Abstract
Prevulcanized natural rubber latex (PVNR) and acrylic emulsion (AR) blends for coating applications were prepared. The influence of blend proportions (30%, 40%, 50%, 60% or 70% AR) on morphological, tensile, adhesion and thermal properties were investigated. The morphology of the blends changed from PVNR continuous matrix type to a rubber particle dispersion when the AR fraction was increased. The greatest increases in tensile strength were achieved with 70% AR, for which partial compatibility between the phases may be occurred. The contact angle test revealed that wettability of the blend increased with AR proportion. This resulted in improved adhesion of the blends on substrates as confirmed by work of adhesion and peeling tests. The greatest peeling forces on wood and metal substrates were achieved at 70% AR, with respective improvements by about 470% and 84% over plain PVNR. Thermogravimetric analysis showed a single derivative degradation peak in the 70% AR sample while others displayed two degradation derivative peaks. Fourier transform infrared spectroscopy suggested possible interactions between the phases involving proteins in the PVNR particles and carbonyl groups of the AR. The results suggest that the PVNR/AR blends may find applications in wood and steel coating, and in adhesive products.
Introduction
Natural rubber (NR) is a polyisoprene polymer generated naturally, being a renewable resource. It is a crucial component in a variety of products. Owing to its distinct physical and chemical properties, it is extensively used in various industries. 1 However, the NR is easily degraded by oxygen and ozone because of the double bonds in the molecular backbones. 2 Further, its non-polar characteristics also make it less effective when exposed to oils and other hydrocarbon liquids. 3
Many strategies exist for producing high-performance rubber that can fulfill the requirements of use in engineered rubber products. Aside from chemical modifications and use of filler, blending different polymers is another effective technique. Blending two polymers is a particularly attractive, simple, and cost-effective method for creating novel materials possibly combining beneficial properties of the individual component polymers. 4 Such blends can be prepared using a variety of techniques, including melt mixing, solution mixing, and latex mixing. 5 Blending in a latex state is more interesting than the other methods, because NR is readily obtainable in latex form. It has been found that there are many special advantages to this blending technique, such as requiring less energy than melt blending and no solvent like solution blending. 6 Furthermore, this approach has demonstrated excellent mechanical properties due to the very fine stable dispersions created. 7 Various types of synthetic polymers have been used in blends with NR in the latex state, to obtain blends with improved properties: for instance, styrene butadiene rubber latex, 8 poly vinyl chloride emulsion, 6 acrylonitrile-butadiene rubber latex,9,10 poly methyl methacrylate, 11 and methyl methacrylate grafted natural rubber. 12
Acrylic rubber is among the most interesting types of rubber. Its molecular structure consists of polar saturated and ester functional groups. 13 It is commercially available as an emulsion, and it tends to offer benefits when combined with NR. Recently, Worlee et al., 14 prepared polymer blends based on prevulcanized-NR latex blended with acrylic resin. They found that the tensile strength increased with addition of acrylic and was maximal at 30/70 (w/w) blend ratio of prevulcanized-NR latex/acrylic resin. This blend was very interesting as it could be utilized as wall paint. However, the effects of acrylic on other properties such as morphology, dynamic mechanical and thermal properties of the blends were not discussed in detail.
To obtain a deeper understanding of this high potential blend, different blend ratios of prevulcanized-NR latex and acrylic emulsion were prepared in this study. The influences of blend ratio of prevulcanized-NR latex and acrylic emulsion are discussed in detail. The effects of blend ratio on morphological formations were investigated by using scanning electron microscopy (SEM). The possible interactions inducing compatibility between phases were investigated by dynamic mechanical analyses (DMA), Fourier transform infrared spectroscopy (FTIR), and thermogravimetric analysis (TGA). The changes in mechanical properties were examined by tensile testing. The wettability and adhesion properties were estimated by means of water contact angle and peeling test, respectively.
Experimental
Materials
Concentrated natural rubber latex (HA with 60% dry rubber content) was purchased from Yala Latex Co., Ltd (Yala, Thailand). Potassium hydroxide solution (10% KOH), potassium oleate emulsion (20% K-Oleate), acrylic emulsion (56%), dispersing agent (2%), zinc-n-diethyl dithiocarbamate (50% ZDEC), zinc 2 -mercaptobenzothiazole (50% ZMBT), Wingstay L (50% Wingstay L), sulfur (50% S), zinc oxide (50% ZnO), calcium carbonate (50% CaCO3), and titanium dioxide (50% TiO2) were all purchased from Chemical Village Co., Ltd. Samutprakarn, Thailand. All chemicals were used as received.
Sample preparation
Prevulcanized-NR latex preparation
Formulation of PVNR in parts per hundred of rubber (phr).

Emulsion preparation
Formulations for preparing emulsions.
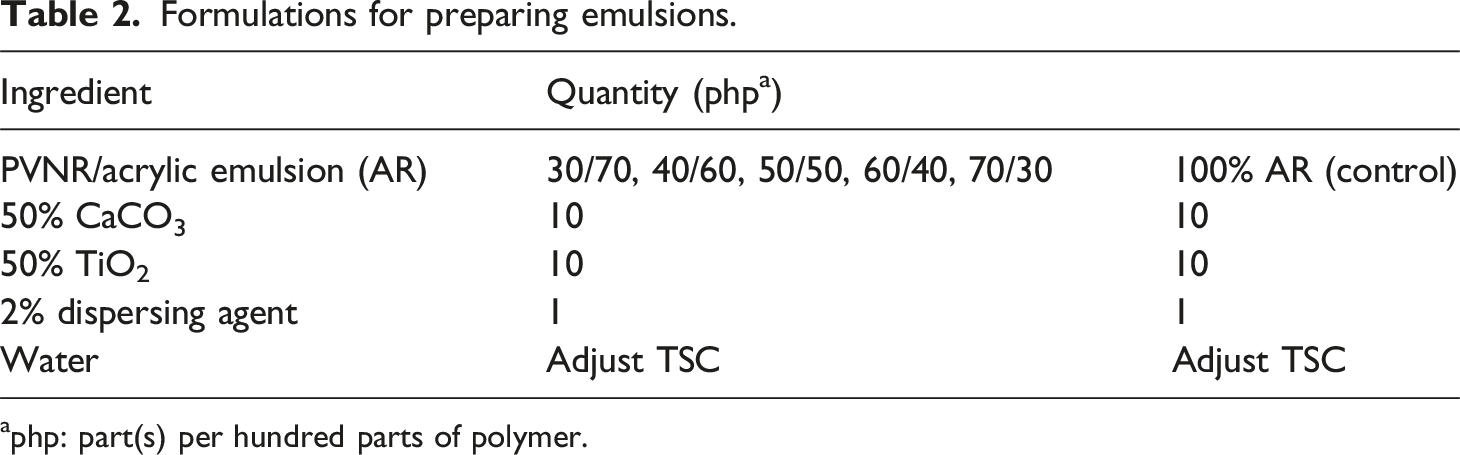
aphp: part(s) per hundred parts of polymer.
Fabrication of thin films
The resultant emulsions were cast in silicon molds. The cast films were then dried in an oven for 72 h at 50°C to obtain about 2 mm thick films for property tests. The blends having different PVNR/AR ratios, that is, 70/30, 60/40, 50/50, 40/60, or 30/70, were labeled with 30% AR, 40% AR, 50% AR, 60% AR and 70% AR respectively. The pure PVNR is labeled PVNR and the pure AR mixed with the ingredients listed in Table 2 was assigned 100% AR label.
Characterization
Morphological property test
The morphologies of specimens with different blend ratios were examined by scanning electron microscopy (SEM, FEI Quanta 400FEG, Netherland) in the back-scatter mode (SEM-BSE). The samples were cryogenically fractured and vapor stained with osmium tetroxide (OsO4) solution before examination.
Tensile property test
Tensile properties of the blend specimens were measured using a universal tensile testing machine, the LR5K Plus (Lloyd Instruments, UK) run with a cross-head speed of 500 mm/min. The test samples were cut into dumbbell shapes with 75 mm length, 4 mm section width, and 2 mm thickness in accordance with ISO 37 (type 2). The measurements were done at room temperature and the averages of five samples are reported.
Contact angle measurement
Contact angle measurements were conducted to evaluate the wettability of blend film surfaces. The test was done with the sessile drop method using a contact angle meter (OCA 15EC, Germany) at room temperature. Deionized water droplet was dispensed onto the film specimen with a micro-syringe. The average contact angle was calculated from at least five measured points on each sample.
Work of adhesion (Wadh) was determined by using the Young-Dupre model
15
as follows:
Peel strength test
Peel strength test was performed to determine the adhesion of PVNR/AR blends on a solid substrate. Two types of substrates were used in this study, that is, wood and metal. 90° peel tests were done by using a universal tensile testing machine, Hounsfield Tensometer H 10 KS (Hounsfield Test Equipment Co., Ltd, U.K.) at room temperature with 50 mm/min pulling rate.
The substrate preparation process varied depending on the type of substrate. For the wood substrate, a rectangular wood board with a 2.54 × 5.4 × 1 cm3 dimension was cleaned with tap water and dried at 80°C for 30 min before being coated with the blends. In contrast, the mild steel plate, with dimensions of 110 × 25 × 3 cm3, underwent grit blasting for 1 min to create surface roughness, ensuring mechanical interlocking between the coating and the substrate. The steel was then cleaned with toluene and dried at 80°C for 1 h.
The PVNR/AR blend emulsions were applied to each substrate using a brush. The coating weight was maintained at 0.5 g for an area of 6.35 × 2.54 cm2. After the coating was applied to the substrate, it was sandwiched with another piece of the same substrate. A 200 g metal weight was then placed on the joint to ensure proper adhesion. The coated substrates were dried in an oven at 50°C for 1 h and subsequently kept at room temperature for 24 h before testing.
Thermogravimetric analysis
Thermal stability of the specimens with varied blend proportions was assessed using a thermogravimetric analyzer, TGA8000 (PerkinElmer, USA). The test was carried out under a nitrogen atmosphere from 50°C to 700°C with a heating rate of 10°C/min.
Fourier-transform infrared spectroscopy (FTIR) analysis
Attenuated total reflectance–Fourier transform infrared (ATR-FTIR) spectra of pure PVNR and AR, and all the blend samples, were recorded using a Vertex 70 spectrometer (Bruker, Germany). All spectra were recorded from 4000 to 1000 cm−1 with resolution of 4 cm−1.
Results and discussion
Morphological properties
To understand the effects of blend ratio on the phase morphology of the PVNR and AR blends, SEM imaging was performed. Figure 1 displays the micrographs of PVNR/AR blends with different blend ratios obtained with SEM-BSE technique. It is clear that the morphology of all the PVNR/AR blends exhibited a two-phase structure, with one dispersed phase and another continuous phase. Since the NR phase was selectively stained by OsO4, the PVNR phase appears brighter whereas the AR appears darker in the images that agree well with a prior report.
16
It is interesting that the rubber domains appear spherical because the latex blending was done with stirring: the shear generated by stirring induced round domain shapes. In the 30% AR, 40% AR and 50% AR samples, the brighter PVNR phase is continuous since the PVNR particles populate the volume densely and have become interconnected. In contrast, some unlinked rubber particles are seen in the 60% AR case, and completely separate rubber domains are noted in the 70% AR sample. So, when the concentration of PVNR is high, the otherwise dispersed particles are forced in contact with each other, leading to the formation of interconnected rubber particles,
17
which contradicts the report
16
that found a co-continuous morphology even when 20% natural rubber was blended with 80% acrylic resin. This difference was due to the different blending techniques on making the blends. SEM-BSE micrographs of PVNR/AR blends having (a) 30% AR, (b) 40% AR, (c) 50% AR, (d) 60% AR, and (e) 70% AR.
Latex blending was used in the current study, whereas melt blending was applied in the prior study. 16 The results clearly indicate that the morphology of a blend depends strongly on the ratio of constituent components. A phase inversion occurred when the amount of AR was more than 60% (40/60 PVNR/AR), changing from continuous rubber matrix to dispersed rubber domains. Phase inversion is a typical phenomenon on change of the blend composition or of viscosity ratio.18,19
The size of rubber particles in various blends was measured by using image J software and the results are also included in Figure 1. The rubber particles varied in size (about 0.9 – 1.1 µm) depending on the proportions of PVNR and AR. The small variation of rubber particle size was tentatively attributed to the presence of a dispersing agent (a surfactant). Surfactants decrease the interfacial tension between phases in a blend, slowing down the coarsening of the domain structures.7,20 However, the smallest size spherical rubber droplets were found in the 70% AR case (about 0.91 µm) where the rubber was the dispersed phase.
Figure 2 presents the micro-fractured surfaces of PVNR/AR blends at different blend ratios. All blends exhibited a rough texture with a few voids across the surfaces, particularly in samples with lower AR proportions (30% AR and 40% AR). As the AR content increased, these voids gradually disappeared. The presence of voids was likely due to moisture in the PVNR phase, which decreased with higher AR content. SEM images of the fracture surface of the blends with different blend proportions (a) 30% AR, (b) 40% AR, (c) 50% AR, (d) 60% AR, and (e) 70% AR.
Tensile properties
Figure 3 demonstrates representative stress-strain curves of the PVNR/AR blends. The results obtained from the tensile tests in terms of 100% modulus, tensile strength, and elongation at break is summarized in Table 3. Clearly, the stress-strain behaviors of the pure PVNR and AR are completely different. The stress of the AR sharply increased at a low strain, and break happened after passing through a yield point because AR is a rigid polymer with a high Tg.
14
In contrast, the stress of the PVNR gradually increased at low strains and drastically increased at a high strain due to the influence of strain induced crystallization. Stress-strain curves of the blends with different blend proportions. 100% modulus, tensile strength, and elongation at break for the blends with different blend ratios.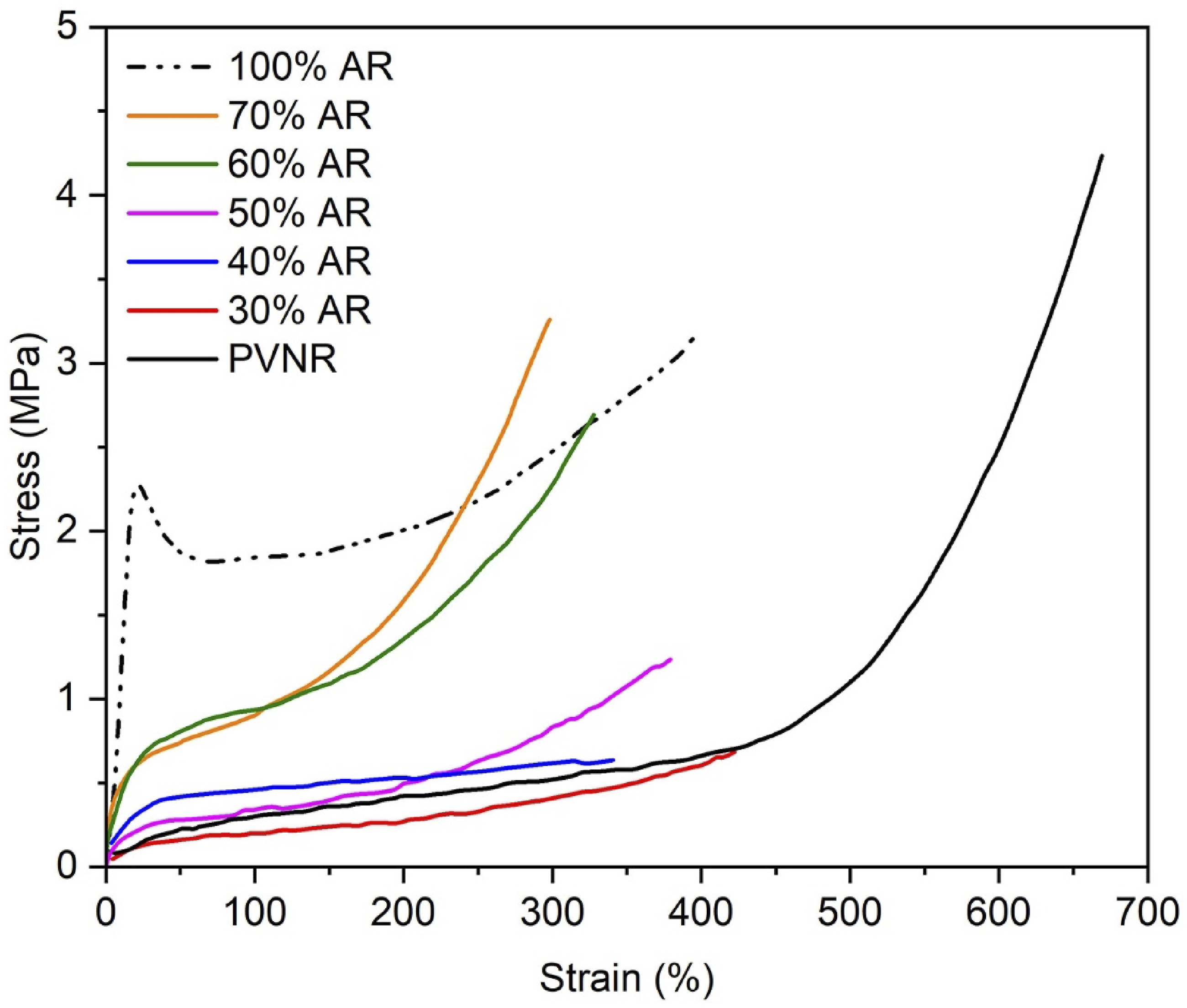
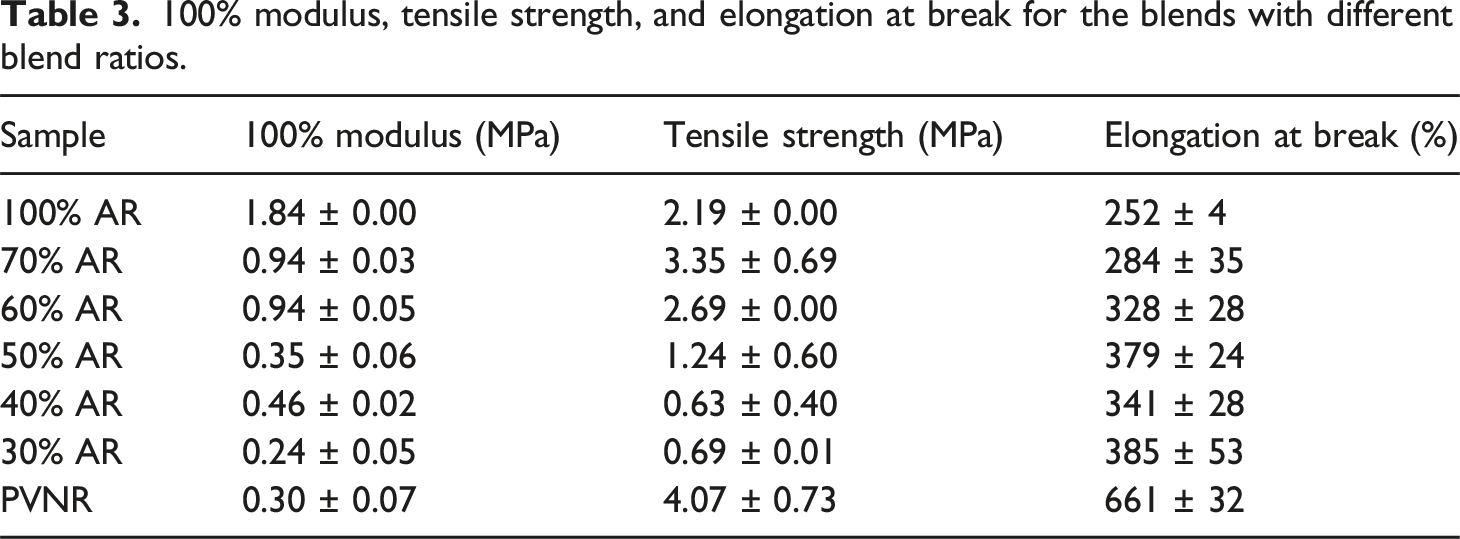
On increasing the AR content, the modulus at 100% strain of the PVNR/AR blend increased over the PVNR while the extensibility decreased due to hardening effect of the AR. Considering tensile strength, it depended strongly on the blend proportions. The highest tensile strength of about 3.35 MPa was found for the 70% AR sample, as this was greater than those of the other samples and even the pure AR, or those of acrylic pseudo paints (∼2.5 MPa). 21 Therefore, the application of this PVNR/AR blend as paint seems possible.
The greatest tensile strength was achieved with 70% AR, for which the morphology showed the smallest sized rubber particles dispersed without touching each other. Such morphology may result in better interaction between PVNR and AR phases due to a greater possibility of the interaction between protein in the PVNR and carbonyl in AR, during emulsion blending as will be discussed later. The tensile strength of the 60% AR was greater than those of the 30%, 40% and 50% AR probably due to partial dispersion morphology.
Water contact angle
Water contact angles of the PVNR/AR blends were characterized to understand the effects of AR fraction on the blend surface polarity. An increased surface polarity would improve the wettability by water on the film surface. Generally, good wettability is achieved when the contact angle is 0 – 90° whereas poor wetting is associated with an angle larger than 90°.
22
Figure 4 shows typical photographs of water droplets on the film specimens with varied blend ratios. It is clearly seen that the shape of a water droplet on the film surface depends strongly on the blend ratio of PVNR and AR. The droplet on the blend film tends to spread more over the film surface when the amount of AR is increased. The water contact angles for the films were quantified and the results are shown in Figure 5. Considering the individual blend materials, the water contact angles of pure PVNR and AR were approximately 98.50° and 50.93°, respectively, in agreement with prior literature.23,24 Since the contact angle of PVNR was greater than 90°, it is considered a hydrophobic substance. In contrast, the AR was a hydrophilic substance with a contact angle lower than 90°.25,26 The water contact angle of PVNR seemed to decrease after blending with the AR, indicating that the wettability of rubber surface was improved. The water contact angles varied from 70.51° to 51.35°, depending on the ratio in which AR was added to PVNR. Addition of AR caused an increase of the polarity of the blend, consequently improving hydrophilicity and thus significantly enhancing wettability. The contact angles of 70% AR were close to those of pure AR, suggesting that the polarity of blend containing 70% AR was comparable to that of pure AR. Increased polarity from that of the PVNR for the PVNR/AR blends may introduce great adhesion on other substrates. This would be a benefit on applying this material as paint on metal and wood. Water droplet profiles on film specimens with different blend ratios. Variation of static contact angle (squares) and work of adhesion (circles) as functions of acrylic content.
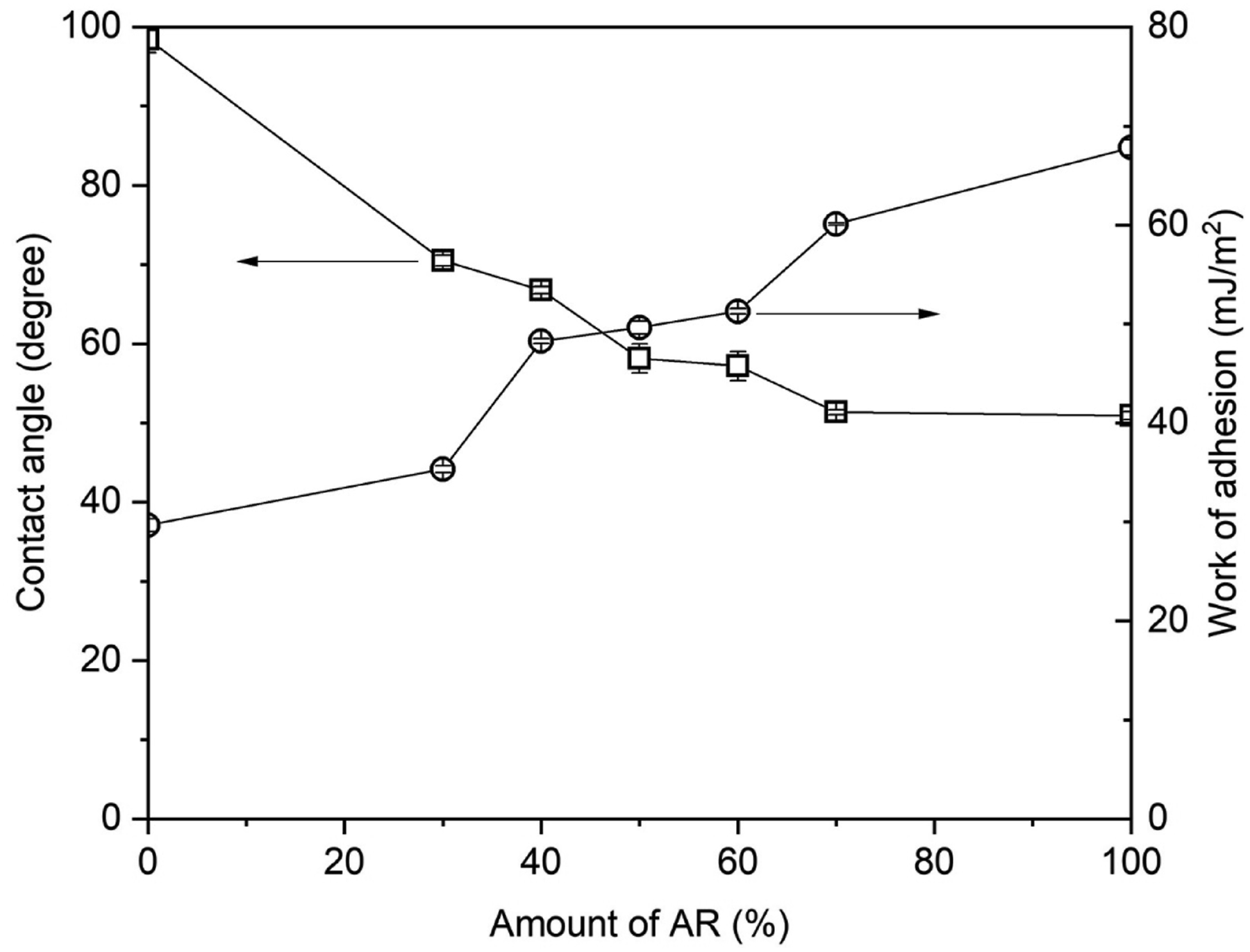
To further confirm the effect of AR content on adhesion between the blend film and a substrate, the work of adhesion (Wadh) was then calculated by applying the Young-Dupre model 15 and the results are also included in Figure 5. As can be seen in Figure 5, the Wadh of the PVNR-based blend films increased with the AR fraction, indicating that a greater adhesion between liquid and the film was achieved. The Wadh of the PVNR has improved by approximately 103% from 29.67 to 60.12 mJ/m2 at the blend ratio of 70% AR. The improved Wadh was due to an increase in the polar component (AR) of the blend, resulting in enhanced polarity of the blend. Higher Wadh suggested that the affinity of the PVNR/AR blend to adhere to a substrate was increased. Based on the contact angle measurement and Wadh, it is expected that the 70% AR case is the most likely one to create a good interfacial contact with a solid substrate, among the blend ratios tested.
Peeling properties
Adhesion of the blend films to substrate can be evaluated by different methods including peeling, pull-off, and shear tests.
27
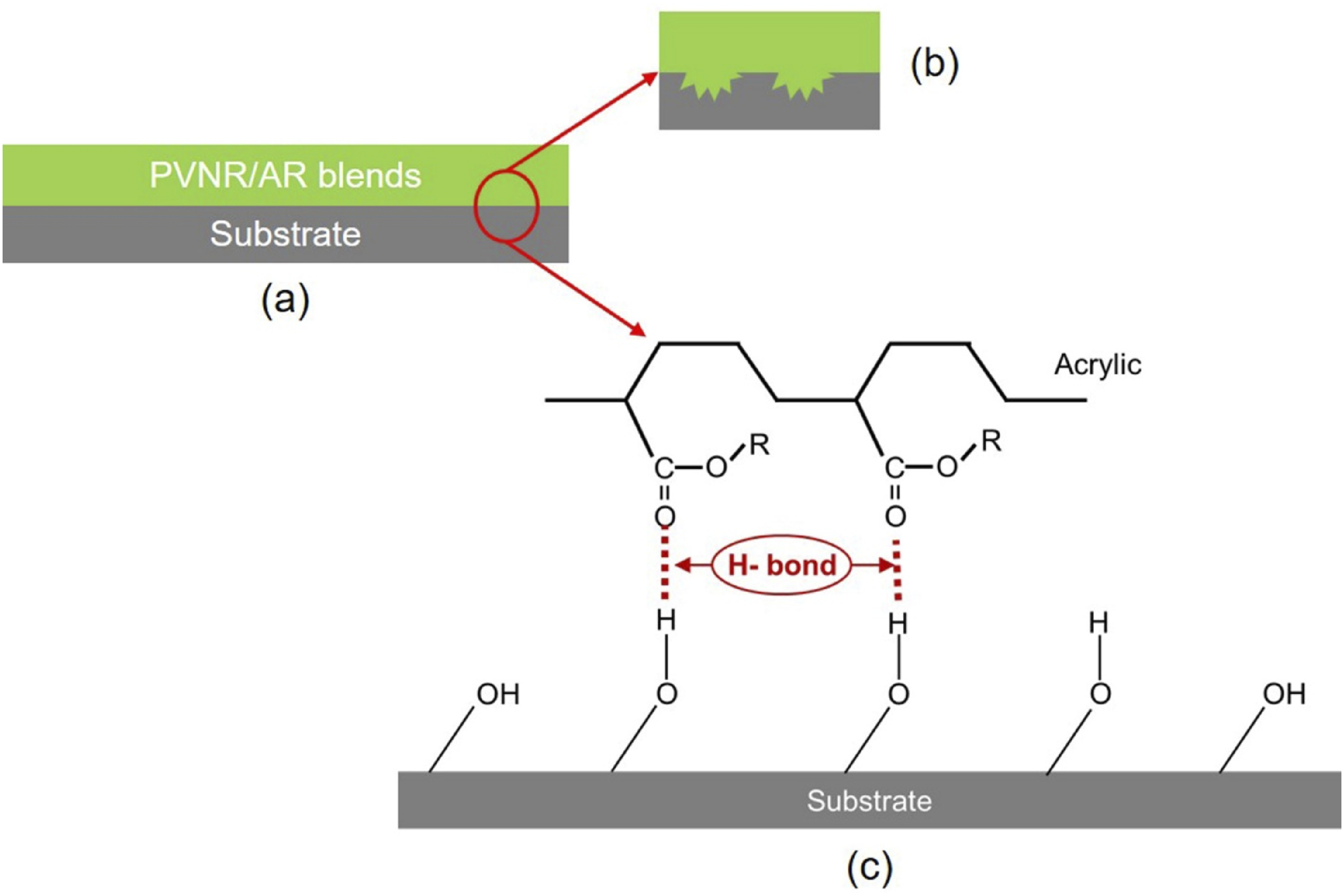
To evaluate the possibility of using the PVNR/AR blends as coating materials, peeling test was chosen. The effect of blend ratio on peel strength of PVNR/AR blends from different substrates is shown in Figure 6. Apparently, the peel strength on both wood and on metal substrates increased with AR content. The increased polarity of the blend significantly enhanced its surface wettability, thereby improving adhesion between the blend and the substrate. In addition to improved wettability, mechanical interlocking and hydrogen bonding between the blend and the substrate were also present, as shown in Figure 7. When the blend and substrate were joined together (Figure 7(a)), some molecular chains of the blend penetrated the pores and irregularities on the substrate surface, creating mechanical interlocking (Figure 7(b)). Simultaneously, the carbonyl functional groups in the acrylic structure interacted with hydroxyl groups on the substrate surface through hydrogen bonding, as both metal and wood surfaces contain hydroxyl groups,28,29 as shown in Figure 7(c). This bonding became stronger with increasing AR content due to the higher availability of carbonyl groups for interaction. Consequently, a higher AR content led to an increase in peel strength. The greatest peel strengths on both wood and metal substrates were achieved with the 70% AR sample. The improvement was by about 470% for the wood and by 84% for the metal over the PVNR. Peel strengths from wood, and metal surfaces as functions of AR content. Possible interaction between blends and substrates (a), substrate and blends mechanical interlocking (b), and hydrogen bonding between the blend and substrate (c).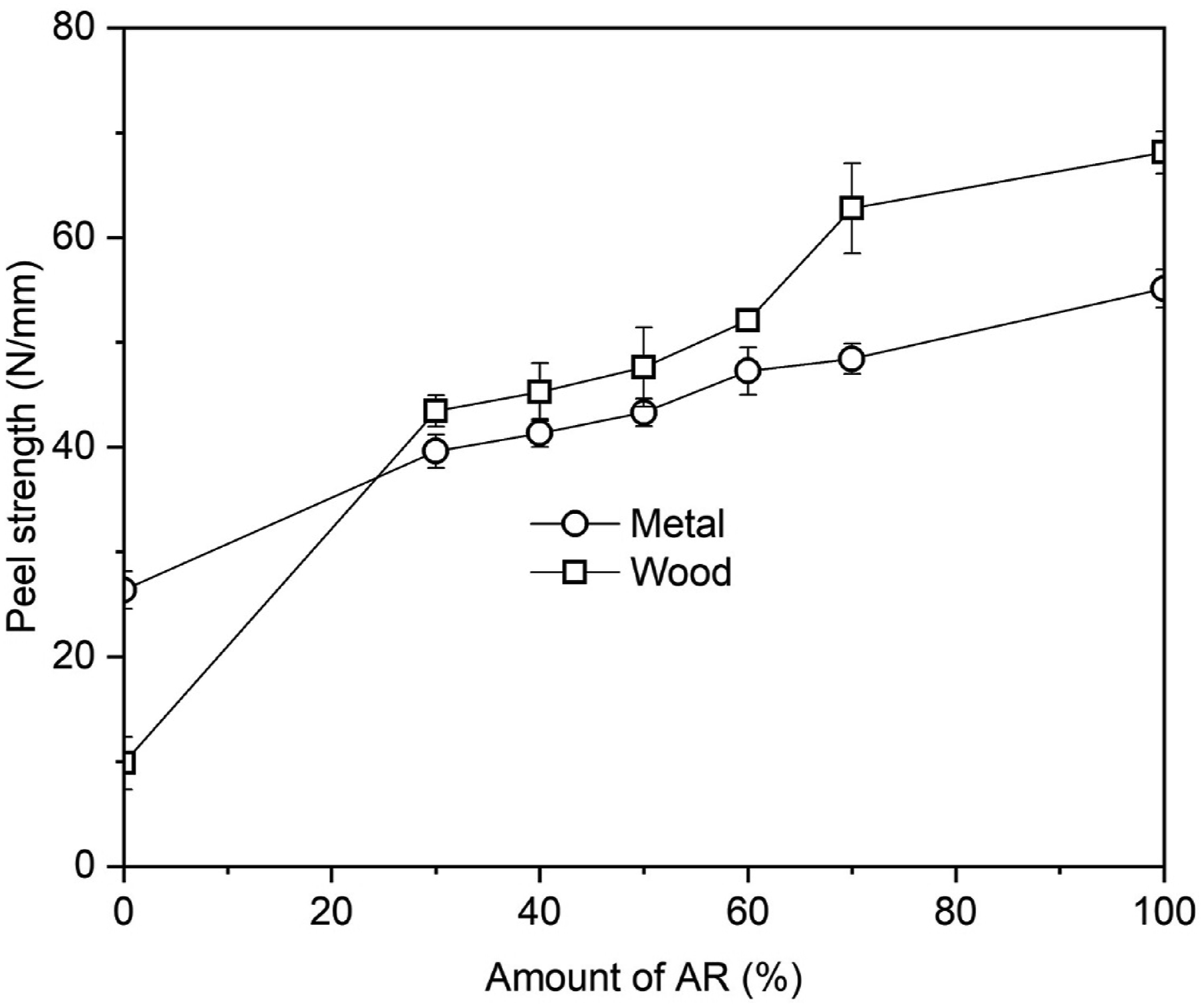
Based on the results obtained from this study, addition of AR into PVNR improved the mechanical properties and adhesion. The greatest enhancements were found for the 70% AR sample due to the well dispersed rubber particles providing the greatest interactions between phases. The wettability, adhesion strength and peel strength were also enhanced on incorporation of AR and the degree of enhancement was almost comparable to the pure AR. Thus, the obtained results suggest that the PVNR/AR blends may find applications in painting, coating and adhesive products.
Thermal properties
To assess the compatibility of the polymers blended further, TGA analysis was performed. The TGA is well recognized as a method for assessing thermal stability, thermal degradation characteristics, and compositional properties of polymers or their blends. However, some studies have shown that the TGA technique is also useful for assessing compatibility in polymer blends by determining use of the derivative thermograms.30,31 Generally, two decomposition peaks in TGA derivative thermograms are seen for incompatible blends, while a single degradation peak would be observed for a compatible blend.30–32 Figure 8 shows the derivative TGA thermograms of the blends with different PVNR/AR ratios. The raw outputs in terms of temperature at maximum rate of weight loss for PVNR and AR (TmaxPVNR and TmaxAR) are summarized in Table 4. Derivative weight loss versus temperature curves for the blends with different blend ratios, (a) 100% AR and PVNR, (b) 70% AR, (c) 60% AR, (d) 50% AR, (e) 40% AR and (f) 30% AR. Temperature at maximum rate of weight loss for pure PVNR and AR (TmaxPVNR and TmaxAR) in for the various blends.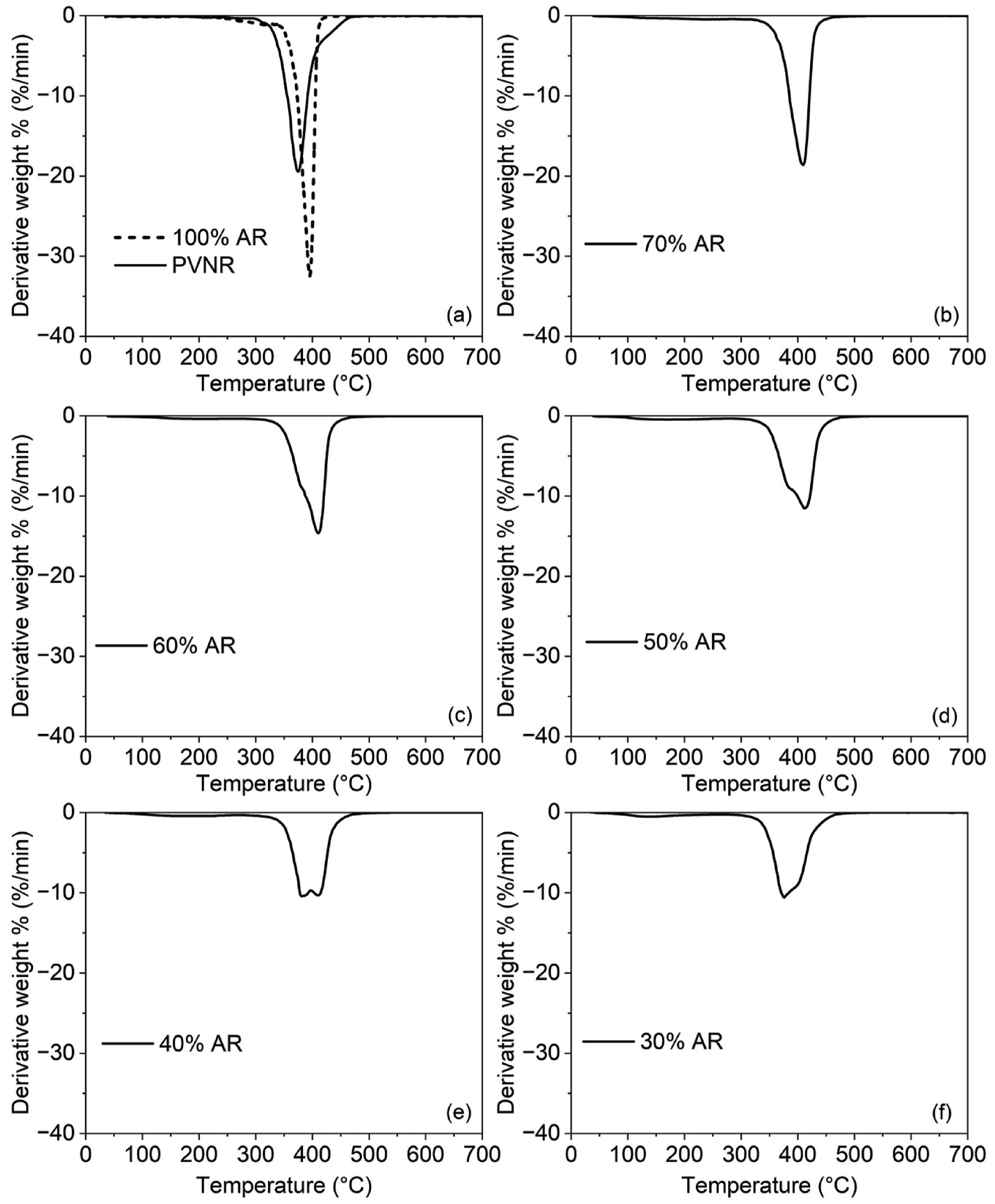
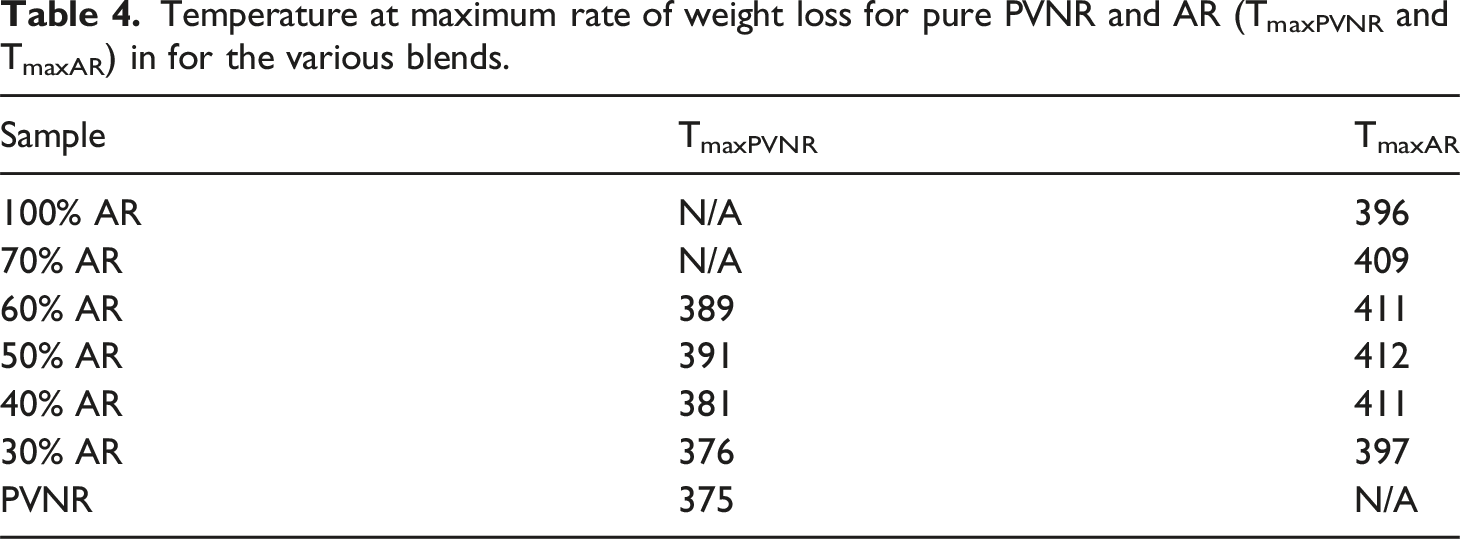
The degradation peaks of the pure PVNR and AR were found at 375°C and 396°C, respectively. From Figure 8 and Table 4, it is clear that a single derivative degradation peak was observed for the 70% AR sample whereas two degradation derivative peaks were found in the other cases. Moreover, that degradation peak was also at a higher temperature than those of the pure components. This may again be due to the partial compatibility of the component phases. Without a proper interaction between the phases, no shift of temperature would happen. The presence of single derivative degradation peak found for the 70% AR sample indicates possible compatibility between the two blend components, induced by some interfacial interactions.
Fourier transform infrared spectroscopy (FTIR)
To evaluate the hypothesis of interactions between PVNR and AR, FTIR was performed. The 70% AR sample was chosen to represent the blends. The FTIR spectrum over the range 4000 – 1000 cm−1 for the 70% AR sample is shown in Figure 9(a). The FTIR spectra of pure PVNR and AR are also shown for comparison. It has been reported that the non-isoprene compounds in the NR cause the bands at 3283 cm−1 for amine, 1748–1738 cm−1 for ester, 1711 cm−1 for carboxyl, 1630 cm−1 for amide I and 1541 cm−1 for amide II.
33
The ester and carboxyl groups are in lipid molecules whereas the others are associated with proteins in the NR.
33
This is because the proteins contain amino acids as repeating units.
34
Since no change in these related bands across the samples was clear, as seen in Figure 9(a), all the spectra were limited to the regions 3500 – 3000 cm−1, 1800 – 1600 cm−1 and 1700 – 1500 cm−1 as shown in Figure 9(b)–(d). Considering Figure 9(b), the band at 3281 cm−1 assigned to amine was found for PVNR whereas AR did not show a peak at the same location. When PVNR was blended with AR (70% AR sample), this amine peak of the PVNR shifted to a higher wavelength. This observation suggests that the chemical environment of amine groups was changed by blending the components. As shown in Figure 9(c), the infrared spectrum of the AR showed a characteristic peak at 1724 cm−1, corresponding to the carbonyl (C=O) in AR.35,36 It should be noted that the PVNR does not show a peak here, although a very small peak is found at a different wavenumber. FTIR spectra of pure PVNR and AR, and their blend, over the wavenumber ranges (a) 4000 – 1000 cm−1, (b) 3500 – 3000 cm−1, (c) 1800 – 1600 cm−1, and (d) 1700 – 1500 cm−1.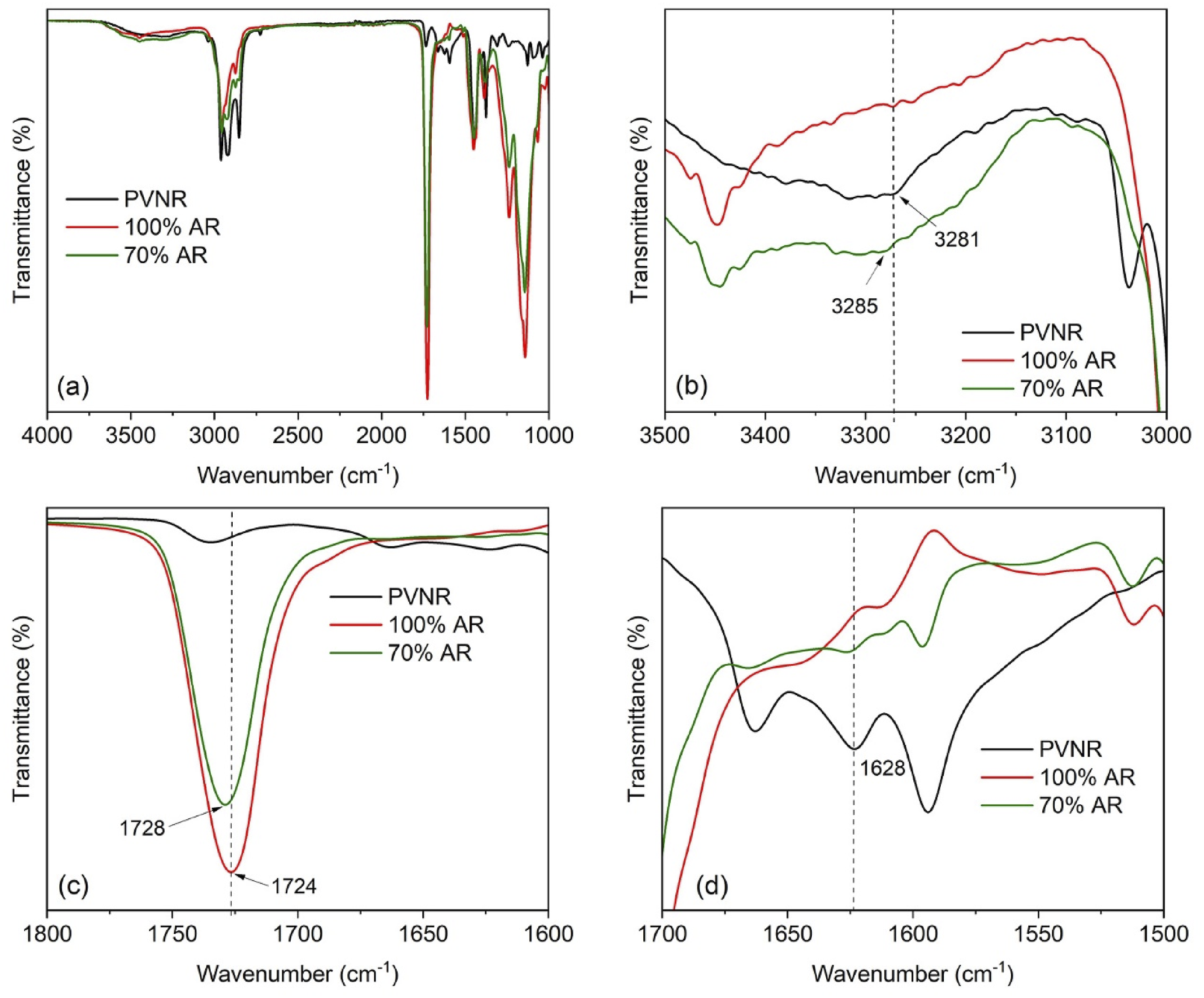
Interestingly, this peak had shifted to 1728 cm−1 after blending. This also suggests that the blending changes the local environment of C=O groups. Figure 9(d) shows FTIR spectra over the range 1700 – 1500 cm−1 indicative of amide I and II. The peak at 1541 cm−1 corresponding to amide II in the PVNR was not clear due to overlaps. However, the peak of amide I was clear at 1628 cm−1 and this peak also shifted to the left on blending PVNR and AR. Based on the FTIR observations, the shifts of peaks in various regions reveal changes to the chemical environment of specific functional groups in the blend of PVNR and AR, clearly proving that the carbonyl in AR, and the amine and amide in PVNR, had chemical interactions probably through the formation of hydrogen bonds between PVNR and AR as illustrated in Figure 10. The amine and/or amide in amino acids of proteins in the PVNR may interact with carbonyl groups in AR via H-bonding. Possible interactions between PVNR and AR.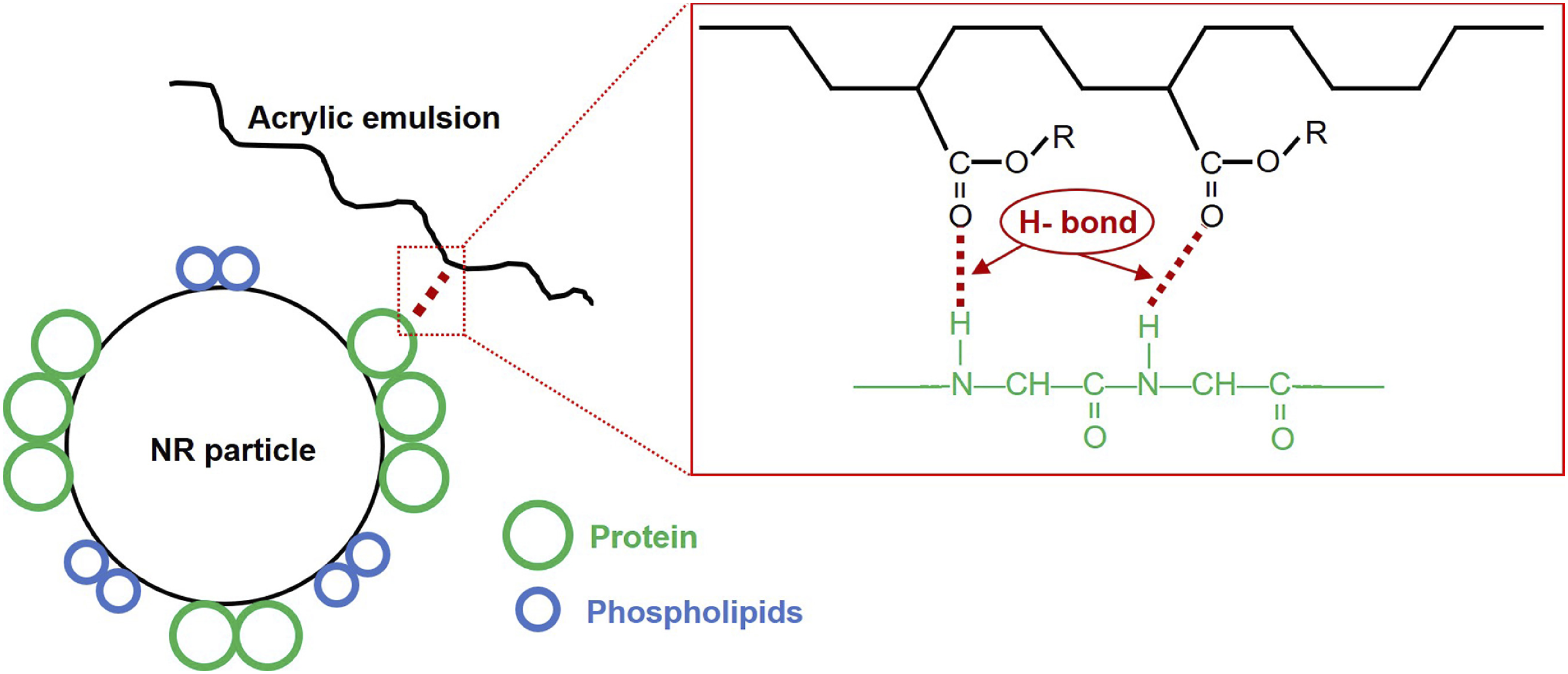
Conclusion
Emulsion blends of PVNR and AR with different blend ratios were prepared in this study. The NR latex was prevulcanized prior to mixing with the AR. The effects of blend proportions (30%, 40%, 50%, 60% or 70% AR) were studied. From a morphological analysis, the PVNR/AR blends exhibited continuous rubber matrix at a low AR fraction, while showing dispersed isolated rubber particles at high AR content. The greatest tensile strength was obtained with 70% AR. The wettability of the blend increased with AR content, resulting in also a better adhesion of the blend on a solid substrate. The greatest peel strength was achieved at 70% AR with improvements by about 470% and 84% over the PVNR on wood and metal substrates, respectively. This was tentatively attributed to a partial compatibility between the two phases. This partial compatibility was confirmed by a single derivative peak in the TGA response. FTIR spectra showed possible interaction mechanisms between the phases, by binding of the proteins in PVNR with the carbonyl groups in AR.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by National Science, Research and Innovation Fund (NSRF) and Prince of Songkla University (Ref. No. UIC6601148S).
Data Availability Statement
Data are contained within the article.
