Abstract
Knowing about structure-properties relationship in polymer nanocomposites, especially for the systems that have not been studied enough, is of great significance in academic and industrial level. In this research, a non-investigated nanocomposite system based on poly(methyl methacrylate) (PMMA) and 30 wt% poly(ethylene-co-vinyl acetate) (EVA) containing 0, 2.5, 5, 7.5 and 10 wt% multi-walled carbon nanotube (MWCNT) loadings were first prepared and then their physical properties were examined using different techniques. The results obtained from the thermal analysis of the materials indicated that the crystallization and melting rates as well as the degree of crystallinity of the nanocomposites steadily decreased, however, the full width at half maximum of the exotherms progressively increased with increasing in the MWCNT loading. The analysis of the rheological data using the generalized Maxwell model (GMM) showed that all the shear moduli and the longest relaxation time increased with the addition of the nanofiller content. The assessment of the average crystallite size of the EVA phase on plane (110) by X-ray diffractometry revealed that the quantity increasingly reduced with the increase in the nanoparticles concentration. It was found from the thermogravimetric experiments that the nanocomposites were more thermally stable, in both air and nitrogen atmospheres, in comparison with those of PMMA and PMMA/EVA blend. Moreover, the thermal stability of the composition containing 7.5 wt% nanofiller was the highest among the materials studied, irrespective of the nature of the atmosphere examined. MWCNT nanoparticles were more effective in ameliorating the thermal stability of the materials under air than that for nitrogen.
Introduction
Polymer nanocomposites (PNCs) can be accounted as one of the most important class of materials to date, because of their superior physical and mechanical performances than those of the pristine polymers. 1 The polymeric matrix or blend, in this advanced group of materials, is often incorporated with one or more nano-sized fillers such as organoclay (OMMT),2,3 single-walled (SWCNT)4,5 and multi-walled carbon nanotube (MWCNT),6–8 halloysite nanotube (HNT),9–11 graphene (GN)12,13 and polyhedral oligomeric silsesquioxane (POSS).14,15 PNCs can be extensively exploited in various areas of application, for instance, electronics, 16 aerospace,17,18 biomedical, 19 automotives,20,21 electromagnetic interference (EMI) shielding, 22 sensing, 23 catalysis, 24 environmental remediation 25 and packaging industry.26,27
Poly(methyl methacrylate) (PMMA)/poly(ethylene-co-vinyl acetate) (EVA) blends and those reinforced with (nano)fillers have been the subject of limited studies. In this regard, the authors tried to uncover the extent of improvement in some physico-mechanical properties of the blends or (nano)composites in comparison with those of the parent polymers or the unfilled blends, respectively. Kojima et al. 28 worked on PMMA/EVA blends fabricated by polymerization-induced phase decomposition. The materials revealed to have a unique two-phase morphology, wherein the particles of PMMA as the major component were dispersed throughout EVA, which played the role of a matrix but it was the minor component. The blends presented much improved impact strength than that of both the melt blends and PMMA toughened by means of a core-shell type impact modifier. Moraes et al. 29 studied the synthesis of poly(EVA-g-PMMA) derived from the free radical polymerization of methyl methacrylate (MMA) in the presence of azobis(isobutyronitrile) (AIBN) as an initiator and mercapto-modified EVA as a chain transfer agent. The functionalized EVA, known as poly(ethylene-co-vinyl alcohol-co-vinyl mercaptoacetate) (EVASH), was prepared by the process of hydrolyzed EVA esterification using mercaptoacetic acid. They claimed that the higher conversion of grafted PMMA could be attained by rising the [SH]/[AIBN] ratio. It was also found that a uniform molecular weight distribution of PMMA segments in the graft and a superior thermal stability of the copolymer could be obtained by controlling the [AIBN]/[SH] ratio. In addition, the degree of crystallinity of the EVASH backbone was not affected by the PMMA segments in the graft copolymer. Chen et al. 30 studied the phase separation of the mixture of MMA/vinyl acetate (VA)/EVA by scanning electron microscopy (SEM) and time-resolved light scattering methods during copolymerization. They reported that when copolymerization in MMA/VA/EVA and MMA/EVA mixtures were carried out, phase separation was taken place through spinodal decomposition (SD). The results indicated that unique morphologies were eventually achieved via the copolymerization of MMA/EVA and MMA/VA/EVA systems during SD phenomenon. PMMA-rich particles having a uniform size and distribution were found to disperse in the matrix of an EVA-rich phase for the polymerized MMA/EVA system. However, MMA/VA copolymer rich particles were finely embedded in an EVA-rich phase with bimodal distribution of sizes for polymerized MMA/VA/EVA mixture. Errico et al. 31 investigated the blends of PMMA and its copolymer with butyl methacrylate (BMA) P(MMA-co-BMA) with EVA using reactive blending technique. The blends and semi-IPN fabricated by the authors showed an attractive optical behavior. They were transparent at ambient temperature whereas turned into opaque at higher temperature. Their optical characteristics revealed that the transition temperature from transparent to opaque was dependent on the material composition, due to the development of complex morphology in the materials and to similar refractive indices of the components because of the formation of an interfacial agent. Wu et al. 32 examined the preparation of PMMA/EVA/OMMT nanocomposites with and without in-situ cross-linking by tetrapropoxysilane and dibutyl tin oxide as a cross-linking agent and as a catalyst, respectively. The experimental results proved that in-situ cross-linking could convert the EVA rheological behavior from a liquid to a viscoelastic solid. They suggested that the majority of the OMMT nanoparticles were clustered in the region of the cross-linked EVA phase. Moreover, an intercalated and incompatible structure was formed. Although, the addition of OMMT and in-situ cross-linking of EVA augmented the modulus of PMMA, the latter slightly decreased the barrier properties of PMMA/EVA/OMMT system. Puthiyottil et al. 33 prepared the PMMA and PMMA/EVA blends doped with europium-β-diketone chelate. The authors reported that the structural properties of the complex doped materials showed that the complex was present in an identical crystalline state in the doped states as it was in the pure status. They proposed that 3 wt% chelate doped macromolecular systems had an impressive emission spectral intensity and the results evidently designated the possibility of producing optoelectronic devices with excellent mechanical and optical properties derived from PMMA/EVA systems doped with europium-β-diketone chelate.
The crystallization kinetics of semi-crystalline polymers is influenced by several factors such as (nano)filler loading, interfacial interaction and crystallization conditions. 34 It was found that the nanoparticles played two competing and contradictory roles in crysallization of polymers; (a) providing nuclei for heterogeneous nucleation, which is normally dominant at low filler concentration and (b) constraining the movements of polymer chains to prevent the crystal growth, which is dominant at high filler loading. The action of nano(fillers) as nucleating agent is designated by higher onset crystallization temperature (Tcon) and crystallization peak temperature (Tcpt) during the non-isothermal crystallization and also shorter induction period as well as smaller spherulite size throughout the isothermal crystallization. 35 Both nucleation and growth are varied by temperature in non-isothermal crystallization. Therefore, the variation of temperature at a constant cooling rate will change both the nucleation and spherulite growth rates simultaneously. 36 In addition, heterogeneous nucleation was reported to be only efficient at low supercooling because homogeneous nucleation could take a lead at high supercooling where its nuclei density would be several orders of magnitude higher than that of the heterogeneous nuclei. 37 By remaining a molten polymer at high enough temperature for a while, the density of nuclei could be reduced which resulted in the formation of spherulites with larger size, at a given crystallization temperature, in comparison with those of its untreated counterpart. 38 Oburoğlu et al. 36 studied the effect of micro-size spherical calcite (CA), HNT and OMMT on non-isothermal crystallization of poly(butylenes terephthalate) (PBT) composites. They reported that CA and HNT improved the crystallization rate of the matrix. This was attributed to the notion that the 3D CA and 1D HNT inorganic fillers imposed less restriction on polymer chains compared with the 2D OMMT filler, which presented significantly higher surface area during crystal growth step. Papageorgiu et al. 39 examined the effect of MWCNT, copper nanofibers, silver nanoparticles and nanodiamonds on the crystallization kinetics of syndiotactic polystyrene (sPS). They suggested that apart from the type, the shapes of nanoparticles could alter the mechanical and crystallization kinetics of sPS. It was found that not only MWCNT encouraged the formation of β-type crystals but also the nanofiller act as the most effective nucleation agents for sPS amid the other nanoparticles studied.
This work is devoted to clarify the influence of MWCNT loadings on crystallization, melting, thermal stability and rheological behavior of PMMA/EVA based nanocomposites. Our aim was to study the impact of the nanofiller content on the above-mentioned properties for this non-studied system, to the best of the authors’ knowledge, to obtain an insight about its structure-properties relationship. For doing that, the PMMA/EVA blend and PMMA/EVA/MWCNT compositions were successfully prepared with 30 wt% EVA and 2.5, 5, 7.5 and 10 wt% MWCNT. Subsequently, they were analyzed using several techniques such as X-ray diffractometry, differential scanning calorimetry (DSC), rheometry and thermogravimetry (TGA).
Materials and Methods
Materials
The nanofiller (MWCNT) with the trade name of NanocylTM NC 7000 was procured from Nanocyl S.A (Belgium) with the purity and mean diameter and length of 90%, 9.5 nm, 1.5 μm, respectively. 40 EVA with the commercial name of EVA VS430 was provided from Hyundai Séetec (South Korea) with the density of 0.94 g/cm3, vinyl acetate content of 19% and melt flow index (190°C, 2.160 kg) of 1.7 g/10 min. PMMA with the trade name of Acryrex CM-205 was procured from Chei Mei Corporation (Taiwan). The melt flow index (230°C/3.8 kg) and density of the polymer were reported to be 1.8 g/10 min and 1.19 g/cm3, respectively. The two polymers were used as in pellet form and all the ingredients were dried at 70°C for 12 h in a vacuum oven before processing.
Nanocomposites and Specimens Preparation
Melt mixing processes were carried out in a Brabender-type laboratory mixer (Germany). The nanofiller and EVA were first mixed at 110°C and 80 rpm for 15 min to fabricate the EVA/MWCNT master-batch followed by cutting the mixture into small pieces. Afterwards, the nanocomposites were prepared with the introduction of the appropriate weight of the master-batch and the unfilled EVA in the same mixer, and mixing was taken place at 180°C and 80 rpm for 7 min before PMMA was added into the mixer. Subsequently, PMMA was introduced into the chamber and mixing continued for an extra 9 min. Finally, the nanocomposites were fabricated with the nanofiller contents of 2.5, 5, 7.5 and 10 wt%. The unfilled blend of PMMA/EVA with 70/30 weight ratio was also prepared for comparison. Each composition was compression molded in the form of 1 mm thick sheet through a compression molding instrument at 210°C, 25 MPa and 5 min. The sheets were then cut into standard specimen sizes and shapes for characterization. The codes of the materials fabricated are denoted as xPyEzC where P, E and C represent PMMA, EVA and carbon nanotubes material and x, y and z indicate the wt% of PMMA, EVA and the nanofiller in each composition, respectively. For instance, 60P30E10C stands for a nanocomposite containing 60 wt% PMMA, 30 wt% EVA and 10 wt% MWCNT.
Characterization
The crystallization and melting behavior of the unfilled and filled materials were studied via a DSC1 Mettler Toledo (Switzerland) at the cooling and heating rate of 10°C/min under a nitrogen atmosphere with the flow rate of 50 mL/min. About 10 ± 0.2 mg of each composition was sealed in an aluminium pan and heated from −50 to 160°C. All samples were then kept at 160°C for 5 min, cooled to −50°C, and after 5 min reheated to 160°C. The dynamic shear rheological measurements of the materials were carried out on a stress-controlled rheometer MCR501 (Anton Paar, Austria) with parallel-plate flow geometry. The disk-shaped samples with the diameter of 25 mm were employed in this work. The strain amplitude of 0.25% and angular frequency (ω) in the range 0.01 to 600 rad/s were used at 180°C. All the measurements were accomplished under an inert nitrogen atmosphere in order to minimize the oxidative degradation of the materials at high temperature while the experiments were in progress. The strain amplitude was found from the strain sweep experiments to explore the linear viscoelastic properties. The thermal stability of the materials was determined by a thermogravimetric analyser (TGA1, Mettler Toledo, Switzerland). All the experiments were accomplished in the scanning mode at the heating rates of 10°C/min. About 15 mg of each sample was placed in a platinum sample pan and heated from 25°C to 600°C under flowing nitrogen and air atmospheres, separately. The balance chamber flow rate and furnace flow rate was set to 20 mL/min and 50 mL/min, respectively. Wide-angle X-ray diffraction (WAXD) experiments were conducted on Philips X’pert X-ray diffractometer (The Netherlands) equipped with the CuKα radiation (λ = 1.54 Å) at 40 kV and 40 mA. The samples were examined over the 2θ range of 5 to 50° and at the rate of 0.02°/s at ambient temperature.
Results and Discussion
Thermal Behaviour
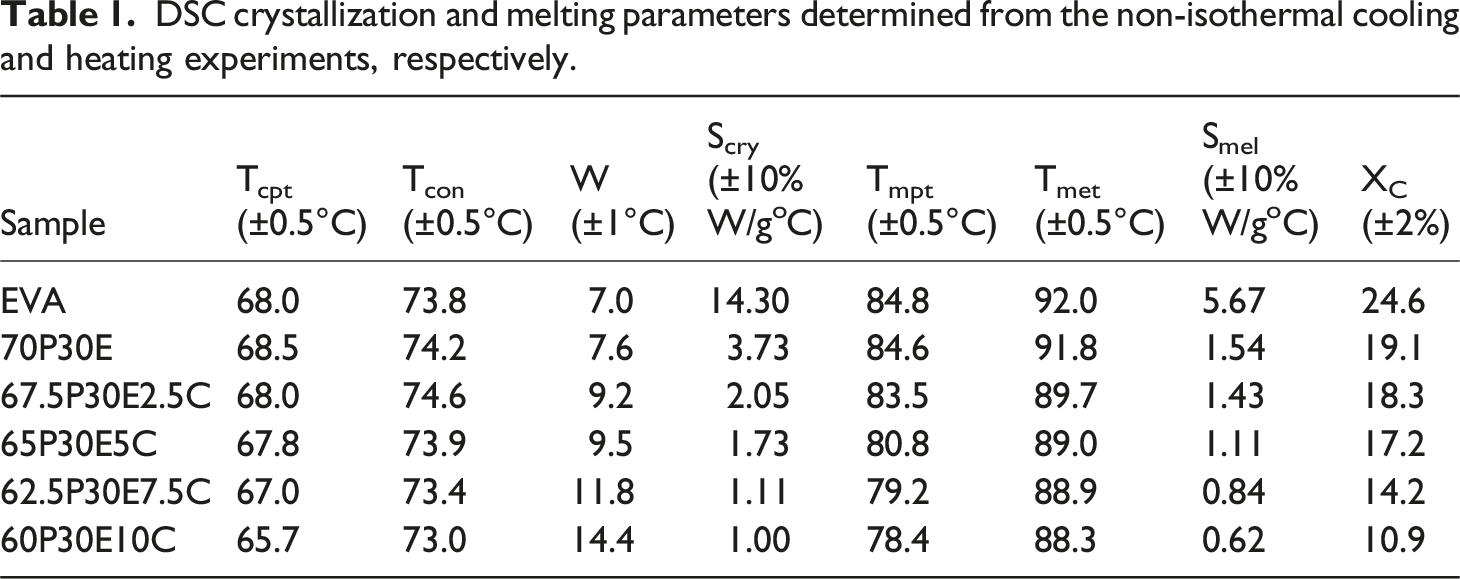
Figure 1(a) and (b) shows the first cooling and second heating thermograms of the crystallizable materials, acquired by DSC, respectively. These thermal responses were employed to obtain the Tcon, Tcpt, melting peak temperature (Tmpt), melting end temperature (Tmet), the degree of crystallinity (XC), the full width at half-maximum of the cooling exotherm (W) as well as the crystallization (Scry) and melting (Smel) rates of the materials. All the values determined are given in Table 1. To obtain the Tcon and Tmet, two tangents at the high-temperature zone of the crystallization and/or melting thermogram as well as the base line were drawn and the intersection point of the lines was taken as the desired temperature. Moreover, Scry and Smel were both determined by calculating the slope of the linear section of the crystallization and melting thermograms at their high-temperature zones, respectively.
41
The results indicated that Tcon remained fairly constant, however, Tcpt, Tmpt and Tmet reduced a little with the increase in MWCNT loading. The crystallization ability of the EVA domains decreased with the increase in the nanofiller content as well as the presence of PMMA continuous phase. The matrix become solid before reaching the crystallization temperature at which the EVA crystallization started and MWCNT progressively increased the viscosity of the mixture with the increase in the nanofiller loading. Therefore, both components imposed obstacles in front of EVA chain movements to the crystallization front by which the chance of crystallization could be diminished. Consequently, the value of XC gradually reduced with the increase in MWCNT content. The values of Scry and Smel also gradually decreased with the increase in the nanofiller concentration in the nanocomposites, reflecting that the crystallization and melting rates both declined with the increase in the MWCNT loading. Nonetheless, the crystallization rate of each composition was always larger than that of its melting rate. Conversely, the values of W, which is associated with the crystallite size distribution,
42
increased with the increase in nanofiller content. This is consistent with the notion that the crystallite size distribution was wider for the blend and become progressively broader as the MWCNT loading was increased in the nanocomposites, in comparison with that of EVA. This clearly points out that the increase in the nanoparticles numbers could destroy their advantage in creating nucleation sites by making the EVA molecular movements more difficult and thus by reducing their diffusions to the crystallization regions. Consequently, the possibility of the formation of more imperfect crystallites with broader dissimilarity is amplified with the increase in MWCNT concentration. It is also worth mentioning that the crystallization peaks become wider and move to lower temperatures with the increase in cooling rate. Accordingly, Tcpt is reduced with increasing in cooling rate. Hence, the lower the cooling rate the higher the temperature at which the crystallization process commences and completes.
43
Consequently, the critical lamella thickness (lc*) distribution curve and its peak point are progressively shifted to lower temperature with the increase in the cooling rate.
44
DSC non-isothermal crystallization (a) exotherms and (b) endotherms of the materials. DSC crystallization and melting parameters determined from the non-isothermal cooling and heating experiments, respectively.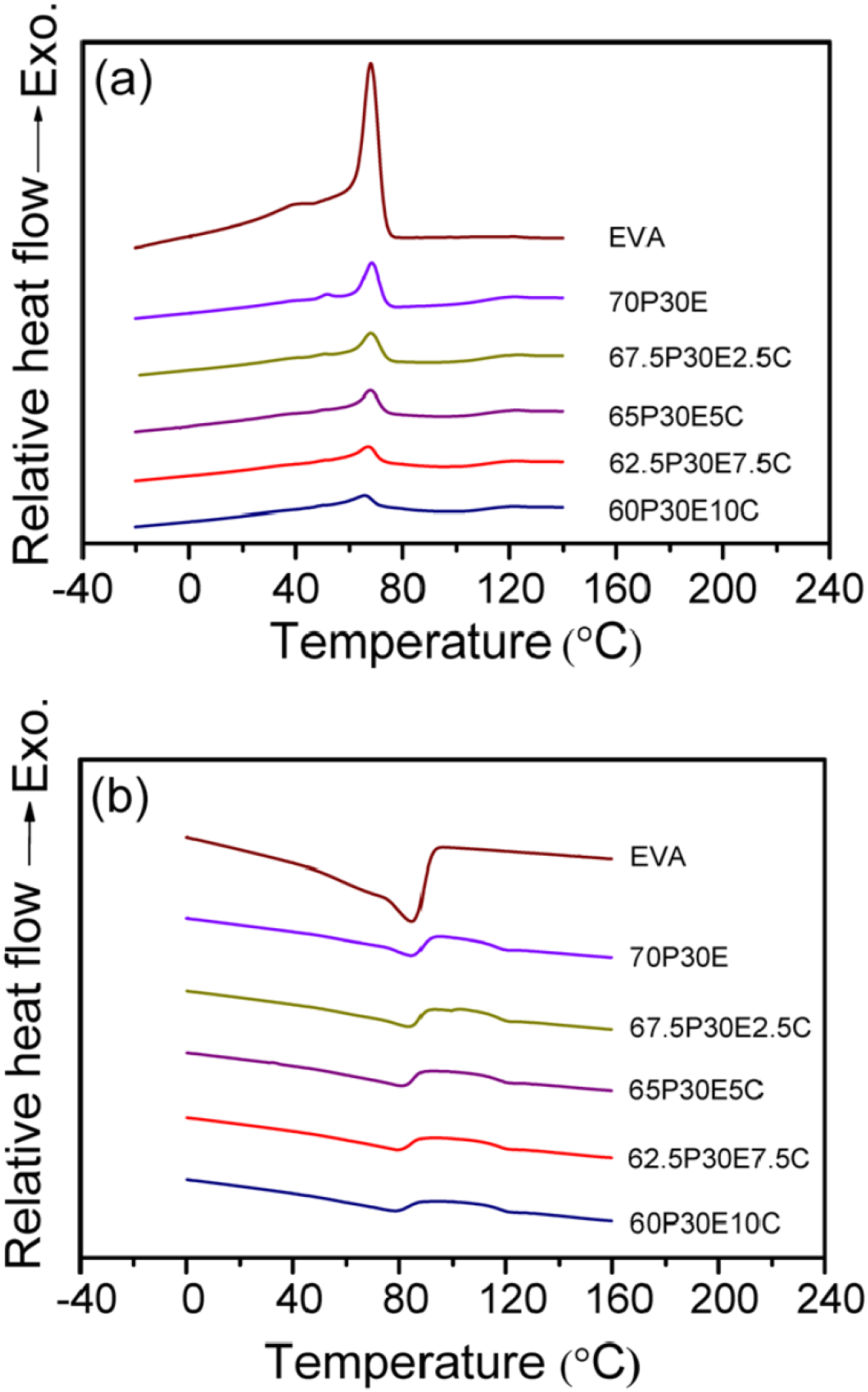
Rheological Experiments
The rheological behaviors of PMMA/EVA/MWCNT ternary nanocomposites at 180°C were analyzed by means of a small amplitude oscillatory shear test. The resulting ω-dependent storage modulus (G') and complex viscosity (η*) are depicted in Figure 2. The ω-dependent values of G' and η* of pristine EVA were found to be far lower than those of PMMA, the unfilled blend and all the nanocomposites. It was illustrated that the molecular mobility of PMMA as the major component was significantly increased with the EVA component, which acted as a plasticizer, in the blend. However, the mobility decreased with the addition of the MWCNT nanofillers in the nanocomposites. All materials behaved as Newtonian fluids at low ω region. The G' and η* of the nanocomposites were changed relatively analogous to that of the 70P30E blend, except for the fact that both values were superior for the nanocomposites having higher MWCNT content, especially in the low ω region. While the G' and η* values for the blend and nanocomposite containing 2.5 wt% nanoparticles were lower than those of PMMA, the two values were progressively increased with the MWCNT loading for the nanocomposites having 5 wt% MWCNT and beyond. The gradually development of the MWCNT network structures within a filled material could normally impose some restrictions on the long-range molecular motions of the polymeric chains and accordingly enlarge the two quantities.
45
Our findings are similar to the most reported results such as those published by Lee et al.
46
for polylactide/polypropylene (PP)/MWCNT and Narimani et al.
47
for PP/ethylene–propylene–diene monomer (EPDM)/SWCNT systems. However, Khasraghi et al.
48
reported that G' and η* of the ultra high molecular weight polyethylene (UHMWPE)/high density polyethylene (HDPE)/MWCNT nanocomposites were lower than those of their unfilled counterparts at low ω region. This behavior was attributed to the adsorption of high molar mass polyethylene chains onto the nanofillers’ surfaces. The plots of η* and G’ against ω for the pristine polymers and unfilled and filled materials at 180°C.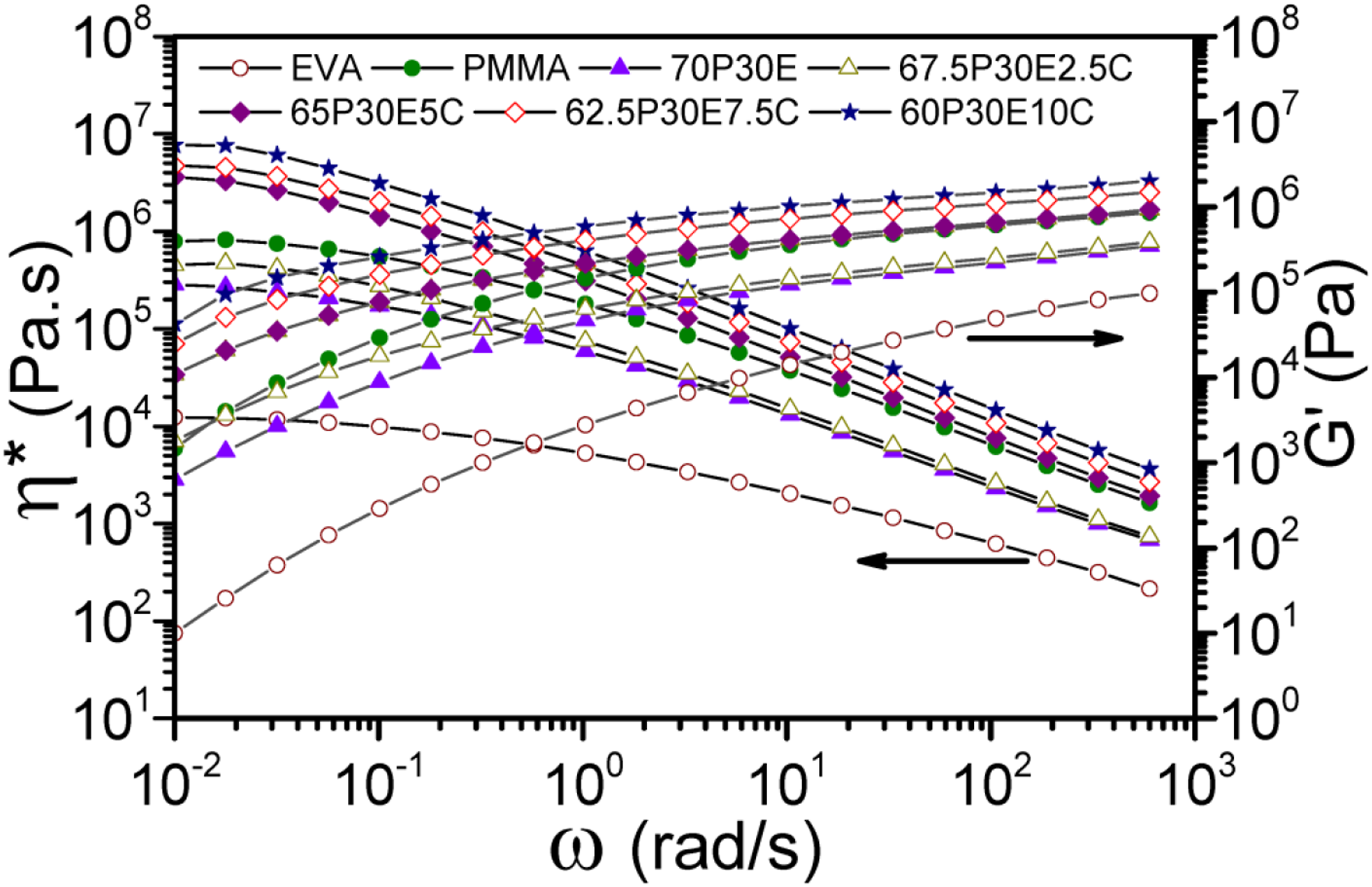
The slopes of G’ and G” and the values of n for the samples.

Tanδ as a function of ω is depicted in Figure 3. It was observed that the tanδ values of the pristine polymers, blend and nanocomposites were larger than 1 at low ω region. However, the value decreased with the increase in ω and finally reached below than 1 for all the materials even for EVA. Tanδ is defined as the ratio of loss and storage modulus, hence, it could be realized that G” > G’ for the materials at low range of ω.
49
Shear deformation resulted in the fractional orientation of the molecules in polymeric chains, leading to the reduction of tanδ of the nanocomposites with the increase in ω. The PMMA/EVA/MWCNT nanocomposites showed smaller tanδ values at lower ω values with the increase in MWCNT concentration, reflecting that the incorporation of MWCNT improved the elasticity of the nanocomposites.
50
The plot of tanδ against ω for the pristine polymers and unfilled and filled materials at 180°C.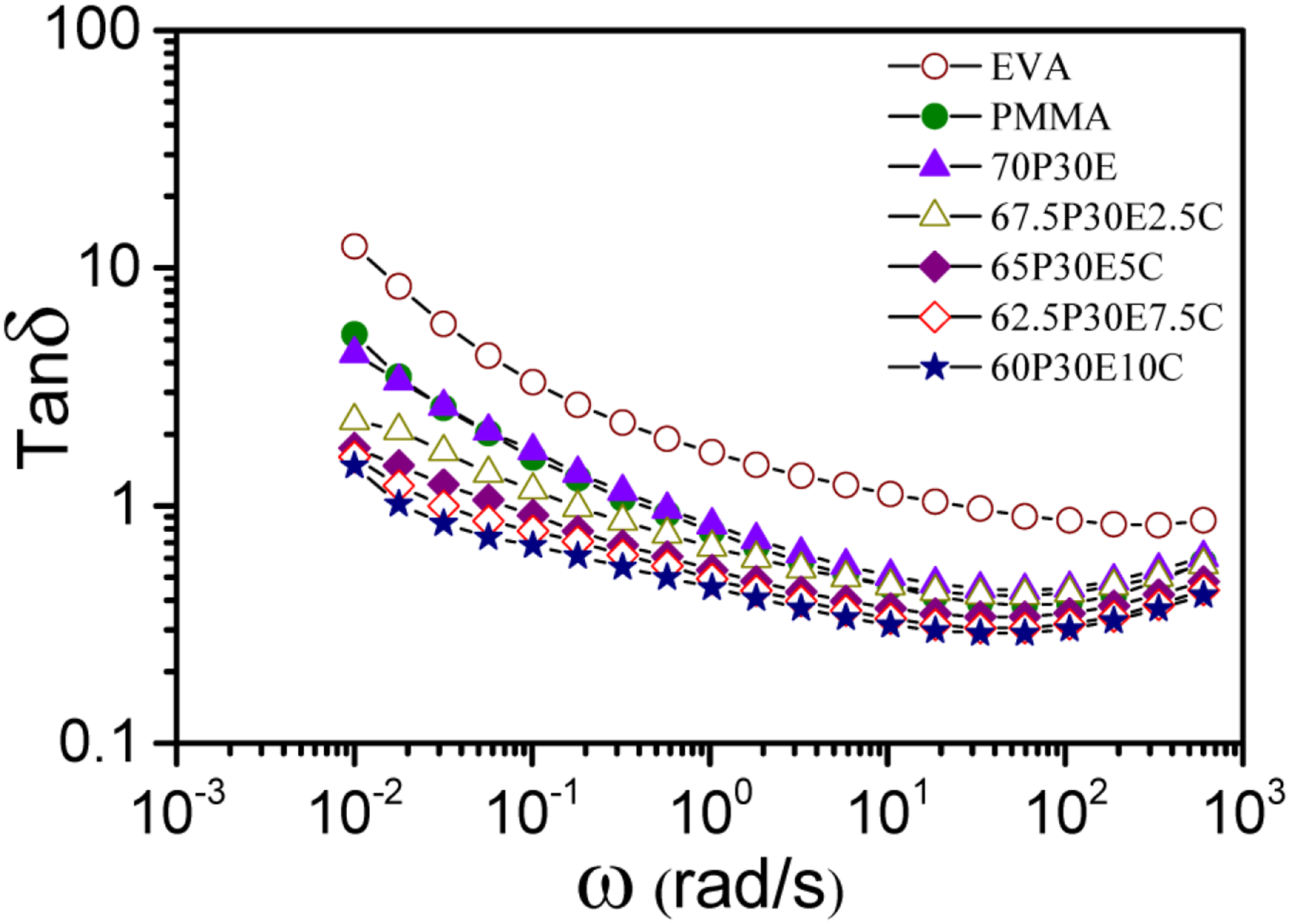
The variations of G’ and G” with MWCNT content at various selected ω are depicted in Figure 4(a) and (b), respectively. The plots revealed that the enlargement of G’ with the nanofiller content was higher than for G”. In addition, it was observed that the increase in both values was non-linear and the increase was more noticeable for the nanofiller loadings lower than 5 wt% than beyond. The behavior was also seemed to be more prominent for low ω. Our results were consistent with those reported by Pötschke et al.
51
for the polycarbonate/MWCNT system. The plots of (a) G’ and (b) G” with the nanofiller loading at various ω.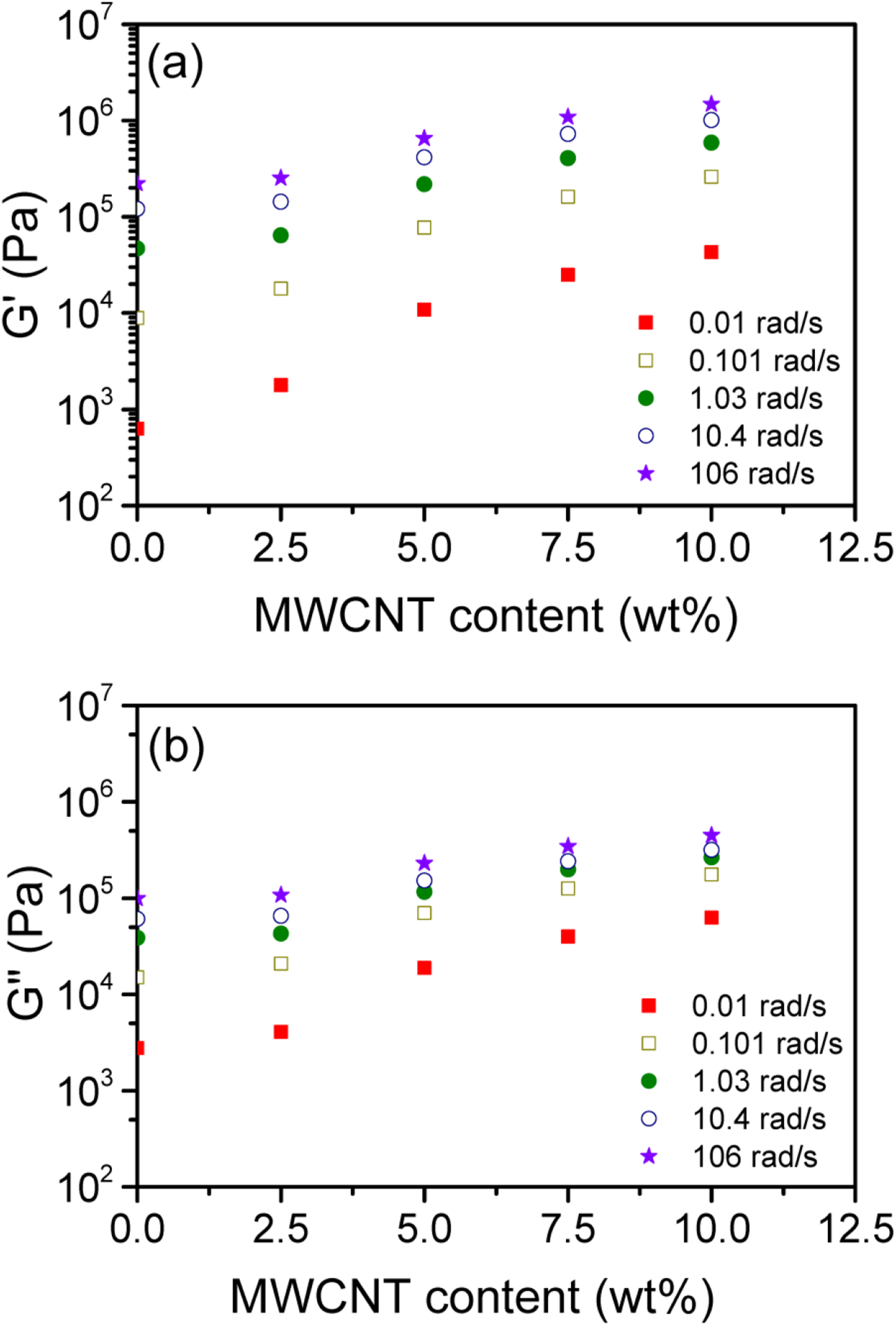
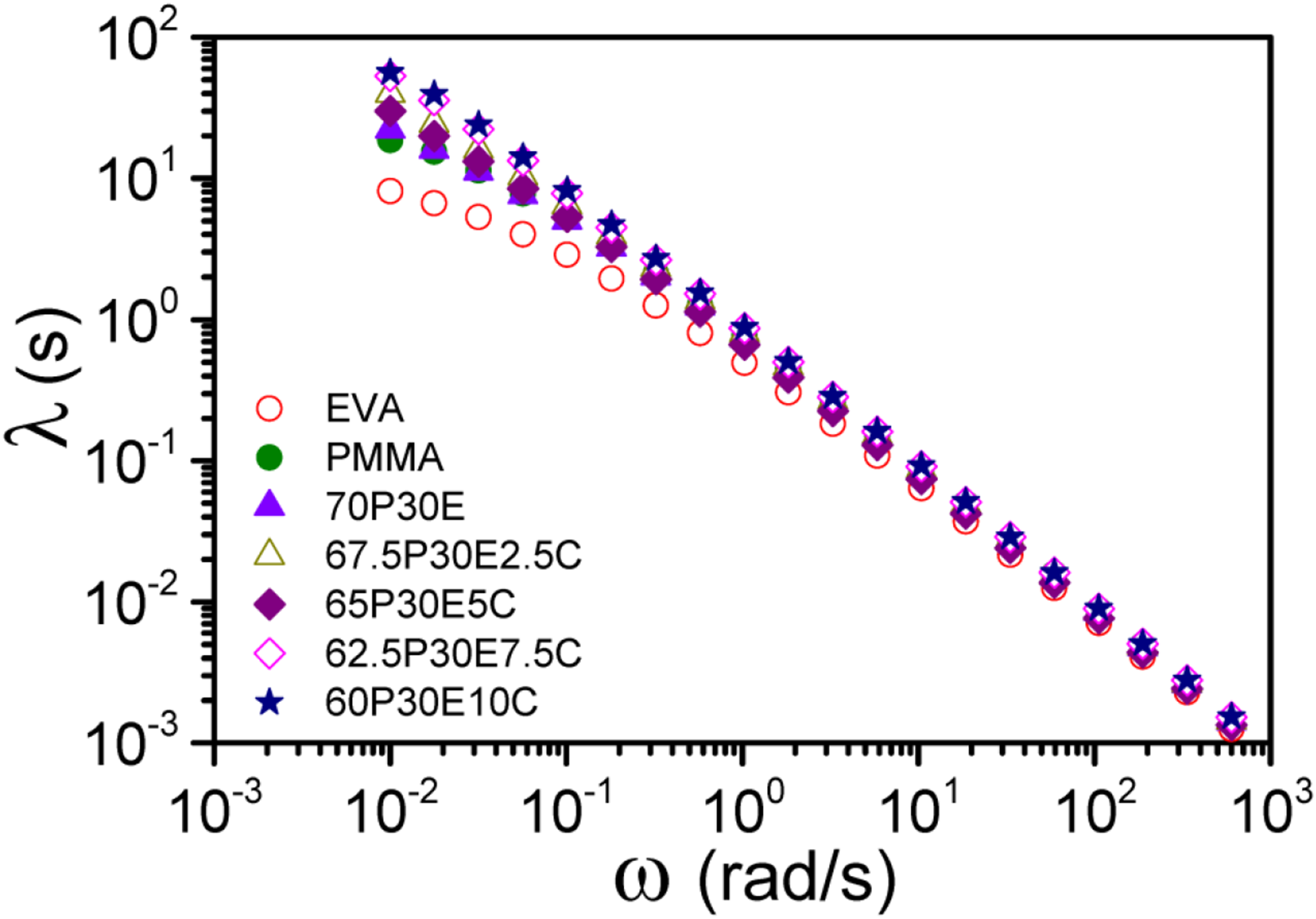
The relaxation time (λ) of the materials under dynamic shear experiments that experienced the pseudo-structures can be evaluated using the following equation
50
; The log-log plot of λ against ω for different materials.
Generalized Maxwell Model
The correlation between stress and deformation for a viscoelastic material at small deformations can be exhibited by the Boltzmann superposition principle as follows;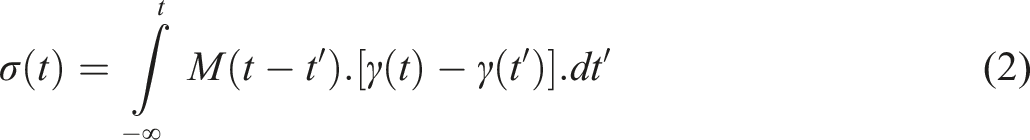

It is required to signify the modulus by a mathematical function, and the function, which is frequently employed with the above model, is the generalized Maxwell model (GMM). A Maxwell element comprises of one spring and one dashpot in series, however, a number of Maxwell units are connected parallel in GMM.
54
The dashpots are employed to resemble the viscous or time dependent property of the part of a substance, which is involved in energy dissipation. The springs are utilized to resemble the elastic property of the part of a substance, which is engaged in the conservation of energy.
55
An extra spring can also be added to the system with the shear modulus of Ge to represent the equilibrium relaxation modulus. Hence, the relaxation model for N Maxwell elements and the single spring could be written as
53
;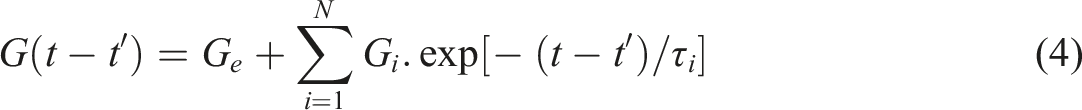
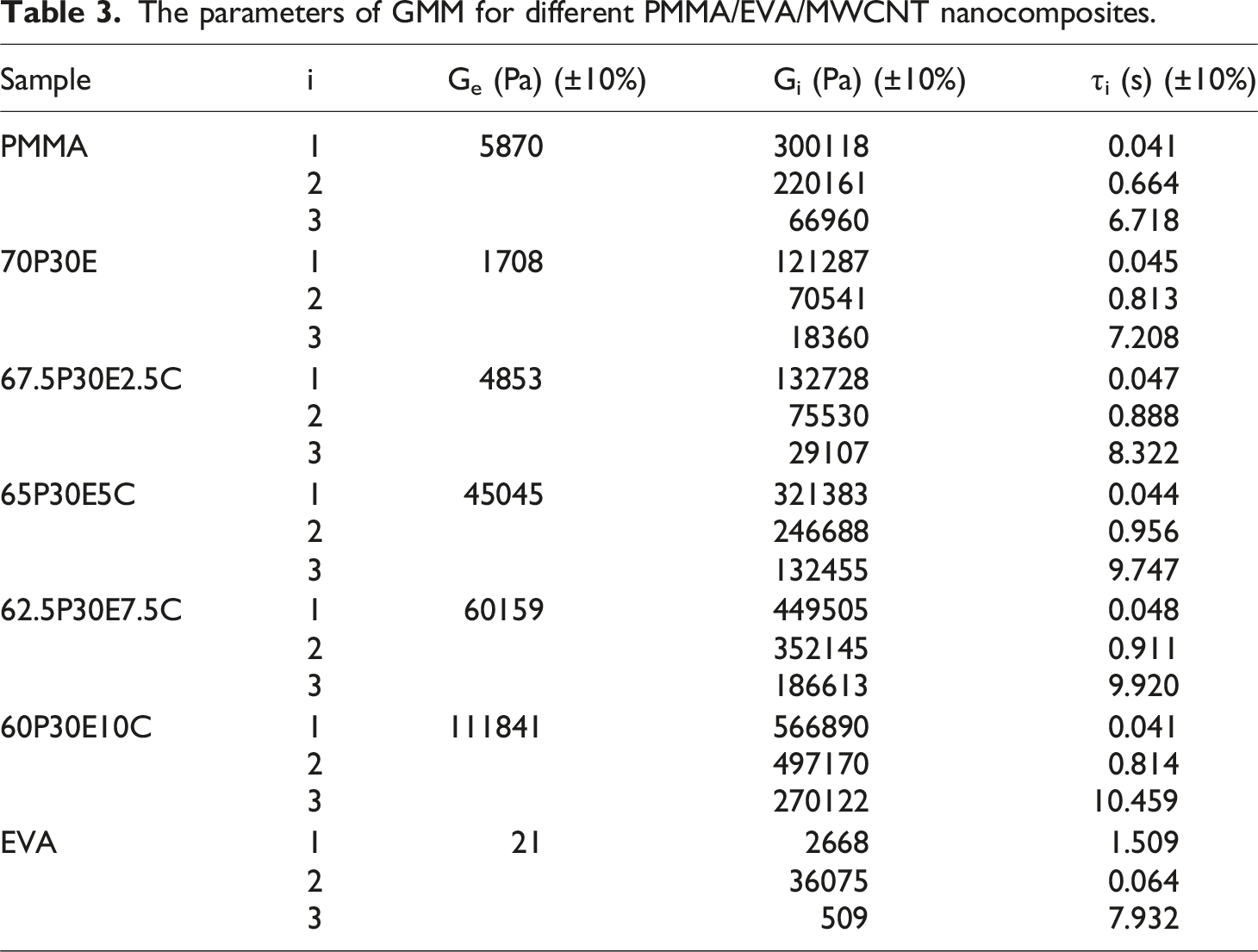
The Schwarzl model,56,57 containing three terms, was used to convert the rheological data to G(t). Then, to find out more knowledge about the materials, the dependence of G(t) on time were analyzed by means of GMM. The semi-log plot of the experimentally obtained G(t) values against time and those of calculated by fitting the GMM model, having three Maxwell units and an additional spring, are represented as symbols and solid lines, respectively, in Figure 6. As it is observed, G(t) is continuously ameliorated with the amounts of the nanotube loading. The difference between the values is more noticeable at longer times when the variation in G(t) is less dependent on time. The values of the parameters of the GMM model are also tabulated in Table 3. It was found that there was a reasonable agreement between the experimental values and the data generated after being analyzed by the GMM model. As it can be observed, the values of Ge, G1, G2, G3 and also τ3 were progressively increased with the addition of MWCNT concentration in the nanocomposites. This was in agreement with the fact that the nanofillers could impose some restrictions on the macromolecular movements. The semi-log plot of G(t) versus time for the materials studied. The parameters of GMM for different PMMA/EVA/MWCNT nanocomposites.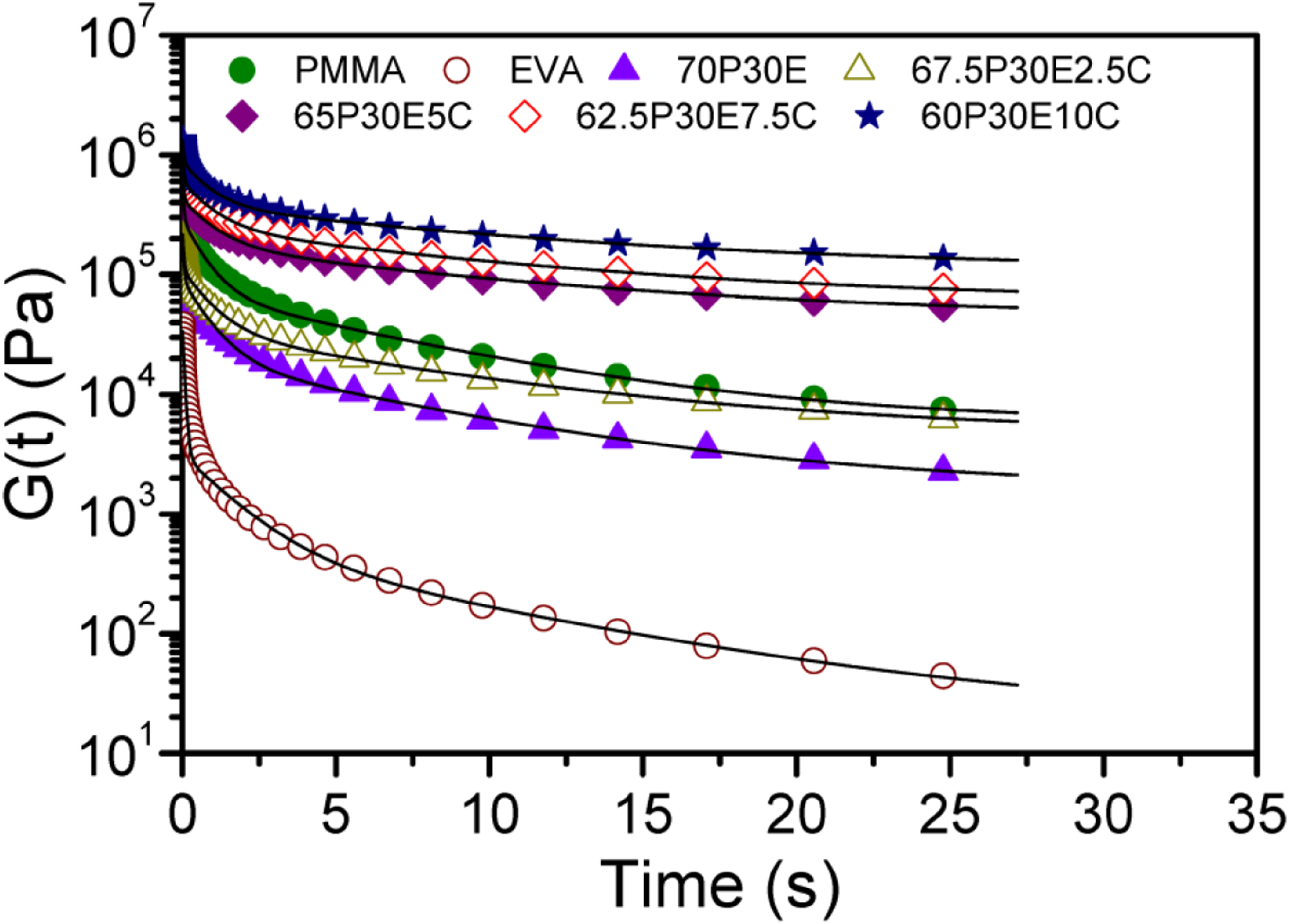
X-Ray Diffraction Analysis
X-ray reflections usually indicate a series of peaks that are related to the crystalline structure of a substance.
58
The X-ray diffraction patterns of the PMMA/EVA blend and their nanocomposites versus the Bragg angle are depicted in Figure 7. EVA is crystallized, similar to polyethylene, with the crystallographic planes (110) and (200) of the orthorhombic unit cell, which are superimposed on its amorphous halo. These crystallographic peaks were appeared at the diffraction angle of about 21.3 and 23.4°, respectively.
59
It could be inferred from the broad background area of EVA diffraction response, in comparison with that for the peaks area, that EVA had a higher portion of the amorphous structure in respect of the crystalline structure.
60
The relatively low XC of EVA, which depends on the amount of VA in the copolymer, could be related to the disordered positioning of ethylenic units, affected by the VA units.
61
However, no distinctive peaks could be observed for PMMA excluding a broadened halo, consistent with the fact that the material was totally an amorphous polymer. The X-ray diffraction spectra of the PMMA/EVA blend and nanocomposites showed similar patterns but with the peaks, which their intensities became progressively smaller as the nanofiller loading was increased. This observation is consistent with the results reported by others in literature.
62
In our case, it might be associated with the delaying effect of the PMMA and MWCNT on EVA crystallization.
63
The former materials acted to decrease the EVA crystallinity in the nanocomposites, which has already been corroborated by DSC in this work. For the nanocomposites, the introduction of the MWCNT results in the improvement of compatibilization, since it is in the amorphous phase that the compatibilization of the two polymers can be taken place.
61
The existence of the crystalline peaks of EVA in the blend and other compositions at relatively the same positions evidently revealed that the crystalline structure of EVA remained unaffected for the blend and nanocomposites. It is also worth mentioning that no detectable reflection could be found at the Bragg angle of 26, which were assigned to the graphite (002) planes of the concentric tubes of the MWCNT.64,65 This could be attributed to the good dispersion of the carbon-based nanoparticles in the matrix. The same was claimed for the dispersion of MWCNT in UHMWPE/HDPE/MWCNT system.
48
XRD spectra of the materials studied.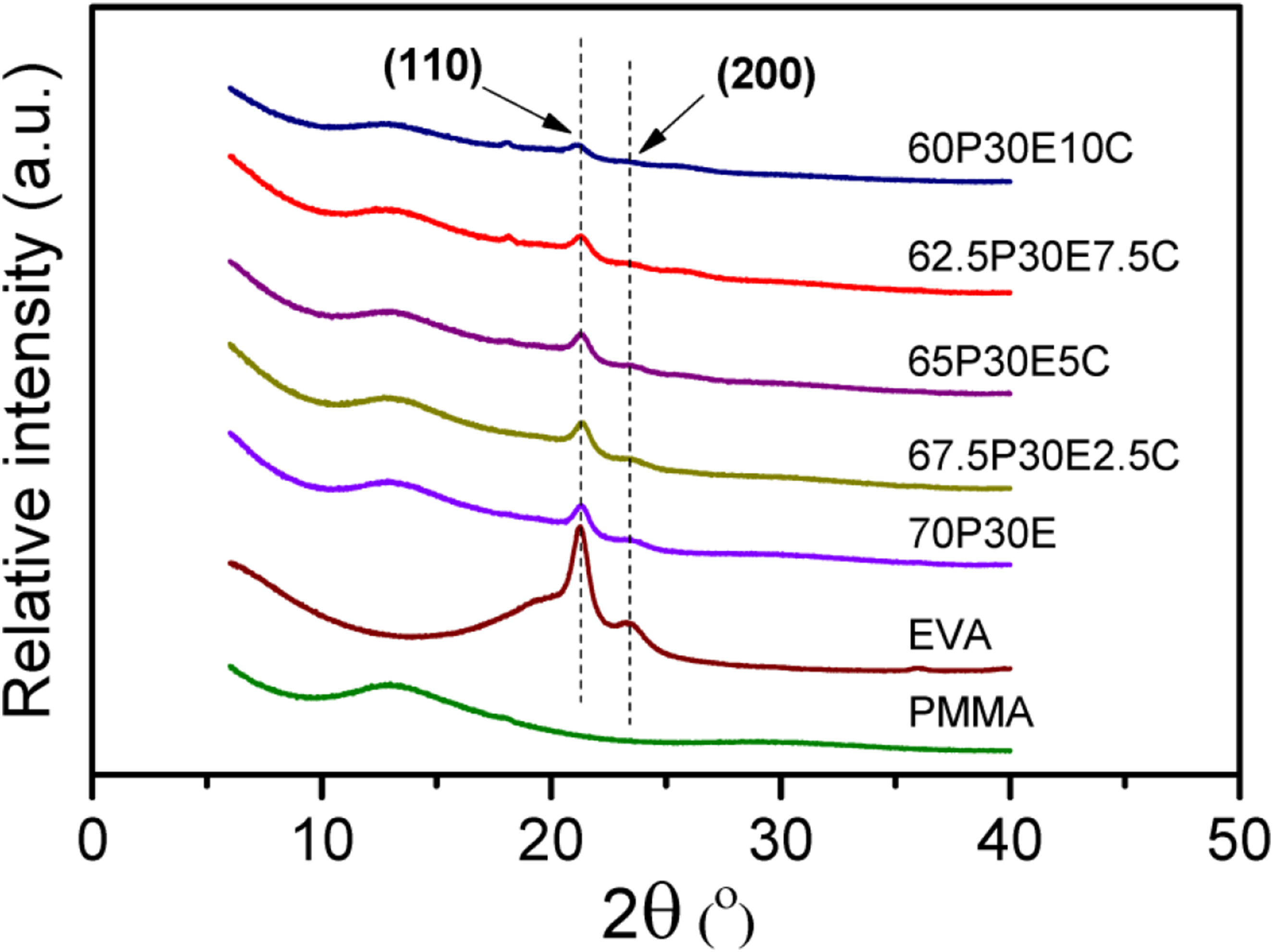
The average crystallite size (LC) was calculated for the blend and nanocomposites from the broadening of the main peak of EVA, plane (110), using the Scherrer equation66,67;
Thermogravimetric Analysis
TGA experiments were carried out as a complementary method to assess the thermal stability and decomposition behavior of the neat polymers and PMMA/EVA/MWCNT nanocomposites. The variation of sample weight with temperature, for each material, in air and nitrogen atmosphere is shown in Figure 8(a) and (b), respectively. To evaluate the thermal stability of these materials, the temperature at 5 wt% (T5), 10 wt% (T10), 20 wt% (T20) and 50 wt% (T50) weight loss in the oxidative and inert atmospheres are tabulated in Tables 4 and 5, respectively. TGA analysis of the materials in (a) air and (b) nitrogen atmosphere. TGA results for different materials in air. TGA results for different materials in nitrogen.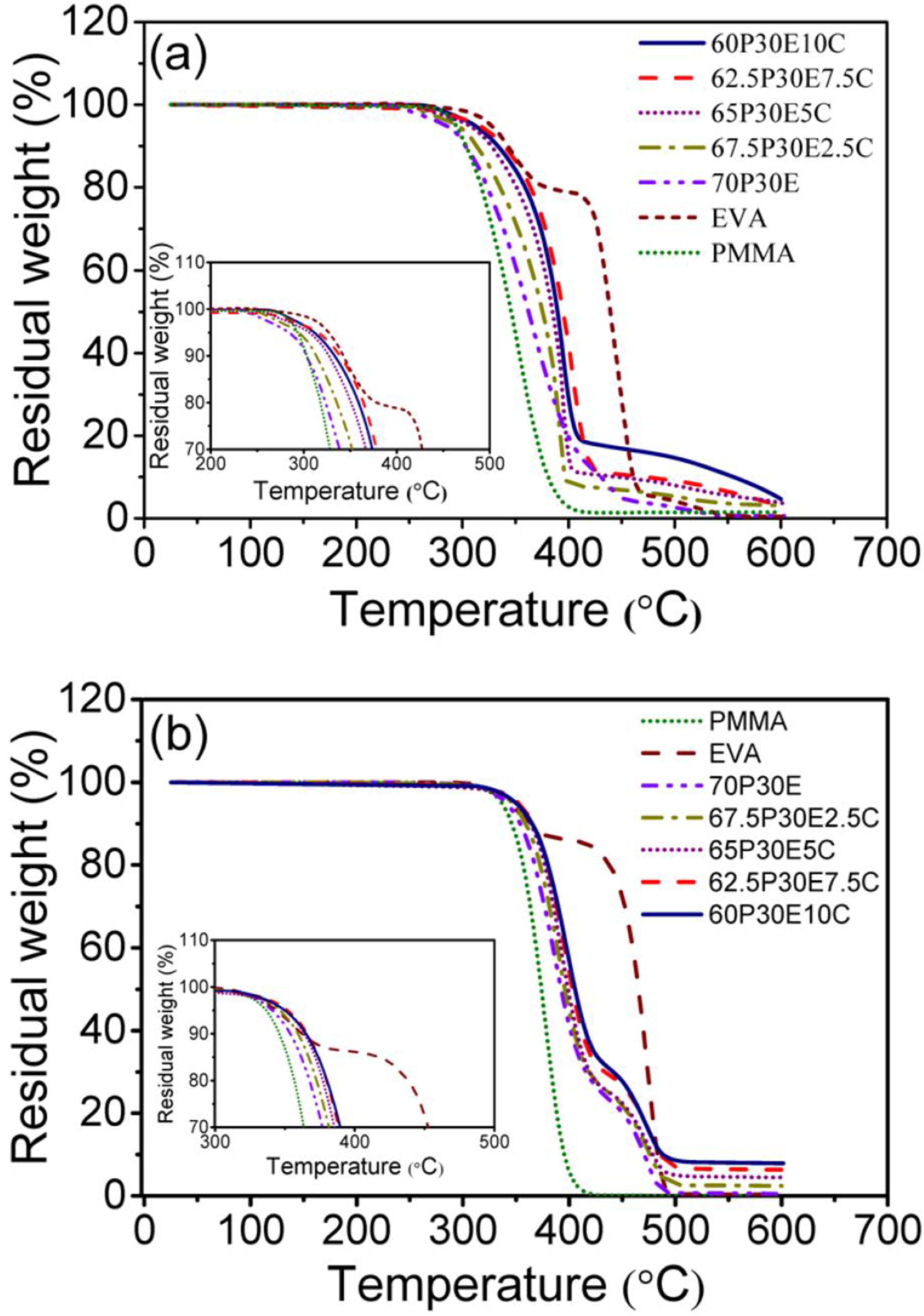
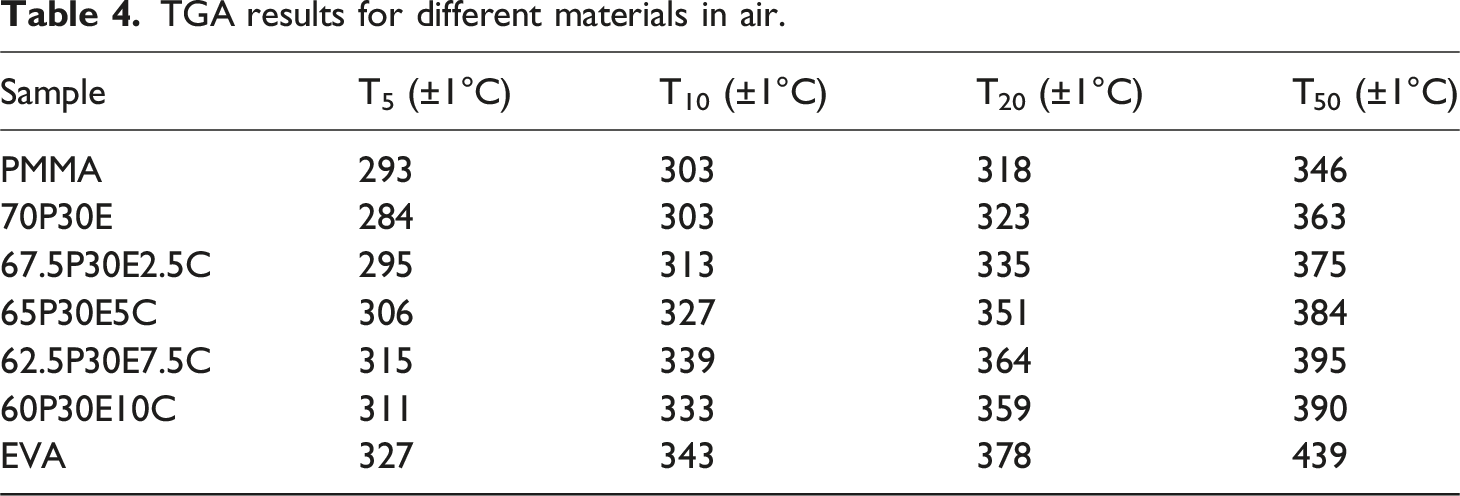
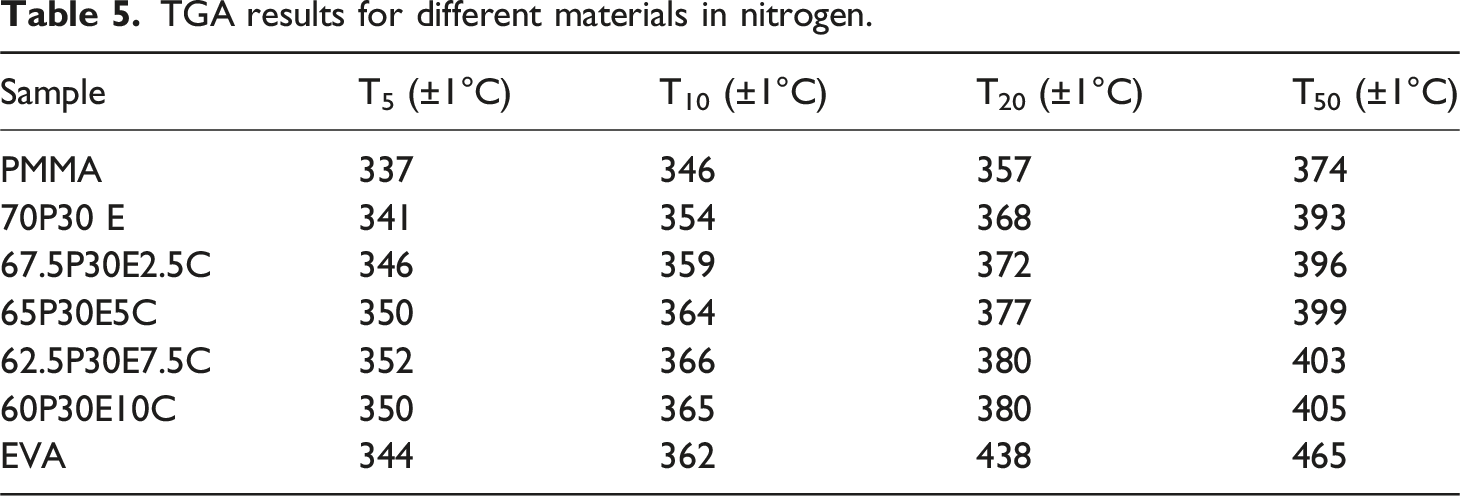
As it can be seen, PMMA and EVA were degraded in one and two steps in both nitrogen and air atmospheres, respectively. PMMA was decomposed in the temperature range 337°C–430°C in nitrogen and 293°C–416°C in air. The former was due to the random C-C bond scission of the polymer backbone and the later was that of the effects of both depolymerization and C-C bond breakage of PMMA chains. 70 However, EVA was decomposed in nitrogen and air in the temperature range 344°C–498°C and 327°C–555°C, respectively. The first degradation step of EVA, was assigned to the removal of acetate side groups as acetic acid (i.e. deacetylation) which resulted in leaving behind an unsaturated polymer chains or polyene in both atmospheres. The second step was related to the allylic chain scission of the polyene in nitrogen and its aromatisation into a char in air. The char was then oxidized into carbon dioxide above 500°C, leading ultimately to the complete decomposition of EVA.71–73 In addition, it was reported that all degradation steps in an inert or oxidative atmospheres reflected endothermic and exothermic effects, respectively. 71 The two pristine polymers almost experienced total degradation with remaining little or no chars in both atmospheres. Thermal stability of the nanocomposites in both environments was higher than those of the pristine PMMA and the unfilled blend. Moreover, the maximum efficiency in increasing the thermal decomposition was achieved for the nanocomposite containing 7.5 wt% MWCNT, regardless of the type of atmosphere employed. This could be attributed to the MWCNT dispersion state as well as its high aspect ratio. The increase in thermal stability of a polymer nanocomposite containing MWCNT could be assigned to several factors. The transportation of degraded polymer volatile products from the condensed phase to the gas phase could be prevented by the barrier effect of the nanofiller network.74,75 The degradation process could be retarded by the act of the MWCNT as an antioxidant due to its strong radical scavenging capability 76 and also its ability in immobilizing the free radical chains formed in the course of degradation. 77 The thermal motion of a polymer could be confined and the activation energy of degradation might be improved by the interfacial interaction between the macromolecular chains and MWCNT nanoparticles. 78 The heat dissipation inside the nanocomposites could be occurred with ease due to the heat conductivity of the MWCNT network and thus the macromolecules degradation could be postponed to higher temperatures.69,79 In addition, MWCNT was found to be more beneficial in increasing the thermal stability of the nanocomposites under oxidative environment such as air than that of nitrogen. For instance, T5, T10, T20 and T50 for 62.5P30E7.5 C increased in air by 31, 36, 41 and 32°C and in nitrogen by 11, 12, 12 and 10°C, respectively, in comparison with those of 70P30E.
Conclusions
The rheological properties, crystallization and melting behavior as well as the thermal stability of the non-studied system of PMMA/EVA/MWCNT nanocomposites, containing different nanofiller concentrations, were investigated in this work. The rheological results of the nanocomposites disclosed that the GMM parameters of Ge, G1, G2, G3 and τ3 were steadily augmented with the increase in the nanofiller loading. X-ray measurements revealed that no practical shift of diffraction reflections could clearly be observed, suggesting that the EVA crystal lattice planes were not altered with the inclusion of the MWCNT. However, the incorporation of the nanofiller content steadily diminished the crystallite or grain size of plane (110). Moreover, it was found that the nanofiller could be more capable in enhancing the thermal stability of the materials in an oxidative than that for an inert atmosphere. The utmost effectiveness in ameliorating of the thermal stability was obtained for the composition having 7.5 wt% MWCNT in both air and nitrogen atmospheres studied. The DSC data analysis disclosed that the values of Scry, Smel and XC of the nanocomposites gradually reduced with the increase in the MWCNT content. However, the opposite trend was observed for the values of W. Conclusively, the results showed that MWCNT was able to successfully fortify and thermally more stabilize the nanocomposites in comparison with the unfilled system. It is now the time to bring up the limitations of this article. Further characterization experiments such as transmission electron microscopy (TEM) and dynamic mechanical thermal analysis (DMTA) should be executed to more scrutinize the performance of the MWCNT in the system. Moreover, the crystallization and degradation kinetics could also be studied in order to extend our knowledge concerning the influence of the reinforcing component on both phenomena.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has been supported by Iran Polymer and Petrochemical Institute (Grant No. 31761207).
