Abstract
This study demonstrated the successful valorization of agro-waste by utilizing spinach biomass extract for the green synthesis of iron oxide nanoparticles (IONPs). The characterization confirmed the formation of spherical, crystalline nanoparticles with an average size of 102 nm, a crystallinity index of 70.4%, and high thermal stability, with a major degradation peak at 230°C. When incorporated into polyvinyl alcohol (PVA) films, the IONPs imparted multifunctional properties. A 3 wt% IONP loading was identified as optimal, resulting in a composite film with a 32.89 MPa Young’s modulus, a 16.8% reduction in moisture content compared to pure PVA, and significant antimicrobial activity. These properties position the spinach-derived IONP/PVA composite as a sustainable and effective material for active food packaging, directly addressing waste valorization and the need for high-performance, biodegradable polymers.
Keywords
Introduction
Nanocomposite films are gaining popularity in the food packaging industry. Nowadays, researchers are increasingly turning to nanomaterials as a filler in polymers. Food packaging systems based on biodegradable polymers are in high demand since they are more environmentally friendly, cheaper, and less harmful than the currently utilized packaging materials. Several polymers have already undergone various modifications to make them more suitable for use in packaging. The film-forming and water-soluble characteristics of the polymer polyvinyl alcohol (PVA) have led to its extensive use in the food and medical industries. The advanced multifunctional packaging sector has taken a keen interest in polyvinyl alcohol (PVA), a biodegradable polymer that has several desirable properties, including high transparency, great oxygen barrier performance, non-toxicity, and outstanding biodegradability. Nevertheless, PVA is extremely sensitive to changes in humidity due to the abundance of hydroxyl groups in its molecular chains. Hence, to encourage its widespread use, it is crucial to improve PVA film’s water vapor barrier performance. There have been recent reports of PVA nanocomposites that incorporate different nanoparticles and exhibit better water vapor barrier performance.
High aspect ratio, excellent mechanical, scratch, barrier, and thermal resistance, and low loadings are all characteristics of nanofillers. To maximize their properties, nanomaterials must be dispersed throughout the polymer matrix, as they tend to clump together. The exceptional characteristics of nanofillers, whether derived from nature or man-made, make them the most intriguing and potentially game-changing materials of the future. Nanofillers include carbon nanotubes, nanofibers, nano metal oxides, graphene, inorganic nanotubes, fullerenes, calcium carbonate, metallic nanoparticles, POSS, and ultra-disperse diamonds (nanodiamonds).
Iron oxide nanoparticles (IONPs) are tiny particles with large surface area. IONPS are known for being biocompatible and having low toxicity. It has many applications such as environmental remediation (water wash treatment and catalyst coatings), constructions (colouring tiles, brick, and concrete), electronics (printed electronics and spintronics), microwave (production and operation), coatings (EM wave consumption), biology and medicine (cancer treatments and drug delivery). 1 Iron oxide is the name for many different natural compounds. There are various forms of iron oxides available which include oxides, hydroxides, and oxides-hydroxides. Iron- Oxide nanoparticles are either maghemite or magnetite. The size of the magnetite nanoparticles ranges from 2 to 20 nm having the super paramagnetism. Some scans such as CT scans and MRI, need a contrast agent to make veins, organs, etc, more visible. Iron oxide nanoparticles are commonly used in many biomedical applications including molecular imaging, hyperthermia treatment, and drug delivery. 2 Having antioxidant properties allows iron oxides to neutralize free radicals and decrease oxidative stress on overall skin. It will protect from premature aging, reduce inflammation, and promote overall skin health. 3
Before civilization, the earth was very clean and non-polluted. However, with the advent of agriculture and industries the earth became polluted and a lot of waste was generated. Some of the agricultural waste is used to feed the animals and to prepare fuels. Other substances such as pesticides and fertilizers, are used during agriculture. 4 Now a days, industries in which these pesticides fertilizers, and manufacturing also cause a lot of air pollution. They also generate a lot of waste that causes pollution to the society. Out of all the resource-based industries, agriculture has the greatest impact on the economy. Additionally, it produces a substantial amount of garbage. Farming, or agriculture, is the practice of raising various kinds of plants and animals for human use or scientific research into their potential medical, nutritional, or aesthetic benefits. 5 Organic waste is the main component of agricultural waste. Slurries and farmyard manures are the extractors used by animals. Included in this category are items originating from plants, such as sugar cane, molasses, and peals, as well as those originating from animals, such as dung and dead bodies. Untreated garbage can contaminate both surface and underground water sources, in addition to adding to air pollution. 6 By producing various composts, waste management can help keep farm animals healthy, lessen the need for commercial fertilizers, and even be used to make fuel. Many poor countries earn a lot of their money from their agro-waste products. According to that spinach waste from marketplaces is very common and is rich in antioxidants. 7 The utilization of them in value-added product development is a research focus of this study.
Spinach is a nutrition-rich leafy vegetable that can be used all over the world. It has dark green leaves and a mild slightly sweet taste. Spinach is a fantastic source of iron, which is important for our health. 8 It is a cool-weather vegetable and grows well in the spring and fall. It is a rich source of nutrition vitamin A, vitamin C, and full minerals like iron, calcium, and magnesium. Spinach improves our health, burn density, and brain function. Some functionalities listed for the spinach extract are; (1) The nitrate content present lowers the chance of developing long-term conditions like diabetes, cancer, and heart disease (2) Improves eye health which is one of the basic important nutrients for healthy vision. (3) It is a good source of calcium which improves the strength of bone and teeth (4) It contains potassium which can help blood pressure and digestion problems (5) It contains high fibre which promotes healthy digestion. Farmers have been growing and enjoying spinach for a very long time. 9
The researcher synthesizes iron oxide nanoparticles using a variety of methods. This study found that iron oxide nanoparticles were reduced using antioxidants present in natural waste material. The IONPs particles are synthesized using spinach extract as capping agents.
10
The reason for the selection of the capping agent (
This study investigates the possibility of using market waste
Experimental
Materials
The source of the wasted spinach (
Method of synthesis of iron oxide nanoparticles
Biosynthesis of iron oxide nanoparticles using spinach extract is presented in Figure 1. The spinaches were washed and let dry in the sun for 12 hours after being harvested. The dried sample was utilized for the preparation of the extract. The reason for the drying process is to eliminate pigments and dust particles. About 200 g of spinach waste was boiled in 1 L of distilled water to extract the antioxidants present. The boiled mix was cooled and filtered for further treatment. To the filtrate, 5%(w/v) sodium citrate was added to avoid fungal and bacterial interactions. The preserved filtrate was utilized as a binder for the ferrous sulphate precursor-based production of iron oxide nanoparticles. The precursor was added to the extract in a dilute form of 0.01 N which was standardized previously. The obtained mixture was heated for 2 h at 60°C. The mixture was set aside for one day for the completion of nanoparticles formation. The solution was then centrifuged in a centrifugation tube at 5000 rpm for 5 h. The nanoparticles obtained were then dried in an oven at 80°C for 4 h. Biosynthesis of iron oxide nanoparticles using spinach extract.
Fabrication of PVA/IONPs nanocomposite film
First, a pure PVA sample was made by mixing 2 g of PVA with 20 mL of cold water and stirring it with a magnetic stirrer at room temperature for 30 minutes at a maximum speed of 1500 revolutions per minute. A glass petri plate with a diameter of 6 cm was used to transfer the dissolved mixture. After that, the petri dish was left to dry for around four or five hours. A silica desiccator was used to keep the dried film once it was peeled off the petri dishes. Following the identical process as the pure PVA film, biofilms reinforced with the IONPs at concentrations of 1%, 2%,3%,4%, and 5% by weight (wt%) were also produced. Depending on the sample, the amount of nanofiller was adjusted. In 20 mL of water, 0.2 g of IONPs and 1.8 g of PVA were added under the same stirring conditions to achieve a 1% solution. The films were placed in a silica desiccator for 12 hours after finishing all of the samples. The filler concentrations (1-5 wt%) were selected to systematically study the effect of low to moderate IONPs loading on the properties of PVA, identifying an optimal concentration that enhances performance without causing significant agglomeration.
Characterization of iron oxide nanoparticles
Fourier transform spectroscopy
The IONP spectra were collected in a KBr matrix using a Perkin Elmer Fourier Transform Infrared (FTIR) Spectrometer. 13 The resolution was 2 cm−1, and the scan frequency was 32 scans per minute. For this investigation, the wave number range that was chosen was 400–4000 cm−1. Before being combined with KBr, the components were ground into a fine powder using a crusher and pestle. When the pressure was applied to the specimen, it pelletized, making it ready to record the FTIR spectra under standard conditions. FTIR spectra of IONPs are used to determine the presence of free functional groups in the sample.
X-ray diffraction analysis
To assess the rate of crystallinity of IONPs, an X-ray diffraction (XRD) investigation was conducted. Cukα radiation (λ = 0.154,056 nm) was used in this investigation, which was conducted using a PW3040/60 X'Pert analyzer (Philips, UK). In this case, the filament was operating at 40 kV with a current of 30 mA.
14
In a range of 5° to 30° of 2θ, the samples were analyzed with a step size of 0.0200°. Equation (3) was used to analyze the Crystalline index (CI). To determine the sample’s crystallinity index, the Origin Pro 2019 software was used to obtain the crystalline peak area value. Using Scherer’s equation (4), which is similar to the one used in the previous step, the size of the crystallite was measured in nanometres (nm), with K standing for Scherer’s constant (0.89), 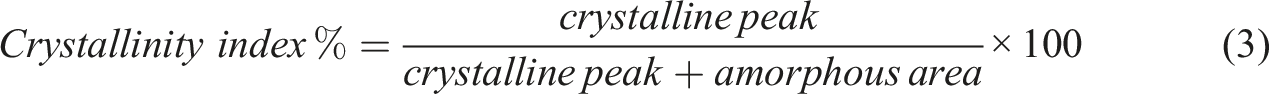
The average crystallite size (nm) in the plane opposite the lattice plane was calculated using the Scherrer equation.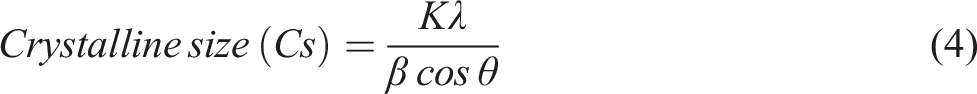
UV spectroscopy
Additional verification of the chemical components of IONPs was conducted by UV-visible spectroscopy examination. 16 The test sample was examined using a Shimadzu UV-2101 PC spectrometer, and the absorption spectra that were produced were found to fall within the 200-700 nm region. To characterize the observed absorption peaks, the optical transparency of several active compounds present in the sample was utilized. 17
EDX analysis
Five distinct places, each ranging in size from 1 to 4 mm2, were examined by utilizing energy dispersive X (EDX) single particle analysis in conjunction with automated scanning electron microscopy (SEM) to analyze spectra. 18 The Zeiss EDX Analyzer is a powerful tool integrated with Scanning Electron Microscopy (SEM) for elemental analysis of materials, including solid nanoparticles detector achieves great efficiency using a batch detector and particle analyser. The electron beam charging during analysis requires non-conductive materials to receive a thin conductive surface made of Au metal or Pt metal or C metal which must be applied by a sputter coater. Following sample placement in the Zeiss SEM chamber users should establish 15-20 kV beam energy as the optimal conditions to analyse nanoparticles. Stable runtime functions together with proper sample preparation methods ensure reliable results for analysing solid nanoparticles. All of the image’s pixels remained at the specified ratio for the study. 19
SEM analysis and particle size analysis
The IONPs’ cross-section was analyzed using a scanning electron microscope (SEM) (ZEISS, USA). The sample was first dissolved in a solvent, then attached to adhesive tape of carbon, and then sputter-coated with gold. 20 To capture the photos, an acceleration voltage of 5 kV was used at various magnifications. Scanning electron microscopy (SEM) images were then used to identify the sample.
Thermal analysis
For the thermal study, a turbo gas analyzer (STA449 F3, Netzsch, Germany) operating in a nitrogen environment at a flow rate of 40 mL/min was utilized. The samples were heated the temperature to 1000°C in an orderly manner. The samples were heated from 25 to 600°C at a rate of 10°C per minute. 21 We evaluated the residual weight at 600°C to find the percentage of weight loss. Thermogravimetric (TGA) and derivative DTG data were obtained for every sample. The present study was conducted in an environment high in nitrogen. It was found that the specimen had a mass of 5.504 mg while kept at room temperature. 22 The next step was to place the specimen in an alumina furnace to heat it. At a rate of 20°C per minute, the sample’s temperature increased throughout the heating process from 30 to 800°C. For this experiment, we used TG data to plot the percent weight loss versus temperature, where m/s is the mass flow rate of the sample measured in degrees celsius increments. When it came to IONPs, the DTG technique was a lifesaver when it came to determining how quickly sample components disintegrated at different temperatures. 23
Particle size and roughness analysis
At 1500×, 1000x, and 500x magnifications, a total of three photographs were taken. The National Institutes of Health’s image processing application, ImageJ, was used to do particle size analysis. 24 The shape descriptors and mean length (µm) were derived from an examination of a total of 25 particles. The size of a sample particle can be easily determined using the Image J software. The SEM image of the sample was incorporated into the software and selected with different ranges for 25 particles. The average of particles was calculated which were selected for the analysis. 25
TEM analysis
The JEOL JM 2100 with Gatan camera is used to identify the particle size and shape of nanoparticle. Distilled water and ethanol and methanol should serve as solvents because they minimize contamination in the system. Sample absorbance was maintained below 0.1 for optimum peak imaging results. The sample concentration was maintained at 10- 6 M by measuring the UV-Vis absorption spectra. A microlitre drop of the diluted solution was placed on the TEM grid. After one minute of natural drying, excess solution was carefully removed from the droplet using a sterile tissue, taking care to keep the tissue away from the nanoparticles. High resolution imaging data were obtained for nanoparticle analysis using the Gatan camera.
Characterization of PLA/IO nanoparticles biobased film material
The thickness of the bio nanocomposite film samples was found using a spiral micrometer a resolution consisting of 0.01 mm. By averaging the thickness from 5 randomly selected sites on each sample, we were able to calculate the mechanical properties of the films.
26
After being cut and weighted to a thickness of 4 × 4 cm2, the film pieces were dried for 24 hours at 105°C. The film reached equilibrium, and its weight (W1) was recorded. The following formula was used to determine the MC: moisture content (MC)
An analytical balance from Shimadzu, model ATX 224, was used to ascertain the film’s weight. The solid specific gravity test involves weighing a film sample in two different environments: air and a liquid with a defined density.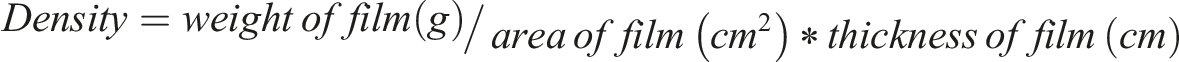
The mechanical properties, which were expressed as TS and EAB, were measured using the XWL Auto Tensile Tester, which was produced by Labthink Mechanical and Electrical Technology Co. Ltd of China. As per the ASTM-D882-02, the experimental parameters were 23 ± 1°C and 50 ± 5% relative humidity. The film samples, which were 100 mm × 15 mm, were initially placed 50 mm away from the machine when it was at room temperature. There were five separate runs, each with a 50 mm/min crosshead speed. The disk diffusion method was also used to assess the fillers’ antimicrobial properties. Plates of GPY agar were used for yeast and Mueller-Hinton Agar for bacteria were infected with the 0.5 McF microbiological suspension (MHA, HiMedia, India). 27 The inoculated agar plate was covered with four sterile discs of six-by-six millimetres filter paper within fifteen minutes of inoculation. Samples were applied to the 6 discs at a concentration of 200 µg/disc. The disc-containing MHA and GPY agar plates were left to incubate at 37°C or 28°C for 24 hours. The sizes of the inhibitory zones were measured after the incubation period. 28
Result and discussions
FT-IR analysis
The results of the FT-IR analysis are displayed in Figure 2. Because the OH group is stretched, the first broadband appears at 3448.35 cm−1. Both the presence of water molecules and the hydroxide production of iron oxide could be responsible for the hydroxyl group. The tiny peak that arises at 2378.24 cm−1 is the reason for the atmospheric carbon dioxide stretching. This will be eliminated after the completion of the purification process. A peak at 1640.28 cm−1 is due to the presence C = C bond stretching. The methylene group is the cause of the peak at 1463.51 cm−1. The peak at 1110.84 cm−1 was also thought to be caused by phytochemicals with C-O interactions. A closer look at the peaks detected between 3500-100 cm−1 reveals the capping agents present in the rejected spinach extract. The formation of iron oxide crystals was confirmed by two prominent absorption bands at 625.76 cm−1 and 511.96 cm−1, which were caused by the formation of Fe-O bonds. Isolated iron oxide nanoparticles from Papaver somniferum L.
29
Only bands linked to iron and hydroxyl absorption were visible in the FTIR spectrum, indicating that the created product was completely devoid of contaminants. The crystalline character of the produced α - Fe2O3 nanoparticles was revealed by the breaking of the double band into bands below 700 cm−1. FT-IR analysis of iron oxide nanoparticles synthesized with spinach extract.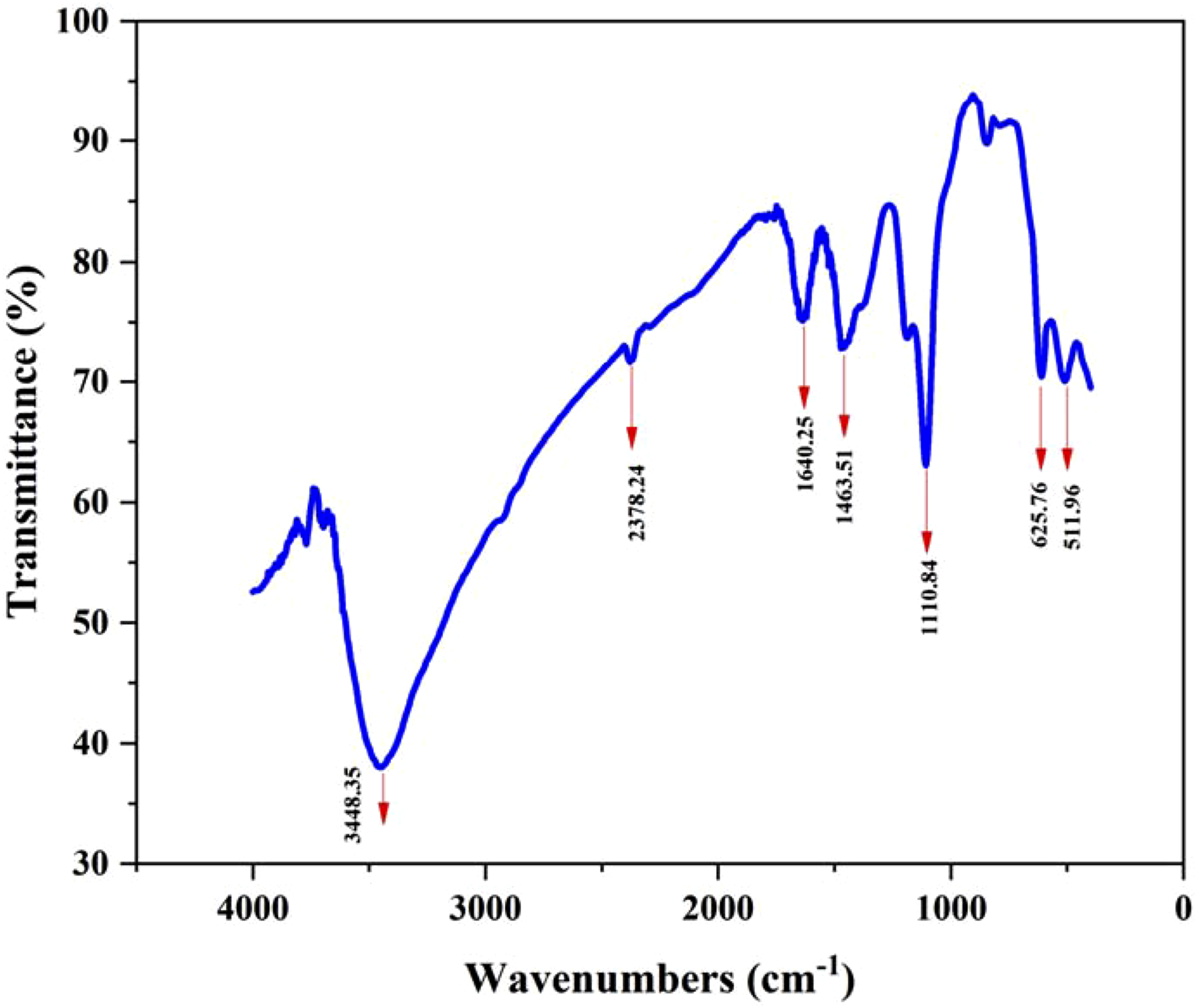
UV analysis
The UV analysis report of iron oxide nanoparticles from spinach leaves is shown in Figure 3. The absorbance peak was visualized and highlighted in the yellow-shaded region. There are two different absorbance peaks were observed between 200-800 nm. The characteristic peaks of iron oxide nanoparticles were 261.60 nm and 340 nm respectively for Fe2O3 and Fe2O4. The biosynthesized nanoparticles contain some capping agents that are responsible for the broadening of peaks in the UV absorbance region. Table 1 displays the peak levels of several materials utilized in the production of iron oxide nanoparticles. The peak 1 of all samples given in the table shows similarities with this study. However, a variation presents in the peak 2 compared to others. Turmeric and lemon grass had higher absorbance peaks of 457 nm and 450 nm respectively compared to others. The waste spinach contains a very low absorbance peak at 340 nm. The variation in peak 2 completely depends on the raw material and method of synthesis involved. The two variant peaks of iron oxide prove the purity and two oxidative (ferrous, ferric) nature. UV analysis of iron oxide nanoparticles synthesized with spinach extract. UV absorbance range of some other iron nanoparticles from different resources.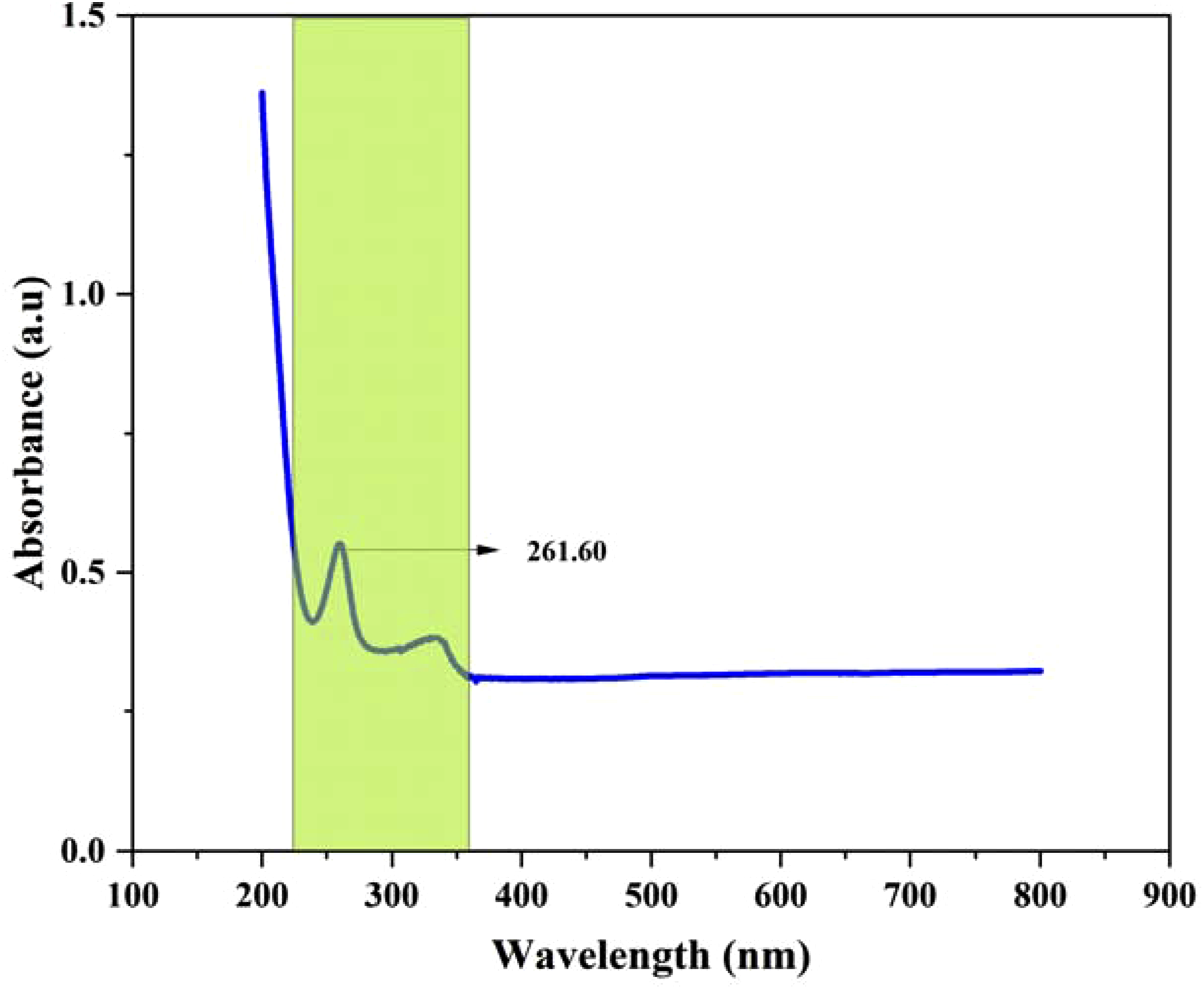
X-ray diffraction analysis
Crystallinity index of some iron oxide nanoparticles from different resources.
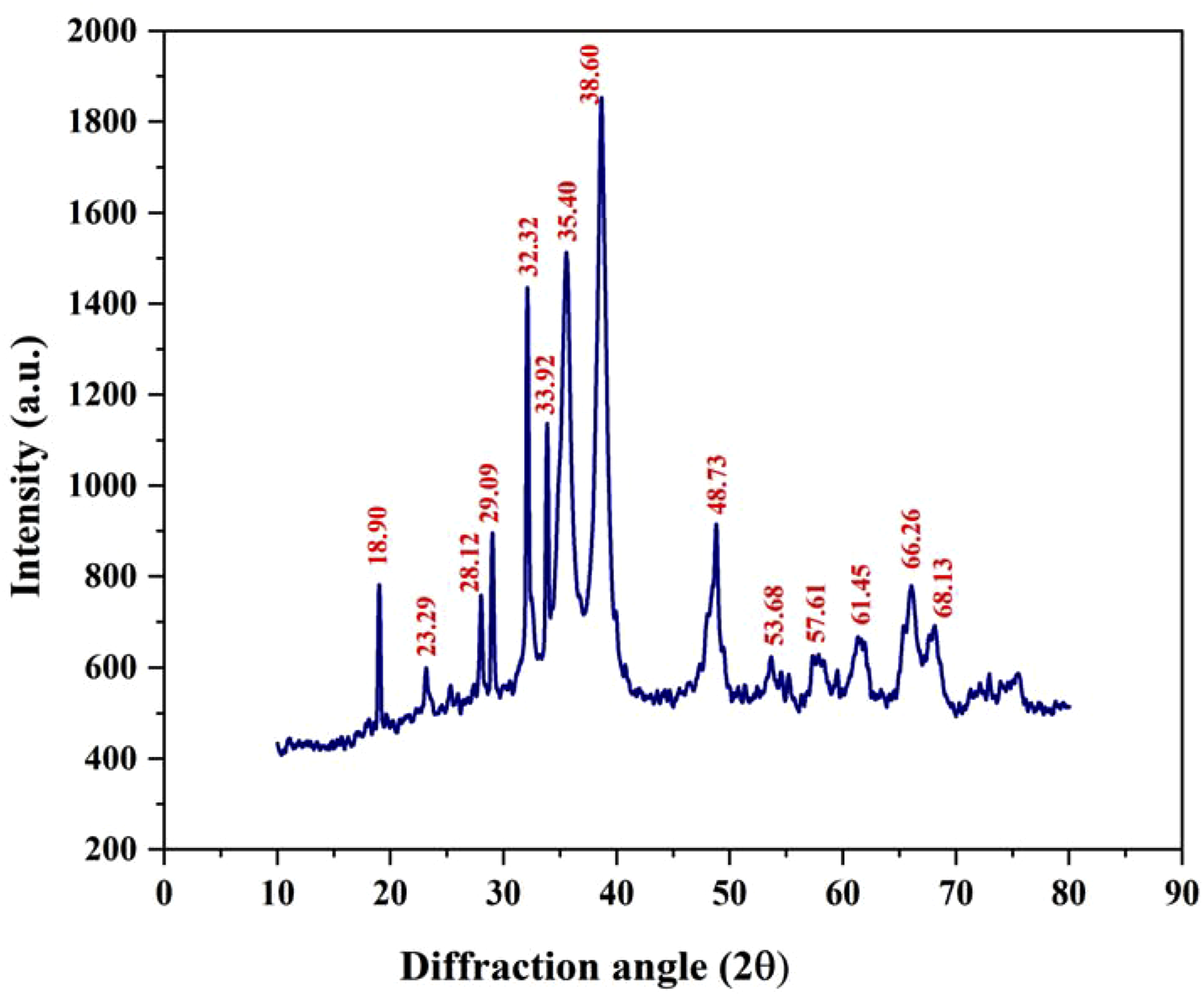
X-ray diffraction analysis of iron oxide nanoparticles synthesized with spinach extract.
SEM analysis
Figure 5 scanning electron microscopy (SEM) gives strong visual support to the successful production of iron oxide nanoparticles (IONPs) by the green synthesis method by use of spinach biomass waste. The images with different magnifications show that the population of nanoparticles is mainly quasi-spherical, however, a high level of asymmetry can be seen in most of them. This is a typical non-sphericity bending in biogenic synthesis since the intricate phytochemicals in the spinach extract (polyphenols, flavonoids, and organic acids) are reducing and capping agents that do not uniformly affect the kinetics of crystallization of the crystal planes. This leads to the rather abnormal and asymmetrical structures observed in the micrographs.
47
SEM analysis of iron oxide nanoparticles synthesized with spinach extract.
One large aspect of all the magnifications is the large agglomeration of the nanoparticles so as to be in large chain-like formations. This is a normal and natural occurrence of iron oxide nanoparticles since they by their properties of dipole-dipole interactions. The fact that the agglomerates form, although it may decrease the specific surface area, indicates that the material under synthesis is magnetic. Moreover, the capping agents made by using biomass (although effective in preventing macroscopic precipitation) might not be enough to fully disrupt these strong magnetic forces at the nanoscale, resulting in the clustered morphology as observed.
The surface morphology of individual particles can be measured better at the highest magnification (100.00 K X). The particles have a relatively smooth texture on their surface and clear edges of the particles. Such smoothness is an indication of successful and coherent growth process in which bioactive compounds of the spinach waste have successfully regulated the nucleation and growth resulting to solid and well-formed particles as opposed to porous and fractured structures. The particle size (which is calculated based on the scale bars) would mostly be in the nanoscale size range with many of the particles being 50-100 nm in diameter, though to measure them precisely would mean using specific image analysis software.
TEM analysis
The size distribution of the nanoparticles ranged from 20 to 180 nm, with an average diameter of 102 nm. Figure 6 clearly shows the IONPs’ spherically formed core-shell. In a lattice image and its equivalent image in ultra-TEM analysis, we highlight the hexagonal forms of IONPs. The hexagonal particle, which was thought to be magnetite (Fe3O4) with main facets composed of {111} planes, had a single crystal free of twin and stacking flaws. TEM analysis a) particle size analysing image b) low magnification shape identification image of iron oxide nanoparticles synthesized with spinach extract.
Particle size analysis
The histogram depicting the size distribution of 25 distinct particles, produced via the intersection technique and threshold capture, provides a visual representation of both quantitative and qualitative fluctuations in particle size (Figure 7). While 25 particles provide a preliminary distribution, it is acknowledged that a larger sample size would yield more robust statistics. The particle size was determined by measuring the equivalent length of each particle from the SEM images using ImageJ software. The average particle size was 105.65 nm with a standard deviation of 35.091 nm. The average particle size is also in the nanometer range. There is a standard deviation of 35.091 and an average particle size of 105.65 nm. Spinach extract undergoes both chemical and physical procedures to establish particle size. From one area to another, the orientation of particles can vary. Particles that gather into larger aggregates, called agglomerate particles, are more easily distinguished from particles that are floating about in the air.
49
Particle size analysis of iron oxide nanoparticles synthesized with spinach extract.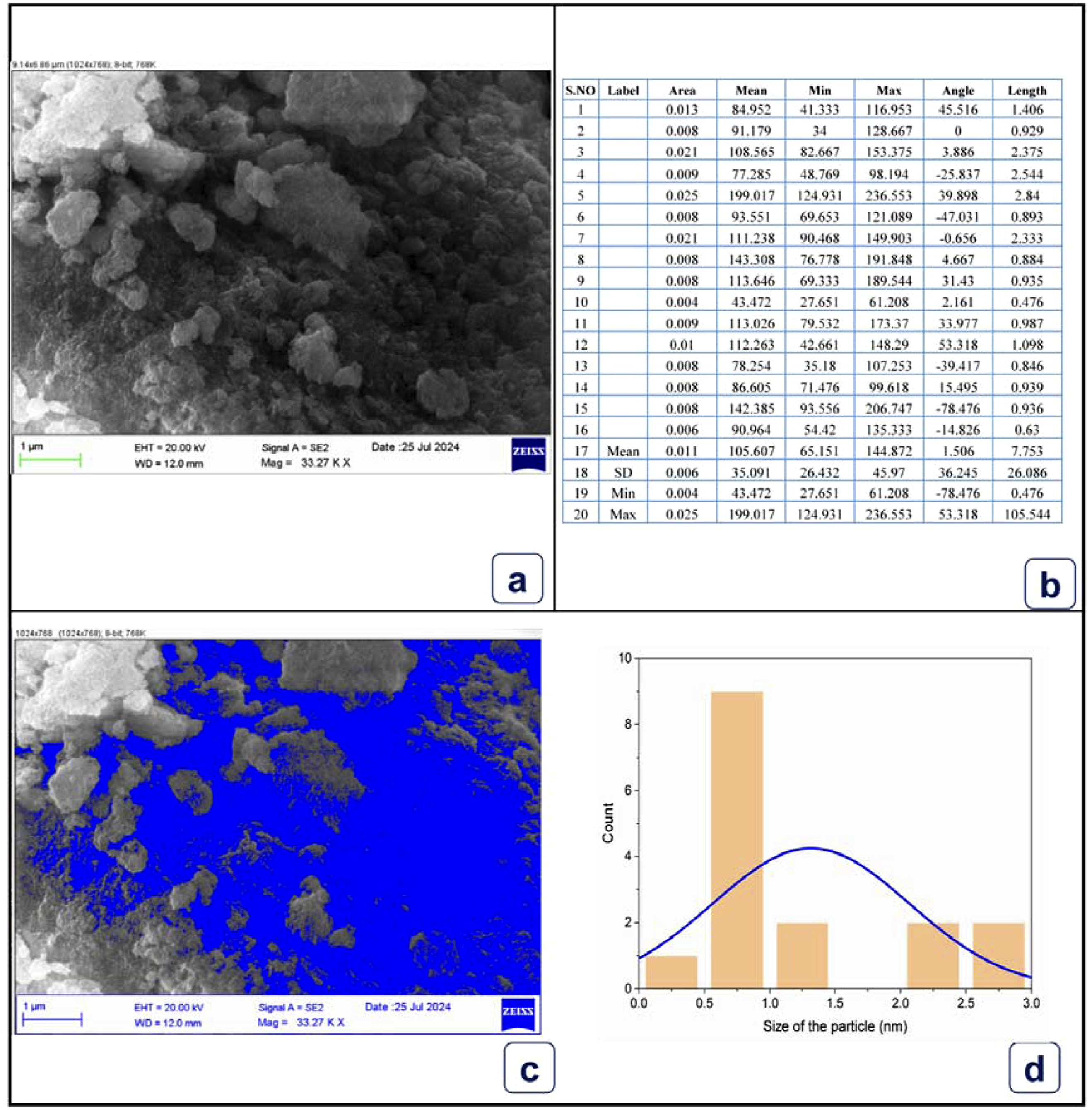
Roughness analysis
Figure 8 shows the in-depth surface roughness study of the Fe2O3 nanoparticles prepared by SEM and 3D topographical profiling. Figure 8(a)) in the form of the SEM micrograph indicates the morphology of the surface of the nanoparticles, which are aggregated in the form of spheres and irregular particles with clear variations in the textures. The line profile (Figure 8(b))) shows that there are changes in the intensity of gray values as factors in changes of surface height in the scanned area, which proves that there were nanoscale irregularities in the surface. Figure 8(c)) is the 3D topographic image, which is used to illustrate the uneven surface texture, whereby, the well-defined peaks and valleys can be seen which contributes to the complexity of the surface. The value of quantitative roughness parameters (Figure 8(d))) was obtained to further analyze the nanoscale topography. The mean roughness (Ra) was 29.2326 nm and the root mean square roughness (Rq) was 25.2959 nm, which is a moderate level of nanoscale unevenness. The skew (Rsk) = 0.4752 value indicates a positive skewness slightly with more peaks than the valleys implying a skewness towards the positive side. The value of kurtosis (Rku) is 0.2094, which shows a rather flat distribution of the peak. The highest point (Rp) and the lowest point (Rv) were found to be −53.0260 nm and 63.4946 nm, respectively, and indicated that there was a considerable surface variation. Other parameters like the depth of the highest valley (Rt = 116.5208 nm) and the average flatness (FAD = 81.4443) are also indicators of the existence of the nanoscale level of surface heterogeneity. In general, the obtained results on surface roughness verify that the Fe2O3 nanoparticles have a stable although irregular topography, which contributes to their interfacial adhesion, dispersion uniformity, and compatibility when being used as a part of the polymer matrices in packaging films. The morphology of rough surface gives it better mechanical strength and barrier properties; thus, these nanoparticles can be used to create highly performing nanocomposite packaging films.
50
Surface roughness analysis of iron oxide nanoparticles synthesized with spinach extract.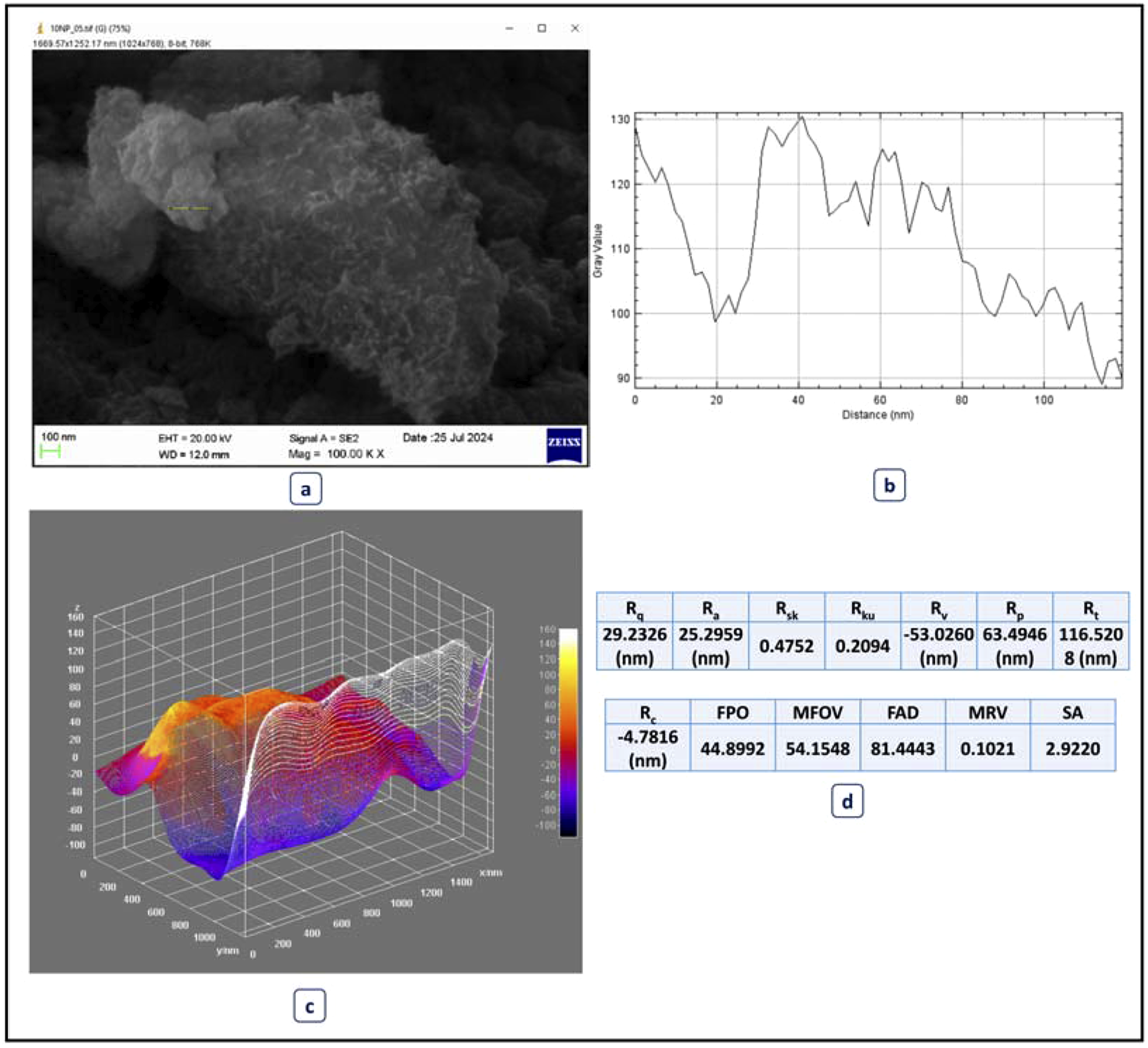
TGA analysis
When using TGA in scanning mode, the mass of a sample can be shown as a function of temperature in Figure 9 (dynamic TGA). It is possible to ascertain the physical and chemical characteristics of materials by monitoring their changes in response to rising temperatures. There have been significant alterations to the materials, as seen by this degradation/decomposition temperature. It was determined that the transition temperature of interest for Fe2O3 was approximately 930°C. Using TG-DSC techniques, it is possible to characterize NPs coatings by evolved gas analysis.51,52 The TGA graph shows that the sample began decomposing at approximately 150°C and finished decomposing at about 700°C, indicating that it was iron (II) oxide. The TGA curve demonstrated a weight loss of approximately 2 g in the trial while the temperature was around 160°C. The thermal stability of the biofilms was demonstrated by the fact that its weight remained nearly constant during the process of water molecules taken from the environment by nanoparticles, which could explain the observed weight reduction. The peak clearly showed that a significant oxide deterioration occurred at 230°C
53
The thermal activity of iron oxide has proven the maximum thermal withstanding nature and can be used as filler in high thermal composite materials. Thermal analysis of iron oxide nanoparticles synthesized with spinach extract.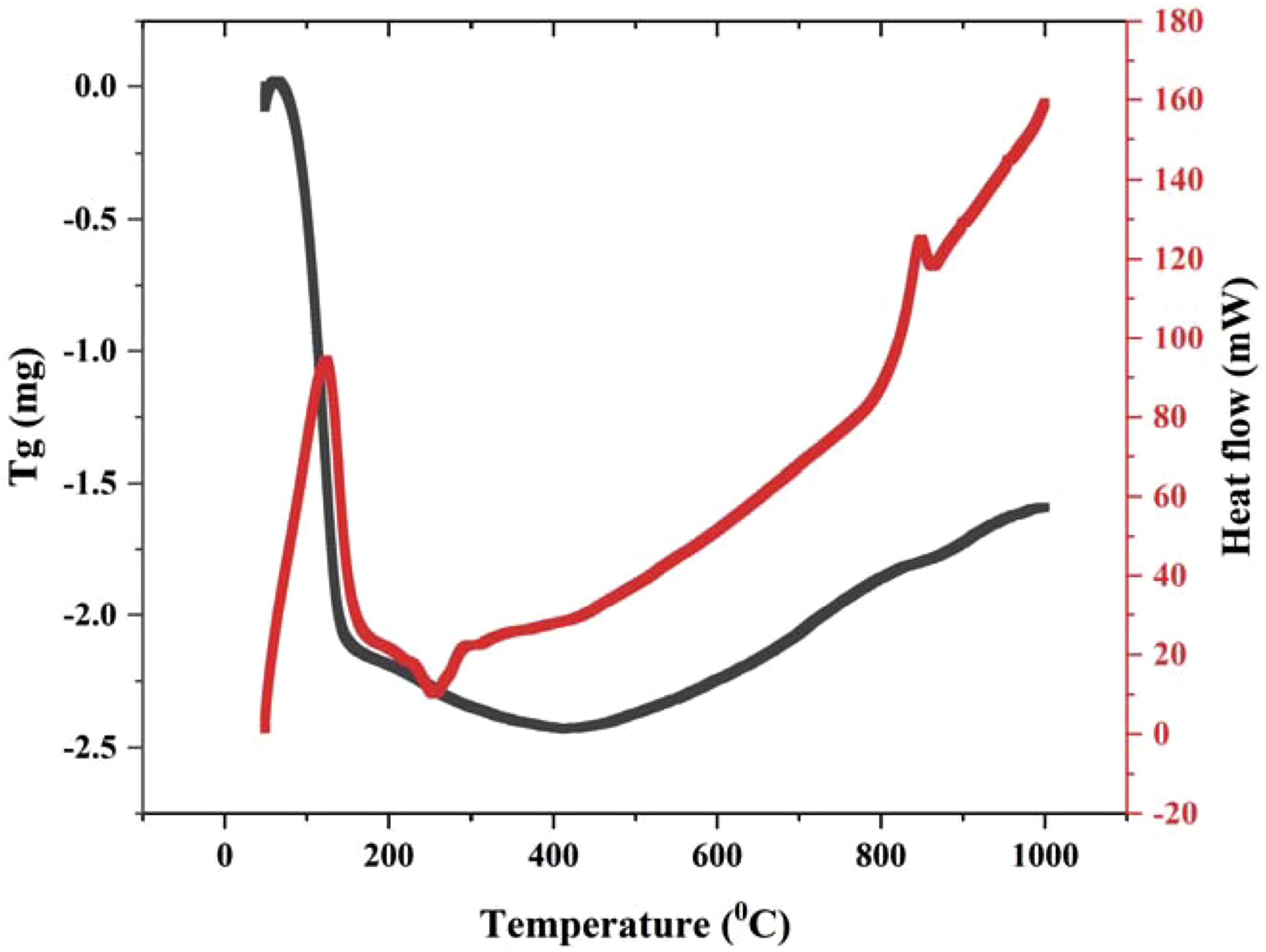
Mechanical properties
Figure 10 shows mechanical behavior of the PVA films that are reinforced with Fe2O3 nanoparticles, such as tensile strength, Young modulus, and break elongation. Figure 10(a)) indicates that tensile strength of pure PVA was 81.11 MPa that steadily declined as the concentration of the nanoparticles increased up to 69.34 MPa at 5 wt contents of Fe2O3. The decrease in the tensile strength of the polymers at increased filler content can be explained by the fact that nanoparticle agglomeration results in the occurrence of stress concentrations and poor interfacial bonding at the polymer matrix interface. On the other hand, Figure 10((b)) reveals that the modulus of Young was increasing at first with pure PVA of 28.74 MPa to a peak of 32.89 MPa with the loading of Fe2O3 of 2 wt percent resulting in stronger stiffness and better intermolecular bonding with homogeneous dispersion of nanoparticles. Figure 10((c)) shows the elongation break was reduced when loading was increased, of 22.12% in pure PVA and 16.71% in 5 wt% Fe2O3, indicating a less flexible material with an increase in filler content. Mechanical property analysis of iron oxide nanoparticles synthesized with spinach extract. A: Tensile strength. B. Elongation at break.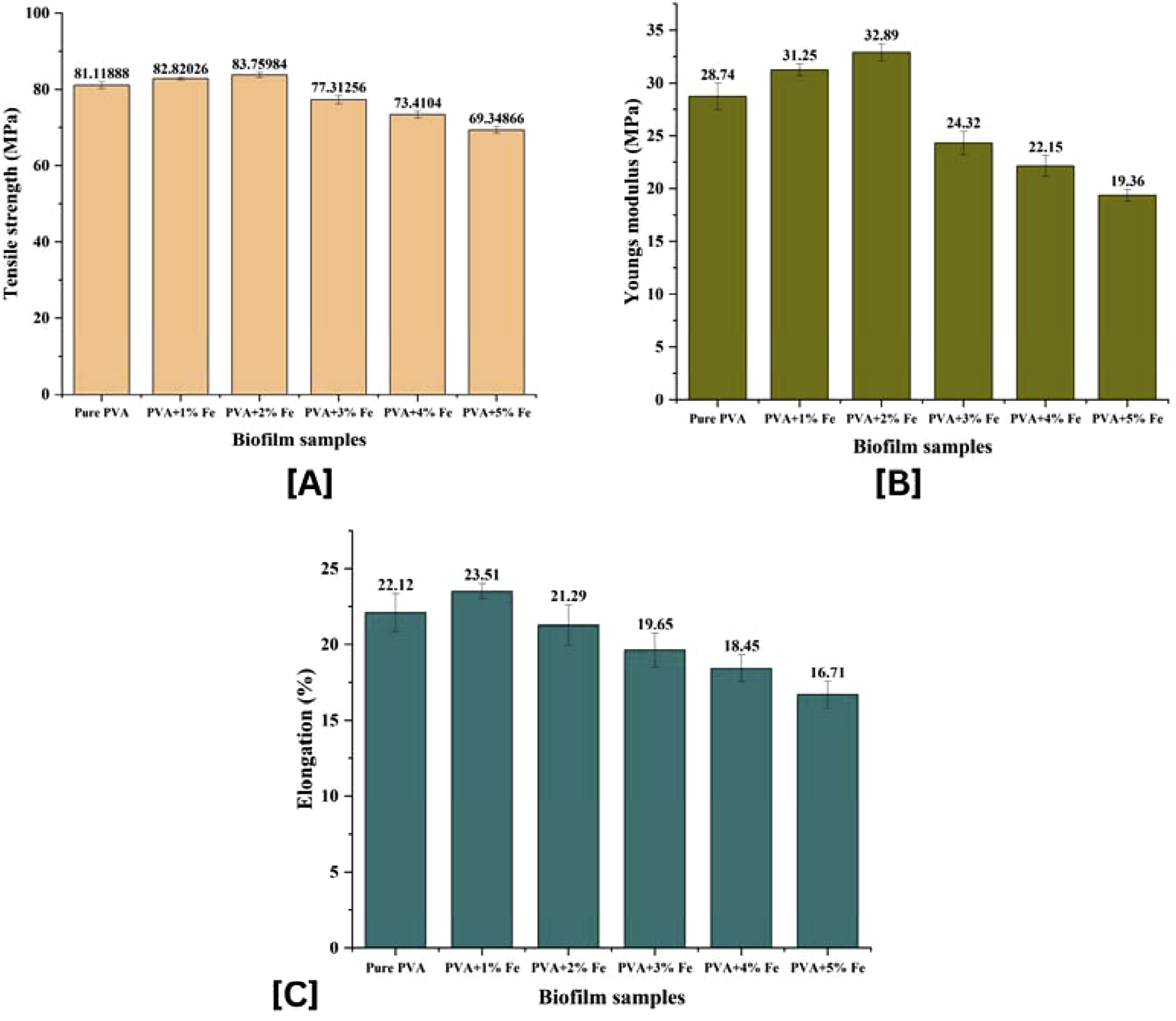
The addition of Fe 2O3 nanoparticles alters the polymer structure by partly rupturing hydrogen bonds and providing new contacts between hydroxyl groups of PVA and the oxygen groups of the iron oxide surface. This interaction increases the mobility of molecular segments, which improves the flexibility and decreases the crystallinity at a lower percentage of filler. The recorded mechanical behavior is in line with previous research results on PVA/Fe2O3 and PVA/chitosan nanocomposites, which found the same strengthened modulus and decreased elongation with increase in filler levels. An addition of 2 wt% of Fe2O3 is the best balance between strength, stiffness and flexibility hence the most suitable composition to be used in bio composites, as a packaging film where good mechanical integrity and structural integrity are needed54,55,. 56
Density, thickness, and moisture content
The density, thickness, and moisture content analysis of the film sample is illustrated in Figure 11. The density of the pure PVA film matrix is approximately 1.30 g/cm3, and the density of the synthesized iron oxide nanoparticles (IONPs) is taken as 5.25 g/cm3 for subsequent calculations. The measured density of the PVA/IONPs films showed a systematic increase as a function of filler content. The close agreement between these values and those calculated via the rule of mixtures indicates a successful integration of the IONPs with minimal void formation, validating the efficacy of the fabrication process. The pure PVA film’s thickness was incrementally increased following the addition of the iron oxide nanoparticle filler to PVA, ranging from 1 to 5 wt percent. After the PVA matrix’s filler level of 35 wt% was reached, the biofilm of PVA/IONPs showed substantial thickness fluctuation. When the filler loading in the PVA matrix was high (5wt%), it caused filler clusters and strips to form on the film surface, which somewhat affected the film’s thickness.
57
Their respective thicknesses were 0.125 mm for purified PVA films and 0.129 mm for PVA/(1-5%) IONPs biofilms. Film sample densities dropped as IONPs concentrations rose. A high IONPs content and a thick structure were associated with a low density. This might be because the filler is denser than PVA, which makes it easier to work with. Due to the presence of IONPs, there were minimal variations among bio nanocomposite films. In contrast to pure PVA film’s moisture content of 1.32%, bio nanocomposite films of PVA/IONPs ranged from 1.524% to 1.291%. It is believed that the hydrophobic properties of IONPs are responsible for these outcomes. Although the hydrophilic nature of the film samples was enhanced by the filler concentration, the uneven dispersion of the film samples was not caused by an excessive amount of IONPs.57,58 A low moisture content is achieved in the film samples following the addition of IONPs. According to research on PVA reinforced with clay nanoparticles, adding filler increases water absorption, making the material unfit for use in packaging. The packaging industry may find use for iron oxide nanoparticles due to their minimal moisture absorption. Physical analysis of iron oxide nanoparticles synthesized with spinach extract.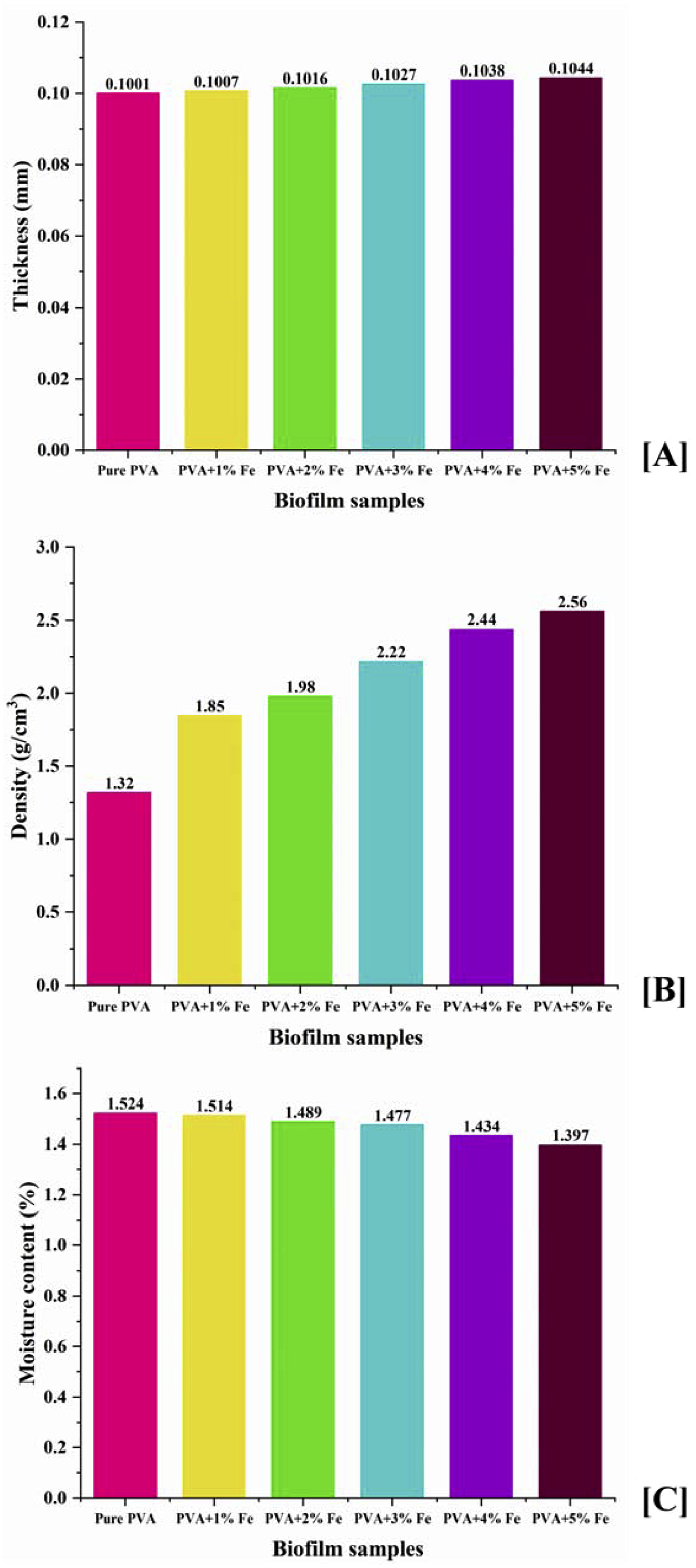
Antimicrobial activity
The antimicrobial activity analysis demonstrates that the 3% IONP composite shows the highest efficacy, which can be explained by the fact that there is an obvious optimum level of loading the spinach-derived IONPs into the PVA matrix. The antibacterial effect has a non-linear shape, which is typical of a sweet spot. Recent studies have shown that the nanoparticle load at lower concentrations (1%,2%) might not be enough to develop a potent antibacterial effect. The optimum concentration at 3% indicates an optimum form of balance where the nanoparticle dispersion and the consequent emanation of the antibacterial ions are maximized. More importantly, the reported decrease in efficacy with higher loadings (4% and 5%), corroborates the assumption that greater agglomeration (as a proven hypothesis by SEM analysis Figure 5) is dominant. This agglomeration minimizes the real surface area of the IONPs which also minimizes interfacial interactions with the bacterial cells. This trend is in agreement with the idea of a percolation threshold in nanocomposites, which occurs beyond which functional properties may become weaker. The spinach iron oxide nanoparticle (IONP) composites are a promising pathway in creating active food packaging. The best 3% IONP film is a biogenic and sustainable solution to suppress microbial growth which improves the food safety and shelf life (Figure 12). Antimicrobial analysis against a) 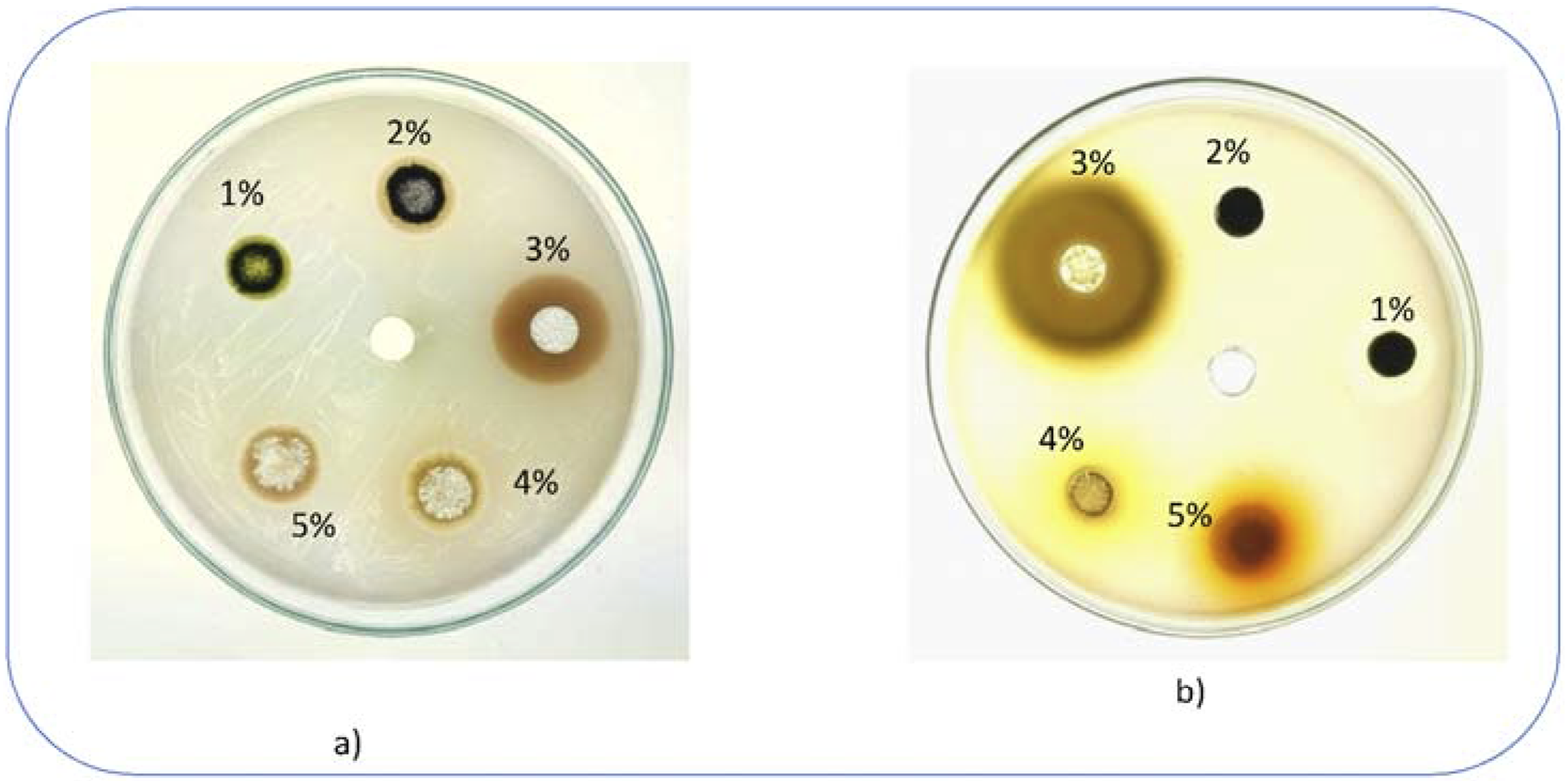
Conclusions
This study conclusively established a green pathway for developing multifunctional packaging films by integrating IONPs synthesized from waste spinach biomass into a PVA matrix. The research successfully produced and characterized IONPs, identifying a 3 wt% loading as the optimal concentration. This optimal composite demonstrated a significant enhancement in antimicrobial efficacy, providing a sustainable solution to inhibit foodborne pathogens. Furthermore, it exhibited a 14% increase in Young’s modulus, a reduction in moisture absorption to 1.29%, and improved thermal stability. By transforming agro-waste into a value-added nanofiller, this work produces a PVA/IONPs biofilm that effectively overcomes the inherent hydrophilicity and lack of antimicrobial activity in pure PVA. The resulting material, with its balanced mechanical integrity, enhanced barrier properties, and active antimicrobial function, is a promising, sustainable candidate for advanced food packaging applications aimed at extending shelf life and improving food.
Footnotes
Acknowledgements
This research was supported by the Sophisticated Testing and Instrumentation Centre (STIC), Department of Mechanical Engineering, Alliance School of Applied Engineering, Alliance University, Bengaluru - 562106, Karnataka, India. In particular, the first author would like to thank Er.K.Suthakar of Rado ChemMAX in Kanyakumari, Tamil Nadu, India, for all of the support he has given during the course of this research.
Author contributions
Kallempudi Murali - Conceptualization, Investigation, Methodology, Writing Original Draft, Visualization and Data Curation, Nirmala Nithya Raju – Resources, Formal analysis, Validation, Writing - Review & Editing, Project Administration, Supervision. Krishna Prakash Arunachalam - Visualization, Investigation, P Senthil Kumar - Writing - Review & Editing, Project Administration, Supervision, Sunesh Narayanaperumal – Formal analysis, Indran Suyambulingam and Nadir Ayrilmis - Writing - Review & Editing, Investigation, Visualization.
Declaration of conflicting interests
The authors declared no competing interest concerning the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
