Abstract
The casting method was used to produce new inexpensive optoelectronic nanocomposite films containing strontium titanate (SrTiO3) and cobalt oxide (Co2O3) nanoparticles in a polyvinyl alcohol (PVA) host polymer matrix. According to the images taken with the optical microscope, the nanoparticles were evenly dispersed throughout the polymer matrix. Looking at the FTIR spectra in comparison to PVA shows that certain peaks have changed intensity and others have moved locations. Results show that the optical constants are proportional to the concentration of (SrTiO3-Co2O3) nanoparticles (NPs), indicating that optical constants increase with concentration and transmittance decreases with further concentration. With a rise in (SrTiO3-Co2O3) nanoparticle concentration. The optical band gap significantly decreased from 4.1 eV to 3.4 eV, facilitating indirect transitions, as the concentration of SrTiO3–Co2O3 increased. This indicates that localized states improved, resulting in enhanced optoelectronic performance. At a loading of 6 wt% of SrTiO-Co2O3, the dielectric constant, dielectric loss, and AC electrical conductivity increased to 1.187, 0.523, and 2.90 × 10−11 S/cm, respectively. This indicates an improvement in charge transfer and interfacial polarization. The antibacterial assays indicated that Staphylococcus aureus had inhibition zones measuring up to 34 mm, while Escherichia coli displayed inhibition zones of up to 26 mm. This confirmed that the antibacterial activity intensified with increasing nanoparticle concentration. The pressure sensing performance was markedly enhanced, with the nanocomposite exhibiting a peak sensitivity of 72.66% at a 6 wt% nanoparticle concentration, highlighting its suitability for flexible pressure sensor applications. In general, the addition of SrTiO3–Co2O3 nanoparticles altered the optical band gap, enhanced the dielectric and electrical properties, and rendered the material significantly more sensitive to pressure and more effective in the elimination of bacteria. The combined functions of PVA/SrTiO3–Co2O3 nanocomposite films suggest that they may be beneficial for antimicrobial applications, flexible pressure sensors, and optoelectronic devices.
Introduction
Nanocomposite materials have recently attracted considerable scientific and industrial interest owing to their exceptional properties and extensive range of potential applications. 1 These materials usually have a polymer matrix with nanoscale fillers like nanoparticles or nanofibers inside them. These fillers greatly improve the mechanical, electrical, and optical properties of the host polymer.2,3 Adding these kinds of nanofillers makes it possible to make materials that can do many things and work better. This makes nanocomposites great for a wide range of fields, such as medicine, products, electronics, optics, food packaging, pharmaceuticals, textiles, automobile engineering, energy storage, and cleaning up the environment. 4 Polymer-based nanocomposites are used in advanced drug delivery systems in biomedical applications, for example, where the nanoscale structure of the composite can be changed to control how the drug is released. 5 In the same way, nanocomposites with better electrical conductivity and mechanical strength make electronic devices more efficient and last longer. Also, their adjustable surface and catalytic properties make them good candidates for cleaning up the environment, like removing pollutants from wastewater and air treatment systems through adsorption and catalytic degradation. As a result, nanocomposites have become the focus of a lot of research because their physical, chemical, and functional properties can be changed and work together. 6 Metal–polymer hybrids are one type of nanocomposite that has gotten a lot of attention because of how they behave differently in terms of light, heat, electricity, and mechanics. Noble metal nanoparticles, in particular, are of great interest in photonics, space technologies, and biomedical applications owing to their exceptional plasmonic and optical responses. 7 The integration of metallic nanoparticles within polymer matrices not only enhances the optical features of the nanometals but also alters the polymer’s structural and mechanical performance. These tailored composites can serve in numerous advanced optical devices, things like light-stabilised colour filters, sensors, polarizers, and solar cells. When the intrinsic properties of polymers are engineered to complement the metallic phase, such hybrid systems become viable materials for space and high-performance optical applications. In addition, controlling particle parameters like size, shape, dispersion, and concentration lets you fine-tune the nanocomposite’s structural, optical, electrical, and thermal properties to fit the needs of certain device applications. Even small changes in the size or loading of nanoparticles can cause big changes in how the composite reacts to light and heat. 8 Even with these benefits, getting nanoparticles to spread evenly throughout the polymer matrix is still a big problem. This is because the overall performance of the nanocomposite depends a lot on the distribution, crystallinity, shape, and interfacial structure of the particles in the matrix. Water-soluble polyvinyl alcohol (PVA) has great physical, electrical, mechanical, and optical properties. 9 Polyvinyl alcohol (PVA) is a polymer that has both carbon and hydroxyl groups in it. Research has been done on PVA polymer because of its amazing qualities, like how well it can absorb water and humidity and how easy it is to make. 10 Strontium titanate (SrTiO3) is a ferroelectric material that stands out from the rest because it has a cubic perovskite structure. It is cheap, good for the environment, and has great photocatalytic, conductivity, and paraelectric properties. 11 Cobalt oxide is a key functional material used in many technologies, including heterogeneous catalysts, anode materials in Li-ion rechargeable batteries, magnetism, optical devices, gas and humidity sensors, solar selective absorbers, pigments for ceramics and glasses, and catalysts for oxygen evolution and oxygen reduction reactions. 12 The integration of nanotechnology with biotechnology has profoundly influenced the fields of biology and biomedicine, leading to the emergence of nanobiotechnology as a multidisciplinary area that combines the principles of both technologies to develop innovative materials and systems. Nanomaterials have garnered significant research interest due to their unique optical, electrical, and structural characteristics, which are markedly different from those of their bulk equivalents. These distinctive attributes make them exceptionally promising for a wide range of industrial, technological, and biomedical applications. There has been a rise in the need for antibacterial materials in areas like textiles, food processing, water purification, pharmaceuticals, and food packaging. 13 Nonetheless, the toxicity of specific organic antimicrobial agents to human health, along with the increasing resistance of various bacterial strains to traditional antibiotics, has necessitated the investigation of alternative antimicrobial materials. In this context, inorganic compounds, especially metals and metal oxides, have become very popular because they are very effective against a wide range of bacteria. Because bacteria can become resistant to antibiotics through many different biochemical pathways, it is very important to create new antibiotics or change the ones that are already available. Nanotechnology provides a potent means to attain this objective by facilitating the precise modulation of the physicochemical properties of materials at the nanoscale, thereby substantially improving their biological efficacy. Nanomedicine, a significant subdiscipline of nanotechnology, has significantly enhanced disease diagnosis, therapeutic monitoring, drug delivery, and treatment regulation. 14 Researchers are still trying to figure out how nanoparticles kill bacteria, but they have come up with a few possible ways they could do it. One main way this happens is by nanoparticles constantly releasing metal ions that can interact with the cell walls and cytoplasmic membranes of bacteria through electrostatic forces. This interaction increases membrane permeability, leading to structural disruption and cell lysis. If these ions are taken in, they could stop the production of adenosine triphosphate (ATP), block respiratory enzymes, and make reactive oxygen species (ROS). Oxidative stress can harm cellular parts, especially nucleic acids. This happens when ions interact with the phosphorus or sulphur atoms in DNA, which leads to mistakes in replication, problems with cell division, and eventually the death of microbes. Ion-mediated denaturation of cytoplasmic ribosomes can also stop protein synthesis, which adds to the antibacterial effect. 15 Pressure sensors are very important in many fields, such as biomedical, aerospace, environmental, and automotive, where they are used to keep an eye on and control many processes and systems. These sensors can work in two main ways when made from nanocomposite materials: piezoresistive or pseudo-capacitive, depending on the materials used and the conditions outside. People often use key performance indicators (KPIs) like pressure range, sensitivity to small and large forces, and overall pressure responsiveness to sort and rate pressure sensors. Piezoresistive pressure sensors are especially interesting because they work by detecting changes in electrical resistance in the sensing element when mechanical stress or pressure is applied. This change in resistance directly measures strain, which makes it possible to accurately detect and measure pressure. 16 The current study is focused on the development of PVA-based nanocomposite films that are augmented with SrTiO3–Co2O3 nanoparticles. The objective is to synergistically integrate the flexibility and processability of PVA with the dielectric, optical, antibacterial, and pressure-sensitive properties of metal oxide nanofillers. The selection of SrTiO3 and Co2O3 is predicated on their complementary electrical and functional properties, which render the resultant nanocomposites suitable for antibacterial applications, pressure sensors, and optoelectronic devices.
Experimental part
Casting made it possible to make movies with strontium titanate (SrTiO3), cobalt oxide (Co2O3), and polyvinyl alcohol (PVA). After dissolving 1 g of PVA in 40 mL of distilled water, the polymers were swirled for 30 minutes at 70°C to make a more uniform solution. We added SrTiO3-Co2O3 nanoparticles to the PVA at 0, 2, 4, and 6 wt percent. After allowing the solution to dry at room temperature for 3 days, we carefully removed the (PVA/SrTiO3-Co2O3) nanocomposites (NCs) from the petri dish. A Nikon 73346 top-view optical microscope was used to examine the (SrTiO3-Co2O3) distribution. The UV/1800/Shimadzu spectrophotometer and Fourier transform infrared (FTIR) spectroscopy (Bruker business type vertex-70, German origin) were used to study the optical properties in the 200–800 nm wavelength range with a varied wave number of 500–4000 cm−1. LCR instruments that could operate between 100 Hz and 5 MHz were used to measure the dielectric characteristics of (PVA/SrTiO3-Co2O3) nanocomposites. The disc diffusion technique was used to assess the effectiveness of the (PVA/SrTiO3-Co2O3) nanocomposite in inhibiting the development of gram-positive staphylococcus and gram-negative bacteria, such as Escherichia coli. To evaluate the pressure sensor of the PVA/SrTiO3-Co2O3 nanocomposites, we measured the parallel capacitance between the specimen’s top and bottom poles at varied pressures (80-160 bar) using an LCR meter.
To get the absorbance, the formula below was used.
17
Here is the equation applied to determine transmittance (T).
18
Using the above equation, we can get the absorption coefficient (α).
19
The symbol A stands for absorbance, whereas the symbol d stands for thickness. Eq. defines the energy gap.
18
This equation finds the refractive index (n).
20
R represents the reflected image. Determine the extinction coefficient (k) by.
21
Finding the wavelength denoted by (λ) is possible. By using the real (ε1) and imaginary (ε2) components of the dielectric constant.
20
By using an equation, the optical conductivity (σop) is defined.
22
We use this equation to get the nanocomposites’ dielectric constant (ε′).
23
The symbol C0 stands for a vacuum capacitor, whereas Cp stands for parallel capacitance.
This equation determines the dielectric loss (ε˝).
24
D stands for displacement. To get the A.C. electrical conductivity, use the following formula.
25
Results and discussion
The Optical microscope of (PVA/SrTiO3-Co2O3) nanocomposites
Figure 1 shows, at a magnification of × 10, optical microscope images of PVA/SrTiO3-Co2O3 nanocomposite films at different concentrations. However, the images (a, b, c, and d) clearly show distinct differences among the nanocomposite films. At a weight percentage of 6%, (SrTiO3-Co2O3) NPs in PVA films form internal networks with other particles in the polymer. Charge carriers are able to move freely inside nanocomposites thanks to the pathways in the network.
26
Optical microscope images for (PVA/SrTiO3-Co2O3) samples: (a) for PVA, (b) 2 wt% SrTiO3-Co2O3 NPs, (c) 4 wt% SrTiO3-Co2O3 NPs, (d) 6 wt% (SrTiO3-Co2O3) NPs.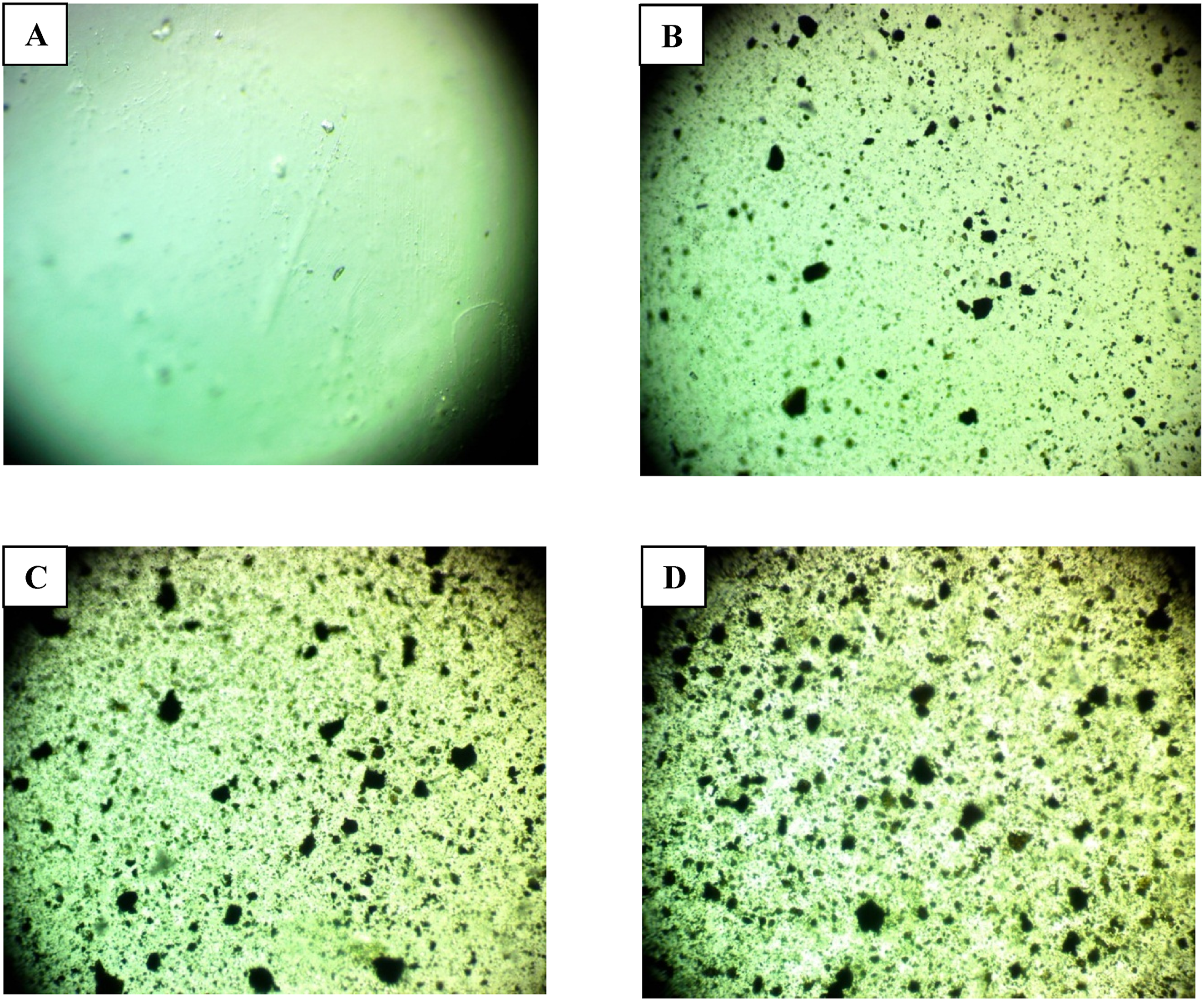
The FTIR Spectra of (PVA/SrTiO3-Co2O3) Nanocomposites
Figure 2 shows the Fourier Transform Infrared (FTIR) spectra of pure PVA and PVA/SrTiO3-Co2O3 nanocomposites with 2%, 4%, and 6% nanoparticle concentrations. The FTIR spectrum of pure PVA shows a number of characteristic absorption bands that match the vibrational modes of the polymer backbone. The O–H stretching vibration, which comes from strong hydrogen bonding between hydroxyl groups along the PVA chains, is responsible for the broad and intense absorption band that can be seen at about 3260–3280 cm−1. The band around 2923 cm−1 is due to the C–H bonds in CH2 groups stretching in an uneven way. The peak around 1415 cm−1 is due to C–H bending vibrations. There is also a sharp absorption band at 1085 cm−1 that is linked to the C–O stretching vibration of the secondary alcohol group in PVA. These characteristics collectively validate the semicrystalline structure of PVA and the prevalence of hydroxyl functionalities that can establish robust hydrogen-bonding networks.27,28 FTIR spectra of PVA/SrTiO3-Co2O3 nanocomposites.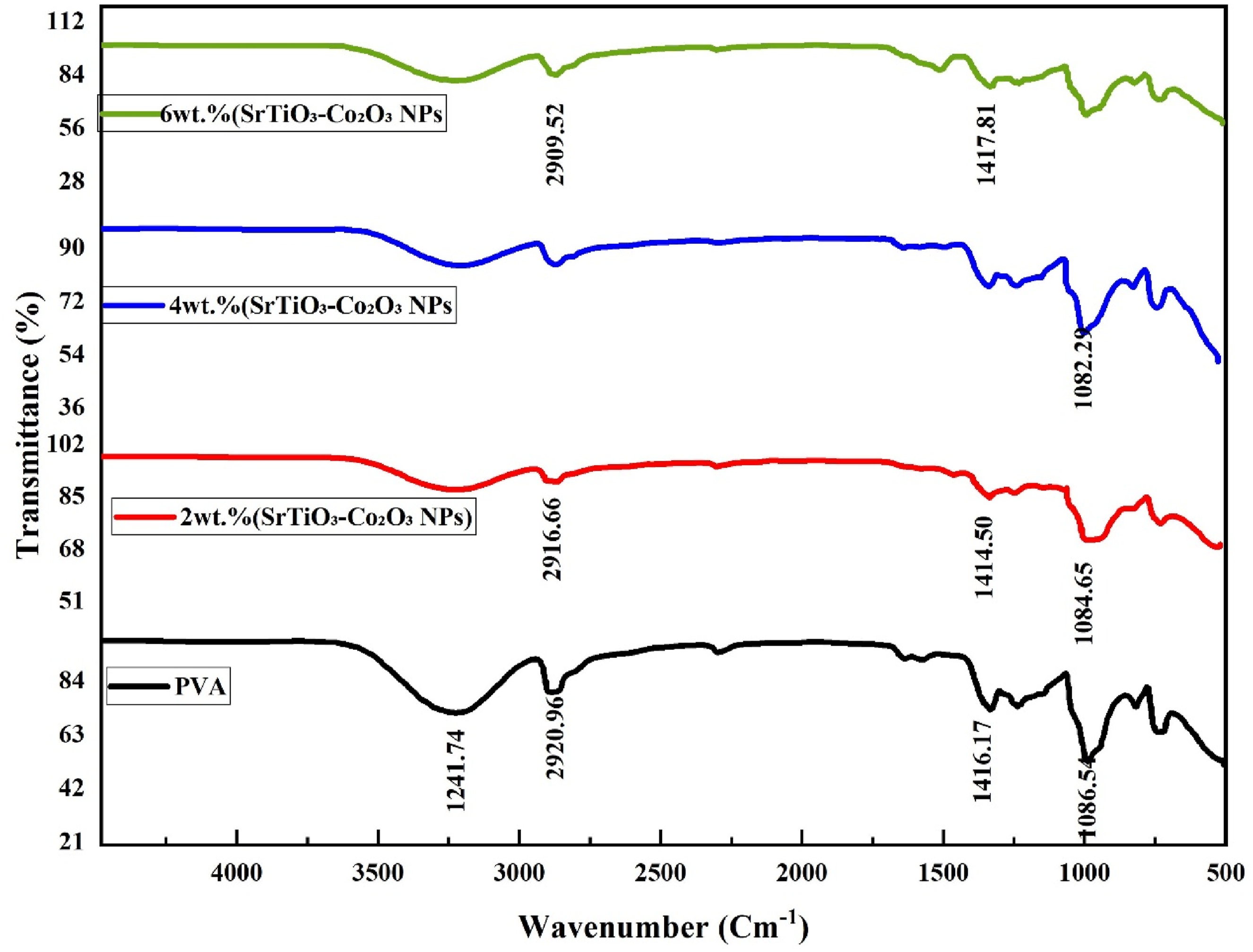
Upon the incorporation of 2 wt% SrTiO3–Co2O3 nanoparticles noticeable modifications appear in the spectral profile. The broad O–H stretching band slightly shifts to a lower wavenumber and becomes narrower, indicating the formation of new hydrogen bonds between the hydroxyl groups of PVA and the surface oxygen atoms of the SrTiO3–Co2O3 nanoparticles. This interaction indicates robust interfacial compatibility between the polymer matrix and the oxide fillers. The C–H stretching band at 2923 cm−1 shows a slight reduction in intensity, reflecting restricted segmental motion of the polymer chains due to physical interactions with the nanoparticles. Meanwhile, the C–O stretching band near 1084 cm−1 exhibits a minor shift and intensity decrease, confirming the possible coordination of oxygen atoms in PVA with Ti4+ or Co3+ ions present at the nanoparticle surface. These spectral changes provide compelling evidence of the initial formation of interfacial bonding between PVA and the SrTiO3–Co2O3 nanofillers.29,30
The FTIR spectra shows more noticeable changes when the nanoparticle loading is increased to 4 wt%. Around 3268 cm−1, the O–H stretching vibration gets wider and a little stronger. This suggests that the hydrogen-bonding interactions between the PVA matrix and the dispersed metal oxide nanoparticles are getting stronger. The C–O stretching band near 1082 cm−1 also moves and gets even wider at the same time. 31 This means that new coordination bonds, like Ti–O–C or Co–O–C linkages, are forming where the polymer and the nanoparticles meet. The weakening of the C–H vibrations shows that the chains are less mobile and the molecules are more tangled up in the composite structure. These results show that when the concentration of nanoparticles goes up, the interactions between the particles become stronger, which makes the polymer network more compact and crosslinked.32–34
When the loading is higher at 6 wt% SrTiO3–Co2O3, the differences in the spectra become even more pronounced. The O–H stretching band around 3259 cm−1 gets wider and moves even more, showing that a strong hydrogen-bonding network has formed between the PVA chains and the oxide nanoparticles. The C–O and C–H bands, which are close to 1417 and 1085 cm−1, also show clear shifts towards lower wavenumbers. This means that there are stronger coordination interactions, and the polymer structure may be changing. The observed decrease in transmittance and enhanced band definition indicate a partial alteration of the polymer crystallinity, aligning with the incorporation of inorganic nanoparticles that disturb the organized PVA chain configuration. 35
The gradual changes in the O–H and C–O stretching regions as the SrTiO3–Co2O3 content rises show that the nanoparticles were successfully added to the PVA matrix and that strong interfacial interactions were formed through hydrogen bonding and metal–oxygen coordination. These interactions at the molecular level are likely to have a big effect on the nanocomposite system’s structure, electrical properties, and ability to sense things. The PVA/SrTiO3–Co2O3 nanocomposites may have better dielectric properties, mechanical stability, and pressure-sensing performance because of better interfacial bonding and less polymer crystallinity. This makes them good candidates for advanced optoelectronic and sensor applications. 36
The Optical properties of (PVA/SrTiO3-Co2O3) nanocomposites
Figure 3 illustrates the absorption spectra of PVA/SrTiO3-Co2O3 nanocomposites as a function of wavelength. Films frequently exhibit minimal absorption values in the visible and near-infrared range. This elucidates why objects absorb light of longer wavelengths, as fewer photons interact effectively with atoms and transmit diminished energy. As the wavelength of the photon decreases, its frequency increases, leading to a greater absorbance when it interacts with matter. This indicates that the unbound electrons will merely absorb the incident light. This results in a rise in absorbance when the weight percentages of SrTiO3-Co2O3 nanoparticles increase.37–39 Difference of absorbance for (PVA/SrTiO3-Co2O3) NCS with wavelength.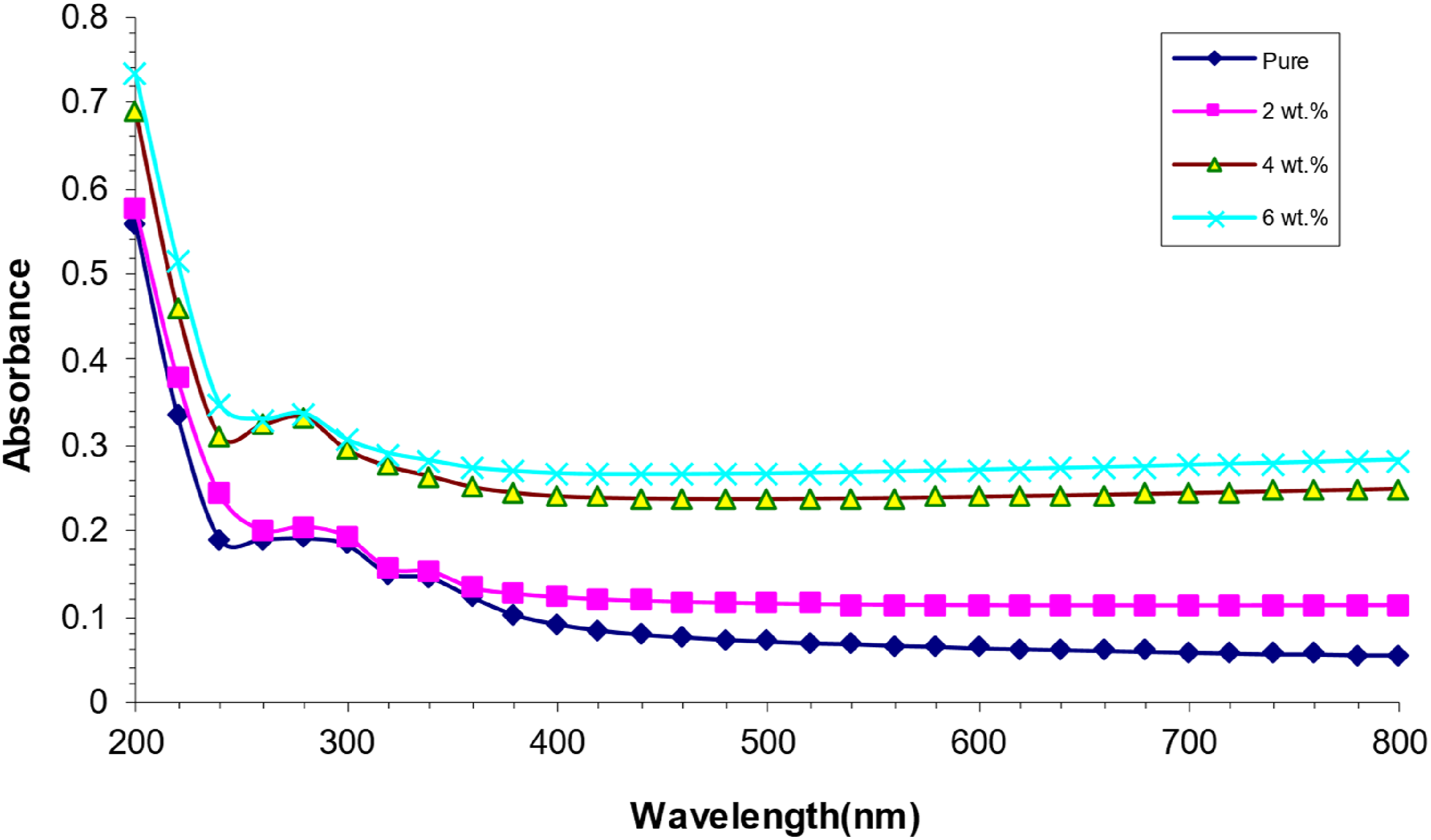
Figure 4 illustrates the variation in transmittance of PVA/SrTiO3-Co2O3 nanocomposites with respect to wavelength. The graph indicates that transmittance diminishes as the concentration of SrTiO3-Co2O3 nanoparticles increases. The SrTiO3-Co2O3 nanoparticles introduce electrons that occupy previously unfilled energy band positions after being elevated to a higher energy level; thus, the process does not result in outer-orbital electron emission, where electrons are susceptible to electromagnetic radiation forces.
40
Pure PVA exhibits limited permeability and high transmittance, permitting docents to traverse it while absorbing a portion of the incident light. The passage of an electron to the conduction band necessitates the breaking of an electron bond, which demands a high-energy photon.
41
Change transmittance spectra of (PVA/SrTiO3-Co2O3) NCS with wavelength.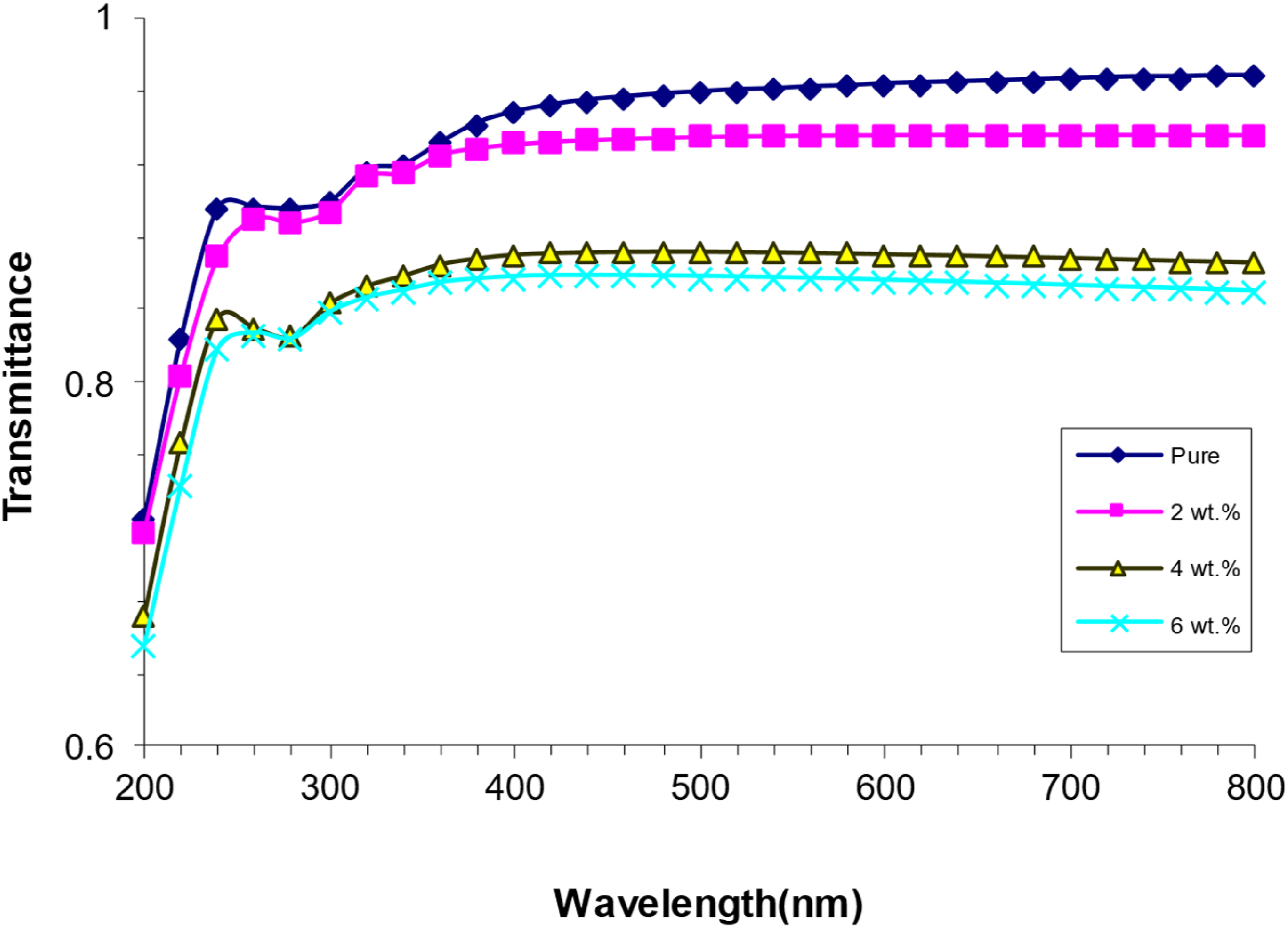
The absorbance coefficient of PVA/SrTiO3-Co2O3 nanocomposites varies with photon energy, as shown in Figure 5. At low energy and high wavelength, the absorption coefficient is the lowest because the incoming photon does not have enough energy to transfer an electron from the valence band (V.B.) to the conduction band (C.B.). Larger absorption at higher energies is indicative of an abundance of possible electron transitions. This forbidden energy difference must be larger than the incoming photon’s energy in order to transfer the electron from the V.B. to the C.B.42,43 At high energies, when the energy and moment are maintained by the (photons and electrons), the absorption coefficient has a major impact on the nature of electron transmission since a direct transition of an electron is predicted. In a phonon-mediated indirect transition, the electronic momentum of an electron is likely to be retained,
44
since absorption coefficients are (of 104 cm−1) at low energies. Absorbance data for PVA/SrTiO3-Co2O3 nanocomposites reveal that the electron transition occurs at wavelengths of less than 104 cm−1. PVA/SrTiO3-Co2O3 NCS absorption coefficient versus photon energy.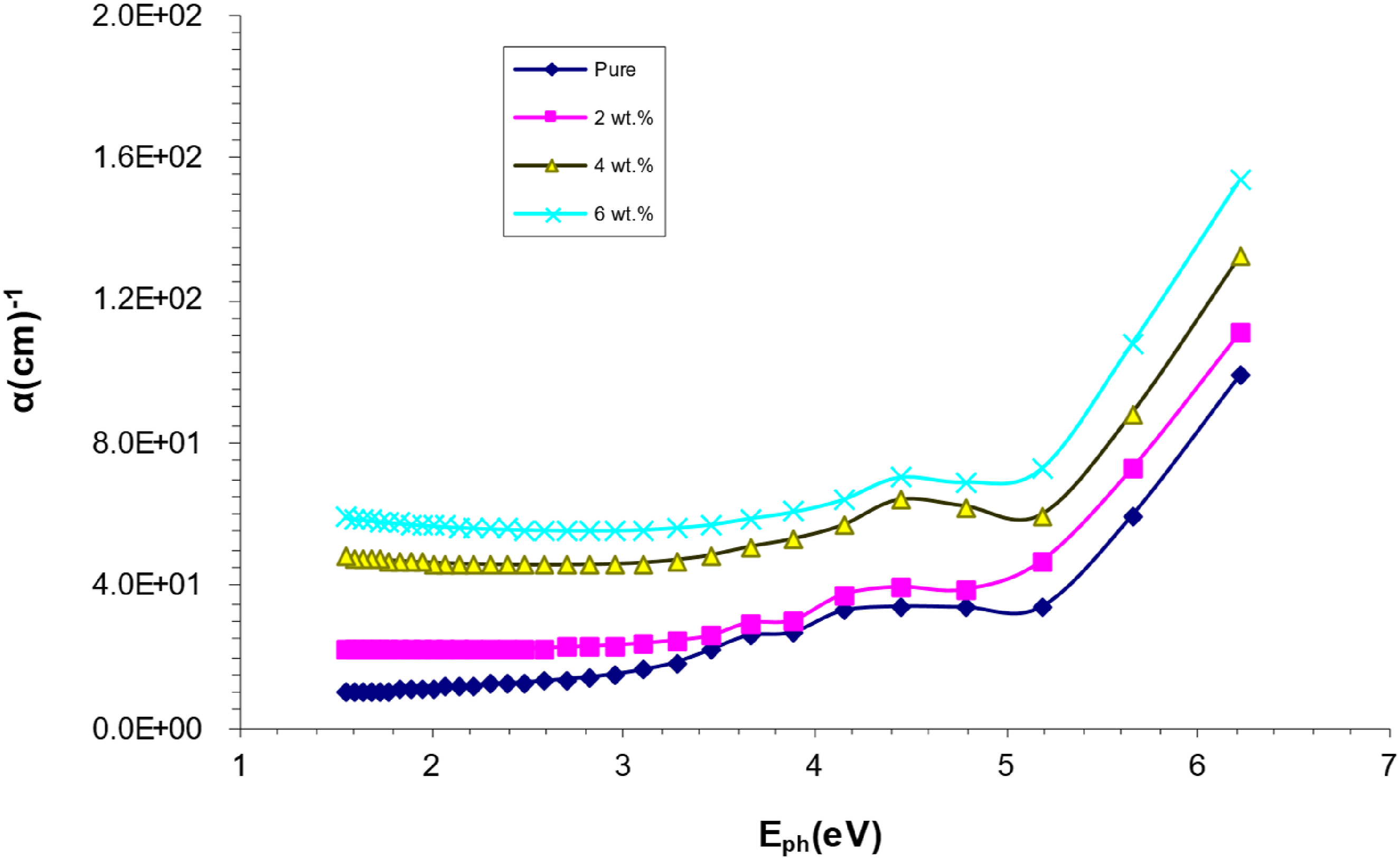
As shown in Figure 6, the photon energy of PVA/SrTiO3-Co2O3 nanocomposites correlates with (hυ)1/2. An indirect energy-gap transition can be achieved by connecting the apex of the curve to the x-axis at the point where (hυ)1/2 equals zero. Optical energy gap decreases as SrTiO3-Co2O3 nanoparticle concentration increases.45,46 This is because, due to the heterogeneous properties of nanocomposites (i.e., electronic conduction is dependent upon the added concentration), the density of localised states increases with increasing concentrations of SrTiO3-Co2O3 nanoparticles.47,48 The correlation between (αhυ)1/2 (cm−1 eV)1/2 and the photon’s energy for (PVA/SrTiO3-Co2O3) NCS.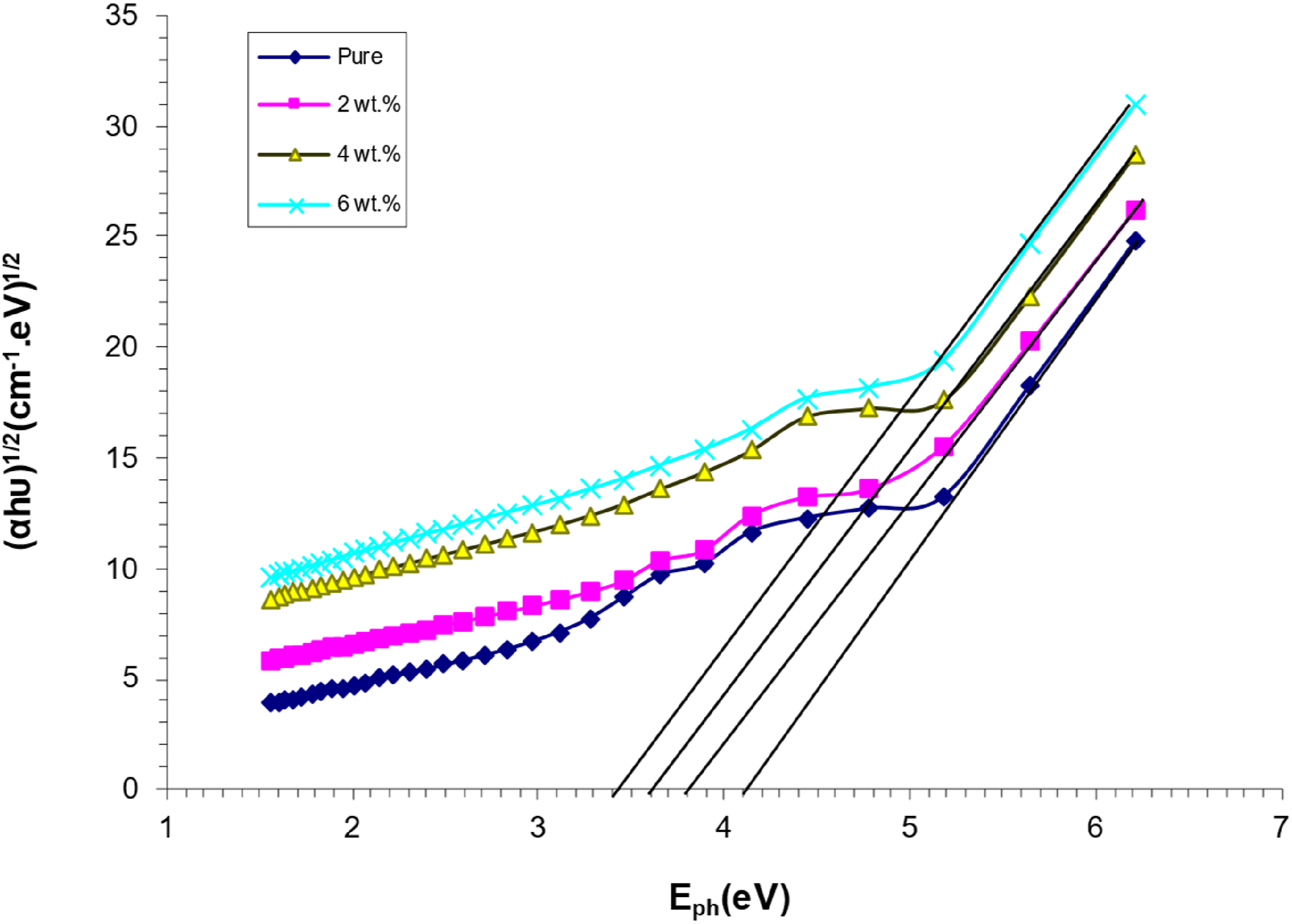
As shown in Figure 7, the photon energy of PVA/SrTiO3-Co2O3 nanocomposites correlates with (hυ)1/3. For the indirect energy gap, a similar approach takes into account the prohibited transition. Table 1 shows values of the energy gap for (PVA/SrTiO3-Co2O3) NCs. The relationship between (αhv)1/3(cm−1 eV)1/3 and photon energy of (PVA/SrTiO3-Co2O3) NCS. The values of the energy gap for (PVA/SrTiO3-Co2O3) NCs.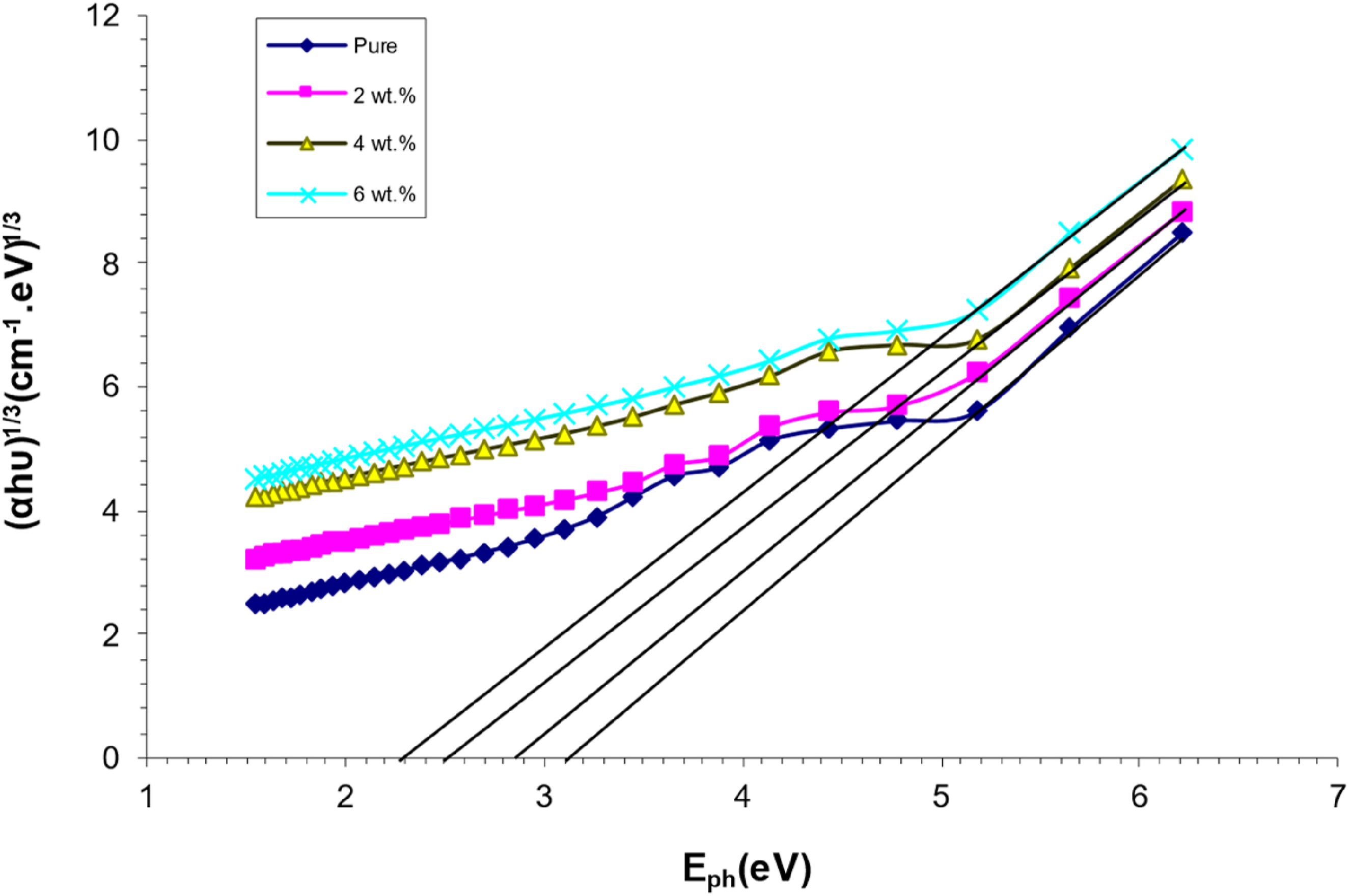
Figure 8 illustrates the correlation between the refractive index and wavelength for nanocomposites composed of PVA/SrTiO3-Co2O3. The figure illustrates that the refractive index increases with the rising weight percentages of (SrTiO3-Co2O3) nanoparticles in the PVA concentration, attributed to the concomitant increase in nanocomposite density. The UV spectrum exhibits elevated refractive index values due to decreased transmission. On the other hand, the visible spectrum exhibits diminished values due to increased transmittance in this region.49,50 Refractive index for (PVA/SrTiO3-Co2O3) NCS variation with wavelength.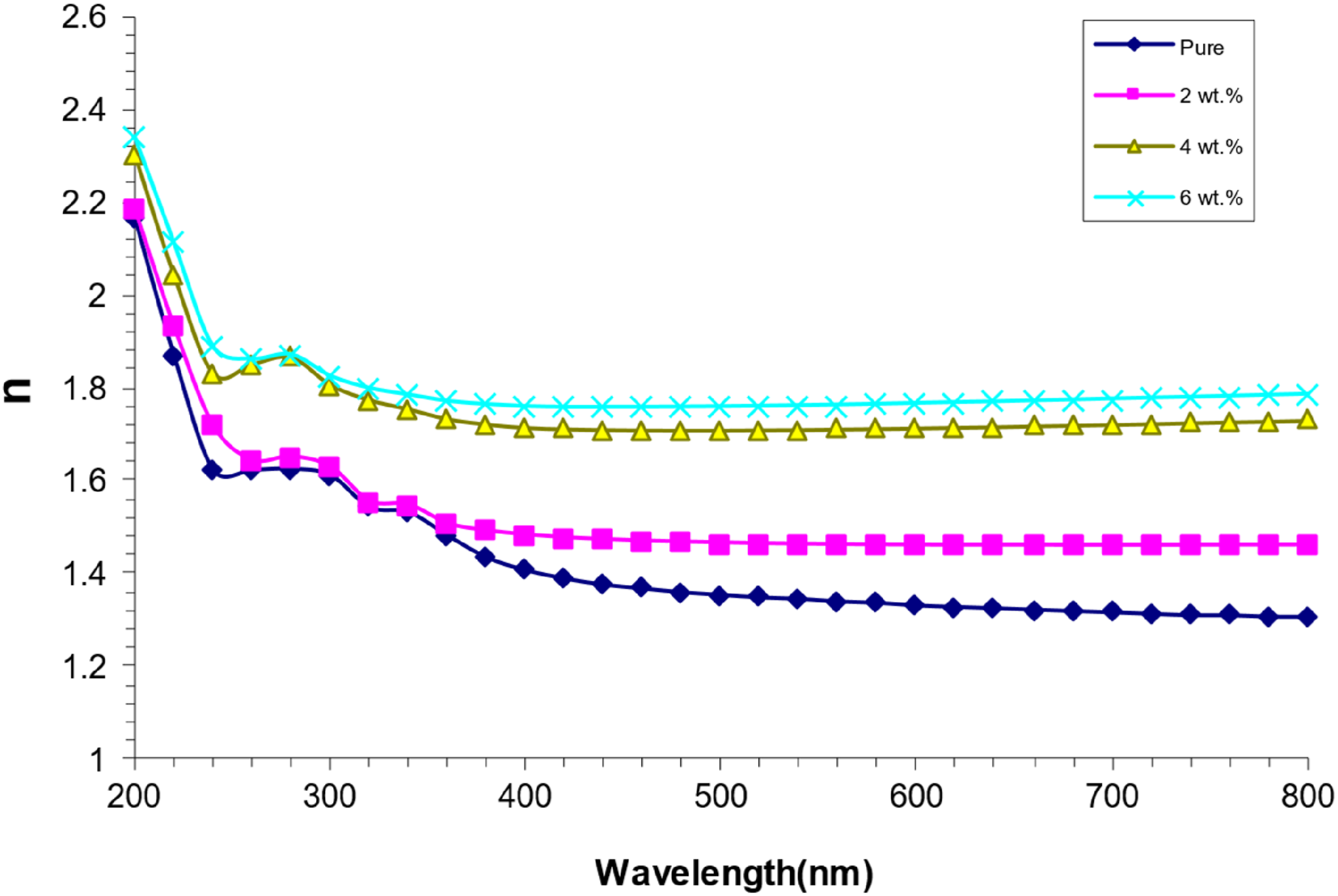
Figure 9 shows the relationship between the extinction coefficient and wavelength for nanocomposites made of PVA/SrTiO3-Co2O3. Increasing the concentration of SrTiO3-Co2O3 nanoparticles causes the extinction coefficient to grow, as shown in this figure. This is because the absorption coefficient is so high. This phenomenon arises from the elevation of the absorption coefficient with higher concentrations of SrTiO3-Co2O3 nanoparticles. This outcome indicates that the polymer’s structure will be influenced by the presence of SrTiO3-Co2O3 nanoparticles.51,52 Variation extinction coefficient with wavelength for (PVA/SrTiO3-Co2O3) nanocomposites.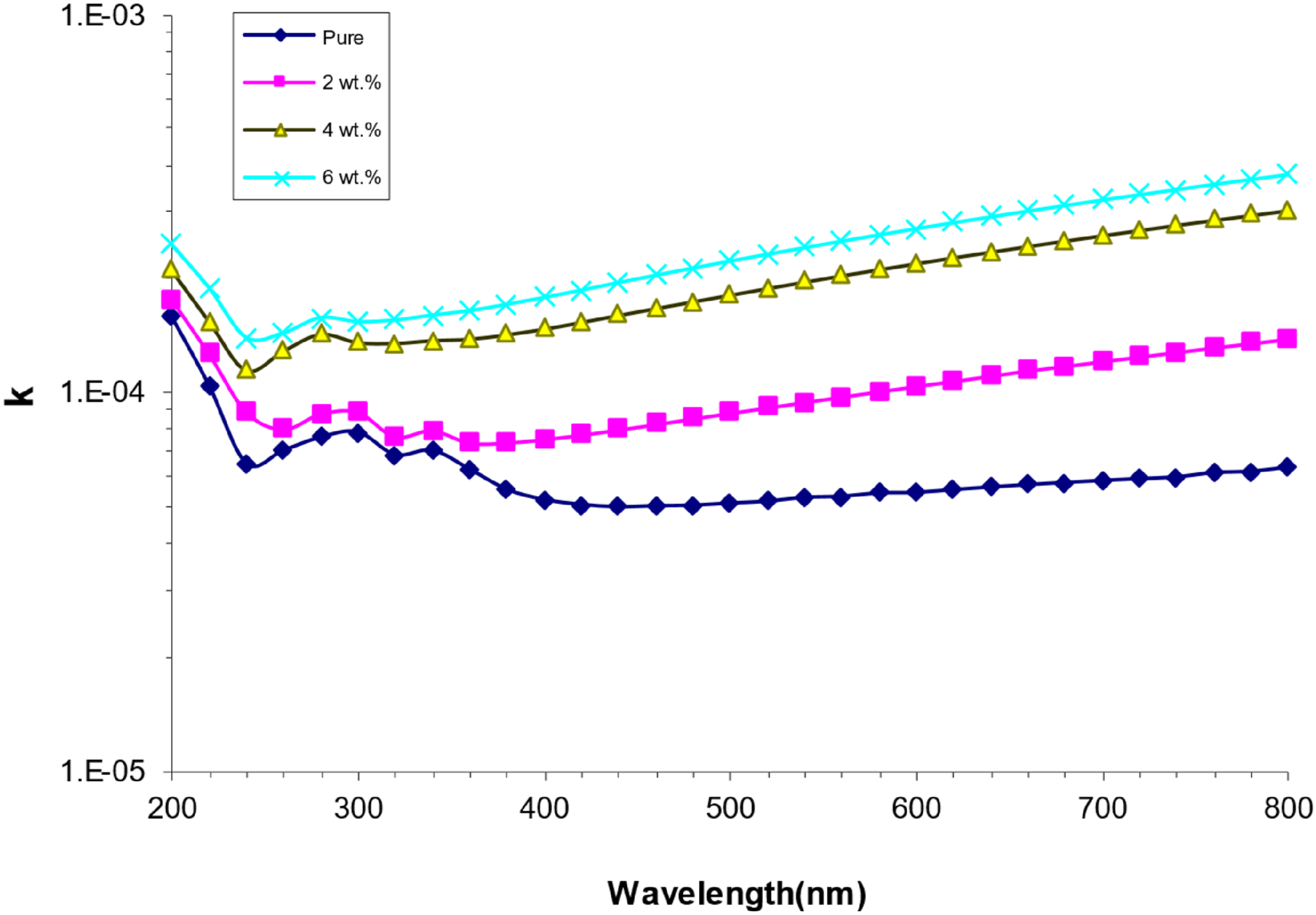
Figure 10 illustrates the fluctuation of the real component of the dielectric constant with respect to wavelength for (PVA/SrTiO3-Co2O3) nanocomposites. This figure demonstrates that the real dielectric constant increases with higher concentrations of SrTiO3-Co2O3 nanoparticles. The reduced value of n2 indicates that ԑ1 is significantly reliant on k2.53,54 Actual dielectric constant (ԑ1) as a function of incident wavelength for (PVA/SrTiO3-Co2O3) NCS.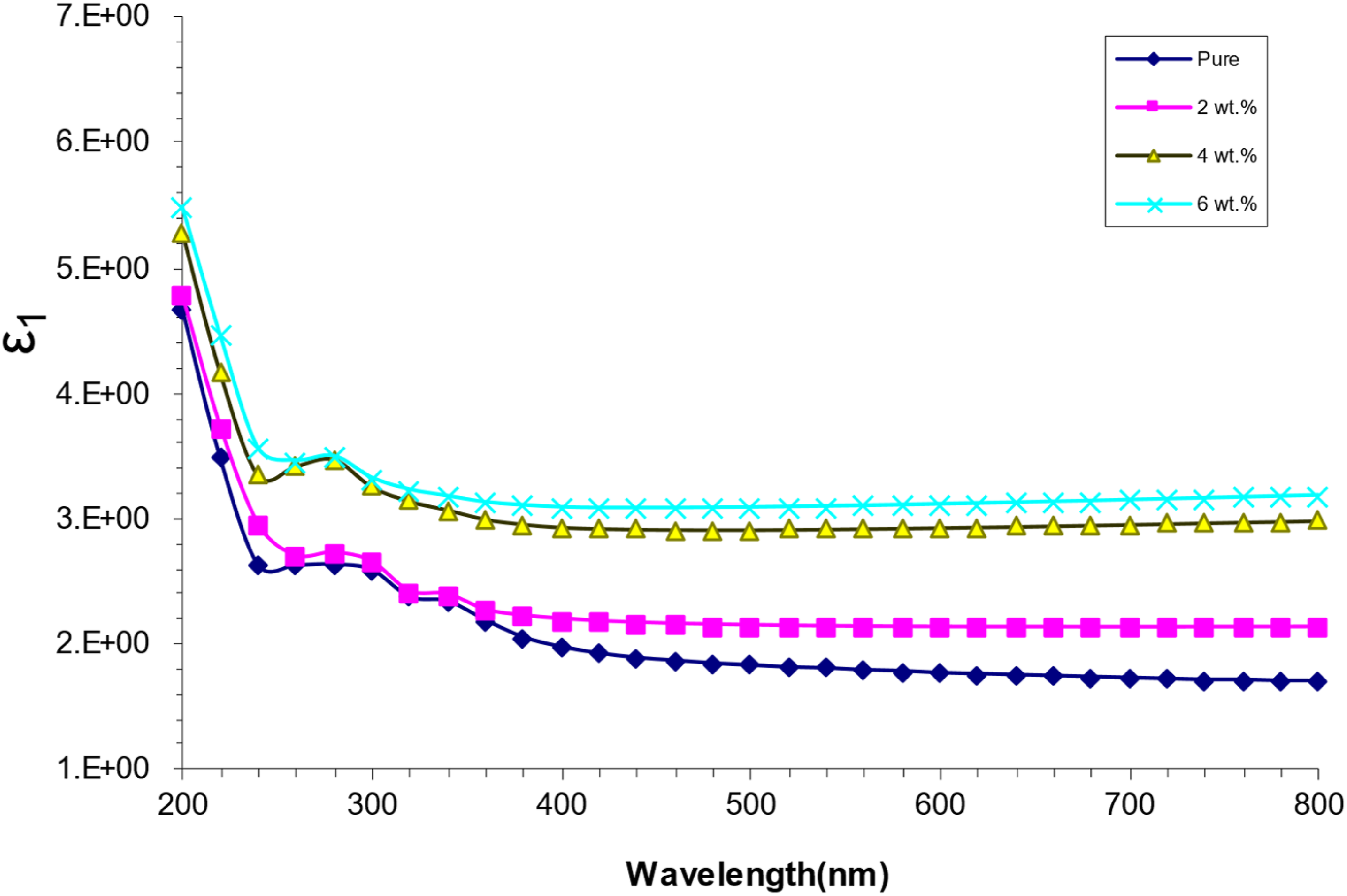
Figure 11 shows how the wavelength and the imaginary part of the dielectric constant are related for (PVA/SrTiO3-Co2O3) nanocomposites. The interaction between (ε2 and k) makes the absorption coefficient change with k, which in turn makes the values of the imaginary component change. The imaginary part of the dielectric constant, on the other hand, gets bigger as the number of nanoparticles goes up. The nanoparticles’ higher electric polarization causes more dipoles to form, which in turn raises the dielectric constant.55,56 Imaginary dielectric constant (ԑ2) as a function of wavelength for (PVA/SrTiO3-Co2O3) NCS.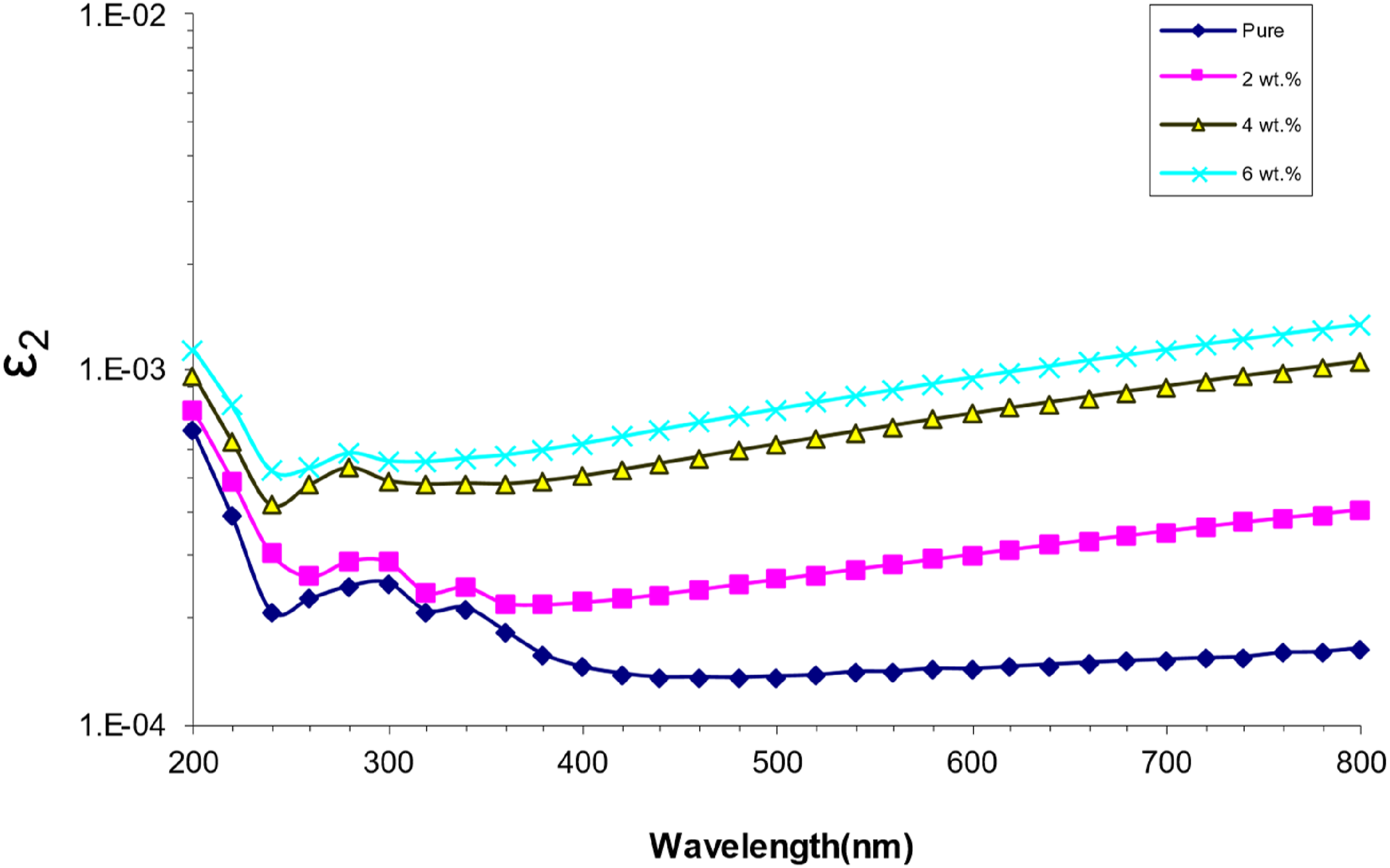
Figure 12 shows how the optical conductivity of PVA/SrTiO3-Co2O3 nanocomposites changes with wavelength. Increasing the SrTiO3-Co2O3 ratio in the PVA improved its optical conductivity. Adding extra band gap levels improves the flow of electrons from the valence band to the conduction band through localized levels. This closes the band gap and makes conductivity better.57,58 Optical conductivity of (PVA/SrTiO3-Co2O3) nanocomposite variation with wavelength.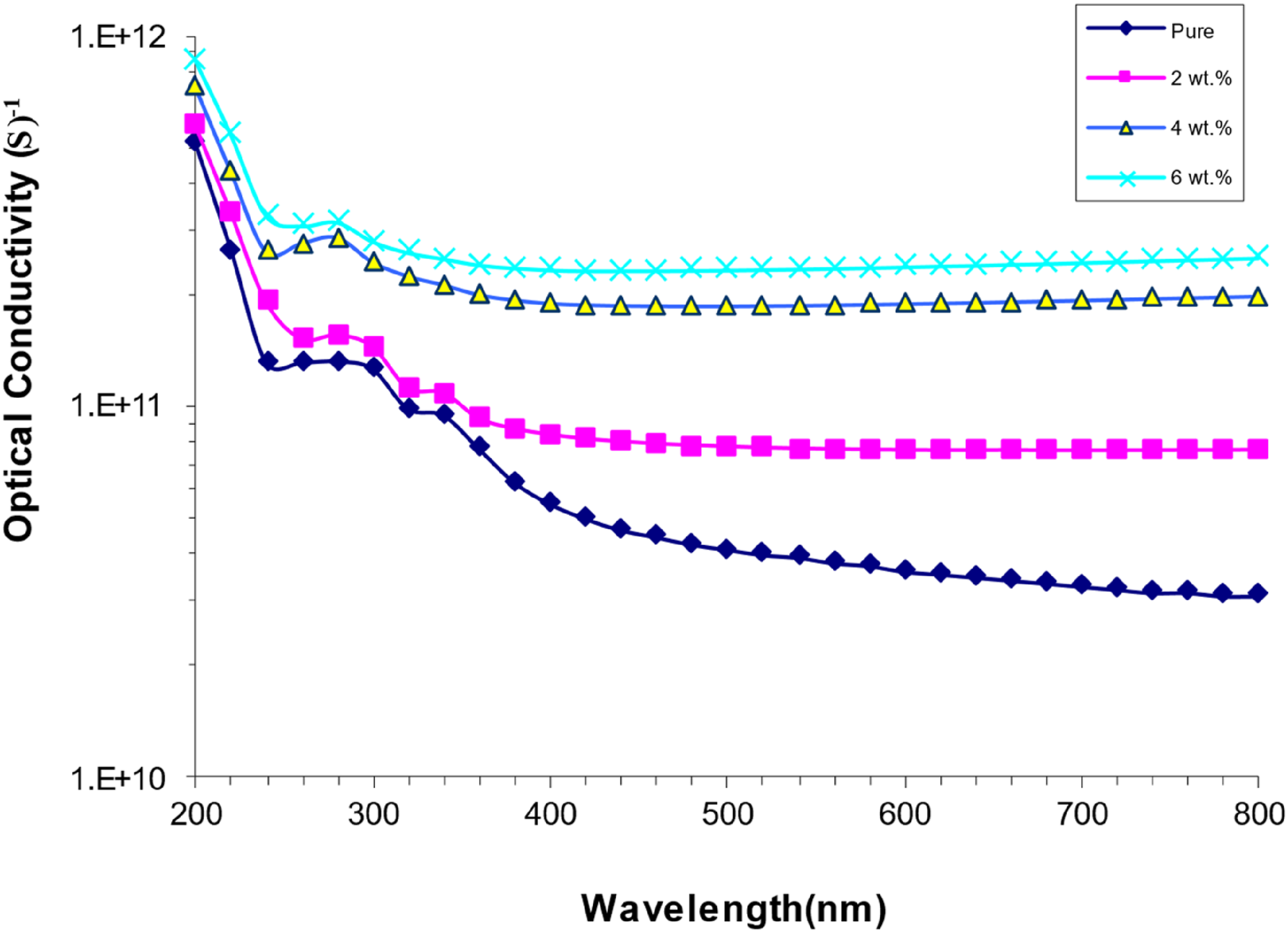
The Dielectric Properties of (PVA/SrTiO3-Co2O3) Nanocomposites
Nanocomposites made of polyvinyl alcohol (PVA), strontium titanate (SrTiO3), and cobalt oxide (Co2O3), called PVA/SrTiO3-Co2O3 nanocomposites, have dielectric properties that change with frequency (as shown in Figure 13). The data indicates that the permittivity (έ) has a negative correlation with frequency (f). The dipoles of the insulating materials align with the applied electric field at low frequencies, leading to charge buildup. As a result of this buildup, polarisation becomes more extreme, and the (έ).59,60 The dipoles are unable to rapidly align with the direction of the applied electric field at higher frequencies, leading to a decrease in polarisation and, therefore, a decrease in the (έ) value. Antennas for communication and microwave components are only two of numerous uses for this property.61,62 Behaviour of (έ) with a frequency for (PVA/SrTiO3-Co2O3) nanocomposites.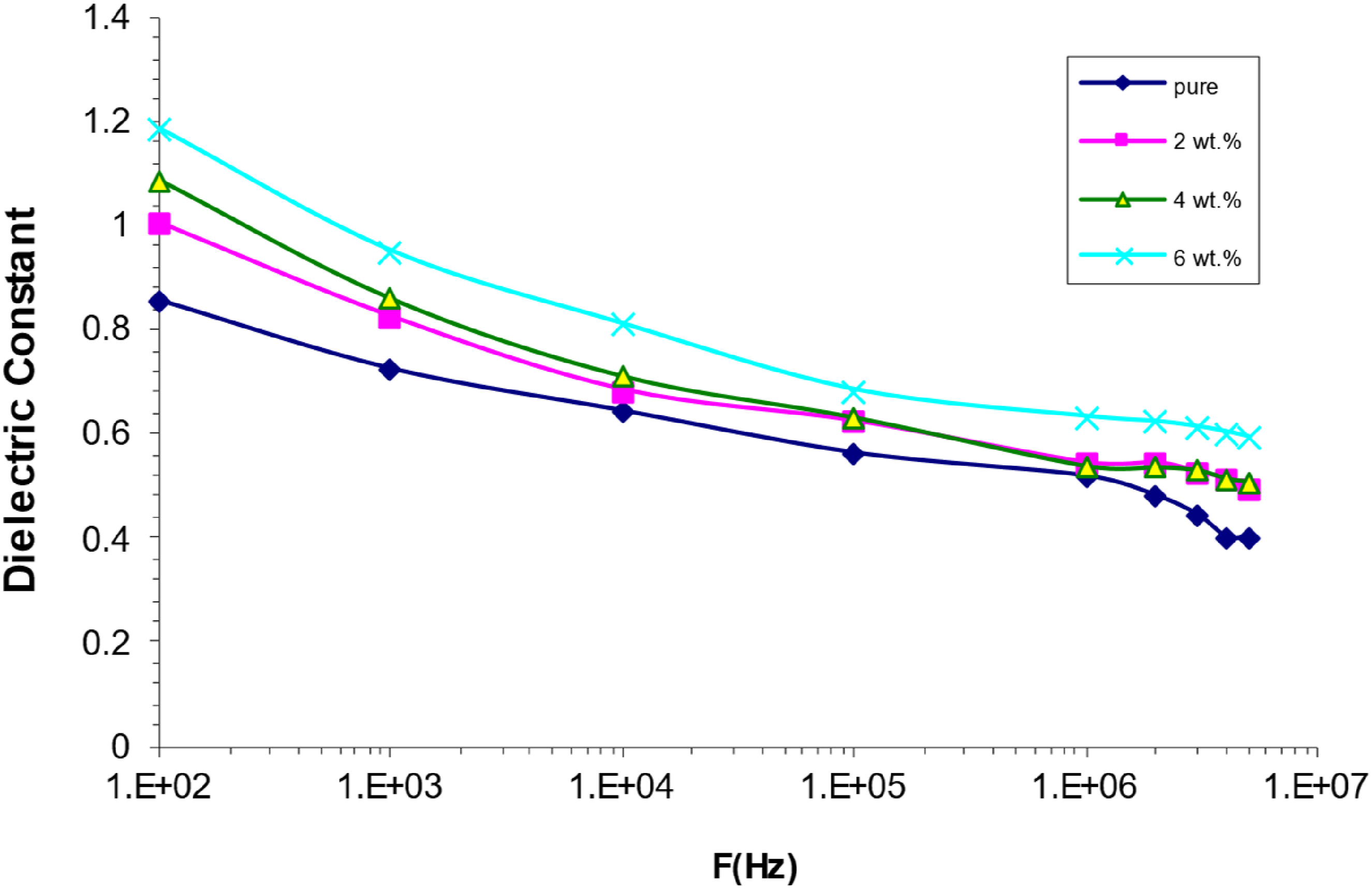
Nanocomposites containing polyvinyl alcohol (PVA), strontium titanate (SrTiO3), and cobalt oxide (Co2O3) exhibit frequency-dependent dielectric loss, as seen in Figure 14. The graph data shows that dielectric loss reduces with increasing frequency, suggesting that the two variables are inversely related. The presence of mobile charges inside the polymer backbone is responsible for this phenomenon. This effect develops as frequency increases and the influence of space charge polarisation decreases.63,64 More electrons cause an increase in dielectric loss of PVA/SrTiO3-Co2O3 NCs, particularly at intermediate frequencies, although higher frequencies cause a reduction.65,66 Behaviour of (ε˝) with a frequency for (PVA/SrTiO3-Co2O3) nanocomposites.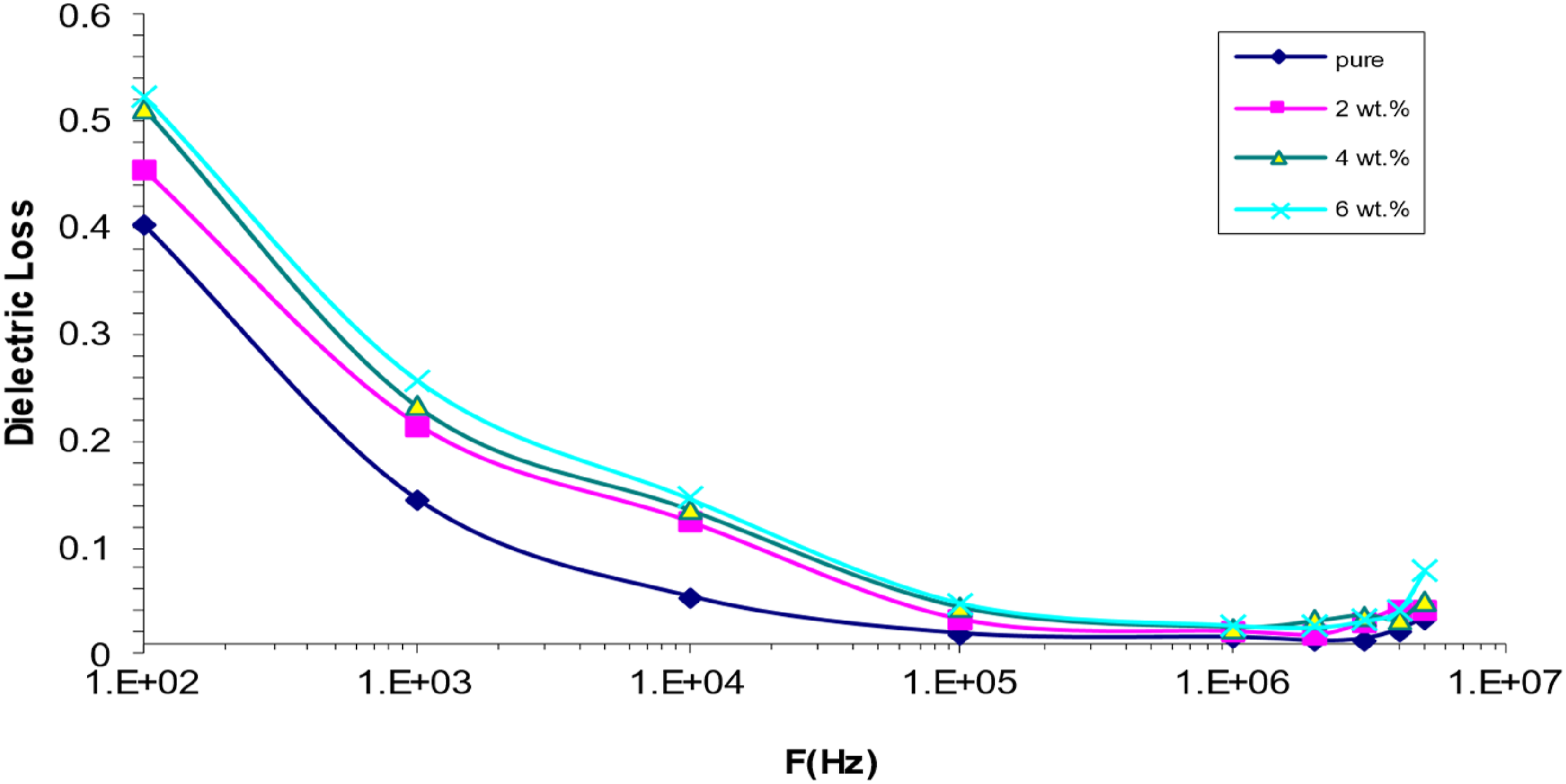
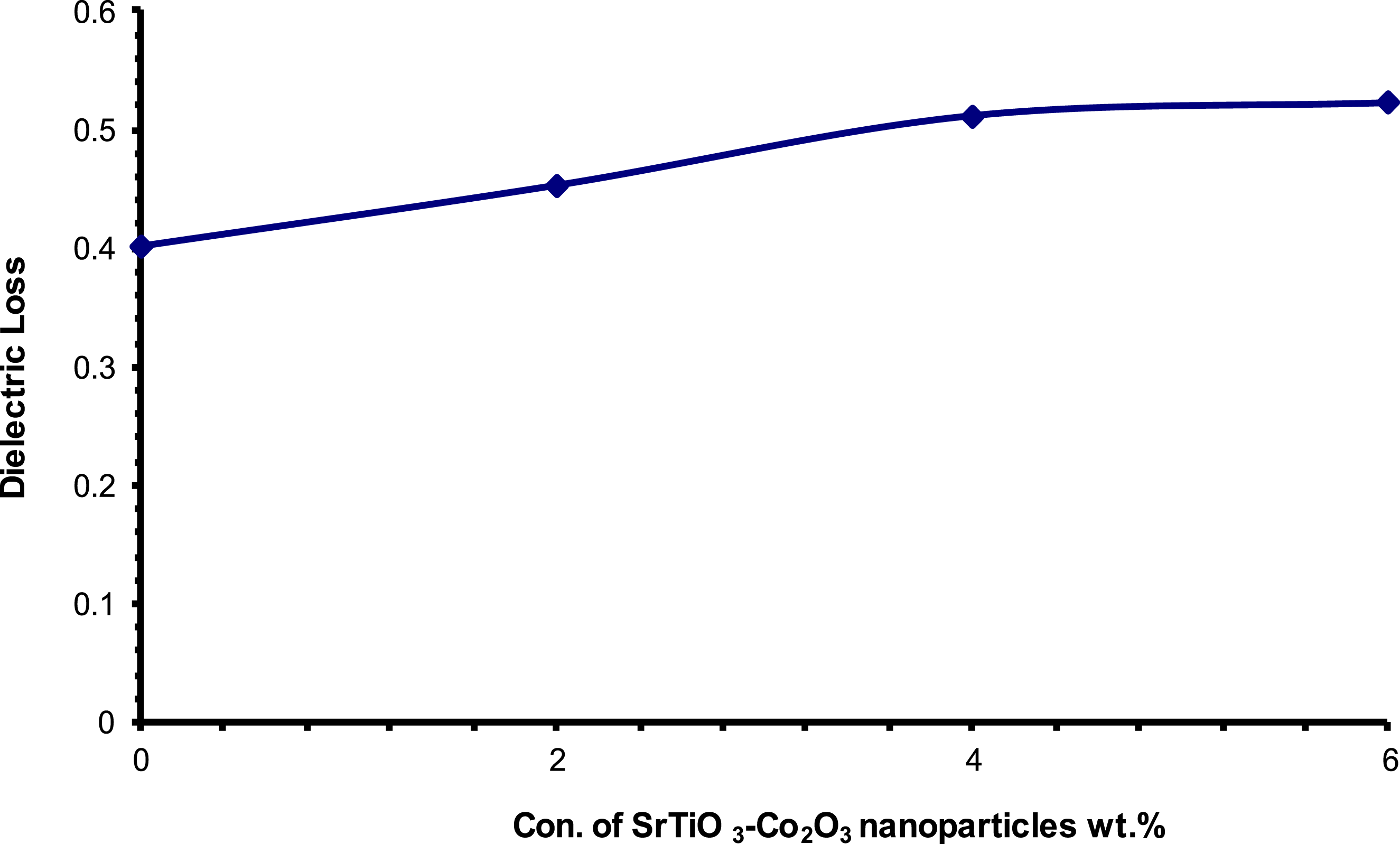
Figures 15 and 16 show the correlation between nanoparticle concentration and dielectric (constant and loss) values in PVA/SrTiO3-Co2O3 nanocomposites, as well as the connection between the two in a general context. According to the results, there is a correlation between the quantity of SrTiO3-Co2O3 nanoparticles and the increases in ( Influence of (SrTiO3-Co2O3) nanoparticle content on the (έ) for (PVA/SrTiO3-Co2O3) nanocomposites at 100 Hz. Influence of (SrTiO3-Co2O3) nanoparticle content on the (ε˝) for (PVA/SrTiO3-Co2O3) nanocomposites at 100 Hz.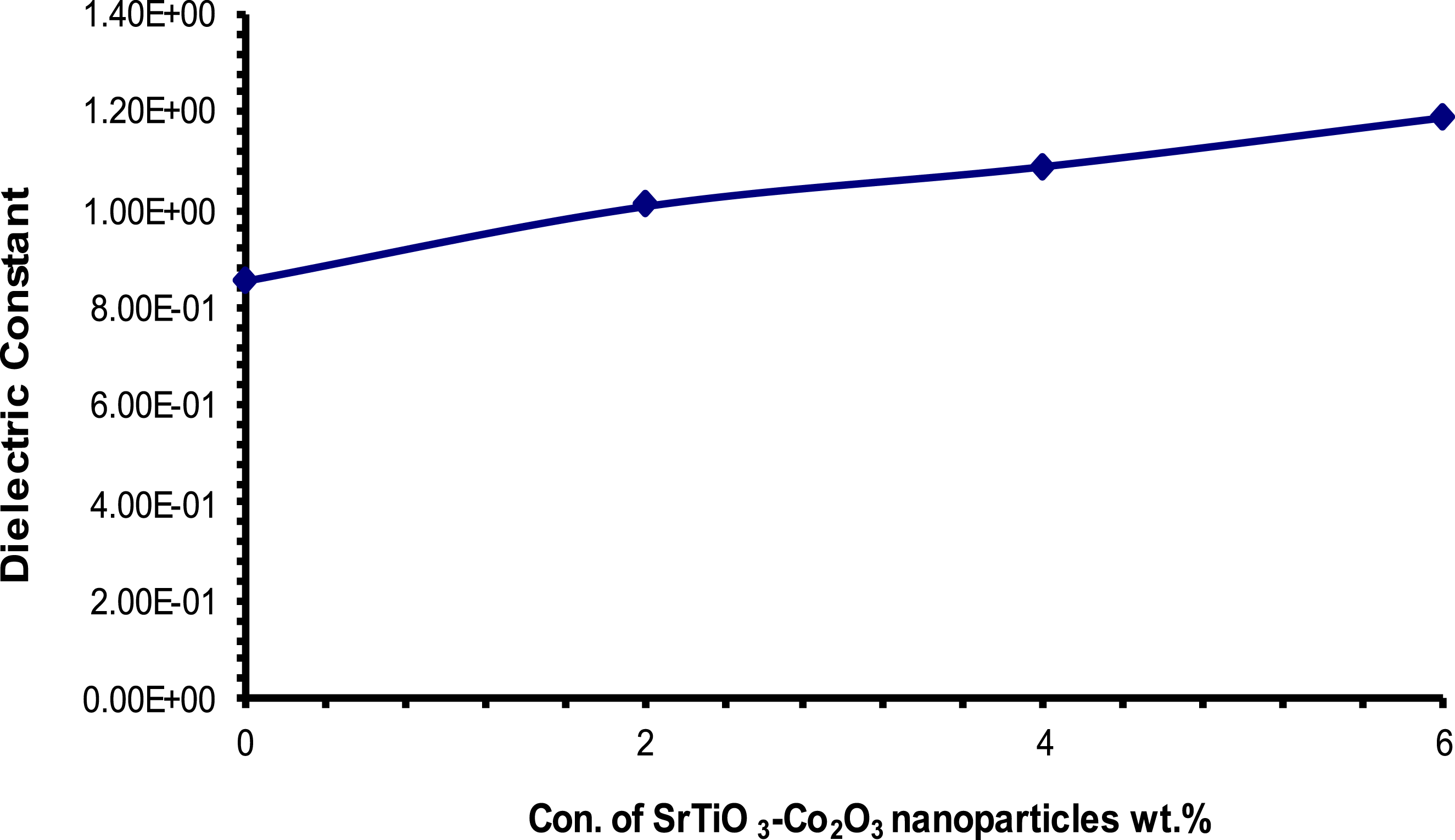
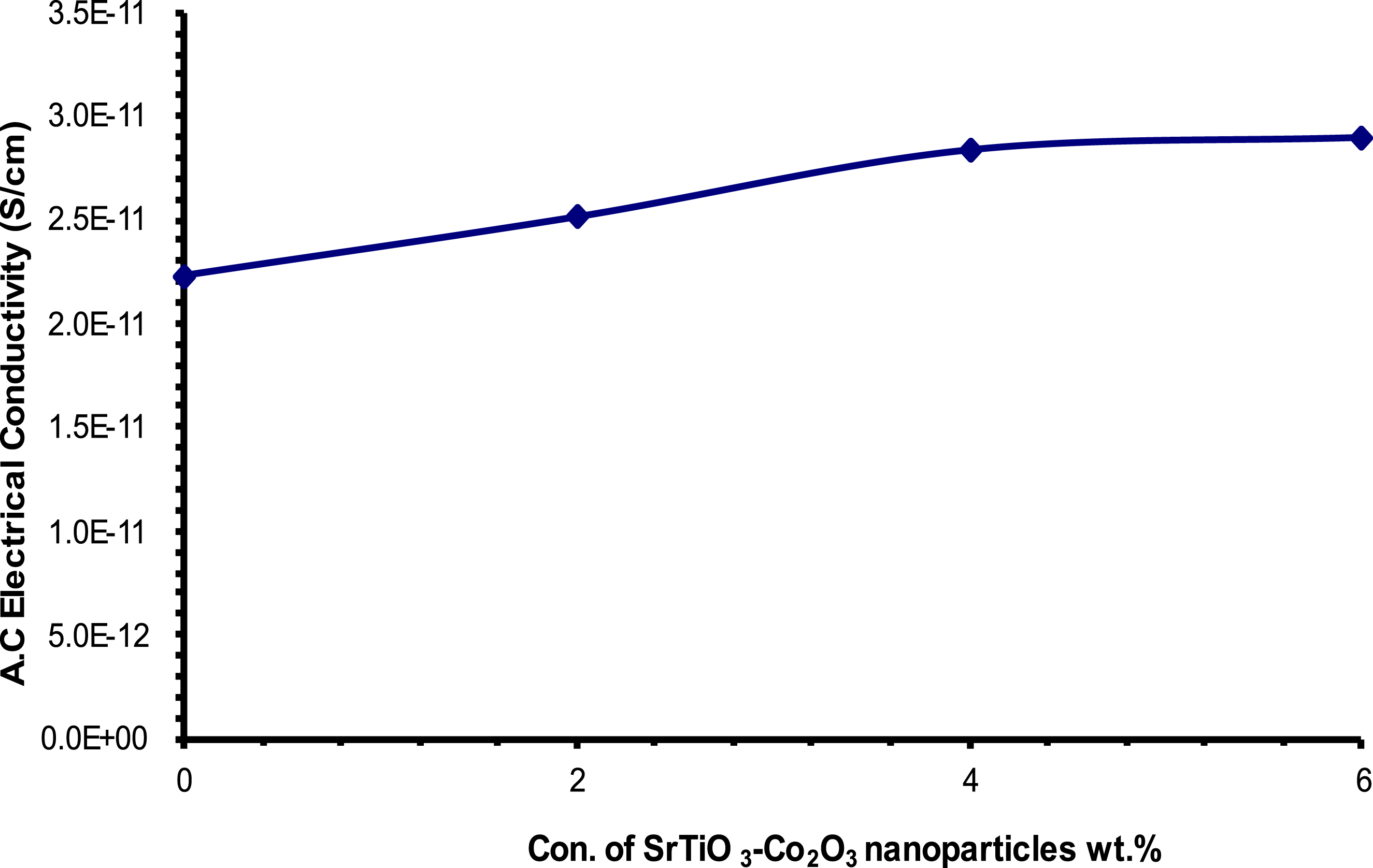
Figures 17 and 18 show the relationship between the A.C. electrical conductivity performance and the (frequency and concentration of SrTiO3-Co2O3 nanoparticles) in the (PVA/SrTiO3-Co2O3) nanocomposites respectively. According to the graph, there is a direct relationship between the electric field frequency and the A.C. electrical conductivity of all the nanocomposite samples. The movement of ions inside the clusters and the mobility of electrically charged particles might account for this phenomenon.71,72 Because fewer ions may travel through the medium at lower frequencies, electrical conductivity decreases as charge builds up at the electrode-electrolyte interface. The conductivity increases with increasing concentrations of SrTiO3-Co2O3 nanoparticles. A rise in electric charge is a byproduct of making fully saturated nanoparticles.73,74 Difference of electrical conductivity for (PVA/SrTiO3-Co2O3) NCs with frequency. Difference of electrical conductivity for (PVA/SrTiO3-Co2O3) nanocomposites with (SrTiO3-Co2O3) nanoparticles.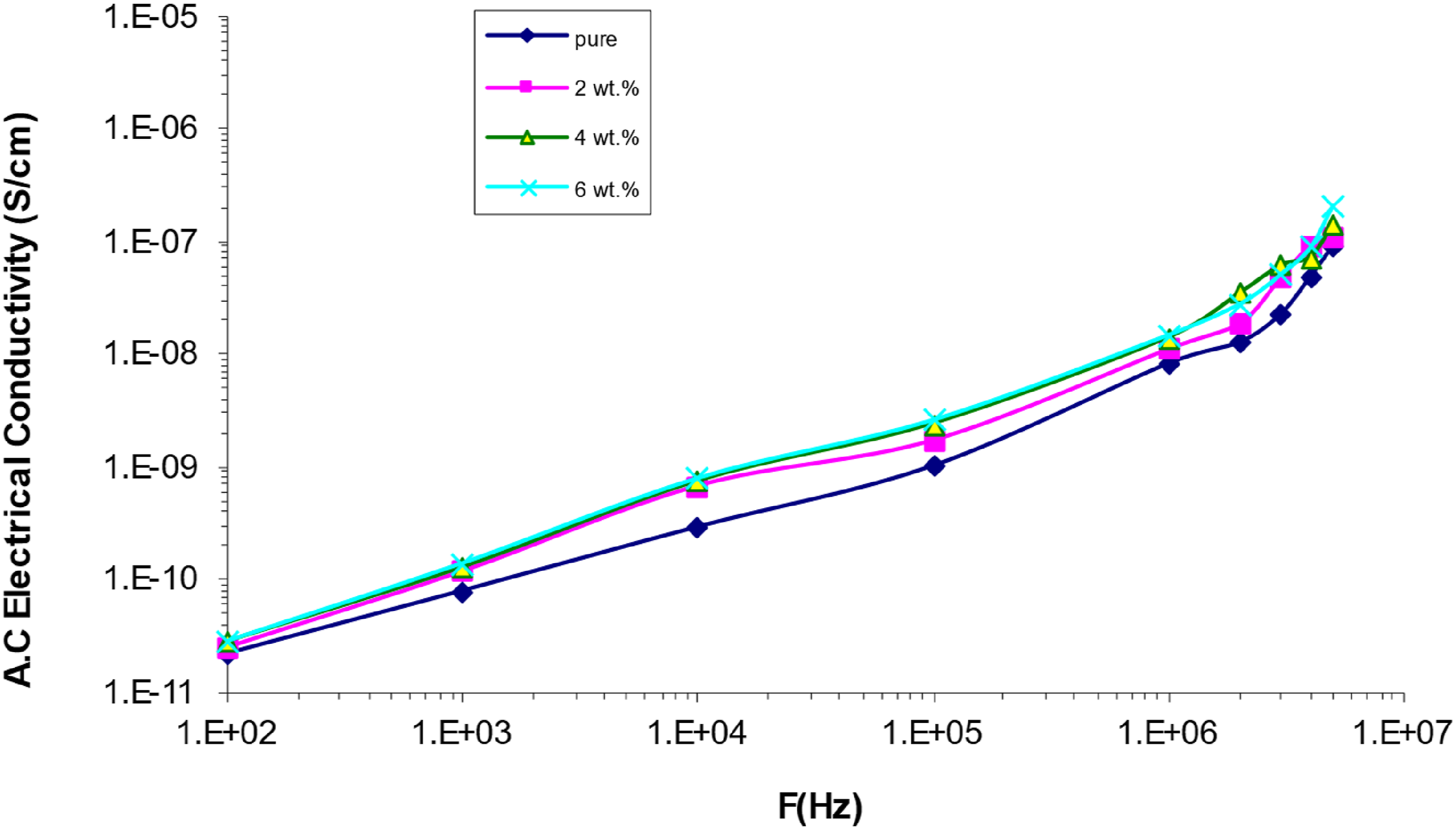
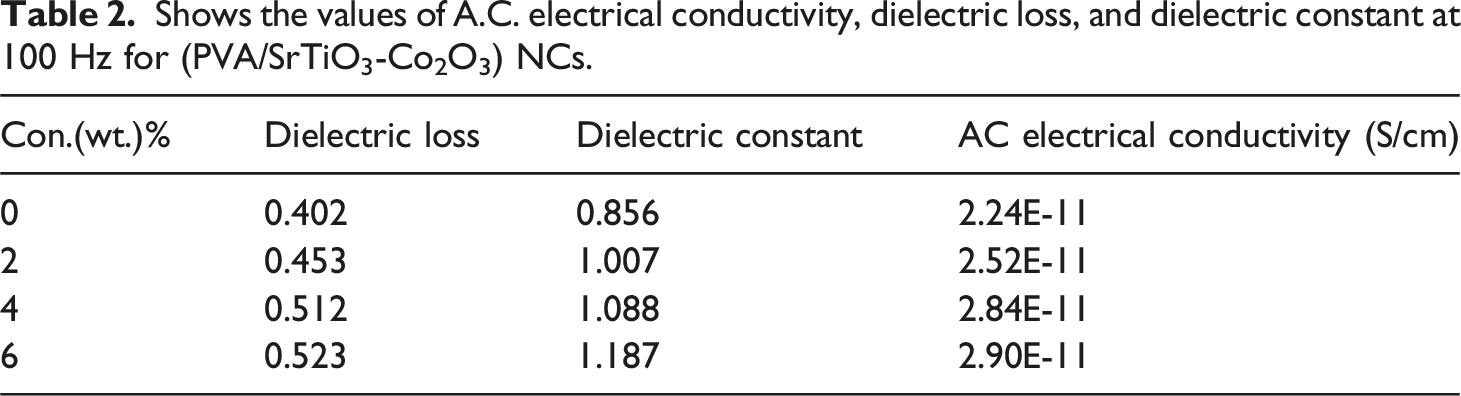
Shows the values of A.C. electrical conductivity, dielectric loss, and dielectric constant at 100 Hz for (PVA/SrTiO3-Co2O3) NCs.
Applications of (PVA/SrTiO3-Co2O3) nanocomposites
Antibacterial activity
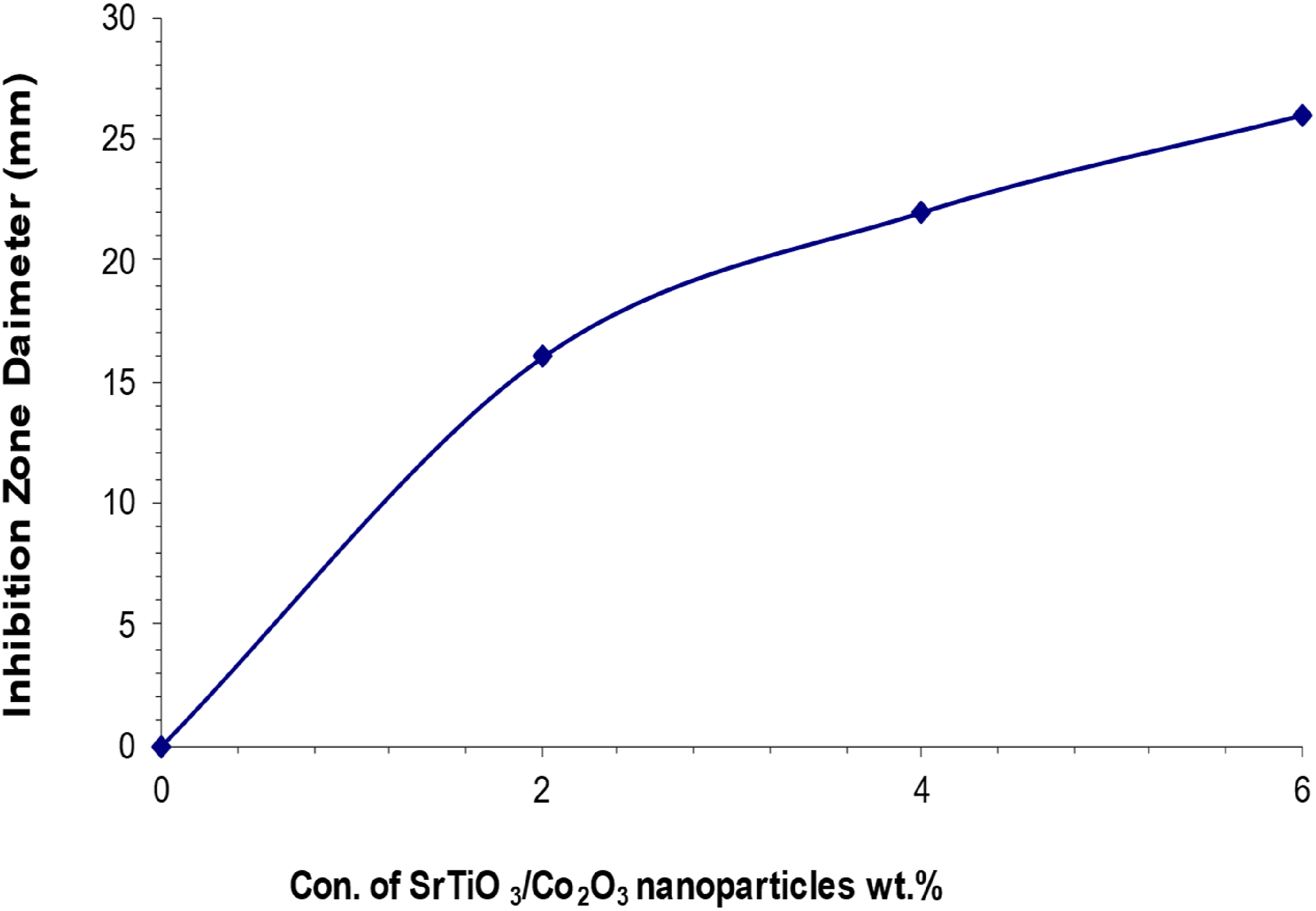
We used the agar diffusion technique to assess the inhibitory zone of nanocomposites and their antibacterial efficacy against two bacterial strains: gram-positive Staphylococcus aureus and gram-negative Escherichia coli. Figures 19 and 20 illustrate the relationship between the width of the inhibitory zone of the (PVA/SrTiO3-Co2O3) nanocomposite system and the concentrations of nanoparticles (NPs). If the concentration of (SrTiO3-Co2O3) inside the polymer matrix goes up, the inhibitory zone gets bigger. At a concentration of 6% SrTiO3-Co2O3 nanoparticles, the diameter of the inhibitory zone gets bigger. The maximal level of these phenomena for gram-positive bacteria (Staphylococcus aureus) is 34 mm, but for gram-negative bacteria (Escherichia coli), it is 26 mm. The results indicate that nanocomposite films inhibit the proliferation of Staphylococcus aureus and Escherichia coli. A lot of research shows that the antibacterial properties of nanocomposite films with nanoparticles are due to the production of reactive oxygen species (ROS) that cause oxidative stress.75,76 Reactive oxygen species (ROS) are things like superoxide (O-2), hydroxyl radicals (-OH), and hydrogen peroxide (H2O2) that happen when oxidative stress happens. These free radicals, particularly singlet oxygen (1O2), are associated with potential damage to bacterial cell DNA and proteins.
77
The strong antibacterial properties of SrTiO3-Co2O3 nanoparticles are what make PVA/SrTiO3-Co2O3 nanocomposites effective against Staphylococcus aureus and Escherichia coli.78,79 Table 3 shows the inhibition zone diameter of (PVA/SrTiO3-Co2O3) nanocomposites. Antibacterial effect of PVA as a function of SrTiO3-Co2O3 NPs concentrations on (Staphylococcus aureus). The antibacterial effect of PVA based on different amounts of (SrTiO3-Co2O3) on (Escherichia coli). Inhibition zone diameter of (PVA/SrTiO3-Co2O3) nanocomposites.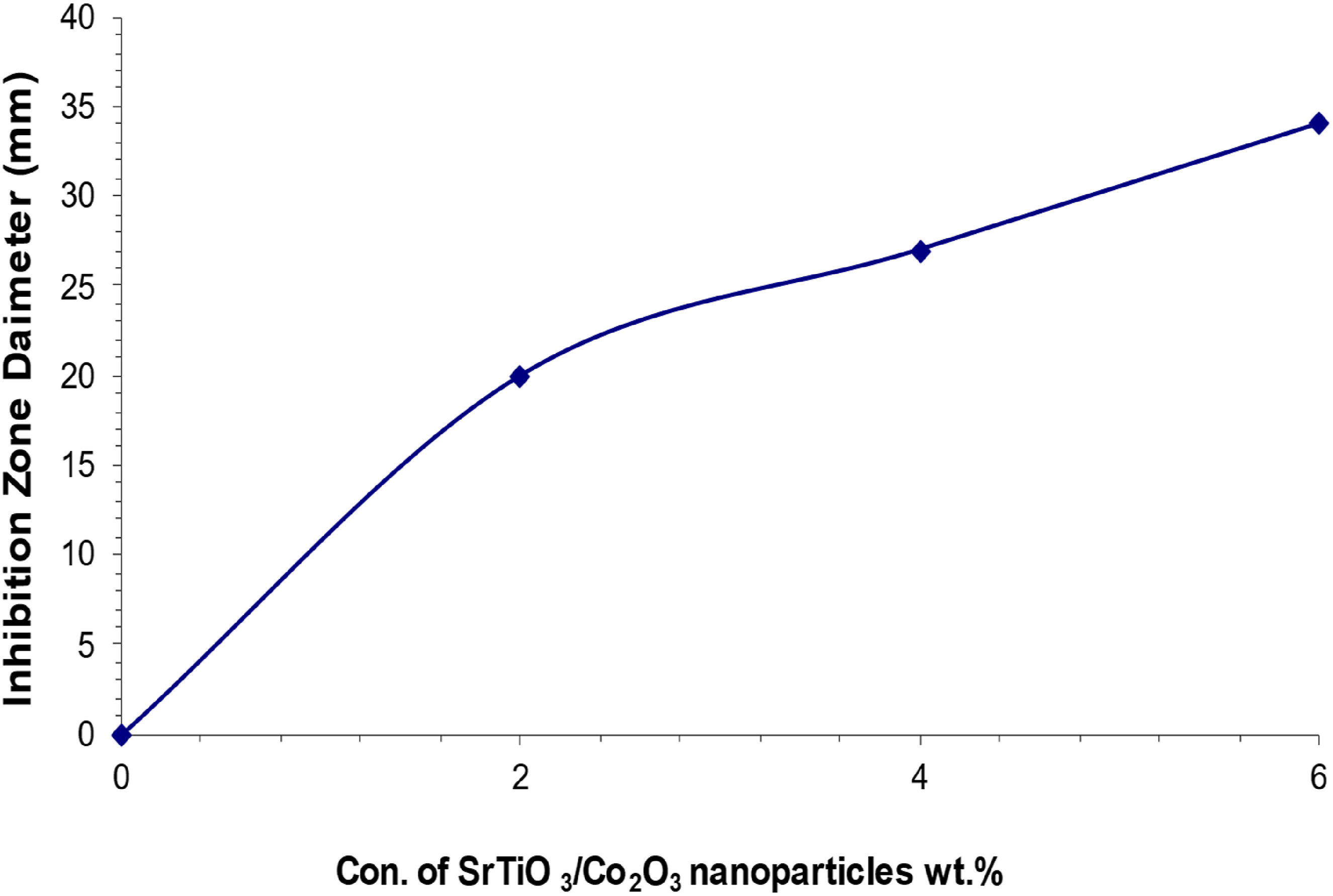

Pressure Sensors
Figure 21 shows that for varying concentrations of (SrTiO3-Co2O3) nanoparticles, the parallel capacitance of (PVA/SrTiO3-Co2O3) nanocomposites changes with pressure. In the graphic, we can see that there is a direct relationship between pressure and capacitance. One possible explanation for the correlation between applied pressure and parallel capacitance is the existence of crystalline regions inside the nanocomposite samples that possess an internal dipole moment. Without any external mechanical or electrical influence, the dipole moments show a stochastic orientation, leading to a zero-amplitude dipole moment.80,81 Stressing the specimens can alter their dipole moments, which in turn can produce an electric field in the proximal distribution. The formation of an electric field results in the accumulation of charges at the upper and lower surfaces of the sample.82,83 Difference of parallel capacitance for (PVA/SrTiO3-Co2O3) nanocomposites with pressure.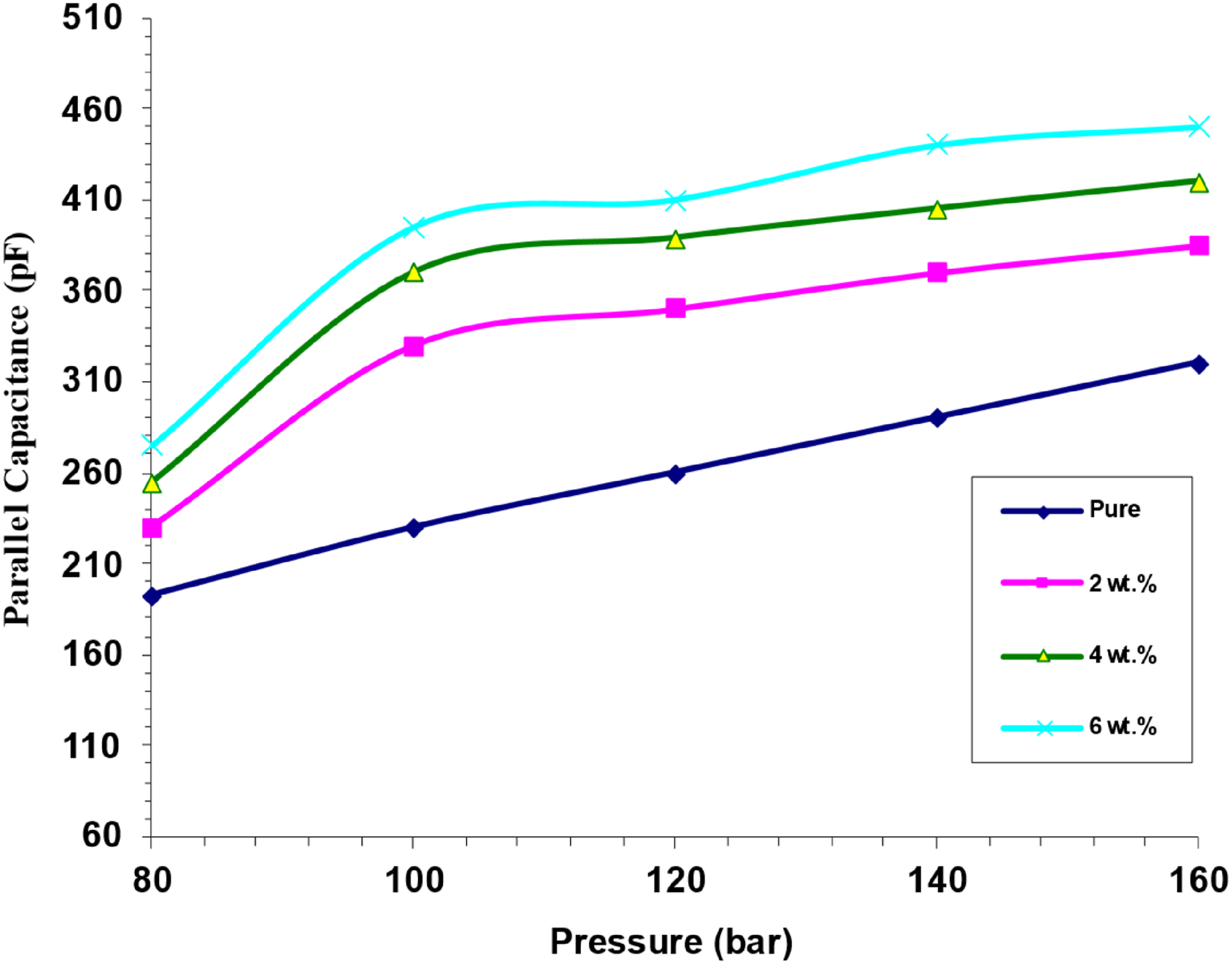
Figure 22 shows the connection between the electrical capacitance (Cp) of (PVA/SrTiO3-Co2O3) nanocomposites and the concentration of SrTiO3-Co2O3 nanoparticles at 80 bar. There is a positive association between the concentration of (SrTiO3-Co2O3) nanoparticles and the electrical capacitance of nanocomposites, as shown in the graph. The higher concentration of charge carriers within nanocomposites may account for the aforementioned occurrence.84–86 Effect of (SrTiO3-Co2O3) nanoparticle concentrations on parallel capacitance for (PVA/SrTiO3-Co2O3) nanocomposites.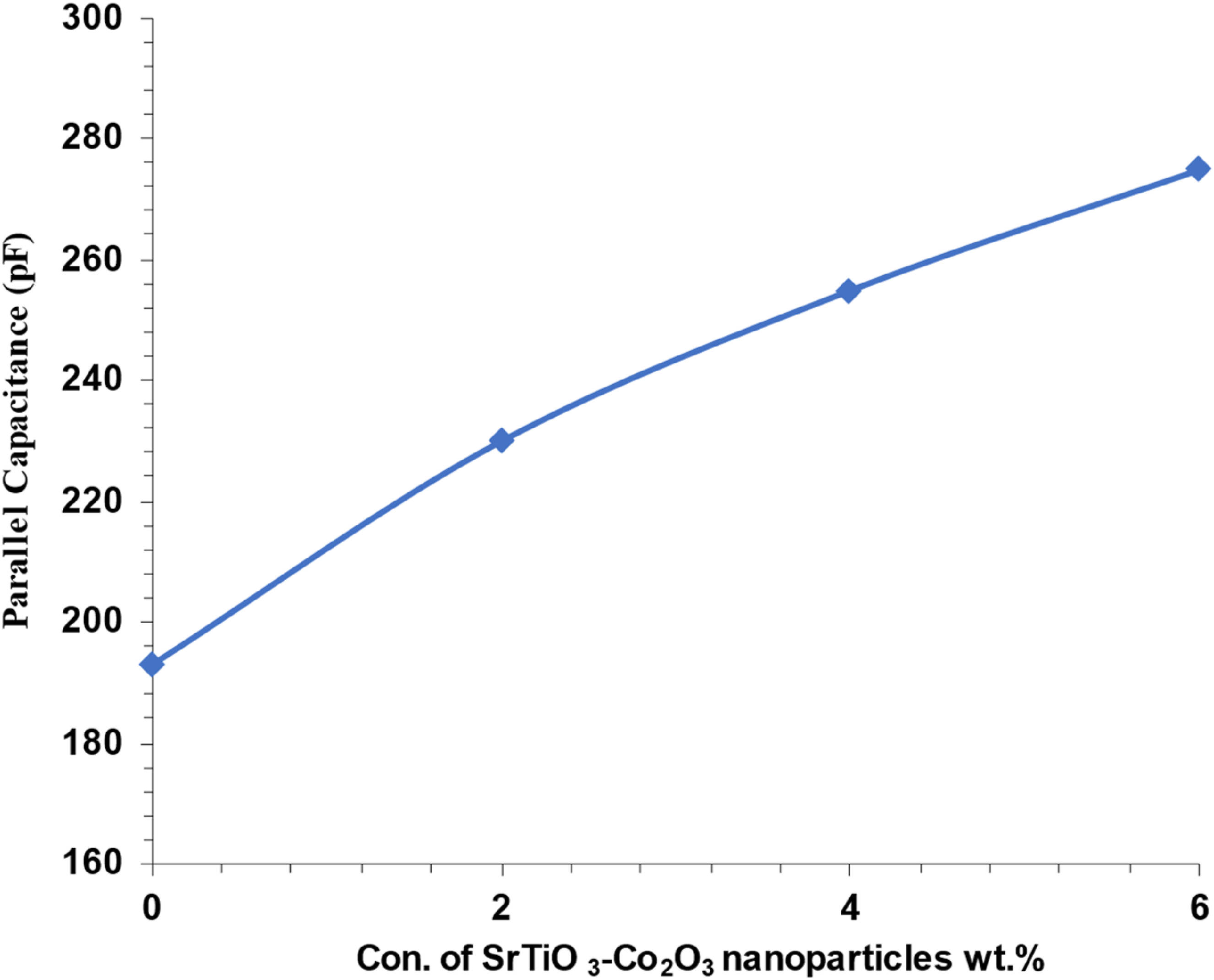
Nanostructures made of (PVA/SrTiO3-Co2O3) are very sensitive to the concentration of (SrTiO3-Co2O3) nanoparticles in the protective layer. The aforementioned processes constrain the applicability of pressure sensing, leading to several applications that require resistance to higher forces. As shown in Figure 23, the sensitivity of (PVA/SrTiO3-Co2O3) nanocomposites is influenced by (SrTiO3-Co2O3) nanoparticles. The graph clearly shows a positive link between the concentration of (SrTiO3-Co2O3) NPs and the sensitivity of nanocomposites. This is likely due to the internal dipole moment, as mentioned earlier.87,88 Table 4 displays the sensitivity values for (PVA/SrTiO3-Co2O3) NCs as a function of concentration of (SrTiO3-Co2O3) NPs. Influence of (SrTiO3-Co2O3) NPs concentrations on sensitivity for (PVA/SrTiO3-Co2O3) nanocomposites. Values of sensitivity with concentration for (PVA/SrTiO3-Co2O3) nanocomposites.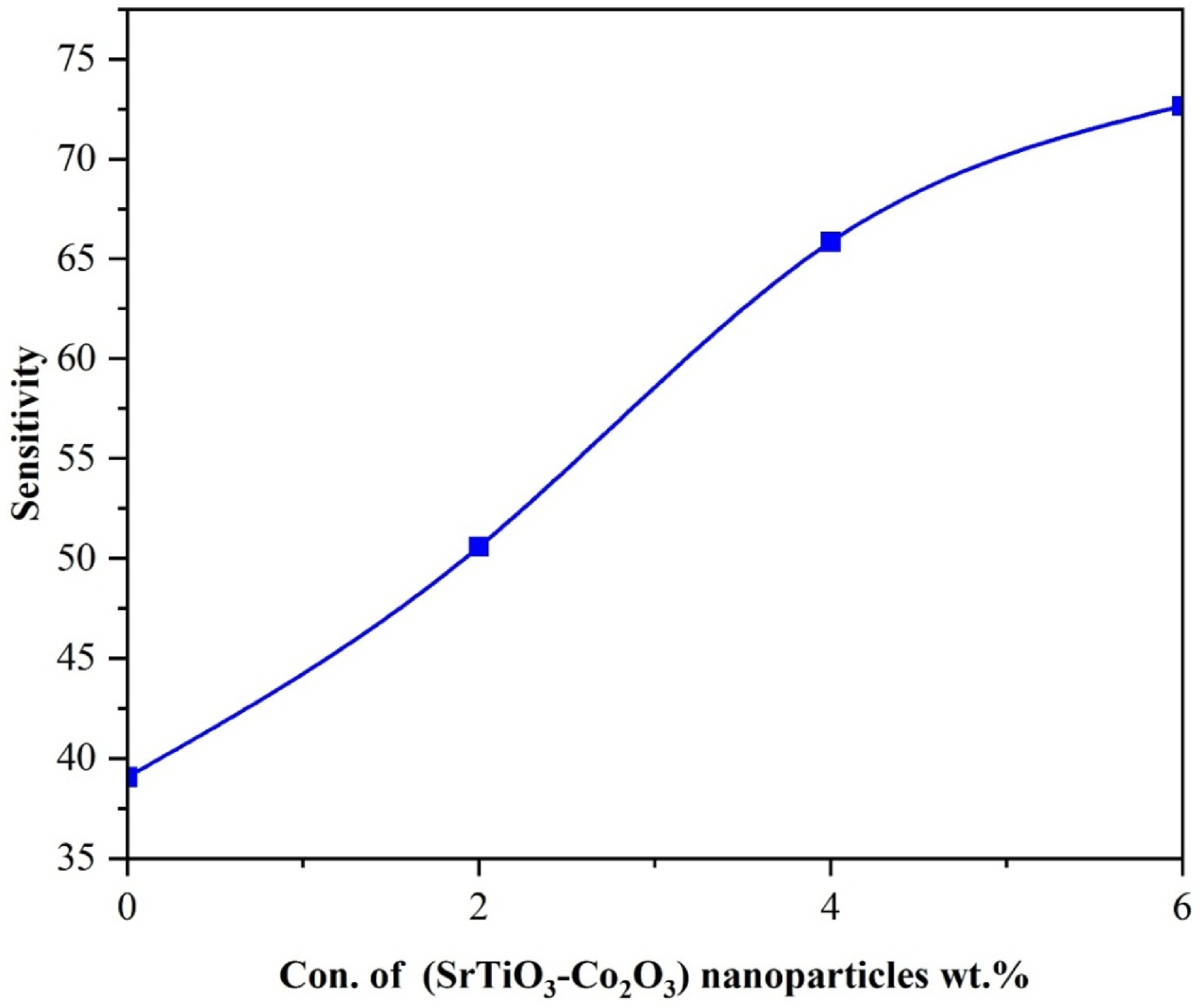

Conclusion
The films used in this experiment were made by using the solution casting process and included (PVA/SrTiO3-Co2O3) NCs. Optical microscopy (OM) images reveal a 6 wt% network of strontium titanate (SrTiO3) and cobalt oxide (Co2O3) nanoparticles embedded in the polymer. Some bands have been rearranged, and others have had their intensities adjusted, according to FTIR spectra. In addition, there is no chemical reaction between the PVA and the SrTiO3-Co2O3 nanoparticles. Research on optical properties has shown that (PVA/SrTiO3-Co2O3) NCs have better optical properties as the concentration of SrTiO3-Co2O3 NPs increases. These properties include a lower transmittance, better absorbance, index of refraction, coefficient of extinction, and dielectric constant (imaginary and real). With a rise in (SrTiO3-Co2O3) nanoparticle concentration, the optical energy gap for allowed indirect transitions drops from 4.1 eV to 3.4 eV and for forbidden indirect transitions from 3.1 eV to 2.3 eV. The optical characteristics of the (PVA/SrTiO3-Co2O3) NCs indicated that they might be useful in different electrical and photonic applications. This behaviour suggests that these materials could be great for use in nanoelectronics, since the dielectric constant, dielectric loss, and AC electrical conductivity all rise with increasing concentrations of SrTiO3-Co2O3 nanoparticles in PVA, from 0.856 to 1.187, 0.402 to 0.523, and 2.24 × 10−11 to 2.90 × 10−11, respectively. Nanocomposites have a higher A.C. electrical conductivity at higher frequencies but a lower dielectric constant and dielectric loss. In addition, the results show that the PVA/SrTiO3-Co2O3 nanocomposites are excellent materials for application in optoelectronic devices. Elevating the SrTiO3-Co2O3 NPs ratio in PVA to 6% resulted in an increase of 26 mm for Staphylococcus bacteria and 34 mm for Escherichia coli. Initial results from antibacterial testing indicate that these (PVA/SrTiO3-Co2O3) NCs spheres have remarkable antibacterial properties against both Staphylococcus and Escherichia coli. The findings demonstrate that PVA/SrTiO3-Co2O3 nanocomposite films are very suitable for usage as antimicrobial food packaging due to their favourable features. These films demonstrate a capacity to prolong the shelf life of packaged foods while maintaining their quality. These nanostructures may have potential as pressure sensors, as the results demonstrated that the dielectric properties of (PVA/SrTiO3-Co2O3) nanostructures improved with increasing pressure. At a concentration of 6 wt%, the nanocomposites demonstrate an exceptional sensitivity of 157.1%. Based on structural morphology and dielectric characteristic data, PVA/SrTiO3-Co2O3 nanostructures have many potential uses in pressure sensors and nanoelectronics.
Footnotes
Acknowledgements
Acknowledgements to University of Babylon.
Author Contributions
All authors contributed to the study’s conception and design. Material preparation, data collection and analysis were performed by, Majeed Ali Habeeb, Ali Hussein Abdel-Amir, Jassim M. AL-Issawe, Mamoun Fellah and Noureddine Elboughdiri. The first draft of the manuscript was written by Majeed Ali Habeeb and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical considerations
The research is not involving the studies on human or their data.
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.












