Abstract
High-density polyethylene (HDPE) nanocomposites with Fe3O4 nanoparticles were synthesized and analyzed for structural, thermal, and magnetic properties. X-ray diffraction showed an increase in Fe3O4 crystallite size from 5.14 nm to 12.01 nm, while the HDPE crystalline size decreased from 14 nm to 4.88 nm for HDPE + 3% Fe3O4 and HDPE + 40% Fe3O4, respectively. Thermogravimetric analysis showed improved thermal stability, with the onset temperature increasing from 243.78°C (HDPE) to 395.13°C (HDPE + 40% Fe3O4). Differential scanning calorimetry indicated a crystallinity rise from 44.45% to 51.95% (HDPE + 40% Fe3O4), while melting and crystallization temperatures remained near 104°C and 94°C, respectively. Magnetic characterization revealed that the saturation magnetization increased from 49.61 × 10−3 emu for HDPE +1% Fe3O4 to 52.98 × 10−3 emu for HDPE +10% Fe3O4, while the coercivity decreased from 17.76 G for HDPE + 5% Fe3O4 to 6.30 G for HDPE + 1% Fe3O4. This reduction in coercivity suggests a transition from a single-domain to a multi-domain state, likely due to nanoparticle aggregation at higher concentrations. These results demonstrate the potential of HDPE/Fe3O4 nanocomposites for thermally stable and magnetically tunable applications.
Keywords
Introduction
Polyethylene is one of the most widely used and cost-effective polymers globally. To expand its range of applications, it is essential to modify its properties.1–3 One effective approach to modifying the properties of polymers is the incorporation of various fillers to produce composite materials. In recent years, significant attention has been devoted to the development of polymer-based nanocomposites, as nano-sized fillers exhibit significantly higher efficiency compared to conventional micron-sized dispersed fillers.4–6
For instance, Merinska et al. (2011) utilized Na+ montmorillonite as a filler in a polyethylene (PE) matrix, assessing its barrier properties with a focus on the permeability of water vapor, carbon dioxide, and oxygen. 7 Similarly, Abou-Kandil et al. (2014) synthesized ZnO nanoparticles of varying sizes and incorporated them into a high-density polyethylene (HDPE) matrix, demonstrating enhancements in both spectral and mechanical properties. Due to these improvements, ZnO-filled nanocomposites are particularly suitable for applications requiring UV absorption, such as food packaging and UV shielding. 8
Zhao (2024) explores the grafting of polyethylene (PE) chains onto α-zirconium phosphate (ZrP) nanoplatelets as a strategy to enhance exfoliation within polyolefin matrices. By carefully adjusting the graft density on the nanoplatelets, exfoliation is preserved throughout the blending process, thereby preventing agglomeration. This method facilitates “PE-confined crystallization,” leading to the formation of oriented PE crystals perpendicular to the nanoplatelets, ultimately improving the nanocomposites’ physical and mechanical properties. The study provides valuable insights into achieving high-performance polyolefin nanocomposites through controlled filler surface modification and crystallization behavior. 9
Dabees (2021) successfully synthesized high-density polyethylene (HDPE)/multi-wall carbon nanotube (MWCNT) nanocomposites with varying weight fractions and examined their tribological and electrical properties. The results indicated that the 3 wt.% MWCNTs/HDPE nanocomposite exhibited the most favorable tribological performance. Further enhancement through the incorporation of 1.2 wt.% nano-alumina resulted in the lowest friction coefficient, attributed to the formation of a stable transfer film. Moreover, the addition of MWCNTs and Al2O3 improved hydrophobicity by increasing the contact angle and reducing surface energy relative to pure HDPE. Electrical analysis identified a percolation threshold between 2 and 3 wt.% MWCNTs, with a sharp increase in conductivity observed at 3 wt.%, marking a transition in electrical behavior. 10
Khan (2023) underscores the versatility of polyolefins, particularly polyethylene, which exhibits desirable characteristics such as reshapability, surface smoothness, chemical resistance, and a lower density compared to metal and glass-based materials. The study emphasizes polyethylene’s significance as a major product within the global plastics industry and its potential for property enhancement through composite formation with various fillers. Among these, nitride-based fillers—specifically group 13 and group 14 nanomaterials—have shown considerable promise in improving polyethylene’s intrinsic properties. The review further examines the effects of filler type, concentration, and interactions with the polyolefin matrix, providing a comprehensive understanding of their influence on nanocomposite performance. 11
Lebedev (2024) systematically investigates the solid-state processing of nanocomposites based on nascent disentangled ultra-high-molecular-weight polyethylene (d-UHMWPE) reactor powders and carbon nanoparticles. The study examines filler distribution optimization, the impact of d-UHMWPE and nanofiller type on electrical conductivity, and the dependence of tensile strength and conductivity on the deformation ratio under homogeneous shear. Findings confirm that high-temperature uniaxial and homogeneous deformation can interchangeably enhance conductivity and mechanical properties. The results highlight the feasibility of directly processing d-UHMWPE powders and nanoparticles into high-strength, conductive composite tapes, providing a model for studying conductive network deformation. 12
Salehi et al. (2019) investigated the effects of surface-modified carbon nanotubes (CNTs) on the morphological, elastic, and thermal properties of HDPE-based nanocomposites. Stearic acid (SA) and nitric acid (HNO3) were used to functionalize the CNT surfaces, improving the dispersion of multi-walled carbon nanotubes (MWCNTs) in HDPE. Tensile test results revealed a 4% increase in yield strength and a 6% enhancement in Young’s modulus following the addition of 5% acid-treated CNTs to HDPE. However, the presence of SA on CNTs slightly reduced the mechanical strength of the nanocomposite. 13
Furthermore, Heidarbeigi et al. (2019) investigated the incorporation of cellulose nanocrystals (CNC) to improve the applicability of HDPE in the food packaging industry. While the addition of CNC did not alter the melting or crystallization temperatures of HDPE, it had a significant impact on its degree of crystallinity. 14 Various other fillers have also been employed to enhance the targeted properties of polyethylene-based nanocomposites.15,16
Nanoparticles with magnetic properties also serve as highly effective fillers for thermoplastic polymers. In particular, iron oxide nanoparticles have garnered significant attention due to their distinctive properties and broad application potential.17–22 Kirchberg et al. (2012) investigated composites based on FeSi, magnetite particles, and a polypropylene (PP) matrix. At filler fractions of x ≥ 0.3, the composites exhibited a substantial enhancement in both magnetic and thermal properties. At x = 0.7, the thermal conductivity of PP increased by a factor of seven. Additionally, FeSi-filled composites demonstrated slightly higher thermal conductivity values compared to those filled with Fe3O4. The magnetic permeability of the composites also increased with filler content. 23
Moghaddam et al. (2018) synthesized magnetic polyurethane nanocomposites and examined the impact of Fe3O4-SiO2-urea nanoparticles on the sound absorption characteristics of polyurethane. The results indicated an improvement in the composite’s sound absorption coefficient across the entire frequency range. Furthermore, the study evaluated the material’s thermal stability and mechanical properties, revealing notable enhancements. 24
Graphene oxide (GO), Fe3O4, and polyamide-imide (PAI)-based nanocomposites were fabricated by Khalili et al. (2019). Thermogravimetric analysis revealed that PAI/Fe3O4@GO exhibits high thermal resistivity. Furthermore, the incorporation of Fe3O4@GO into the polymer matrix resulted in a substantial enhancement of its mechanical properties. 25
A new class of electromagnetic interference (EMI) shielding materials based on polyaniline was introduced by Zahid et al. (2021). Their study extensively explored polyaniline nanocomposites incorporating dielectric nanomaterials (graphene) and magnetic nanomaterials (γ-Fe2O3). 26 Similarly, Jayakrishnan et al. (2022) investigated nanocomposites based on poly-anthranilic acid-co-indole polymer and Fe3O4 particles. Their findings indicated that nanoparticle incorporation led to an increase in the saturation magnetization of the nanocomposites, which exhibited superparamagnetic behavior. Additionally, the inclusion of Fe3O4 nanoparticles significantly improved the thermal stability and glass transition temperature of the nanocomposites, as confirmed by differential scanning calorimetry (DSC) and thermogravimetric analysis (TGA). 27
Surface-modified Fe3O4 + PE systems were synthesized by Gao et al. (2024) using the metathesis polymerization technique. The resulting nanocomposites demonstrated photothermal properties, thermally induced shape memory behavior, and superparamagnetic characteristics. 28
Despite extensive research in the field, there remains a limited body of work focused on developing a simple and efficient preparation method for HDPE matrix and Fe3O4 nanoparticle-based structures. Additionally, studies investigating the magnetic properties of polymer nanocomposites in relation to the domain structure of magnetite, as well as the thermal behavior of these nanocomposites at higher filler concentrations, are relatively scarce.
Furthermore, discrepancies exist among the available literature. For instance, Ginting et al. (2016) examined the morphological, mechanical, and thermal properties of HDPE/Fe3O4 nanocomposites, reporting an increase in thermal stability and crystallinity with higher filler content. 29 In contrast, Ahangaran et al. (2017) found that the incorporation of Fe3O4 nanoparticles into a high-density polyethylene matrix resulted in an enhancement of the dielectric constant but a reduction in the degree of crystallinity. 30
In contemporary literature, despite the abundance of research on materials incorporating magnetite nanoparticles, the magnetic properties of PE + Fe3O4 nanocomposites remain largely unexplored. This study addresses that gap by providing the first detailed investigation of both the thermal and magnetic properties of PE + Fe3O4 nanocomposites, while also elucidating how changes in their structure influence these properties. Specifically, HDPE-based nanocomposites were synthesized via an ex-situ approach that combined hot pressing and solution mixing. Their structural features were evaluated through X-ray diffraction (XRD), scanning electron microscopy (SEM), and atomic force microscopy (AFM), and their thermal and magnetic behaviors were systematically characterized. The experimental findings indicate that Fe3O4-based nanocomposites exhibit remarkable properties, underscoring their potential for the development of advanced materials across various applications.
Experimental part
Materials
The materials used in this study include iron (II) sulfate (FeSO4·7H2O, PLC 141362, 98% chemically pure), iron (III) chloride (FeCl3·6H2O, PLC 141358, 98% chemically pure), and ammonia solution (NH4OH, PLC 141129). Additionally, polyethylene glycol powder 6000 (PLC 163325), high-density polyethylene (HDPE) granules (MW = 200,000, SOCAR, ‘Azerikimya’ Production Union, ‘Etilen-Polyethylene’ plant, 15803-020), and carbon tetrachloride (PLC 141245, 99.5%, CAS No. 56-23-5) were utilized. Deionized water was used as the solvent in all synthesis processes.
Synthesis of Fe3O4 nanoparticles
Magnetite (Fe3O4) nanoparticles were synthesized via chemical co-precipitation using ferric chloride (FeCl3·6H2O) and ferrous sulfate (FeSO4·7H2O) salts, with an ammonium solution serving as the precipitant. Ferric and ferrous salts, in a stoichiometric ratio of 3:2, were dissolved in 100 mL of distilled water. A total of 100 mL of iron ion solutions (50 mL Fe3+ and 50 mL Fe2+) along with 0.5 g of polyethylene glycol (PEG) powder were introduced into the flask and stirred for 10 minutes. PEG acted as a surfactant, aiding in the stabilization of the magnetite nanoparticles.
Subsequently, 100 mL of a 25% ammonium solution was added to the reaction mixture and stirred for 1 hour at 80°C. The appearance of a black precipitate upon ammonium solution addition indicated the formation of Fe3O4 nanoparticles. The synthesis was considered complete once the pH of the solution reached the desired level.
After the reaction, the Fe3O4 nanoparticles were thoroughly washed with distilled water using ultracentrifugation to eliminate residual non-reacting ions. The purified nanoparticles were then subjected to ultrasonic treatment using a VSX 500 ultrasonicator to disrupt potential aggregates and agglomerates. Finally, the nanoparticles were transferred to Petri dishes for air drying over several days, and the resulting dried powder was finely ground using a mortar and pestle.
Preparation of magnetic polymer nanocomposites
HDPE + Fe3O4 nanocomposites were synthesized using a combination of ex-situ and solution-casting methods. First, 0.5 g of polyethylene granules were dissolved in 100 mL of carbon tetrachloride (CCl4) at its boiling point (70°C). Subsequently, pre-synthesized Fe3O4 nanopowders in varying concentrations (3%, 5%, 10%, and 40%) were gradually introduced into the dissolved polymer and vigorously stirred using a magnetic stirrer for 1 hour to ensure uniform dispersion.
The resulting polymer-nanoparticle mixture was then transferred to Petri dishes and allowed to dry under ambient conditions for several days. To eliminate residual carbon tetrachloride, the nanocomposite films were placed in a vacuum oven at 100°C for 1 hour. Finally, thin films of HDPE + Fe3O4 nanocomposites, with a thickness of 100 μm, were fabricated using the hot-pressing technique on a laboratory pressing machine at a pressure of 10 MPa and a temperature of 160°C.
Characterization
Rigaku Mini Flex 600 XRD diffractometer was used for X-ray diffraction analysis at ambient temperature. SEM analysis of HDPE + Fe3O4 nanocomposites was performed on a JEOL JSM-7600F SEM (Japan) in SEI mode at a working distance of 4.5 mm. AFM analysis of samples was analyzed on an NT-MDT Integra Prima atomic force microscope (Russia, Zelenograd) in the tapping mode. TGA analysis of HDPE + Fe3O4 nanocomposites was accomplished on an SDT Q600 TA Instruments in a nitrogen atmosphere with heating in the temperature range from −40 to 200°C. VSM analysis of HDPE + Fe3O4 nanocomposites was executed using a Lake Shore 7400 VSM magnetometer.
Results and discussion
Figure 1(a) displays the XRD pattern of the pure HDPE polymer. The HDPE diffractogram shows diffraction peaks at 2θ = 21.7° and 24.1°.
29
The nanocomposite diffractogram reflects both the HDPE and Fe3O4 diffraction lines. The characteristic lines at 30.36°, 35.68°, 43.3°, 57.36°, and 62.95° values of 2θ angle are observed for iron oxide nanoparticles and correspond to Miller indices (440), (220), (311), (400), (511) (card number DB 01-073-9877), respectively.
30
XRD diffractograms of HDPE (a) and HDPE + Fe3O4 based nanocomposites (b): HDPE + 3% Fe3O4 (1); HDPE + 5% Fe3O4 (2); HDPE + 10% Fe3O4 (3) and HDPE + 40% Fe3O4 (4).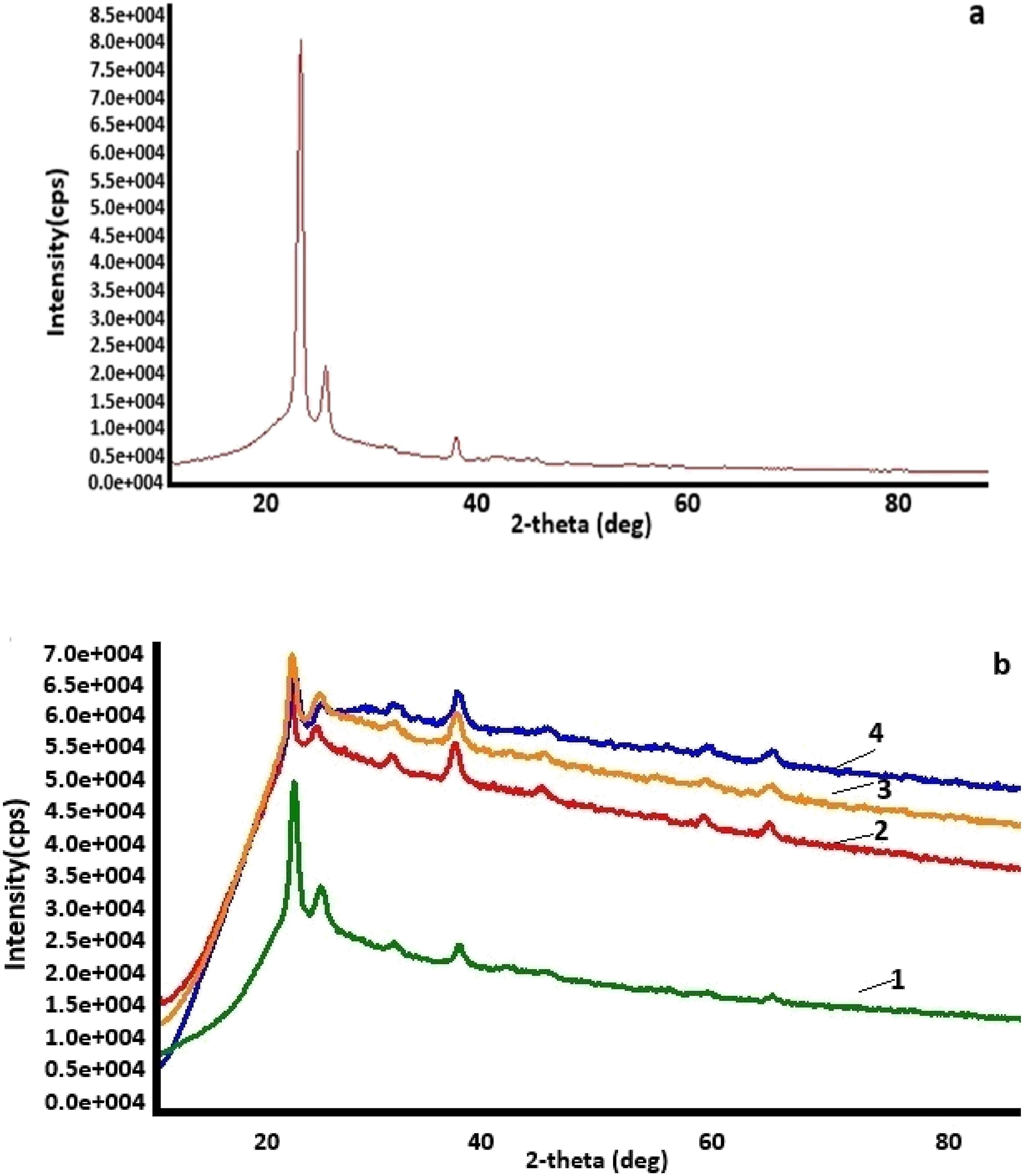
The average crystallite size was calculated from XRD spectra using the Debye-Scherer formula
It was observed that as the concentration of Fe3O4 nanoparticles in the polymer matrix increased, the average size of the dispersed nanoparticles exhibited a slight increase. This phenomenon can be attributed to the nanoparticles’ inherent tendency to agglomerate at higher concentrations due to their high surface energy. Simultaneously, the crystalline size of HDPE in the nanocomposite decreased upon the incorporation of nanoparticles.
Data of HDPE + Fe3O4 based nanocomposites calculated from XRD diffractograms.
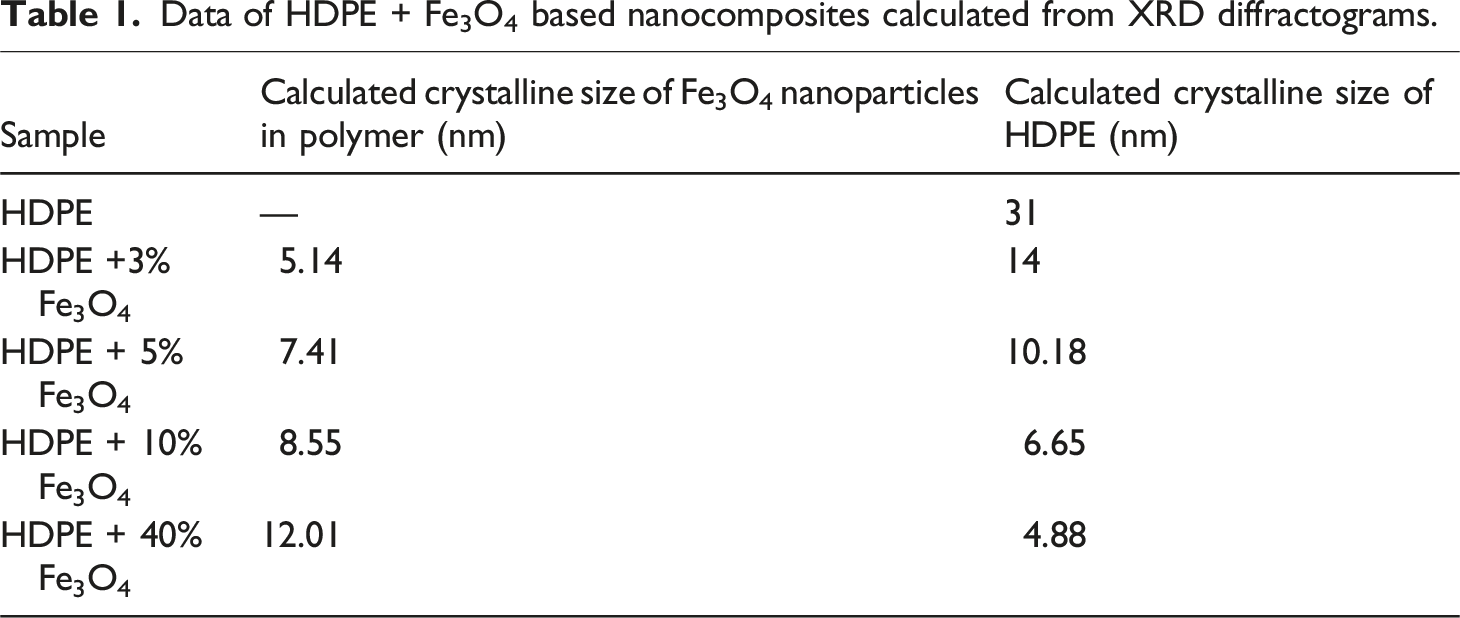
The SEM images of pure Fe3O4 nanoparticles and HDPE + Fe3O4 nanocomposites are presented in Figure 2. The size of pure nanoparticles is about 5-10 nm (Figure 2(b)). The size of the original nanoparticles remains largely unchanged after their incorporation into the polymer matrix. Figure 3 provides a histogram depicting the size distribution of nanoparticles within the polyethylene matrix. The average size of magnetite nanoparticles in the polyethylene matrix is 13 nm, while the majority of particles fall within the 8–10 nm range. SEM images of Fe3O4 nanoparticles (a, b), HDPE + Fe3O4 based polymer nanocomposites: HDPE + 3% Fe3O4 (c); (b) HDPE + 5% Fe3O4 (d); (c) HDPE + 10% Fe3O4 (e); (d) HDPE + 40% Fe3O4 (f). Size distribution of Fe3O4 nanoparticles in HDPE + 5% Fe3O4 nanocomposite.
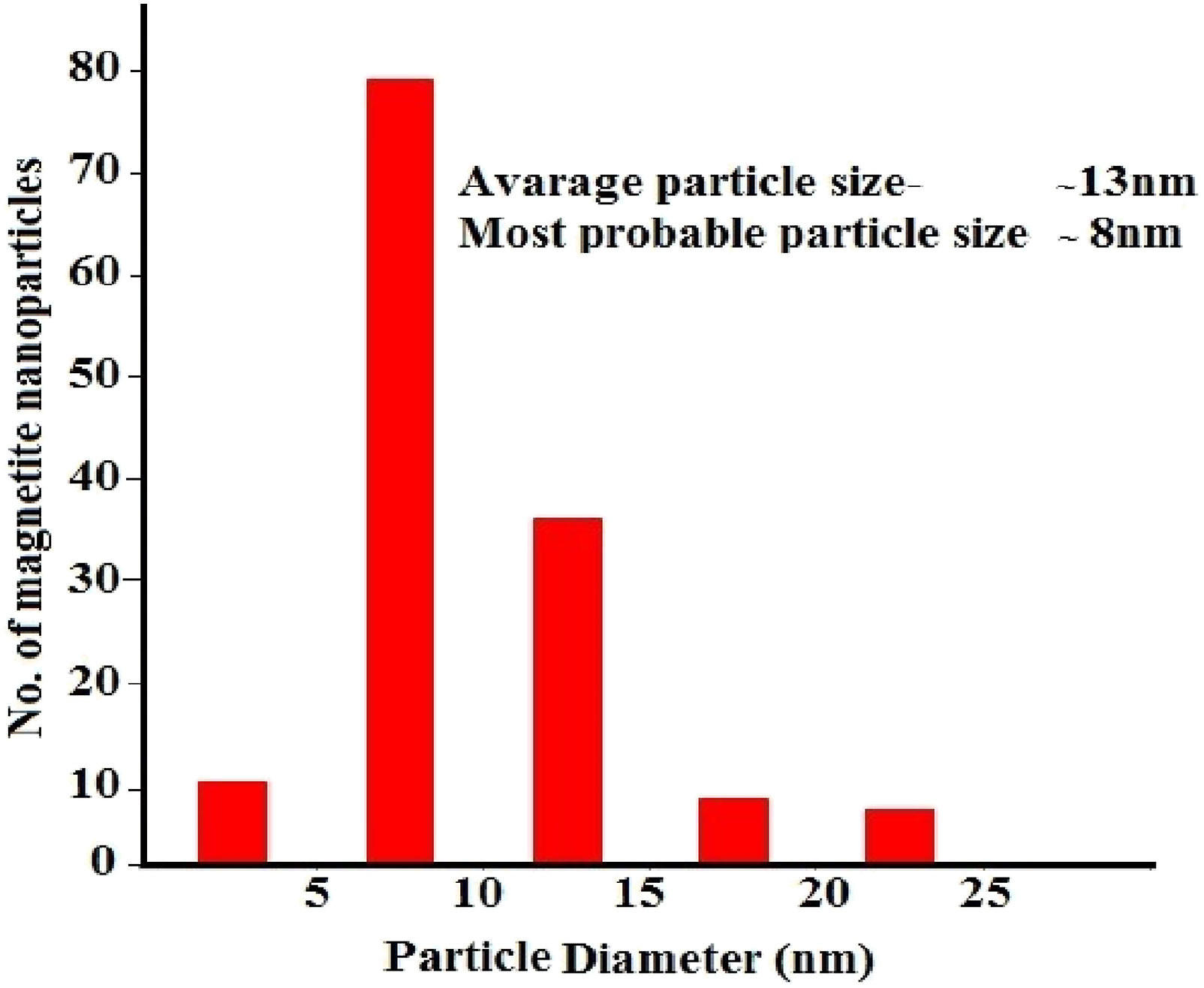
Ideally, the average particle size should closely correspond to the size of the majority of particles. However, the arithmetic mean is inherently sensitive to the presence of a small fraction of larger particles within the sample. If the majority of nanoparticles are clustered around 8–10 nm, but a minor proportion exhibit sizes of 20 nm or more, these larger particles can disproportionately increase the overall average, shifting it to approximately 13 nm. In statistical terms, the mode (the most frequently occurring size) represents the dominant particle population, whereas the mean can be influenced by the presence of outliers or skewed distribution, leading to a higher calculated average size.
Figure 4 presents AFM 3D images and surface roughness histograms for pure HDPE, HDPE + 5% Fe3O4, and HDPE + 10% Fe3O4 nanocomposites, illustrating significant structural modifications induced by Fe3O4 incorporation. AFM analysis reveals that the surface roughness values are approximately 100–300 nm for pure HDPE, 80–100 nm for HDPE + 5% Fe3O4, and 40–60 nm for HDPE + 10% Fe3O4 nanocomposites. These findings are consistent with XRD data, which indicate a reduction in HDPE crystallite size. Histogram of surface roughness and AFM 3D images of HDPE (a) and HDPE + 5% Fe3O4 (b) and HDPE + 10% Fe3O4 (c) based nanocomposites.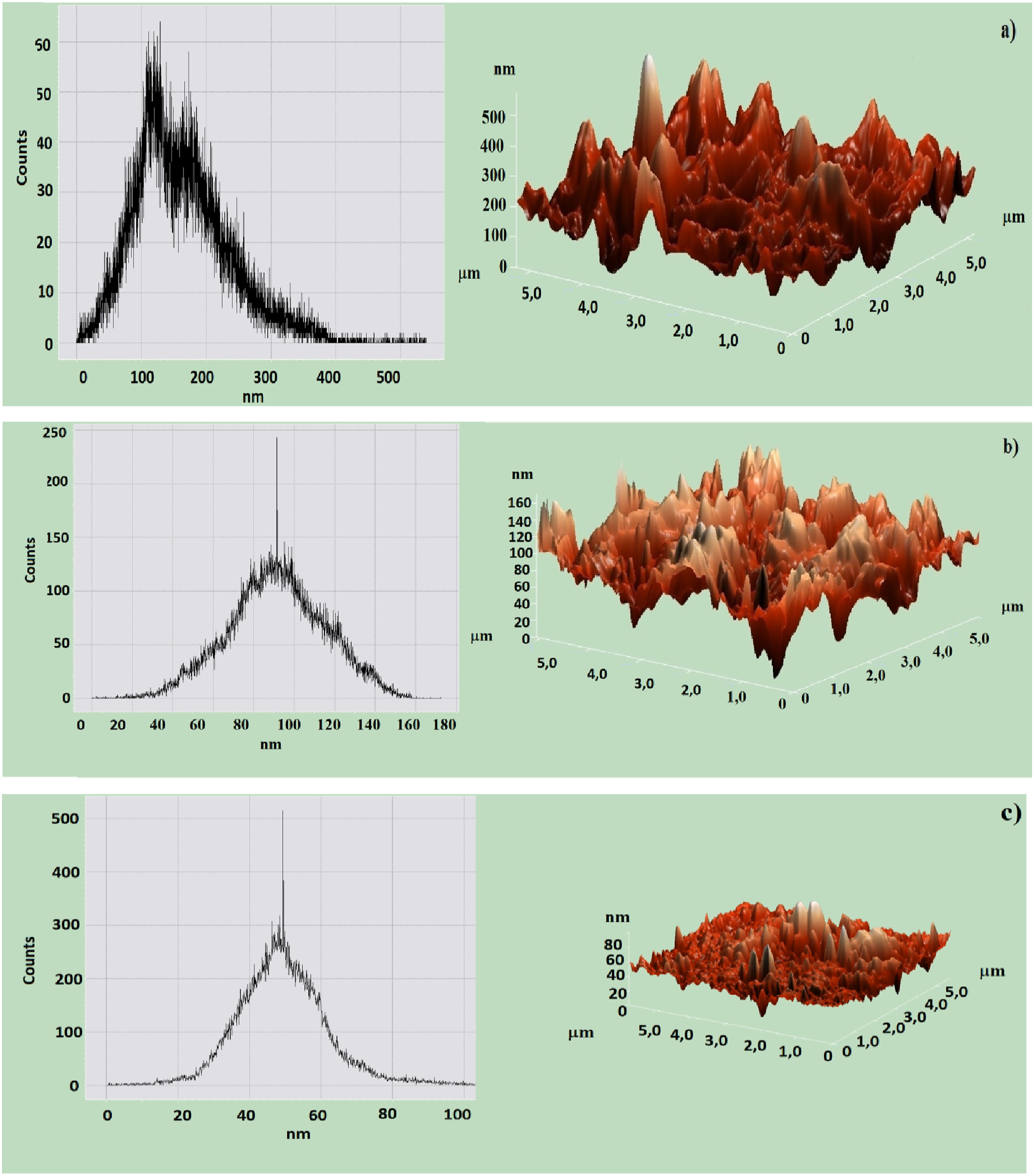
The observed decrease in surface roughness can be attributed to several factors. Primarily, Fe3O4 nanoparticles act as nucleation sites, facilitating the formation of smaller, more uniformly distributed crystalline regions, thereby resulting in a smoother surface. Furthermore, XRD analysis confirms a decrease in HDPE crystallite size, suggesting that the presence of nanoparticles disrupts the growth of large crystalline domains, leading to a finer structural organization. 35
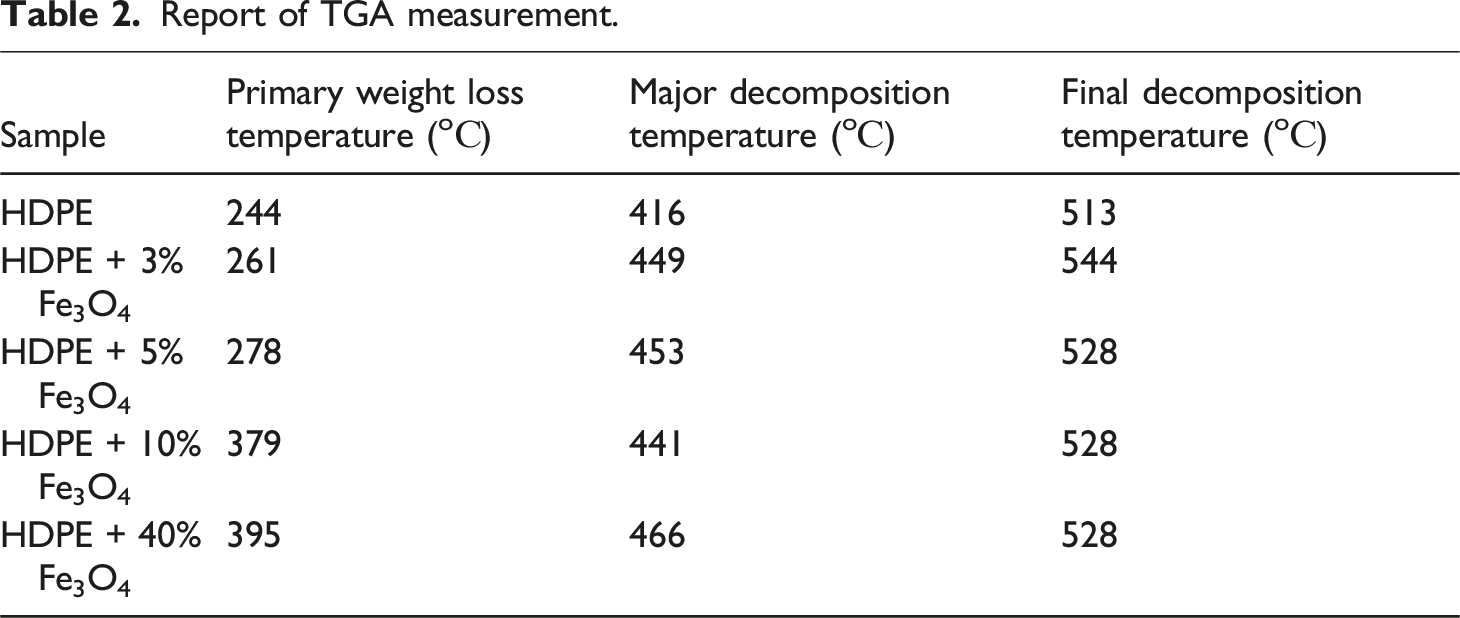
The thermal stability of HDPE + Fe3O4 nanocomposites was investigated as a function of nanoparticle concentration. Thermogravimetric (TGA) and calorimetric analyses were conducted to evaluate the thermal degradation behavior and possible structural modifications induced by the filler. Figure 5 presents the TGA curves of both the host polymer and the Fe3O4-based nanocomposites, while Table 2 summarizes the TGA data. TGA patterns of HDPE (1), HDPE + 3% Fe3O4 (2), HDPE + 5% Fe3O4 (3), HDPE + 10% Fe3O4 (4) and HDPE + 40% Fe3O4 (5) nanocomposites. Report of TGA measurement.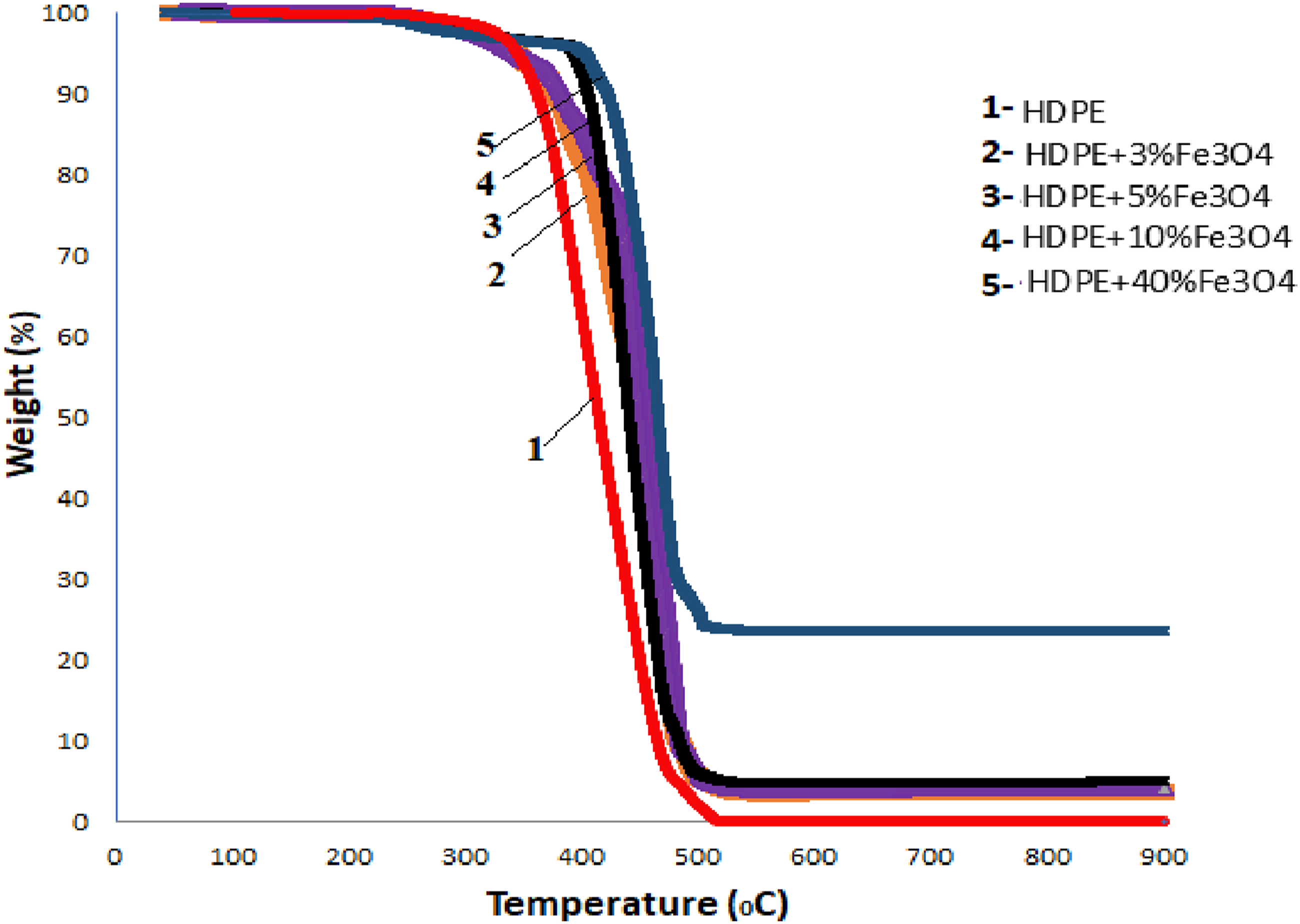
For all samples, weight loss initially proceeded at a slow rate before accelerating steadily as the degradation process progressed, ultimately reaching the primary decomposition stage. In HDPE, mass loss occurs after the initial breakdown of polymer bonds and the vaporization of light gas products. 36 Kaleel et al. (2011) demonstrated that the incorporation of TiO2 nanoparticles enhances the thermal stability of polyethylene, 37 while Abou-Kandil et al. (2014) reported that the addition of ZnO nanoparticles to HDPE results in only a marginal improvement in thermal stability. 8
The thermo-oxidative degradation of polyethylene occurs between 244°C and 416°C (final decomposition zone), with 99% mass loss observed at 448°C, beyond which the remaining mass stabilizes. As evident from Figure 5, the incorporation of Fe3O4 nanoparticles enhances the thermostability of the nanocomposites, leading to an increase in the onset temperature of thermal degradation. The primary decomposition temperature ( Tonset) values for HDPE + 5% Fe3O4, HDPE + 10% Fe3O4, and HDPE + 40% Fe3O4 nanocomposites were determined to be 261°C, 278°C, 379°C, and 395°C, respectively. Notably, the thermostability of the nanocomposites improved by approximately 150°C upon the introduction of Fe3O4 nanoparticles into the HDPE matrix (Table 2).
The enhancement in the thermal stability of the material upon the incorporation of magnetite into the HDPE matrix can be attributed to the role of iron oxide nanoparticles as heat transfer centers within the nanocomposite. These nanoparticles facilitate the dissipation of thermal energy, thereby delaying the onset of thermal degradation. Additionally, the formation of a supramolecular structure around the iron oxide nanoparticles contributes to the stabilization of the polymer matrix, further improving its thermal resistance. This structural modification also promotes an increase in the degree of polymer crystallization by acting as nucleation sites for crystalline growth. 38
The degree of crystallinity, as well as the melting and crystallization temperatures of the material, can be evaluated using Differential Scanning Calorimetry (DSC). Figure 6(a) and (b) present the melting and crystallization curves of the HDPE + Fe3O4 nanocomposite, respectively. DSC patterns of HDPE and HDPE + Fe3O4 nanocomposites (a) exothermic phase (b) endothermic phase.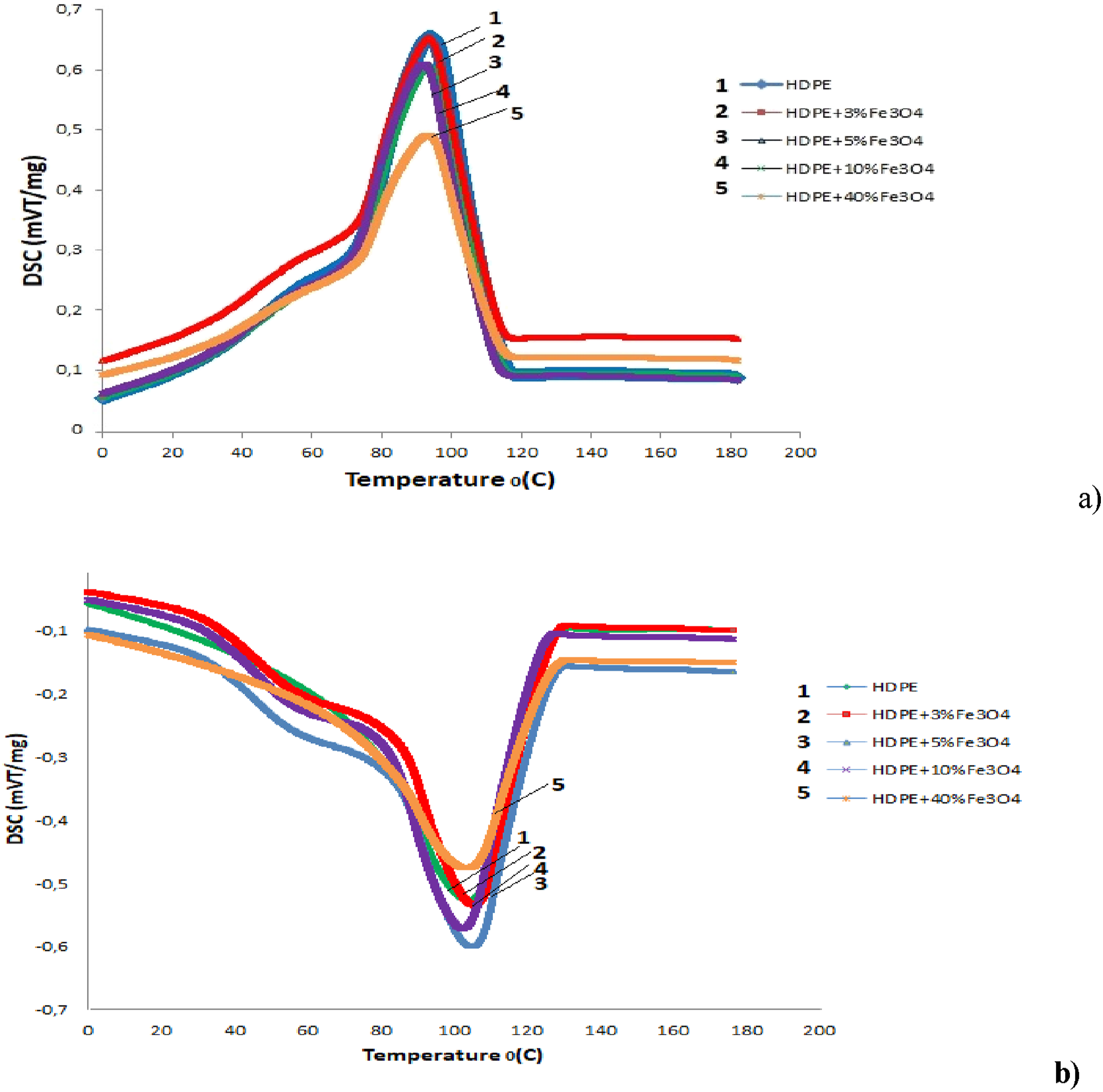

Report of DSC measurement.
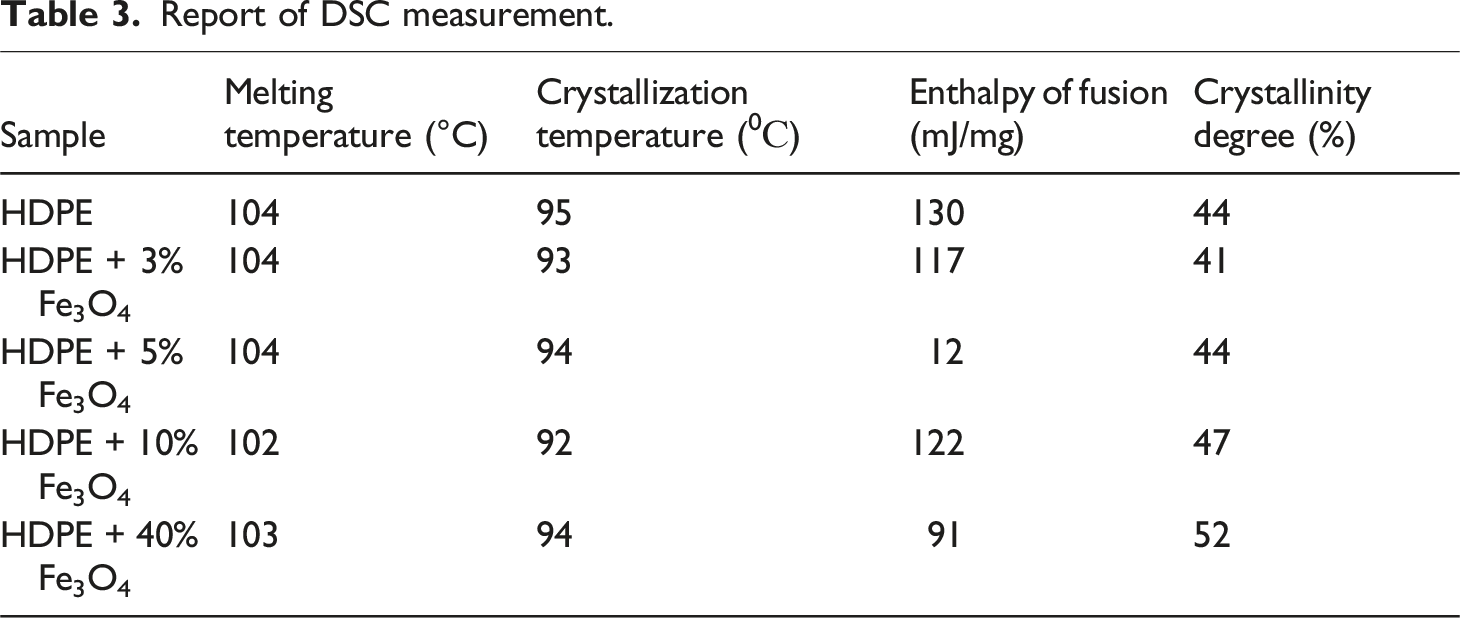
HDPE is characterized by a low degree of branching, meaning that its linear polymer chains are closely packed together. This structural arrangement results in a highly dense and crystalline material due to the strong intermolecular forces between polymer chains. The fact that Tmelting and Tcrystallization remain nearly unchanged at low Fe3O4 concentrations suggests that ultrafine, well-dispersed nanoparticles do not significantly alter the intermolecular interactions within the tightly packed polymer matrix. However, at higher Fe3O4 concentrations (40 wt.%), nanoparticle aggregates act as nucleation centers, promoting polymer crystallization and leading to a measurable increase in the crystallinity degree.
The dependence of magnetization on magnetic field intensity for HDPE + Fe3O4 nanocomposites is illustrated in Figure 7. The magnetic properties of the HDPE + 1% Fe3O4, HDPE + 5% Fe3O4, and HDPE + 10% Fe3O4 samples are summarized in Table 4. The dependence of the magnetization of HDPE + Fe3O4 nanocomposites on the magnetic field intensity. Report of VSM measurement.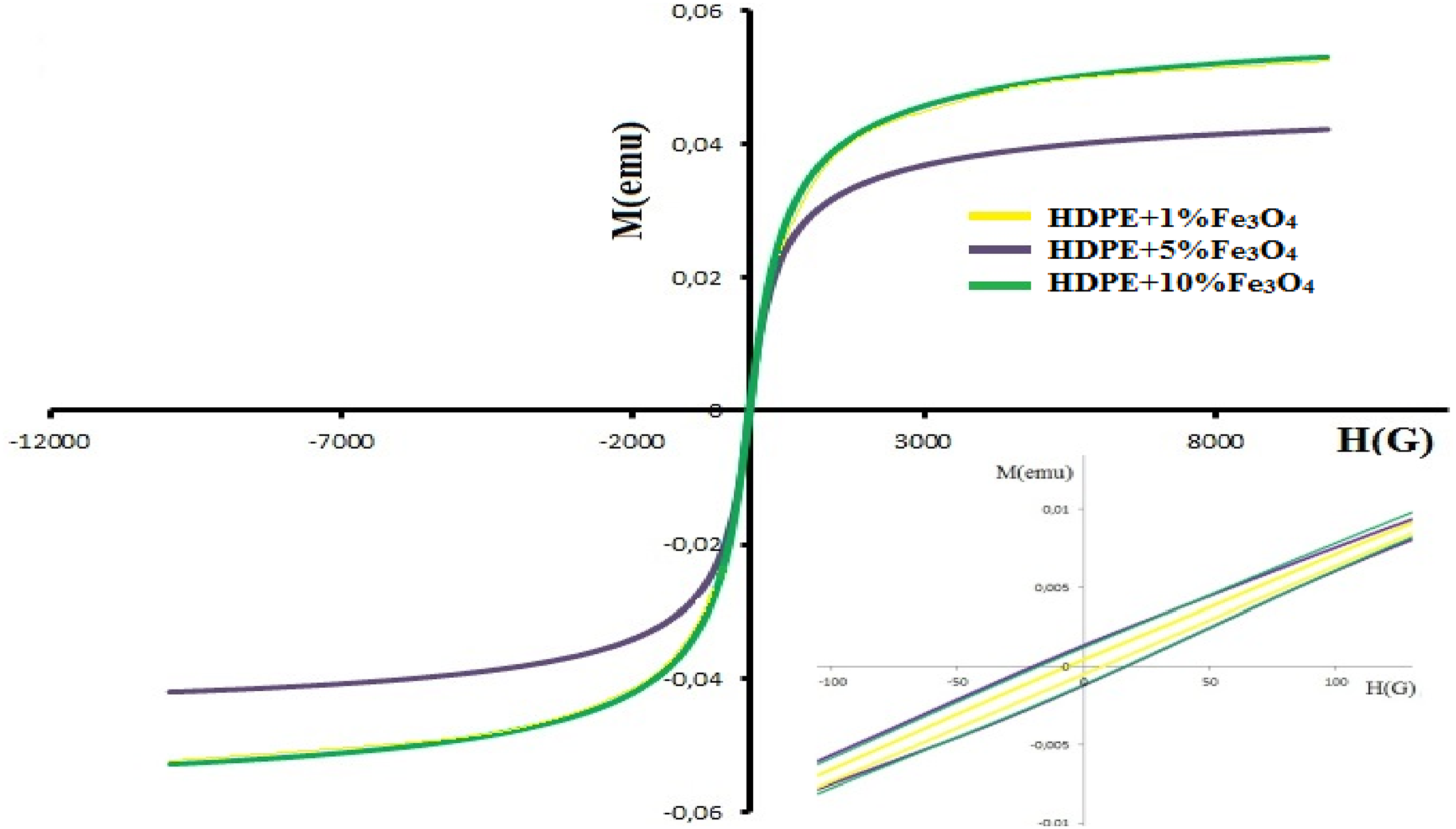
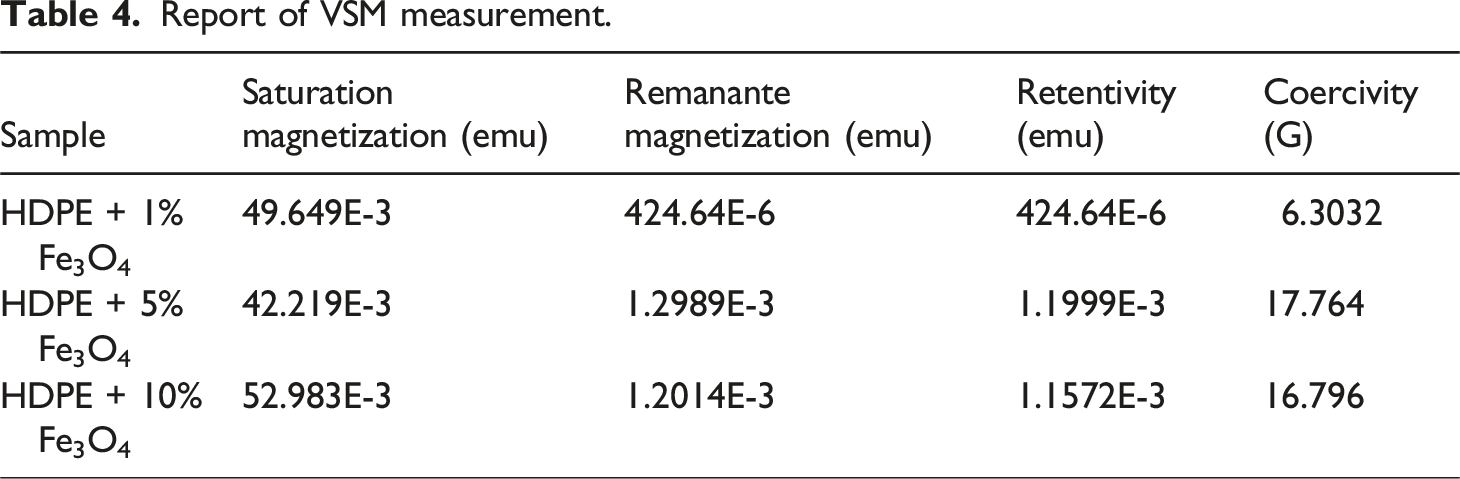
As presented in Table 4, the saturation magnetization values for HDPE + 1% Fe3O4, HDPE + 5% Fe3O4, and HDPE + 10% Fe3O4 nanocomposites are 49.6 × 10−3 emu, 42.2 × 10−3 emu, and 52.9 × 10−3 emu, respectively. While the saturation magnetization remains relatively consistent across different Fe3O4 concentrations, the coercivity (Hc) exhibits a significant variation. Specifically, the coercivity values for HDPE + 1% Fe3O4 and HDPE + 5% Fe3O4 nanocomposites are Hc = 6.3 G and Hc = 17.7 G, respectively. However, with a further increase in Fe3O4 content, the coercivity value slightly decreases.
The observed reduction in coercivity at higher Fe3O4 concentrations can be attributed to the transition of magnetite nanoparticles from a single-domain to a multi-domain state, which occurs due to nanoparticle coagulation at elevated concentrations.22,40,41 This phenomenon can be theoretically analyzed using models such as the Néel and Brownian relaxation theories, which allow for the prediction of the critical particle size at which Fe3O4 transitions from a single-domain to a multi-domain state. The critical size for Fe3O4 nanoparticles typically falls within the 20–30 nm range, above which the formation of multiple magnetic domains leads to a reduction in coercivity. 41
The Néel relaxation time describes the time required for the magnetic moment of a nanoparticle to flip direction due to thermal fluctuations. This model is particularly applicable to superparamagnetic particles in the single-domain state, where magnetic moments can rapidly reorient under thermal agitation.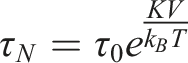
Here, K represents the anisotropy energy density of Fe3O4, which is typically 1.35 × 104 J/m3. 42 V is the volume of a nanoparticle, given as V = 1.15 × 10−24 m3. kB denotes Boltzmann’s constant, where k_B = 1.38 × 10−23 J/K, and T = 300 K represents the ambient temperature. τ0 is the characteristic time constant, typically ranging between 10−9 and 10−10 seconds. 43
For a spherical Fe3O4 nanoparticle with a diameter of 13 nm, the Néel relaxation time (τN) is calculated as 42.7 ns. This result suggests that the nanoparticle may exhibit superparamagnetic behavior at room temperature, where its magnetic moment can rapidly flip due to thermal fluctuations, given the relatively short relaxation period.
To determine the critical particle diameter at which Fe3O4 transitions from a single-domain to a multi-domain state, it is necessary to equate the anisotropy energy (KV) to the thermal energy (kBT). This condition defines the threshold at which thermal fluctuations overcome magnetic anisotropy, leading to a transition in magnetic behavior.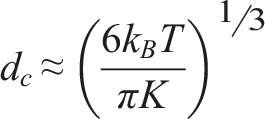
The critical particle size at which Fe3O4 transitions from a single-domain to a multi-domain state is approximately 8.46 nm at room temperature. This suggests that Fe3O4 nanoparticles smaller than 8.46 nm will predominantly remain in the single-domain state, while larger nanoparticles are more likely to transition into the multi-domain state.
The observed decrease in coercivity in HDPE + Fe3O4 nanocomposites at higher filler concentrations can be attributed to this single-domain to multi-domain transition. This result aligns well with the SEM histogram, which shows that although the average particle size is 13 nm, the majority of Fe3O4 nanoparticles have a diameter of approximately 8 nm. Consequently, the shift in magnetic domain state leads to a reduction in coercivity, as evidenced in Table 4.
The correlation between the critical particle size, Fe3O4 nanoparticles in the HDPE polymer matrix, and coercivity is illustrated in Figure 8. A schematic representation coercivity (Hc) behaviour of a HDPE + Fe3O4 nanocomposites depending on filler concentration related with particle size.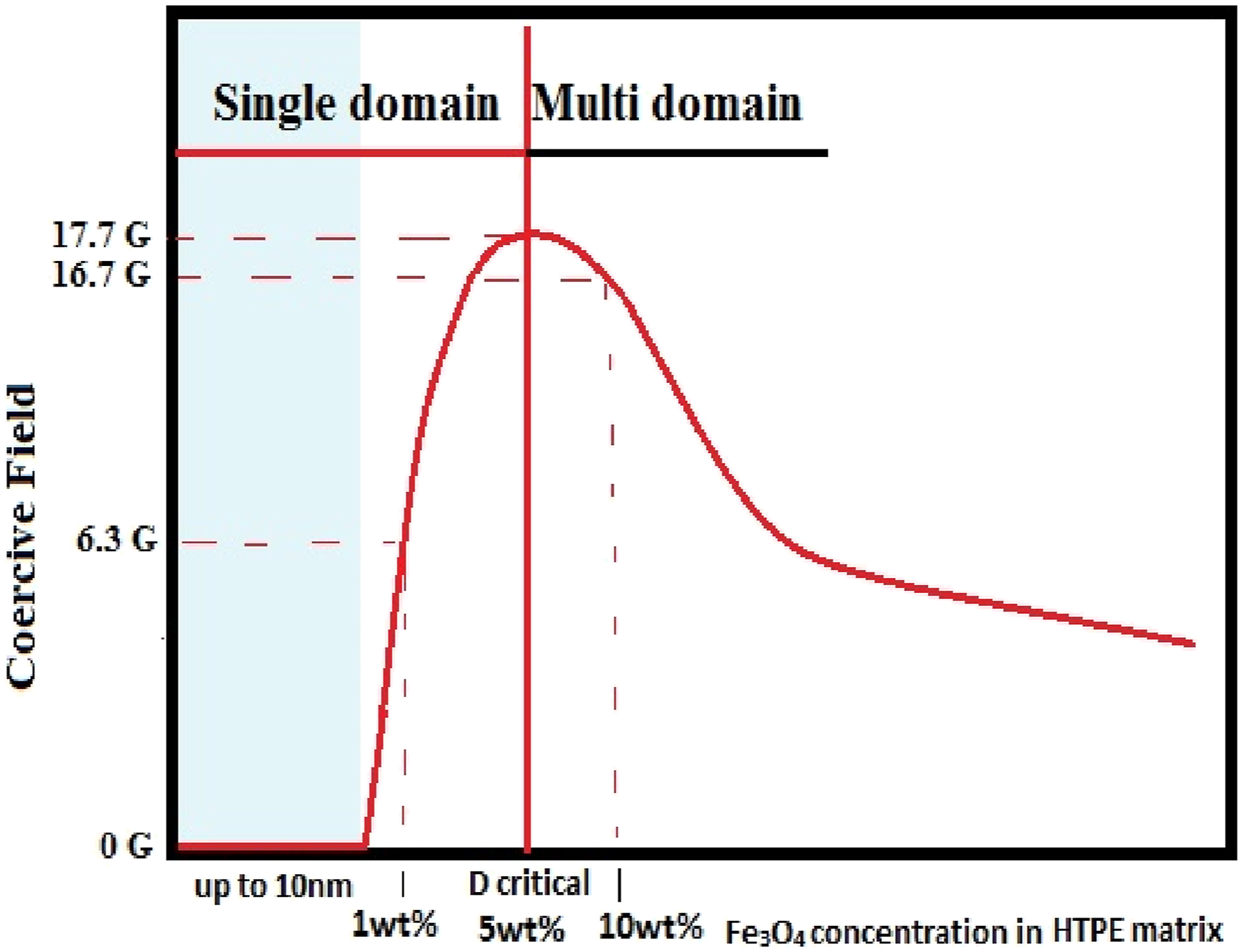
Conclusion
In this study, HDPE + Fe3O4 nanocomposites were synthesized and systematically characterized. SEM analysis revealed that while the average size of magnetite nanoparticles in the polyethylene matrix is 13 nm, the majority of the particles fall within the 8–10 nm range. Additionally, AFM 3D images and corresponding histograms confirmed that the incorporation of iron oxide into polyethylene significantly alters its supramolecular structure. These findings are further supported by XRD data, which exhibit strong agreement with both SEM and AFM results.
The thermal stability of the HDPE + Fe3O4 nanocomposites was analyzed as a function of Fe3O4 concentration. It was determined that the introduction of Fe3O4 nanoparticles into HDPE enhances the thermostability of the material by approximately 150°C.
DSC analysis indicated that the incorporation of Fe3O4 nanoparticles has a negligible effect on both the melting temperature (Tmelting) and crystallization temperature (Tcrystallization) of HDPE. Moreover, at low Fe3O4 concentrations, the degree of crystallinity remains largely unchanged. A significant change in the crystallization degree is observed only at 40 wt.% Fe3O4, suggesting that high nanoparticle loading influences the crystalline structure of the polymer matrix.
Furthermore, magnetic measurements demonstrated that Fe3O4 nanoparticles remain in a single-domain state at concentrations up to 5 wt.% Fe3O4 in the HDPE matrix, confirming their superparamagnetic behavior at these lower concentrations.
The HDPE/Fe3O4 nanocomposites exhibit improved thermal stability and magnetic properties, making them suitable for applications requiring high-temperature resistance and magnetic responsiveness. Potential applications include: Electromagnetic shielding materials: Due to their magnetic properties, these nanocomposites can be used in EMI shielding applications. Smart packaging materials: The increased thermal stability and structural modifications could be beneficial in food packaging or industrial containers that require durability. Magnetically responsive polymers: They can be used in biomedical applications, such as drug delivery systems or hyperthermia treatment, where magnetic response is essential. Structural and automotive components: The enhanced mechanical properties and resistance to degradation make them useful in automotive or construction applications where polymers are exposed to mechanical and thermal stresses. Sensor materials: The integration of Fe3O4 nanoparticles can enable the development of magnetic and thermal sensors.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

