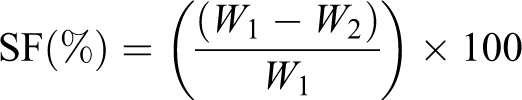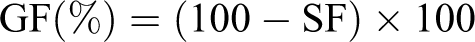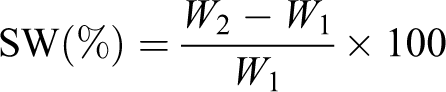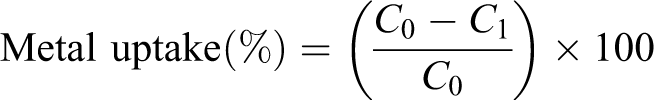Abstract
Superabsorbent hydrogels based on natural polymer, carboxymethyl cellulose (CMC)/sodium alginate (SA) were prepared by 60Co γ radiation as a source of initiation of cross-linking. The effect of different ratios of SA (20, 30, 40, and 50%) on the physical properties of the CMC/SA hydrogel such as gel fraction percentage (GF%), and swelling percentage (SW%) were investigated. It was found that the GF % decreases with increasing SA content in the hydrogel. While the SW% of CMC/SA hydrogel tends to increase with increasing SA content in the hydrogel at constant irradiation dose (2.5 kGy). Morphology of the hydrogels was examined using scanning electron microscopy, which indicates compatibility between CMC and SA. Thermal properties were also investigated using thermogravimetric analysis. It was found that the thermal properties of the hydrogel having different composition were almost the same. The results obtained from ultraviolet–visible spectrophotometric analysis show that the prepared hydrogels can be used in the removal of heavy metals from waste water. The ability to absorb these heavy metals is of great importance from the point of view of environmental pollution.
Keywords
Introduction
Superabsorbent hydrogels are three-dimensional cross-linked hydrophilic, linear, or branched polymers with the ability to absorb large quantities of water, saline, or physiological solutions compared with general absorbing materials. 1,2 Because of their excellent hydrophilic properties, high swelling ratio, and biocompatibility hydrogels have been widely used in agriculture, 3 biomedical areas as antibacterial materials, 4 tissue engineering, 5 and biosensors, 6,7 and sorbents for the removal of heavy metals 8 and drug delivery. 9,10
Superabsorbent hydrogels prepared with natural materials, such as cellulose, 11 starch, 12 dextran, guar gum, 13 and chitosan, 14 because of their abundant resources, low production cost, and biodegradability, have attracted great attention. 15 It is worth noting that natural polymers have better biocompatibility and less latent toxic effect than most synthetic polymer hydrogels. 16,17 So, pure natural polmer hydrogels would be more suitable for biomaterials. 18,19 Carboxymethyl cellulose (CMC), as a water-soluble cellulose derivative is easily cross-linked with solutions of multivalent metal cation to produce hydrogels, is a particular carrier for pesticides, 20 while CMC-sodium (Na) was previously believed to undergo degradation upon radiation treatment, 21 the primary event, upon the CMC-Na exposure to high-energy radiation in the dry form includes the formation of ions resulting from the scission of the main polymer backbone bonds C–C, C–O, or C–H. These ions are rapidly converted into free radicals, which are responsible for the subsequent polymer degradation. 22
Alginate is a natural biopolymer derived from brown seaweed. 23 Also, it is hydrophilic, biocompatible, and relatively economical. It has been widely used in medical applications such as wound dressings, scaffolds for hepatocyte culture, and surgical or dental impression material, even if the allergic reaction to skin has been occurred. 24,25
Recently, some attempts were made to cross-link cellulosic materials by radiation in the presence of water. 26 –29
Generally, the properties of resulting blends can be studied to explore further applications for biomedical and pharmaceutical devices. Furthermore, Na-CMC, a cellulose derivative, has been used in wound dressing since it is biocompatible, hydrophilic, nontoxic, and nonallergenic. 30,31 Because both sodium alginate (SA) and CMC are water-soluble polymers, they are compatible polymers due to the formation of hydrogen bonds. There are numerous studies reported on the material properties for the respective pullulan, alginate, and CMC films. 32,33
Increased SA concentration in the polyvinyl alcohol/SA hydrogel matrix decreased the gelation percentage, maximum strength, and elongation at break, but it resulted into an increment in the swelling ability, elasticity, and thermal stability of hydrogel film. 34
CMC/SA blend has been prepared by casting solution method. The effect of different doses (2.5, 5, 10, 15, and 20 kGy) of γ rays on the physical properties such as gel fraction percentage (GF%) and swelling percentage (SW%) of CMC/SA blends containing different ratios of SA was investigated. 35
In this study, the effect of the addition of different concentration of SA to the CMC hydrogel on the properties of CMC/SA hydrogel was examined. The cross-linking density of the prepared hydrogel was determined in terms of GF%. The swelling property of the prepared hydrogels was measured to identify its hydrophilic property. The thermal property in terms of thermogravimetric analysis (TGA) and morphology of the hydrogels by scanning electron microscopy (SEM) indicates that there is compatibility between CMC and SA. The ability to absorb some heavy metals from waste water was determined.
Experimental
Material
Na salt of CMC (pure polymer) in the form of granules, molecular weight 180,000, was supplied by El-Nasr Pharmaceutical Chemical-Prolabo (Egypt). N,N-Methylene bisacrylamide (MBAM) supplied by Aldrich (Madison, Wisconsin, USA) was used as a cross-linking agent. SA of chemical grade was purchased from Shanghai Chemical Reagents Company (Shanghai, China).
Preparation of CMC/SA hydrogel
CMC/SA hydrogel was prepared by dissolving different compositions of CMC/SA (100, 80/20, 70/30, 60/40, and 50/50%) in a known volume of distilled water at room temperature. MBAM as cross-linking agent (0.1 wt%) was added to the polymer solutions, then mixed with continuous stirring until complete miscibility, were poured onto petri dishes, and finally exposed to 20 kGy γ radiation.
γ Irradiation
The irradiation process was carried out using a Co-60 γ cell (made in Russia) at the National Center for Radiation Research and Technology, Cairo, Egypt, with a dose rate 6.92 kGy h−1. Irradiation was carried out under atmosphere.
Gel fraction percentage
A known weight (W
1) of the dried and weighted hydrogel was extracted using water in a Soxhlet apparatus for 24 h and then dried to a constant weight in vacuum (W
2). The soluble fraction (SF) was determined according to the following equation:
Thus, GF% was calculated using the following equation:
Swelling percentage
A known dry weight of the insoluble part of the hydrogel (W 1) after calculation of the soluble fraction was immersed in distilled water at room temperature for 24 h. The swollen part of the hydrogel at equilibrium was weighed (W 2).
SW% is determined using the following equation:
where W 1 and W 2 represent the weights of the dry and wet sample.
Thermogravimetric analysis
TGA studies were carried out using a TGA-30 apparatus (Shimadzu, Kyoto, Japan) at a heating rate of 10°C min−1 in air over a temperature range from room temperature to 600°C.
Scanning electron microscopy
The surface morphology of the CMC/SA hydrogel was examined using a JSM-5400 SEM (JEOL, Japan).
Sorption of heavy metal ions
An attempt was made to use the CMC/SA hydrogel for removal of some heavy metals such as copper (Cu2+), nickel (Ni2+), and Co2+ from waste water. Different metal salt solutions (cupric sulfate pentahydrate (CuSO4 5H2O), nickel sulfate hexahydrate (NiSO4 6H2O), and cobalt sulfate hexahydrate (CoSO4 6H2O)) of known concentrations (500 ppm) were first prepared. A sample of known weight (0.5 g) of the hydrogel was then immersed in these solutions for different time intervals ranging from 1 to 24 h. The residual metal solution was measured using ultraviolet (UV) spectrophotometer. The metal uptake (%) was determined using the following equation:
where C 1 and C 0 are the concentrations of the metal solution before and after sorption by modified polymers, respectively.
Results and discussion
Gel fraction percentage
The dependency of GF% on CMC/SA hydrogel ratio was given in Figure 1. From this figure, it was found that pure CMC possesses a high value of GF % when compared with CMC/SA hydrogel with different compositions. Furthermore, increasing the SA ratio in the hydrogel is accompanied with a decrease in the GF% at a fixed dose of 2.5 kGy. 35 Finally, it can be concluded that there is a marked increase in the GF% of hydrogel especially for hydrogel rich in CMC due to cross-linking as for example GF% was about 64.1, 69.4, 71.5, 73.5, and 77% at 50, 60, 70, 80, and 100% CMC, respectively.
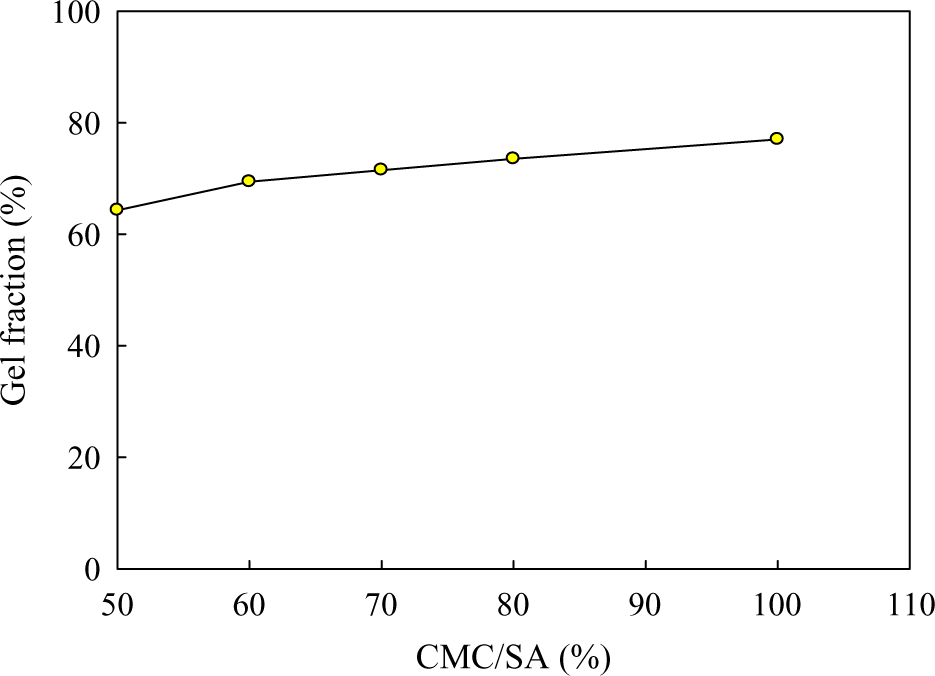
GF% of pure CMC and CMC/SA hydrogels with different compositions at 2.5 kGy. GF%: gel fraction percentage; CMC: carboxymethyl cellulose; SA: sodium alginate.
Swelling percentage
The SW% of CMC/SA hydrogel has been studied at different compositions of CMC (50, 60, 70, 80, and 100%). Figure 2 shows that the SW% of CMC/SA hydrogel nearly tends to increase with increasing SA content in the hydrogel. The amount of absorbed water by the hydrogel highly depends on SA content.. The reason for lowered SW% is due to crystallinity of CMC chains that can cause the percolation of water molecules around the insoluble crystals and this impedes the diffusivity of water molecules through the hydrogel. 36
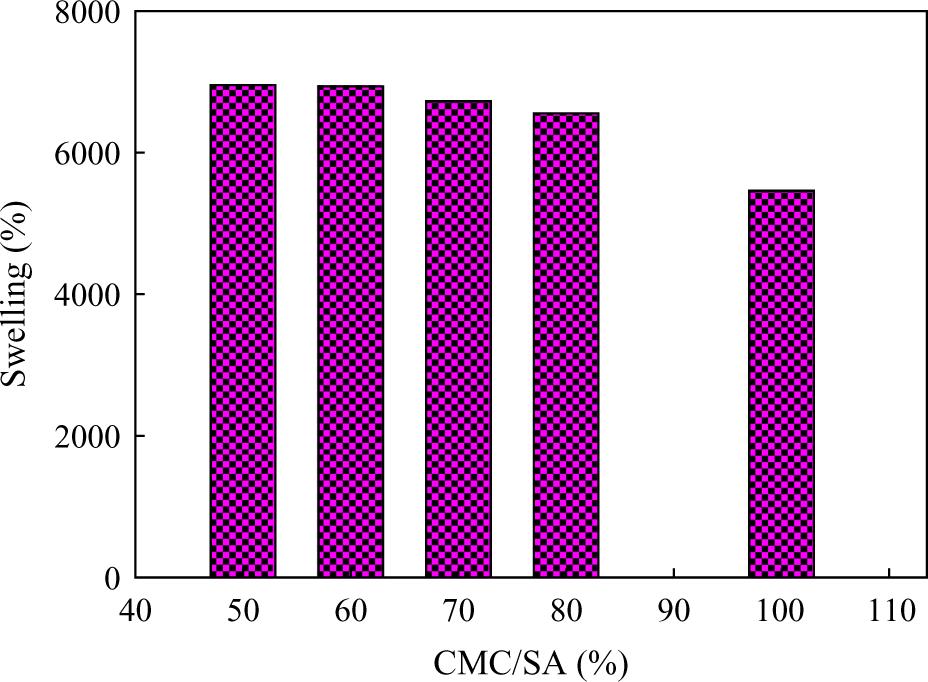
Effect of pure CMC and CMC/SA hydrogel with different composition on SW%. CMC: carboxymethyl cellulose; SA: sodium alginate; SW%: swelling percentage.
Thermogravimetric analysis
Thermal properties for hydrogel at various elevated temperatures are shown in Figure 3 and Table 1. From this figure, it can be deduced that pure CMC and different compositions of CMC/SA hydrogel have nearly the same weight loss. For example, at 280°C, the weight loss for pure CMC, 80% CMC,60% CMC, 50% CMC was equal to 10,12, 15, and 20, respectively.
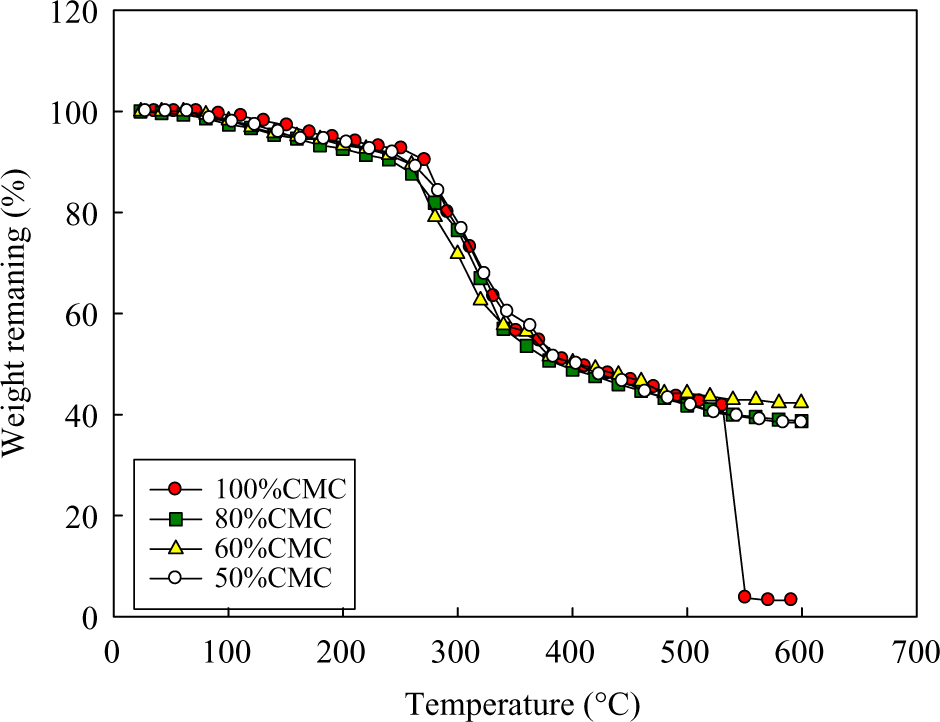
Thermal analysis of pure CMC and CMC/SA with different compositions. CMC: carboxymethyl cellulose; SA: sodium alginate.
Weight loss percentage at different decomposition temperatures for pure CMC and CMC/SA hydrogel with different compositions.
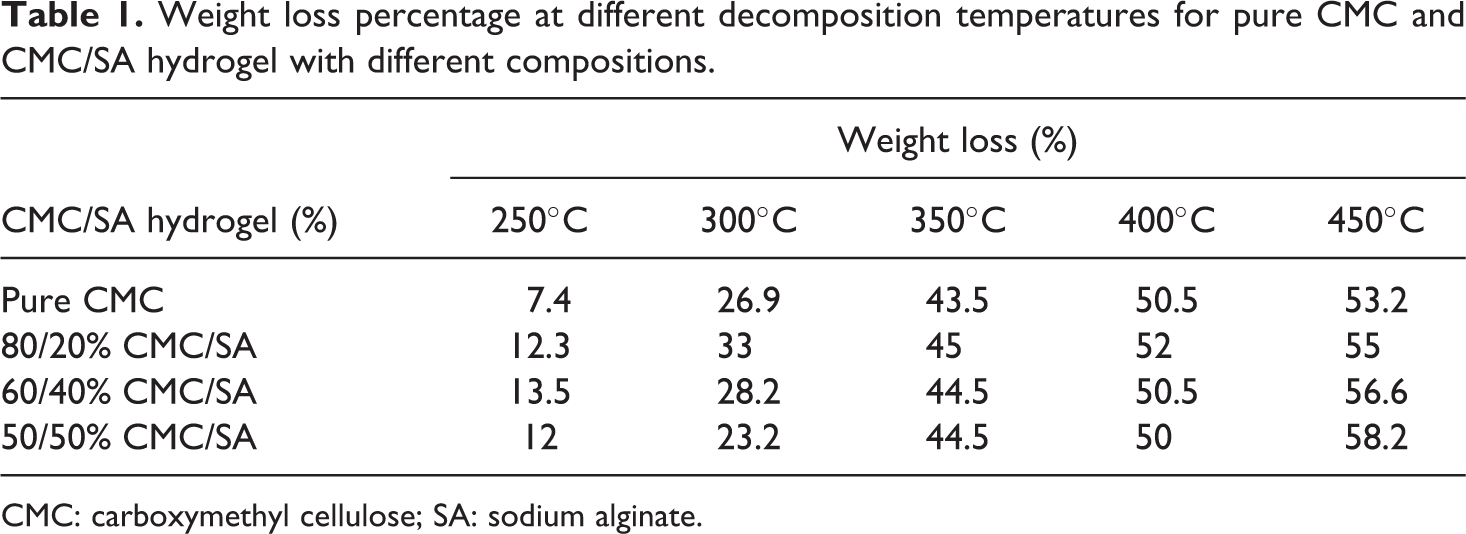
CMC: carboxymethyl cellulose; SA: sodium alginate.
It is suggested that in the initial stage of the thermal decomposition in the temperature range from 0 to 100°C, the weight loss is due to the dehydration process of water contained in such hydrophilic hydrogel. At the second stage, that is, in the temperature range of 100–380°C, there is decomposition in the side groups and the branches of the hydrogel. However, at the third stage, above 380°C, the weight loss is due to the main chain scission in the hydrogel chain.
Results showed that all the prepared CMC/SA hydrogels with different composition possess good thermal stability suitable for all industrial applications.
Scanning electron microscopy
The surface morphology of pure CMC and CMC/SA hydrogels with different compositions were investigated using SEM as shown in Figure 4. The SEM micrographs of pure CMC is characterized with corn flakes texture and also is characterized with the presence of cavities and holes as shown in Figure 4(a). On the other hand, the SEM micrographs of CMC/SA hydrogels showed a different morphology. This different morphology is due to the presence of different compositions (80, 70, 60, and 50%) of SA in the hydrogel as shown in Figure 4(b) to (e). This different morphology indicated that the compatibility between CMC and SA increases by increasing the ratio of SA in the hydrogels.

SEM micrographs of different hydrogels (a) Pure CMC, (b) 80%CMC, (c) 70%CMC, (d) 60%CMC, and (e) 50% CMC). SEM: scanning electron microscopy; CMC: carboxymethyl cellulose.
Metal ions uptake
From the practical point of view, it is valuable to use some hydrogels for minimizing environmental pollution. 37 The hydrogel prepared in this work is investigated for the treatment of waste water containing some heavy and toxic metals. The efficiency of hydrogel is investigated by considering the following parameters: maximum metal uptake, selectivity toward different metal ions such as Cu, Ni, and Co.
The metal ions uptake percentage was determined using UV/visible spectrophotometric analysis based on measuring the absorption of different colored solution for different metal ions before and after metal uptake process.
Figures 5 to 7 show the relationship between metal ion uptake percentage and immersing time For Ni2+ , Cu2+, and Co2+, respectively. It can be seen that the metal uptake percentage increases with time to reach a certain limiting value, which is called the maximum metal uptake capacity. The maximum uptake is reached at certain immersion time depending on the type of metal ion.
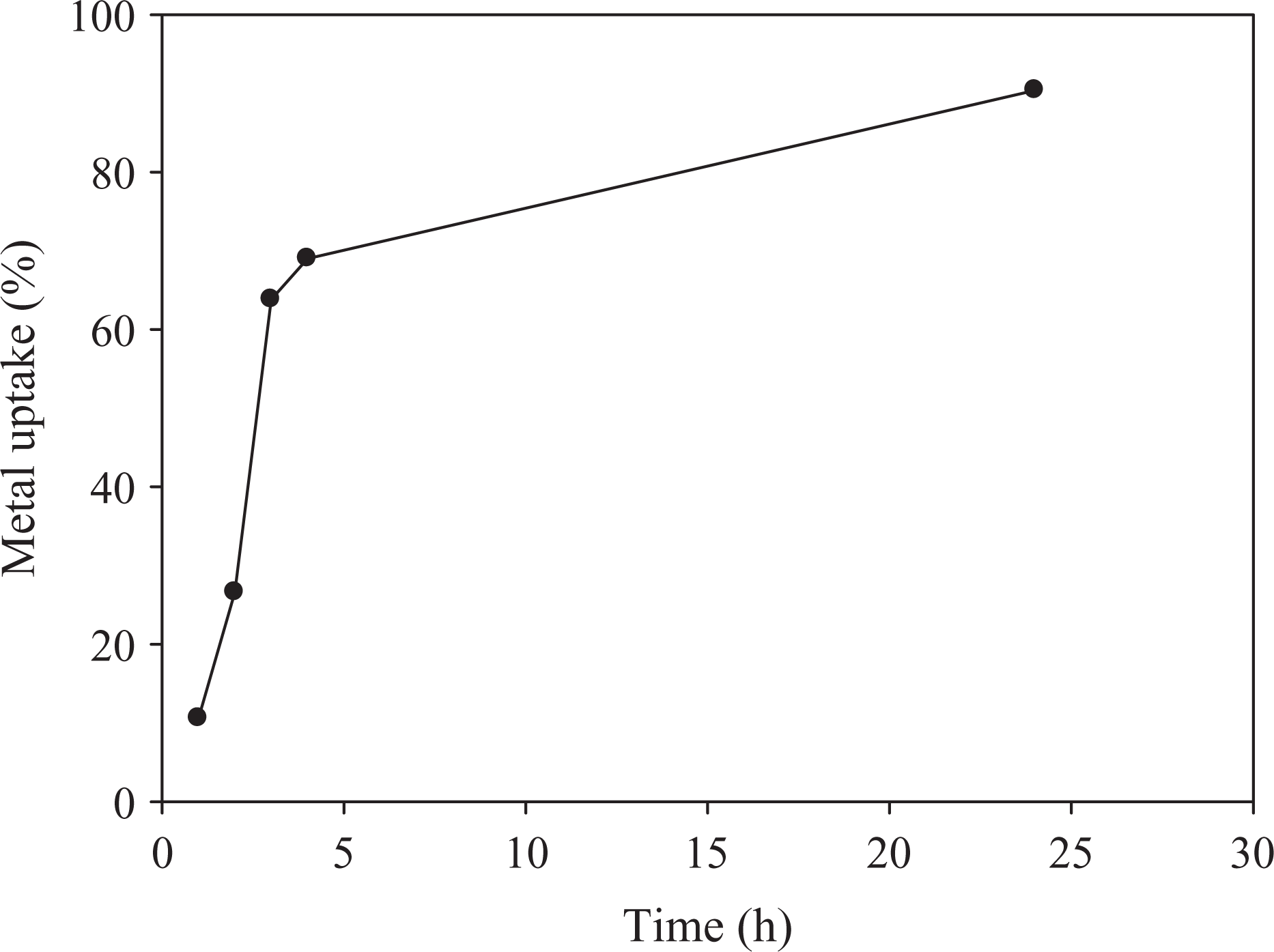
Metal uptake of Ni by 80% CMC and 20%SA. Ni: nickel; CMC: carboxymethyl cellulose; SA: sodium alginate.
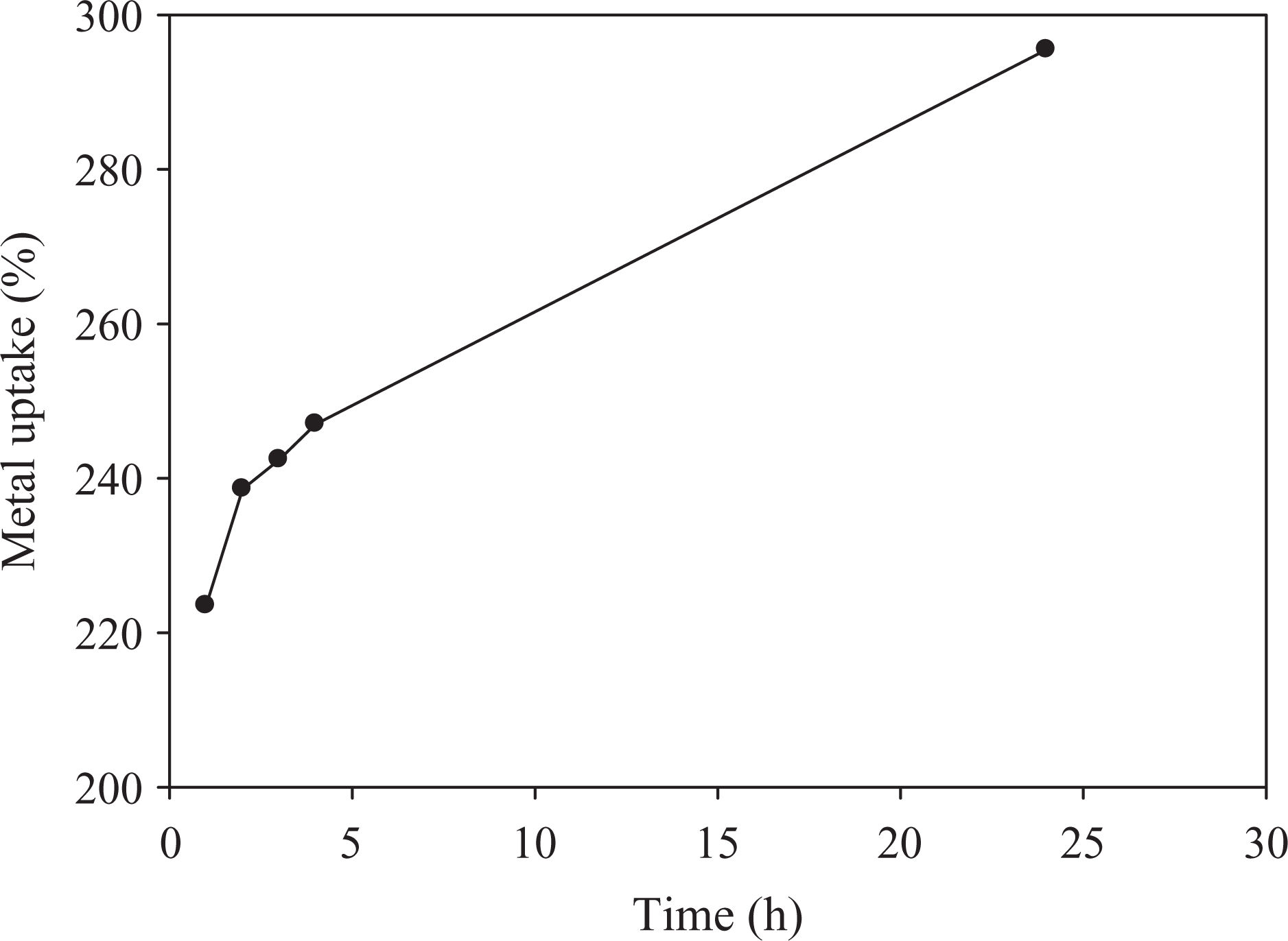
Metal uptake of Cu by 80% CMC and 20%SA. Cu: copper; CMC: carboxymethyl cellulose; SA: sodium alginate.
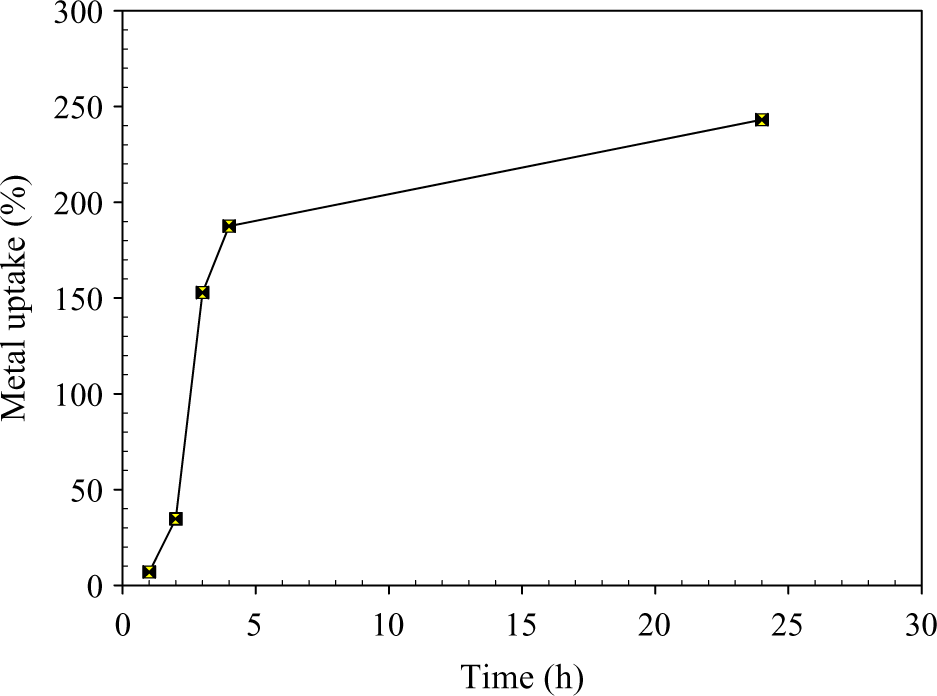
Metal uptake of Co by 80% CMC/20%SA. Co: cobalt; CMC: carboxymethyl cellulose; SA: sodium alginate.
The defeasibility of such metals through the pores of the hydrogel and the stability of chelation of metal ions to polymer hydrogel depends on atomic size, ionic valency, and ionic radii of metal ions. The affinity of CMC/SA hydrogel toward such metal ions may be attributed to the presence of free hydroxyl groups along its molecular structure which permit the occurrence of ion-exchange reaction. These superabsorbent hydrogels were successfully used in waste water treatment. This is because the modified hydrogel can absorb some heavy metal ions by coordination, formation of hydrogen bonding and/or dipole association, or through hydrogen bonding of water molecule.
Results show that the hydrogels have a tendency toward metal ions according to the following order; Cu2+ > Co2+ > Ni2+.
Conclusion
Hydrogels of CMC/SA was prepared by radiation cross-linking using 60Co γ irradiation and the gelation dose was 2.5 kGy. The optimum concentration of CMC/SA was found to be 80%/20%.
The SW% increases with increasing the SA ratio in the hydrogel. TGA thermograms were used to study the thermal stabilities of the hydrogel. It was found that the prepared hydrogel posseses good thermal stability suitable for all industrial applications.
SEM was used to identify the compatibility of the hydrogel for pure CMC and CMC/SA with different ratio. The application of the prepared hydrogels as superabsorbents for recovery of Cu2+, Co2+, and Ni2+ from waste water was studied. It was found that the prepared hydrogel has a great ability to recover the metal ions from their solution in the following order Cu2+ > Co2+ > Ni2+.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.