Abstract
Due to the organic porous nature of expanded polystyrene (EPS) foam concrete, the bond strength between it and cement is weak, leading to a weakened interface transition zone and low strength. However, most studies on this issue have been limited to phenomenological approaches, neglecting the role of atomic-level mechanisms. Therefore, this study combines macroscopic and microscopic experiments, as well as molecular dynamics, to investigate the mechanisms of a silane coupling agent (SCA) enhancing the interface performance of EPS-concrete. Macroscopic mechanical tests demonstrate that SCA can improve the compressive and flexural strength of EPS concrete. Combined with the test and analysis of variance, the addition of 9% SCA has the best enhancement effect on the mechanical properties of EPS composite concrete. The compressive strength increased by 24% and 17% on day 7 and day 28, respectively, and the flexural strength increased by 39% and 16% on day 7 and day 28, respectively. At the microscopic scale, scanning electron microscopy, X-ray diffraction, and Fourier transform infrared spectroscopy are used to analyze the effects of SCA on the microstructure and functional groups of the EPS-cement interface. Microscopic analysis shows that SCA optimizes the composition of cementitious products, effectively repairing and improving the defects of the EPS-cement interface. Ultimately, molecular dynamics simulations and analysis are conducted on two interface models, one including and one excluding SCA, at the nanoscale. The results indicate that SCA enhances the hydrogen bonding, H-Ca ion bonding, and interface interactions between EPS-C-S-H, resulting in a tighter and more stable bond between the two phases, effectively enhancing the mechanical properties of EPS composite concrete. This study provides valuable insights into the strengthening mechanisms of EPS composite concrete and proposes a promising improvement method.
Keywords
Introduction
Due to the swift progression of industrialization and the development industry in many nations, traditional concrete is now inadequate to fulfill construction needs because of its considerable heaviness and proneness to cracking. Therefore, there is an urgent need for new types of concrete with higher quality and better performance to fill the research and development gap.1,2 Compared to traditional concrete, recycled aggregate concrete uses reprocessed materials to partially or completely replace coarse aggregate, reducing the overall weight of the concrete structure.3–5 Modified concrete can increase the properties of concrete through the use of modifiers. Additionally, it improves the meso-structure of concrete, providing a guarantee for enhancing the mechanical properties of concrete.6–8 Among them, expanded polystyrene (EPS) concrete is a typical new type of recycled aggregate concrete and modified concrete. Due to its lightweight properties, effective insulation, impressive sound absorption capabilities, and energy-efficient qualities, this material is extensively utilized in construction industries of different nations. Additionally, its ability to reduce noise pollution and carbon emissions make it a popular choice among builders.9–11 However, EPS concrete has some problems such as poor porosity, toughness and low strength. Moreover, the adhesion between EPS beads and cement is poor, resulting in weak interfacial transition zone.12,13 This greatly limits the further development of polystyrene foam new concrete in the fields of structure and construction.
Polystyrene foam and cementitious materials are two distinct phases, with significantly different physicochemical properties. Furthermore, EPS concrete belongs to the category of lightweight aggregate concrete. Lightweight aggregate concrete not only exhibits poor bonding properties, but also faces challenges related to uneven mixing.14,15 These problems result in numerous weak interfacial transition zones inside the polystyrene foam concrete, which, in turn, lead to premature failure due to poor interfacial contact between the two materials, resulting in unsatisfactory mechanical properties of the specimens.16–18 To solve this problem, this study used modified additives to improve weak interfaces and further enhance the performance of the composite material. SCA is a modifier used to improve the bonding strength and compatibility between concrete and other composite materials. Utilizing this method is not only highly efficient and cost-effective, but it also serves as an eco-conscious approach to enhancing the mechanical characteristics of polymer concrete. Additionally, it effectively addresses the issue of inadequate interfacial transition zones between polystyrene foam and cement-based materials.19,20 The hydrolysable and organic functional groups present in SCA have the ability to interact with both inorganic and organic substances. This interaction plays a vital role in enhancing the interfacial bonding between silicon-based materials and improving the mechanical characteristics of composite concrete systems.21,22 Falliano et al. 23 discovered that SCA has the ability to increase the adhesion between fiber-cement composites and bolster the compressive and flexural properties of the experimental samples. Liu et al. 24 explored how varying levels of SCA impact the compressive strength and elastic modulus of EPS samples using experimental analysis. The findings indicate that incorporating 9% SCA notably enhances the performance of EPS samples.
Previous studies have shown that silane coupling agents (SCAs) can improve the performance of EPS concrete. Previous research is limited to macroscopic phenomenological engineering methods, especially in the study of micro-nano improvement mechanisms, which is still in the initial stage. Therefore, in order to fully analyze the mechanism of its improvement, it is necessary to continue to explore at the nanoscale. Numerous academics have verified that molecular dynamics (MD) simulation is a valuable computational tool for studying material characteristics and forecasting mechanical reactions. 25 Chowdhury, SC et al. 26 used MD software to prepare a glass-epoxy resin interface with a single layer of glycidyl oxyel trimethoxysilane by changing the silane number density between glass and epoxy resin and established a method to predict the strain-rate-dependent mixed mode traction-separation response of the glass-epoxy resin composite interface by using MD simulation. Yeon, J et al. 27 simulated the surface reactivity and hydrolysis reaction of quartz glass and S glass using MD to study the temperature conditions of annealing on four surfaces (300 K, 700 K, 1000 K, and 1300 K). The results show that the density of surface defects and hydroxyl groups is higher at lower surface annealing temperatures. Compared with S glass, silicon glass shows more physical adsorption of water and surface energy. Kang et al. 28 utilized a blended strategy of experimentation and simulation to examine the alteration impact of SCA on the interface of composite rubber concrete. The results showed that introducing SCA molecules into weak interfaces can effectively repair interface defects between rubber beads and cementitious materials, thereby improving their mechanical properties. Cui et al. 20 studied the interface properties of three different functional group SCAs grafted onto silica and asphalt binders through molecular dynamics simulations. They found that there is a thick transition zone between the modified asphalt and aggregate, and the interface bonding performance is significantly improved after modification with SCA.
With its benefits of being extremely lightweight, easy to build with, and eco-friendly, EPS concrete is widely utilized in the construction sector.29,30 It is applied in a variety of ways, including load-bearing precast concrete elements, thermal insulation panels, cushioning or structural insulation panels, composite floors, pavement bases, building envelope structures, and offshore floating structures. These constructions exhibit great strength, superb insulation properties, high resistance to fire, excellent water repellency, cost-effectiveness, and strong abilities for absorbing sound and reducing noise. Nevertheless, the disadvantages of polystyrene, such as hydrophobicity and porosity, lead to the unsatisfactory performance of EPS composite concrete.
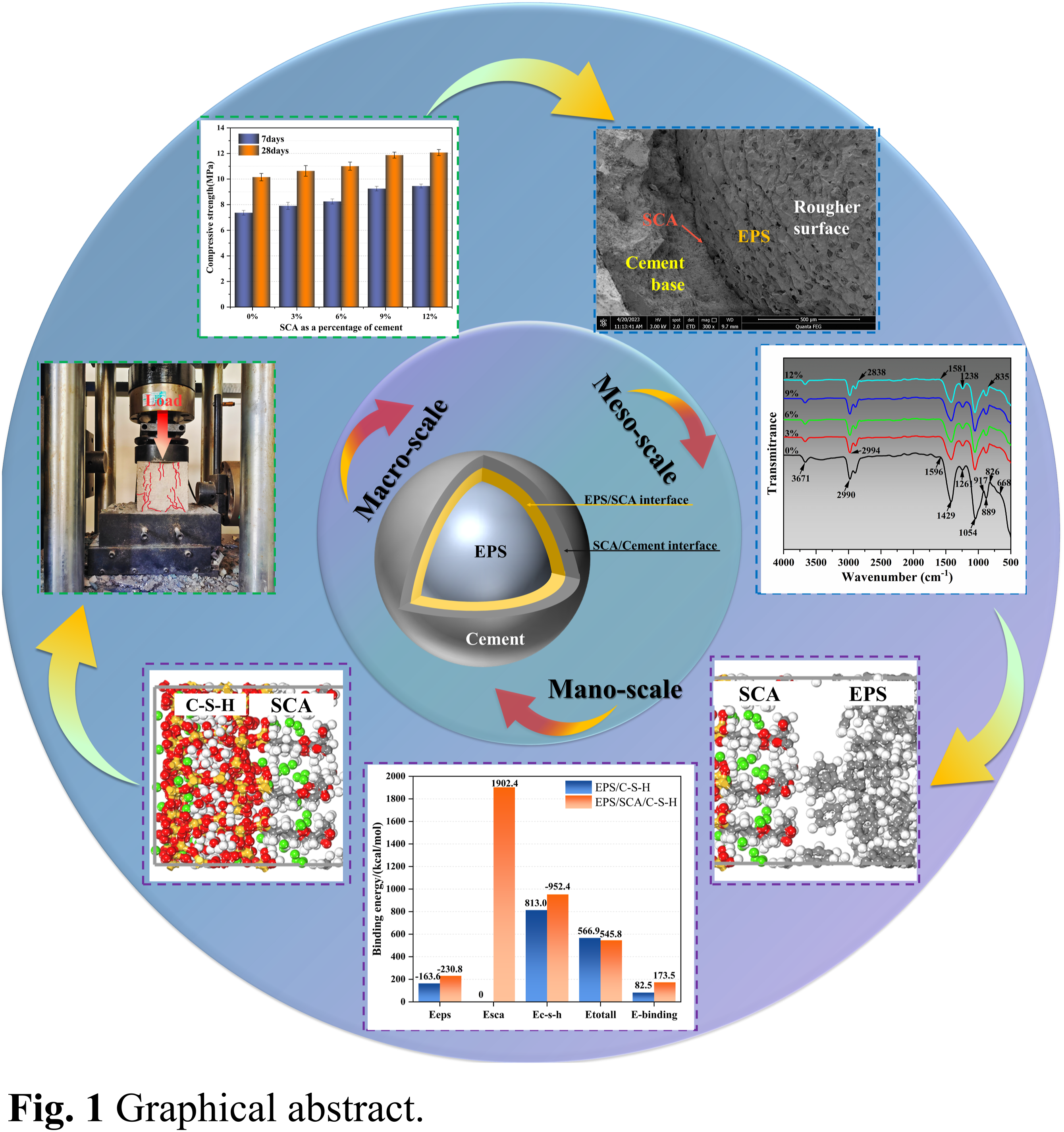
In order to further expand its prospects in engineering applications and meet the requirements of modern engineering, it is necessary to enhance the workability and mechanical performance of EPS concrete. In this study, through a combination of experiment and simulation, the mechanism and method of SCA strengthening EPS concrete are studied from a multi-scale and multi-angle perspective, in order to provide reference for the preparation of more stable and excellent performance EPS concrete. This research delves into the fortifying process of SCA on EPS composite concrete through a comprehensive exploration across various levels, utilizing macro and micro experiments alongside molecular dynamics simulation. As shown in Figure 1, firstly, SCA (KH560) is used to modify EPS beads on a macro scale (>10−3m), and the properties of the modified EPS concrete are tested by designing compressive and flexural tests. Secondly, at the microscopic scale (10−3m–10−9m), the interface structure between SCA-modified EPS material and cementitious matrix is characterized by SEM. XRD and FTIR experiments are performed to identify the types of functional groups and chemical composition in the interface following the modification with SCA. Additionally, at the nanoscale level (<10−9m), this research utilizes MD optimization simulation to examine and interpret the factors contributing to the insufficient compatibility between EPS and cement matrix. Furthermore, this study explores the impact of SCA on the interaction at the EPS-cement interface through analysis of the static interface structure, dynamic properties, and calculation of binding energy. Schematic diagram of multi-scale analysis.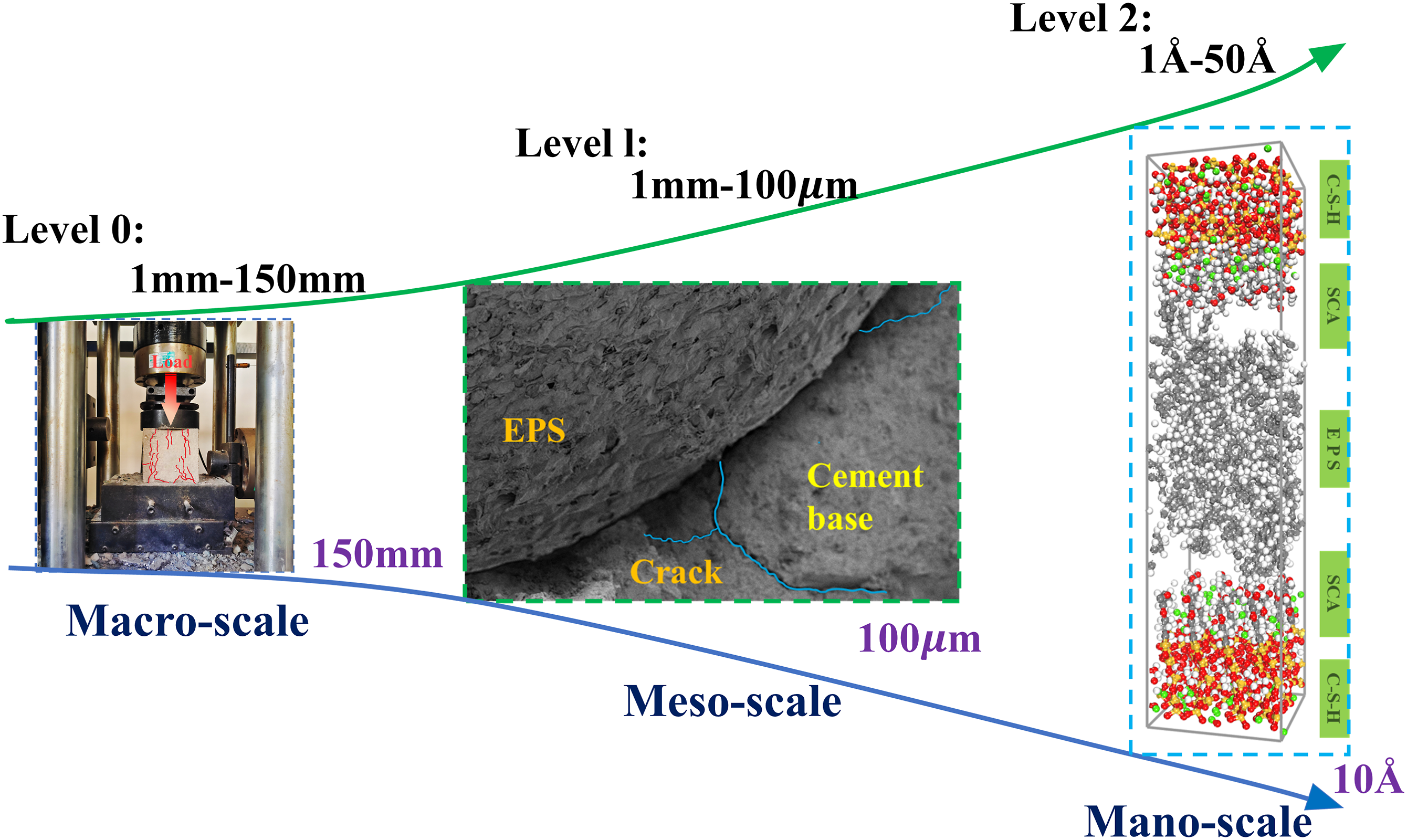
Materials and methods
Main materials
The main materials used in this experiment are fine sand, stones, cement, expanded polystyrene (EPS) beads, and SCA (KH560), (Figure 2). Main materials.
Chemical composition of cement.

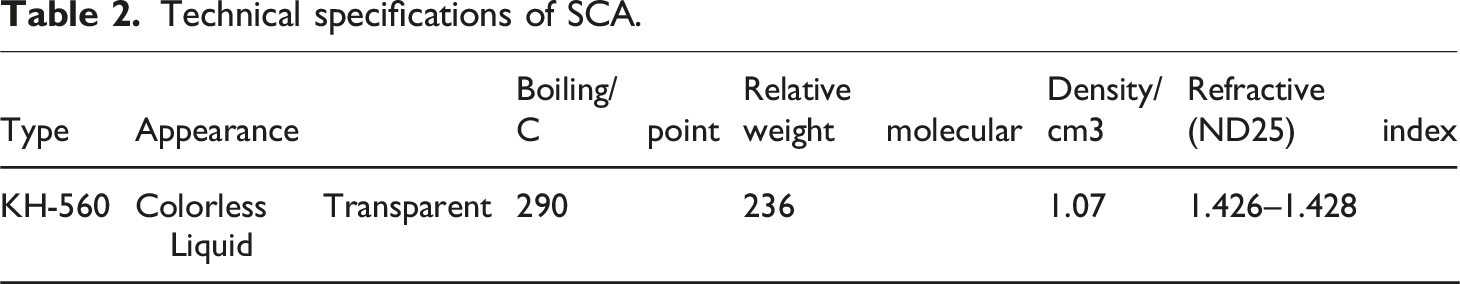
Technical specifications of SCA.
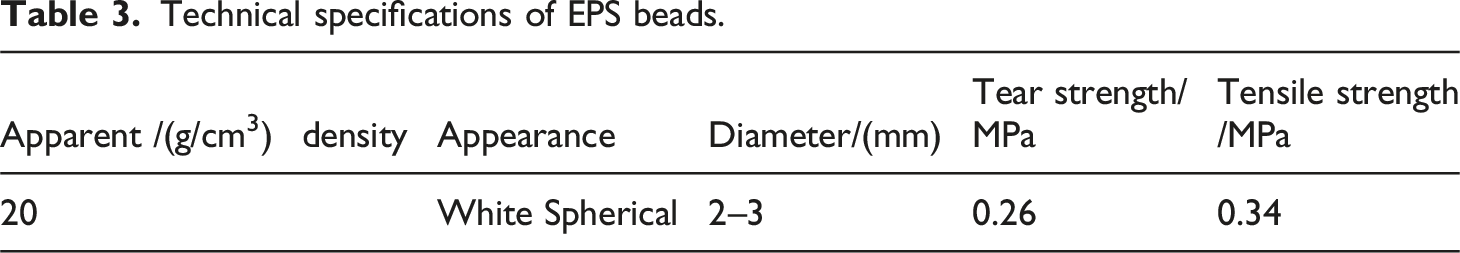
Technical specifications of EPS beads.
Treatment method and mechanism
As for the surface pretreatment method of EPS beads, this study modified EPS beads based on the method studied by Yu, Jiao and Feng, Yong.31,32 In the modification process of EPS beads, in the first step, (Figure 3), the EPS beads were placed in anhydrous ethanol to remove impurities. After soaking for 12 h, they were dried to constant weight. In the second step, a 95:5 volume ratio of deionized water and anhydrous ethanol solution was prepared, and a certain amount of SCA coupling agent was added. After stirring evenly, acetic acid was slowly added until the solution pH reached 4.0. In the third step, the coupling agent solution prepared in the previous steps is slowly poured into the container with EPS beads and fully stirred for 5 min. Finally, the mixture was left at room temperature (20°C) for 6 h, and then dried at 40°C until a constant weight was achieved for later use. EPS beads treatment process.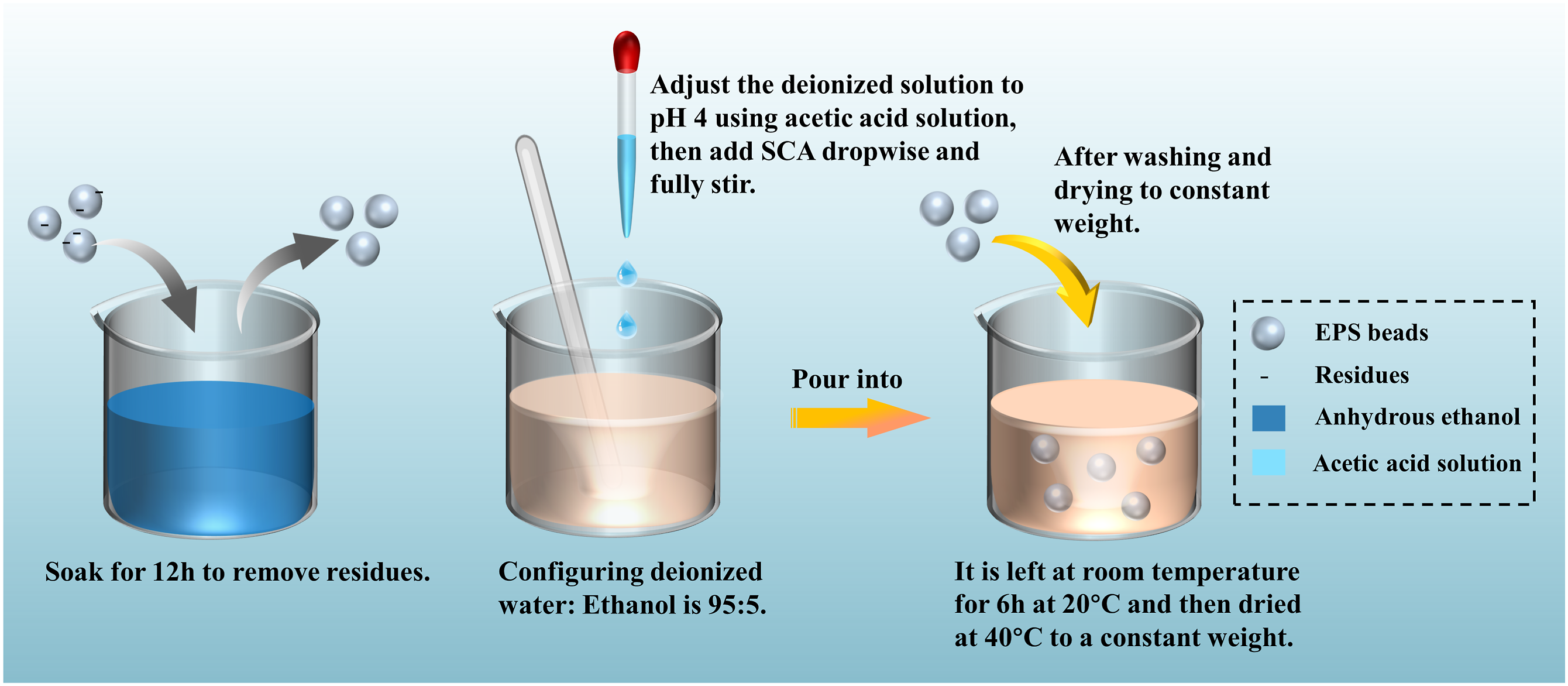
The EPS modifier used was SCA-KH560 (3-(2,3-epoxypropoxy)propyltrimethoxysilane), which is synthesized from two compounds, allyl glycidyl ether (C6H10O2) and trimethoxysilane (C3H10O3Si), (Figure 4(a)), The KH560 molecule structure contains a reactive epoxy group (C9H20O5Si), which can undergo addition reaction with compounds, thereby helping to form a more robust bond with organic polymers. Moreover, the methoxy (-OCH3) group present in SCA is a group that can undergo hydrolysis, yielding Si-OH and -OH bonds upon contact with water (Figure 4(b)). It should be emphasized that overuse of SCA may trigger self-condensation of -OH groups, leading to the creation of Si-O-Si bonds (Figure 4(c)).33–35 The (a) molecular structure, (b) hydrolysis process and (c) condensation reaction process of SCA.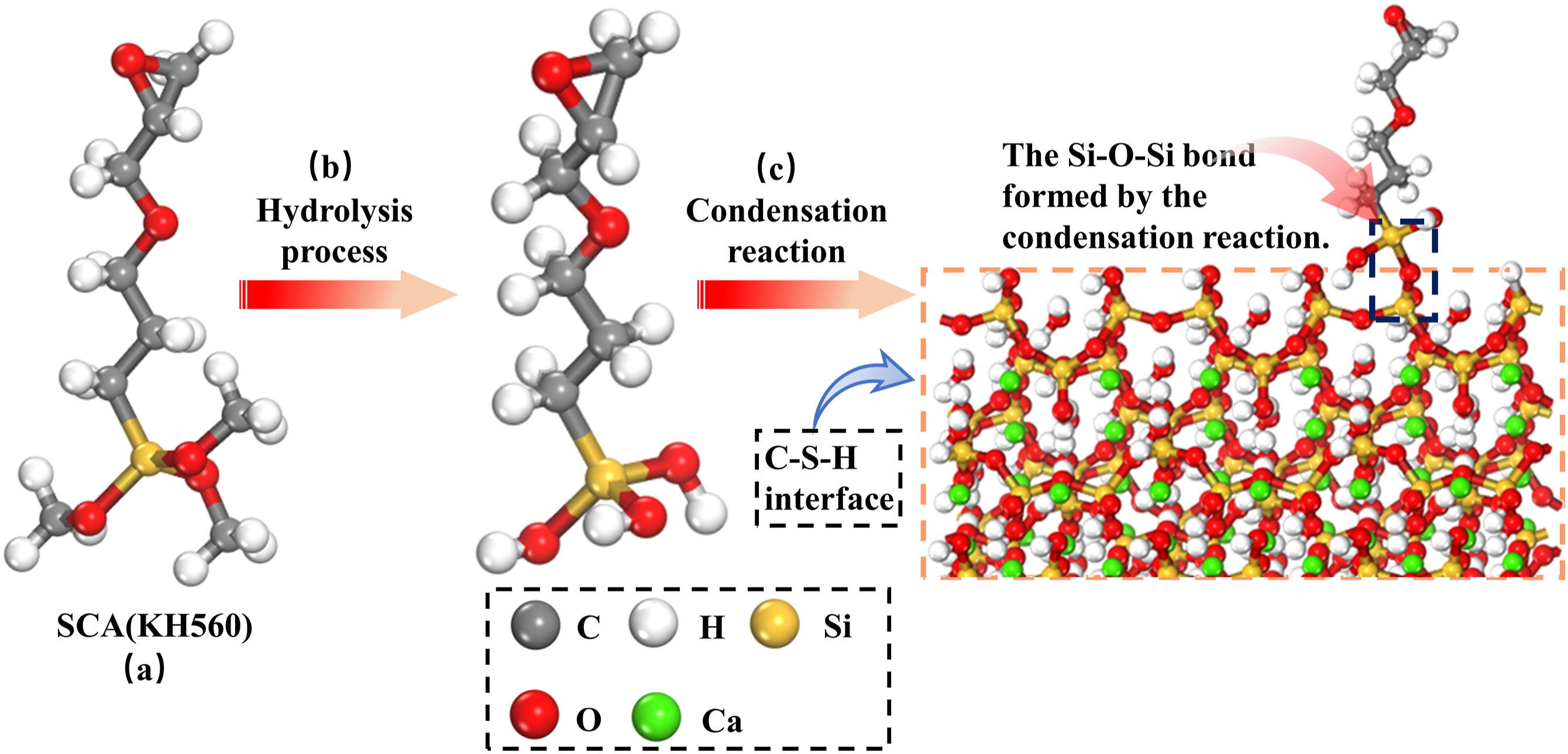
Mix proportion and sample preparation
In order to investigate the mechanism by which SCA coupling agent affects the mechanical properties of EPS concrete at the macroscopic level, this study followed the procedures outlined in the “ Standard Test Methods for Mechanical Properties of Concrete” (GB/T50081-2019) to prepare and test the mechanical properties of the samples. 36 Five different mix ratios of compressive (100 mm × 100 mm × 100 mm) and flexural (40 mm × 40 mm × 160 mm) EPS concrete specimens were prepared. In strict accordance with the “Common Concrete Mix Design Code” (JGJ55-2011) and combined with the research of Zhao X 37 and Xiong Z, 38 the mix ratio of this test was determined. 39 Among them, the water-cement ratio is 0.45, and the content of cement, fly ash, fine sand, water reducing agent, and Superplasticizer is 382 Kg/m3, 127 Kg/m3, 140 Kg/m3, 5.91 Kg/m3, and 31.62 Kg/m3 respectively. In addition, the EPS beads equal volume replacement of fine aggregate content of 8%. Xu et al.'s research shows that this content can meet the strength requirements of basic lightweight aggregate concrete.40,41 The only variable is set to be SCA, whose content is specified as 3%, 6%, 9% and 12% of the cement mass, respectively. In addition, to reduce experimental errors, this study strictly followed the specifications and prepared three identical specimens for each sample.
The preparation process of EPS composite concrete is carried out in strict accordance with the specifications. First, the fine sand, fly ash, cement, water and modified EPS beads were mixed in proportion and stirred for 30 s. Then, all the cement was added and stirred for 30 s until the EPS beads were evenly distributed in the cement. Subsequently, the entirety of the water and water-reducing compound were incorporated, and the amalgamation was stirred consistently for 90 s before being poured into a cast treated with a release compound. Subsequent to vibrating on the shaking table for 1 min, the specimen was allowed to cure at ambient temperature for 24 h, de-molded, and transferred to a chamber for curing for periods of 7 days and 28 days.
Laboratory test method
Mechanical properties
The mechanical properties test of EPS concrete samples shall be carried out with reference to ASTM C495/C495M-12 standard test method for compressive strength of concrete,
42
ASTM C348-21 Standard test method for flexural strength of Concrete
43
and China’s Standard Test Method for Physical and Mechanical Properties of Concrete (GB/T50081-2019).
36
According to Figure 5, the Y250 digital display electric bending testing machines were utilized to examine the compressive strength and flexural strength on 100 mm cube and 40 mm × 40 mm × 160 mm prism respectively. (a) Diagram of the apparatus for the compressive macro test; and (b) diagram of the apparatus for the flexural macro test.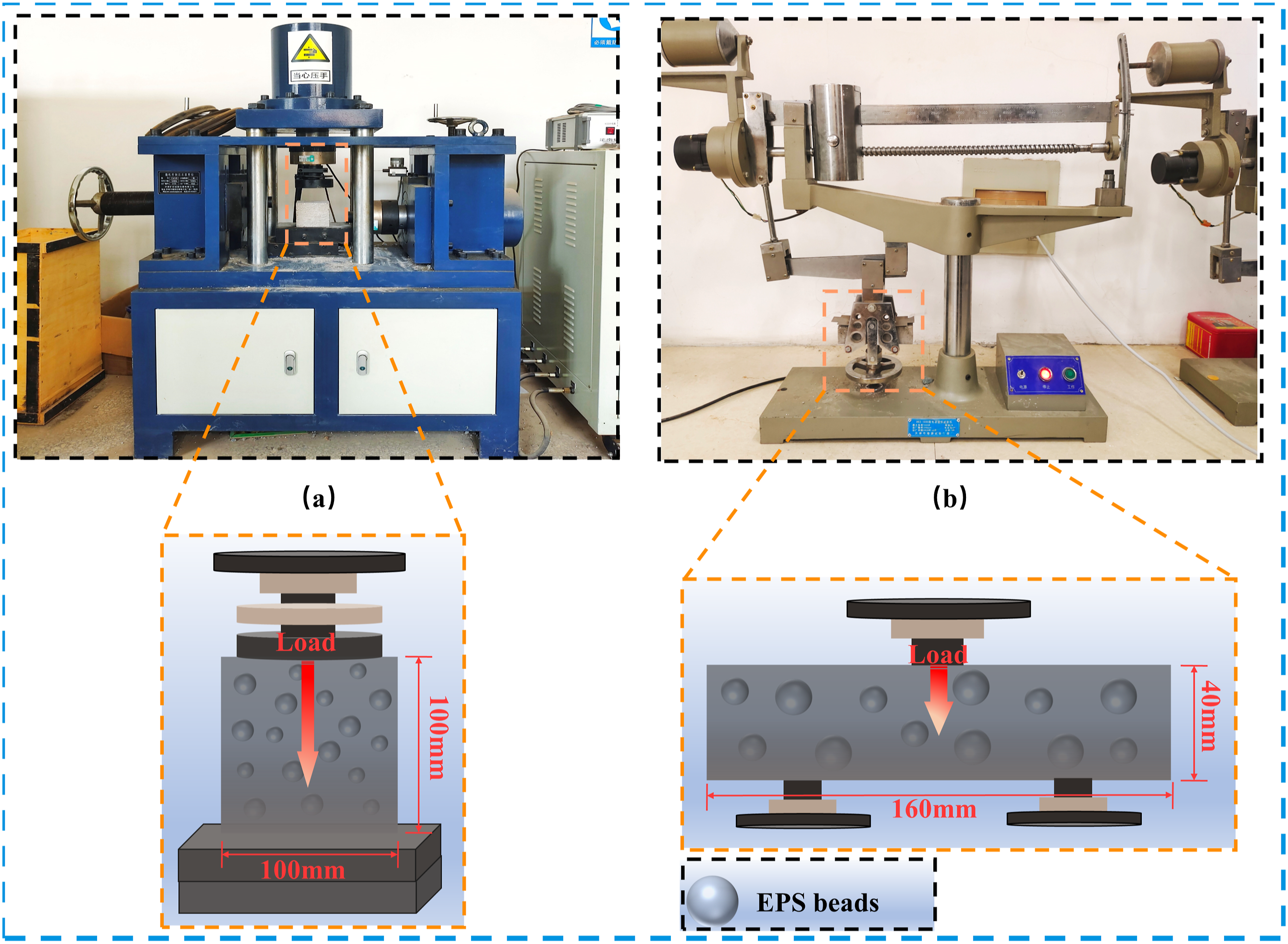
Each dosage of compressive and flexural tests includes three samples. The final test results are based on the average values of compressive and flexural strength taken from the three samples. Prior to the compression test, ensure that both the specimen’s pressure surface and the pressure plate on the equipment are vertical and secure, then apply pressure to the test block at a loading speed of 0.6 kN/s. Stop loading immediately when the specimen is damaged and experiences rapid deformation, and record the failure load at that point. The flexural test is conducted using the center loading method. Begin by adjusting the position of the instrument’s support frame and indenter, setting the rotating wheel to 10 MPa, and ensuring the swinging weight is at zero. Level the balance plate before placing the specimen into the loading slot. Start the test, observing the specimen until it breaks. Stop the test and record the load strength at that moment. Figure 6 shows the schematic diagram of some test blocks placed after cement curing and forming with different SCA content, including some test blocks used in compressive and flexural tests before and after failure. Schematic diagram before and after failure of part of the sample.
SEM
Scanning electron microscopy (SEM) was used to study and analyze the microstructure of the transition area at the boundary both pre and post treatment with SCA, in order to explore further. The test was conducted accordance with ASTM C1356-07 specification of the United States. 44 Initially, the desired samples were sliced into 10 mm × 10 mm × 10 mm cubes with a cutting instrument, and the interface was refined using 2000 grit sandpaper. Subsequently, any remaining powder residue on the surface was eliminated through the use of anhydrous ethanol via ultrasound, followed by drying at a temperature of 60 ± 2°C for a duration of 5 h. Finally, the surface of the uncompressed EPS concrete specimens containing EPS microspheres was scrutinized using SEM.
XRD
In this study, an X-ray diffraction (XRD) test was conducted to analyze the phase composition of EPS composite concrete. The test was conducted accordance with ASTM C1365-18 specification of the United States. 45 Initially, the crushed EPS composite concrete, which was not under direct stress during the mechanical performance test, was immersed in anhydrous ethanol to prevent further hydration. Following that, the solidified cement residue was delicately eliminated from the exterior of the revealed EPS composite concrete fragments using a blade, and then finely pulverized into a powder approximately 200 mesh. Afterward, the powdered specimen was moved to a vacuum drying chamber and dehydrated at 50°C for 24 h. Lastly, the dehydrated samples were analyzed with an X-ray powder diffractometer to identify their makeup.
FTIR
In order to further determine the changes of interface functional groups, Fourier transform infrared spectroscopy (FTIR) was performed on the samples in this study. The test was conducted accordance with ASTM C1365-18 specification of the United States. 46 Through an examination of the creation and vanishing of distinct functional units within the specimens, a qualitative assessment was conducted on the adjustment of the EPS beads interface. The measurement precision was set at 4 cm−1, with a wavelength span ranging from 4000 to 500 cm−1, and the scans were carried out a total of 32 times.
Model building and simulation methods
Computational models
In this study, macroscopic and microscopic experimental research and analysis were conducted on EPS concrete before and after modification with SCA. To further study and understand the mechanism of SCA, this paper used Materials Studio (MS) software to perform molecular dynamics simulation analysis on two EPS cement interface models with and without SCA at the nanoscale. 47 It has been shown that the size of the interface model may affect the simulation results.48,49 This study aims to study the influence mechanism of SCA on the EPS/C-S-H interface, so the influence of model size on the results can be eliminated as much as possible without changing the model size. The MS software can establish an effective model and algorithm from the atomic level. It can be combined with macroscopic and microscopic experiments to further study new composite materials. 50
The excellent performance of concrete is primarily influenced by the hydration products of cement, which are mainly composed of calcium silicate hydrate gel (C-S-H), an incomplete crystal widely used in the study of C-S-H structure and dynamics.
51
The Tobermorite molecular structure model has been used to characterize the evolution of C-S-H properties and is therefore selected as the basic model for this study.52–54 A gel pore was constructed as the cement-based model by cutting, extending, and combining along the crystal plane (0 0 1), with crystal plane parameters of U = 22.00 Å and V = 22.00 Å. EPS beads with a density of 1.0 g/cm3 were selected based on experimental raw materials, with an amorphous monomer unit composed of 8 EPS polymer chains, each containing 16 EPS monomers.41,55 Utilizing the bonding process of SCA and cementitious substances as previously discussed, SCA was linked to non-bridged oxygen atoms on the inner side of C-S-H matrix nanopore by Si-O-Si chemical bond.56,57 The EPS/C-S-H model (a = 22.00 Å, b = 22.00 Å, c = 72.02 Å) and EPS/SCA/C-S-H model (a = 22.00 Å, b = 22.00 Å, c = 92.69 Å) were constructed using the Build Layer module. In addition, EPS and SCA were not fixed to a specific surface in the model, as they can be optimized to a more suitable energy position. The process of constructing the EPS/C-S-H model is show (Figure 7). Modeling of the C-S-H/EPS interface.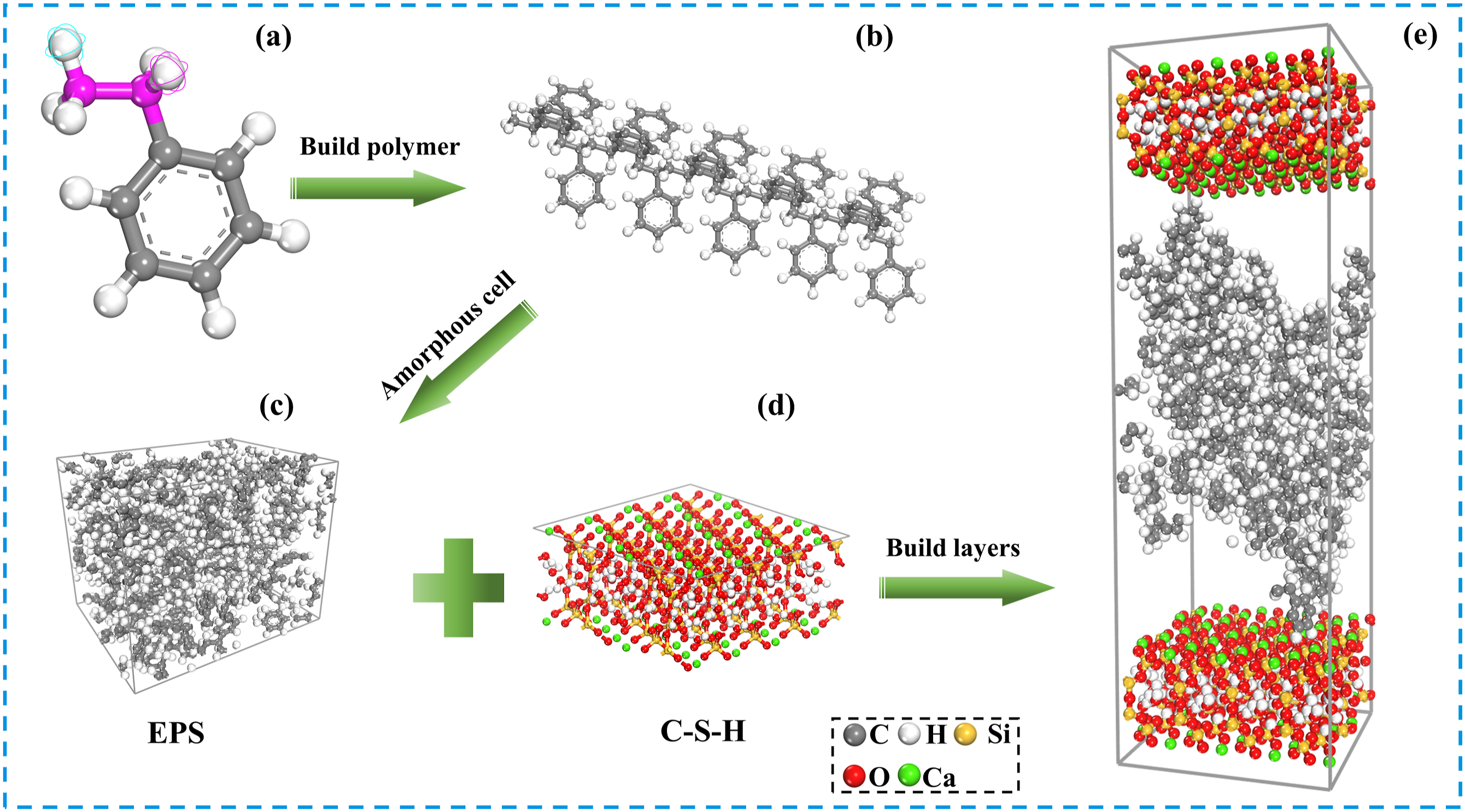
Force field and simulation details
This study employed the Materials Studio (MS) 2020 software to perform simulation analysis on all models. In molecular dynamics simulation calculations, the choice of force fields is critical as it precisely affects the accuracy of the computational results. After consulting relevant research, this study utilized the Compass force field.
58
Contained within this force field is the energy analysis of the majority of silicate substances and the potential energy of the connections between various atoms or molecules within.59,60 Moreover, the force field may be represented by a reasonable model when addressing both organic and inorganic systems.47,61 It has been proven that COMPASS has good agreement with the test data and can be used to simulate the hydration products of cement.
62
Therefore, this study employed the Compass force field for subsequent simulation analysis. To eliminate the illogical framework during model development, this research employed the geometric optimization feature in the Forcite component of MS software. The COMPASS force field was chosen, and the algorithm was set to Smart with precision set to Fine for the optimization of the interface models. Subsequently, a balanced operation was carried out using the Forcite module under the NPT system. The COMPASS force field was again selected, with accuracy set to Fine, and time steps of 300 ps and 1 fs were utilized to transition the model system to a more stable state at 300 K. Lastly, to ensure the reliability of the simulation data, the final model underwent a relaxation process of 1 ns under the NVT ensemble, (Figure 8). Throughout the simulation, the system’s temperature and pressure were monitored and controlled by the Nose-Hoover-Langevin thermostat and Andersen voltage regulator, respectively.
60
The final interface model size of EPS/C-S-H is a = 22.00 Å, b = 22.00 Å, c = 72.02 Å, and the total number of atoms is 3472. The final interface model size of EPS/SCA/C-S-H is a = 22.00 Å, b = 22.00 Å, c = 92.69 Å, and the total number of atoms is 4238. (a) EPS/C-S-H interface model (b) EPS/SCA/C-S-H interface model.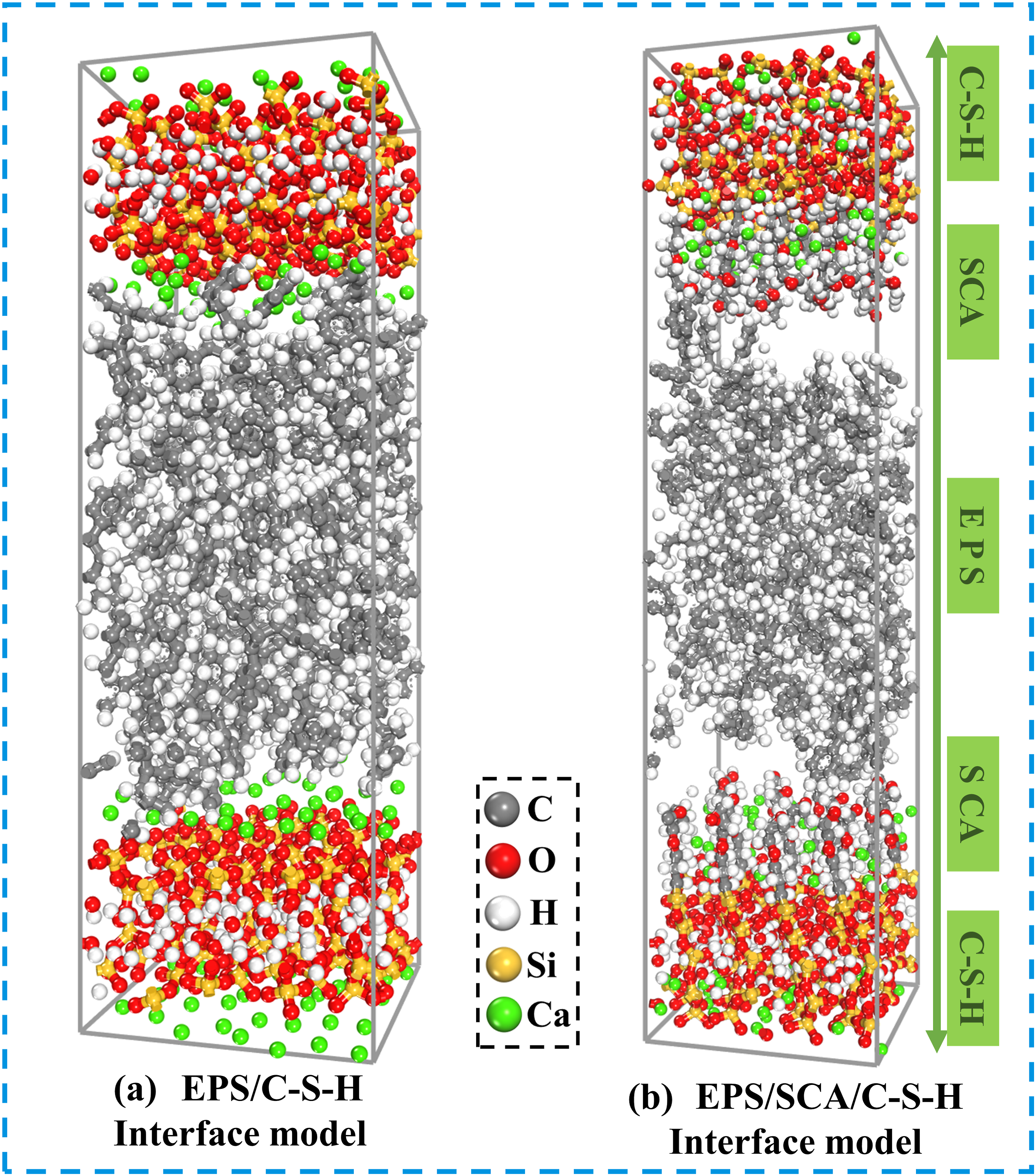
Results and discussion
Mechanical properties
This research utilized traditional compression and flexural mechanical tests to examine the mechanical characteristics of samples with varying mix ratios. Five different mix ratio samples were tested using traditional macroscopic mechanical tests to confirm the overall representativeness of the experimental data. The effects of SCA on the compressive strength of EPS composite concrete at 7 days and 28 days for different mix proportions were investigated (Figure 9). The experimental results indicated that the compressive strength of the specimens increased continuously with increasing SCA (KH560) content. Compared with the blank control group (0% SCA), EPS blocks containing 3%, 6%, 9%, and 12% SCA showed an increase in compressive strength of 7%, 12%, 24%, and 27%, respectively, after 7 days. The flexural strength increased by 8%, 23%, 39%, and 43%, respectively. In addition, EPS blocks containing 3%, 6%, 9%, and 12% SCA showed an increase in compressive strength of 5%, 8%, 17%, and 19%, respectively, after 28 days. The flexural strength increased by 6%, 10%, 16%, and 20%, respectively. (a) Compression test process and results; (b) Flexural test process and results.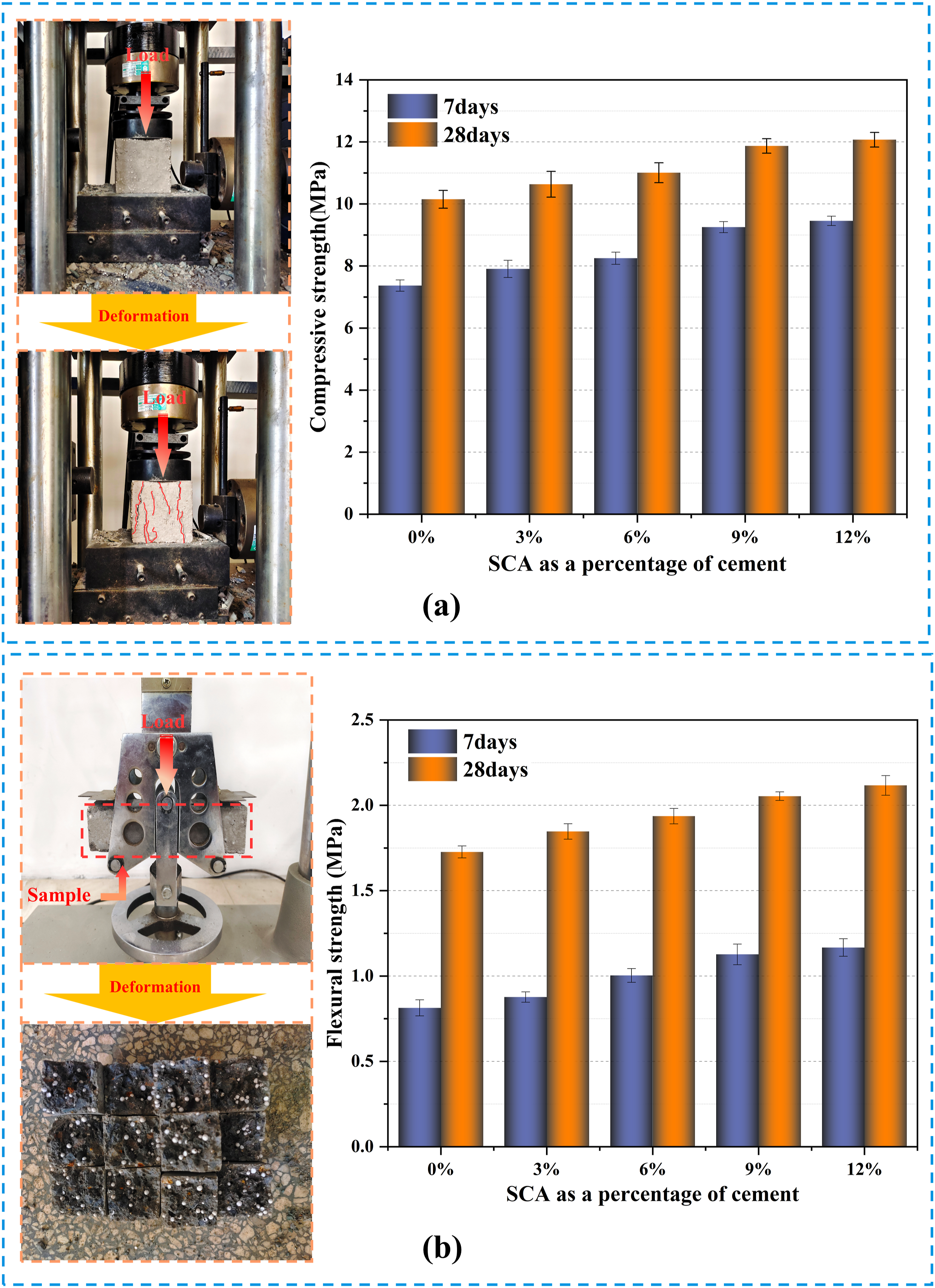
It is worth noting that the compressive strength of SCA-modified EPS composite concrete exceeds the flexural strength (Figure 9). Furthermore, the research revealed that incorporating SCA can enhance the adhesion between EPS and the cement base interface, thereby retarding the propagation of concrete fissures. The highest compressive and flexural strength of EPS composite concrete containing 12% SCA was observed on the 7th and 28th days respectively, however, the rate of strength increase was lower compared to other EPS composite concrete mixes with SCA. Simply put, as the SCA content rises, the reinforcement effect on EPS concrete will diminish gradually. This preliminarily shows that when the SCA content is 9%, the reinforcement effect of EPS composite concrete may be the most effective. However, the difference in results is not very clear, which will be further demonstrated by ANOVA in the next chapter.
Analysis of variance
Analysis of total variance for each parameter.
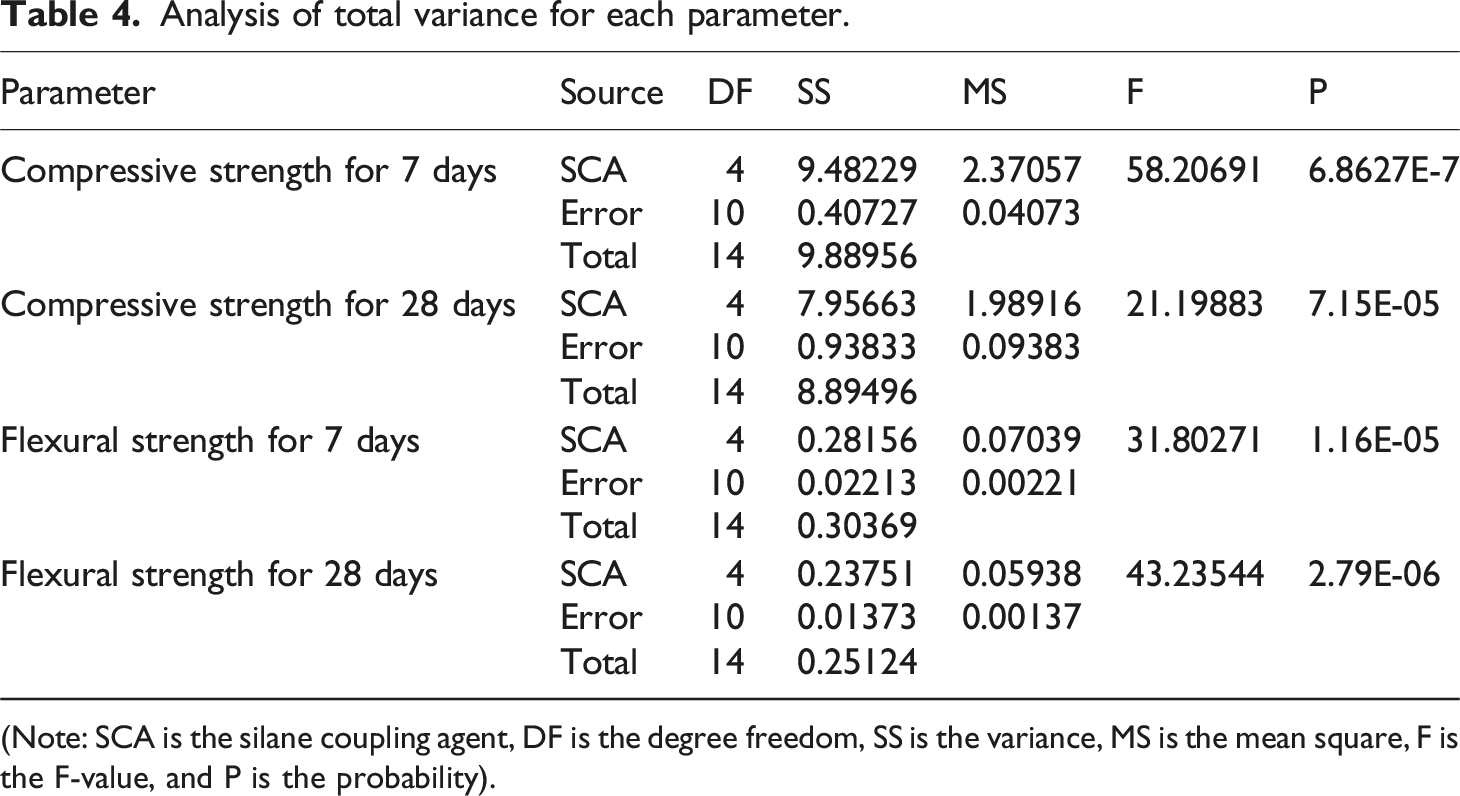
(Note: SCA is the silane coupling agent, DF is the degree freedom, SS is the variance, MS is the mean square, F is the F-value, and P is the probability).
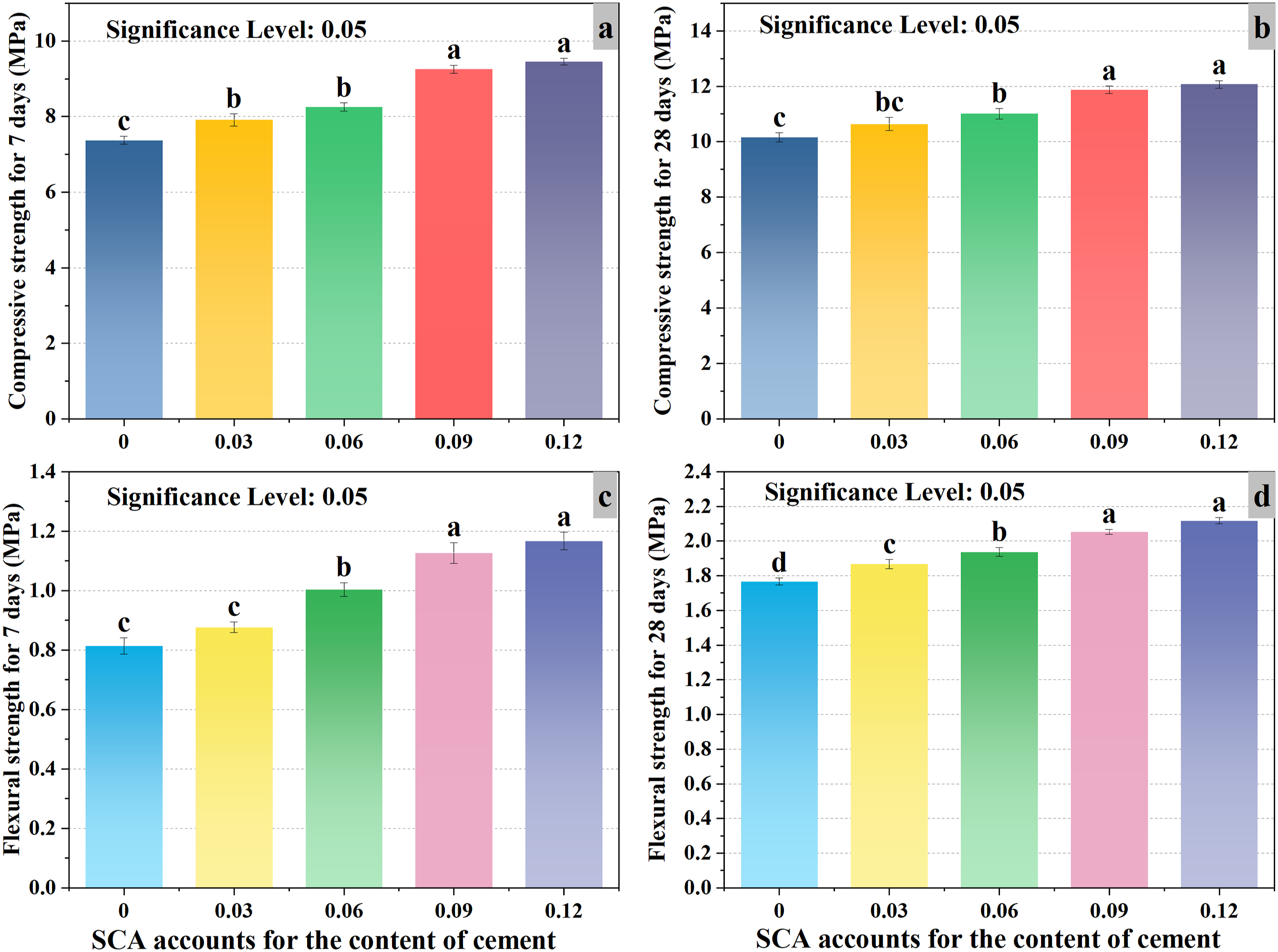
Histogram of data variance analysis results.
In addition, it can be seen from Figure 8 that when the SCA content is 9% and 12%, the compressive strength and flexural strength of the two groups of data at 7 days and 28 days are labeled with the same letter (a). This shows that the content of SCA at 9% and 12% does not have a significant difference. However, compared with the SCA content of 0% in the two sets of data, the difference in letters indicates significance. The analysis shows that with SCA at 12%, the effect on improving EPS concrete is similar to when SCA is at 9%. In other words, with an SCA content of 12%, although the performance of EPS concrete can still be enhanced, the improvement trend will gradually decrease with the increase of SCA content. This is similar to the research results of previous scholars, and further confirms that the reinforcement effect of EPS concrete is the best when SCA is 9%.64–66
SEM
To investigate the microstructural changes at the EPS-cement interface caused by SCA, SEM images of the interface before and after SCA modification were compared in this study. Unmodified and SCA-modified EPS composite concrete samples, prepared as described, were scanned and their SEM images were magnified by 300 times. In Figure 11(a) unmodified EPS beads and cement particles at the interface were visibly separated, indicating insufficient and uneven cement hydration. The extent of cement hydration directly affects the mechanical properties of EPS-cement composite materials, and the absence of hydration products leads to many weak interface transition zones in EPS composite concrete, which can easily connect and cause a decrease in the mechanical properties of the concrete when subjected to stress. However, as shown in Figure 11(b), the interface between EPS and cement after SCA modification is uniform, and the contact area between EPS and cement matrix is large without obvious gap. In addition, the surface of the EPS treated with SCA is rougher, which will help it bond with the cement base. The findings from the experiment indicate that the use of SCA can effectively enhance the deficient interfacial bond region of EPS-cement, restrain the progression of cracks, strengthen its bonding capacity, and enhance the mechanical characteristics of EPS composite concrete. SEM photographs.
XRD
To further analyze and verify the presence of SCA at the EPS-cement interface and its changes before and after modification, this study conducted X-ray diffraction (XRD) analysis on blank control groups (0% SCA) and EPS-cement test blocks containing 3%, 6%, 9%, and 12% SCA, according to the experimental method described in the previous section. The experimental results are shown in Figure 12. The findings revealed that the absorption peaks in XRD spectra exhibited slight fluctuations with a consistent shape and position, with no presence of extra diffraction peaks. This suggests that while SCA does not alter the composition and nature of cement hydration products, it does impact their quantity and crystallization pace. With an increase in SCA content, the intensity of the diffraction peaks relating to the cement hydration product [Ca(OH)2] decreased gradually, whereas the characteristic peaks of the homogenized minerals in hydration (Alite and Belite) increased steadily.
67
This further demonstrates that SCA prolongs the cement hydration reaction. This is due to the initial hydrolysis of SCA, which produces silanols that subsequently engage in condensation reactions with hydroxyl groups present in cement to generate alkylsiloxanes on the surface of the cementitious material, thereby delaying the cement hydration reaction.
68
XRD patterns of specimens with varying SCA content.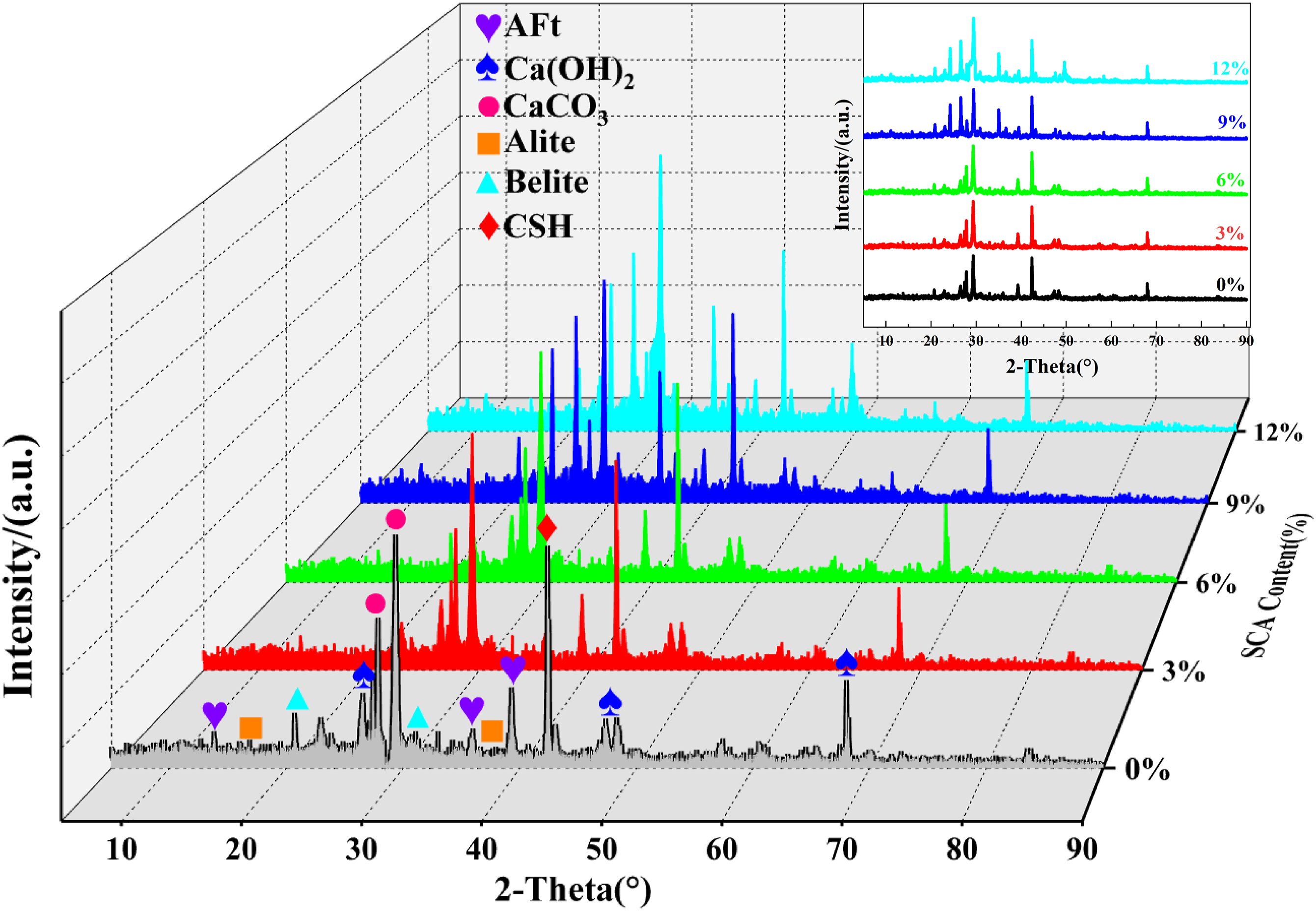
FTIR
As shown in Figure 13, the infrared spectroscopy test results of EPS composite concrete samples modified with different levels of SCA are presented. The results indicate that SCA has an influence on the type and quantity of functional groups at the EPS beads-cement interface. The infrared vibration bands of the modified EPS beads are weaker than those of the unmodified EPS beads. The absorption peak at 1054 cm−1 in the spectrum is related to the Si-O-Si stretching of the siloxane oligomers, indicating that SCA molecules may undergo hydrolysis and condensation reactions. Importantly, SCA molecules hydrolyze and form intermediates with one, two, or three hydroxyl groups, which then undergo condensation reactions to form siloxane oligomers containing Si-O-Si chemical bonds. This ultimately improves the bonding between the organic (EPS beads) and inorganic (cement) materials at the direct interface, thereby improving the mechanical properties of EPS composite concrete. Additionally, with the rise in SCA content, the presence of cement silicates in the cement slurry diminishes, while the vibration band strength of the hydrated product of cementitious gel C-S-H (917 cm−1) in the region of cement hydration fingerprint (1200 ∼ 400 cm−1) steadily grows.69,70 Infrared spectra of specimens.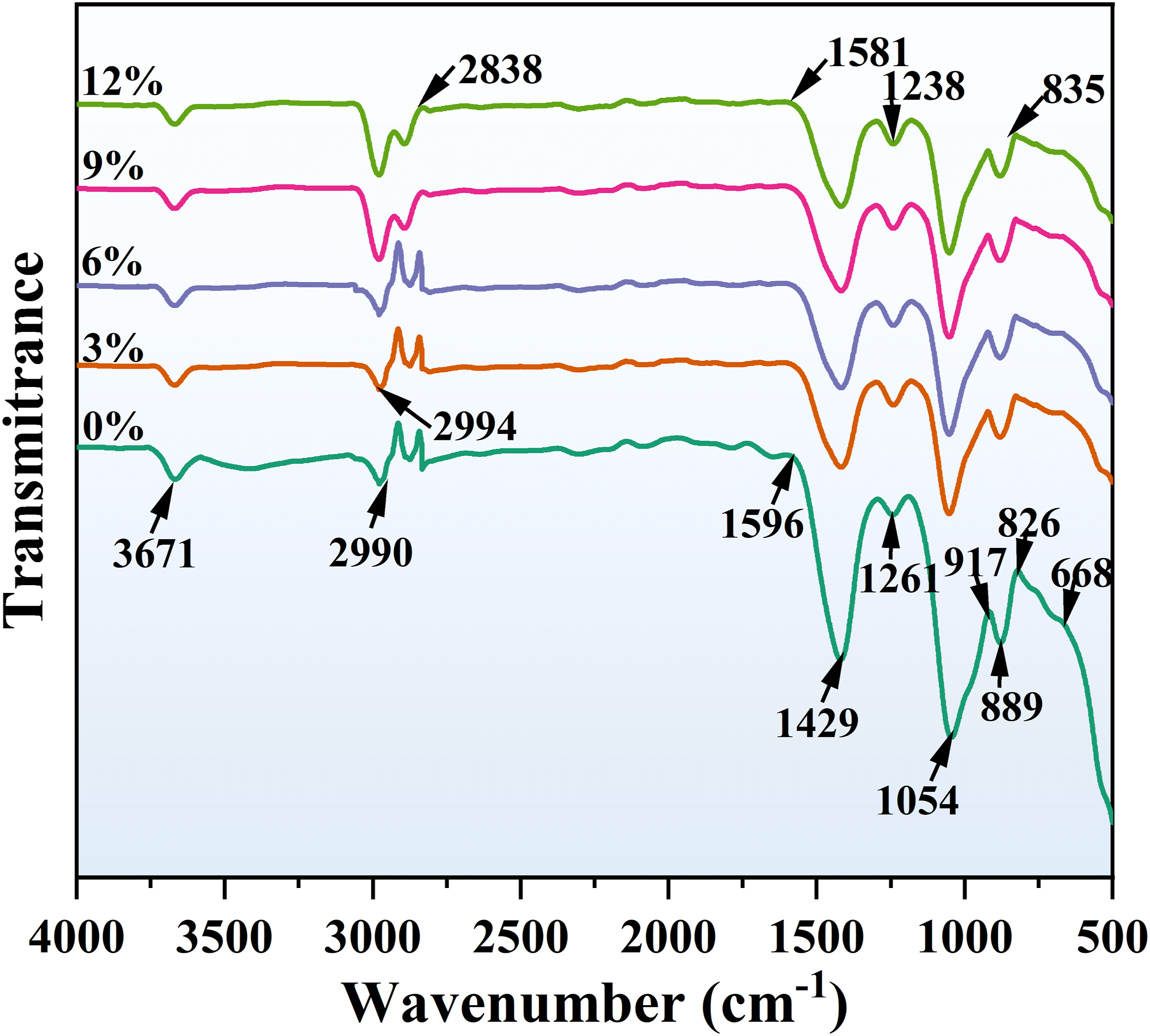
Molecular dynamics simulation
Interface static structure
The concentration distribution curve can reflect the specific density of atoms in a specific direction at the nanoscale.
71
Therefore, we calculated the concentration distribution of each atom to further understand and analyze the local structural characteristics between SCA, EPS, and cement interfaces. The expression for the relative concentration equation is as follows (1).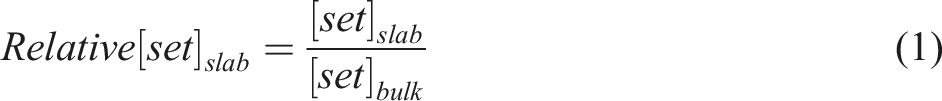
The model established in this study was oriented in the Relative concentration distribution along the Z-direction of the system for (a) C-S-H/EPS and (b) C-SH/SCA/EPS. (Si1 and hcw represent silicic and hydrogen atoms in water molecules, respectively; Ceps and Heps represent carbon and hydrogen atoms in EPS molecules, respectively; and Csca, Osca, and Hsca represent carbon, oxygen, and hydrogen atoms in SCA molecules, respectively).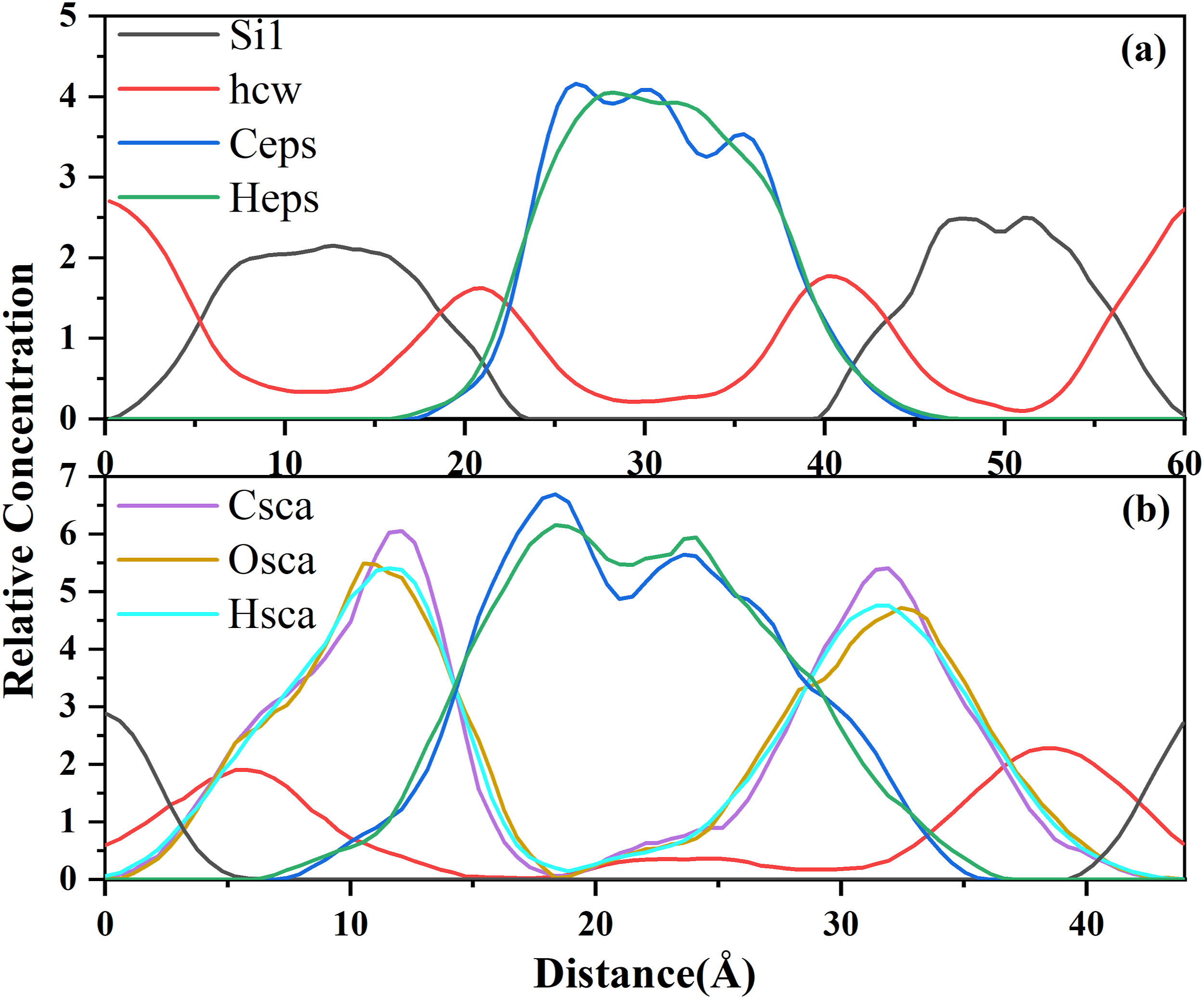
Figure 14(b) reveals that the concentration profiles of Csca, Hsca, and Osca overlap with the hcw water molecules in hydrated calcium silicate within the 10–20 Å and 25–30 Å areas, indicating the hydrophilic nature of the SCA polymer. This characteristic is significant in enhancing the bonding capability between EPS-C-S-H and improving the bond strength between EPS and cement slurry. Furthermore, within the same range, an irregular distribution of atomic density was observed in the SCA system. Moreover, the atomic concentrations of Ceps and Heps have peak values on both sides of the nanopores and are also disordered. This suggests the possible interactions between EPS and SCA atoms at the interface.
Furthermore, the thickness between the addition of EPS to C-S-H after the addition of SCA was determined by reading the range of phase densities over 90% in the Z direction (Heps and Si) from the concentration distribution curve.72,73 From the Figure 14, it can be seen that at the relative concentration of about 4, before SCA is added, Heps fluctuates for the first time at the position 25 Å −35 Å in the thickness direction; that is, the thickness layer is about 10 Å. After the addition of SCA, Heps first fluctuated in the 15 Å −23 Å position, that is, the thickness layer is about 8 Å. Similarly, at a relative concentration of about 2, Si first fluctuated and accumulated at 8 Å −17 Å with a thickness of about 9 Å before SCA was added. After the addition of SCA, it is highly aggregated for the first time at 0 Å −3 Å, with a thickness of about 3 Å. Thus, it can be seen that the addition of SCA makes the EPS and C-S-H closer and makes the internal structure of concrete more stable.
The radial distribution function (RDF) describes the probability of finding a particle at a distance r from another particle.
74
In order to investigate the local structure between EPS/C-S-H interfaces, we calculated the RDF between atoms in the EPS, SCA, and C-S-H system using the formula (2).
As shown in Figure 15, the bond length corresponds to the first peak-valley position in the RDF curve. The peaks caused by the intramolecular interactions were omitted during the calculation. From Figure 15(a) and (b), we found that the RDF curve for Heps-Ca1 has a significant peak and is higher than the peaks corresponding to Heps-Ocw and Heps-Hcw. Additionally, the RDF curve peaks in EPS/C-S-H (Figure 15(a)) are slightly higher than those in EPS/SCA/C-S-H (Figure 15(b)). The peaks of the Heps-Ocw and Heps-Hcw curves in EPS/SCA/C-S-H (Figure 15(b)) are lower than those in EPS/C-S-H (Figure 15(a)), indicating that SCA enhances the connection of hydrogen bonds. RDF curves for (a) Heps-Ca1, Heps-Ocw, and Heps-hcw in the EPS/C-S-H system; (b) Heps-Ca1, Heps-Ocw, and Heps-hcw in the EPS/SCA/C-S-H system; (c) Hsca-Ca1, Osca-Ca1, Osca-Ocw, and Hsca-Ocw in the SCA/C-S-H system; (d) Hsca-Heps in the SCA/EPS system; and (e) local C-S-H-EPS in the EPS/C-S-H and EPS/SCA/C-S-H system.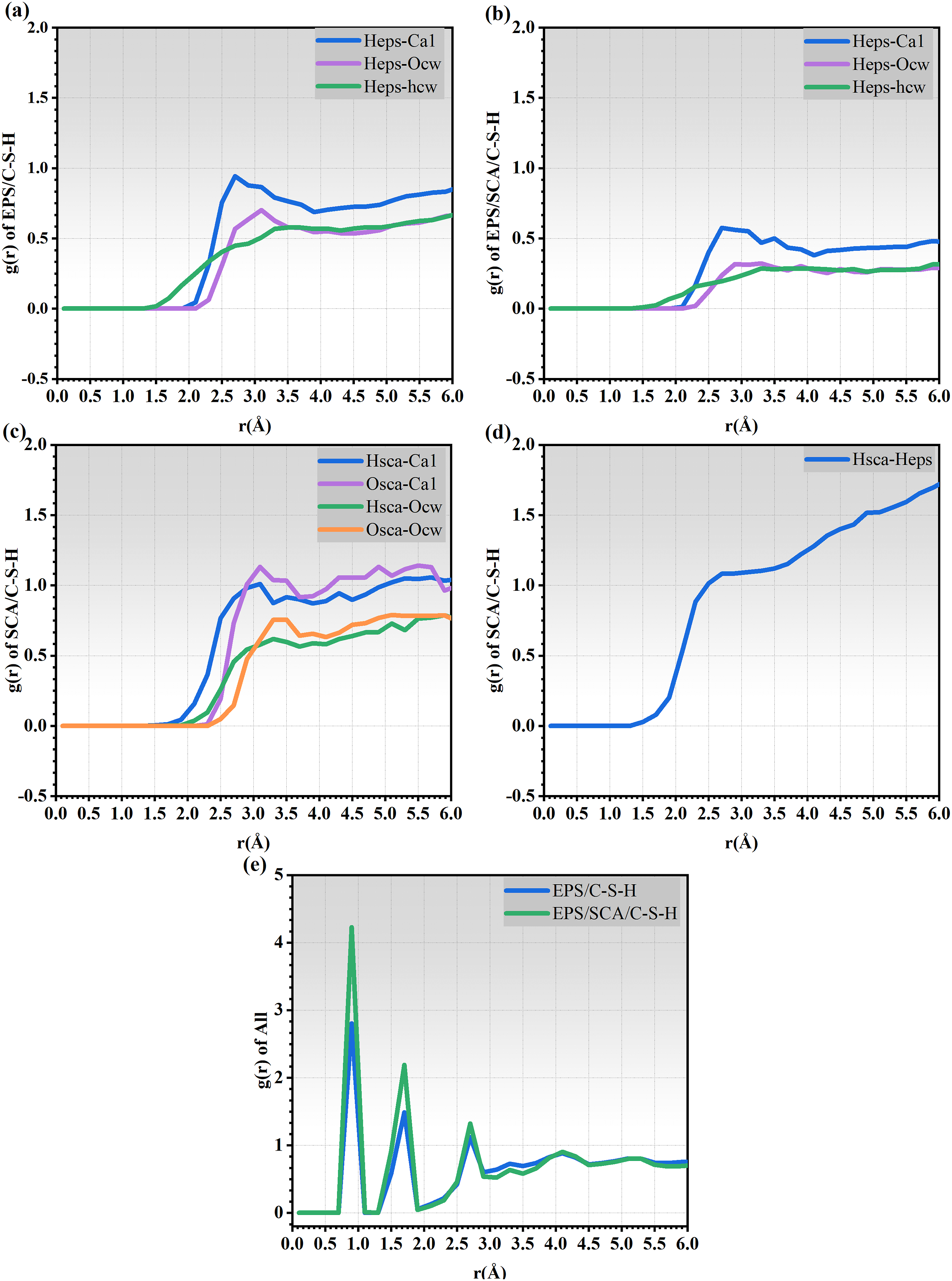
In Figure 15(c), there is a peak at 3.25 Å in the Osca-Ca1 curve, which is higher than the other curves in In Figure 15(c). This is due to the interaction between hydrated calcium silicate and polymers such as EPS and SCA, and is one of its characteristics. In Figure 15(d), the peak value of the Hsca-Heps curve is higher than the Heps-Ocw and Heps-Hcw curves in Figure 15(b), indicating that the hydrogen bond effect of the former is stronger than the latter. This may be because the polystyrene chain in the EPS polymer contains many benzene rings, which provide more hydrogen bond acceptors for hydrogen atoms, further increasing the interaction effect with H and O atoms in SCA. More prominently, in Figure 15(e), the RDF calculation of C-S-H-EPS in the EPS/SCA/C-S-H system is higher than that in the EPS/C-S-H system. This confirms that SCA enhances the interfacial bonding ability of C-S-H/EPS.
Interface dynamic characteristics
The root mean square displacement (MSD) is the average of the square displacement of particles, which can be used to evaluate the motion characteristics of atoms.
75
It is a useful tool for analyzing the dynamic properties of the system interface before and after SCA modification. In determining the MSD, we analyze the disparity between the atom’s current position and its starting position over a period of time, illustrating the atom’s motion over time. This provides a more intuitive way to analyze the dynamic behavior of the system interface. Formula (3) below shows the calculation formula for MSD.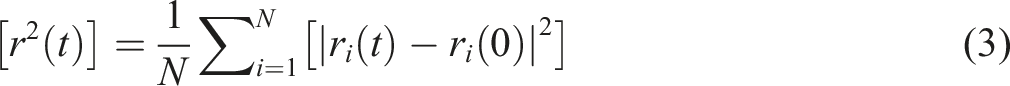
As shown in Figure 16, the MSD is a time function. It can be observed that Ca1, Och, Ceps, and Heps in the EPS/SCA/C-S-H system are all higher than those in the EPS/C-S-H system. Additionally, it is worth noting that the MSD curve for H atoms is generally higher than that of other atoms in both the EPS/C-S-H and EPS/SCA/C-S-H systems, while the MSD curve for Ca1 atoms is generally lower than that of other atoms. This indicates that the hydrated calcium silicate on the interface is stable and has a stable framework structure that is not easily affected by other atoms. The high MSD curve for H atoms indicates that they are highly unstable. The results show that SCA increases the hydrogen bond interaction at the EPS/C-S-H system interface, further increasing its stability. Additionally, it can be observed in Figure 16(c) that the MSD curve of the SCA atoms is similar to that of the C-S-H atoms, but higher than that of the EPS atoms. This suggests that the interaction between SCA and C-S-H is easier, and the SCA atoms are more free in the EPS/SCA/C-S-H system. The easier interaction between SCA and C-S-H and the greater freedom of SCA atoms further enhance the bonding strength of the C-S-H/EPS interface. Major atomic mean square displacement.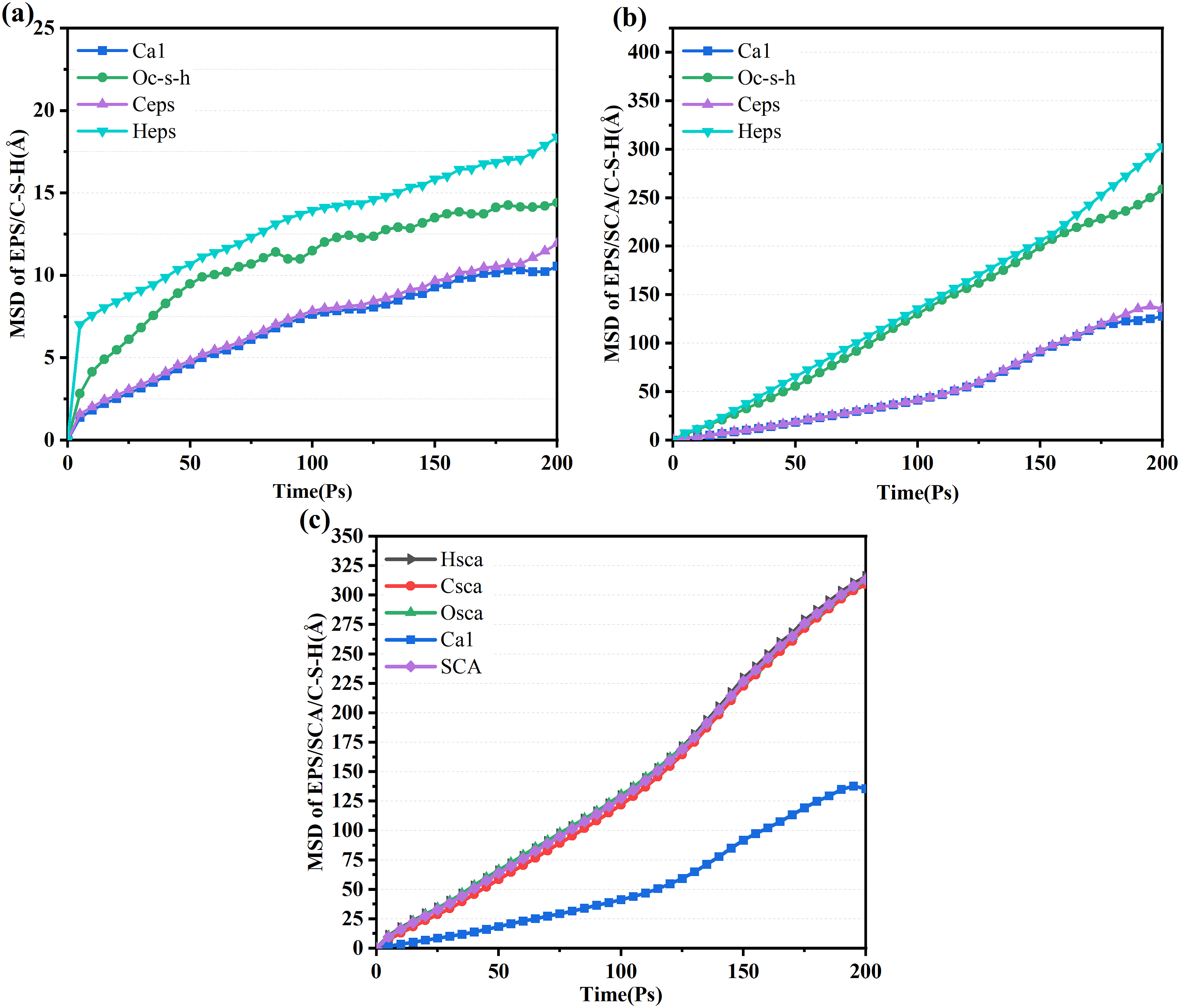
The bonding of hydrogen to calcium is seen as a crucial link between the water-saturated calcium silicate gel (C-S-H) and various polymers at the interface.
76
In order to delve deeper into the interactions of calcium ions and hydrogen within the EPS/SCA/C-S-H system, we utilize time correlation functions (TCF) to examine the ongoing stability of chemical bonds and perform a detailed assessment of the calcium ion connections and hydrogen bonds within the system.
77
The calculation formula for the time correlation function is shown in formula (4).
At each time step, the TCF value C(t) is equal to 1 if the ionic bond is connected and 0 if it is not. The decomposition and recombination of chemical bonds cause the TCF value to fluctuate between 1 and 0 over time.
In Figure 17(a), it can be seen that the TCF values of the ionic bonds are fluctuating above 0.9. This indicates that the ionic bonds produced by Ca have stable connection strength. From Figure 17(b), compared with Hsca-Heps, the TCF value of Heps-hcw is lower and more fluctuating. This suggests that the hydrogen bonds in Heps-hcw are easily lost, which also indicates that the interaction between water molecules and EPS in the cement matrix is small. Finally, in Figure 17(c), it can be found that compared with Osca-hcw, the TCF values of Hsca-Ocw and Heps-Ocw are relatively stable and flat. Therefore, it can be concluded that the addition of SCA enhances the EPS/C-S-H interface mainly through Hsca-Ca1 and Heps-Ca1 ionic bonds, followed by hydrogen bonds in Hsca-Heps, Hsca-Ocw, and Heps-Ocw. The time correlation functions value of (a) Hsca-Ca1, Heps-Ca1, and Osca-Ca1; (b) Heps-hcw and Hsca-Heps; and (c) Hsca-Ocw, Osca-hcw and Heps-Ocw in the EPS–SCA–C-S-H system.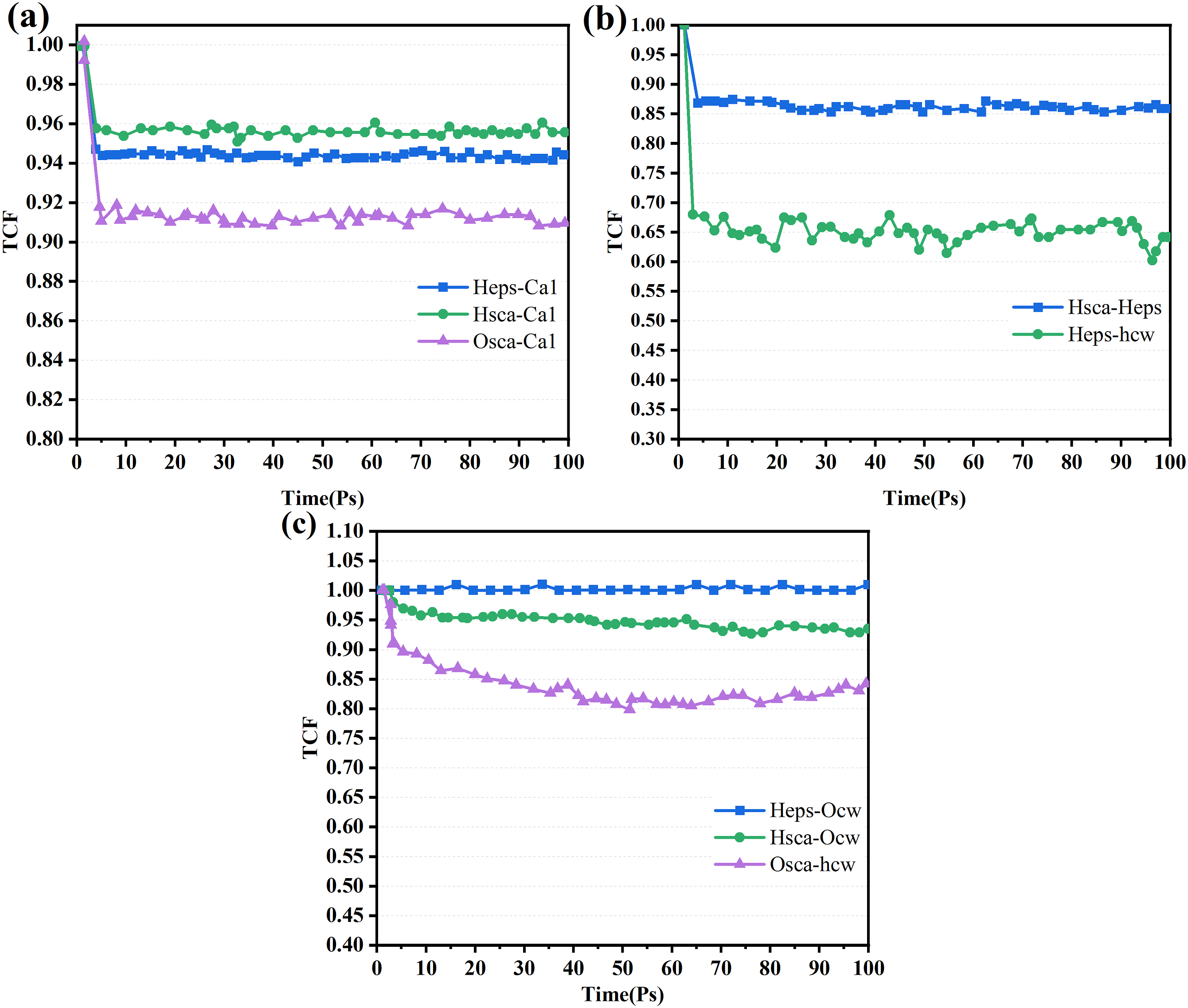
Analysis of interface adhesion mechanism
In order to further investigate whether the system containing SCA has stronger interface adhesion than the system without SCA, the binding energy of two interface systems (EPS/C-S-H and EPS/SCA/C-S-H) was calculated. The binding energy serves as a clear indicator of the attraction or repulsion between two phases, portraying the energy contrast between the overall system and the individual parts.
78
A positive outcome suggests a mutual attraction between the phases, whereas a negative outcome indicates mutual repulsion. The greater the absolute value of the binding energy, the more robust the interaction force among the systems, leading to a more stabilized system. We used the EPS/C-S-H system as an example to calculate the binding energy (5).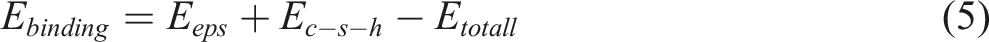
As shown in Figure 18, it can be observed that the binding energy (E-binding) of both systems is positive, indicating mutual attraction between the two phases. Furthermore, this indicates that the SCA does not lead to mutual exclusion between EPS-C-S-H interfaces. In addition, the bond energy of EPS/SCA/C-S-H is larger than that of EPS/C-S-H, indicating that the existence of SCA enhances the bond strength between components and makes the EPS-C-S-H system more stable. The results show that SCA can enhance the interface cohesion of the EPS/C-S-H structure, thereby enhancing its structural integrity and mechanical properties. Binding energy of two systems.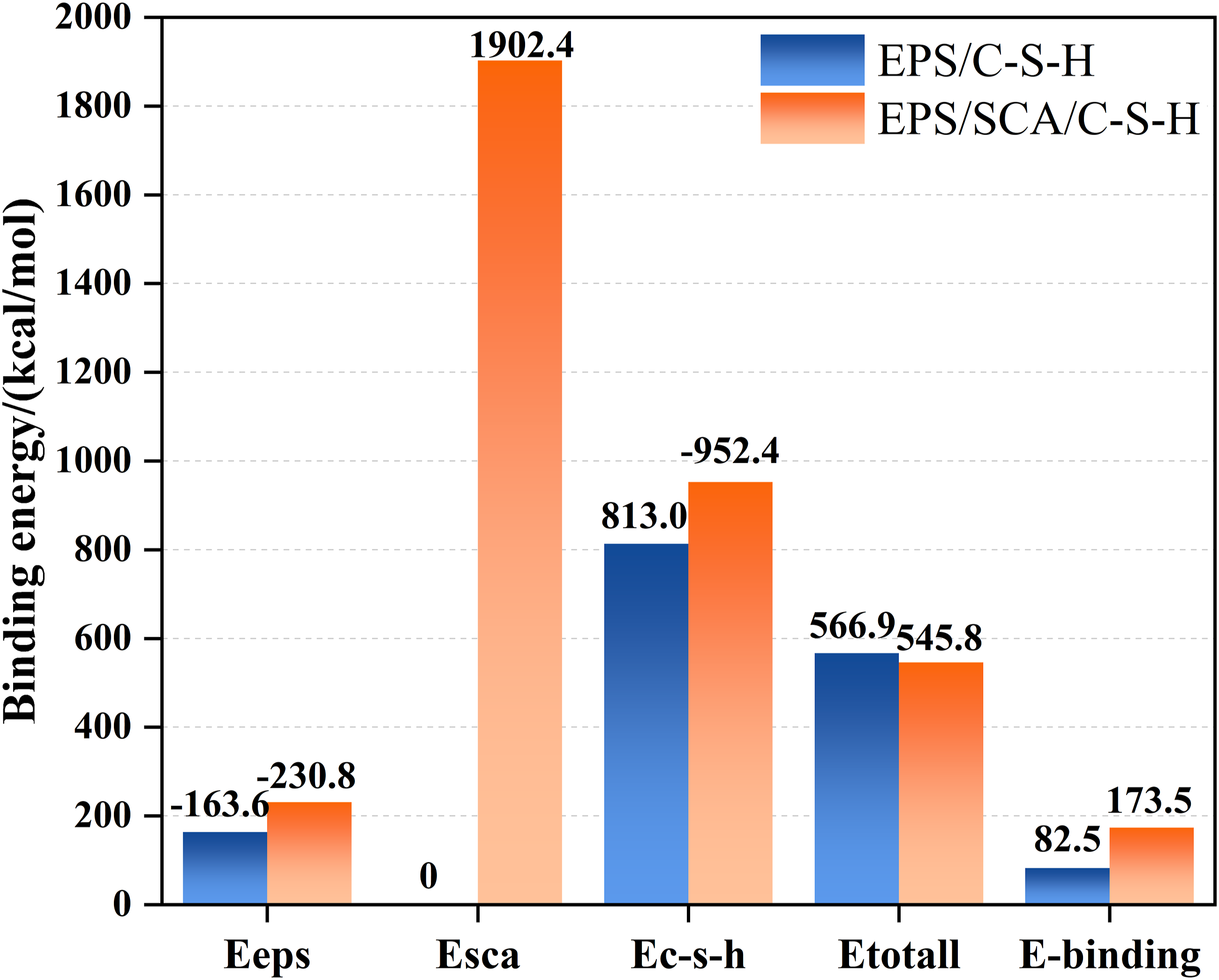
The reason why the system modified by SCA becomes more stable is that there are a large number of -OH bonds on the surface of EPS and C-S-H due to the addition of SCA. The combination process between SCA, cement, and EPS is shown in Figure 19. The addition of SCA and C-S-H undergoes a series of chemical reactions, including the silanol generated by the hydrolysis of SCA itself, as described in the above modification method and mechanism, as shown in Figure 19 (1). A part of the silanol molecules will self-condense among themselves, as shown in Figure 19 (2), and another part of the silanol molecules will condense with cement hydration products, as shown in Figure 19 (3). That is, the Si-OH group in SCA can condense with the -OH group in the hydration product and form a Si-O-Si bond.
79
This bond can increase the interfacial interaction force, and it can also react with inorganic materials containing hydroxyl to form a stable Si-O-Ca bond. Moreover, the hydrogen bond between hydroxyl groups is further transformed into a covalent bond during cement hardening, which can further improve the interface adhesion energy. In other words, SCA not only increases the contact area between the EPS and cement slurry but also shortens the distance between them and the cement matrix, acting as a bridge for interface interactions.
80
The reaction mechanism between SCA and C-S-H.
Reinforcement mechanism analysis
Microstructure-mechanical property relationships
From the combination of macroscopic and microscopic scales, the macroscopic bending test results and SEM images of the EPS-cement-based interaction show that the addition of SCA plays a crucial strengthening role, (Figure 20). Following the SCA modification, the surface of EPS beads becomes more uneven, enhancing the connection at the interface between EPS and the cement base, resulting in a larger contact area with no visible gaps. This strengthens the bond with the cement base. The addition of SCA further promoted the hydration of cement at the interface of organic polymer and inorganic silicate, formed more abundant gels, filled the weak pores of cement, made the internal structure of EPS concrete more compact, and thus improved the macroscopic mechanical properties of EPS concrete. The SCA acts as a “bridge” that enhances direct interaction between EPS beads and cement, transferring some stress from the fragile cement material to the EPS beads. This effectively enhances the strength of the composite material under stress.
81
The relation between mechanical properties and microstructure changes.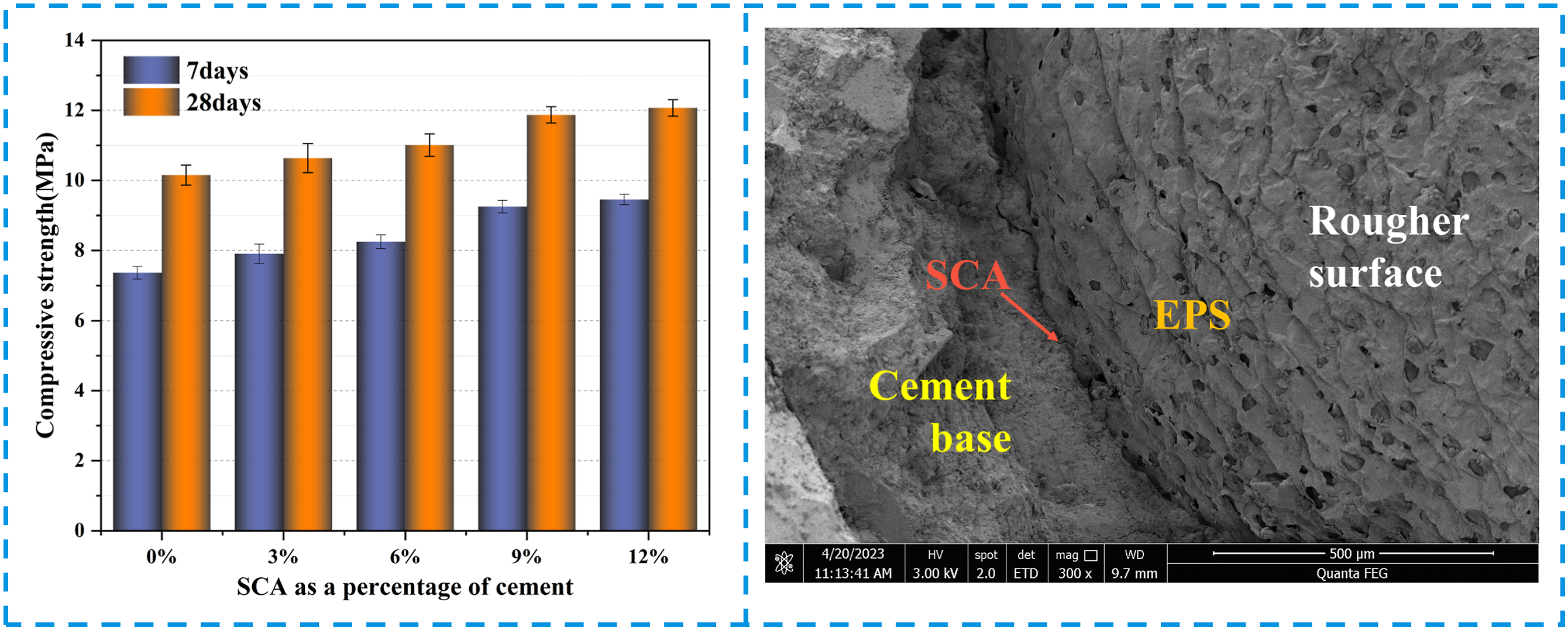
Chemical bonds -mechanical property relationships
As illustrated in Figure 21, following SCA adjustment, the EPS beads’ surface exhibits a plethora of -OH linkages, along with -OH bonds and Ca(OH)2 byproducts within their cement hydration constituents. This facilitates the establishment of a chemical connection at the junction between the organic substance and the cement hydration byproduct, wherein the Si-OH cluster in SCA can interact with the -OH cluster in the hydration byproduct to create a Si-O-Ca linkage. The hydrogen bonds with Ca are seen as vital connectors between diverse polymers at the interface.
33
The possible interaction mechanisms between the interface of (a-1,2) EPS beads and (b-1,2) cementitious.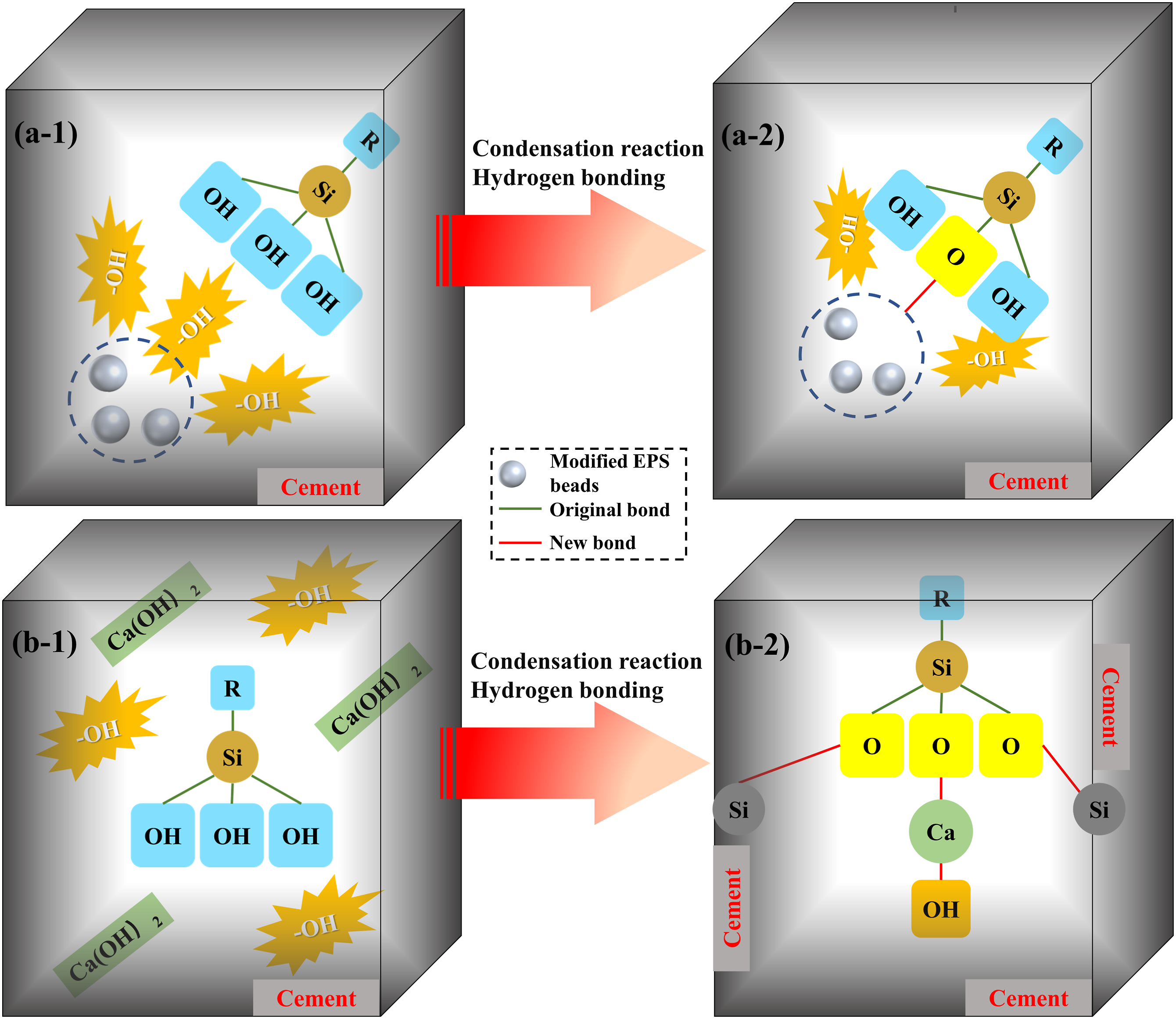
Combined with the results of molecular dynamics analysis, it can be seen that the RDF curve peaks of Heps-Ocw and Heps-Hcw in the EPS/SCA/C-S-H system are lower than those in the EPS/C-S-H system, which indicates that the addition of SCA enhances the hydrogen bonding between different phases. In addition, the peak value of the Hsca-Heps curve is higher than that of other curves, such as Heps-Hcw, which indicates that the SCA further increases the number of hydrogen bonds, thereby increasing the bonding force between different phases. However, the MSD curve of Heps is found to be larger than that of other atoms, which indicates that the newly formed hydrogen bond is not very stable. This is also supported by the computational analysis of TCF. In general, it can be seen that the addition of SCA mainly enhances the interface of EPS-C-S-H through the ionic bonds of Hsca-Ca1 and Heps-Ca1, followed by the hydrogen bonds in Hsca-Heps, Hsca-Ocw, and Heps-Ocw. The chemical bonds between the major atoms in SCA are shown in Figure 22. Chemical bonding of the main atoms in SCA.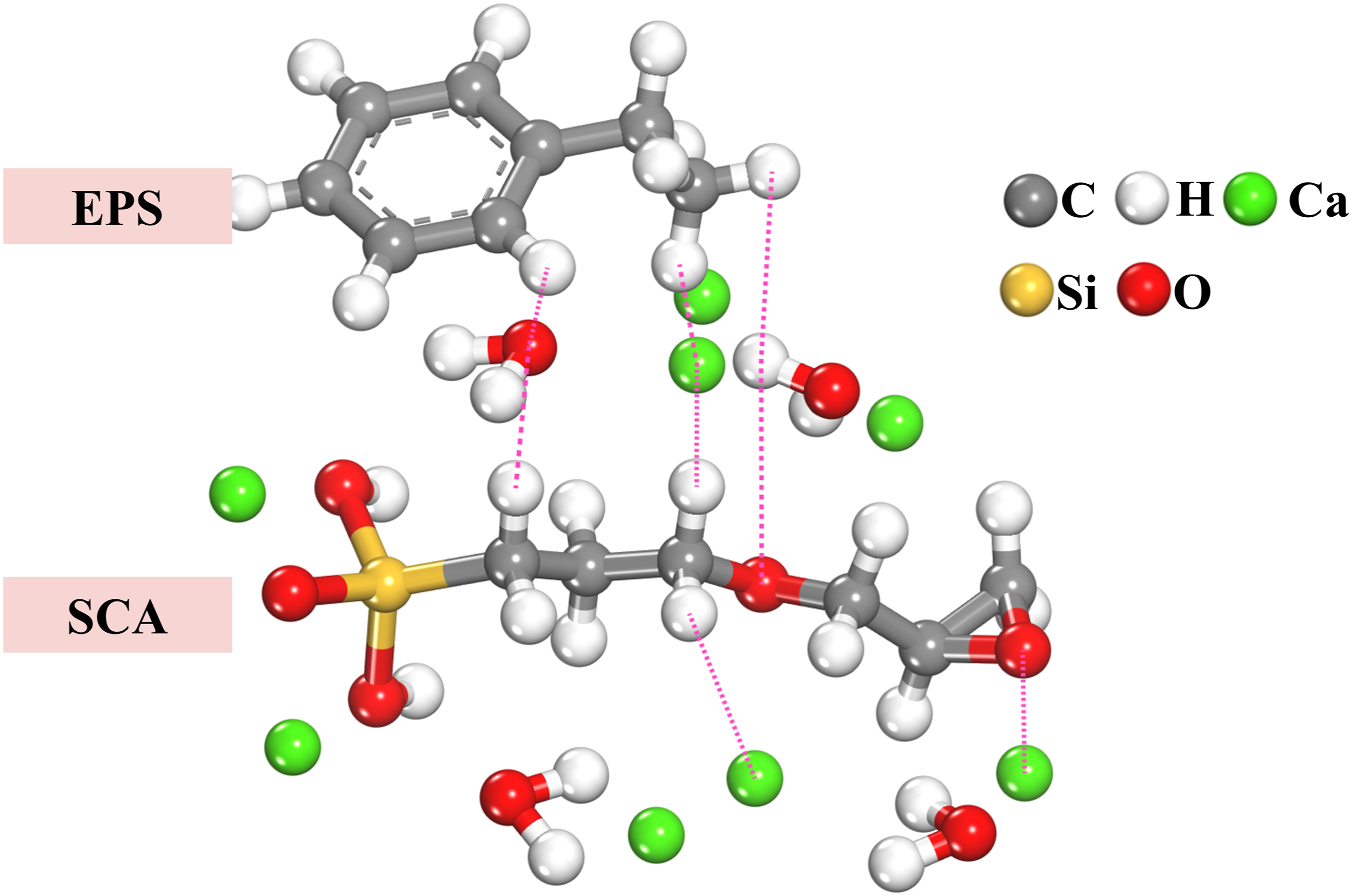
Based on the macroscopic, microscopic, and nano-level studies, the addition of SCA not only expands the surface contact between EPS cement grout but also reduces the distance of EPS cement grout in the gelling matrix by enhancing the interfacial hydrogen bond, Ca-H ionic bond, and other chemical bonds. Studies on ionic and hydrogen bonds show that the connection is not very stable; however, microscopic experiments show that the addition of SCA causes the interface to form a large number of Si-O-Si and Si-O-Ca chemical bonds. 82 Combined with molecular dynamics, the strong existence of SiSCA-OSCA-SiC-S-H and SiSCA-OSCA-CaC-S-H, which coexist with a large number of ionic and hydrogen bonds in the form of “bonding bridges,” strengthens the connection of different phases and tightly attaches EPS beads to the gelatinous matrix. Therefore, the SCA-modified EPS microbeads significantly improved the compressive and flexural strength of EPS composite concrete on the 7th and 28th days, and the effect was significant. SCA modification can enhance the interface bonding between EPS microbeads and the cementing matrix on multiple scales and significantly improve the performance of EPS concrete, making the structure more stable.
Conclusions
This study utilized a combination of macroscopic and microscopic experiments along with molecular dynamics simulation analysis to explore the impact of SCA-modified EPS beads on the interfacial characteristics of the cementitious matrix across various scales. Initially, on a macroscopic level, we evaluated the influence of different SCA (KH560) concentrations on the mechanical properties of EPS composite concrete through tests measuring macroscopic mechanical performance. Furthermore, variance analysis was conducted on the experimental data. Subsequently, at a microscopic level, we examined and analyzed the effects of SCA on the microstructure of EPS-cement interfaces using scanning electron microscopy, XRD, and FTIR analyses. Finally, at the nanoscale, molecular dynamics simulations were carried out to analyze two interface models, one with SCA and one without SCA.
Based on the comprehensive analysis and research, the following conclusions were drawn: 1. The macroscopic mechanical properties test results indicated that the addition of SCA led to an enhancement in the mechanical properties of EPS composite concrete. Macro test results and variance analysis revealed that a SCA content of 9% produced the most significant improvement effect on EPS composite concrete. This resulted in a 24% and 17% increase in compressive strength on day 7 and day 28 respectively, as well as a 39% and 16% increase in flexural strength on day 7 and day 28 respectively. 2. SEM observations demonstrated the presence of visible cracks at the interface between unmodified EPS and the cementitious matrix, which were less apparent after modification. These cracks were effectively filled, rectifying the defects of the EPS-cementitious matrix interface and creating a more compact and stable structure. 3. XRD and FTIR analysis indicated that the addition of SCA contributed to a reduction in the crystallinity of hydration products, delayed cement hydration, and decreased the production of Ca(OH)2. However, it also increased the quantity of hydrated calcium silicate gel and optimized the composition of interfacial cement hydration products. 4. Molecular dynamics simulation analysis results revealed that the inclusion of SCA enhanced the interaction forces of hydrogen bonds and Ca-H ionic bonds at the EPS/C-S-H interface, thereby enhancing the stability of the EPS/C-S-H interface. 5. By calculating the binding energy of the two system interfaces (EPS/C-S-H and EPS/SCA/C-S-H), it was determined that the addition of SCA bolstered the interaction force between the two phases, leading to a more tightly stable and compatible EPS-C-S-H interface.
Footnotes
Acknowledgements
The authors sincerely acknowledge the strong support from the School of Civil Engineering, Henan University of Technology during the research stage.
Author contributions
All authors contributed to the study conception and design.
Zehua Li wrote the main manuscript text.
Yong Feng critically reviewed the content of the study.
Chen Zhao, Jingjie Feng and Wang Chen, made a substantial contribution to data acquisition, material preparation, analysis of data.
All the authors revised and check the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Innovative Funds Plan of Henan University of Technology (No. 2020ZKCJ21) and the Zhengzhou Collaborative Innovation Project (No. 21ZZXTCX09), as well as the 2023 Henan Province Science and Technology Research Project (232102320173, 232102111126).
Data availability statement
All data generated or analyzed during this study are included in this published article.
