Abstract
This study investigates the enhancement of the properties of mixtures based on hydrogenated butadiene nitrile rubber (HNBR) under thermal, radiation and thermo-radiation effects by introducing into them the composition of chlorine-containing triazine and peroxide compounds (HCPX and ABTST) into the composition. It has been shown that radiation-chemical cross-linking and the formation of effective chemical cross-links in HNBR depend both on the absorbed dose of radiation and on the addition of vulcanizing agents. It has been established that the activity of hexachloroparaxylene (HCPX), dicumyl peroxide (DP) and triazine (ABTST) is not the same; the content of the gel increases markedly with increasing absorbed dose. A higher crosslinking density is observed for images subjected to thermo-radiation crosslinking. The change in the molecular structure of HNBR, observed both during heating and irradiation, has the same nature. It has been shown that the physical and mechanical properties of saturated radiation and thermal vulcanizates are practically inferior to thermo-radiation ones in strength.
Introduction
A wide range of elastomers requiring enhanced thermal stability and resistance to aggressive environments can be obtained on the basis of hydrogenated butadiene nitrile rubber (HNBR), whose properties are significantly superior to SKN-26.1–4 To cross-link HNBR macromolecules, it is prospective to use low-molecular additives that provide high temperature resistance, resistance to thermal and radiation aging and various types of energy effects and do not require the development of a complex vulcanizing system. For the vulcanization of HNBR, various organic cross-linking agents are used.5–7 The properties of crosslinking agents for HNBR and the principles of their classification were described previously. 8 It is necessary to consider the type and structure of elastomers when selecting crosslinking agents for HNBR,
Some cross-linking agents are toxic such as tetramethylthiuram disulfide, mercaptobenzothiazole and diphenylguanidine. Dusty mixtures of sulfur and amine-containing compounds are explosive; vapors irritate the mucous membranes of the upper respiratory tract and eyes. 9
It is known that radiation-chemical cross-linking and the formation of effective chemical cross-links in HNBR depend both on the absorbed radiation dose and the addition of cross-linking agents.10,11 Radiation-induced cross-linking uses high-energy radiation to form cross-links between polymer chains, enhancing the material’s performance. This process generates free radicals that initiate cross-linking, resulting in a stronger molecular network. Optimizing radiation doses is essential, as it significantly influences cross-link density and the mechanical properties of HNBR, underscoring the importance of understanding these mechanisms for advancing its applications. Gamma and electron-beam radiation can induce cross-linking in hydrogenated nitrile-butadiene rubber (HNBR), offering an alternative to chemical curing methods. 12 Cross-linking density increases linearly with radiation dose, with a yield of about 2.5 crosslinks/100 eV. The process leads to increased gel content, dynamic storage modulus, and glass transition temperature. However, chain scission also occurs, with 6-9 scission acts per 100 crosslinking acts. While radiation-induced cross-linking improves some properties, it can also cause degradation of mechanical properties at higher doses, such as decreased tensile strength and elongation at break. The formation of double bonds, carbonyl, and ether groups has been observed in irradiated HNBR. Nitrile butadiene rubber can achieve radiation resistance up to 4 MGy. To increase the degree of cross-linking and physical-chemical properties of vulcanizates, accelerators and sensitizers of chlorine-containing and triazine compounds are utilized. 13
It was of interest to study the possibility of improving the properties of mixtures based on saturated hydrogenated butadiene nitrile rubber (HNBR) under thermal, radiation and thermo-radiation effects by, the introduction of hexachloroparaxylene (HCPX), 2-amino-4,6-bis-trichloromethyl-sym- triazine (ABTST), dicumyl peroxide (DP).
To select the most radiation-intensive ways for radiation vulcanization of HNBR, studies were carried out to establish the general patterns of the influence of γ-irradiation on the structure and properties of the elastomer of the developed formulation. At the same time, we took into account the influence of the structure and composition of HNBR on the radiation-chemical yield; the number of effective cross-links nc arising as a result of the interaction between low-molecular components and the elastomer, which ultimately determined the properties of the vulcanizates.
To ensure satisfactory physical and mechanical properties of radiation vulcanizates, epoxy resin (ED-40), zinc oxide and carbon black (P324) were introduced as a modifier. 14
To form an additional chemical C-S-C bond, 0.2 parts by weight of sulfur was additionally added to the thermal vulcanizates. For a comparative assessment of the cross-linking effect of HNBR, dicumyl peroxide (DP) was used.
Experimental Part
Materials and Preparation
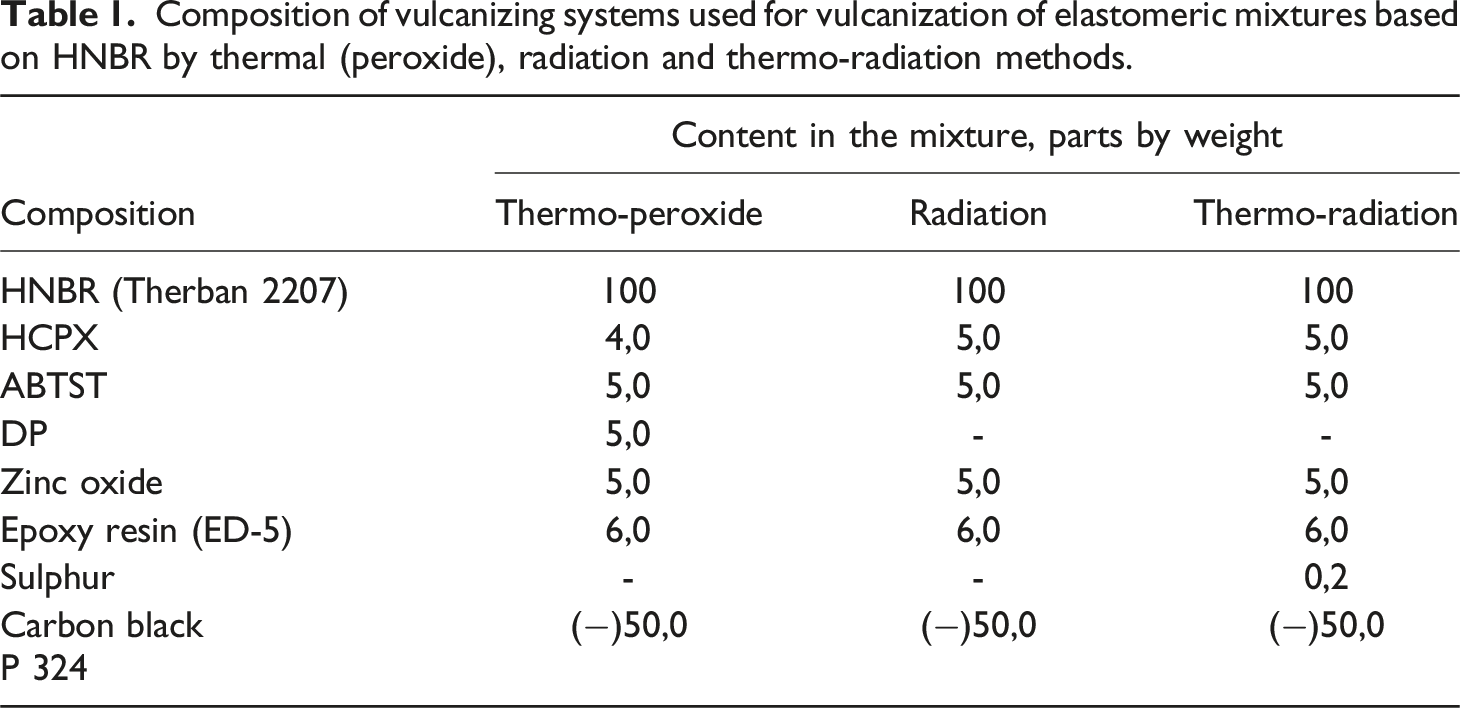
Composition of vulcanizing systems used for vulcanization of elastomeric mixtures based on HNBR by thermal (peroxide), radiation and thermo-radiation methods.
The radiation-chemical yield of effective cross-links (nс), the number of cross-linked molecules (
Calculation of cross-linking density by sol-gel analysis in an organic solvent (Flory-Rehner)
The Flory-Rehner equation was utilized to ascertain the cross-linked density of the compounds.
16
After weighing the samples to a mass of roughly 0.25 ± 0.05 g, they were submerged in toluene and kept in a dark environment for 5 days, or until equilibrium was attained. After that, the samples were taken out, dried to get rid of extra solvent, and weighed. Following a 24-h period at 80°C in the oven, the samples were weighed. Equation (1), which was created by Flory-Rehner, was used to calculate the cross-linked density using these mass values. The parameters that were utilized were 106.4 cm3 mol−1 for the molar volume of toluene (V0) and 0.39 for the Flory Huggins interaction parameter (χ).
17
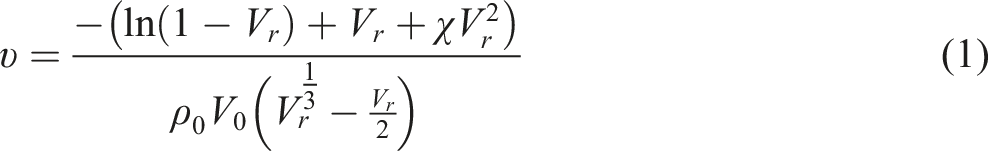
Pre-vulcanization characteristics of elastomer samples were also investigated by Mooney viscometer according to ASTM D1646-19a standard.18,19 Scorch time and cure rate was obtained from analysis and results are discussed later in result section.
Gel content calculation
Gel contents of three samples were investigated using extraction method by solvent (toluene).20–22 Gel content is calculated with following methodology. A 0.2 g specimen was precisely cut and weighed. Then, sample added to flask with 50 mL of toluene inside and placed magnetic stirrer and stirred for 1 day in the dark until resulting weight remained stable. Further, gel-containing solution was filtered through 400-mesh stainless steel filter bag. Then residue on filter was weighed with filter bag and the percent gel content was found by following equation Eq. (2):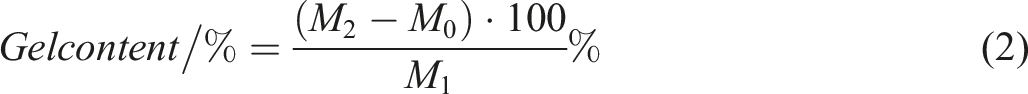
Tensile strength and compressive residual stress test
Tensile test of every vulcanized sample with different compositions was done using Liyi 1000N Universal Tensile Testing Machine (Dongguan Liyi Environmental Technology Co., Ltd). Geometry of specimens was in accordance to ISO 37 type 2.23–25 All geometry and dimensions are mentioned in Figure 1. For each kind of elastomer two identical specimens were prepared and test was carried out twice. Tensile rate was 500 mm min−1. Furthermore, following data was successfully obtained: Stress at elongation (300 %), tensile strength, elongation at break. Geometry and dimensions of dumbbell-type samples for tensile test. L = Minimum total length = 75 mm, GL = Length of gauge section = 25 ± 1.0 mm, B = Width of ends = 12.5 ± 1.0 mm, W = Width of gauge section = 4.0 ± 1.0 mm, R1 = Transition radius (inner) = 8.0 ± 0.5 mm, R2 = Transition radius (outer) = 12.5 ± 1.0 mm.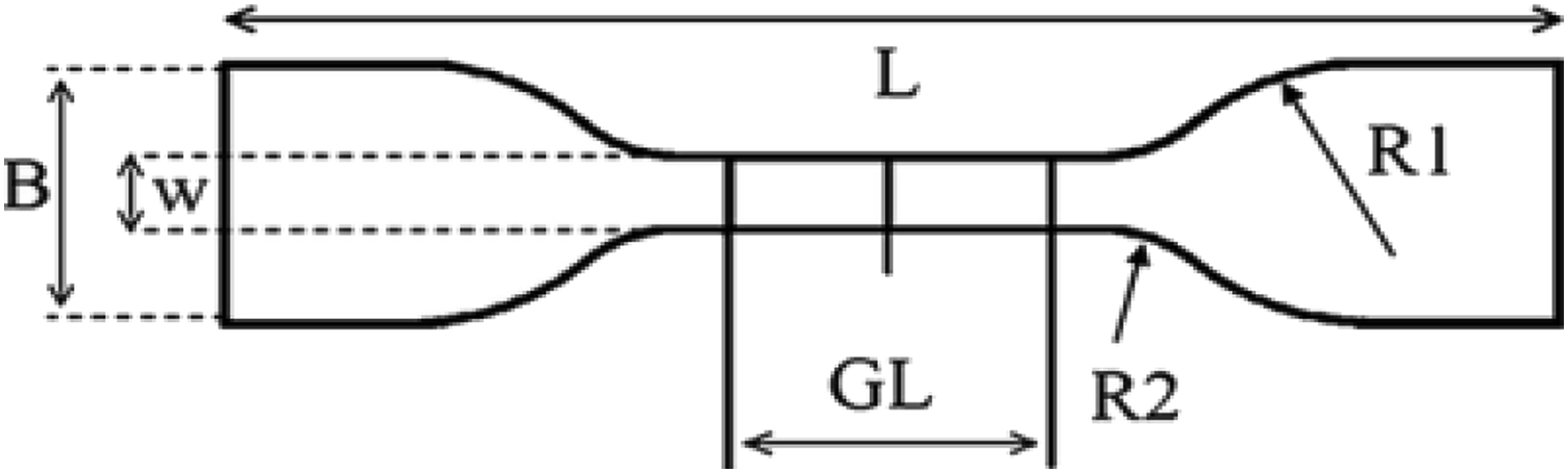
In order to explore mechanical properties of samples compressive residual stress analysis was also carried out in accordance to GOST 9.029–74.26,27 Specimens are prepared in a cylindrical form with dimensions of 29.0 ± 0.5 mm diameter and 12.5 ± 0.5 mm height. Samples were immersed in sea water for 72 hr duration at 150°C. After aging process elastomers were taken out and held in room temperature for 2 hr to cool down. The height change of sample was recorded and test results later were calculated by following equation Eq. (3):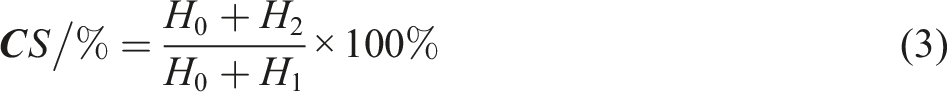
The compression tests conducted on HNBR elastomer samples revealed a permanent reduction in thickness following prolonged compression at elevated temperatures. Unlike materials that exhibit reversible elastic deformation, the HNBR samples did not return to their original thickness after removal of the compressive load. This behavior suggests that the material undergoes irreversible molecular rearrangements during compression, particularly under the conditions of high temperature and extended duration.
The observed permanent thickness reduction is likely due to the realignment and compaction of polymer chains within the HNBR matrix. At elevated temperatures, the increased molecular mobility facilitates a denser packing of polymer segments, which leads to residual deformation that the material cannot recover upon cooling. Furthermore, the cross-link density achieved through radiation-induced cross-linking plays a significant role in limiting the elastomer’s ability to return to its initial dimensions. A higher cross-link density, while enhancing tensile strength and stability, restricts chain mobility, thereby reducing elasticity and contributing to permanent deformation under compressive loads.
FTIR Spectra of Elastomers
Fourier Transform Infrared (FTIR) spectra were obtained on VARIAN 640-IR FTIR spectrophotometer. Changes in the molecular structures of HNBR after irradiation were assessed by Fourier transform infrared spectroscopy. The spectra were recorded in the range of 700-2400 cm−1. The interpretation of the spectra was carried out in accordance with correlation tables and the manual intended for physical and mechanical properties of radiation vulcanizates in accordance with literature data.28–30
Chlorine Content Calculation
The method 31 outlined for calculating chlorine content in rubber involves weighing roughly 1-2 g of a dry rubber sample and subjecting it to combustion at temperatures between Tcombust, oxidizing organic matter and leaving inorganic residues, notably chlorine. Combustion gases are at that point then passed through a silver nitrate (AgNO3) solution, where chlorine (Cl2) reacts with AgNO3 to form silver chloride (AgCl), according to the reaction:
2AgNO3 + Cl2 →2AgCl+2NO2 + O2
To find out the mass of chlorine absorbed (mabsorbed) gravimetrically, after the reaction of chlorine with silver nitrate to form silver chloride, the precipitate is isolated, washed, and dried. The dried precipitate is then weighed accurately, with its mass representing the absorbed chlorine. This mass is used to calculate the percentage of chlorine in the rubber sample.
The percentage of chlorine (%Cl) in the rubber sample is calculated by following equation Eq. (4):
Quality control measures are important, including the analysis of standard reference materials and repetition of analyses to ensure accuracy and precision, while adherence to safety protocols is essential due to high-temperature operations and chemical handling.
Results and Discussions
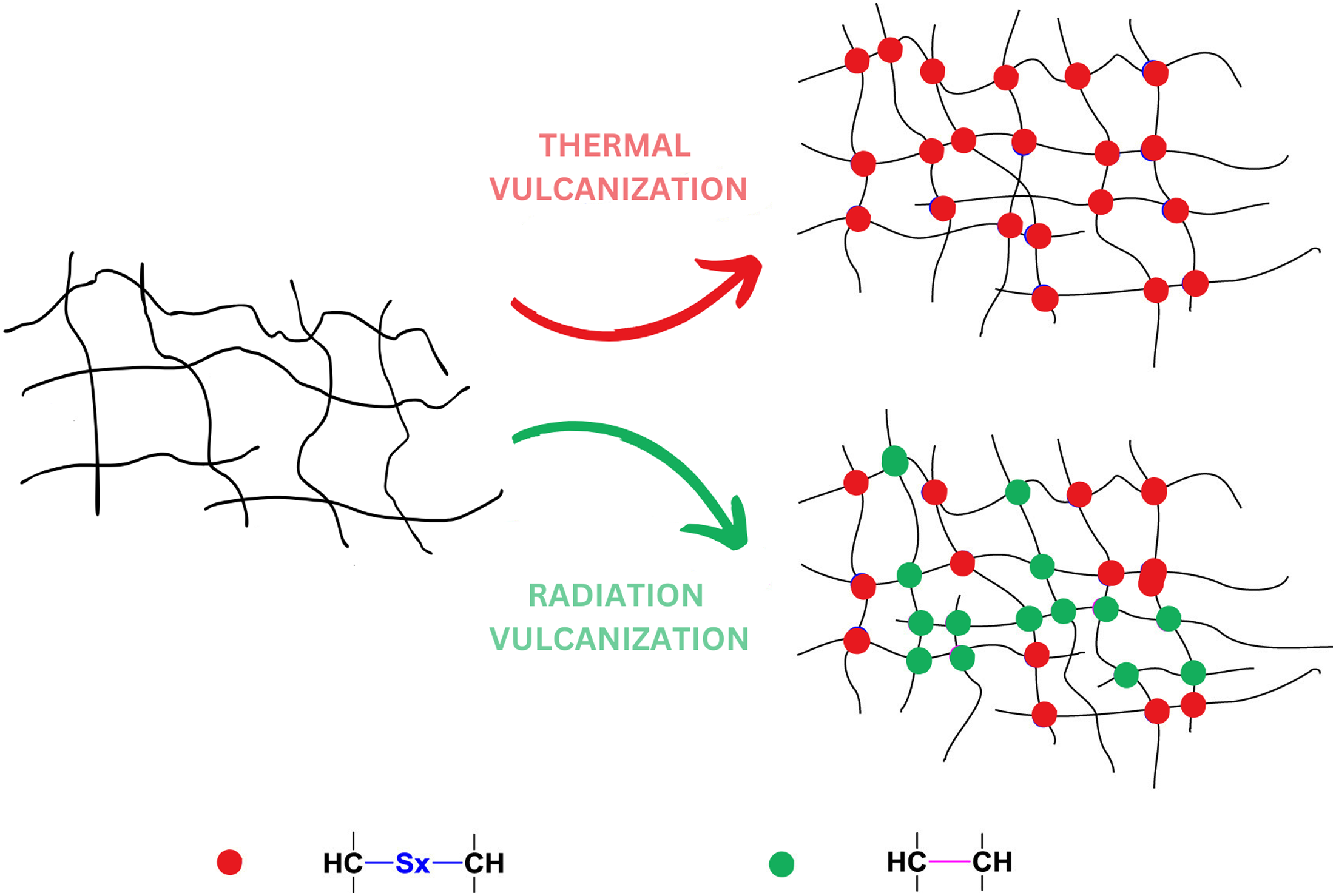
The nature of the interaction of the polymer chains of the elastomer with low-molecular compounds plays a large role in the process of radiation cross-linking of the elastomer. This interaction is due to the formation of both physical and chemical bonds, the number and ratio of which can vary depending on the properties of the interacting components and the nature of the interaction of HNBR under the influence of ionizing radiation. The vulcanization process via heat and radiation is shown in Figure 2. It has been observed that during radiation vulcanization more stable bonds (C-C) with higher energy were formed compared to heat vulcanization process (C-Sx-C). Vulcanization process by heat and radiation.
12
Influence of crosslinking type and vulcanizing system on scorch time τ5, cure index Δτ (τ35-τ5) and concentration of effective cross-links nc (150 оС x 40`, Dose = 300 kGy).

In this study, the cross-link density of HNBR samples was calculated using sol-gel analysis with the Flory-Rehner equation and presented in Table 2. The theoretical upper bound of cross-link density for hydrogenated butadiene nitrile rubber (HNBR) is primarily limited by two factors: the availability of reactive sites (double bonds) in the polymer backbone and the saturation capacity of cross-linking agents. Due to the high hydrogenation level in HNBR (typically around 96%), only a small proportion of the original double bonds remain available for cross-linking. This high hydrogenation is beneficial for enhancing the rubber’s thermal stability and resistance to aging but also reduces the potential sites for chemical bonding. Therefore, the upper limit of cross-link density in HNBR is inherently lower than in less-saturated rubbers, where more unsaturation allows for denser cross-linking.
Additionally, the saturation point of the cross-linking agents used, such as hexachloroparaxylene (HCPX) and 2-amino-4,6-bis-trichloromethyl-sym-triazine (ABTST), also restricts the maximum achievable cross-link density. These agents, while effective, reach a limit in bonding efficiency as the concentration of available reactive sites diminishes. Once saturation is reached, additional cross-linking agents do not contribute significantly to increasing cross-link density, as there are fewer unreacted double bonds to form further cross-links. Thus, the values reported in Table 2 represent near-optimal cross-link densities for HNBR, taking into account both the inherent molecular structure and the practical limits of the cross-linking agents used. This theoretical upper bound contextualizes our findings, illustrating that the cross-link densities achieved align well with the material’s intended performance properties and are consistent with maximum achievable values for HNBR under the study’s radiation-induced cross-linking conditions.
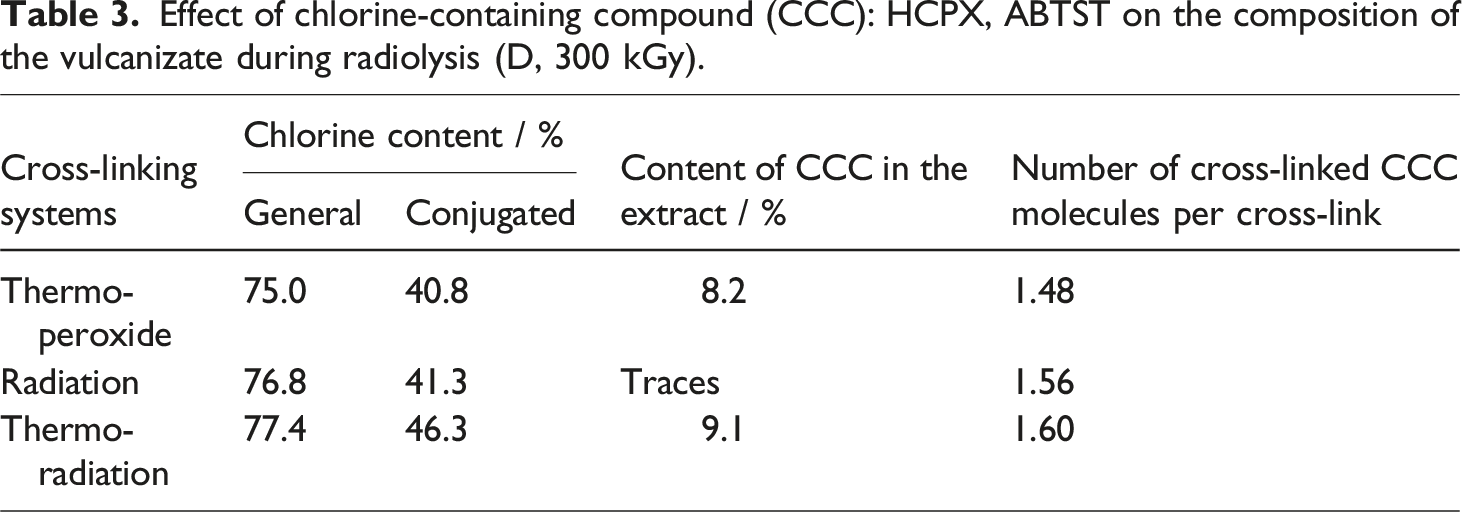
As expected, the gel content increases noticeably with increasing absorbed radiation dose (see Figure 3). Dependency of gel content on time in thermo-peroxide vulcanizing system and radiation dose in radiation and thermo-radiation vulcanizing system was investigated. From the results it is clearly seen that, in thermo-peroxide vulcanization method ideal time for obtaining maximum gel content is 60 min. After exceeding 60 min destruction is observed. In other two methods samples are exposed to various radiation doses and between 100 and 400 kGy it showed an increase. Over 300 kGy radiation dose gel content starting to decline due to decomposition in polymer chain. The maximum gel contents in the model mixture and in the peroxide unsaturated sample are close and indicate that the studied chlorine-containing compounds and epoxy resins take part in the crosslinking process. As the concentration of effective cross-links (nc) in samples with the studied chlorine-containing compounds is higher, also their activity is the higher (see Table 2). A higher crosslinking density is observed for samples subjected to thermo-radiation crosslinking. In the case of thermo-radiation cross-linking, the density of the resulting spatial network is lower than that of other studied samples. The change in the molecular structure of the HNBR itself, observed both during heating and irradiation, has the same characteristics. Kinetics of gelation during vulcanization of HNBR rubber (a) thermo-peroxide, (b) radiation, (c) thermo-radiation method using the suggested cross-linking systems.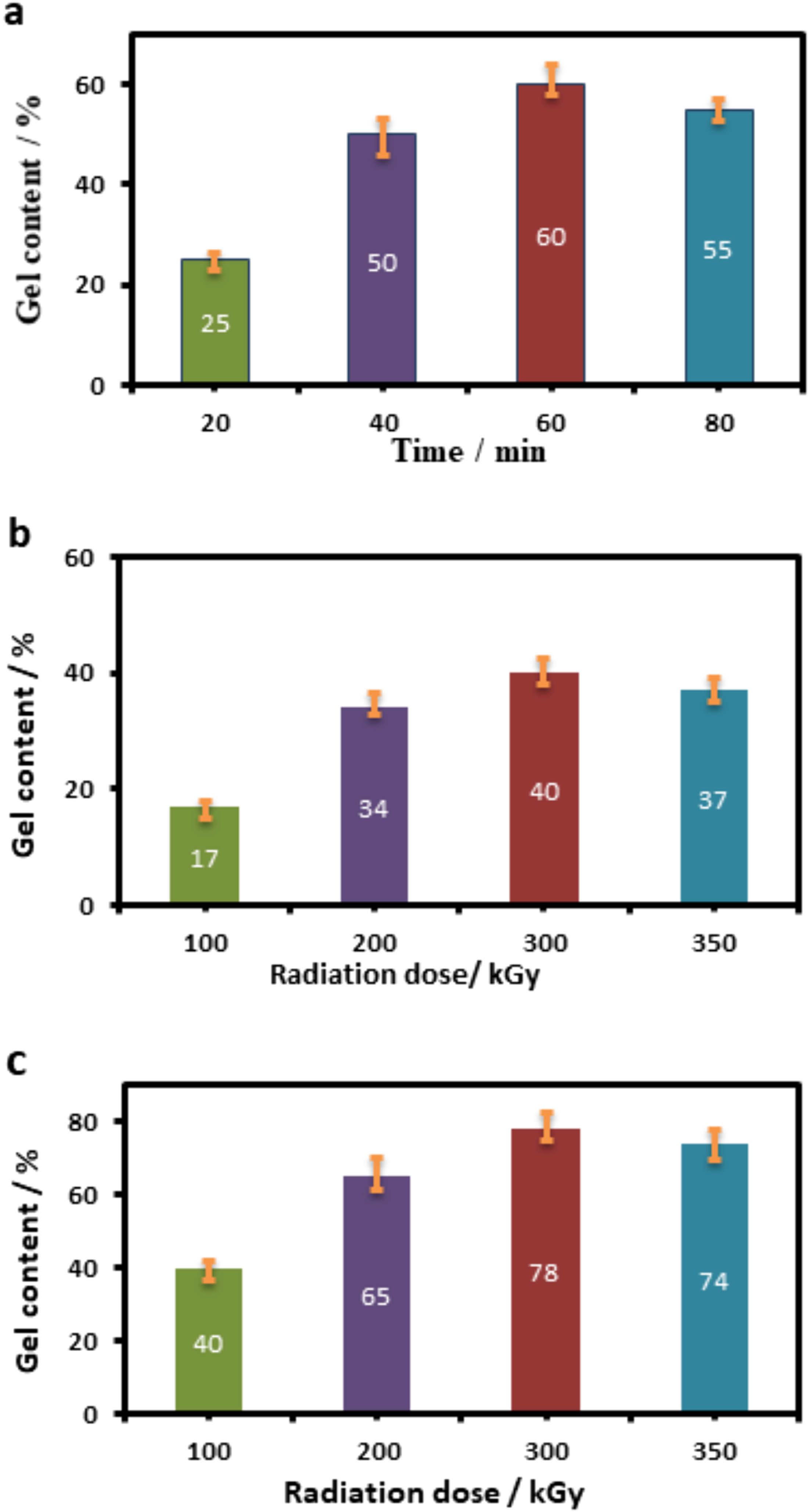
The interaction between polymeric peroxide and hydrogenated nitrile butadiene rubber (HNBR) was examined through analysis of infrared (IR) spectra alterations. A new band observed at 720 cm−1 is related with vibrations of the C-Cl group. The IR spectroscopic data revealed that the polymeric peroxide lacked absorption bands corresponding to carbonyl groups and also a doublet within the range of 1805-1765 cm−1, indicative of -CO-OO-CO- groups. Notable changes observed in the final state samples included a reduction in the intensity of the band at 1450 cm−1, associated with deformation vibrations of the C-H bond in the CH2 group, as well as diminished intensities of the bands at 970 and 1340 cm−1, linked to out-of-plane and deformation vibrations of the C-H group in the trans configuration of C = C bonds.
Literature suggests that cross-linking of HNBR likely happens through the bonding of polymeric peroxide to double bonds (C = C) after a 120-min heat treatment of HNBR. The presence of spectral bands at 1805 and 1765 cm−1, attributed to -CO-RO-CO- and COO bonds involved in cross-linking. Thus, the cross-linking is carried out not only by 2% saturation, but the structure HNBR has a significant impact on the rate of cross-linking. Furthermore, the emergence of a peak around 2235 cm−1 is linked to vibrations of CN groups. It is documented that the radiation-induced cross-linking of HNBR with chlorine-containing compounds is activated by metal oxides Figure 4. IR spectra of HNBR before (a) and after (b) thermo-peroxide vulcanization.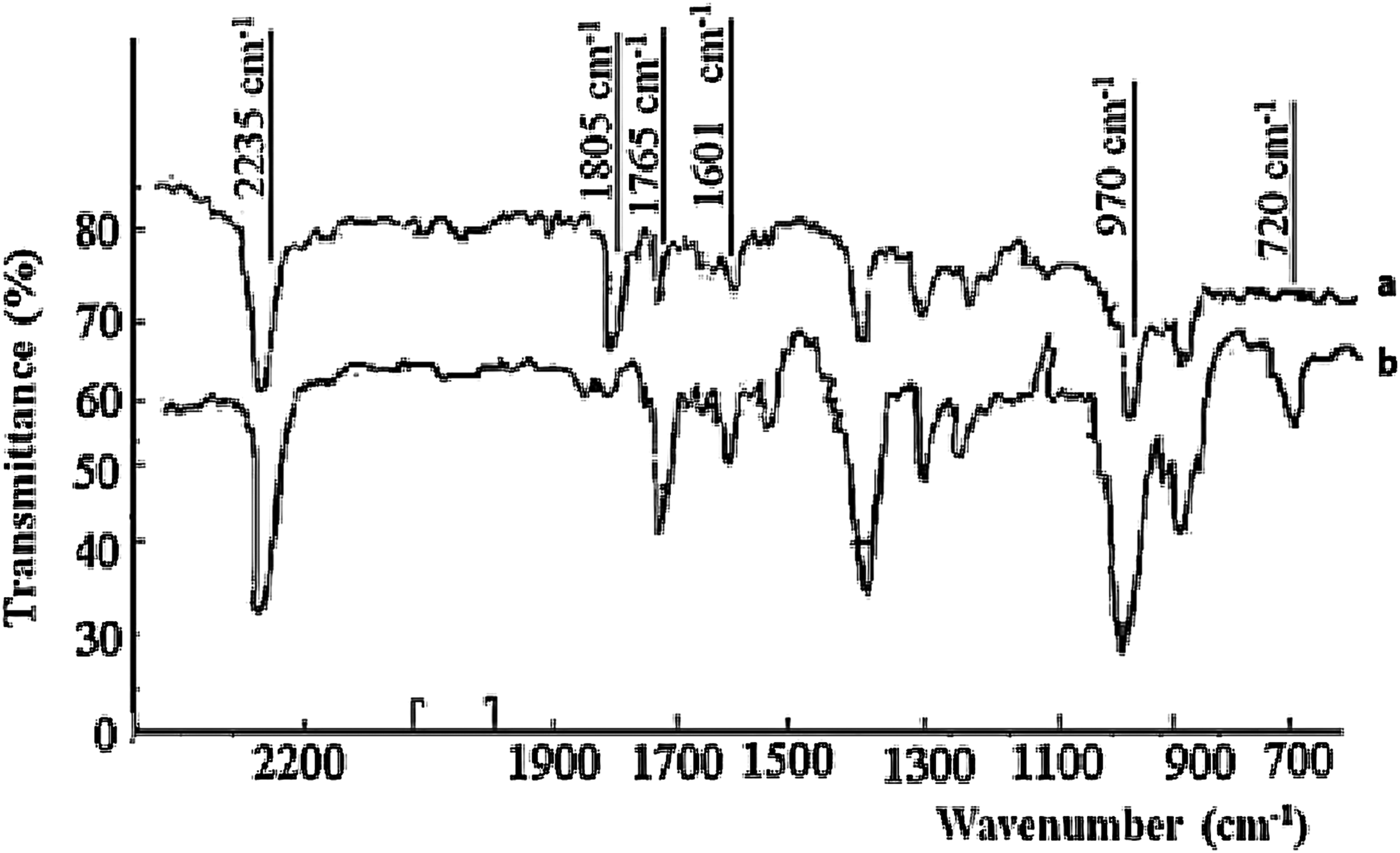
Here are the results obtained from IR Spectroscopy analysis:
Effect of chlorine-containing compound (CCC): HCPX, ABTST on the composition of the vulcanizate during radiolysis (D, 300 kGy).
Effect of crosslinking type on the physical and mechanical properties of vulcanizates filled with carbon black P324 based on HNBR (vulcanizing system 3).
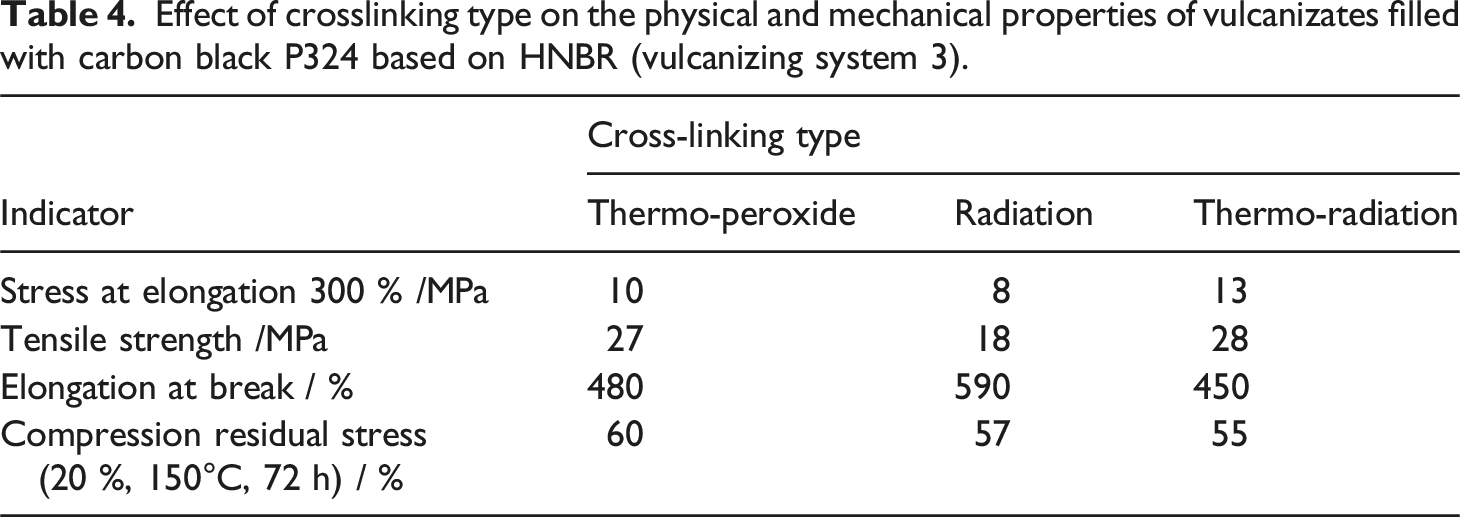
The data obtained indicate that in the presence of the studied vulcanizing systems, peroxide and thermo-radiation processes are most effective. 32 To crosslink saturated systems based on HNBR under conditions of combined exposure, it is advisable to use 0.2 parts by weight of sulphur for the formation of the C-S-C chemical bond in the HNBR molecule and it is more effective under thermo-radiation exposure. The same composition of vulcanizing systems based on these compounds provides nearly identical properties of the samples both under peroxide and thermo-radiation exposure.
Thus, the efficiency of cross-linking compositions based on HNBR using the thermo-radiation cross-linking method depends on the type of cross-linking action, the type of chlorine-containing compound and the composition of the vulcanizing system.33,34 The most effective methods of crosslinking elastomer compositions based on HNBR, ensuring the production of samples with an optimal set of strength properties, are thermo-peroxide and thermo-radiation using the specified crosslinking systems.
Conclusion
The introduction of active low-molecular cross-linking agents (HCPX, ABTST, DP) of various chemical characteristics into the molecular chain of HNBR and their interaction with HNBR ensures radiation cross-linking of hydrogenated butadiene nitrile rubber. The conducted studies made it possible to identify the influence of HCPX, ABTST and DP on the efficiency of radiation vulcanization of HNBR and to establish the structuring action of low molecular weight products. The introduction of zinc oxide, epoxy resin and carbon black into the composition of thermo-peroxide, radiation and thermo-radiation vulcanizates activates cross-linking and the yield of effective cross-links and the number of cross-linked molecules.
The positive results achieved help to observe a change in the molecular structure of HNBR upon irradiation with a 300 kGy radiation dose and the appearance of a new band at 720 cm−1 is associated with vibrations of the C-Cl group, which indicates the participation of chlorine-containing compounds. Additionally, the influence of polymer peroxide was proven by peaks observed at 1805 cm−1 and 1765 cm−1 which related to -CO-RO-CO- and COO bonds.
The use of these low-molecular compounds as vulcanizing agents for HNBR, which determines the structural and physical characteristics, also determines significant relationships between changes in the chemical structure of molecular dynamics and reactivity.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
