trochar device. Possible complications with this catheter design include bleeding, bowel or bladder perforation, obstruction due to the small side holes and lumen and leakage of dialysate. The incidence of peritonitis increases with time the catheter is left in the abdomen. 23 Comparison of these catheters with flexible PD catheters in children showed significantly higher rates of catheter malfunction. 20,24
Improvised catheter use has been reported in many centres where rigid or Tenckhoff catheters are unavailable. Reported cases describe nasogastric tubes with side holes cut into the tube prior to insertion surgically. These have been very effective, although no comparative data are available. Other options include intercostal drainage tubes, haemodialysis catheters and percutaneous cavity drainage catheters. It should be noted that none of these options are recommended as the first line; however, they have been shown to be lifesaving and so it is suggested that they are used if no other option exists. 25,26
Guideline 2.5–2.8 – Method of insertion
A far more in-depth review of this topic is available in the International Society for Peritoneal Dialysis (ISPD) guidelines on optimal peritoneal access 2019. 27 The key to effective PD is a catheter which allows rapid inflow and outflow of fluid to minimise drain and fill time and maximise the dialysate dwell time and contact of the dialysate with the peritoneal membrane. This is predominantly dependent on the catheter used (see above) but will be influenced by the position of the catheter and any interference from the omentum or adhesions. Leakage of fluid through the surgical wound necessitates reduction in fill volumes or even stopping PD for as long as necessary for the leakage to stop. The pros and cons of different catheter designs and implantation techniques are summarised in Tables 1 and 2. Preparation of the patient prior to insertion of the catheter will assist achieving optimal outcomes. Examples include bowel preparation using either oral or rectal solutions prescribed for colonoscopy and ensuring the bladder is catheterised.
Advantages and disadvantages of flexible, rigid and other peritoneal access.

Advantages and disadvantages of different catheter implantation techniques.

Percutaneous placement of PD catheters compare favourably with both laparoscopic and open surgical techniques and the ISPD guidelines on peritoneal access recommend that one should use a technique that one is most familiar with.
27
Blind insertion with a Seldinger technique is generally contraindicated in those patients in whom intra-abdominal adhesions might be expected because of the increased risk of bowel perforation. This would include those with a midline laparotomy scar or previous significant peritonitis. Patients with obesity may present technical difficulty and possible infectious complications and therefore should be considered for surgical insertion if readily available. The above can be assessed using pre-procedural ultrasound to determine the presence of visceral slide and skin to peritoneal distance of <5.5 cm. This technique identified a number of patients with prior abdominal surgery who were deemed suitable for percutaneous insertion due to a lack of adhesions.
22
The procedure should preferably be performed by an experienced clinician in a room specially designed for catheter placement or a sterile, calm environment. Under these conditions, the reported complication rate is low.
28
–30
Henderson et al. compared 283 percutaneous with 104 surgically inserted catheters. The incidence of leak (6% vs. 10%,
Al-Hwiesh et al. reported the outcomes of percutaneous catheter insertion without a peel-away sheath and demonstrated that rates of migration, leak and poor outflow were low (7.5%, 2.5% and 12.5%, respectively, with a success rate of 97.5%). 8,33
The laparoscopic technique allows direct visualisation of the peritoneal cavity and placement of the tip of the PD catheter. There is no significant difference in outcomes of laparoscopic versus open surgical placement unless one uses the advanced laparoscopic techniques including musculofascial tunnelling, omentopexy and tip suturing. 27,34 The advantage of laparoscopic insertion over open laparotomy is the relatively small incisions required and the ability to suture the port sites thus reducing the risk of leak in the acute PD patient.
Given the clear evidence of safety, we recommend that PD catheters can be inserted percutaneously by appropriately trained nephrologists. This has the significant practical reason that it limits the time required from diagnosis of dialysis requiring AKI to initiation of treatment, especially if the catheter can be inserted in the emergency room or procedure room rather than waiting for operating theatre. The smaller incision and serial dilation approach further reduces the risk of leak.
In conclusion, the method of flexible catheter placement should be that suited to the unit, balancing skills, resources and cost-effectiveness. Patients with previous midline surgical scars or high risk of peritoneal adhesions should have the catheter inserted using a technique which allows direct vision.
Guideline 2.9: Prophylactic antibiotics
Colonisation of the Tenckhoff catheter and/or contamination at the time of insertion increases the risk of subsequent peritonitis and needs to be avoided through strict sterile technique. The most appropriate place for insertion of the catheter will depend on the clinical setting of the patient. For example, in a patient with multi-organ failure and shock, the most appropriate place may be at the bedside in the ICU, whereas a stable patient should be transferred to a surgical theatre, radiology suite or dedicated procedure room. There are no trials answering this question; however, the experience of many clinicians is that bedside insertion is safe and does not lead to increased peritonitis risk as long as strict sterile technique is adhered to.
Prophylactic antibiotics do not prevent infections if the above precautions are not followed. However, when used in conjunction with sterile technique, there is a decrease in the incidence of peritonitis. 35 –38 The decision of which antibiotic to use is dependent on local bacterial sensitivities, timing of the procedure and availability. It is generally accepted that the most important organisms to protect against are the gram-positive organisms. However, given the small risk of bowel injury, some clinicians use an agent which would also cover gram-negative bacteria.
Prophylactic antibiotics need to have adequate tissue levels prior to the initial incision. It therefore makes agents which require a long infusion time unsuitable for patients who need emergent dialysis.
Guidelines 2.10 and 2.11
Disconnecting systems allow the removal of the fluid infusion set (and infusion and drainage containers) from the patient between dwells. A Y-set using a double bag and the disconnect system are associated with lower peritonitis rates compared to the standard spiking system in chronic patients, and there is no reason to suspect this would not be the case in acute PD. 39 To use the disconnect systems, there needs to be an adequate supply of closure devices to ensure that the proximal end of the catheter does not become contaminated between exchanges. If these are not available, it may be safer to leave the ‘bag’ connected to the patient and perform a ‘reverse’ exchange (i.e. fill the peritoneum and leave the patient connected for the dwell, then drain and disconnect, attaching the new bag prior to the next fill).
When commercially produced solutions are not available, then it may be necessary to improvise the system being used. Some proprietary devices are available which have a spike system to attach intravenous (IV) fluid containers and this can be attached to a three-way tap on the PD catheter with a drainage tube on the other port.
If three-way taps are not available, then the fluid can be attached to a standard IV fluid administration set (without a one-way valve). Fluid is instilled and the clamp is closed. Once the dwell is complete, then the empty bag is dropped to the floor and the clamp is opened allowing fluid to drain back into the container. This will only be an option with flexible plastic bags as they have the capacity to expand and absorb the excess ultrafiltrate.
Cycler
Automated cycler PD is the term used to refer to all forms of PD that employ a mechanized device to assist in the delivery and drainage of dialysate. A volume of dialysate is prescribed as well as the therapy time and fill volume. The advantage of this system is that it can be set up by a trained staff member once per day to reduce the risk of complications. It also reduces nursing time as all cycles are automatic. There are conflicting reports of whether there is a reduction in peritonitis with cyclers, but on balance, there appears to be no difference compared to the manual system in chronic PD. This may be different in acute PD where the number of exchanges is increased, hence the increase in potential contamination episodes.
Tidal automated PD (APD)
This is where a small volume of fluid is left in the abdomen at all times which may reduce mechanical complications and pain associated with complete fluid drainage. It may have a benefit in critically ill patients in that there is always some fluid in contact with the peritoneum, and therefore large molecular weight toxins formed as part of the inflammatory process may be cleared better. This has been demonstrated most dramatically for higher dialysate flow rates. 40
Automated cyclers have been used extensively for PD in AKI; however, in a resource poor setting, cyclers may prove too expensive. 41 Occasionally, the fixed hydraulic suction of APD machines may worsen mechanical obstruction in catheters with already tenuous fluid flow in which case it is sometimes helpful to change to manual exchanges.
A further disadvantage of cyclers is that there may not be support after hours for inexperienced nurses using the cyclers and there is the risk of the machines being turned off during the night to avoid alarms.
Acute PD in patients with acute respiratory distress syndrome has prompted the question of the suitability in patients in the prone position. There are two aspects to consider. First is the impact of raised intraabdominal pressure on lung mechanics and organ perfusion. Second is the effect of the prone position on flow characteristics. There are no studies which address this; however, prone positioning increases the intra-abdominal pressure by approximately 1–3 mmHg. Acute PD increases the intra-abdominal pressure by a further approximately 2 mmHg. Intraabdominal pressures above 18 mmHg should be avoided due to reduced organ perfusion and diaphragmatic splinting; this is unlikely to occur in the absence of other causes of intra-abdominal hypertension (IAH). If IAH is suspected, then the abdominal pressure can be measured. This is most easily performed using a bladder manometer; however, if this is not available, a three-way connector placed between the PD catheter and the PD solution can be connected to a standard vascular pressure transducer. The latter approach carries the risk of causing peritonitis. Abdominal pressure in the prone position can be reduced by placing a pillow under the hips and chest, thus allowing the abdomen to be suspended.
As the PD catheters in these patients are placed and used acutely, attention to immobilisation of the catheter to prevent inadvertent removal is essential.
Guideline 3: Peritoneal dialysis solutions for acute PD
(3.1) In patients who are critically ill, especially those with significant liver dysfunction and marked elevation of lactate levels, bicarbonate containing solutions should be used (
(3.2) Commercially prepared solutions should be used
(3.3) Once potassium levels in the serum fall below 4 mmol/L, potassium should be added to dialysate (using strict sterile technique to prevent infection) or alternatively oral or intravenous potassium should be given to maintain potassium levels at 4 mmol/L or above
(3.4) Potassium levels should be measured daily
Rationale
Guideline 3.1
The high mortality rate among critically ill patients with AKI remains an unresolved problem despite the use of all modes of RRT. Increasing evidence from clinical studies in adults and children suggests that the new less bio-incompatible solutions may allow for better long-term preservation of peritoneal morphology and function. Formation of glucose degradation products (GDPs) can be reduced and even avoided with the double compartment bags allowing separate glucose heat sterilisation in an acid environment. Due to the separation of components such as calcium, it is also possible to create solutions whose buffer is bicarbonate and not lactate. Lactate is normally converted to bicarbonate in the liver; however, in critically ill patients and those with liver dysfunction, there is accumulation of the lactate and the inability to buffer appropriately. A randomized controlled trial including 20 AKI patients compared the effectiveness of bicarbonate versus lactate-buffered PD solutions with a dwell time of 30 min.
42
In shocked patients treated with bicarbonate-buffered solution, there was a more rapid increase in serum bicarbonate (21.2 ± 1.8 vs. 13.4 ± 1.3 mmol/L) and blood pH (7.3 ± 0.03 vs. 7.05 ± 0.04,
Guideline 3.2 – Commercial versus locally mixed solutions
Commercial solutions are produced to high standards with strict asepsis and careful monitoring for bacterial and endotoxin contamination. Locally prepared solutions carry the potential risks of contamination and mixing errors which may be life threatening. The use of hospital pharmacy-prepared solutions has previously been reported in children with good peritonitis rates and outcomes. 43,44 More recent publications from Africa have demonstrated good outcomes using bedside preparation of PD fluids made from commercially available IV fluid. 12,45,46 Palmer et al. performed a retrospective review of all acute PD patients and showed no difference in peritonitis rates between those treated with commercial solutions and those using locally mixed solutions. 45 A retrospective review of 49 children from Cape Town, using bedside prepared PD solutions, made from IV fluids showed a low peritonitis rate and no complications. 47
Commercial solutions often have closed drainage systems to prevent accidental contamination, whereas makeshift connections may be needed for locally prepared solutions.
Cost is often a factor which may limit utilisation of commercially produced solutions in low-resource settings, particularly if patients are paying for their own care. The costs include both the cost of purchasing the solutions and the costs for transportation, taxes and bureaucratic assessments.
The ISPD recommends the following types of fluid in order of preference: Commercially prepared solutions Locally prepared fluid made in an approved and certified aseptic unit/pharmacy. These products would have a limited expiry time as approved by the manufacturing unithttp://www.ashp.org/DocLibrary/BestPractices/PrepGdlCSP.aspx. Solutions prepared in a clean environment with a minimum number of punctures and the least number of steps. This fluid should be used immediately.
Appendix 1 provides examples of how to mix solutions using commonly available IV fluids to approximate commercially prepared solutions as presented in Tables 3 and 4.
Commercially available intravenous fluids.
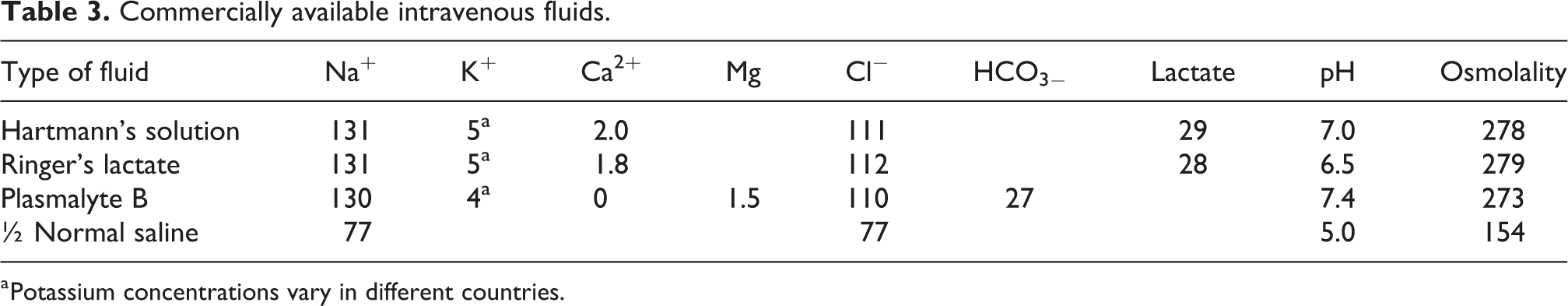
a Potassium concentrations vary in different countries.
Typical compositions of some commercially available PD fluids.
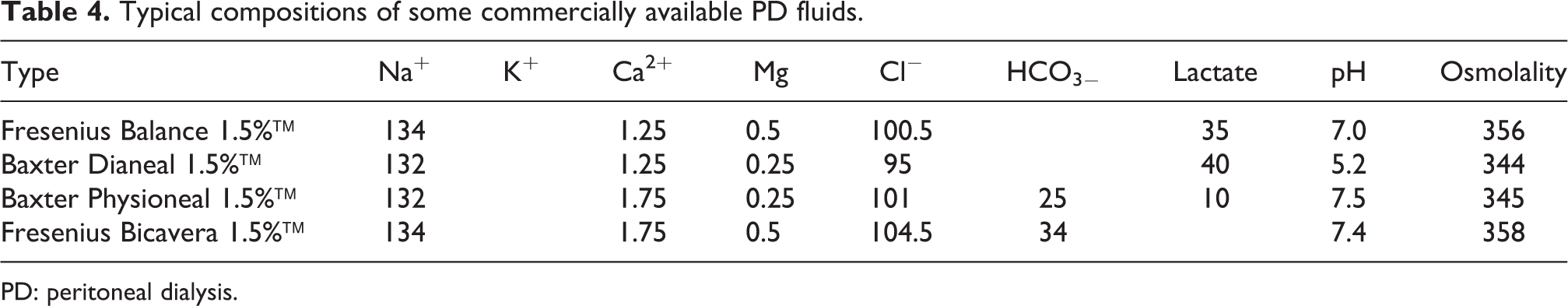
PD: peritoneal dialysis.
It should be noted that in making solutions using the above approach, calcium and magnesium may be present. In general, this is not a problem for acute PD, which is usually of short duration.
Many of the plasma expanders contain potassium, and although after the first 24 h this may be beneficial, it may be counterproductive initially.
General rules when preparing dialysis solutions: The concentrations of the well-known IV solutions may vary from country to country so check concentrations before mixing, Maintain absolute strict sterile technique when mixing solutions, The fewer components added to the solution, the lower the risk of infection and error, Avoid mixing bicarbonate and calcium as they will precipitate.
Guidelines 3.3 and 3.4
Losses of potassium can be high in acute PD; such removal may cause serious potassium depletion and cardiovascular instability. This might be prevented or corrected by adding potassium chloride to the dialysis solution to create a solution containing 3–4 mmol/L of potassium
Large studies on PD in AKI patients demonstrated serum potassium control was obtained after a 24hr session of high-volume PD, and when serum potassium was lower than 4 mmol/L, potassium (K) 3.5–5 mmol/L was added to dialysis solutions to avoid hypokalemia. 7,48 It is important that sterile technique be maintained when potassium is added and that nurses are carefully instructed to make certain the amount added is appropriate. Measurement of potassium on a daily basis is the safest method of monitoring these patients. However, in many low-income countries, it is not possible to measure the potassium, and therefore, extrapolating from the above studies, it seems prudent to add potassium to the fluid after 24 h. It should be noted that as the potassium is subject to diffusion down a concentration gradient, it should not be possible to drive the serum potassium greater than the concentration which is in the PD fluid unless there is significant exogenous administration thereof or shifts of potassium from intracellular compartments. Hence, adding 4mmol/L should be safe, although it may limit clearance of potassium from the serum if it is still elevated.
Guideline 4: Prescribing and achieving adequate clearance in acute PD
(4.1) Targeting a weekly
(4.2) Cycle times should be dictated by the clinical circumstances. Short cycle times (1–2 h) are likely to more rapidly correct uraemia, hyperkalaemia, fluid overload and/or metabolic acidosis; however, they may be increased to 4–6 hourly once the above are controlled to reduce costs and facilitate clearance of larger sized solutes
(4.3) The concentration of dextrose should be increased and cycle time reduced to 2 hourly when fluid overload is evident. Once the patient is euvolemic, the dextrose concentration and cycle time should be adjusted to ensure a neutral fluid balance
(4.4) Where resources permit, creatinine, urea, potassium and bicarbonate levels should be measured daily; 24 h
(4.5) Interruption of dialysis should be considered once the patient is passing >1 L of urine/24 h and there is a spontaneous reduction in creatinine
Rationale
Guideline 4.1–4.4
There remains controversy as to the most appropriate dose of PD that should be prescribed for patients with AKI. The factors influencing this are the relative need for small versus large molecule and fluid clearance, the rate of equilibration of molecules and the relative contribution of convection versus diffusion. Until now, all outcome studies have measured daily/weekly
K
t/V
urea
Daily
The most effective dose of PD for patients with AKI remains uncertain, mainly due to a limited number of trials, the existence of methodological flaws in some studies and the fact that the doses of dialysis used varied widely. We also have little knowledge regarding the inter-individual variability of membrane function in critically ill patients. A very small study in infants showed marked differences in Dialysate/Plasma (D/P) creatinine in patients determined by the cause of the AKI. 49
A study by Ponce-Gabriel et al. compared acute PD using a cuffed catheter (36–44 L per session, 18–22 cycles, 2 L per cycle, weekly delivered
Other studies have shown very good outcomes with much lower doses (16–24 L per session, 8–16 cycles, 1–2 L per cycle). 9,51 However, since these studies were non-randomized, the problem of a positive reporting bias needs to be kept in mind.
The same Brazilian investigators subsequently published a study randomising 79 patients with AKI on the ICU to differing doses of treatment and compared survival.
52
The achieved weekly
The volume of fluid required to achieve the clearances mentioned above would be prohibitively expensive in many of the countries where PD will be used to treat AKI. As a result, the previous ISPD guidelines also recommended a second dosing target of a weekly
Following the previous guidelines on PD for AKI, Parapiboon et al. randomised 80 critically ill patients with AKI to 2 regimens recommended in the ISPD guidelines and aimed at achieving target weekly
The primary hypothesis was that intensive treatment would result in a 10% reduction in mortality. However, the number of patients needed to power this study was >700, and as such, it was underpowered for the primary end point. Mortality, however, was compared using Kaplan-Meier survival analysis.
The dropout rate was low, with one patient in the low-intensity arm changing to HD and two of the high-intensity patients being transferred to low intensity due to either hyperglycaemia or hypokalaemia.
Baseline characteristics were not significantly different between the two groups. However, it must be noted that as this study was performed in Asia, the mean body weight was low (60.1 kg ± 11.1) compared with that seen in other countries, and the dwell volumes were low because of this. The patients were similar to those in the study by Ponce-Gabriel et al., with 88% on mechanical ventilation, 69% on inotropic support and a mean APACHE II score of 26. 1,2 Pre-dialysis blood urea nitrogen (BUN) values were lower than those seen in the Brazilian studies approximately 75 mg/dL and therefore may represent earlier initiation of dialysis.
Seventy-five patients were included in the analysis. The achieved
The mortality was 72% in the high-intensity and 63% in the low-intensity groups (
The above studies have used manual or APD with maximum drainage of each dwell. There has been concern raised that the rapid cycling using this method results in significant periods when the peritoneum is not in contact with fluid, thus reducing efficiency. Al-Hwiesh et al. published a study randomising 125 critically ill patients to tidal APD or continuous veno-venous haemodiafiltration (CVVHDF). The use of tidal APD allows some residual PD fluid to remain in contact with the peritoneum as it is not drained completely.
8
The patients in this study were similar to those mentioned above, with APACHE II scores of 21–22 and >60% on mechanical ventilation. The volume of fluid used was 25 L/24 h, and it must be mentioned that low GDP, bicarbonate-based solutions were used, thus making this study and previous studies impossible to compare. The trial though compared mortality, recovery of renal function and other outcome measures with patients on CVVHDF who achieved effluent rates of 23 mL/kg/h, which corresponds with current best practice. There is concern that although these effluent levels were achieved, serum levels of creatinine remained higher than expected. The study was underpowered to detect the 20% mortality difference hypothesized. Analysis of mortality on Kaplan-Meier curve showed significantly lower mortality in the tidal APD group (30.2 vs. 53.2%,
Randomized controlled studies comparing PD versus extracorporeal therapies and PD with high versus low dose.
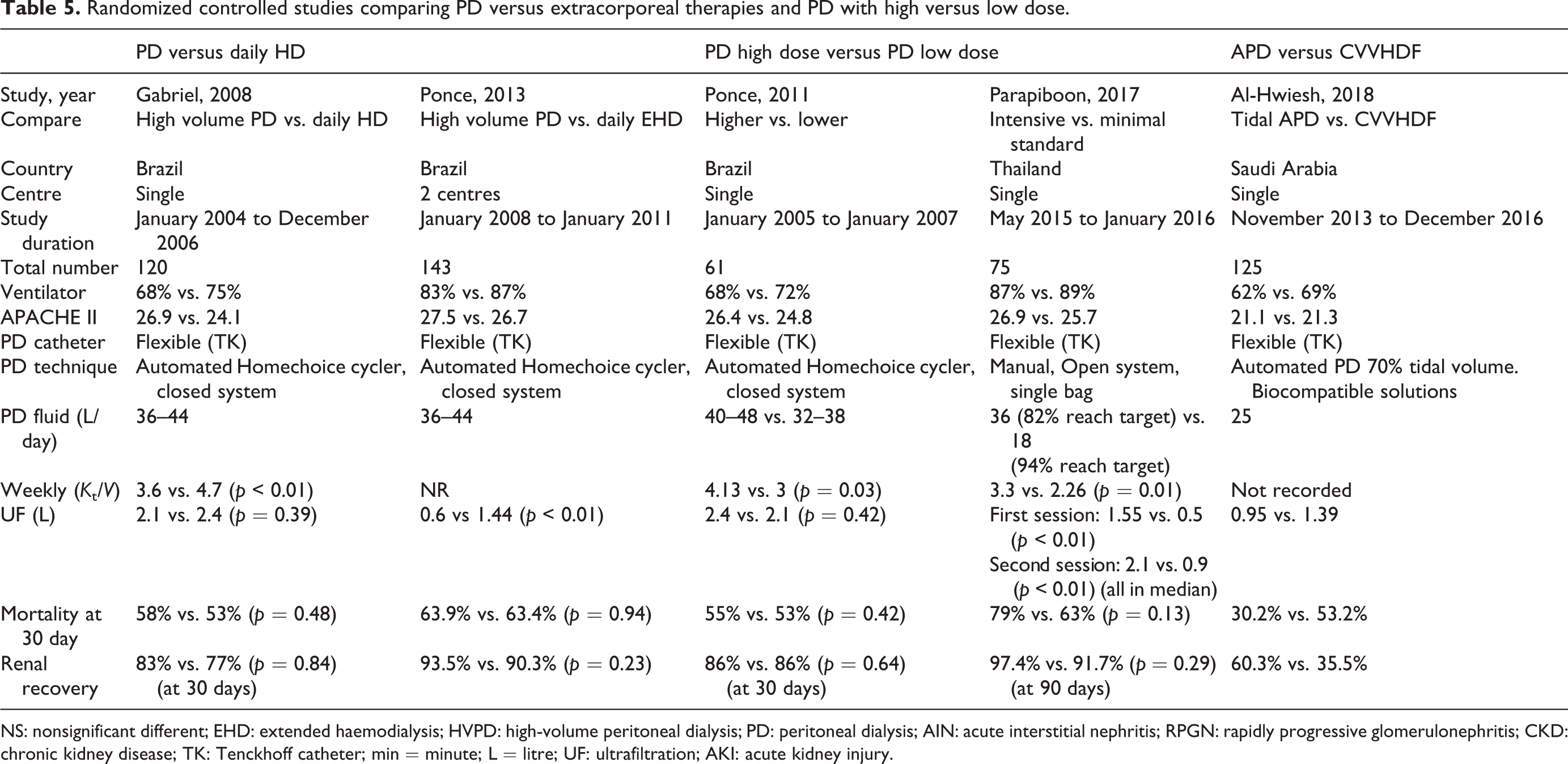
NS: nonsignificant different; EHD: extended haemodialysis; HVPD: high-volume peritoneal dialysis; PD: peritoneal dialysis; AIN: acute interstitial nephritis; RPGN: rapidly progressive glomerulonephritis; CKD: chronic kidney disease; TK: Tenckhoff catheter; min = minute; L = litre; UF: ultrafiltration; AKI: acute kidney injury.
Much attention has focused on solute clearances, but there is increasing evidence that fluid overload is also harmful and should be avoided or corrected. In principle, regular assessment of volume status and the prescription of ultrafiltration and fluid balance targets are necessary for all patients receiving RRT, including PD. 55 Relatively large amounts of fluid can be removed by PD, that is, up to 1 L in 2–4 h when using a 4.25% PD solution. Although this may cause hyperglycaemia, the risks of hypertonic solutions are negligible in the short term. The convective clearances associated with this increased ultrafiltration may offer improved middle molecule clearance and again further research is necessary in this area.
Additional attention needs to be paid to the dosing of various medications (such as antibiotics) depending on the peritoneal clearances achieved with acute PD, particularly with high-volume therapy; unfortunately, there is very little in the literature to guide this, but it can be assumed that one would achieve clearances in the order of those seen with daily haemodialysis. 56
Recommendations for clinical practice
During the initial 24–48 h of acute PD, the duration of cycle time needs to be determined based on the clinical circumstances (see Figure 1). Short cycle times (every 1–2 h) may be necessary in the first 24–48 h to correct uraemia, hyperkalaemia, fluid overload and/or metabolic acidosis, mainly in critically ill patients and when PD starts late. The algorithm below gives an approximate weight-based prescription guideline. Thereafter, the cycle time may be increased to 4–6 h depending on the clinical circumstances. Suggested management of adult patients requiring peritoneal dialysis to treat AKI. AKI: acute kidney injury.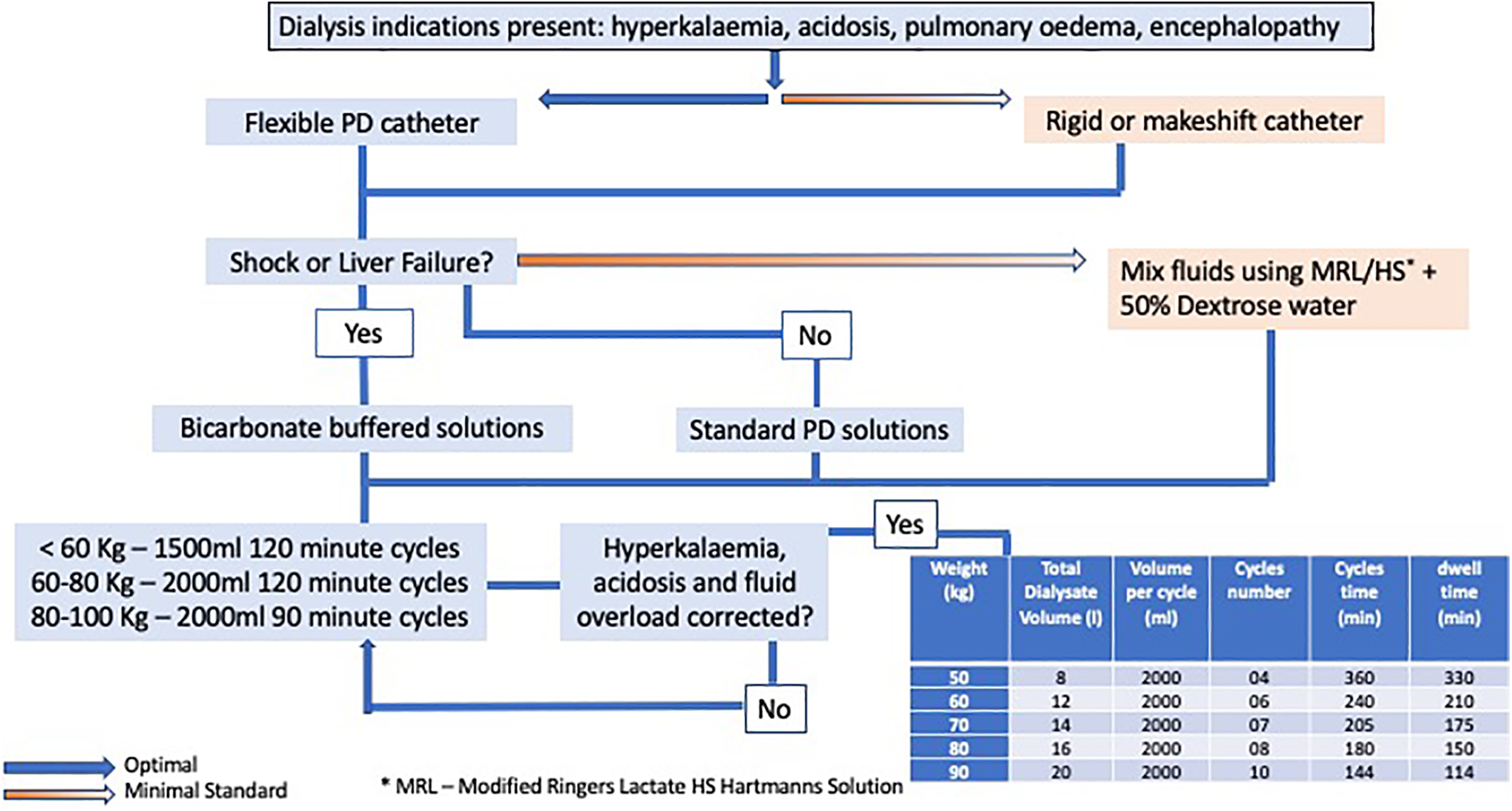
To treat or avoid fluid overload, ultrafiltration can be increased by raising the concentration of dextrose and/or shortening the cycle duration. When the patient is euvolemic, the dextrose concentration and cycle time should be adjusted to ensure a neutral fluid balance.
Research recommendations
Determine whether Creatinine clearance or other markers of solute removal predict survival better than
Comparison of patient comfort, appetite and mobilisation with PD and CRRT/IHD.
To compare PD with weekly
Compare tidal APD with manual PD achieving
Compare biocompatible versus standard solutions.
Determine the solute clearances of substances with varying molecular weight and protein binding in patients with critical illness of varying cause to ascertain whether this may impact on prescription.
Evaluate antibiotic and other drug clearance in high-volume PD.
Further studies comparing outcomes with PD compared to extracorporeal therapies in the treatment of AKI in different patient populations.
Managing complications in PD for AKI
There are a number of potential complications associated with the use of acute PD. Although an in-depth discussion on these is beyond the scope of these guidelines, the following will be discussed briefly:
peritonitis,
mechanical complications,
protein loss,
hyperglycaemia.
Peritonitis
The diagnosis and management of peritonitis in AKI may be challenging but should be based on the recommendations from the ISPD guidelines for infectious complications. 57 The diagnosis is made based on the presence of abdominal pain, cloudy dialysate and a leukocyte count of >100 cells µL (or polymorphonuclear cells >50%) after a 2-h dwell. These signs may be masked by the overall illness, and it is therefore reasonable to perform a leukocyte count daily for peritonitis surveillance in patients on acute PD. In resource limited settings, this may not be feasible and an alternative method is to perform a urine leukocyte esterase dipstick test daily, which if >2+ should prompt treatment while waiting for a confirmatory leukocyte count and cultures. This method has shown good sensitivity and specificity in small studies, but other features such as abdominal pain and fever should also prompt further investigation. 58,59
Treatment of peritonitis is beyond the scope of these guidelines, and it is recommended that the ISPD infectious complication guidelines be consulted. 57 Although no antibiotic regimen has proven to be superior to others as empirical treatment, 60 the combination of a glycopeptide (vancomycin or teicoplanin) plus ceftazidime was considered to be superior to other regimens in a proportional meta-analysis. 61 Importantly, both gram-positive and gram-negative coverage should be used for empiric therapy.
The recommended dosage of most commonly used antibiotics for the treatment of PD-related peritonitis is available in the ISPD guidelines for treatment of peritonitis. 57
Although antibiotics are often given once daily in continuous ambulatory peritoneal dialysis (CAPD), this regimen relies on subsequent exchanges achieving therapeutic intraperitoneal levels through back-diffusion from the bloodstream and this is determined by the length of the dwell. 62 In cases of rapid cycling, it has been shown that certain antibiotics are unable to achieve therapeutic levels, and therefore antibiotics should be added to all bags, if possible, using the continuous dosing recommendation in the ISPD guidelines. 57
Patients in the critical care environment have a high incidence of systemic candidiasis and there needs to be a high index of suspicion for fungal peritonitis in these patients.
Mechanical complications
PD access dysfunction
Another important complication is mechanical or catheter-related problems. In one study, this resulted in discontinuation of PD in over 10% of the patients randomized to the PD arm. Ponce et al. studied 204 patients on acute PD and found a mechanical complication rate of 7.3% with interruption of treatment in 2.6%. 7 Catheter flow dysfunction is usually manifested as outflow failure; therefore, the volume of drained dialysate is substantially less than the inflow volume. The most common cause of outflow dysfunction is constipation. 63 This may be manifested as either migration of the catheter out of the pelvis due to a loaded rectosigmoid colon or mechanical obstruction of fluid return to the pelvis. Extrinsic bladder compression on the catheter due to urinary retention occurs less frequently. 64 Mechanical kinking of the catheter tubing or an intraluminal fibrin clot is usually accompanied by two-way obstruction. An abdominal radiograph is often helpful in identifying a faecal-filled rectosigmoid colon, catheter displacement or a kink in the catheter tubing.
Treatment of mechanical failure due to constipation involves aggressive attempts to clear the bowel and is usually only achieved with the use of agents reserved for bowel preparation for colonoscopy.
If the abdominal X-ray excludes tubing kinks or displacement, bladder distention is excluded, and flow function is not restored with correction of constipation, then an attempt should be made to repeatedly flush the catheter with 20–50 mL of 0.9% saline. This must be performed by an experienced nurse or doctor in a sterile manner and aspiration should not be done or not be aggressive as it is possible to draw intra-abdominal organs or omentum into the catheter. If flushing is unsuccessful, fibrinolytic therapy with tissue plasminogen activator (tPA) may be attempted to clear presumed intraluminal fibrin or blood clots; 8 mL of tPA (1 mg/mL) is slowly injected into the catheter. This is left for 1 h and followed by gentle aspiration and flushing of the catheter. This can be repeated if there is partial resolution. If catheter obstruction is due to a fibrin or blood clot, recovery of flow function with tPA has been reported at nearly 100%. 65,66 Once the catheter is cleared, then 500 units of heparin should be added to each litre of PD solution.
If the catheter has migrated out of the pelvis and there has been no improvement with treating the constipation, then it is possible that the catheter has been entrapped in omentum or loops of bowel.
The least invasive method of correction via catheter repositioning is with the use of fluoroscopy and a flexible guidewire to manipulate the catheter into the correct position. There are numerous techniques which have been effective and it is recommended these are tried before proceeding to surgery if facilities permit. 67– 69
If fluoroscopy is unavailable or unsuccessful, then surgical options need to be entertained. The first option is to open the midline incision and slowly withdraw the catheter until the first side hole is visualised. Using a percutaneous PD catheter insertion kit, the guidewire can be advanced through the first side hole down the catheter into the peritoneal space. The catheter can then be withdrawn from the abdomen and fibrin can be cleaned out of the catheter if present. A peel-away sheath dilator can be introduced using the guidewire and then the catheter can be replaced using this method.
The alternative is to perform surgical manipulation. This should preferably be performed laparoscopically and the catheter can be mobilised and replaced in the pelvis under vision. Open surgical manipulation is possible; however, this usually requires a minilaparotomy with the risk of leakage if used immediately.
Leakage
Leakage of peritoneal fluid occurs occasionally. It does not appear to be related to fluid volumes or pressures and may be more related to insertion technique and patient factors. It is recommended that in the first week if a patient is mobilized, they should have a dry or minimally filled abdomen; however, in many cases, these patients are bed ridden, and therefore this may not be a common problem. If leakage occurs once the patient is more stable, then it may be possible to rest the abdomen for 24 h and restart PD with smaller volumes. If it occurs early, then reducing fill volumes may help. Fibrin glue and tissue adhesive have been used in a number of cases with some success. 70 –72
Metabolic complications
Loss of protein from the peritoneum in patients on chronic PD varies in different studies from 6.2 g to 12.8 g per 24 h. However, this has been known to increase to as high as 48 g during episodes of peritonitis. 51,73,74 A study from Brazil measured protein loss in 31 patients on high-volume acute PD over 208 sessions. They showed that protein loss was 4.2 (±6.1) g/24 h, and there was no correlation with albumin levels. Peritonitis did however increase protein loss. 75 Care should be taken to ensure that adequate protein intake occurs aiming for approximately 1.2 g/kg of protein per 24 h. There is an association with increased mortality in those patients with a negative protein balance, but whether this is related to disease severity rather than inadequate intake is uncertain. 76
Due to the high glucose concentration in PD fluid, there is a tendency towards hyperglycaemia in acute PD. This decreases the osmotic gradient between PD fluid and serum and should be treated to enable optimal ultrafiltration. Maintenance of normoglycaemia has also been shown to significantly improve survival in critically ill patients. 77
Footnotes
Author note
We have read and understood Peritoneal Dialysis International’s policy on conflicts of interest disclosure and declare no conflict of interests.
Author contributions
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
ORCID iD
Appendix 1
Suggestions for the preparation of locally mixed peritoneal dialysis fluids.a
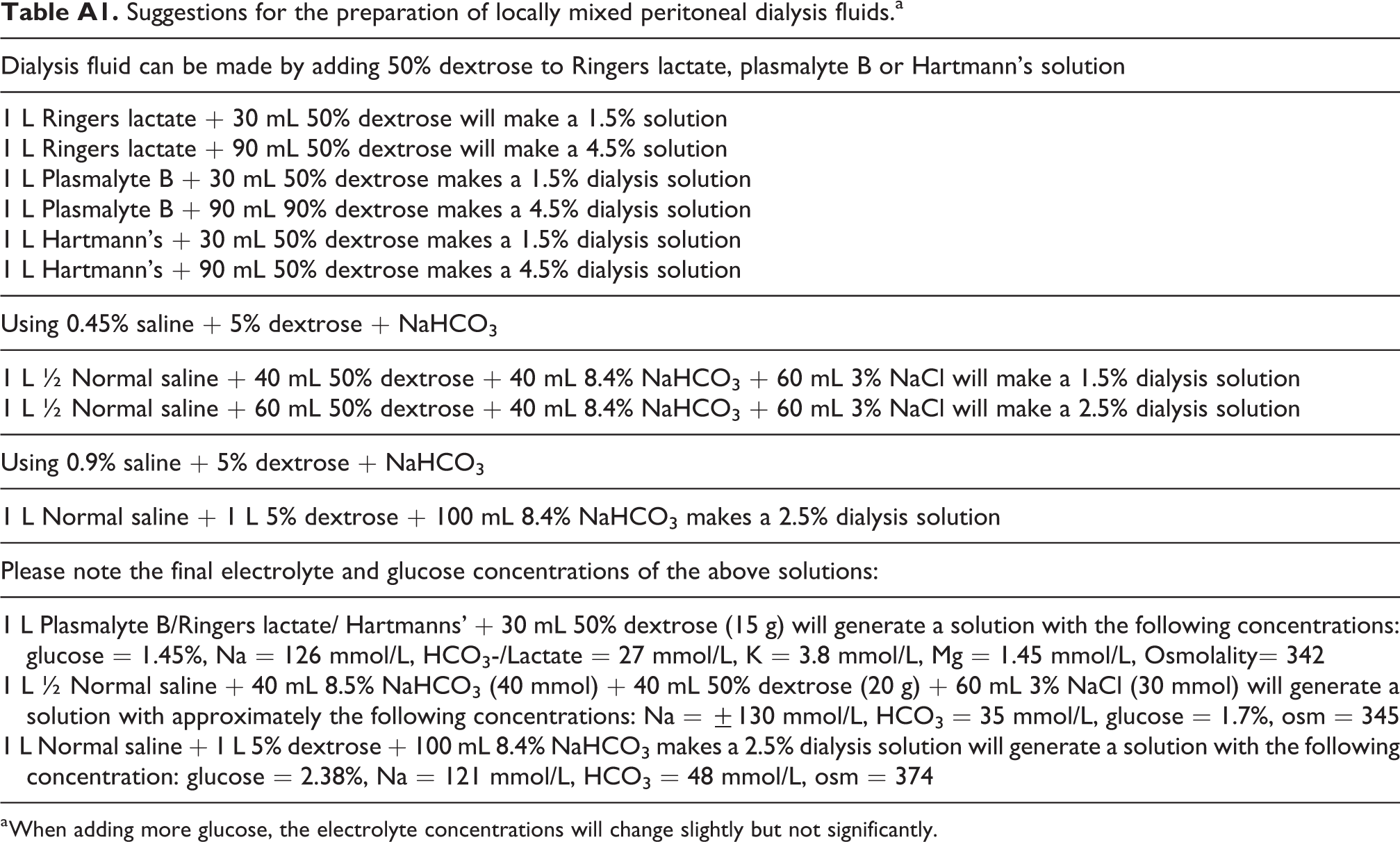
| Dialysis fluid can be made by adding 50% dextrose to Ringers lactate, plasmalyte B or Hartmann’s solution |
|---|
| 1 L Ringers lactate + 30 mL 50% dextrose will make a 1.5% solution |
| 1 L Plasmalyte B + 30 mL 50% dextrose makes a 1.5% dialysis solution |
| 1 L Hartmann’s + 30 mL 50% dextrose makes a 1.5% dialysis solution |
| Using 0.45% saline + 5% dextrose + NaHCO3 |
| 1 L ½ Normal saline + 40 mL 50% dextrose + 40 mL 8.4% NaHCO3 + 60 mL 3% NaCl will make a 1.5% dialysis solution |
| Using 0.9% saline + 5% dextrose + NaHCO3 |
| 1 L Normal saline + 1 L 5% dextrose + 100 mL 8.4% NaHCO3 makes a 2.5% dialysis solution |
| Please note the final electrolyte and glucose concentrations of the above solutions: |
| 1 L Plasmalyte B/Ringers lactate/ Hartmanns’ + 30 mL 50% dextrose (15 g) will generate a solution with the following concentrations: glucose = 1.45%, Na = 126 mmol/L, HCO3-/Lactate = 27 mmol/L, K = 3.8 mmol/L, Mg = 1.45 mmol/L, Osmolality= 342 |
| 1 L ½ Normal saline + 40 mL 8.5% NaHCO3 (40 mmol) + 40 mL 50% dextrose (20 g) + 60 mL 3% NaCl (30 mmol) will generate a solution with approximately the following concentrations: Na = ±130 mmol/L, HCO3 = 35 mmol/L, glucose = 1.7%, osm = 345 |
| 1 L Normal saline + 1 L 5% dextrose + 100 mL 8.4% NaHCO3 makes a 2.5% dialysis solution will generate a solution with the following concentration: glucose = 2.38%, Na = 121 mmol/L, HCO3 = 48 mmol/L, osm = 374 |
a When adding more glucose, the electrolyte concentrations will change slightly but not significantly.
Appendix 2
Authors:
Introduction: Brett Cullis, Fredric Finkelstein
Access and fluid delivery: Abdullah Al-Hwiesh, Brett Cullis
Fluids: Peter Nourse, Kajiru Kilonzo, Mignon McCulloch, Brett Cullis
Dialysis prescription: Daniela Ponce, Watanyu Parapiboon, Brett Cullis
Complications: Niang Abdou, Frederic Finkelstein, Abdullah Al-Hwiesh, Brett Cullis
