Abstract
The use of polymer matrix thermoplastic composite materials in the aviation sector has been increasing due to the superior properties. Materials used in airplanes are expected to have good fire resistance properties, and so composite materials offer an advantage to engineer properties. In this study, the surface of a commercially obtained 50% carbon fiber (CF) reinforced polyphenylene sulfide (PPS) composite plate was coated using a spray coating method with reduced graphene oxide (r-GO) and zinc borate, both known for their flame-retardant and green properties. The structural characterization, contact angle measurements, surface morphology determination, combustion analysis, thermal analysis, and mechanical analysis of both coated and uncoated samples were conducted. The limit oxygen index (LOI) value of the uncoated 50CF/PPS composite plate was measured as 59.2 ± 0.25%. The LOI value of the r-GO coated sample was increased to 61.0 ± 0.25%, while the LOI value of the sample coated with both r-GO and zinc borate was increased to 61.8 ± 0.57%. Thus, it was concluded that co-coating of r-GO and zinc borate synergistically enhances the LOI value of the composite material. The results of the study are believed to be industrially applicable.
Introduction
Polymer composite materials have been preferred in the aerospace industry due to their light weight, reasonable strength and low fuel consumption. Compared to the earliest aircrafts, Airbus A350 and Boeing Dreamliner are now made up of 50% composite materials. 1 Thermoset polymers have been already used as matrixes in aerospace applications due to their high chemical, thermal and structural durability. On the other hand, thermoplastic polymer matrixes have been recently considered as one of the promising alternatives offering shorter
Autoclave times, easy in processing, and higher impact resistance compared to the thermosets. Especially semi-crystalline thermoplastics such as polyetheretherketone (PEEK) and polyphenylene sulfide (PPS) have mostly used matrixes due to their inherent high toughness and flame retardancy. 2 Thermal durability and flame retardancy are very critical criteria for aerospace applications to work safely under rush conditions such as extraordinary tribological conditions. For example, drones used for fire extinguishing purposes must be protected from high temperatures to keep their structural and electronic integrity. Similarly, it is known that aircraft accidents taken place by fire will cause loss of life and property. In the report of Transportation Safety Board of Canada, it was announced that 193 of the 290 aircraft accidents were significant and 11% of these accidents were caused by fire. To overcome these drawbacks of aerospace technology materials, it is important to enhance the flame retardancy, surface morphology and other tribological properties of composite materials.
PPS is one of the high-performance thermoplastic polymers with aromatic ring structure containing sulfur bonds. Although PPS matrixes show lower Tg (85°C) and Tm (285°C) than PEEK (Tg = 143°C and Tm = 343°C), they have superior solvent and chemical resistance.3,4 In addition, PPS is inherently flame retardant due to its chemical structure and char formation. It has been known that about 6 tons of thermoplastic materials were used in the structure of Airbus A380 aircraft, of which about 2.5 tons consisted of PPS. It was also recommended to use high standard non-flammable and self-extinguishing as large area materials for aerospace applications. 5 The limiting oxygen index (LOI) of epoxy, PEEK and PPS polymers were reported to be 21%, 6 38% 7 and 44%,8,9 respectively indicating flammable resistance. However, self-extinguishing property can be improved by increasing LOI value, and thus, a fiber reinforced composites are prepared in the literature such as carbon fiber (CF).10,11
Fibers are the most common reinforcement agents for aerospace materials owing to their outstanding mechanical and corrosion resistive properties. Carbon, glass and aramid fiber– reinforced polymers are commercially available materials. 12 Carbon fibers (CF) are known for their high strength-to-weight ratio and stiffness; thus, they can be used in wings and motion stabilizers in aircrafts. It was reported that CF reinforcement is more advantageous than metallic materials in terms of their high strength, stiffness and corrosion resistance. 13 Additionally, LOI values of CF reinforced polymers were enhanced to be 30% for CF/epoxy 6 and 47% for CF/PEEK. 14 Moreover, it was also reported that the decomposition temperature 485°C of PPS increased to 492°C by addition of thermally stable CF to the structure. 15
The studies about CF reinforced PPS reported to show improved tensile and impact properties. 16 On the other hand, the enhancement in thermal stability of CF/PPS composite is not detrimental for aerospace applications. 17 Thus, it indicates a need to increase thermal and flame retardancy properties of CF/PPS composites.
Halogens, metal hydroxides, phosphorus, borates and inorganic materials are common examples for flame retardants. Each of these materials suppress combustion of the surface by acting through different mechanisms. The use of halogen-containing flame retardants has been restricted or banned in some countries due to their harmful effects on the environment and human health. The 2014 conference ‘Meeting High Performance Flammability Requirements for Aviation’ reported that it is important to use environmentally friendly and high performance flame retardant materials. For this purpose, new types of flame retardants in accordance with the principles of green chemistry and Paris green deal agreement are needed. 18 Graphene and zinc borate are suitable candidates to meet these requirements, and many studies have been reported on this topic. It was reported that the use of graphene on the combustion of polymer nanocomposites led to increase in the heat dissipation rate, increased char formation, which inhibits the heat and mass transfer during combustion. 19 Further, zinc borate turns into a glassy phase with heat and forms an insulating barrier against flame. It was also reported that LOI value of the wood increased from 15% to over 30% by the addition of zinc borate. 20
Many methods have been proposed in the literature for the production of composite materials. If composites are formulated correctly, superior tribological properties may be obtained. 21 Flame retardant additives are usually added during the in-situ composite production stage. However, some problems are encountered when composite structures consisting of at least two different materials are in the production platform. These can be listed as the fact that the materials used are not distributed homogeneously in the composite structure, the cost of the materials is high and long production processes are required. Therefore, the surface coating can be promising method in order to improve combustion properties without changing applicable process and price for the production of composite material.22–25
In this study, surfaces of the commercially available 50%CF/50%PPS plates were coated with reduced graphene oxide (r-GO) and r-GO/zinc borate micro-particles, respectively via spray coating technique to produce high performance flame retardant and environmentally friendly aerospace materials. Structural, surface, thermal and mechanical characterizations of CF/PPS/r-GO and CF/PPS/r-GO/zinc borate hybrid composites were carried out and the synergistic effects of r-GO and zinc borate coating on flame retardancy of the materials are analyzed.
Experimental
Materials
Graphite (<20 µm), zinc borate (3ZnOB2O3.7/2H2O), and all the other chemicals were purchased from Merck (Germany) and used as received. CF/PPS composite plate with 50% by mass CF content was kindly supplied by Turkish Aerospace Industries, Inc. (TUSAŞ) with the specifications of; Fabric: Carbon-Flat weave J300 JB, 200 g/m2, PPS Fiber volume content: 50 ± 3%.
Synthesis of GO and r-GO particles
Graphene oxide (GO) was prepared by a modified Hummers method using pure graphite powder. 26 For this purpose, 10 g of graphite was expanded with 5 g of K2S2O8 and 5 g of P2O5 in concentrated H2SO4 at 80°C for 6 h. Then, 2 g of expanded graphite and 6 g of KMO4 mixed concentrated H2SO4. Reaction was terminated with addition of water and H2O2(aq). Product GO was washed three times with distilled water until pH 5. Brown GO was obtained and dried under vacuum oven.
100 mg GO was dispersed in 100 mL of water by ultrasonicator for 2 h, and then pH was adjusted to 10. Into the dispersions, 200 mg of NaBH4 was added at 8°C and stirred for 6 h. The product r-GO was washed with water and collected by centrifugation. Black r-GO was dried under vacuum oven.
Preparation of r-GO/zinc borate dispersions
Zinc borate (3ZnOB2O3.7/2H2O) was dispersed in water by 5 wt.%. Then, dispersion of r-GO with 5 wt.% was added into zinc borate dispersion to obtain r-GO/zinc borate ultimate dispersion.
Etching of CF/PPS composite surfaces
CF/PPS plates were cut into certain dimensions via a water-jet device for further characterizations (Figure 1(a)). Then, the specimens were fixed to the floor with a tape to prevent Slipping during the etching. They were abraded with 120 grit sandpaper using a Deburr machine and it was important to ensure that each area of the sample was abraded homogeneously (Figure 1(b)). CF/PPS composite specimens of (a) water-jet cut, (b) abrasion with sandpaper, and (c) thermal annealing after spray coating.
Spray coating of r-GO and r-GO/zinc borate onto CF/PPS surface
R-GO and r-GO/zinc borate dispersions were spray-coated separately onto the surfaces of CF/PPS plates after surface etching processes. A simple spray gun was used for coating. The dispersions were sprayed for 3 min at a distance of 30 cm with angles of 45° and 90° to obtain 5 µm thickness then subjected to air drying. Then, the coated specimens were cured in a 120°C oven for an hour (Figure 1(c)).
Structure and surface analyses
Zeta (ζ)-potentials of r-GO, zinc borate and r-GO/zinc borate particles were measured in aqueous dispersions with 0.1 g L−1 concentration at pH 7 and 25°C by using Malvern Zeta-sizer Nano-ZS (England) instrument equipped with 4-mW He-Ne laser operating at λ = 633 nm and non-invasive backscatter system working for 173° backscatter lights. ζ-potentials were given as averages of 300 times repeated measurements.
ATR-FTIR analyses (Shimadzu, Japan) of CF/PPS, CF/PPS/r-GO and CF/PPS/r-GO/zinc borate samples were carried out to identify their chemical structures.
Contact angles (SEO Phoenix 300 10/220 V, 50/60 Hz, Korea) of the specimens were investigated by imaging a water droplet on the surfaces to determine the hydrophilicities. Surface roughness values of the specimens were determined as μm Ra by Mitutoyo instrument (China). SEM (Tescan, Czech) images and energy-dispersive X-Ray (EDX, Bruker, Germany) analysis of the specimens with gold coatings (Cressington Sputter Coater, USA) were performed for morphological characterization of the surfaces.
Thermal and flame retardancy analyses
Thermal characterizations of the specimens were performed by DSC (DSC7020, Hitachi, Japan) under N2 atmosphere in the temperature range of 25°C−600°C with a scan rate of 10°C/min. TGA (Hitachi STA 7300, Japan) thermograms were also obtained under N2 atmosphere in the temperature range of 25°C–600°C with a scan rate of 10°C/min. Transition temperatures, decomposition temperatures, decomposition rates and mass losses were obtained and compared for two specimens.
LOI test was performed via Fire Testing Technology (China) instrument by using the specimens with ASTM D2863 standard provided by the manufacturer (dimension: 120 × 15 × 2 mm). 27
Vertical combustion analyses by UL-94 test (Fire Testing Technology, China, 220 V) was carried out by following ASTM D3801-00 standard procedure.
Mechanical analyses
Tensile and three-point bending tests (Electronic Universal Testing Machine, WDW-100, UK) were performed with specimens sized 100 × 25 × 2 mm according to EN ISO14125 standard.
Result and discussion
Structure and surface analyses
CF and PPS have non-polar aromatic groups28,29; thus, the hydrophobic and π-π stacking interactions between CF/PPS surface, r-GO and r-GO/zinc borate particles in aqueous dispersions are expected. 30 Evaporation of water also enhanced the hydrophobic interactions between surface of CF/PPS and r-GO particles. 31 Additionally, physical etching process on CF/PSS surface leads to increase in the surface-to-volume ratio and the number of π-π stacking points with r-GO particles. 32 To understand the surface properties of r-GO, zinc borate, and r-GO/zinc borate hybrid, ζ-potentials were investigated in aqueous dispersions: ζ-potential (r- GO) = −15 ± 1 mV, ζ-potential (zinc borate) = +14 ± 1 mV, and ζ-potential (r-GO/zinc borate) = −3 ± 2 mV. The negative surface potential of r-GO can be attributed to the unreduced units of GO structures. It was also concluded that there are some strong electrostatic interactions between r-GO (ζ = −15 mV) and zinc/borate (ζ = +14 mV) structures. Moreover, r-GO/zinc borate particles have slightly negative charge (ζ = −3 mV) indicating available r-GO units on its surfaces, which can interact with CF/PPS surfaces.
ATR-FTIR spectra of CF/PPS, CF/PPS/r-GO and CF/PPS/r-GO/zinc borate surfaces were comparatively analyzed (Figure 2). For CF/PPS, sharp peak at 1000 cm−1 is due to the stretching vibrations of C-S bond of PPS.
33
The ring stretching peak of PSS is observed at 1450 cm−1.
34
In addition, the peaks in 2850–2900 cm−1 region are corresponding to the stretching vibrations of C-H bond of both PSS and CF components.
35
For CF/PPS/r-GO and CF/PPS/r-GO/zinc borate, the intensity of C-H bond vibrations reduced which may be attributed to the mechanical etching and the coating of surfaces. In the literature, it has been reported that perfluoroalkoxy particles coated on aluminum surfaces led to a decrease in the FTIR peak intensities.
36
Same kind of reductions in ATR-FTIR signal intensities were also observed for CF/PPS/r-GO and CF/PPS/r-GO/zinc borate samples which may be attributed to the successful coatings of r-GO and zinc borate onto the surfaces of CF/PPS plates. FTIR spectra of the surfaces of CF/PPS, CF/PPS/r-GO and CF/PPS/r-GO/zinc borate samples.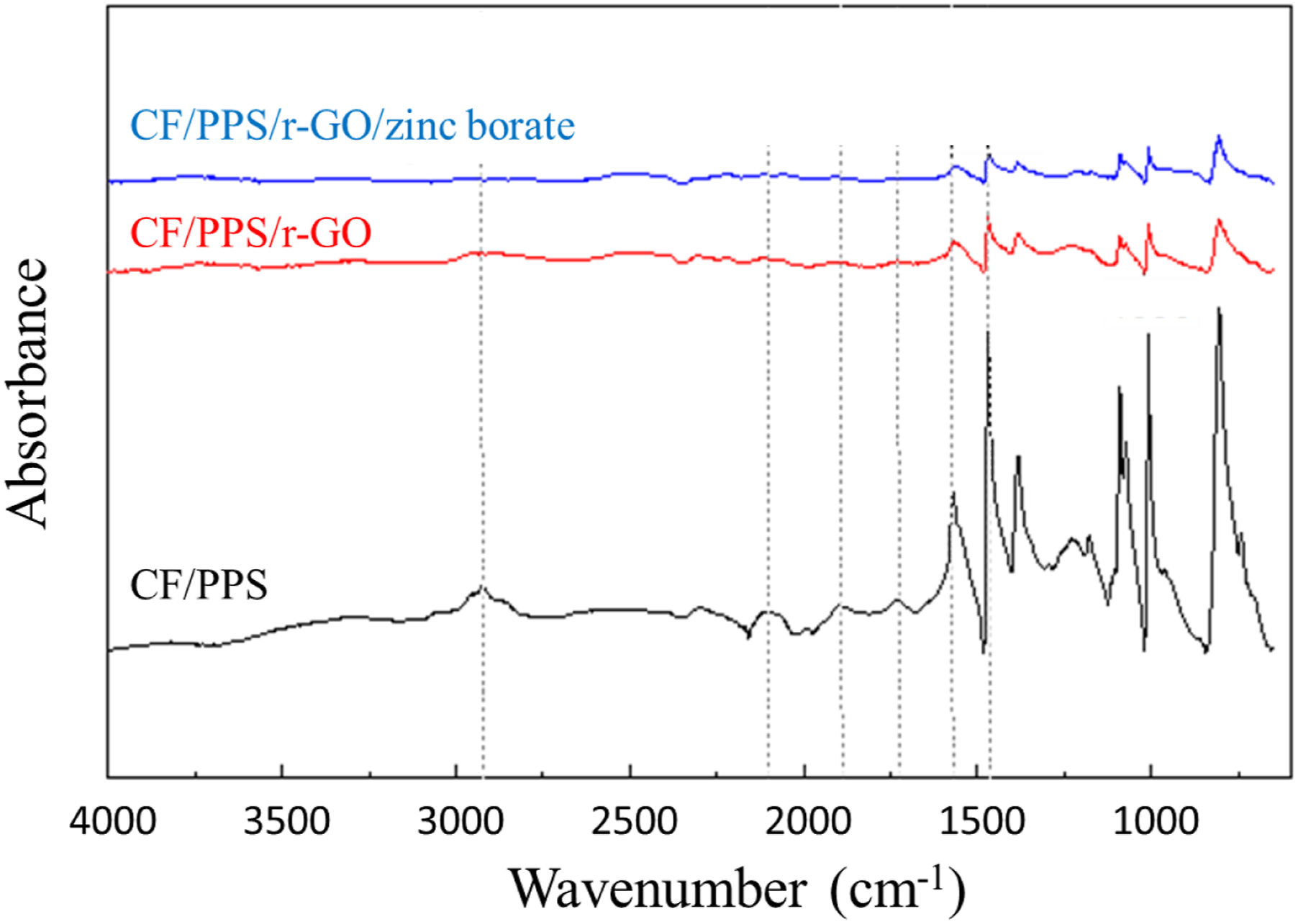
Contact angle (CA) measurement results of CF/PPS, CF/PPS/r-GO and CF/PPS/r-GO/zinc borate surfaces are given in Figure 3. The CA of CF/PPS sample was measured as 74° ± 1° and determined to be slightly hydrophilic nature due to polar –OH, –COOH and –S– groups.
29
As a result of mechanical etching applied to the surface of the CF/PPS plate, the CA was observed to increase to 102° ± 3°, and this enhancement in the hydrophobic character of the plate was attributed to the rising surface roughness.37,38 After coating with r-GO and r-GO/zinc borate, the sample surfaces observed to show more hydrophilic character having a contact angle of 34° ± 1° and 45° ± 1°, respectively. Although graphene is considered as hydrophobic material, it was reported that thermal annealing produces hydrophilic graphene surfaces.
39
Additionally, ζ-potential of r-GO showed that −15 mV surface charge is due to the unreduced GO units, which is considered has a hydrophilic character. The CA of CF/PPS/r-GO/zinc borate (45°) was higher than CF/PPS/r-GO (34°) sample.
40
This may be attributed to the low ζ-potential (−3 mV) of r-GO-zinc borate particles and high thermal degradation stability (520°C) of zinc borate preventing the degradation of r-GO during the thermal annealing.40,41 Thus, it was concluded that after surface coating, CF/PPS plates became more suitable for possible aerospace applications. Contact angle measurements of (a) CF/PPS, (b) etched CF/PSS, (c) CF/PPS/r-GO and (d) CF/PPS/r-GO/zinc borate samples.
The surface roughness is important for coating processes to decide the quality of samples and the processing performance of the material. 42 The surface roughness of the untreated CF/PPS plate was determined to be 8 Ra indicating an almost smooth morphology. After mechanical etching with a sandpaper, the surface roughness increased to 101 Ra in accordance with the CA results. This indicates increase in surface-to-volume ratio, as expected. Thus, hydrophobic and π-π stacking interactions are enhanced, resulting in better adhesion of r-GO and r-GO/zinc borate coatings onto the surfaces of CF/PPS plates. Increasing surface roughness to 121 Ra and 135 Ra after deposition of r-GO and r-GO/zinc borate, respectively approve the successful coating.
The morphological and structural analysis of CF/PPS, CF/PPS/r-GO and CF/PPS/r-GO/zinc borate surfaces were examined by SEM-EDX measurements (Figure 4). The micrograph of CF/PPS sample showed smooth surface with uniformly stacked CF. EDX results revealed high amount of S and O atoms due to the presence of PPS. For CF/PPS/r-GO sample, the smoothness of CF/PPS surface was disrupted in consistence with the surface roughness measurements. Moreover, EDX patterns showed that the amount of S and O atoms decreased as a result of coating with r-GO micro-particles. After coating of surfaces of CF/PPS plates with a dispersion composed of r-GO/zinc borate mixture, the surface roughnesses were observed to increase. This may be attributed to the needle-like geometry of zinc borate particles.
43
Additionally, this approves the co-coating of r-GO and zinc borate as targeted. Similar to CF/PPS/r-GO, the amount of S and O atoms decreased for CF/PPS/r-GO/zinc borate when compared to the virgin CF/PPS plate. On the other hand, O atom content slightly increased after zinc borate coating of CF/PPS/r-GO surface, as expected. SEM micrographs and EDX results of (a) CF/PPS, (b) CF/PPS/r-GO and (c) CF/PPS/r-GO/zinc borate samples.
Thermal and flame retardancy analyses
Glass transition (Tg), melting (Tm) and crystallization temperature (Tc) results obtained from DSC thermograms for CF/PPS, CF/PPS/r-GO and CF/PPS/r-GO/zinc borate samples are given in Figure 5. The onset Tg of CF/PPS composite was determined to be 40°C, which is smaller than the previously reported value (85°C) of Tg of PPS.
44
This may be due to the increasing free volume of PSS by introduction of carbon fibers into the composite structure.
45
Similarly, the addition of r-GO and zinc borate decreased the values of Tg to 38°C for CF/PPS/r-GO, and to 24°C for CF/PPS/r-GO/zinc borate samples. The plasticizing effects of flame retardant additives were reported in the literature.46–49 For example, increasing zinc oxide content in borosilicate glass caused a decrease in Tg upto a certain concentration, and then caused an increase on Tg of the material.46 Therefore, lower Tg values in current study may be attributed to the coating of small amounts of r-GO and zinc borate onto the surfaces of CF/PPS. DSC thermograms of CF/PPS, CF/PPS/r-GO and CF/PPS/r-GO/zinc borate samples.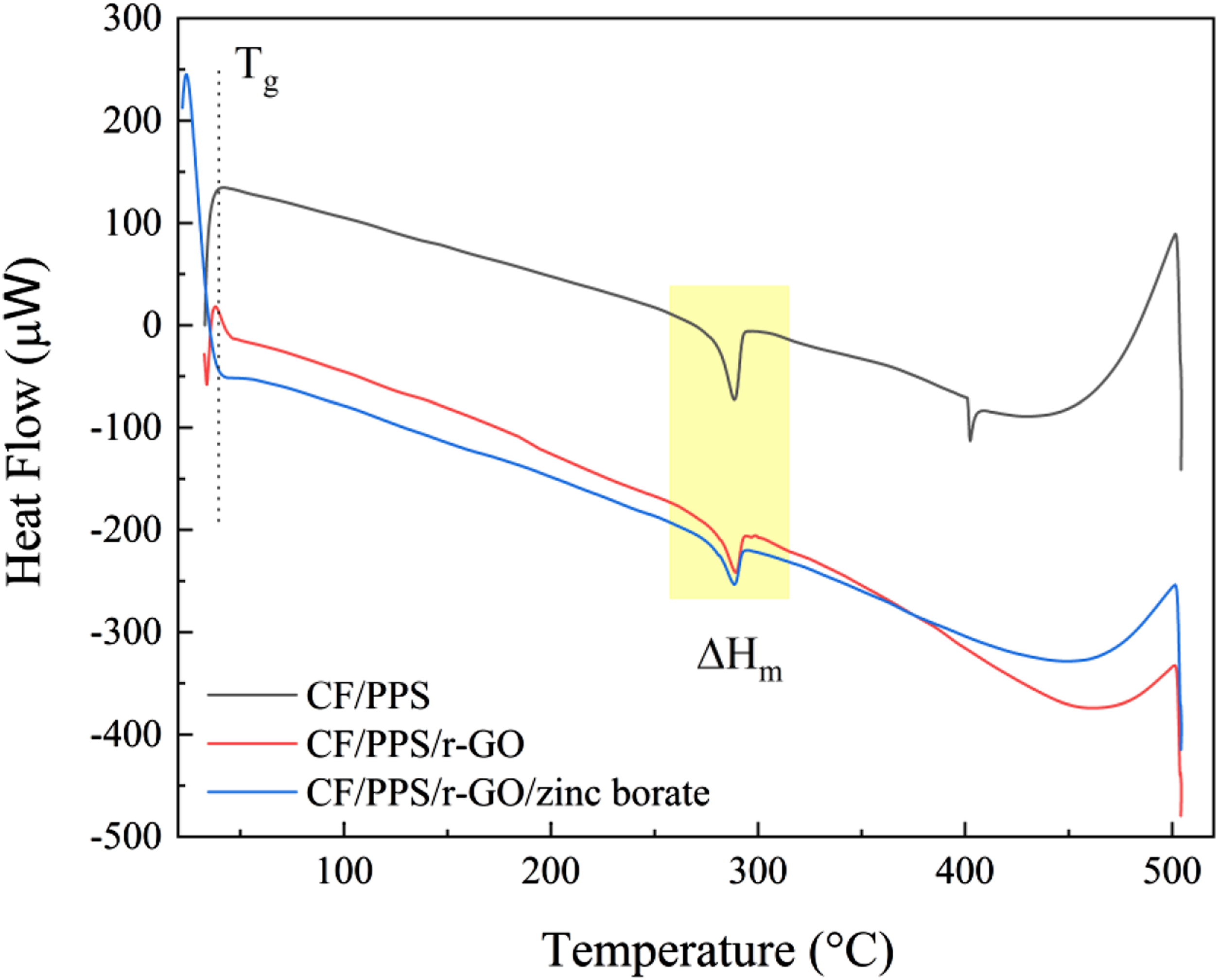
The melting point (Tm) was observed for CF/PPS at 288°C. A similar high Tm value was reported in the literature for PPS due to its high crystallinity. 50 The second thermal degradation point at 400°C may be attributed to the isothermal aging of PPS chains.51,52 This analysis revealed that the annealing temperature of 120°C, which is above Tg (40°C), below Tm (288°C) and Tdecomposition (400°C) leads to both evaporation of the water and homogenization of the surface by rearranging the PPS chains. 53 Similar to CF/PPS, the melting zone at 288°C was also observed for CF/PPS/r-GO and CF/PPS/r-GO/zinc borate samples. However, the second degradation zone at 400°C was disappeared for CF/PPS/r-GO and CF/PPS/r-GO/zinc borate. This may be attributed to enhanced thermal retardancy of CF/PPS surfaces after coating with r- GO and zinc borate dispersions. The fusion enthalpies were calculated to be −555 J/g for CF/PPS, −272 J/g for CF/PPS/r-GO, and −325 J/g for CF/PPS/r-GO/zinc borate. Although r-GO resulted in significant decrease in the enthalpy of fusion, zinc borate coating recovered the thermal property of the sample.
TGA thermograms for CF/PPS, CF/PPS/r-GO and CF/PPS/r-GO/zinc borate samples are given in Figure 6. No apparent mass losses were observed for all the samples before 400°C indicating high thermally stability. CF/PPS sample started to decompose at 442°C as a result of desulfonation and destruction of the PPS backbone.
54
The second decomposition at 563°C may be attributed to the decomposition of CF units.
55
The total mass loss of CF/PPS in this process was 30%. Additionally, the main mass losses for CF/PPS/r-GO sample occurred at high temperatures of 425°C and 566°C corresponding to 29%. This revealed that r-GO coating slightly enhanced the second decomposition temperature from 563°C to 566°C. On the other hand, CF/PPS/r-GO/zinc borate sample was decomposed at higher temperatures of 450°C and 570°C with 29% mass loss. As a results of this, highly thermally stable r-GO
56
and zinc borate coatings57,58 improved the thermal degradation behavior of CF/PPS structure by dissipating the heat accumulating on the surface of the sample and the best thermal stability was achieved after co-coating of CF/PPS surfaces by r-GO/zinc borate dispersion as targeted. TGA thermograms of (a) CF/PPS, (b) CF/PPS/r-GO and (c) CF/PPS/r-GO/zinc borate samples.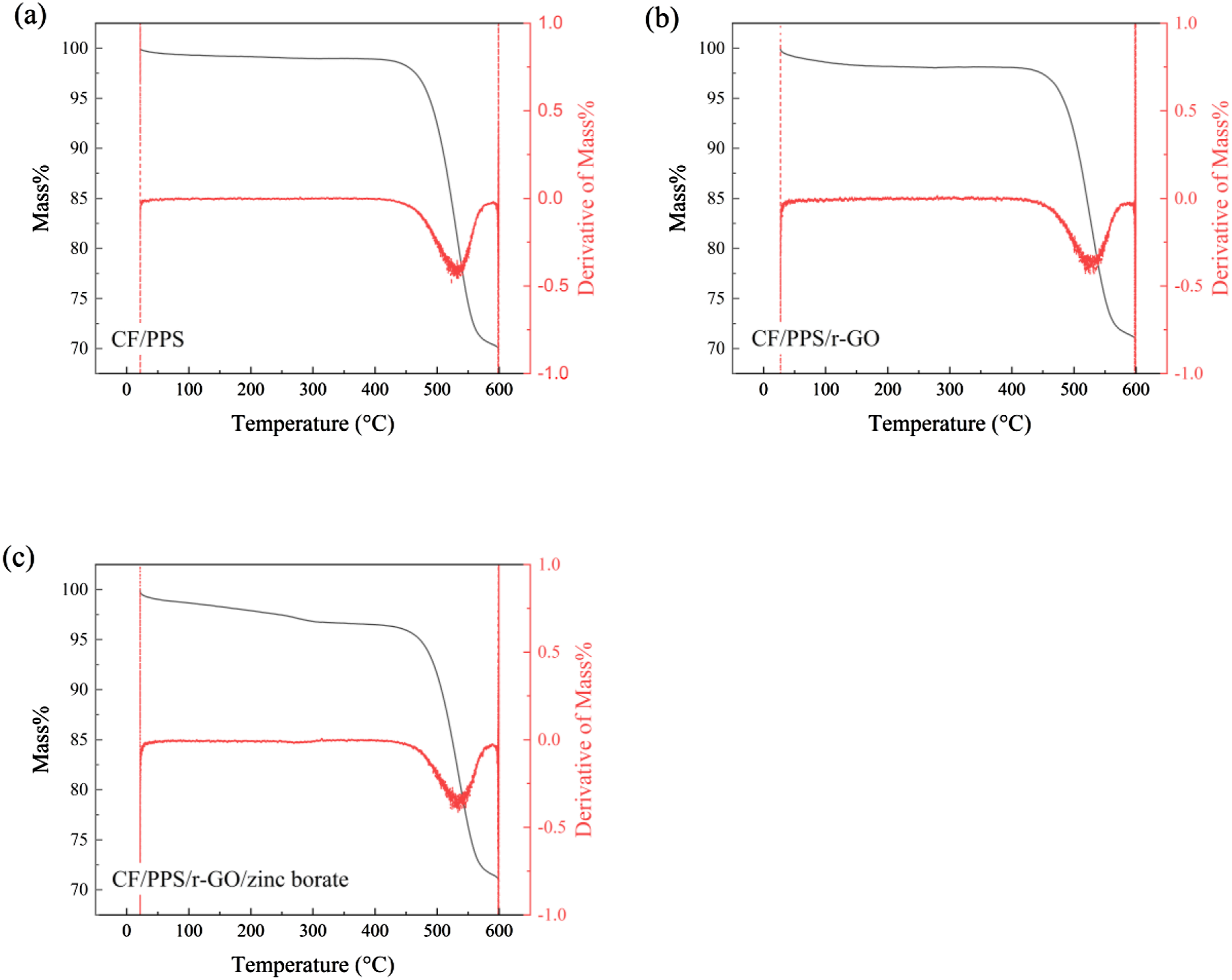
UL-94 test was performed according to ASTM D3801-00 standard to examine the flammability and flame spread of CF/PPS, CF/PPS/r-GO, and CF/PPS/r-GO/zinc borate samples in vertical positions. During the combustion test, no burning and dripping were occurred for all the samples and the results are recorded as V0, non-flammable. CF/PPS plate has already had flame retardancy property (V0); thus, enhancing the flame retardancy of the CF/PPS plate presents a significant challenge. The improvements observed through the evaluation of LOI values will be analyzed in detail.
The LOI test is based on measuring the minimum amount of O2 that must be present in the environment for a material to start burning. The LOI value of CF/PPS composite sample was determined to be 59.2 ± 0.25%. The coating with r-GO on CF/PPS surface improved the LOI value to 61.2 ± 0.25%. Additionally, LOI value of CF/PPS/r-GO/zinc borate sample slightly increased to 61.8 ± 0.25% within the experimental limits, as targeted. Although it is very difficult to achieve an enhancement in the LOI values of the samples which already have high LOI values, co-coating with r-GO/zinc borate dispersion has shown a synergistic effect as targeted. The highest LOI value of 52% was reported in the literature for glass fiber and CaAl layered double hydroxide modified PPS material. 59 To the best of our knowledge, a LOI value as high as 61% for PPS composites has not hitherto been reported in literature.60,61
When the applied flame was withdrawn, self-extinguishing of the samples were observed after a while (Figure 7). The length of the burned area in the CF/PPS rectangular in shape plate was measured to be 35 mm. For CF/PPS/r-GO, the length of char layer is observed to reduce to 25 mm. The reason of this decrease on the char layer may be attributed to the formation of a physical insulating layer on the surface of CF/PPS/r-GO plate by the chemical reactions, including dehydration, cross-linking, and carbonization reactions of r-GO.62–64 Carbonaceous material r-GO increases the melt viscosity of material under treatment by trapping gaseous components of the combustion process and also inhibiting the transfer of heat giving rise to the intumescence of char which prevents the further progress of the flame.65,66 Moreover, r- GO/zinc borate coating provided only 16 mm char residue on the surface of CF/PPS/r-GO/zinc borate plate. It was also reported on a study carried out on “effect of zinc borate as a flame retardant formulation on some tropical woods” that zinc borate thermally decomposes into boron oxide (B2O3), which forms a protective layer acting as an oxygen barrier and inhibiting volatility of the flame.
20
The effect of this layer can be seen in Figure 7(c) as trapped gases. This also indicates the protection of the interior CF/PPS matrix through reducing the combustion rate, the smoke and the heat releases.
67
In accordance with LOI values, char yield properties indicate that the co-coating of r-GO and zinc borate onto CF/PPS surface results in a synergistic effect, as targeted. These results also meet the requirements of using materials in certain parts of the aircraft. In conclusion, for the aerospace applications even 1% fire resistance enhancement is a critical achievement which provides a few minutes during fire as mentioned in the literature.
68
LOI test images of (a) CF/PPS, (b) CF/PPS/r-GO and (c) CF/PPS/r-GO/zinc borate samples.
Mechanical analyses
Tensile strengths of CF/PPS, CF/PPS/r-GO, and CF/PPS/r-GO/zinc borate specimens are investigated and results obtained are depicted in Figure 8. The tensile strength and elongation% of CF/PPS sample were calculated to be 632 MPa and 3.6%, which is similar with the previously reported literature.
69
After coating of CF/PPS surfaces with r-GO and r-GO/zinc borate, tensile strength was observed to decrease to 451 and 520 MPa, respectively with the elongation of 0.7% for the both samples. This reduction in tensile strengths may be attributed to the decreased interaction of CF with PPS matrix after mechanical etching.
70
Similar to the thermal properties obtained from TGA, toughness (areas under the curves) was decreased with addition of r-GO but improved by addition of r-GO/zinc borate coating. The better interactions of polar zinc borate with polar PPS chains may lead to enhancement in the toughness and also tensile strength of CF/PPS/r-GO/zinc borate when compared to CF/PS/r-GO plate.
71
However, there are some reports in the literature carried out on polyethylene/rubbers/magnesium oxide
72
and on wood flour/polypropylene composites mentioning that zinc borate reduces the mechanical properties of materials.
73
Tensile test results of CF/PPS, CF/PPS/r-GO and CF/PPS/r-GO/zinc borate samples.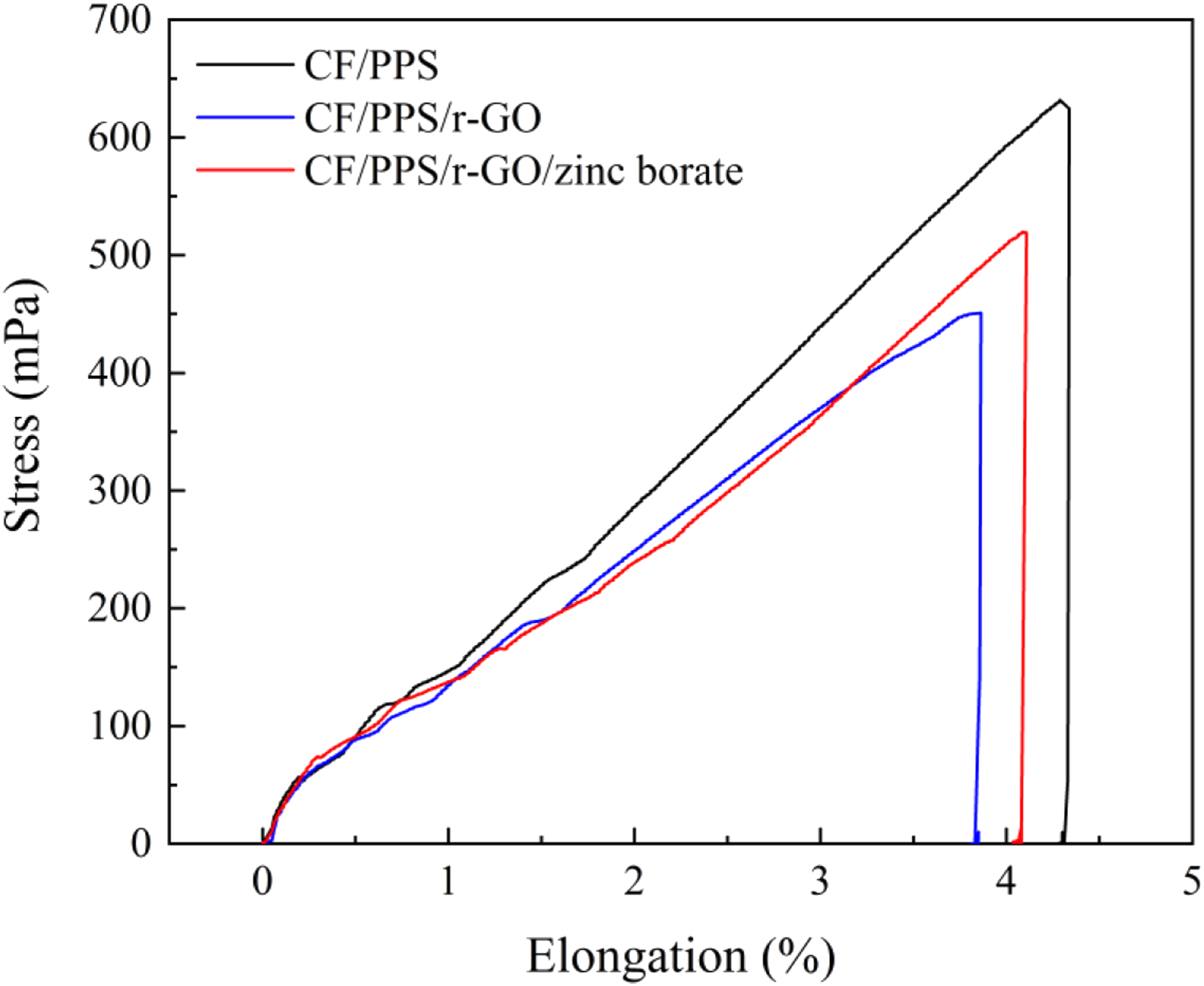
Fracture behaviors of the samples were analyzed by three-point bending test. The results showed that CF/PPS, CF/PPS/r-GO and CF/PPS/r-GO/zinc borate plates broke at 250 s, 280 s and 285 s, respectively. Stress-strain curves in Figure 9 revealed that CF/PPS plate start to failure at 10 mm under 14 MPa. However, r-GO coating caused slightly decrease in the elasticity and the failure stress (12 MPa) in bending. In accordance with both thermal and tensile test results, the elasticity and the failure stress (13 MPa) in bending was again recovered after coating with r-GO/zinc borate. Three-point bending stress-strain results of (a) CF/PPS, (b) CF/PPS/r-GO and (c) CF/PPS/r-GO/zinc borate samples.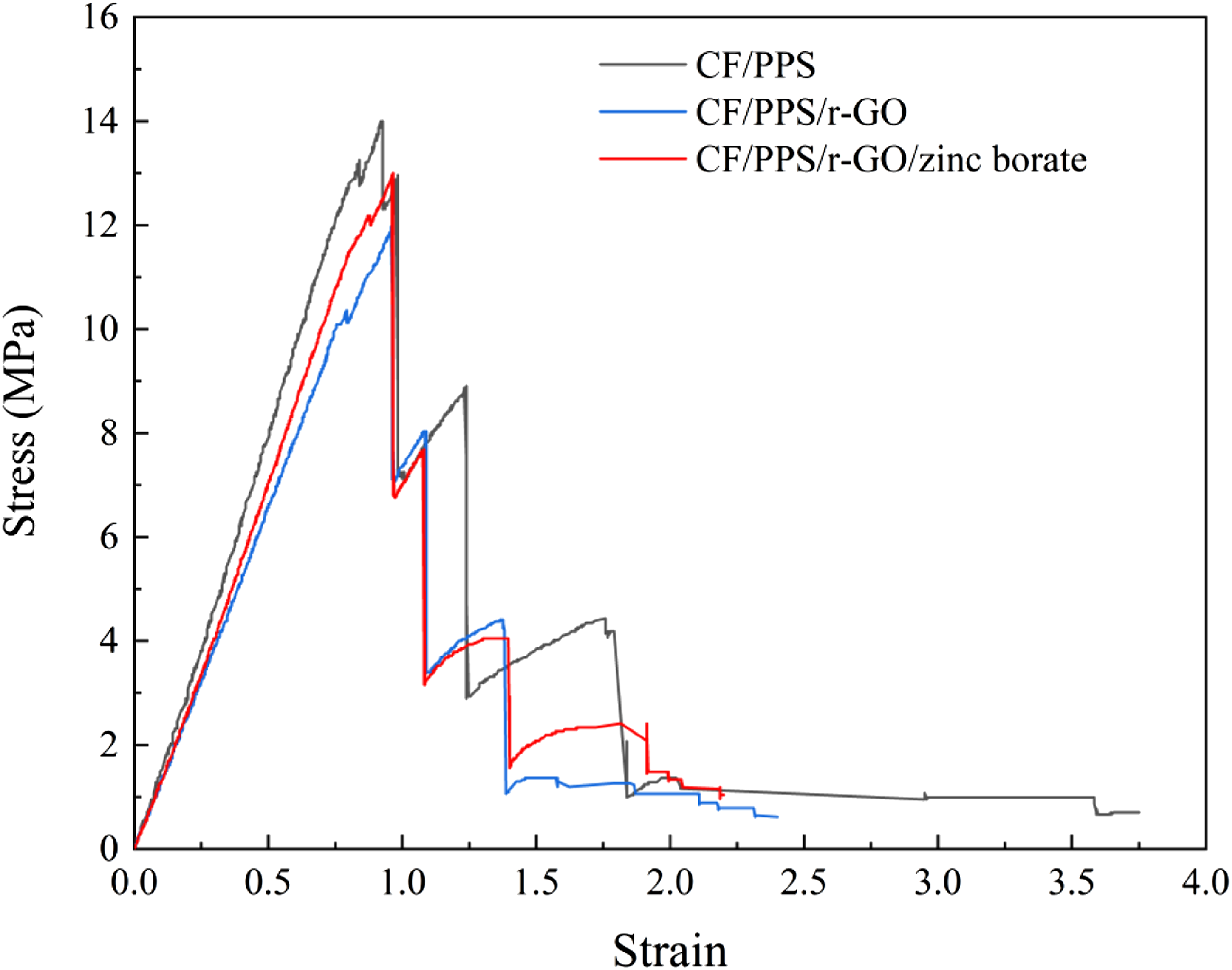
Conclusion
Surface of the CF/PPS composite plate was coated with r-GO and r-GO/zinc borate dispersions by spray coating method. ATR-FTIR and SEM-EDX results revealed the successful coatings of r-GO and zinc borate onto the surfaces of CF/PPS plates. Contact Angle and surface roughness analyses approved that CF/PPS/r-GO and CF/PPS/r-GO/zinc borate surfaces have hydrophilic properties with high surface roughnesses. DSC results indicated decreases in Tg, Tm, and ΔHm values after coating of CF/PPS surfaces with r−GO and r−GO/zinc borate dispersions. However, decomposition temperature of CF/PPS improved by coating according to the TGA thermograms. Significant improvement was obtained for r-GO/zinc borate co- coating, as targeted. Similarly, LOI value of CF/PPS increased from 59.2 ± 0.25% to 61.2 ± 0.25% by coating after r-GO and to 61.8 ± 0.25% after co-coating with r-GO/zinc borate. These results are higher than the LOI values of the materials already in use in the fuselage structures of the airplanes. Additionally, tensile and bending tests showed that the mechanical strength of CF/PPS was slightly decreased after r-GO and r-GO/zinc borate coatings which may be attributed to the etching process to create roughness on the surfaces of plates for the sequential processes. However, addition of zinc borate provided distinctive recoveries in mechanical properties. As a result of the studies carried out, it is suggested that less damaged and homogeneously coated surfaces may be obtained and enhanced flame resistance and durable mechanical strengths can be achieved by using CF/PPS/r-GO/zinc borate hybrid composite plates.
Footnotes
Acknowledgements
Authors acknowledge the support of Turkish Aerospace Industries (TUSAŞ) of Ankara. Authors also thank to Prof. Dr Fatih Üstel from Sakarya University of Türkiye for ATR-FTIR and SEM-EDX measurements and to Boron Institute of Türkiye for LIO and UL94 measurements.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Turkish Aerospace Industries (TUSAŞ).
