Abstract
Chitosan is a natural polymer that is well known for its inherent antimicrobial ability and natural healing properties and hence it has been developed into fibres that are used for wound dressing applications. To increase its antimicrobial potency, these fibres are treated with copper sulphate. In a wound environment, copper ions have potent antimicrobial abilities due to their propensity to bind with proteins and ability to produce hydrogen peroxides. In this article, copper-treated chitosan fibres of different concentrations have been tested and analysed to determine the most effective antimicrobial dose for potential wound care dressings. Selected common skin microflora, i.e. Staphylococcus aureus, Staphylococcus epidermidis and Micrococcus luteus, have been used to test the chitosan–copper combinations. The analysis, based on the test methods of zone of inhibition, spectrophotometry and plating technique, has shown that copper concentration of 0.3 gm/ml was highly effective against the three bacteria and could potentially be most suitable formulation for wound dressing applications.
Keywords
Introduction
Despite its long history of use, the antimicrobial capabilities of chitosan against a spectrum of Gram-positive and Gram-negative bacteria were formally recognised in 1979 [1] and a whole range of studies began [2–10]. The antimicrobial ability of this natural animal-based polysaccharide relies on a number of factors including pH, microorganism species, metal ion content, molecular weight, degree of deacetylation and so forth [11–13]. Chitosan fibres are of particular interest due to their large surface area, flexibility and their subsequent ease of conversion into nonwovens used for wound dressing applications [14–19]. As a wound dressing, chitosan serves a number of other functions including haemostasis, stimulation of macrophage activities responsible for wound repair and ultimately the healing process [20–26]. Inclusion of metal ions such as zinc and copper into chitosan fibres could potentially enhance their antimicrobial potency even further, given the high surface area and the good chelating ability of chitosan fibres. A number of researchers have worked in this area and have overall confirmed the additional benefits of metal ions in promoting the antimicrobial properties of chitosan [27–33]. In our previously reported article [34], we have demonstrated how chitosan fibres treated with CuSO4 5H2O had shown significant weight gain, i.e. from 9.89% to 46.82% for 0.03 to 0.3 gm/ml copper content. Furthermore, we have shown that once the chitosan fibres were loaded, the rate of copper ion release in solution A and protein solution with time was particularly significant for the protein solution, given the strength of binding between Cu (II) ions and the amino acid groups on the protein molecules. The antimicrobial potency of treated and untreated chitosan fibres against Escherichia coli was also quantified and the superior antimicrobial property of the treated fibres was verified. In this study, chitosan fibres have been treated with various concentrations of copper sulphate and their antimicrobial properties compared and contrasted using three types of common bacteria and three different techniques of assessments. The antibacterial nature of the copper has been studied since early science and it is thought to be dependant on various phenomenona at the microbe–metal interface. It is generally believed that the three-dimensional structure of protein within the microorganisms is altered by copper and its normal function is curtailed [33], thus resulting in inactivation of the microrganisms [11]. Copper ions tend to form radicals [12,13], disrupt enzyme structure [14] and form superoxide radicals [15]. On their interaction with lipids, they tend to cause peroxidation, thereby making holes in the cell membrane [16]. In the metabolism, copper tends to damage the respiratory chains [17], thereby causing impairment to respiratory pathways.
Materials and methods
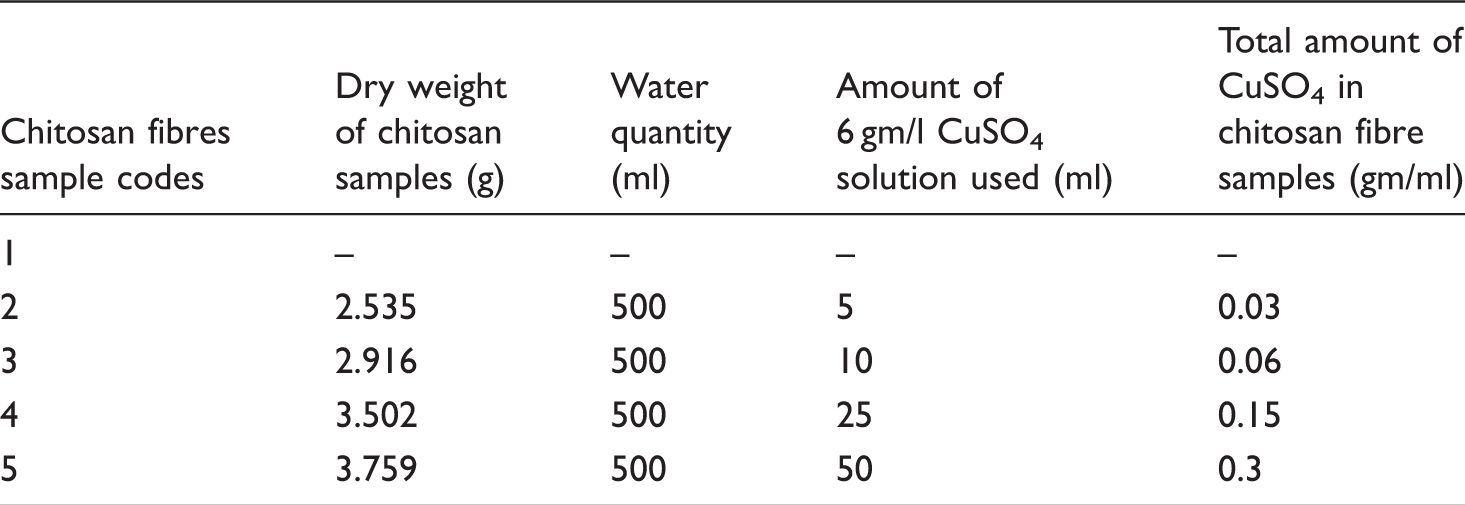
Synthesis of copper-containing chitosan fibres
Various concentrations of copper for chitosan fibre samples.
Bacterial cultures
Staphylococcus aureus ATCC 29213 and Staphylococcus epidermidis ATCC 12228 were obtained from ATCC and Micrococcus luteus B110 Sciento-Manchester was obtained from Sciento-Manchester. Mueller–Hinton Agar (II) LAB039-A and Mueller–Hinton Broth LAB114-A were from Lab M (Bury, UK).
Zone of inhibition (I)
A total of 20 μl of bacterial cultures grown overnight in Muller–Hinton (MH) broth were streaked on the Mueller–Hinton agar plates using spread plate method. Fibre discs of 1 cm diameter were made from chitosan–copper fibres by matting the fibres together by hand and placed in the centre of the agar plate. MH broth and agar was used because they lack proteins and salts; this property enables the antimicrobial elements of the fibres to diffuse smoothly through the agar and the broth used. The plates were incubated in an inverted position at 37°C for 24 h. The tests were conducted in triplicates and the mean of the value noted [22].
Antimicrobial analysis by turbidometric estimation using spectrophotometer at 600 nm (II)
A total of 10 ml of MH broth was dispensed into small capped glass tubes. Chitosan–copper ion fibre discs of 1 cm diameter were made using a cork borer and added into the broth tubes. The single fibre discs weighed approximately 0.015 g. The tubes were inoculated with bacterial cultures and incubated at 37°C for 24 h. Tubes of MH broth along with disc fibres without bacterial inoculation were also tested for turbidity. Tubes of pure cultures and blanks containing only Mueller–Hinton broth were also incubated. The tests were conducted in triplicate [22]. After incubation, the broth turbidity was examined for antimicrobial activity using a spectrophotometer at 600 nm.
Estimation of antimicrobial properties using modified dilution plating technique (III)
A total of 10 ml of MH broth was dispensed into small capped glass tubes. Single fibre discs of 1 cm diameter and weighing 0.015 g were added to the broth tubes. The tubes were inoculated with bacterial cultures and incubated at 37°C for 24 h; after incubation, 20 μl of bacterial culture was plated out on MH agar plates [22]. The plates were incubated at 37°C for 24 h in an inverted position. The tests were conducted in triplicate. After incubation, colony count was performed and the readings were noted.
Results
Zone of inhibition (I)
Of the four copper-loaded samples, only sample 5, i.e. the sample with the highest copper content, showed tangible antimicrobial potency when subjected to the zone of inhibition test. These results are depicted in Figure 1(a)–(c) for the three bacteria used.
Zone of inhibition analysis for: (a) Staphylococcus aureus, (b) Staphylococcus epidermidis and (c) Micrococcus luteus for sample 5. A is control and B represents the zone of inhibition, i.e. clear ring around the sample.
Measurements of zone of inhibition, i.e. potency diameter encircling the samples for different sets of sample 5, showed that Micrococcus luteus was most sensitive to the copper-treated chitosan, Staphylococcus epidermidis was moderately sensitive and Staphylococcus aureus was least sensitive. Figure 2 shows the diameter measurements of the zone of inhibition for the different sets when exposed to these common bacteria. In the second set, it was noted that Staphylococcus aureus had shown lower sensitivity in the zone of inhibition, which can be interpreted as a slight fluctuation in the collected data. In the third set, however, the distinct pattern of sensitivity of bacteria remained the same. The overall sensitivity in the zone of inhibition shows a slight decrease. This, however, does not discredit the authenticity of the results, bearing in mind that there are other factors that could influence experimental results.
Examination of antimicrobial properties by zone of inhibition method for chitosan–copper fibres (discs made from fibre materials) with common skin microflora.
Antimicrobial analysis by turbidometric estimation using spectrophotometer at 600 nm (II)
To verify the validity of the data collected from the zone of inhibition, further antimicrobial examinations were carried out for all the samples by turbidometric estimations using a spectrophotometer at 600 nm.
Statistical analysis of turbidometric estimations of chitosan–copper sample 5.

The controls were inoculated with bacteria without the disc fibres. The transmittance in the uninoculated MH broth that contained only the fibre discs was observed to decline as the copper concentration of the sample increased: from the control samples (broths without any fibre discs) to sample 5 (with the maximum). The decrease in transmittance is attributed to the increase in copper concentration that imparts the blue colour to the solution, thereby increasing the turbidity values. This decrease in transmittance due to colour impaction can be nullified for all sample values considering the uninoculated MH broth. In the same figure, for Staphylococcus aureus, the low transmittance for samples 1–3 indicates high growth factor and thus low antimicrobial activity. High transmittance for samples 4 to 5 signify low density of microbial growth and correspondingly high antimicrobial activity. For Staphylococcus epidermidis, maximum inhibition is observed for samples 3–5 and for Micrococcus luteus, inhibition is also observed to occur for samples 3–5. Figure 3 shows the amount of transmittance for the antimicrobial analysis.
The % transmission for antimicrobial analysis of turbidity using a spectrophotometer at 600 nm.
The estimation of absorption indicates the amount of visible light absorbed at 600 nm by the broth. Values correspond directly to the density of the broth. From Figure 4 it can be observed that the MH broth with the fibre disc had little absorption since there was no bacterial inoculation. The small increase in the values in samples 4 and 5 in the MH broth samples can be attributed to the blue colour of copper at higher concentration. The controls were inoculated with bacteria without the disc fibres. It was observed that for Staphylococcus aureus growth increased for samples 1 and 2, inferring that the bacteria was actually utilizing the low concentration of copper for its growth. Furthermore, as the concentration of copper increased, growth was observed to be slightly inhibited in sample 3. Growth for samples 1, 2 and 3 was observed to be relatively high since growth for Staphylococcus aureus declined sharply for samples 4 and 5, showing antimicrobial properties of the samples. In the same figure, for Staphylococcus epidermidis, growth declined from sample 1–3, the rise in values for samples 4 and 5 can be attributed to the blue colour development and not the bacterial growth. Further examination using the plating technique, which is discussed later in this article, confirms this fact. Growth of Micrococcus luteus was also observed to decline from samples 1 to 3, the rise of small values in sample 4 and 5 is again attributed to the blue colour development and not the bacteria growth.
Spectrophotometric estimation of absorption at 600 nm for analysis of antimicrobial properties of chitosan–copper fibres.
Estimation of antimicrobial properties using modified dilution plating technique (III)
The dilution plating was performed from the same tubes that were used for the spectrophotometer analysis; culture samples were first sampled.
Staphylococcus aureus
Figure 5 shows the results of the plating technique for Staphylococcus aureus. It is observed that in the control sample A, there were numerous colonies; this sample did not contain any fibre in the broth culture. In plates B and C (samples 1 and 2) also there were numerous colonies; these were also a form of control since the concentration of copper in the chitosan fibres was very low. Plate D represents the results of the antimicrobial activity of sample 3 with Staphylococcus aureus. Here there was a slight decrease in the density of bacterial growth although the colonies were still found to be numerous. Plate E represents the antimicrobial activity of sample 4 and here a sharp decline in bacterial growth was observed, i.e. 63 colonies and the dilution factor was 6.3 × 10−2000/ml of culture broth. The drop in bacterial count can be attributed to the increasingly potent concentration of the antimicrobial copper in the chitosan fibres. There was a further reduction in colonies in plate F, tolling up to 27 colonies, 2.7 × 10−2000/ml of culture broth, which represents the antimicrobial activity of sample 5. From the results obtained for the plating of the antimicrobial activity of chitosan–copper, it may be concluded that the fibre samples up to sample 3 had minimum or negligible antimicrobial effect on Staphylococcus aureus, while samples 4 and 5 had the most effect. The above analysis confirms that that the concentration of copper in chitosan samples 4 and sample 5 are potent antimicrobials against Staphylococcus aureus.
Assessment of antimicrobial properties for chitosan–copper with Staphylococcus aureus using the plating technique. A: control, number of colonies, numerous; B: sample 1, number of colonies, numerous; C: sample 2, number of colonies, numerous; D: sample 3, number of colonies, numerous; E: sample 4, number of colonies, 63 and F: sample 5, number of colonies, 27.
Staphylococcus epidermidis
Figure 6 shows the results of the plating method used for analysis of antimicrobial properties of chitosan–copper fibres with Staphylococcus epidermidis; the control plate A showed numerous colonies since the broth contained no fibres. Plate B (sample 1) also showed numerous colonies since it hardly contained any copper. However, once the copper concentration had risen to 0.06 gm/ml, i.e. plate C (sample 2), a large reduction in the number of colonies, i.e. to 2000 (200 × 10−2000/ml) was observed. There was total reduction of the number of the colony-forming units in plates D, E and F, representing samples 3, 4 and 5. From the above observations it is evident that there was a high level of antimicrobial activity against Staphylococcus epidermidis by the chitosan–copper fibres at and beyond 0.06 gm/ml of copper.
The plate count results for the antimicrobial properties of Staphylococcus epidermidis with chitosan–copper fibres (sample 5). A: control, number of colonies, numerous; B: sample 1, number of colonies, numerous; C: sample 2, number of colonies, 2000; D: sample 3, number of colonies, nil; E: number of colonies, nil and F: number of colonies, nil.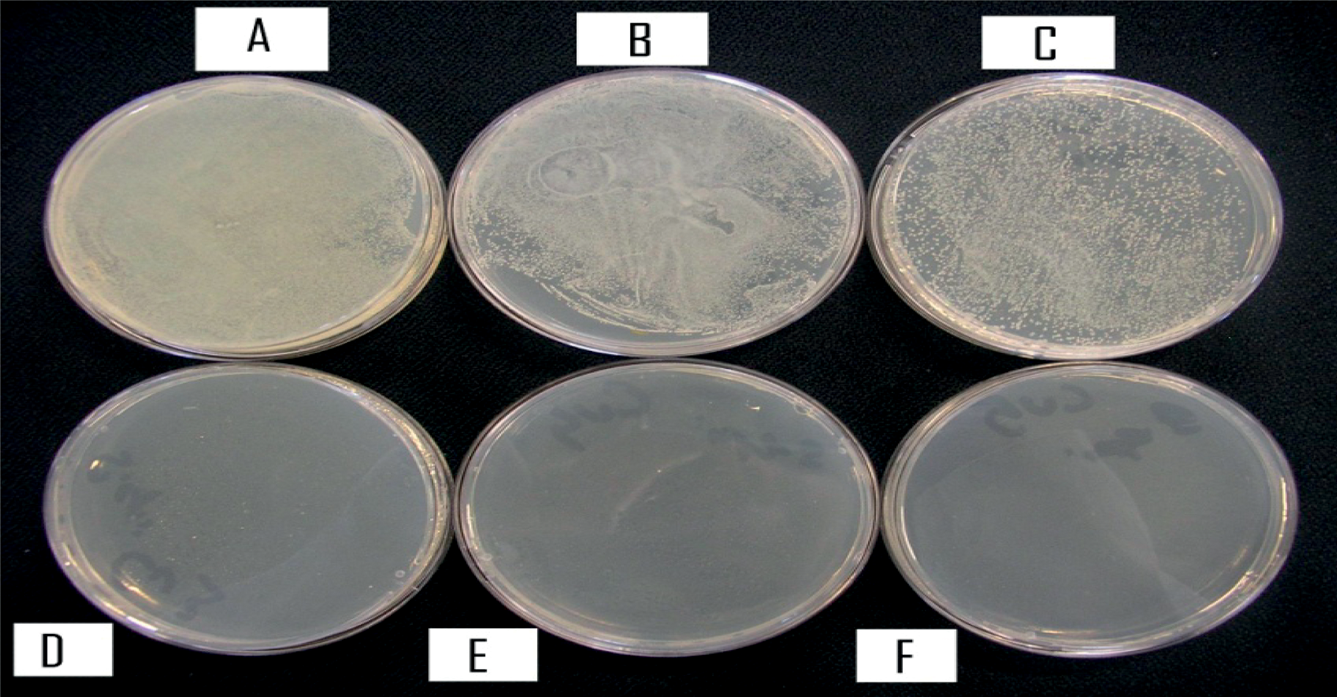
Micrococcus luteus
Figure 7 shows the results of the plating method for the analysis of antimicrobial properties of chitosan–copper fibres with Micrococcus luteus. As before, numerous colonies in plate A represent the control plate. Plates B and C likewise showed numerous colonies due to insufficient availability of copper ions. However, when copper content reached 0.06 gm/ml (sample D) and beyond, the number of colonies initially fell to 2300 (230 × 10−2000/ml) and then to zero for samples 4 and 5. From the above observations it is evident that there was significant antimicrobial activity against Micrococcus luteus by the chitosan–copper fibres in samples 4 and 5.
Antimicrobial properties of chitosan–copper fibres with Micrococcus luteus. A: control, number of colonies, numerous; B: sample 1, number of colonies, numerous; C: sample 2, number of colonies; numerous; D: sample 3 number of colonies, 2300; E: sample 4, number of colonies, nil and F: sample 5, number of colonies, nil.
Conclusions
From the analysis of the results derived by the various methods of antimicrobial assessments, it is evident that the chitosan–copper fibres are effective antimicrobial fibres when sufficient quantities of copper ions are present. The zone of inhibition analysis has demonstrated that the antimicrobial properties were confined to the highest concentration, i.e. 0.3 gms/ml (sample 5). This has provided visible and measurable demonstration of the antimicrobial properties for Staphylococcus aureus, Staphylococcus epidermidis and Micrococcus luteus. Furthermore, in the spectrophotometric analysis, it was found that the chitosan–copper fibres were antimicrobial against the three bacterial strains, i.e. samples 4 and 5 (0.15–0.3 gm/ml). The spectrophotometric analysis is more sensitive because in the broth cultures, bacterial cells come into direct contact with the antimicrobial element in the liquid; hence the antimicrobial potency at lower concentration becomes evident. Similarly, in the plating technique, antimicrobial effect was observed for Staphylococcus aureus at copper concentrations of 0.15 and 0.3 gm/ml, i.e. samples 4 and 5. Samples 3, 4 and 5 were found to be significantly antimicrobial against Staphylococcus epidermidis, i.e. antimicrobial ability is active even at 0.06 gm/ml of copper content. Micrococcus luteus showed antimicrobial resistance for samples 4 and 5, i.e. 0.15 and 0.3 gm/ml of copper concentration. In the overall conclusion, it can be safely assumed that when copper concentration of 0.3 gm/ml is used in these fibres, effective antimicrobial resistance against the three common skin microflora, i.e. Staphylococcus aureus, Staphylococcus epidermidis and Micrococcus luteus can be expected.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
