Abstract
A nanospherical composite based on poly(2-chlorobenzenamine) and silver chloride (POCBA/AgCl NS-composite) was synthesized through a facile one-step, one-pot approach, providing a simple and efficient route to integrate conducting polymers with silver halides for advanced energy storage applications. The composite was thoroughly characterized, with XRD confirming its high crystallinity and semiconducting nature, while XPS validated the successful chemical integration and revealed the electronic environment of the constituent elements. Morphological analyses demonstrated the formation of uniform, porous nanospheres (∼180 nm), which facilitate rapid ion diffusion and effective electroactive surface utilization. Electrochemical evaluation in a pseudo-supercapacitor configuration revealed a high specific capacitance of 105 F g−1 at 0.1 A g−1, retaining 86 F g−1 at 0.2 A g−1, with an energy density of 8.8 Wh kg−1. The device also exhibited low internal resistance (7.5 Ω) and outstanding cycling stability, maintaining 98.3% capacitance over 1000 charge–discharge cycles. Compared to previously reported polymer/silver halide composites, the single-step synthesized POCBA/AgCl NS-composite uniquely combines facile fabrication, high electrochemical performance, and excellent long-term durability, highlighting its potential as a next-generation pseudocapacitive material. This work demonstrates a novel strategy to engineer polymer–metal halide nanostructures, paving the way for efficient, reliable, and scalable energy storage devices.
Keywords
Introduction
The rapid advancement of pseudo-supercapacitors has expanded their potential in energy storage, particularly through reversible Faradaic processes.1–4 Within this field, conducting polymers have emerged as highly attractive electrode materials. In particular, polyaniline-based derivatives stand out due to their unique combination of high electrical conductivity, fast and reversible redox activity, and tunable structural characteristics that enable enhanced charge storage capabilities.2,5,6 However, these materials face several inherent limitations, including poor mechanical integrity and progressive capacity fade under electrochemical cycling, restricting their practical application in high-performance energy storage devices.7,8 Therefore, continued research endeavors are essential to advance conducting polymer-based nanocomposite architectures with enhanced mechanical integrity and superior electrochemical performance characteristics.
Metal halides, particularly silver chloride (AgCl), with its unique properties, are widely proposed as a conductive bridge for conducting polymers9,10 The integration of AgCl nanoparticles with conductive polymer matrices has been explored to address these limitations through the enhancement of charge storage, electrical conductivity, and pseudocapacitive contributions via redox-active sites. For instance, Fathalipour and Massoumi 11 developed AgCl/PANI nanocomposites within a PVA matrix through a two-stage synthetic protocol. The acidic medium proved critical for composite formation, with structural and morphological alterations observed upon HCl removal or substitution with camphorsulfonic acid. The resulting AgCl/PANI/PVA nanocomposite, when immobilized on glassy carbon electrodes, demonstrated enhanced electrocatalytic performance for dopamine oxidation in neutral media. In related work, Zhou et al. 10 fabricated a PANI/AgCl hybrid electrochemical sensor demonstrating superior electrocatalytic performance for dopamine detection. Electrochemical characterization via cyclic voltammetry exhibits that the PANI/AgCl/GCE electrode exhibited significantly reduced oxidation overpotential and enhanced peak currents compared to individual AgCl/GCE or the pristine GCE electrodes, attributed to synergistic interactions between the organic polymer matrix and inorganic AgCl nanoparticles.
Conventional methodologies for polymer-inorganic composite fabrication are characterized by prolonged reaction times, extensive purification requirements, and potential for inadequate interfacial adhesion between constituent phases.12–14 On the other hand, single-step synthetic methodologies provide advantageous processing efficiencies through optimized interfacial coupling, enhanced thermodynamic stability, and superior batch-to-batch reproducibility. These integrated synthesis approaches facilitate economically viable scale-up pathways for high-performance electrode architectures with industrial manufacturing potential.
Recent advances in this field demonstrate the potential of such integrated approaches. For example, Raiz et al. 12 developed KCuCl3/PANI halide perovskite composites via solvothermal and antisolvent methodologies, demonstrating electrochemical properties in 1.0 M KOH. Poudel et al. 13 fabricated polyaniline-encapsulated silver-decorated manganese dioxide (PANI/Ag@MnO2) nanorods for application as positive electrode materials in supercapacitor systems. The results demonstrated superior electrochemical activity of the PANI/Ag@MnO2 nanocomposite compared to binary Ag@MnO2 and pristine MnO2 electrodes. Also, several polyaniline and derivatives-based composites have been explored for supercapacitor applications, including polyaniline/WO3, polyaniline/ZnO-ZrO2, polymethylaniline/Ag-Ag2O, polyaniline/TiO2, poly m-toluidine/NiO, polyaniline/Fe2O3, or poly-O-chloroaniline/Se nanocomposites. These materials have exhibited significant improvements in capacitance, conductivity, and stability compared to pure polyaniline or metal oxides alone. Despite extensive research on these composite materials, no studies have reported on the development of silver halide/poly-o-chlorobenzeneamine nanocomposites for supercapacitor applications.
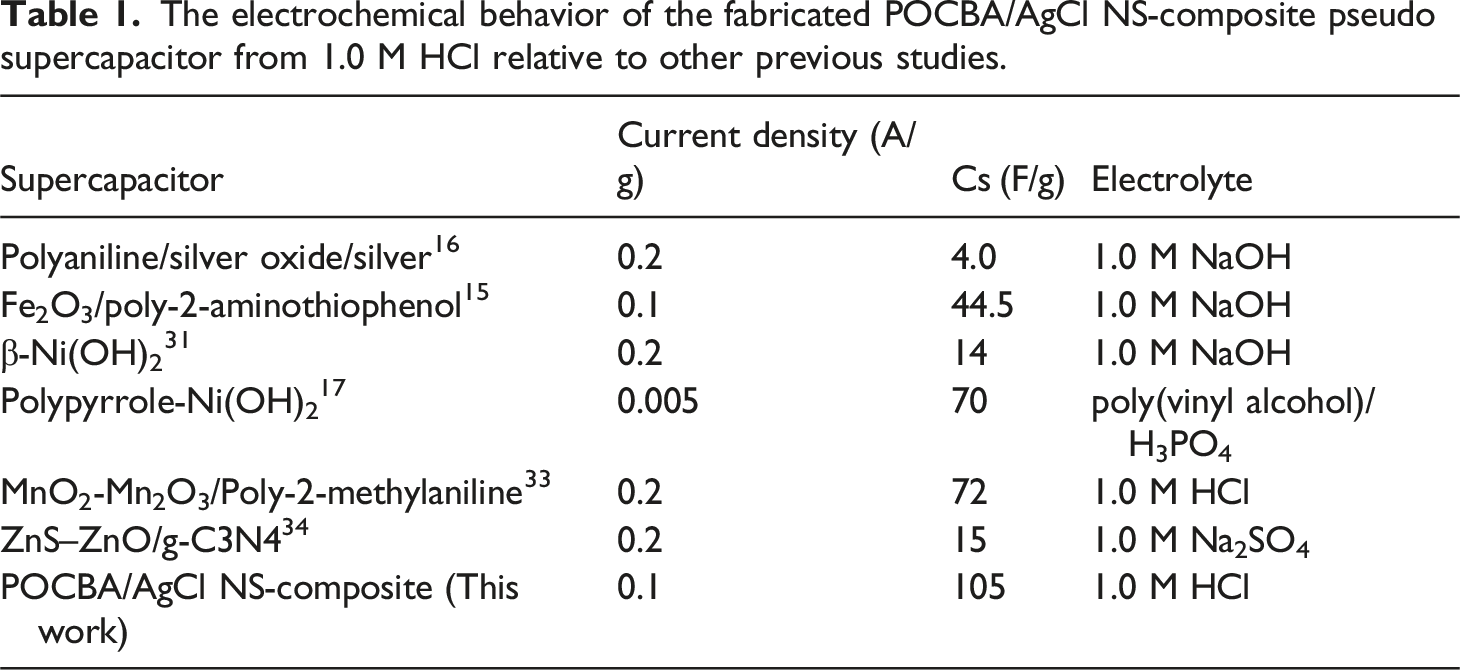
Examples of Cs values include 44.5 F/g, which was obtained for Fe2O3/poly-2-aminothiophenol 15 when NaOH was used as the electrolyte. In contrast, other studies reported Cs values of less than 20 F/g for polyaniline/silver oxide/silver composites. 16 Additionally, the Polypyrrole-Ni(OH)2 composite 17 demonstrated a higher capacitance, reaching 70 F/g.
Recent studies have highlighted the exceptional conductivity and electrochemical properties of silver-based materials, including silver oxides and halides. Silver oxide nanoparticles, in particular, exhibit several properties, such as a porous structure, excellent thermal stability, and favorable wettability with storage behavior. These features make silver a highly suitable electroactive material for supercapacitors. Furthermore, silver halide, known for its semiconducting behavior and chemical stability, can provide the electrochemical feature of the material composites to charge transfer kinetics, and provide additional redox-active sites.
Herein, we report, for the first time, the successful synthesis of a novel nanospherical composite of poly(2-chlorobenzenamine) and silver chloride (POCBA/AgCl NS-composite) via a simple one-pot approach, and its application as a high-performance pseudocapacitive electrode. The physicochemical and morphological properties of the composite were systematically characterized using XRD, XPS, FT-IR, and SEM, confirming its crystallinity, chemical composition, and uniform nanosphere morphology. The electrochemical performance was comprehensively evaluated through cyclic voltammetry (CV), electrochemical impedance spectroscopy (EIS), and galvanostatic charge–discharge (GCD) measurements, demonstrating the composite’s efficient charge storage behavior and enhanced pseudocapacitive characteristics. This study highlights a novel strategy to combine conducting polymers with silver halide nanostructures in a single-step process, offering a promising route toward next-generation, high-performance pseudocapacitive energy storage materials.
Experimental section
Materials
O-chlorobenzenamine (99.9%) and hydrochloric acid (36%) were obtained from Merck, Germany. Acetic acid (CH3COOH, 99.9%), silver nitrate (AgNO3, 99.9%), graphite powder (99.9%), and ammonium persulfate ((NH4)2S2O8, 99.9%) were supplied by Pio-Chem, Egypt. Nafion solution (5% in methanol) was purchased from Sigma-Aldrich, USA, and used as the binder during electrode fabrication.
One-pot synthesis of POCBA/AgCl NS-composite
The synthesis of the POCBA/AgCl NS-composite is performed using a simple one-pot direct oxidation of O-chlorobenzenamine using AgNO3 and (NH4)2S2O8. Initially, 0.06 M of the monomer O-chlorobenzenamine is thoroughly dissolved in 0.8 M acetic acid. Concurrently, separate solutions are prepared with 0.07 M AgNO3 and 0.07 M (NH4)2S2O8. These solutions are then sequentially poured over the monomer solution, with AgNO3 being introduced first. This step is crucial as it initiates the formation of AgCl, followed by the simultaneous polymerization process through which the one-pot synthesis of the POCBA/AgCl NS-composite is estimated.
Pseudosupercapacitor device fabrication
The fabrication of a pseudocapacitor based on the POCBA/AgCl NS-composite involves creating a paste cast on a carbon sheet (0.5 mm thick). This paste is prepared by dissolving the nanocomposite in ethanol and adding Nafion in quantities of 0.75 and 0.1 mL, respectively. After stirring this mixture at 1000 r/min for 2 days, the resulting paste is ready for application. It is then spread onto the carbon sheet and dried for 2 h at 40°C.
For the electrochemical measurements of the pseudocapacitor, CHI608 E is used. Besides the carbon sheet with the nanocomposite paste acting as the working electrode, the setup includes an additional carbon rod and calomel electrodes. The cyclic voltammetry is performed at 0.0 to 1.0 V. Additionally, the chronopotentiometry test is performed to evaluate the charge/discharge behavior of the pseudocapacitor (Figure 1. The CS of the pseudocapacitor is calculated using equation (1). Key factors in this estimation include the mass of the paste applied to the carbon sheet, which is 0.003 g, and the measurement of Δt and ΔV for the discharge time voltage, respectively. These values are essential for determining the Cs, which represent the capacitance per unit mass of the active material. The procedure of synthesis of POCBA/AgCl NS-composite and the electrochemical estimation of the pseudo supercapacitor device through a three-electrode cell.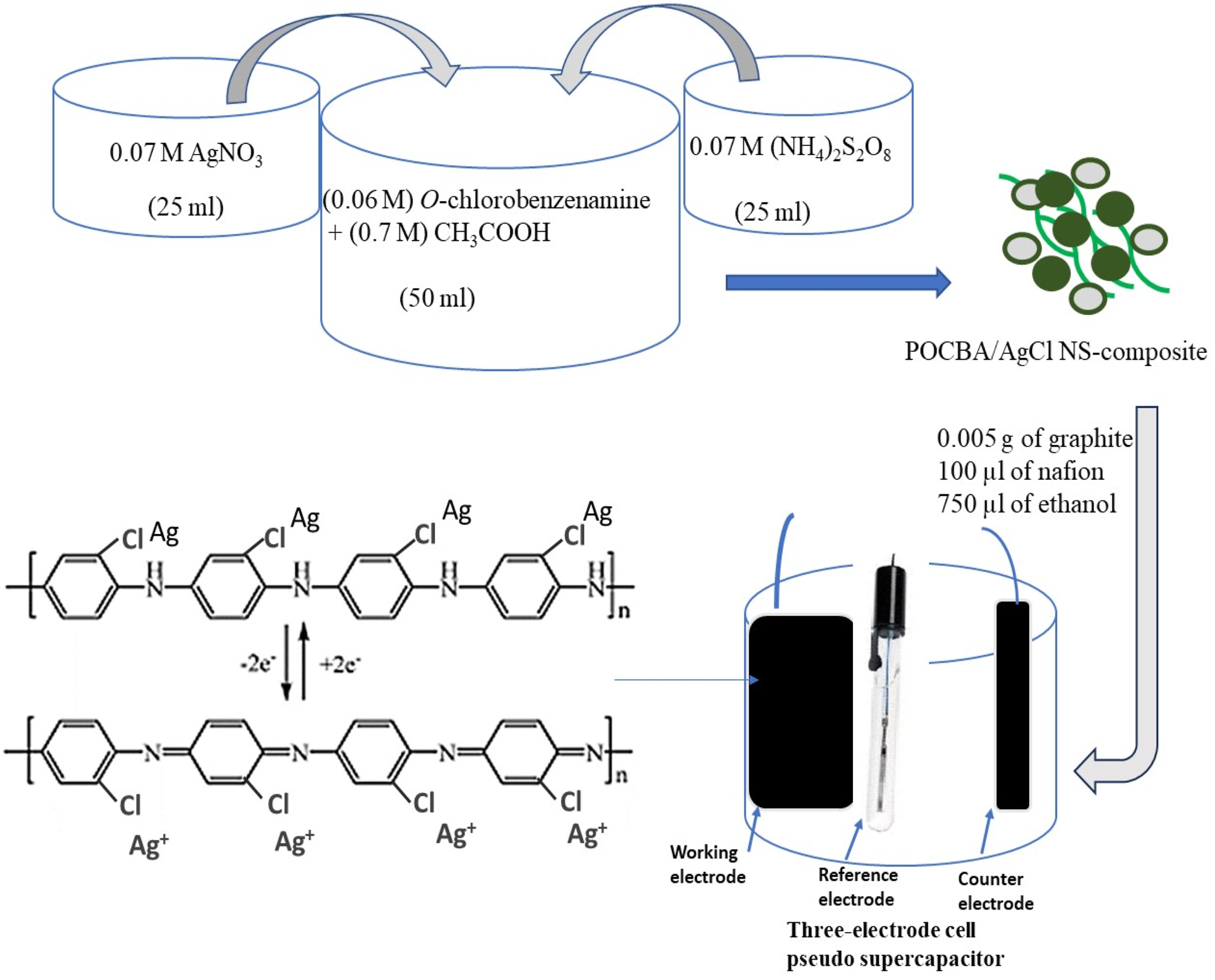
Once the CS is known, the E value of the pseudocapacitor can be estimated using equation (2). This equation used
Results and discussion
Analyses
The chemical structure and crystalline behavior of the synthesized POCBA/AgCl NS-composite, compared to the pristine POCBA, were evaluated using XRD analyses, as exhibited in Figure 2(a). The significant differences between these two materials are evident through the highly defined, sharp peaks observed in the composite’s XRD pattern, indicating superior chemical properties. These sharp peaks suggest a high degree of crystallinity, which in turn implies an enhanced ability of the composite to store charge within its crystalline structures. The chemical analyses for the synthesized POCBA/AgCl: (a) XRD, (b) XPS for Ag element, (c) XPS survey, and (d) FTIR.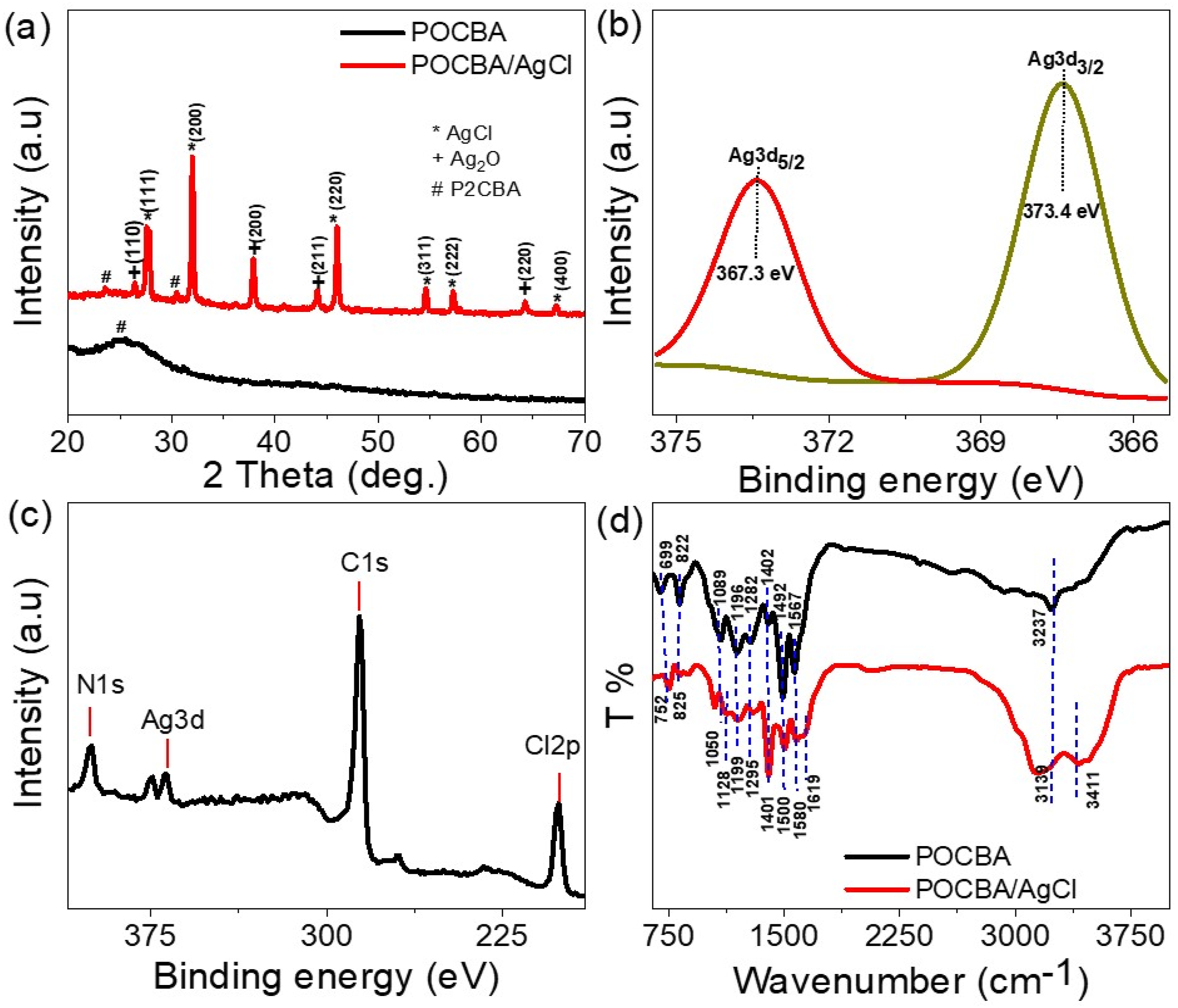
The XRD analysis evaluated the peaks at 2θ values of 27.6°, 32.1°, 45.9°, 54.6°, 57.3°, and 67.4°, for (111), (200), (220), (311), (222), and (400) of AgCl, respectively. 18 Additionally, other peaks were identified, corresponding to Ag2O at 26.3°, 37.8°, 44.0°, and 64.2°, for (110), (200), (221), and (220), respectively. Although these peaks are present, their intensities are lower compared to those of the AgCl-related peaks, highlighting that AgCl is the dominant crystalline phase in the composite. While the pristine POCBA exhibited a broad peak in its XRD pattern, indicative of its amorphous nature. After forming the composite, additional peaks of crystalline structures emerged, reflecting an increase in crystallinity within the polymer matrix.
The crystalline size (D) of the POCBA/AgCl NS-composite is evaluated using equation (4).19,20 Based on the peak at 2θ = 45.9°, the calculated D was found to be 43 nm. This calculation, which considers the full-width half maximum (β) of the peak, underscores the excellent properties of the composite. The presence of small crystals within the composite is particularly beneficial for charge storage, as the increased surface area and crystallinity facilitate enhanced electrochemical performance.
The oxidation states of the elements within the POCBA/AgCl NS-composite were determined through XPS analyses (Figure 2(b)) for the Ag element and Figure 2(c) for the survey of the POCBA/AgCl NS-composite. For the Ag element, the binding energies for Ag3d5/2 and Ag3d3/2 were observed at 367.3 eV and 373.4 eV, respectively. 21 This indicates the silver inside the composite. Additionally, the presence of chlorine within the composite is confirmed by a characteristic peak at 200 eV, indicating the formation of AgCl. The survey also revealed other elements inherent to the composite. Specifically, nitrogen (N) and carbon (C) were evaluated at 400 eV and 286 eV, respectively, which correspond to their s orbitals. Figure 2(d) presents the FT-IR spectroscopic analysis of POCBA/AgCl nanospheres and pristine POCBA composite. The POCBA spectrum exhibits distinctive vibrational modes at 3237 cm−1 for N-H. Characteristic bands at 1567 cm−1 and 1492 cm−1 are for C = C and C = N of quinonoid and benzenoid ring structures,22,23 respectively. Absorption features spanning 1402-1196 cm−1 are attributed to aromatic C-N stretching vibrations characteristic of aromatic amine linkages. Supplementary bands at 1089 cm−1 and 822 cm−1 are assigned to N = Q = N (quinonoid) and C-H out-of-plane deformation vibrations typical of disubstituted benzene rings. The one at 699 cm−1 is for the C-Cl group originating from the chloro-substituted aromatic moieties.10,24 These vibrational fingerprints substantiate the successful polymerization of the 2-chlorobenzenamine matrix. Significantly, the fundamental vibrational frequencies of POCBA exhibit bathochromic shifts in the POCBA/AgCl composite, indicating intermolecular interactions between the polymeric backbone and AgCl nanoparticles during composite formation. Additionally, the composite spectrum reveals bands at 3411 cm−1 and 1619 cm−1, attributed to O-H and H-O-H modes of physisorbed water molecules, respectively.25,26
The consistency between the XPS data and the XRD results validates the POCBA/AgCl NS-composite chemical structure. The XPS results provide detailed insights into the composition and oxidation states, complementing the crystallographic information obtained from XRD. Together, these analyses confirm the formation and composition of the POCBA/AgCl NS-composite.
The morphology of the POCBA/AgCl NS-composite was examined using SEM (Figure 3(a)–(b)), revealing a uniform porous spherical (PS) structure with an average particle diameter of ∼200 nm that consists of smaller particles. This nanospherical and porous architecture plays a crucial role in the composite’s electrochemical performance. The high surface area associated with the porous spheres provides a large number of accessible redox-active sites, facilitating efficient faradaic reactions during charge–discharge cycles. Additionally, the interconnected pores and nanoscale dimensions significantly enhance ion diffusion, allowing electrolyte ions to rapidly penetrate and interact with the interior of the spheres, which improves charge storage kinetics and overall pseudocapacitance. The SEM of POCBA/AgCl NS-composite: (a-b) SEM at different scal bare, and (d) theoretical cross-section of this composite surface. While (c) SEM of pristine POCBA.
In contrast, pristine POCBA (Figure 3(c)) displays a fibrous morphology, with fibers extending over 1000 nm in length and 20–150 nm in thickness. While these fibers possess intrinsic surface activity, their larger dimensions and less porous structure limit ion transport and reduce accessibility to redox sites compared to the nanospherical composite.
Cross-sectional SEM images of the POCBA/AgCl NS-composite (Figure 3(d)) further reveal that smaller nanoparticles (∼20 nm) aggregate to form larger porous spheres (∼200 nm). The rough surfaces and abundant pores indicate the effective integration of inorganic AgCl within the polymer matrix, creating numerous active sites and short diffusion pathways that together enhance electrochemical charge–discharge efficiency.27,28
The TEM image reveals the formation of the POCBA/AgCl nanocomposite particles, which exhibit a porous, spherical morphology with diameters ranging from approximately 10 to 100 nm, as estimated in Figure S1. Notably, the larger spherical structures are composed of an assembly of numerous smaller nanoparticles, indicating a hierarchical architecture within the composite.
The pseudosupercapacitor testing
The performance of the POCBA/AgCl NS-composite pseudo supercapacitor estimates the stored charges within the composite paste. This charge storage process relies on the redox behavior of AgCl and POCBA, which can undergo redox reactions involving electron acceptance or loss. Through these redox reactions, charges are inserted and stored. The electrochemical reactions are conducted using a three-electrode cell setup, with the fabricated NS-Composite/graphite sheet (working electrode). The energy storage capacity is assessed using a highly polarized HCl solution, which facilitates the movement of H+ ions during the redox reactions. This setup allows for efficient estimation of the composite’s energy storage performance.
Figure 4(a) demonstrates the behavior of the fabricated pseudo supercapacitor, showcasing its capability to either store charges or discharge them as electrons across a range of 0.1 to 0.5 A/g. The data reveal that the discharge time increases with notable improvements in discharge time observed at lower current densities. Specifically, discharge times of 210 s and 84 s were achieved at 0.1 and 0.2 A/g, respectively. These impressive results highlight the device’s ability to store charges for extended periods related to the highly active surface area of the POCBA/AgCl NS-composite used in its construction. This behavior is further analyzed using cyclic voltammetry, as depicted in Figure 4(b). The cyclic voltammetry results exhibit prominent redox peaks on both the positive and negative sides, indicating robust redox activity in the fabricated pseudo-supercapacitor. The intensity of these peaks increases with higher scan rates from 50 to 300 mV/s. This enhancement in peak values is a testament to the POCBA/AgCl NS-composite’s ability to respond effectively to potential changes for charge storage and release under negative potentials. The electrochemical performance of the POCBA/AgCl NS-composite pseudo supercapacitor through the (a) charge-discharge and (b) cyclic potentiometry.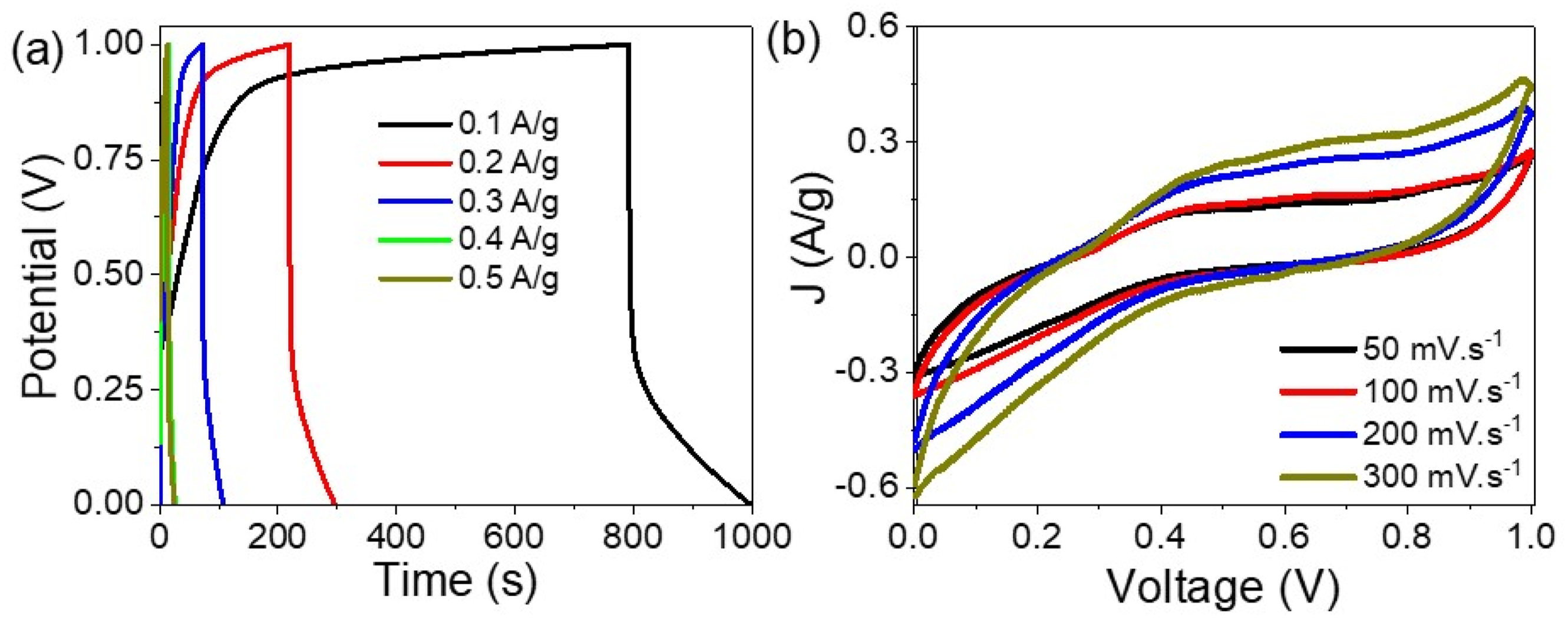
The enhanced discharge times at lower current densities, along with the significant redox peaks observed during cyclic voltammetry, underscore the high performance of the POCBA/AgCl NS-composite pseudo supercapacitor. The material’s porous structure promotes charge accumulation, contributing to the prolonged charge retention capabilities of the device. Furthermore, the cyclic voltammetry analysis highlights the dynamic redox behavior of the supercapacitor29,30 increasing scan rates lead to higher peak intensities, which in turn demonstrate the composite’s responsiveness to potential variations.
Overall, the combination of long discharge times, significant redox peaks, and the highly active surface area of the POCBA/AgCl NS-composite indicates that this fabricated pseudo-supercapacitor is a great device for efficient energy storage utilization. The findings presented in Figure 4(a) and (b) collectively illustrate the superior performance characteristics of the device, driven by the advanced materials and structural design employed in its construction. This enables the supercapacitor to effectively store and discharge energy, making it suitable for a variety of practical applications where long-term charge retention and efficient energy management are critical.
Figure S2 presents the galvanostatic charge–discharge profiles of the pristine POCBA polymer, employed as a pseudo-supercapacitor electrode, measured at varying current densities (0.1, 0.2, 0.4, and 0.5 A g−1). The potential is plotted as a function of time during the charging and discharging cycles. At the lowest current density of 0.1 A g−1, the POCBA electrode exhibits a specific capacitance of 12 F g−1. Upon incorporation of AgCl, the POCBA/AgCl nanocomposite demonstrates notably longer discharge durations and higher specific capacitances at the same current densities. This improvement is attributed to the enhanced electron transport and the introduction of additional redox-active sites provided by AgCl, highlighting the synergistic effect of the composite in boosting electrochemical performance.
The behavior of the fabricated POCBA/AgCl NS-composite pseudo supercapacitor is assessed using Figure 5(a) and equation (1) to determine the produced CS values related to the redox reaction and subsequent charge accumulation in the nanocomposite. The optimal CS is observed at 0.1 A/g, reaching 105 F/g, and then decreases to 46.0 F/g at 0.2 A/g. This provides a great indication that the supercapacitor effectively stores charges at lower current densities, as a result of the strong response of the AgCl and POCBA oxidation states at these low currents. The semiconducting properties of silver chloride (AgCl) play a crucial role in enhancing both the capacitance and stability of the POCBA/AgCl NS-composite. AgCl provides electronically conductive pathways within the polymer matrix, which facilitates rapid electron transport during charge–discharge cycles. This improves the utilization of redox-active sites in the poly(2-chlorobenzenamine) polymer, thereby increasing the overall pseudocapacitance. Additionally, AgCl contributes to the structural stability of the composite. Its rigid inorganic framework helps maintain the integrity of the nanospherical architecture during repeated cycling, reducing polymer swelling or degradation that typically limits long-term performance. The combination of enhanced electron conductivity, abundant active sites, and mechanical reinforcement from AgCl ensures efficient charge storage and fast ion diffusion. Thus, the semiconducting AgCl acts both as an electronic facilitator and a structural stabilizer, synergistically improving the electrochemical performance of the POCBA/AgCl NS-composite electrode. The POCBA/AgCl NS-composite pseudo supercapacitor efficiency: (a) CS, (b) E, (c) P, and (d) Ragone plot.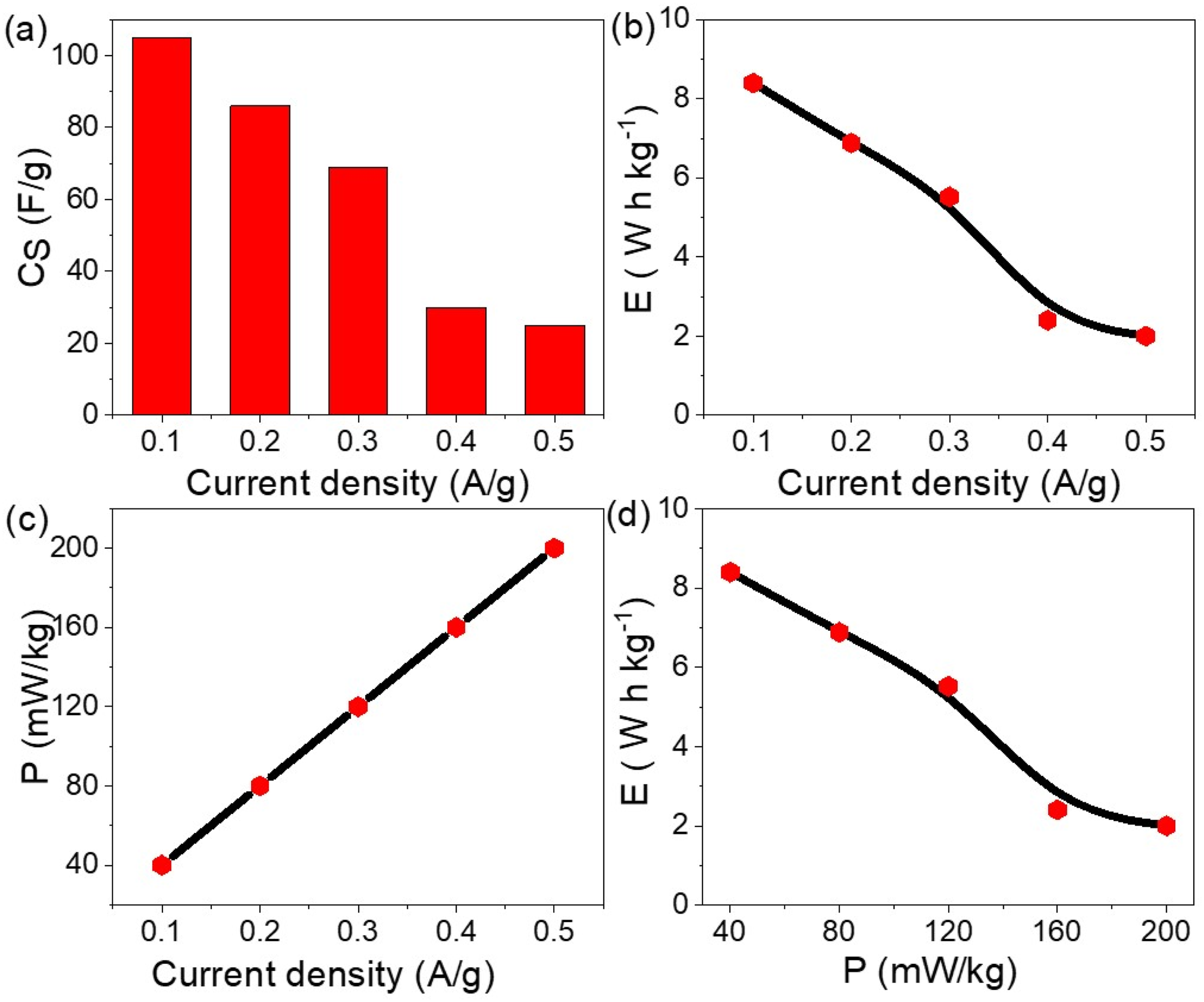
Similarly, the energy (E) values exhibit high performance at lower current densities, with a peak value of 8.8 W h/kg at 0.1 A/g. This behavior is described by equation (2), where the maximum potential values are the key factor for this evaluation. As the current increases, the evaluated energy decreases to 2.2 W h/kg at 0.5 A/g. Despite this decrease, it remains a good value for charge storage, demonstrating the device’s capability to release stored charges when connected to other high-technology devices.
The power (P) value is also crucial, with the pseudo supercapacitor delivering 200 mW/kg at 0.5 A/g, as shown in Figure 5(c) and calculated using equation (3). To further understand the charge storage and release behavior, a Ragone plot is presented in Figure 5(d).
These findings are further supported by the Ragone plot, showcasing the device’s potential for practical applications. The comprehensive analysis highlights the advanced performance of the POCBA/AgCl NS-composite pseudo supercapacitor, evaluating its energy storage and release in high-technology devices. With superior charge storage compared to previous polymer composites, such as polyaniline/Ag2O, 16 G-C3N4, 31 and Fe2O3/poly-2-aminothiophenol, 15 which have CS values of about 4 to 40 F/g.
To estimate the behavior of the POCBA/AgCl NS-composite pseudo supercapacitor, we examine the charge transfer and the ability of POCBA and AgCl materials to undergo redox reactions in response to the graphite supporting materials. This assessment is depicted in the Nyquist plot (Figure 6(a)), which reveals a Rs of 7.5 Ω and a charge transfer resistance (RCT) of 24 Ω. The small diameter of the semi-circle indicates excellent performance, reflecting efficient charge transfer.30,32 The estimated (a) Nyquist plot and (b) the retention stability for the fabricated POCBA/AgCl NS-composite pseudo supercapacitor from 1.0 M HCl.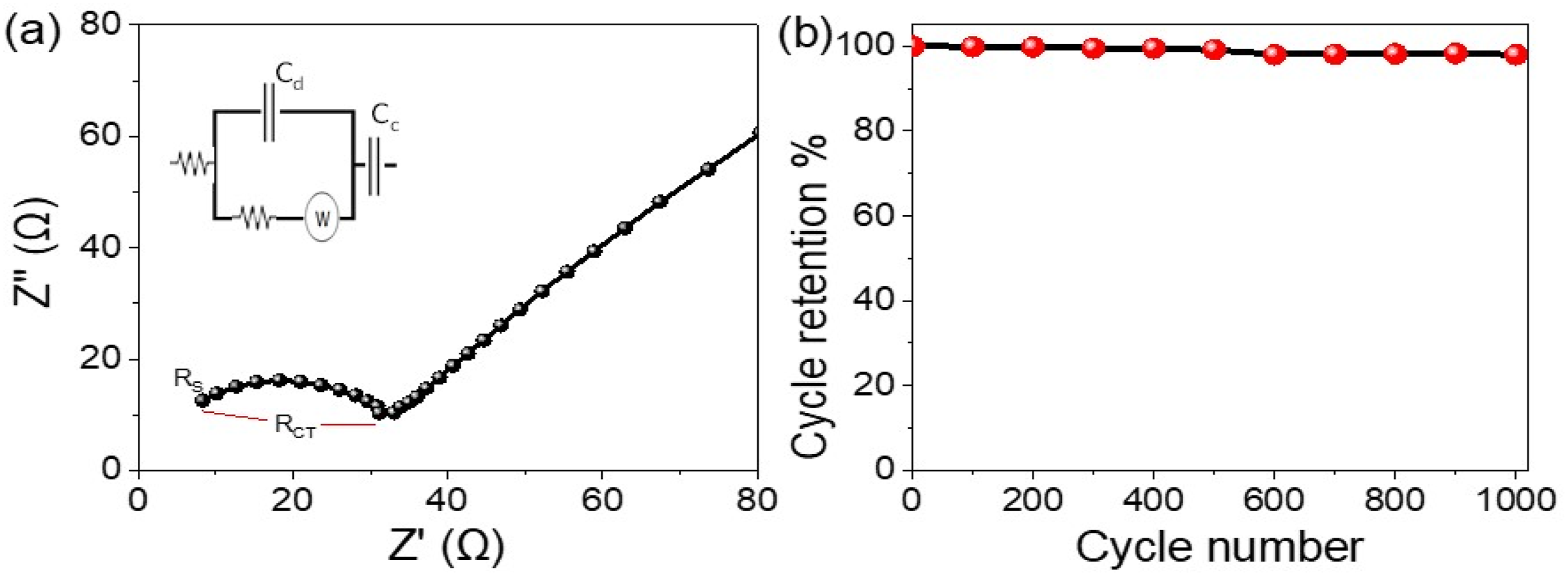
Additionally, this device’s stability is remarkable, retaining 98.3% of its capacity after 1000 cycles. This high stability is attributed to the efficient charge transfer during the composite materials’ oxidation and reduction processes. The chemical inclusion of AgCl into the polymer network adds synergistic storage for the capacitor’s longevity, benefiting from the polymer’s excellent anticorrosion properties and high electrical conductivity in protonic acid. The use of HCl significantly improves the EIS results and cycle retention.
The novel POCBA/AgCl NS-composite pseudo supercapacitor demonstrates outstanding performance. It combines mass production potential with low-cost manufacturing techniques.
The electrochemical behavior of the fabricated POCBA/AgCl NS-composite pseudo supercapacitor from 1.0 M HCl relative to other previous studies.
Conclusions
In this study, a POCBA/AgCl nanospherical composite was successfully synthesized through a simple one-pot approach and thoroughly characterized to evaluate its structural and electrochemical features. XRD confirmed its high degree of crystallinity, with an estimated crystallite size of about 63 nm, reflecting the material’s promising semiconducting behavior. Microscopic examinations using SEM and TEM, supported by theoretical insights, demonstrated that the composite forms a porous spherical architecture with an average particle diameter of roughly 180 nm, providing a large surface area favorable for charge transfer and storage. Electrochemical investigations revealed that the fabricated material functioned effectively as a pseudo-supercapacitor. At a current density of 0.1 A g−1, it delivered a specific capacitance of 105 F g−1, along with an energy density of 8.8 Wh kg−1, indicating strong charge storage capability. More importantly, the electrode exhibited exceptional durability, retaining 98.3% of its capacitance after 1000 continuous charge–discharge cycles, emphasizing its excellent structural integrity and cycling stability. Overall, the superior electrochemical performance, high stability, and facile synthesis route highlight the POCBA/AgCl nanospherical composite as a promising candidate for next-generation energy storage systems, offering great potential for practical and scalable applications in commercial supercapacitor technologies.
Supplemental Material
Supplemental Material - Single-step fabrication of poly(O-chlorobenzenamine)/silver halide nanosphere composite for pseudocapacitive energy storage
Supplemental Material for Single-step fabrication of poly(O-chlorobenzenamine)/silver halide nanosphere composite for pseudocapacitive energy storage by Mohamed Rabia, Farid Fadhillah, Ahmed A. Abdel-Khalek, Amine Aymen Assadi, Abdulziz I Alromaeh, Saba A. Aladeemy in Journal of Thermoplastic Composite Materials.
Footnotes
Author contributions
Mohamed Rabia: Experimental, writing, and analysis.
Farid Fadhillah: Writing, analysis, and Funding
Ahmed A. Abdel-Khalek, Amine Aymen Assadi, Abdulziz I Alromaeh, and Saba A. Aladeemy: Revision, supervision, and ordering the work.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported and funded by the Deanship of Scientific Research at Imam Mohammad Ibn Saud Islamic University (IMSIU) (grant number IMSIU-DDRSP2503).
Ethical considerations
This study does not include any human or animal studies.
Data Availability Statement
All data generated or analyzed during this study are included in this article
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.




