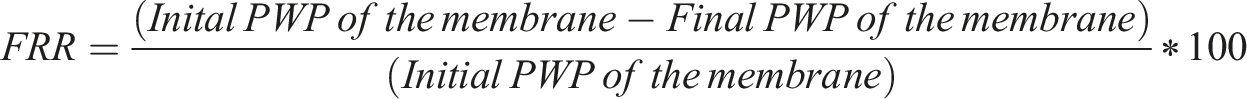Abstract
The growing global population contaminates the growing requirement for clean process water for Industries and sanitary pure water for drinking. Ultrafiltration is an established approach for producing water for domestic and industrial use, and it shows significant potential in eliminating pollutants from surface water. In the present work, the copper-chitosan layer was coated over a Polysulfone membrane with varying concentrations of copper in the top coating layer, and the membranes were characterized. Scanning electron micrographs demonstrated that the membrane’s top surface impact was modified by an appropriately dense layer of chitosan coating. Zeta potential examinations revealed that the membrane surface charge enhanced in absolute terms with chitosan covering and increased further with copper concentration. Removal of cationic dye was greater as compared to the removal of the anionic dye by the coated membranes based on the surface charge With increasing Copper concentration in the top coating layer, the increasing bactericidal properties against Gram-negative bacteria were observed when the membrane was treated with the bacterial solution. The highest concentration of copper loading in the membrane was associated with the lowest bacterial count (cfu/ml) after swabbing the membrane surface to evaluate the number of colony forming units attached to the membrane surface. Thus, the present paper demonstrates a novel way of forming an antimicrobial layer over the conventional ultrafiltration layer to achieve the desired separation of dye and antibacterial performance.
Practitioner points
• The manuscript addresses the need for an antimicrobial coating over ultrafiltration membrane • It demonstrates decreased bacterial adhesion over the modified membrane by this novel coating • Zeta potential increased in absolute terms from −31.37 mv to −9.63 mv demonstrating the positive charge imparted by the coating • Charge-based separation is possible as demonstrated by the near complete removal of Methylene blue dye from water by the modified membrane
Introduction
Water scarcity has become a serious worldwide issue, prompting the development of new and creative technologies for mitigating the water demand. 1 While the conventional treatment technologies for example filtration, sedimentation, flocculation, coagulation, activated carbon (charcoal) adsorption, and biologically active sludge technology are used in water/wastewater treatment, the membrane based technologies have very significant role both in water and wastewater treatment. 2 Membrane-based desalination provides the water essential for any process plant and thus became a key to process plant sustainability to mitigate the water demand in any area, particularly coastal areas. 3
When it comes to wastewater treatment, membrane-based systems offer many advantages for example high efficiency, smaller footprint, cost-effectiveness, no phase change, lower energy requirements, flexibility in combining with other processes, and excellent selectivity. 4 There are a few crucial requirements that must be satisfied for a membrane process such as high quality with high throughput and stable flux, lower fouling and low-pressure operation, and avoiding a long elaborate pre-treatment. Dependable pollutant removal capability without producing any hazardous by-products will be important in membrane applications for wastewater treatment. 3 The ramification of using membrane is an unwanted accumulation of colloids, salts, macromolecules, and residual particles on the membrane surface causing fouling. 5 Fouling is a common issue with polymeric membranes, which has a direct impact on their performance such as a reduction in water flow, increased passage of the compound to be removed, increased energy consumption, and operating expenses. 6 The fouling results in pore blockage, layer formation, and interaction with the membrane top surface leading to extreme depletion in membrane flow and increase in energy requirements for filtration. 2
Biofouling is a particularly significant problem because it severely limits the use of membrane processes. 7 It has been also known as the unwelcome growth of microorganisms (mostly bacteria) and the formation of biofilm, which is a significant barrier to membrane separation and the application of membrane processes in biotechnology related applications. 8 There are many ways of water disinfection already implemented, including chlorination, ozonation, and photochemical inactivation utilizing ultraviolet (UV) light in pre-treatment. 5 The antibacterial activity of the membrane is influenced by many factors for example roughness of the surface, surface charge of membrane and pore diameter, and presence of anti-bacterial material on top layer.3,9
While ceramic and polymer based membranes are being used; the ceramic membranes offer the advantages of chemical resistance and lower feed water temperature sensitivity, the polymeric membranes have better processability, better selectivity and productivity.10,11 Polysulfone is widely used as a membrane material because of its chemical stability, mechanical strength, and flexibility, which makes it a popular choice for ultrafiltration and nano filtration applications.10,12,13 In the last few years, many agents like copper oxide, zinc oxide, titanium dioxide, graphene oxide, carbon nanotubes, hallo site nanotubes, etc. Have emerged as the candidates to make the mixed matrix membrane for anti-biofouling applications. 5
Contact angle is the necessary test by which determination of the flowrate and surface nature of the membrane can be evaluated so as contact angle is high that means the membrane is hydrophobic in the nature which represents less flow and it means the membrane is hydrophilic which means that flow through the membrane will be high. 14
Tannic acid is reported to inhibit the adhesion and colonization of microbes and may be used for surface coating of the metal-phenol complex for antibacterial surface coating.15,16 Metal-Organic frame work is attracting researchers towards it because of the new formulation of the metals and organics by this combination we can categorized the new type of material which can give us the high surface area and active material. 17 Copper is used in various places like effective urea electrocatalytic oxidation where copper is metal-organic framework is done by utilizing tetracaroxilic acid ligand that incorporates –NH2 group. 18 In another paper authors have noted that Cu doped CoS2 NNs material is very useful in the hydrogen evolution reaction because this material exhibit ultralow over potential and long-term stability during the process. 19
Chitin, a modified polysaccharide containing nitrogen, and chitosan, a polymer specifically mentioned as having strong bacteriostasis, are other polysaccharides that are isolated from Dictyophora indusiata (DIP). 20 It is possible to naturally extract Chitosan and chitin from crustacean shells, lobster, and crwfish chemicall by demineralizing and deproteinizing the shells using acid and base or biologically by employing microorganism or a proteolytic enzyme. Chitosan and chitin are broken down into tiny powders once it has been isolated and purified and then used in industrial, biological, and Research Area. 21 Research on chitosan, a byproduct of chitin, has been quite interesting. Since chitosan contains amino and hydroxyl functional groups, it has outstanding biological features like good adhesion, coating ability, high water permeability, nontoxicity, and biocompatibility.7,12 Numerous research have successfully used chitosan to remove metals and dyes from the pollutants. Because they are nontoxic and biocompatible with mammalian cells, hybrid copper sulfate, copper oxide, and chitosan have found extensive application in the medical, research, and food industries. Additionally, well-known substances with antibacterial qualities similar to copper metallic include copper oxide and copper sulfate.22,23 The copper atom helps to prevent the bacterial development. Bacterial cells undergo oxidative stress due to damage from copper atoms to their cell wall, and DNA and RNA structures. 24 As a result hydrogen peroxide is released from that and bacteria are been killed. Chitosan-copper coated ultrafiltration membranes with crosslinking demonstrated good performance for the removal of Arsenate and emerging contaminants when operated in Micellar-enhanced ultrafiltration mode.
Copper has several antibacterial qualities compared to other metals like gold, silver, and iron because it releases ions quickly. These ions assist in breaking the cell walls of microbes and degrading their RNA structure, which can ultimately result in the microorganism’s destruction. 25 In addition to being genotoxic to microorganisms, copper can also help in their demise. This copper metal may also disrupt the enzyme structure of the bacteria and therefore due to this bacteria start dying. Copper metal is itself toxic for the bacteria so that’s why when it comes in the contact with the bacteria it will create oxidative stress which disrupts its cell wall and the bacteria is killed.
For the measuring antibacterial nature of the membrane different methods are been used bacterial adhesion method, petri dish diffusion method, etc. Petri dish diffusion method is done with the membrane circle pattern in the plate of the bacteria and zone of the membrane will be created this shows the antibacterial nature of the membrane. 26
Food and water contamination by faecal bacteria is still a prevalent issue that has an effect on local and national economies as well as public health. 27 Escherichia coli (E. coli) is a rod-shaped, gram negative, gamma proteobacterium in the Enterobacteriaceae family of bacteria that is a membrane of the fecal coliform group. In warm-blooded animals like humans, the lower intestine is considered to be E.coli’s preferred residence. 28 Since consuming any one of the E.coli serotypes can cause illness, this organism is the focus of intensive study and monitoring efforts.18,19 Incorporation of nanomaterial such as Boron nitride, and copper nitride may trigger temperature responsive behavior in permeate flux of the membrane. 10 Biofouling of such nanocomposite membranes need to be investigated.
The present work addresses the research gap in forming a robust anti-microbial layer based on biomaterial over a membrane that can facilitate the charge based separation. The present study demonstrates that the copper-chitosan based antibacterial coating can significantly improve the antibacterial nature of the membrane and demonstrate very high rejection of cationic dye and thus may find applications in dye wastewater treatment.
Materials and methods
Materials
Polysulfone pellets (Solvay), Nonwoven technical fabric- Polyester (Nordlys TS-100), Dimethyl formamide (Central Drug House (P) Ltd), Copper Sulfate (Sisco Research Laboratory Pvt. Limited), Chitosan (150-500 mPa-S, 90% DA, Sisco Research Laboratory Pvt. Limited), Sodium hydroxide (Sisco Research Laboratory Pvt. Limited) Nutrient agar (Hi-Media), Nutrient broth (Hi-Media), cotton swabs (Hi-Media), 0.9% saline, Methylene blue (Sisco Research Laboratory Pvt. Limited), Methyl orange dye (Nice chemicals Pvt. Limited), Polyethylene glycol and Polyvinyl alcohol (TCI Chemicals (India) Pvt. limited), Egg Albumin (Sisco Research laboratory Pvt. Limited)
Methods
Membrane preparation
Polysulfone (PSF) pellets were dried in an oven at the temperature of 70°C until the moisture was completely removed from the pellets. N, N-Dimethylormamide (DMF) was taken in a flask and pre-heated up to 60°C for 1 hour, thereafter; Polysulfone pellets were added slowly until they dissolved in DMF to prepare 15% (w/v) solution. The polyester non-woven technical fabric was rolled over the rollers of the membrane casting machine and the solution was spreaded over the fabric and thickness was controlled by a precision knife placed over the roller with a desired gap. The linear speed of rollers was maintained at 2 m/minute during casting and the membrane was prepared by phase inversion with the non-solvent deionized water in a gelation bath. The prepared membrane was washed thoroughly with deionized water and was preserved in deionized water for further experiments.
The chitosan solution (1%) was prepared by dissolving chitosan in 1% glacial acetic acid solution. The solution was stirred at 50°C until the chitosan was dissolved fully and the solution became clear. This solution was used to prepare the copper-chitosan complex by dissolving 0.1%, 0.2%, 0.3%, and 0.4% CuSO4.5H2O with stirring at the constant speed.
The Polysulfone membrane was carefully stuck over the clean glass plate and all the sides were sealed with adhesive tape. Thereafter, the prepared solutions that is 1% Chitosan solution, 1% Chitosan with 0.1% up to 0.4% CuSO4.5H2O were poured over the membrane surface to dip the membrane surface completely for 1 hour. Thereafter, the solution was decanted off and the membrane was cured in an oven at 70°C until the membrane became dry completely. The dried membranes were taken out and dipped in 5N NaOH solution for 1 hr to coat the chitosan over the membrane and remove the excess material. The prepared membranes were rinsed with deionized water to remove NaOH and then preserved in deionized water for further experiments/analyses.
Biofouling
Membranes (PSF membrane, 1% chitosan, different doping of copper ranging from 0.1% to 0.4% in chitosan) of the size 2.5 cm × 2.5 cm were taken for biofouling study. Each membrane was autoclaved at a temperature of 121°C and for 15 minutes for sterilization purpose. On the other side, Escherichia coli (E-coli) was inoculated in nutrient broth taking 13 g nutrient broth in 1000 mL deionized water and the solution was sterilized at 121°C for 15 minutes and allowed to cool to 25°C. Further, E.coli bacteria was added into it and put in a shaker incubator at the temperature of 37°C and 120 r/min for 12 hours. Following a 12-h growth period, the culture was centrifuged at 1000 r/min for 10 minutes at 25°C. The supernatant from the centrifuge was disposed of, and the biomass pellet was rinsed with 0.9% saline and then re-suspended in 0.9% saline solution. Membranes were placed over the petri dish in such a way that its active side remained on top. Then, broth inoculated with E.coli was poured over the active side of the membrane and the initial sample was collected for measuring CFU/ml at zero time. After 24 hours, 48, and 72 hours, the bacterial load was counted in CFU/mL (Colony Forming Unit/ mL). To count the number of bacteria adhered over the membrane surface; it was rinsed with sterile distilled water to remove any loosely attached bacteria. Thereafter, the bacteria attached to the membrane surface were removed from the membrane using a cotton swab. The cotton swab was then dipped in 0.9% saline solution, vortexed, and finally, 100 µl of this solution was spread out on nutrient agar plates. The plate was then incubated for an overnight period at 37°C. Thereafter, the Colony forming units per ml (CFU/mL) were counted.
Flux recovery ratio (FRR)
The flux recovery ratio approach has been widely utilized for studying membrane fouling. For the study of the membrane fouling pure water permeation data of each membrane is required. Take the initial pure water permeance of each membrane at 50 PSI before fouling it. To make the membrane foul a 3000 mg/L solution of egg albumin is used. Membranes are placed inside the testing kit, and using a 3000 mg/L egg albumin solution, the membrane testing kit is started at 150 PSI for 6 hours. A sample of the volume obtained through membrane permeate is taken each 30 minutes. Membranes need to be gently cleaned after 6 hours, and then a final Pure Water Permeance test at 50 PSI needs to be performed. Once the membrane’s initial and final PWP has been noted, calculate the flux recovery ratio using the formula below.
Characterization
The prepared membranes were characterized for knowing the top surface morphology by Field emission-scanning electron microscope (FE-SEM, JEOL JSM 7100F). The membranes were analyzed by Atomic force microscope (Model no. NTEGRA AURA) to understand the surface roughness and associated features.
The prepared membranes were analyzed for pure water permeance by testing them in a cross-flow testing kit. Membrane coupons were placed in the kit and deionized water was used for testing. The compaction was done at 5.17 bar pressure for 45 minutes and thereafter, the permeate was collected at 3.45 bar pressure. The average value of the permeate collected per hour divided by membrane surface area is pure water flux. It was divided by applied pressure to get the permeance in LMH/bar.
Membranes were characterized for the surface charge by zeta potential measurement using a zeta potential measuring instrument (CAD instruments ZETACAD).
Dye removal study
The prepared membranes were taken for anionic and cationic dye removal. Methylene blue was chosen as cationic dye and Methyl orange was chosen as anionic dye for the study. The solution concentration was 100 ppm in each case. The membranes were placed in a cross-flow membrane testing kit and 100 mg/L dye solution was filtered through prepared membranes at 10.342 bar pressure. The permeate was collected and the concentration was measured by UV-VIS spectrophotometer (UV-2700 SHIMADZU) analyses. The absorbance of methylene blue and methyl orange was recorded at 665 nm and 464 nm respectively.
Results and discussion
Bacterial adhesion on the membranes
CFU/mL of adhered bacteria to membrane at different time intervals.
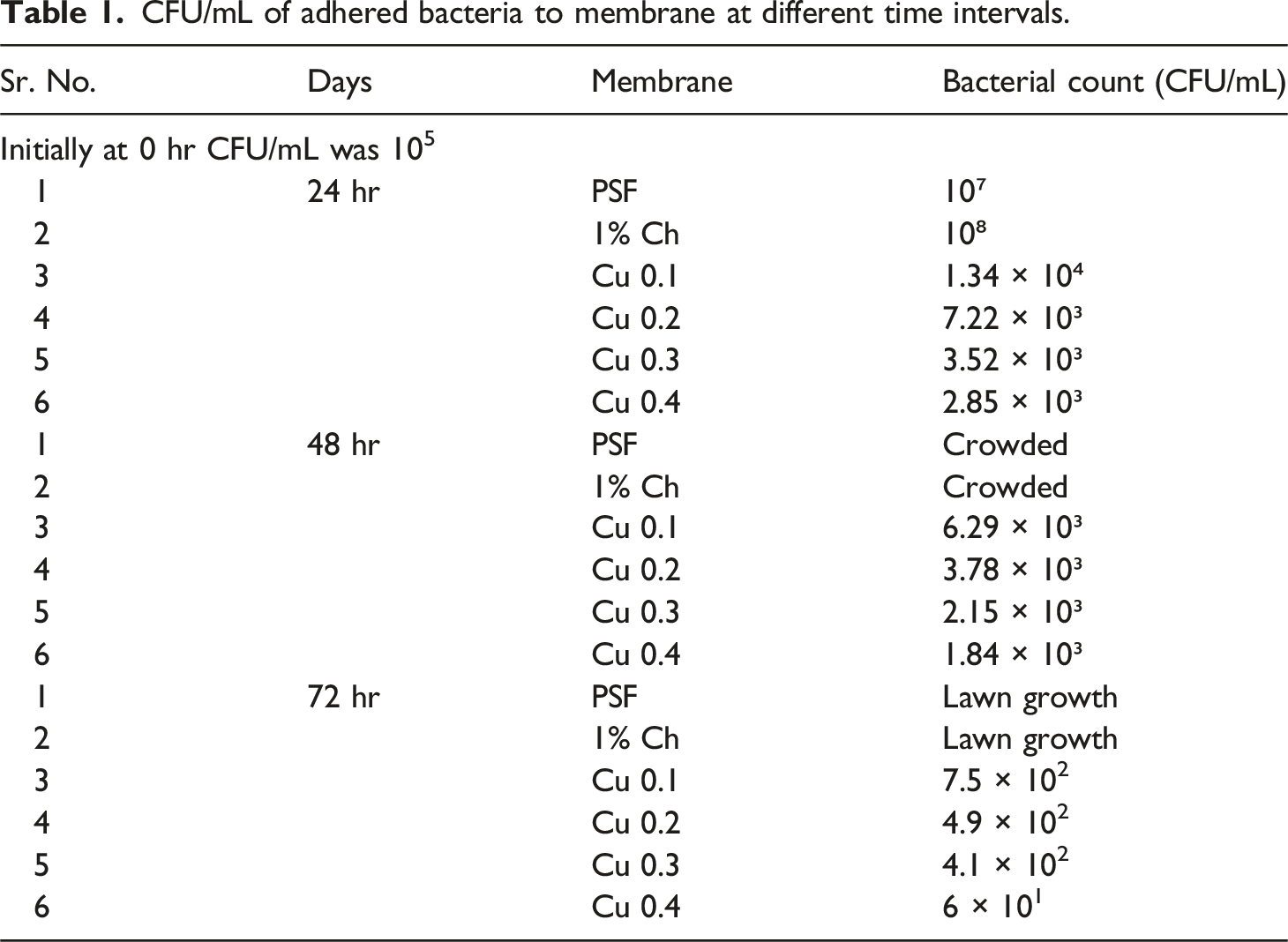
As can be seen in the above table, the initial CFU/mL of the culture was 105. After applying the culture to six different membrane samples, the CFU/mL was calculated after 24 hours, and it is evident that the bacterial colonies on the PSF Membrane and 1% chitosan have increased compared to the CFU/mL of the initial culture, and in the case of copper sulfate loading, the CFU/mL (Bacterial Count) has decreased in the first 24 hours. Due to the overgrowth of the bacterial colony, PSF and 1% chitosan became crowded after 48 hours as can be seen from Table 1. As the doped copper percentage increases, 0.1% to 0.4%, bacterial concentration in the solution as measured by CFU/mL decreased, while comparing the same period of exposure in each case in comparison to the 24 hours, and 72 hours yields a lower bacterial count than the 48 hours. The greatest amount of bacteria was killed by 0.4% copper sulfate; at 0 hours, it was 105, and after 72 hours, it dropped to just 60.
Bacterial sticking on petri plate
Figure 1 demonstrate how the bacteria’s culture fluid is poured onto the membrane’s surface and how, over time, the membrane becomes more and more congested. However, when copper sulfate loading rises, it exhibits strong antibacterial action, and throughout 24 to 72 hours, there are fewer and fewer bacteria on the membrane’s surface. As illustrated in Figure 1(r), 0.4% CuSO4 has good antimicrobial activity when compared to all other membranes. After 72 hours, the final CFU/mL is only 60, and the colony counts are quite low. CFU/mL of adhered bacteria to the membrane at 24 Hr, 48 Hr, 72 Hr. (a)-24 Hr PSF, (b)-48 Hr PSF, (b)-72 Hr PSF, (d)-24 Hr 1% Chitosan, (e)-48 Hr 1% Chitosan, (f)-72 Hr 1% Chitosan, (g)-24 Hr 0.1% CuSO4, (h)-48 Hr 0.1% CuSO4, (i)-72 Hr 0.1% CuSO4, (j)-24 Hr 0.2% CuSO4, (k)-48 Hr 0.2% CuSO4, (l)-72 Hr 0.2% CuSO4, (m)-24 Hr 0.3% CuSO4, (n)-48 Hr 0.3% CuSO4, (o)-72 Hr 0.3% CuSO4, (p)-24 Hr 0.4% CuSO4, (q)-48 Hr 0.4% CuSO4, (r)-72 0.4% CuSO4.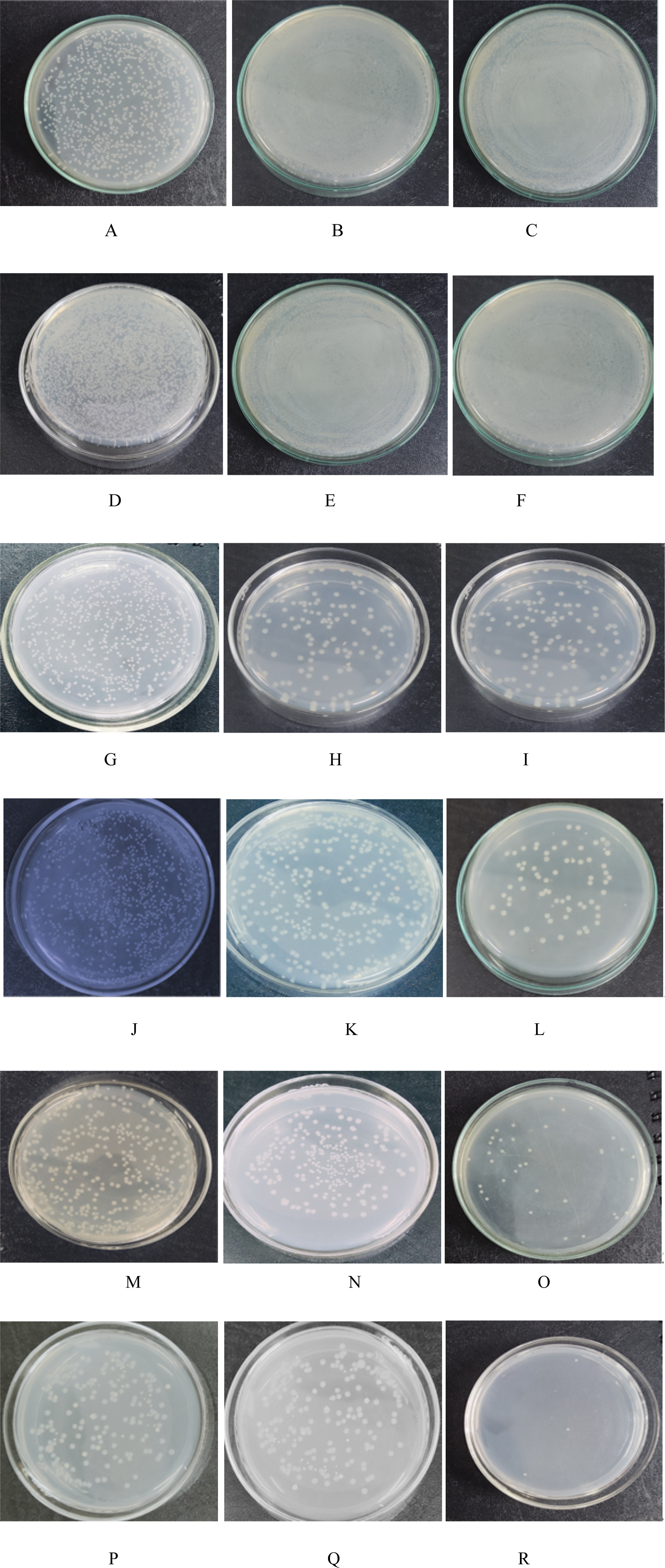
Scanning electron micrography of membranes
Figure 2 shows the scanning electron micrographs of the different membranes. SEM Images of normal and biofouled membrane. (a) PSF membrane, (b) PSF membrane Biofouled, (c)- 0.4% CuSO4, (d)- 0.4% CuSO4 Biofouled.
The top surface morphology of different membrane surfaces is depicted by FE-SEM images as shown in Figure 2(a) through Figure 2(d). As can be seen from Figure 2(a), the pores on the top surface are visible in the Polysulfone membrane, which are not visible when it is coated with the 0.4% CuSO4 as shown in Figure 2(c). The reason for spotting the debris of microorganism in copper-doped membranes is the dead micro-organism over the surface. The zeta potential value of the membrane increases in absolute terms with increasing the copper doping. Since, the bacteria are gram negative with a highly negative charge on the surface, when the membrane surface charge (zeta potential) increases in absolute terms, the bacteria approaching the membrane are killed over the membrane surface because of the presence of copper on the membrane surface. These findings are consistent with the decline in CFU/ml of the solution as shown in Table 1.
Atomic force microscope imaging
Atomic force microscope images have been shown in Figure 3. AFM Inmages of Normal and Biofouled membrane. (a) PSF membrane, (b) PSF membrane Biofouled, (c)- 0.4% CuSO4, (d)- 0.4% CuSO4 Biofouled.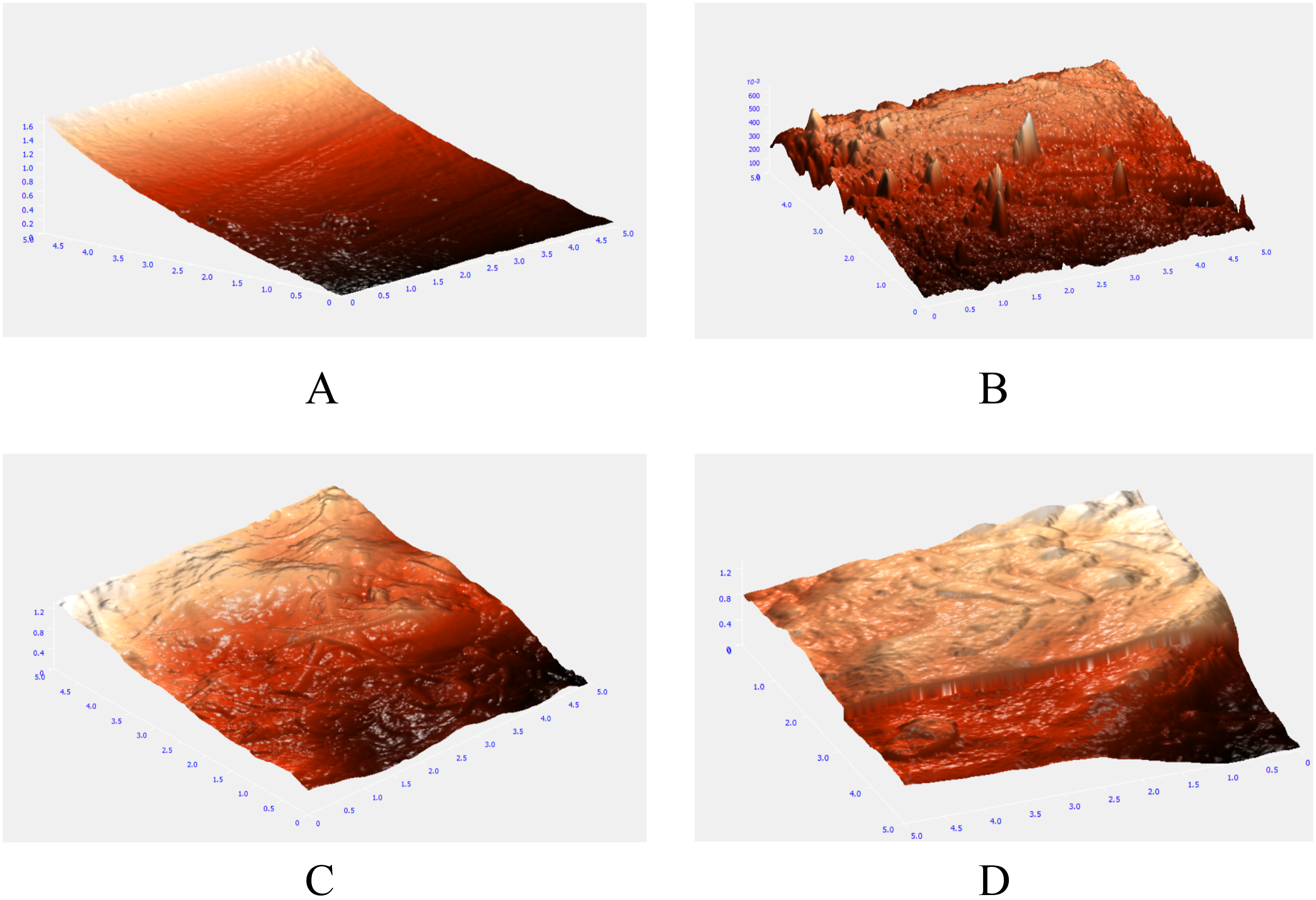
The AFM images show the top surface of the membrane with ridge and valley structure. As shown in Figure 3(b), biofouling of Polysulfone membrane has altered its top surface and at a few points the ridges have been formed. The 0.4% CuSO4 coated membrane has been shown in Figure 3(c), whose Biofouled version showed the changes in top surface features.
Study of membrane surface roughness is important for understanding the extent of biofouling. Because of fouling, the valleys from the ridge and valley structure of the membrane are filled and roughness decreases. The average roughness (in micron) for different copper-doped membranes and biofouled (BF) membranes are shown in Figure 4 Normal PSF membrane had high average roughness, as shown in Figure 4. When the normal PSF membrane was compared to biofouled PSF, the roughness decreased significantly from 295.4 nm to 88.66 nm. As can be seen from Figure 4, the roughness did not change much in case of 1% chitosan coated membrane. In case of 0.1% and 0.2% copper-doped membranes, the roughness did not decline after biofouling but increased slightly. This may be because of lower biofouling and debris of dead bacteria at few places. Moreover, there was a little change in roughness in case of 0.3% and 0.4% copper-doped membranes. In general, the change in roughness with biofouling was attenuated in case of copper-doped membranes, which can be attributed to lower adhesion of bacteria in these membranes (Figure 5). Average Roughness of different membranes. (b-d): XPS analysis of 1% Chitosan-coated membrane. (a). XPS Survey of 1% Chitosan, (b). C1s Scan of 1% Chitosan, (c). N1s Scan of 1% Chitosan, (d). O1s Scan of 1% Chitosan.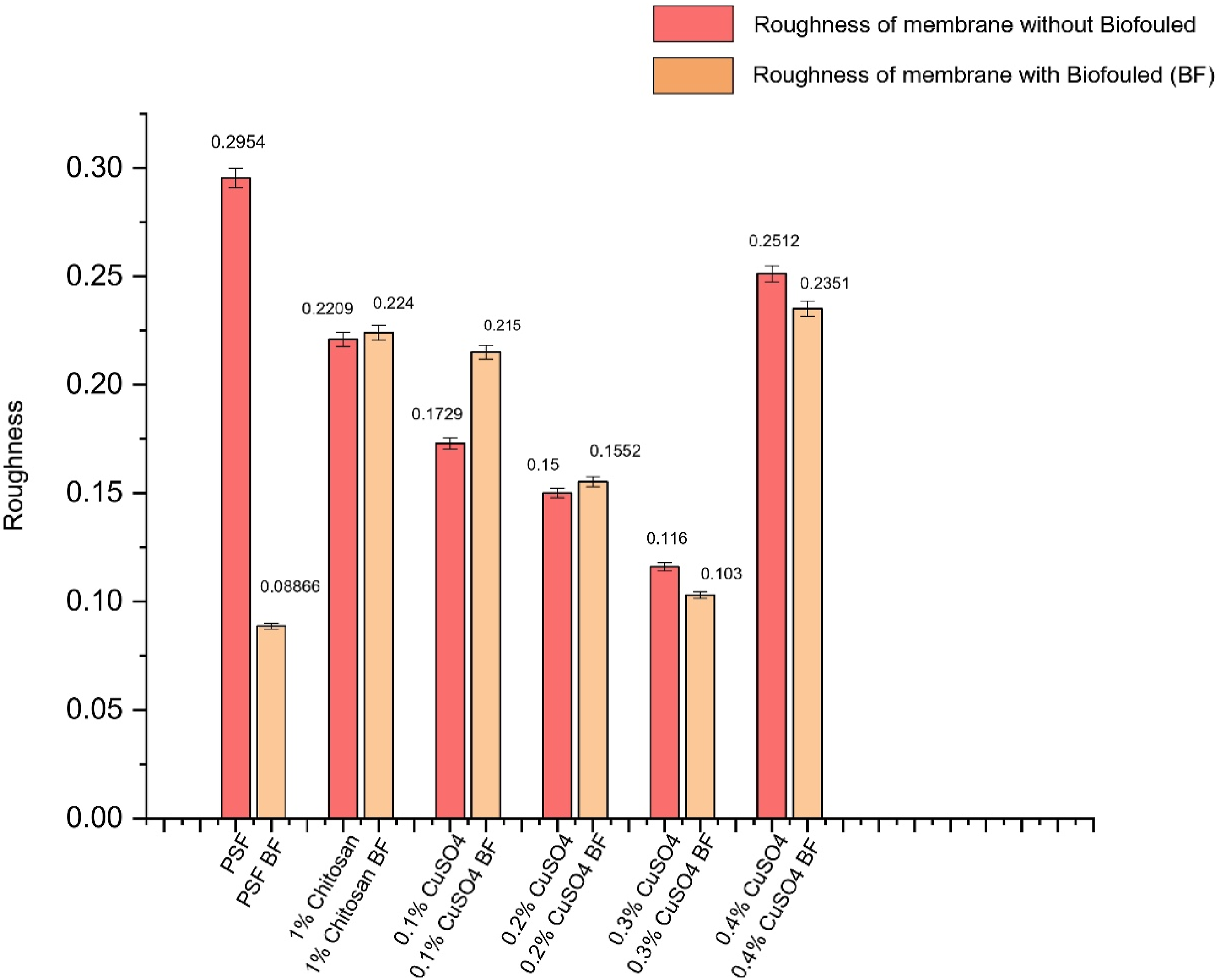
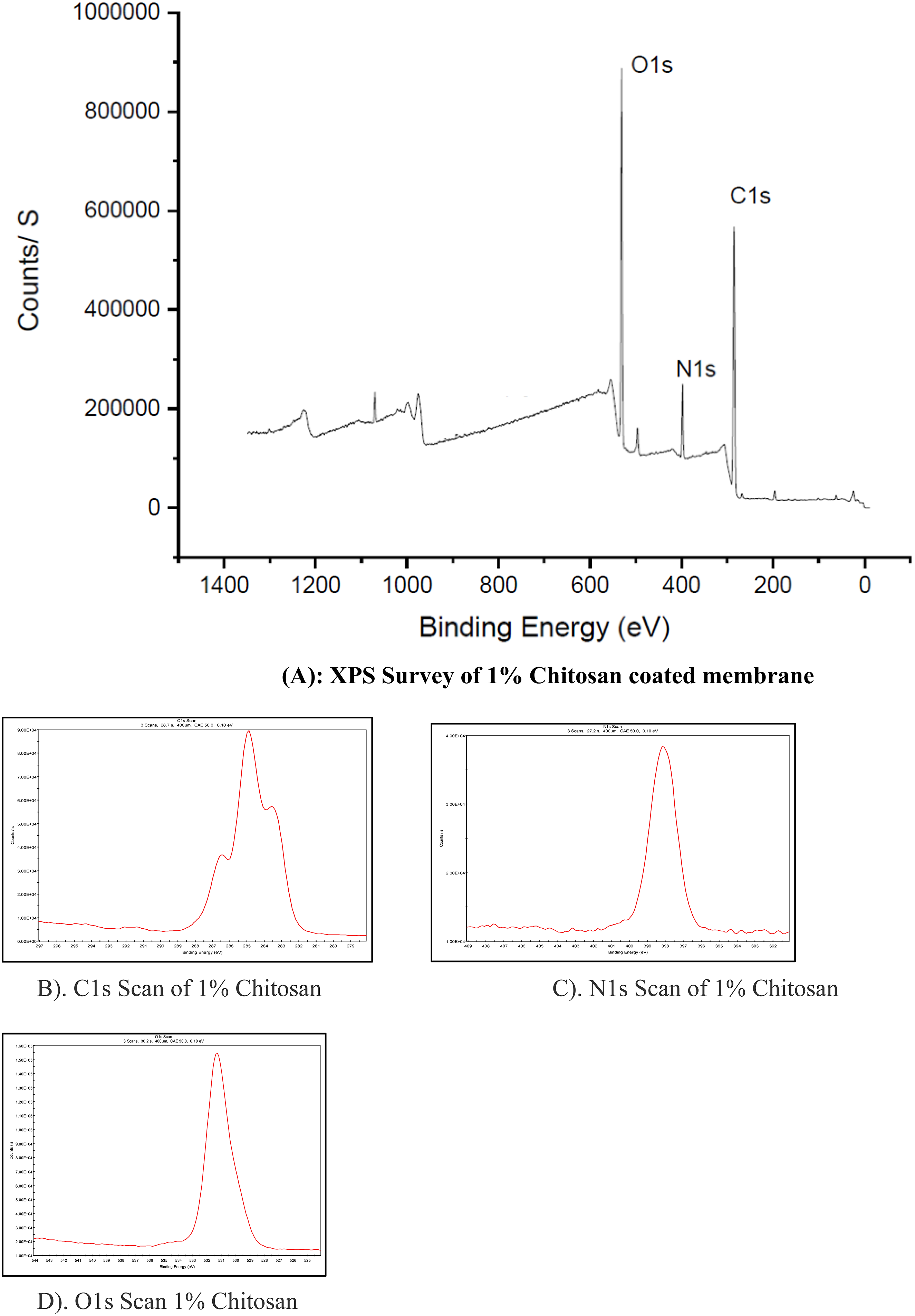
X-Ray photoelectron spectroscopy of membranes
The first peak of C1s falling at Binding Energy 284.76 eV contributed to C-C. The N1 binding energy was associated with the amino group NH2 present in the chitosan. The binding energy 531.11 eV is associate with O1s.
Figure 6(a) shows the presence of copper by the peak shown at about 932.33 ev. Cu2P scan as shown in Figure 6(e) demonstrated the peaks at the interval of 19.27 ev corresponding to Cu2P3 and Cu2P1. Major Peak of copper here is Cu2P3 at its Binding Energy of 932.33 eV and the second peak is of Cu2P1 and its Binding Energy is 951.6 eV. The difference between two successive peaks is Δ = 19.27 eV. Thus, based on the interval of 19.27 and the peak analysis, the oxidation state of copper is +1; that is copper is present in the Cu(I) state.
29
(b-e): XPS analysis of 1% Chitosan and 0.1% Cu coated membrane. (a). XPS Survey of 1% Chitosan+0.1% Cu, (b). C1s Scan of 1% Chitosan+0.1%Cu, (c). N1s Scan of 1% Chitosan+0.1% Cu, (d). O1s Scan of 1% Chitosan+0.1% Cu, (e). Cu2P Scan of 1% Chitosan+ 0.1% Cu.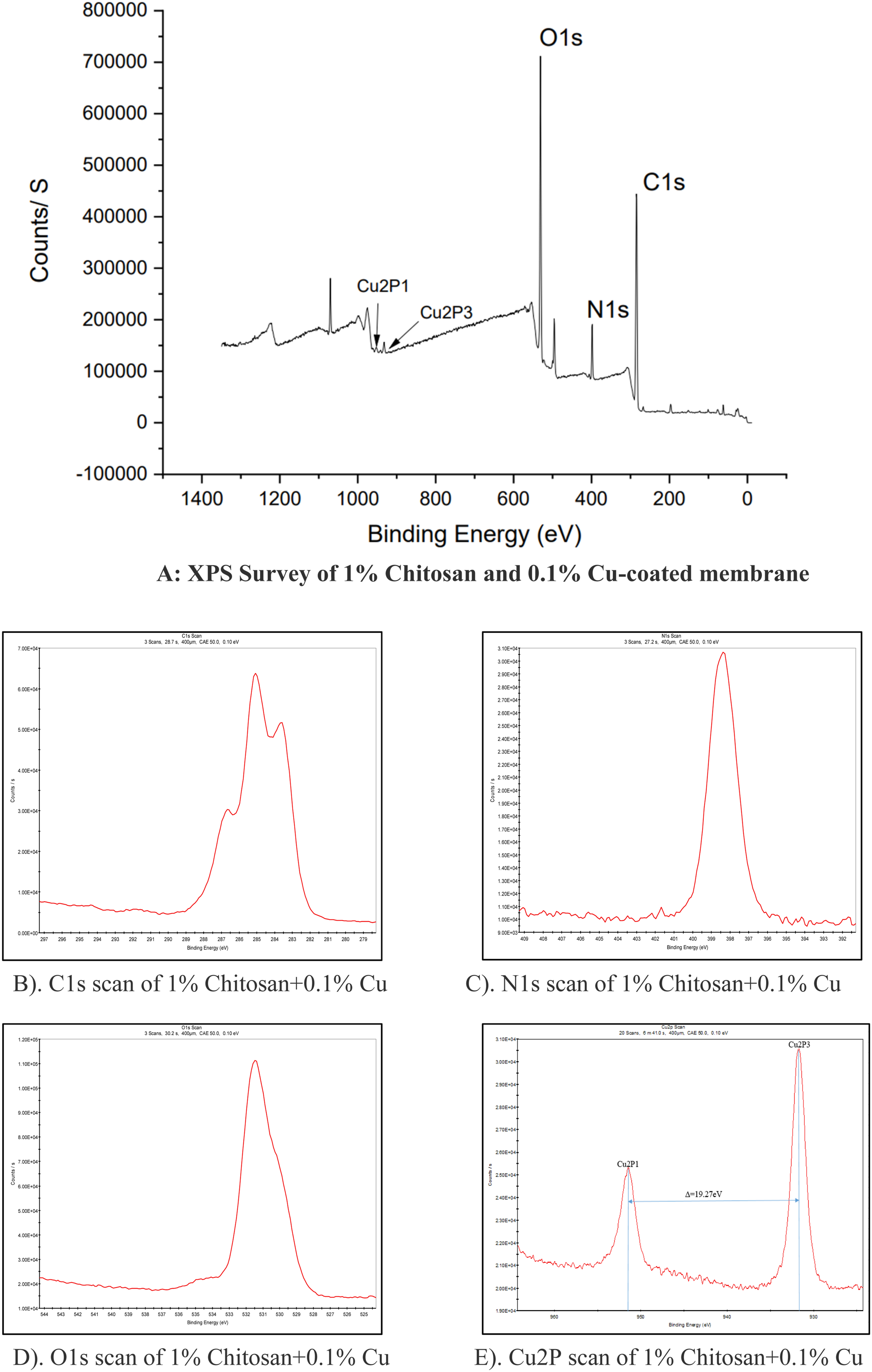
XRD analyses of membranes
Figure 7 shows the XRD patterns of different membranes. This XRD pattern for pure base membrane and copper-chitosan doped samples for 2θ parameter are obtained in the range of 0° to 90°. The XRD spectrum of the Pure PSF membrane shown in figure indicates the peak at 17.756° that confirms amorphous structure of PSF. As coating of 1% chitosan is applied on the PSF Surface the graph is slight shifted. When copper is added to the 1% Chitosan the graph starts broadening as copper concentration is increased and there is slight shift of the graph is also seen. This broadening of the graph conforms that the material is becoming more amorphous in nature. When diffraction happens, there will be high chances of having broadening of the X-ray diffraction pattern.
30
As compared to 0.1% Cu the graph of the 0.4% Cu is becoming more broaden and the peaks are also shifted. XRD patterns of the membranes.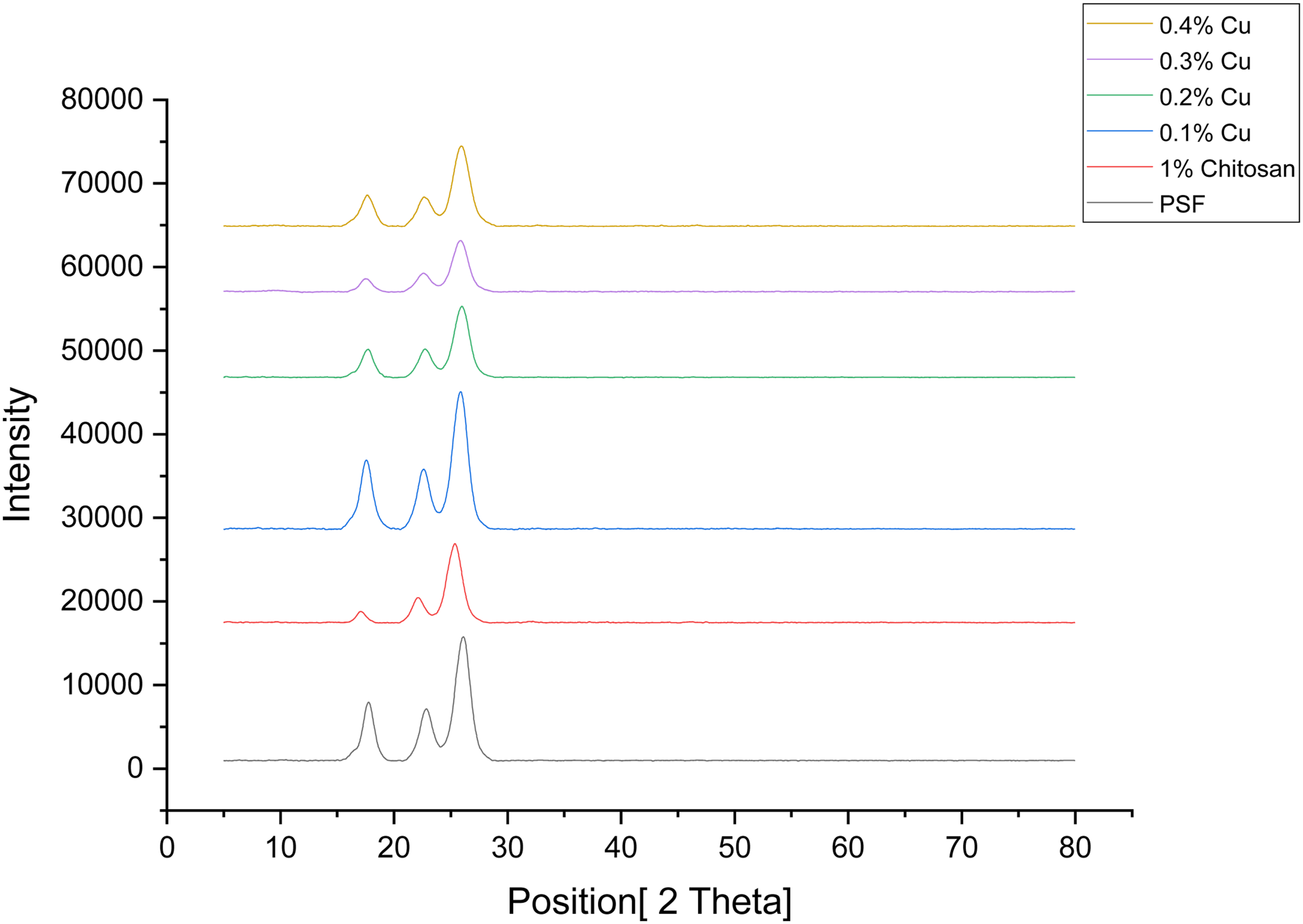
Pure water permeance (PWP) of the membranes
Pure water permeance of different membranes.

The pure water permeance data of all prepared membranes are presented in Table 2. The pure water permeance declined from 9.62 LMH/bar to 3.01 LMH/bar with coating of chitosan because of additional layer formation over the membrane. However, with incorporation of copper in chitosan layer, there is a steady increase in pure water permeance as compared to chitosan coated membranes and it increased from 3.01 LMH/bar upto 11.42 LMH/bar in case of 0.4% CuSO4 doping in chitosan. The increase in Copper doped chitosan coated membrane is because of increased hydrophilicity and decline in contact angle as shown in Figure 7.
Zeta potential analysis
Figure 8 shows the zeta potential analyses of the different membranes. Zeta potential analysis.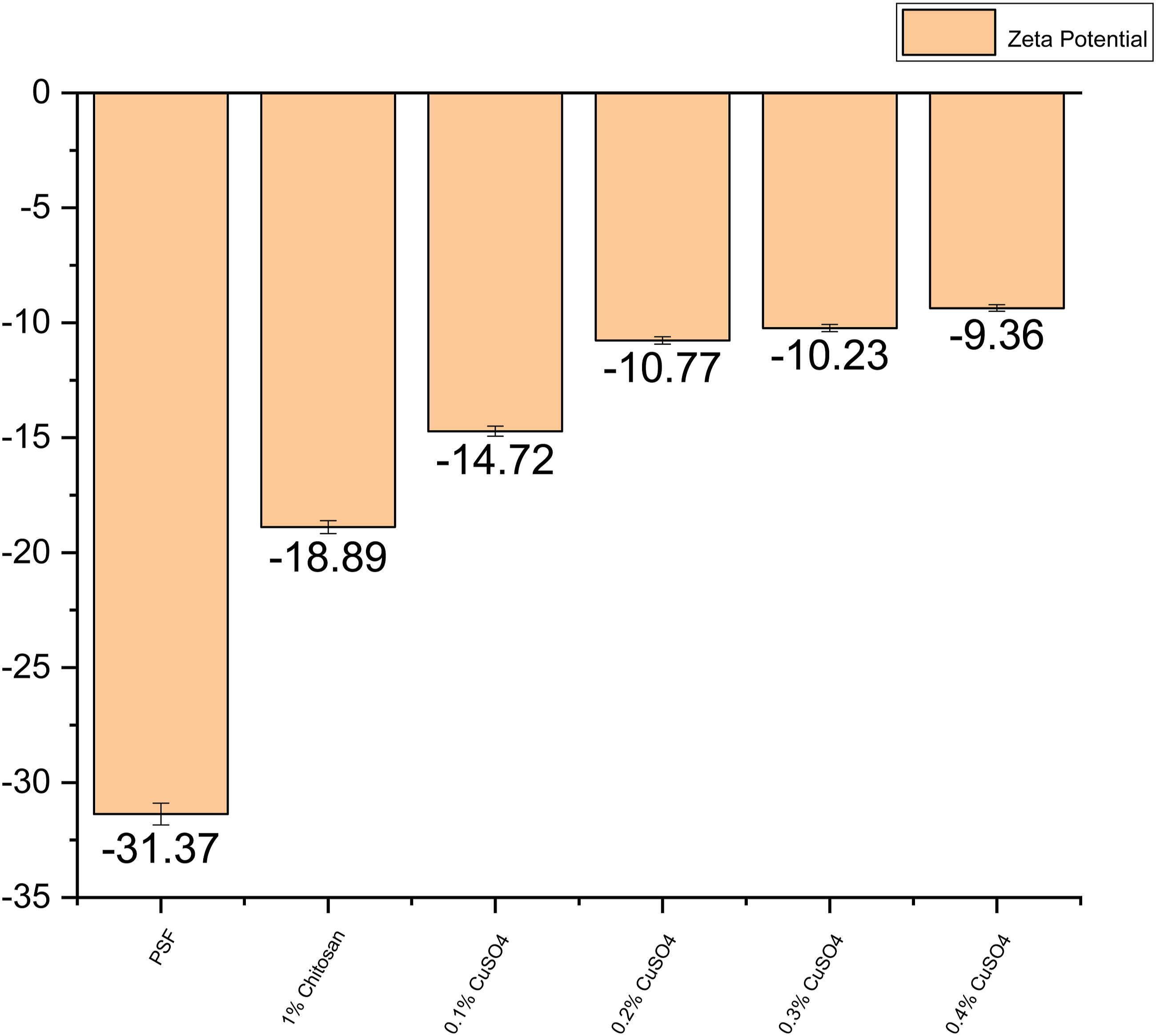
By measuring the Zeta charge, which indicates the charge present on the membrane, we may determine the total amount of charge present on the membrane’s surface. The figure indicates that the PSF surface has a height-negative charge. When chitosan is applied to the PSF surface, the membrane’s charge increases in terms of absolute value, and as the copper loading is increased the charge on the membrane surface also increased in absolute terms.
Dye removal performance of the membranes
Figure 9 shows the dye (Methylene blue and Methyl orange) removal performance of different membranes. Dye Rejection performance of the membranes.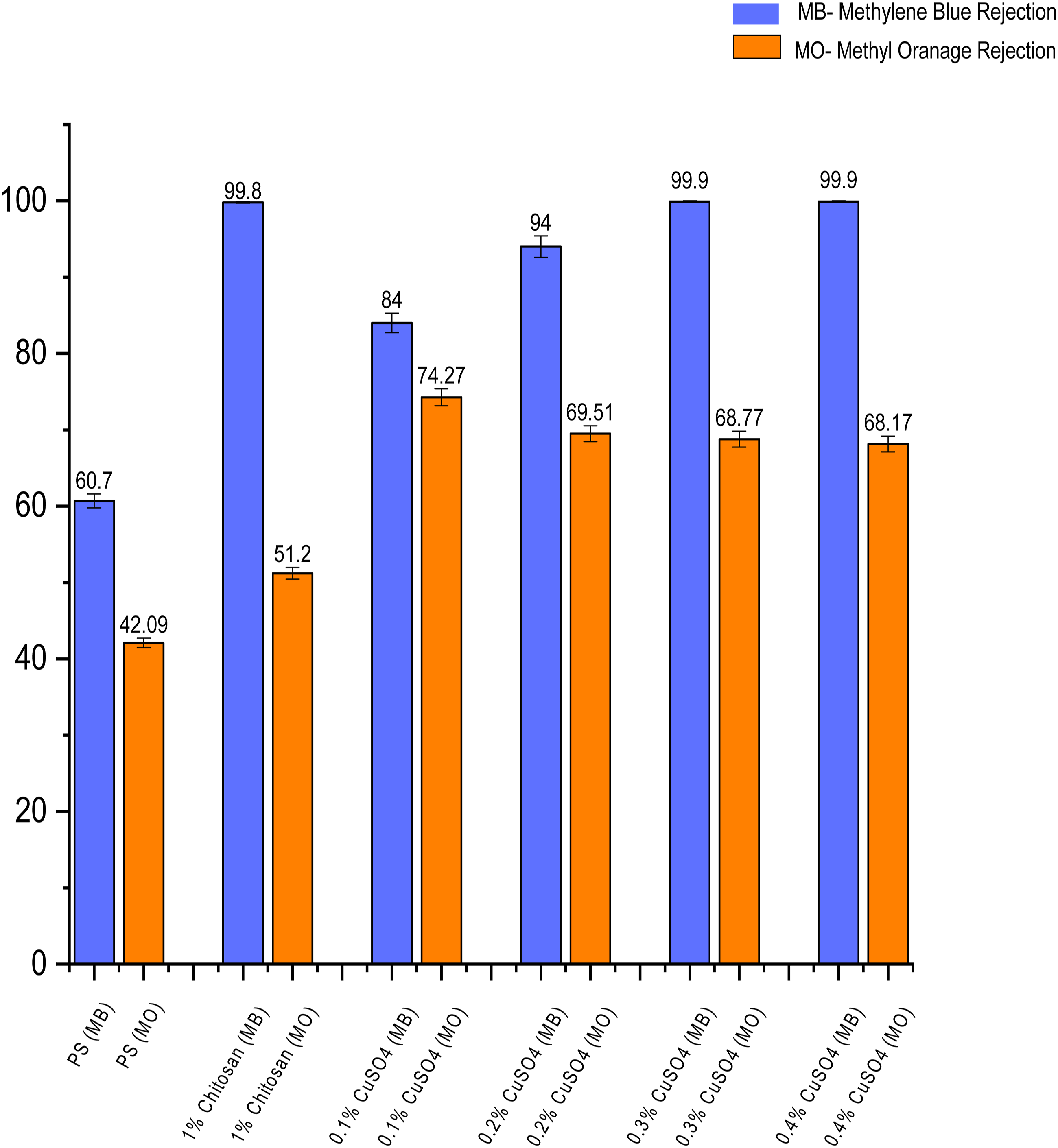
This bar chart represents the separation performance of two different dyes for example Methyl Orange (MO) - anionic and Methylene blue (MB) –cationic by different membranes Methyl orange is an anionic dye with a negative charge, and methylene blue is a cationic dye with a positive charge. While the average rejection of MB by PSF membrane was 60.7%, it increased to about 99.8% in case of Chitosan coated membrane because of the positive charge imparted on membrane surface and additional coating layer over the membrane surface. The average MO rejection by PSF membrane was 42.09%, which increased to 51.2% in case of chitosan coated membrane because of the additional coating layer above the membrane surface.
With increasing copper concentration in the top coating layer, the rejection of MB remains high reaching upto 99.9% in case of 0.3% and 0.4% Copper sulfate loaded membrane, which demonstrated nearly complete removal of MB. The average MO rejection was 74.27%, 69.51%, 68.77% and 68.17% in case of 0.1%, 0.2%, 0.3% and 0.4% CuSO4 loaded membranes as compared to 42.09% as demonstrated by PSF membrane. This is because of the additional layer coated on the membrane surface.
The increment in absolute value of the surface charge contributed to increased rejection of cationic dye due to repulsion, while the additional coating layer contributed to increase in rejection of anionic dye. However, the rejection of cationic dye is significantly higher as compared to anionic dye because of the surface charge.
Contact angle analysis
Figure 10 provides information on the contact angle for different membranes. The increment in contact angle with chitosan layer was observed. However, when copper concentration was increased in the chitosan solution, the contact angle decreased. This indicates that the membranes became more hydrophilic/wettable with increasing copper loading. Contact angle analysis.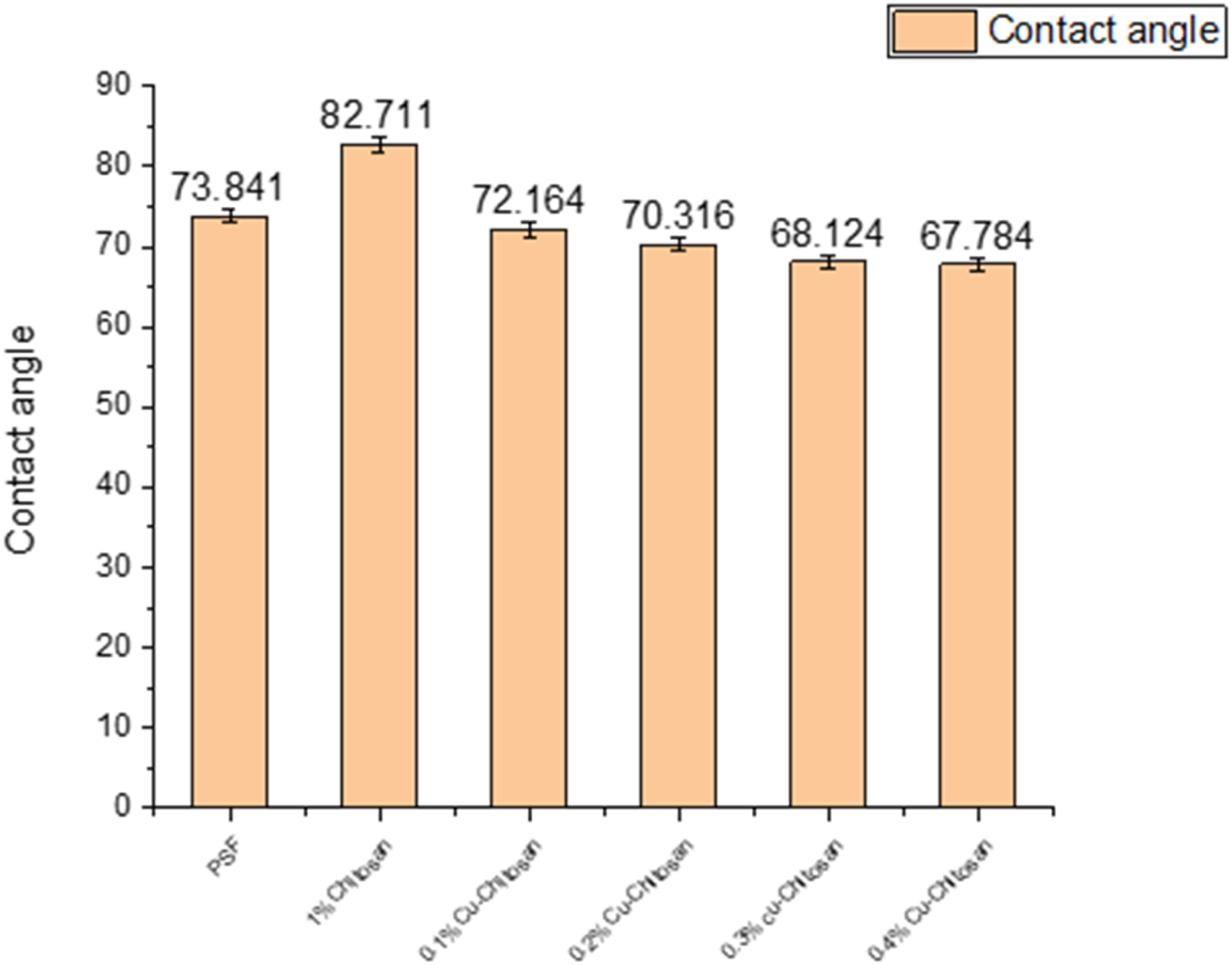
Flux recovery ratio (FRR)
The flux recovery ratio of the modified membranes are shown in the Figure 11. It can be observed that the flux recovery ratio of Polysulfone membrane was 18.65%, which was lower than 1% chitosan-coated membrane that is 36.27%. Moreover, with copper doping the flux recovery ratio increased further for 0.1% and 0.2% copper doping and decreased to 79.41% and 80.95% respectively and then decreasd. However, the modified membranes showed higher flux recovery ratio as compared to virgin Polysulfone membrane. This showed that the coated layer over the membrane prohibits the stubborn foulant layer formation when subjected to organic foulant such as egg albumin. Table 3 compares this work with other reported work. Flux Recovery Ratio of different membranes. Comparison of this work with other reported works.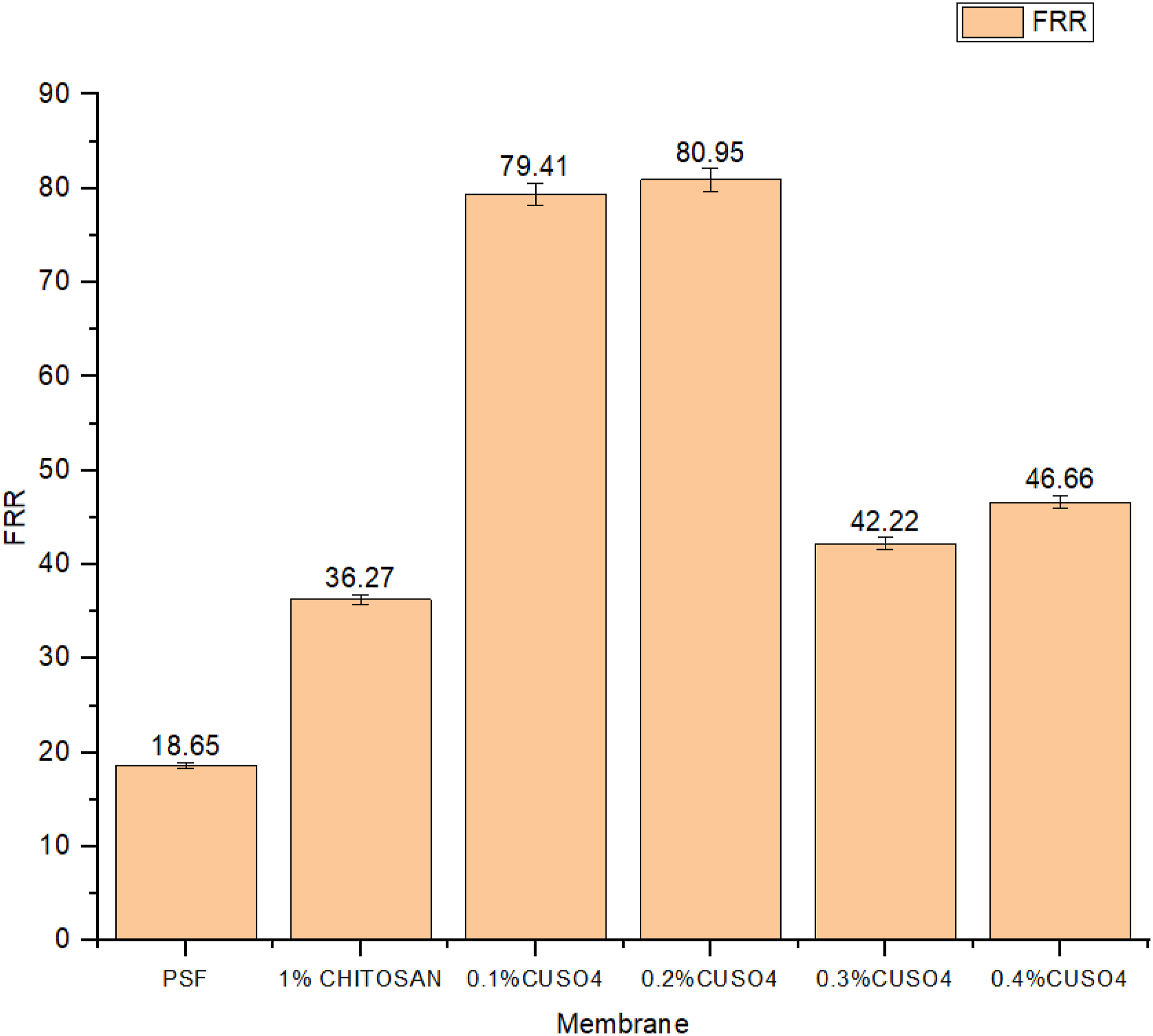

Conclusion
This paper comprises of the study on antibacterial properties of the membrane by making an antibacterial coating and the applications of membranes in dye separation. As copper-chitosan coated membranes have the very good antibacterial properties and this can be seen by the results. The highest antibacterial property was seen in 0.4% Copper-chitosan coated membrane where the adhered bacterial concentration was 60 CFU/mL, which is 99.94% lower than control. This bacterial killing was due to high copper concentration and this was seen by the zeta potential charge of the membranes. Zeta potential of virgin PSF was −31.37 mv and as copper concentration increased in the top coating layer, the zeta potential has increased in terms of absolute value and became −9.36 for 0.4% Copper-chitosan coated membrane. By this phenomenon of the charge difference, the dye rejection also got affected and it can be observed that as copper concentration was increased in the membrane, the rejection of the methylene blue increased due to repulsion owing to the positive charge on dye and the highest rejection was noted 99.9% on 0.4% Cu. For the negatively charged dye that is methyl orange; as copper concentration was increased in the membrane, the rejection declined upto 68.17%. While comparing the roughness of the membrane before and after bacterial adhesion, it was found that the roughness did not change much, which signified lower adhesion of bacteria on membrane surface.
This paper paves the way for future work in the area that is synergistic effects of copper and chitosan in antibacterial surface formation, application of these coatings on different bacterial species, other micro-organism etc. Moreover, the future scope of work may include the study of mechanism of microbial adhesion to membrane surface/role of copper in this area and evaluation of membrane life with this modification.
Footnotes
Acknowledgements
CSIR-CSMCRI PRIS registration number 01/2024. Authors thank the Department of Science and Technology, India for funding support (grant no. DST/TDT/WMT/fouled Membranes/2021/01). Authors thank the open-source software “Grammarly” for grammar/language checking and Marine elements and Marine environment division-Centralized Instrument Facility, CSIR-CSMCRI for the needful instrument and analytical support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Authors thank the Department of Science and Technology, India for funding support (grant no. DST/TDT/WMT/fouled Membranes/2021/01).