Abstract
This study shows how montmorillonite clay (C30B) and titanium dioxide (TiO2) can be incorporated into EVA polymer through melt blending, utilizing clay:TiO2 ratios of 1:0, 2:1, 1:1, 1:2, and 0:1. WAXD, SEM, TGA, and stress-strain tests were used to evaluate the structure, morphology, thermal stability, and mechanical properties of the materials. Additionally, a three-hour ultraviolet irradiation study was conducted using an accelerated aging chamber equipped with eight UV lamps. Post-irradiation products were analyzed using FTIR and DRX. The results indicated a widespread distribution of clay layers within the polymer; however, the dispersion of TiO2 particles was minimal. Despite this, thermal stability remained unchanged, while mechanical properties showed improvement. Irradiation experiments revealed photodegradation of the polymer after just 3 hours of exposure to UV light. We propose that the polar and non-crystalline nature of vinyl acetate (VA) allows interactions with the reactive oxygen species generated by TiO2, facilitating the photodegradation of EVA polymer. The results suggest that versatile polymer materials can be created by blending clay and TiO2 with EVA. TiO2 can leverage its photocatalytic properties to decompose volatile compounds, pollutants, or microorganisms under controlled UV irradiation. Ultimately, the materials can undergo self-photodegradation after their use and disposal, offering environmental benefits.
Introduction
Ethylene-vinyl acetate (EVA) copolymer is a polymer with wide uses, such as thermoplastic elastomer for cable sheathing, foamed material, and hot melt adhesive. 1 The variety of properties that the polymer can achieve is due to the ethylene-vinyl acetate ratio in its structure. Furthermore, since EVA is safe for the body and not harmful, it has been studied for making materials used in medicine, like devices for delivering drugs,2,3 and orofacial protectors. 4 Despite its wide range of applications, some researchers have combined EVA with several inorganic compounds to enhance specific properties. For instance, EVA has been blended with montmorillonite clay to improve the flame retardancy5,6 and the gas barrier. 7 Some works have also combined EVA with titanium dioxide (TiO2). TiO2 is a compound that has been exploited for its photocatalytic capabilities.8–10 When this compound is exposed to ultraviolet light, it creates electron-hole pairs that react with water and oxygen in the environment to produce reactive oxygen species (ROS). 11 These chemical species can cause the degradation of volatile organic compounds, solvents, dyes, or microorganisms. To improve its surface properties and photocatalytic ability, some studies have attempted to combine TiO2 particles with montmorillonite clay.12–14 Conveniently, other studies have incorporated montmorillonite and TiO2 into polyethylene to improve its characteristics for food packaging applications.15–18 Yousefi et al. reported that mixing clay and TiO2 with starch biopolymer improves the mechanical and thermal properties and the water vapor barrier. 19 In a previous study, we described how TiO2 nanoparticles were attached to montmorillonite clay layers, and then the clay-TiO2 combination was mixed into natural rubber. 20
Based on the information provided, this work aimed to examine the inclusion of organomodified montmorillonite clay and TiO2 in the EVA polymer. The study describes how the structural and morphological behaviors of the EVA/clay-TiO2 mixtures change and how they react to ultraviolet light. The results presented here aim to lay the groundwork for creating medical devices that can fight bacteria or cancer using the beneficial properties of TiO2 and the safe nature of the EVA polymer. Additionally, after use and disposal, the material may undergo photodegradation under sunlight, thereby minimizing its environmental impact. Our thorough review of existing studies reveals that there is only one report about mixing clay and TiO2 with EVA polymer, which explains how gamma irradiation is used to attach glycidyl methacrylate to the surface of TiO2 and clay to make them work better with EVA. 21
Experimental
Materials
Ethylene-vinyl acetate (EVA) copolymer with 40 wt% vinyl acetate and a melt index of 52 g/10 min was acquired from DuPont Co. under the name Elvax 40w. An organomodified montmorillonite clay, called Cloisite 30B (C30B), was purchased from Southern Clay Products. The organic modifier in the clay is methyl, tallow, bis-2-hydroxyethyl, quaternary ammonium, where tallow consists of carbon chains with 65% C18, 30% C16, and 5% C14. P25 titanium dioxide (Degussa Co.) with a high content of the anatase phase and a specific area of 50 ± 1 m2/g was used.
Nanocomposite processing
A Beutelspacher single-screw extruder with a diameter of 19 mm and an L/D ratio of 30:1 was used to obtain the EVA/clay-TiO2 nanocomposites. Three heating zones were set at 85, 100, and 105°C, and a screw speed of 40 r/min was used. Two processing cycles were used to obtain nanocomposites. In the first cycle, EVA was blended with clay. Then, the product (EVA/clay) was processed with TiO2 in a second cycle. The ternary blends were made under clay:TiO2 ratios of 1:0, 2:1, 1:1, 1:2, and 0:1. In all cases, a 3% total weight of the reinforcing agents (clay plus TiO2) was considered. Therefore, we consistently maintained the EVA copolymer content at 97% by weight. To compare the results, the polymer labeled EVA, sample E/CT-10 (which indicates that the polymer contains only clay), and sample E/CT-01 (which contains only TiO2 as a reinforcing additive) were established as controls within the series of experiments. The other samples contained the indicated additive ratios.
Preparation of test specimens
For stress-strain tests, type IV specimens were prepared according to ASTM D638. For this purpose, the products were compression molded at 100°C for 3 min under a pressure of 10 tons and die-cast. We used some of the specimens for other characterizations.
Test of irradiation with ultraviolet light
The samples were placed in an accelerated aging chamber, using eight ultraviolet light irradiation lamps. The aging tests were carried out at 1, 2, and 3 hours of irradiation.
Characterization
Wide-angle X-ray diffraction (WAXD)
The low-angle diffraction signal of clay was examined using a Siemens model D-500 X-ray diffractometer, which has a CuKα radiation generator and a Ni filter. The working conditions were set from 2 to 10° in 2θ, with increments of 0.004° and a scanning speed of 6 seconds per step.
X-ray diffraction (XRD). A Bruker AXS X-ray diffractometer, model D8 Advance, was used to determine the changes in the crystalline behavior of the ternary system. The tests were performed in the 2θ range from 5 to 60°, with increments of 0.1, at a scanning speed of 4 seconds per step and a sample rotation of 30 r/min.
Field emission scanning electron microscopy (FE-SEM)
The morphology was analyzed using a field emission scanning electron microscope (FE-SEM) model JSM-740H, set to 5 kV and positioned at a working distance of 6 mm. The samples were previously fractured with liquid nitrogen and coated with a thin layer of gold-palladium alloy.
Mechanical tests
The stress-strain tests were carried out on an Instron 4301 Universal machine at a speed of 20 in/min, and applying a load of 500 N. A statistical analysis based on four experimental replicates was performed using one-way ANOVA with Fisher´s test at p < .05 in Minitab 19.
Thermogravimetric analysis (TGA)
The mass loss curves of the samples were created using a TA Universal V4.5A thermogravimetric analyzer. The analyses were carried out at a heating rate of 20°C/min and within a temperature range of 30 to 600°C under a nitrogen atmosphere.
Fourier-transformed infrared spectroscopy (FTIR)
The impact of ultraviolet light on the polymer was measured using a Perkin Elmer infrared spectrometer model Spectrum 100, equipped with an ATR accessory. The analyses were performed at 100 scans, using a resolution of 4 cm−1 and in a range of 4000 to 500 cm−1.
Results and Discussion
Structural and morphological behavior
The separation of the clay layers in the polymer was examined using WAXD. We conducted this analysis to verify the diffraction peak associated with the clay’s (001) crystallographic plane. The signal correlates with the distance between the adjacent surfaces of the stacked clay layers. If a new peak appears at low angles, with a 2θ value lower than that of the peak for the (001) plane of pure clay, it indicates that the clay layers have been partially separated. This separation is usually due to the confinement or intercalation of polymer chains in the clay galleries. The absence of any peak in the diffraction pattern indicates that the (001) plane is no longer present. This behavior suggests that the clay layers have become fully disordered and separated, leading to a nanodispersion of individual clay layers within the polymer. Figure 1 shows that pure clay has a peak at 4.8°, which is related to the (001) plane; however, this peak is not present in any of the EVA/clay-TiO2 nanocomposites. Therefore, this behavior signifies a complete separation and dispersion of the clay layers throughout the polymer. Similar results were reported by Duan et al., who combined EVA with an organomodified clay.
22
In similar works, we have shown the good dispersion of organomodified clay on the EVA polymer. We have attributed this phenomenon to polar interactions between the surface of the clay and the vinyl acetate in EVA, which favors compatibility between the two phases.23–25 In the present study, in addition to clay, there is TiO2, and according to the results, the TiO2 particles do not affect the interaction behavior between EVA and clay. Diffraction patterns obtained by WAXD of the clay and the EVA/Clay-TiO2 nanocomposites with the indicated clay-TiO2 ratio.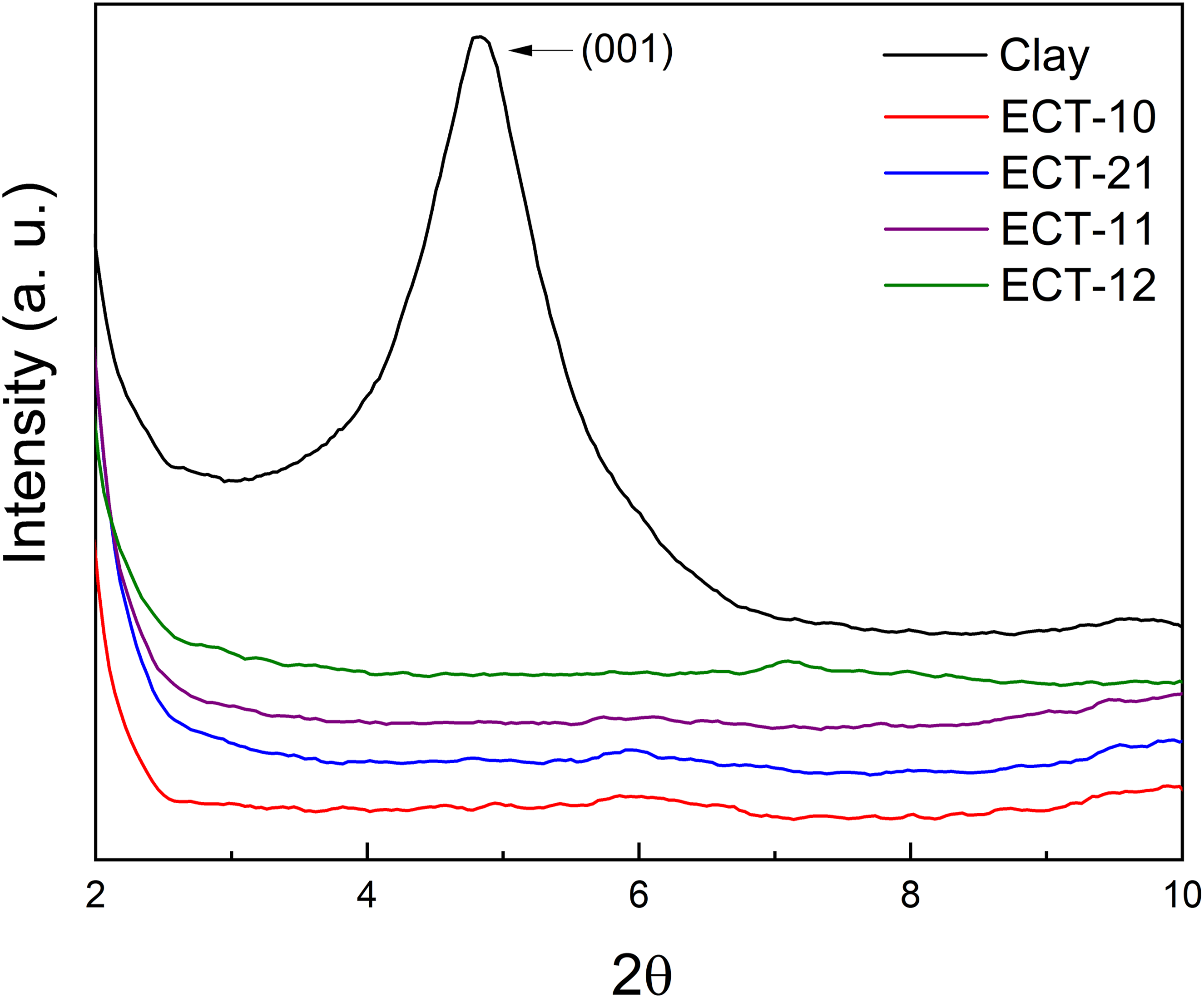
The morphological behavior of the clay was verified using electron microscopy, as shown in Figure 2. Initially, as a raw material, the clay consists of ordered stacked layers that form agglomerates with sizes greater than 50 microns. However, during the extrusion process and when it interacts with the polymer, the clay undergoes unfolding and delamination, resulting in the formation of disordered individual layers (Figure 2(a)). Consequently, the WAXD results demonstrate the loss of the crystallographic plane associated with the layer stacking, as shown in Figure 1. The double processing cycle creates high shear stresses that break the clay agglomerates and mix them better with the polymer. Additionally, because of the attractions between the surface of the clay and the vinyl acetate in the EVA, the polymer chains were confined between the clay layers and efficiently separated them (Figure 2(b)). In contrast, the TiO2 particles were not sufficiently dispersed in the EVA polymer, as shown in Figure 3(a). Even though TiO2 (P25) is formed by nanoparticles with an average size of 35 nm (see Figure 3(b)), the strong cohesive forces between these particles cause their agglomeration.
26
Therefore, neither the polar characteristics of the EVA polymer nor the clay influenced the dispersion behavior of the TiO2 particles. Despite this, the processing conditions allowed partial breakage of the TiO2 agglomerates, thereby achieving a less coarse dispersion in the polymer. In a previous study, we showed that TiO2 particles can interact favorably with organomodified clay in the presence of natural rubber. The result was a consequence of weak attractive forces between the polymer and clay, allowing interactions between the clay and TiO2.
20
Subsequently, the results shown here indicate that the interactions between EVA and clay prevail over those generated between TiO2 and clay or between TiO2 and the polymer. Therefore, the characteristics of the polymer significantly contribute to achieving good compatibility when adding two inorganic additives. FE-SEM micrographs of clay on the EVA polymer. (a) Unstacking of the clay layers through mechanical mixing and molecular interactions, and (b) delamination of clay layers, generating exfoliation on the polymer matrix. FE-SEM micrographs of a nanocomposite containing TiO2. (a) Clay layers and an agglomerate of TiO2 particles on polymer matrix, and (b) TiO2 nanoparticles with an average size of 35 nm.

As previously stated, the strong attractive forces between TiO2 particles make their dispersion in the polymer difficult. In the literature, several strategies have been reported to improve the dispersion of TiO2 in a polymer matrix. In some cases, TiO2 has been modified to combine it with a polymer blend (HDPE, LDPE, PP, and PVC) and wood flour, 27 or to improve dispersion with a poly (amide-imide) using a blending process in solution. 28 In other cases, TiO2 has been surface-functionalized for blending with carbon fiber and HDPE 29 or with polyamide six via a multiple extrusion processing. 30 Another study reported the surface treatment of TiO2 with HCl for blending with PMMA. 31 Additionally, the use of modifying agents 32 or a twin-screw extruder33,34 have also been reported. Therefore, according to the literature, there are several strategies for combining TiO2 with a polymer. However, in a ternary system such as the one in this study, the degree of dispersion depends on both reinforcing additives, that is, on the competition between attractive and repulsive forces among the three chemical species present in the system. This, together with the processing conditions used during extrusion mixing, can improve the dispersion of the inorganic additives in the polymer. In our case, the established processing conditions were based on preliminary tests of temperature profiles, screw speed, feeding method, and processing cycles. Therefore, based on the preliminary results, two processing cycles were considered, along with the most suitable processing parameters to obtain physically homogeneous materials with high nanometric clay dispersion. As other authors have reported, extrusion conditions and processing cycles improve the dispersion of the reinforcement within the polymer.30,34 In some cases, up to seven processing cycles have been used. 30 However, when multiple processing cycles are employed, thermal and shear degradation phenomena must be considered. Although processing conditions help improve mixing and dispersion, the dispersion of particles at the nanometer scale is primarily dependent on the compatibility behavior between the reinforcing additives and the polymer. In our system, due to the high compatibility of the clay with the polymer, the clay dispersion results predominate over the dispersion of the TiO2 particles. As indicated, the polar groups in EVA promote melt blending with the -OH groups of the clay, which prevents greater interaction with TiO2.
Mechanical properties
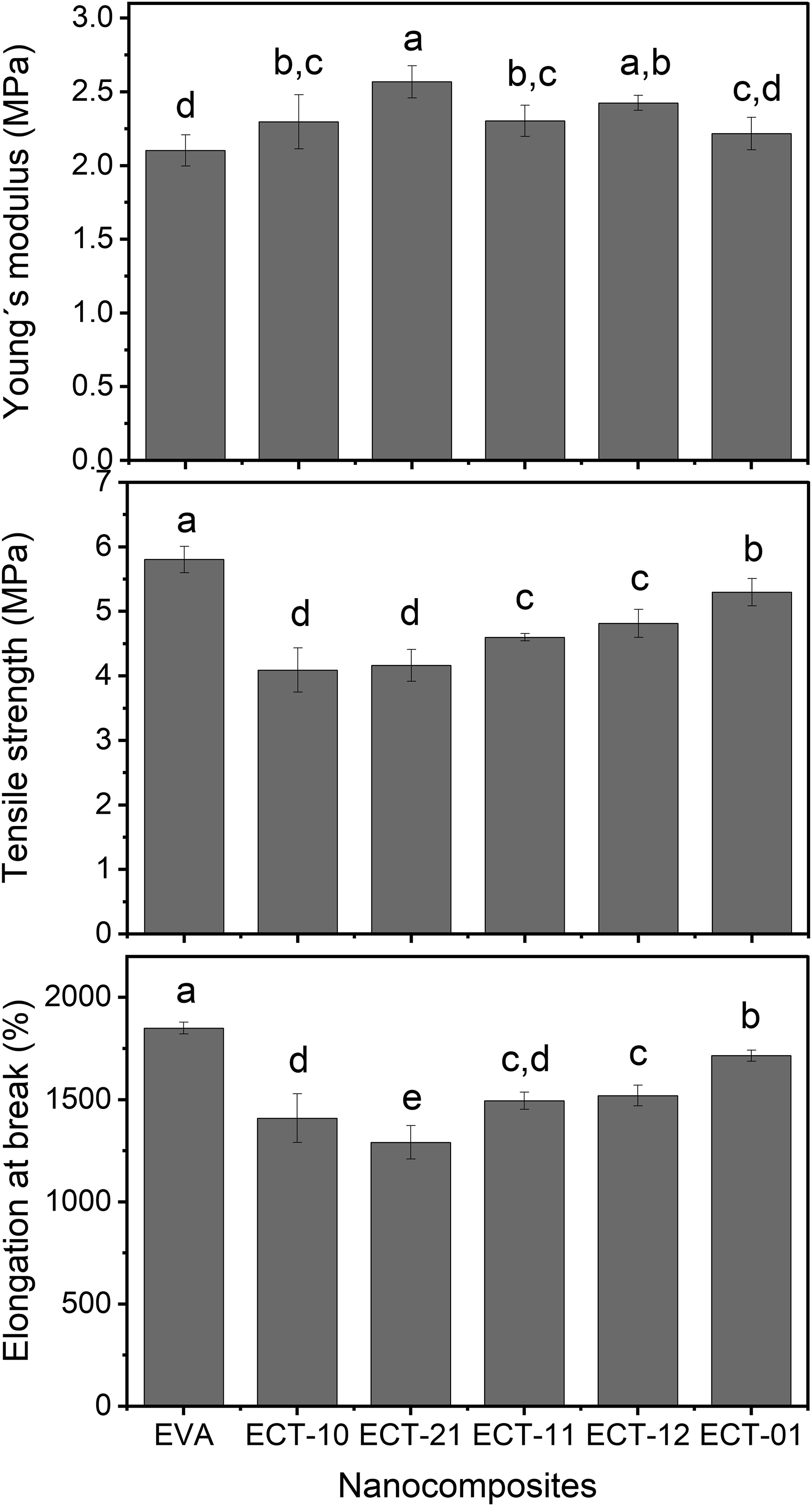
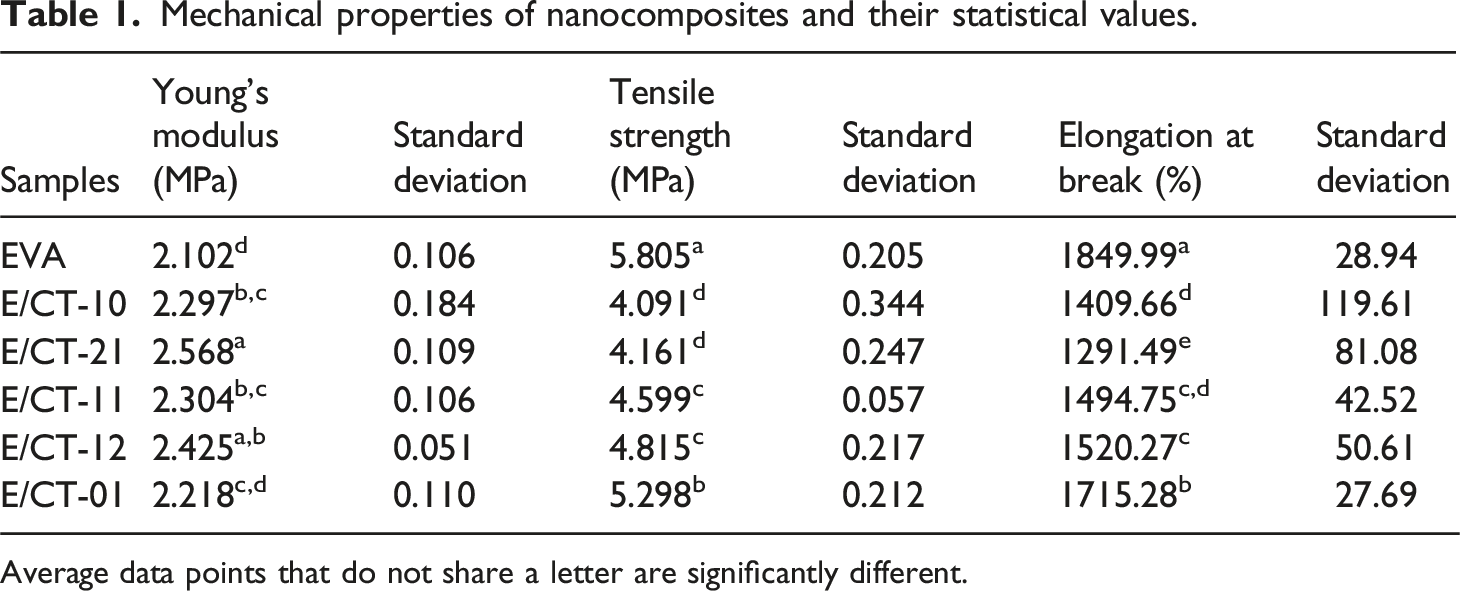
The stress-strain curves are shown in Figure 4. Apparently, the additives increase the Young’s modulus (see inner box) and decrease the strain at break. To confirm this behavior, Figure 5 shows the results for Young’s modulus, tensile strength (maximum stress), and elongation at break (strain at break). The Young’s modulus changes with inorganic additives. Generally, clay increases the modulus, and its behavior depends on the degree of nanometric dispersion of the clay layers. Apparently, the phenomenon also occurs with TiO2 particles. However, although Figure 5 shows a trend towards an increase in Young’s modulus, the change is slight, with some values remaining close to the standard deviation (Table 1). The results are consistent with those reported by Farhoodi et al., who showed slight increases in Young’s modulus when they incorporated nanoclay or nanoTiO2 into PET.
33
Similarly, Zhenhua et al. reported that incorporating TiO2 into PPMA can improve the stiffness of the polymer.
32
According to Figure 5, the 2:1 clay:TiO2 combination produces the clearest increase in Young’s modulus. Conversely, the tensile strength decreased with the addition of inorganic components but increased in the presence of TiO2 (ECT-01). Other authors have reported that the inclusion of clay aims to increase tensile strength, and the effect is enhanced if the clay is dispersed.
35
On the other hand, as shown in Figures 4 and 5, the elongation at break of the nanocomposite decreases in the presence of clay (ECT-10, red line Figure 4), but increases again with TiO2. This result is consistent with what was reported by Bodaghi et al., who showed an increase in the strain at break of PE with TiO2.
18
As expected, in elongation at break, the polymer without additives (EVA) exhibits a higher value, which decreases with the presence of inorganic additives due to the restriction of chain mobility caused by the nanometric particles. This behavior occurs primarily with clay. However, in the samples with the highest TiO2 content (ECT-12 and ECT-01), the elongation at break increases again. Other authors have reported an increase in strain at break with the incorporation of TiO2, indicating improved ductility with TiO2 and reduced ductility with the addition of clay.
33
The effects observed in the mechanical properties are related both to the dispersion behavior of the clay to the agglomeration of TiO2 particles within the ternary system. As other authors have reported, the agglomeration of TiO2 particles affects the properties of a composite material.27,30,33,34 Agglomeration leads to poor distribution within the matrix, reduces the surface area of the reinforcement, and can generate heterogeneity in the material properties. Stress-strain curves of EVA/Clay-TiO2 nanocomposites with the indicated clay-TiO2 ratio. Mechanical properties of the nanocomposites and their statistical analysis. Mechanical properties of nanocomposites and their statistical values. Average data points that do not share a letter are significantly different.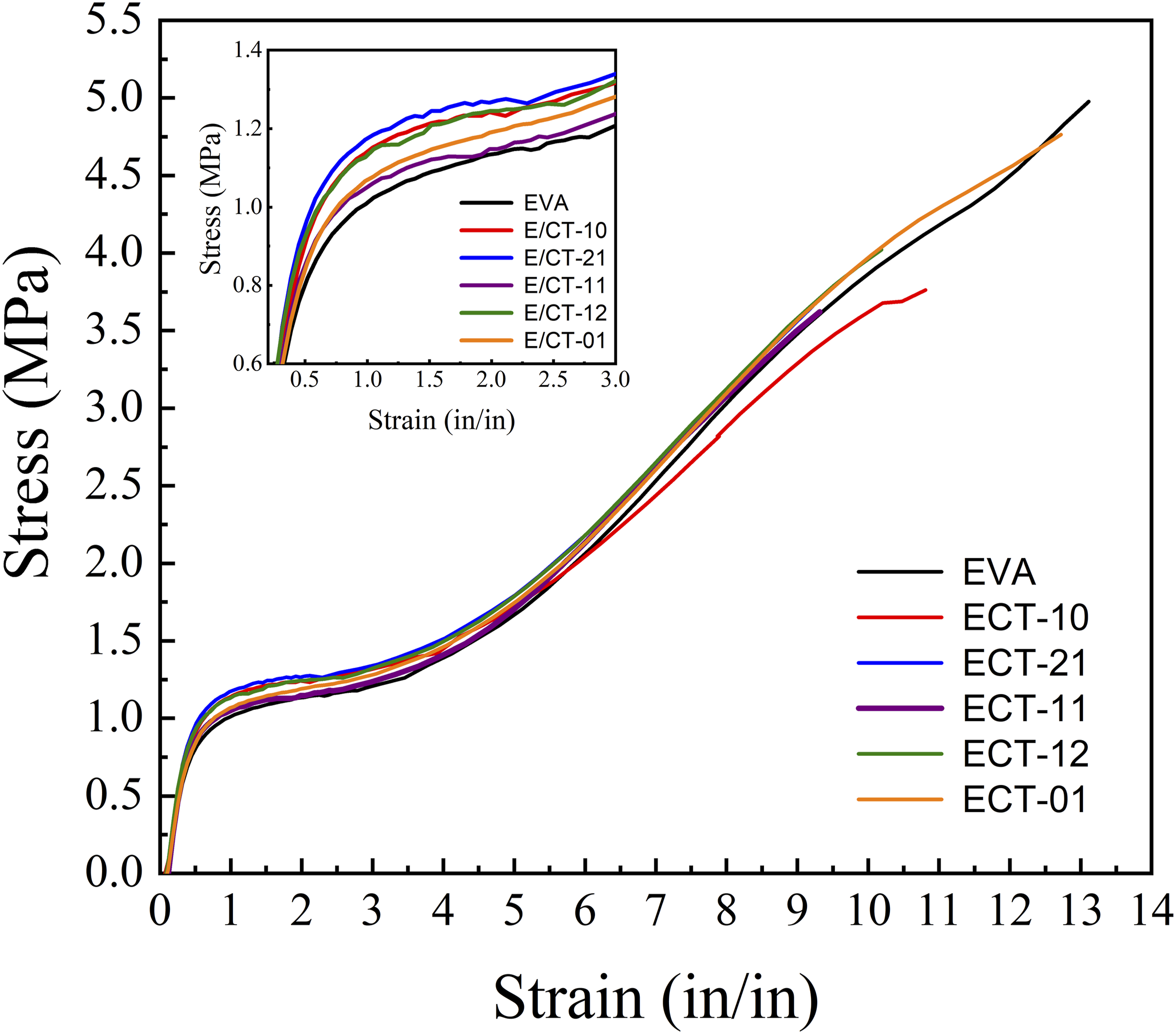
Thermal behavior
Figure 6 displays the TGA results. Slight variations can be seen along the weight loss curves (Figure 6(a)). However, the derivatives obtained from the TGA data show exactly the differences in weight loss temperatures (Figure 6(b)). The degradation curve of the EVA polymer shows two weight loss states. The first state, around 350° C, corresponds to the deacetylation reaction of vinyl acetate with the elimination of acetic acid. The second state takes place at approximately 470°C and is associated with the chain scission of the copolymer ethylene component.17,36 Even though the shifts in the degradation temperature of the EVA copolymer are not that significant, it is evident that changes in its stability occur. Morphology of dispersed layers with a high aspect ratio and the intrinsic ceramic condition of the clay can create a heat barrier effect, delaying the degradative behavior of the macromolecules (see EC-10, red line). On the contrary, TiO2 particles cannot create a greater thermal barrier to the polymer due to the nanometric size of each particle and their spherical geometry (see ECT-01, orange line). TGA Data of the nanocomposites with the indicated clay-TiO2 ratio. (a) Weight loss curves, and (b) derivatives calculated from the weight loss curves.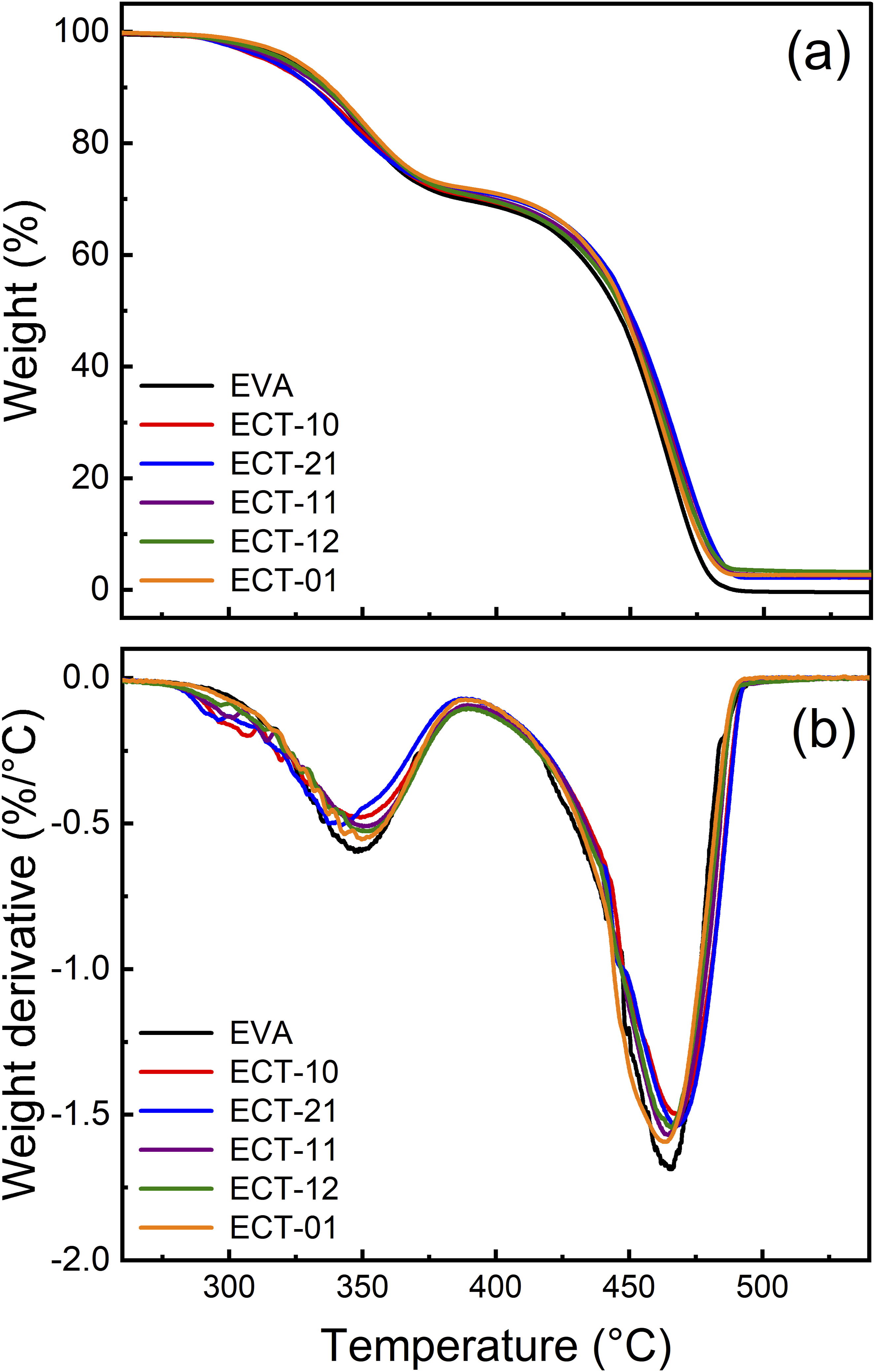
Response to ultraviolet light irradiation
TiO2 is a semiconductor with a band gap energy around 3.2 eV.37,38 This compound acts with ultraviolet light under wavelengths less than 380 nm. Irradiation excites electrons from the valence band to the conduction band, generating electron-hole pairs, which interact with water and oxygen molecules in the environment, producing reactive oxygen species (ROS). These chemical species can degrade organic pollutants, dyes, microorganisms, and other systems.39,40 If TiO2 is added to a polymeric matrix, ROS could also cause degradation of the host polymer chains through susceptible functional groups. Figure 7 shows photographs of samples of nanocomposites obtained with different clay-TiO2 ratios, exposed to UV irradiation for 1, 2, and 3 hours. Although Figure 7 shows only qualitative results, the effects on the macrostructure achieved over time are notable. The physical change, such as the waviness in the central region of the samples, which appears to be a localized effect, is due to the arrangement of the samples within the aging chamber. The samples were placed on hollow plates, with their ends held in place by the plates and the remainder of each sample was exposed to irradiation. As a result, the photodegradative effect occurs from the center of the specimen toward the ends of the samples, causing a wavy distortion, as observed in the irradiated samples. On the other hand, the comparative photodegradation effect may be linked to internal phenomena such as dispersion, homogeneity, or some other factor. At the end of the aging tests, it was possible to perform some characterizations of the products, but it was not possible to measure the mechanical properties because the irradiated samples were not easily handled for mechanical testing, as they exhibited adhesive behavior at the end of testing time, due to the temperature increase caused by the irradiation. Photographs of the nanocomposites irradiated under ultraviolet light for 1, 2, and 3 hours.
Based on Figure 7, TiO2 enhances the photodegradative behavior of the polymer. This behavior is evident in the sample with the highest amount of TiO2 (see photograph of ECT-01 at 3 h). As other authors have reported under different testing conditions, TiO2 shows a strong photodegradative behavior towards some polymers.41–43 In our case, we have a copolymer as the matrix, where the ethylene part provides chemical and mechanical resistance. Therefore, we consider that the action of TiO2 occurred in the vinyl acetate (VA) region. The absence of crystalline order and the susceptibility of the acetate group to attack by free radicals generated by TiO2 particles favored the photodegradation of the polymer. On the other hand, in the photographs of Figure 7, it is also observed that the nanocomposite containing only clay as a reinforcing additive (ECT-10) suffers greater deformation due to the photodegradative effect than the sample without clay (pure EVA). This behavior implies that the clay also catalyzes the photodegradation reaction of the polymer, although it does so at a slower rate than TiO2. Some reports in the literature have demonstrated the photocatalytic capacity of montmorillonite clay in other polymers,44,45 which is consistent with this result.
The photodegradation behavior of the nanocomposites was also monitored by FTIR. Figure 8 shows the spectra at different regions of infrared light absorption for the samples irradiated for 1, 2, and 3 hours (a, b, and c, respectively). Although the nanocomposites contain EVA, clay, and TiO2, due to the concentration of the polymer in each system, the polymer signals predominate in the spectra. The C-O absorption signal from EVA overlaps the most relevant absorption peak of the clay, around 1000 cm−1, which corresponds to the stretching vibration of Si-O bonds.
46
Therefore, the absorption bands at 1734, 1235, and 1018 cm−1 correspond to symmetric or asymmetric stretching vibrations of C=O and the C-O-C bond in EVA,
47
which originate from the vinyl acetate in the copolymer. The signal at 1641 cm−1 corresponds to an amine additive contained in the EVA. The peaks at 1563, 798, 720, and 604 cm−1 are related to the stretching and bending vibrations of the C-C and –CH2 bonds in the ethylene part of the copolymer. The changes in the absorption peaks are evident as a function of the irradiation time. Considering the sample with the longest irradiation time (Figure 7(c)), a higher TiO2 content (ECT-01, orange line, and ECT-12, olive green line) shows lower intensity of the peaks related to vinyl acetate (1734, 1235 cm−1) and higher intensity of the peaks related to the ethylene part of EVA (1563, 720, 604 cm−1). As previously stated, the action of ROS impacts the vinyl acetate region, altering the structure of the copolymer. In contrast, the ethylene component exhibits greater resistance to photodegradation. Therefore, the results indicate that the EVA copolymer undergoes partial degradation. However, photodegradation depends on the exposure time to UV light, as shown in the photographs in Figure 7. Vibration bands of the spectra obtained by FTIR of the nanocomposites exposed to UV irradiation for 1, 2, and 3 hours.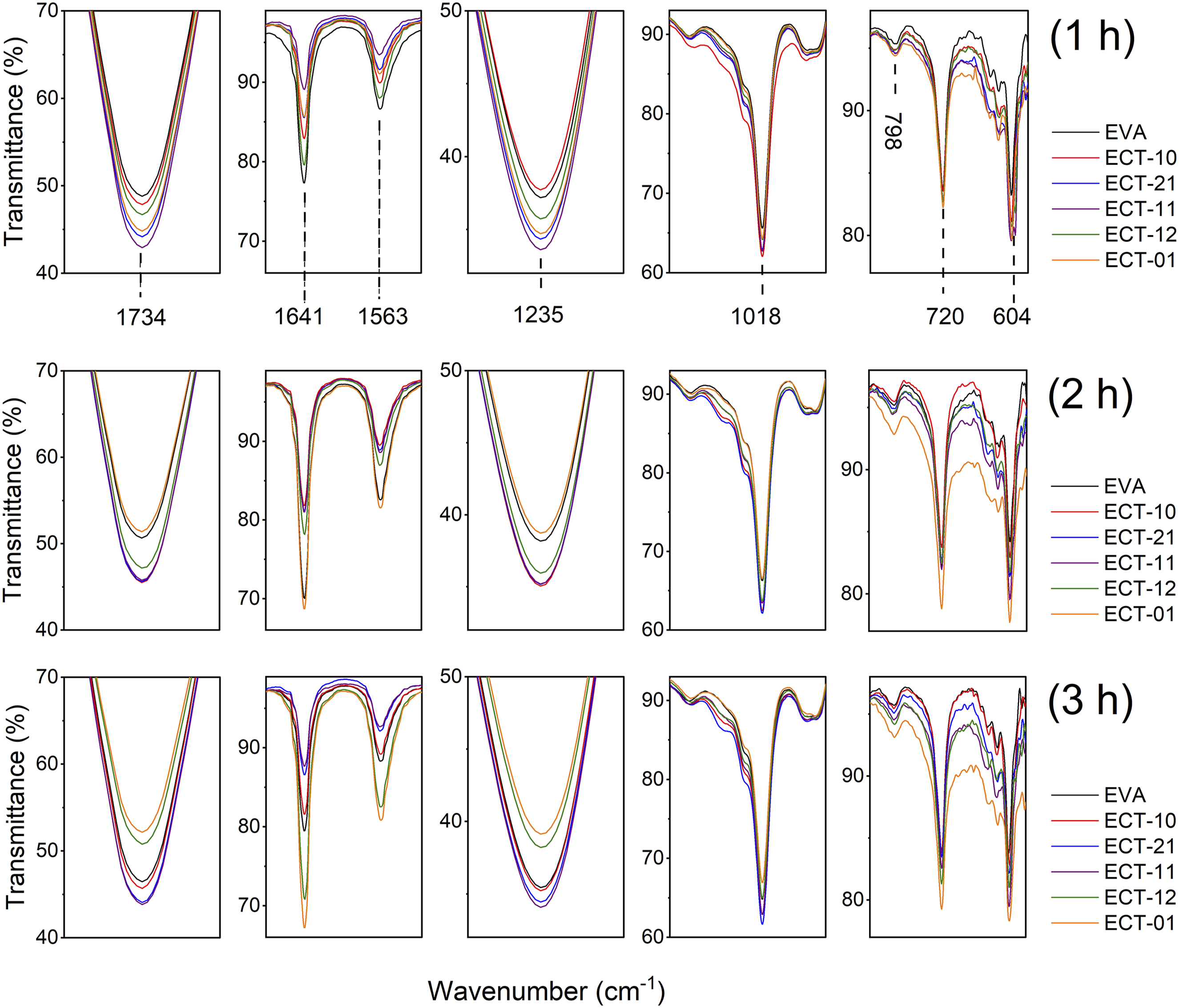
Figure 9(a) shows the diffraction patterns of the nanocomposites after 3 hours of UV irradiation. The predominant peak is observed at around 20° in 2θ. This peak corresponds to the crystalline domains of ethylene within the EVA copolymer. Similarly to the FTIR spectra, the crystalline signals of the clay are overlapped by the EVA peak, as shown in Figure 9(b). The graph omits the signal of the clay with the maximum intensity, which occurs at an angle of less than 10° in 2θ and corresponds to the (001) plane associated with the interlayer spacing of the clay. We previously examined the behavior of this crystallographic peak in relation to the WAXD results (refer to Figure 1). Figure 9(b) also displays the diffraction pattern of TiO2, revealing distinctive peaks of the anatase and rutile structures, as documented by earlier researchers.
48
However, because the TiO2 used contains 70% of anatase, the most noticeable signals correspond to this structure. Consequently, the peak (101), characterized by the maximum intensity in anatase, is evident in the nanocomposites ECT-21, ECT-11, ECT-12, and ECT-01 as depicted in Figure 9(a). The results indicate that UV irradiation does not completely decompose the nanocomposites. The crystalline structure of the ethylene zone offers some protection to the samples. However, this protective effect diminished as the TiO2 content increases, as evidenced by the ECT-01 diffraction pattern (orange line), which shows a decrease in the intensity of the peak or the crystalline degree in the EVA. Thus, we conclude that the photodegradative effect induced by TiO2 primarily affects the amorphous portion of the copolymer (vinyl acetate). Nonetheless, depending on the amount of TiO2 and the duration of irradiation, the crystalline domains may also undergo photodegradation. X-ray diffraction patterns of: (a) the nanocomposites at the indicated clay-TiO2 ratio after 3 hours of UV irradiation, and (b) the raw materials EVA, clay, and TiO2. The inner box marks the peak related to the anatase structure of nanocomposites containing TiO2.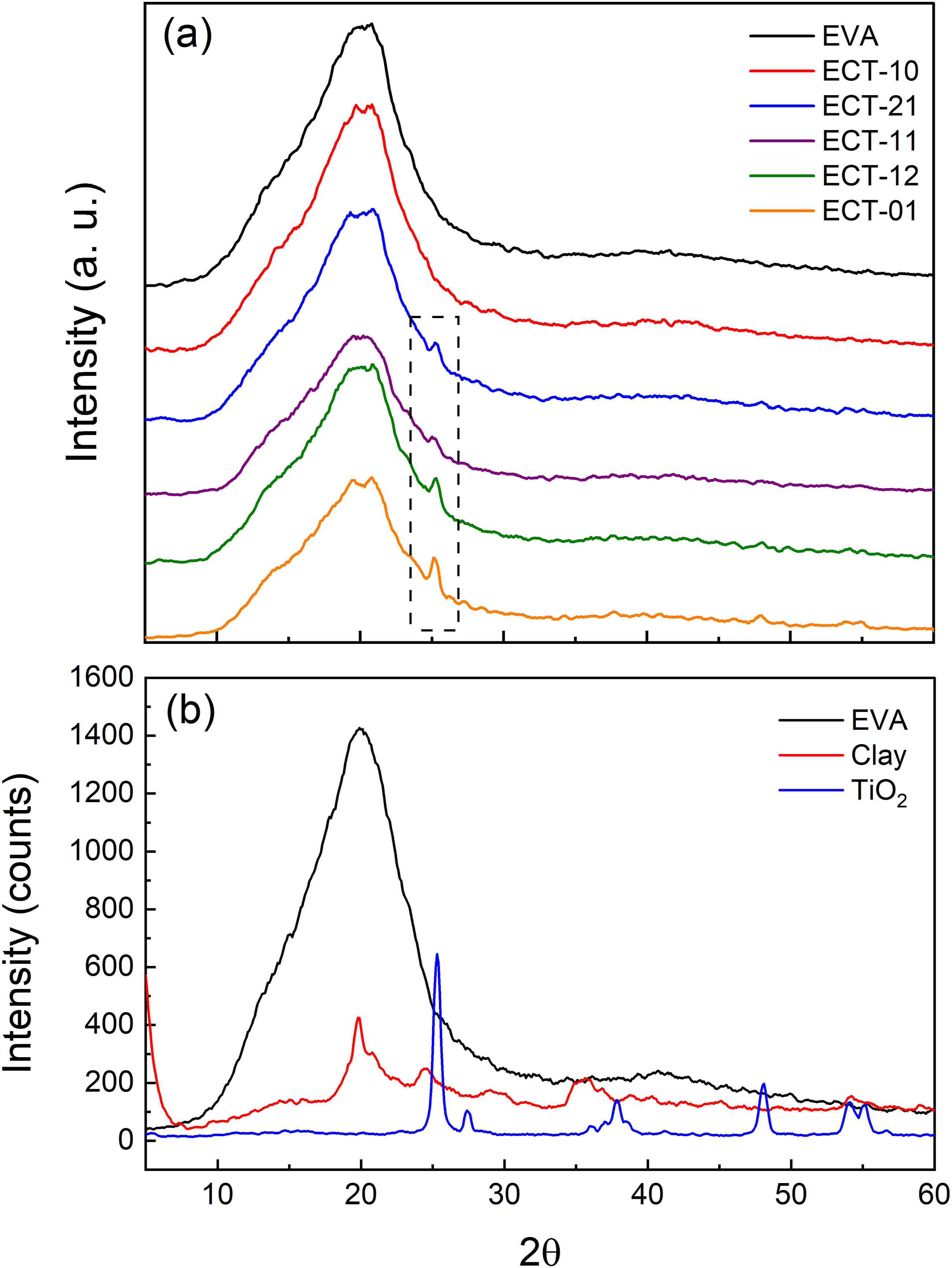
Conclusions
The combination of the EVA copolymer with TiO2 and a modified organoclay was studied. The results showed the nanodispersion of clay layers in the polymer, but not the deagglomeration of TiO2 particles in the polymer matrix. This implies that the interfacial attraction between clay and EVA is stronger than the attraction between clay and TiO2 or between EVA and TiO2. The results are therefore dependent on the interplay of attractive and repulsive forces between the three components. Based on this result, future studies aim to seek a greater affinity between TiO2 and clay to achieve a high dispersion of TiO2 particles on individual clay layers without affecting the nanodispersion already obtained by the clay layers on the polymer matrix. On the other hand, the UV irradiation results indicated that the deformation of the nanocomposites occurs as a function of the TiO2 content and the irradiation time. The effect was generated in the amorphous zone of the copolymer, which corresponds to the vinyl acetate. The ethylenic part of the copolymer provides greater protection against photodegradation. However, the protection is dependent on the irradiation time and the amount of TiO2 present in the system. These results suggest that these materials could be used to create devices that utilize the photocatalytic capabilities of the polymer additives, such as medical devices like catheters and antimicrobial patches. Sunlight can self-photodegrade the devices after use and disposal, preventing environmental contamination from the previously used plastic device.
Footnotes
Acknowledgements
B. Castro-Hilario thanks SECIHTI for the scholarship awarded for these master´s studies. The authors thank M.C. Cantú-González, B.M. Huerta-Martínez, and J.A. Cepeda-Garza for their technical assistance.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
