Abstract
In this study, to solve the problem of poor interfacial bonding between carbon fiber (CF) and fluororubber (FKM), the polyacrylonitrile (PAN) coating and high-temperature carbonizing method were proposed to modify the CF. In this method, the PAN nanospheres were compounded with carbon fiber by coating process, and then melted and carbonized to a thin carbon layer attached to the CF surface during the high-temperature carbonizing process. The rough and irregular PAN carbonized layer on C-PAN-CF can greatly enhance the mechanical performance of fluoroelastomer by mechanical riveting effect. The tensile strength, modulus at 100% strain and hardness of FKM/C-PAN-CF improved by 122.0%, 123.5% and 29.2%, respectively. And the compression set reduced to 29.5%. Furthermore, the C-PAN-CF could also raise the FKM’s thermal stability, thermal management performance and Electromagnetic shielding performance (EMI SE). Particularly in the X-band, the composites’ shielding mechanism was mainly based on reflection. In summary, such a simple and effective technique for preparing C-PAN-CF hybrid fibers provides a certain reference for the preparation of reinforcing fillers for high-performance FKM composites.
Keywords
Highlights
1. C-PAN-CF fiber was prepared by PAN coating and high-temperature carbonization. 2. The C-PAN-CF surface was covered by a thin PAN carbonized layer. 3. The C-PAN-CF can greatly enhance the mechanical performance of FKM. 4. The tensile strength and modulus at 100% strain improved by 122.0% and 123.5%. 5. C-PAN-CF could also raise FKM’s thermal performance and EMI SE in X band.
Introduction
Fluororubber are widely used in cutting-edge applications such as aerospace, oil extraction, transportation, medical materials, and semiconductor packaging due to their remarkable resistance to corrosion, high temperatures and oil. 1 However, it still suffers from poor mechanical, thermal, and electrical properties. To overcome these problems, researchers usually use carbon nanofiller doping modification strategies to strengthen and enhance FKM. 2
With high strength, high modulus, low specific gravity, and excellent thermal and electrical conductivity, CF is an ideal reinforcing filler for FKM elastomer.3–5 But its low affinity, smooth and chemically inert surface usually results in weak interfacial interaction between FKM and reinforcing CF, which limits its enhancement effect.6,7 Moreover, interfacial connection performance is widely recognized for their critical role in regulating load transfer between fibers and matrix, as well as influencing the strength and toughness displayed by composite materials. To solve these issues, it is necessary to modify the CF surface to enhance the composites’ interfacial bonding. 8
Currently, there are two typical surface modification mechanism for CF. One mechanism is to increase the surface roughness of CF to facilitate mechanical riveting with FKM, resulting in a good interfacial bond between CF and FKM. The other one is to chemically graft a large number of active functional groups, typically hydroxyl, carboxyl, amino, etc., onto the surface of CF, resulting in a chemical bonding with FKM. 9 So far, researchers both domestically and internationally have employed chemical grafting, plasma treating, coating, and other modification techniques to modify CF according to the above two CF modification mechanism. For example, Gao et al. 10 preoxidized CF and then grafted them using polyelectrolyte polyethyleneimine and trimethoxy (1H,1H,2H,2H-heptadecafluorodecyl) silane, resulting in a 173% improvement in tensile strength. Wang et al. 11 grafted CF with 4,4-methylenediphenyl diisocyanate coupling agent, which significantly enhanced the silicone rubber’s tensile strength (0.62 MPa) and elongation at break (92.85%). However, liquid contamination and fiber damage in the above cases is inevitable in chemical grafting treatment. Gravis et al. 12 modified CF with O2 plasma treatment, which introduced acidic (COO) and ether (C-O-C) groups to its surface, and showed the excellent potential for enhancing the rubber’s mechanical properties. However, the plasma modified CF need to be immediately compounded with FKM because they are prone to lose their surface functional groups when exposed to air. Gu et al. 13 coated CF with dopamine, and the composites demonstrated an improvement in tensile strength and elongation at break of 61.7% and 68.4%, respectively. The coating method are simple and cost-effective techniques for modifying CF,14–16 but the interface connection between CF and coating was mainly via weak Van der waals’ force, which also limits its application. Given the above modification method still have so many limitations, so a new method that is simple and effective method for CF modification is urgently needed.
The high-temperature carbonizing method is an effective and simple method to form a uniform carbon layer on the CF surface after polymer coating treatment. So far, there is no research report on the modification of carbon fiber by coating method and high temperature carbonization method. In addition, there is no report on filling modified FKM with chopped CF as filler. In this paper, carbon fibers were coated with PAN and then carbonized at high temperature. The formed nanorod-like structured PAN carbonized layer on CF surface could increase CF’s diameter and surface roughness. After compounding with FKM, the rough surface of C-PAN-CF could enhance its mechanical riveting with FKM and resulting in excellent mechanical performance. In addition, the morphology and surface chemical composition of CF, and the mechanical, thermal and electromagnetic shielding performance of FKM composites were systematically investigated.
Experimental
Materials
FKM with a fluorine content of 70% was purchased from DuPont (Wilmington, DE, USA). Chopped CF (T800) with the lengths of 1 mm was sourced from Toray Industries (Tokyo, Japan), and removed the sizing in acetone at 80°C for 24 h prior to use. 2,5-Dimethyl-2,5-bis (tert-butylperoxy) hexane (Triceratops®101-50D) was obtained from Yuyao Mingri Chemical Co Ltd (Yuyao, China). Triallyl isocyanurate (TAIC) was supplied by Rhein Chemie (Qingdao) Co. Polyacrylonitrile (PAN) and methylene dichloride (CH2Cl2) was facilitated by Shanghai Jizi Biochemistry Technology Co Ltd (Shanghai, China). Sulfuric acid (sulfuric acid, purity 95%∼98%) and nitric acid (nitric acid, purity 65%∼68%) were produced by Zhejiang Zhongxing Chemical Reagent Co. Ltd (Lingzhi, China).
Preparation of acidized carbon fibers
A specific amount of CF was introduced into a mixture of sulfuric acid and nitric acid (3/1, v/v), and the mixture was magnetically stirred at 70°C for 6 h. Then the deionized (DI) water was added to the mixed solution to dilute it until the pH was higher than 3. Next, the mixed solution was filtered and the obtained acidized CF was repeatedly washed with DI water until the filtrate became neutral. Finally, the acidized CF were filtrated and dried in an oven at 80°C.
Preparation of PAN-CF and C-PAN-CF
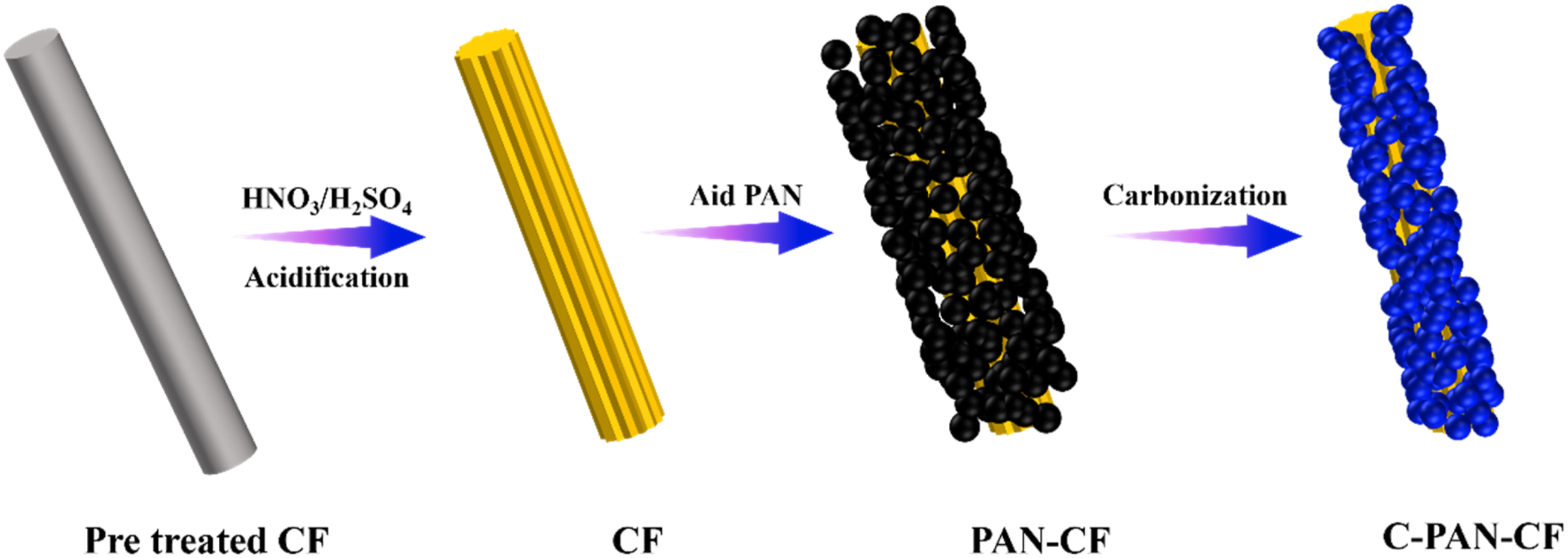
Dissolving 4 g of PAN in 200 mL of CH2Cl2 and thoroughly stirring, then 2 g of acidified CF was added. After ultrasonic dispersion for 20 min and dried by oven drying until the quality did not change, the PAN-coated acidified CFs were obtained and coded as PAN-CF. Subsequently, parts of PAN-CF were placed in a tube furnace and burned at 500°C for 40 min under a nitrogen atmosphere. Finally, the carbon layer coated acidified CF was obtained and coded as C-PAN-CF. The detailed manufacturing process is shown in Figure 1. And the schematic diagram of CF modification is shown in Figure 2. Flow chart for the preparation of C-PAN-CF. Schematic diagram of CF modification.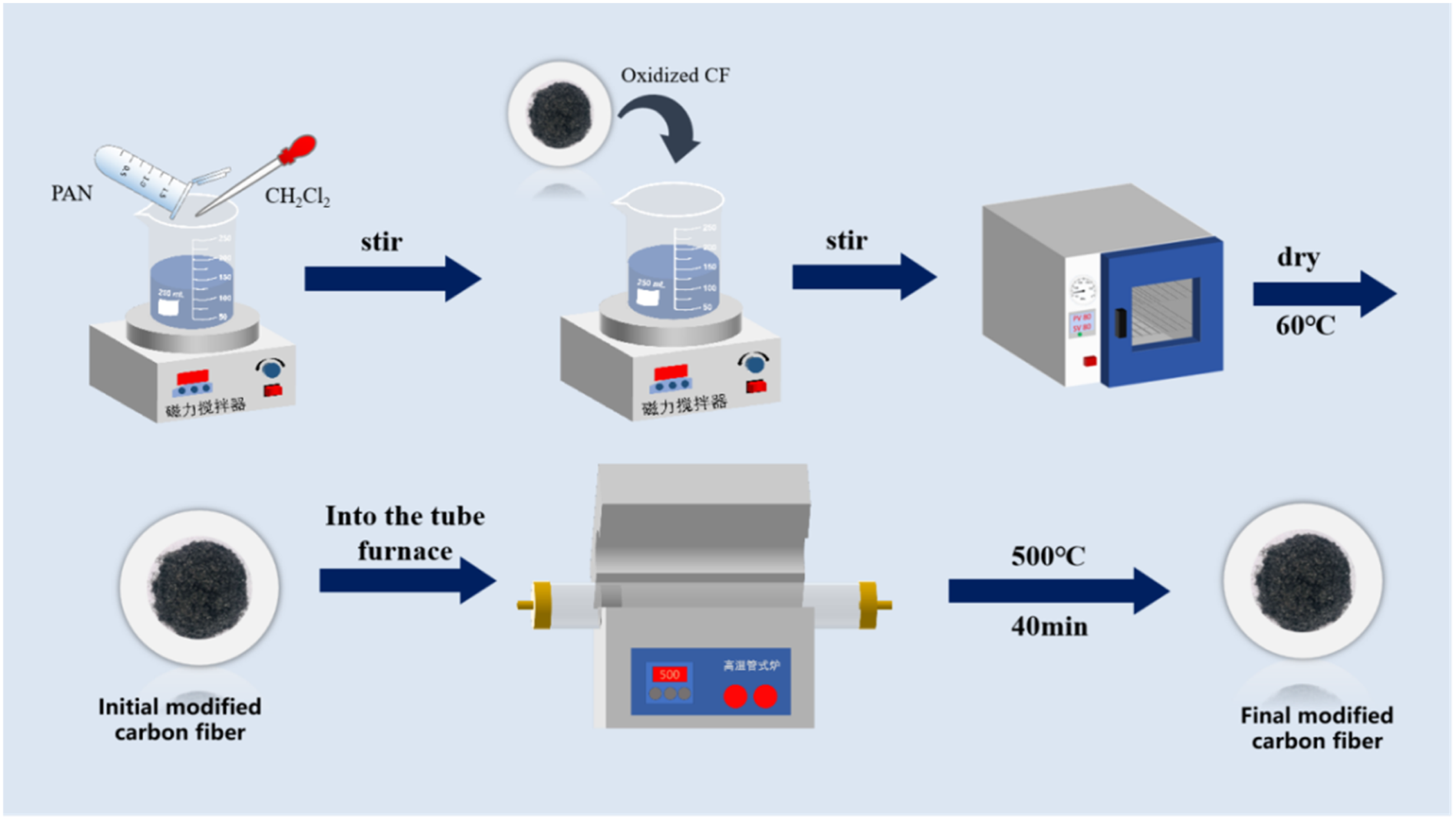
Preparation of various FKM composites
In order to ensure uniform dispersion of C-PAN-CF in FKM, firstly, the pristine FKM was added to the mixer with a roll temperature of 50°C, a roller speed of 20 r/min and a roller distance of 1 mm. Then the C-PAN-CFwas added evenly. After the FKM was rolled and cut three times on the open mill, the TAIC and Triceratops®101-50D were were introduced in sequence, then rolled and cut for six times on the open mill until it became homogenized. The one-stage-curing FKM composite were obtained by press molding with a flat vulcanizer (170°C, 10 MPa, 10 min). After post-curing treatment in a constant temperature oven at 200°C for 24 h,
17
the FKM/C-PAN-CF composite were obtained. The related preparation process is shown in Figure 3. As a comparison, the FKM, FKM/CF, FKM/PAN-CF and FKM/C-PAN-CF composites were also produced following the same procedure. The specific formulas for these composites are listed in Table 1. Schematic diagram of FKM composite’s preparation process. Raw material composition of different FKM composites.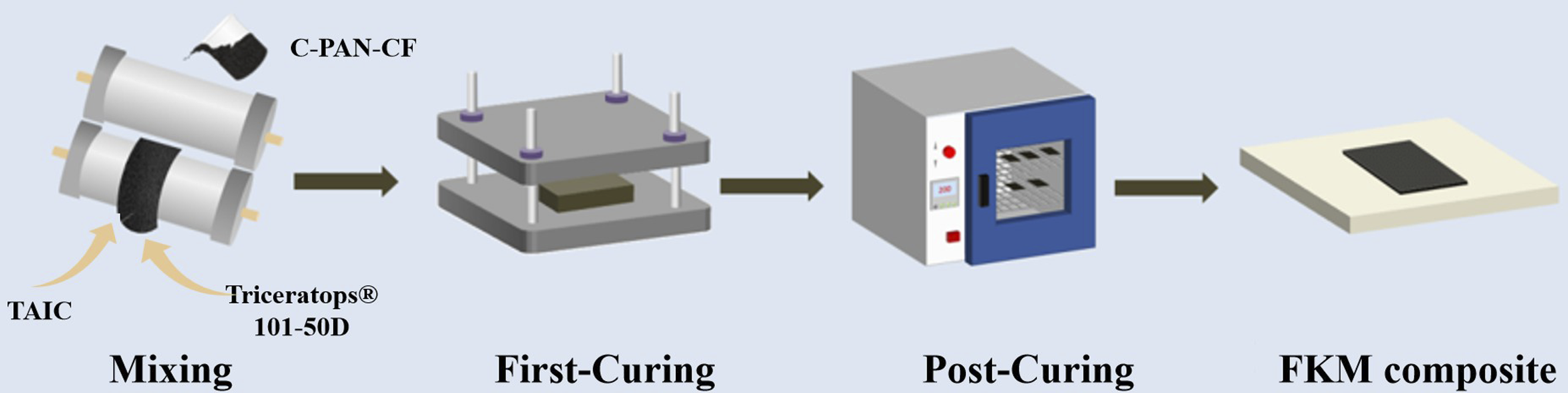
Characterization
The morphology of modified CFs and various FKM composites’ fracture surfaces was characterized by scanning electron microscopy (SEM, Sigma300, ZEISS, Germany) with an acceleration voltage of 5 kV.
The chemical composition of the CFs before and after modification was analyzed by the X-ray photoelectron spectroscopy (XPS, Thermo Fisher, ESCALAB 250Xi, USA).
The tensile characteristics of various FKM composite were evaluated by using a universal testing machine (AI-7000-LA, GOTECH, China) following ISO 37:2005 standard with the crosshead speed was 200 mm/min. Dumbbell-shaped samples with a thickness of 2 mm and a total length of 50 mm were used for the test, and the average value of five samples was taken as the test result.
Additionally, the hardness was determined by using a sclerometer (HTS-800A, YZJINGMI, China) following the ISO 7619 standard. The thickness of the test sample is 6 mm, the testing time is 3 s and the result is the average of five samples.
According to the ASTM D395 standard, a compression permanent deformer was used to characterize the compression set. The test was conducted at a temperature of 200°C and lasted for 70 h. And the pre-compression ratio is set to 25%, the dimension inner diameter of O-ring is 24.99 mm and the line diameter is 3.53 mm.
The electrical conductivity of various FKM composites was assessed by a precision digital ultra-high resistance microcurrent-measuring instrument (EST121, EST, Beijing, China). To enhance the reliability of the results, each test was conducted on five specimens, and the average value was recorded.
The thermal stability of various FKM composites was evaluated by using a Thermogravimetric Analyzer (Pyris 1 TGA, PerkinElmer, Waltham, MA, USA) under a nitrogen atmosphere with a scan rate of 10°C/min, and a maximum temperature of 850°C.
The thermal management performance of various FKM composites was measured by an infrared camera (Model HM-TP7WL-HB, HIKMICRO, China) and a precision stainless steel hot plate (DB-1AB, Shanghai Kuntian, China). In the process of heating or cooling, samples with a size of 10 mm × 10 mm × 2 mm are placed on the hot plate, and their infrared images are recorded by pyrometer.
The electromagnetic shielding performance of various FKM composites was tested by using an network analyzer (VNA, N5232 A, Agilent, USA) in X band with the specimen dimensions of 22.9 mm × 10.2 mm.
The hydrophobicity of various FKM composites was measured with a contact angle meter (LSA100, LAUDA Scientific, Germany), using 5.0 μL DI water droplets.
The transmittance and haze of various FKM composites were tested by using a high-precision spectral haze meter (TH-100, CHN Spet, China) with a light source of D65.
Results and discussion
Surface morphology analysis
The morphology and micro-structure of CF, PAN-CF, and C-PAN-CF were examined by SEM, and the results are displayed in Figure 4(a)-(c). From Figure 4(a), the surface of pristine CF was clean and smooth. After compounding with PAN, large number of the PAN microspheres with a diameter of 30⁓60 μm appeared, and rarely adhere to the CF surface (Figure 4(b)). However, as shown in Figure 4(c), after high temperature carbonizing treatment, the CF surface was covered by a thin PAN carbonized layer and formed a novel nanorod-like structure, which increased the diameter of CF and made the fiber surface become rough. In addition, the rough surface of C-PAN-CF with nanorod structure signify strong mechanical riveting ability and excellent interface bonding strength.
17
The SEM images of various CF: (a) CF, (b) PAN-CF, and (c) C-PAN-CF.
Chemical composition analysis
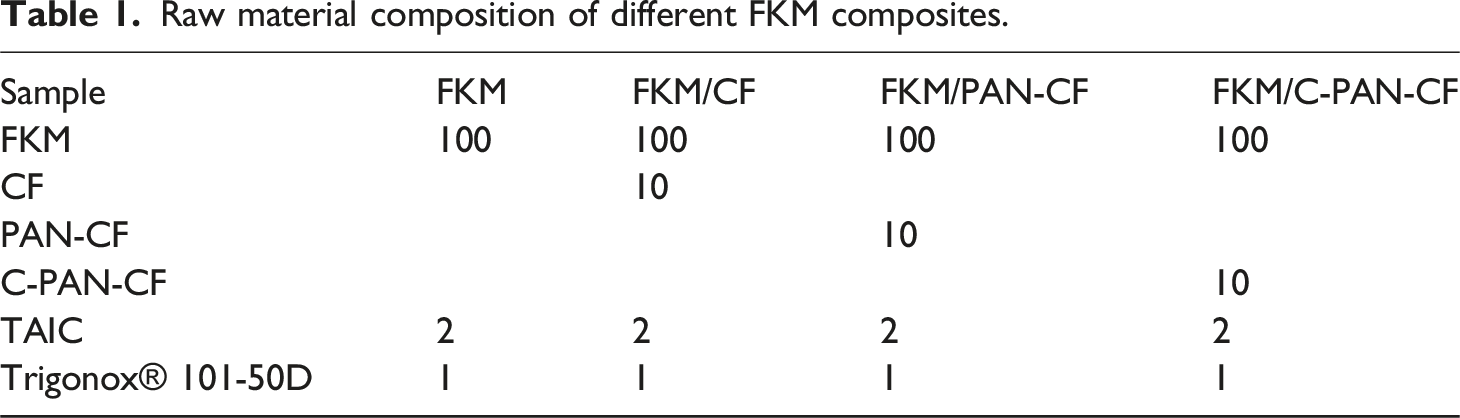
To obtain a deep understanding of the changes in the CF after modification treatment, the XPS was used to scrutinize the chemical composition of CF, PAN-CF and C-PAN-CF. As Figure 5(a)-(c) illustrates, for CF, only two peaks were found, which represent C1s and O1s. However, for PAN-CF, a new N1s peak appeared at 400 eV, which proved that the successful introduction of PAN. For C-PAN-CF, the N1s peak weakened, indicating that the PAN was successfully carbonized. Wide-scan XPS spectra and C1s high-resolution XPS element spectra of (a, d) CF, (b, e) PAN-CF, (c, f) C-PAN-CF.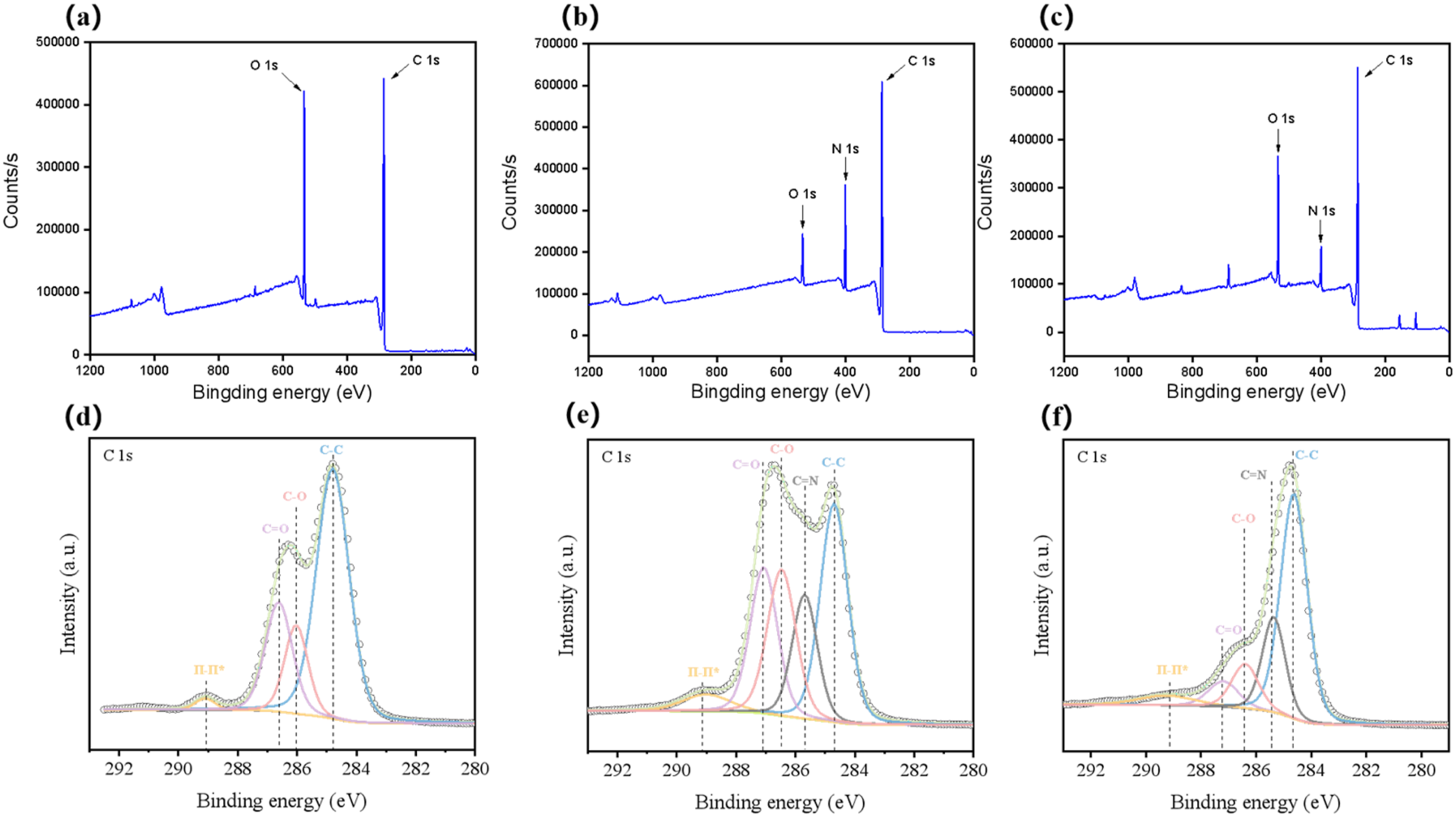
To quantify analyze chemical environment of carbon atom, the high resolution C1s spectra was performed and the results were displayed in Figure 5(d)-(f). For CF, three peaks were found, which stand for C = O (286.3 eV), C-C (284.5 eV), and C-O (286 eV). When PAN was introduced, an extra peak appeared at 285.7 eV.18–22 After high temperature carbonizing treatment, the percentage content of C-O, C = O and C = N in C-PAN-CF was reduced compared to PAN-CF, indicating that the high temperature carbonizing treatment was successful.
As can be seen from the Figure 6, the peak intensity of CF at low angle (in the range of 10°–30°) is relatively low, indicating that its graphitization degree or crystal order degree is relatively limited, and the peak near 25.2° is the diffraction peak of graphite (002) crystal face, with a wide peak type, which reflects that the grain size of carbon fiber is relatively small. PAN is a typical organic polymer, and its structure is amorphous or low crystallinity. PAN is a typical organic polymer with amorphous or low crystallinity. After doping CF, the curve in the figure has an obvious broad peak at 17°, corresponding to the (100) crystal plane of PAN. The peak near 25.2° is the graphite phase diffraction peak of CF, but its intensity is different from that of pure CF, indicating that the doping of PAN has changed the crystal environment of CF. High-temperature carbonization will lead to pyrolysis and graphitization of PAN and interface fusion with CF, and its XRD characteristics are as follows: The overall peak intensity is significantly increased, indicating that the crystal order is greatly improved, and the diffraction peak pattern of (002) crystal plane near 25.2° is sharper and higher in intensity, reflecting the increase of grain size and graphitization degree. Compared with PAN-CF, the amorphous broad peak disappears. It is proved that high temperature carbonization can completely transform PAN from organic polymer into graphite-like carbon phase, and further fuse with the crystal structure of carbon fiber to form a more unified carbon-based crystal system. XRD of CF, PAN-CF, C-PAN-CF.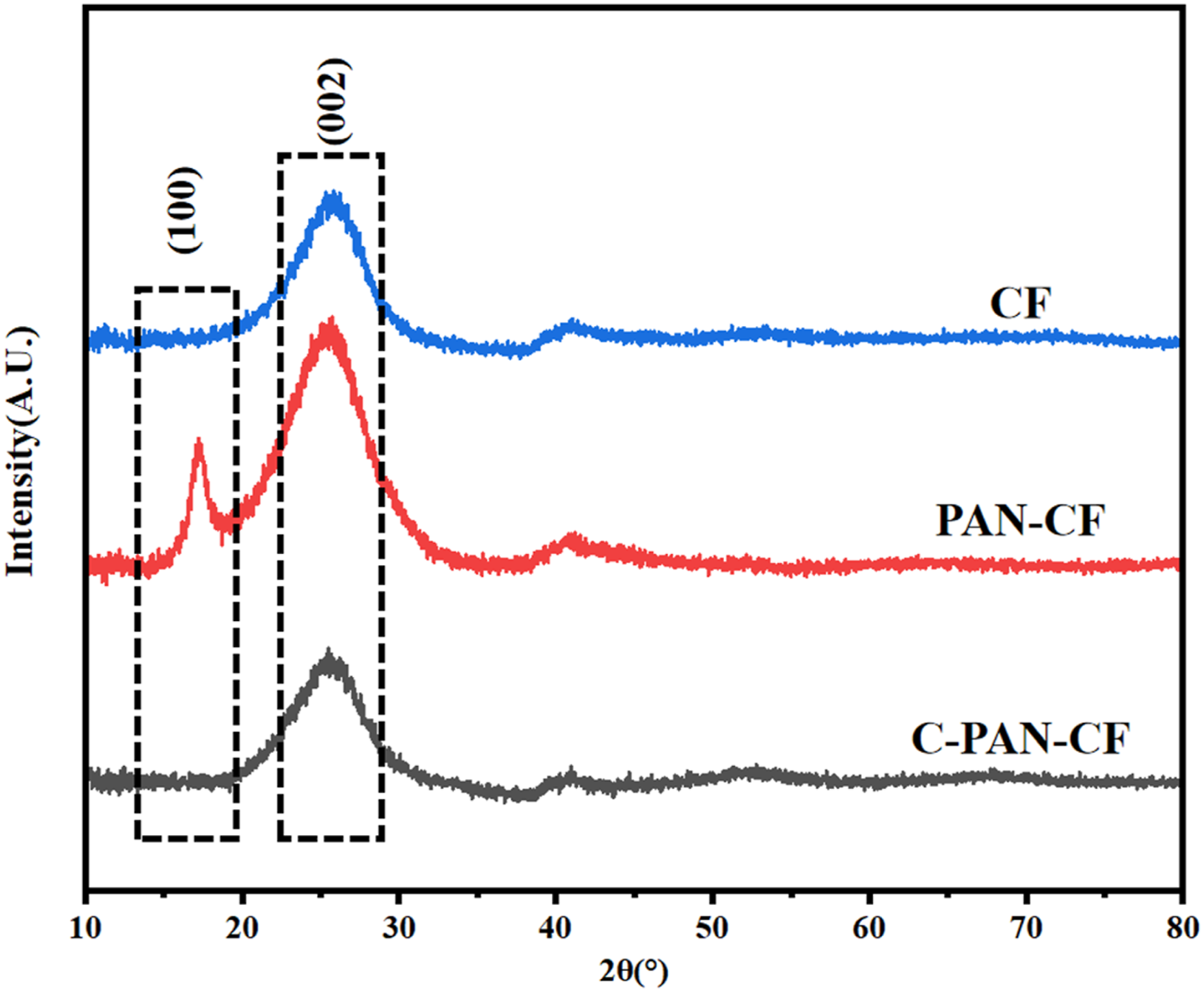
Mechanical properties analysis
To investigate the effect of fiber modification on the mechanical properties of various FKM composite, the tensile properties, hardness and compression set was tested, and the results were demonstrated in Figure 7. From Figure 7(a)-(d), it can be seen that the mechanical properties of FKM can be considerably enhanced by the addition of modified CF. For example, the original FKM exhibits the lowest tensile strength and modulus at 100% strain. After addition of CF, the tensile strength and modulus at 100% strain of FKM/CF were sharply enhanced by 57.6% and 41.6%, respectively. This notable improvement can be ascribed to CF’s innate exceptional mechanical strength, which gives the FKM composites a robust “supporting framework” and strengthens the mechanical properties of the materials as a whole.23,24 When addition of PAN-CF, the tensile strength and modulus at 100% strain of FKM/PAN-CF were further enhanced, which was due to the PAN micro-spheres and CF filler’s synergistic enhancement effect. However, when modifying the FKM with C-PAN-CF, the tensile strength and modulus at 100% strain of FKM/C-PAN-CF were the highest, which achieved an increment of 122.0% and 123.5%, respectively. The reason was that the irregular PAN’s carbon layer on C-PAN-CF improves its surface roughness and strengthens mechanical riveting with FKM matrix.
25
Furthermore, a three-dimensional network structure developed between the rubber matrix and the filler, also significantly enhancing the composites’ mechanical properties. Mechanical properties of different FKM composites: (a) stress-strain curve, (b) tensile test process, (c) tensile strength, (d) modulus at 100% strain, (e) hardness, (f) compression set.
Figure 7(e) displays the hardness of several FKM composites. It is evident that adding CF, PAN-CF and C-PAN-CF could greatly raise the hardness of FKM. For FKM/C-PAN-CF composite, the hardness improved by 29.2% compared with the original FKM, which was due to the high hardness of modified CF itself and the good mechanical riveting with FKM matrix, which absorb energy under external stresses. However, for FKM/CF, FKM/PAN-CF and FKM/C-PAN-CF composites, the hardness didn’t show big difference.
Furthermore, the impact of CF modification on FKM composites’ compression set was also examined, and the outcomes were displayed in Figure 7(f). It is evident that adding CF, PAN-CF, and C-PAN-CF can greatly increase FKM’s capacity to withstand compression, resulting in outstanding resistance to compression deformation. The possible explanation for this could be that the addition of CF, PAN-CF, and C-PAN-CF can create a three-dimensional network inside the FKM 26 and raise its cross-linking density, which prevents the rubber molecular chains from sliding relative to one another and makes it more difficult to cause permanent deformation when subjected to outside forces.
To further prove the tensile properties enhancing function of modified CF, the fracture surface morphology of various FKM composites’ tensile specimens was examined by SEM, and the images was shown in Figure 8. The comparatively smooth fracture surface of FKM, as seen in Figure 8(a)), suggests that the tensile strength of FKM rubber was low. However, the composites’ fracture surfaces became rough and fibers were pulled out when CF, PAN-CF, and C-PAN-CF were added to the FKM matrix. The spherical material in Figure 8(c) is PAN, which is separated from CF and can strengthen the interfacial forces between the filler and the rubber matrix. In addition, the relatively smooth surface of CF. as shown in Figure 8(b) indicates the existence of weak van der Waals forces and poor interfacial interaction between the carbon fiber and FKM. As shown in Figure 8(d), the surface of CF is covered with PAN carbonized layer, forming a unique rough structure, which significantly enhances the mechanical riveting between fiber and matrix.
27
At the same time, a large number of fibers can be observed on the fracture surface instead of pulling out, which indicates that a strong interface bond is formed between C-PAN-CF and FKM matrix, which effectively promotes stress transfer, thus significantly improving the tensile properties of the composites. These morphologies are highly consistent with the test results of mechanical properties of materials, which further proves that the introduction of C-PAN-CF can significantly improve the tensile properties of FKM composites by improving the interface bonding and enhancing the stress transfer efficiency (Figure 8(e)). SEM images of the (a) FKM, (b) FKM/CF, (c) FKM/PAN-CF, (d) FKM/C-PAN-CF composites and (e) Strengthening mechanisms of FKM/C-PAN-CF.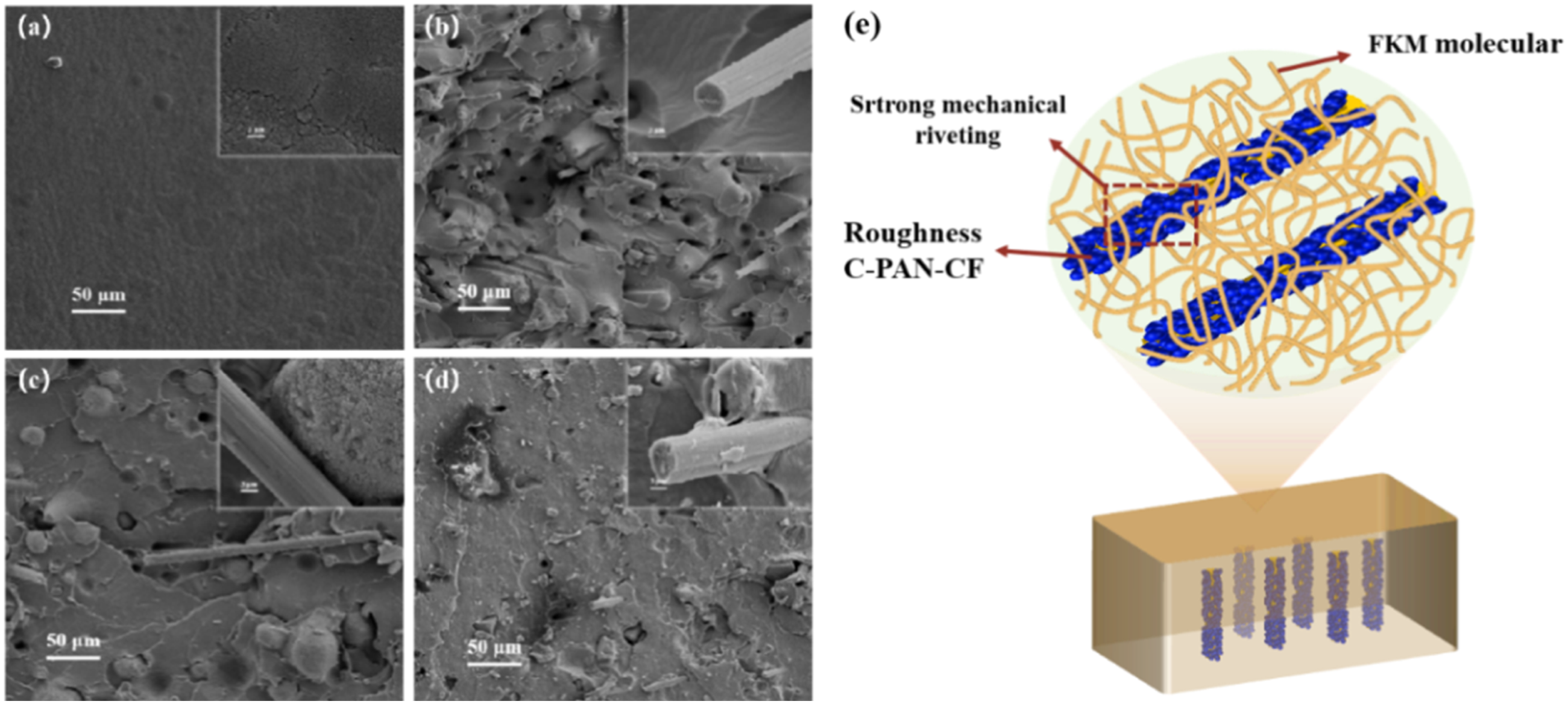
Thermal performance analysis
The TG curves of various FKM composites were displayed in Figure 9(a). It can be seen that the addition of CF and C-PAN-CF could raise the FKM’s initial thermal decomposition temperature, which signify better thermal stability. However, for FKM/PAN-CF composite, the initial thermal decomposition temperature was slightly reduced due to the thermal decomposition of PAN. (a) TG and (b) DTG curves of different FKM composites.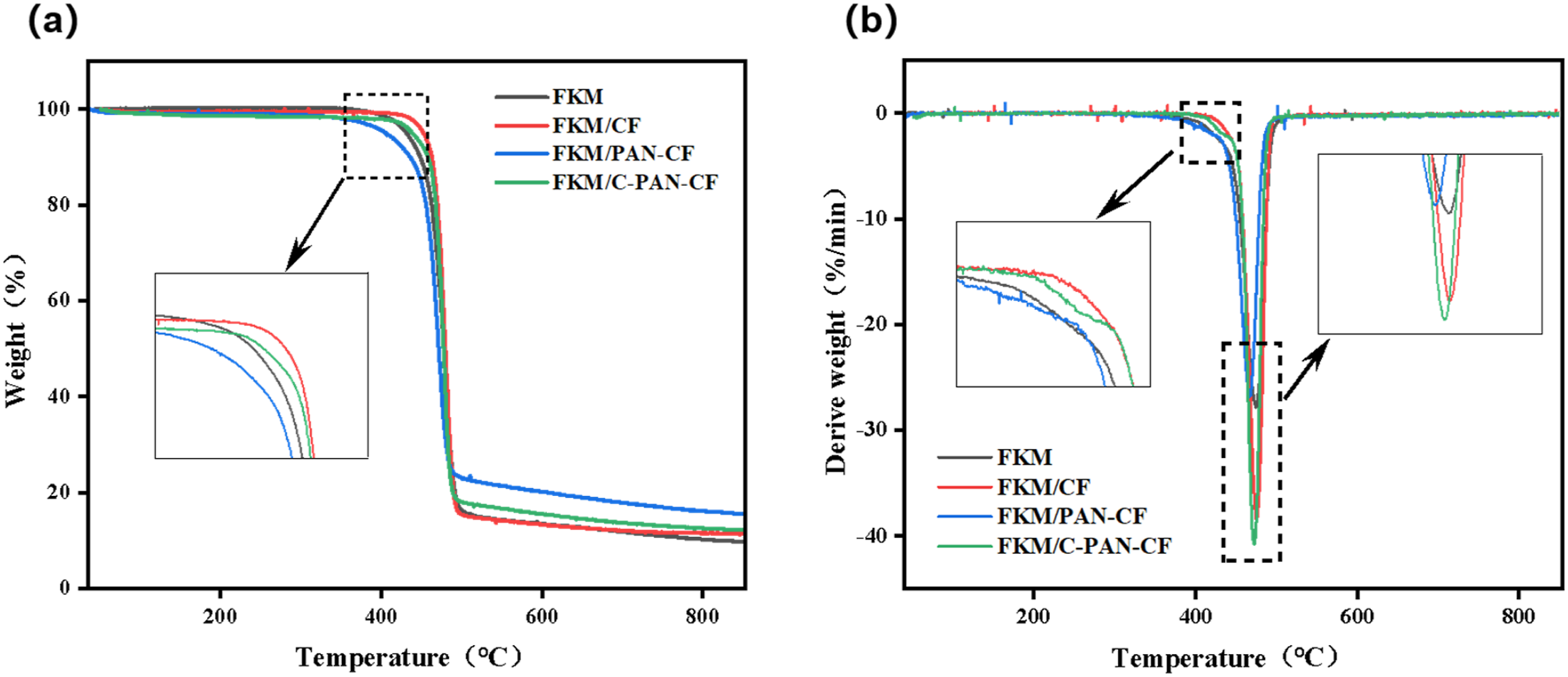
The DTG curves of several FKM composites were displayed in Figure 9(b). The composites exhibit two distinct breakdown mechanisms. The volatilization of additives, water, or solvents causes the first peak in the DTG after 400°C, which indicates the breakdown of non-rubber components. The pyrolysis of the FKM side groups and the C-C bonds on the main chain is shown by the second peak, which marks the start of the composite’s fast breakdown in the 425°C⁓500°C range. It is evident that CF and C-PAN-CF can raise FKM’s first breakdown temperature, improving its thermal stability. 28
To investigate the thermal manage performance of the FKM composites, the temperature change of the sample surface during the heating and cooling processes was demonstrated by using an infrared thermography, and the high and low temperatures are represented by the light and dark colors, respectively. 29
As shown in Figure 10(a)-(c), after 100 second’s heating treatment, the FKM, FKM/CF, FKM/PAN-CF and FKM/C-PAN-CF composites’ temperatures increased to 66.9°C, 86.3°C, 69.8°C, and 84.5°C, respectively. While after 300 second’s cooling treatment, the FKM, FKM/CF, FKM/PAN-CF and FKM/C-PAN-CF composites’ temperatures reduced to 31.5°C, 26.1°C, 29.3°C, and 28.4°C, respectively. It can be clearly seen that FKM/PAN-CF composite shows the relatively lower heating and cooling speed, which was due to the low thermal conductivity of PAN nanospheres. However, the FKM/CF and FKM/C-PAN-CF composite exhibits relatively higher heating and cooling speed, which could be ascribed to the high thermal conductivity of CF and C-PAN-CF. In short, compared with the PAN-CF, the carbonizing treatment could enhance the heat transfer and heat dissipation ability of C-PAN-CF. In order to further verify the thermal properties of the composites, the thermal conductivity of the composites was tested by a thermal conductivity meter, and the results are shown in Figure SI 1. Surface temperature variations of different FKM samples: (a) infrared images during heating and cooling process, (b) temperature variations during heating process, and (c) temperature variations during cooling process.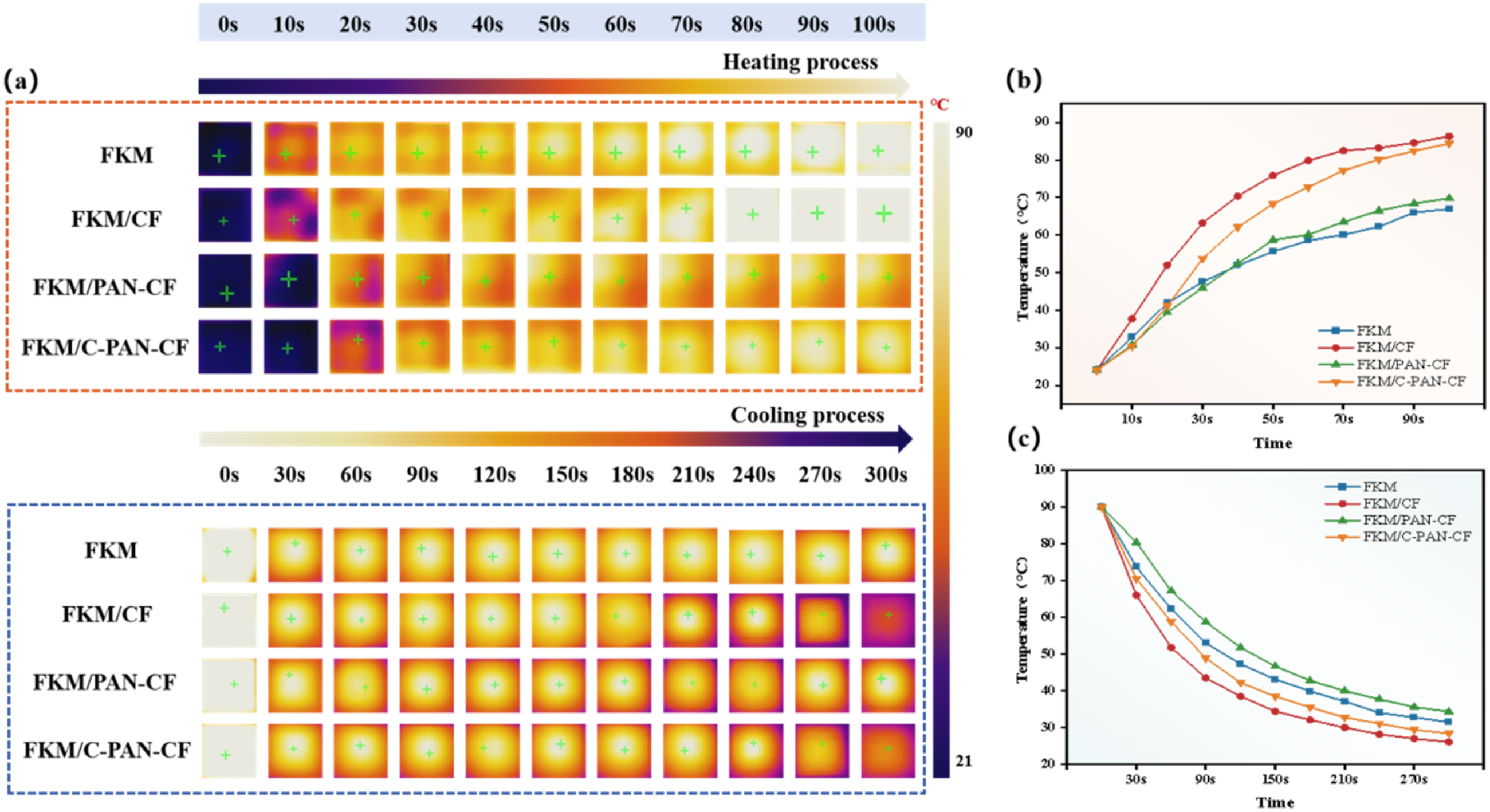
Electromagnetic shielding performance analysis
The EMI shielding effectiveness of several composites in the X-band is displayed in Figure 11. Figure 11(a) illustrates the rise in EMI SE values after addition of CF, PAN-CF, and C-PAN-CF, indicates that CF or modified CF is the primary source of the composites’ EMI SE. The absorption coefficient (SEA), reflection coefficient (SER), and total coefficient (SET) of various composites are displayed in Figure 11(b).30,31 The results show that SER is greater than SEA, indicating that the composite reflects the majority of electromagnetic waves and absorbs a small portion. This suggests that SER is the primary factor affecting SET. Furthermore, the composite’s SER, SEA, and SET after fiber addition are all greater than those of the FKM. The reason was that the filler could attenuate the electromagnetic waves that enter the samples, improving their electrical conductivity. Figure 11(c) shows the transmission (T), reflection (R), and absorption (A) power coefficients of several FKM composites, respectively. It can seen that the composites’ shielding mechanism was mainly based on reflection.32,33 This finding is consistent with the conclusion drawn in Figure 11(b) and indicates that the composites are electromagnetic shielding materials based on reflection mechanism. Figure 11(d) presents the water contact angles of fluororubber composites. The water contact angles of FKM, FKM/CF, FKM/PAN-CF, and FKM/C-PAN-CF are 99°, 104.8°, 113°, and 115.3° respectively. It can be observed that the water contact angle of the composite material increases with the addition of carbon fibers, which is attributed to the fact that CF enhances the roughness of the FKM composite material at the microstructural level.34,35 After the addition of PAN, the water contact angle is further increased, which may be caused by the hydrophobicity of PAN. (a) EMI SE, (b) SET, SER, SEA, (c) R, T, A of different FKM samples,and (d) water contact angle.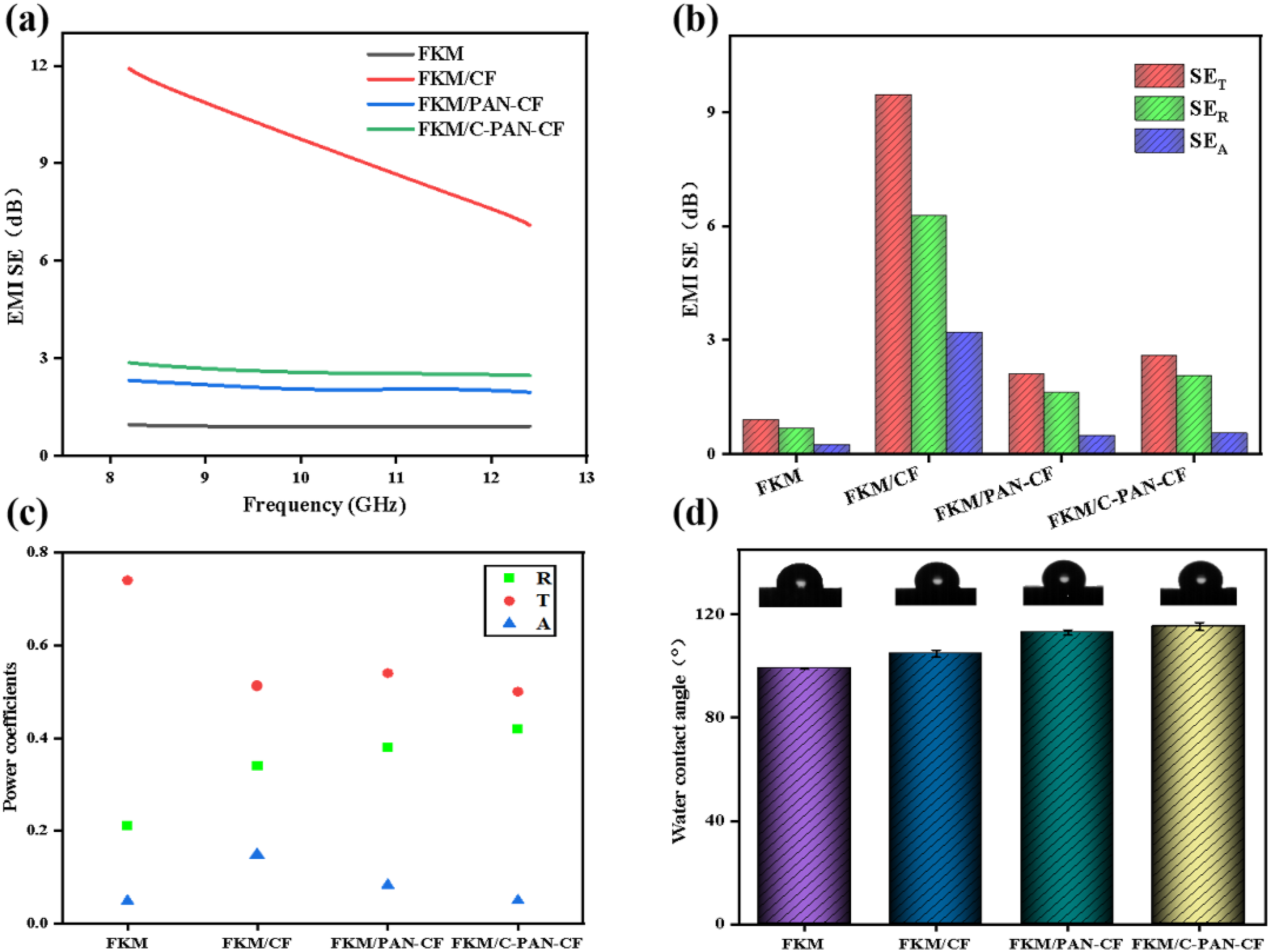
Conclusion
In this study, a C-PAN-CF hybrid fiber was prepared by PAN coating treatment followed by a high-temperature carbonizing process. The rough and irregular PAN carbonized layer on C-PAN-CF can greatly enhance the mechanical performance of FKM by mechanical riveting with FKM. The tensile strength, modulus at 100% strain and hardness of FKM/C-PAN-CF improved by 122.0%, 123.5% and 29.2%, respectively, compared with the original FKM. And the compression set was reduced to a relatively lower value of 29.5%. In addition, the C-PAN-CF could also raise the FKM’s initial thermal decomposition temperature and enhanced its thermal stability. The thermal management performance and the EMI SE were also improved, particularly in the X-band, the composites’ shielding mechanism was mainly based on reflection. In addition, the addition of CF and PAN increased the water contact angle of the composites, which contributes to the hydrophobicity of the composites. In summary, such a simple and effective technique for preparing C-PAN-CF hybrid fibers provides great potential in industrial applications.
Supplemental Material
Suppplemental Material - Modification of carbon fiber by polyacrylonitrile coating and high-temperature carbonizing treatment for enhancing fluororubber’ mechanical and electromagnetic shielding performance
Suppplemental Material for Modification of carbon fiber by polyacrylonitrile coating and high-temperature carbonizing treatment for enhancing fluororubber’ mechanical and electromagnetic shielding performance by Xiaohong Li, Ping Shen, Yiyan Liu, Xinyi Ma, Yadong Wu, Kanstantsin Vishneuski, Shun Wang, Huile Jin in Journal of Thermoplastic Composite Materials
Footnotes
Acknowledgements
We are deeply grateful for the financial assistance we have received from various funding institutions. Special thanks to the support of National Natural Science Foundation of China, with approval numbers of 51872209, 52072273 and 51972239.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
