Abstract
Reduced graphene oxide (RGEO) and N-[4-(chlorocarbonyl)phenyl]maleimide-functionalized reduced graphene oxide (MFRGEO) were used as nanofillers for polymethyl methacrylate (PMMA) matrix nanocomposites to enhance thermal stability. Methyl methacrylate containing nanofiller of four different weight percent (0.2, 0.4, 0.6, and 0.8) was polymerized using ultrasonic radiation-assisted bulk polymerization. The Fourier-transform infrared spectra showed the absence of chemical interaction between the filler and the matrix phase. Morphology of nanocomposites studied using scanning electron microscope confirmed the assistance aided by ultrasonication in the uniform dispersion of nanofiller in the PMMA matrix. Thermogravimetric (TG) study revealed the presence of MFRGEO enhanced the thermal stability of PMMA by shifting the entire degradation to higher temperature. The thermal stability of PMMA nanocomposite was improved by as much as 40°C at just 0.8 wt% loading of MFRGEO. Differential TG study also supported the role of maleimide functionalization on RGEO in the enhancement of thermal stability of PMMA by means of retarding the degradation rate of unsaturated chain ends in the PMMA matrix. Unlike MFRGEO, RGEO failed to enhance the thermal stability of PMMA.
Keywords
Introduction
Graphene is a potent nanofiller for polymer matrix composites 1 having better thermal stability and mechanical properties. Since it is hydrophobic in nature, its dispersion in monomer for making polymer composite is difficult. These necessitate the need for functionalized graphene.
In order to make thermally stable and flame resistant graphene with improved dispersibility in polar media, graphene oxide is functionalized with N-[4-(chlorocarbonyl)phenyl]maleimide (Cl-CPMI). Mono and bismaleimides are widely used to make materials for high-temperature applications. 2,3 On exposure to high temperature, they start to melt and undergo polymerization without the evolution of volatile products. The Cl-CPMI-functionalized reduced graphene oxide 4 (MFRGEO) is being highly polar and its dispersibility in polar medium is good.
In this study, polymethyl methacrylate (PMMA) is chosen as a matrix phase. PMMA is a most promising material used in construction, automotive, and medical fields because of its mechanical properties, transparency, and compatibility with human tissue, respectively. 5,6 Poor thermal stability of PMMA reduces plasticity and induces transparency loss, powdering, cracking, and so on. 7 –9 When exposed to heat, PMMA degrades into monomer through unzipping with the release of volatile fuel. 10,11 One of the possible ways to enhance thermal stability without altering mechanical properties, electrical, and thermal conductivity is to incorporate nanoscale fillers into the polymer matrix. 12 –14 The polar ester group in methyl methacrylate (MMA) monomer is responsible for its polar nature. 15
A variety of functionalized graphene nanofillers 16 –21 have been reported to improve the thermal stability of PMMA nanocomposites prepared via conventional in situ polymerization. Without altering the thermal degradation path, homogeneously distributed nanofillers enhanced the thermal stability of PMMA nanocomposites by shifting the onset degradation temperature to higher temperature. In addition to the improvement in the glass transition temperature, tensile strength, Young’s modulus, and mechanical strength were also improved. Rajender and Suresh 19 prepared PMMA-acrylated graphene oxide nanocomposites having higher thermal stability, rheological, and mechanical properties via surface-initiated atom transfer radical polymerization. Significant reduction in the elongation at break of PMMA nanocomposites versus neat PMMA was reported by Paydayesh et al. 20 Poor quality of dispersion at higher loadings (>1 wt%) of nanofillers was also reported.
In the present work, a series of PMMA-reduced graphene oxide (RGEO) and PMMA-MFRGEO nanocomposites were prepared with high level of dispersion using ultrasonication-assisted conventional in situ polymerization. The effect of RGEO and MFRGEO on the thermal stability of PMMA was studied using thermogravimetric (TG) analyzer. Further surface morphology of the nanocomposites was also investigated using scanning electron microscope (SEM).
Experimental
Materials
MMA supplied by Alfa Aesar, Karlsruhe, Germany, was extracted with about 20 mL of 0.5% sodium hydroxide solution in a 250 mL separatory funnel to remove phenolic inhibitors. 22,23 The extraction was repeated till the aqueous layer was colorless, that is, the monomer, MMA, was completely freed from the phenolic inhibitors and then washed thrice with distilled water. Washings were rejected. Finally, the inhibitor-free MMA was dried over anhydrous magnesium sulphate and filtered. MMA freed from inhibitor was distilled under reduced pressure of 95 mm Hg at 84°C. 2,2′-Azobisisobutyronitrile (AIBN) obtained from Merck, Mumbai, Maharashtra, India, was recrystallized from methanol.
Synthesis of nanofillers
Nanofillers, RGEO and MFRGEO, were prepared according to the procedure reported. 4
Preparation of PMMA-RGEO and PMMA-MFRGEO nanocomposites
MMA flushed with oxygen-free dry nitrogen gas was used. MMA was bulk polymerized into PMMA using approximately 0.125 wt% of AIBN as the initiator. The outline of the apparatus setup used for bulk polymerization is depicted in Figure 1(a). Colloidal suspension of RGEO (0.0, 0.2, 0.4, 0.6, and 0.8 wt%) in MMA containing AIBN taken in a clear Borosil glass test tube was placed in a thermostated ultrasonic water bath kept at 20°C and ultrasonicated for 30 min. After 30 min, the temperature of the ultrasonic bath was raised to 60°C at a heating rate of 1°C min−1 and the polymerization was carried out for 150 min at 60°C under ultrasonication. After that, sonication was stopped to avoid the formation of air voids and the polymerization was continued for further 48 h at 45°C. The neat glass clear PMMA and black-colored PMMA-RGEO nanocomposite rods were carefully removed from the glass tube and post cured at 70°C in a hot air oven for 6 h. PMMA-MFRGEO nanocomposite rods depicted in Figure 1(b) were also prepared using the procedure adopted to prepare PMMA-RGEO nanocomposites.
(a) Outline of the apparatus used for bulk polymerization and (b) photograph of PMMA-MFRGEO nanocomposites.
Methods
The Fourier-transform infrared (FTIR) spectra were recorded on a SHIMADZU-8400 S infrared spectrophotometer using KBr pellet technique. The absorption bands in FTIR spectra were used to ascertain the presence of RGEO and MFRGEO in the PMMA matrix. The morphological properties of the materials prepared were studied using Carl Zeiss’s Auriga cross beam SEM. The thermograms of PMMA-RGEO and PMMA-MFRGEO were recorded in an inert nitrogen atomosphere at a heating rate of 10°C min−1 using TG analyzer Model Q50 supplied by Waters (India) Pvt. Ltd. (TA Instruments Division), Bengaluru, Karnataka, India.
Results and discussion
Spectral characterization
The structural characterization of the prepared neat PMMA, PMMA-RGEO nanocomposites, and PMMA-MFRGEO nanocomposites was done with the recorded FTIR spectra presented in Figure 2.
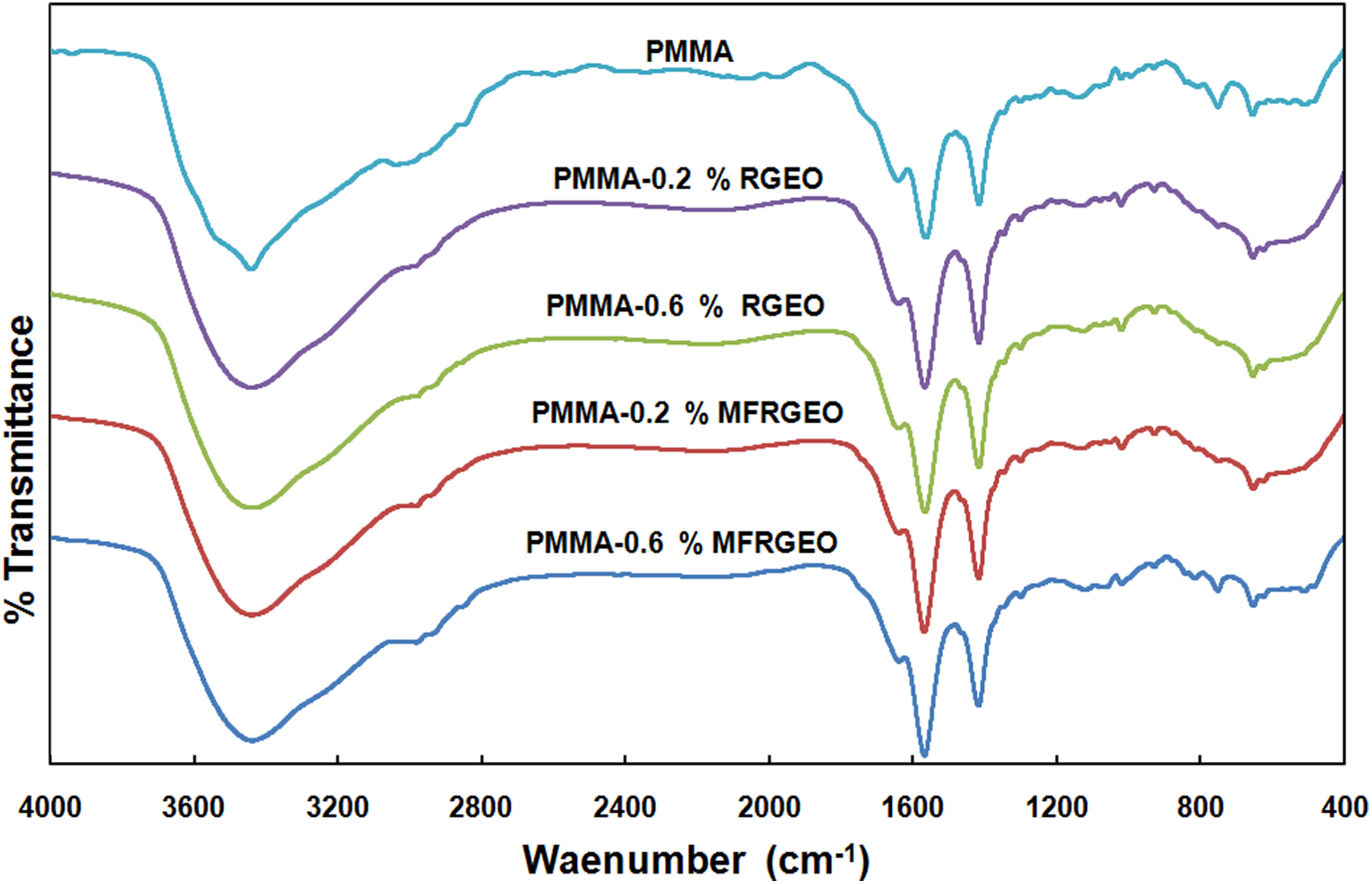
FTIR spectra of neat PMMA, PMMA-RGEO and PMMA-MFRGEO nanocomposites.
The FTIR spectrum of neat PMMA (Figure 2) confirms the functional groups present. The bands at around 3400 cm−1 and 1640 cm−1 are responsible for physisorbed moisture. Distinct absorption bands for C–O–C are seen in the regions 1250–1050 cm−1 (stretching) and 960–880 cm−1 (deformation). A sharp intense peak at 1731 cm−1 is due to the stretching vibration of ester carbonyl group.
24,25
The deformation vibrations associated with –CH3 and –CH2– groups are seen in the wave number region 1450–1395 cm−1. The C–H stretching vibration is seen in the wave number region 3050–2990 cm−1. The bands at 1388 cm−1 and 754 cm−1 can be attributed to
The bands responsible for the functional groups present in the RGEO and MFRGEO fall in the wave number region assigned for functional groups present in the PMMA. 4 The percent loading of nanofiller in the PMMA matrix is less than one, and hence, the presence of RGEO and MFRGEO nanofillers was not clearly detected in the FTIR spectra. 17
Morphological study
The homogeneity in the dispersion of suspended nanoparticles, RGEO and MFRGEO, in the PMMA matrix was studied using SEM imaging. The SEM images of PMMA, PMMA-0.6% RGEO, PMMA-0.2% MFRGEO, and PMMA-0.6% MFRGEO are presented in Figure 3.

SEM images of (a) PMMA, (b) PMMA-0.6 wt% RGEO, (c) PMMA-0.2 wt% MFRGEO, and (d) PMMA-0.6 wt% MFRGEO.
It was observed that PMMA polymer matrix produced a bright contrast, whereas the nanoparticles produced a dark contrast. On comparing the SEM images, one can find the uniform dispersion of nanoparticles in the PMMA matrix. Hazy appearance in the SEM images of PMMA-0.2% MFRGEO (Figure 3(c)) and PMMA-0.6% MFRGEO (Figure 3(d)) is due to the presence of maleimide unit in the RGEO. The absence of bigger aggregation confirms the assistance aided by ultrasonication and maleimide functionalization on RGEO in the uniform dispersion of nanofillers RGEO and MFRGEO in the PMMA matrix. Potts et al. 1,17 reported the assistance aided by sonication in the suspension of graphene oxide in MMA and found that low loadings (0.5 wt%) of nanofiller does not lead to significant agglomeration.
TG study
TG analysis provides information about the char yield and mass loss against temperature rise. The decomposition behavior of polymers during flaming combustion is thought to be anaerobic. Therefore, the study of the decomposition behavior of polymers was performed under inert nitrogen atmosphere.
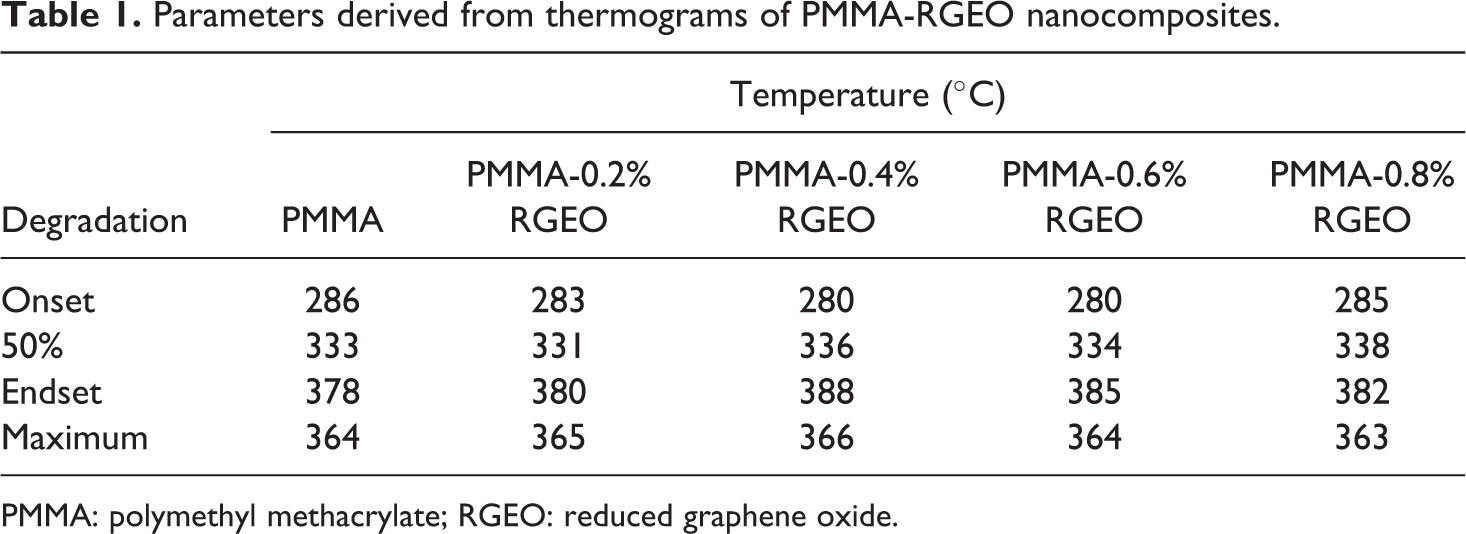
The thermograms of neat PMMA and PMMA-RGEO nanocomposites are shown in Figure 4. The thermogram of neat PMMA showed major weight loss falls in the temperature region 250–375°C with the degradation maximum at 324°C for depolymerization at unsaturated chain ends and 364°C for depolymerization through main chain scission. Irrespective of the loading percent of RGEO, the thermograms of PMMA-RGEO nanocomposites overlap with each other. The parameters derived from the thermograms of PMMA-RGEO are tabulated in Table 1.
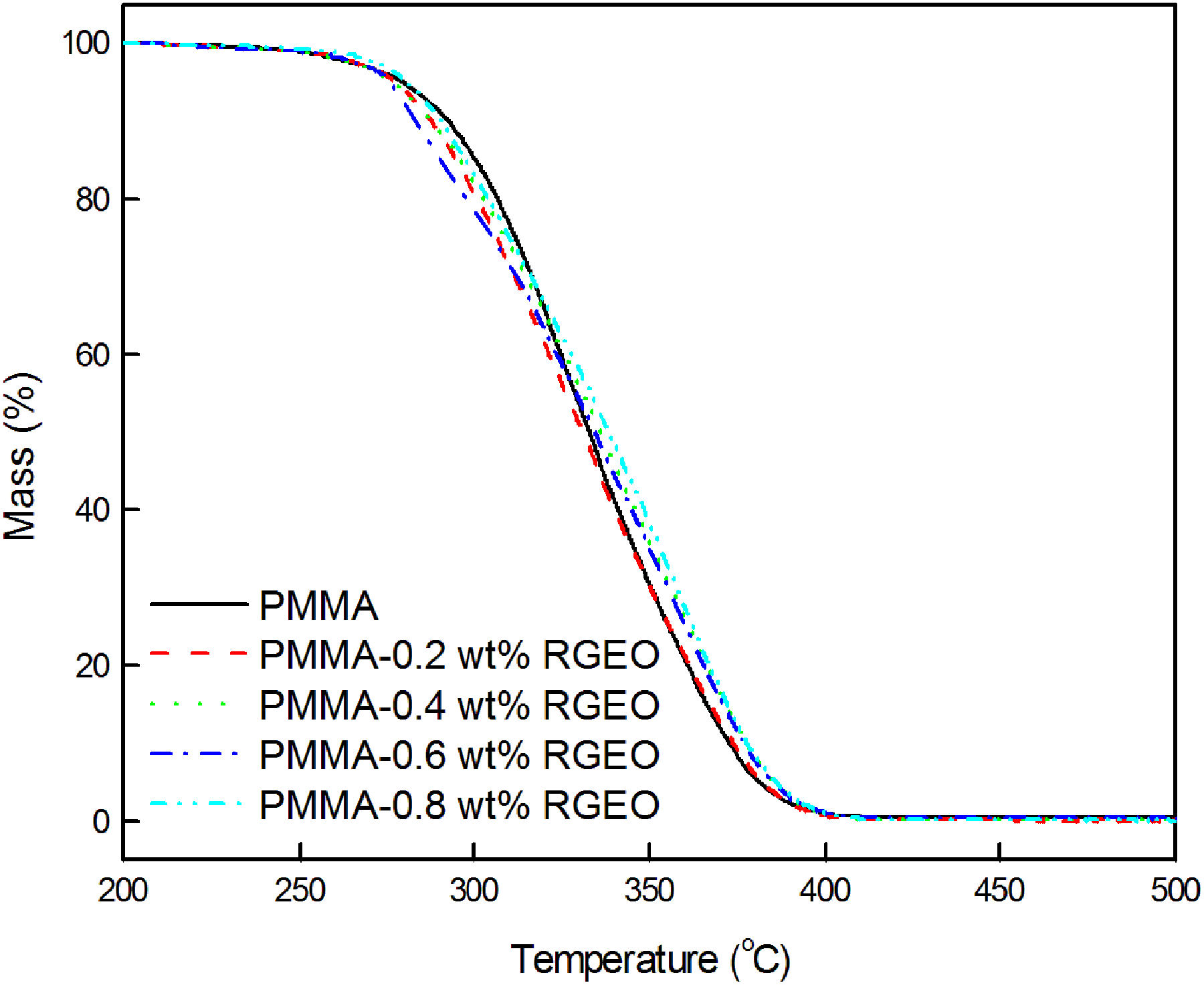
Thermograms of PMMA-RGEO nanocomposites.
Parameters derived from thermograms of PMMA-RGEO nanocomposites.
PMMA: polymethyl methacrylate; RGEO: reduced graphene oxide.
The temperatures responsible for onset, endset, and maximum degradation (
The thermograms presented in Figure 5 showed PMMA-MFRGEO nanocomposites underwent thermal degradation like neat PMMA. The incorporation of MFRGEO to PMMA shifted the degradation as a whole to higher temperature. The parameters derived from the thermograms of PMMA-MFRGEO nanocomposites (Table 2) showed an increase in the degradation temperature of PMMA by loading MFRGEO as nanofiller.
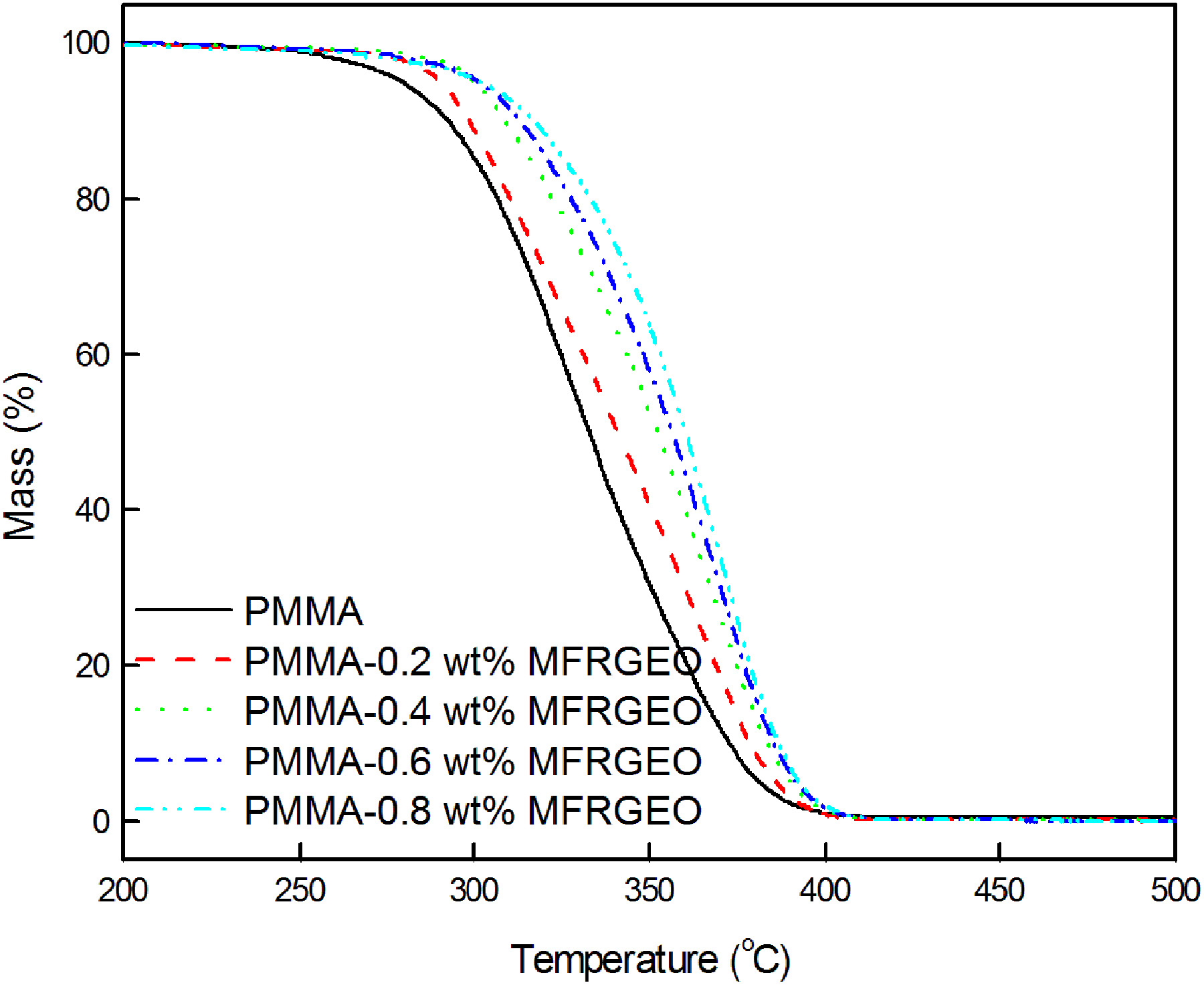
Thermograms of PMMA-MFRGEO nanocomposites.
Parameters derived from thermograms of PMMA-MFRGEO nanocomposites.
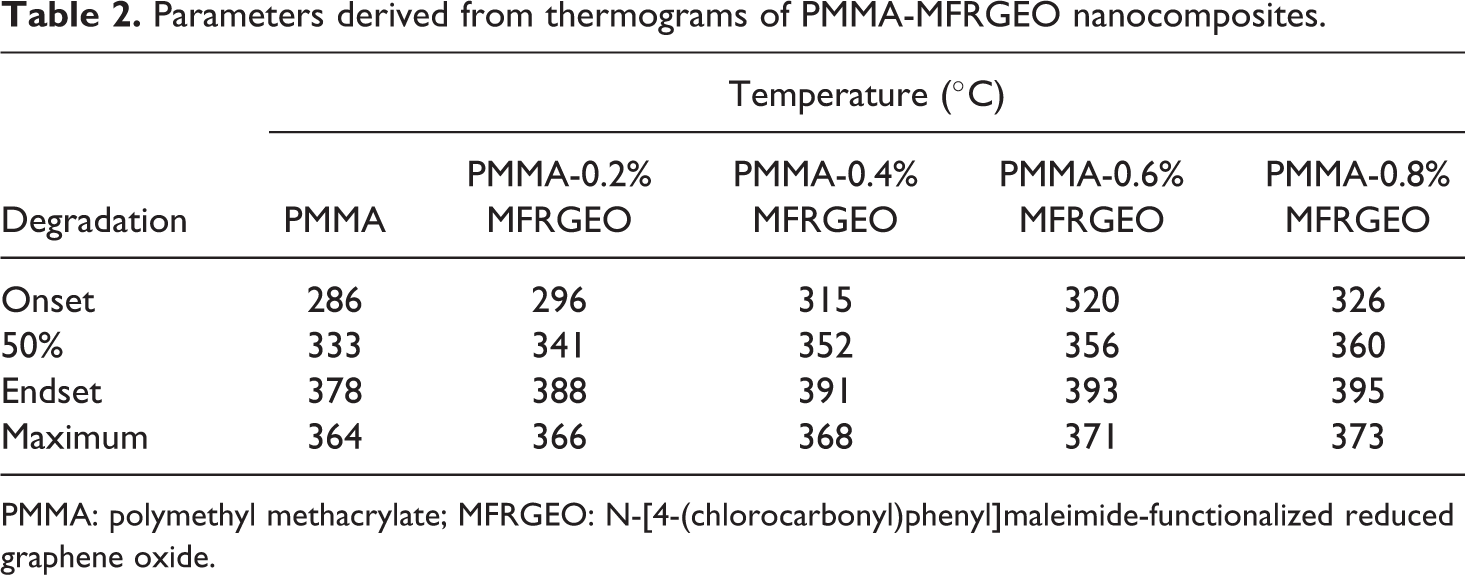
PMMA: polymethyl methacrylate; MFRGEO: N-[4-(chlorocarbonyl)phenyl]maleimide-functionalized reduced graphene oxide.
MFRGEO loading to PMMA increased the onset temperature to an extent of nearly 40°C. The shift in the temperature for 50% weight loss (
Differential TG study
The differential thermograms of PMMA-RGEO (Figure 6) and PMMA-MFRGEO (Figure 7) show degradation in two stages similar to neat PMMA. 26
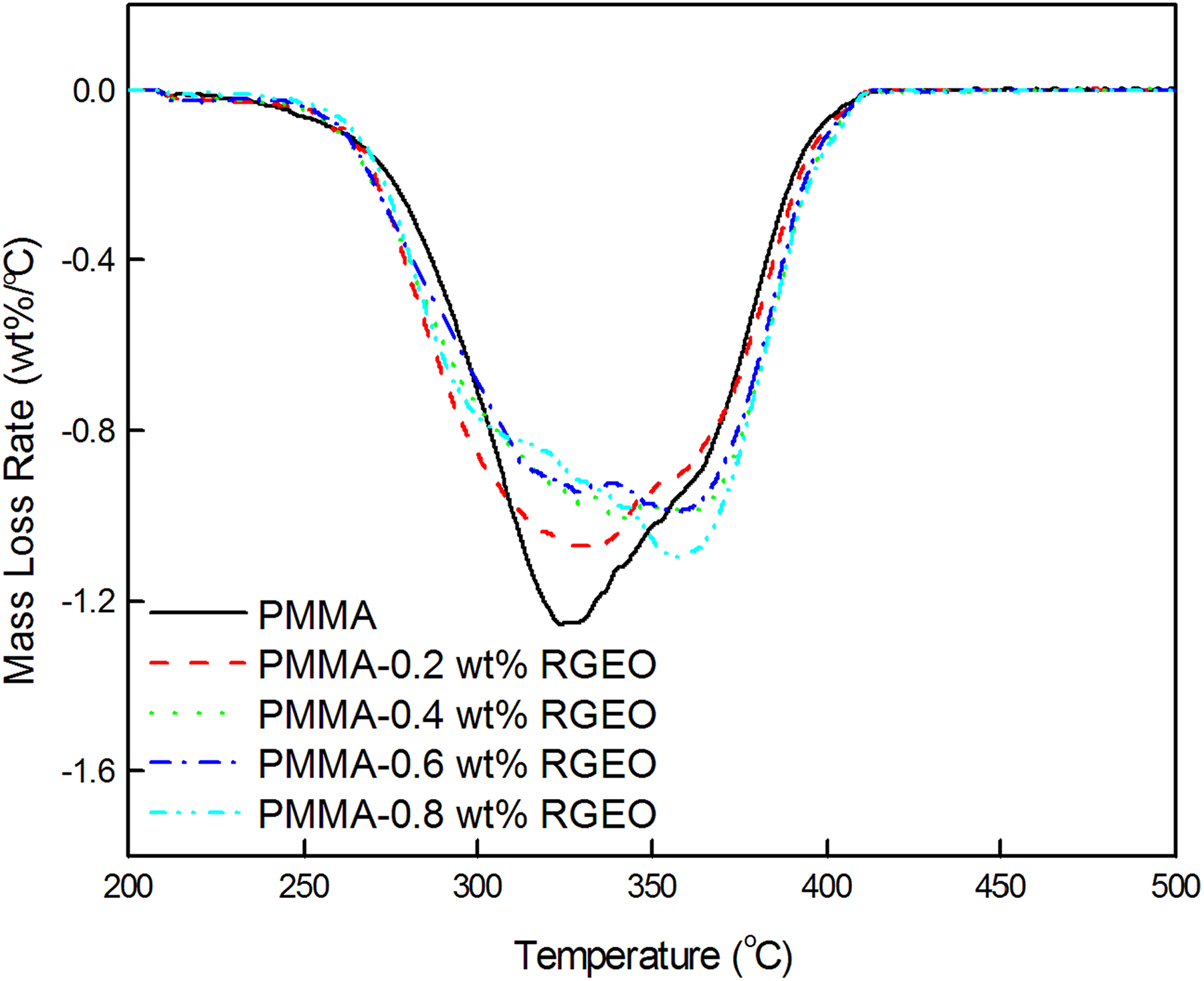
Differential thermograms of PMMA-RGEO nanocomposites.
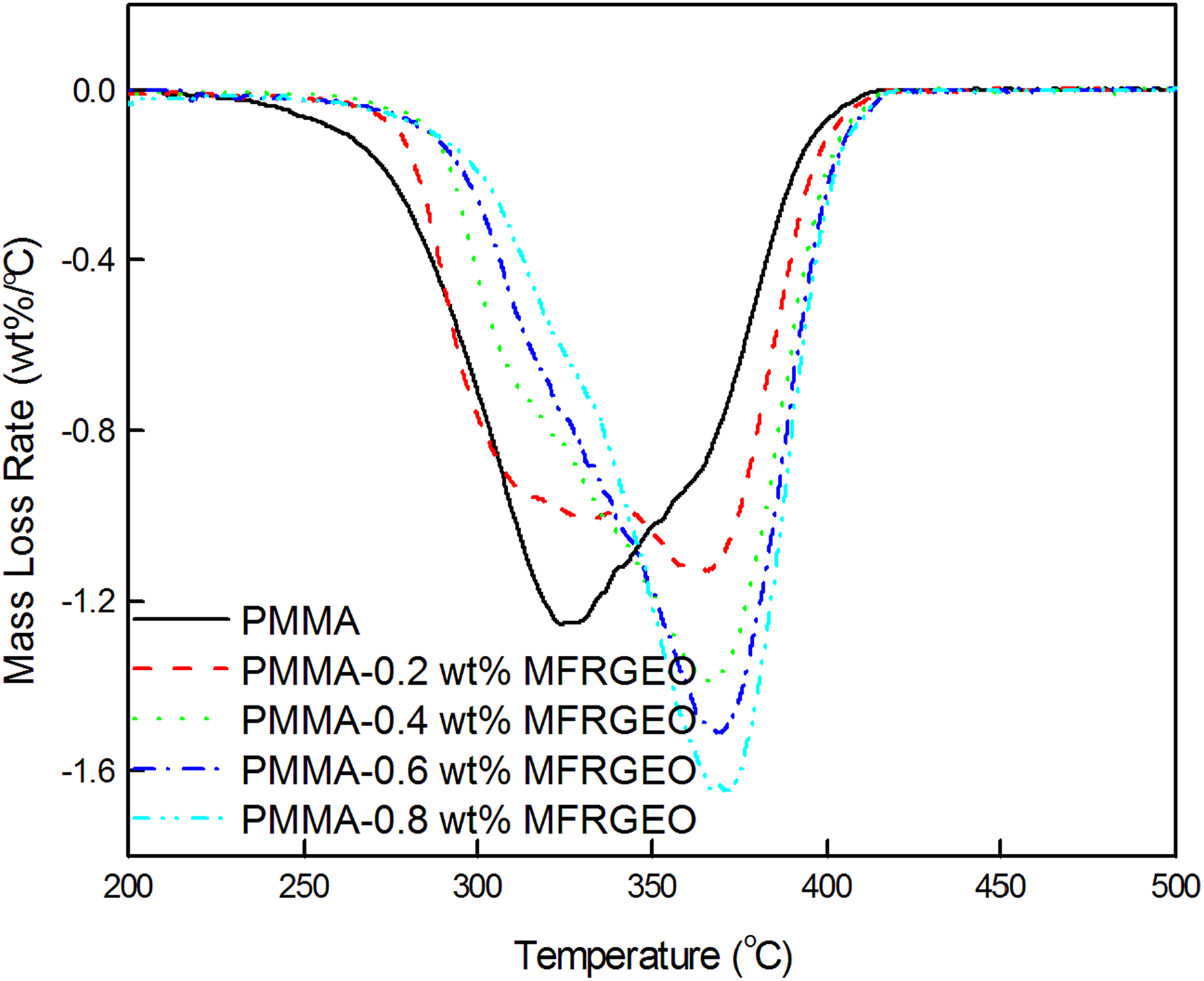
Differential thermograms of PMMA-MFRGEO nanocomposites.
Both the stages are responsible for depolymerization. Irrespective of the loading of RGEO or MFRGEO to PMMA, mass loss rate for the first-stage degradation occurring with
On the basis of TG and DTG studies, it can be concluded that better thermal stability of PMMA can be achieved by loading 0.8 wt% of MFRGEO nanofiller. Unlike graphene-based nanofillers reported for the enhancement of thermal stability of PMMA nanocomposites prepared via in situ polymerization, MFRGEO not only enhanced the onset degradation temperature but also enhanced the temperatures corresponding to degradation maximum, degradation of half of the material, and endset of degradation by delaying depolymerization at unsaturated chain end in PMMA matrix.
Conclusions
The proposed polymer nanocomposites of PMMA with two nanofillers, RGEO and MFRGEO, were separately prepared using free radical in situ bulk polymerization of MMA containing nanofillers and an initiator, AIBN. The FTIR study of PMMA and nanofiller-loaded PMMA revealed no variation in the spectral band positions. SEM imaging technique confirmed the assistance aided by ultrasonic radiation in the prevention of agglomeration of nanofillers in the PMMA matrix. The TG study indicates that PMMA-MFRGEO nanocomposites are having better thermal stability than PMMA-RGEO nanocomposites and neat PMMA. Enhanced thermal stability of PMMA-MFRGEO is attributed to uniform distribution of nanofiller which could dissipate heat uniformly, polymerization of maleimide unit attached to the graphitic plane, and hinderance to depolymerization at unsaturated chain ends. This study also suggests that MFRGEO nanofiller shifted the degradation maxima corresponding to depolymerization of PMMA main chain to higher temperature whereas RGEO nanofiller do not. The extent of thermal stability of PMMA nanocomposite increases with the increase of MFRGEO content. The MFRGEO could be used as a potent nanofiller in several polymer matrix composites for not only enhancing the thermal stability but also imparting flame retardancy.
Footnotes
Acknowledgements
The authors thank the Principal and Head of the Chemistry Department of Rajah Serfoji Government College (Autonomous), Thanjavur, Tamil Nadu, India, for their constant encouragement and providing facilities to carry out this work successfully.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Tamil Nadu State Council for Higher Education in a frame of the research project entitled “Synthesis and Characterization of Functionalized Graphene Oxide-Polymer Nanocomposite” (Project No. 1083/2015A dated September 24, 2016).
