Abstract
This article evaluates the physicochemical and antimicrobial properties when silver-coated silica (SiO2-Ag) nanoparticles as active nanofiller are incorporated into the polypropylene (PP) thermoplastic matrix. The silica (SiO2) nanoparticles were prepared by precipitation method using sodium silicate. After that, silver-coated silica (SiO2-Ag) nanoparticles were synthesized by gamma radiation technique. X-ray diffraction (XRD), Infrared spectroscopy analysis (FTIR) and Transmission electron microscopy (TEM) analysis clarified the formation of SiO2-Ag nanoparticles. SiO2-Ag nanoparticles has a particle size with an average of 70 nm. The melt mixing procedure operated to fabricate PP thermoplastic nanocomposites with various ratios of 1.0, 2.0, and 3.0 part per hundred resin (phr) of the SiO2-Ag nanoparticles. To examine the effect of ionizing radiation on the prepared PP/SiO2-Ag nanocomposites, the samples were exposed to 20 kGy of gamma-irradiation. FTIR, XRD, mechanical analysis, thermogravimetric analysis (TGA), and scanning electron microscope (SEM) were utilized to characterize the physico-chemical alterations of the PP when loaded with SiO2-Ag nanoparticles. It is found that PP/1.0 phr SiO2-Ag nanocomposite revealed superior physico-chemical characteristics than the other two components. The irradiated specimens revealed superior tensile strength (TS) and elastic modulus (EM) over unirradiated ones, whereas inverse effects were predominant in case elongation at break (E%). Tmax of the native PP increased from 335°C to nearly 370°C of PP/1.0 phr SiO2-Ag nanocomposite. It is established that the fabricated PP/SiO2-Ag nanocomposites exhibited potent antimicrobial activity and can be a good candidate for food packaging applications.
Keywords
Introduction
Polymers have attracted great attention due to their unique properties such as high mechanical strength, light-weight, flexibility, corrosion resistance, long-term stability, barrier properties and a good host matrix to be used in various applications. Polypropylene (PP) is a common thermoplastic due to its excellent properties such as lower density, easy processability, respectable virtuous moisture barrier, and its cost-effectiveness. PP is commonly employed in a variety of plastic products in which toughness, flexibility, light weight, and heat resistance are required.1,2
Recently, polymeric nanocomposites (PNCs) have concerned great interest owing to their superior properties over the conventional bulk materials. PNCs produced by adding inorganic nanoparticles as nanofiller into polymeric matrix. Nanofillers materials, small substances with dimensions ranging from one to one hundred nanometers, exhibit unique physical and chemical features due to their small size and high surface to volume ratio that are not present in bulk materials. As a result, low weight percentage of nanomaterials can significantly affect the properties of polymeric matrix, producing lightweight and economical materials. Nanofillers provide outstanding opportunity for designing polymeric materials with advanced functions.3,4
Silica is an oxide of silicon with the chemical formula SiO2, commonly found in nature as quartz. It is in the form of silicic acid polymer with interconnected tetrahedral SiO4 units. 5 Natural and synthetic SiO2 is present as the crystals (quartz, cristobalite, and tridymite) and amorphous form, respectively.6,7 The amorphous SiO2 types are biologically safe materials to be used in the food and drug industry without any clear health problem.8,9 They have a high degree of both thermal and mechanical stability.10,11
Silica nanoparticles (SiO2 NPs) is one of the most promising inorganic nanofillers in polymer nanocomposites due to its properties such as adjustable morphology, ease of functionalization, higher specific surface area, great thermal stability and rigidity. Moreover, the preparation procedures of the SiO2 NPs are cost effective and relatively simple and this qualifies them for use in various fields such as in catalysts, chemical sensors, polymers, coating, ceramics and medical applications. Silica nanoparticles are used for the reinforcement of polymeric materials for developing high performance materials with great industrial importance.12,13 Due to their non-toxicity, versatile applications in the medical and biological systems are defined for them such as it can be used as hosts for the encapsulation of contrast agents in medical imaging and can be utilize as a stabilizer for the aspirin tablets.14–16 On the other hand, additional antibacterial applications for SiO2 NPs can be originated after adsorbing different kinds of ions like silver and zinc. 17 Recently, it has been observed that silver-functionalized SiO2 nanomaterials exhibit antibacterial effect compared to unfunctionalized SiO2 nanostructures due to the distinct bactericidal effect of Ag nanostructures. Recently, Mourad et al added Ag-SiO2 core-shell particles for enhancing both physico-mechanical and antibacterial properties of natural rubber. 18
Recently, the active packaging, especially packaging containing biocidal materials, is considered a solution for the main problems that occur due to the microbiological contamination in foods and which have implications for public health due to diseases occurred by contaminated foods. In this context, the primary research challenge involves searching for effective ways to prevent the growth of microbes in food. Packaging aims to preserve food quality, reduce food waste in light of global crises, and maximize economic returns. Advances in polymeric nanocomposites have made a significant contribution to the food packaging materials by introducing new materials with improved physical, chemical and biological properties.19–21
The radiation-induced synthesis of nanoparticles is considered as one of the most promising and cost-effective methods. The irradiation process takes place at ambient temperature without producing toxic reductant byproducts or using excessive reducing agents, so it is a more convenient and clean technique where the other chemical and physical techniques require toxic chemicals, high energy and are expensive.22–25
Moreover, radiation technology offers many advantages to polymers and gives them new chemical and physical properties. Ionizing radiation leads to cross-linking between polymeric chains with a sterilization effect in the same time.26–29
The present work aims to synthesize SiO2-Ag nanoparticles by gamma radiation technique as a more convenient and clean technique and using it as active nanofiller for the PP thermoplastic matrix to be a good candidate for packaging applications. XRD, TEM, and FTIR investigated the SiO2-Ag nanoparticles. The mechanical, thermal, and antimicrobial properties of the prepared PP/SiO2-Ag nanocomposites were studied. The physicochemical alterations of PP thermoplastic nanocomposite were investigated for gamma-irradiated species at an ambient dose, of 20 kGy.
Experimental
Materials
Polypropylene (PP) pellets was provided from Sidi Kerir Petrochemical Co. (SIDPEC), Egypt with melt flow index (MFI) was 2.3 g/10 min (at 230°C, 2.16 kg), density 905 kg/m3 and crystallinity of 93.1%. Commercial liquid Sodium silicate (Na2O 11.7 wt%, SiO2 32.8 wt% and H2O 55.5 wt%) was produced by Silica Egypt Company, Burg Al-Arab, Alexandria, Egypt. Concentrated hydrochloric acid solution (HCl, 33%, density 1.18 g/mL) and sodium dodecyl sulphate (SDS) were provided from El-Nasr Pharmaceutical Chemicals Co., Egypt. Silver nitrate (AgNO3, molar mass 169.87 g/mol, assay 99.9 %) was purchased from Merck (Darmstadt, Germany). Bidistalled water was used throughout the experiment.
Preparation of SiO2NPs
The silica nanoparticles were prepared by precipitation method using sodium silicate as a source of silicon.30–32 Firstly, 100 mL sodium silicate was added to 200 mL magnetically stirred bidistilled water for 2 h at room temperature until complete homogeneity. Then, 1 M HCl was added dropwisely to sodium silicate solution with constant stirring at room temperature until achieving neutral pH. The formed gel white powder was recovered by filtration followed by washing with hot bidistilled water in order to be free from chloride ions using dilute AgNO3 solution as tested experiment and finally dried at a temperature of 150°C for about 4 h. The obtained white silica particles were ground before use.
Preparation of SiO2-Ag nanoparticles
The SiO2-Ag nanoparticles were synthesized by the radiation method. In a typical procedure, 15.0 g of SiO2 nanoparticles was dispersed in 300 mL deionized water containing 1% SDS and vigorously stirred for 2 h. Then, 0.1 M AgNO3 was added to the SiO2 aqueous suspension and the mixture was further stirred for 2 h. After that, 30 mL of isopropanol as a hydroxyl radical scavenger were added to this dispersed solution and finally irradiated with 25 kGy gamma radiation using 60Co radiation facility at a constant dose rate (0.8 kGy/h) at room temperature. 33 The radiation process occurs in the National Centre for Radiation Research and Technology (NCRRT), Egyptian Atomic Energy Authority (EAEA), Egypt. SiO2-Ag nanoparticles were separated by centrifugation at 6000 r/min and washed three times with water and ethanol, and then dried at room temperature for 2 days.
Fabrication of PP/SiO2-Ag nanocomposites
Nanocomposites were formed by the melt blending process via a Brabender Plasticcorder mixer apparatus (PlasticorderPL2100, Germany). Firstly, for melting PP pellets, it injected in the hot mixer at a temperature of 175°C–180°C for 5 min. After that, the various percentages of SiO2-Ag nanoparticles (1.0, 2.0, and 3.0 part per hundered resin (phr)) were added into the molten PP and the mixing procedure was continued for a further 5 min at the same temperature to achieve complete homogeneous mixing. Finally, the as-prepared PP/SiO2-Ag nanocomposites were quickly taken from the twin screw of the hot mixer and then withdrawn between the open two-roll mills for shaping and obtaining suitable sheets that are easy to press. Afterward, the samples were hot pressed at about 175°C below 10 MPa for 5 min into sheets of appropriate thickness and size for investigation. The fabricated nanocomposites were exposed to 20 kGy of gamma irradiation to study the effect of ionizing radiation on the fabricated specimens.
1
A schematic representation of the SiO2-Ag nanoparticles and PP/SiO2-Ag nanocomposites fabrication is given in Figure 1. Schematic illustration of SiO2-Ag nanoparticles and PP/SiO2-Ag nanocomposites fabrication.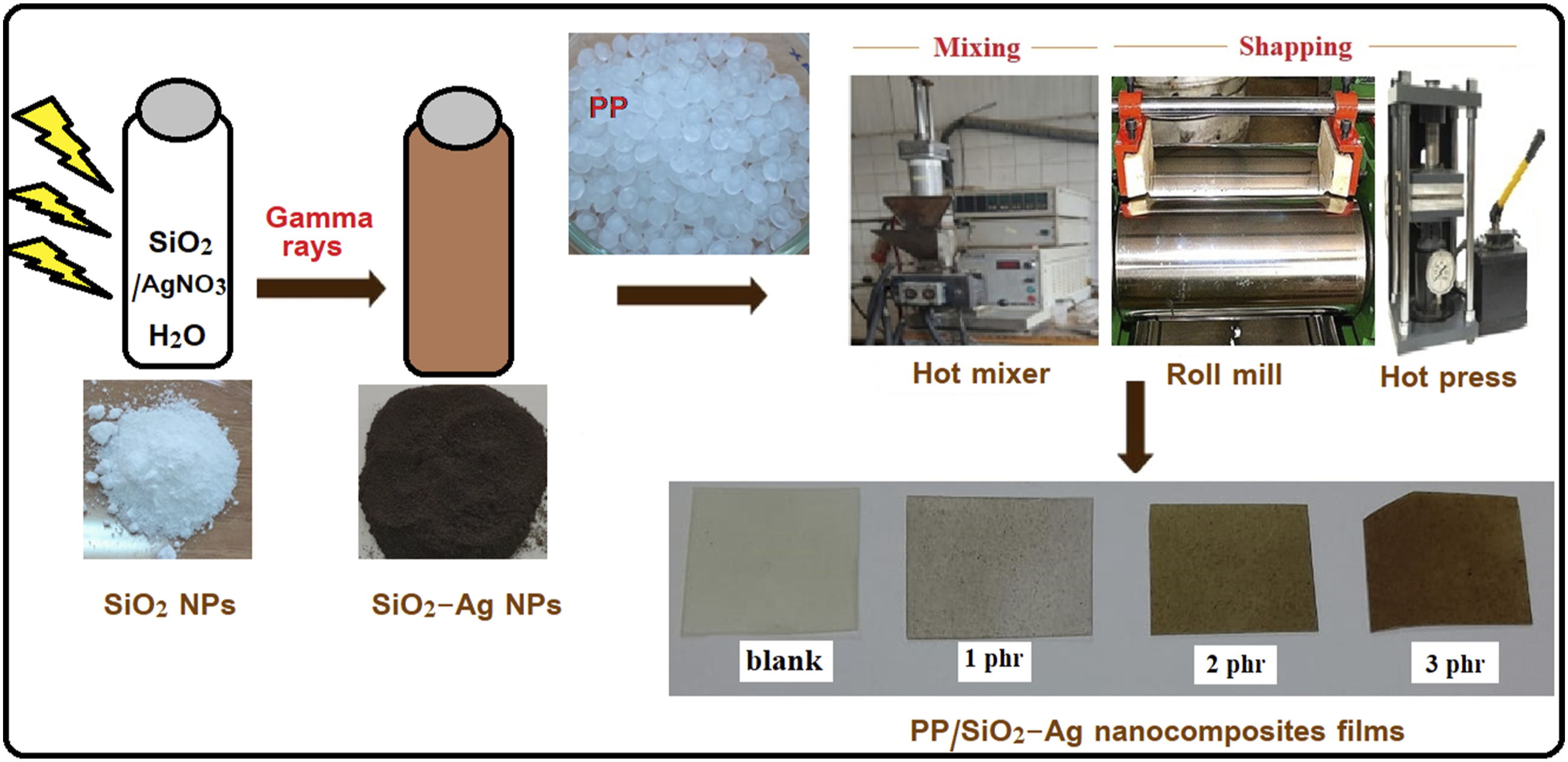
Characterization
The X-ray diffraction analysis the synthesized samples was tested using a Shimadzu 6000 diffractometer standard with Cu-kα radiation. The wavelength of the X-rays was 1.54 A. the infrared spectra was taken within the frequency range from 500 cm−1 to 4000 cm−1 using Attenuated total reflectance-Fourier transform infrared (ATR-FTIR) spectroscopy (Vertex70, Bruker Optik GmbH, Ettlingen, Germany). Both size and shape of the synthesized nanoparticles was obtained by Transmission electron microscopy (TEM) (a JEOL JSM-100 CX model instrument worked at 80 kV accelerating voltage). The surface morphology of pure PP and nanocomposites were obtained using a ZEISS EVO-15 scanning microscope operated at an acceleration voltage of 30 kV. For this purpose fractured surfaces were coated with a thin layer of gold to avoid charging under the electron beam.
Mechanical measurements
The mechanical analysis of PP/SiO2-Ag nanocomposite film was obtained for a dumbbell-shaped examination sections at a crosshead speed of 300 mm/min at 25 ± 2°C using a tensile testing machine Qchida computerized testing instrument, Dongguan Haida Equipment Co., Ltd. China. The ISO 527-2 was monitored. The average value of the mechanical factors was taken via at least three testers.
Antimicrobial activity assays
The antimicrobial activity of the synthesized SiO2, SiO2-Ag nanoparticles, PP control and PP/SiO2-Ag nanocomposite films were determined by using the disk diffusion method against (Staphylococcus aureus ATCC 25923 and Staphylococcus epidermidis ATTC 12228) as model of Gram-positive bacteria and Gram-negative bacteria like (Escherichia coli ATCC11229, Klebsiella pneumoniae ATCC13883, Pseudomonas aeruginosa ATCC6538). Additionally, antifungal activity was tested towards unicellular fungi as (Candida albicans ATCC 90028 and Candida tropicalis ATCC 1456). The bacterial inoculums were adjusted at 0.5 McFarland (1-2) × 108 CFU/ml, using UV–Vis spectrophotometer at 600 nm. Furthermore, Candida inoculums were adjusted at (1-4) × 106 CFU/ml, Clindamycin and Nystatin used for as positive control.
34
Plates were incubated at 37°C for 18-24 h, the antimicrobial activity was evaluated by measuring the inhibition zones (clear areas around the disk) against the test microorganism’s conventional antibiotic disks (6.0 mm diameter) like Clindamycin (DA;2 μg/mL) and Nystatin (NS;100 μg/mL)35,36 using the following equation.
37
Result and discussion
Characterization of SiO2-Ag nanoparticles
X-ray diffraction analysis
For gaining information aboutthe nanomaterials’ crystal structure and crystal lattice, XRD analysis was performed. The XRD spectrum of the SiO2 (Figure 2) showed distinctive strong and wide peak at 22° indicates the amorphous structure of SiO2.
38
On the other hand, the XRD pattern of SiO2-Ag nanoparticles demonstrated the same peak of the SiO2 with a lower intensity and other new four peaks at 38.1°, 44.5°, 64.7° and 77.5° which were indexed to (111), (200), (220) and (311) crystal planes, respectively, related to silver with a face-centered cubic (FCC) phase structure (JCPDS file number 4-0783).23,39,40 The crystallite size of silver NPs (D) was calculated based on the main plane of (111) using Scherer formula (D = kλ/βcosθ), where k is the shape or geometry factor which equals 0.9, λ is the X-ray wavelength (λ = 0.1541 nm), β is the full width at half maximum (FWHM) of diffraction peak and θ is the diffraction angle.) Using the FWHM of the strong and sharp diffraction peak (111) it was found to be approximately 27 nm. XRD patterns of SiO2 nanoparticles and SiO2-Ag nanoparticles.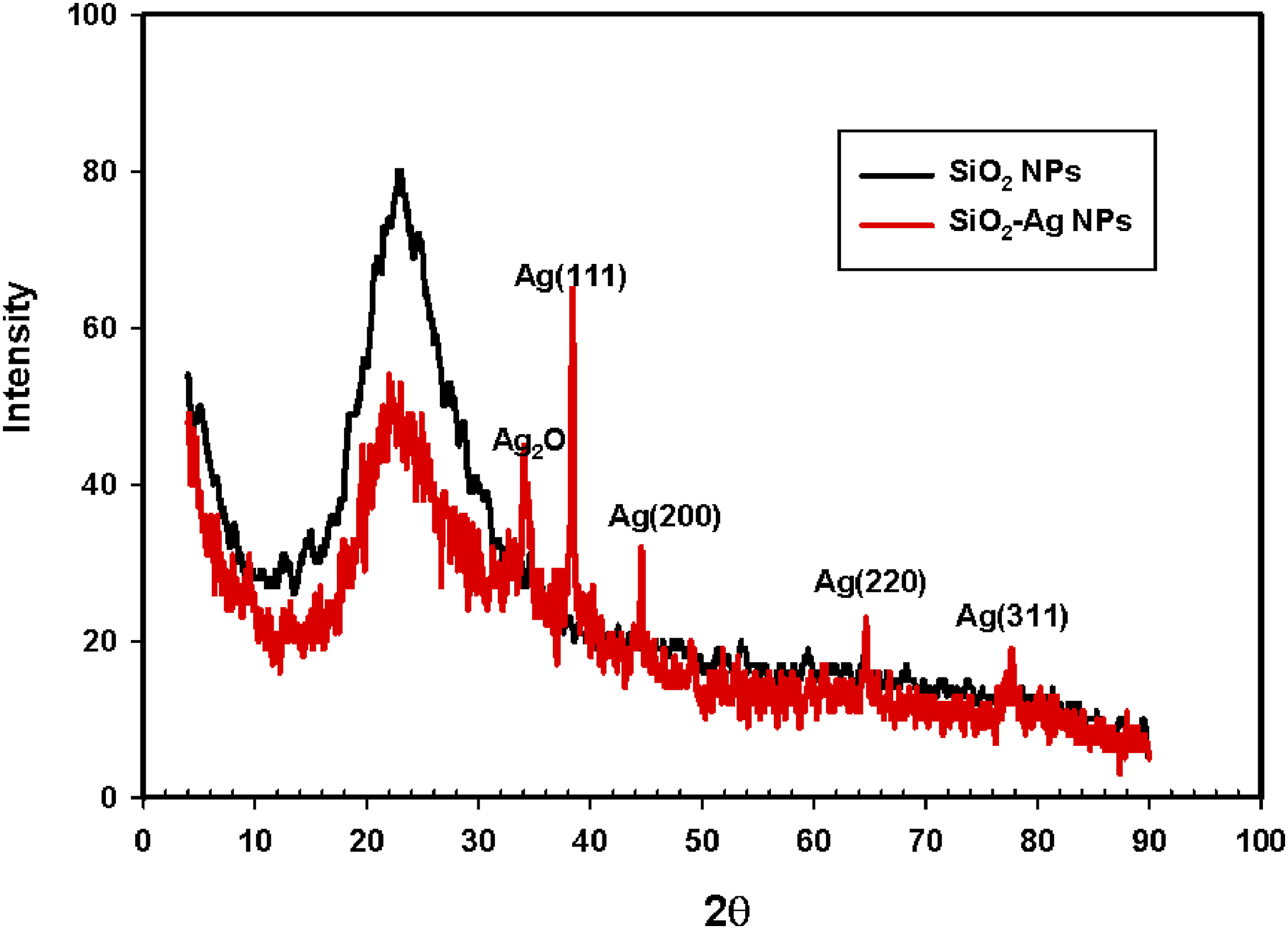
FTIR investigation
The FT-IR spectra for SiO2 nanoparticles and SiO2-Ag nanoparticles are represented in Figure 3. The characteristic peaks of SiO2 were observed at 1056, 790 and 445 cm−1 which coincide with Tran et al.
41
and Hassan et al.
42
The peak at 1056 cm−1 was attributed to the asymmetric stretching vibration of siloxane bond (Si–O–Si). The peak at 790 cm−1 represented the symmetric stretching vibration of Si–O–Si bond. The peak at 445 cm−1 corresponded to the Si–O–Si bending (SiO4) tetrahedral. Furthermore, the shoulder at 955 cm−1 is due to the bending vibration mode of the silanol group (Si–OH). The weak broad peak between 3000 and 4000 cm−1 is characteristic to the stretching vibration of hydroxyl (OH) group of silanol group and adsorbed water. The peak at 1636 cm−1 belonged to H–O–H bending.31,43,44 The weak peak at 618 cm−1 is assigned to ring structure.
42
On the other hand, the spectra of SiO2-Ag nanoparticles exhibit the same features of SiO2 nanoparticles but the intensity of the peaks was slightly changed due to the Ag anchored into the SiO2 nanoparticles. FTIR of SiO2 nanoparticles and SiO2-Ag nanoparticles.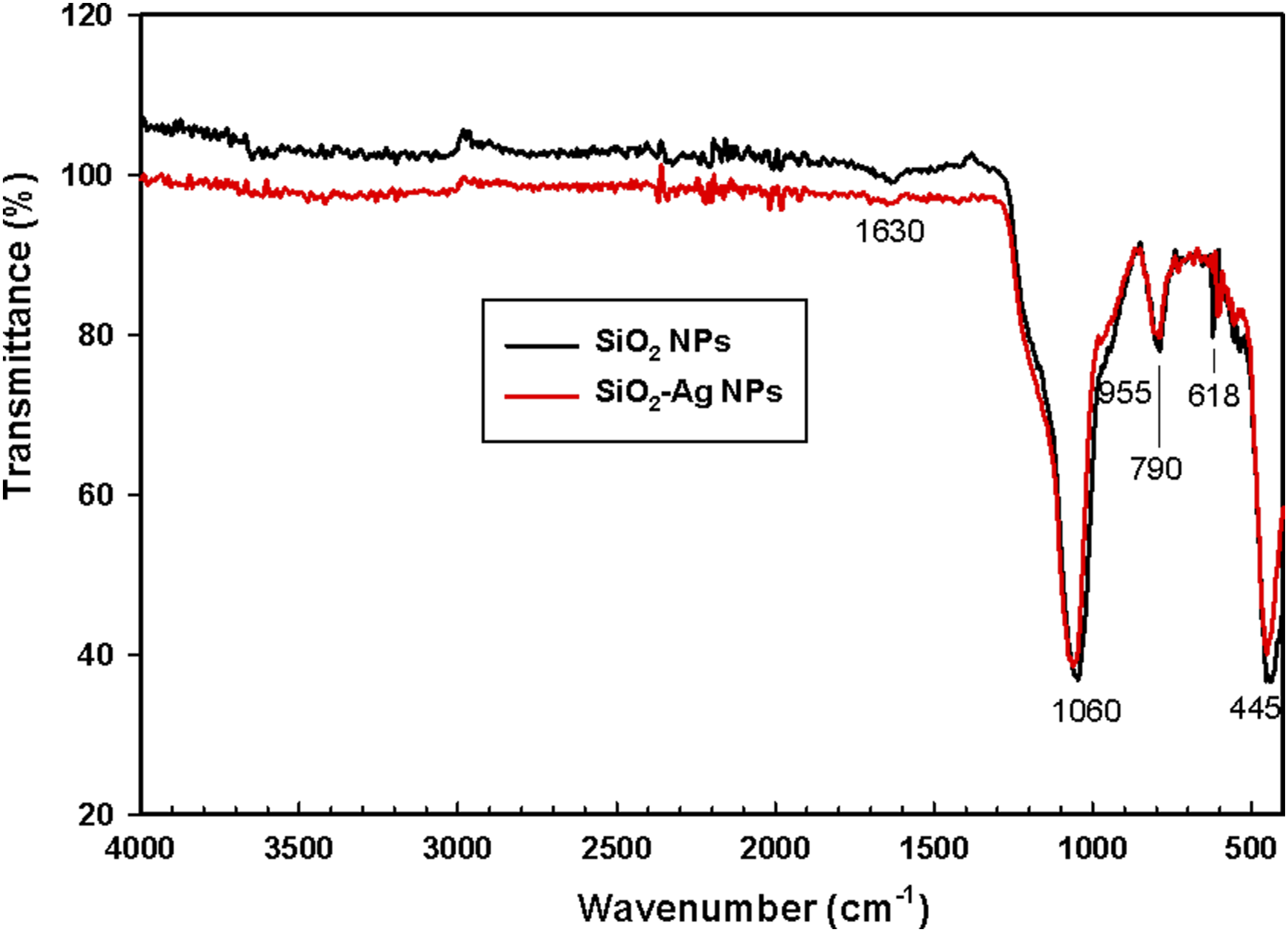
TEM analysis
The morphology and size of SiO2 nanoparticles and SiO2-Ag nanoparticles are shown in Figure 4. It is observed that the resulting SiO2 nanoparticles (Figure 4(A)) are monodispersed and appear as spherical shape withan average diameter nearly 30 nm. On the other hand, it is clear from SiO2-Ag nanoparticles images (Figure 4(B) and (C)) that the silver nanoparticles were deposit on the surface of SiO2 nanoparticles, which means that these grains contain the two component materials and thus the silver/silica bi-structure were formed. The SiO2-Ag nanoparticles with an average particle size of 70 nm. TEM images of: (A) SiO2 nanoparticles and (B-C) SiO2-Ag nanoparticles with different magnification.
Characterization of PP/SiO2-Ag nanocomposites
Infrared spectroscopy analysis
The characteristic absorption bands of the native PP in Figure 5(A) showed a distinct peak at 2933 cm-l that was credited to the stretching vibration of –CH groups. Whereas, the absorption vibration peaks of symmetrical and asymmetrical –CH appeared at 1445 and 1370 cm−1.1,45 FTIR of (A) native PP, (B) PP/1phr SiO2-Ag and (C) irradiated PP/1phr SiO2-Ag nanocomposites.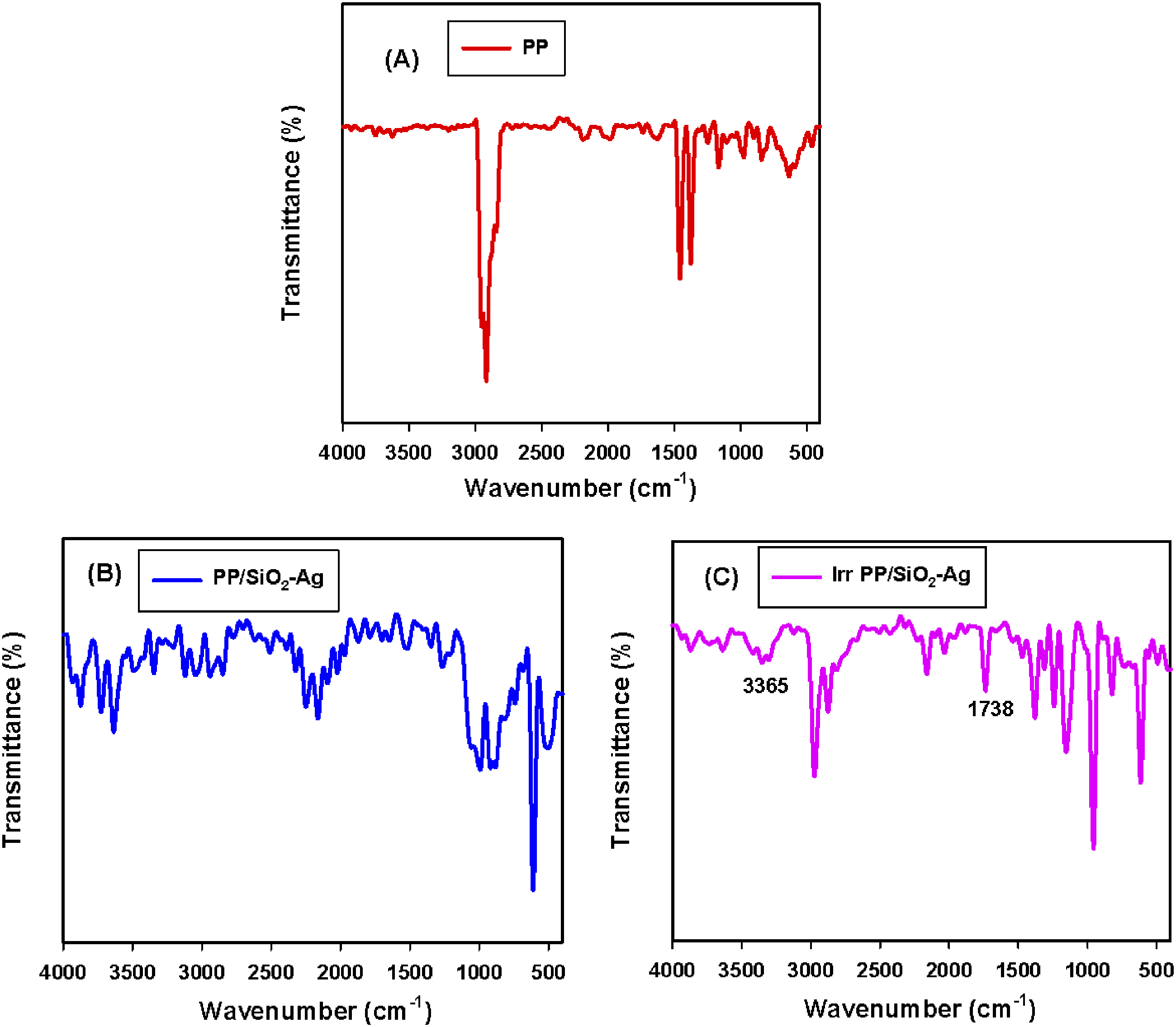
After the interference of SiO2-Ag nanoparticles into the PP skeleton, PP/SiO2-Ag nanocomposites were recognized by FTIR spectra as shown in Figure 5(B). As revealed the Si–O–Si absorbance of stretching vibration is detected at about 1098 cm−1 for the filled species,46,47 approving the presence of silica in the PP matrix. 48 Furthermore, the appearance of the peak around 465 cm−1 specified the bending types of SiO2,38,49 and also all the peaks assigned for C-CH3 stretching of PP changed in their nature after PP adsorption onto SiO2-Ag nanoparticles. The major bands after SiO2-Ag intrusion into PP appeared at 3467-2871 cm−1, the presence of the bands within the range of 2853-2934 cm−1 which are assigned to C−H stretching vibration type of long-chain carbon on SiO2-Ag nanoparticles‒SDS surfactant fragments. 50 After irradiation, as clear from FTIR in Figure 5(C), the bands within range 2853-2934 which specified C‒H stretching were changed in nature when compared to their counterparts in Figure 5(B). The induced crosslinking into the polymer matrix and the promoted adhesion between SiO2-Ag nanoparticles and PP may be improved due to irradiation. Also, after irradiation, both absorption bands of hydroxyl groups (OH) at 3365 cm−1 and carbonyl group (C = O) at 1738 cm−1 appeared in PP nanocomposite. This result is qualified to the occurrence of oxidative phenomena during irradiation due to the presence of oxygen surrounding in gamma irradiation cell and occurrence of some chain scission.51,52
Mechanical measurements
The mechanical investigation is a suitable procedure to assess the structural alterations of polymeric materials owing to filler intrusion and radiation effects. Tensile strength (TS) is the maximum load a specimen can support without breakage but being stretched. Dividing the stress over strain is identified as modulus of elasticity (EM), whereas elongation at break (E %) defines the resistance of cracking of the samples upon elongating and altering in shape. Stress-strain curves and the different mechanical results, including tensile strength, elongation at break, and elastic modulus, of PP and its composites with nano SiO2-Ag, were investigated and displayed in Figures 6 and 7, respectively. The mechanical studies of unirradiated and irradiated specimens revealed superior mechanical values for the PP/1.0 phr SiO2-Ag nanocomposite compared to the others. This result is attributed to strong interfacial interaction and a good dispersion of 1.0 phr SiO2-Ag nanocomposite.
53
Castell et al
51
found that 20 KGy gamma-irradiation promote crosslinking in the PP matrix and improve the adhesion of the nanofiller and the mechanical properties of the nanocomposites. Stress-Strain curve of (A) unirradiated (B) irradiated PP and PP/SiO2-Ag nanocomposites exposed to 20 kGy. (A) Tensile strength, (B) Elongation at break, (C) Elastic modulus of PP and PP/SiO2-Ag nanocomposites exposed to 20 kGy.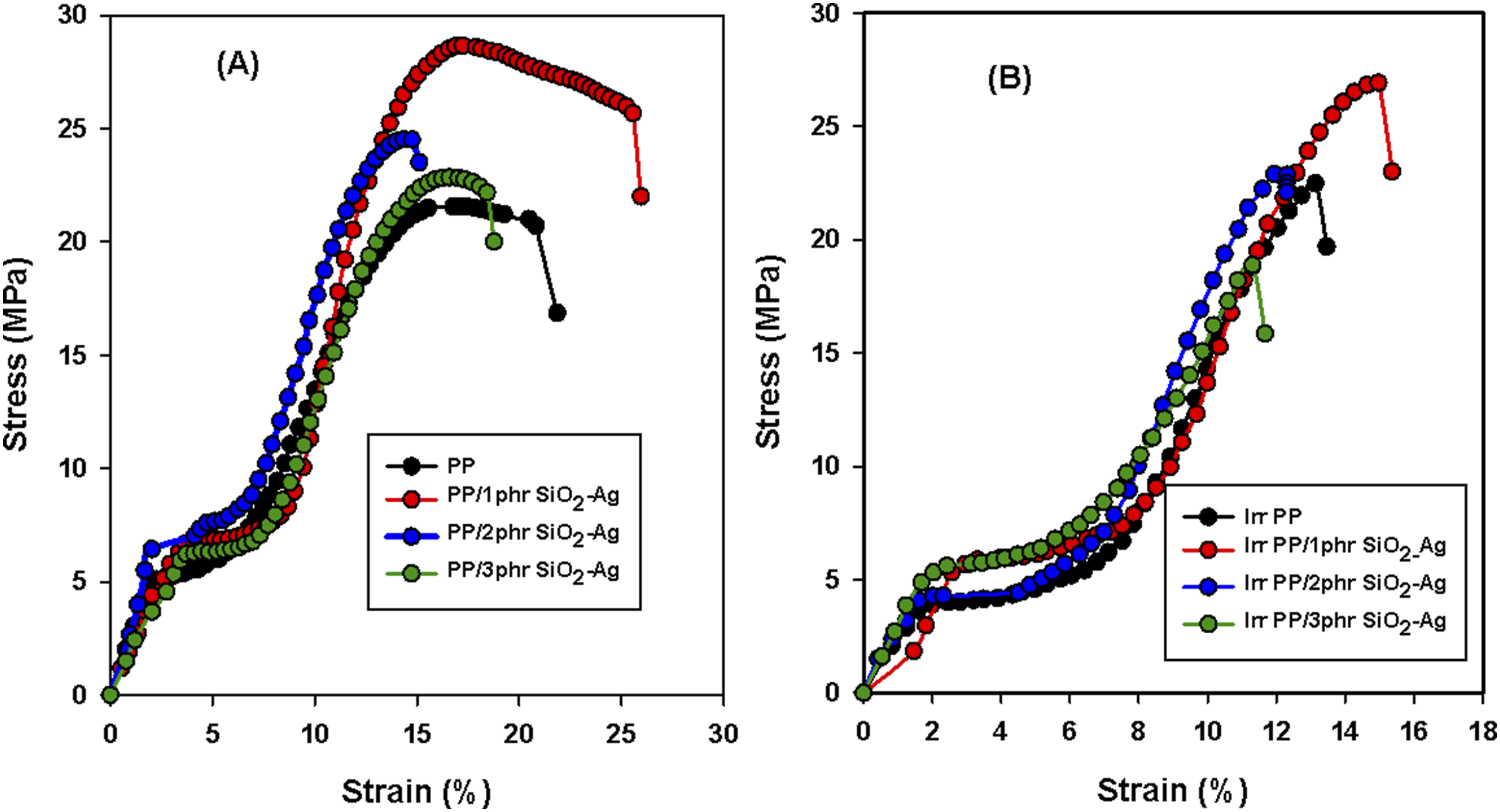
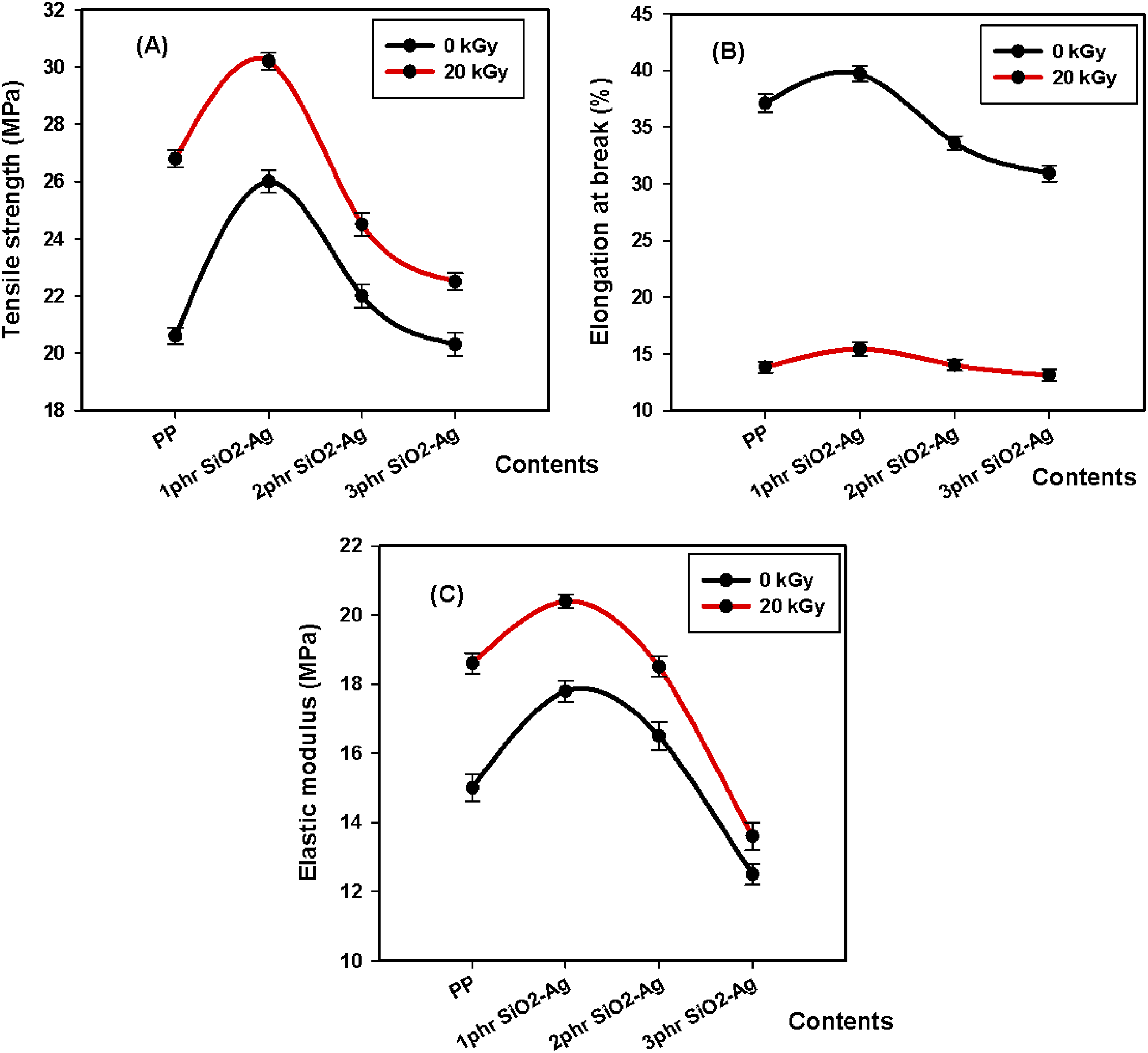
Tumnantong et al 54 studied the effect of SiO2 nanocomposites on the mechanical properties of natural rubber (NR) with concentration equivalent to 0%, 1%, 1.5%, 2% and 2.5% silica. They found that with the addition of low concentration of (1%–1.5%), the tensile strength of nanocomposites was enhanced by 39.1% and 15.6% from the unfilled NR (19.2 MPa) where silica provide a reinforcing effect on the NR at low concentration due to the homogeneous SiO2 distribution. With the addition of SiO2 at 2%–2.5%, the tensile strength of NR nanocomposites was decreased by 12.0% and 17.7%. This behavior was attributed to the high loading that leads to small aggregates and a lower the filler-rubber interaction. Zolfaghari et al 55 reported that the PP samples exhibited an improvement in the tensile strength when reinforced with only 1.0% silica. They suggested that the thin walls of the cavities in the silica particles resulted in fast degradation under tensile loads. The reduction in TS for higher silica fillings would be because of the agglomeration for the utmost silica contents and the subsequent stress concentration. These results agree with our study, therefore, both the strength and the elasticity of the PP thermoplastic matrix have been improved with SiO2-Ag filling, suggesting a grander interface and blending of the nanoparticles into the PP texture at 1phr. Also, it is shown below in XRD section that 1phr SiO2-Ag act as a nucleation for β crystal form of PP. The presence of the β-phase within the crystalline portion of the material enhances the mechanical properties. 56 Over 1phr of SiO2-Ag loading into the PP texture decreased all mechanical parameters. This means that the agglomeration was brought into the PP nanocomposite matrix at 2 and 3phr of the SiO2-Ag interface. The same behavior is clear for irradiated samples at 20 kGy.
At the same time, the irradiated specimens revealed superior tensile strength and elastic modulus over unirradiated samples due to induced crosslinking produced by irradiation. 51 Inverse effects were predominant in case elongation at break studies due to the restriction impact. 57 Bonds between the thermoplastic and the SiO2-Ag nanoparticles caused the reduction of the polymeric chain mobility of the PP and a decline in the elongation at break %.
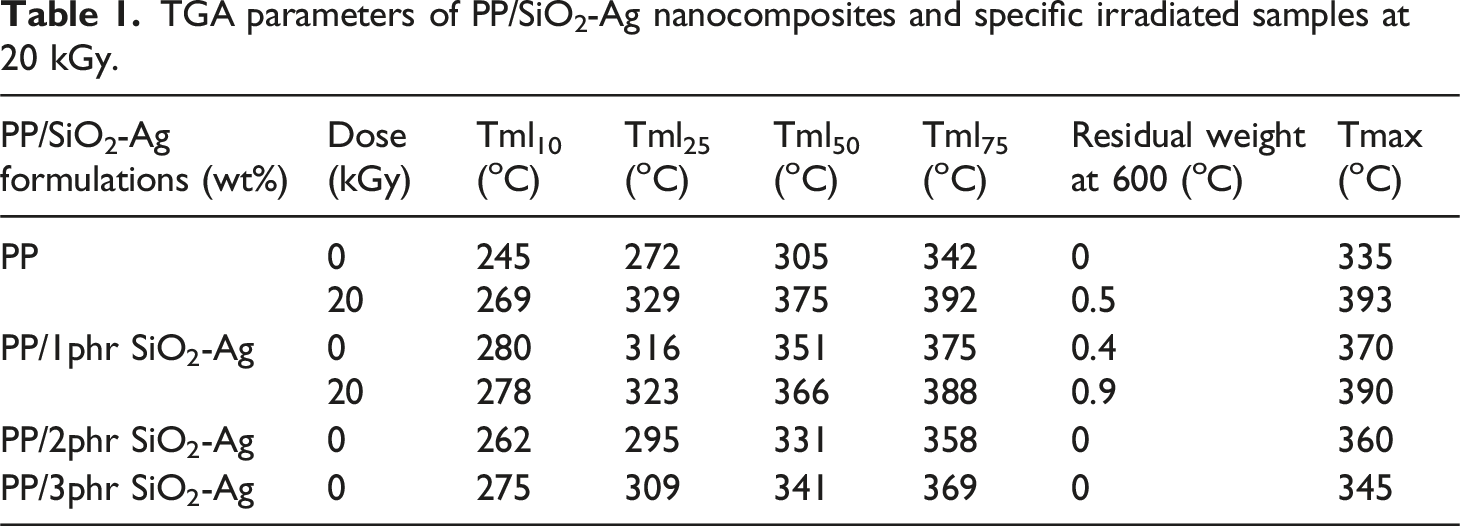
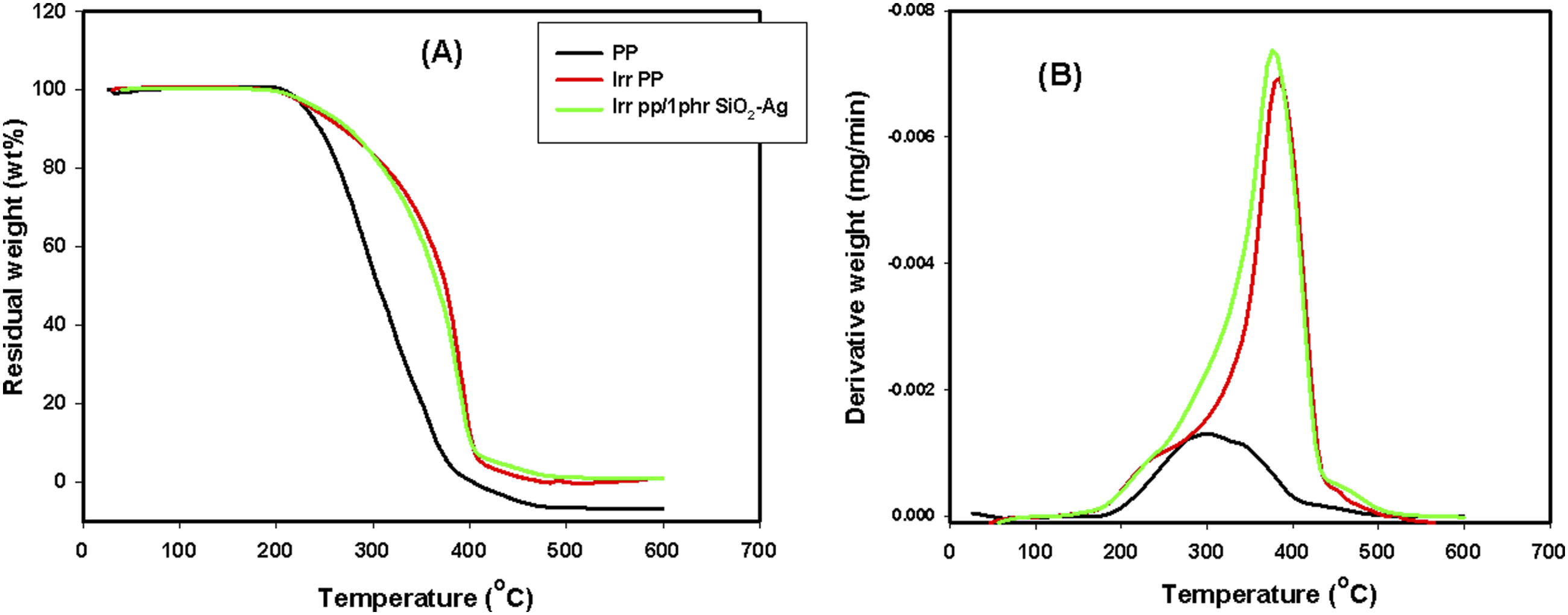
Thermogravimetric analysis
In a nitrogen atmosphere, Figure 8(A) and (B) respectively displays the thermogravimetric analysis (TGA) and the derivative of the mass loss (DTG) as a function of temperature of the PP/SiO2-Ag nanocomposites. The numerical data of TGA and DTG from room temperature up to 600°C are presented in Table 1. It is clearly seen that the thermal stability of the native PP significantly increases with the content of SiO2-Ag in the matrix and the percent of the nanoparticles at 1 phr was the best. Where, all the stages of the temperature mass loss (Tml10, Tm25, Tm50, and Tm75) of the pure PP considerably increase. For example, the Tm50 of the native PP increased from 305°C to 351°C of PP/1phr SiO2-Ag nanocomposite. The enhancement of the thermal stability of the native PP when loaded with 1 phr of SiO2-Ag can be qualified for the improved dispersion of the nanoparticles between the PP chains.
58
While the increase in the thermal stability of PP when loaded with 2 and 3 phr of SiO2-Ag nanoparticles could be credited to the interface between the SiO2-Ag nanoparticles and the matrix that hindered the segmental movement of the thermoplastic molecules. In the same manner, the Tmax of the native PP increased from 335°C to nearly 370°C of PP/1 phr SiO2-Ag nanocomposite.
59
TGA and DTG of the irradiated samples represented in Figure 9(A) and (B), to study the influence of irradiation dose at 20 kGy on the thermal stability of the fabricated species, we select the native PP and also PP/1 phr SiO2-Ag nanocomposite as the best component have thermal stability. Tmax of the pure PP and PP/SiO2-Ag were improved with irradiation dose. In which, the three-dimensional space structure formed in the PP and PP/SiO2-Ag nanocomposites caused by irradiation, blocks efficiently the movement of the heat transfer and origins a reduction in the dispersion of the degraded product of the polymer matrix to the gas phase that successfully delays the nanocomposites thermal decomposition.
1
(A) TGA and (B) DTG of PP, PP/1phr SiO2-Ag, PP/2phr SiO2-Ag and PP/3phr SiO2-Ag nanocomposites. TGA parameters of PP/SiO2-Ag nanocomposites and specific irradiated samples at 20 kGy. (A) TGA and (B) DTG of PP, irradiated PP, irradiated PP/1phr SiO2-Ag nanocomposite.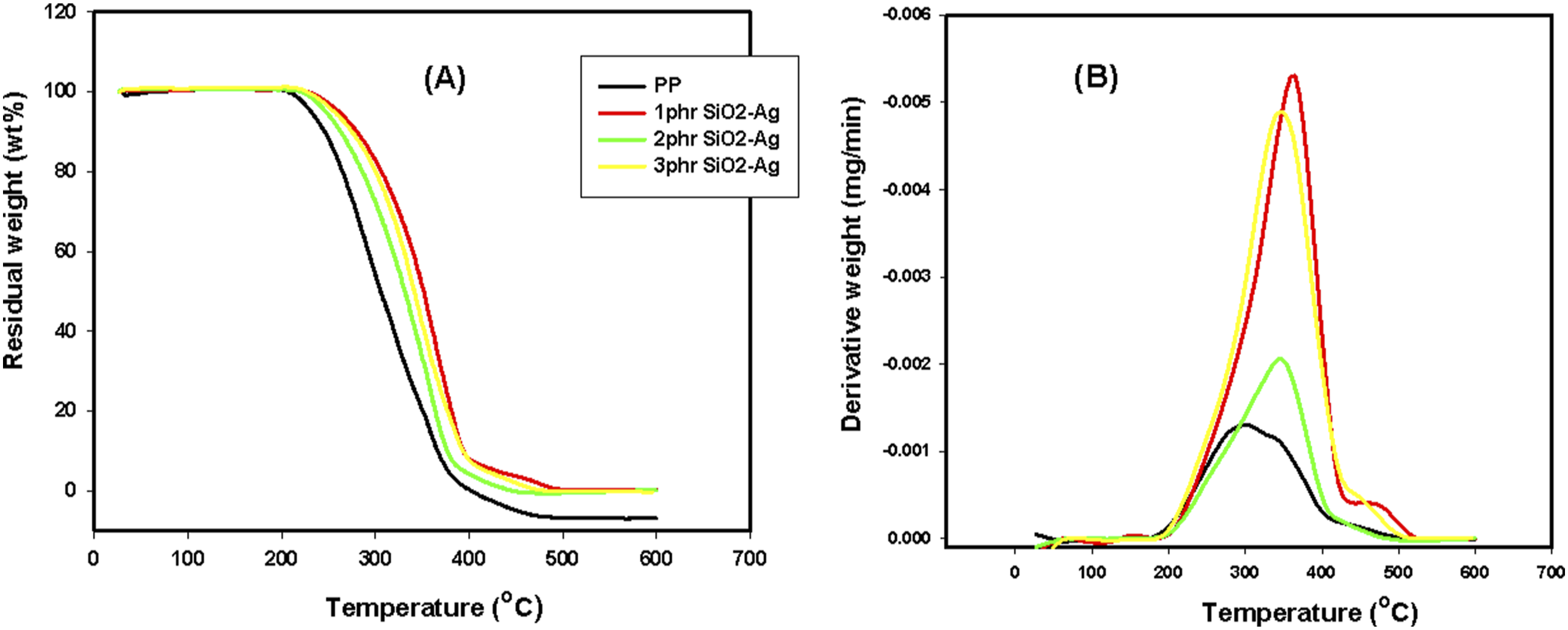
XRD of PP/SiO2-Ag nanocomposites
Figure 10 shows the XRD patterns of PP/SiO2-Ag nanocomposite with different concentration of SiO2-Ag nanoparticles. The diffraction peaks at 2θ = 13.1°,15.9°,17.6°,20.9°,24.6°,27.7° and 41.9° are assigned to the lattice planes of α-form isotactic structure of PP.
45
Upon addition of SiO2-Ag into pp matrix, a new diffraction peak was appeared corresponding to SiO2-Ag nanoparticles (see inset figures). With the increasing the content of SiO2-Ag, the intensity of the diffraction peak increased. At 1 phr of SiO2-Ag, its peak intensity is very low due to the high dispersion of nanofiller into the PP molten, while at 2 and 3 phr of the nanoparticles, the intensity of SiO2-Ag was appeared slightly sharp due to the high concentration and agglomeration inside the PP matrix. For the PP/1 phr SiO2-Ag nanocomposite, a peak can be observed at 2θ = 15.3°, indicating the presence of β crystal form. This result indicates that SiO2-Ag nanoparticles at low concentrations acted as nucleating agent to develop a small amount of β crystals.
60
In addition, the decrease in the crystallinity of the PP at 3phr SiO2-Ag nanoparticles (see intensity scale) is due to the agglomeration between the SiO2-Ag filler and polymer chains. X-ray diffraction patterns of pristine PP and PP/SiO2-Ag nanocomposites containing different concentrations of SiO2-Ag nanoparticles.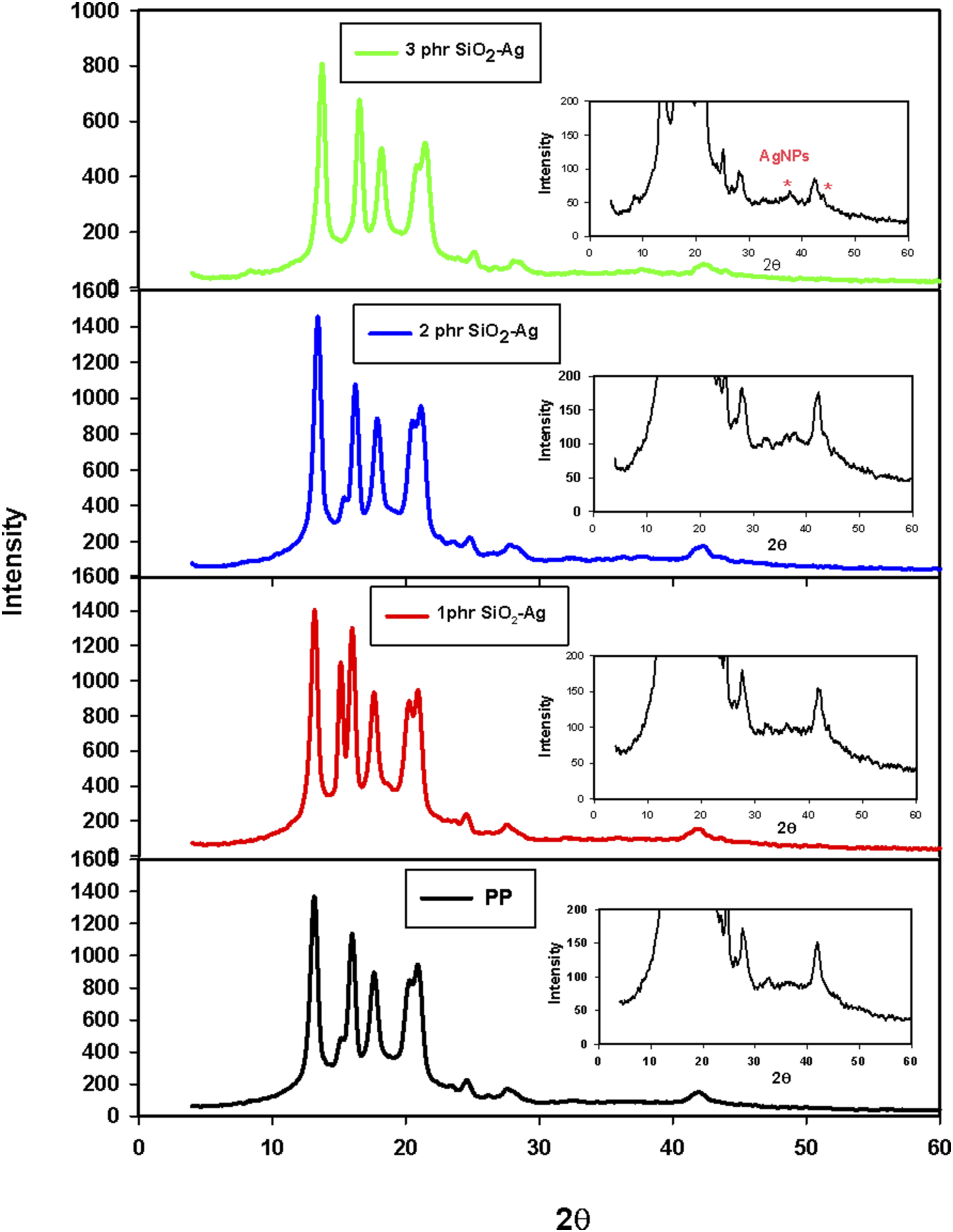
The XRD spectra for unirradiated and 20 kGy gamma irradiated PP/1 phr SiO2-Ag nanocomposite are shown in Figure 11. As shown, the XRD spectra of PP/SiO2-Ag nanocomposite before and after exposure to gamma irradiation nearly have the same spectra with marginally change in intensity that is, the overall crystallinity remained relatively intact after irradiation. This behavior is attributed to the polymer chains reorganization due to radiation induced degradation and crosslinking. The radiation does not influence on the polymer regions to the same extent where, the amorphous regions which contain the tie molecules that join crystallites are more susceptible to radiation, while the crystalline regions are relatively less affected. When the tie molecules are degraded by gamma irradiation, the stress on the crystallites may be relieved, facilitating the reorganization of crystalline and amorphous zones to some extent. Also, the degrading of these bonds and the local mobility of free radicals thus being generated can cause their displacement amongst the crystalline planes. As a result, some of the crystalline planes may deplete in the atoms and others may get enriched. This fact illustrates the decrease in the intensity of some plane and the increase in the intensity of other plans. Furthermore, the breaking of chains in amorphous regions makes them shorter, which makes their re-orientation easy.51,61,62 X-ray diffraction patterns unirradated and 20 kGy gamma irradiated PP/SiO2-Ag nanocomposites containing 1phr SiO2-Ag nanoparticles.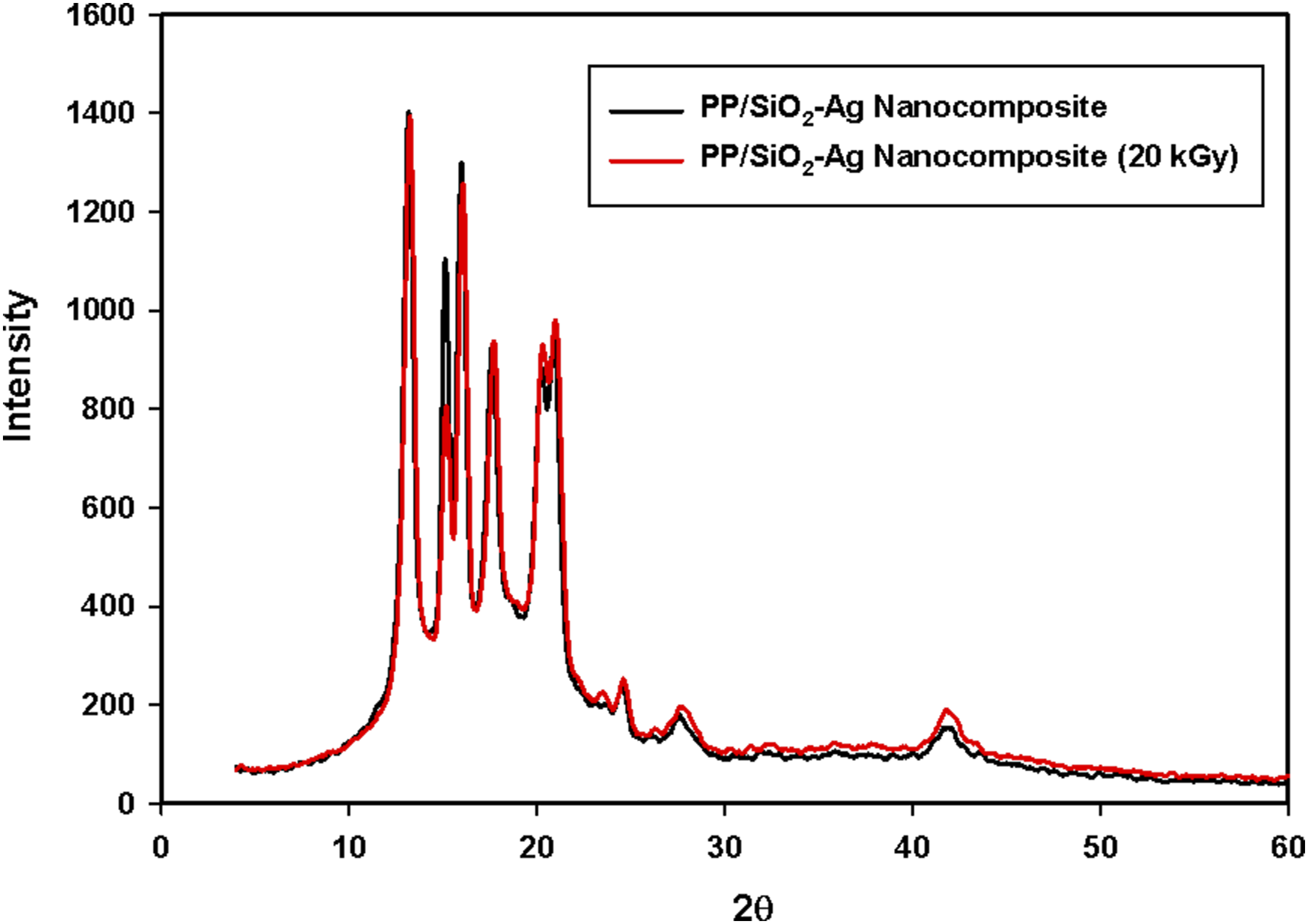
Scanning electron microscope
Figure 12(A)–(E) shows the surface morphologies of the PP and PP/SiO2-Ag (1phr) nanocomposite. As shown in Figure 12(A) and (B), the surface roughness of the unirradiated PP film decreases with the sample irradiation indicating radiation crosslinking process. Also, the surface of irradiated PP film contains low degree of roughness due to some scission of PP chains arising from the fact that both crosslinking and degradation process occurs simultaneously. It can be observed that (Figure 12(C)–(E)), SiO2-Ag (1phr) nanocomposite is uniformly dispersed and sphere-like structures indicated the presence of SiO2-Ag in the main polymeric film. SEM images of (A) pristine PP, (B) Irradiated PP and (C-E) PP/SiO2-Ag (1phr) nanocomposite with different magnification.
Antimicrobial activity
Improving effective biocidal polymers that can suppress pathogenic bacteria and yeast in touch has received significant attention, as some pathogenic bacteria often have unusual abilities to survive on everyday polymeric materials. The activity of microbial growth was examined and shown in Figure 13 and Table 2. It can be shown that PP and SiO2 NPs have no activity against both strains. On the other hand, both SiO2-Ag and PP/SiO2-Ag nanocomposites films showed antibacterial activity. The addition of SiO2-Ag developed that activity of PP. Antimicrobial activity of pristine PP and PP/SiO2-Ag nanocomposite. [1 = SiO2,2 = SiO2-Ag NPs, 3 = PP control, 4 = PP/1phr SiO2-Ag nanocomposite film, 5 = PP/2phr SiO2-Ag nanocomposite film, 6 = PP/3phr SiO2-Ag nanocomposite film, 7 = Clindamycin (DA; 2 μg/mL), Nystatin (NS;100 μg/mL)]. The antimicrobial activity of the SiO2-AgNPs and PP/SiO2-Ag nanocomposite films with different concentration SiO2-AgNPs. Calculated mean is for triplicate measurements from three independent experiments ± SD, a-f means with different superscripts in the same column for each sample are considered statistically different (LSD test, p ≤ .05), Nil = No activity (6 mm).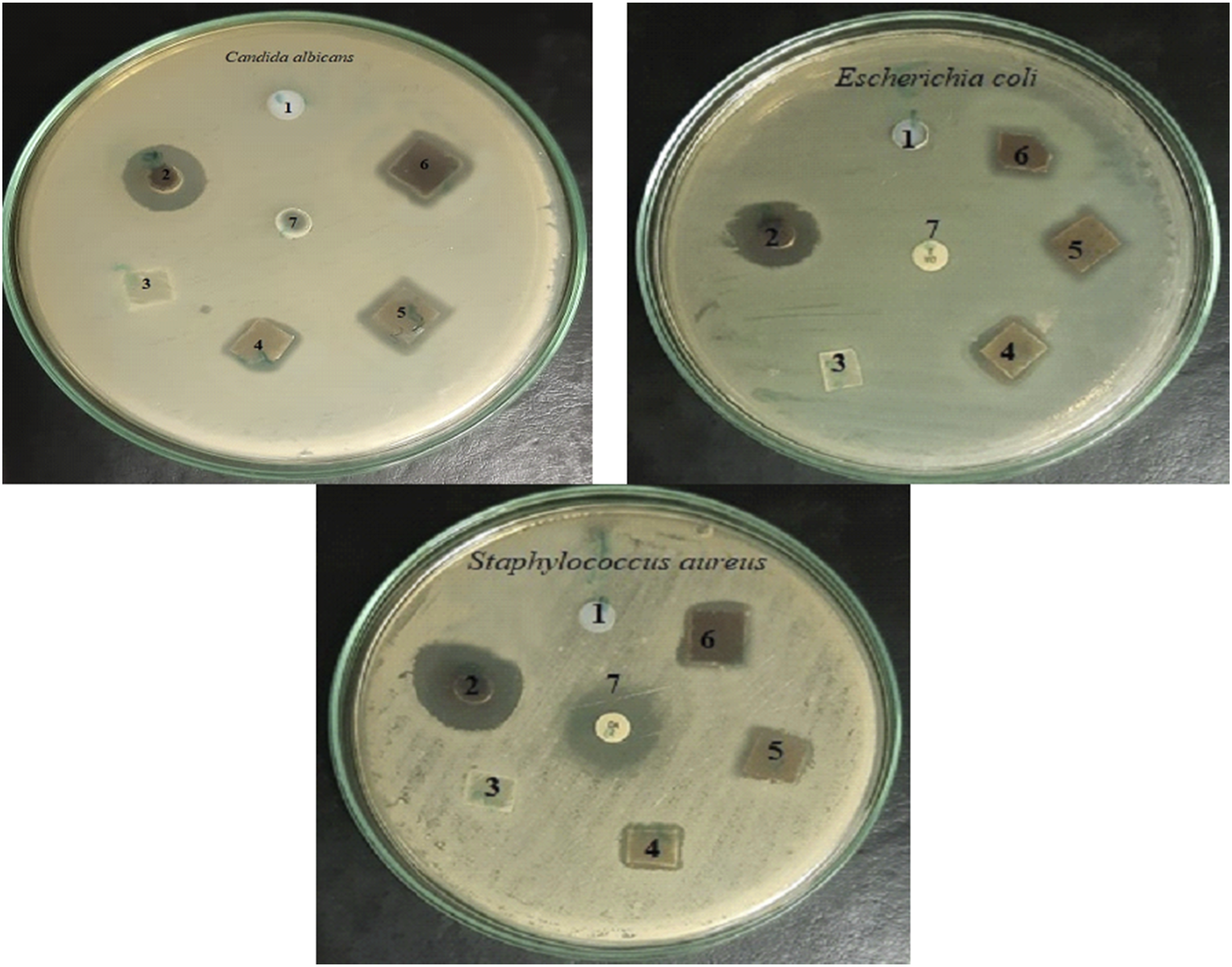
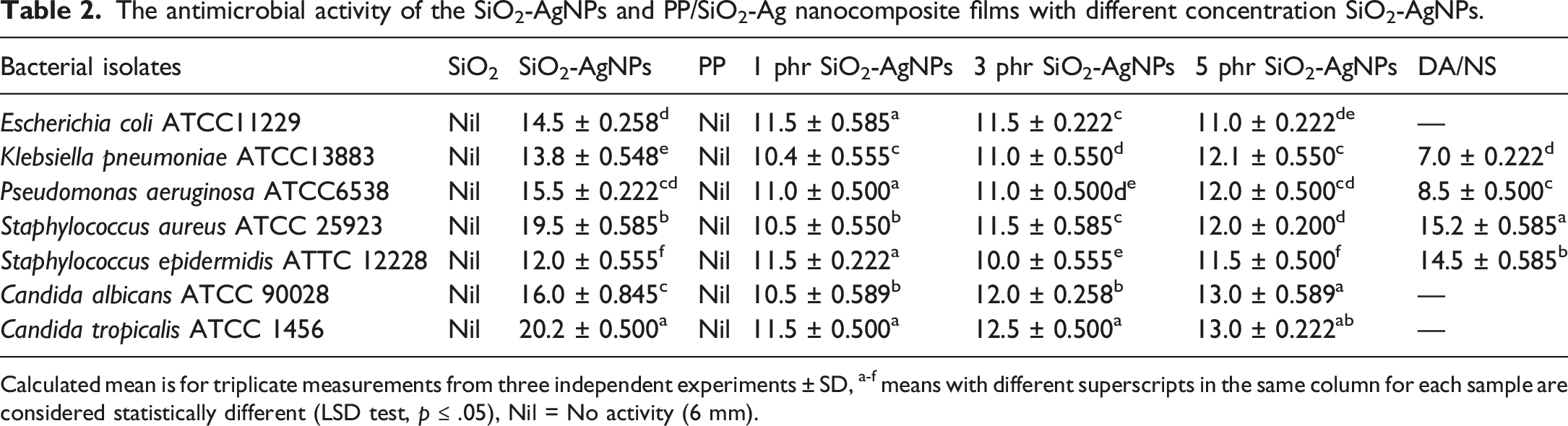
According to the previous studies, the adhesion of silver nanoparticles on the outer surface of the cell wall changes the bacterial surface and cellular behavior, ultimately changing the membrane permeability and stimulating the production of oxidative pressure response genes in the bacterial cell. This action disrupts the cell functions and finally causing the microorganism dies. Also, the penetration of silver nanoparticles into the cells causing both cell wall damage and breaking of intracellular composition (vacuoles, mitochondria, ribosomes) and biomolecules (lipids, protein, and DNA) resulting cell destruction and finally killing of the pathogenic microbes. 63
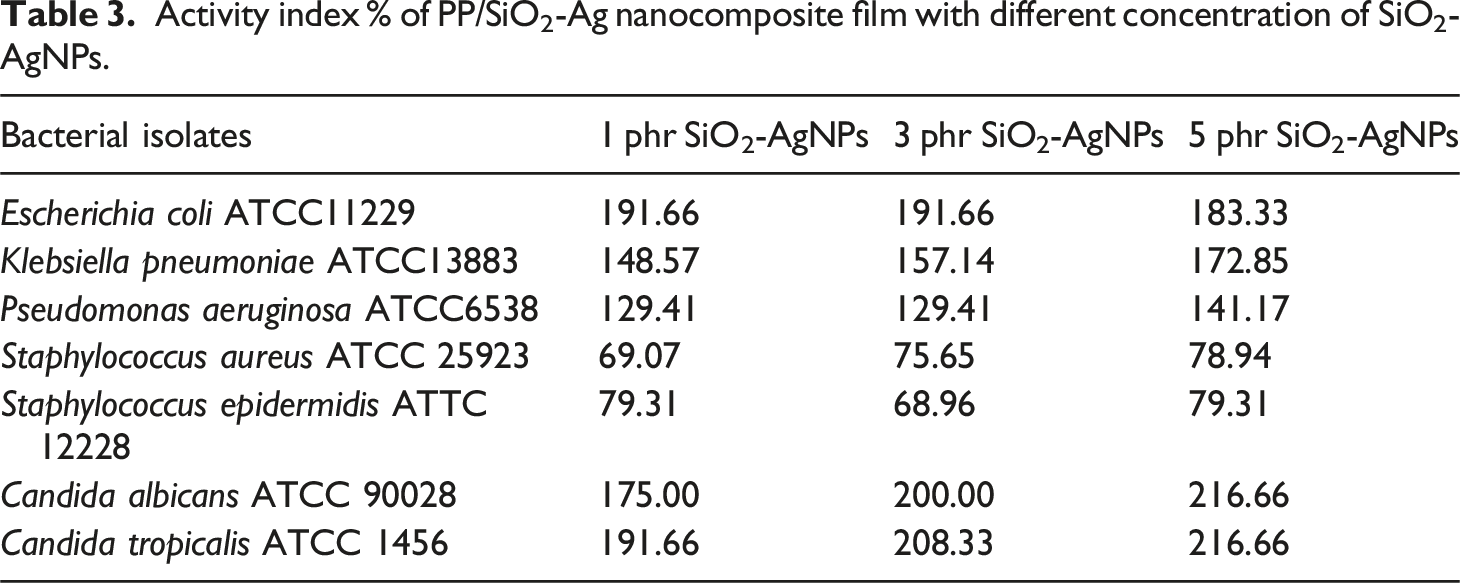
Activity index % of PP/SiO2-Ag nanocomposite film with different concentration of SiO2-AgNPs.
Conclusions
In this study, silver-coated silica (SiO2-Ag) nanoparticles were successfully synthesized to be combined with polypropylene via a melt mixing technique to improve both the physicochemical and bactericide effects of the thermoplastic. Silica nanoparticles were fabricated by precipitation method. After that SiO2-Ag nanoparticles were synthesized by gamma radiation. The XRD pattern and FTIR evidenced the formation of SiO2-Ag nanoparticles and TEM clarified the nano size of the prepared filler. In which, the XRD pattern of SiO2-Ag nanoparticles confirmed both SiO2 peak and other new five peaks at 38.1°, 44.5°, 64.7°, 77.5° and 81.3° which were indexed to (111), (200), (220), (311) and (222) crystal planes, respectively, correlated to silver. TEM supported that SiO2-Ag nanoparticles has a particle size with an average of 70 nm.
FTIR revealed that stretching vibration of the Si–O–Si absorbance is detected at about 1098 cm−1 for the PP-filled species approving the presence of silica in the PP matrix. Both the tensile strength and the elasticity of the PP thermoplastic matrix have been improved with SiO2-Ag filling at 1.0 phr, above this percent of the loading the mechanical parameters of PP were reduced. The irradiated specimens revealed superior tensile strength and elastic modulus over unirradiated samples. On the other hand, adhesion between the thermoplastic and the SiO2-Ag nanoparticles caused the reduction of the polymeric chain mobility of the PP and a decline in the elongation at break %. All the stages of the temperature mass loss (Tml10, Tm25, Tm50, and Tm75) of the pure PP considerably increased with SiO2-Ag nanoparticles filling reflecting significantly improved in the thermal stability of the native PP and 1.0 phr SiO2-Ag was the best. For instance, the Tm50 of the native PP increased from 305°C to 351°C of PP/1% SiO2-Ag thermoplastic nanocomposite. On the other hand, Tmax of the pure PP and PP/SiO2-Ag were improved with irradiation dose.
XRD revealed the diffraction peaks at 2θ = 13.1°,15.9°,17.6°,20.9°,24.6°,27.7° and 41.9° are assigned to the lattice planes of isotactic structure of PP. Upon addition of SiO2-Ag into the PP matrix, new diffraction peak appeared corresponding to SiO2-Ag.The use of SiO2-Ag nanoparticles gives important antibacterial properties. The fabricated PP/SiO2-Ag nanocomposites hold a notable antimicrobial potential for some pathogenic bacteria, yeast, and fungi which can be used in food packaging, biomedical and industrial applications.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

