Abstract
Additive manufacturing of bone scaffolds enables the fabrication of patient-specific inter-connected porous structures that facilitate tissue generation with closely matched mechanical and biological properties of natural bone. This study examines how TPMS architecture, porosity, and material composition impact the compressive strength and stiffness of 3D printed PLA and PLA/nHA scaffolds for bone repair. Filaments are produced via a single-screw extrusion system and 3D printed into ASTM D695 specimens with 50%, 60%, and 70% porosity. Compression tests evaluated the effects of TPMS designs, porosity, and material composition on strength and elastic modulus. IWP scaffolds exhibited the highest mechanical properties, while FRD showed the lowest. The addition of nHA increased the compressive strength by 7.76% and the elastic modulus by 24.79%, with improved crystallinity shown in the XRD analysis. However, SEM images showed the formation of pores in the composite filaments, which may act as stress concentrators and limit the mechanical performance. ANOVA results confirmed TPMS design and porosity as the most significant factors influencing mechanical performance, while material composition had a lesser effect, particularly on compressive strength. TPMS-based PLA/nHA scaffolds show strong potential for bone regeneration, with IWP and gyroid structures exhibiting higher mechanical strength even at high porosity levels.
Introduction
Bones are dynamic tissues which undergo remodelling where old bone is resorbed by osteoclasts and new bones are formed by osteoblasts. 1 Bones are those specialized composites made up of approximately 60% collagens and 40% hydroxyapatite. They provide exceptional overall body strength and perform key functions such as structural support for movement, protecting vital organs and regulating the mineral balance of the body.2,3 The ability of bone cells to attach to the bone surface is essential for their development and function, demonstrating the essential connection between bone tissue and human cells. The demand for more advanced treatment options than the conventional autograft and allograft techniques has increased due to the rise in bone related injuries and degenerative diseases.4,5 The challenges include donor site restrictions, immunological rejection, and infections. Regenerative medicine stimulates the body’s inherent healing processes to repair damaged tissues using biological processes. Therefore, regenerative medicine has become a viable alternative for bone restoration and repair. 6 The technological advancements in the manufacturing sector have widened the scope of bone tissue engineering for promoting regenerative medicine. Additive manufacturing is a popular technology which can fabricate patient-specific implants overcoming the above-mentioned limitations. 7 Drug delivery, surgical guides, implants, surgery pre-planning, and training are other possible scopes of 3D printing in bone tissue engineering. 8
The creation of bioengineered scaffolds that replicate the composition and functionality of natural bone is one of its most prominent applications. These scaffolds offer a short-term structure for tissue regeneration, cell adhesion, and proliferation and over time, new bone gradually replaces them.9,10 There are many drawbacks to the traditional methods of producing bone scaffolds, including electrospinning, gas foaming, salt leaching, hydrogel creation, phase separation, freeze-drying, and cryogelation. This includes insufficient connectivity, difficulty regulating the dimensions of pores and design, limited reproducibility, and the application of toxic solvents.11–13 Additive manufacturing provides accurate control over the size and design of bone lattices, boosting its potential in biomedical fields. Different lattice architectures, including strut-based layouts, triply periodic minimal surface (TPMS) designs, and Voronoi lattices, are being used to manufacture bone scaffolds.14,15 The scaffolds for tissue engineering applications must have essential requirements such as: • Biodegradable and biocompatible – It must break down within the body without causing any toxic effects ensuring new tissue regeneration. • Osteoinductive – It should encourage stem cells to differentiate into osteoblasts promoting bone growth. • Porosity and interconnected architecture – It is essential to maintain a porous structure with interconnectivity to ensure cell adhesion, nutrient diffusion and vascularization. • Good mechanical strength – The scaffold must possess sufficient structural strength to withstand external forces and impact. • Non-toxic – It should not release harmful substances that could damage surrounding tissues or organs.
16
Rahatuzzaman et al. 17 demonstrated the design and manufacturing of polylactic acid (PLA) and acrylonitrile butadiene styrene (ABS) scaffolds with a two lay-down pattern mimicking bone structure. The results showed a better scope for PLA in bone tissue engineering applications with a maximum compressive strength value of approximately 134 MPa for a 0/90° pattern. ABS exhibited an ultimate compressive strength value of approximately 91 MPa for a 0/90° pattern. Bakhtiari et al. 18 investigated the compressive and fatigue properties of gyroid structured PLA with 60% porosity. Nozzle temperature had the highest influence on the fatigue resistance. They obtained the highest compressive strength of 7 MPa and modulus of elasticity of 215 MPa for the extrusion thickness of 0.65 mm. Gremare et al. 19 investigated the effects of pore sizes on mechanical properties and cellular growth on PLA scaffolds. The study showed excellent mechanical strength and cellular growth independent of pore sizes. Although PLA is an approved material for biomedical applications due to its biodegradable and biocompatibility properties, its inert characteristics limit human cell growth hindering its application in bone regenerative studies. Dukle et al. 20 proposed two effective approaches to overcome the above-mentioned limitation by either incorporating bioceramic reinforcements into the polymer matrix or coating the polymer surface with biologically active substances such as ceramics and biopolymers. Various studies have examined the effects of incorporating different fillers into the polymer matrix for bone scaffolding applications. The influence of the addition of magnesium hydroxide to PLA is studied in 21. The tensile and compressive strength enhanced by approximately 21% and 64% respectively with the addition of 5 wt% magnesium hydroxide powders to PLA. The composite also showed better cellular responses due to the release of magnesium ions. Keshtiban et al. 22 examined the mechanical and biological characteristics of graphene oxide coated fused filament fabricated PLA for bone scaffolds. Although the results showed lower mechanical strength for TPMS structures with graphene oxide coatings, there was an improvement of 220% in cell viability during dynamic cell culture investigations.
Ceramic powders are commonly combined with polymers to create rigid and tough composite bone scaffolds that offer improved cellular responses. The most used ceramics are hydroxyapatite (HA) and tricalcium phosphate (TCP).23–25 These materials typically degrade at a slower rate, preserving their structural integrity throughout the process of bone regeneration. Heo et al. 26 investigated the benefits of incorporating HA powders into polycaprolactone (PCL) bone scaffolds. They conducted a comparative study to evaluate the performance of both nano and micro-HA powders in PCL scaffolds. The PCL/nHA scaffold exhibited a compressive strength of approximately 3.2 MPa, while the micro powders resulted in a strength of approximately 1.4 MPa. The hydrophilic properties of the PCL scaffolds were also improved by incorporating nHA which facilitated enhanced cell attachment and differentiation. The compressive strengths of a 3D printing polyvinylidene fluoride (PVDF) composite (90% PVDF, 8% HA, 2% Chitosan by weight) produced through fused filament fabrication for application in partially absorbable canine implants were studied in 27. Emadi et al. 28 developed a good substitute material for bone scaffolds by adding Baghdadite powders into PCL polymers. They demonstrated a faster degradation rate and better bone regeneration with the composite material. Sahmani et al. 29 developed calcium phosphate (CaP) incorporated PLA bone scaffolds with cubic, honeycomb and spherical cellular architecture. Cubic samples dipped in body fluids demonstrated maximum permeability whereas honeycomb lattices showed least permeability. PLA/HA bone scaffolds promote cell growth without osteogenic stimuli and the degradation studies demonstrated that HA can regulate the acidic environment created by PLA breakdown, which in turn facilitates the release of calcium and phosphate ions. 30 The addition of carbon nanotube powders into PLA was successfully demonstrated with good compressive strength and cell growth. 31
Namasivayam et al. 32 examined PLA/HA composite filaments developed for biomedical applications by material extrusion 3D printing. The filaments were also heat-treated to evaluate the effect of heat treatment on mechanical performance. Mechanical characterization of ASTM standard samples indicated that PLA/HA showed better properties than standard PLA due to enhanced crystallinity. PLA/HA and PLA filaments were also used to fabricate and evaluate bone plates based on ASTM F543 standards, establishing their feasibility for real-time biomedical and orthopaedic implant applications. The reinforcement mixture composed of 4.97 wt% HA was selected after experimental confirmation, since it possessed better mechanical properties. This result is proof for the existence of a critical threshold, after which an increase in HA content adversely affects mechanical performance. The wettability, wear behaviour, and morphology of 3D printed PLA composites reinforced with molybdenum disulfide (MoS2) and silicon carbide (SiC) were investigated in 33. The minimum specific wear rate was 0.00,141 g/Nm for the samples printed using filaments manufactured at 160°C and 7 r/min screw speed and printed at 210°C and an infill density of 40%. The contact angle tests revealed that the reinforcement with MoS2-SiC increased the surface roughness and promoted hydrophilicity.
Qin et al. 34 investigated the rheological and mechanical properties of calcium carbonate (CaCO3) and tricalcium phosphate (TCP) infused PLA lattice structures. Cubic and TPMS diamond lattices were selected for the comparative study. The TPMS diamond lattice demonstrated higher load bearing capacity and energy absorption efficiency than the cubic structure. Deformation analysis showed that the pore structure of the diamond lattice remained intact while the cubic structure failed as vertical struts fractured. TPMS lattice structures are those with a high surface-to-volume ratio enabling higher permeability for bone tissue engineering applications. 35 Gid et al. 36 studied the mechanical behaviour of polyether ether ketone (PEEK) diamond and gyroid lattices with 50% porosity. Cai et al. 37 examined the effect of porosity on the compression properties of PLA diamond TPMS structures. They found that low porosity designs had yield strengths three times higher and moduli 2.5 times greater than high porosity designs. TPMS lattice structures were fabricated using polymer composites such as thermoplastic polyurethane (TPU) blended with PLA and poly (butylene adipate-co-terephthalate) (PBAT) combined with polyglycolic acid (PGA). 38 The compression test results showed that the diamond lattices exhibited greater strength and enhanced energy dissipation compared to the gyroid and primitive structures. Studies have shown enhanced mechanical properties for TPMS lattice structures with improved cell growth when compared to strut-based lattices. 39 Yaxin et al. 40 compared the mechanical performance of PBAT/PLA based TPMS gyroid and primitive lattice scaffolds. The findings showed that PBAT/PLA filaments exhibited strong flexibility, thermal stability, rheological characteristics, and high printability. Gyroid structures showed superior load bearing capacity and energy absorption compared to the primitive lattice structures.
Firoz et al. 41 explored the impact of magnetic nano Fe3O4 on PLA scaffolds of varying infill volume. The nanoparticles were found to be optimal in a variety of applications such as bone repair, tissue engineering, targeted drug delivery, and hyperthermia therapy of cancer. The research showed that 3D printed PLA/Fe3O4 composite scaffolds possess controllable mechanical properties, ferrimagnetic behaviour, and shape memory effect, with shape recovery up to 100% in nonporous PLA and 85% in porous gyroid structure. Decrease in infill density was found to increase shape recovery, and the mechanical characteristics of 50% porous composites were found comparable to native cancellous bone, and the results indicate great potential for individualized bone repair applications. Incorporation of Fe3O4 nanoparticles into PLA resulted in interactions such as bonding between the ester group of PLA and the metallic cations of Fe3O4 and hydrogen bonding involving citric acid on the nanoparticle surface; these interactions were validated by Raman spectroscopy through carbonyl (C = O) band shifts of PLA. PLA gyroid scaffolds with unit cell dimensions of 2 mm (G2), 2.5 mm (G25), and 3 mm (G3) were produced by fused deposition modeling and subjected to testing of their structural and mechanical performance in the building and transverse directions. Scaffolds were found to be highly open porous - 86.1 ± 1.4% (G2), 89.4 ± 1.0% (G25), and 90.3 ± 0.4% (G3) and exhibited structural anisotropy of 3.8, 2.0, and 1.0, respectively, with a higher isotropy as the cell size increased. Compressive elastic modulus and yield strength in the building direction were 118–180 MPa and 106–138 MPa, respectively, and were significantly lower in the transverse direction at 2–8 MPa and 2.5–6 MPa, respectively. Tensile testing showed moduli of 51–63 MPa and yield strengths of 11–17 MPa in the building direction and 1.5–4.5 MPa and 1–5 MPa transversely. The G2 scaffold showed the best overall performance with the highest mechanical strength and stiffness in both directions. 42 Mall et al. 43 conducted a critical assessment of TPMS based bone scaffolds of Primitive, Gyroid, and Double Diamond structures made from a hybrid of bioactive ceramic HA and biodegradable PLA. The study tests the material and structural performance of the scaffolds and determines the most appropriate hybrid TPMS structure that most closely mimics the mechanical and biological behaviour of human bone. The compressive strength of TPMS 3D printed structures in the following order is Primitive < Gyroid < Double Diamond, with the highest energy absorption characteristic in the gyroid structure. The increase in the number of unit cells in TPMS lattices maximizes the surface area to volume ratio with given porosity and volume, which is favourable for biological functions such as vascularization.
Previous studies have highlighted the significance of composite materials in improving mechanical strength, along with the benefits of hydroxyapatite for enhanced cellular differentiation and proliferation. TPMS based bone scaffolds have also demonstrated superior mechanical properties even at lower porosities. Further investigations into a wider range of TPMS designs are essential to explore the potential of bone scaffolding using fused filament fabrication. The aim of this investigation is to analyze the effects of different TPMS design structures such as diamond, Fischer Koch S (FKS), Forstner Random Dots (FRD), gyroid, neovius, primitive, I-graph Wrapped Package (IWP) and Polyhedral Modified-Y (PMY) on the compression properties and modulus of elasticity of PLA and nHA reinforced PLA for bone scaffolding applications. The impact of porosity on the compression behaviour is also studied to understand the relationship between strength and material density. The detailed investigations of the effects of TPMS designs and porosity on the mechanical properties of PLA/nHA make it novel. These results could be used by researchers to advance the field through in vivo testing of the most promising scaffold designs exhibiting high interconnectivity and high surface area to volume ratios required for effective bone regeneration.
Materials and Methods
The initial step in material preparation involved thoroughly drying the raw materials. The nHA powder is vacuum-dried at 100°C while the PLA granules are heated at 70°C for a full day. Two types of filaments are produced: one composed solely of PLA and another containing 95% PLA and 5% nHA by weight. The white coloured nHA powders having less than 100 nm size with purity greater than 99% are used for creating the composite filament. The powder had a molecular weight of 502.31 g/mol with a melting temperature of 1100°C. The nHA powders is selected due to its superior bioactivity, higher surface area, and better dispersion within polymer matrices, which enhances mechanical reinforcement and promotes cellular responses beneficial for bone regeneration. Moreover, due to the use of a 0.2 mm nozzle for 3D printing of fine TPMS scaffolds, micron-sized HA posed a significant risk of clogging, particularly due to agglomeration. Clear PLA granules having a purity greater than 99% with a melting temperature of 155°C are used as the base material in the filament manufacturing process. The density of PLA and nHA is 1.24 g/cm3 and 1.038 g/cm3 respectively at 25°C. A dry blender is used to ensure uniform dispersion of nHA within the polymer. This involved using a high-speed mixer to achieve homogeneous distribution and prevent agglomeration, which could affect the extrusion process. The prepared blend is then added to the hopper of a single-screw extruder. The extrusion equipment, a Suzhou ACC model, operated at 15 kW and featured a screw with a 28:1 length-to-diameter ratio and a diameter of 5 mm. The material is gradually heated to approximately 180°C, which is greater than the melting temperature of PLA. The heating profile consisted of four progressive zones set at 165°C, 170°C, 175°C, and 180°C. The rotating screw generated shear forces, aiding in the dispersion of nHA within the molten polymer. These forces helped maintain a uniform distribution of the filler within the PLA matrix. The composite is extruded through a die into filament form after reaching the molten state. Precise control over temperature and screw speed is required to achieve a consistent diameter of 1.75 mm suitable for most commercial 3D printers. The extruded filament is then cooled using a two-stage water bath: first at 60°C, followed by ambient temperature water. Continuous monitoring is performed throughout the manufacturing process to ensure dimensional consistency. The same procedure is repeated for producing pure PLA filaments of the same diameter.
Process parameters for fused filament fabrication.
Shimadzu AGX plus universal testing machine with a maximum load of 10 kN is used to obtain the compression strength and modulus of elasticity of the PLA and PLA/nHA TPMS structures. The crosshead speed of the platform is maintained at a rate of 1 mm/min during the compression testing. Filament manufacturing, 3D printing of TPMS structures and compression testing are shown in Figure 1. Scanning electron microscopy (SEM) is used to examine both pure PLA filaments and those containing 5 wt% nHA to analyze the surface characteristics. The imaging is performed with a Jeol 6390LA SEM, which operates within a voltage value of 0.5–30 kV having a resolution of 136 eV, and provides the highest magnification of 300,000. The system utilizes a tungsten electrode with small filament samples mounted onto carbon tape affixed to the sample holder. A thin gold coating is applied using a JFC 1600 ion sputtering system to enhance imaging clarity which ensures uniform coverage for improved visibility under the electron beam. X-ray diffraction (XRD) analysis is performed to study the crystalline structure and phase composition of both filament types. The analysis is carried out using a Bruker D8 Advance diffractometer, which operates by directing X-rays at the sample while detecting the diffracted beams to assess crystallinity and structural characteristics. The diffraction patterns are recorded within a 2θ range of 3° to 80°, allowing for precise measurement of peak intensities and structural properties of the materials. These measurements provided valuable information on how the addition of nHA influenced the crystalline nature of the PLA filaments. (a) Filament manufacturing, (b) fused filament fabrication of TPMS structures, (c) compression testing, and (d) 3D printed TPMS structures.
Results and Discussion
Compressive strength and modulus of elasticity of 3D printed PLA and PLA/nHA using nominal cross-sectional area.
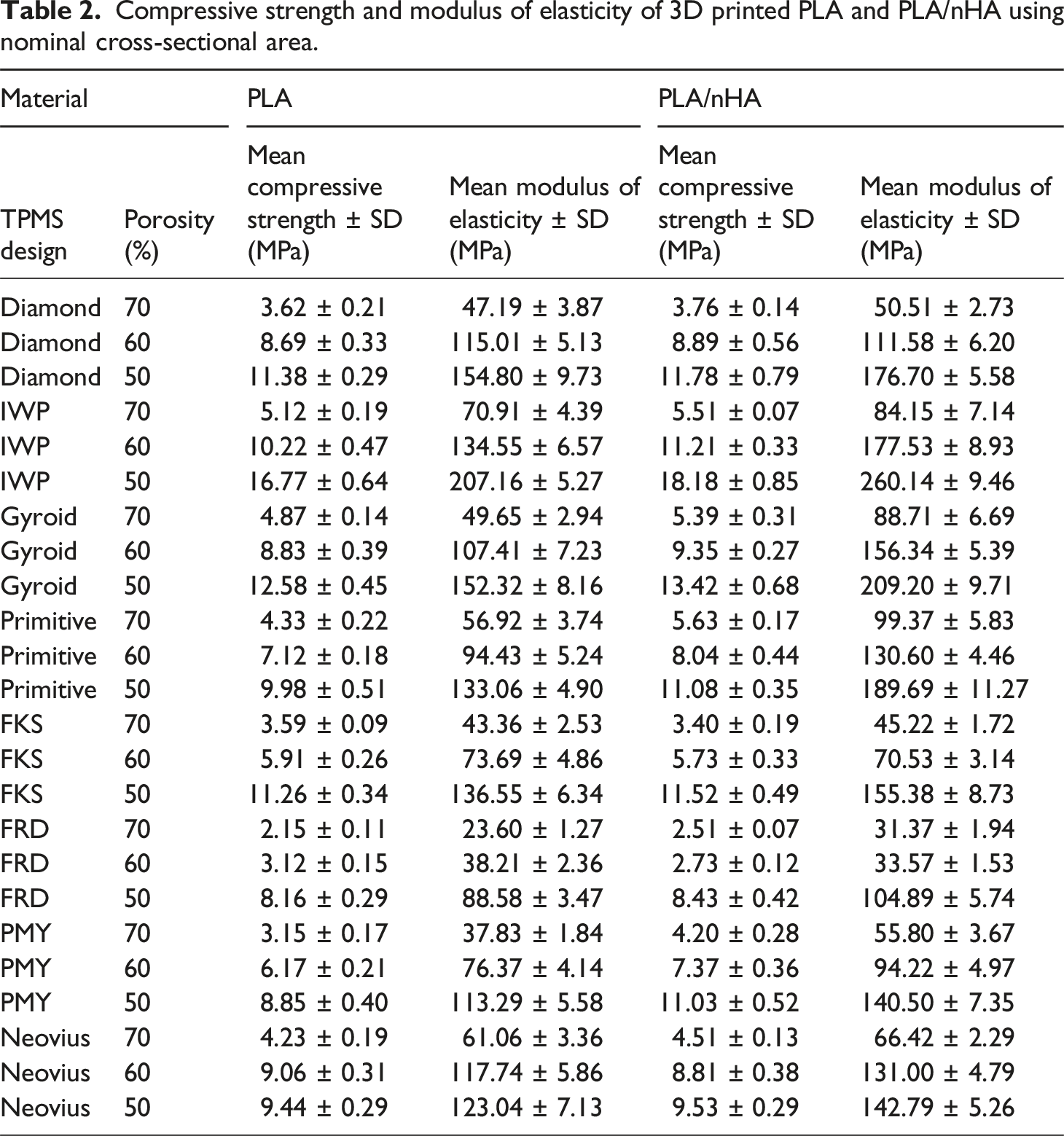
ANOVA for compressive strength using nominal cross-sectional area.
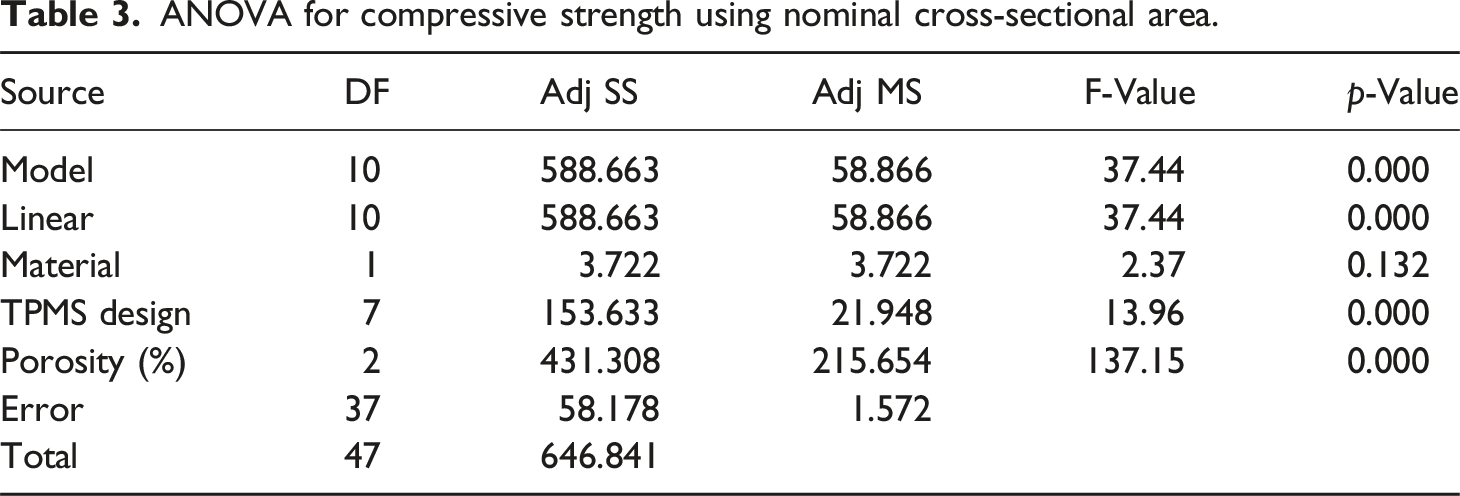
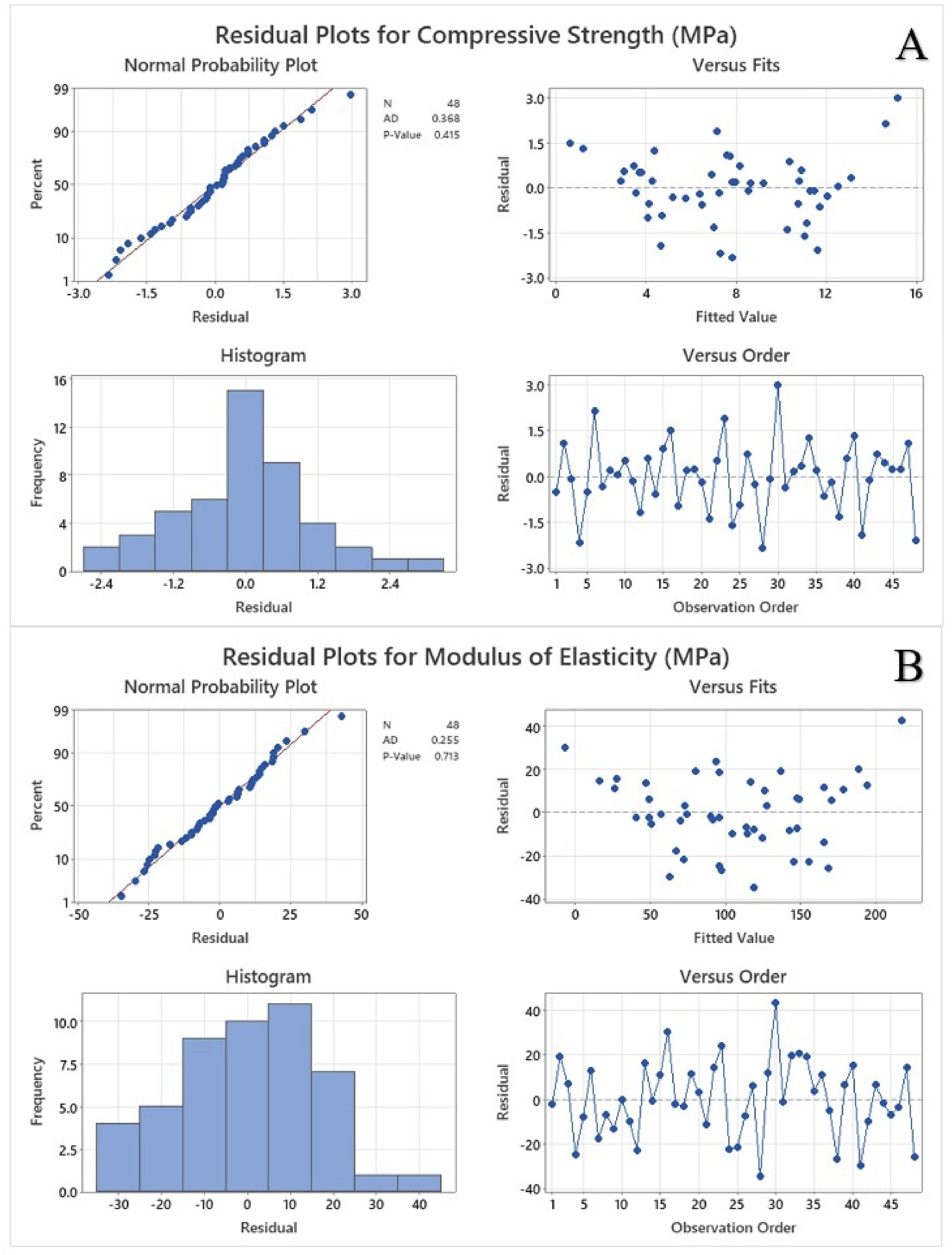
ANOVA for modulus of elasticity using nominal cross-sectional area.
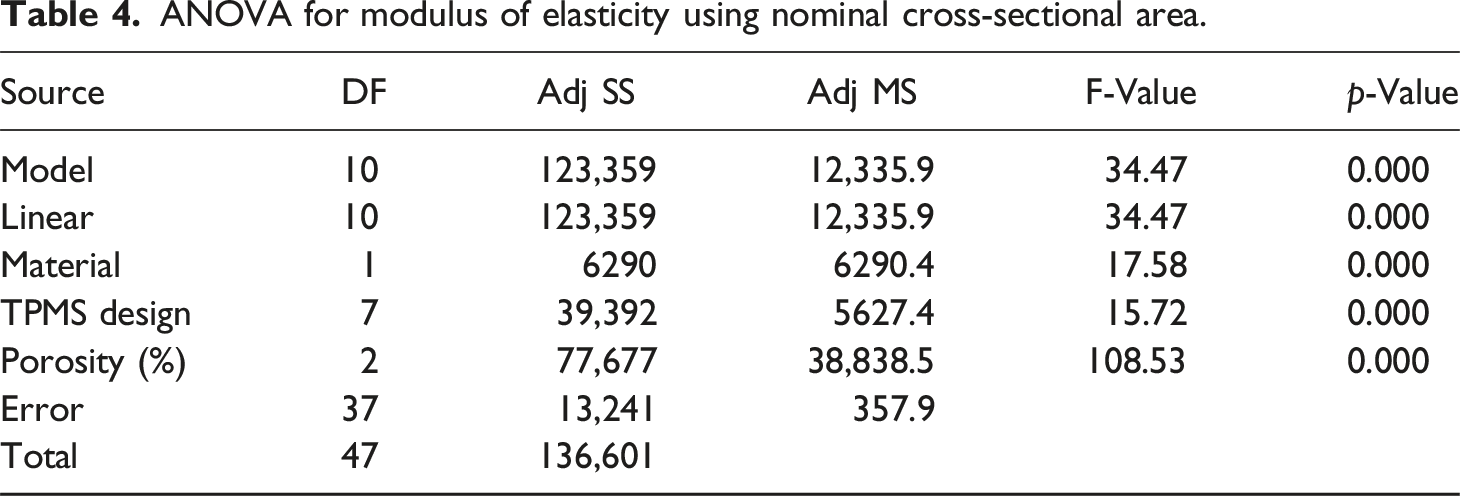
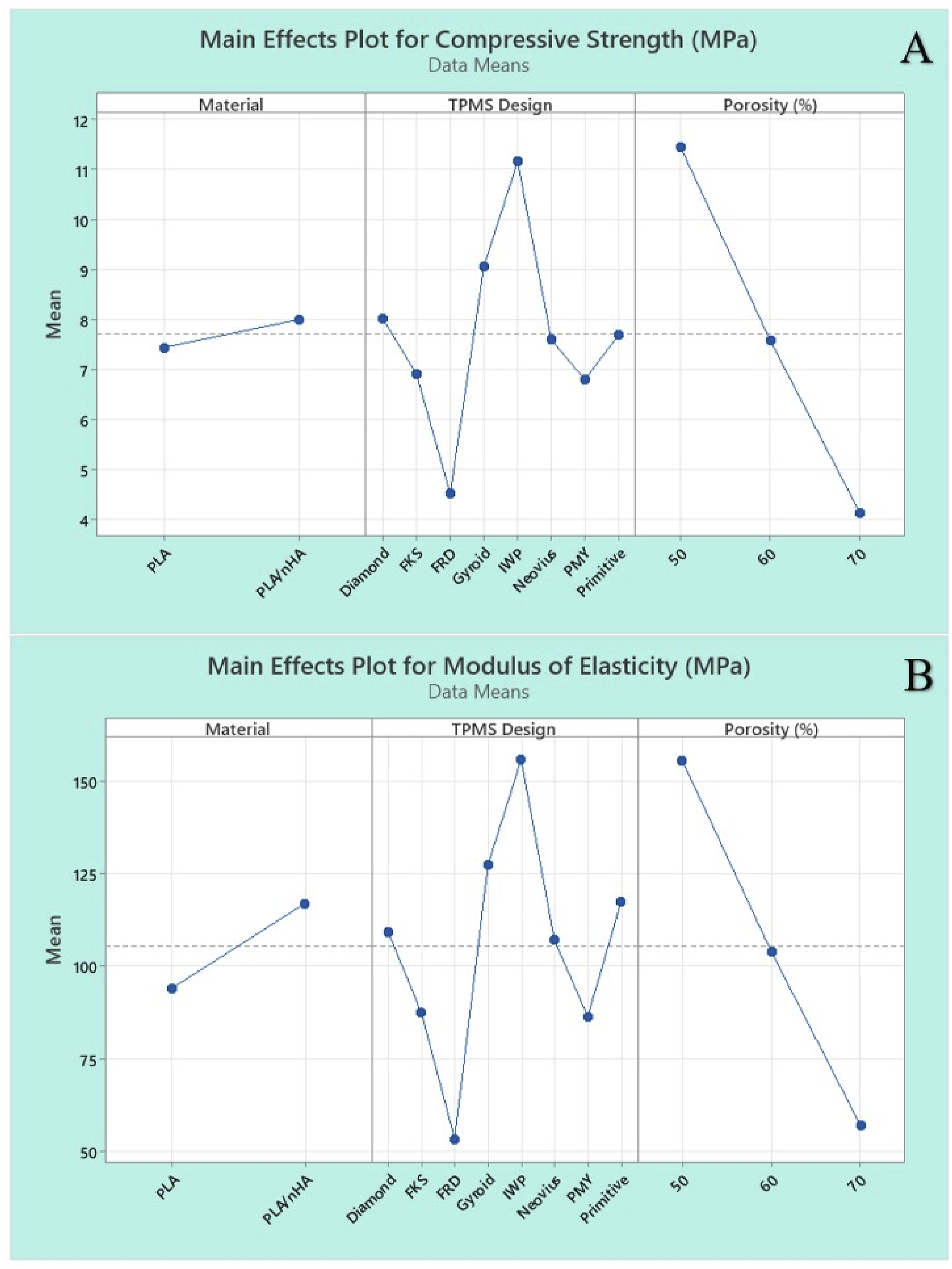
Main effects plot for (a) compressive strength, and (b) modulus of elasticity using nominal cross-sectional area.
The Pareto chart of standardized effects shown in Figure 3 illustrates the relative influence of the material, TPMS designs and porosity on the mechanical properties at a significance level of α = 0.05. The red dashed vertical line represents the critical t-value of 2.03. Any factor exceeding this value significantly affects the mechanical strength. Porosity has the largest standardized effect followed by TPMS designs indicating its critical influence on the response factors. Material has the smallest effect and does not exceed the threshold value for compressive strength. Figure 4 shows the residual plots for compressive strength and modulus of elasticity using nominal cross-sectional area. Residual plots are used to check the assumptions of regression models, such as normality, independence, and homoscedasticity. The normal probability plot tests whether the residuals follow a normal distribution. The residuals closely follow the reference diagonal line demonstrating approximate normality. The Anderson-Darling (AD) test statistic is 0.368 and the p-value is 0.415 for compressive strength whereas the AD value is 0.255 and the p-value is 0.713 for modulus of elasticity. A high p-value (> 0.05) suggests that the null hypothesis is that the residuals are normally distributed and cannot be rejected. There are no extreme deviations or outliers. The residuals appear randomly scattered around zero without any clear pattern and there is no funnel shape, which means homoscedasticity is likely satisfied. The histogram is approximately bell-shaped and symmetrical, and most residuals are centred around zero, with few extreme values. There is no significant skewness or multimodal distribution, and the histogram supports the assumption of normality. The randomness shows that the residuals are independent over time and no autocorrelation is detected, thus the model appears valid and reliable for making inferences. Pareto chart for (a) compressive strength, and (b) modulus of elasticity using nominal cross-sectional area. Residual plots for (a) compressive strength, and (b) modulus of elasticity using nominal cross-sectional area.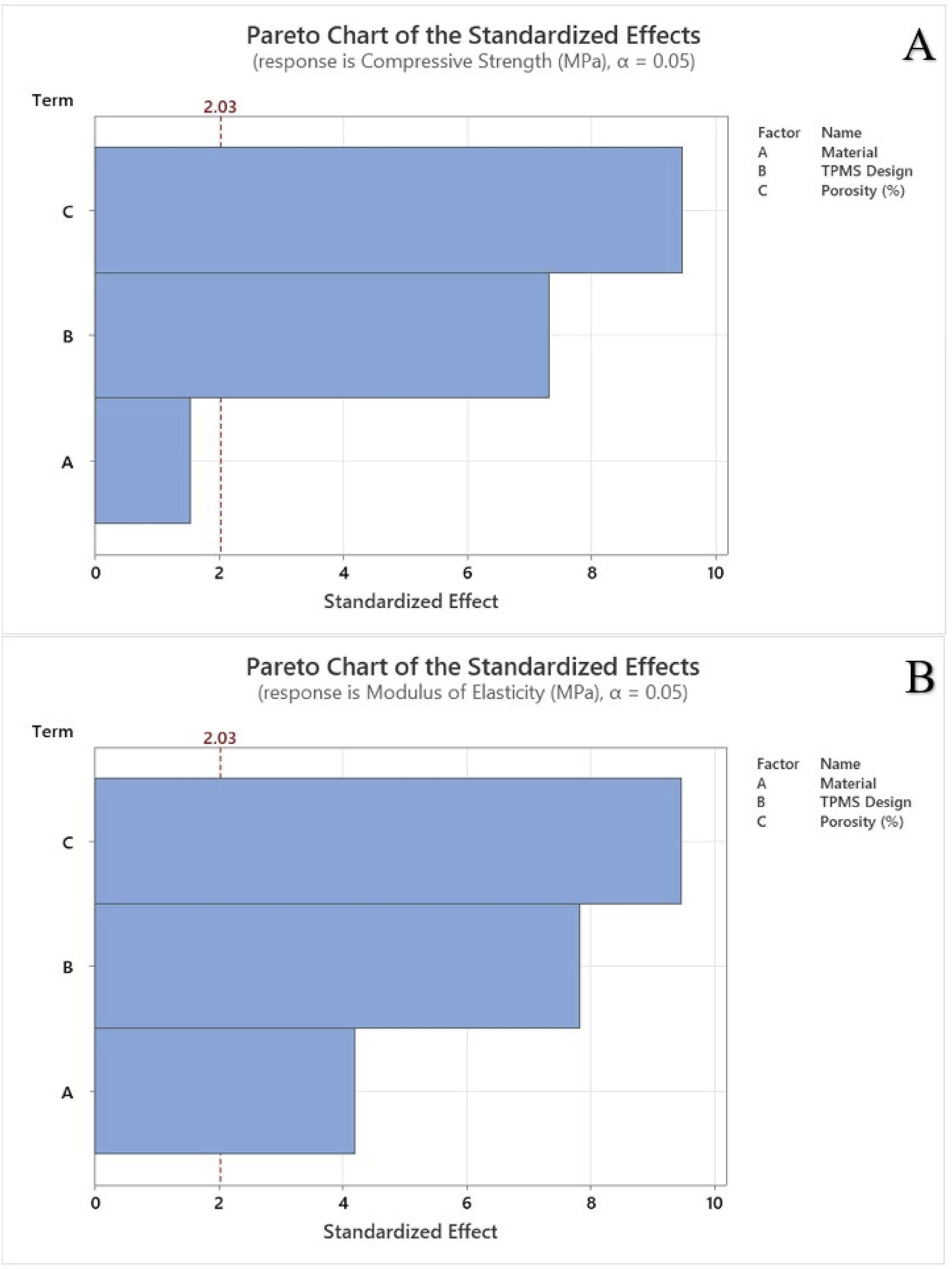
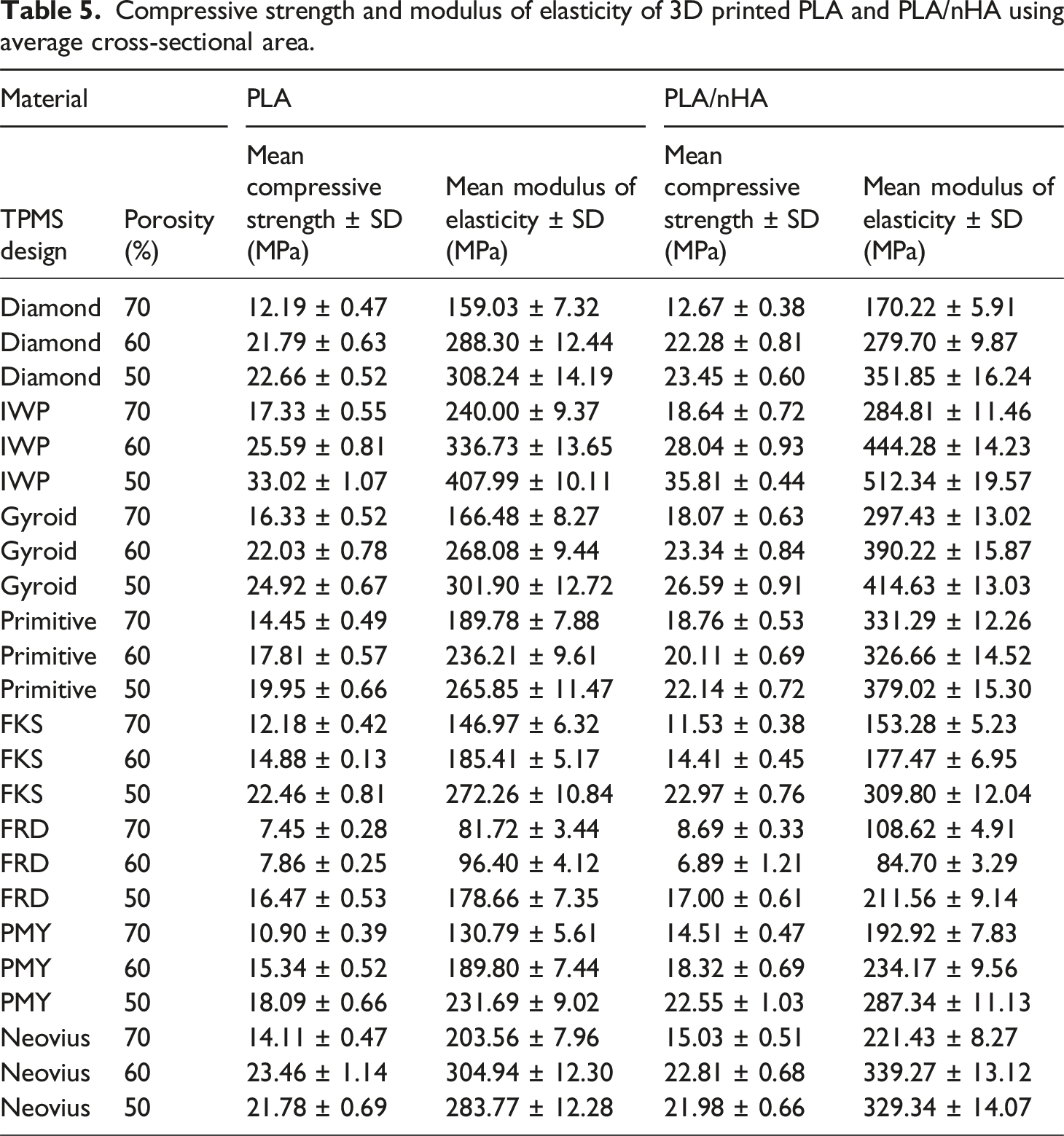
Compressive strength and modulus of elasticity of 3D printed PLA and PLA/nHA using average cross-sectional area.
ANOVA for compressive strength using average cross-sectional area.
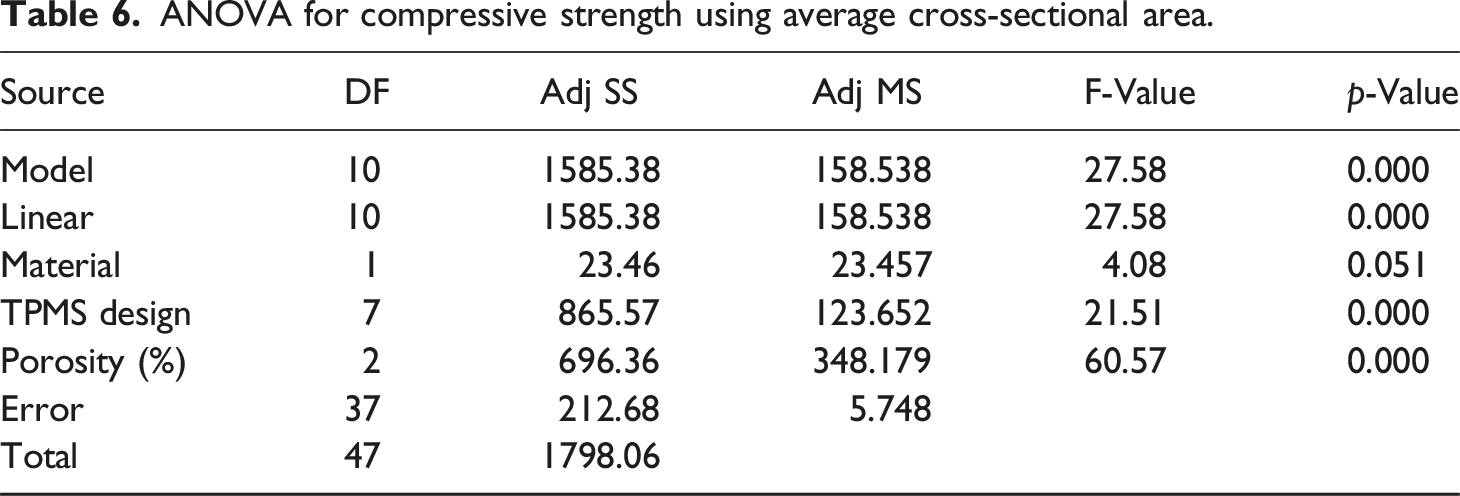
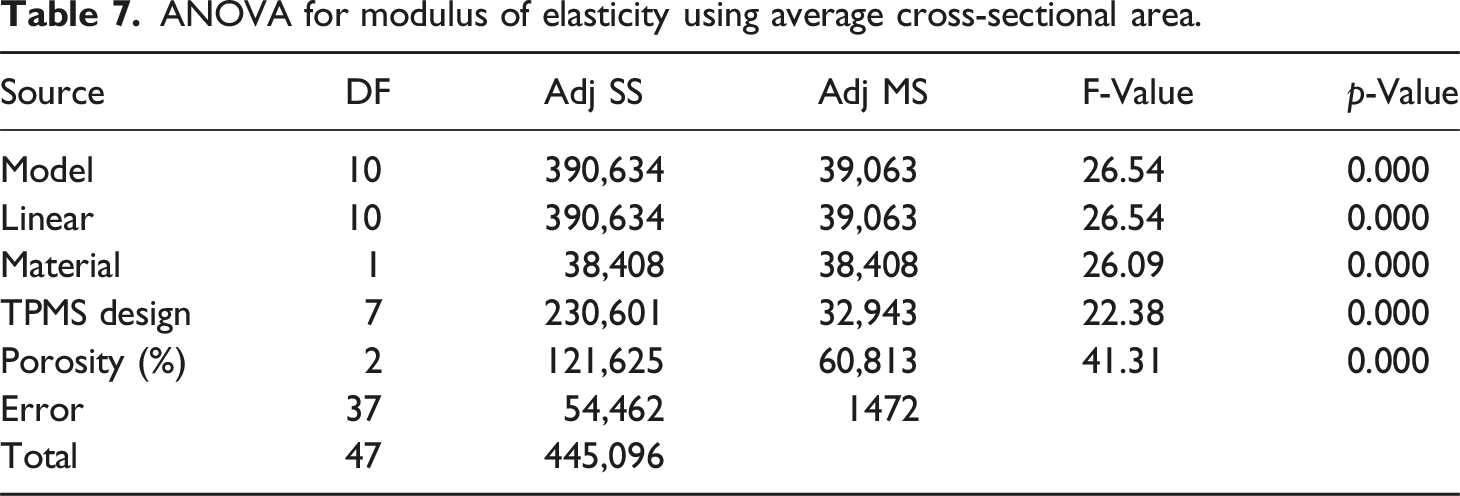
ANOVA for modulus of elasticity using average cross-sectional area.
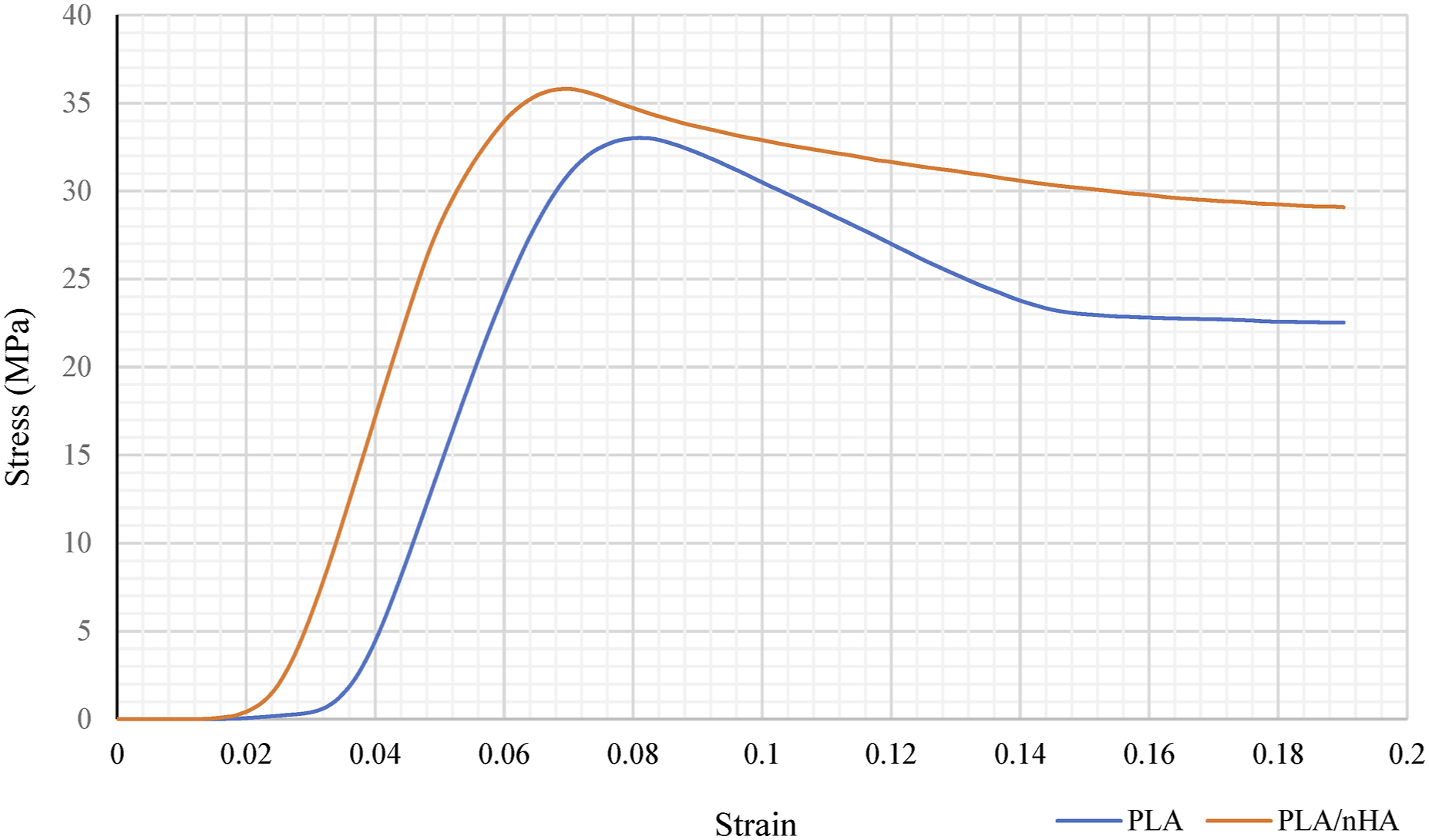
Stress strain curve of IWP TPMS structures for both PLA and PLA/nHA.
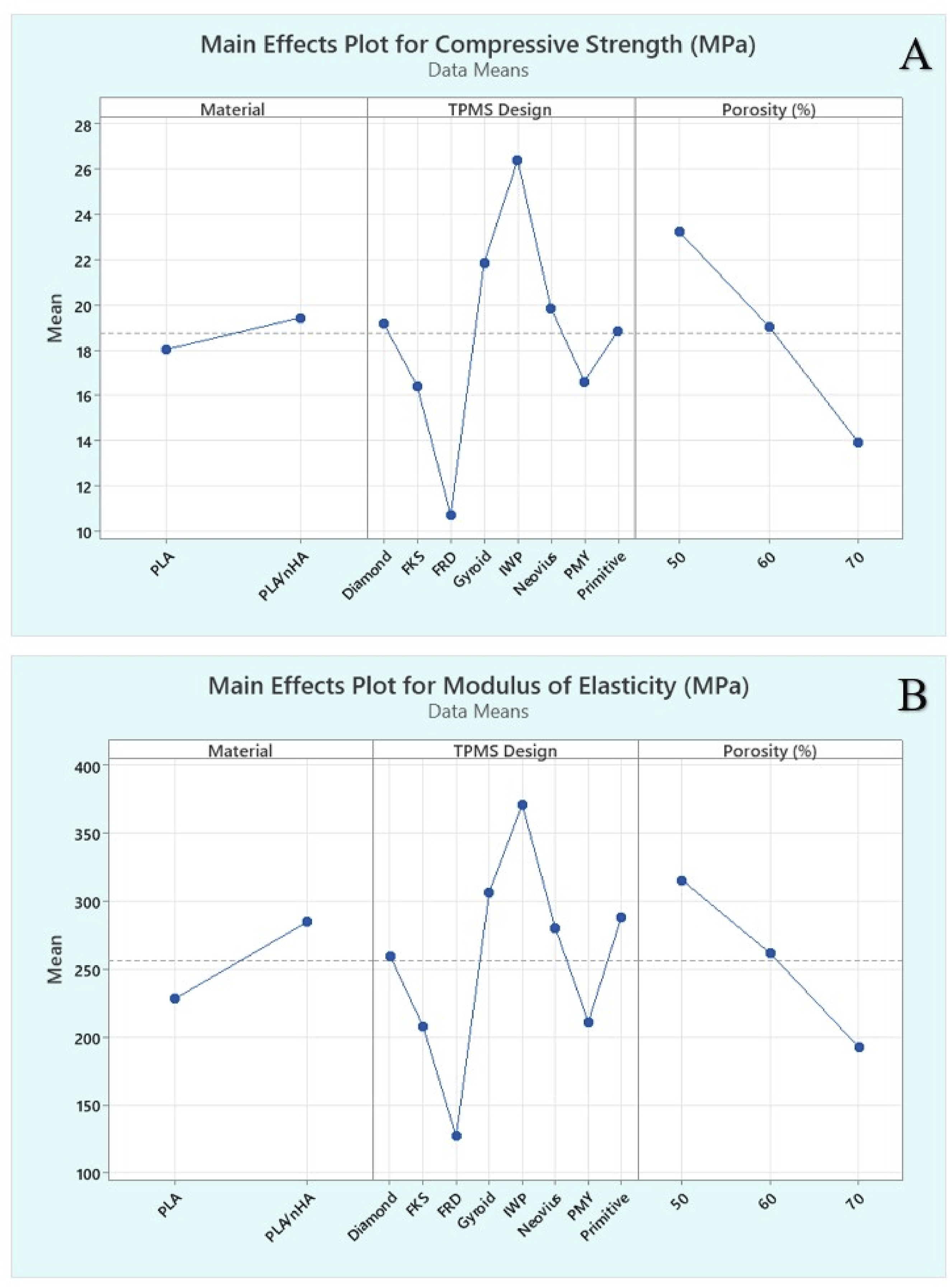
Main effects plot for (a) compressive strength, and (b) modulus of elasticity using average cross-sectional area.
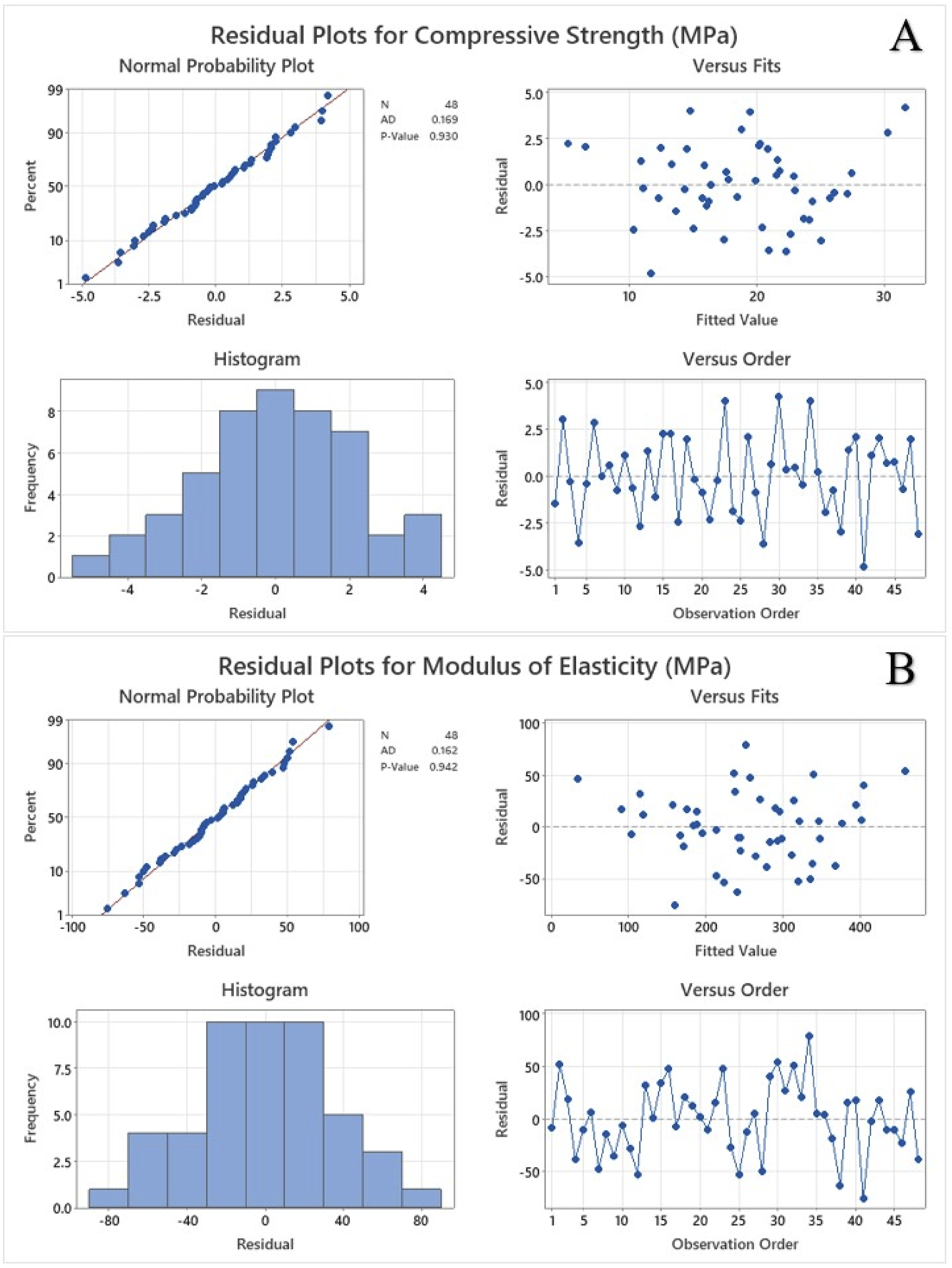
Figure 7 represents the Pareto chart of standardized effects, highlighting the influence of material, TPMS designs, and porosity on mechanical properties at a significance level of α = 0.05. The red dashed vertical line indicates the critical t-value of 2.03, with any factor surpassing this threshold having a significant impact on mechanical strength. Porosity had the highest standardized effect on compressive strength whereas TPMS designs showed the largest effect on elastic modulus. The material exhibited the least effects in both cases. The residual plots for compressive strength and modulus of elasticity using average cross-sectional area are illustrated in Figure 8. Residual plots help in verifying the assumptions of regression models, such as normality, independence, and homoscedasticity. The normal probability plot showed that the points closely coincide with the reference diagonal line demonstrating approximate normality. The AD test statistic for compressive strength is 0.169 with a p-value of 0.93, while for modulus of elasticity, the AD value is 0.162 with a p-value of 0.942. The residuals fluctuate randomly over the observation order, indicating no specific trends or autocorrelation. The high p-value obtained demonstrated that the null hypothesis cannot be rejected, and the residuals are normally distributed. The residuals are randomly distributed around zero without a distinct pattern, and the absence of a funnel shape suggests that the homoscedasticity is likely met. The histogram is roughly bell-shaped and symmetrical, with most residuals concentrated around zero. Pareto chart for (a) compressive strength, and (b) modulus of elasticity using average cross-sectional area. Residual plots for (a) compressive strength, and (b) modulus of elasticity using average cross-sectional area.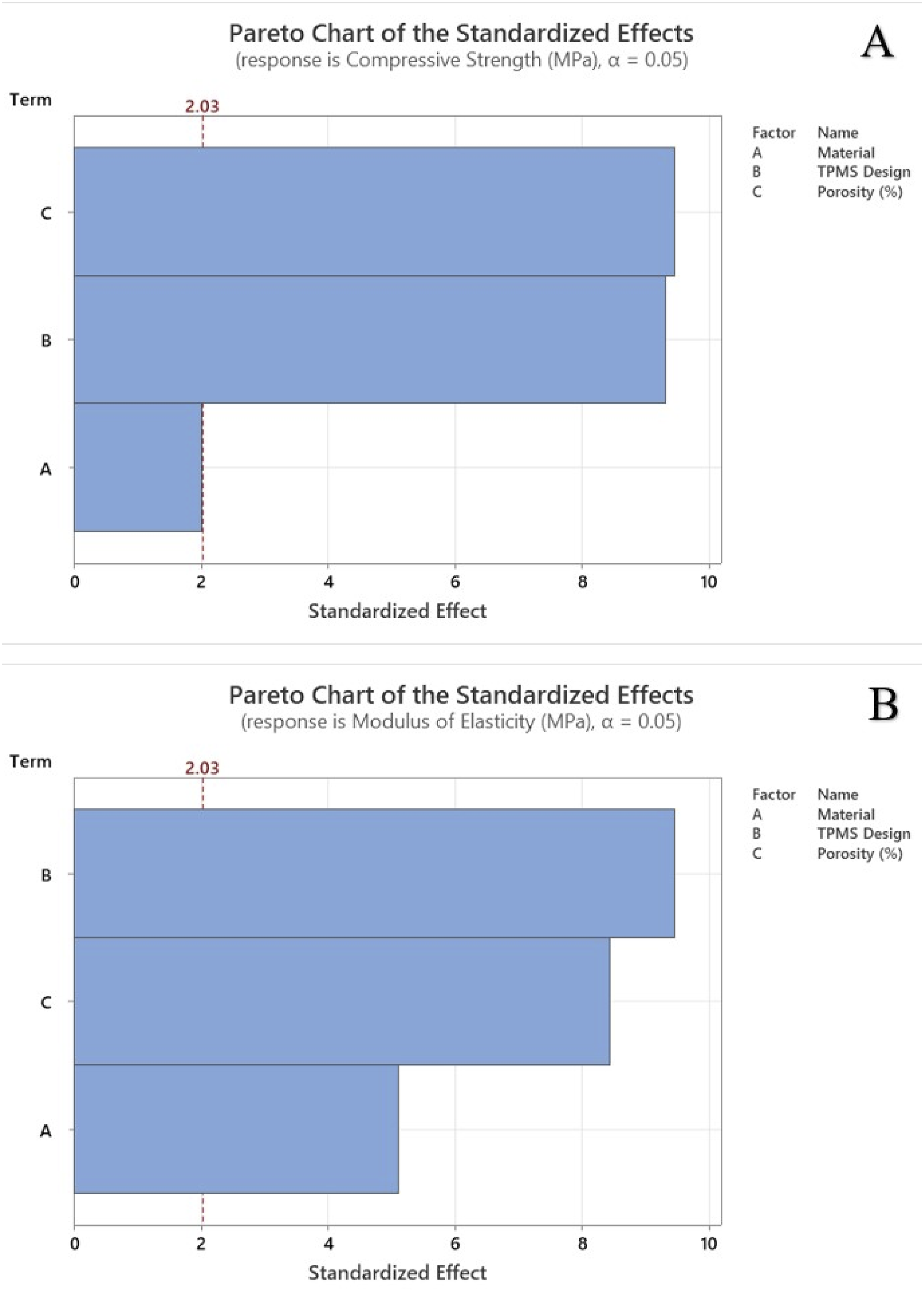
The IWP TPMS structure achieved a compressive strength of 35.81 MPa at 50% porosity using PLA/nHA, and 18.64 MPa at 70% porosity. At 50% porosity, the IWP structure exhibited an elastic modulus of 512.34 MPa, while the Primitive TPMS structure demonstrated an elastic modulus of 331.29 MPa at 70% porosity. These values are comparable to those of cancellous bone. Human cortical bone has compressive strengths of 130–290 MPa and tensile strengths of 90–190 MPa, compared to cancellous bone, which has compressive strengths of 2–38 MPa.
45
If pore size increases at a certain porosity value, the elastic modulus and compressive strength value fall. Elastic moduli of cancellous and cortical bones are 0.02–2 and 3–30 GPa, respectively.
46
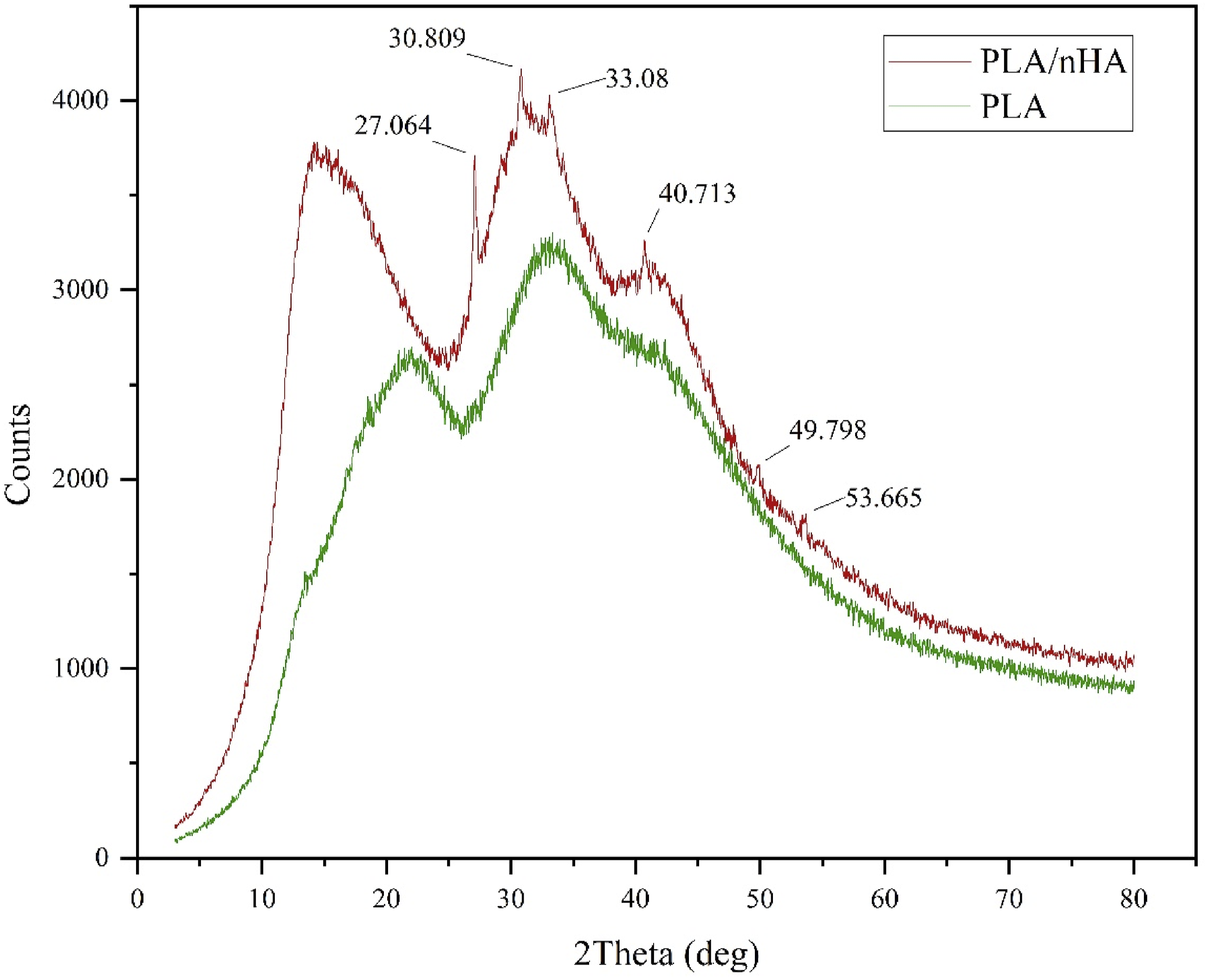
The SEM images of PLA and PLA/nHA filaments are shown in Figures 9 and 10 respectively. The diameter of the filament is 1.75 mm to be used in most industrial 3D printers. The microscopic images clearly display the smooth PLA filament whereas pores and voids are seen on nHA reinforced PLA filaments. It is observed that there was only a slight improvement in the compressive strength with the incorporation of nano ceramic powders. The pores generated during the filament manufacturing process might serve as stress concentrators during external loading. The XRD patterns for both materials are illustrated in Figure 11. The nHA reinforced PLA filament showed more crystalline behaviour with peaks highlighted in the image. The most intense peaks are formed at 27.064°, 30.809°, and 33.08°. The composite filament attained better crystallinity percentage compared to PLA. TPMS structures such as IWP and gyroid exhibited good mechanical properties even at high porosity levels, comparable to natural bones. The homogenous distribution of nHA within the polymer matrix is thus confirmed with the SEM analysis, XRD crystallinity changes and extrusion process control. The addition of nHA into polymer matrix has been shown to enhance the biological activity during bone regeneration. These findings expand the potential for developing high-performance, patient-specific TPMS bone scaffolds using PLA/nHA. SEM images of PLA 3D printing filament. SEM images of PLA/nHA 3D printing filament. XRD patterns of PLA and PLA/nHA 3D printing filament.

Conclusions
This study presents a comprehensive investigation of the mechanical properties of various 3D printed TPMS structures using PLA and PLA/nHA for bone scaffolding applications. It highlights the effects of porosity, TPMS designs and material on the compressive strength and elastic modulus of the fabricated standards. IWP exhibited the best mechanical performance followed by gyroid structures. FRD had the lowest strength, followed by FKS and PMY. This might be due to its thinner wall thickness, which results from a shape that has more surface area at the same porosity. The average cross-sectional area methodology provided more accurate results in determining both compressive strength and modulus of elasticity. The addition of 5 wt% nHA to PLA resulted in enhancements in compressive strength (7.76%) and elastic modulus (24.79%) with improved crystallinity as observed in the XRD analysis. However, SEM images showed the formation of pores and voids in the PLA/nHA filaments, which may act as stress concentrators and limit the mechanical performance. The manufacturing and extrusion process of the composite filament needs further optimization to eliminate such defects. ANOVA results confirmed that TPMS design and porosity significantly influenced both compressive strength and elastic modulus, while the type of material had a lesser effect, particularly on compressive strength. The single-screw extrusion system was capable of producing 5 wt% of nHA filaments with a 1.75 mm consistent diameter. Higher ratios of bioceramics led to the variation in filament diameter due to improper extrusion. This could be overcome with the manufacturing of composite pellets using a twin screw extrusion system and thereafter using it for manufacturing filament for 3D printing. Future studies should focus on optimizing the filament manufacturing process to minimize void formation and better dispersion of nHA in the polymer matrix. The investigation could further highlight the possibility of higher nHA loading percentages or alternative bioceramic fillers for improving bioactivity and strength. The in vitro cell culture and degradation studies are essential to evaluate the biological performance and long-term biocompatibility of these scaffolds. Customized patient-specific designs using CT/MRI data and employing multi-material printing approaches could significantly advance the clinical relevance of TPMS based bone scaffolds in regenerative medicine.
Footnotes
Acknowledgements
ADU author acknowledges the financial support received from the United Al-Saqer Grants Initiative through Project Number 19300842.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by United Al-Saqer Grants Initiative (19300842).
Data Availability Statement
All data supporting the study’s findings are included in the paper.
