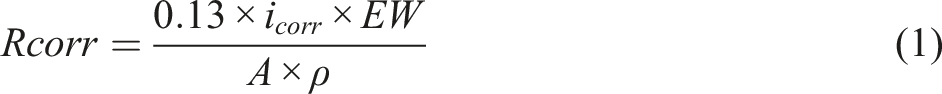Abstract
Polyurethanes (PU) are widely used in corrosion protection, anti-biofouling coatings, and self-cleaning glass, yet their low hydrophobicity poses durability challenges. This study addresses this by enhancing PU coatings’ hydrophobicity by refining surface roughness and forming tailored porous structures. Using drop, spin, dip, and spray coating techniques, hydrophobic PU coatings were fabricated on 304 steel plates. To further enhance hydrophobicity, 0.5 w/v% graphene nanoplatelets (GNP) were introduced into the coatings. Additionally, stainless-steel 500 mesh was incorporated to create patterned surfaces, increasing surface roughness and tailoring the porous structure. The analysis included optical and scanning electron microscopy, water contact angle (WCA) measurements, Fourier transform infrared spectroscopy, electrochemical corrosion tests, and atomic force microscopy. Incorporating GNP notably increased WCA by 20–23° and reduced corrosion rates in a 3.5% NaCl solution. Likewise, applying stainless-steel mesh improved surface roughness, raising WCA by 3-8° and decreasing corrosion rates. The study observed changes in porous structure, with pore size reduction correlating with increased roughness and decreased corrosion rates upon mesh application and GNP addition. This comprehensive examination highlights the potential of PU coatings for various applications and underlines the importance of tailored porous structures in enhancing their performance and versatility. Spin coating emerged as the optimal technique, yielding uniform, defect-free coatings with the highest WCA.
Keywords
Introduction
Steels, valued for their tensile strength, affordability, and availability, 1 are susceptible to corrosion from environmental factors like humidity, saltwater, acid rain, and pollutants. Metal corrosion results in significant annual economic losses worldwide, with the World Bank estimated a global cost of USD 6 trillion in 2021. 2 Corrosion leads to significant economic losses globally and threatens infrastructure integrity. 3 Protective coatings, particularly organic coatings such as polyurethane (PU), are widely used to combat corrosion by providing a barrier against environmental exposure. 1
Organic coatings, such as epoxy and polyurethane (PU), have emerged as promising candidates for corrosion protection.1,4 PU coatings, in particular, offer numerous advantages, including flexibility, strong adhesion, environmental stability, and corrosion resistance. 5 However, their effectiveness can be further enhanced by improving hydrophobicity, a property crucial for preventing water ingress and inhibiting corrosion initiation. One common strategy to enhance hydrophobicity is by increasing the roughness of organic surface,6,7 achieved through various means such as the incorporation of nanoparticles and replication processes.8,9 While this approach has shown promise, it is essential to acknowledge that the resulting coatings may contain pores, which could potentially compromise their performance in certain applications. Despite the presence of pores, coatings can still find utility in various applications where impermeability is not a primary concern. For example, in applications such as anti-biofouling coatings and self-cleaning surfaces, the presence of pores may not significantly impact effectiveness.10,11 Pores can also contribute to enhanced surface roughness, which is beneficial for promoting air retention and inhibiting the adhesion of contaminants.
Past research has explored the incorporation of fillers, including graphene nanoplatelets (GNP), into PU coatings to achieve superhydrophobic properties and improved corrosion resistance.12–14 Moreover, surface patterning, including the use of meshes, has demonstrated effectiveness in enhancing surface roughness and corrosion resistance. 9 However, there remains a significant gap in the literature regarding the systematic study of how different coating techniques influence the surface properties of PU/GNP coatings. Techniques like spray, spin, dip, and drop coating can uniquely alter the surface structure, roughness, and energy, thereby impacting the overall performance of the coating.15–18 To date, no comprehensive research has been conducted on the effects of different coating techniques combined with surface patterning on PU/GNP coatings.
This study seeks to fill this gap by systematically investigating the impact of coating techniques on the surface morphology, hydrophobicity, and corrosion resistance of PU coatings containing GNP and patterned with stainless-steel mesh. By employing drop, dip, spin, and spray coating methods, this work aims to elucidate the relationship between the coating technique and the final properties of the coatings. Each technique was selected based on its ability to uniquely influence the film’s surface characteristics, which are critical for optimizing the coating’s performance. A thorough comparison of these techniques provides valuable insights into how the chosen method affects the final properties of PU/GNP coatings. This work contributes to the field by offering insights into the optimization of PU/GNP coatings for enhanced performance across diverse applications, despite potential porosity issues.
Materials and methods
Preparation of PU and PU/GNP coatings
304 stainless steel (Chromium-Nickel austenitic alloy, Sunrise Stainless Steel Sdn Bhd) was cut into 10 mm × 10 mm × 5 mm dimensions. Before application of PU, the steel plates were etched with a 3:1 ratio of HNO3 (Merck) and HCl (Sigma Aldrich, Selangor) solution. The etched steel plates were then cleaned with ethyl alcohol, and rinsed with deionized water. 400 mm2 316 steel 500 mesh was ultrasonically rinsed in the water bath at 25°C for 10 min. Then the steel-mesh was dried using an air gun. PU solution was prepared by the solution mixing method. PU pellets (Fabbxible Technology) were dissolved at a concentration of 15 w/v% in dimethylformamide (DMF, Sigma Aldrich) and kept stirring for 1 h by magnetic stirring. To prepare the PU/GNP solution, the GNP powder (Sigma Aldrich) was first dispersed at 0.5 w/v% in DMF for 30 min by ultrasonication. The 0.5% GNP concentration was selected based on previous studies that demonstrated excellent corrosion resistance at this level. 19 Then, 15 w/v% TPU pellets were dissolved in the GNP dispersion solution and mixed thoroughly using a magnetic stirrer for 1 h to obtain a homogeneous PU/GNP solution.
The hydrophobic polyurethane coating was prepared using PU and PU/GNP solution via different fabrication methods; drop casting (1 drop of PU), spin coating (3 drops of PU, spinning speed 3000 r/min), dip coating (dipping time 1 min), and spray coating (distance 15 cm, 5 times spraying). After the samples were coated, steel-mesh was laid on the coating surface for 10 s to create a patterning effect on the coating before drying the coatings. Then the samples were left for solvent evaporation.
The selection of drop, spin, dip, and spray coating techniques was based on their distinct advantages in controlling film morphology, thickness, and uniformity, which are crucial for enhancing the hydrophobicity and corrosion resistance of PU/GNP coatings. Drop coating was chosen for its simplicity and ability to form thick films, ideal for evaluating bulk properties. Spin coating, known for its precision in controlling thickness and uniformity, was employed to create smooth, thin layers and assess the distribution of GNP. Dip coating, which ensures uniform coverage over complex geometries, was selected for consistent PU/GNP deposition, critical for corrosion protection. Lastly, spray coating, with its scalability and efficiency in covering large areas, was included to evaluate how rapid deposition affects surface roughness and overall coating performance.
Surface morphology
The surface morphology was investigated via an Olympus POS polarizing microscope with 15 magnification and a Field Emission Scanning Electron Microscope (FESEM). FESEM model Zeiss Supra 35 VP was employed to monitor the surface morphology of the coatings at 1000 magnification. The samples were first coated with gold-palladium using the sputtering method before the observation. The average pore size of the samples was measured using ImageJ software.
Water contact angle (WCA)
WCA of the coatings was evaluated via Ossila optical contact angle goniometer model L2004A1. The measurement was carried out by dropping one drop of deionized water on the coating surface. The contact angle of coatings was measured after 1 min of deposition using DSA4 software. One-way ANOVA was performed using Minitab version 19 using α = 0.05.
Fourier transform infrared (FTIR)
Perkin Elmer Spectrum One FTIR spectrometer was employed to examine the type of functional groups present in the samples. The drop casted sample was scanned 32 times with wavelength numbers between 4000 cm−1 to 550 cm−1.
Electrochemical corrosion test
Electrochemical measurement was conducted using the Frequency Response Analysis (FRA) autolab model PGSTST302 N. The electrochemical workstation was constructed with a three-electrode arrangement cell and 3.5 wt% NaCl electrolyte. A saturated calomel electrode served as the reference electrode, a graphite electrode as the counter electrode, and coated samples were employed as the working electrode. The coated samples had a surface area of 100 mm2. Tafel analysis was conducted through different immersion intervals of coated samples in NaCl solution. The analyzed frequency ranges between 0.01 Hz and 100 kHz with an amplitude of 10 mV. The corrosion potential (Ecorr), corrosion current density (icorr), and corrosion rates (Rcorr) were determined using equation (1) as described in ASTM standard G102:
Surface roughness
The surface roughness of PU and PU/GNP coatings prepared via the spin coating method were measured using Atomic Force Microscopy, AFM (SPM-SPA400) in non-contact mode with a scanning area of 5 × 5 µm2.
Results and discussion
Surface morphology
The surface morphology was examined using an optical microscope to assess the homogeneity of samples from four different coating techniques (Figure 1). Coatings formed via drop casting and dip coating exhibited uneven surfaces, primarily due to blistering defects observed in the drop-casted sample and the presence of excess, high-viscosity coating solution adhering to the substrate during the drainage step of the dip coating technique. Conversely, spray-coated samples exhibited high porosity, as evidenced by SEM micrographs (Figure 2). Notably, spin-coated samples displayed the best surface finish among all techniques. OM images of PU and PU/GNP coatings: (a) drop casting, (b) dip coating, (c) spin coating, (d) spray coating, (e) drop casting-GNP, (f) dip coating-GNP, (g) spin coating-GNP, and (h) spray coating-GNP (4 magnification). SEM images of PU and PU/GNP coatings with and without mesh: (a) Drop casting, (b) Drop casting-mesh, (c) Drop casting-GNP, (d) Drop casting-GNP-mesh, (e) Dip coating, (f) Dip coating-mesh, (g) Dip coating-GNP, (h) Dip coating-GNP-mesh, (i) Spin coating, (j) Spin coating-mesh, (k) Spin coating-GNP, (l) Spin coating-GNP-mesh, (m) Spray coating, (n) Spray coating-mesh, (o) Spray coating-GNP, and (p) Spray coating-GNP-mesh (1000 magnification).
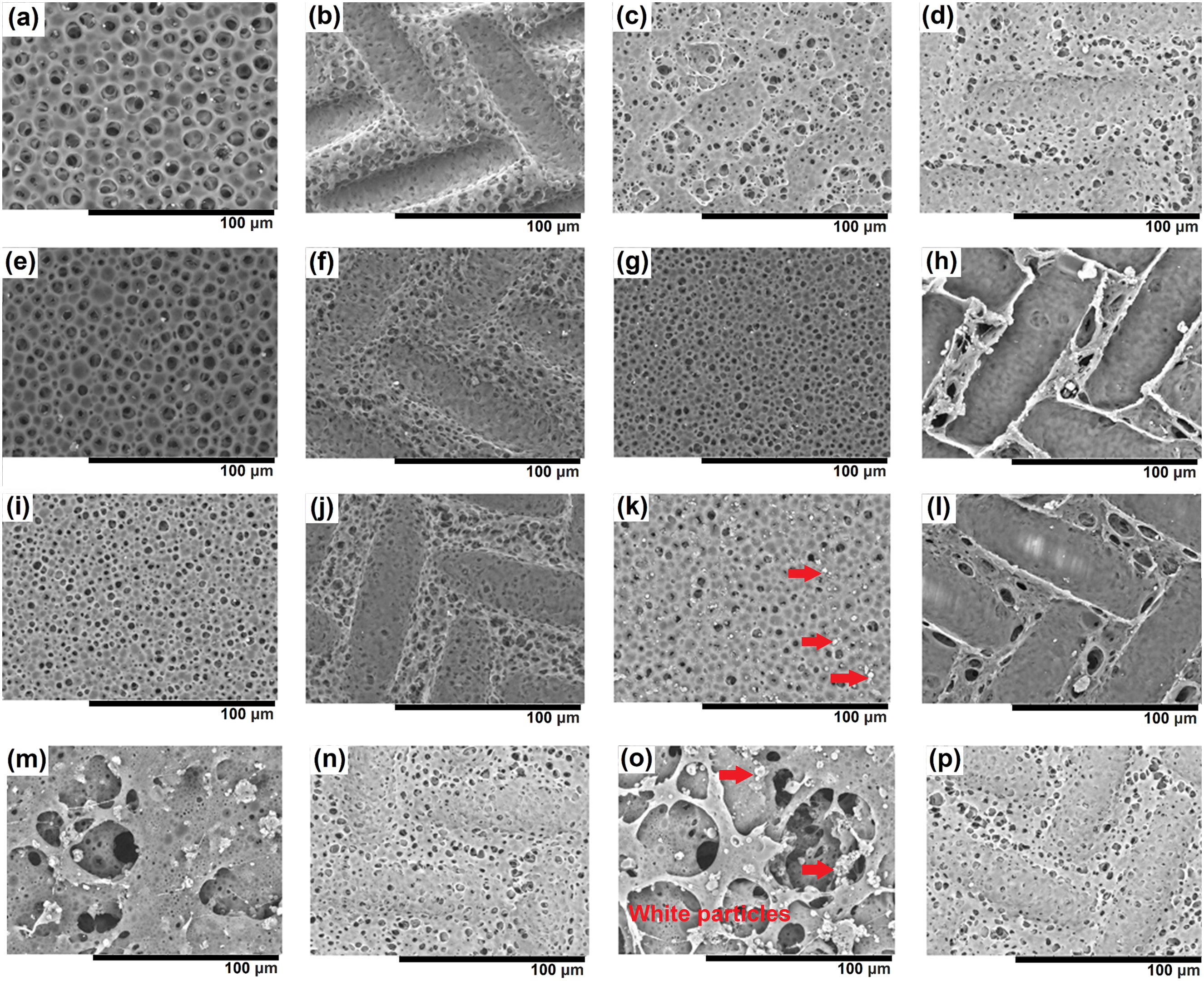
The surface morphology of the coatings prepared using different techniques varies significantly, as illustrated by Figure 2. These variations are largely attributed to the unique deposition mechanisms inherent to each method. The drop-casting technique results in an uneven surface with visible blistering and less uniform pore distribution (Figure 2(a)), likely due to trapped air or residual solvent during the drying process. This irregular drying pattern creates pockets within the coating, leading to a rougher, less uniform surface. Similarly, the dip coating method exhibits an uneven surface, though the pores appear more uniformly distributed than those in drop casting pores (Figure 2(e)). This is often due to the excess high-viscosity solution that remains on the substrate during the draining step, which can pool and create non-uniform film thickness and structural irregularities as the solvent evaporates. Both methods produce surfaces with substantial defects that may negatively affect the coating’s overall performance. On the other hand, spin coating produces much more homogeneous surface morphology pores, as the centrifugal force applied during the spinning process evenly distributes the coating material across the substrate. This leads to finer and more uniform pores, with minimal defects observed (Figure 2(i)). The resulting surface is smooth and consistent, making spin coating the most effective method for achieving a high-quality finish. In contrast, spray coating results in a highly porous structure with larger, more irregular pores (Figure 2(m)). The high velocity of the spray droplets and rapid solvent evaporation contribute to the formation of these large pores and voids, resulting in a less uniform coating.
The incorporation of graphene nanoplatelets (GNP) into the PU matrix significantly altered the surface morphology of the coatings, particularly in samples prepared by drop casting, spin coating, and dip coating (Figure 2). While there were minimal changes in the surface morphology of spray-coated samples after the addition of GNP, other techniques showed distinct alterations. White particles, highlighted by arrows in Figure 2(k) and (o), were observed in PU/GNP coating samples, attributed to the presence of GNP distributed evenly throughout the samples. Furthermore, the surface of PU coatings exhibited a homogeneous distribution of larger pores, which were notably reduced in size upon the addition of GNP. The average pore size reduction ranged from 10.4% to 58.3% after the incorporation of GNP, as calculated using ImageJ (Figure 3). The incorporation of GNP reduces pore size due to its ability to alter the microstructure of the polymer matrix. GNP, with its high surface area and layered structure, can disperse within the PU matrix to physically occupy pores, form continuous networks, and bridge gaps, thereby reducing pore size and creating a more uniform surface. A previous study reported that dispersed graphene fills the pores in waterborne epoxy coatings, effectively sealing them.
20
In addition, nanofillers such as cerium oxide, silica, and zinc oxide are used as anti-corrosion agents in polymer coatings.21–23 These inorganic nanofillers enhance the barrier properties of the coatings by occupying the pores and reducing the diffusion of corrosive substances. Average pore size of PU and PU/GNP coating with and without mesh prepared using (a) drop casting and dip coating, (b) spin and spray coating.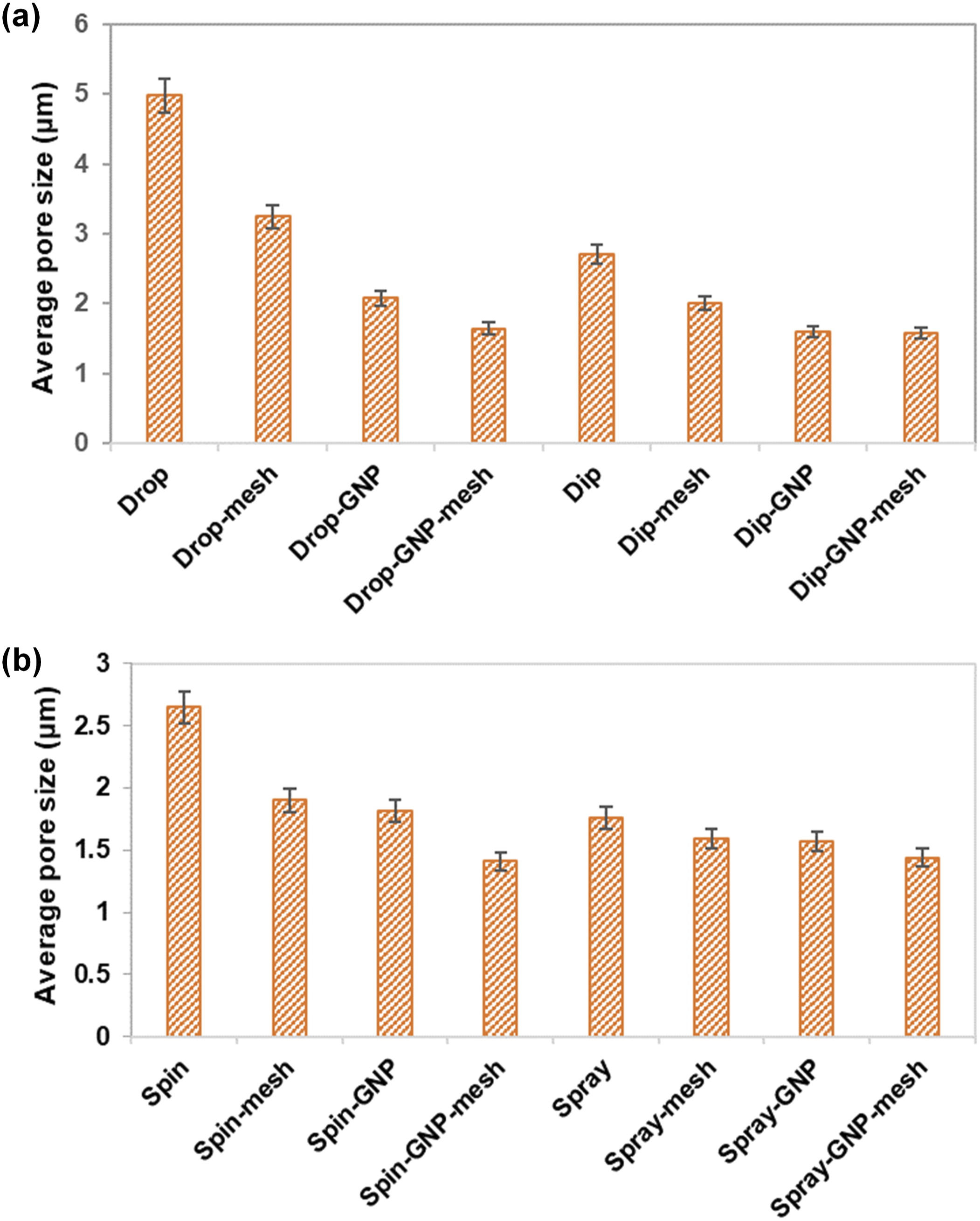
Moreover, the application of stainless-steel mesh resulted in further changes to the surface morphology of PU and PU/GNP coatings. It promoted micro-scale surface roughness with an intricate topographical structure and a notable reduction in pore size observed across all coating techniques. The application of mesh appears to introduce a physical constraint on the surface, potentially acting as a template that controls the spreading and distribution of the polymer during the coating process. The mesh likely influences the flow of the polymer, leading to a more controlled deposition, which in turn limits the formation of larger pores by providing a structural boundary. 24 When both GNP and mesh are applied, the combined effect results in enhanced control over the polymer’s deposition and microstructure. The mesh constrains the surface topography, while the GNP further tightens the network structure within the polymer, leading to a significant reduction in pore size. The interaction between the GNP and the structured mesh promotes an even more compact matrix by simultaneously controlling the surface and bulk properties.
The observed alterations in surface morphology, particularly the reduction in pore size following the addition of GNP and the application of stainless-steel mesh, have significant implications for corrosion protection. Smaller pore sizes can potentially improve the coatings’ ability to act as a barrier against corrosive agents by reducing the pathways for ingress. Furthermore, the uniform distribution of GNP throughout the coatings may enhance their mechanical properties, leading to improved resistance against environmental degradation. These findings suggest that the modifications made to the surface morphology of the coatings have the potential to enhance their corrosion resistance, thus contributing to their suitability for diverse applications requiring robust protection against corrosion.
Water contact angle (WCA)
The WCA measurements of PU and PU/GNP coatings prepared using four distinct techniques are presented in Figure 4. Across all coating techniques, the incorporation of GNP and application of mesh pattern significantly enhanced the WCAs of the PU coatings. Initially, the WCAs of the PU coatings ranged from 90° to 103°, indicating the inherent hydrophobic nature of pure PU. This relatively low hydrophobicity is attributed to the abundance of surface polar urethane functional groups, which tend to interact with polar water molecules.
14
WCA measurement of PU and PU/GNP coatings, with and without mesh prepared using (a) drop casting and dip coating, (b) spin and spray coating.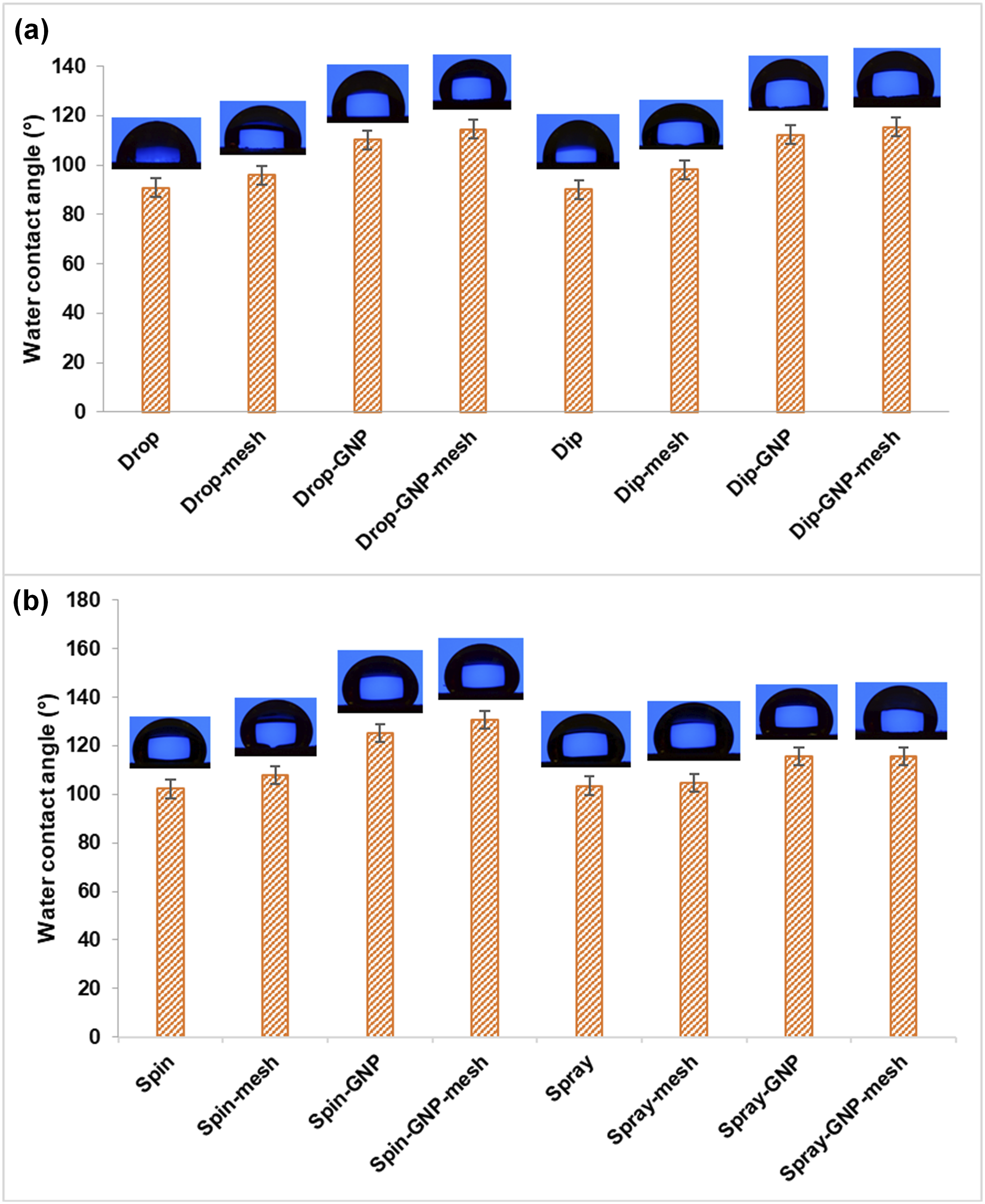
The introduction of graphene nanoplatelets (GNP) notably elevated the hydrophobicity of PU by 20–22°. This enhancement is attributed to the non-polar and high surface area properties of GNP, which possess only minimal carboxylic groups at their edges. 25 The incorporation of 0.5 wt% GNP effectively covered a substantial surface area within the PU matrix, resulting in an overall increase in hydrophobic properties. Additionally, the application of stainless-steel mesh to the coatings further improved the WCA of both PU and PU/GNP nanocomposite coatings by approximately 3–8°. The application of the mesh pattern induces a micro-scale texture on the surface of the coatings. This increased roughness facilitates the formation of air pockets at the interface between the water droplet and the surface, thereby effectively reducing the contact area. When the surface exhibits sufficient roughness, the water droplet can rest atop these air pockets, resulting in an elevated contact angle. However, the WCA increment observed in PU and PU/GNP nanocomposite coatings produced by the spraying technique was negligible after the application of stainless-steel mesh. This observation suggests that the surface roughness of the coatings fabricated through the spraying technique was maintained even after the incorporation of the mesh.
A one-way ANOVA was performed to compare the water contact angles across the four coating methods (drop, dip, spin, and spray coating), revealing a statistically significant difference (F (3,4) = 157.68, p < .001), as the p-value is smaller than σ (0.05). Among the four coating techniques, the spin coating method resulted in the highest WCA values, ranging from 102° to 130°. This is likely due to the formation of finer and more uniform pores. These microstructures lead to a smoother, well-dispersed coating with enhanced hydrophobicity, as smaller and more uniform pores create fewer areas for water molecules to penetrate, promoting the water droplet’s stability on the surface. The drop coating method exhibited a less uniform pore distribution, which could result in blistering defects and larger, more irregular areas on the coating surface. These defects provide potential entry points for water, reducing the overall hydrophobicity. Similarly, despite having more uniform pores, the dip coating method involves excess viscous solution during the drainage step, leading to an uneven surface and lowering the WCA. Additionally, the WCA of coatings formed by the spraying method ranged from 103° to 115°, suggesting that the spraying method can produce a coating surface with low surface energy, forming a micro-roughness surface structure that consequently improves the hydrophobicity of the coating.25,26 Considering these structural differences, it is clear that the spin coating method, with its finer and more uniform pores, provides a more favorable microstructure for enhancing hydrophobic properties.
A subsequent one-way ANOVA analysis was performed on the spin coating method with the application of mesh and the addition of GNP (mesh, GNP, and GNP-mesh). The analysis revealed a significant difference in water contact angle (F (3,4) = 410.51, p = .000, σ = 0.05, indicating that these additions significantly enhance the hydrophobicity of the spin-coated surfaces. The incorporation of GNP and the application of stainless-steel mesh in spin-coated PU coatings significantly enhance their hydrophobicity by modifying the coating’s microstructure. GNP addition reduces the average pore size, creating a tighter polymer matrix, which limits water penetration and increases the WCA. The mesh introduces micro-scale surface roughness, forming air pockets between the water and the surface, further enhancing the WCA. When both GNP and mesh are applied together, they create a synergistic effect, with the mesh controlling surface topography by introducing roughness, while GNP refines the internal structure by reducing pore size. This combination produces a well-structured, highly roughened surface with minimal pore size, resulting in the highest hydrophobicity, as evidenced by the highest WCAs in the Spin-GNP-mesh coating.
Comparison of pore characteristics and water contact angles of various coating techniques.

FTIR analysis
Figure 5 shows the FTIR spectra of PU and PU/GNP coating formed via spin coating. It reveals the characteristic peaks that correspond to the major functional groups found in polyurethane. The peak at 3325 cm−1 is attributed to the -NH urethane group, the peak at 2950–2800 cm−1 region is due to aliphatic hydrocarbon (C-H), and the peak at 1700 cm−1 is referred to the C = O stretching.14,27 The bands at 1220 cm−1, and 1065 cm−1, 765 cm−1 indicate the typical peaks of PU in the range of 500-1500 cm−1, which are attributed to C-O-C and C-H stretching vibrations, respectively.28,29 The characteristic bands of PU are also observed in the PU/GNP nanocomposite. FTIR spectra of PU and PU/GNP coating formed by spin coating.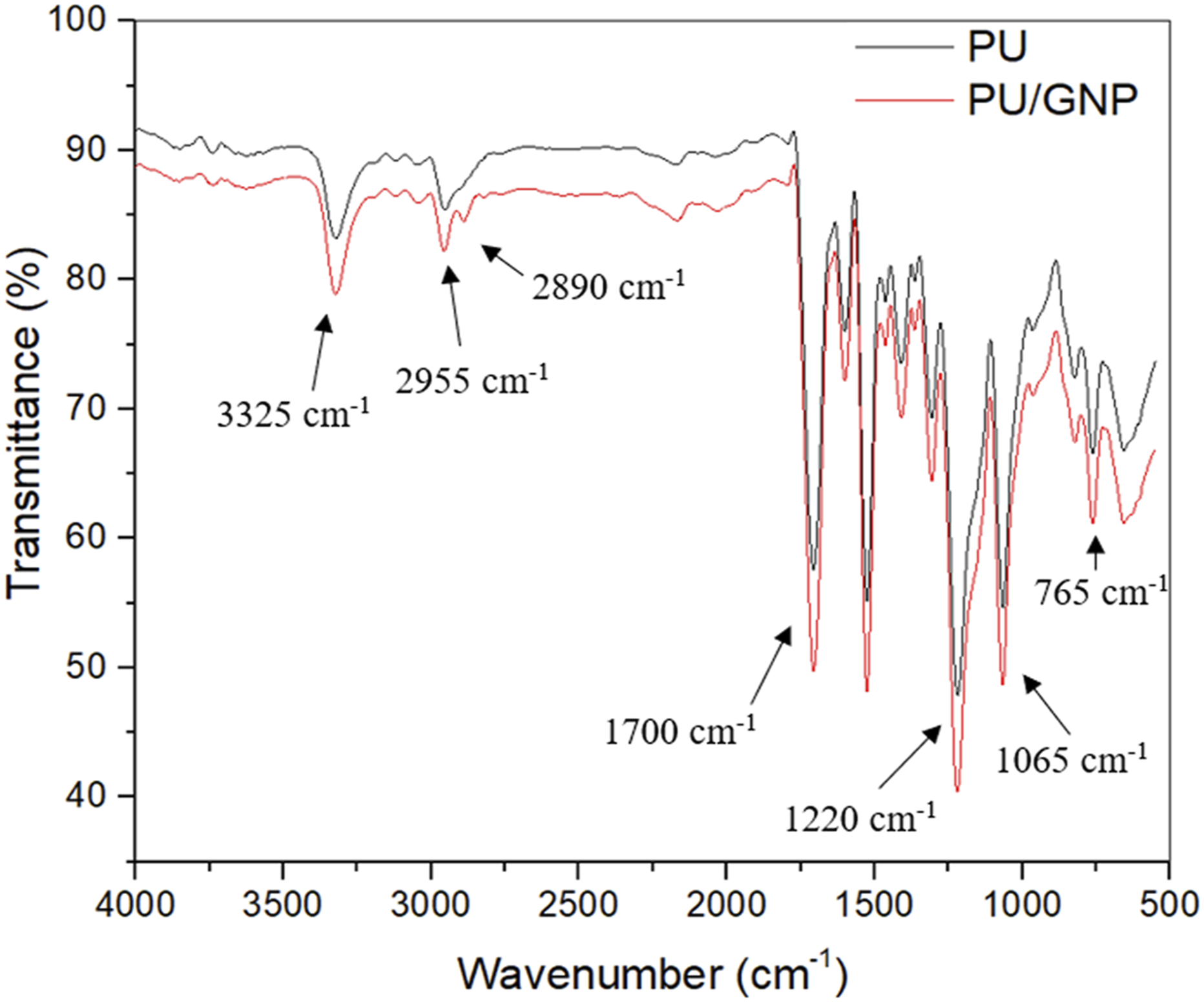
Corrosion resistance
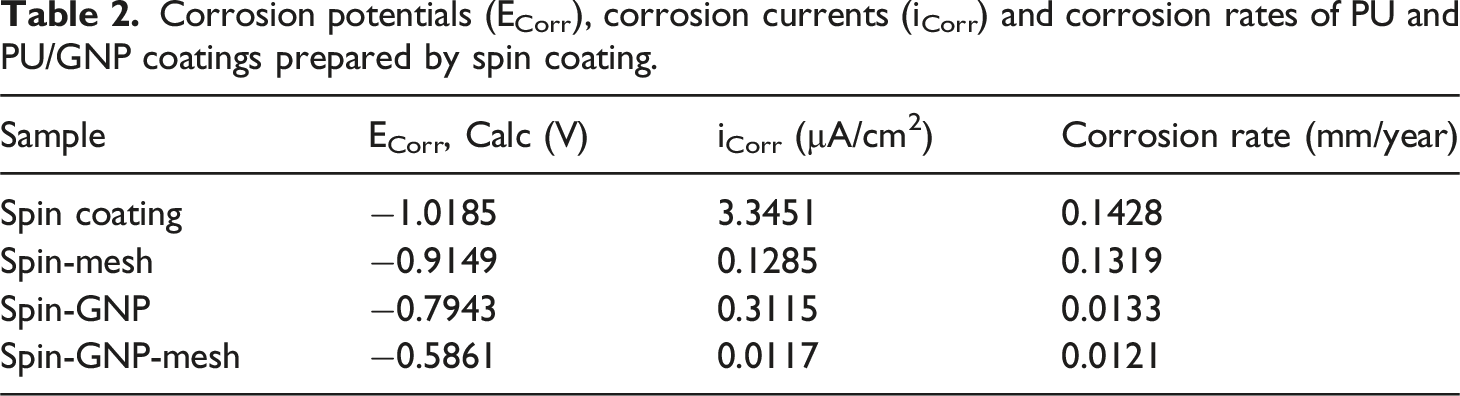
Tafel plot analysis (Figure 6) was employed to assess the corrosion rates of PU and PU/GNP coatings prepared via the spinning method, chosen due to their superior range of water contact angle (WCA) values. Despite the presence of porosity observed in the coatings, the application of mesh onto the coatings and the incorporation of GNP into the PU matrix resulted in a positive shift in corrosion potential (Ecorr), indicating enhanced corrosion resistance. This shift suggests increased difficulty for the corrosion reaction to occur, thus reducing the likelihood of encountering corrosive media. Tafel plots of PU and PU/GNP coatings prepared by spin coating.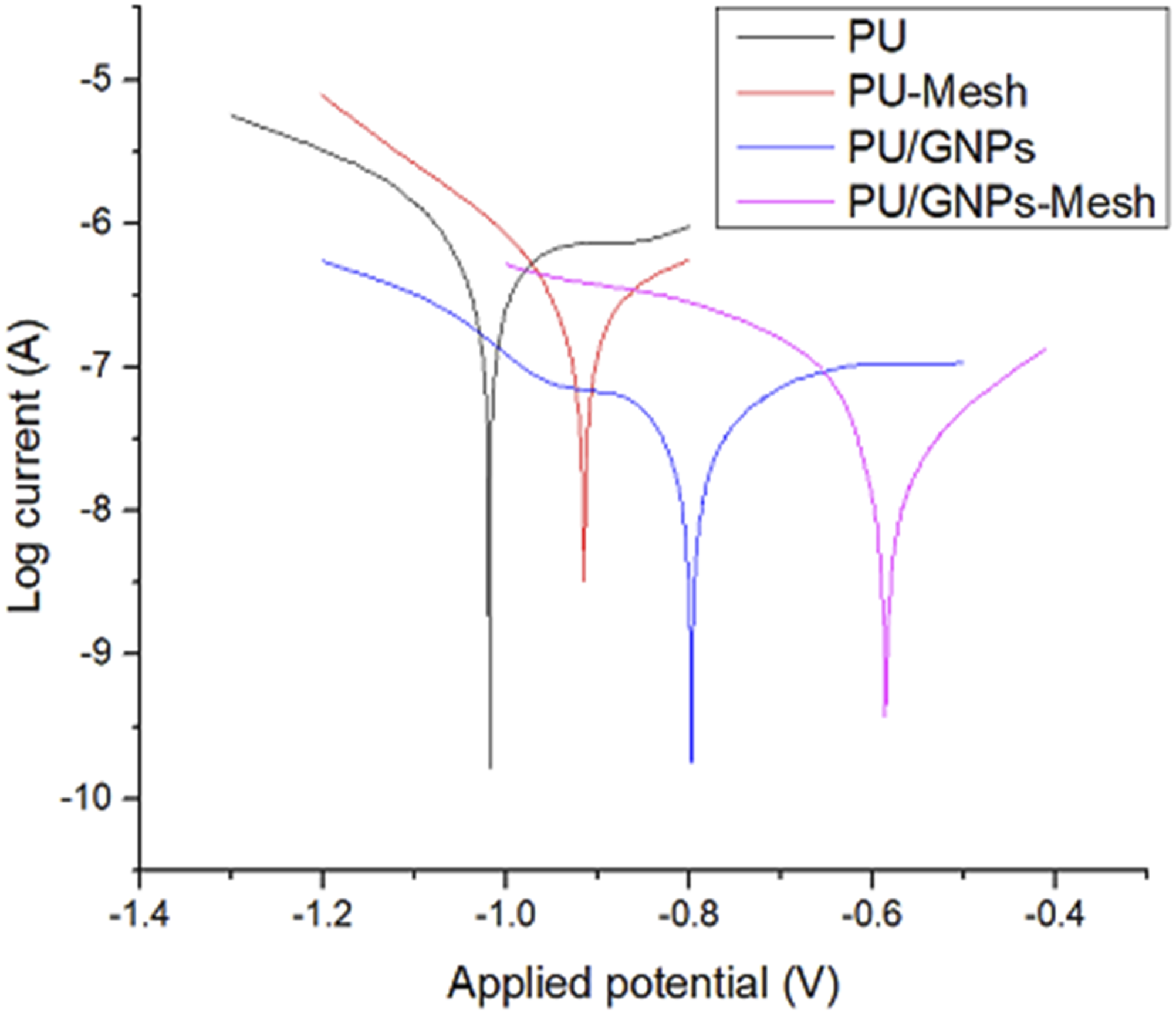
The introduction of GNP into the PU matrix served a dual purpose: filling existing gaps and pores while impeding the corrosive medium’s access to the substrate. By acting as a barrier, GNP hindered the diffusion pathways of oxygen, effectively enhancing the coatings’ resistance to corrosion. This strategy addresses the challenge posed by porosity, as the GNP fills the pores, reducing their size and limiting the penetration of corrosive agents. In another study, Liu et al. prepared and investigated graphene-reinforced waterborne epoxy coatings, finding that the dispersed graphene effectively blocked pores within the coating, thereby reducing electrolyte penetration to the metal surface and improving protection. 20
Additionally, the application of stainless-steel mesh onto the coatings contributed to increased surface roughness, facilitating the retention of air on the hydrophobic surface. This retained air layer acted as a passivation layer, effectively inhibiting the corrosion process by reducing the contact area between the hydrophobic surface and the corrosive media. The unique mesh patterning introduces a hierarchical roughness that creates micro-scale features, enhancing the surface’s ability to trap air. This architecture not only increases the stability of the air layer but also promotes the formation of a protective barrier against corrosive agents. Consequently, the presence of the air layer prevented corrosive agents like chloride ions from reaching the substrate surface, providing an additional layer of corrosion protection. 30
Corrosion potentials (ECorr), corrosion currents (iCorr) and corrosion rates of PU and PU/GNP coatings prepared by spin coating.
Surface roughness
The surface roughness of PU and PU/GNP coatings (with and without mesh) prepared via the spinning method were evaluated by AFM to observe their topography (Figure 7). Figure 8 compares the average surface roughness (RMS) of the coatings where the surface roughness increases upon application of mesh and addition of GNP, and GNP with mesh. The surface roughness increases with the application of mesh and incorporation of GNP in the PU coating. The RMS value increased slightly (24%) after the application of mesh on PU coating whereas upon incorporation of GNP, the RMS value of PU/GNP coating increased by 121%. The RMS value increased dramatically from 15.7 to 47.9 nm (205%) after the application of mesh for PU/GNP coating. AFM topography image of coatings formed by the spinning method. (a) Spin coating, (b) Spin coating-mesh, (c) Spin coating-GNP, (d) Spin coating-GNP-mesh. Average surface roughness of coatings formed by the spinning method.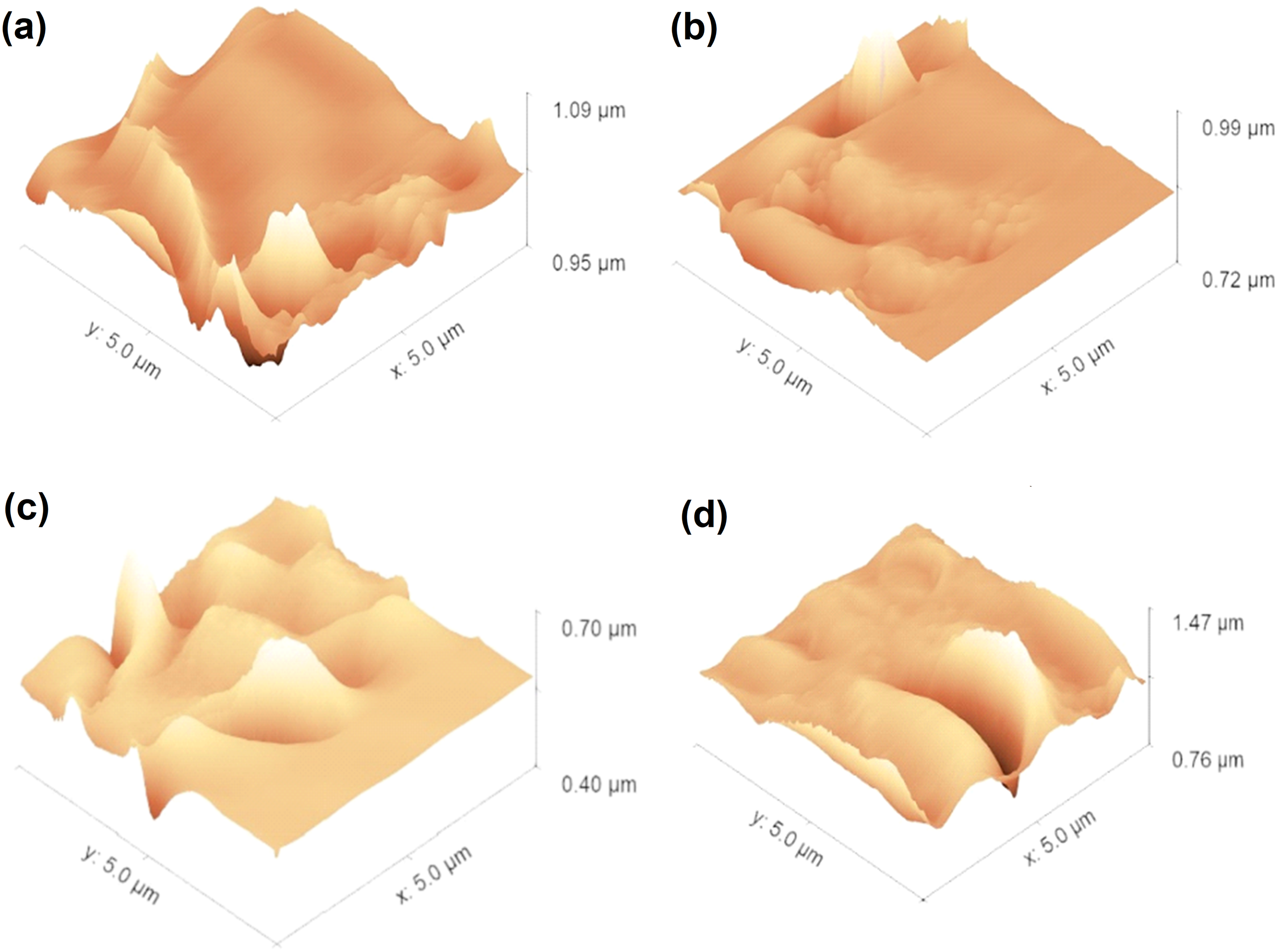
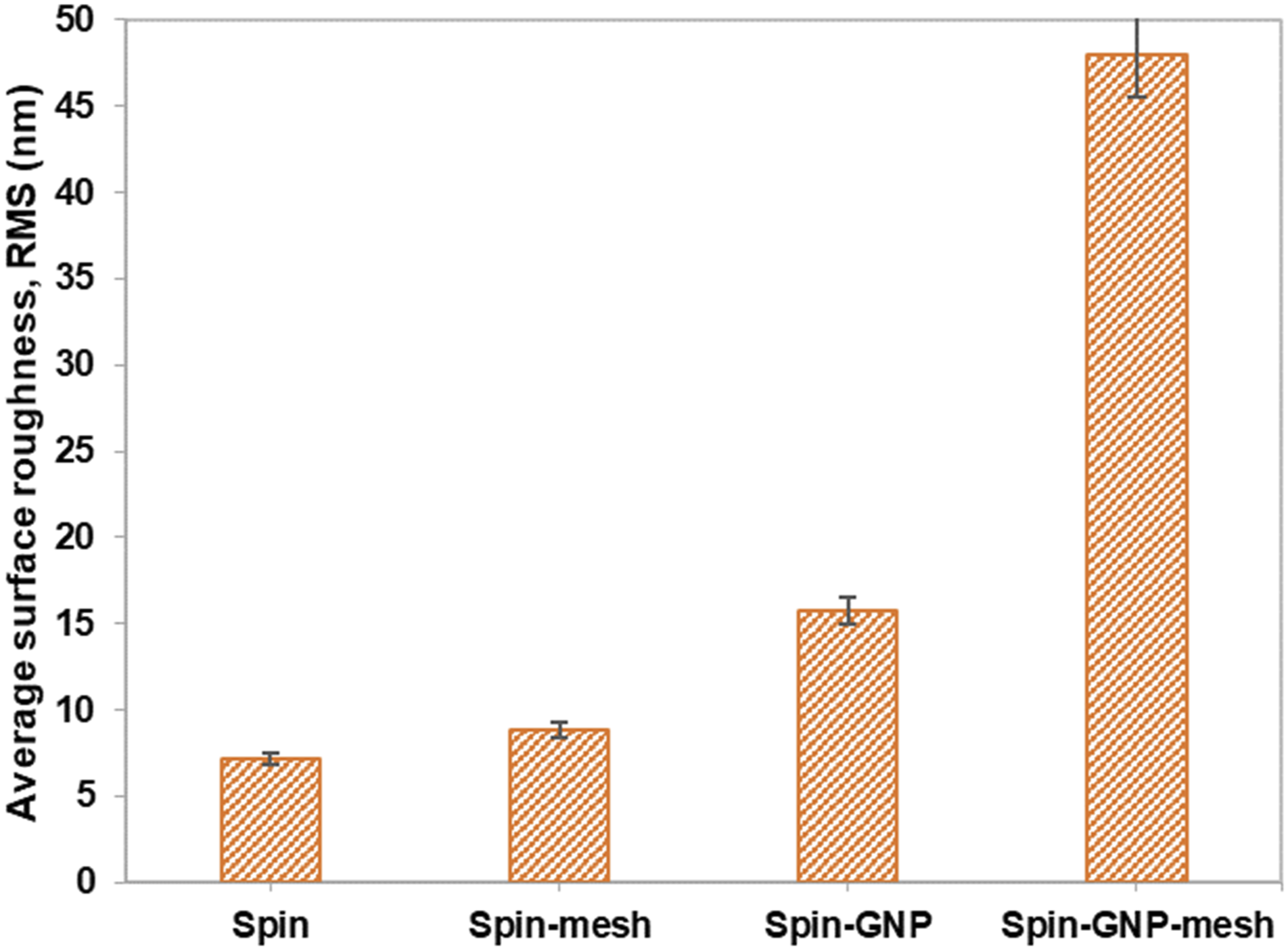
The results reveal an interesting trend where pore size reduction (Figure 3) correlates with increasing surface roughness. The application of the stainless-steel mesh enhances the surface texture by introducing additional micro-scale features, which contribute to a more complex topography. This increased roughness allows for better air retention and aids in masking or filling the pores within the coating. As observed in the AFM images, the coatings with added GNP and mesh exhibit a more uniform surface texture, indicating a reduction in pore size and a corresponding increase in surface roughness. This is consistent with the concept that the presence of pores contributes to surface irregularities, which are reflected in higher surface roughness values.
The coating microstructure, particularly the presence of pores, surface roughness, and surface energy, significantly influences corrosion rates by dictating how corrosive agents interact with the substrate. The reduction in pore size, combined with increased surface roughness, plays a crucial role in enhancing corrosion protection. Uniformly distributed smaller pores, formed through the addition of GNP and the application of mesh, slow down corrosion by trapping air and limiting the diffusion of corrosive species. Additionally, coatings with higher surface roughness and lower surface energy are more hydrophobic, reducing water ingress and corrosion initiation. The observed correlation between pore size reduction, increased roughness, and decreased corrosion rates indicates that the addition of mesh and GNP, both separately and combined, improves the coatings’ resistance to corrosion. While these structural improvements enhance corrosion protection, excessive porosity or surface roughness could negatively affect other properties, such as mechanical strength and long-term durability. High roughness may create stress concentration points, compromising the coating’s performance under mechanical or environmental stress.
The ideal coating microstructure in this study consists of small, uniformly distributed pores and increased surface roughness, achieved by incorporating both GNP and mesh to enhance hydrophobicity and corrosion resistance. This structure traps air and limits water ingress, slowing the diffusion of corrosive agents. The findings suggest that among the four methods prepared, spin coating offers the best performance, with superior hydrophobic properties and reduced pore size, which improves corrosion resistance. However, for industrial applications, scalability, cost-effectiveness, and ease of application must also be considered. Techniques like spray and dip coating, though less optimal in performance, excel in these practical aspects. Thus, combining the advantages of spin coating with the practicality of spray or dip coating could lead to the development of advanced coatings that perform well both in controlled settings and real-world applications.
Conclusion
In this study, we successfully developed polyurethane coatings with enhanced hydrophobicity and corrosion resistance using a variety of techniques, including drop casting, dip coating, spin coating, and spray coating. By incorporating graphene nanoplatelets (GNP) into the PU matrix and applying stainless-steel mesh onto the coatings, we aimed to improve their performance for demanding marine applications. Among the methods explored, spin coating emerged as the most effective, yielding coatings with the highest hydrophobicity, as evidenced by an increase in water contact angle (WCA) from 102° to 125° upon the addition of GNP. Additionally, the application of stainless-steel mesh further augmented hydrophobicity, leading to a 5° increase in WCA for coatings prepared via spin coating. Structurally, the introduction of GNP led to a reduction in pore size, while the application of mesh served a dual role by diminishing and constricting pore structures while enhancing overall surface roughness. This combination of pore size reduction and surface roughness enhancement contributed to improved corrosion resistance of the coatings. Analysis of corrosion parameters showed significant improvements with both mesh application and GNP incorporation, resulting in lower corrosion current density (icorr) and decreased corrosion rates.
While spin coating demonstrated superior hydrophobic properties, spray and dip coating methods are more practical for industrial applications. Future research could focus on integrating the advantages of spin coating with the scalability and practicality of spray or dip coating techniques. The novelty of this work lies in the synergistic use of GNP and stainless-steel mesh to enhance both hydrophobicity and corrosion resistance in polyurethane coatings. Additionally, optimizing formulation and processing parameters to improve mechanical properties and long-term durability, exploring alternative nanofillers, and assessing performance under dynamic marine conditions would further expand the potential applications of these coatings. Evaluating the scalability of these techniques for industrial use will ensure they meet the demands of sectors such as anti-biofouling and corrosion-resistant coatings for marine environments.
Footnotes
Acknowledgements
The authors express appreciation to the School of Materials and Mineral Resources of Engineering, Universiti Sains Malaysia for its facilities.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.