Abstract
This study investigates the magnetic, thermal, and electrical properties of Co0.72Sr0.07Ni0.21Fe2O4 ferrite nanoparticles under different conditions, including as-prepared, irradiated (at a dose of 100 kGy in CO2 atmosphere), and aged (at 1000°C). The magnetic properties are analyzed using M-H loops, revealing that the aged sample exhibits the highest magnetization values. The observed decrease in magnetization after irradiation and increase after aging is consistent due to the presence of a new phase (γ-FeOOH) in the irradiated sample that XRD confirms. Electrical conductivity measurements demonstrate that the aging sample exhibits the highest electrical conductivity due to increased grain boundaries, while the irradiated sample shows increased conductivity attributed to oxygen vacancies. As well as the nanocomposite of PVP and Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles aged (at 1000°C) is found to be effective in degrading the Toluidine Blue (TB) dye through catalytic oxidation and photodegradation mechanisms. The catalytic degradation of TB dye provides valuable insights into the potential application of these ferrite nanoparticles in environmental remediation and wastewater treatment. Also, nanocomposite demonstrated significant antimicrobial activity against five pathogenic bacterial strains commonly found in contaminated water, with superior effectiveness against Gram-negative bacteria, particularly Salmonella enterica and Pseudomonas aeruginosa, suggesting its potential as an effective water treatment agent. The novelty and the aims are to analyze the changes in magnetization and conductivity of the nanoparticles under different conditions, including as-prepared, irradiated, and aged samples. Additionally, the catalytic efficiency of the aged nanoparticles in degrading Toluidine Blue (TB) dye is examined, providing insights into their potential application in environmental remediation and wastewater treatment.
Keywords
Introduction
In recent years, there has been a growing interest among scientists and researchers in the synthesis and investigation of efficient and versatile nanomaterials. One such class of nanomaterials that has gained significant attention is the Sr-Co nano ferrite particles, which exhibit remarkable properties and applications in various fields 1 including waste water treatment including electronics, data storage, waste water treatment, 2 photocatalysis, 3 and energy. These nanocrystalline magnetic oxide nanomaterials, specifically the Sr-Co nano ferrites, have shown exceptional sensitivity to electromagnetic waves, particularly in the microwave frequency. They possess high absorption and attenuation capabilities, making them excellent candidates for applications as microwave attenuators. By effectively suppressing electromagnetic waves in the microwave range, these nanomaterials have the potential to revolutionize technologies related to telecommunications, signal processing, and wireless communication. 4 One area where the synthesis of nanocrystalline and soft magnetic Cobalt ferrite particles holds great significance is in the development of advanced industrial and technical applications. These include nano-drug delivery systems, magnetic contrast agents for medical imaging, electromagnetic wave signal suppressors, microwave devices, permanent magnets, improved catalytic activity,5–7 and water treatment technologies.2,8,9 The unique magnetic properties, chemical stability, and biocompatibility make Sr-Co nano ferrite particles highly attractive for these applications. 10 The study of magnetic materials and dielectric properties of green synthesized Ag doped NiFe2O4 spinel ferrite. 11 The surface area (BET) of the sample NiFe2O4 was higher compared to Ni0.3Zn0.7Fe2O4 equal 5 nm. 12 The study of magnetic materials, electrical, and ferroelectric transformation in bismuth doped nanosized cobalt ferrite under the effect of Gamma irradiation. 13 The impact of doping sodium tantalate (NaTaO3) with Cu or Al metal ions on the frequency and temperature dependence of the electrical conductivity σ(f,T) is investigated through complex impedance measurements in the frequency range of 200 Hz–2 MHz and at different temperatures from 30°C to 90°C 14
The study of magnetic materials and their properties has attracted significant attention due to their wide range of applications in various fields, 15 including electronics, data storage, waste water treatment,16–19 medicine, antibacterial,20–22 catalytic,23–25 photocatalysis,26–30 and energy.12,31 Among these magnetic materials, cobalt ferrite CoFe2O4 nanoparticles have emerged as promising candidates for numerous technological advancements. Cobalt ferrite nanoparticles possess excellent magnetic properties, high chemical stability, and biocompatibility, making them suitable for various applications.32–35 In recent years, researchers have been exploring the effect of incorporating different dopants into cobalt ferrite nanoparticles to enhance their properties and tailor their functionality for specific applications. One such dopant is bismuth (Bi), which has shown interesting results in modifying cobalt ferrite nanoparticles’ structural, electrical, and magnetic properties. 36 In addition, it is well known that both Co- and Ni are inverse spinels, that exhibit homogeneous distribution in the A and B lattice sites; thus, their magnetic features are attributed to antiferromagnetic coupling amongst the tetrahedral and octahedral sublattices. 37 Furthermore, nickel substituted cobalt ferrites demonstrate good chemical stability and thus found applications as a binder and as magnetic filler in electromagnetic shielding nanocomposites. 38 Moreover, it is generally anticipated that rare-earth substituted spinel ferrites, in many cases, would show a superior electric and magnetic character when compared with the pristine spinel counterpart. 39
However, besides chemical doping, irradiation is another approach to modifying the properties of magnetic nanoparticles. Gamma irradiation, as a powerful and controlled source of ionizing radiation, can induce significant changes in the structure and properties of materials. Subjecting cobalt ferrite nanoparticles to gamma irradiation makes it possible to manipulate their properties and explore the potential for improved performance in various applications.
40
Radiolysis of CO2 and H2O can be induced reactive species such as (CO2→ CO,CO3,CO+
Studies have demonstrated that PVP encapsulated and gamma-irradiated ferrite nanoparticles exhibit excellent catalytic activity for dye degradation, achieving unprecedented dye removal rates of up to 95–99% in certain cases. This underscores the great potential of this methodology. Certainly, PVP encapsulation is critical in developing highly efficient ferrite-based nanocatalysts for treating dye-contaminated wastewater and other pollutants. By optimizing the polymer coating and gamma irradiation dose, it is possible to create stable, reusable, and non-leaching nanocatalysts with unusual catalytic activity. Catalytic degradation of dyes has gained significant attention due to the environmental concerns associated with their release into water bodies. Various strategies have been explored to develop efficient and eco-friendly catalysts for dye degradation, including nanocomposites. One such nanocomposite of interest is the (PVP/Co0.72Sr0.07Ni0.21Fe2O4) nanocomposite, which has shown promising catalytic activity in the degradation of dyes. The (PVP/Co0.72Sr0.07Ni0.21Fe2O4) nanocomposite is a composite material consisting of polyvinylpyrrolidone (PVP) and spinel Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles. The PVP component acts as a stabilizer, preventing the aggregation of nanoparticles and providing a favorable environment for catalytic reactions. The Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles, synthesized through a high-temperature annealing process at 1000°C, possess desirable properties for catalytic applications. The catalytic activity of the (PVP/Co0.72Sr0.07Ni0.21Fe2O4) nanocomposite has been investigated for the degradation of TB dyes, a commonly used textile dye, is known for its high stability and resistance to degradation. Therefore, finding an effective and efficient catalyst for its degradation is of great importance. 56 Water contamination by pathogenic bacteria poses significant health risks globally, particularly concerning organisms like Escherichia coli, 57 Salmonella enterica, 58 Pseudomonas aeruginosa, 59 Shigella flexneri, 60 and Enterococcus faecalis. 61 These bacteria can cause severe gastrointestinal illnesses, infections, and other health complications, especially in vulnerable populations. Developing effective antimicrobial treatments for water purification is therefore crucial for public health protection and disease prevention. 62
In this study, we evaluate the catalytic performance of the (PVP/Co0.72Sr0.07Ni0.21Fe2O4) nanocomposite in the degradation of TB dye under UV light. We investigate the adsorption kinetics of TB dye onto the nanocomposite using pseudo-first-order and pseudo-second-order kinetic models. Additionally, we explore the isotherm models, such as Langmuir and Freundlich, to understand the adsorption behavior of TB dye on the nanocomposite. Generally, the (PVP/Co0.72Sr0.07Ni0.21Fe2O4) nanocomposite demonstrates good catalytic activity for the degradation of TB dye. The enhanced adsorption capacity and good adsorption process can be attributed to the chelation between the TB dye and the nanocomposite’s oxygen- and nitrogen-containing groups.
Furthermore, their effect on deactivation pathogenetic bacteria is such that they are both Gram-negative and Gram-positive bacteria. The test organisms include four Gram-negative bacteria: Escherichia coli (ATCC25922), Shigella flexneri (ATCC 12022), Salmonella enterica (ATCC25566), and Pseudomonas aeruginosa (ATCC 27853), along with one Gram-positive bacterium, Enterococcus faecalis (ATCC 19433). 63 These strains are selected as representative pathogenic bacteria commonly found in contaminated water. This research contributes to developing efficient and sustainable catalysts for dye degradation, addressing the environmental concerns associated with dye pollution.
Experimental
Materials
Cobalt(II) chloride (CoCl2) (CAS Number:7646-79-9, Purity:99.995%), Strontium chloride hexahydrate (SrCl2.6H2O) (CAS Number:10025-70-4, Purity:99.995%), Nickel chloride hexahydrate (NiCl2.6H2O) (CAS Number:7791-20-0, Purity:99.9%), Iron(III) chloride (FeCl3) (CAS Number:7705-08-0, Purity:99.99%), Sodium hydroxide (NaOH) (CAS Number:12200-64-5, Purity:97%), PVP (CAS Number:9003-39-8) was purchased from Sigma Aldrich Co. 64
Preparation of Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles
The preparation of Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles by the chemical co-precipitation method is as follows. Dissolve stoichiometric amounts of CoCl2, SrCl2.6H2O, NiCl2.6H2O, and FeCl3 in distilled water. The amounts used are based on the desired composition of Co0.72Sr0.07Ni0.21Fe2O4. Maintain the solution at 10°C and stir constantly using a magnetic stirrer for 1 h. Monitor the pH of the mixed solution and gradually add NaOH solution drop by drop until the pH reaches 12. 65 This is done to achieve the desired pH for precipitation. Once the pH reaches 12, heat the solution and maintain it at 90°C for 2 h under continuous stirring. This step promotes the formation of the desired nanoparticles. After the reaction time, the sediments formed in the solution must be separated. Thoroughly wash the precipitates with distilled water until the washings become free from sodium chloride. This washing step is crucial to remove impurities and unwanted by-products. After washing, the residues are dried for a few days at room temperature. This allows the water to evaporate, leaving behind the solid nanoparticles. 66 Once dried, the samples are ground thoroughly in an agate mortar to obtain an ultra-fine powder. This step helps achieve a finer particle size and better homogeneity.
Following these steps, the resulting product should be Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles. Spinel Co0.72Sr0.07Ni0.21Fe2O4 nano ferrites were prepared by the chemical co-precipitation method according to the equation; 0.72 CoCl2 (l) + 0.07 SrCl2.6H2O(l) + 0.21 NiCl2.6H2O(l) + 2 FeCl3(l) + 8 NaOH(l)
These nanoparticles can be further characterized and used for various applications, 67 such as magnetic materials, 68 catalysts, 69 or biomedical applications. 70
Exposure of Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles to gamma irradiation at a dose of 100 kGy in CO2 atmosphere using Co-60 cell
The appropriated weight Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles powder is exposed to gamma irradiation in CO2 atmosphere at a dose of 100 kGy using a Co-60 cell at ambient conditions, several effects can be expected. Here are some potential outcomes examined in this article, such as (i) crystal structural changes performed by (XRD) analysis: Gamma irradiation at such a high dose can induce significant structural modifications in the nanoparticles. It can cause lattice defects, such as vacancies, interstitials, or dislocations, and may lead to changes in the crystal structure or phase transformation. The irradiation can also result in grain growth or aggregation of nanoparticles. (ii) Size and morphology changes performed by (TEM) analysis: The irradiation can affect the size and morphology of the nanoparticles. High doses of gamma radiation can lead to particle growth, aggregation, or fragmentation, altering the nanoparticles’ size, distribution and shape. (iii) Magnetic properties were performed by (VSM) analysis: Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles exhibit magnetic behavior, and gamma irradiation can impact their magnetic properties. The irradiation-induced structural changes can influence the magnetic interactions between atoms, affecting parameters like saturation magnetization, coercivity, and magnetic anisotropy. (iv) Thermal stability and the activation energy were performed by (TGA) analysis. Gamma irradiation may influence the thermal stability of the nanoparticles. It can cause changes in the onset temperature, weight loss rate, or decomposition extent compared to the non-irradiated samples. These changes can indicate radiation-induced structural modifications or defects affecting thermal behavior. (v) The optical properties and band was performed by uv-vis spectrophotometer; The effect of gamma irradiation on the optical properties and band gaps of Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles can be studied using UV-Vis spectrophotometry. This technique measures the absorption or reflection of light by a sample over a range of wavelengths in the ultraviolet (UV) and visible (Vis) regions of the electromagnetic spectrum. Gamma irradiation can affect the energy levels and band structure of the nanoparticles. A shift in the absorption edge or changes in the absorption onset wavelength can provide insights into alterations in the band gap or electronic transitions due to irradiation-induced structural modifications.
A comprehensive characterization would be necessary to fully evaluate the effects of gamma irradiation at 100 kGy on Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles. Techniques such as X-ray diffraction (XRD), transmission electron microscopy (TEM), vibrating sample magnetometry (VSM), and UV-Vis spectrophotometer (UV-Vis) can provide valuable insights into the structural, morphological, magnetic, and chemical changes induced by the irradiation. It’s important to conduct a comprehensive study that would provide more precise insights into the effects of gamma irradiation on Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles powder, such as the effect of the aging process.
Aging Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles at a temperature of 1000 C
Aging Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles at a temperature of 1000°C was carried out by heating the appropriate weight of nanoparticles in a muffle furnace at a temperature of 100 C for 2 h. Various effects can be observed, such as; (i) Phase transformation was performed by (XRD) analysis: At high temperatures, phase transitions may occur in Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles. Depending on the composition and conditions, the nanoparticles may undergo crystal structure changes, such as spinel-to-cubic or phase segregation. (ii) Grain growth of nanoparticles can be performed by (TEM): Elevated temperatures can promote grain growth in the nanoparticles. This can lead to an increase in the average particle size and changes in the size distribution. The nanoparticles can fuse, forming larger aggregates or even bulk-like structures. Larger particles may exhibit altered magnetic, electrical, or optical properties compared to the initial nanoparticles. (iii) Chemical structure change was performed by UV-Vis spectrophotometer: The high temperature can induce chemical reactions within the nanoparticles, forming new compounds or reacting with the surrounding environment. This can change the nanoparticles’ stoichiometry, surface chemistry, or oxidation states. (iv) Magnetic properties were performed by vibrating sample magnetometry (VSM): sintering at 1000°C, Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles may experience agglomeration or fuse, forming larger aggregates or even bulk-like structures. This process can affect the specific surface area and may influence the magnetic, electrical, or catalytic properties.
To fully understand the effects of aging Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles at 1000°C, it is important to perform a comprehensive characterization using techniques such as X-ray diffraction (XRD), scanning electron microscopy (SEM), transmission electron microscopy (TEM), energy-dispersive X-ray spectroscopy (EDS), vibrating sample magnetometry (VSM) and various spectroscopic methods. These techniques can provide insights into the nanoparticles’ structural, morphological, chemical, and electronic changes.
Characterization
Transmission electron microscope (TEM) images
The average nanoparticle size was determined using JEOL JEM–100 SX transmission electron microscope (TEM). The sample powder was first exposed to ultrasonic waves for 45 min to separate the nanoparticles from each other. Sample Preparation: Prepare a thin sample of Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles suitable for TEM analysis. This typically involves dispersing the nanoparticles in a suitable solvent and depositing a drop of the suspension onto a TEM grid, a thin support film. Instrument Setup: Place the prepared TEM grid onto the TEM sample holder and load it into the TEM instrument. Ensure the instrument is set to the appropriate operating conditions, including the desired electron beam voltage and imaging mode (bright-field or dark-field). Imaging: Use the TEM instrument to acquire images of the Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles. In bright-field imaging, the transmitted electrons create a bright image on a dark background, allowing the nanoparticles’ contrast to be visualized. Dark-field imaging uses scattered electrons, providing contrast based on the nanoparticle diffraction pattern. Measure the size of the nanoparticles using TEM images. This can be done manually by measuring the particle diameter or employing image analysis software to automate the process. Statistical analysis of the particle size distribution can provide insights into the uniformity of the nanoparticles. TEM analysis of Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles can provide crucial information about their structural and morphological characteristics, aiding in understanding their properties and potential applications in various fields, including catalysis, energy storage, and magnetic devices.
Thermal analysis (TGA)
This study conducted simultaneous thermo-gravimetric analysis (TGA) on the nano ferrite Co0.72Sr0.07Ni0.21Fe2O4 samples. The TGA experiments were performed using a Perkin-Elmer STA 6000 thermal analysis system under a N2 atmosphere. The samples were subjected to a heating rate of 15°C/min. TGA is a technique used to measure a sample’s weight change as a function of temperature. It provides information about materials’ thermal stability, decomposition, and weight loss. By analyzing the weight change of the Co0.72Sr0.07Ni0.21Fe2O4 samples as they are heated, we can gain insights into the thermal behavior and decomposition characteristics of the nanoparticles. Using a N2 atmosphere during TGA helps prevent oxidation or other reactions that may occur in the presence of air. This ensures accurate measurement of the weight loss of the nano ferrite samples without interference from external factors.
The Perkin-Elmer STA 6000 thermal analysis system is a widely used instrument for TGA experiments. It provides precise control over the heating rate and accurate measurement of weight changes, allowing for reliable analysis of the thermal properties of materials. By conducting TGA on the nano ferrite Co0.72Sr0.07Ni0.21Fe2O4 samples, we can obtain crucial information about their thermal stability and behavior, contributing to a comprehensive understanding of their properties and potential applications.
The X-ray powder diffraction (XRD)
The X-ray powder diffraction (XRD) spectra of Co0.72Sr0.07Ni0.21Fe2O4 samples were obtained using a Bruker AXS D8-advance X-ray diffractometer. This instrument is specifically designed for XRD analysis and is commonly used for material characterization and phase identification. The X-ray source used in this instrument is Cu Ka radiation, with a wavelength of 1.5418 Å (angstroms). By utilizing the Bruker AXS D8-advance X-ray diffractometer with Cu Ka radiation, researchers can obtain detailed information about the analyzed materials’ crystal structure, phase composition, and lattice parameters. The XRD spectra collected using this instrument enable the identification and characterization of various crystalline materials, contributing to understanding their physical and chemical properties.
Vibrating-sample magnetometer (VSM) analysis
The magnetic properties of the Co0.72Sr0.07Ni0.21Fe2O4 samples were measured using a BHV-55 vibrating sample magnetometer (VSM). The BHV-55 VSM is a commonly used instrument for characterizing magnetic materials. It allows for precisely measuring magnetic properties such as magnetization, coercivity, and magnetic susceptibility. The BHV-55 VSM operates based on the principle of vibrating sample magnetometry. The sample, typically a small solid or powder, is placed on a sample holder and subjected to an oscillating magnetic field. The magnetic response of the sample is measured by detecting the changes in the induced voltage caused by the sample’s magnetization.
AC conductivity excrement setup
To perform AC conductivity measurements of the Co0.72Sr0.07Ni0.21Fe2O4 samples can be performed by applying an AC voltage signal to the sample through the electrodes and measuring the resulting current using the impedance spectrometer based on our previous work. 71
Synthesis of (PVP/Co0.72Sr0.07Ni0.21Fe2O4) nanocomposite and photodegradation setup
Sonochemical and gamma irradiation synthesis was employed as the experimental method for preparing the (PVP/Co0.72Sr0.07Ni0.21Fe2O4) nanocomposite catalyst. The synthesis involved the following steps: A 7% PVP solution was prepared by dissolving 7 grams of polyvinylpyrrolidone (PVP) in a water solvent and subjected to sonication using a sonication probe. The sonication process was carried out for 30 min, during which high-frequency sound waves were applied to the solution. By subjecting 0.5 g of Co0.72Sr0.07Ni0.21Fe2O4 treated with 1 mL of 3-mercaptopropionic acid to sonication, the PVP molecules can effectively encapsulate the Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles, resulting in the formation of a nanocomposite catalyst (PVP/Co0.72Sr0.07Ni0.21Fe2O4). The sonication process helps achieve a uniform dispersion of the nanoparticles within the PVP matrix, leading to improved catalytic properties. The solution of (PVP/Co0.72Sr0.07Ni0.21Fe2O4) nanocomposite catalysts was subject to γ-ray at the dose of 30 kGy, then filtrated with washing several times and dried in an oven at a temperature of 50°C. PVP enables the enhanced catalytic activity of Co0.72Sr0.07Ni0.21Fe2O4 because PVP has a high affinity for dyes and can adsorb onto their surfaces. This adsorption process helps immobilize the dye molecules onto the catalyst surface, forming a stable dye-catalyst complex. The adsorption of the dye by PVP enhances the adherence of the dye molecules to the catalyst particles, ensuring a high degree of contact between the dye and the Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles. By adhering the dye molecules to the catalyst’s surface, PVP improves the stability of the dye-catalyst system and enhances the catalytic activity. The proximity of the dye molecules to the Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles facilitates the efficient transfer of reactants, promotes the generation of reactive species, and increases the general efficiency of the catalytic degradation process.
Furthermore, the adsorption of the dye by PVP can prevent re-aggregation or re-deposition of the dye molecules, ensuring their continuous exposure to the catalytic sites on the Co0.72Sr0.07Ni0.21Fe2O4 surface. This promotes a sustained and efficient degradation of the dye. The presence of PVP in the PVP/Co0.72Sr0.07Ni0.21Fe2O4 nanocomposite not only acts as a stabilizer and dispersant but also enhances the catalytic activity by adsorbing the dye and promoting its adherence to the catalyst surface. This synergistic effect between PVP and Co0.72Sr0.07Ni0.21Fe2O4 contributes to the improved degradation efficiency of the TB dye. In the experimental part, the TB degradation process using PVP/Co0.72Sr0.07Ni0.21Fe2O4 nanocomposite catalyst was analyzed using the first and second pseudo-order reaction models to determine the reaction kinetics, additionally, based on our previous work 72 The Langmuir and Freundlich adsorption models were employed to investigate the adsorption isotherms and understand the adsorption behavior of TB dye on the nanocomposite catalyst. All the experimental studies in this article are conducted in the Faculty of Science and Humanities - Jubail, Imam Abdulrahman Bin Faisal University, Jubail, Saudi Arabia and the Department of Chemistry, College of Science, Northern Border University (NBU), Arar, Saudi Arabia.
Evaluation of (PVP/Co0.72Sr0.07Ni0.21Fe2O4) to deactivated pathogenic bacteria in water
The antimicrobial activity evaluation of PVP/Co0.72Sr0.07Ni0.21Fe2O4 nanocomposite begins with sample preparation. The nanocomposite is dissolved in 3.0 mL DMSO to achieve a 40 mg/mL concentration. This stock solution serves as the primary test material for the antimicrobial assays. The experimental setup requires two types of growth media: nutrient agar and nutrient broth. Both media contain identical base ingredients including yeast extract (2.0 g/L), peptone (5.0 g/L), meat extract (1.0 g/L), and NaCl (5.0 g/L), maintained at pH 7.4 ± 0.2. The nutrient agar medium additionally contains 15.0 g/L of agar for solidification purposes. The antimicrobial activity testing protocol is designed to evaluate effectiveness against both Gram-negative and Gram-positive bacteria. The test organisms include four Gram-negative bacteria: Escherichia coli (ATCC25922), Shigella flexneri (ATCC 12022), Salmonella enterica (ATCC25566), and Pseudomonas aeruginosa (ATCC 27853), along with one Gram-positive bacterium, Enterococcus faecalis (ATCC 19433). These strains are selected as representative pathogenic bacteria commonly found in contaminated water. The experimental procedure employs the well diffusion method according to established protocols. 73 Fresh bacterial cultures are prepared in nutrient broth and incubated at 37°C. The bacterial inoculum is standardized by adjusting to match the 0.5 McFarland standard, corresponding to approximately 1.5 × 108 CFU/mL. For each test, precisely 75.0 µL of the standardized bacterial suspension is inoculated onto nutrient agar plates containing 20.0 mL of medium. The well diffusion technique involves creating wells of 0.9 cm diameter in the inoculated agar plates using a cork borer. Each well receives 75.0 µL of the nanocomposite solution. To ensure proper diffusion of the test compound, the plates are kept in a refrigerator for 1 hour before incubation. Following this diffusion period, the plates are incubated at 37°C for 24 h under controlled conditions. Quality control measures include using Gentamicin (10 mcg) as a reference antibiotic standard for comparison. The antimicrobial activity is quantified by measuring the zones of inhibition (ZI) in millimeters around each well after incubation. All experiments are conducted under sterile conditions, carefully maintaining consistent pH, temperature, and other environmental factors throughout the testing period. This standardized methodology allows for quantitative assessment of the nanocomposite’s antimicrobial properties while ensuring the reproducibility and reliability of the experimental results. The protocol follows established microbiological testing procedures as referenced in the current literature74,75
Results and discussion
Effect of gamma irradiation and aging temperature on the physiochemical properties of Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles
The effect of gamma irradiation (100 kGy) and aging temperature (1000°C) on the physiochemical properties of Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles compared to the as-prepared sample can provide valuable insights into their stability, structural changes, magnetic behavior, and potential applications. We can better understand these effects by examining various characteristics of the nanoparticles before and after the irradiation process and aging at 1000°C. Some of the properties to study include:
Some of the properties that can be investigated include. Investigate the stability of the Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles by assessing any changes in their physical and chemical properties after gamma irradiation and aging. This can include analyzing changes in particle size, morphology, crystal structure, and phase composition. Examine the structural changes induced by gamma irradiation and aging temperature. Investigate any surface modifications induced by irradiation and aging. Surface analysis techniques such as transmission electron microscope (TEM). Techniques such as X-ray diffraction (XRD) can be employed to analyze any alterations in the crystal lattice, shifts in peak positions, and changes in peak intensities. Study the magnetic properties of the Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles before and after irradiation and aging. Magnetic measurements, such as vibrating sample magnetometry (VSM), can help determine magnetization, coercivity, and magnetic ordering changes. Assess the thermal stability of the nanoparticles by examining changes in their thermal behavior, such as thermal decomposition or phase transitions, after irradiation and aging. Thermal analysis techniques like thermogravimetric analysis (TGA) can be employed.
TEM analysis of Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles
TEM analysis of Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles can provide crucial information about their structural and morphological characteristics, aiding in understanding their properties and potential applications in various fields, including catalysis, energy storage, and magnetic devices. The morphology of the as-prepared Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles, sintered at 1000°C and irradiated at a dose of 100 kGy is clearly shown in Figure 1. It is depicted that the nanoparticles as-prepared are agglomerated, which may point to the formation of ordered nanoclusters, as shown in Figure 1(a). The aggregation of Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles, as observed in Figure 1(a) could be attributed to their magnetic properties. Magnetic nanoparticles attract and aggregate due to magnetic forces between the particles.
76
These magnetic interactions can cause the nanoparticles to form ordered nanoclusters or agglomerates. Magnetic properties in the nanoparticles can play a significant role in their self-assembly and aggregation behavior. The aggregation of magnetic nanoparticles can have positive and negative effects depending on the application. In Magnetic Particle Imaging (MPI) context, the aggregation of magnetic nanoparticles can lead to signal loss and reduced sensitivity. MPI relies on detecting the magnetization response from the nanoparticles, and large aggregates can hinder the generation and detection of magnetic signals, thereby affecting imaging performance.
77
However, the aggregation of magnetic nanoparticles can benefit drug delivery applications. Aggregated nanoparticles can enhance the targeting and retention of the drug at the desired site of action. The aggregates’ larger size and increased magnetic moment can facilitate their accumulation in specific regions, improving the localized drug delivery efficiency.
78
It is important to note that controlling and optimizing the aggregation behavior of magnetic nanoparticles is crucial in different applications. Strategies such as surface modifications, controlling the particle size, and utilizing external magnetic fields can be employed to tailor the aggregation properties of magnetic nanoparticles for specific purposes. Therefore, understanding the aggregation behavior of magnetic nanoparticles is crucial for their successful application in various fields. TEM images of (a) the as-prepared (AP) spinel Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles and (b) the Co0.72Sr0.07Ni0.21Fe2O4 nano ferrite particles aging at 1000°C (AG) and (c,d) the Co0.72Sr0.07Ni0.21Fe2O4 nano ferrite particles irradiated at 100 kGy (Ir).
On the other hand, Figure 1(b) shows the TEM image of Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles aged at (1000°C). Based on the TEM analysis, it is observed that the aggregation of the aging ferrite nanoparticles (65.43 nm) is larger compared to the as-prepared sample (21.31 nm). This difference in agglomeration size suggests that the aging process at a temperature of 1000°C has led to the formation of larger particle clusters or agglomerates. The increase in average nanoparticle size upon aging suggests that the nanoparticles undergo a growth process during the aging treatment. 79 The higher temperature of 1000°C likely promotes particle coalescence and sintering, forming larger agglomerates. This phenomenon can be attributed to the increased mobility of particles at higher temperatures, allowing for their rearrangement and fusion. The aggregation and increased particle size can have implications for the properties and performance of the nanoferrite material. 80 It may affect parameters such as magnetic properties, surface area, and reactivity. Understanding and controlling the agglomeration behavior is important for optimizing the properties and applications of magnetic nanoparticles, as it can impact their functionality and performance in various fields such as catalysis, magnetic recording, and biomedical applications. In Figure 1(c) and (d), for irradiated sample at 100 kGy the cubic morphologies of irradiated Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles with size of (40–55 nm) are observed, and it is noted that there is less aggregation compared to the aging sample. This indicates that the gamma irradiation has not only influenced the morphology but also reduced the extent of particle aggregation. The reduced aggregation of irradiated nanoparticles can be attributed to the radiation-induced effects on the surface chemistry and particle interactions. Gamma irradiation can lead to the generation of reactive species, such as free radicals and charged particles, which can modify the surface properties of the nanoparticles. These modifications may result in improved dispersion and reduced particle-particle interactions, leading to less aggregation.
Observing cubic morphology and reduced aggregation in irradiated Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles is significant as it suggests the potential for improved dispersibility and stability of the nanoparticles in various applications. The reduced aggregation can enhance the performance of the nanoparticles in areas such as catalysis, magnetic resonance imaging, or biomedical applications where dispersion and uniformity are crucial. However, further characterization and investigation are necessary to understand the underlying mechanisms and fully exploit the advantages of the cubic morphology and reduced aggregation in irradiated Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles.
XRD analysis of Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles
Figure 2 shows the X-ray diffraction (XRD) pattern of Co0.72Sr0.07Ni0.21Fe2O4 as prepared, aging at a temperature of 1000°C and irradiated by gamma rays at dose of 100 kGy. The XRD of blank Co0.72Sr0.07Ni0.21Fe2O4 sample as shown in Figure 2(a) matches the ferrite cubic spinel structure (ICDD n°98-002-8108). The crosspoding XRD peaks located at 2θ of 13.16, 30.4o, 35.49o, 36.20o, 41.38o, 43.5, 53.42o, 58.48o and 62.90o and 75.18o. Conversely, after aging at a temperature of 1000°C the XRD in Figure 2(b) exhibits very sharp and intensity peaks due to increasing size and crystallization of Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles which agree with the TEM observations in Figure 1(b). The X-ray diffraction patterns of aging sample are located at 2θ = 20.96o, 30.29o, 32.56o, 34.37o, 35.62o, 37.33o, 43.28o, 47.03o, 47.44o, 57.07o and 62.84o. As seen, the XRD peaks compared to Figure 2(a) is made some shift in 2θ value after aging and new XRD peaks were observed at 2θ = 20.96o, 32.56o, 34.37o, 37.33o, 43.28o, 47.03o and 47.44o, that could attribute to the phase transition induced by aging temperature (1000°C) and appeared of new phases. XRD images of (a) the as-prepared (AP) spinel Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles and (b) the Co0.72Sr0.07Ni0.21Fe2O4 nano ferrite particles aging at 1000°C (AG) and (c) the Co0.72Sr0.07Ni0.21Fe2O4 nano ferrite particles irradiated at 100 kGy (Ir).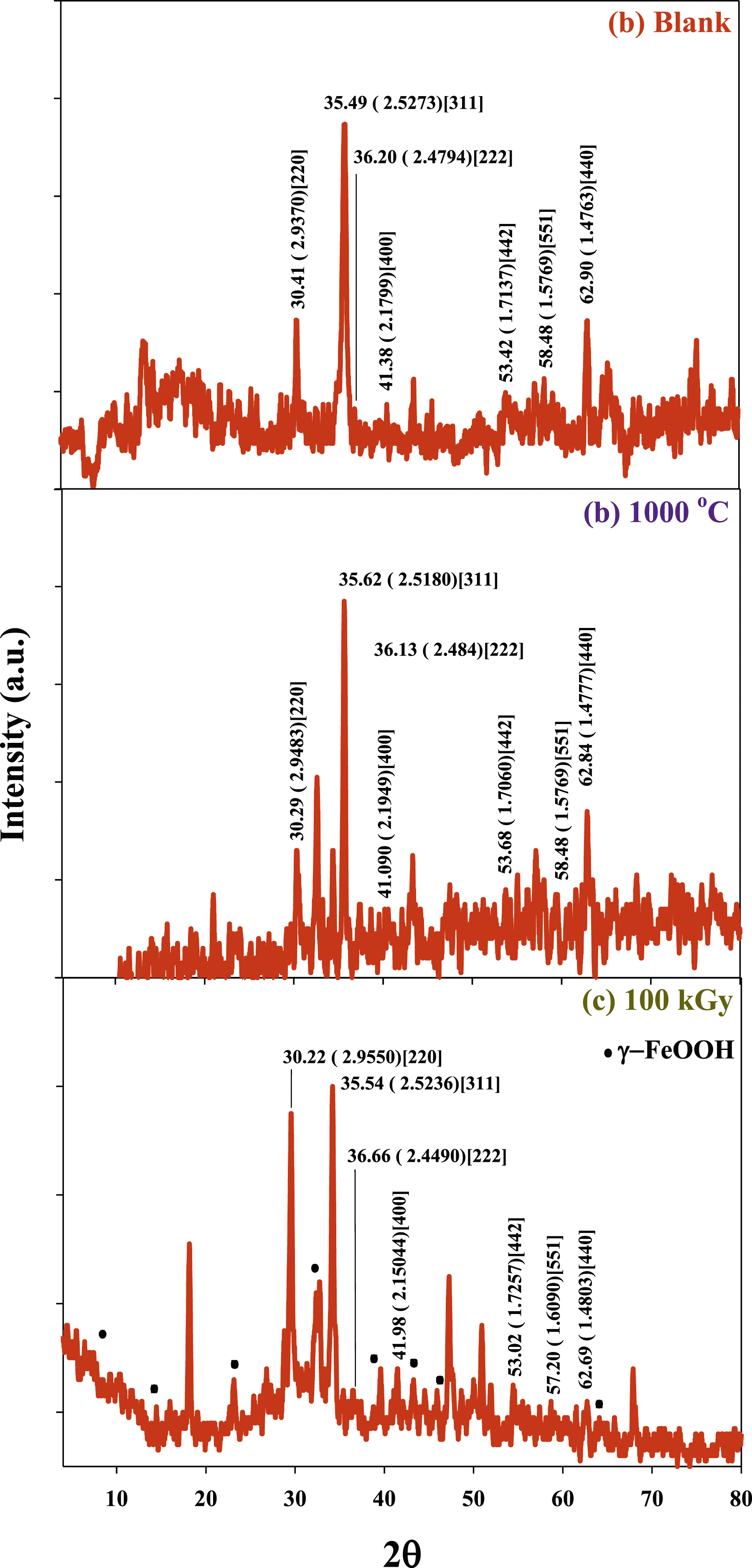
The observed shifts in the peak positions indicate a change in the lattice parameters and crystal structure. The peak positions of the aged (AG) sample, such as 2θ = 20.96°, 32.56°, 34.37°, 37.33°, 43.28°, 47.03°, and 47.44°, are different from the corresponding peaks in the XRD pattern of the blank sample (Figure 2(a)). The appearance of new peaks at specific 2θ values indicates the presence of new phases or the crystallization of additional crystalline components in the (AG) nanoparticle sample. The changes occurring during the aging process are observed in further literature such as by Huixia et al., 19 he observed that the crystallite size of cobalt ferrite increased with increasing aging and calcination temperature. The increase in crystallite size can be attributed to several factors. Firstly, at high temperatures, the diffusion of atoms becomes more favorable, leading to the rearrangement and recrystallization of the material. This allows the crystallites to grow and merge, resulting in larger crystallite sizes. Secondly, the high-temperature treatment can induce sintering, where the particles undergo interparticle bonding and consolidation. This process can lead to the aggregation and growth of the nanoparticles, contributing to the increase in crystallite size.
Figure 2(c) shows the XRD patterns of irradiated Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles samples by gamma rays (100 kGy) that exhibited a slight shift of the XRD peak position that indicated the cubic spinel structure of blank samples in Figure 2(a) the 2θ = 18.4°, 20.96°, 22.77°, 30.29°, 30.54°, 32.56°, 33.23°, 34.37°, 35.11°, 35.62°, 38.63°, 39.52°, 40.10°, 43.03°, 47.03°, 49.2°, 47.34°, 53.68°, 54.96°, 59.33°, 62.84°, 65.14°, 66.08° and 77.25°. Comparing Figure 2(a) and (c), some peak shifts indicate the influence of gamma irradiation on the crystal structure. However, some peaks in Figure 2(c) coincide with the XRD pattern observed after aging in Figure 2(b), suggesting that gamma irradiation’s effect is similar to aging at a high temperature. Furthermore, a new 10 peaks are observed at 2θ = 8.3°, 12.7, 15.8°, 22.8°, 25.1, 33.4°, 39.2°, 42.4°, 46.4°, 53.8°, 57.2° and 63.82°, which are attributed to the presence of γ-FeOOH.81–83 In addition, some XRD peaks are associated to several phases of FeOOH, such as α-FeOOH (goethite) giving XRD peaks at 2θ = around 12.0°, 24.1°, 33.1°, 39.7°, 41.1°, and 61.5°. as well as, β-FeOOH (akaganeite) give XRD peaks at 2θ = around 12.2°, 24.3°, 29.5°, 33.0°, 35.6°, and 61.6° suggests the coexistence of multiple phases of FeOOH in the sample. Several kinds of the literature suggest that the irradiation process promotes the formation or stabilization of the γ-FeOOH phase.84,85 This explains why the γ-FeOOH is domaine in Figure 2(c).
Gamma irradiation can cause the generation of solvated electrons, H • and oxygen radicals with hydroxyl radicals, which act as strong reducing and oxidizing agents, respectively. Solvated electrons, H • undergoes competitive reactions with oxygen and hydroxyl radical. In the case of Fe(III) ions, solvated electrons generated by gamma irradiation can effectively reduce them to Fe(II) ions, as observed in the study by Dano et al. 86 However, due to the irradiation process being carried out in the solid phase so the generation of solvated electrons, hydrogen radicals (H•), and hydroxyl radicals (•OH) is limited compared to oxygen radicals.87–89 In solid-phase irradiation, the dominant species generated are oxygen radicals from the atmosphere, which have a higher concentration than solvated electrons and hydroxyl radicals. These oxygen radicals can act as strong oxidizing agents and tend to oxidize the Fe(II) ions back to Fe(III) ions. Therefore, in the absence of solvated electrons and with a higher concentration of oxygen radicals, the oxidation of Fe(II) ions to Fe(III) ions is more pronounced. This cyclic process of reduction and oxidation can lead to the formation of γ-FeOOH, which is a hydrated form of iron(III) oxide.84,90
In radiation chemistry, it is indeed recognized that solvated electrons and holes can undergo relaxation or solvation processes. Like electrons, holes can be trapped or localized to different degrees before undergoing full adiabatic solvation. The concept of holes in radiation chemistry refers to the absence of an electron in an atomic or molecular orbital, resulting in a positive charge. When a molecule or material is irradiated, energy is transferred to the system, creating electron-hole pairs. These holes can migrate through the material and participate in various chemical reactions. The relaxation or solvation of holes involves their interaction with surrounding solvent molecules or other species in the system. This interaction can influence the reactivity and fate of the holes. The various reported values of Vo (formation volumes) often describe the degree of trapping or solvation of holes before they reach a fully solvated state. It is important to note that the solvation processes and behavior of holes can be complex and depend on various factors, such as the nature of the material, solvent, and surrounding environment. Further research is ongoing to understand the dynamics and mechanisms of hole solvation in radiation-chemical processes. 91 Solvated electrons are produced in all ionizing radiation processes such as gamma irradiation and electron beams, and their radiation chemistry depends on their energy, state of solvation, and the nature of the medium, which influence the activity of solvated electrons such as temperature, pressure, and solvent properties. The solvated electron is a key species in radiation chemistry, electron transfer processes, redox phenomena, electrochemistry, and photochemistry. Due to the short lifetime and low concentration of solvated electrons, their structure is accessible only by transient measurements. 92 Without stabilizing groups or molecules, solvated electrons and holes formed during the ionization process will not be effectively stabilized. Solvated electrons have a short lifetime and low concentration, making them highly reactive and prone to reactions with other species in the medium. Similarly, holes can also undergo relaxation or solvation processes, but their stabilization depends on the presence of suitable stabilizing agents or reaction partners. Without stabilization, solvated electrons and holes can react with nearby molecules, leading to various chemical reactions, including radical formation, bond dissociation, and oxidation/reduction processes. These reactions can have important implications in radiation chemistry, as they contribute to the generally radiation-induced chemical changes in the system. 93 Therefore, the absence or presence of solvated electrons plays a crucial role in radiation-chemical processes, and their behavior depends on various factors. Some key factors that influence the behavior of solvated electrons in radiation-chemical processes such as (i) The energy of the radiation source determines the initial formation of solvated electrons. Higher energy radiation can lead to the generation of more solvated electrons. (ii) The solvation state of solvated electrons refers to the degree of trapping or stabilization by surrounding solvent molecules. Solvated electrons can exist in different solvation states, ranging from weak to fully solvated, depending on the medium and conditions. (iii) The nature of the medium in which the radiation-chemical process occurs, such as the solvent properties (polarity, viscosity, dielectric constant), temperature, and pressure, can influence the behavior of solvated electrons. These factors can affect the solvation and reactivity of solvated electrons. (iv) Solvated electrons can interact with various molecules and species in the medium. The nature and concentration of these reaction partners can influence the reactivity and selectivity of solvated electrons. Solvated electrons can participate in redox reactions, electron transfer processes, and other chemical transformations. Here, tha lack of solvated electrons leads to the formation of a new phase introduced of γ-FeOOH crystal leading to the intensity increase of the peaks (220), (222) and (400). In addition, the gamma irradiation decreases the intensity of the peak (311) and (440). These results confirmed the modification of the cubic lattice of Co0.72Sr0.07Ni0.21Fe2O4 takes place., were reported by different authors and they attributed that to the distortion that occurred in the cubic lattice after γ-irradiation. 94
Crystal size was calculated by Scherrer’s equation and the results were for the samples (a) the as-prepared (AP) spinel Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles and (b) the Co0.72Sr0.07Ni0.21Fe2O4 nano ferrite particles aging at 1000°C (AG) and (c) the Co0.72Sr0.07Ni0.21Fe2O4 nano ferrite particles irradiated at 100 kGy (Ir) equal to (9.1 nm), (18 nm), and (20 nm) respectively.95,96
VSM analysis of Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles
In Figure 3, the room temperature magnetization versus magnetic hysteresis (M-H) loops for the blank, irradiated (at a dose of 100 kGy), and aging Co0.72Sr0.07Ni0.21Fe2O4 samples at 1000°C are shown. The M-H loops provide information about the magnetic behavior of the samples and can reveal important characteristics such as saturation magnetization, coercivity, and remanence. These parameters indicate the magnetic properties and potential applications of the nanoparticles. By analyzing the M-H loops, one can determine the saturation magnetization, representing the maximum magnetization the material can attain in an applied magnetic field. A higher saturation magnetization indicates stronger magnetic properties. The three ferrite samples are ferromagnetically property, and the aging ferrite sample has the highest magnetization values. Table 1 provides measurements of various parameters for the blank, irradiated (at a dose of 100 kGy), and sintered Co0.72Sr0.07Ni0.21Fe2O4 samples. These parameters include Hc (coercive force), Br (retentive force), and Bs (saturated inductive magnetization). The Hc, Br, and B units are given in Gauss (G) and emu/g (electromagnetic units per gram), respectively. Based on Figure 3, it is observed that the blank and irradiated Co0.72Sr0.07Ni0.21Fe2O4 samples exhibit a fat hysteresis loop, indicating a higher coercive force (Hc) and lower saturation magnetization (Bs). On the other hand, the sintered Co0.72Sr0.07Ni0.21Fe2O4 sample shows a thin hysteresis loop, suggesting a lower coercive force (Hc) and higher saturation magnetization (Bs). Furthermore, The observed decrease in magnetization after irradiation and increase after aging in the case of the Co0.72Sr0.07Ni0.21Fe2O4 samples is consistent with findings reported in other literature. Similar trends have been observed in various studies where the magnetic properties of materials are influenced by external factors such as irradiation or changes in processing conditions. A study on the radiolytic formation of Fe3O4 nanoparticles showed that the saturation and remanence magnetization decreased when the radiation dose increased.
97
Irradiation can introduce structural defects or disorder in the material, disrupting the alignment of magnetic moments and leading to a decrease in magnetization. This is supported by detecting a new phase (γ-FeOOH) in the irradiated sample, as indicated by XRD analysis in your previous comment. The presence of this new phase may contribute to the reduction in magnetization. On the other hand, the aging process at high temperatures can lead to changes in the particle size and crystallinity of the material. In the case of the Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles, the increase in particle size and improved superexchange interactions between Fe3+ ions can enhance the material’s magnetization. This is in line with the observed increase in magnetization after aging. As seen in Table 1 The increased coercivity of the irradiated sample can be attributed to (i) Irradiation can introduce structural defects or disorder in the material, leading to an increase in coercivity. These defects can disrupt the alignment of magnetic moments and make it more difficult to switch the magnetization direction, resulting in higher coercive force.13,98–100 (ii) Irradiation can affect the magnetic interactions between particles or within the crystal structure. It can enhance magnetic anisotropy and create more rigid magnetic domains, increasing coercivity.101,102 (iii) Irradiation-induced changes in the domain structure can contribute to increased coercivity. Forming smaller or more irregular magnetic domains can require higher applied magnetic fields to reverse the magnetization, resulting in higher coercive force.103,104 (iv) Depending on the irradiation conditions and the specific material, irradiation can induce changes in the chemical composition or redistribution of cations, which can influence the magnetic properties.
105
Alterations in the chemical composition can affect magnetic interactions and contribute to increased coercivity. On the other hand, The decreased coercivity of the aging sample can be attributed to several factors such as (i) During the aging process, the nanoparticles can undergo grain growth, increasing particle size. Larger particles typically have a lower magnetic anisotropy, which results in a decrease in coercivity. The reduced magnetic anisotropy makes it easier to switch the magnetization direction, resulting in a lower coercive force.
106
(ii) Aging at high temperatures can promote the crystallization and improvement of the crystal structure. A more ordered crystal structure with fewer defects can lead to a reduction in coercivity. The improved crystallinity allows for easier reorientation of the magnetic moments, lowering the energy barrier for magnetization reversal.107,108 (iii) The aging process can also affect the magnetic interactions between particles or within the crystal structure. It may lead to a change in the magnetic domain structure, resulting in a decrease in coercivity. Changes in the magnetic interactions can promote the alignment of magnetic moments and reduce the resistance to magnetization reversal.
109
(iv) Aging at elevated temperatures can induce chemical changes, such as cation redistribution or phase transformations. These changes can influence the magnetic properties of the material, including coercivity. Alterations in the chemical composition can affect the magnetic interactions and produce a lower coercive force.
110
The M–H loops of the Co0.72Sr0.07Ni0.21Fe2O4 samples. Table 1 shows the magnetic characteristics of the three samples: AP (as prepared), Ir (100 kGy irradiated), and AG (aged at 1000°C).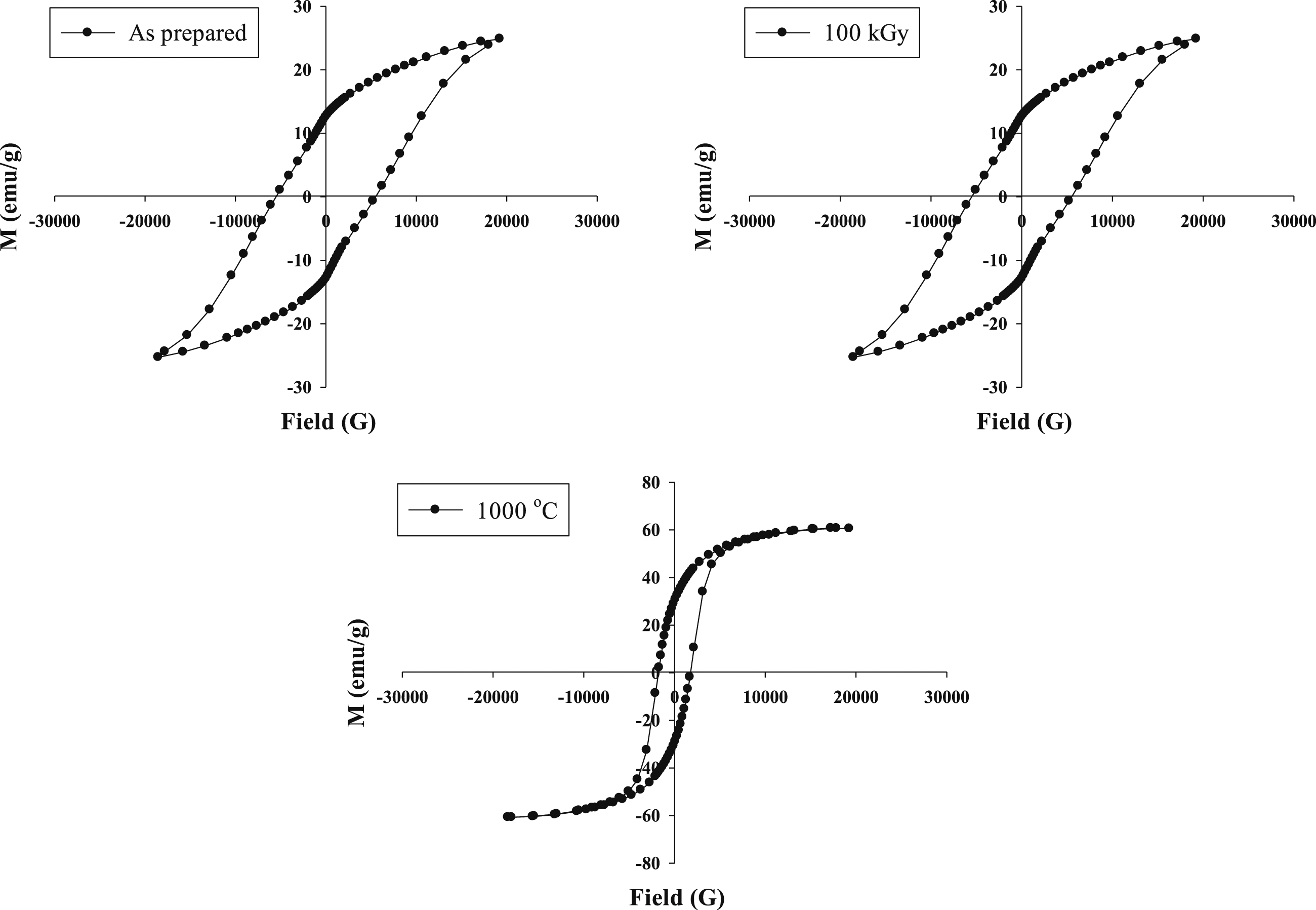

The interesting observation from the magnetic study is that the aging process of Co0.72Sr0.07Ni0.21Fe2O4 samples at 1000°C leads to an enhancement in magnetization, while the irradiation process results in a decreased magnetization. In the sintering process, the increase in particle size leads to an increase in the strength of the superexchange interaction between Fe3 + ions in the A- and B-sites. This enhanced interaction contributes to a general increase in magnetization.
On the other hand, the irradiation process introduces structural disorder and cation redistribution in the material. This leads to a new phase of γ-FeOOH, as detected by XRD analysis. This new phase and the structural disorder caused by irradiation result in decreased magnetization. γ-FeOOH is an antiferromagnetic material, meaning that the magnetic moments of neighboring iron ions align in an antiparallel manner. γ-FeOOH known as “Goethite” exhibits antiferromagnetic behavior below the Morin temperature (about 120 K) due to the antiparallel alignment of magnetic moments that caused reduced magnetic properties of the Co0.72Sr0.07Ni0.21Fe2O4 sample after gamma irradiation.111,112
Figure 4 displays the FTIR spectra of Co0.72Sr0.07Ni0.21Fe2O4 samples in different conditions: as-prepared, aged at 1000°C, and irradiated at 100 kGy. Notably, two distinct peaks were observed at 3421 cm−1 and 1628 cm−1.
113
These peaks correspond to O-H groups’ stretching and bending vibrations, respectively. These peaks indicate the involvement of O-H functional groups in the chemical structure of the ferrite nanoparticles. The two peaks at 2923 cm-1 and 2842 cm−1 in FTIR spectroscopy are typically attributed to asymmetric and symmetric vibrations of C-H bonds in organic compounds. However, in the case of Co0.72Sr0.07Ni0.21Fe2O4 samples, no organic compounds are present. Therefore, the two peaks at 2923 cm-1 and 2842 cm-1 may be attributed to the asymmetric and symmetric vibrations of M-H bonds. This issue has been faced by other researchers as well, such as Swapna et al. (2014)
114
and Mahamuda et al. (2014),
115
who also observed these peaks in their samples despite the absence of organic compounds. The FTIR peak located at 2359 cm−1 is attributed to CO2.
116
The two peaks at 1551 cm−1 and 1373 cm−1 are exitance only in sample of as prepared is likely due to bending O-H bonds arise from water molecules.117,118 The absorbed water is presumably removed upon heating and irradiation, explaining the disappearance of this peak in the other samples. It is expected the water molecules assist in formation of γ-FeooH of irradiated sample by radiolysis reaction as confirmed by XRD analysis. The formation of γ-FeOOH likely contributes to the changes in metal-oxygen bonding observed in the FTIR curve of irradiated sample, as indicated by new peaks of -O-O- bonds. The two peaks of 916 cm−1 and 1231 cm−1 could be attributed to peroxides and superoxide -O-O- bonds
119
that could be formed of FeOOH phases. A group of peaks in the range of 1155 cm−1, 1089 cm−1, and 1029 cm-1 has been observed only in the irradiated Co0.72Sr0.07Ni0.21Fe2O4 sample. These peaks correspond to stretching vibration of the of (C-O-O-C) endoperoxide bridge,120,121 Fe-O, and Ni-O bonds, respectively. This indicates that the irradiation likely altered the metal-oxygen bonding in the spinel structure of Co0.72Sr0.07Ni0.21Fe2O4. FTIR chart of Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles.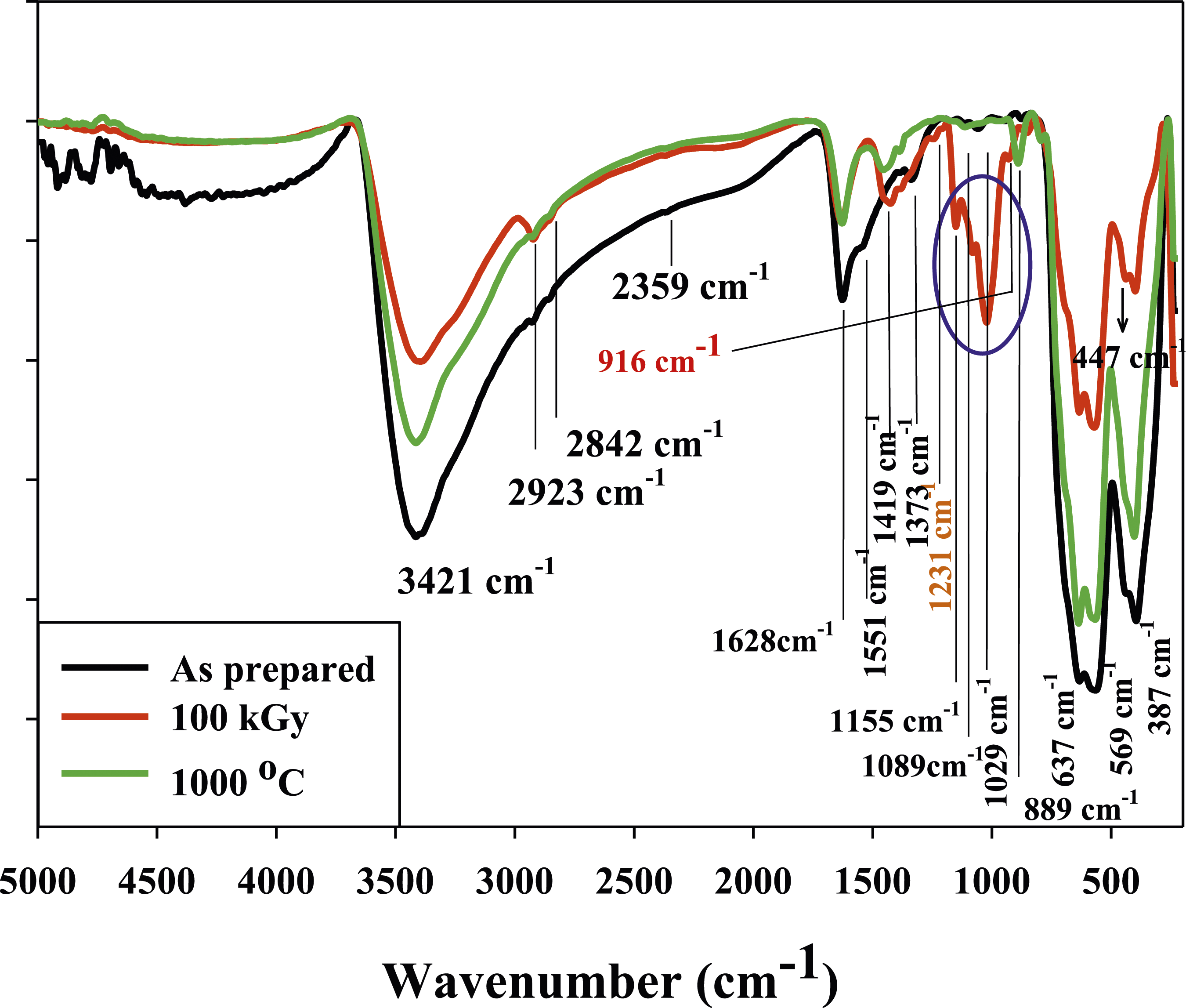
Additionally, the formation of peroxides (-O-O-) indicates the presence of oxygen radicals, which are typically generated through irradiation processes. At the same time, irradiation could cause -O-O- cleavage to produce (C-O-O-C, Fe-O, and Ni-O) bonds. The peak at 1419 cm−1 assigned to C-O stretches from endoperoxide bridges, 122 which further supports the forming of new structures due to irradiation. The appearance of peaks associated with C-O-O-C, Fe-O, and Ni-O bonds suggests modifications in the coordination environment of the metal ions, possibly due to the interaction of radiation-induced species with the material. The -O-O- cleavage and subsequent bond reconfigurations could be caused by reactive oxygen species generated during irradiation. These findings indicate that the irradiation has induced alterations in the metal-oxygen bonding, potentially affecting the structural and chemical properties of Co0.72Sr0.07Ni0.21Fe2O4.
Further analysis and characterization techniques may be necessary to fully understand the nature and implications of these changes. The FTIR peaks are located at 889 cm−1 for Fe-O, 637 cm−1 for Ni-O, 569 cm−1 for Co-O, 447 cm−1 for Sr-O, and 387 cm−1 for stretching vibration of Fe-O-Fe bonds in the spinel structure. The intensities of the FTIR peaks assigned to Fe-O, Ni-O, Co-O, Sr-O and Fe-O-Fe bonds are weaker in the irradiated sample compared to the other samples. This indicates that gamma irradiation likely disrupts or modifies the metal-oxygen bonds in the spinel structure, reducing the number of these bonds. The cleavage of existing metal-oxygen bonds and formation of new non-metal-oxygen bonds, as indicated by other proposed FTIR peak assignments, would explain the spinal structure could change into new structure after irradiation process. In particular, the formation of C-O, C-O-O-C and O-O bonds at the expense of metal-oxygen bonds would account for the decrease in intensities of the Fe-O, Ni-O, Co-O, Sr-O and Fe-O-Fe peaks for the irradiated sample. Changes in the coordination environments of the cobalt, strontium, iron and nickel ions and defective spinel structure formed upon irradiation could also contribute to the weakening of the metal-oxygen bond peaks. According FTIR analysis Gamma irradiation could induce (i) Disrupts or modifies existing metal-oxygen bonds (ii) the formation of new non-metal-oxygen bonds (iii) Changes the metal ion coordination environments within the spinel structure. Does this analysis of how the changes in intensities of the metal-oxygen FTIR peaks provide insights into the effects of irradiation make sense.
Thermal analysis of Co0.72Sr0.07Ni0.21Fe2O4
TGA thermal analysis for the as-prepared Co0.72Sr0.07Ni0.21Fe2O4 spinel nanoparticles was performed by changing the temperature to 1000°C in the N2-atmosphere at a heating rate of 15°C/min. Figure 5 shows the experimental DTA-TGA curves for these nanoparticles. TGA curve for (a) the as-prepared spinel Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles, (b) Co0.72Sr0.07Ni0.21Fe2O4 irradiated nanoparticles and (c) Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles annealed at 1000°C.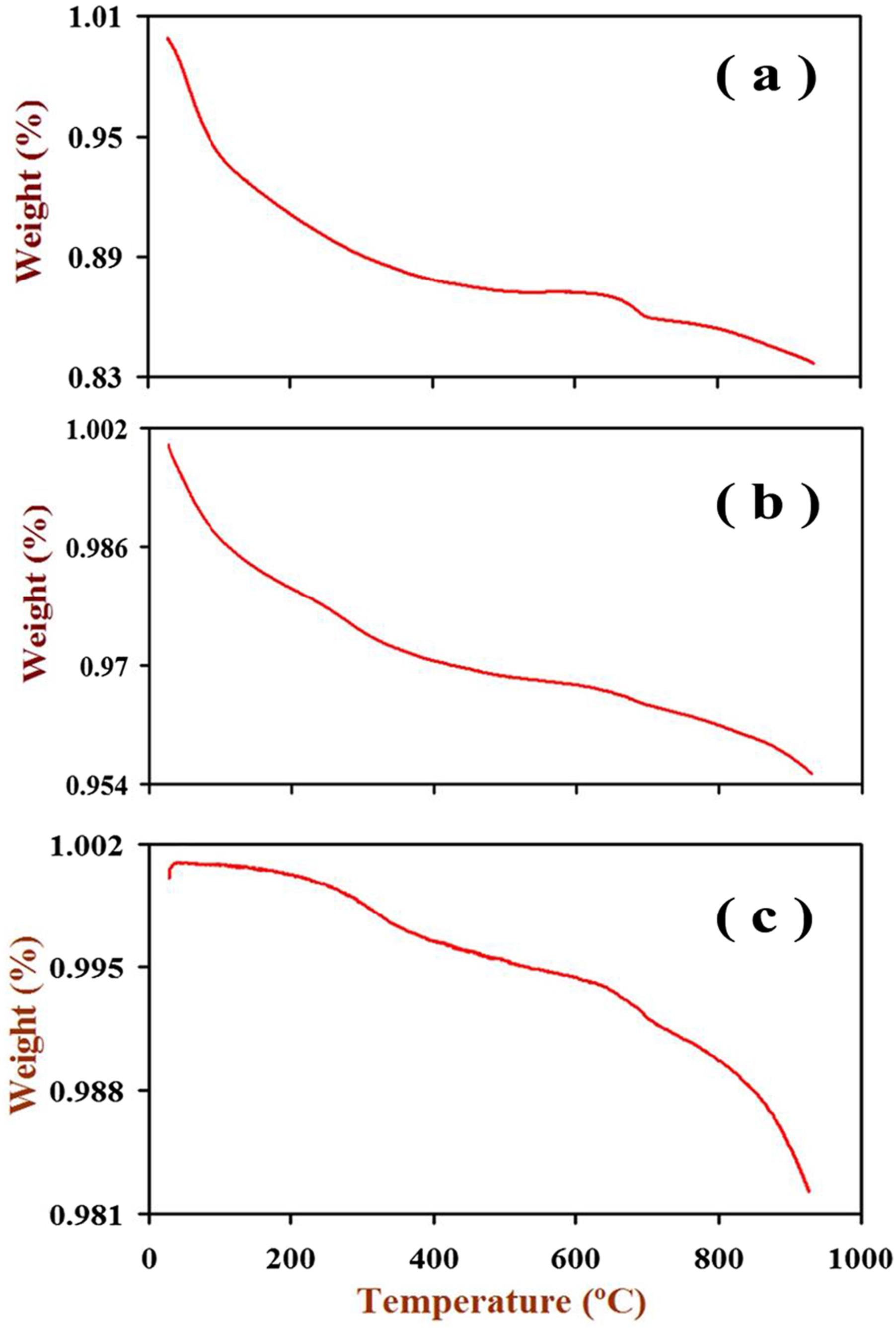
The observed weight loss (%) with temperature.
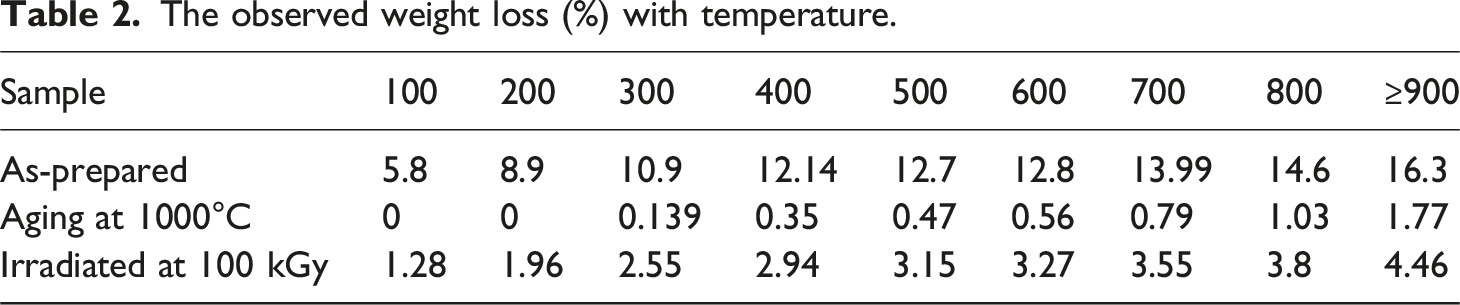
For the “As-prepared” sample: At 100°C, a weight loss of 5.8% is observed, attributed to the evaporation of absorbed water molecules. Between 100°C and 200°C, the weight loss increases to 8.9% as metal hydroxides form. From 200°C to 300°C, the weight loss continues to increase to 10.9% due to the conversion of metal hydroxides into metal oxides and the formation of the nano ferrite phase. The weight loss gradually increases from 300°C to 800°C, reaching a maximum of 16.3% at temperatures greater than or equal to 900°C. For the “Aging at 1000°C″ sample: As expected, no weight loss is observed at temperatures up to 200°C. A slight weight loss of 0.139% and 1.77% is observed at 300°C up to 900°C, respectively, which could be attributed to minor reactions or changes in the nanoparticle structure. For the “Irradiated at 100 kGy” sample: The weight loss is equal 1.28% at 100°C could be attributed to the moisture. From 100°C to 300°C, the weight loss cumulative 5.79% as various transformations occur within the nanoparticles due to FeOOH may convert to FeO or Fe2O3. The conversion of γ-FeOOH to FeO or Fe2O3 can involve the release of water and oxygen, decreasing the general weight of the nanoparticles. The weight loss cumulative reaching 23.72% from 300°C to 900°C could be due to the evolution of oxygen vacancies that gain due to the irradiation process in the nanocrystalline structure of these nano ferrite particles. The gain of oxygen vacancies during the irradiation process can promote the formation of oxygen-deficient phases or facilitate oxygen diffusion from the nanocrystalline structure. This can result in increased weight loss as the nanoparticles are heated to higher temperatures.
FT Raman of Co0.72Sr0.07Ni0.21Fe2O4
Figure 6(a) shows the FT Raman spectra of the as-prepared Co0.72Sr0.07Ni0.21Fe2O4 ferrite nanoparticles. The characteristic Raman modes at 192 cm−1 (A₁g mode, symmetric stretching of oxygen in tetrahedral sites), 316 cm−1 (Eg mode, bending of oxygen in octahedral sites),
123
478 cm−1 (T₂g mode, stretching of oxygen in octahedral sites), 528 cm−1 (T₂g mode, symmetric stretching of oxygen in octahedral sites), and 672 cm−1 (A₁g mode, symmetric stretching of oxygen in tetrahedral sites) are visible.124,125 These narrow, well-resolved peaks indicate high crystallinity and homogeneous cation distribution within the inverse spinel lattice. Figure 6(b) shows the FT Raman spectra of the irradiated Co0.72Sr0.07Ni0.21Fe2O4 ferrite nanoparticles. Compared to the as-prepared sample, the Raman modes exhibit significant broadening and reduced intensity, indicating increased structural disorder and the presence of defects such as cation vacancies, interstitials, and anti-site defects due to the 100 kGy irradiation. The broadening of the peaks suggests a broader distribution of bond lengths and angles. (a) Co0.72Sr0.07Ni0.21Fe2O4 ferrite nanoparticles as prepared. (b) Co0.72Sr0.07Ni0.21Fe2O4 ferrite nanoparticles irradiated (at a dose of 100 kGy in CO2 atmosphere). (c) Co0.72Sr0.07Ni0.21Fe2O4 ferrite nanoparticles aged (at 1000°C).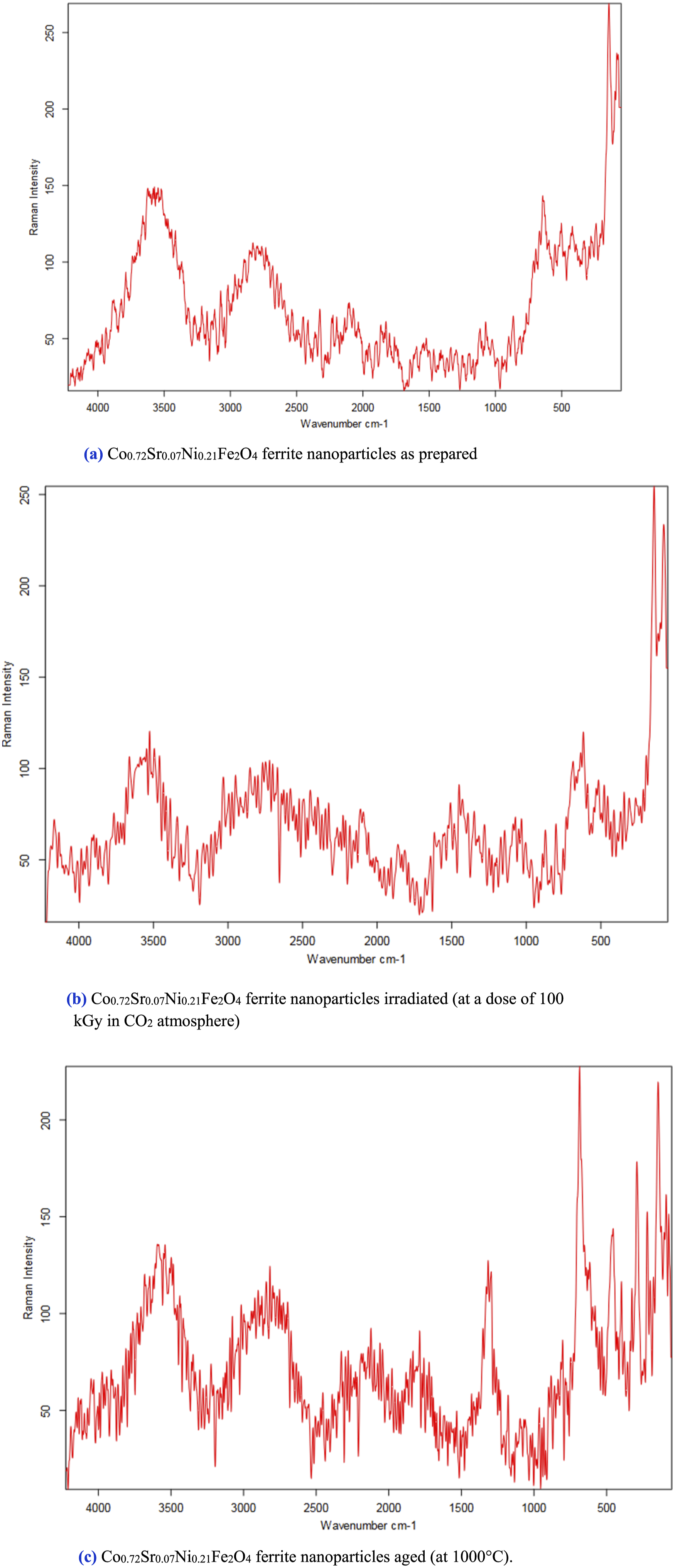
In contrast, the shifts in peak positions reflect local lattice distortions and changes in the force constants of the oxygen-cation bonds. The shift and broadening of the peaks, such as the 672 cm−1 peak shifting to 665 cm−1, indicate local crystal lattice distortions by point defects. New peaks around 322 cm−1 also suggest chemical modifications and possible oxidation or reduction of metal cations. These changes indicate that irradiation has disrupted the long-range structural order, leading to a more disordered and defect-rich crystal structure. Figure 6(c) presents the FT Raman spectra of the Co0.72Sr0.07Ni0.21Fe2O4 ferrite nanoparticles after high-temperature (1000°C) aging treatment. The Raman modes are much sharper and better resolved than the irradiated sample, suggesting a recovery of the crystal structure and increased structural order. The sharp, intense Raman peaks imply that high-temperature aging has annealed out the irradiation-induced defects, leading to a more homogeneous and well-ordered spinel structure with optimized cation distribution. This reduced structural disorder and defect concentration restore the characteristic Raman modes associated with the inverse spinel lattice. The FT Raman spectroscopy provides valuable insights into the structural evolution of Co0.72Sr0.07Ni0.21Fe2O4 ferrite nanoparticles under different processing conditions, highlighting how high crystallinity is disrupted by irradiation but can be recovered through subsequent high-temperature aging.
SEM of Co0.72Sr0.07Ni0.21Fe2O4
Figure 7 shows the SEM images of the Co0.72Sr0.07Ni0.21Fe2O4 ferrite nanoparticles in three different states: as-prepared, irradiated, and after high-temperature (1000°C) aging treatment. The comparison reveals distinct morphological changes across these states. In the as-prepared sample, the nanoparticles exhibit a relatively uniform size and shape, indicating a well-controlled synthesis process. Upon irradiation, the nanoparticles appear more irregular, suggesting an increase in structural defects and possibly some degree of aggregation due to the introduction of high-energy radiation. This increased defect density likely contributes to the observed particle size and shape irregularity. Shows the SEM images of the Co₀.₇₂Sr₀.₀₇Ni₀.₂₁Fe₂O₄ ferrite nanoparticles in three states: as-prepared (a), irradiated at 100 kGy, and (c) after high-temperature (1000°C) aging treatment.
Interestingly, the SEM images of the nanoparticles after the high-temperature aging treatment at 1000°C show a significant reduction in particle size compared to both the as-prepared and irradiated samples. The aging process appears to facilitate the annealing of defects and a restructuring of the nanoparticles, resulting in a more compact and refined morphology. The size reduction could be attributed to the sintering effect during the high-temperature treatment, which can break larger particles into smaller, more thermodynamically stable ones. Overall, these SEM observations highlight the impact of irradiation and thermal treatment on the structural properties of Co0.72Sr0.07Ni0.21Fe2O4 ferrite nanoparticles, providing valuable insights into their morphological evolution under different processing conditions.
AC conductivity of Co0.72Sr0.07Ni0.21Fe2O4
Figure 8 represents the σAC electrical conductivity (Ln σac (S/cm)) against frequency (Lnf (Hz.)) of as-prepared spinel Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles, irradiated nanoparticles at 100 kGy and aging at 1000°C. It is observed that the aging sample had higher electrical conductivity than other samples at any given frequency despite the increased electrical conductivity of the Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles with the increase in frequency. The increase in the aging sample could be attributed to the aggregation, and increased particle size can have implications for the grain boundary. This means that the aging process leads to aggregation and increased particle size, resulting in more grain boundaries, which can promote enhanced conduction along the grain boundaries. Grain boundaries can act as highly conductive paths for electron transport, and an increased number of grain boundaries can contribute to higher electrical conductivity. At the same time, an irradiated sample at (100 kGy) induces oxygen vacancy formation in the material. Oxygen vacancies act as charge carriers and can significantly increase the material’s electrical conductivity. These vacancies provide additional mobile charge carriers that contribute to electrical conduction. In addition, the irradiation process increases defect concentration, such as vacancies or interstitials, within the material. These defects can act as charge carriers and facilitate electrical conduction. The higher defect concentration in the irradiated sample compared to the as-prepared samples (0 kGy) can increase electrical conductivity. However, (100 kGy) is still relatively low compared to aging materials due to the γ-FeOOH goethite phase existing in the (100 kGy) sample behaving as an insulator and exhibiting low electrical conductivity. It has high resistivity, meaning it restricts the flow of electric current. As the frequency increases from kilohertz (kHz) to megahertz (MHz) range, goethite’s electrical conductivity may increase due to charge carrier mobility. However, it is still relatively low compared to materials specifically designed for electrical conduction. It’s important to note that goethite is primarily known for its semiconducting and magnetic properties rather than its electrical conductivity. Its conductivity is typically considered in the insulator or weak semiconductor range. Shows the AC conductivity of Co0.72Sr0.07Ni0.21Fe2O4 samples.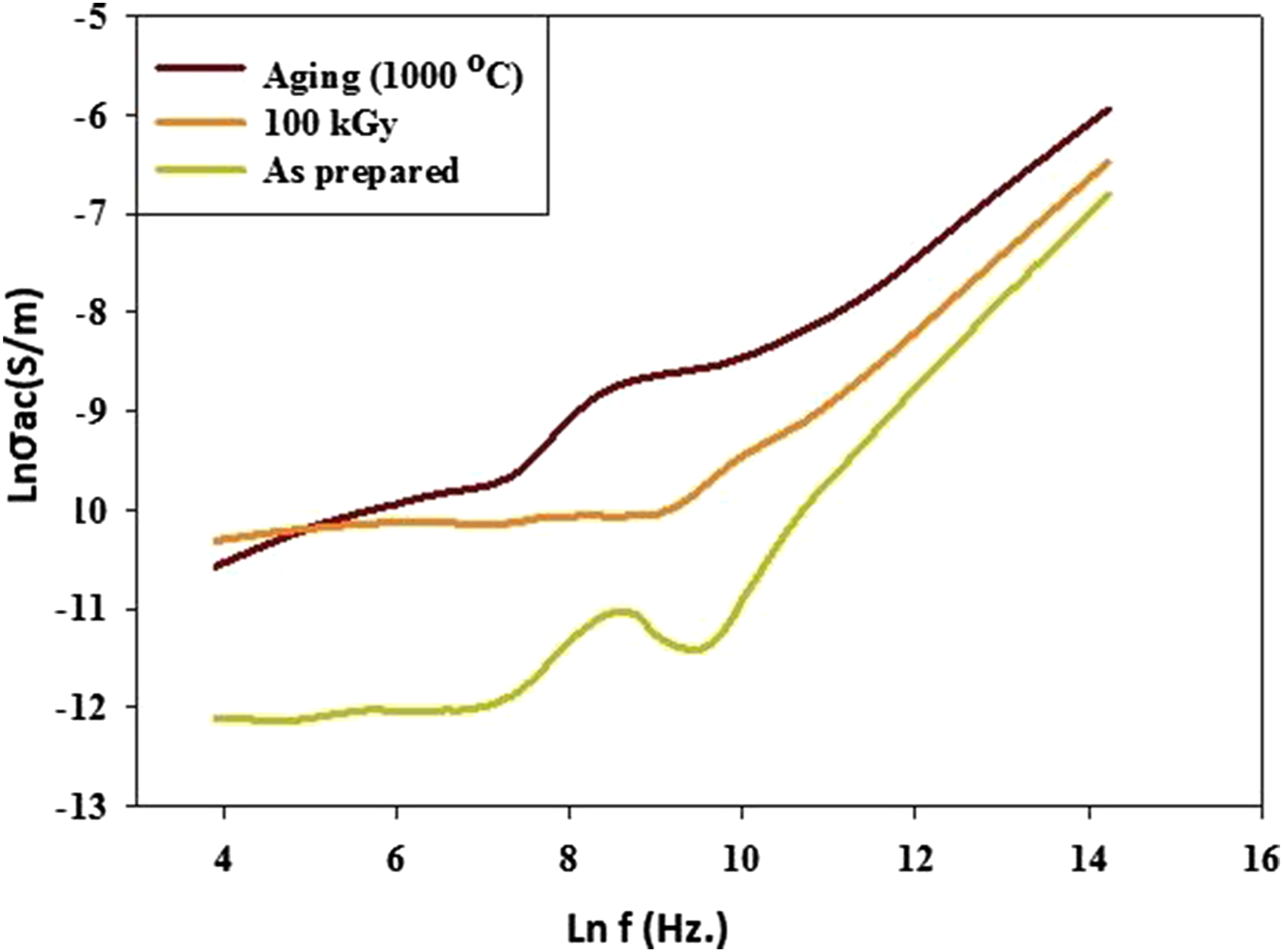
Photodegradation of (TB) by (PVP/Co0.72Sr0.07Ni0.21Fe2O4) nanocomposite
Figure 9(a) represents the setup formation of Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles encapsulated by PVP through a two-step process involving sonication and gamma irradiation-induced PVP polymerization. Initially, a mixture containing Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles and a 7% PVP solution are prepared. The mixture is then subjected to sonication using a sonication probe. Sonication involves the application of high-frequency sound waves to the mixture, leading to the dispersion of the nanoparticles in the PVP solution and the formation of a uniform suspension. After sonication, the PVP/Co0.72Sr0.07Ni0.21Fe2O4 nanocomposite suspension is exposed to gamma irradiation. Gamma irradiation is a high-energy radiation process that can induce chemical changes in materials. In this case, gamma irradiation triggers the polymerization of PVP, forming a polymer coating around the nanoparticles. This encapsulation process helps stabilize the nanoparticles and enhance their properties for catalytic applications. (a) Show the gamma irradiation-induced synthesis of PVP/Co0.72Sr0.07Ni0.21Fe2O4, (b) their catalytic photo degradation of TB dye, and (c) the real photograph serves as visual evidence of the effectiveness of the PVP/Co0.72Sr0.07Ni0.21Fe2O4 nanocomposite as a catalyst for TB dye photodegradation and demonstrates the process of photodegradation and subsequent magnetic separation.
Gamma irradiation can significantly alter the properties of ferrite nanoparticles, which can be harnessed for various applications like catalysis, electronic devices, biomedical applications, etc. Proper encapsulation by PVP is required to achieve desirable changes in the properties of ferrite nanoparticles through gamma irradiation for various applications like catalysis for water treatment. PVP encapsulation of ferrite nanoparticles before gamma irradiation is crucial to achieving desirable properties for catalytic and other applications. Properly optimized, this method can lead to highly efficient nanocatalysts for wastewater treatment and other environmental remediation applications. PVP encapsulation of ferrite nanoparticles has several benefits for their catalytic application in dye degradation in aqueous solutions. It enhances dispersibility in water: As discussed previously, gamma irradiation of PVP-encapsulated ferrite nanoparticles can enhance their hydrophilicity and dispersibility in water. This allows greater availability of surface active sites for the dye molecules, leading to higher catalytic activity. PVP encapsulation allows gradual catalytic activity optimization by protecting the catalyst surface from impurities in the reaction mixture. This prevents the poisoning of active sites on the catalyst surface, allowing for higher catalytic activity. This helps avoid excess defect formation, which can harm catalytic performance. PVP encapsulation prevents the leaching of metal ions by retaining the metal ions like Fe3+ within the ferrite lattice and prevents their leaching into the solution during the catalytic reaction. This prevents contamination of the treated water. Due to their larger aggregate size, the encapsulated nanoparticles can be easily separated from the reaction mixture using filtration or centrifugation. This allows the catalyst to be reused for multiple cycles. PVP encapsulation prevents the individual nanoparticles from detaching from the aggregates, thereby retaining their catalytic activity over reuse cycles. PVP-encapsulated nanoparticles form stable colloidal dispersions in water that show minimal sedimentation over long periods. This high stability is crucial for real applications.
Studies have shown that PVP encapsulated and gamma-irradiated ferrite nanoparticles exhibit excellent catalytic activity for dye degradation with up to 95-99% dye removal in some cases. This highlights the great promise of this methodology. Thus, PVP encapsulation is extremely useful for developing highly efficient ferrite-based nanocatalysts for treating dye-contaminated wastewater and other pollutants. Proper optimization of polymer coating and gamma irradiation dose can lead to stable, reusable, and non-leaching nanocatalysts with high activity. The degradation of dyes, such as TB (Toluidine Blue), using nanocomposites composed of PVP (Polyvinylpyrrolidone) and Co0.72Sr0.07Ni0.21Fe2O4 can be an effective approach. The nanocomposite combines the properties of PVP, which acts as a stabilizer and dispersant. PVP also enhances the dispersibility of the nanocomposite in the dye solution, promoting efficient contact between the dye molecules and the catalyst surface and Co0.72Sr0.07Ni0.21Fe2O4, which acts as a catalyst for dye degradation.
Figure 9(b) visually represents this experimental setup, illustrating the interaction between the PVP/Co0.72Sr0.07Ni0.21Fe2O4 nanocomposite and the TB dye under UV light for photodegradation. The first part of the photograph in Figure 9(c) depicts the initial state of the aqueous dye solution containing the TB dye. The dye solution appears with its characteristic color, indicating the presence of the dye molecules. The second part of the photograph showcases the photodegradation treatment process. The magnetic PVP/Co0.72Sr0.07Ni0.21Fe2O4 nanocomposite is added to the dye solution, exposing the mixture to UV light. This stage aims to initiate the catalytic degradation of the TB dye by the nanocomposite under UV light irradiation. After photodegradation, the final part of the photograph demonstrates the magnetic separation step. After the photodegradation treatment, a magnet separates the PVP/Co0.72Sr0.07Ni0.21Fe2O4 nanocomposite from the solution. The magnetic properties of the nanocomposite allow for easy collection and separation by applying a magnetic field. The clear solution in this part of the photograph indicates that the dye has undergone photodegradation, while the nanocomposite is collected and separated for potential reuse or further analysis. A real photograph showing the use of the magnetic PVP/Co0.72Sr0.07Ni0.21Fe2O4 nanocomposite used in our study as catalyst to degradation TB dye as see the aqueous dye solution before photodegradation and after photodegradation treatment and magnetic separation of PVP/Co0.72Sr0.07Ni0.21Fe2O4 after photodegradation treatment.
The degradation occurs through various mechanisms, including photodegradation and/or catalytic oxidation. When the TB dye molecules come into contact with the nanocomposite, the Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles catalyze and generate reactive oxygen species (ROS) such as hydroxyl radicals (•OH). These ROS have strong oxidizing properties and can effectively break down the dye molecules and breakdown of the dye molecules into smaller, less harmful compounds. The time-dependent removal of the (TB) dye at the concentration of 4 × 10−5 mol using 0.1 g of the (PVP/Co0.72Sr0.07Ni0.21Fe2O4) nanocomposite catalyst at 100 mL of dye solution was examined by a UV spectrophotometer. As known TB is a basic dye. The adsorption capacity of the TB adsorbate onto the PVP adsorbent is provided by ionic dipole–dipole, or hydrogen bond interactions. Figure 10(a) and (b) show that the absorption spectra of the water-soluble TB dye are strongly affected by catalytic degradation induced by The UV-Vis spectrophotometer (a, b) and the decolorization (%) (c) of 4 × 10−5 mol TB photodegradation with time using 0.1 g of the (PVP/Co0.72Sr0.07Ni0.21Fe2O4) nanocomposite under UV light.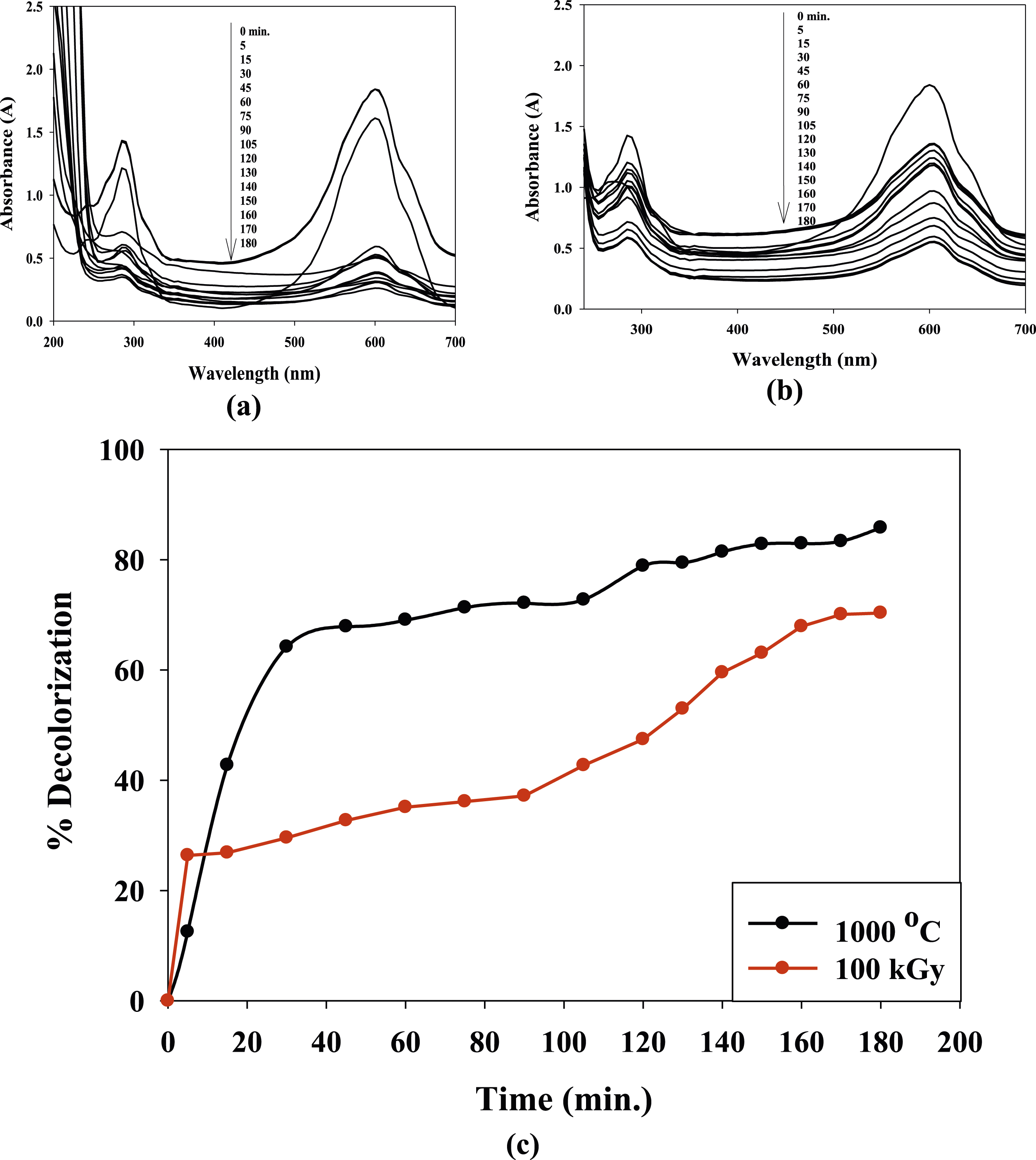
These results indicate that the aged and irradiated nanocomposites show effective decolorization of the TB dye. The higher decolorization percentages observed at 1000°C suggest that the aging process enhances the catalytic activity of the nanocomposite, resulting in more efficient degradation of the dye. However, even the nanocomposite irradiated at 100 kGy shows significant decolorization activity, highlighting the potential of gamma irradiation to modify the nanocomposite’s catalytic properties. In the first 5 min, the decolorization percentages for the (PVP/Co0.72Sr0.07Ni0.21Fe2O4) nanocomposite aged at 1000°C and irradiated at 100 kGy are as follows: For the nanocomposite aged at 1000°C, the decolorization percentage is 12.4897%. For the nanocomposite irradiated at 100 kGy, the decolorization percentage is 26.3444%. These results indicate that the nanocomposite irradiated at 100 kGy exhibits a higher decolorization percentage in the first 5 min than the nanocomposite aged at 1000°C. The nanocomposite irradiated at 100 kGy initially exhibits a higher decolorization percentage due to the formation of γ-FeOOH during gamma irradiation.
γ-FeOOH is known for its high catalytic activity, which contributes to faster decolorization in the first 5 min. However, as the reaction progresses, the nanocomposite aged at 1000°C shows a higher decolorization percentage due to its improved crystallinity and particle size. On the other hand, the enhanced catalytic activity of the aged nanocomposite at 1000°C. The higher decolorization percentage for the aged nanocomposite can be attributed to the structural changes and improved properties of the nanocomposite during the aging process. The annealing process at 1000°C promotes the growth of particles in the (PVP/Co0.72Sr0.07Ni0.21Fe2O4) nanocomposite, resulting in larger particle sizes. This increase in particle size can enhance the superexchange interactions between the Fe3+ ions, leading to improved magnetic properties, as observed in the VSM analysis. The enhanced superexchange interactions and improved magnetic properties can contribute to increased catalytic activity in the nanocomposite aged at 1000°C. The larger particle size allows for more active sites on the catalyst surface, providing more opportunities for the adsorption and degradation of the TB dye molecules.
Additionally, the improved orientation of magnetic moments in the nanocomposite can facilitate electron transfer and promote the generation of reactive species, further enhancing the catalytic activity. It is worth noting that the increased catalytic activity observed in the nanocomposite aged at 1000°C results from the combined effects of particle growth, enhanced superexchange interactions, and improved orientation. These factors work synergistically to create a favorable environment for the catalytic degradation of TB dye.
Adsorption kinetics
The kinetics of catalytic photodegradation of TB adsorbed on (PVP/Co0.72Sr0.07Ni0.21Fe2O4) nanocomposite powder aging at 1000 C and irradiated at 100 kGy were evaluated to determine the mechanism of adsorption using Pseudo-first order and Pseudo-second order kinetics models as shown in Figure 11(a)–(d), respectively. Kinetic degradation of TB on the (PVP/Co0.72Sr0.07Ni0.21Fe2O4) nanocomposite follows a pseudo-second-order reaction. The higher R2 value for the pseudo-second-order model suggests a better fit of the experimental data to the model, indicating that the degradation process is better described by second-order kinetics. Photodegradation kinetic models for TB using Co0.72Sr0.07Ni0.21Fe2O4 for Pseudo-first order (a) and (b) Pseudo-second order.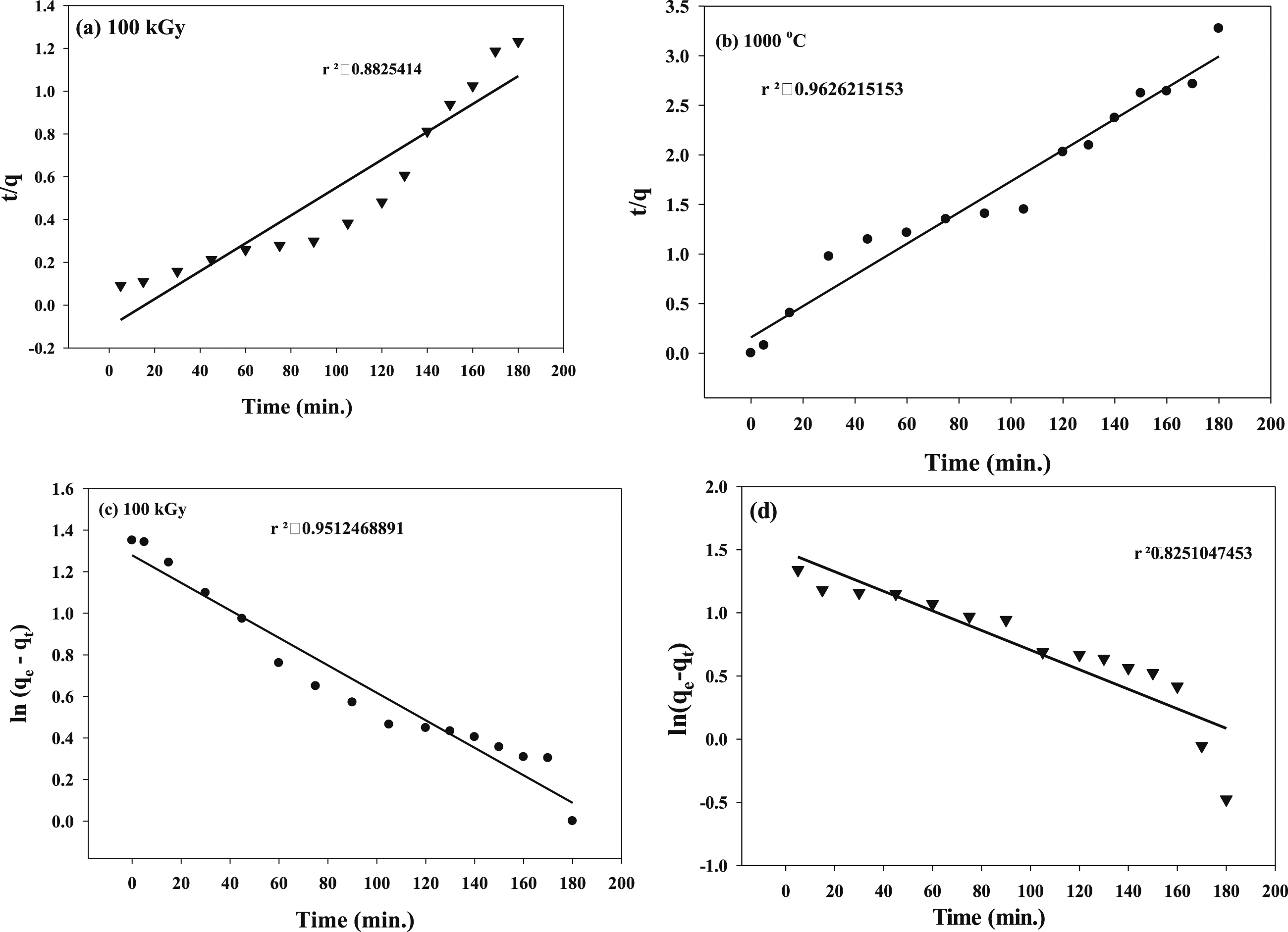
The pseudo-first-order model assumes that the degradation rate is proportional to the concentration of the TB dye remaining on the nanocomposite. However, the lower R2 value (0.82) indicates a poorer fit of the experimental data to the model, suggesting that the pseudo-first-order model does not accurately describe the degradation kinetics.
In contrast, the pseudo-second-order model considers the interaction between the TB dye and the nanocomposite surface, where the degradation rate depends on the dye’s concentration and the available active sites on the nanocomposite. The higher R2 value (0.96) suggests a strong correlation between the experimental data and the pseudo-second-order model, indicating that the degradation of TB follows a second-order reaction.
The results indicate that the adsorption and degradation of TB on the (PVP/Co0.72Sr0.07Ni0.21Fe2O4) nanocomposite are influenced by multiple factors, such as the surface area, active sites, and surface properties of the nanocomposite. The pseudo-second-order kinetics model better represents the degradation process and suggests that the rate-limiting step is likely the chemical reaction between the nanocomposite and the TB dye.
These findings have implications for the design and optimization of catalytic systems for the degradation of organic pollutants, as understanding the kinetic behavior can help determine the appropriate reaction conditions, catalyst dosage, and reaction time for efficient pollutant removal.
Adsorption isotherms
Two adsorption isotherms models of (Langmuir and Freundlich) for TB photodegradation under UV using (PVP/Co0.72Sr0.07Ni0.21Fe2O4) aging at 1000°C and irradiated at 100 kGy.

The difference in favorability of the Langmuir and Freundlich models for the samples aged at 1000°C and irradiated at 100 kGy can be attributed to the specific adsorption characteristics of the nanocomposite under different conditions. In the Langmuir model, the assumption is that adsorption occurs on a homogeneous surface with a limited number of identical sites available for adsorption. The model assumes monolayer adsorption, where once a molecule occupies a site, no further adsorption can take place at that site. The Langmuir model is more favorable for the sample aged at 1000°C because the higher R2 value (0.95) indicates a better fit of the experimental data to the model. This suggests that the adsorption process on the nanocomposite surface is well-described by the Langmuir model, and there is a limited and specific number of active sites available for TB dye adsorption. The higher qmax value (4.1903 mg/g) further indicates that the nanocomposite has a higher adsorption capacity for TB dye in the monolayer adsorption process.
On the other hand, the Freundlich model assumes multilayer adsorption on a heterogeneous surface with a wide range of adsorption sites and energies. It does not have a specific limit on the number of molecules that can be adsorbed on the surface. The Freundlich model is more favorable for the sample irradiated at 100 kGy because the higher R2 value (0.92) indicates a better fit of the experimental data to the model. This suggests that the adsorption process on the nanocomposite surface is better described by the Freundlich model, which accounts for a wider distribution of adsorption sites with varying affinities for TB dye molecules. The kF value (1.842) indicates the adsorption capacity of the nanocomposite, and the 1/n value (0.903) suggests a higher intensity of adsorption, indicating a more heterogeneous surface with varying adsorption energies.
The difference in favorability of the Langmuir and Freundlich models for the two samples can be attributed to the structural and surface changes induced by the aging process at 1000°C and gamma irradiation at 100 kGy. These processes can alter the surface properties of the nanocomposite, including surface area, active sites, and surface energy, leading to variations in the adsorption behavior. The specific conditions of the aging and irradiation processes influence the adsorption mechanisms and, therefore, the applicability of the Langmuir and Freundlich models.
Figure 12 demonstrates the photocatalytic stability and reusability of a nanocomposite material consisting of polyvinylpyrrolidone (PVP) and Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles for the degradation of (TB) dye under UV light irradiation. Figure 12 indicates that even after multiple catalytic cycles of dye degradation and recovery of the photocatalyst, the nanocomposite retains its high photocatalytic activity towards TB dye degradation. This suggests that the PVP/Co0.72Sr0.07Ni0.21Fe2O4 nanocomposite is highly stable and can be reused multiple times without significantly losing its catalytic performance. Maintaining high photocatalytic activity over multiple cycles is a crucial factor for practical applications of photocatalysts in wastewater treatment or environmental remediation processes. The stability and reusability of this nanocomposite can be attributed to the synergistic effects of the polymer (PVP) and the spinel ferrite nanoparticles (Co0.72Sr0.07Ni0.21Fe2O4). The photocatalytic stability and reusability of the (PVP/Co0.72Sr0.07Ni0.21Fe2O4) nanocomposite under UV light degradation of 4 × 10−5 mol TB dye.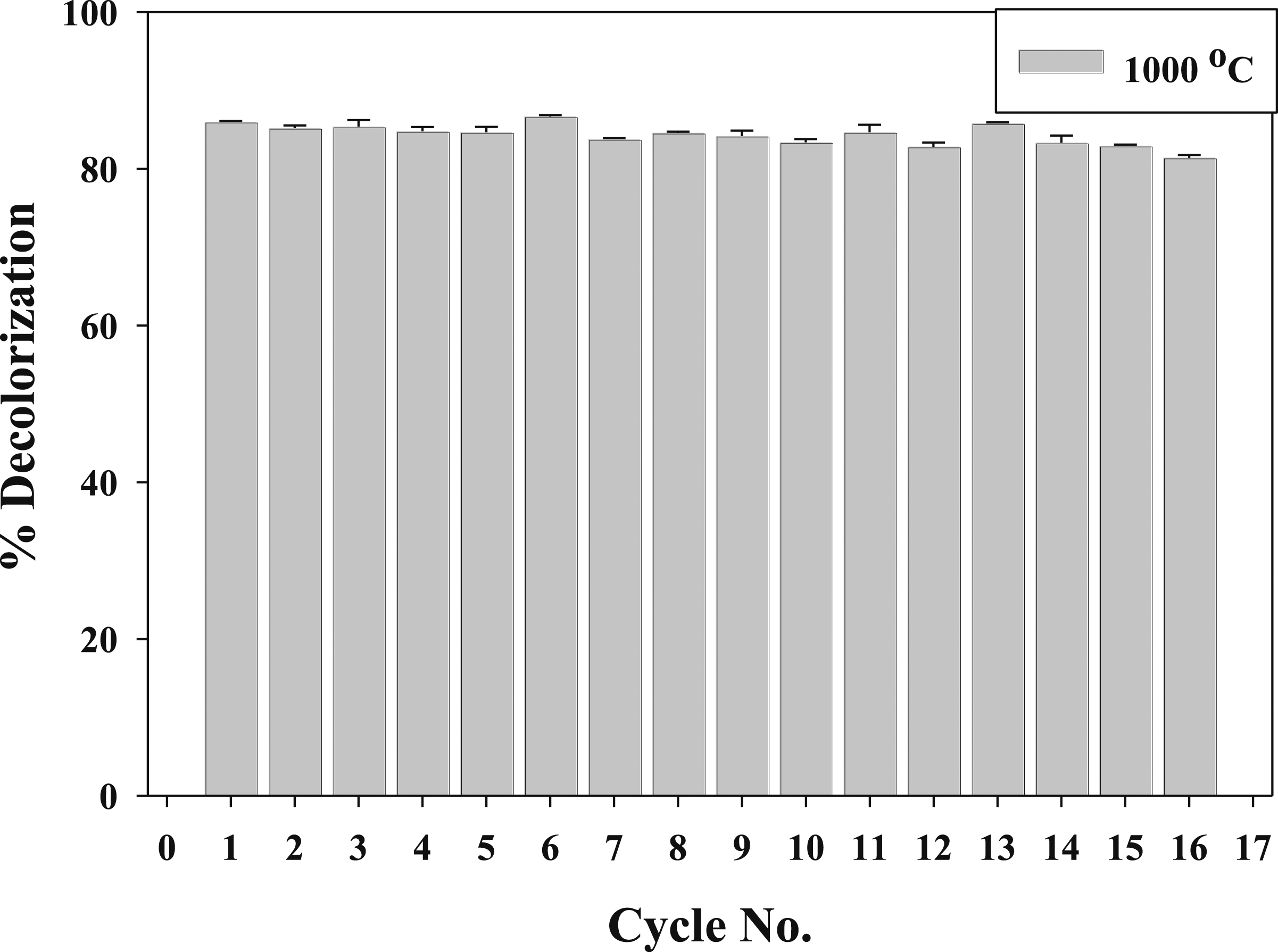
Antimicrobial efficacy of PVP/Co0.72Sr0.07Ni0.21Fe2O4 nanocomposite against waterborne pathogenic bacteria
Figure 13 presents a critical selection of pathogenic bacteria commonly found in contaminated water, encompassing both Gram-negative bacteria (Escherichia coli, Shigella flexneri, Salmonella enterica, and Pseudomonas aeruginosa) and Gram-positive bacteria (Enterococcus faecalis). This selection represents a comprehensive spectrum of bacterial threats in water systems, with each species possessing distinct cell wall structures and pathogenic mechanisms. The antimicrobial activity data in Table 4 reveals interesting patterns when analyzed in the context of bacterial cell wall structures. For Gram-negative bacteria, which possess a complex cell wall with an outer membrane, the PVP/Co0.72Sr0.07Ni0.21Fe2O4 nanocomposite demonstrated varying effectiveness. Most notably, Salmonella enterica showed the highest susceptibility with inhibition zones of 17–18 mm across (A, B, and C), significantly exceeding the reference antibiotic Gentamicin (13 mm). This enhanced activity against Salmonella enterica suggests the nanocomposite may have specific mechanisms for penetrating the complex Gram-negative cell wall structure. The response pattern of Pseudomonas aeruginosa (15–16 mm inhibition) and Escherichia coli (14–15 mm inhibition) indicates that despite their robust outer membrane structures, these Gram-negative bacteria remain susceptible to the nanocomposite’s antimicrobial action. Particularly interesting is the case of Shigella flexneri, where the inhibition zone increases significantly from 13 mm to 16 mm at concentration C, suggesting a concentration-dependent effectiveness. Shows the test organisms, which include four Gram-negative bacteria: Escherichia coli (ATCC25922), Shigella flexneri (ATCC 12022), Salmonella enterica (ATCC25566), and Pseudomonas aeruginosa (ATCC 27853), along with one Gram-positive bacterium, Enterococcus faecalis (ATCC 19433). These strains are selected as representative pathogenic bacteria commonly found in contaminated water. Antimicrobial activity of the sample (PVP/Co0.72Sr0.07Ni0.21Fe2O4) nanocomposite by measuring the Inhibition zone diameter (millimeter) after 24 h incubation period and at 37°C applying the well diffusion technique. *Reference: CN: Gentamicin 10 mcg (standard antibiotic disc) Bioanalyse.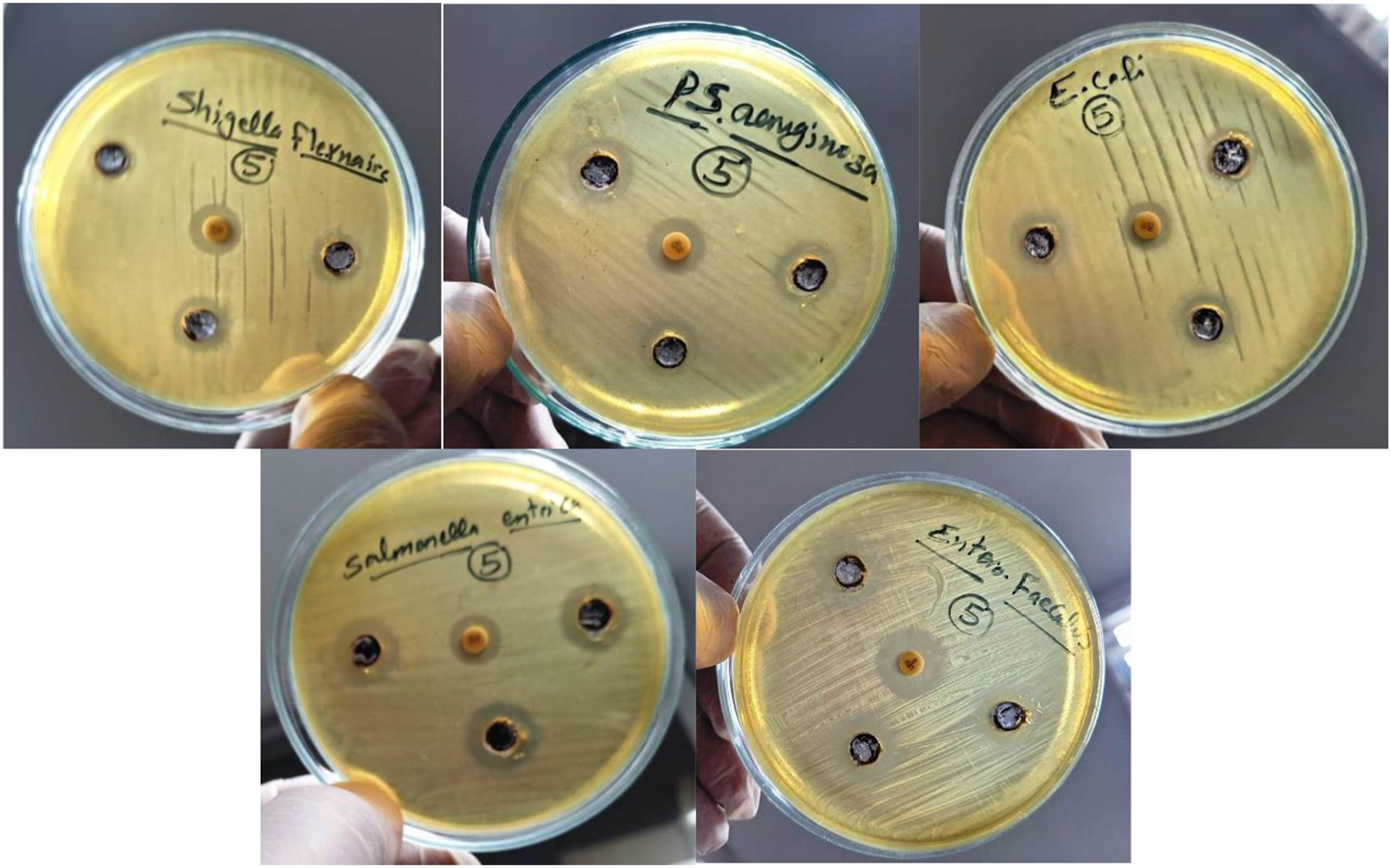
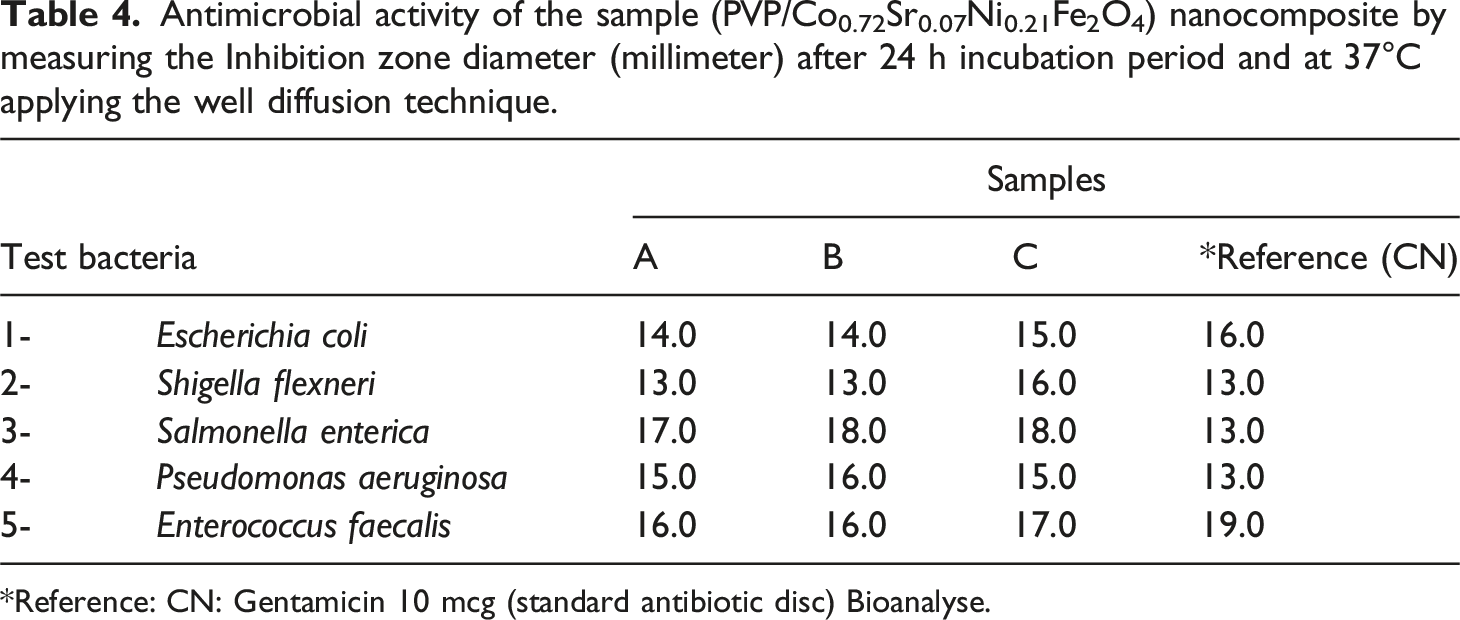
The nanocomposite showed consistent and strong inhibition for the Gram-positive bacterium Enterococcus faecalis, which has a thick peptidoglycan layer but no outer membrane (16–17 mm). While slightly less effective than Gentamicin (19 mm) against this particular strain, the results demonstrate the nanocomposite’s ability to act against different bacterial cell wall types. This finding reveals that the PVP/Co0.72Sr0.07Ni0.21Fe2O4 nanocomposite exhibits broad-spectrum antimicrobial activity, particularly effective against Gram-negative bacteria. This is especially significant given that Gram-negative bacteria are typically more resistant to antimicrobial agents due to their additional outer membrane barrier.
Conclusion
As outlined in your detailed description, the physiochemical properties of Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles can be influenced by gamma irradiation and aging temperature. These factors play crucial roles in altering the stability, structural configuration, magnetic behavior, and potential applications of the nanoparticles. Surface analysis techniques such as transmission electron microscopy (TEM), X-ray diffraction (XRD), and magnetic measurements are essential for elucidating these changes and understanding the nanoparticles’ behavior before and after irradiation and aging processes. The observed reduction in aggregation and cubic morphology of irradiated Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles suggests the potential of gamma irradiation as a technique for enhancing dispersibility and stability, particularly advantageous in applications such as catalysis, where dispersion and uniformity are pivotal. The XRD patterns reveal the cubic spinel structure of the nanoparticles post-irradiation, with the emergence of (FeOOH) phases, and indicate the intricate structural changes induced by gamma irradiation. This process generates solvated electrons, H•, and oxygen radicals, which act as strong reducing and oxidizing agents, influencing the oxidation state of iron ions and forming γ-FeOOH.
Moreover, aging at high temperatures contributes to crystallization and growth, resulting in larger and more well-defined crystallites. This process enhances magnetization, catalytic activity, and electrical conductivity due to increased grain boundaries and superexchange interactions within the ferrite lattice. Encapsulation of Co0.72Sr0.07Ni0.21Fe2O4 nanoparticles with PVP further enhances their catalytic properties, particularly in the degradation of Toluidine Blue (TB) dye under UV light. PVP encapsulation improves dispersibility, protects catalytic active sites, prevents metal ion leaching, and facilitates a pseudo-second-order kinetic model for adsorption, indicating a chemical adsorption mechanism.
The Langmuir isotherm model’s high correlation coefficient and favorable dimensionless separation factor underscore the homogeneous adsorption of TB dye onto the (PVP/Co0.72Sr0.07Ni0.21Fe2O4) nanocomposite. In summary, the aged and encapsulated nanoparticles exhibit structural integrity and sustained catalytic activity, offering promising prospects for photodegradation applications. These findings highlight the importance of understanding and controlling the physiochemical properties of nanoparticles for their effective utilization in various fields. The PVP/Co0.72Sr0.07Ni0.21Fe2O4 nanocomposite demonstrates broad-spectrum antimicrobial activity, particularly effective against Salmonella enterica and Pseudomonas aeruginosa. In several cases, the compound’s ability to match or exceed the performance of a standard antibiotic (Gentamicin) suggests its potential as an effective water treatment agent. The consistent performance across Gram-positive and Gram-negative bacteria indicates versatility in its antimicrobial mechanisms, making it a promising candidate for water purification applications.
Footnotes
Acknowledgments
The authors extend their appreciation to the Deanship of Scientific Research at Northern Border University, Arar, KSA for funding this research work through the project number NBU-FFR-2024-3049-03.
Author contributions
All authors shared equally in writing the manuscript and accepted the responsibility of the entire contents of this manuscript and approved the submission.
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors extend their appreciation to the Deanship of Scientific Research at Northern Border University, Arar, KSA for funding this research work through the project number NBU-FFR-2024-3049-03. The authors would like to thank Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2024R9), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia for supporting this project.
Research data policy
The authors stated that all data exists and is available.
