Abstract
A unique morphology was fabricated using melt mixing of polysulfone (PSU) and nylon 6, 6, as well as organically modified clay to produce two blended nanocomposite compositions (80/20 and 60/40 w/w) of polysulfone and nylon 6, 6. The morphology of PSU/Nylon 6, 6 blend nanocomposites with various amounts of clay has been examined using scanning electron microscope (SEM), transmission electron microscope (TEM), and wide-angle X-ray diffraction (WAXD). In the case of 80/20 (w/w) PSU/Nylon 6, 6 without clay, the Nylon 6, 6 is dispersed in the PSU matrix with an average particle size of about 6.81 micrometers (μm). After adding clay (2%, 4%, and 8%), the domain size of nylon 6, 6 decreases, although the decrease rate is much slower than initially observed. However, we discovered that when the organoclay level exceeds 2%, the matrix-domain structure transforms into a co-continuous morphology for the 60/40 (w/w) blends. The TEM studies clearly demonstrate that the organoclay preferentially positions itself in the nylon 6, 6 phase, exhibiting a high degree of exfoliation, while the PSU phase of the nanocomposites remains devoid of clay, irrespective of the amount present. This study indicates that the size of clay platelets dispersed in the PSU/Nylon 6, 6 blend plays an important role in determining the morphology and stability of these blends. Moreover, the co-continuous structures were stable against further annealing at high temperatures, thus inhibiting the coalescence of the dispersed phase in addition to reducing interfacial tension.
Keywords
Introduction
In recent decades, polymer blends have emerged as a key pathway to novel, high-performance polymeric materials.1–6 On the other hand, most polymer blends create multiphase structures due to their immiscibility and undesirable interactions caused by their large molecular weight. The morphology of the formed polymer chains influences the properties of multiphase polymeric materials, in addition to the characteristics of the individual polymers. There are two major types of morphology observed in melt-blended immiscible polymers, namely matrix-dispersed structures 7 and co-continuous structures. 8 Researchers are more interested in co-continuous blends because this morphology can offer a better combination of component properties than the dispersed structure.9–16
In contrast, nanocomposites incorporating organoclays into a polymer matrix have garnered significant attention because of their superior physical qualities in comparison to unmodified polymers. Additionally, it has been found that layering clay nanofillers are efficient polymer blend modifiers because of their ability to affect interphase phenomena as well as the formation of the structure of the dispersed phase component within the matrix.17–23 An improvement in the performance of composite products filled with aluminosilicates has been observed, attributed to variations in the morphology of polymer dispersion from batch to batch. Researchers have extensively studied several polymer matrices to understand how the addition of nanoclay affects their structure and properties. Polystyrene (PS), 24 polycarbonate (PC), 25 polyamide-6 (PA-6), 26 polyacrylamide (PAA), 27 polyurethane, 26 polyvinyl alcohol 28 and poly (ethylene terephthalate) (PET) 29 are some of the matrices commonly found in these systems. It is noteworthy that these polymer-clay nanocomposites offer several economic advantages, including their lower weight due to their lower clay content (2–5 wt%), which is an appealing feature for industrial applications. On the other hand, only a small amount of research has focused on organoclay-based nanocomposites in polymer blend systems.30–34 Gelfer found that adding organoclay to PS/PMMA blends made the PS domain size smaller. This was because the high surfactant content in organoclays made the blends more compatible, which led to the higher viscosity. 30 Conversely, with the PS/polypropylene (PP) combination, Wang et al. demonstrated comparable outcomes. 31 It has been shown that the PS domain size can be significantly reduced by adding organoclay to it. They reasoned that this phenomenon must be due to two block copolymer-like immiscible polymer chains intercalated with the clay platelets. In a study conducted by Khatua et al, researcher discovered that, even with a very small amount of organoclay added to the PA6/ethylenepropylene rubber (EPR) nanocomposite mix, the size of the EPR phase in the matrix of PA6 was dramatically reduced. 32 This effect was characterized by their explanation that the scattered domains of the exfoliated clay platelets were unable to coalesce.
This study employed direct melt extrusion to manufacture an organically modified clay, PSU, and nylon 6, 6 blend nanocomposite. Various concentrations of organoclay were systematically added to the blend nanocomposite to investigate its morphology. This study found two main things. Firstly, despite using a one-step mixing technique to make the nanocomposites, the researchers observed an even distribution of individual organoclay platelets within the nylon 6, 6 phase. Secondly, the researcher noticed that the original matrix-domain morphology changed into a co-continuous structure when the clay content in the mixture exceeded a particular threshold level. These findings hold the potential to pave the way for a novel processing method for fabricating polymeric nanocomposites with exceptional performance. It is possible to improve the processing and impact strength of polymer blends by selectively placing clay platelets in one phase. This lowers the concentration of organoclay in the nanocomposite system. Studies have shown that inorganic clay degenerates polymer material toughness35,36 as well as increasing its melt viscosity.37,38 On the other hand, it is expected that a co-continuous-like structure will aid in the formation of polymer blends that exhibit a good balance between stiffness and toughness. 39
Experimental
Materials and sample preparation
The supplier of the Nylon 6, 6 used in this investigation was Sigma Aldrich Chemical Pvt. Ltd. Nylon 6, 6 is desirable not just because of its excellent physical properties but also because its predecessors are cheap. The commercial product known as polysulfone (PSU) was purchased from Toray Industries of Japan. Polysulfones are a family of high performance thermoplastics. The technical information of the polymer (PSU) such as melting temperature, Density and tensile strength are 250 – 410OC, 1.13 - 1.66 g/cc and 44.8 - 179 MPa. The stability and durability of these polymers are well-known properties at high temperatures. Technically produced polysulfones contain an aryl-SO2-aryl component. Southern clay product Inc. Supplied the organoclay known as Closite 30B for this study. During the modification of the montmorillonite, dimethyl dehydrogenated tallow ammonium was added to enhance the spacing of the galleries in the original sodium montmorillonite in order to increase its strength. There are 95 mequiv/100 g of cation exchange capacity in the clay. In order to blend PSU and Nylon 6, 6, in 80/20 w/w and 60/40 w/w ratios with varying quantities of organoclay, a twin screw extruder (Haake-minilab) was used. The mixture was then heated to 240°C at a speed of 100 r/min for 10 min. Prior to mixing, the clay and polymers were each dried for a minimum of 24 h in a vacuum oven set at 80°C. Using the same technique, PSU and Nylon 6, 6 nanocomposites were also created. Scheme 1 displays the chemical structures of these materials. Chemical Structure of (a) Polysulfone (PSU), (b) Nylon 6, 6 (c) Closite 30B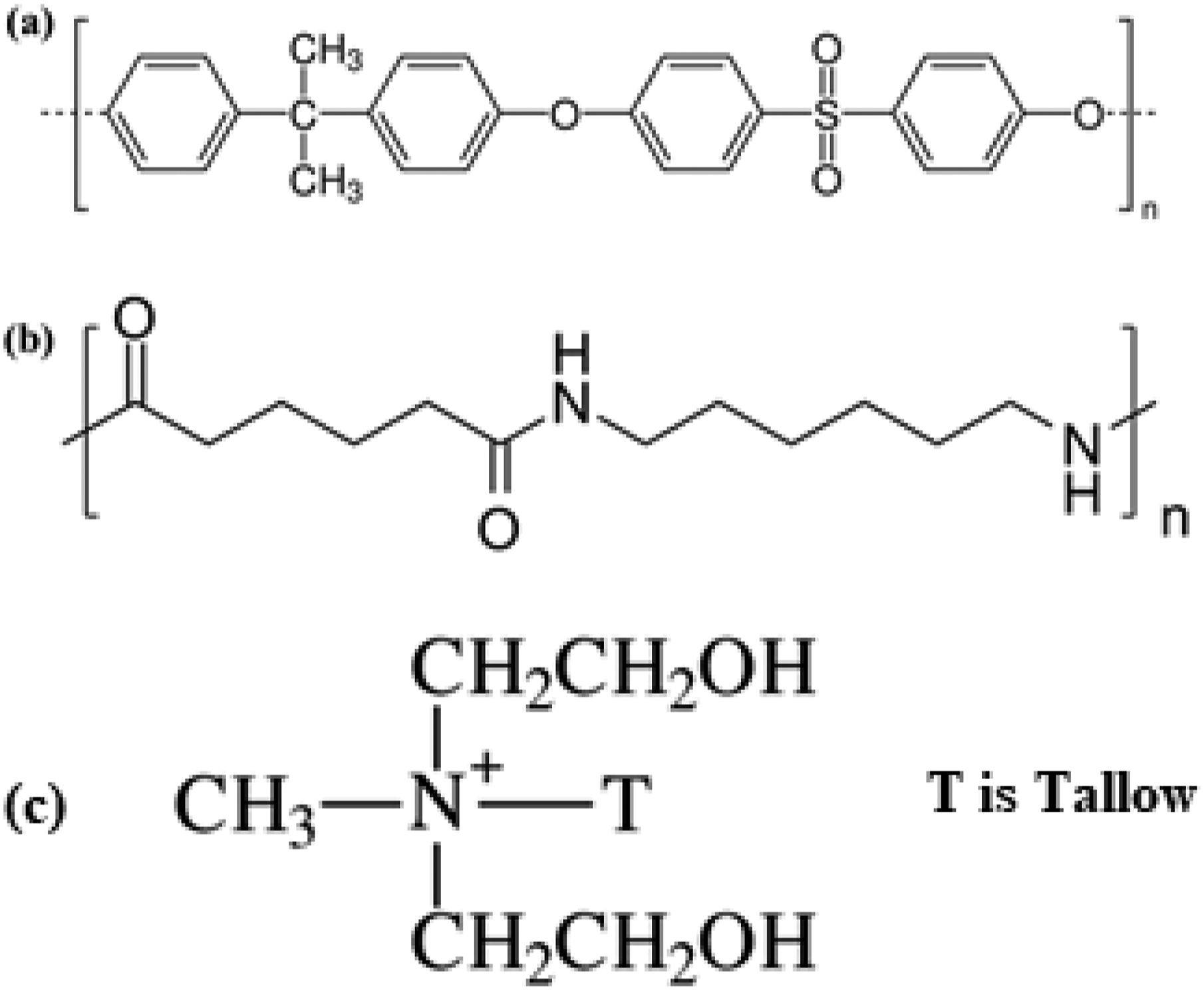
Characterization
Scanning electron microscopes (SEMs) were used to elucidate the morphology of PSU/Nylon 6, 6 blends and their nanocomposites. The following sample preparation techniques were utilized for SEM characterization. The testing procedure included exposing the extruded polymer strands to liquid nitrogen for an extended period of time, which caused the sample to become brittle and shatter. It was then necessary to etch the fracture surface using either of two methods in order to expose the fractured surface. As an etching agent, a solution of formic acid was used to dissolve the nylon 6, 6 phase, and a solution of dimethyl formamide (DMF) was used to dissolve the PSU phase from the nanocomposites’ surface. After completing the etching process, the specimens were dried at room temperature in a vacuum oven, before being coated with a thin layer of gold. The gold-coated samples were studied using a scanning electron microscope (SEM) (Philip XL-20) operating at 10 KV. According to the following relation, the number-average diameter of sea-island domains was determined:
In this case, Ni is defined as the total number of dispersed domains from the SEM images of a diameter of Di. There are a total of 200 – 300 particles in the analysis.
To assess the structure and/or extent of clay particle expansion and exfoliation in the nanocomposites, wide-angle X-ray diffraction (WAXD) was carried out on the nanocomposites. WAXD was performed on a Rigaku diffractometer (Rint 2500VH/PC, Rigaku) running at 40 kV and 100 mA, Using Cu Ka radiation (k = 1.541 Å). The scanning range was 3 – 30° with a scanning speed of 1°/min.
To investigate the dispersion of clay platelets in a blended materials, a transmission electron microscope (TEM) (JEOL JEM 1230) with an accelerating voltage of 120 KV was used with an objective of dispersion analysis. The ultra-microtomization was performed at room temperature on blended nanocomposite samples in order to produce a segment with a thickness of about 60 nm, a thin section of the sample was created and then stained for 20 min with OsO4.
To quantify the mechanical characteristics of polymer nanocomposites that are dynamically affected by temperature changes, dynamic mechanical thermal analysis (DMTA) is used. These findings may be used to determine the crystallization parameters and glass transition temperature (Tg) of polymers. A Rheovibron DDV-25FP-S device from Orientec Corp. Was used to conduct a dynamic mechanical assessment of the nanocomposites (5 cm × 1 cm × 2 mm) in an air environment from 150°C to 250°C at a frequency of 10 Hz, in accordance with ASTM E1640. The samples were subjected to three-point bending and heated at a rate of 5°C/min.
Results and Discussion
Phase Morphology Study of the PSU/Nylon 6, 6 Blend and its Nanocomposites
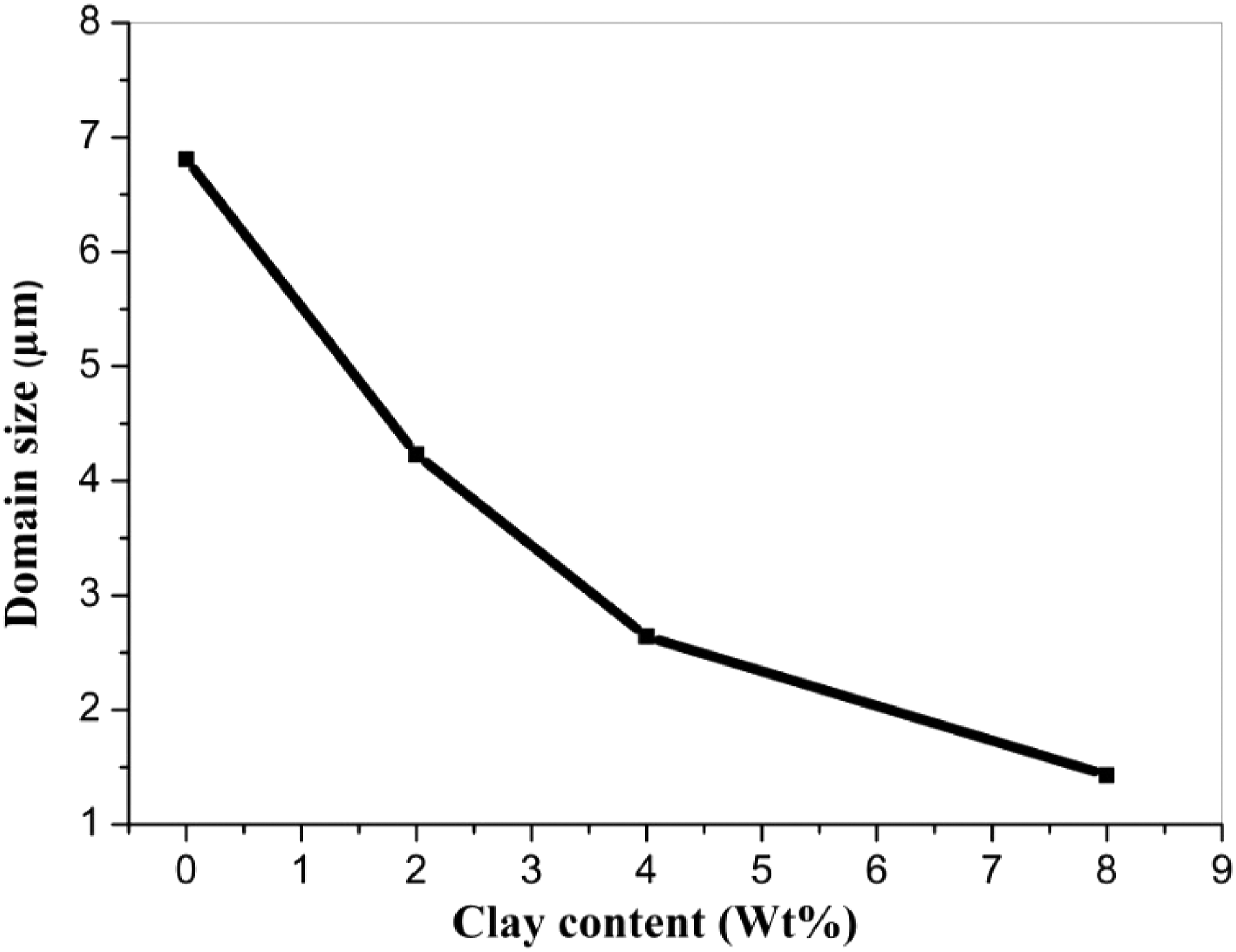
For phase morphology studies, two types of blend compositions, that is, 80/20 (w/w) and 60/40 (w/w), have been considered as samples. The SEM pictures of blends of 80/20 (w/w) PSU/Nylon 6, 6 with varied clay contents are displayed in Figure 1. A small amount of organoclay (2%), was found to reduce the dispersed domain size (D) of nylon 6, 6 phase by roughly a quarter compared to it’s without clay value. It should be noted that as clay concentration increases, the D decreases, although the decrease rate is much slower than it was initially. It is clear from the results of this experiment that the presence of the organoclay in the blend of PSU with nylon 6, 6 plays a significant role in reducing the size of the dispersed domains in the blend. Figure 2 shows a plot of the amount of clay versus the number-average (Dn) domain diameters based on the SEM images. It was observed that D decreased rapidly at lower amounts of clay, which was followed by a slow but gradual decrease in D with the further increase in clay, which is consistent with what was observed at lower amounts of clay. Interestingly, this curve bears a resemblance to the emulsification curve, as reported for an immiscible blend containing a block copolymer (or graft copolymer).
40
This composition, however, did not result in a co-continuous structure, which is what we were trying to achieve. The above is a set of SEM images of PSU/Nylon 6, 6 blends (80/20 w/w) with various amounts of clay included: (a) 0%, (b) 2%, (c) 4%, and (d) 8%. Plot of Dn in the 80/20 (w/w) PSU/Nylon 6, 6 blend versus the amount of clay.
A phase morphology analysis of the phases of the PSU/Nylon 6, 6 blend (60/40 w/w) and its nanocomposites with various amounts of organoclay was carried out after extraction of the blends with DMF in order to obtain the co-continuous structure as shown in Figure 3. Observations of the PSU/Nylon 6, 6 blend without clay show that it exhibits characteristic sea-island morphologies where the nylon 6,6 domains are dispersed throughout the PSU matrix due to the relatively small fraction of nylon 6,6 in the blended composite compared to the PSU matrix alone. However, adding a small amount of organoclay to the blend phase caused a noticeable change in its morphology. Extraction tests were performed to obtain more information of structure of the co-continuous network. As shown in Figure 3(b), the phase morphology analysis of a blend nanocomposites containing 2% organoclay was conducted after their extraction with DMF to determine their phase morphology. It was possible to etch the PSU phase with DMF since it is an excellent solvent for PSU, but it will not dissolve nylon 6, 6. According to the experiments that were carried out within the nanocomposites, both PSU and nylon 6, 6 phased out as continuous phases within the nanocomposites, which indicates that the sea-island structure has been transformed to become a co-continuous structure in the presence of 2% organoclay. Thereby proving that after addition of clay, the composition reaches the percolation threshold where the concentration of the dispersed phase reaches a point at which co-continuity is set in, and a phase network starts to form as a result. In spite of the fact that the blended nanocomposites remained co-continuous even after a significant increase in the organoclay loading, but the phase size of the composites decreased with increasing clay loading, as shown in Figure 3(c) and (d). The Scanning Electron Micrographs (SEM) of PSU/Nylon 6, 6 (60/40 w/w) blend with different concentrations of clay: (a) 0%, (b) 2%, (c) 4%, and (d) 8%.
Clay’s Dispersion in the PSU/Nylon 6, 6 Nanocomposites
The dispersion of clay throughout the blend is vital to understanding the process by which clay creates morphological changes in the blend when it is added. Figure 4 displays the WAXD profiles of the PSU, nylon 6, 6 and PSU/Nylon 6, 6 (60/40 w/w) blend nanocomposites, along with the organoclay. There was 4% organoclay in each nanocomposite. The diffraction pattern of the pure organoclay (001) at 2θ = 8.63° revealed an interlayer spacing of 1.86 nm. This observation was made in the composites of PSU/organoclay, in which the characteristics’ peak (001) of the clay moved to 2θ = 7.89°, which corresponds to a d spacing of 3.12 nm. As a result, it appears that some molecular chains in the PSU are able to form an intercalated structure through their intercalation between clay galleries. However, the absence of any clay diffraction peaks in the WAXD for the nylon 6, 6 nanocomposites indicates that the clay platelets were fully exfoliated within the Nylon 6, 6 phase, which is in line with the findings that were previously published.41,42 Finally, there were no discernible diffraction peaks in the WAXD profile of the PSU/Nylon 6, 6 blend nanocomposite, which was identical as the profile of the nylon 6, 6 nanocomposite. The clay has a greater affinity for nylon 6, 6 than PSU, according to the WAXD data. The polar nature of the modifier of this organo-modified nanoclay is responsible for its selective affinity towards the Nylon 6,6 phase. In the blend nanocomposites, clay may therefore preferentially be located in the nylon phase. The different WAXD patterns shown here are for (a) pure organoclay, (b) PSU/clay nanocomposite, (c) Nylon 6, 6/clay nanocomposite, and (d) PSU/Nylon 6, 6/clay blend nanocomposite. In all nanocomposites, the clay concentration is 4%.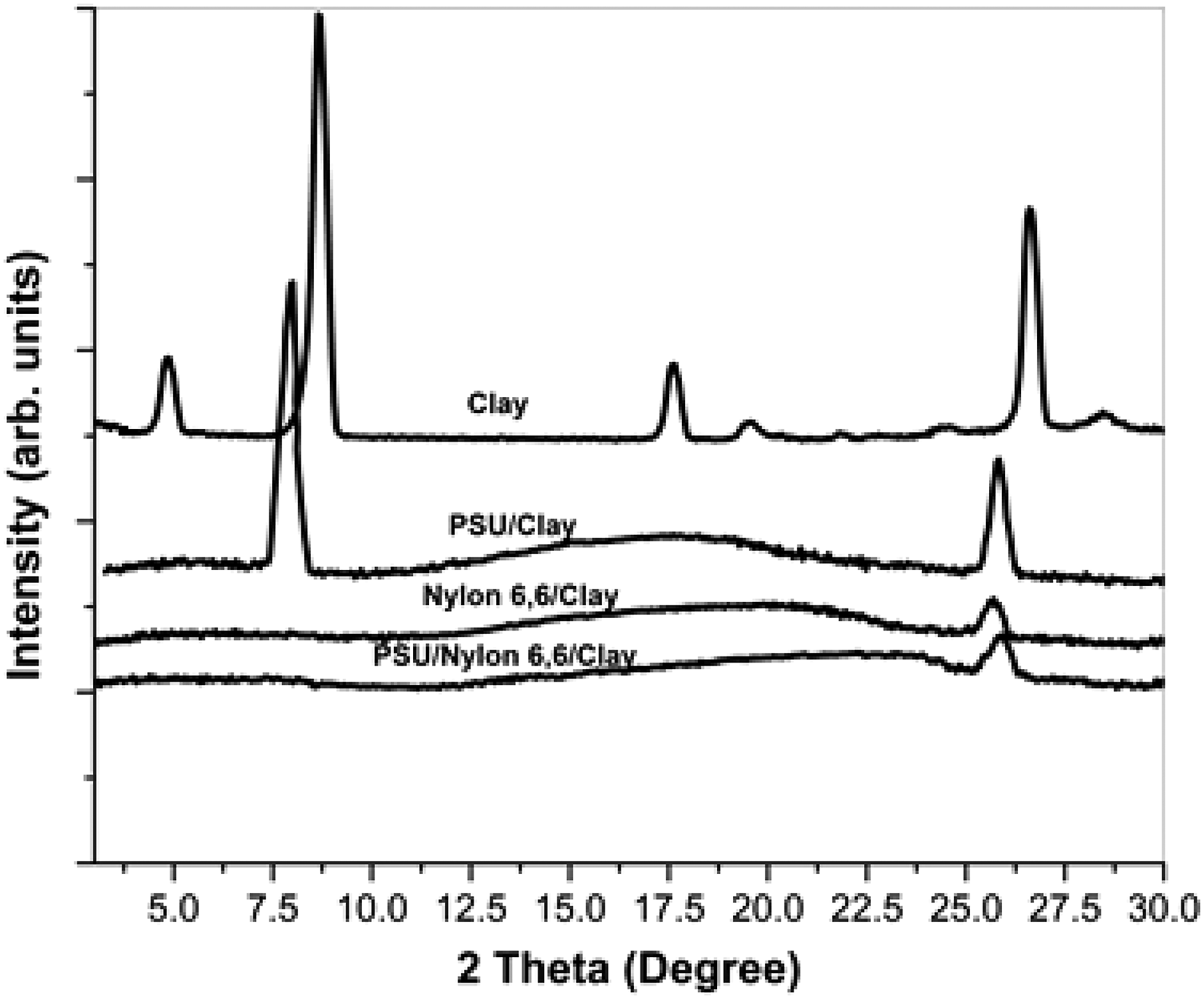
The micrographs in Figure 5 show TEM images of the blend nanocomposite that contains approximately 4% organoclay, after it has been stained with OSO4 to increase phase contrast under various magnifications of light. The PSU phase is the black, small-domain portion, while the nylon 6, 6 phase is the other portion. As can be seen in the graph, the dark line in the nylon 6, 6 phase represents the organoclay phase. The phase size and blend shape are consistent between the TEM micrographs and the SEM image. The higher magnification TEM image (Figure 5(b)) clearly reveals the organoclay’s specific location in the nylon 6, 6 phase, where it has heavily exfoliated into single platelets. However, these are not apparent in the PSU phase TEM micrographs of PSU/Nylon 6, 6 (60/40 w/w) nanocomposite with 4% clay, (a) lower magnification, and (b) high magnification. The portion with tiny domains and black color is PSU phase, whereas the remainder is nylon 6, 6 phase. The dark line that exists in the nylon 6, 6 phase is the organoclay. Schematic diagram for the co-continuous PSU/Nylon 6, 6 blend nanocomposite. A black line represents the organoclay, a blue section represents nylon 6, 6 phase, and a gray section represents PSU phase.
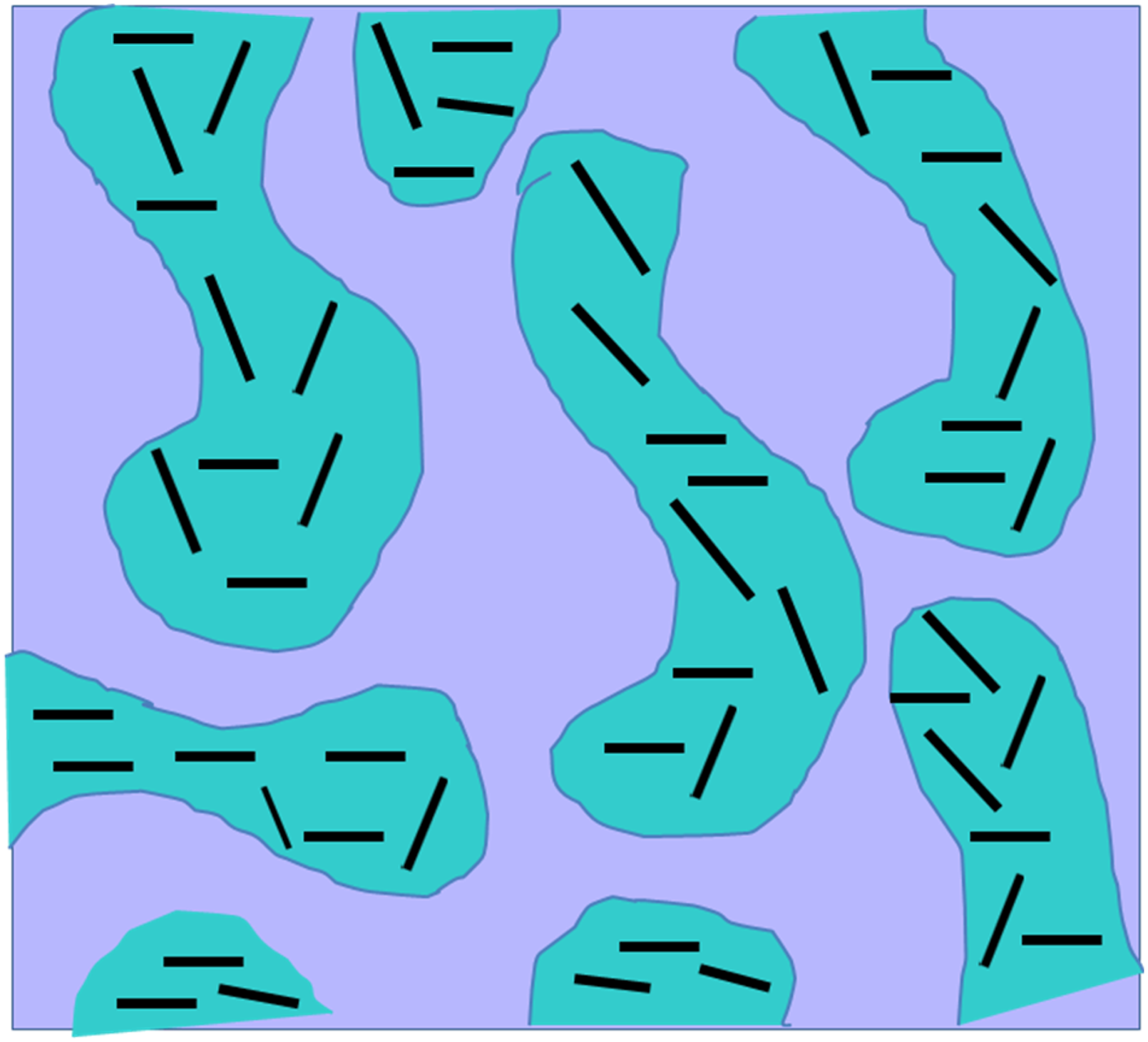
Generally, co-continuous structures can be formed in an area around a phase inversion composition, and the strength of the co-continuous structure will be determined by the viscosity ratio between the phases involved in the inversion composition.43,44 The rheological behavior of polymers has been found to be significantly impacted by exfoliated clay platelets; specifically, they have been found to cause an increase in melt viscosity as well as an increase in shear thinning behavior in regions with high shear rates.45,46 Here, the precise location of clay platelets changes the viscosity of the nylon 6, 6 phase but their impact on the PSU phase’s melt behavior is unaffected. Consequently, a small amount of organoclay will cause a change in the ratio of viscosity between the two phases, which results in the formation of a unique co-continuous morphology when the two phases are combined. However, during melt mixing, the high aspect ratio of the clay platelets that are spread out in the nylon 6, 6 phase might slow down phase coalescence. This will make the structure more stable. Figure 6 depicts a schematic diagram of the co-continuous phase structure of the PSU/Nylon 6, 6 nanocomposites. According to the morphological data obtained by SEM and TEM, it has been predicted that the co-continuous phase structure of the PSU/Nylon 6, 6 nanocomposites would effectively combine the properties of these two components to produce a co-continuous nanocomposites. As a consequence of the fact that the exfoliated clay platelets are only distributed within the nylon 6, 6 phase where they have a positive influence on the modulus and heat resistance of nylon 6, 6 parts. It can be argued that the morphology of the PSU/Nylon 6, 6 nanocomposites represents a good structural paradigm for multi-component polymeric systems, where each component is capable of contributing to the material properties in an independent way.
Dynamic Mechanical Properties (DMA)
In this study, the dynamic mechanical characteristics of the PSU/Nylon 6, 6 blend and its nanocomposites with varying concentrations of organoclay were investigated. The effect of temperature on the storage modulus, E, is displayed in Figure 7. In comparison to the blend without clay, the blend nanocomposites exhibited a notable increase in modulus over the temperature range examined. It was also observed that the increase in the loading of organoclay contributed to an increase in modulus when the loading content of organoclay with a wide aspect ratio was increased. However, Figure 7 shows notable variations in the PSU/Nylon 6, 6 blend with and without clay, particularly in the high temperature zone (over 220°C). The nylon 6, 6 domains were distributed in the PSU phase of the PSU/Nylon 6, 6 blend without clay, with PSU acting as the matrix. The matrix becomes soft at temperatures greater than the glass transition temperature (Tg) (206°C) and starts to flow at temperatures even higher than that. As a result, at temperatures above 220°C, the blend did not show any strength. The DMA curve for the nanocomposite with 2% organoclay, however, looked quite different. In the high temperature range, this sample could sustain itself until the melting point of nylon 6, 6 (about 268°C) was achieved. In Figure 3, it can be seen how a sea-island structure, which was present in the PSU/Nylon 6,6 blend without clay, changed a co-continuous morphology when more than 2% organoclay was added to the blend. In the nanocomposites, the phases Nylon 6, 6 and PSU were both continuous. In spite of PSU’s softness in the high temperature range, nylon 6,6 retains its mechanical properties until it melts. The difference in thermal behavior between the PSU/Nylon 6, 6 blend with 2% clay and without clay demonstrates phase inversion, in line with the SEM findings presented in Figure 3. The dynamic storage modulus, E, for PSU/Nylon 6, 6 (60/40 w/w) nanocomposites with different quantities of organoclay as a function of temperature.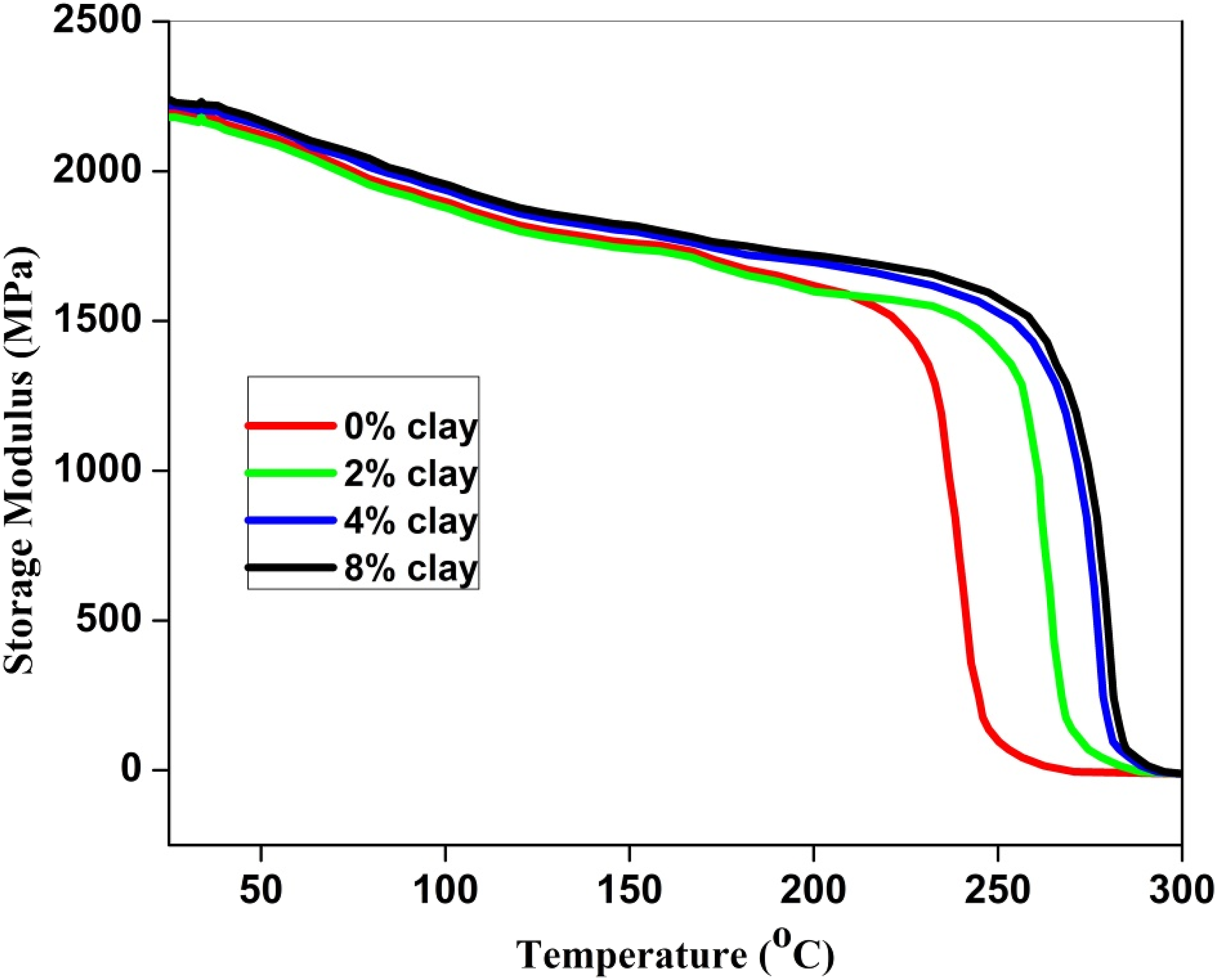
Viscosity
An analysis of the dynamic viscosity curves for the nylon 6, 6, Nylon 6, 6/clay, and PSU systems is presented in Figure 8 in order to examine how changes in viscosity are influential on the observed morphological evolutions. It is evident that adding clay to the nylon 6, 6 phase caused the melt viscosity to slightly rise when measured at a processing temperature of 260°C. As a result of taking into account the shear rate induced by the Haake mixture, which is estimated to be between 8.7 s−1 (deep section of mixture) to a maximum of 34.2 s−1 (gap of the mixture), the increase in nylon 6, 6 viscosity induced by clay addition is found to be minimal (Figure 8). It must be noted that although the nylon 6, 6 phase has a significant difference in viscosity from the PSU phase due to its higher viscosity. With the addition of clay in the PSU/Nylon 6, 6 blend nanocomposites system the viscosity ration of the two phases is decreased and the co-continuous phase structure appears.32,47 Furthermore, because the clay particles belong to the nylon 6, 6 phase, they cannot act as defects to facilitate the PSU phase’s breakup. In light of the above, the clay particles in the nylon 6, 6 phase only act as a barrier in order to retard the process and reduce the amount of phase coalescence. Melt viscosity as a function of shear rate for (a) pure PSU, (b) PSU/Nylon 6, 6 (60/40 w/w) nanocomposites with 8% clay, (c) PSU/Nylon 6, 6 (60/40 w/w) nanocomposites with 4% clay, and (d) pure Nylon 6, 6.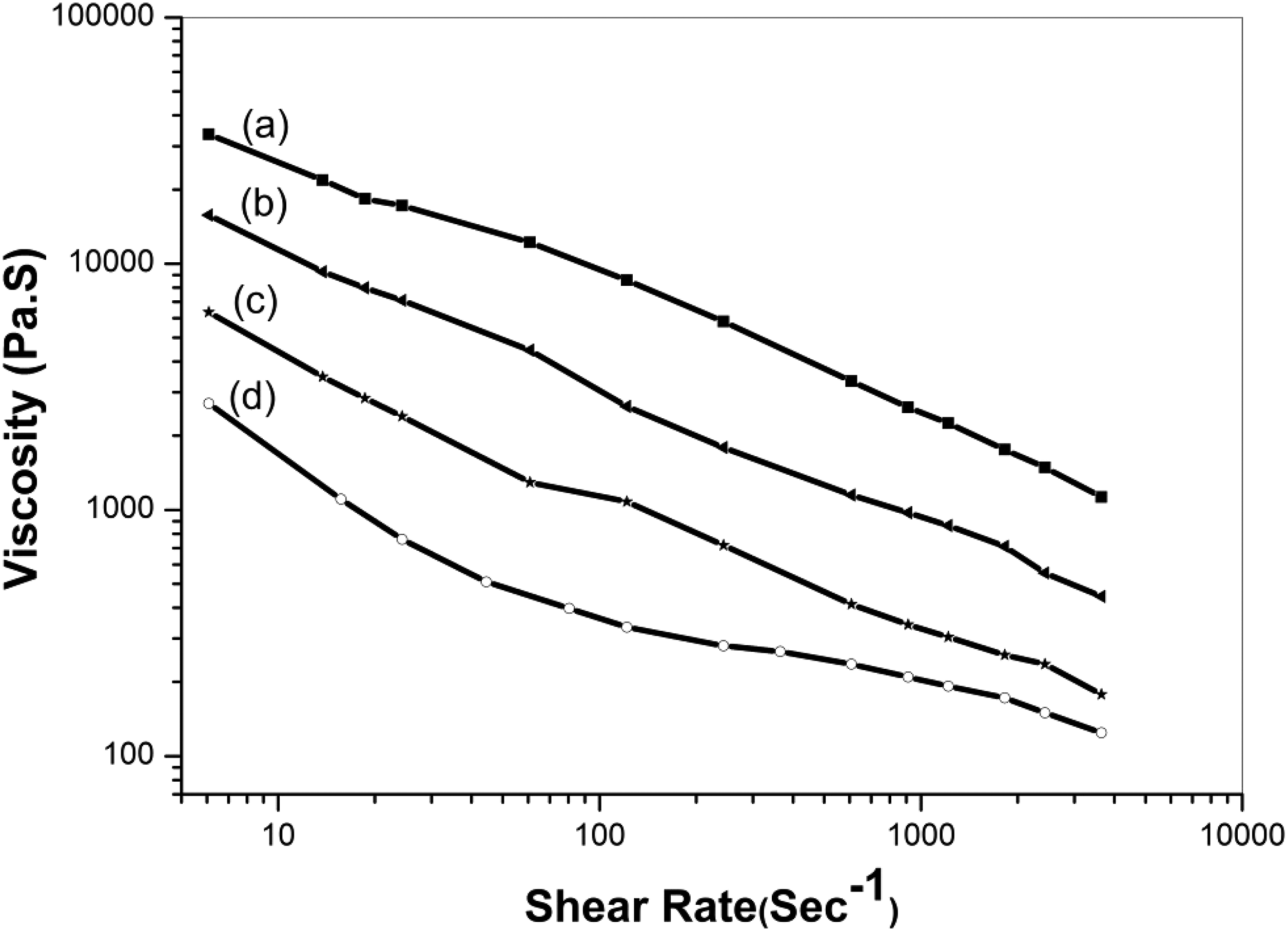
In addition, it is imperative to take into consideration the high aspect ratio of clay platelets, which may in turn have a significant effect on phase coalescence during the melting process. The development of morphology during melt processing can be mainly attributed to flow-induced processes. The particles in the dispersed phase will undergo a series of deformations, break up, coalesce, and finally reach a dynamic equilibrium state as a result of this process.48,49 Due to the large aspect ratio of clay platelets, they can act as a barrier, preventing coalescence of the PSU phase, which leads to a change in the morphology of the blend when clay platelets are incorporated into the blend. The annealing experiment on the nanocomposites provides proof of this effect. As shown in Figure 9, this graph illustrates the annealing of the blends without and with clay, that is, 0%, 4%, and 8% clay, at different annealing times without applying pressure. After annealing, there was a notable increase in the diameter of the PSU domains in the blend without clay. However, there was almost no change in the diameter of the co-continuous phase when 4% and 8% clay were present in the blend. Plots of phase size versus annealing time of PSU/Nylon 6, 6 blend with 0%, 4%, and 8% clay.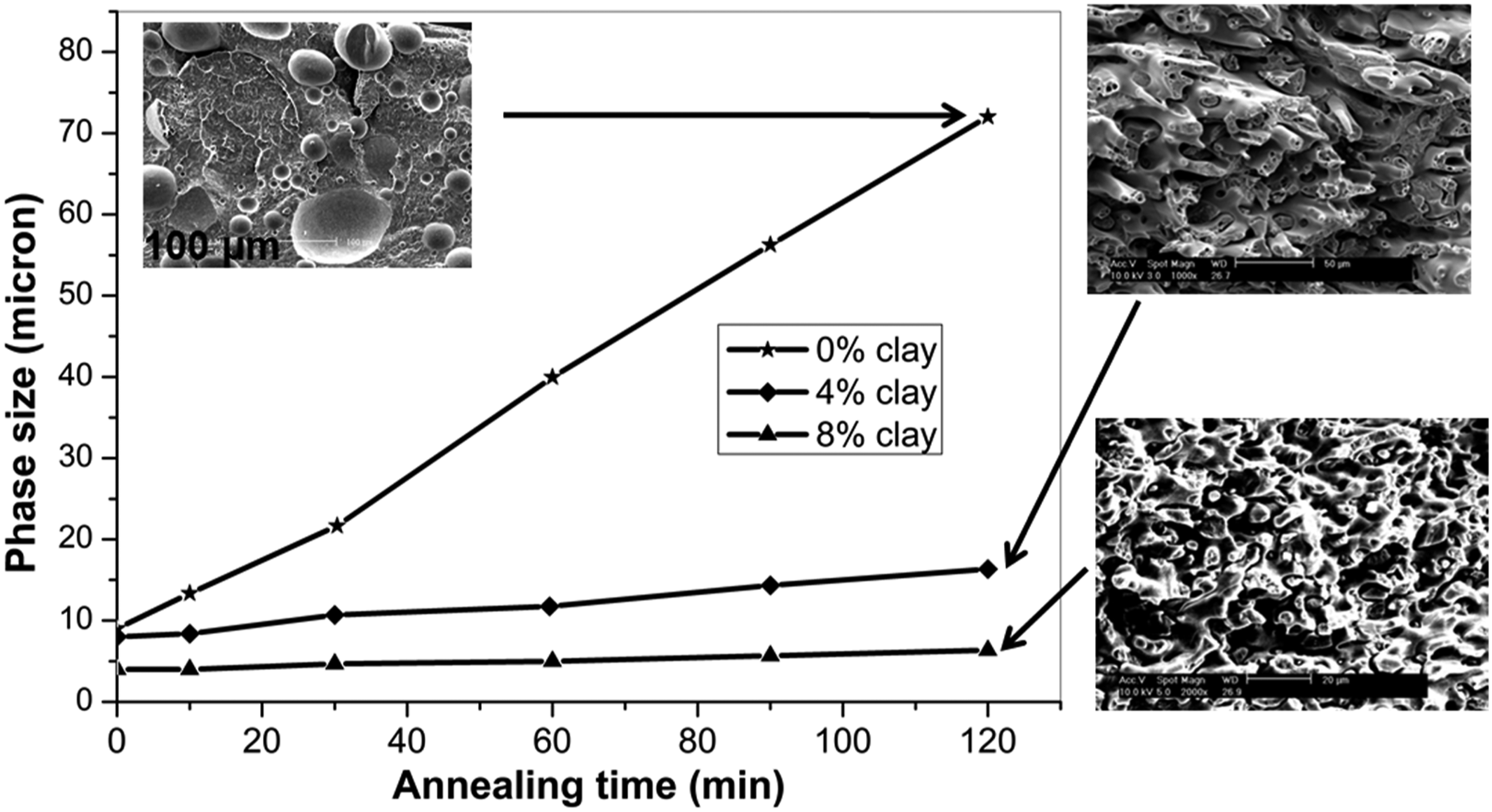
Conclusion
This study investigates the morphology of PSU/Nylon 6, 6 blend nanocomposites with varying clay contents. The findings show that selectively locating clay platelets that have been peeled off in the nylon phase changes the viscosity ratio between the nylon 6, 6 and PSU phases. This makes nylon 6, 6 more viscous and makes it harder for the PSU phase to stick together. As a result, adding a modest amount of clay causes a significant reduction in the average diameter of the scattered PSU domains. Higher clay levels have been seen to change the shape of the sea-island into a co-continuous structure, where the nylon 6, 6 and PSU phases are continuous throughout the sea-island structure. Based on the results of the dynamic mechanical analysis, it can be stated that the nanocomposites made up of co-continuous blend possess significantly better mechanical properties than the pure blend nanocomposites.
Footnotes
Author contributions
Tanmoy Rath: conceptualization, formal analysis, investigation, methodology, visualization, writing – original draft, writing – review and editing. Ibrahim A. Alnaser: investigation, methodology and editing. Asiful H. Seikh: investigation, methodology and editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the King Saud University, Riyadh, Saudi Arabia for funding their research with the grant number (RSPD2024R597).
