Abstract
This work presents the influence and improvement of the properties of linear and non-linear optical polystyrene (PS) nanocomposites by adding lanthanum hydroxide La(OH)3 NPs at different concentrations (0, 1, 2, and 3 wt%). The solution casting procedure was employed to fabricate the PS/La(OH)3 nanocomposites. XRD, FTIR, SEM, EDX, and Raman spectroscopy were used to analyze the structure of the prepared samples. The pure PS film exhibits an absorption edge of 3.29 eV, which gradually decreases to 2.80, 2.39, and 2.01 eV with increasing concentrations of La(OH)3. Additionally, the optical band gap of pure PS is 3.83 eV, respectively, decreased to 3.67, 3.55, and 3.39 eV with increasing La(OH)3 NPs loading from 1 to 3 wt%. Furthermore, the volume and surface energy loss functions (VELF and SELF) values increased with the increasing content of La(OH)3 NPs within the PS polymer matrix. Furthermore, the third-order nonlinear optical susceptibility χ (3) value increased from 1.03 × 10−14 for the pure PS film to 1.14 × 10−9 esu for PS/3% La(OH)3 nanocomposite film. Meanwhile, the nonlinear refractive index n 2 value enhanced from 2.68 × 10−13 esu for the pure PS film to 1.23 × 10−9 esu for the PS/3%La(OH)3 nanocomposite films, respectively.
Keywords
Introduction
Polymer composite materials have garnered significant interest due to their ability to integrate the beneficial characteristics of nanofillers and polymers, resulting in materials with enhanced functionalities.1,2 Polymer nanocomposites enriched with metal oxides are utilized for advanced applications, including optoelectronics, radiation shielding, sensors, and energy storage.3–11 Khasraw et al. reported the development of polyethylene oxide (PEO) nanocomposites doped with V2O5 nanoparticles, exhibiting improved structural, optical, and dielectric properties. 12 Similarly, Mamand et al. 13 investigated PEO nanocomposites incorporating Fe3O4 nanoparticles, achieving tailored optical and magnetic characteristics. Additionally, Dyari M. Mamand et al. 14 reported improved optical properties in chitosan polymer doped with orange peel dye. One of the remarkable polymers that garnered an enormous degree of scientific interest is polystyrene (PS). PS achieved particular attention attributable to its exceptional thermal and chemical stability, as well as its unique optical properties. PS possessed exceptional mechanical durability, low density, outstanding thermal resistance, superior electrical insulation, and simplicity of processing and molding, in addition to its exceptional optical transparency.15,16 All of these benefits render it suitable for a diverse range of industrial and technological applications, such as electronics, insulation, and packaging. 17 The integration of nanofillers has emerged as a crucial field of research to meet the increasing demand for novel materials with unique properties. Abdelghany et al. 18 reported that adding 5 wt% nanosilica reduced the direct optical band gap of PVC/PS (50/50 wt%) from 3.88 eV to 3.49 eV. Rahimli et al. 2 have studied the impact of ZnO nanoparticles (NPs) on the dielectric and thermal attributes of PS/ZnO nanocomposites. Abulyazied et al. 19 reported the radiation shielding, optical, and structural properties of PS/Dy2O3 composites.
Lanthanum compounds (lanthanum (III) hydroxide La(OH)3 and lanthanum (III) oxide La2O3) are of significant research interest due to their potential applications across various technological domains. 20 R.S. Yadav et al. 20 synthesized a Eu, Sm co-doped La(OH)3 nanocrystalline red-emitting phosphor via a combustion process. Leidinger et al. 21 produced (La(OH)3) hollow spheres by employing a water-in-oil microemulsion technique. Wang et al. 22 prepared La(OH)3 nanorods via a hydrothermal process and evaluated their photocatalytic performance through the degradation of Congo red dye. Khound et al. 23 have synthesized a La2O3/polyvinyl phenol thin film for transistor applications. Alhassan et al. 24 have outlined the linear/non-linear optical properties of PVC/La2O3 nanocomposites. La2O3 exhibits high hygroscopicity and readily absorbs water to produce La(OH)3, which is notably stable. 20
The solution casting method is simple and allows reasonable control over film thickness. However, its large-scale use is limited by high solvent consumption, long processing time, and the risk of nonuniform filler distribution due to gravity-driven sedimentation during drying. 25
This work presents the influence and improvement of the properties of linear and non-linear optical PS nanocomposites by the addition of La(OH)3 NPs at different fractions (0, 1, 2, and 3 wt%). The solution casting approach was employed to fabricate PS/La(OH)3 nanocomposites. The structural characteristics of the samples were evaluated using several characterization techniques, such as EDX. SEM, and XRD. The optical characteristics of the synthesized PS/La(OH)3 nanocomposites were also examined.
Materials and methods
Polystyrene and lanthanum hydroxide (La(OH)3) nanopowder were purchased from ADWIC Co. A polystyrene (PS) solution was prepared by dissolving 1 g of PS in 10 mL of toluene and stirring it for 30 min using a magnetic stirrer. La(OH)3 NPs were added to the PS solution with different concentration ratios (0, 1, 2, and 3 wt%) and stirred via a magnetic stirrer for 45 min. The PS, PS/1% La(OH)3, PS/2% La(OH)3, and PS/3% La(OH)3 nanocomposites were dried in air. The PS, PS/1% La(OH)3, PS/2% La(OH)3, and PS/3% La(OH)3 nanocomposites were renamed as PS, PS/La1, PS/La2, and PS/La3, respectively. The thickness of the PS/La(OH)3 nanocomposite films was measured to be 350 ± 20 µm. The structural and morphological characterization of PS and PS/La(OH)3 nanocomposite films was performed using various analytical techniques. The crystal structure was analyzed by X-ray diffraction (XRD) using a Shimadzu 6000 diffractometer. The functional groups were identified using Fourier Transform Infrared (FTIR) spectroscopy with a Nicolet iS10 spectrometer. Surface morphology and elemental composition were investigated by scanning electron microscopy (SEM) coupled with energy-dispersive X-ray analysis (EDX) using an Inspect S instrument (FEI, Holland). Optical properties of the samples were examined using a UV–Vis–NIR spectrophotometer (Jasco V-570).
Results and discussion
Structural analyses
The X-ray diffraction and FTIR spectra of La(OH)3 NPs are illustrated in Figure 1. The La(OH)3 NPs’ main crystal structure and purity degrees are revealed by the XRD patterns (Figure 1(a)). The pure hexagonal phase can be identified by the diffraction peaks observed at the (100), (110), (101), (201), (200), (211), and (112) reflection planes (JCPDS card No. 36-1481).
26
The high crystallinity of La(OH)3 NPs was verified by the stronger and identified diffraction peaks of the NPs. The nanostructures of the La(OH)3 NPs are suggested by the broader character of XRD peaks.
27
(a) X-ray diffraction pattern, (b) FTIR spectra of La(OH)3 NPs.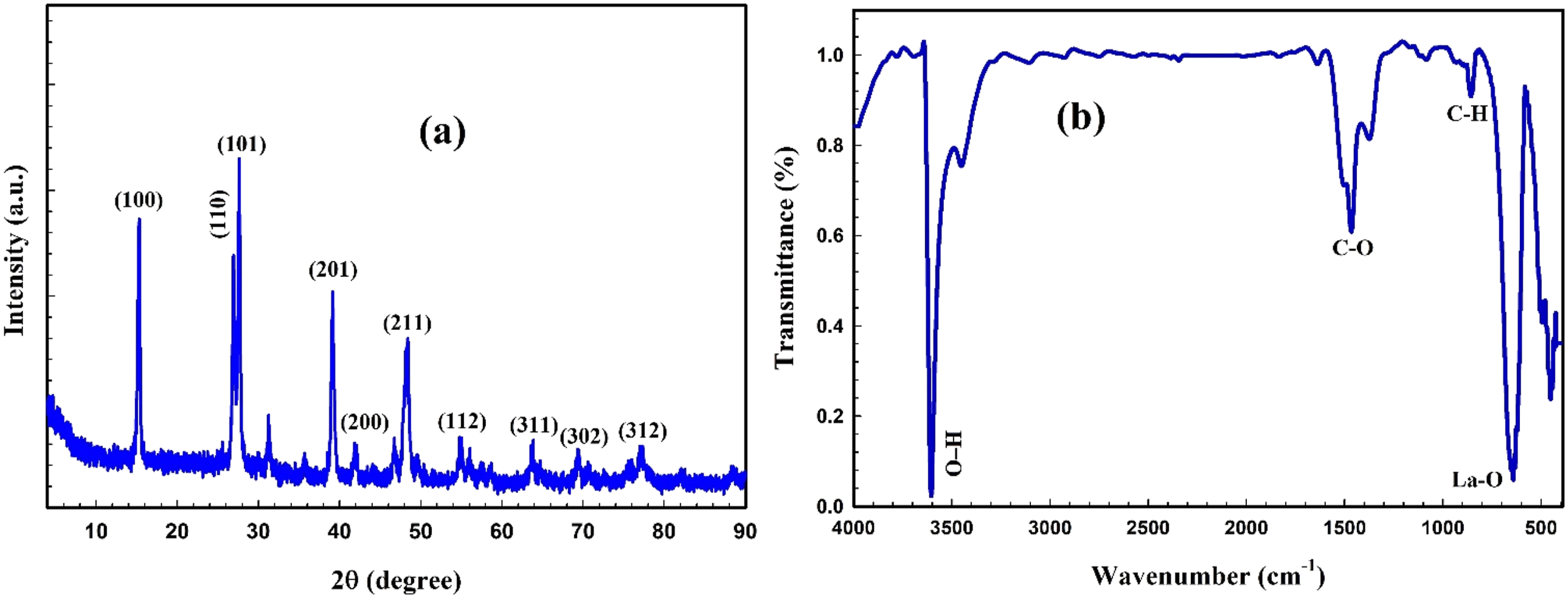
Scherrer’s formula was implemented to assess the crystallite size (D) of the La(OH)3 NPs
28
:
Figure 1(b) illustrates FTIR spectra of La(OH)3 NPs in the wavenumber range of 3500–400 cm−1. The detection of O–H stretching vibrations resulting from moisture absorption on the surface of the samples has been verified by a wide band observed at 3604 cm−1. The well-defined band appearing at 857 cm−1 is related to C–H bending vibrations, while the peak at 1465 cm−1 was created by the asymmetric stretching of C-O−functional groups. The broadband at 641 cm−1 is attributed to La–O stretching vibrations, whereas the narrow band at 494 cm−1 results from La–O bending vibrations. 27
The SEM image of La(OH)3 NPs is illustrated in Figure 2(a). The figure revealed that the La(OH)3 NPs have plate-like and rod-shaped particles with an aggregated distribution. Figure 2(b) shows EDX spectra of La(OH)3 NPs. EDX spectra revealed the elemental composition (La and O elements) and purity of La(OH)3 NPs. (a) SEM image and (b) EDX spectra of La(OH)3 NPs.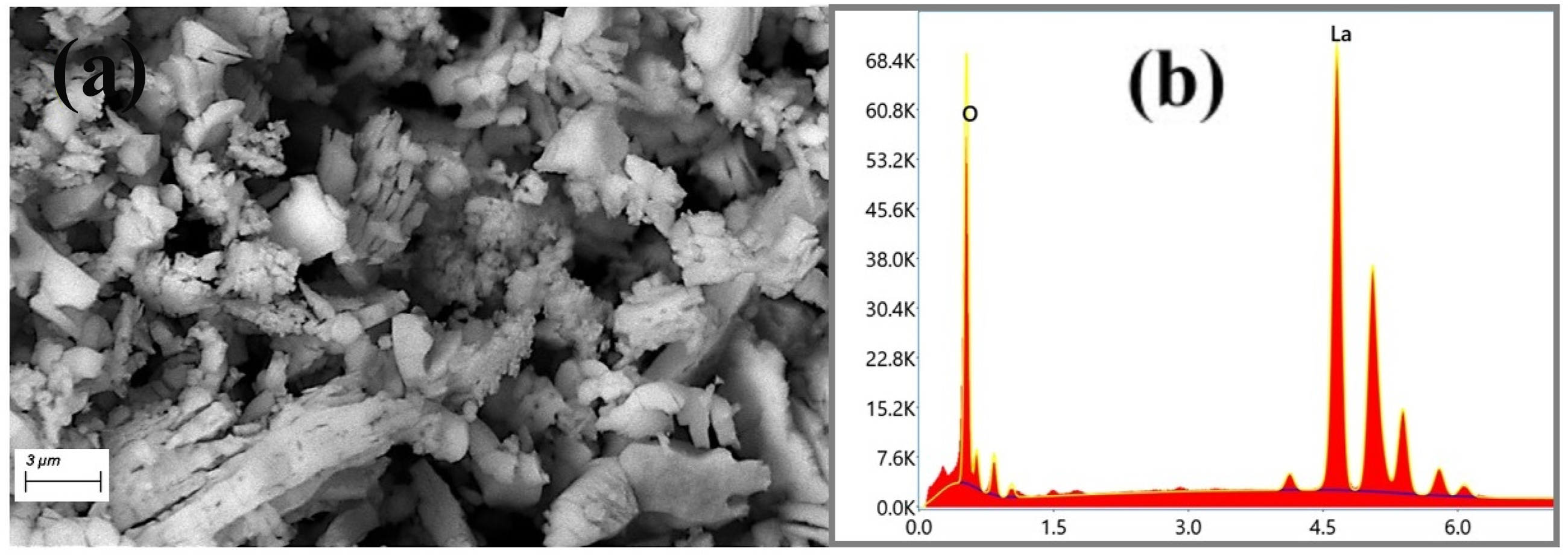
Additionally, Figure 3(a) illustrates the XRD patterns of polystyrene (PS) and PS-based nanocomposites as the concentration of La(OH)3 increases to 1%, 2%, and 3%. The amorphous structure of pure PS is characterized by a broad peak at approximately 20°. As the content of La(OH)3 rises, distinct peaks that correspond to La(OH)3 are observed. As the concentration of La(OH)3 rises, these peaks become more distinct, suggesting that the La(OH)3 NPs are effectively incorporated and have an improved crystallinity profile. The FTIR spectra of PS and PS/La(OH)3 nanocomposite films are illustrated in Figure 3(b). FTIR spectrum of the pure PS film revealed characteristic absorption peaks at 3025 cm−1 and 2919 cm−1, which are attributed to the aromatic C-H and aliphatic C-H stretching vibrations, respectively. The bands identified at 1490 cm−1 were characterized as resulting from C = C bending vibrations. The vibrational phase of CH2 bending is located at 1448 cm−1.29,30 (a) X-ray diffraction patterns and (b) FTIR spectra for the pristine PS and La(OH)3-mixed PS films.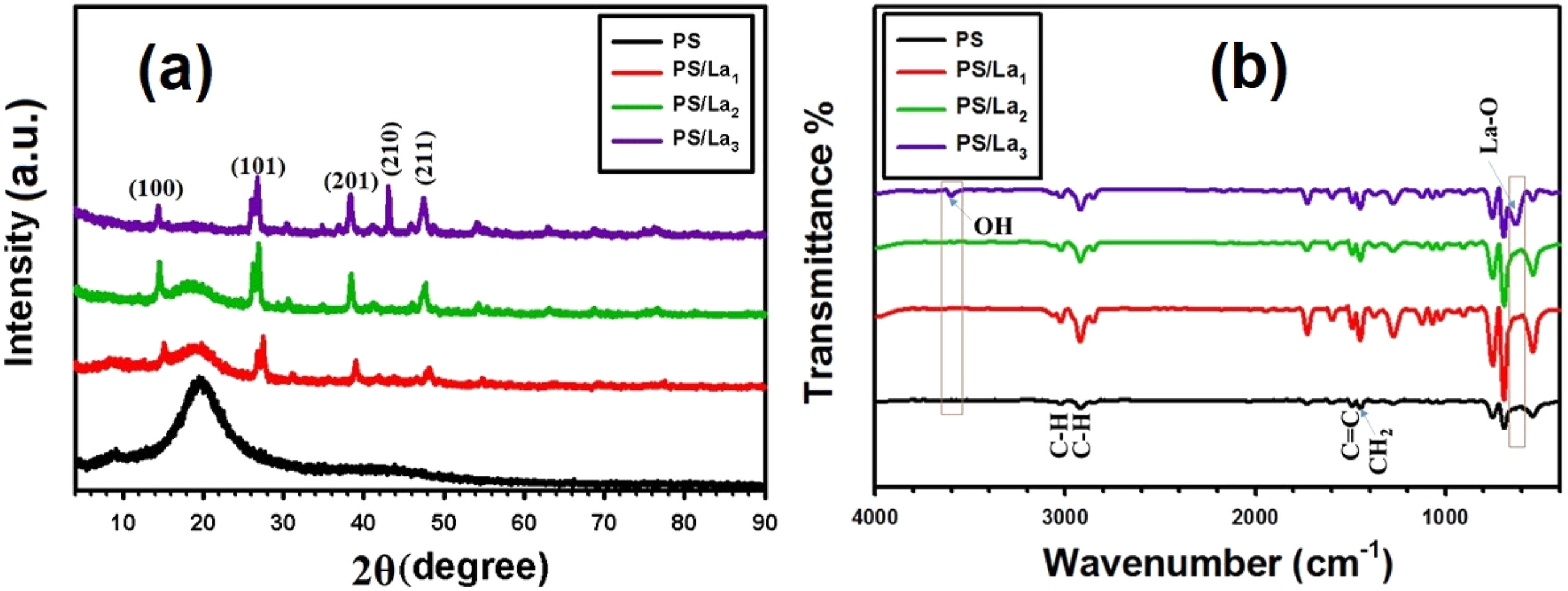
Impressively, by the addition of a high percentage 3% of La(OH)3 NPs into the PS, new characteristic bands appeared at a wavenumber of about 3604 and 641 cm−1 owing to O–H group and La–O stretching vibrations of the La(OH)3 NPs. Moreover, the inclusion of La(OH)3 NPs into the PS matrix also led to an increase in the intensity of the bands for all nanocomposite films, suggesting an interaction between the polymeric chain and La(OH)3 NPs. Possible physical and chemical interactions may occur between La(OH)3 and the PS matrix. La(OH)3 nanoparticles can interact with PS chains through weak van der Waals forces, and hydrogen bonding may also form between surface oxygen atoms of La(OH)3 NPs and hydrogen atoms of the PS chains.31–35 These findings confirm the good preparation of PS/La(OH)3 nanocomposite films.
Raman analysis is a practical and efficient method to ascribe the structure of nanocomposite films. Figure 4 shows the Raman spectra of PS/La1 and PS/L3 films. It can be seen that the spectra present several Raman modes intrinsic to PS, including peaks at 609, 998, 1028, and 1595 cm−1, which correspond to ring deformation, aromatic groups, C-H bending, and C = C aromatic ring stretching, respectively. Moreover, the peaks at 794, 2900, and 3053 cm−1 are related to a weaker single bond C-C, C-H stretching of aromatic, and C-H stretching of aliphatic carbon. Meanwhile, these spectra also present small peaks, which are more pronounced in the case of the PS/L3 film at 208, 354, and 447 cm−1, assigned to La–O bending vibration, La–O stretching vibration, and Egν1 mode, respectively. These results provide strong evidence of the successful loading of La(OH)3 on the PS.36–39 The Raman spectra of PS/La1 and PS/L3 films.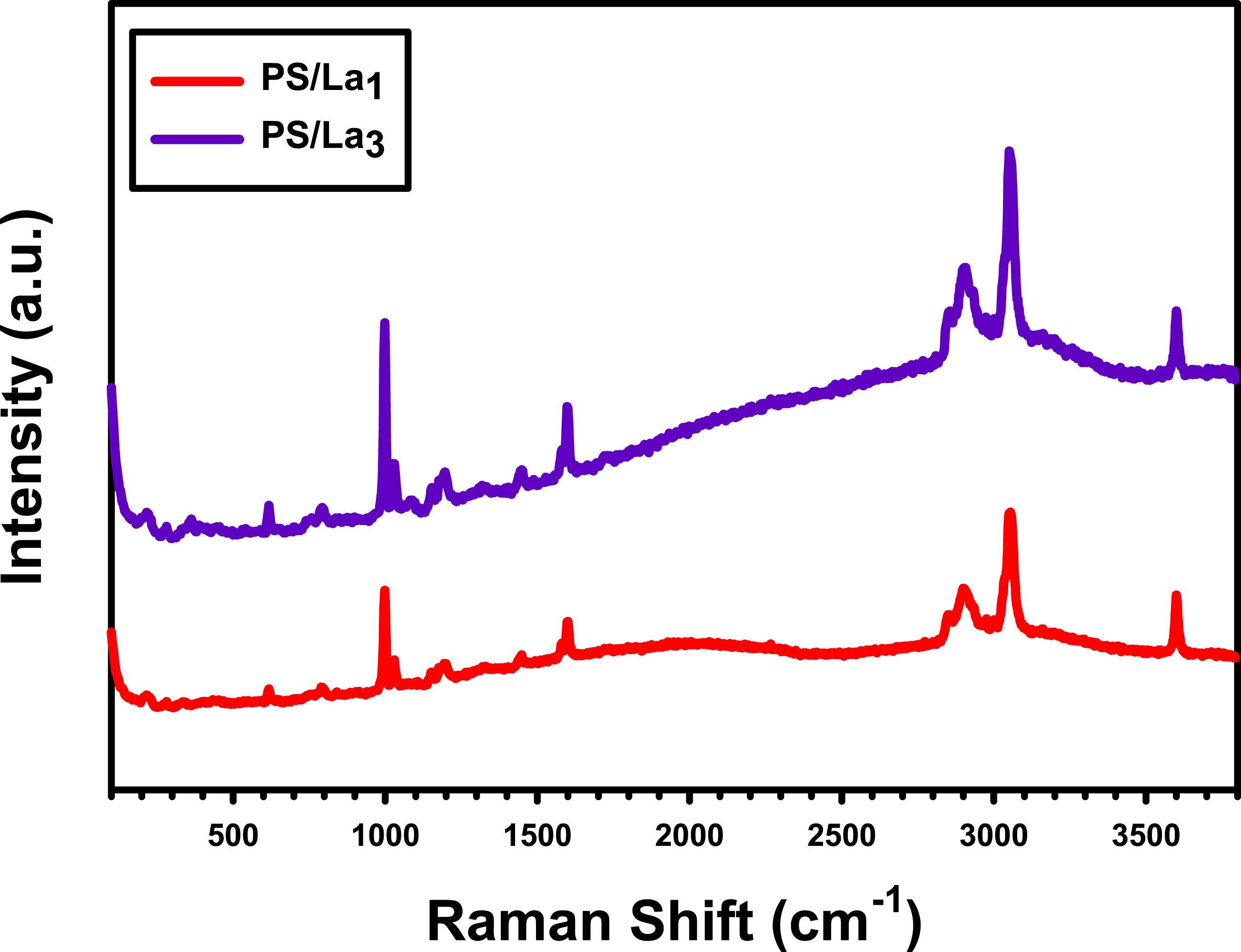
Optical properties
Studying the optical properties, such as absorption, absorption coefficient, energy gap, reflectance, and refractive index, provides insight into the electronic configuration and energy levels of the polymers, which are crucial for understanding their functionality in various applications. The absorbance (A) and reflectance (R) are measured using UV-vis spectroscopy. Figure 5(a) displays optical absorbance spectra for pure PS and La(OH)3-loaded PS films. All spectra present a strong absorption region in the UV region between 190 and 350 nm, which may be owing to the sufficient photon energy for interacting with atoms.
40
It can be seen that the pristine PS spectrum exhibits an absorption band at 250 nm, which is attributed to the π→π* electronic transitions of the PS matrix.41,42 This figure also shows that increasing the amount of La(OH)3 NPs in the nanocomposite films enhances the absorbance, which is attributed to the nanoparticles aggregating, thereby increasing the number of charge carriers.
41
The enhanced absorbance is beneficial for applications where strong light absorption is essential, such as in optical devices.
43
The dependence of reflectance spectra (R) on wavelength (λ) for both pure and doped nanocomposite films is displayed in Figure 5(b). As observed, the reflectance spectra for all films decrease with increasing wavelength, reaching a constant value at higher wavelengths. Moreover, the reflectance values gradually increased as the La(OH)3 NPs content improved in the PS matrix. Such an increase may stem from enhanced light scattering resulting from the formation of molecular bonds between the polymer and the incorporated filler.
24
(a) The absorbance (A) versus wavelength (λ) and (b) the reflectance (R) versus wavelength (λ) for the pristine PS and La(OH)3 -mixed PS films.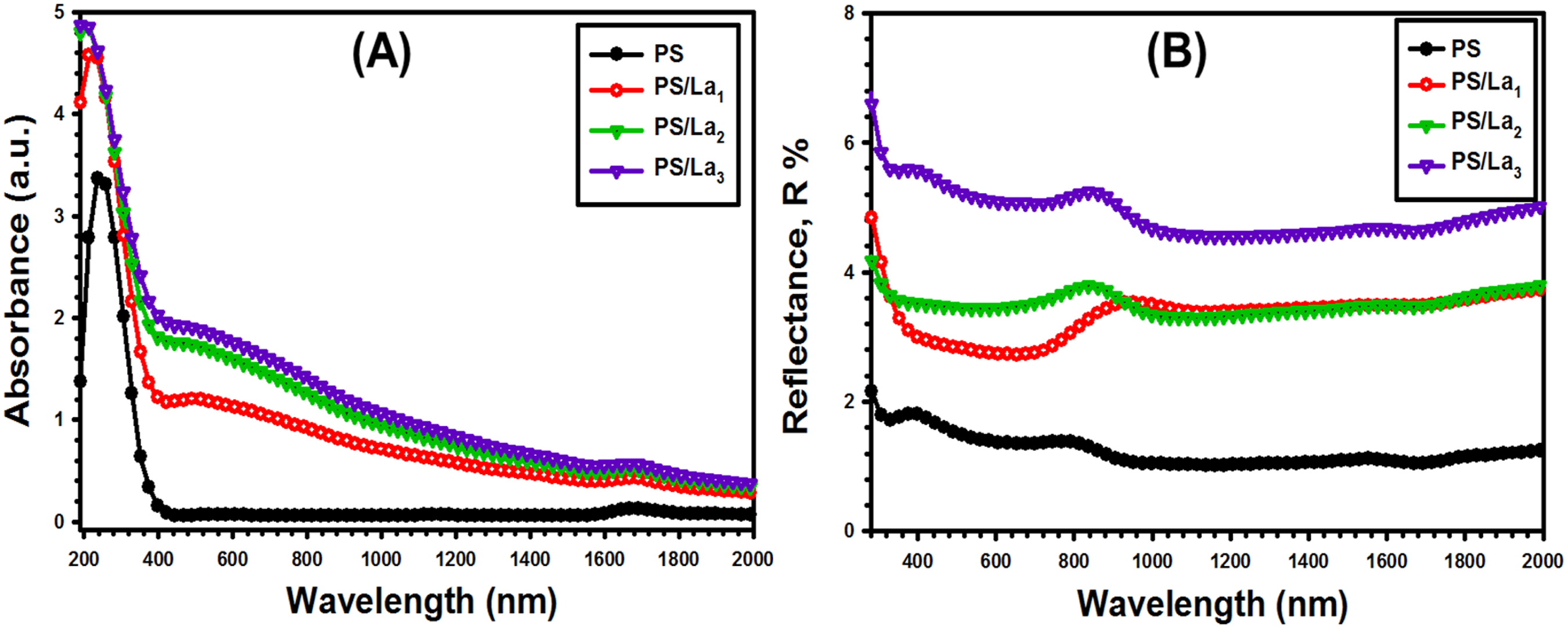
The linear absorption coefficient (α) of pure PS and PS-doped films by La(OH)3 can be given from the absorbance values and thickness of films (d) according to the Beer-Lambert expression
44
:
Figure 6(b) reveals changes in linear absorption coefficient (α) with photon energy (hν) for the prepared films. There is a gradual improvement in the absorption coefficient after incorporation of the La(OH)3 NPs with the polymer matrix. This improvement in the absorption coefficient is attributed to the formation of molecular complexes between the La(OH)3 NPs and the PS polymer, which creates new energy levels and enhances photon absorption.
45
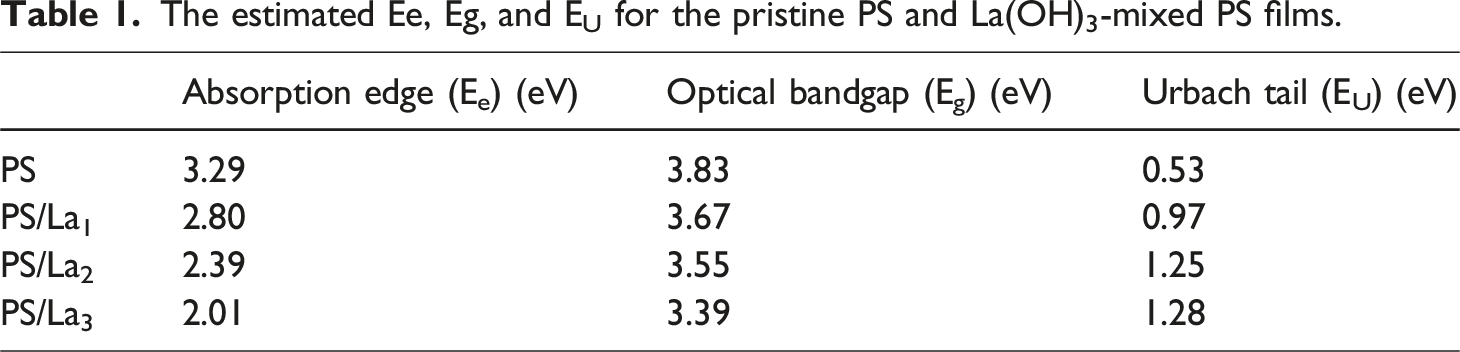
Meanwhile, the absorption edge values can be extracted from the pure PS and PS-doped films from the extrapolation of the linear parts of the curves to a point α (cm – 1) = 0 and were listed in Table 1. According to the table, the pure PS film exhibits an absorption edge of 3.29 eV, which gradually decreases to 2.80, 2.39, and 2.01 eV for the PS/La1, PS/La2, and PS/La3 samples, respectively. This result confirms a red shift of the absorption edge after inserting different amounts of the nanofiller, indicating interactions between the nanoparticles and the polymer matrix and reducing the optical band gap. (a) α Vs. hν (b) (αhν)2 versus hν for the pristine PS and La(OH)3-mixed PS films. The estimated Ee, Eg, and EU for the pristine PS and La(OH)3-mixed PS films.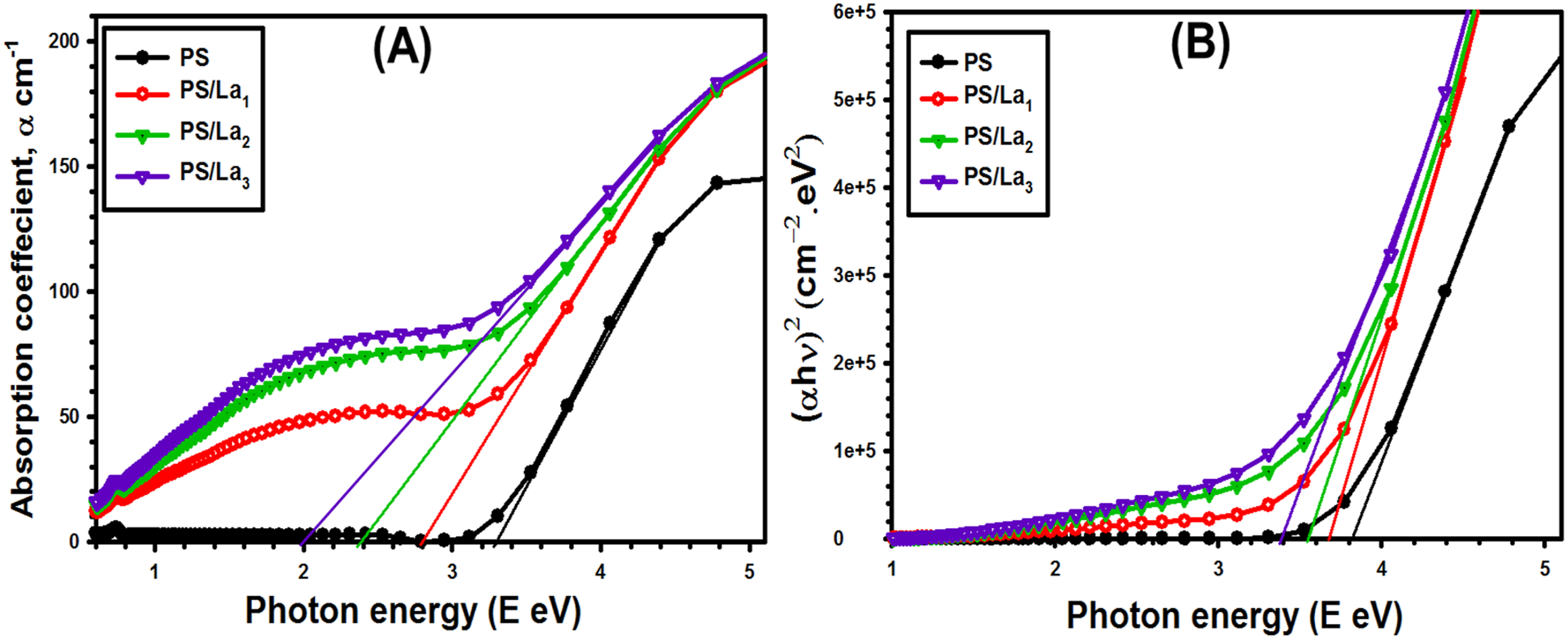
According to Mott and Davis, the optical band gap energy (Eg) values of the pure PS and PS-loaded films were computed by considering the following dependence of α on the photon energy (hν)
46
:
The parameter x, known as the band tailing factor, is assigned a value of 1/2 for direct allowed transitions and 2 for indirect allowed transitions. Figure 6(b) presents the Tauc relation between (αhν)2 versus hν for the pristine PS and three nanocomposite films of PS/La(OH)3 with various weight amounts of La(OH)3. Using this figure and equation (3), the allowed direct optical band gap can be found by extrapolating the linear portion of the obtained curves to the x-axis, where α = 0. The values of the band gap were summarized in Table 1. The optical band gap of pure PS is 3.83 eV, respectively, decreased to 3.67, 3.55, and 3.39 eV with increasing La(OH)3 NPs loading from 1 to 3 wt%. Specific defects in the films induced this reduction due to the insertion of La(OH)3 nanoparticles. 45 This decrease indicates that the additional La(OH)3 NPs enhance the nanocomposite films’ ability to conduct electricity by creating defects that facilitate charge carrier generation across the nanocomposite’s band gap.
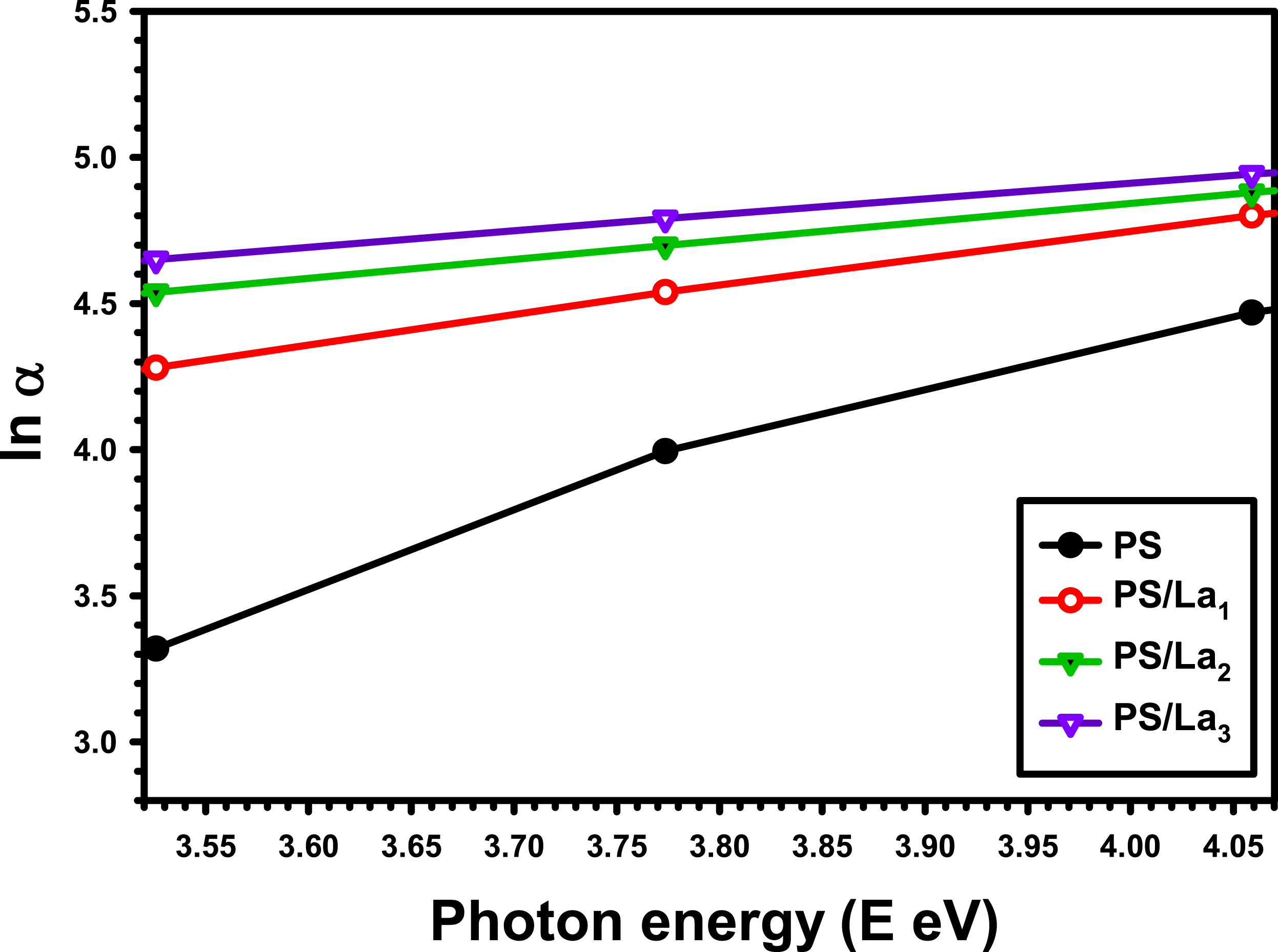
The Urbach energy (EU), a measure of band tail width, can be calculated from the absorption coefficient spectra based on the following formula.
47
Ln α versus h 
Based on the wavelength (λ) and absorption coefficient (α), the extinction coefficient (k) can be evaluated using the relation below
49
:
The variation of k as a function of λ for the pure PS and La(OH)3-loaded PS films is disclosed in Figure 8(a). As shown, incorporating La(OH)3 NPs with an increased ratio gradually increases k, which is explained by the increase in charge carriers and defect states.
50
The refractive index (n) is a fundamental optical property, indicating the extent to which light slows down in a given medium. The refractive index of the pure and nanocomposite films can be extracted from the Fresnel’s relationship
51
: (a) The extinction coefficient (k) versus wavelength (λ) and (b) the refractive index (n) versus wavelength (λ) for the pristine PS and La(OH)3-mixed PS films.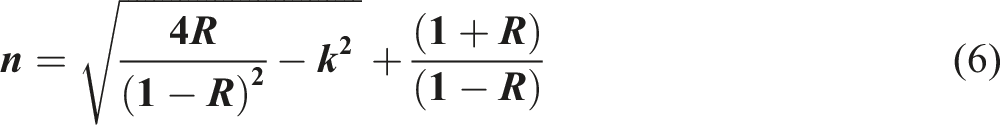
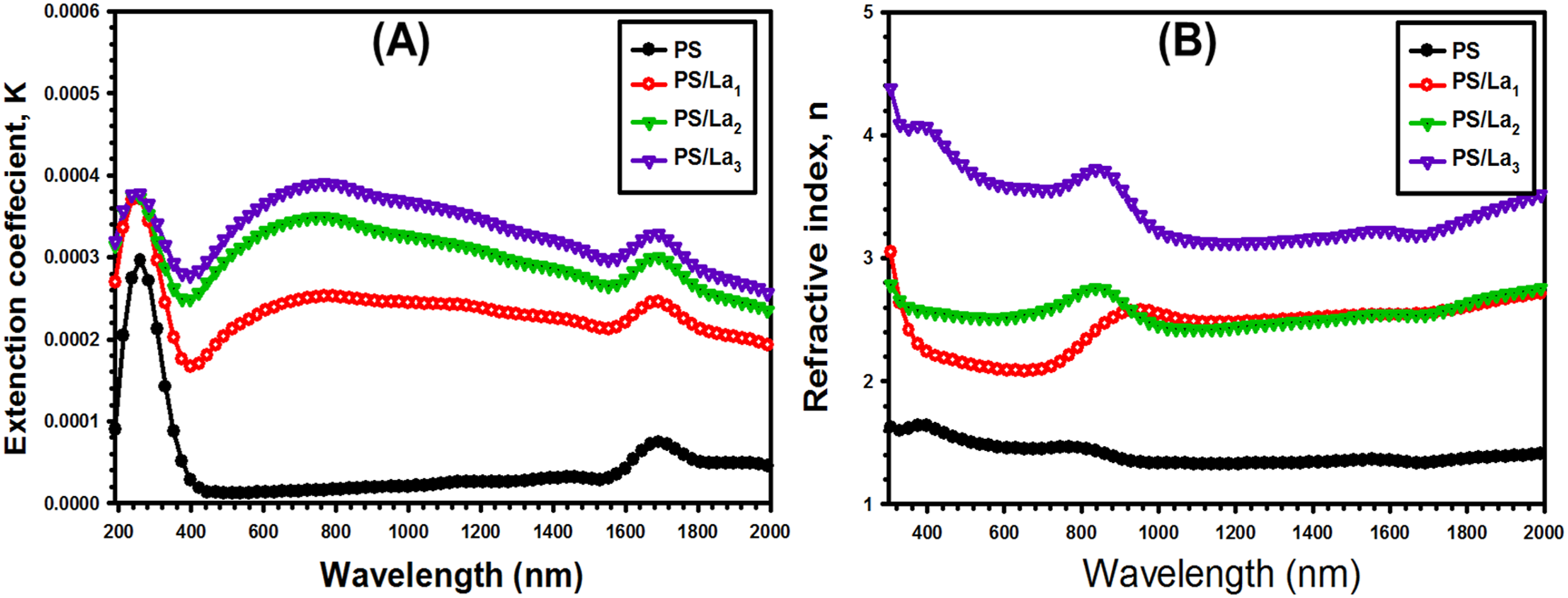
Figure 8(b) depicts the refractive index alteration against the wavelength of pure PS and La(OH)3-doped PS films. The refractive index of virgin PS and PS/La(OH)3 films declined substantially with increasing wavelengths. Further, as the concentration of La(OH)3 NPs increases, a higher refractive index is observed, due to the higher dipole and density arising from the cross-linked structure and the presence of La(OH)3 in the PS matrix. 52 Meanwhile, incorporating the La(OH)3 NPs into the PS matrix may lead to the formation of localized states in the band gap area. 53 There is a growing need for optical materials with a wide range of refractive indices in various applications, such as filters, optical adhesives, antireflection coatings, and relatively high-reflecting surfaces. 54
Moreover, dielectric properties demonstrate the materials’ response when interacting with an electric field, resulting in polarization. The real dielectric part (εr) and the imaginary part (εi) can be deduced from the k and n values by the following formulas
55
:

Figure 9 shows both the real and imaginary components of the dielectric constant for pure PS and La(OH)3-loaded PS films. It is noted that the real dielectric constant values are higher than the imaginary dielectric constant. As the La(OH)3 NPs concentration is increased, both the real and imaginary dielectric constant gradually increase. The increase in dielectric constant can be attributed to the enhanced density of states in the band gap of La(OH)3/PS films, resulting in greater polarization.
56
(a) The real dielectric (ε
r
) versus wavelength (λ) and (b) the imaginary dielectric (ε
i
) versus wavelength (λ) for the pristine PS and La(OH)3-mixed PS films.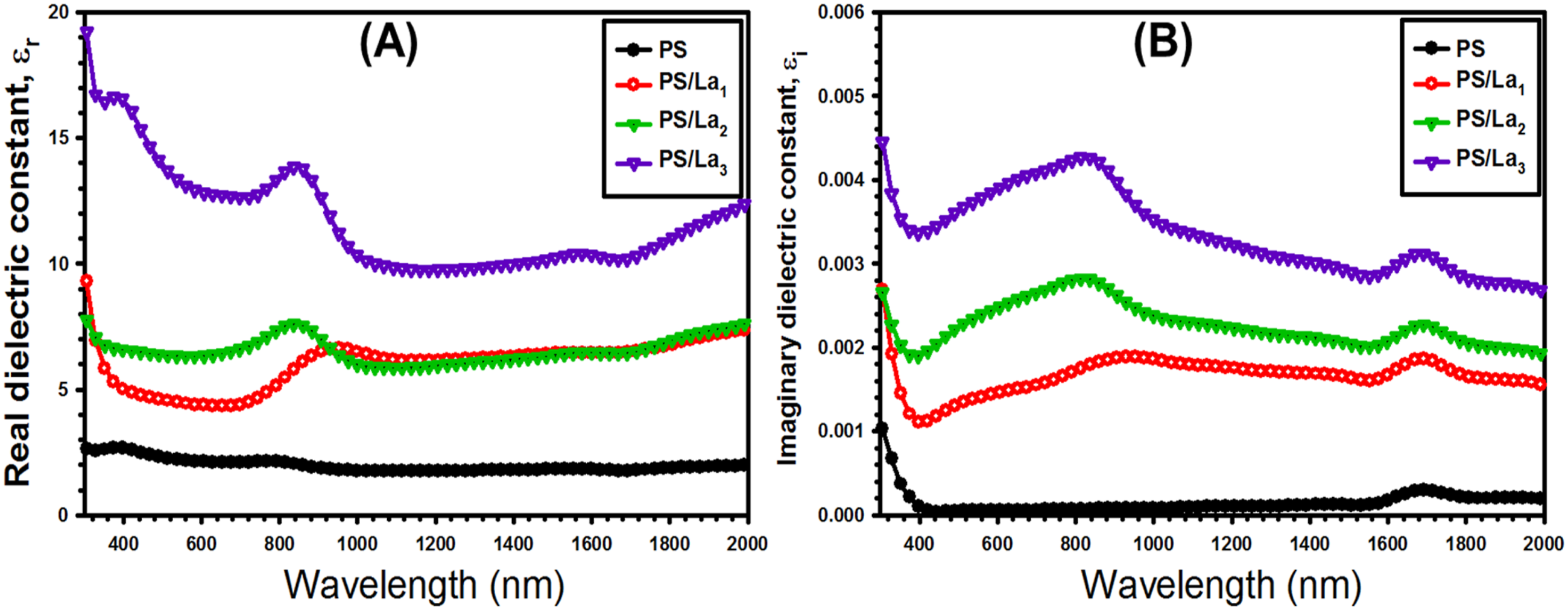
Fast electron energy loss within a material and at its surface can be quantified by the volume and surface energy loss functions (VELF and SELF), respectively. In which these two functions can be deduced using values of εr and εi by the following equations
57
:

Figure 10(a) and (b) shows the variation in the VELF and the SELF for the pure PS and filled PS by La(OH)3 NPs. It can be seen that both VELF and SELF have similar behaviors. Furthermore, VELF and SELF exhibited growth with an increase in the amount of La(OH)3 NPs within the PS polymer matrix, which may be due to improvements in the electronic structure and polarization processes of the polymer molecules under the applied electromagnetic field.
58
The enhancement of VELF and SELF renders these PS/La(OH)3 films suitable for electro-optical uses. (a) VELF versus hν and (b) SELF versus hν for the pristine PS and La(OH)3-mixed PS films.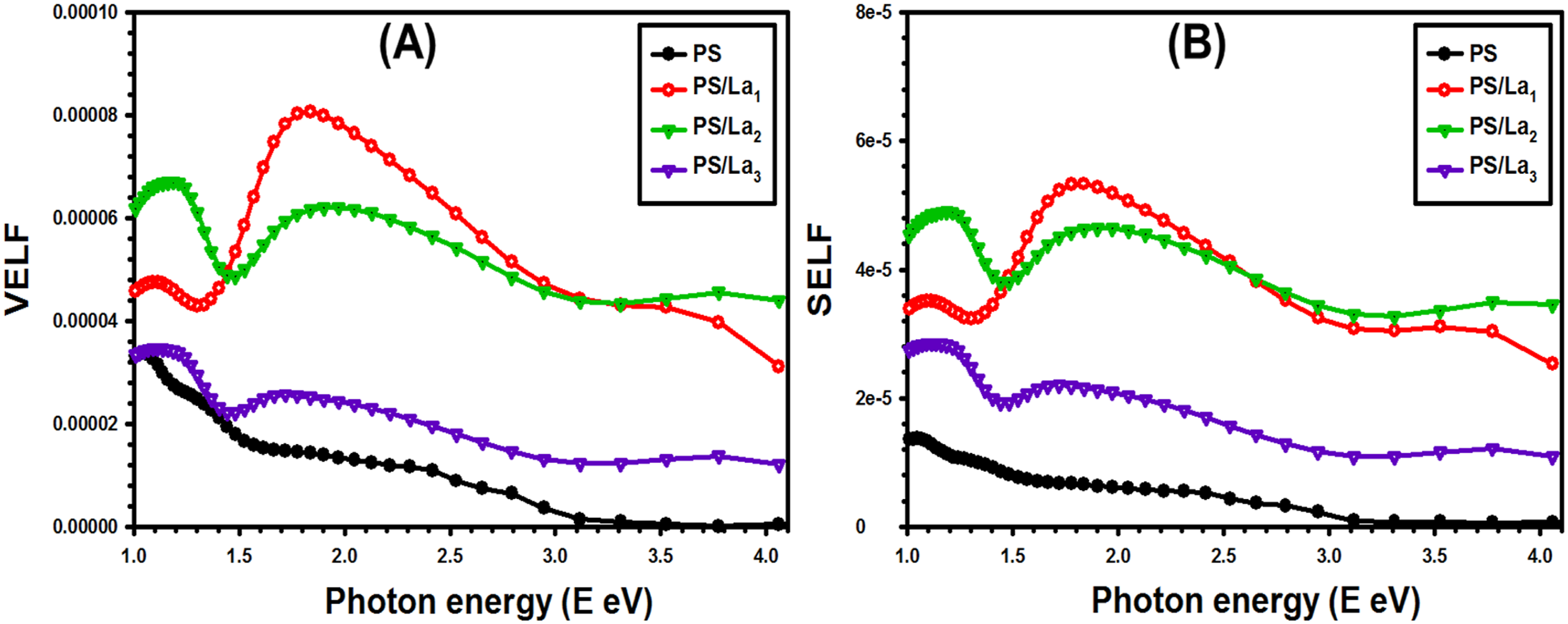
The Wemple–DiDomenico single-oscillator model was applied to describe the wavelength-dependent behavior of the refractive index within the normal dispersion region
59
:
E
d
. refers to the dispersion energy (average strength of interband optical transitions), and E
o
denotes oscillator energy. These two parameters can be estimated from the slope and intercept of the linear part of the curves that appeared in the plot between (n
2
−1)
−1
and (hν)
2
(Figure 11). The numerical values of E
d
. and E
o
for the pure and filled films are listed in Table 2. The E
o
values are approximately equal to 1.05, 1.37, 1.92, and 1.66 times the direct band gap for the pure PS, PS/La1, PS/La2, and PS/La3 nanocomposite films, respectively. Furthermore, the optical transition strength increases as the Ed. rises, particularly with a higher percentage of La(OH)3 NPs in the PS matrix. This can be explained by the fact that a charge transfer complex develops between the La(OH)3NPs and the PS matrix.
60
In addition, the other two factors, including the static refractive index (n
o
) and the oscillation strengths (f) for the pure PS and La(OH)3-filled PS films, can be estimated using the following relations61,62: (n
2
-1)
−1
versus (hν)
2
for the pristine PS and La(OH)3-mixed PS films. The optical parameters and χ
(1)
, χ
(3)
, and n
2
for the pristine PS and La(OH)3-mixed PS films.

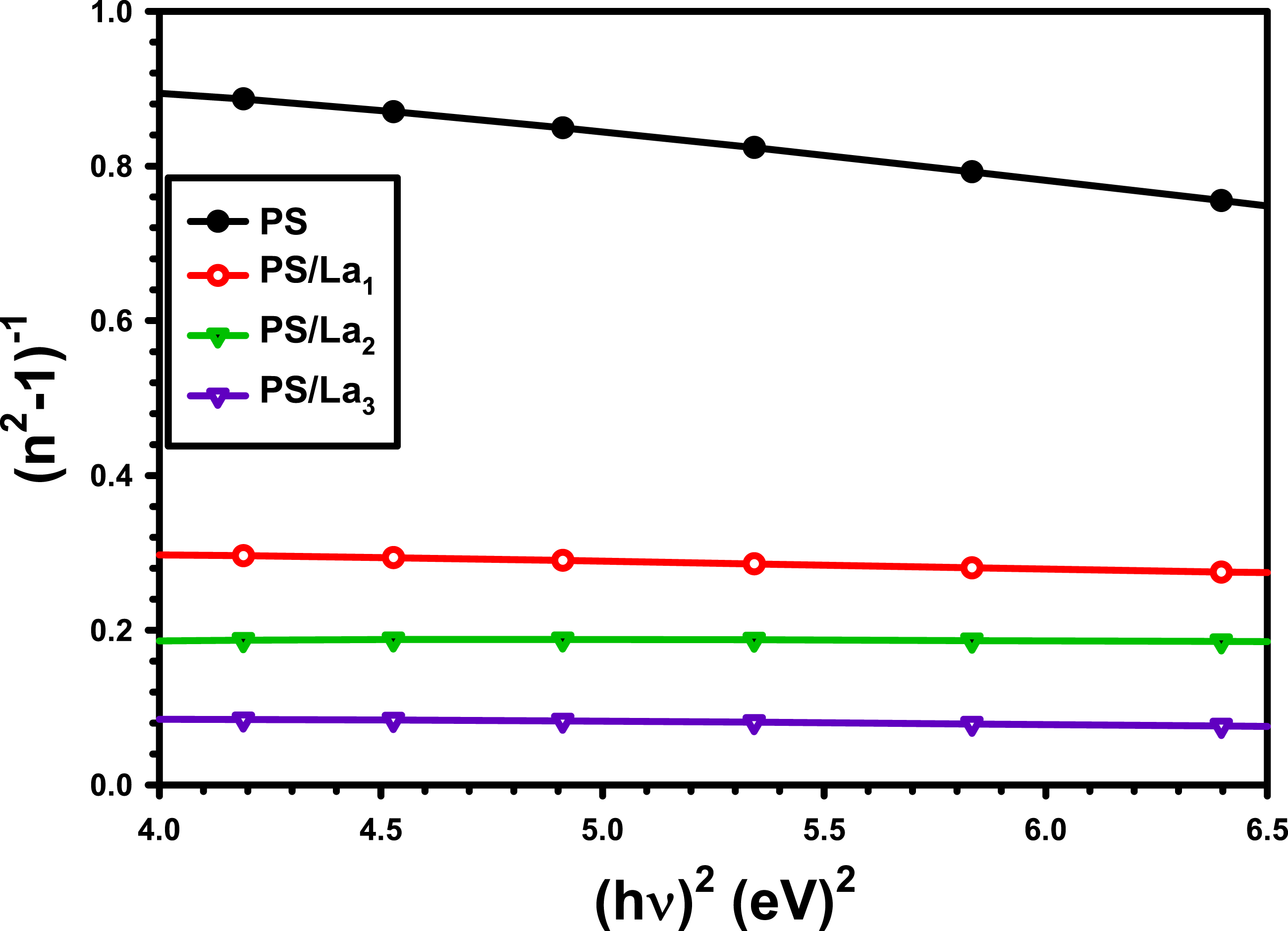
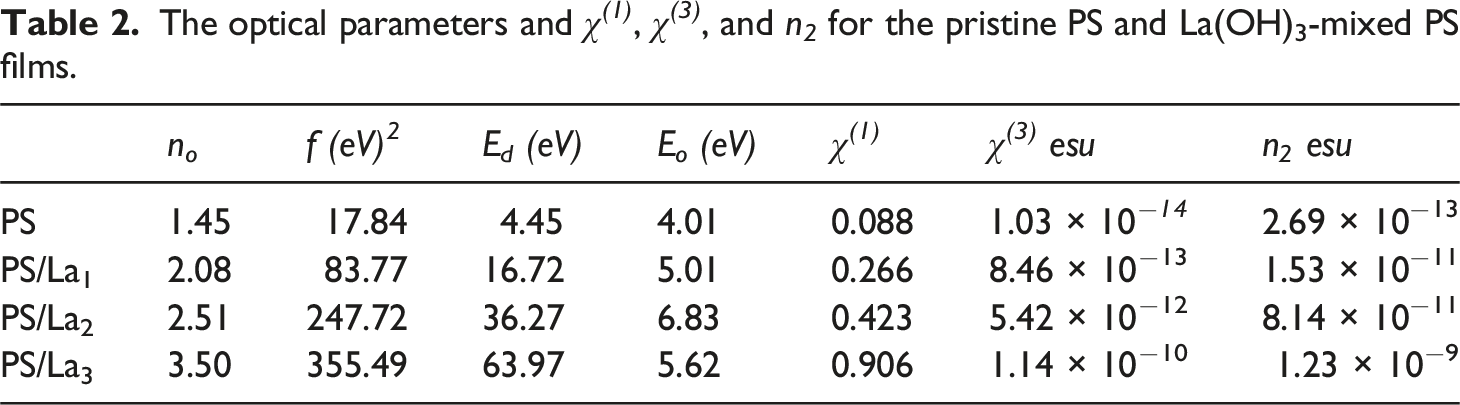
The assessed values of n o and f were listed in Table 2. The n o value ranged between 1.45 and 3.50 for PS/La3 nanocomposite film, and f started from 17.84 (eV)2 to 355.49 (eV)2. This increase in no and f values is due to new structural defects created by incorporating La(OH)3NPs with the PS backbone. 63
The nonlinear optical characteristics of materials are fundamental to the operation of many advanced optical devices, including all-optical switching and integrated photonic devices. The first-order linear optical susceptibility can be given for the pure PS and doped PS films with La(OH)3 NPs as follows
64
:
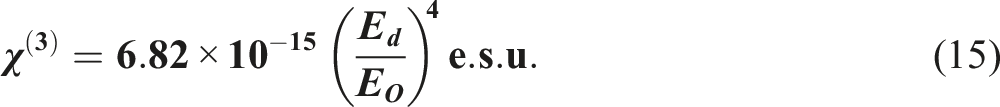

Conclusions
Using the solution casting method, novel and flexible optoelectronic nanocomposite films were produced based on PS and La(OH)3 NPs. XRD analyses reveal that La(OH)3 NPs are effectively incorporated into the PS matrix, exhibiting a pure hexagonal phase with a crystallite size of ∼26.32 nm. FTIR spectra indicated the functional groups of pristine PS and PS/La(OH)3 nanocomposite films. From the linear optical characteristics, the absorbance and absorbance coefficient of the PS increased with the increase in the amount of La(OH)3 NPs in the nanocomposite films. The optical band gap of the virgin PS film is 3.83 eV, respectively, and decreases to 3.67, 3.55, and 3.39 eV with increasing La(OH)3 NPs loading from 1 to 3 wt%. Additionally, the Urbach energy value of the pure PS increased with the increase in nanofiller concentration in the nanocomposite films. Furthermore, the VELF and SELF values were enhanced with higher amounts of La(OH)3 NPs within the PS matrix. For the dispersion parameters, the static refractive index (n o ) was boosted from 1.45 of the pure PS to 3.50 for PS/La3 nanocomposite film, and the oscillation strengths (f) increased from 17.84 (eV)2 to 355.49 (eV)2. A significant increase was noted in the nonlinear χ (3) value of the pure film from 1.03 × 10−14 to 8.46 × 10−13, 5.42 × 10−12, and 1.14 × 10−10 esu for PS/La1, PS/La2, and PS/La3 nanocomposite films, respectively. The results provide evidence of improved linear and nonlinear optical properties in PS after the addition of La(OH)3 NPs; therefore, the nanocomposite films are expected to excel in optoelectronic devices.
Footnotes
Authors contributions
A.M. Elbasiony: Writing – review & editing, Conceptualization, Writing – original draft, Funding, and Formal analysis. Nuha Al-Harbi: Data curation, Conceptualization, Writing – review & editing, Software, Resources, and Investigation. M. M. Abdelhamied: Writing – original draft, Methodology, Investigation, Formal analysis, Conceptualization, and Supervision.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors extend their appreciation to the Deanship of Scientific Research at Northern Border University, Arar, KSA for funding this research work through the project number “NBU-FFR-2025-3049-11”.
Data Availability Statement
Data will be made available on request.


