Abstract
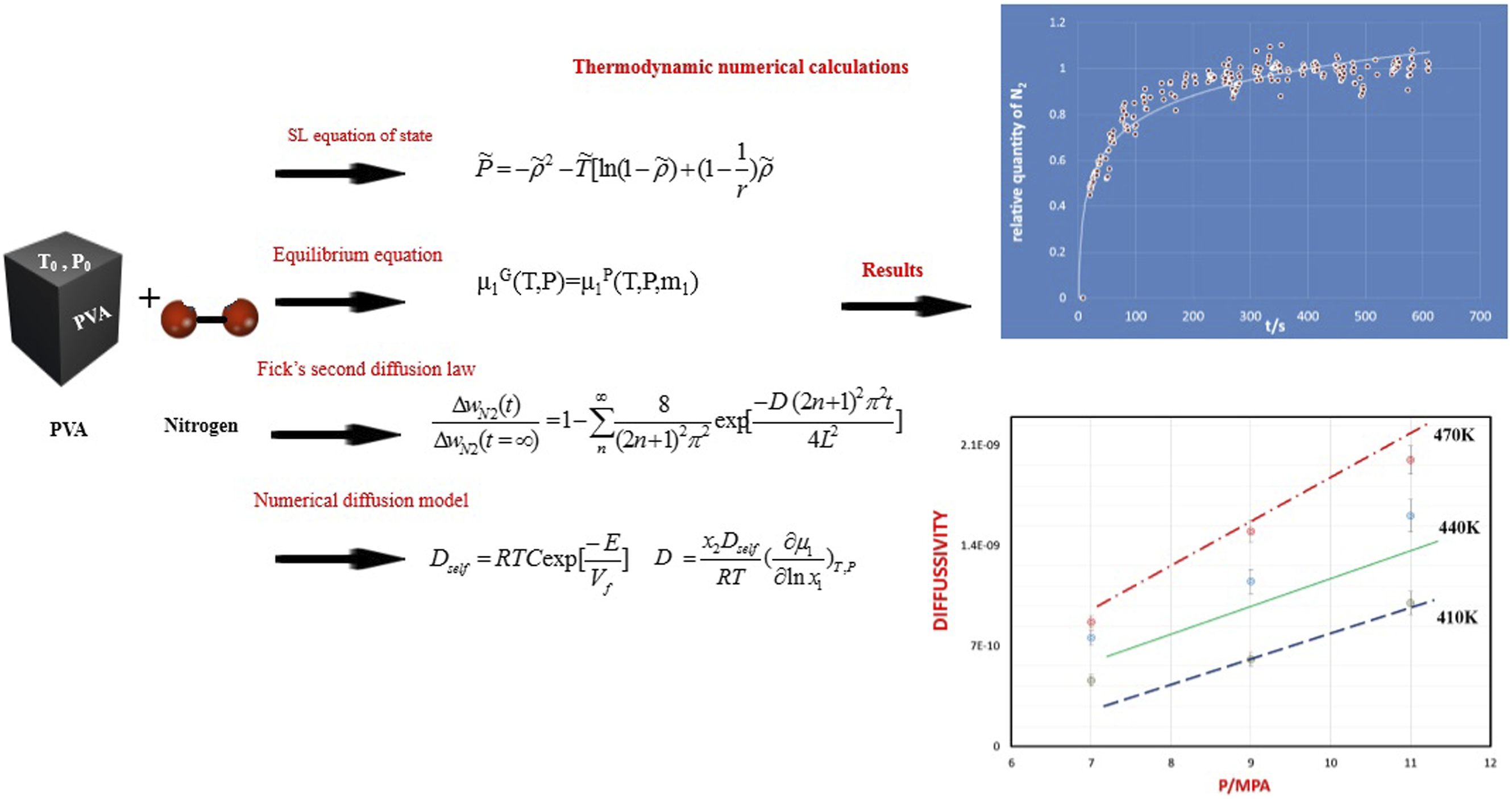
In this work, we recommended a numerical model in which the diffusivity of nitrogen in poly (vinyl alcohol) (PVA) could be determined from the specific volume of polymer/gas system. In this study, PVA with applicable properties was used as polymer matrix and supercritical nitrogen (N2), was selected as a gas or blowing agent. Firstly, the solubility and diffusivity of nitrogen into PVA were calculated via a magnetic suspension balance (MSB) system. The effects revealed that the thermodynamic parameters like solubility and diffusivity of nitrogen improved with an increase in pressure. Furthermore a diffusion model was proposed, considering the thermodynamic aspects of PVA/N2 system. It has been proven that there was an appropriate overlapping among the experimental and expected amounts from the model. Also it was shown that the model effectively predict the diffusion coefficient of nitrogen in PVA whenever the chemical potential derivative and the absolute specific free volume of N2 in PVA were known.
Keywords
Introduction
Poly (vinyl alcohol) (PVA) is a biopolymer that have desirable residences, including polarity, physical and appropriate mechanical properties.1–3 Different high pressure gases such as supercritical carbon dioxide or nitrogen are used in the production of polymer foams. 4 So that in the first step, the polymer is under high pressure of the desired gas and then the required saturation time is given. Finally, the foaming process starts with a rapid pressure drop.4,5 Therefore, it is very important to study how the desired gas is dissolved and diffuse in the polymer. As well, to calculate the modified solubility and the gas diffusion coefficient in the polymer, one equation of state such and the Fick equation are used, respectively.5,6 Also, by considering the gas diffusivity data in the polymer, the free volume of the polymer/gas system and the movement of the polymer chains, the gas diffusivity in the polymer could be modeled. In this way, various studies on measuring the solubility and diffusivity of different gases in various polymers have been investigated.7–9 Solubility and diffusivity of gas in polymers are main parameters in foaming of polymers. 8 A few models have been suggested to determine the diffusivity of gases in polymers.10,11 For instance, Kiran et al. reviewed literature records of the solubility and diffusivity of CO2 and N2 in different polymers like poly (methyl methacrylate), polystyrene, and polyolefin that the Sanchez-Lacombe equation of stat and the Fick’s law have been used for solubility and diffusivity measurements. 12 Also the sorption kinetics or desorption techniques was estimated. 12 Li et al., investigate thermodynamic parameters of CO2 and N2 and polyurethane thinking about the results on cellular nucleation of in batch foaming. 13 In another work, Wong et al, considered the interaction of CO2 and N2 in polystyrene foaming which the foaming mechanisms become regarded via referring to the foaming results with solubility information. 14 The ideal CO2–N2 composition to reap an most beneficial foaming effects was determined. Sebők et al., studied on the thermodynamic factors of N2, Ar, H2 and CO2 with poly Tetra fluoro ethylene considering various parameters. 15 Recently, for the experimental measurement of solubility and diffusivity data, the Magnetic Suspension balance (MSB) system has been used, and its working methods are given in different works.16–18 The MSB consists of a figuring out chamber and a stability wherein the balance is located out of chamber. An excessive pressure and temperature was set in chamber. The majority of the models evolved so far have proposed as an exponential feature of the free volume. 8 For apply the models to estimate the gas diffusion in a polymer, firstly the solubility and diffusivity of nitrogen in PVA and specific free volume of the polymer/gas mixture should be considered. In the case of PVA/gas structures, we studied the alternative factors of the PVA/gas system in our preceding work 18 wherein the bubble nucleation and growth of supercritical CO2 in PVA become experimentally and numerically investigated. We are going to investigate the diffusivity of gas into PVA which the selected gas for this work was N2. This work will be useful for any study about the foaming process of PVA/gas mixtures. Firstly, the solubility and diffusivity of N2 in PVA was calculated at different temperatures and pressures using the MSB system. For prediction of diffusivity, we used a numerical model that the diffusivity of nitrogen in PVA can be predicted from the chemical potential of N2 in PVA and specific volume of PVA/N2 system.
Experimental
Material
The properties of used materials.

Experiments
The solubility and diffusivity information for nitrogen have been determined by using the MSB system under different temperatures and pressures.
19
Additionally, the PVT data had been calculated by means of an excessive-pressure device (GNOMIX, Inc., USA) with a certain temperature range and pressure as much as 40 MPa. For defining the specific volume of N2, 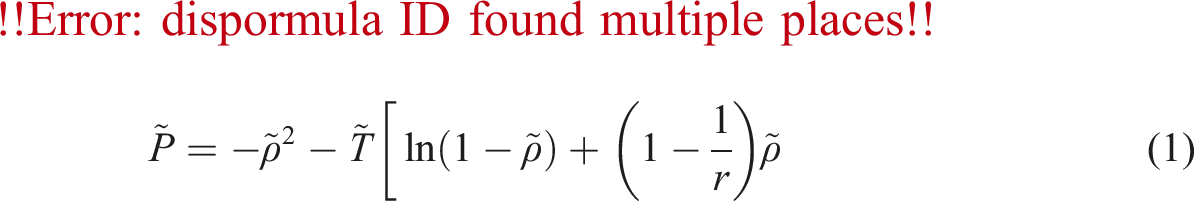
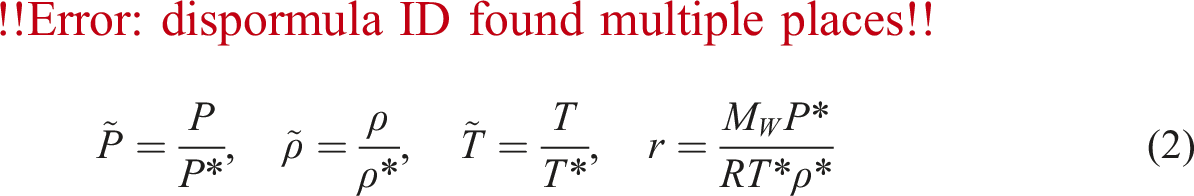
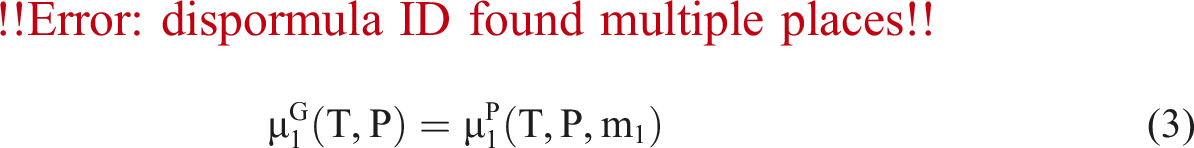

For diffusivity model of N2 into PVA, we used a numerical model. In this model it is estimated the mobility (M) of PVA and N2 could be calculated by the absolute specific free volume of the system and it is shown by20,21:
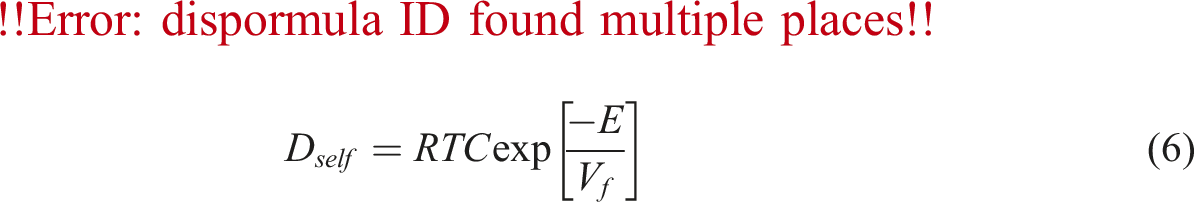
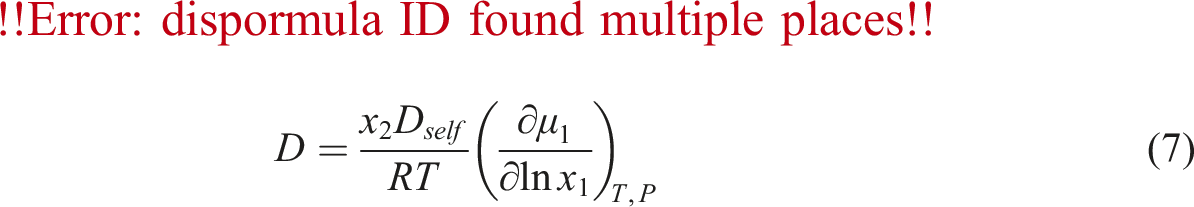
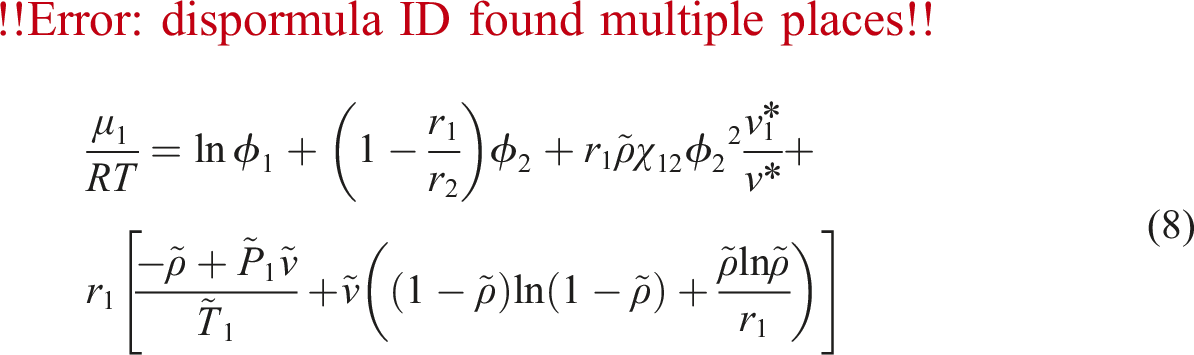
The specific volume at zero temperature, 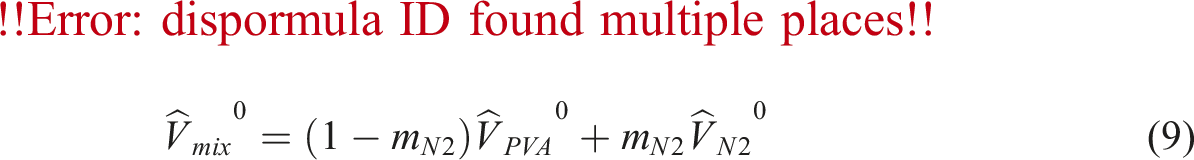
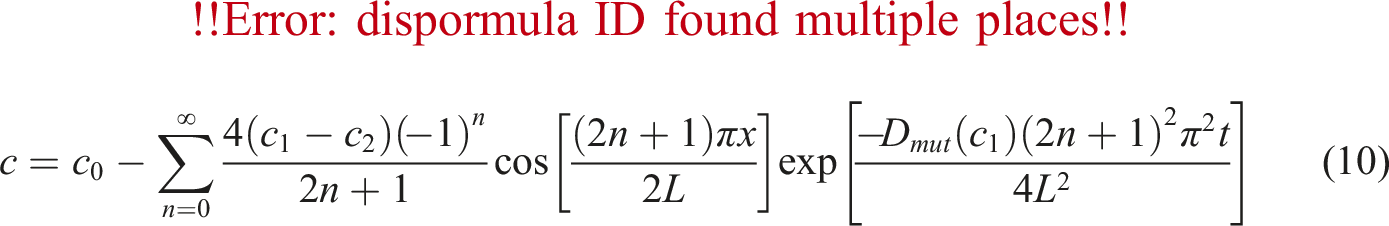
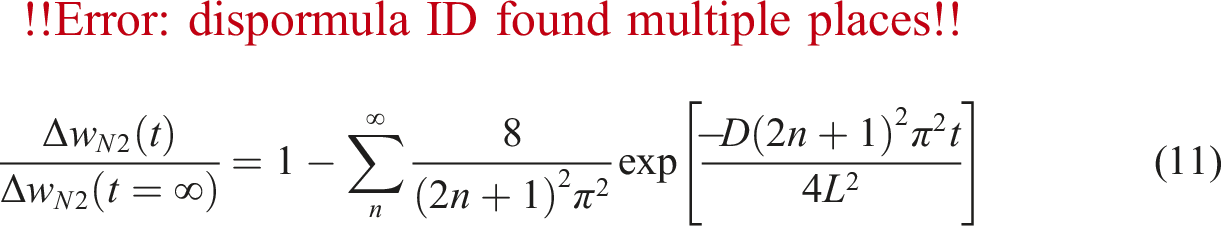
Results and discussion
The characteristic parameters of samples.

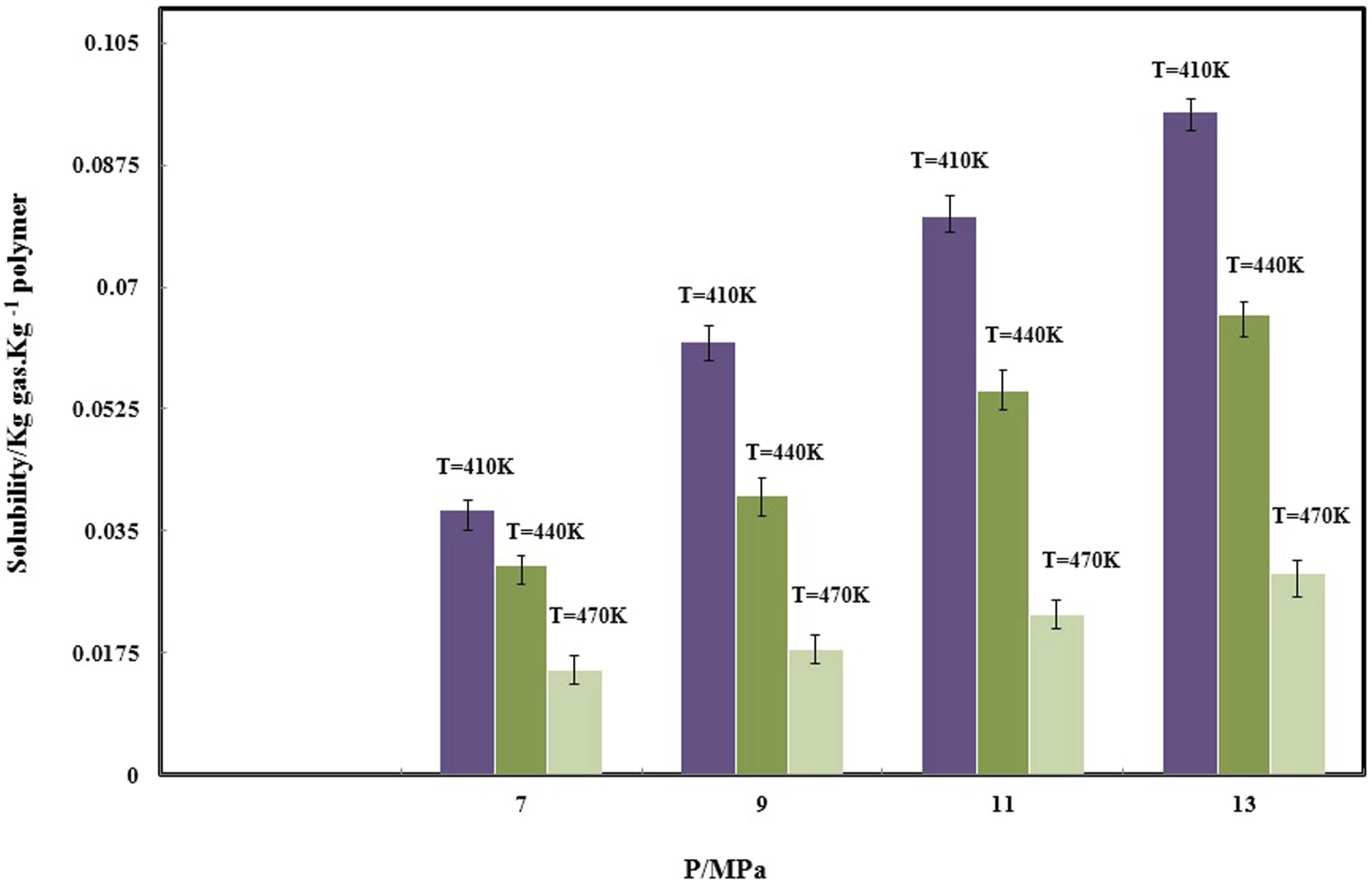
The N2 solubility in PVA.
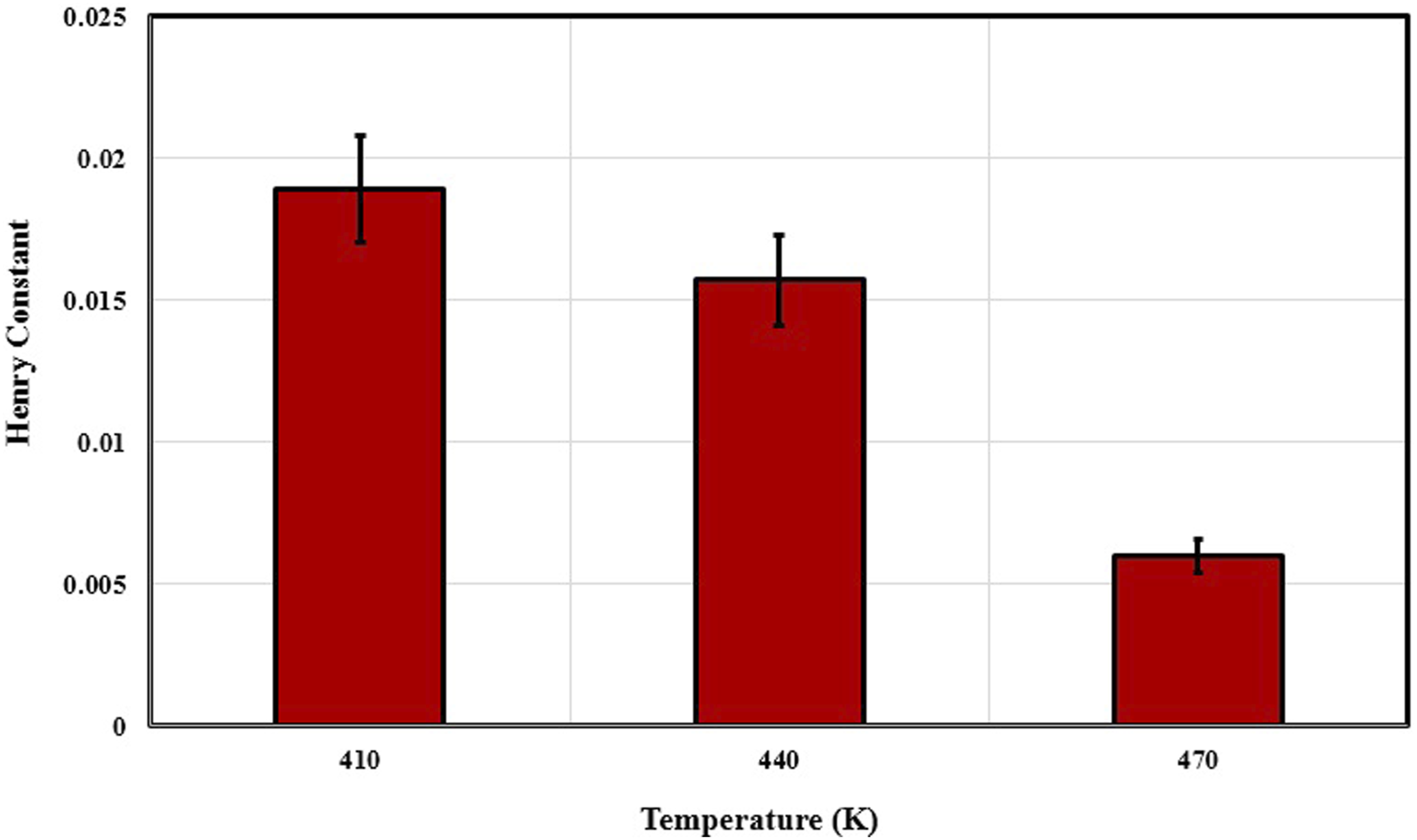
The calculated Henry constants for PVA/N2 system.
As mentioned before, there is free volume in the polymer/gas system, which will be the basis for calculating the gas diffusion coefficient in the polymer. First, this parameter should be calculated by the SL equation of state equation (9). The free volume depends on the amount of gas dissolved in the polymer and the temperature. It could be seen in Figure 3 that with the increasing of the of nitrogen percentage in PVA, the amount of free volume of the system increases. Also, with the temperature increment, due to the increase in the movements of polymer chains and nitrogen, the free volume has increased. Determined PVA/N2 specific free volume.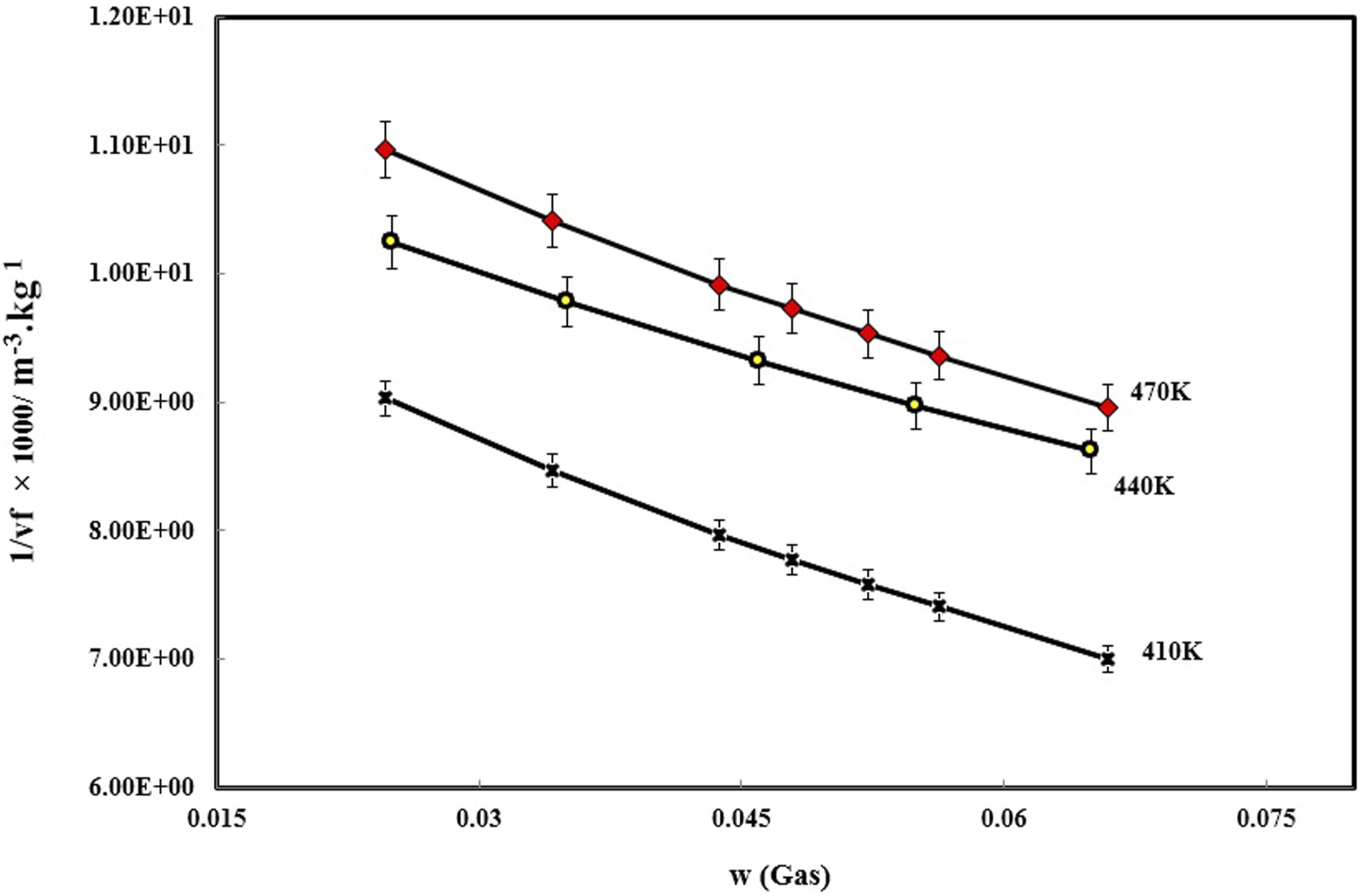
The relative quantity of N2 (R) in PVA with time is shown for PVA/N2 in Figure 4. The symbols are the experimental data and solid line shows the calculation results. Quantity of nitrogen (R) against time which the solid lines are predicted by equation (10).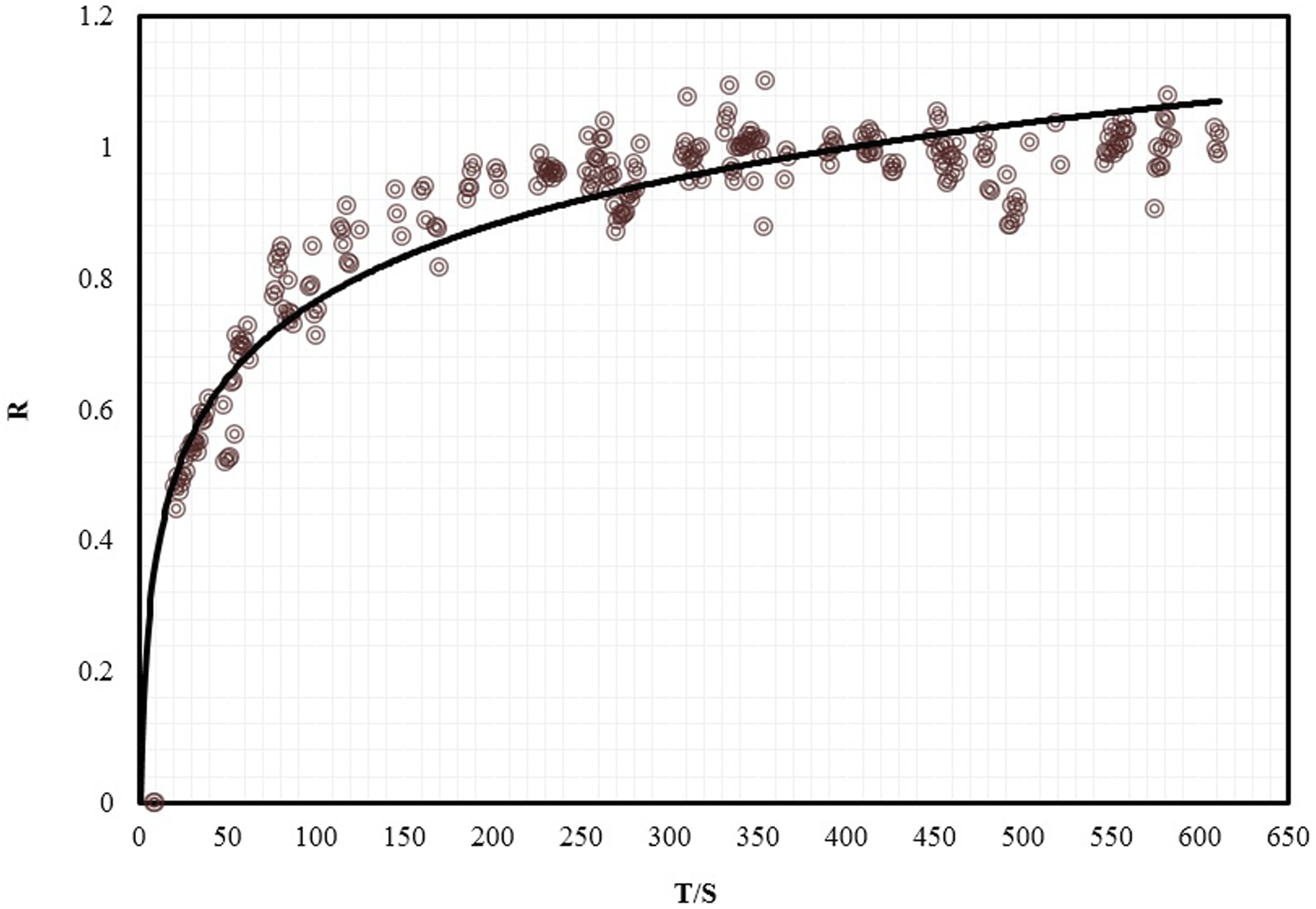
To calculate the diffusivity of gas in PVA at different temperature and pressure, using above Equations the factors C and E in equation (6) must be calculated. Firstly, N2 chemical potential and derivative were determined for PVA using equation (8) and SL equation. Then, the self-diffusivity of N2 for PVA was estimated by equation (7) from the calculated chemical potential derivative, weight fraction of polymer, calculated diffusion coefficient, and temperature. As well, the absolute specific free volumes of PVA/N2 was determined. The self-diffusion coefficients are plotted against the absolute specific free volume as shown in Figure 5. Self-diffusion coefficients of N2 (Dself) versus converse of the absolute specific free volume in pressure range 7 to 11 MPa (the dach line signifies predictions calculated by the numerical model).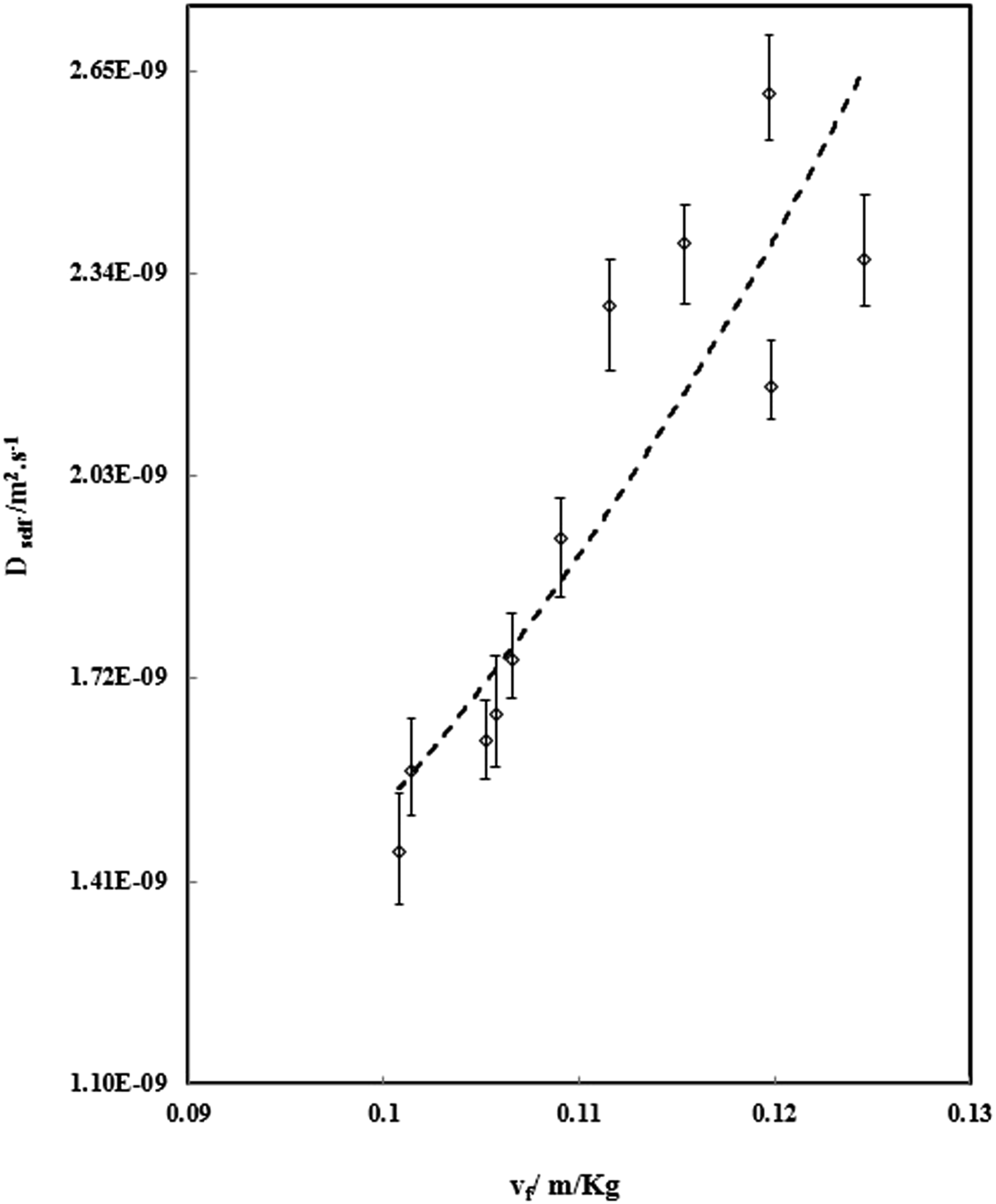
The factors C and E could be calculated by plotting of it to the curve of the N2 diffusivity in PVA determined at different temperature and pressure range, which the solid line characterizes the fitting plot of equation (6). Therefore, the diffusivity of N2 could be predicted whenever that the specific free volume of PVA/ N2 and the N2 chemical potential derivative were calculated. The results of diffusivity prediction are plotted in Figure 6. The symbols are the experimental data and solid lines signify the diffusivity predicted by equations (6) and (7). As it is shown, it is clear, there is a suitable covering between the experimental and predictable values, revealing the numerical model could effectively predict the diffusion coefficient of N2 in PVA at various conditions. In Figure 6 the diffusivity of N2 in PVA are improved with increasing of the temperature. Diffusivity of N2 in PVA, at various temperatures.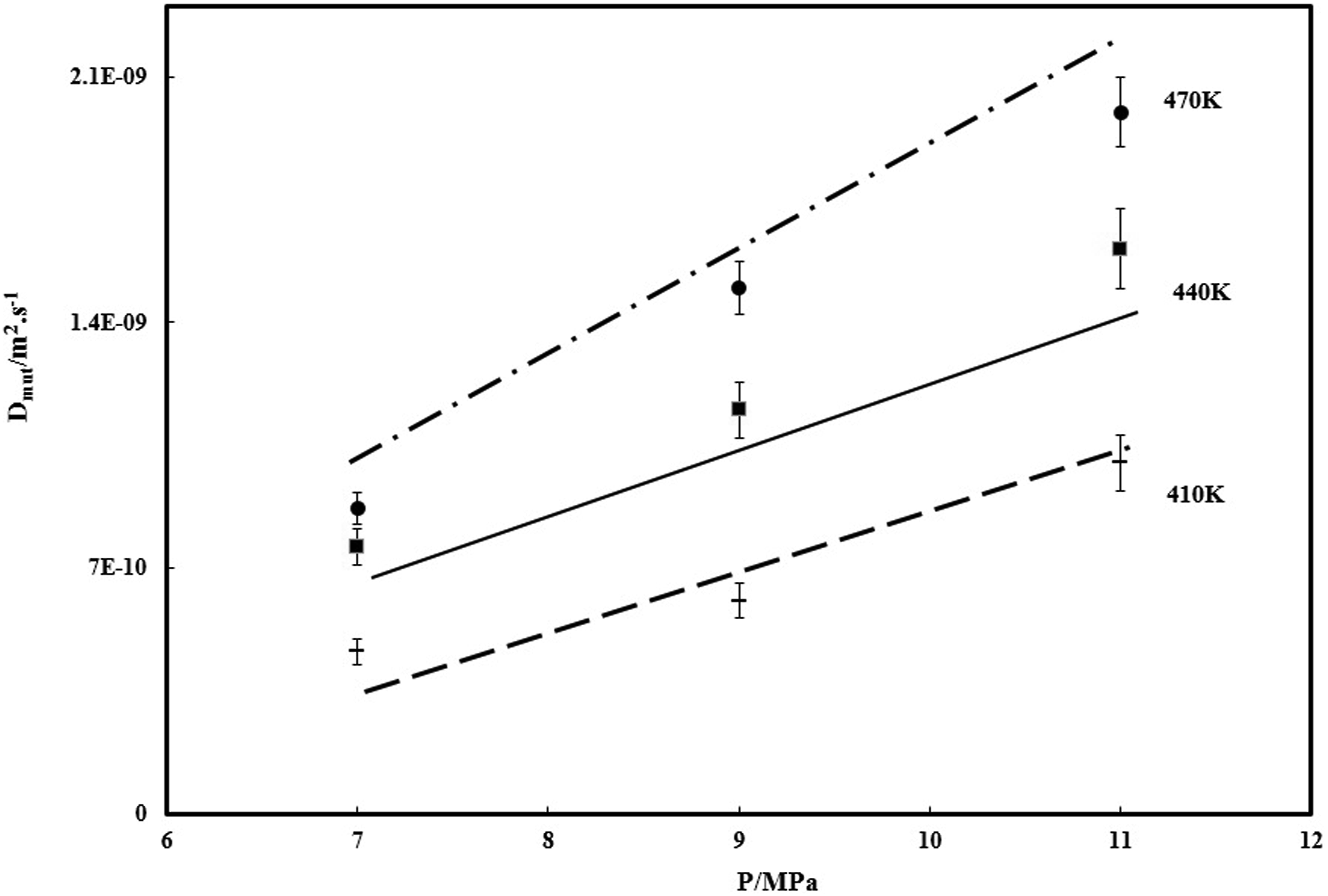
Conclusion
In this study, at the first the thermodynamic parameters of N2 in PVA were determined at certain temperatures and pressures using the MSB system. The solubility of N2 in PVA reduced with temperature increasing and improved with increasing of the pressure. The association between the diffusivity and the absolute specific free volume of sample was examined to develop a numerical model. We used numerical model to predict the N2 diffusivity in PVA. The model can estimate the diffusivity of N2 in PVA, when the absolute specific free volume and the chemical potential derivative of N2 were recognized.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has been financially supported by Azarbaijan Shahid Madani University under the grant number of 1603/1402.
