Abstract
In this study, the structural, optical, and radiation shielding characteristics of a PVA/PVP blend loaded with varying ratios (0%, 1%, 2%, and 3 wt%) of Cr2O3 nanoparticles (NPs) are studied. This research is driven by the increasing demand for lightweight, flexible, and transparent shielding materials as alternatives to traditional, heavy, and hazardous substances such as lead. This study’s novelty arises in establishing a thorough correlation between the microstructural modifications induced by Cr2O3 NPs and their subsequent optical and theoretically assessed radiation shielding features. The X-ray diffraction (XRD) analysis of the produced Cr2O3 NPs revealed a crystallite size of 42 nm. The optical band gap value of the PVA/PVP mix was 3.51 eV, which decreased to 1.97 eV with the incorporation of 3% wt. of Cr2O3 NPs. The PVA/PVP mix shows an Urbach energy value of 1.42 eV, which incrementally increases to 3.98 eV when the concentration of Cr2O3 NPs in the PVA/PVP blend escalates from 0 to 3% by weight. The impact of Cr2O3 NPs concentration on the radiation shielding characteristics of PVA/PVP/Cr2O3 nanocomposites has been theoretically assessed using the Phy-X tool and the XCOM database. The HVL value varied from 0.475 cm to 29.137 cm for PVA/PVP at 0.015 MeV and 15 MeV, respectively. At the same time, the HVL value ranged from 0.259 cm to 28.161 cm for the PVA/PVP/3%Cr2O3 nanocomposites at 0.015 MeV and 15 MeV, respectively. The exposure buildup factor (EBF) of PVA/PVP/Cr2O3 nanocomposites has been analyzed as a function of photon energy. Furthermore, the equivalent atomic number (Zeq) values for PVA/PVP/Cr2O3 nanocomposites have been studied. The optical features and radiation shielding performance of PVA/PVP/Cr2O3 nanocomposites make them suitable for radiation shielding applications.
Introduction
Researchers have been interested in wide-band-gap semiconductors for decades due to their potential uses in electrical and optoelectronic devices as well as their unexpected properties. 1 Nanostructures of semiconductor metal oxides are intriguing to nanoscience and nanotechnology despite their unusual structural geometry, due to their frequently exhibiting unique characteristics compared to their coarse-grained counterparts.1,2 Polymer nanocomposites incorporating diverse additives, including metal oxides, carbon-based nanomaterials, and high-Z metal nanoparticles, have garnered considerable interest for industrial applications due to their enhanced mechanical, electrical, and thermal properties.3–21 Trivalent chromium oxide (Cr2O3) is the most stable among other chromium oxide phases, including hexavalent (CrO3) and tetravalent (CrO2). 2 Cr2O3 exhibits antiferromagnetism and possesses a substantial energy gap of up to 3.4 eV, finding utility in several applications, including the production of pigments for ceramics, coatings, paints, and printing. 1 Moreover, it has been examined as a lithium cathode, solar absorber, catalyst, and hydrogen sorbent.22,23 Additionally, it is utilized in electrohydrogenation, dehydrogenation, dye removal, biomedical applications, and gas sensing.24–26 Chromium oxide-based polymer nanocomposites, such as nano-Cr2O3, are increasingly recognized for their ability to combine the processability and flexibility of polymers with the superior thermal, optical, and dielectric properties of chromium oxide fillers, thereby facilitating the development of materials with multiple applications for advanced technologies. Cr2O3-doped polymer blends in flexible electronic and energy-storage devices exhibit enhanced dielectric constant, loss, and AC conductivity with increasing nanoparticle loading and frequency, rendering them suitable for storage capacitors, solid-state semiconductors, and other electrical uses. The integration of Cr2O3 nanoparticles into polymers diminishes the optical energy band gap, enhances UV absorption, and modifies refractive index dispersion, facilitating the tuning of optical constants for UV-blocking coatings, optoelectronic components, and photonic devices.27,28
Polyvinyl alcohol (PVA) is a semicrystalline polymer that provides numerous applications due to the presence of hydroxyl groups and hydrogen bonding. Due to its affinity with biological systems, it can also serve as a medicinal agent. Furthermore, PVA exhibits solubility in organic solvents, hydrophilicity, crystallinity, and self-lubricating characteristics.29–31
In the same way, polyvinylpyrrolidone (PVP) is readily soluble in water and an environmentally friendly polymer that possesses exceptional properties, including compatibility, a superior dielectric constant, and low environmental impact. The interaction between PVA and PVP occurs through hydrogen bonding between the hydroxyl group of PVA and the carbonyl group of PVP. Minor incorporations of PVP are considered to improve miscibility, strengthen hydrogen-bonding interactions, and promote the uniform dispersion of nanoparticles, whereas larger PVP concentrations can negatively impact the mechanical strength of PVA-based films. 32 A consistent distribution of fillers in the polymers would be achieved by merging polymers with nanofillers.33,34
Many nanocomposites of PVA/PVP with different fillers have been studied, including silicon dioxide, titanium dioxide, zinc oxide, cadmium sulfide, copper oxide, manganese oxide, and magnesium oxide. 35 Dhatarwal and Sengwa 31 have documented the optical characteristics of PVP/PVA/Al2O3 nanocomposite films. In our previous work, we prepared PVA/PVP (80/20 wt%) loaded with V2O5 NPs for optoelectronics. 36
Our review of the relevant literature indicates that no effort has been conducted to investigate the optical characteristics and radiation shielding properties of the PVP/PVA mix loaded with Cr2O3 nanoparticles (NPs). Therefore, taking into consideration these facts, the purpose of this paper is to investigate in depth the optical behavior of the PVP/PVA/Cr2O3 films with a fixed polymer blend compositional ratio (90/10 wt%) and varying amounts of loaded Cr2O3 NPs (0, 1, 2, and 3 wt%) to investigate their potential applications in optoelectronic devices. In addition, the influence of the concentration of Cr2O3 NPs on the radiation shielding properties of PVA/PVP/Cr2O3 nanocomposites has been theoretically estimated using the Phy-X program and the XCOM database. Additionally, the exposure buildup factor (EBF) and the equivalent atomic number (Zeq) values for PVA/PVP/Cr2O3 nanocomposites have been studied.
Experimental
Using the co-precipitation method, we have synthesized pure Cr2O3 NPs. Chromium (III) nitrate nonahydrate chemical reagents of 99.9% purity were utilized as Cr precursor. The magnetic stirrer continuously agitated 0.1 M of Cr precursor solution for half an hour. The pH value was subsequently regulated by adding an aqueous ammonia solution dropwise. At a pH of 9, the green precipitates commence to form. The soluble impurities were removed by filtering and washing the precipitates formed with distilled water multiple times. Next, the Cr2O3 NPs precipitate was dried at 200°C and annealed at 900°C in a muffle furnace for 3 hours. The PVA/PVP films doped with Cr2O3 NPs were prepared according to the steps described in our previous work. 36
The mass attenuation coefficient (MAC), the half-value layer (HVL), the tenth-value layer (TVL), the mean free path (MFP), the effective atomic number (Zeff), and the effective electron density (Neff) values were calculated according to37–40.
Results and Discussions
Structural Analyses
Figure 1(a) displays the X-ray diffraction patterns (XRD) of the synthesized powder Cr2O3 NPs. The appearance of sharp peaks in the Cr2O3 pattern corroborates the presence of nanoparticles. The angular positions are 24.4°, 33.63°, 36.15°, 41.1°, 50.22°, 54.8°, 63.3°, and 65.10°corresponding to the planes (012), (104), (110), (113), (024), (116), (214), and (300), correspondingly. The observed peak positions correspond well with JCPDS file number 1308-38-9, indicating rhombohedral lattice planes with parameters a = 4.958 Ǻ and c = 13.594 Ǻ.
41
Figure 1(b) shows EDX spectra of Cr2O3 NPs, which affirmed the presence of Cr and O elements with no foreign elements. Additionally, EDX mapping images confirmed the uniform distribution of Cr and O elements over the sample (Figure 2). (a) XRD profile and (b) EDX spectra of Cr2O3 NPs. EDX mapping images of Cr2O3 NPs.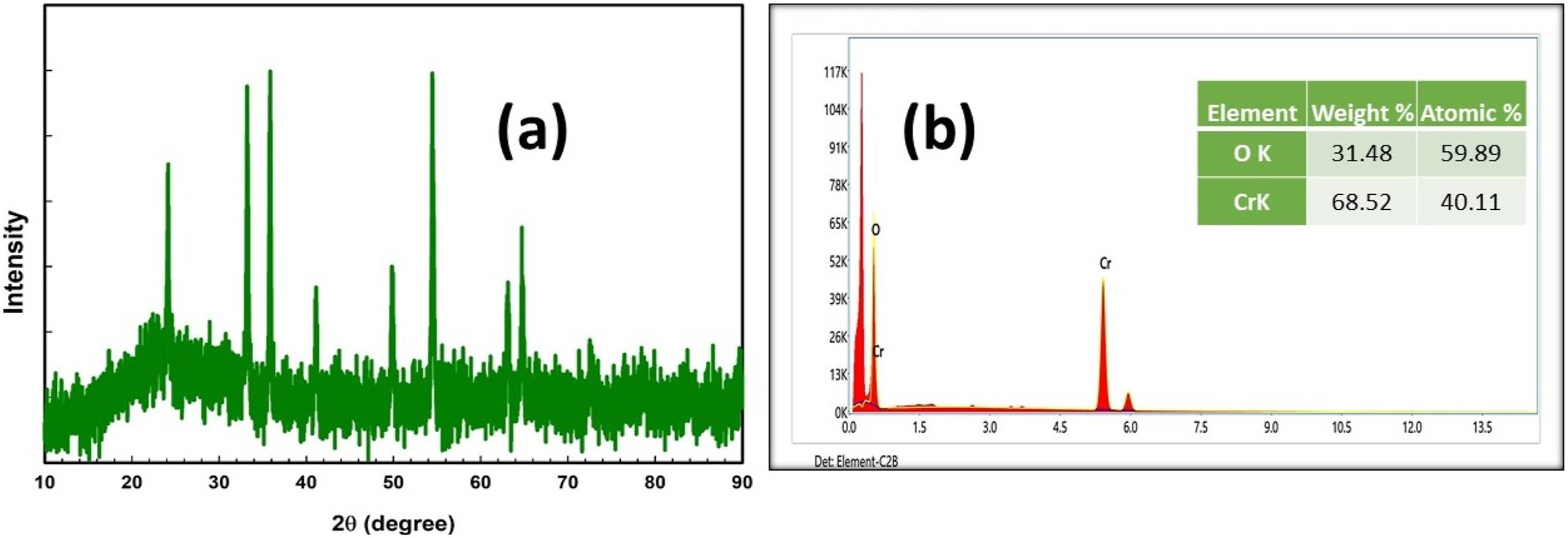

Figure 3 displays XRD patterns of the PVA/PVA pure and PVA/PVP/x wt% Cr2O3 (x = 1, 2, and 3). A relatively broad peak at 2θ = 19.26° can be observed, which may be attributed to the amorphous polymer matrix of the PVA/PVP blend.
33
Moreover, the diffraction patterns of PVA/PVP/x wt% Cr2O3 (x = 1, 2, and 3). The prepared films exhibit both the diffraction peak of PVA/PVP and the diffraction peaks of Cr2O3, which means that characteristic peaks demonstrate that Cr2O3 NPs indeed occur in the amorphous phase of PVA/PVP. The figure illustrates that the relative intensity of the peak of PVA/PVP (2θ = 19.26°) diminishes following the incorporation of Cr2O3 NPs into a PVA/PVP matrix. At a doping concentration of 1 wt% Cr2O3, new peaks at 2θ values of 24.4, 33.6, 36.6, and 54.8° corresponding to the crystal planes (012), (104), (110), and (116) of crystalline Cr2O3
42
begin to emerge with impressive, heightened intensities. The distinctive peaks indicate that Cr2O3 NPs are present in the amorphous phase of PVA/PVB. Simultaneously, as the content of Cr2O3 NPs rises, the severity of these peaks escalates. XRD patterns of PVA/PVP/x wt% Cr2O3 (x = 0, 1, 2, and 3).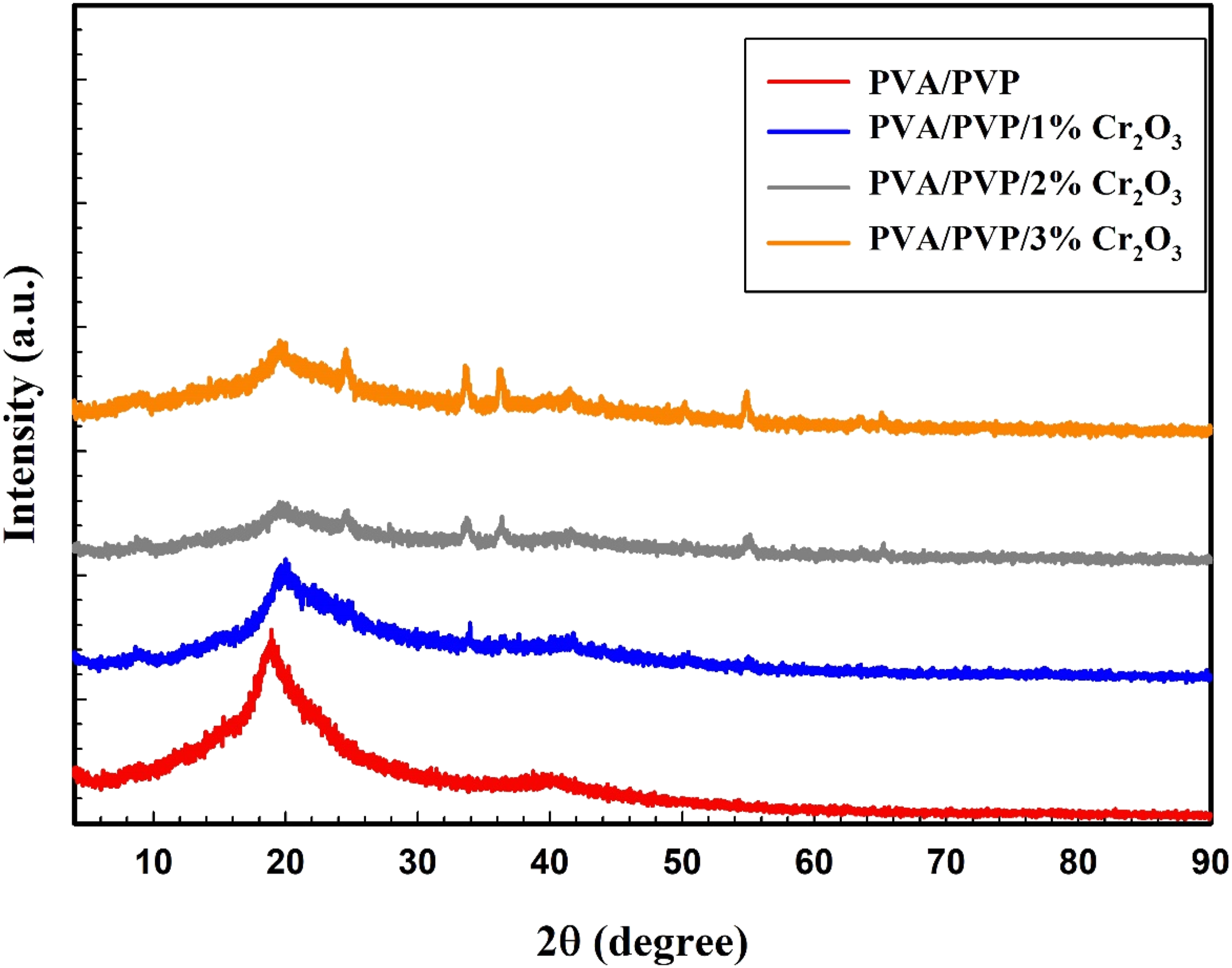
Additionally, the average grain size of the acquired phase D was estimated by applying Debye–Scherrer’s formula provided below
43
: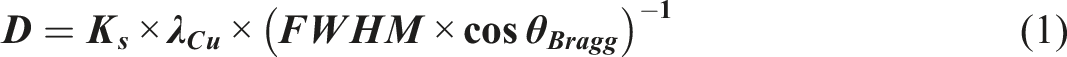
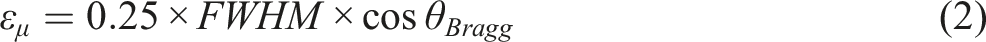
Moreover, the pertinent connection is employed to compute the dislocation density of the crystallization phases in the Cr2O3 and PVA/PVP/x wt% Cr2O3 (x = 1, 2, and 3)
44
:
The crystallite size (D), micro-strain (ε), and dislocation density (δ) of pure Cr2O3, and PVA/PVP/x wt% Cr2O3 (x = 1, 2, and 3).
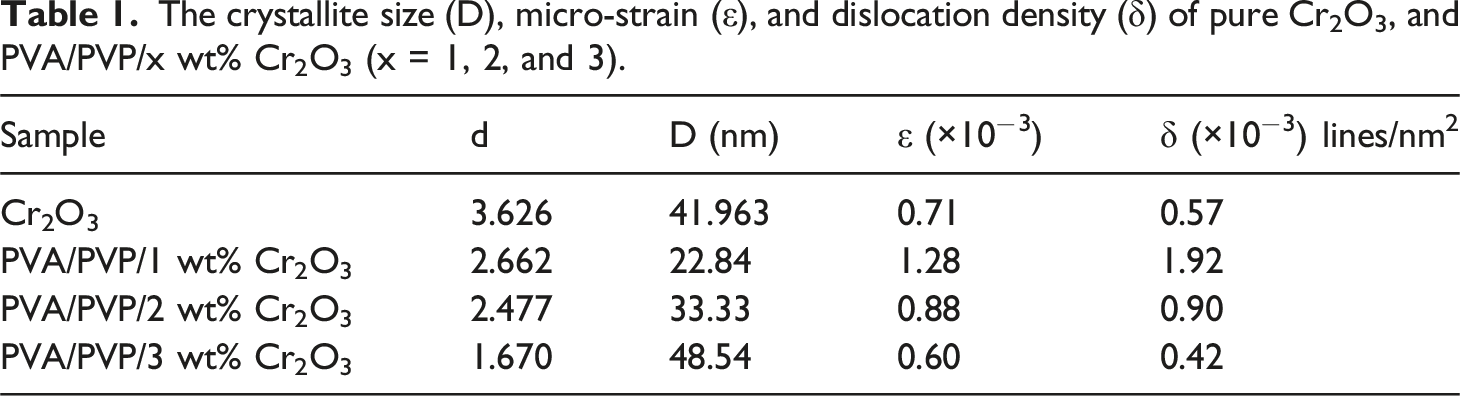
The augmentation in crystallite size indicates a strengthened contact between the polymer chains and Cr2O3 nanoparticles, facilitating the rise of crystallite size, as previously documented. 45 This signifies an improvement in the interaction between the Cr2O3 nanofiller, increasing the size of the Cr2O3 nanoparticle coalescence in the polymers. Conversely, this reduces micro-strain and dislocation density within the system as the Cr2O3 content escalates. 45 The structural properties of the obtained films will influences both the optical and radiation shielding properties.
Optical Characteristics
Figure 4 depicts the absorbance spectra of the PVA/PVP/Cr2O3 nanocomposites. A markedly reduced absorbance was noticed over the visible spectrum. It is noteworthy that absorbance rises when λ drops and with a boost in the concentration of Cr2O3 NPs in PVA/PVP. Increasing the Cr2O3 NPs loading produces a gradual redshift in the PVP/PVA/Cr2O3 absorption bands, moving the spectral features to longer wavelengths as the concentration rises. This behavior is because of the rising Cr2O3 NPs concentrations, which produce more localized energy levels and defects that promote light absorption.
36
Multiple investigations identified these shifts in behavior.46–50 The unpaired electron pairs of oxygen atoms in PVA and the n→π* and π→π* transitions related to the carbonyl group (C = O) in PVP are identified by their UV absorbance behavior, which depends on the wavelength.
36
The absorbance spectra of the PVA/PVP/Cr2O3 nanocomposites.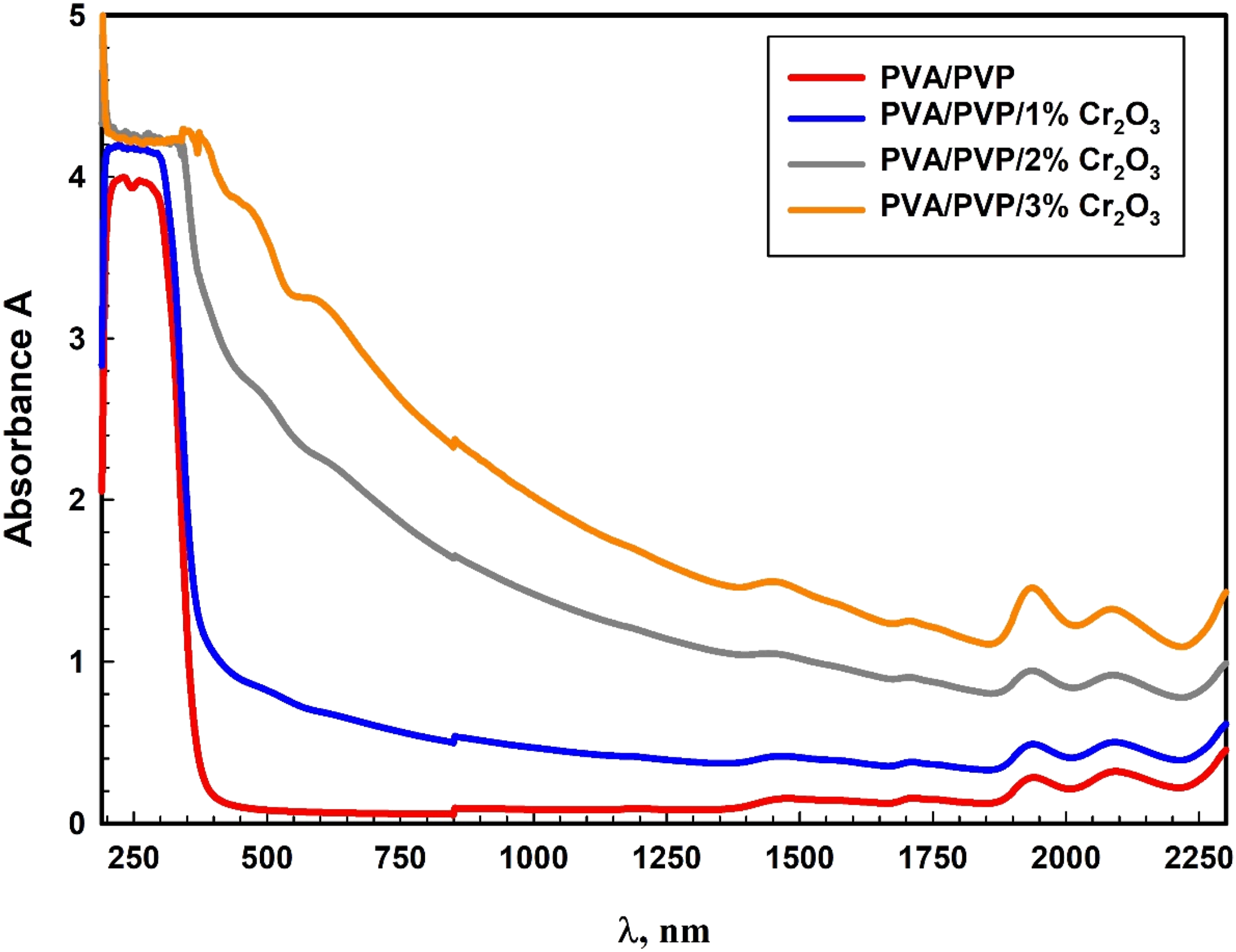
Tauc’s relation can be employed to determine the direct band gap Eg from the absorbance edge, as indicated by the absorption spectra
51
:
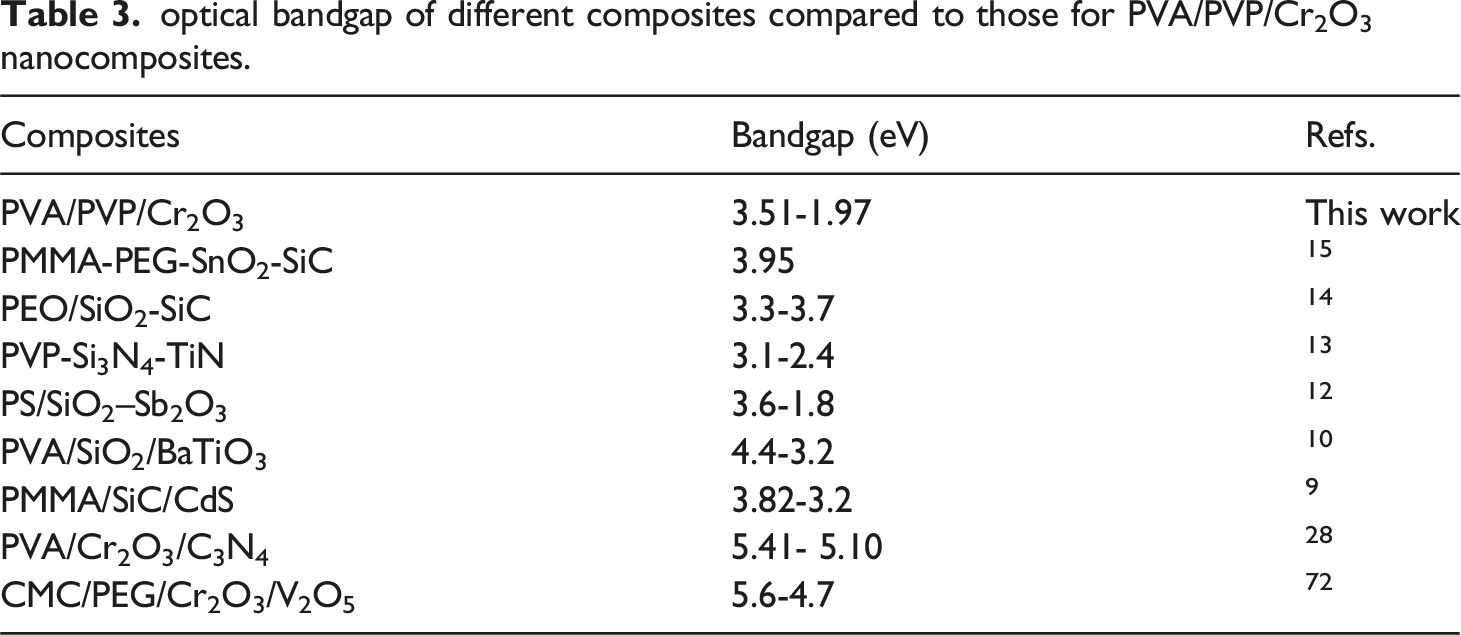
The extrapolation procedure, previously described,47,52,53 is used to ascertain the Eg values of PVA/PVP/Cr2O3 nanocomposites. Figure 5(a) illustrates the (a) Optical band gap and Urbach energy Eu for PVA/PVP/Cr2O3 nanocomposites. optical bandgap of different composites compared to those for PVA/PVP/Cr2O3 nanocomposites.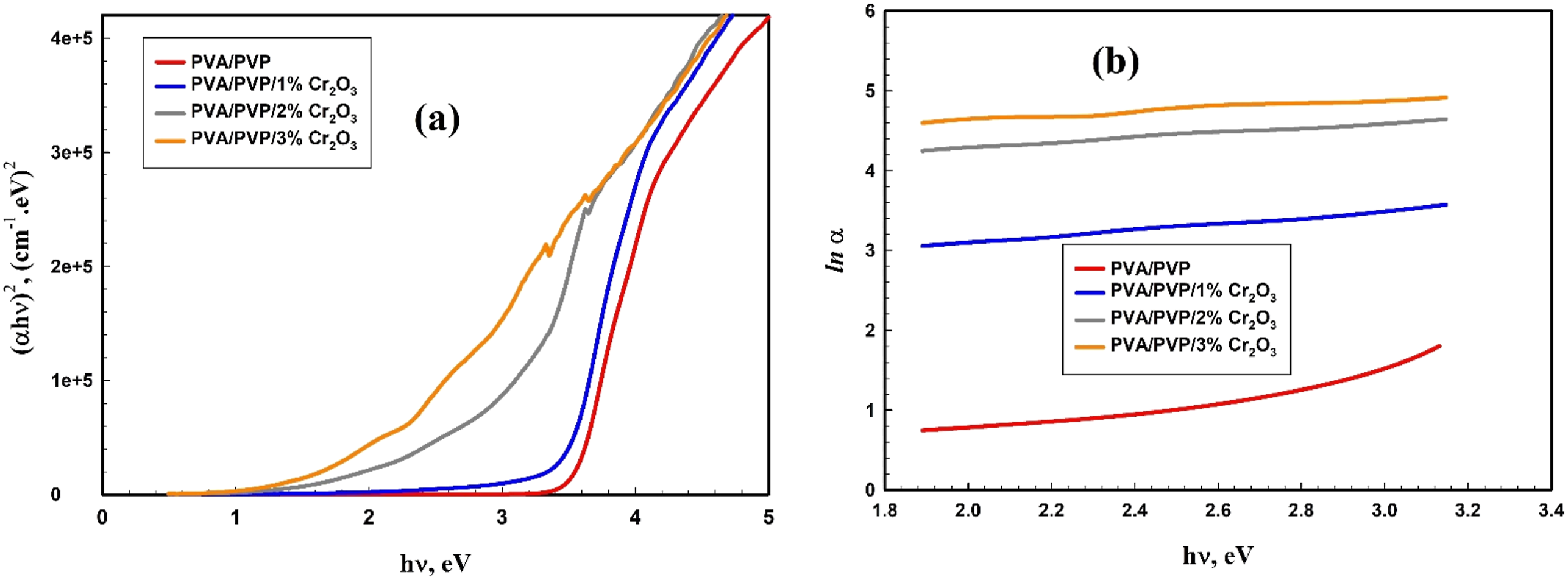
Urbach energy Eu refers to the exponential variation in defects in the PVA/PVP/Cr2O3 nanocomposites.
59
The correlation between the photon energy and Urbach energy can be expressed as
60
:
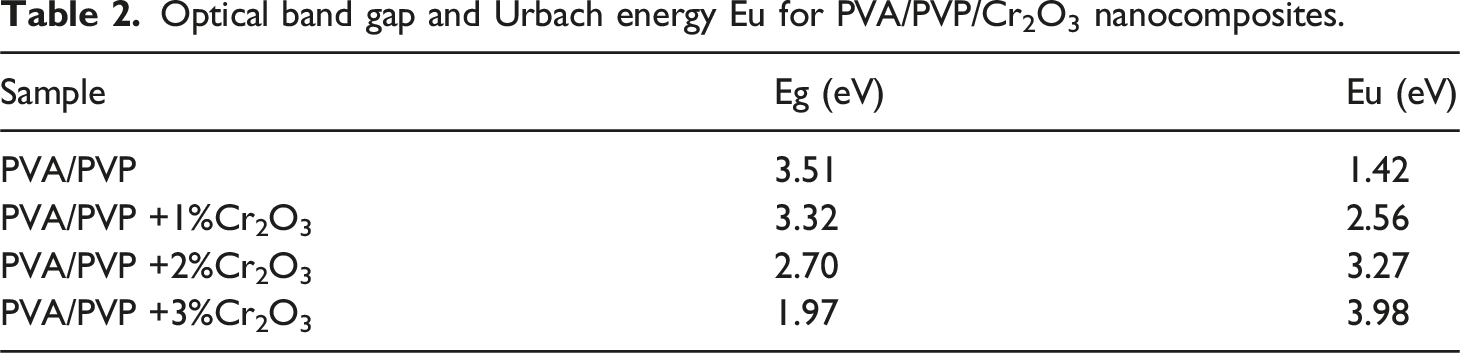
Figure 5(b) illustrates the relationship between ln α and hν for the PVA/PVP/Cr2O3 nanocomposites. Consequently, the Urbach energy values were calculated and shown in Table 2. The PVP/PVA blend exhibits a Urbach energy value of 1.42 eV, which increases progressively to 3.98 eV when the Cr2O3 NPs concentration in the PVP/PVA rises. This result indicates that the energy band gap of PVA/PVP/Cr2O3 nanocomposites is marginally reduced, since electronic transitions are notably enhanced by increased structural disorder and the formation of localized states.31,52
The refractive index (n) and the extinction coefficient (k) are optical features that significantly influence the advantageous technological applications of nanocomposites in optical technology and optoelectronic systems.31,61,62 The refractive index (n) and the extinction coefficient (k) have been calculated as follows63–68: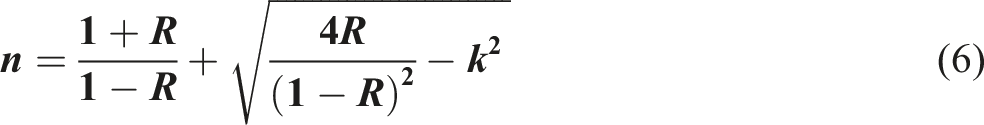

The relation between reflectance (R), refractive index (n), and the extinction coefficient (k) and versus λ plots for the PVA/PVP/Cr2O3 nanocomposites is illustrated in Figure 6. Extinction coefficient plots, as shown in Figure 6(a), were remarkably comparable to absorbance curves in outline. Figure 6(b) shows the reflectance (R) spectra for the PVA/PVP/Cr2O3 films. Furthermore, as shown in Figure 6(a) and 6(c), the extinction coefficient of the PVA/PVP/Cr2O3 films is substantially much less than their refractive indices. It was established that the The relation between (a) k, (b) R, and (c) n versus λ plots for the PVA/PVP/Cr2O3 nanocomposites.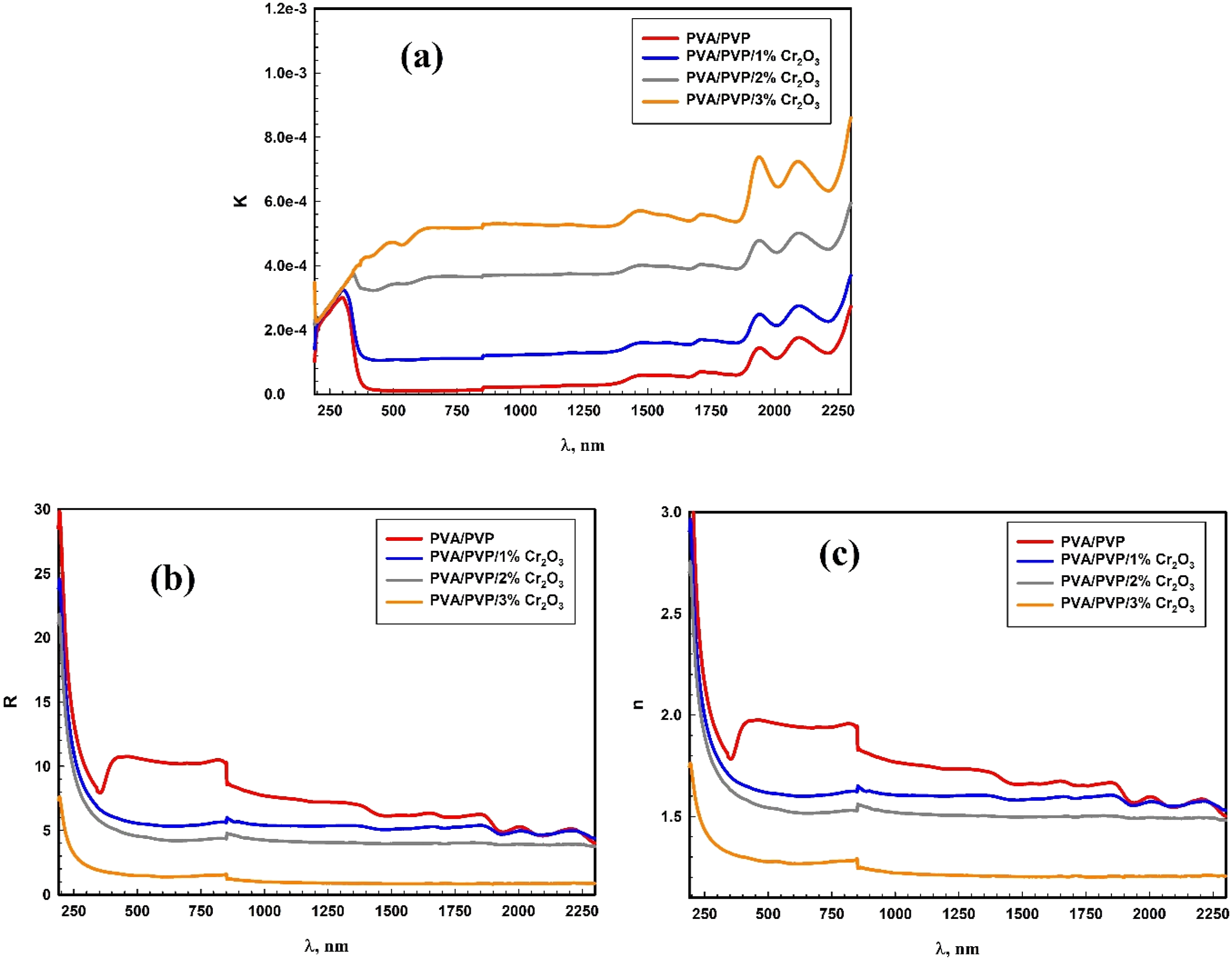
Radiation Shielding
Chemical composition ratios and density of the PVA/PVP/Cr2O3 nanocomposites.
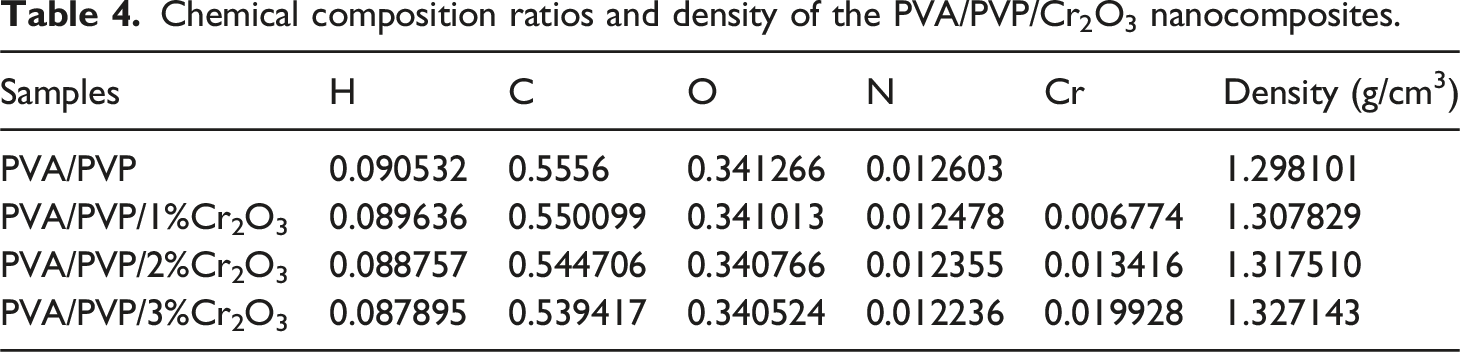
Figure 7 exhibits the variation of MAC with gamma energy for the PVA/PVP/Cr2O3 nanocomposites. The results show that the MAC in the low energy region is greater than at high energy. The MAC increases with the increase in doping with Cr2O3. This is because gamma rays’ interaction with matter depends on their energy and the density of matter, or, in other words, the atomic number of matter. The effect of Cr2O3 is apparent in low gamma energy (<0.1 MeV) because the photoelectric absorption process is dominant. The key players for the probability are strongly dependant on the energy of the gamma rays as well as the atomic number. The effect of Cr2O3 decreases at intermediate energies (>0.1 MeV) because the effect of the photoelectric absorption procedure is reduced, and the Compton scattering is dominant. Herein, the achived probability related to this process depends weakly on the energy of gamma rays as well as the atomic number. It depends on the electron density, which is proportional to Z/A and nearly constant for the PVA/PVP/Cr2O3 nanocomposites. The effect of Cr2O3 decreases at high energies, where the pair production is the most dominant interaction process. The probability of pair production varies approximately as the square of the atomic number Z and differs slightly.73,74 Table S1 shows the mass attenuation coefficient (μ/ρ) (cm2/g) for the PVA/PVP/Cr2O3 nanocomposites at various energies. The change of mass attenuation coefficient (MAC) for the PVA/PVP/Cr2O3 nanocomposites.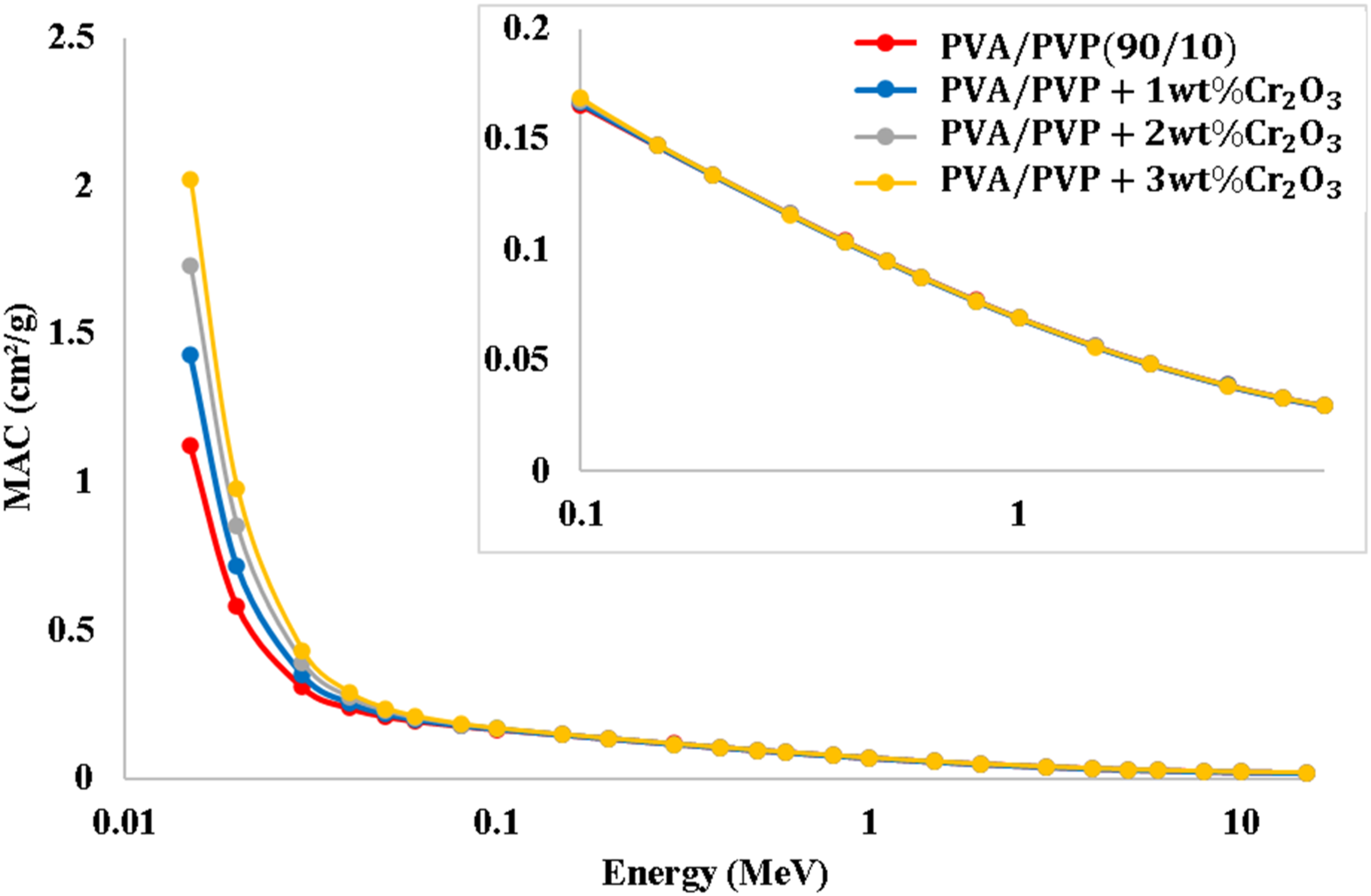
Figure 8 shows the variation of LAC for the PVA/PVP/Cr2O3 nanocomposites with gamma energy. The LAC shows the same behavior as the MAC. Additionally, the half-value layer (HVL) represents the utmost frequently employed factor for explaining the penetration capability of the various types of radaitions or specific radiations as well as the penetration through specific objects. The change of linear attenuation coefficient (LAC) for the PVA/PVP/Cr2O3 nanocomposite films.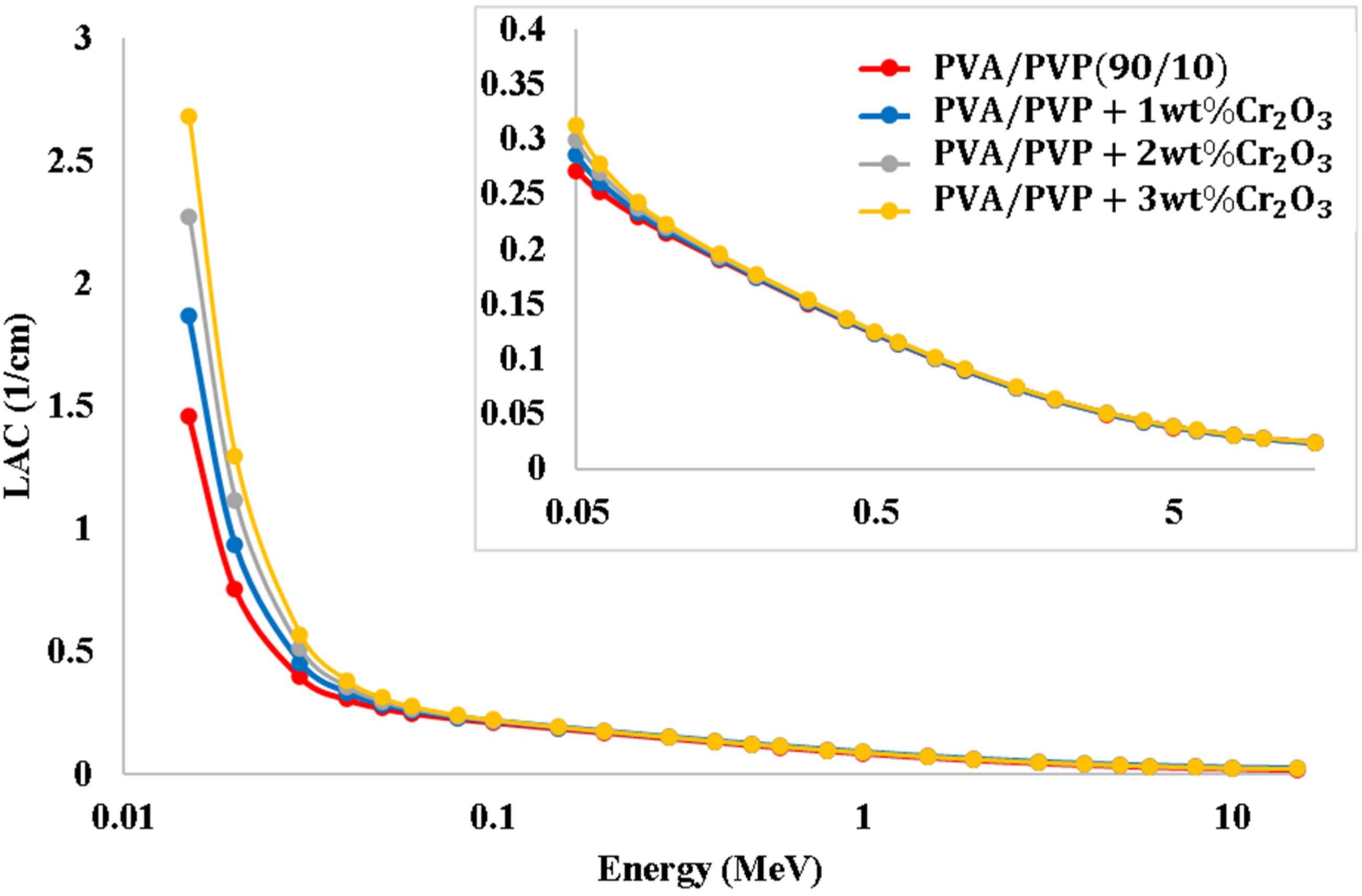
Figure 9 (a) shows the change in HVL for the PVA/PVP/Cr2O3 nanocomposites with photon energy. In this figure, the HVL decreases slightly with an increase in the Cr2O3 NPs ratio and increases with an rise in the photon energy. A value of HVL changed from 0.475 cm to 29.137 cm for PVA/PVP at 0.015 MeV and 15 MeV, respectively. The HVL value changed from 0.371 cm to 28.802 cm for the PVA/PVP/1%Cr2O3 at 0.015 MeV and 15 MeV, respectively. Also, the HVL value changed from 0.305 cm to 28.478 cm and from 0.259 cm to 28.161 cm for the PVA/PVP/2%Cr2O3 and PVA/PVP/3%Cr2O3, respectively, at 0.015 MeV and 15 MeV. The change of the tenth value layer (TVL) and mean free path (MFP) for the PVA/PVP/Cr2O3 nanocomposites is shown in Figure 9(b) and (c). It should be noted that, the diffences in TVL as well as MFP with the energy has the same trend as that detected for the interpretation of HVL. (a) The change of the HVL, (b) TVL, and (c) MFP values for the PVA/PVP/Cr2O3 nanocomposites.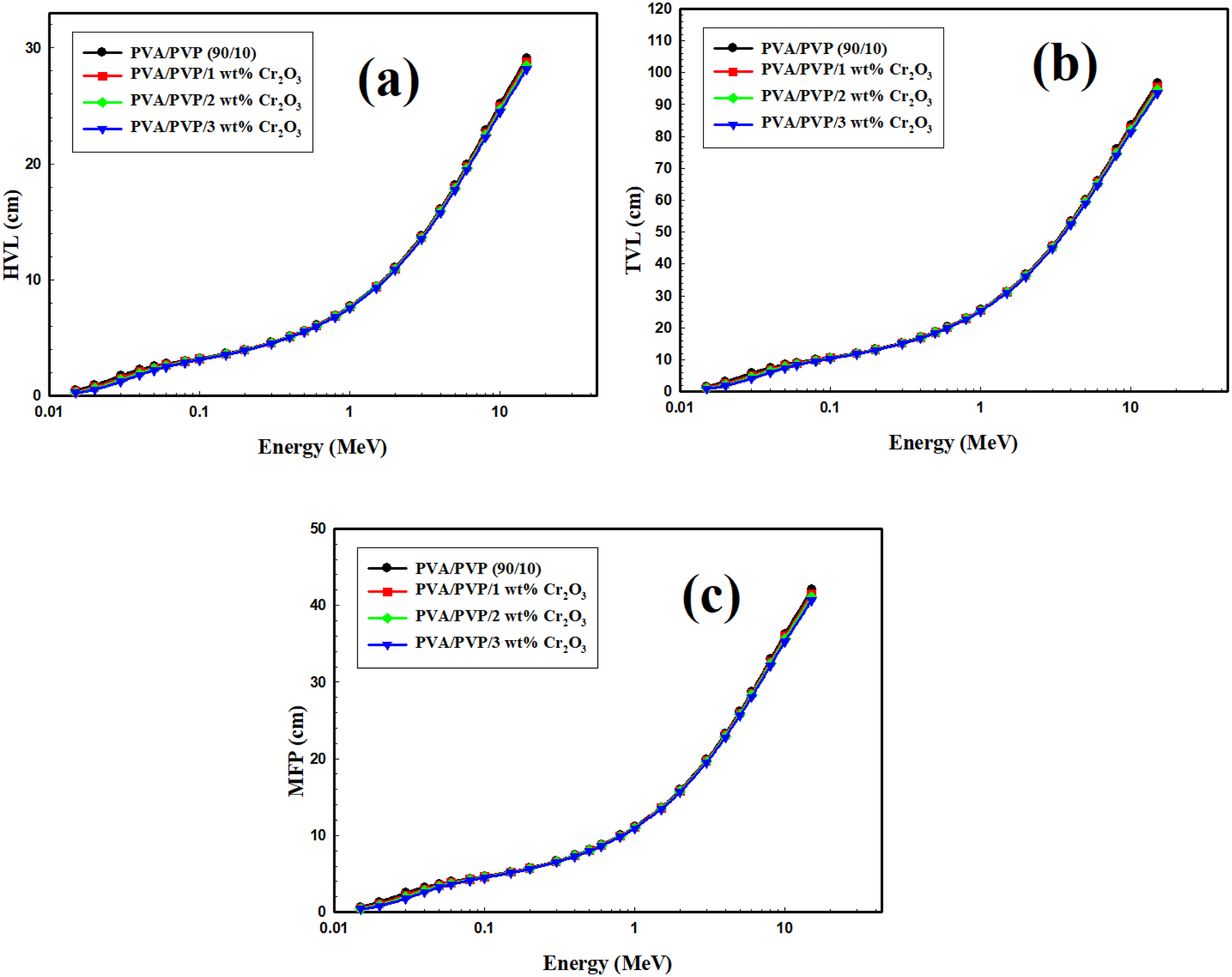
Figure 10 illustrates the Zeff and Neff variation for the PVA/PVP/Cr2O3 nanocomposites with different weight percentages of Cr2O3 subjected to gamma-ray radiation. The mass attenuation coefficients of the compound, the elements, and the corresponding proportions significantly influence the variation in Zeff and Neff. Therefore, the Zeff and Neff escalated with increased Cr2O3 into PVA/PVP, indicating a rise in the mass attenuation coefficient. These results validate the radiation-shielding potential of the PVA/PVP/Cr2O3 nanocomposites. The variation of (a) Zeff and (b) Neff for the PVA/PVP/Cr2O3 nanocomposites.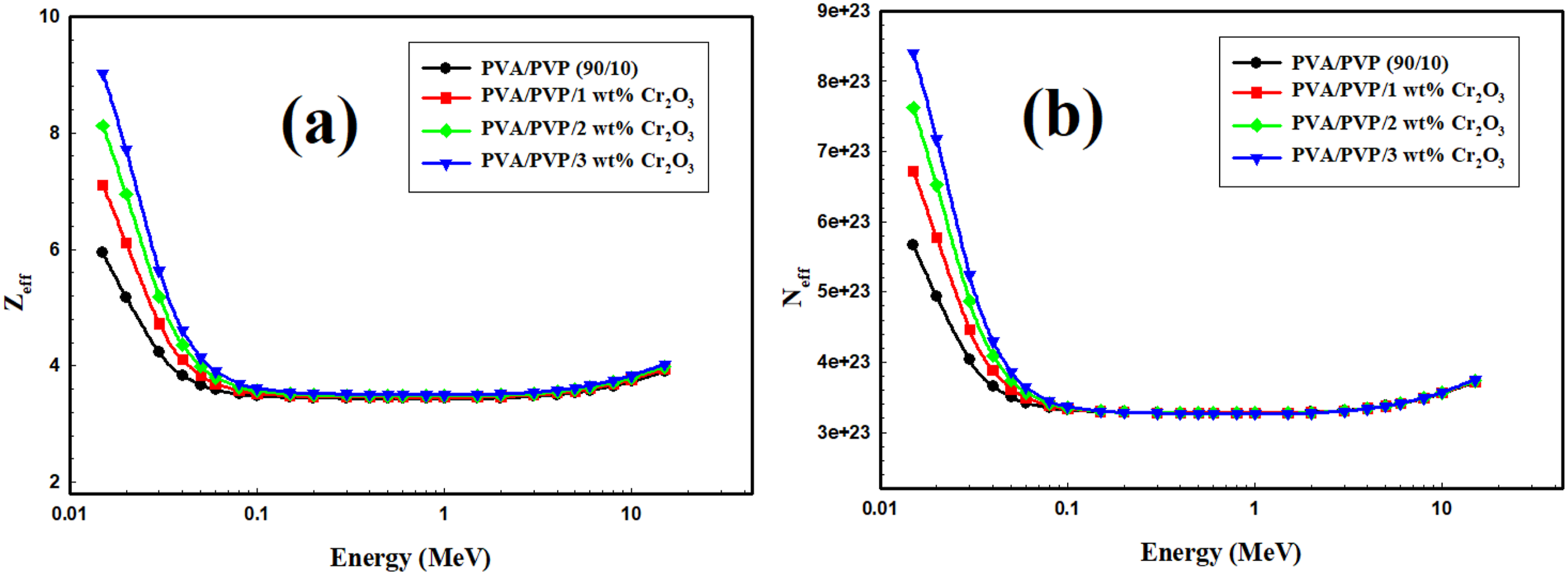
Figure 11 illustrates the exposure buildup factor (EBF) of the PVA/PVP/Cr2O3 nanocomposites as a function of photon energy and the films’ thickness in mean free path (MFP). The EBFs generally have small values across the higher and lower energies, with a peak appearing at middle energies. Furthermore, due to the predominance of the photoelectric effect (PE) at lower energies, many photons ultimately dissipate or deplete all of their energy. This results in a decrease in the number of photons building up. Compton scattering (CS) is a primary component of intermediate energy levels.
75
The exposure buildup factor (EBF) of the PVA/PVP/Cr2O3 nanocomposites as a function of photon energy.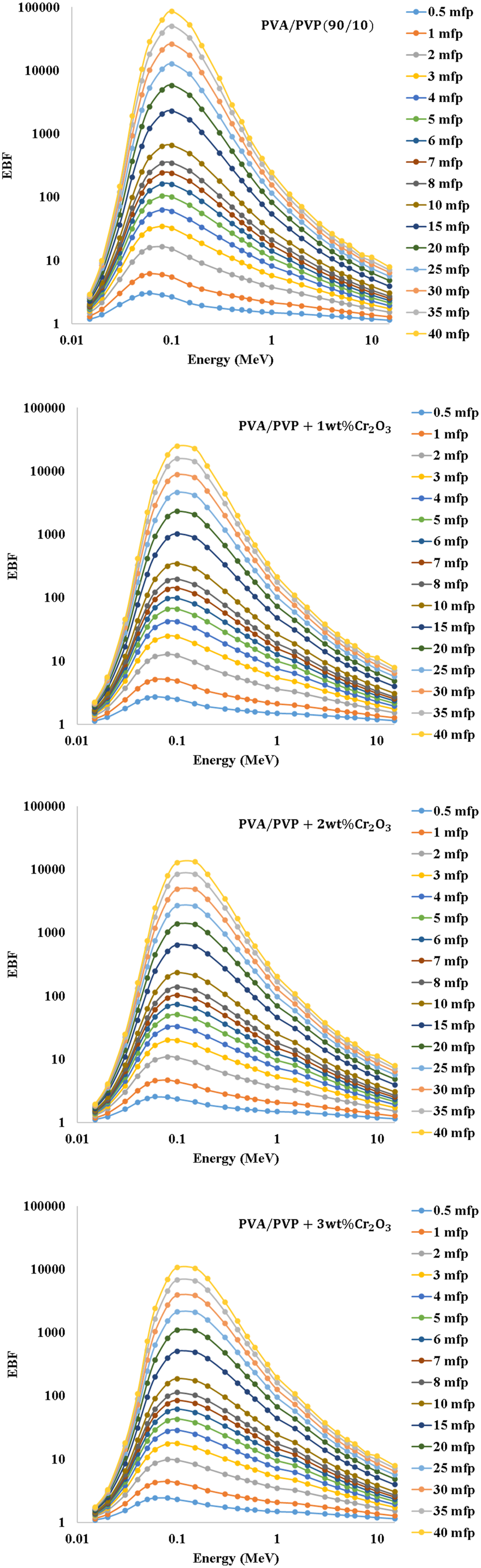
The estimated equivalent atomic number (Zeq) for PVA/PVP/Cr2O3 nanocomposites, with differing quantities of Cr2O3, was presented in Figure 12. The equivalent atomic number reaches the peak values of PVA/PVP/Cr2O3 nanocomposites when the photoelectric effect (PE) predominates. The estimated Zeq of PVA/PVP/Cr2O3 nanocomposites.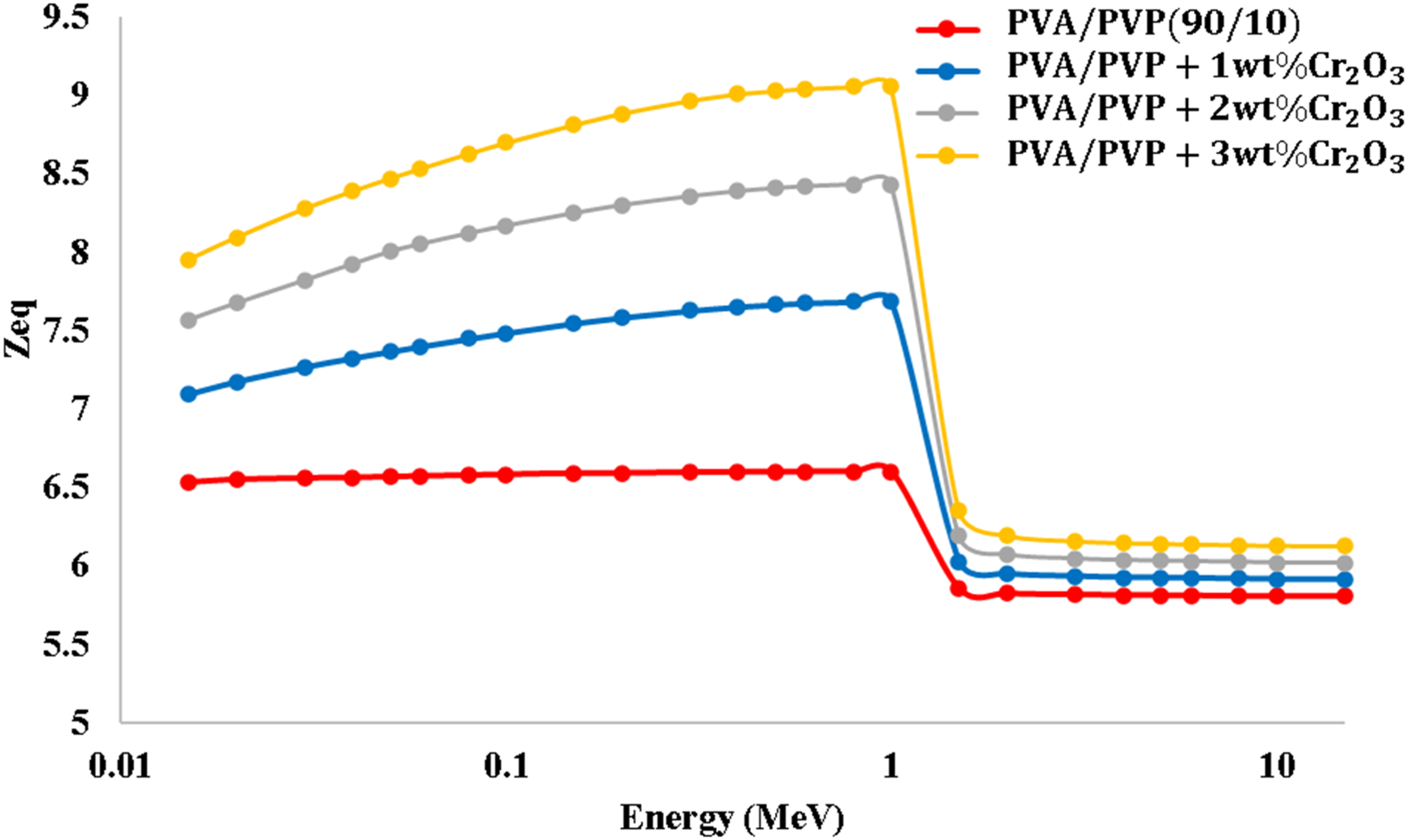
Conclusion
This work investigates the optical characteristics and radiation shielding properties of a PVP/PVA mixture loaded with varying ratios of Cr2O3 nanoparticles (0%, 1%, 2%, and 3%). XRD of the synthesized powder Cr2O3 NPs exhibited a crystallite size of 42 nm. The PVA/PVP film’s Eg value was 3.51 eV; with the addition of 3% wt Cr2O3 NPs, it was lowered to 1.97 eV. The PVA/PVP blend exhibits an Eu value of 1.42 eV, which increases progressively to 3.98 eV when the Cr2O3 NPs concentration in the PVA/PVP rises to 3% wt. In addition, the influence of the concentration of Cr2O3 NPs on the radiation shielding properties of PVA/PVP/Cr2O3 nanocomposites has been theoretically estimated using the Phy-X program and the XCOM database. The value of HVL changed from 0.475 cm to 29.137 cm for PVA/PVP at 0.015 MeV and 15 MeV, respectively. The HVL value changed from 0.371 cm to 28.802 cm for the PVA/PVP/1%Cr2O3 at 0.015 MeV and 15 MeV, respectively. Also, the HVL value changed from 0.305 cm to 28.478 cm and from 0.259 cm to 28.161 cm for the PVA/PVP/2% Cr2O3 and PVA/PVP/3%Cr2O3, respectively, at 0.015 MeV and 15 MeV. The exposure buildup factor (EBF) of the PVA/PVP/Cr2O3 nanocomposites as a function of photon energy has been studied. The equivalent atomic number (Zeq) values for PVA/PVP/Cr2O3 nanocomposites have also been estimated. It should be noted that the obtained experimental and theoretical results confirm the ability for employing such materials in various optical applications and applications besides their capability for used them as radiation shielders.
Supplemental Material
Supplemental Material - Correlated structural, optical, and radiation shielding characteristics of PVA/PVP/Cr2O3 nanocomposites
Supplemental Material for Correlated structural, optical, and radiation shielding characteristics of PVA/PVP/Cr2O3 nanocomposites by B. Alayed, A. Z. Mahmoud, Mohammed O. Alziyadi, Asma Alkabsh, Amani Alruwaili, M. S. Shalaby in Journal of Thermoplastic Composite Materials
Footnotes
Acknowledgements
The authors extend their appreciation to the Deanship of Scientific Research at the Northern Border University, Arar, KSA, for funding this research work through the project number NBU-FFR-2024-885-08.
Author contributions
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Deanship of Scientific Research at the Northern Border University, Arar, KSA, for funding this research work through the project number NBU-FFR-2024-885-08.
Data Availability Statement
All data generated or analysed during this study are included in this published article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
