Abstract
Heavy metals are considered to be a significant pollutant in water bodies, adversely affecting human health by causing various severe diseases after passing down the food chain. The rise in environmental problems due to the usage of non – biodegradable materials leads to the necessity of eco–friendly materials. The abundant and eco-friendly nature of the nanocellulose makes them promising substitutes for non-sustainable materials, nowadays. It is also possible to find the chemical components (cellulose, hemicellulose, and lignin) present in a source and the cellulose yield. In this context, nanocellulose has gained considerable attention among nanomaterials as a promising candidate for the adsorption of toxic heavy metal ions because of its large surface area, light weight, low cost, biocompatible nature, etc. Moreover, the numerous surface hydroxyl groups present in its surface make them suitable for the wide range of surface functionalization with different groups. They can thus be used individually or in combination with other materials for excellent adsorption towards various toxic heavy metal ions. The state of research on modified nanocellulose as an adsorbent for heavy metals is principally discussed in this review. Mainly two types of plant-based nanocelluloses; cellulose nanofibers (CNFs) and cellulose nanocrystals (CNCs), are discussed in detail in this review. The extraction of nanocellulose via a green approach was also covered. This review comprises comprehensive details on the modifications and other relevant properties of nanocellulose which would facilitate the adsorption of toxic heavy metals.
Keywords
Introduction
Nowadays our ecology is severely and dangerously impacted by water contamination due to the various toxins in the water bodies. Heavy metal ions are the primary cause of contamination in waterways. The majority of heavy metal ions are carcinogens that endanger human health as well as the ecological equilibrium. 1 Heavy metals are naturally occurring elements with a high atomic weight and a density at least five times greater than that of water. At extremely low concentrations, these metals are necessary for a number of physiological and biochemical processes in living things; nevertheless, when their quantities above a threshold, they become hazardous. The most common heavy metals found in waste water are arsenic, cadmium, chromium, copper, lead, nickel, and zinc. All of these metals are hazardous to the environment and human health. 2 The energy levels can be decreased by heavy metal poisoning, which can also harm vital organs including the kidney, liver, lungs, brain, and blood. Long-term exposure can cause slowly advancing muscular, neurological, and physical degenerative processes that resemble illnesses including multiple sclerosis, Parkinson’s disease, Alzheimer’s disease, etc. Continual long-term exposure possibly results in cancer. 2 Different techniques have been utilized to remove heavy metals including chemical precipitation, coagulation/flocculation, ion exchange, reverse osmosis, electrochemical treatment, and adsorption. Adsorption stands out among them as a quick, simple, and affordable way to purify water. Cost-effectiveness, large surface area, porosity, distribution of functional groups, and polarity are important selection factors for adsorbents. 3 Activated carbon, zeolites, graphene, and fullerene, as well as carbon nanotubes, are among the common and widely used adsorbents. The most often employed adsorbents are carbons and their derivatives because of their high adsorption effectiveness. Activated carbon is the widely used adsorbent but they are expensive to produce, the used activated carbon is problematic to dispose of, and the regeneration process is time-consuming and inefficient. 4 As a result, there are a lot of investigations have been carried out on inexpensive adsorbents that could be abundantly available and sustainable. Among those materials, cellulose has the advantages of availability, high yield, non–toxicity, and a high content of surface hydroxyl groups which are conducive for further modification. 5 Generally, nanocellulose has a high surface area which results in the enhanced exposed surface hydroxyl groups which are capable of surface modifications. Consequently, nanocellulose has gained considerable attention as a potential candidate for excellent adsorbent materials against warborne pollutants. This can improve the adsorption capacity. Therefore, here we are discussing an efficient eco–friendly material from the environment, that is cellulose, and its use to protect the environment and the public from toxic contaminants. 6
Cellulose (Figure 1) is a profuse green material that forms engrossing applications in the history of the human race. Cellulose is a polysaccharide composed of β (1, 4)– Extraction process and the structure of cellulose.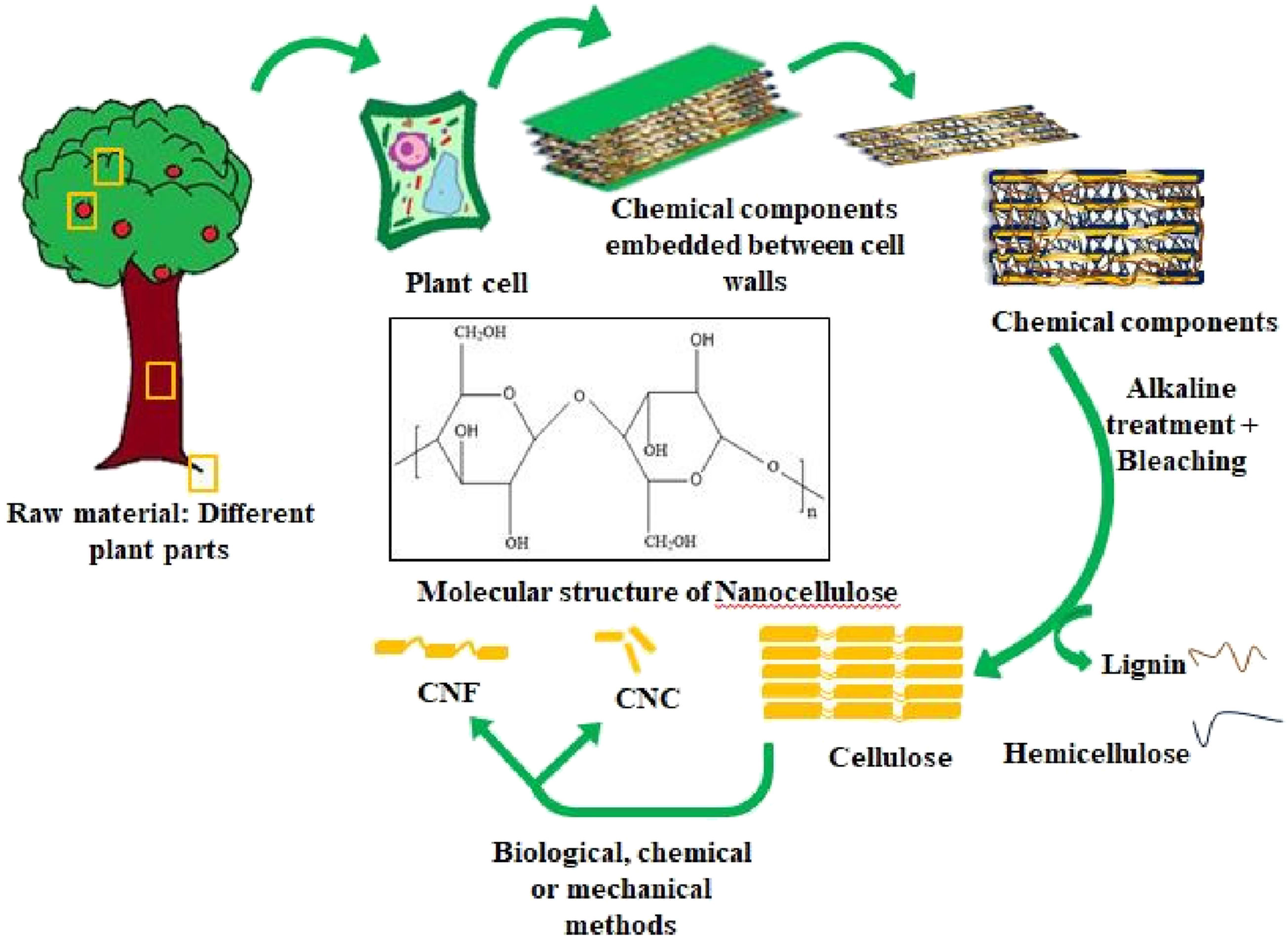
Cellulose can be used to develop new eco-friendly alternatives for conventional plastics 7 as they have various applications such as thermal insulation, packaging, water purification, etc. It can be extracted by both bottom-up and top-down processes. Figure 1 demonstrates that the cellulose can be extracted from various plant parts (top-down process), in which the alkaline treatment and bleaching help to remove lignin and hemicellulose, materials that bind the cellulose. Bundling cellulose chains together can further enhance its benefits by generating highly ordered regions that can be isolated as nano-particles. These particles, also referred to as nanocelluloses, are considered a promising class of materials for the future because of their physicochemical characteristics. They have qualities in addition to being plentiful and renewable: chemical inertness, high strength, great stiffness, low coefficient of thermal expansion, low density, dimensional stability, and the capacity to change their surface chemistry. 8 Depending upon the different biological, chemical, or mechanical treatments, nanocrystals or nanofibers are obtained. Mariano et al. proved that the basic bleaching process using sodium hydroxide solution could provide higher crystalline and thermally stable nanocrystals than that produced from acidic bleaching using peracetic acid. 9 To achieve the green extraction of nanocellulose, acid moieties should be avoided during the bleaching and other processes. Cellulase pretreatment of bleached soft kraft pulp reduced the acid concentration, which helps to obtain nanocellulose crystals (CNC) having similar homogeneity and appearance to those of the untreated samples with 64 wt % sulphuric acid concentration, which is the most widely employed acid. 10 Pre-treatment is done to break down the lignin that is present in the intricate structure of the cellulose and hemicellulose. Cellulase pre-treatment is an enzymatic process that makes it easier for the subsequent acid hydrolysis to yield CNC of comparable quality to the control but with less sulfuric acid. Xingye et al. carried out the cellulase pretreatment around 55°C for 8°h with varying enzyme dosage in a slightly acidic medium, and 4.8 unit/g was the optimized enzyme dosage as it gives high carbonyl contents that can be oxidized to carboxylic groups, thereby increasing the charge density of the cellulose particles. 11 The varying acid concentrations were used to prepare nanocellulose with optimized enzyme dosage, and 40% sulfuric acid concentration gives the same quality as that obtained from untreated pulp sample with 64% acid concentration, thus proving that it could reduce the disadvantages that can occur due to normal acid hydrolysis process such as environment pollution, high cost for equipment.
Apart from the extraction of nano cellulose, other aspects such as its modification, characteristics, and applications play a vital role and deciding factor for sustainable product development. Vizireanu et al. used submerged liquid plasma (SLP) with an ultrasonication method to modify microcrystalline cellulose, 12 which is considered an inexpensive and environmentally safe method. The broad range applicability of nanocellulose in diverse fields is due to their high possibility of surface modifications. Asoori et al. modified the cellulose nanofibers with dithizone for analyzing the trace quantities of manganese ions in industrial wastewater samples. 13 The sensors based on environmental monitoring applications using this biodegradable material, among its various applications, become more enchanting in conserving the green environment. Orelma et al. developed optical cellulose fibre for water-censoring applications by analyzing the change in light attenuation. 14
The applications of nanocellulose have increased as it is a fundamental material for the solution of environmental pollution; as a result, more platforms are necessary for discussing its recent history. Here, we discuss the latest advancements in the usage of nanocellulose for the adsorption of heavy metal ions.
Types of Plant-Based Nanocellulose
Nanocellulose from plant sources is mainly divided into two types: cellulose nanocrystals (CNCs) or cellulose nanowhiskers (CNWs), which have a short rod-like shape or whisker shape containing only crystalline region and cellulose nanofibers (CNFs) which is fibril in shape containing both crystalline and amorphous regions. CNCs were extracted from plant sources via strong acid hydrolysis, whereas CNFs were extracted using mild acid hydrolysis.
Cellulose Nanocrystals (CNCs)
Cellulose nanocrystals are unique nanomaterials that have received significant interest because of their mechanical, optical, chemical properties, etc. Although they are hydrophilic, they may be surface functionalized to fit a variety of difficult criteria, including the generation of high-performance nanocomposites, employing hydrophobic polymer matrices. Three types of commercial wood pulp were used to extract cellulose nanocrystals. 15 So it can be concluded that the nature of cellulose sources could control the aspect ratio and length of CNC as it has little effect on crystalline properties, surface charge, and sulfur content. Nanocrystals’ initial aggregation behavior depends on their dimensions, aspect ratio, and cross-section characteristics. CNCs were used to enhance the thermal insulation properties of rigid polyurethane foam without affecting its density. 16 A solvent-free ultrasonication method was opted for the incorporation of nanocrystals. The thermal conductivity reduction was 5% when adding 0.4 wt percentage nanocrystals.
Pine cones are a cheap, abundant, and effective source for isolating cellulose nanocrystals. 17 The potential of CNCs obtained from grape pomace to reduce waste management costs in the wine industry. 18 It was proved that this material was not toxic via cytotoxicity evaluation studies using in vitro methodologies, and hence it can be used in food and pharmaceutical applications. The potentiality of the blend of cellulose nanocrystals and polyvinyl alcohol/carboxymethyl cellulose was checked in food packaging applications. 19 Incorporation of CNCs into the polyvinyl alcohol/carboxymethyl cellulose positively affects the properties of bio–nanocomposite film due to the relatively high aspect ratio of nanocrystals. Arjmandi et al. incorporated CNC grafted Poly L – Lactide into the polylactic acid (PLA) to form nanocomposite films, and the characterizations such as mechanical, thermal, and optical properties were carried out. 20 It was found that the CNC: L – Lactide ratios could affect the grafting efficiency and thermal, mechanical, and optical properties of bio-nanocomposites.
Cellulose nanowhiskers (CNWs) were added to the montmorillonite/polylactic acid composite to investigate the biodegradability, thermal, tensile, and water absorption properties, etc. 21 This hybrid nanocomposite was prepared by solution casting, and the biodegradability, thermal stability, crystallinity, tensile strength, ductility, and Young’s modulus were increased with the addition of CNWs. It also gave improved water resistance. Saba et al. proved that nanowhiskers could increase the thermal and mechanical properties of PLA/cellulose nanowhiskers composites fabricated via the solution casting technique. 22
Cellulose Nanofibers (CNFs)
The world’s most sophisticated biomass component, cellulose nanofiber (CNF), is produced from wood-derived fibre (pulp) that has been micro-refined to the nano level of several hundredths of a micron and smaller. Due to its lightweight properties, the modulus of elasticity is as strong as that of the aramid fibre, a high-strength fibre with glass-like levels of thermal expansion. Additionally, CNF is a superior gas barrier against oxygen. Like cellulose nano whiskers, cellulose nanofibers (CNFs) also act as good reinforcing agents 23 for most polymer matrices. Okahisa et al. reported the effects of cellulose nanofibers on thermal and dynamic mechanical analysis of epoxy composites as a function of temperature. 24 The results showed that the thermal properties and dynamic mechanical properties increase in a composite when compared to neat epoxy.
Oil palm wastes and oil palm tree trunks were used to produce cellulose nanofibers. 25 All these raw materials have different chemical components and cell wall structures; as a result, the yield percentage of cellulose obtained from each source varies. 26 So the degree of polymerization, structure, morphology, and surface properties of the resulting CNF are also different. So, they have different behaviors towards mechanical and chemical treatments. For, both chemical and mechanical treatments cause the cellulose to degrade, and the more severe the treatment, the more severe the degree of polymerization (DP) decreases. Acid hydrolysis, TEMPO oxidation, and grinding all considerably decreased the DP of cellulose fibre. But pure mechanical processing could not destroy the construction of fibres. 27 Jiao et al. extracted CNFs with different charge densities by varying reaction times and conditions. 28 Due to this, CNFs with different lengths and similar cross-sections were obtained. CNFs were prepared by TEMPO–mediated oxidation systems and mechanical homogenization, which could reinforce cement by increasing the flexural and compressive strengths of the resultant composites. 29 The properties of nanocomposite film, which was prepared by using Sengkang starch and cellulose nanofiber, can be examined and the effects of filler loading were studied. 30 The properties such as moisture, and thermal resistance were improved. Wang et al. extracted the cellulose nano whiskers and cellulose nanofibers from the mulberry pulp, and it was used to make composite films in which alginate was used as the matrix. 31 The size of the reinforcing fillers decreased to the nanosize with the increase in crystallinity, which was proved by morphology and XRD analysis. Moreover, significant improvement in film property and tensile strength was shown by nano whiskers.
Extraction of Nanocellulose from Under-utilized Sources
Extraction of Nanocellulose from under – utilized sources.
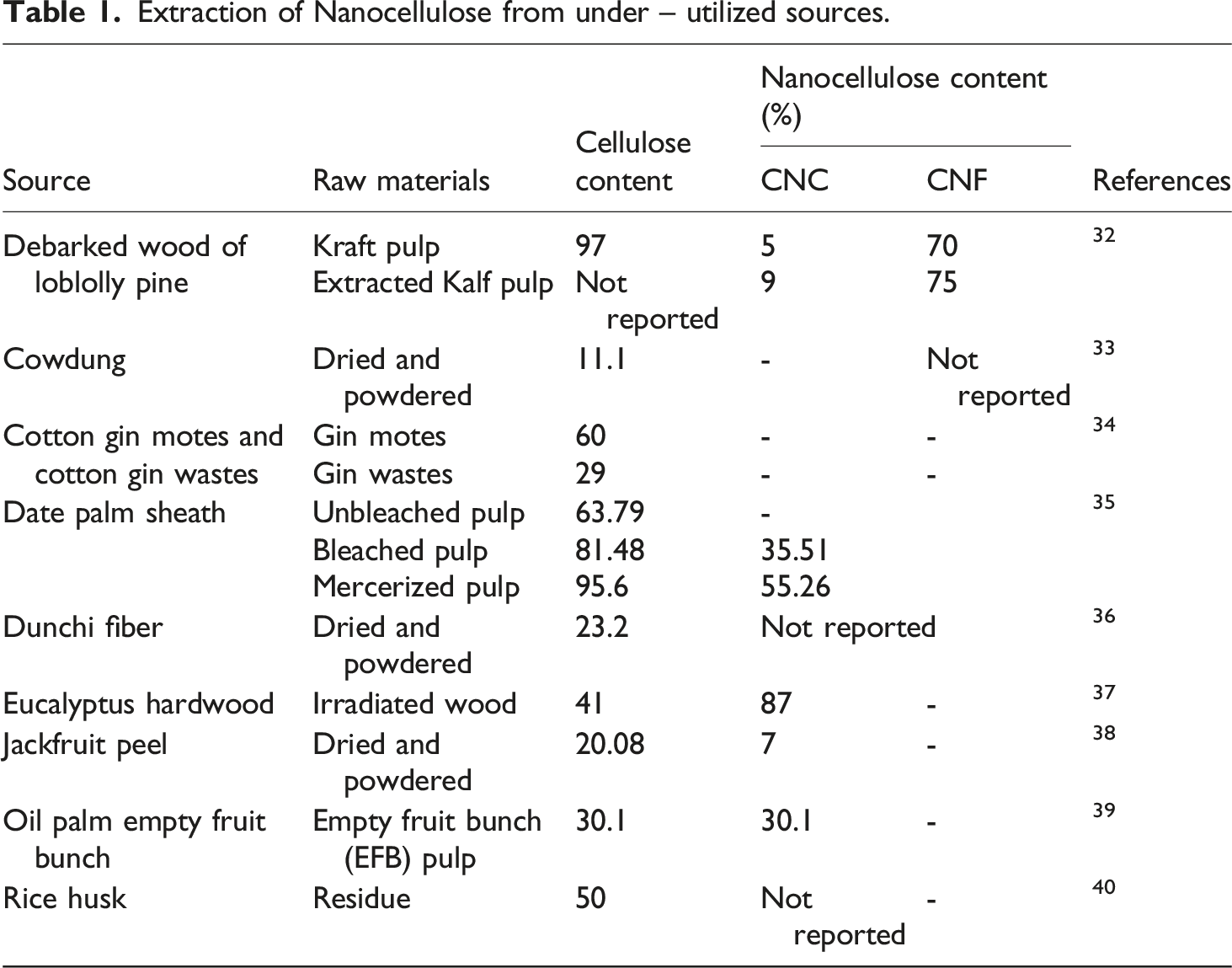
Rajan et al. used debarked loblolly pine wood as the source of nanocellulose. Here kraft pulp was extracted using a 1 L stainless steel an et al. used debarked loblolly pine wood as the source of nanocellulose. 32 Here kraft pulp was extracted using a 1 L stainless steel Parr reactor at 150⁰C, 60 min, 0.5% sulphuric acid, and was designated as EKP. The one without extraction was termed as control (KP). CNC (cellulose nanocrystals) yield in KP and EKP was less because of the short residence time of kraft pulps in concentrated acid. Cellulose was also obtained by drying and bleaching washed pulp. Puri et al. optimized the conditions 15% NaOH, 10% Na2S, 160 ⁰C, 1 h. Their bleaching process comprises (1) Hypochlorite bleaching, (2) EDTA bleaching, (3) Peroxide bleaching, and (4) EDTA bleaching. 33
Jordan et al. carried out the chemical treatments such as alkali bleaching to produce cellulose and cellulose nanocrystals successively using acid hydrolysis effectively from 40 mesh-size sieved samples of cotton gin motes and gin waste. 34 The mercerization treatment could enhance the removal of hemicellulose and lignin from the date palm sheath samples. 35 Oxidized nanocellulose was prepared by using ammonium persulfate (APS) solution with a concentration of 0.5 – 1.5 M for a duration of 8 – 20 h at 50 – 800⁰C and got the highest yield of the bleached sample with a concentration of 1.25 M at 600⁰C, 16 h and that of the mercerized sample with a concentration of 1 M at 600⁰C for a duration of 10 h. For the extraction of nanocellulose from Dunchi fibres, Khan et al. used n-hexane, ethyl acetate, and methanol treatment for a duration of 3h following the immersion in distilled water to remove extractives and wax content successively as a primary step. 36 Zhang et al. produced CNC from irradiated Eucalyptus wood via bleaching and ultrasonic disintegration process using γ – Valerolactone (GVL)/H2O and ethanol/H2O as organosol mixture solutions. Cellulose is simultaneously crystallized and subjected to irradiation oxidation to trigger fast breakdown and produce an approximate 87% yield of CNC. 37
CNC yield percentage is given as follows,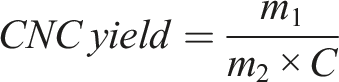
Trilokesh et al. used three methods for isolating cellulose
38
vis a vis sodium chlorite method, acetic acid plus nitric acid treatment, and formic acid treatment. Sajab et al. employed a telescopic approach containing lignin extraction, catalytic oxidation, and mechanical shearing processes to reduce cellulose loss and cellulose isolation.
39
The yields of cellulose and CNFs were improved without affecting the quality by this approach. The percentage yield of pre–hydrolyzed sample and extracted sample were determined by using the below equation’s.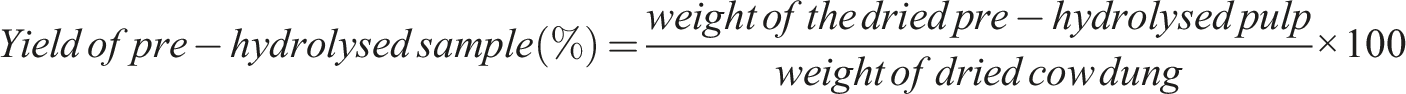
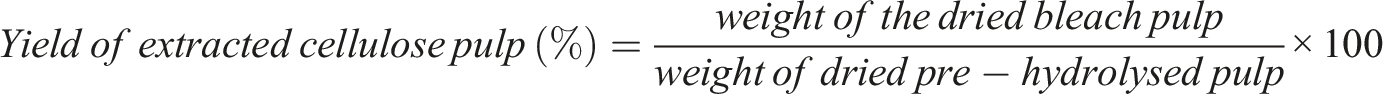
The production of cellulose nanofibers with similar properties to commercial nanofibers from agricultural residues sugarcane bagasse, along with the simple eco-friendly method 40 is a purifying technique for a green environment. Shahi et al. adopted a two-step process which includes Alkaline Hydrogen Peroxide (AHP) hydrolysis to remove lignin and hemicellulose and ultra-sonication for the extraction of CNF. 41 In the AHP hydrolysis process, the powdered material was continuously stirred with AHP solution in a water bath. 2N sulfuric acid is used to neutralize the pH at the end of the process. Then it was ultrasonically irradiated with 1% hydrogen peroxide solution. Kang et al. prepared cellulose nanocrystals without harsh chemicals or organic solvents and conquered higher thermal stability. 42 It was prepared by ball milling with water, followed by centrifugation.
Nanocellulose Modifications for the Removal of Heavy Metal Ions
Inter and intra-molecular hydrogen bonds in nanocellulose, due to the abundant hydroxyl groups present in it, help to react with various functional groups. Jiang et al. reported that the modifications of nanocellulose are possible in three different ways as given in Figure 2.
43
Modifications of nanocellulose (Modified).
43
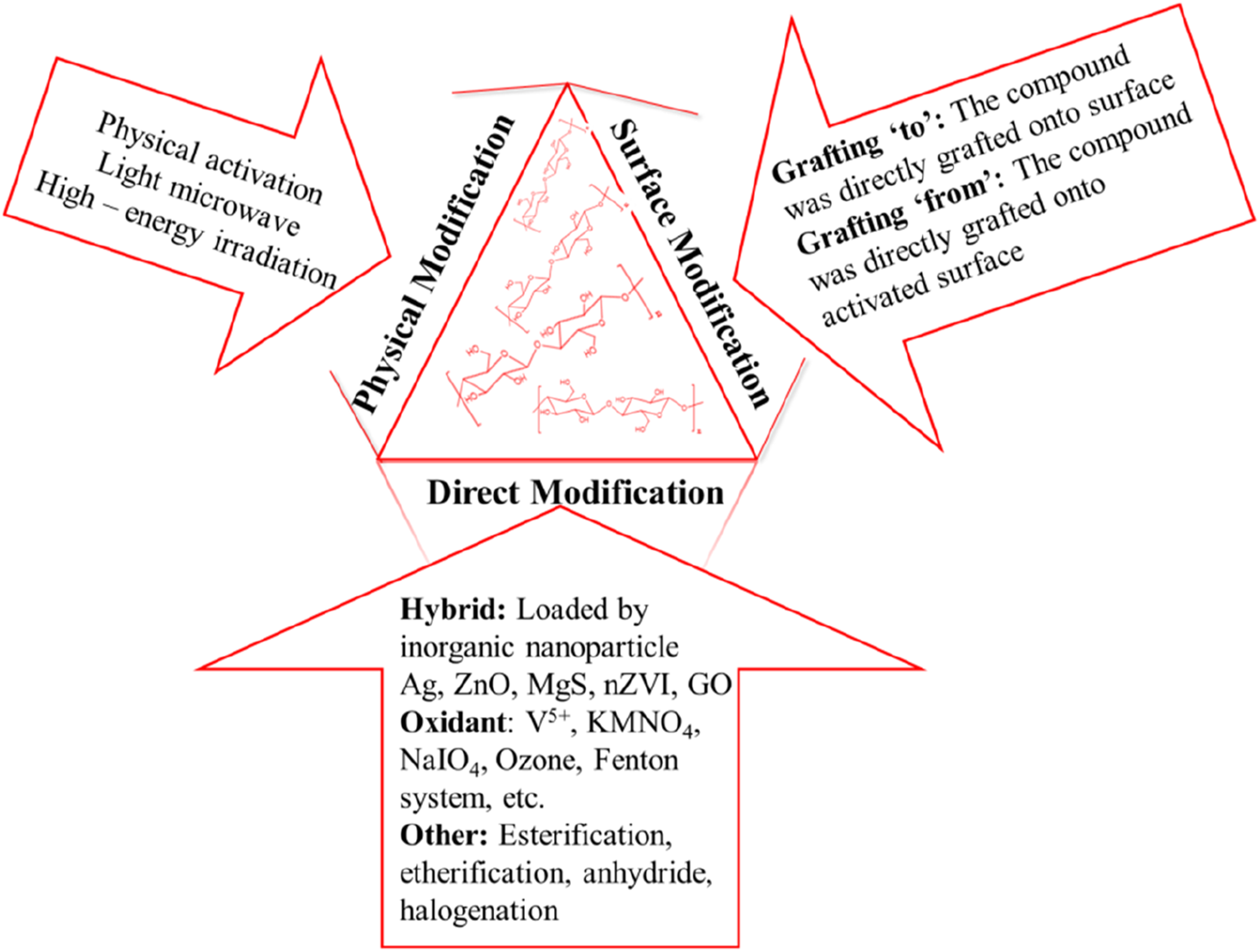
The three -OH groups on the C-2, C-3, and C-6 atoms of the six-member glucose unit of nanocellulose provide reactive sites for modification. Mainly there are two major types of chemical modification approaches, namely indirect modification & direct modification. Direct modification constitutes halogenation, esterification, and oxidation. A chemical process known as “direct modification”, which facilitates the hydroxyl groups, could react with other reactive moieties that might chelate or bind heavy metals. It was discovered that the etherification process of the –OH group on C-2 was two times as quick as that on C-3 and that the esterification of -OH on C-6 was 10 times faster than in other places. The reactivity of C2-OH and C3-OH is generally lower than that of C6-OH for the three hydroxyl groups. 44 Surface grafting modification, which may be classified as grafting “to” and grafting “from,” adds monomers or reactive moieties to the main chain of nanocellulose by cross-linking chemicals or not after being activated by initiators, allowing it to adsorb heavy metals. This is due to the surface chemical alteration of the long chain of cellulose nanomaterials consisting of grafting agents with a lengthy compatibilizing tail and a reactive end group which increases the apolar character of the nanocellulose.
Direct Modification
The glucose unit on nanocellulose would immediately react with the chemicals used for direct modification, affecting the morphology rather than the fundamental structure of nanocellulose. 45
Hybrids
Hybridization is when inorganic nanoparticles like Ag, ZnO, MgS, MgO, and graphene oxide (GO) are added to nanocellulose surface. The nanocomposites now have specific different characteristics thanks to the hybrid alteration. For instance, a mixture of GO and nanocellulose exhibits a high electrical conductivity. 45 Organic solvent resistance is increased in the hybridized cellulose nanofibers with silver and titanium dioxide nanomaterials. 46 Heavy metal ions can be effectively removed by hybridizing nanocellulose with metal compounds. Using several interference ions such as NO3-, Cl−, etc. with a pH range of 3-5, the composite of cellulose nanocrystals attached with nZVI had the potential for applications such as nickel ion removal, and the maximum removal efficiency reached 98.5%. 47 Compared to CNFs doped with FeS alone (66.7 mg/g), cellulose nanofibers doped with iron sulfide and magnesium sulfide had an adsorption capacity for hexavalent chromium ions of 142.8 mg/g.
Oxidation
The oxidative modification technique was susceptible to the acidity or alkalinity of the oxidizing media and the oxidizing reagent due to the hydroalcoholic nature of cellulose nanomaterials. By oxidizing the C6-OH group into the carboxyl group, potassium permanganate (KMnO4) can transform cellulose nanomaterial into a brown-black adsorbent with a high manganese concentration. 48 The modified nanocellulose has an 80.1 mg/g Pb2+ adsorption capacity. 49 Direct oxidation and selective breaking of the NaIO4 occurred at the alcoholic –OH on C-2 and C-3. A cellulose nanomaterial oxidized by NaIO4 and fixed with a -CHO group showed a maximum capacity of 384.62 mg/g for lead ion adsorption. 50 Alcoholic -OH groups on C-6 were intended to be converted to -COO groups using the TEMPO/NaClO/NaBr oxidation system. 51 Pb2+ was able to bind to TEMPO-oxidized cellulose nanofibrils with an adsorption capacity of 157.73 mg/g, 52 and copper ions and lead ions (above 75 mg/g) were better able to bind to TEMPO-oxidized cellulose hydrogels (TOCNF). 53
Other Methods
Other direct techniques besides oxidation modification are esterification, etherification, hybridization, and alkylation. These techniques increased the absorbability of nanocellulose to heavy metals by directly loading it with some organic molecules that bind to heavy metals. For heavy metal ion cleanup, for instance, alkali-washed nanocellulose esterified with citric acid demonstrated outstanding performance. The clearance rate of Cr6+, in particular, was 81.3%. 54 The maximal As3+ adsorption capacity was reported to be 357.14 mg/g in several investigations using cysteine-esterified CNCs or cysteine-esterified CNFs. 55 Cysteine-CNCs had an Hg2+ adsorption capacity of 849–923 mg/g,56,57 whereas cysteine-CNFs had a Hg2+ adsorption capacity of 117 mg/g. 58
Surface Modification
Three techniques—grafting “to,” “from,” and “through”—were used to covalently connect monomers or large molecules to the surface of nanocellulose. It is possible to think of the grafting “through” approach as a middle step between the grafting “from” and grafting “to” procedures. 59
Grafting to
The surface-initiated grafting technique included striking the monomer against the active sites of the nanocellulose primary chain and causing the monomer to polymerize. A cross-linking agent worked to combine the monomer and the active site. The reaction took place anywhere in the nanocellulose, middle region, or at either end of the chain. The organic monomers had chemical linkages such as ester bonds, anhydride bonds, etc. and functional groups such as amino, carboxyl, sulfhydryl, etc. For instance, CNF surface grafted with ethylene diamine tetraacetic acid dianhydride was employed as a dye adsorbent. As a hydrophobic adsorbent, 4, 4′-diphenylmethane diisocyanate was grafted on cellulose nanofibers. A cellulose paper-based humidity sensor of CNCs grafted with glycidyl trimethyl ammonium chloride was employed. 60 As an oil-in-water emulsion stabilizer, cellulose was grafted with octenyl succinic anhydride. 61
Because of its great changeable qualities, low cost, and wide availability, this method has the potential to be used in heavy metal adsorption. According to the Langmuir equation, A. Zhang et al.61,62 used an agent called epichlorohydrin to cross-link polyethyleneimine to nanocellulose. The resulting 3D network-structured, polyethyleneimine-functionalized nanocellulose can lower the Cr6+ content to <0.05 mg L-1. According to Ali et al, 63 the order of the strength of the heavy metal ions adsorbed by grafting ‘to’ modified nanocellulose is as follows: Hg2+> Ni2+> Cr3+> Co2+> Pb2+. Removing heavy metals by nanocellulose significantly improved by adding carboxyl and amide functional groups. Zhang et. al proved that the PEI – CF composite has good selectivity towards Cu2+, Pb2+, Fe3+, Zn2+, Cr3+, SO42-, Cl−, NO3-, H2PO42- and S2-. 64
Grafting From
Initiators should be required for the ‘grafting from’ technique, often referred to as free radical polymerization, to enhance the grafting conditions and activate nanocellulose at the beginning of modification. Adsorption sites were oxidatively activated by inorganic or organic initiators. The –OH group on the nanocellulose has been oxidized to the carbon-based matching structure after the initiator reaction process, such as the ketone at C-2 and C-3 or the aldehyde or carboxyl moiety at C-6. Any reactive group, including -OH, -NR2, and -CONR2, could be attached to the monomers. The chain propagation is promoted by combining monomers to a growing radical. The chain growth stops when the propagating radicals combine, and the growth could be terminated. The surface-initiation techniques comprised gamma radiation, UV radiation, redox processes, and the breakdown of peroxide materials following dehydrogenation, oxidation, and depolymerization. Using a TEMPO oxidation method as an initiator, Xing et al. 65 activated the cellulose to create a TEMPO-oxidized cellulose hydrogel (TCH). Then, glutaraldehyde was used to cross-link polyethyleneimine to TCH, resulting in the creation of cross-linked product of TEMPO – oxidised cellulose hydrogel TCP. TCP had substantial adsorption capacities of 109.89 and 279.32 mg/g towards Cu2+ and Pb2+ heavy metal ions respectively. TEMPO oxidization helps to generate a typical network structure. After grafted with polyethyleneimine (PEI), the TCP was evenly surrounded by a PEI layer, creating several tiny pores between the TCP branches. The grafting of PEI improved the specific surface area of TCP, which would improve metal ion accessibility to the TCP. Xing et al. 66 prepared a Cr6+ adsorbent based on amino-dense nanocellulose (4.4 mmol/g). As an initiator, carbamate was utilized to esterify the nanocellulose. The nanocellulose was then grafted with polyethyleneimine.
Physical Modification
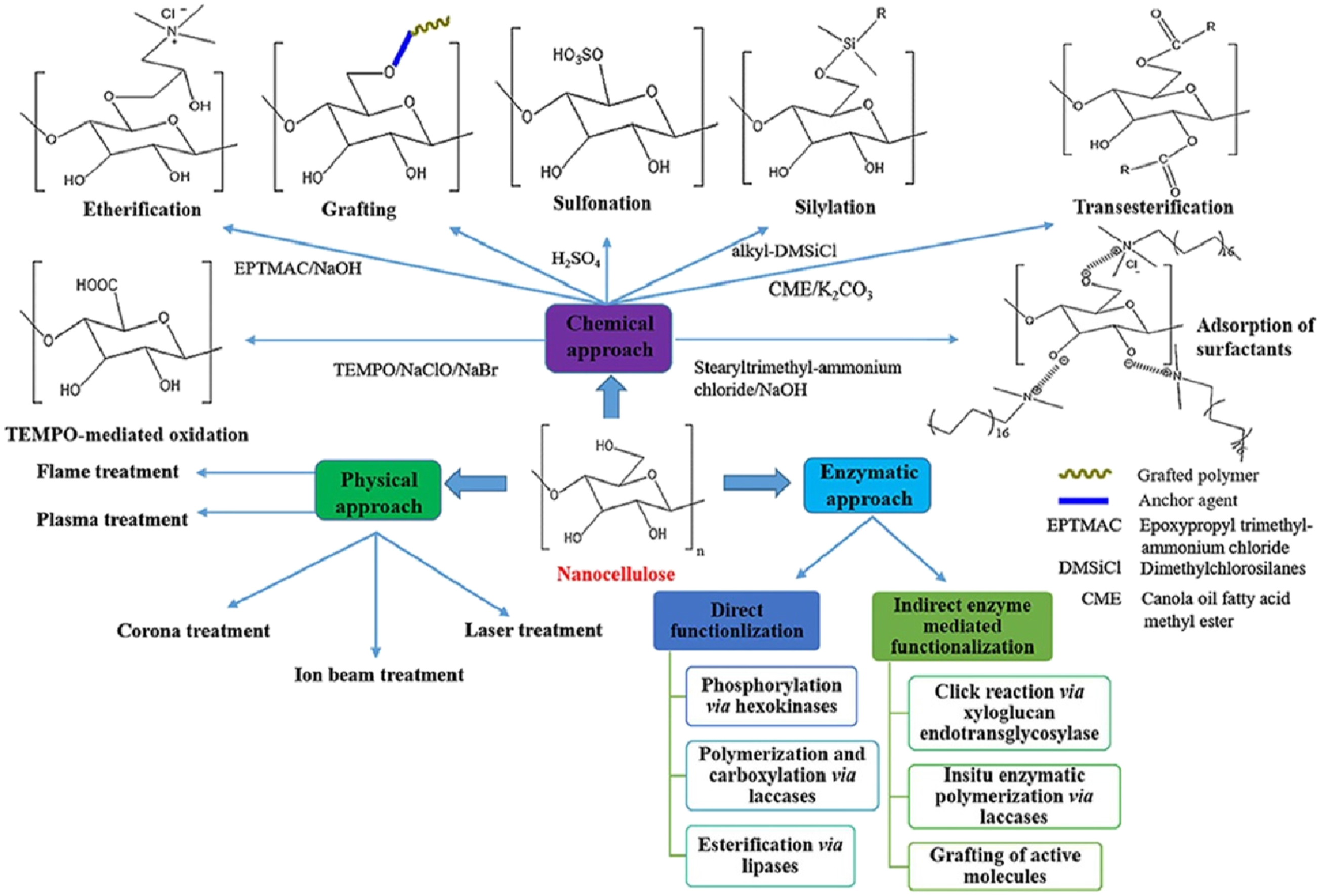
Physical stimulation could also modify nanocellulose without the use of chemical initiators. Different vinyl monomers could be introduced into nanocellulose using light grafting. Gelatin epoxidized natural rubber and polyacrylamide are functional polymers that can be grafted onto nanocellulose using microwaves or high-energy radiation. Even a large number of modifications are available; this paper only discusses some significantly essential modifications through covalent bond formation for heavy metal ion adsorption. The various surface modification routes of nanocellulose using different functional groups are represented in Figure 3. Each functional group improves the properties of nanocellulose in its own way.
67
Surface modification routes of nanocellulose using different functional groups. Reproduced with the permission from Elsevier Ltd.
67
Silane Modification
Silylation of nanocellulose is a most common modification not only for improving hydrophobization, antimicrobial activity, and compatibilization but also for improving adhesion. Thus, it can be widely used in coating applications. Three types of silane modification used are listed below.
70
• Epoxy silane (gamma glycidoxy propyl trimethoxy silane) • Methacryloxy silane (gamma methacryloxy propyl trimethoxy silane) • Amino silane (gamma amino propyl trimethoxy silane)
For this, silane solutions were prepared using ethanol as the solvent, and each solution was added to nanocellulose suspended in water. At the fixed ratio of pure silane to dried nanocellulose (1:1), various silanes’ effects in nanocellulose after stirring for 2h at room temperature were studied. The preparation of silane-modified cellulose using different types of silanes, (a) epoxy silane (gamma glycidoxy propyl trimethoxy silane), (b) methacryloxy silane (gamma methacryloxy propyl trimethoxy silane), and (c) amino silane (gamma amino propyl trimethoxy silane) is represented in Figure 4. These silanes contain dissimilar functional groups in which all the groups are involved in the chemical reaction. Preparation of silane-modified nanocellulose. Reproduced with the permission from Elsevier Ltd.
68
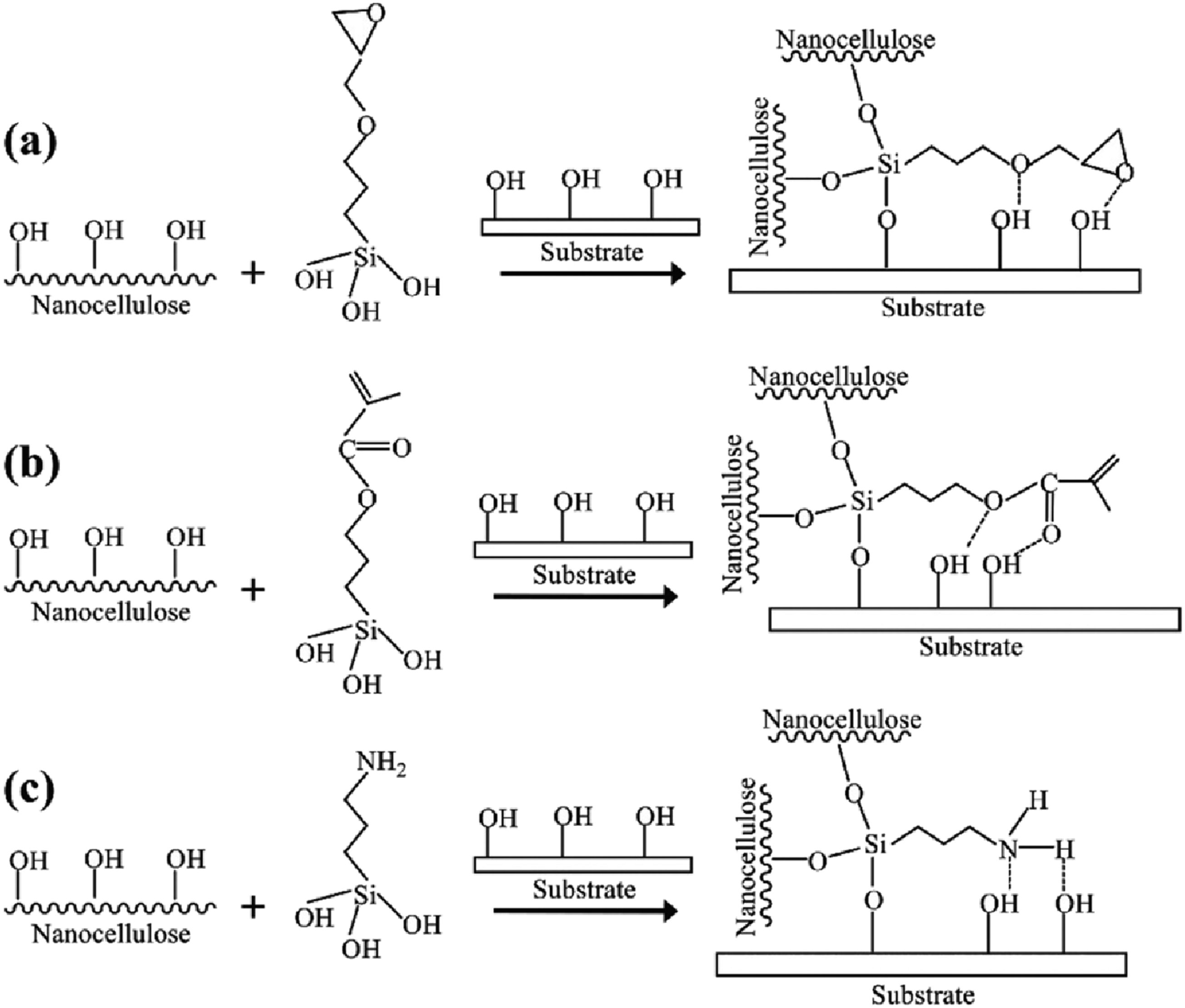
Excellent adhesion property is given by nanocellulose modified with amino silane groups rather than epoxy and methacrylic silane. Also, there is a tendency to increase adhesion properties with higher amino silane ratios. However, the elongation and crack resistance are higher in the case of epoxy and methacryloxy silane than amino silane-modified film.
Carboxylate Modifications
To broaden the application of natural biodegradable PEs by preparing highly efficient water-absorbing natural PEs, the dicarboxylate group was incorporated into the nanocellulose by a two-step oxidation process. The introduction of carboxylate groups into the nanocellulose can help to improve water uptake capacity.
69
The steps involved in the carboxylate modification are represented in Figure 5. Preparation of dicarboxylate NCC.
69
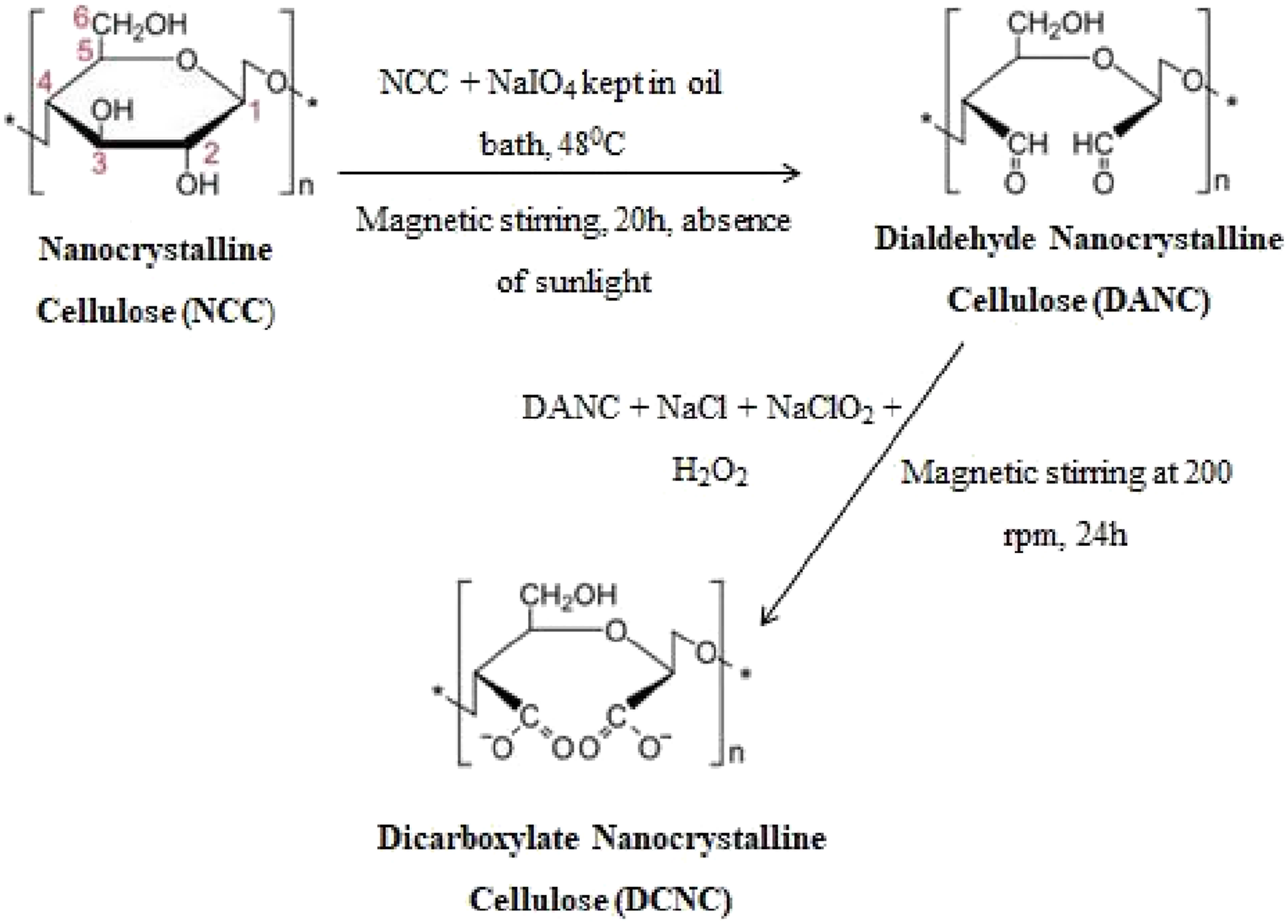
The authority that granted approval and the associated ethical approval code must be mentioned for interventional studies involving humans or animals as well as for other investigations that need ethical clearance.
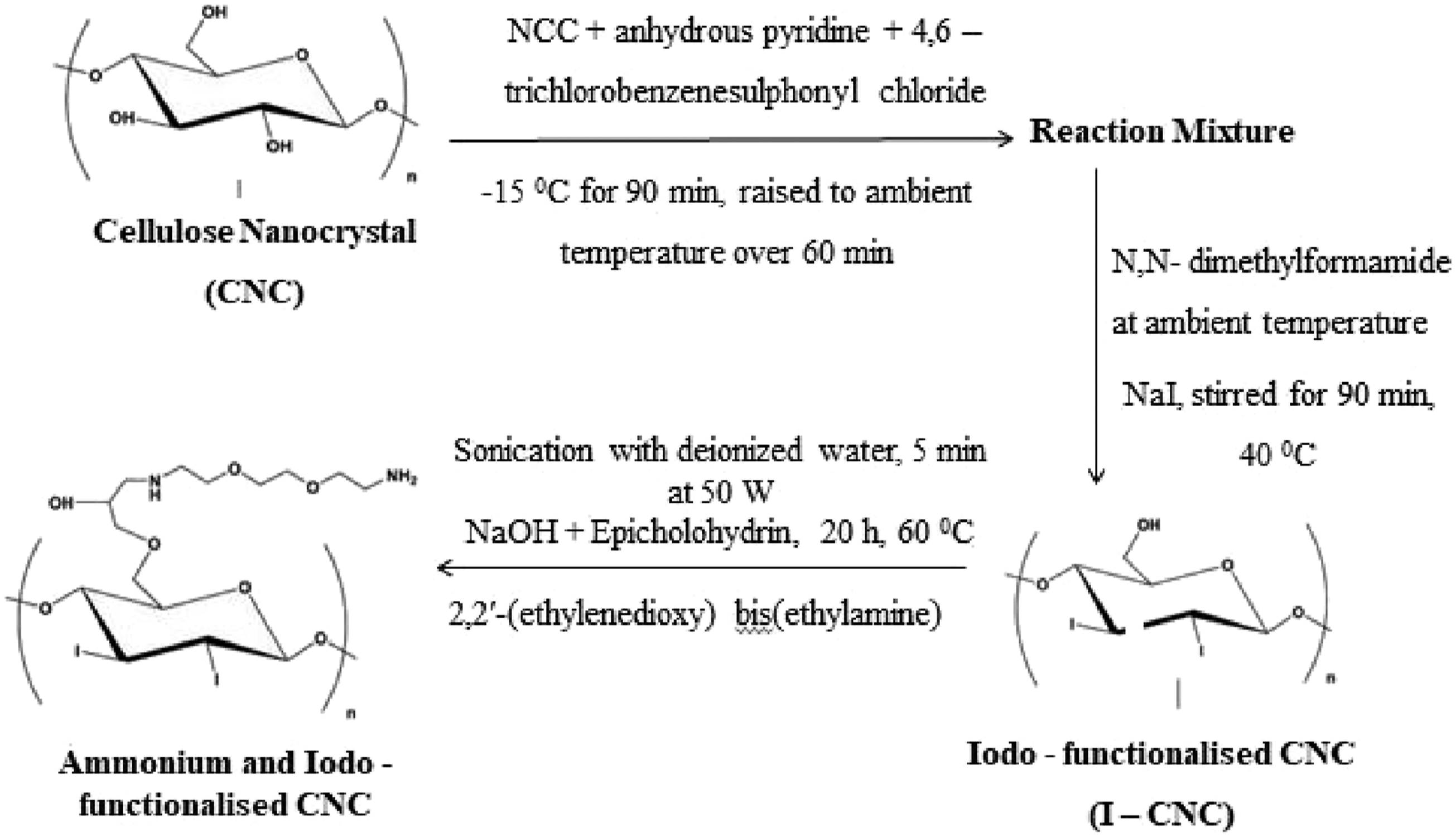
Ammonium Modifications
A covalent chemical design approach was explained by introducing ammonium groups into cellulose nanocrystals to improve aqueous solubility.
70
A two-step process is required for their preparation which includes the following steps, (1) Formation of Iodo-functionalized cellulose nanocrystals (2) Formation of solid ammonium and Iodo-functionalized nanocrystals
This modification showed that this material could exhibit unique fibrillar nanomaterial properties in vivo because of its water-soluble functionalized derivatives. The modifications of nanocellulose give controllability to its hydrophilic and hydrophobic nature. Therefore, nanocellulose can be used in composites for various applications. The steps involved in the ammonium modification of cellulose are represented in Figure 6. Preparation of ammonium and Iodo-functionalized CNC.
70
Adsorption Properties of Nanocellulose
Nanocellulose is a potential material in adsorbent applications as it has high adsorption properties, which depend on various factors. It is possible to correlate the chemical structures, functionalization degree, morphology, and surface hydrophobic/hydrophilic character of nanocellulose with the chemical structures, polarity, shape & size of target molecules in the case of chemical adsorption. 71 The primary aim of the use of nanocellulose adsorption properties is to make the potential to detect the most threatful materials. The hydroxyl groups offer the property of nanocellulose for metal ion adsorption.
While using carboxylic nanocellulose, the carboxyl groups became more effective in adsorption process.
72
The maximum adsorption capacity of carboxyl groups can be estimated theoretically by using the following equation: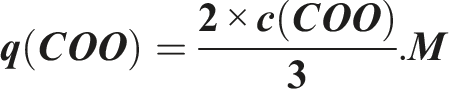
The maximum adsorption capacity of carboxyl groups can also be estimated theoretically by using the equation: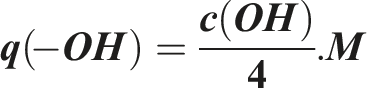
The nanocellulose in aerogels has oil and organic solvent adsorption capacity. However, nanocellulose aerogels in combination with reduced graphene oxide and nano chitosan are more effective. 73 Nanocellulose, along with sodium dodecyl sulfate (SDS) in the form of aerogel foam, gives a higher adsorption capacity towards ethyl acetate, cyclohexane, and vacuum pump oil than that of conventional nanocellulose aerogels. 74 Modifications of nanocellulose with chitosan are essential methods for adsorbent development. Hydrogel beads using carboxyl chitosan and carboxyl nanocellulose offer more lead adsorption capacity. 75 A magnetic nano cellulosic aerogel using iron oxide and oleic acid is an effective adsorbent of oil spills and organic solvents and can be used in magnetic-related applications. 53
Kinetic models like Langmuir and Temkin were used to determine the adsorption kinetics of cellulose-based biosorbents (bleached almond shell, lyophilized bleached almond shell, and TEMPO-oxidized cellulose nanofibers) from the almond shell and checked with experimental kinetic data. The bleached almond shell was fitted with the Temkin model, while all others were fitted with the Langmuir model. 76 Nanocellulose-based composites with reduced graphene oxide and amino-functionalized carbon nanotubes were utilized to develop excellent oil-adsorbent aerogel. 77
As heavy metal pollution is one of the significant environmental issues that cause serious health problems among humans, nanocellulose – iron oxide hybrid aerogel was used to detect heavy metals in water.
78
The adsorption capacity of hybrid aerogel can be estimated by using the equation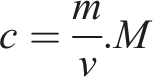
C (mg/mL) = heavy metal ion concentration; m (mg) = mass of the heavy metal ion in the solution; v = solution volume
Absorption amount of heavy metal ions at equilibrium can be estimated by: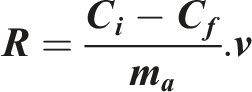
Factors Affecting the Heavy Metal Adsorption of Nanocellulose
Crystallinity
A crystalline region and an amorphous region make up the nanocellulose. The molecules were grouped in a perfectly regular and compact form, and the crystal region had a significant intermolecular binding force. Thus, the crystallization zone was influenced by nano cellulose’s strength. In contrast, the arrangement of cellulose molecules in the amorphous zone was loose and uneven with significant intermolecular spacing; as a result, the area has a lot of free hydroxyl groups. The crystallinity of nanocellulose is significantly influenced by the ingredients, processing, and modification techniques. The high crystallinity structure enhanced the regulating variables and the heavy metal adsorption performance during the subsequent modification process. For example, the most significant capacity of cysteine-modified CNCs for copper ions was 92.07 mg/g, and created polyamide-grafted cellulose nanocrystals with an 87% crystallinity. 79
Specific Surface Area (SSA)
The adsorptive activity is impacted by SSA. A greater amount of adsorption sites should be exposed due to the increased SSA. The greatest adsorption capacity for cadmium ions was 54.71 mg/g, and the specific surface area of vulcanized cellulose nanocrystals-based adsorbents was 10.93 m2/g. 80 The greatest Cr6+ adsorption capacity was 205.0 mg/g, and the specific surface area of cellulose nanocrystals grafted with dopamine rose from 15.6 to 101.9 m2/g. 81 For instance, as the specific surface area dropped, the adsorption efficiency increased. The fact that the adsorption site of the modified nanocellulose with low specific surface area was significantly bigger than that of the unmodified nanocellulose, while the specific surface area reduced after modification, could be one explanation for this outcome.
Aspect Ratio
The aspect ratio is a crucial factor in separating CNCs from CNFs. Strong acid was used to remove the CNCs, which had an aspect ratio of 5 to 50. The cellulose nanofibers generated by the mechanical method has an aspect ratio greater than 50 and keeps its original structure. 82 Both CNCs and CNFs have varying starting capabilities for adsorbing heavy metals. For instance, the maximal Cd2+ adsorption capacity of cellulose nanocrystals was around 6 mg/g, but that of cellulose nanofibers was 7.81 mg/g. 83 It was investigated that cellulose nanocrystals have a substantially higher maximum adsorption capacity for Cu2+ (47 mg/g) than cellulose nanofibers (14 mg/g). 84
Modified Nanocellulose: Adsorption Mechanism
To improve the material’s capacity to bind heavy metals, reactive surface components such as carboxyl, amino, quaternary ammonium salt, phosphate, etc. were successfully grafted onto nanocellulose after it had been modified. At high pH levels, the carboxyl group is more likely to adsorb heavy metal ions that are positively charged and cause deprotonation. Adsorption of negatively charged heavy metal ions was enhanced by positively charged amine groups because they protonated at low pH of two to 3. It was simpler for amine functional groups to attract heavy metal ions in their acid anion form.85–87 In the proper pH range, the majority of the reactive groups should non-selectively adsorb heavy metal ions.
Oxidization modification had shown a greater adsorption capability for CNCs in comparison to surface modification. This occurred because, in contrast to oxidized CNFs, the -COOH and -CHO groups produced by oxidation were more stable, and the structure of CNCs didn’t aggregate readily. However, the upper limit of the oxidized CNCs has received little attention, and it is significantly lower than that of surface grafting modification. The grafting chemicals in grafting reaction increased the nano cellulose’s adsorption site and may significantly improve the adsorption performance for both cellulose nanocrystals and cellulose nanofibers. Based on statistical data, the ‘graft to’ approach’s median adsorption capacities for cellulose nanocrystals and cellulose nanofibers (156.93 and 141.45 mg/g) were greater than that of the ‘graft from’ method (103.19 and 89.22 mg/g). Therefore, the ‘graft to’ method was more appropriate for the cellulose nanofiber based adsorbent strategy, with a maximum adsorption capacity of 1302.3 mg/g. 88 Nevertheless, because of the maximal adsorption capacity of 2207 mg/g and the high ceiling (abnormal point) the potential of CNCs altered by the ‘graft from’ technique is greater. 89
Heavy Metal Ions
List of heavy metals that have the potential to harm humans and the environments.

Clay, charcoal, polymers, and activated carbon are the main components of adsorbents. These adsorbents offer significant adsorption capabilities for particular heavy metal ions, but they also come with drawbacks, like undesired non-biodegradability, expensive production or regeneration energy costs, and secondary contamination. Thus, it remains desirable to find an efficient heavy metal ion adsorption technique and a simple process for bioadsorbent regeneration in order to promote green/sustainable growth. Because of their large specific surface area, excellent mechanical properties,98,99 and robust biocompatibility, nanocellulose-based adsorbents have become more and more popular in recent years. This makes them especially well-suited for the construction of heavy metal ion adsorbents. 100
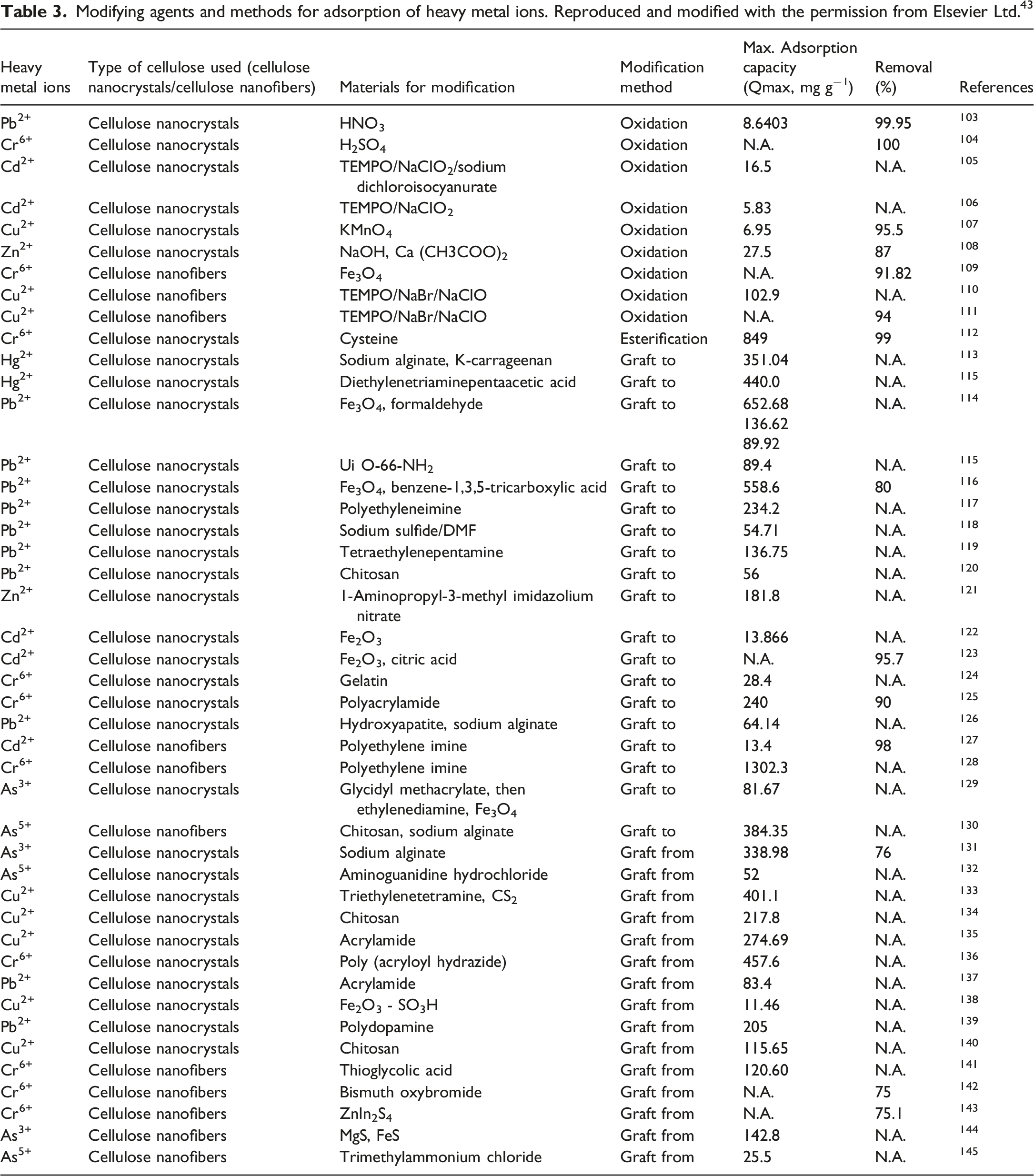
Modifying agents and methods for adsorption of heavy metal ions. Reproduced and modified with the permission from Elsevier Ltd. 43
Sensing of Heavy Metal Ions
Environmental monitoring is the sampling of air, water, and soil in a systematic manner. To assess the pollution effect, monitoring should be carried out; thus, sensors are essential. The presence of reactive hydroxyl groups and a large specific surface area makes the nanocellulose suitable for sensing applications. Cellulose materials like cellulose acetate can be doped with fluorescent materials to get pH sensors. Rhodamine B – functionalized core-shell ferrous nanoparticles are such type of fluorescent material. 146 Inherent adsorption and incorporated fluorescent properties of nanocellulose can be used in sensing heavy metals.
The nano cellulose-based fluorescent hydrogel is put in the aqueous solution containing heavy metals. Due to the natural behavior of cellulose nanofibers, the hydrophilic group induction causes the accumulation of heavy metals on the surface of the hydrogel. The hydrogels’ high-density amide, hydroxyl, and carboxyl groups are actively involved in accelerating the accumulation and adsorption of the heavy metals. The adsorption capacity of nanocellulose towards heavy metals was increased by the carboxylation and carbon dots grafting. 147 The nanocellulose hydrogels combined with the graphene quantum dots (GQD) give high sensitivity and great potential in photoluminescent sensing applications. 148 Reduced graphene oxide and nanocellulose blend were combined with NR latex to form an ordered 3D conductive structure that can be used as a chemical sensor for environmental applications. 149
Conductive hydrogels can be used in highly - sensitive temperature and strain sensors produced from Polyacrylamide – CNF/CNT – Glycerol. 150 Here, the Polyacrylamide acts as an elastic network, whereas CNF/CNT acts as a supportive and conductive network. The cationic nanocellulose – carbon nitride – Polyacrylamide hydrogel, which has a high sensitivity to detect human body motion, speech, and exhalation, was synthesized. TEMPO-oxidized CNF/CNT - Polyacrylamide hydrogels have fast responsiveness, long-term stability, and strain and pressure sensing ability. 151 The strain sensor with nanocellulose combining Polyacrylamide, sodium alginate, and calcium chloride has high mechanical properties, conductivity, and anti-freezing properties. 152
It is desirable to develop a tactile sensor that can sense the temperature and pressure simultaneously even though the temperature sensor and pressure sensor operate on different principles. 153 Nanocellulose can also detect 2, 4, 6 – trinitrotoluene (TNT) fabricated by incorporating gold nanoparticles and silver nanocubes on bacterial cellulose aerogels for several applications such as environmental monitoring and detection in research laboratories. 154 The iron oxide and CNCs were used as ultrasensitive sensors to detect NO2 because of their distinctive physical and electrical properties.155,156 Another essential type of sensor is a humidity sensor which can be used for monitoring humidity in industrial fields. A paper-based humidity sensor using cellulose nanofibers and carbon nanotubes as a sensitive layer and a microporous paper substrate was studied.
As different types of nanocellulose sensors are available, they require conductive properties for sensing applications. So it is essential to incorporate the conductive materials to induce sensing properties as discussed above.
Current Market Scenario, Challenges, and Prospects
The inclusion of nanocellulose promotes qualities like as biodegradability, low toxicity, enhanced strength and stiffness, and low weight to finished products. Nanocellulose could replace petroleum-based packaging, metallic parts, and other non-renewable materials because of their fascinating properties. The development of sustainable and eco-friendly bio-based products is an emerging trend that could create a big global impact on company investments and business decisions. In this context, nanocellulose-based functional constructs and composites are highly desirable in varying technological applications, especially in water treatment technologies. However, the research and development teams in the industries have been working to control the nanocellulose-based membranes on the homogeneity and pore size.
The present review reports the potential of nanocellulose as an excellent candidate for the adsorption of heavy metal ions from water. The major challenges associated with nanocellulose include the utilization of organic solvents, less dimensional stability due to its inherent hydrophilicity, mass production, and the commercialization of cellulose processable forms like aerogels, nanopapers, etc. Organic solvents are frequently used in the processing of nanocellulose for functionalization, modification, and dispersion. These solvents can raise manufacturing costs and present health and environmental problems. To meet this problem, it is imperative to create environmentally benign and sustainable solvent systems or discover substitute processing techniques that reduce the amount of solvent used. Nanocellulose exhibits high hydrophilicity due to its abundant hydroxyl groups, which can lead to dimensional instability in certain applications, especially when exposed to moisture. Strategies to enhance the dimensional stability of nanocellulose-based materials include chemical modification, cross-linking, or blending with hydrophobic polymers to reduce water uptake and improve mechanical properties.
While nanocellulose can be produced from various sources using different methods, scaling up production to meet industrial demand remains a challenge. Developing cost-effective and scalable production processes, optimizing raw material utilization, and improving process efficiency are essential for mass production of nanocellulose while maintaining product quality and consistency. Nanocellulose can be processed into various forms such as aerogels, nanopapers, films, and coatings with unique properties and applications. However, commercialization of these cellulose-based products faces several hurdles. Establishing standardized processing protocols, optimizing material properties, ensuring product reproducibility, and meeting regulatory requirements are critical steps in the successful commercialization of nanocellulose-based products.
Additionally, developing markets and creating awareness among potential end-users about the benefits and applications of nanocellulose products are essential for market penetration and adoption.
To prove that the nanocellulose materials are feasible and launch them into the market, more research needs to be done, specifically to: (1) optimize the entire process and develop new methods to produce new nanocellulose based materials; (2) use life cycle assessment to some environmental aspects of nanocellulose based materials; and (3) reduce the energy consumption process associated with the extraction of nanocellulose. Despite the difficulties outlined above, we anticipate that advancements in the next generation of materials will result in nanocellulose-based materials improving people’s quality of life.
Conclusion
Nanocellulose-based adsorbents, a new materials platform for removing heavy metal ions, have numerous benefits. Nanocellulose exhibits excellent adsorption properties, inherent non-toxicity, and biodegradability. Because of its vast source, large specific surface area, and great modification potential, nanocellulose has emerged as the current research hotspot in heavy metal adsorption from both a financial and environmental standpoint. Adsorbents based on modified nanocellulose have been demonstrated to remove heavy metals more efficiently than other adsorbents. Due to the large surface area and an abundance of functional groupings, nanocellulose can interact with various molecules. As a result, it can be used in various applications such as ultrafiltration, supercapacitors, memory devices, organic transistors, sensors, etc. Different types of sensors based on nanocellulose include humidity sensors, strain sensors, temperature sensors, pH sensors, gas sensors, pressure sensors, etc. for environmental monitoring, military, medical and food industry etc.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the The Kerala State Higher Education Council (Nava Kerala Post Doctoral Fellowship), Cochin University of Science and Technology (UJRF Fellowship).
