Abstract
Biodegradable thermoplastic polylactic acid (PLA) has recently been limited in its broader development and application due to its high cost. Therefore, it is crucial to develop low-cost and high-performance PLA-based composites by adding fillers. In this study, surface-modified incinerated sewage sludge ash (M-OH-ISSA) was employed as a reinforcing filler in PLA to prepare PLA/M-OH-ISSA thermoplastic composites with varying M-OH-ISSA ratios (10–30 wt%), and the mold mechanism for the PLA/M-OH-ISSA composites was explored. The surface of incinerated sewage sludge ash (ISSA) was modified to improve interfacial adhesion by NaOH alkali treatment and KH570 silane grafting, which successfully grafted silane groups onto the surface of the ISSA and converted the surface from hydrophilic to hydrophobic, strengthening the compatibility between the ISSA and PLA. The effects of the M-OH-ISSA content on the mechanical and thermal properties of the PLA composites were investigated. The mechanical test results showed that the tensile and flexural properties of the 15 wt% M-OH-ISSA-filled PLA composites were excellent compared to those of pure PLA, with strengths of 33.67 MPa and 73.70 MPa, respectively, which are all 1.6 times greater than those of pure PLA. Morphological analysis via SEM indicated that the composites with 15 wt% M-OH-ISSA exhibited good adhesion between the M-OH-ISSA and the matrix. The thermal performance of the composites was determined by using differential scanning calorimetry (DSC) and thermogravimetric analysis (TG/DTG). The results showed that the crystallinity of the composites increased with increasing M-OH-ISSA content and that the thermal stability between 290°C and 400°C decreased slightly with increasing M-OH-ISSA. However, the composites have good thermal stability near 200°C and do not undergo thermal degradation if processed at this temperature. As a filler in composites, M-OH-ISSA can reduce PLA usage and improve polymer properties, applying PLA/M-OH-ISSA composites in packaging and 3D printing applications.
Keywords
Introduction
The market demand for plastics has substantially increased during the current period of rapid economic growth, leading to a growing reliance on nonrenewable petroleum resources. 1 Being green, ecologically friendly, and sustainable has recently become popular, and as a result, academics have given biodegradable plastics much attention. Polylactic acid (PLA), a linear aliphatic thermoplastic polymer made from renewable resources such as corn and potato starch, is one of the most important alternatives to nondegradable plastics. 2 PLA has the advantages of high transparency, high modulus, high strength, biodegradability, and biocompatibility and has been extensively used in packaging applications, 3D printing, and medical devices. 3 However, compared with those of other petroleum-based polymers, the processing ability and wide application of PLA are restricted by its brittleness and high cost. 4 As a result, the development of degradable thermoplastic composites, using a valid method to improve the properties of composites, is highly important.
The commonly used modification methods for PLA include blending, incorporating nucleating agents, and performing thermal annealing treatment. 5 Combining treatment with a specific quantity of affordable materials is a practical approach for ensuring the high cost of PLA. 6 There have been numerous studies on mixing PLA with organic materials and inorganic substances. For instance, Altun et al. prepared PLA/pistachio shell biocomposites by pretreating pistachio shells with alkaline silane and adding them to PLA. 7 There has been significant research into introducing biomass degradation particles into polymer bodies, such as peanut shell powder, rice hulls, and soya bean powder; moreover, while these materials are promising, there are some drawbacks associated with the relatively high cost of manufacturing and poor performance when compared to polymers derived from non-renewable sources. 8 Razali et al. prepared PLA biocomposites using waste filler calcined shell (CSh) particles and studied the mechanical and thermal properties of the composites. 9 Loyo et al. modified CaO organically with oleic acid. They incorporated both into PLA at concentrations of 5 and 8 wt.% by a melting process and studied the antimicrobial and degradation properties of PLA/CaO composites. 10 Most studies on preparing PLA composites with added fillers have focused on natural fiber fillers. In contrast, few researchers have used solid waste inorganic materials as fillers including the incinerated sewage sludge ash used in this study.
Incorporating inorganic fillers as reinforcements into the polymer matrix has proven to be an effective way to improve the properties of composites.11,12 Among the many inorganic fillers employed as reinforcements for composites, solid waste fillers are receiving increasing interest. 13 Because of its favorable features, such as heat resistance, excellent mechanical strength, and chemical resistance, solid waste fillers can considerably increase the mechanical properties and heat resistance of polymer composites as reinforcing fillers. For example, Maurya and Manik found that the addition of fly ash in the preparation of polypropylene composites further enhanced the interfacial interaction between PP and nylon fibers, which resulted in a 29 % increase in tensile strength and a 49% increase in flexural strength of the composites. 14 Incinerated sewage sludge ash (ISSA) is a solid waste generated from municipal sludge incineration that is readily available, produced in enormous quantities, and primarily disposed of in landfills with minimal resource use. 15 ISSA comprises multifunctional active oxides, including silica and alumina, and its composition, mineralogy, and surface chemical activity all play a role in its application. 16 As a result, incorporating ISSA in a polymer matrix is a novel application direction. The mechanical and thermal performance of composites can be improved by adding a suitable quantity of solid waste filler, which also helps with the use of solid waste resources, the transformation of trash into gold, and the lowering of the price of plastics in some cases. 17 There have been studies on the addition of solid waste fillers to PP, epoxy resin, PVC, and other matrices.18–21 However, studies on the addition of ISSA to the PLA matrix are rare.
However, the interfacial compatibility was found to be insufficient; a surface modification of the ISSA was still needed. Although inorganic fillers, including ISSA, have shown great potential as mechanical reinforcing fillers, the hydrophilic hydroxyl groups on their surface could lead to poor compatibility with hydrophobic polymer matrices. 22 This poor compatibility is a great obstacle, that prevents inorganic fillers from reaching their theoretical potential. The ISSA needs to be thoroughly dispersed in the PLA matrix using the right dispersants to enhance the interfacial properties between the ISSA and polymer matrices. However, the hydroxyl groups on the surface of ISSA also make it possible to introduce modifiers for chemical grafting on the surface of ISSA. The most common and typical surface modification is the silane coupling agent, which has been shown to significantly increase dispersion when it reacts with the hydroxyl groups on the surface of the ISSA. 23 The silane coupling agent acts as a “bridge” to connect the ISSA and PLA, improving the interfacial compatibility of the two phases.
In this study, ISSA was treated with NaOH and a silane coupling agent (3-methacryloxypropyltrimethoxysilane, KH570). Then, using the melt blending method, PLA/M-OH-ISSA composites were fabricated by adding 10%, 15%, 20%, 25%, and 30% M-OH-ISSA to PLA. Fourier transform infrared spectroscopy (FTIR), contact angle measurements, and scanning electron microscopy (SEM) were used to determine the surface characteristics of the M-OH-ISSA. Mechanical property tests, differential scanning calorimetry (DSC), thermogravimetric tests (TG/DTG), and SEM were used to determine the mechanical characteristics, thermal properties, and fracture morphology of the PLA/M-OH-ISSA composites. Moreover, a comparison with composite including the raw ash without the treatment was made Finally, the PLA/M-OH-ISSA composite cost and molding mechanism were explored.
Experimental
Materials
Polylactic acid (PLA) Ingeo™ 4032D grade was obtained from China (density 1.24 g/cm3) and was produced by LLC Nature Works (USA). The silane coupling agent (3-methacryloxypropyltrimethoxysilane, KH570), anhydrous ethanol, hydrochloric acid (HCl), and sodium hydroxide (NaOH) were purchased from Sinopharm Chemical Reagent Co., Ltd, (China). Incinerated sewage sludge ash (ISSA) was supplied by Xin Kun Bioenergy Cogeneration Co. in China.
Preparation
Figure 1 shows a schematic diagram of the preparation of the PLA/M-OH-ISSA composites. There are two main steps: first, alkali silane modification of the ISSA is performed, and then the PLA/M-OH-ISSA composites are prepared using surface-modified ISSA. Schematic diagram of the preparation of the PLA/M-OH-ISSA composites. (a) Preparation of M-OH-ISSA. (b) Preparation of PLA/M-OH-ISSA composites.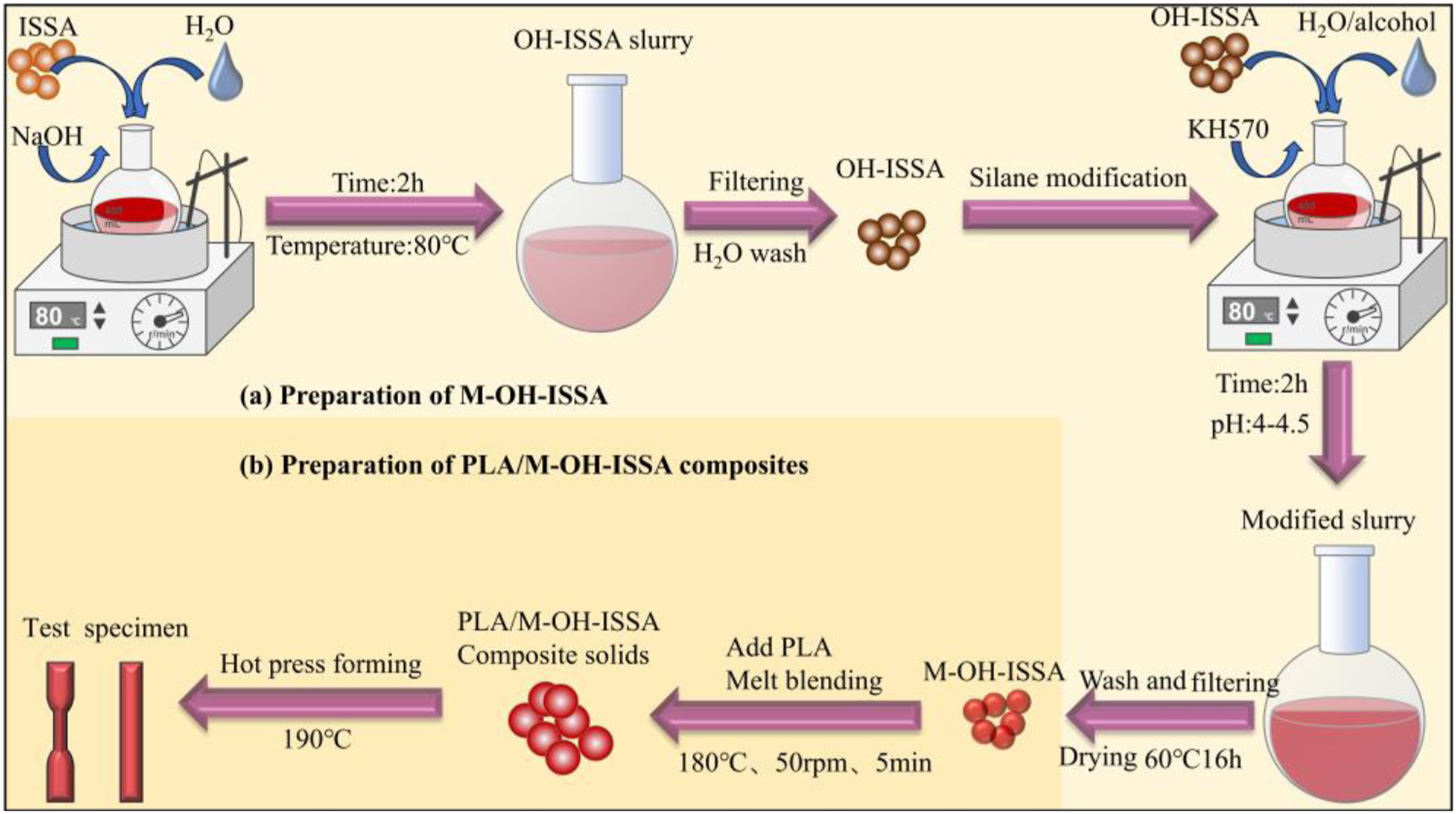
Preparation of alkali-silane-modified incinerated sewage sludge ash (M-OH-ISSA)
The process of alkali-silane treatment was similar to that previously described by Xue et al. for the treatment of fly ash, 24 but we have simplified the method of operation and used different modifiers and dosages. First, 0.1 mol NaOH was added to a round-bottom flask containing 400 mL of deionized water, and the OH-ISSA slurry was held at a constant temperature in an 80°C water bath with rapid stirring for 2 h. The OH-ISSA slurry was filtered and washed with deionized water until the pH of the filtrate was neutral. Next, an appropriate amount of HCl was used to prepare an ethanol solution (1:1 vol ratio of ethanol to water) with a pH of approximately 4, and 5 wt% of the silane coupling agent KH570 (relative to ISSA) was hydrolyzed in the ethanol solution. Then, a certain amount of OH-ISSA was sequentially added to the round-bottomed flask, and the modified slurry was stirred at 80°C for 2 h. Finally, the reaction products were washed with ethanol and deionized water successively and filtered to obtain a wet sample (M-OH-ISSA), which was dried at 60°C in an electrically heated constant-temperature blast drying oven.
Preparation of PLA/M-OH-ISSA composites
Formulations of the composite specimens.

Characterization of ISSA and M-OH-ISSA
The ISSA was characterized using a laser particle sizer (Model: Bettersize2600) and X-ray diffraction (XRD) analysis to determine its particle size and crystal structure. The ISSA was well dispersed with deionized water and the background was tested after the laser particle sizer had reached the appropriate refractive index. Then, the ISSA was added dropwise to the ultrasonic chamber. Continuous testing was performed, and the data were exported. XRD analysis was carried out with an XRD instrument (Model: Smart Lab SE) with a Cu-Kα radiation source, working voltage, and current of 40 kV and 30 mA. The ISSA was scanned with a Bragg angle (2θ) ranging from 5° to 90° to obtain the standard diffraction peaks.
Fourier transform infrared (FTIR) spectroscopy was performed to determine the structures of ISSA and M-OH-ISSA. The FTIR spectra were obtained from Thermo Fisher Scientific Ltd (China). High signal-to-noise spectra in the 4000–500 cm−1 region could be obtained with 32 scans and a resolution of 0.5 cm−1.
A contact angle meter (JC2000D1, Shanghai Zhong Chen Digital Technology Equipment Co., Ltd, China) was used to test the hydrophilicity/hydrophobicity of ISSA and M-OH-ISSA to characterize the modification effect. The powder samples to be tested were pressed on a tablet press at a pressure of 15 MPa to form a smooth surface and tight flakes and the contact angle was measured using a distilled water droplet. Each sample was tested three times, and the average value was taken.
A scanning electron microscope (SEM) (Model: su1510) was used to examine the morphology of the ISSA and M-OH-ISSA. A small layer of gold was applied to the surface of each sample.
Mechanical characterization of PLA/M-OH-ISSA composites
The tensile and flexural tests of the composites were carried out with a universal material tester (Model: E43, Iseed Automation Instruments Ltd, China.). According to the ISO-527-2-2012 1BA standard, the tensile strength of the specimens was tested, and the specimens were dumbbell-shaped, with a size of 75 × 10 × 2 mm, and a test speed of 5 mm/min. Similarly, for the bending test following the ISO178-2010 standard, the specimen size was 80 × 10 × 4 mm and the speed of the bending strength test was 10 mm/min. The reported values were obtained from five independent tests.
A scanning electron microscope (SEM) (Model: su1510) was used to examine the fracture morphology of PLA and the composites with the best mechanical performance. To improve electrical conductivity for SEM imaging, a small layer of gold was applied to every sample.
Thermal characterization of the PLA/M-OH-ISSA composites
The melting and crystallization properties of the composites were studied using a differential scanning calorimeter (Q2000, TA, USA) under a nitrogen gas stream at 50 mL/min. Experiments were carried out using 3–10 mg samples in an aluminum crucible. First, the samples were held at 30°C for 5 min and then scanned from 30°C to 200°C at a heating rate of 10°C/min. Subsequently, the samples were cooled to 30°C at a rate of 10°C/min. The following equation was used to calculate the crystallinity of the composites: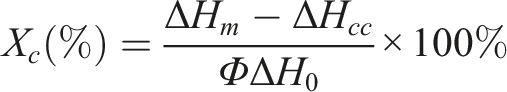
Thermal stability testing was performed on a fully automated thermogravimetric analyzer (Mettler TGA2, Mettler-Toledo, Switzerland) at a 50 mL/min flow rate under a nitrogen atmosphere. Samples of 3–10 mg were heated from 30°C to 600°C at a rate of 10°C/min. The relative mass loss of the material during heating was recorded as a function of temperature.
Rheological characterization of PLA/M-OH-ISSA composites
The rheological properties of these materials with small amplitudes were tested by a rotary rheometer (Physica MCR301, Austria). Tested with dynamic frequency sweeps. The parallel plate fixture was 25 mm long and the plate spacing was 2 mm. The frequency scanning parameters were set at 200°C, a strain amplitude control of 1%, and a frequency scanning range of 10–100 rad/s.
Results and discussion
Characterization of ISSA and M-OH-ISSA
Particle size analysis
The particle size distribution of ISSA is depicted in Figure 2 and summarized in Table 2. 10% of the particles are smaller than 4.38 μm, 50% are smaller than 24.67 μm, and 90% are smaller than 71.86 μm. On average, the particle size of ISSA is 24.67 μm. In a previous study, Alghamdi developed high-density polyethylene (HDPE)/fly ash composites and found that the use of small (50–90 µm) particles of fly ash could give rise to the tensile modulus (∼95%) and tensile strength (∼7%) of their reinforced composites when compared to neat HDPE materials.
26
This makes it possible to reinforce PLA composites with ISSA (24.67 μm) as filler. Particle size distribution of ISSA. Particle size distribution data for ISSA.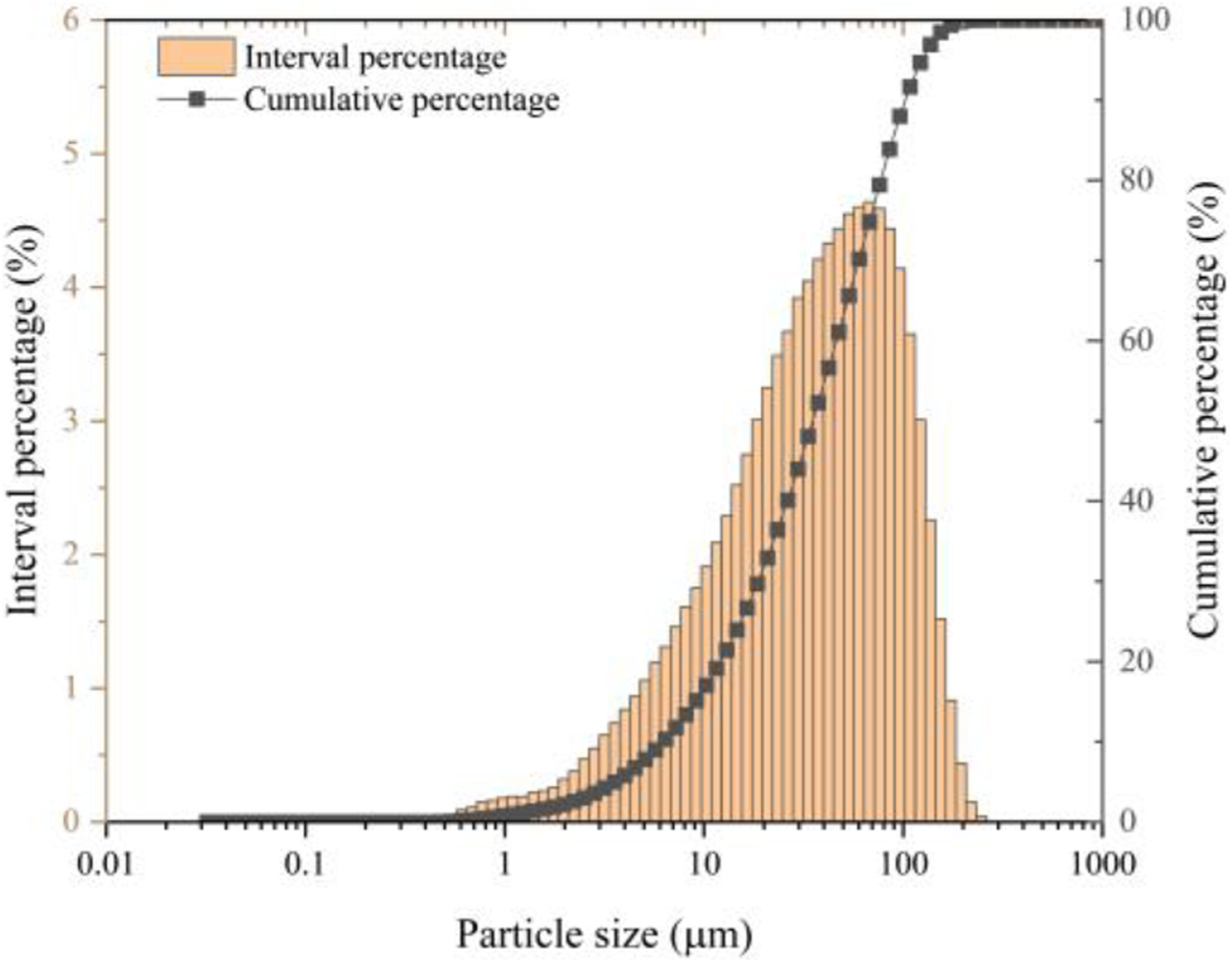

X-ray diffraction (XRD) analysis
X-ray diffraction characterization was performed to elucidate the crystal structures present in ISSA. Figure 3 displays the XRD pattern, which shows prominent characteristic peaks corresponding to the three phases of SiO2, Fe2O3, and Al2O3. Additionally, a minor amount of calcium sulfate is observed. Specifically, the SiO2 phases exhibit significant 2 theta peaks at 21°, 26.7°, and 49.7°, while the Al2O3 phase manifests major peaks at 33.3°, 35.7°, and 49.7°. Fe2O3, which exists in the form of magnetite and hematite, has 2 theta peaks at 33.3°, 35.7°, and 54.3°.
27
Remarkably, these findings align closely with those reported in the literature concerning fly ash, indicating consistency and reliability in the characterization results.
28
X-ray diffraction patterns of ISSA.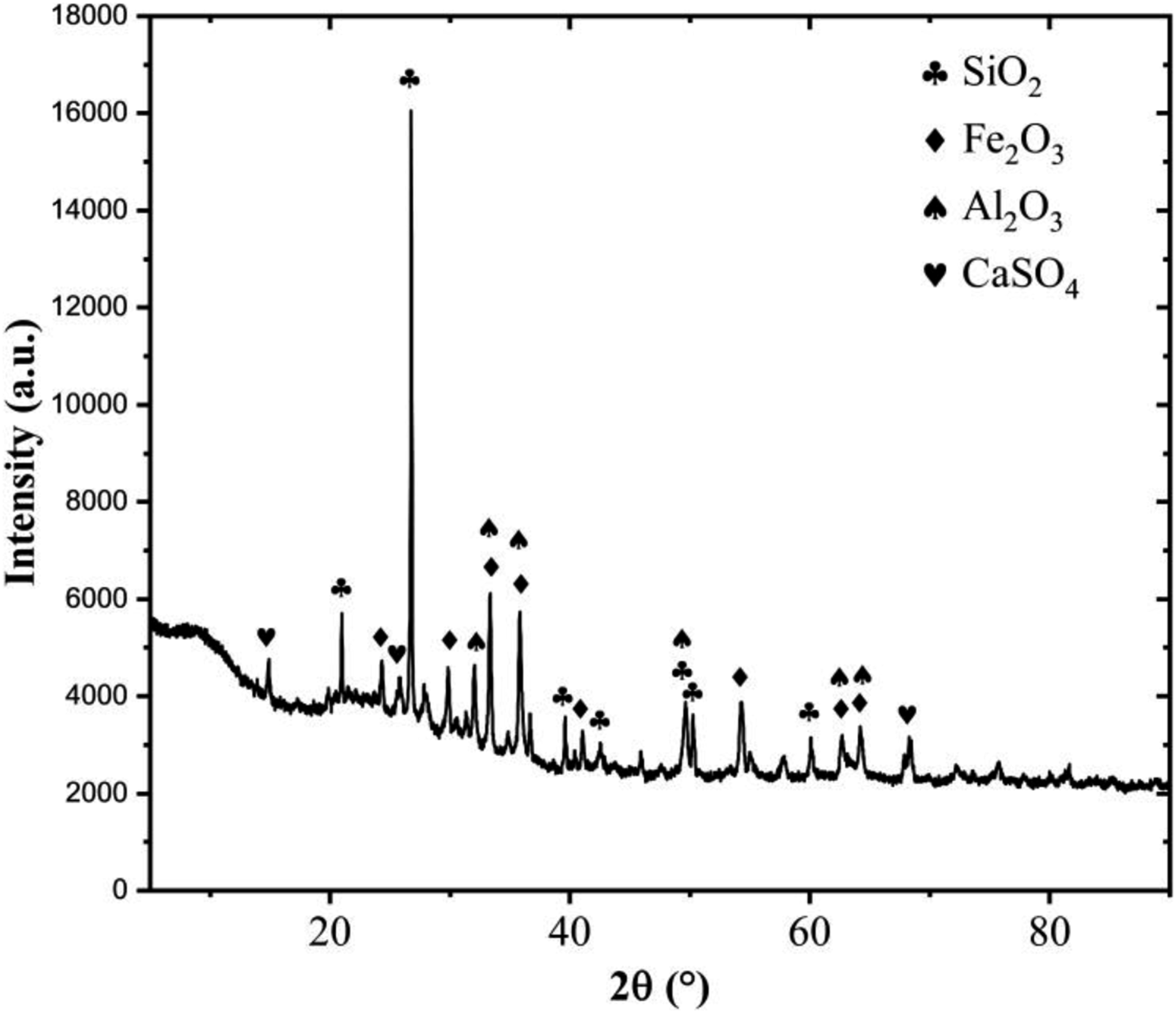
Fourier transform infrared (FTIR) spectroscopy analysis
The infrared spectra of ISSA and M-OH-ISSA are shown in Figure 4(a), and the local magnification in the 1600–1740 cm−1 wavelength range is shown in Figure 4(b). The identical peaks at 1110 cm−1 and 500–800 cm−1, correspond to the asymmetric stretching vibration peaks of Si–O–S,
29
the vibrational peaks of the Al–O bond,
30
and the Fe-O bond,
31
respectively, because there is no change in the main components of ISSA before and after modification. The telescopic and bending vibrational peaks of the hydroxyl group are at 3447 cm−1 and 1636 cm−1, respectively.
32
Clearly, the presence of hydroxyl groups in both ISSA and M-OH-ISSA results from the presence of some of these groups on the surface of ISSA and from the persistence of some O-H bonds following hydrolysis and reaction of the coupling agent with ISSA. More significantly, a distinctive KH570 peak was observed for M-OH-ISSA. These results indicate that KH570 was successfully grafted onto the surface of ISSA. The lower intensity peaks at 2929 cm−1 and 2858 cm−1 correspond to the C-H stretching of the methyl group, and the C-H stretching of the methylene group, respectively. The vibrational peak corresponding to the double bond was at 1650 cm−1, and the vibrational peak corresponding to the formyl group was at 1714 cm−1.
33
Fourier transform infrared spectra at 500–4000 cm−1 (a) and localized magnification at 1600–1740 cm−1 (b) of ISSA and M-OH-ISSA.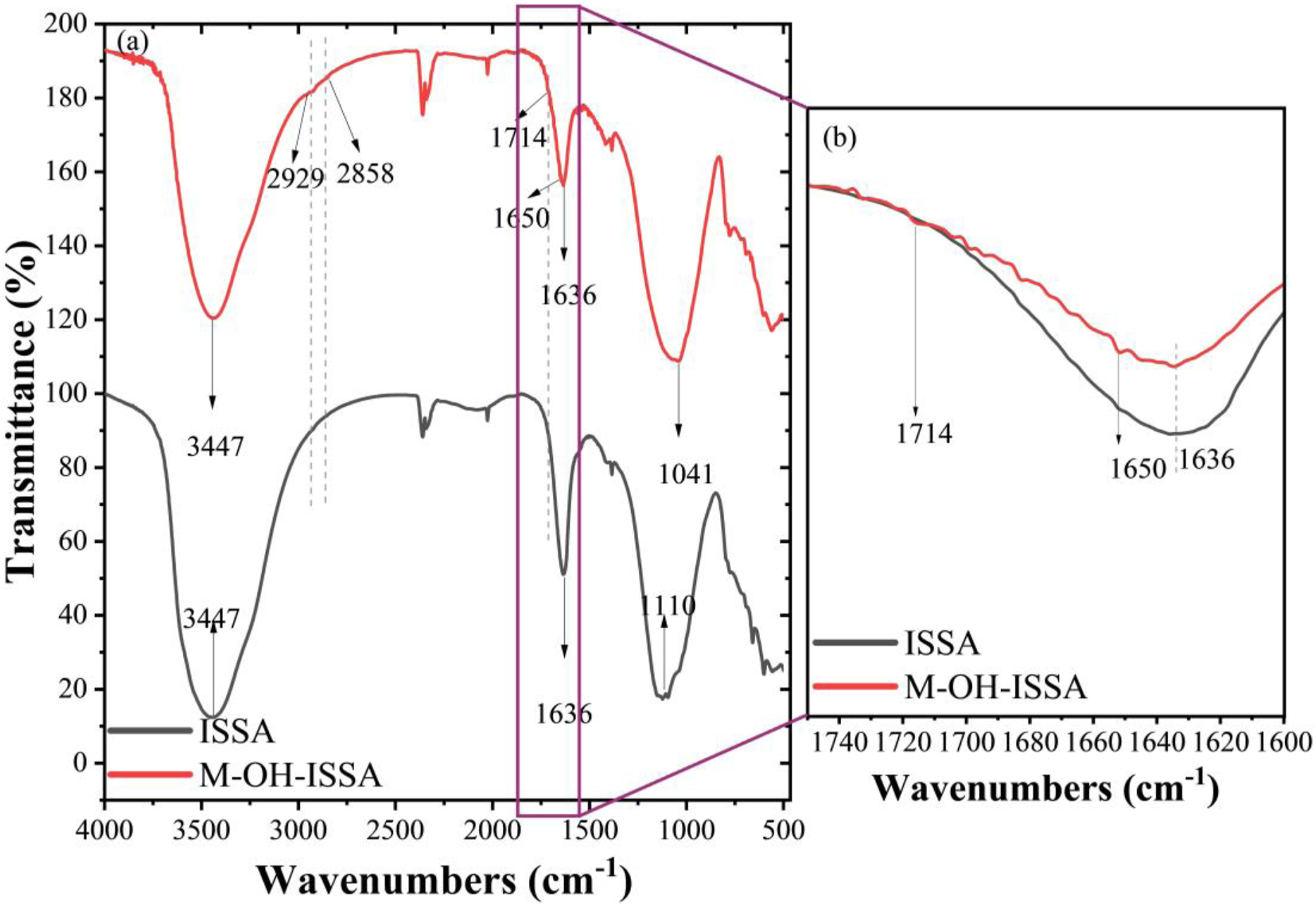
Hydrophilic/hydrophobic
Incinerated Sewage Sludge Ash (ISSA), characterized by its porous nature and inherent polar groups,
34
exhibits pronounced hydrophilicity with a contact angle of 13.57°. This hydrophilic behavior undergoes a notable transformation, as illustrated in Figure 5, where the contact angle of the surface-modified ISSA substantially increases to 97.35°, indicative of enhanced hydrophobicity. This shift toward hydrophobicity is attributed to the introduction of the KH570 silane coupling agent. The amphiphilic properties of this coupling agent play a pivotal role in altering the water absorption characteristics on the surface of ISSA upon grafting.
35
This modification not only mitigates water absorption but also contributes to the development of a hydrophobic surface. Simultaneously, the porous structure of ISSA underwent surface modification, further amplifying its hydrophobic properties. Contact angles of the ISSA and M-OH-ISSA.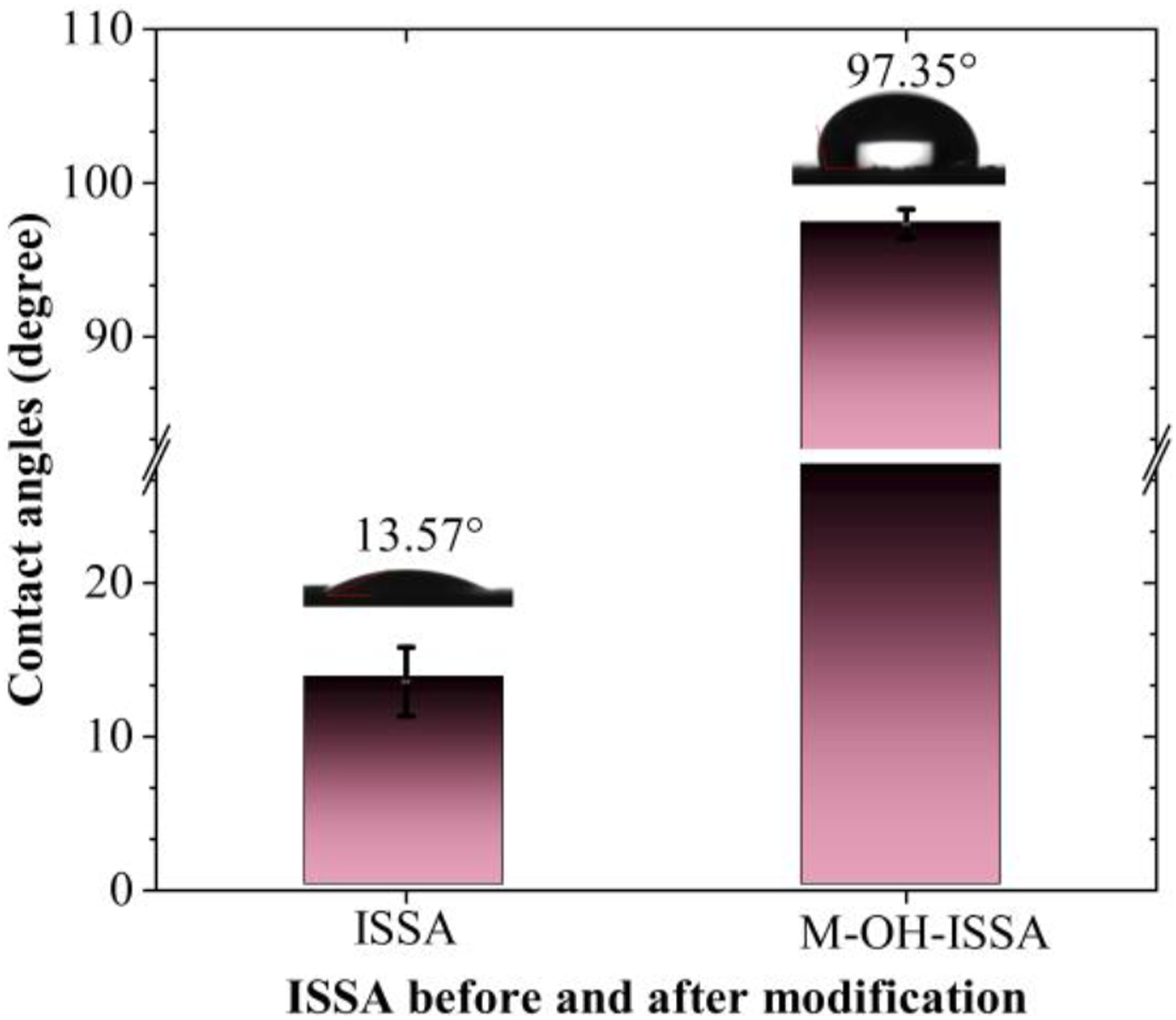
Morphological analysis
Scanning Electron Microscopy analysis was employed to scrutinize the surface morphologies of ISSA before and after surface modification. Figure 6(a) reveals the presence of numerous minute particles on the surface of untreated ISSA. After surface modification, the texture on the M-OH-ISSA surface becomes discernible, as depicted in Figure 6(b). Notably, the M-OH-ISSA exhibited a roughened surface with reduced granularity, indicative of the successful modification. Scanning electron microscopy images of the ISSA(a) and M-OH-ISSA(b).
Furthermore, the SEM images highlight the presence of a distinct coupling agent layer on the surface of M-OH-ISSA. This layer is visible, suggesting that the coupling agent, likely the KH570 silane coupling agent, effectively adhered to the ISSA surface. The introduction of this coupling agent contributed to the observed changes in surface morphology, leading to a rougher texture and reduced granularity.
Mechanical properties of the PLA/M-OH-ISSA composites
Tensile properties
Figure 7 displays the tensile qualities of the PLA/M-OH-ISSA composites, including the tensile strength, tensile modulus, and strain at break. PLA/M-OH-ISSA composites have better mechanical properties than untreated composites. The poor compatibility of hydrophilic ISSA with hydrophobic PLA matrix may lead to particle aggregation or agglomeration. Aggregates produce defective sites in the composites and thus no improvement in mechanical properties is observed.
36
The tensile strength of the PLA/M-OH-ISSA composites increases with increasing M-OH-ISSA content when less than 15 wt%. This is due to the significantly improved M-OH-ISSA dispersion in the PLA matrix following surface modification, as well as the efficient reaction between the methyl acryloyl oxy groups on the M-OH-ISSA particles and the groups on the PLA, which increases the amount of M-OH-ISSA. Above 15 wt%, the tensile strength of the composites decreases with increasing M-OH-ISSA content. This is because as the amount of filler increases, aggregation will occur more easily. Aggregation-induced stress-concentrated regions will propagate cracks at lower stresses, and these defects will obstruct the effective transfer of stresses during tensile testing, ultimately causing the composites to fracture.
37
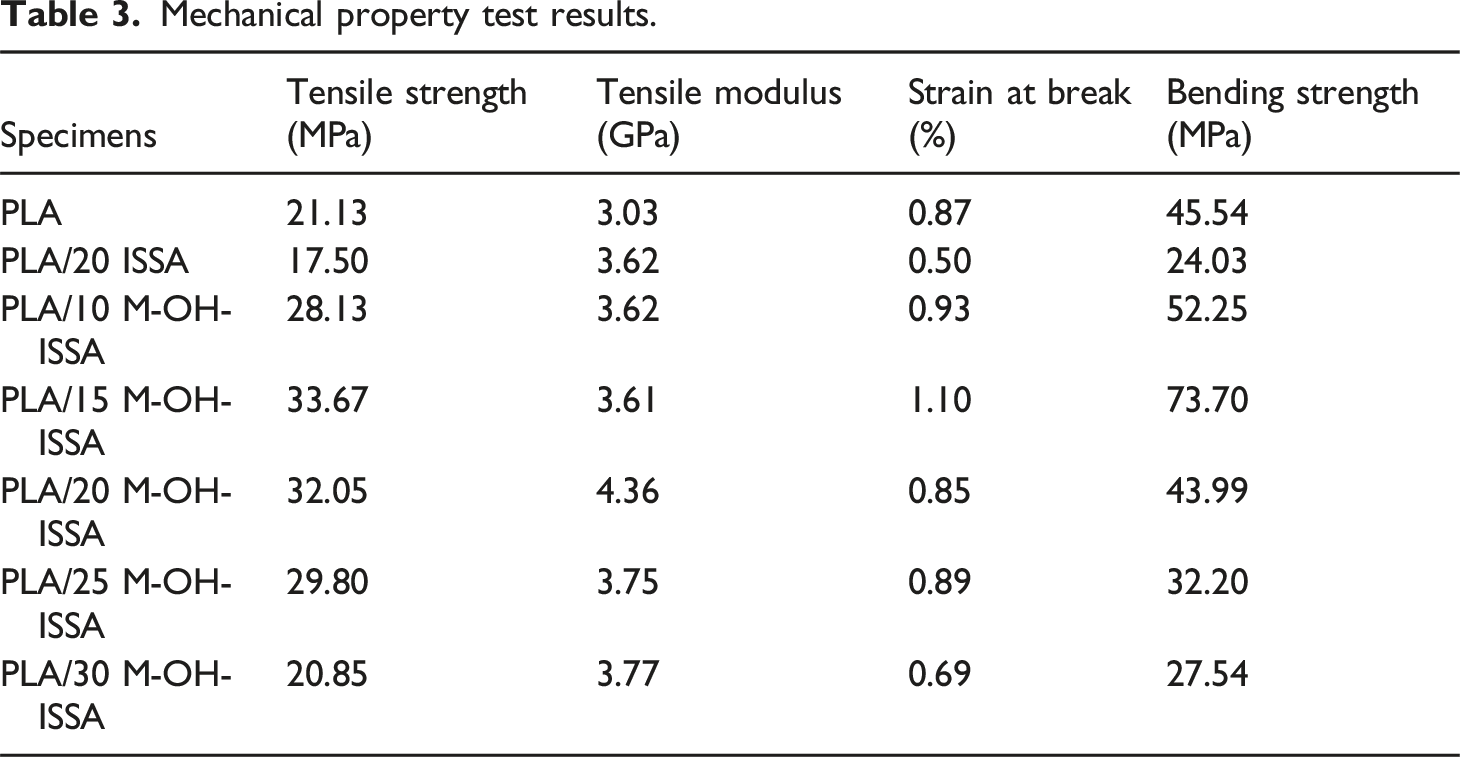
The maximum tensile strength of 33.67 MPa for the PLA/M-OH-ISSA composites is achieved at the M-OH-ISSA content of 15 wt %, which is 1.6 times greater than that of pure PLA. Table 3 lists all the mechanical property analysis values. Tensile properties of the PLA/M-OH-ISSA composite. Mechanical property test results.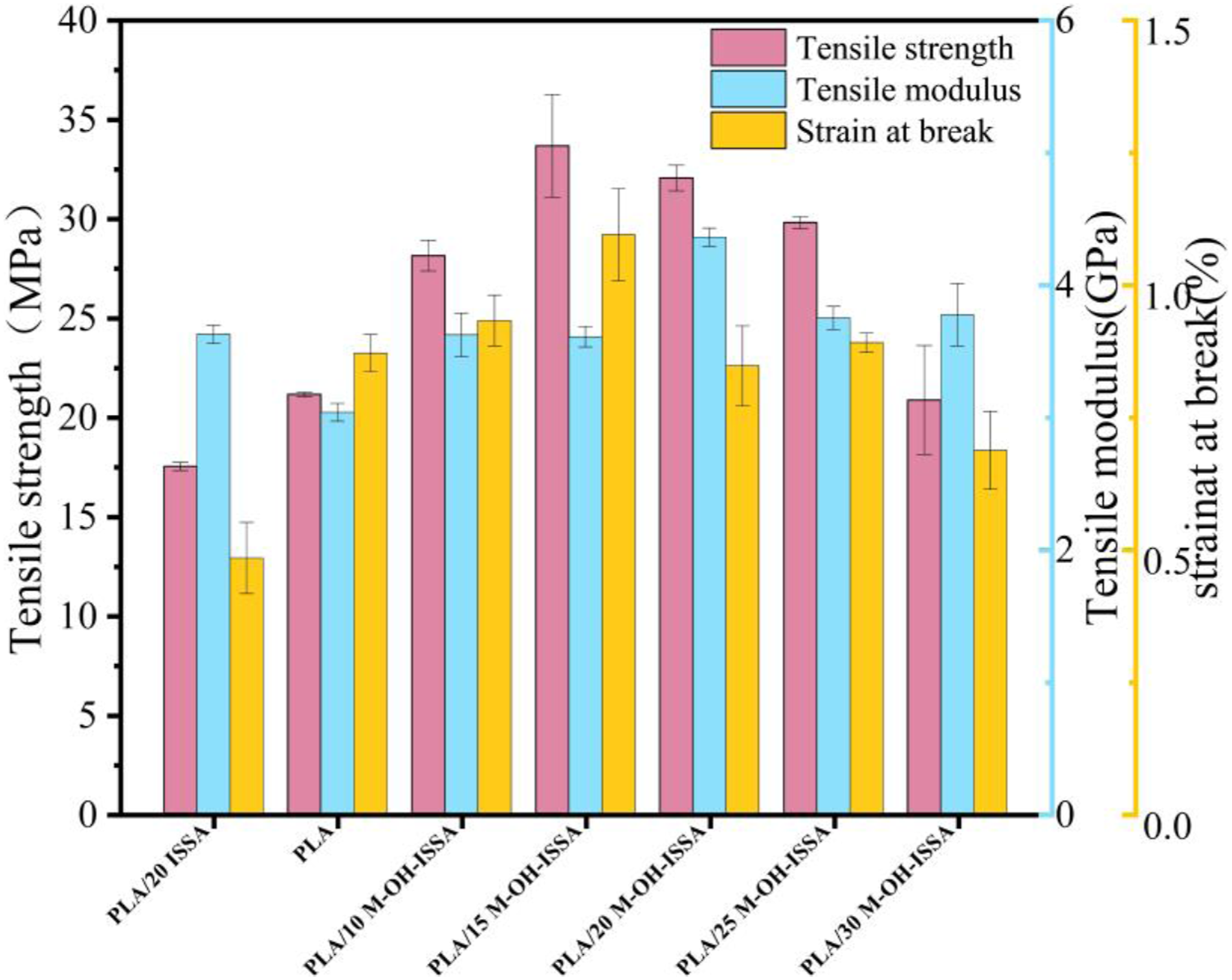
Strain is a parameter used to measure the toughness of composites. 38 As the M-OH-ISSA content increases, the strain at break exhibits the same trend as the tensile strength. These findings suggest that the interfacial properties between M-OH-ISSA and PLA improved. The silane molecules grafted between the M-OH-ISSA and the PLA molecules played a bridging role, facilitating the transfer of stress and strain under tension. 39 Additionally, the trend in tensile modulus is not obvious in contrast to the increases in strain at break. The performed tests showed a significant increase in stiffness, as indicated by the tensile modulus in highly filled samples, which is consistent with the findings of Andrzejewski et al. in talc filler. 40 However, the tensile modulus was still higher than that of pure PLA.
Flexural strength
The results of the bending strength tests for the composites are displayed in Figure 8. The tendency of the flexural strength to increase was similar to that of the tensile strength, in the study of natural fiber-reinforced polymers similar conclusions were obtained.
41
The bending strength of the PLA/M-OH-ISSA composites increases with increasing M-OH-ISSA content when less than 15 wt%. This is attributed to the fact that alkali silane modification improves the M-OH-ISSA surface adhesion characteristics by grafting large numbers of silane groups onto the surface of M-OH-ISSA.
42
Increasing the PLA/M-OH-ISSA interfacial compatibility provides effective resistance to bending and thereby improves the strength of these composites. When the M-OH-ISSA content was greater than 15 wt%, the flexural strength of the composites decreased with increasing M-OH-ISSA content. This finding demonstrated that the aggregation of M-OH-ISSA particles began at a 20 wt% content. The addition of a high filler content increases the probability of filler agglomeration, which leads to the poor dispersion of M-OH-ISSSA particles within PLA. A maximum bending strength of 73.70 MPa was obtained for the PLA/M-OH-ISSA composites at the M-OH-ISSA content of 15 wt%, which is 1.6 times greater than that of pure PLA. Due to the rigidity of the ISSA itself, the flexural strength of these composites is increased, and the rigidity of the whole composite is improved after the addition of M-OH-ISSA.
43
Flexural properties of the PLA/M-OH-ISSA composite.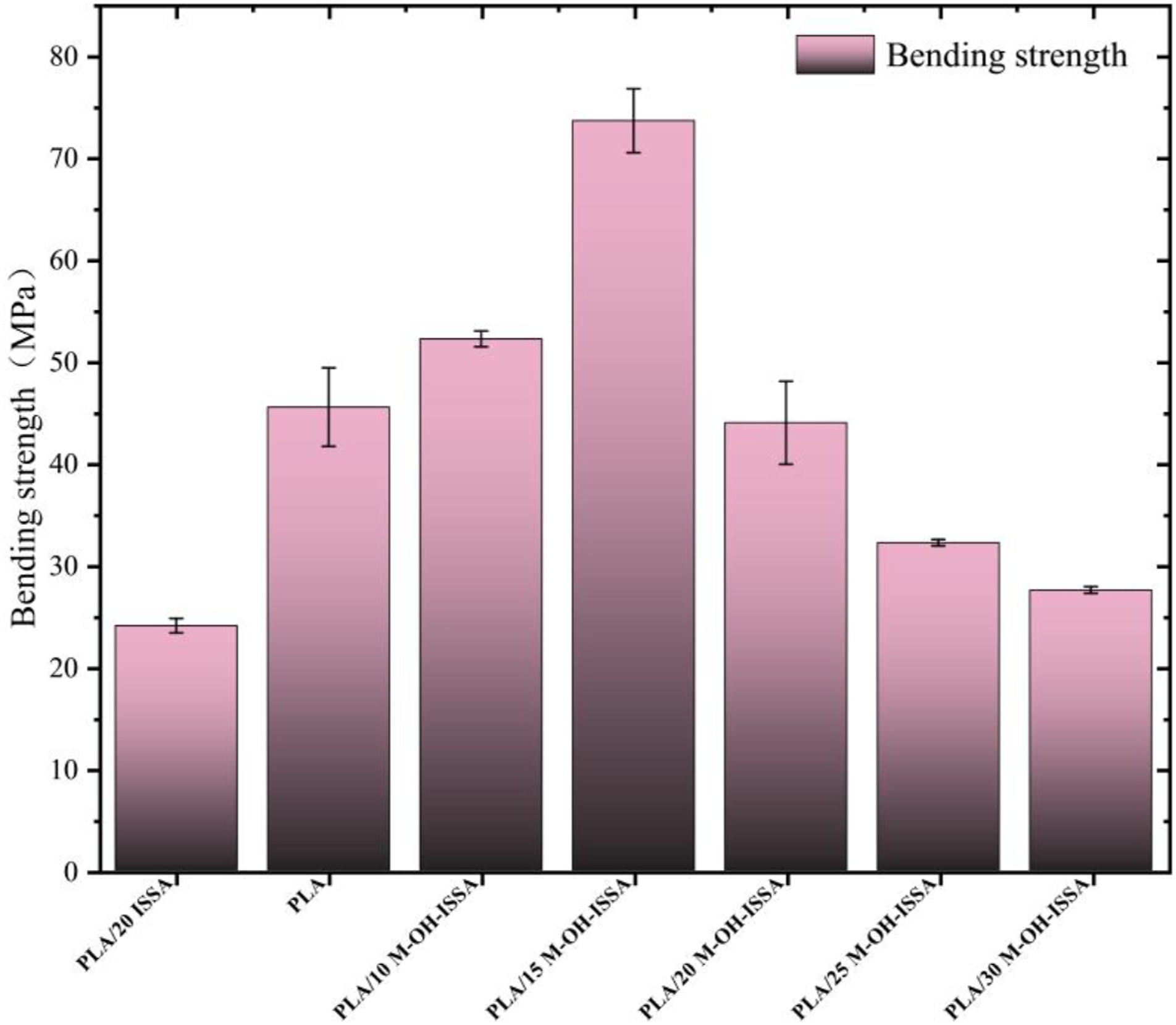
Morphological analysis
It was found that PLA/M-OH-ISSA composites with a content of 15 wt% demonstrated the best mechanical properties based on the tensile and flexural findings of the composites with varying M-OH-ISSA contents. The fracture morphology of PLA/15 M-OH-ISSA is shown in Figure 9(a) and (b). SEM micrographs of PLA/15 M-OH-ISSA revealed that the surface of M-OH-ISSA was wrapped with the PLA matrix thus indicating improved wettability of M-OH-ISSA particles. Due to sufficient surface wetting properties, the structure of the M-OH-ISSA is relatively dense, and the polymer matrix has a high holding capacity.
44
This phenomenon can be explained by the similar facts of silane treatment of ISSA which provides a bridging effect with the polymer matrix.
45
The improved interfacial interaction between M-OH-ISSA and PLA can also be corroborated by the mechanical findings reported in the earlier sections. Moreover, Figure 9(b) shows that ISSA was homogeneously dispersed in the PLA matrix. This is because the coupling agent modification promotes the dispersion of the filler particles by changing the hydrophilicity of the inorganic filler and reducing the specific surface energy.
46
However, significant interstitial gaps can be observed in the fracture surfaces of the untreated PLA/ISSA composites as shown in Figure 9(c). Moreover, the SEM image of virgin PLA exhibited a brittle fracture pattern as shown in Figure 9(d). There were some distinct river markings on its fractured surface. The smooth, laminated surfaces confirm the inherent brittle character of pristine PLA.
47
The mechanical properties of the composites with a given amount of M-OH-ISSA addition were significantly affected by the decrease in porosity and increase in ISSA-polymer interfacial adhesion. Scanning electron microscopy images of the PLA/15 M-OH-ISSA (a) (b); PLA/20 ISSA (c) and PLA (d).
Thermal properties of PLA/M-OH-ISSA composites
Differential scanning calorimetry (DSC)
The effect of the M-OH-ISSA as a filler on the crystallization and melting behavior of the composites was explored by DSC. Figure 10 displays the DSC plots of PLA/M-OH-ISSA composites with various ratios of M-OH-ISSA. Table 4 displays the thermal parameters, including glass transition temperature (T
g
), cold crystallization temperature (T
cc
), melting temperature (T
m
), enthalpy of melting (ΔH
m
), enthalpy of crystallization (ΔH
cc
), the degree of crystallinity (X
c
). DSC melting curve of PLA/M-OH-ISSA composites (a) and DSC cooling curve of PLA/M-OH-ISSA composites (b). DSC data of the PLA/M-OH-ISSA composites.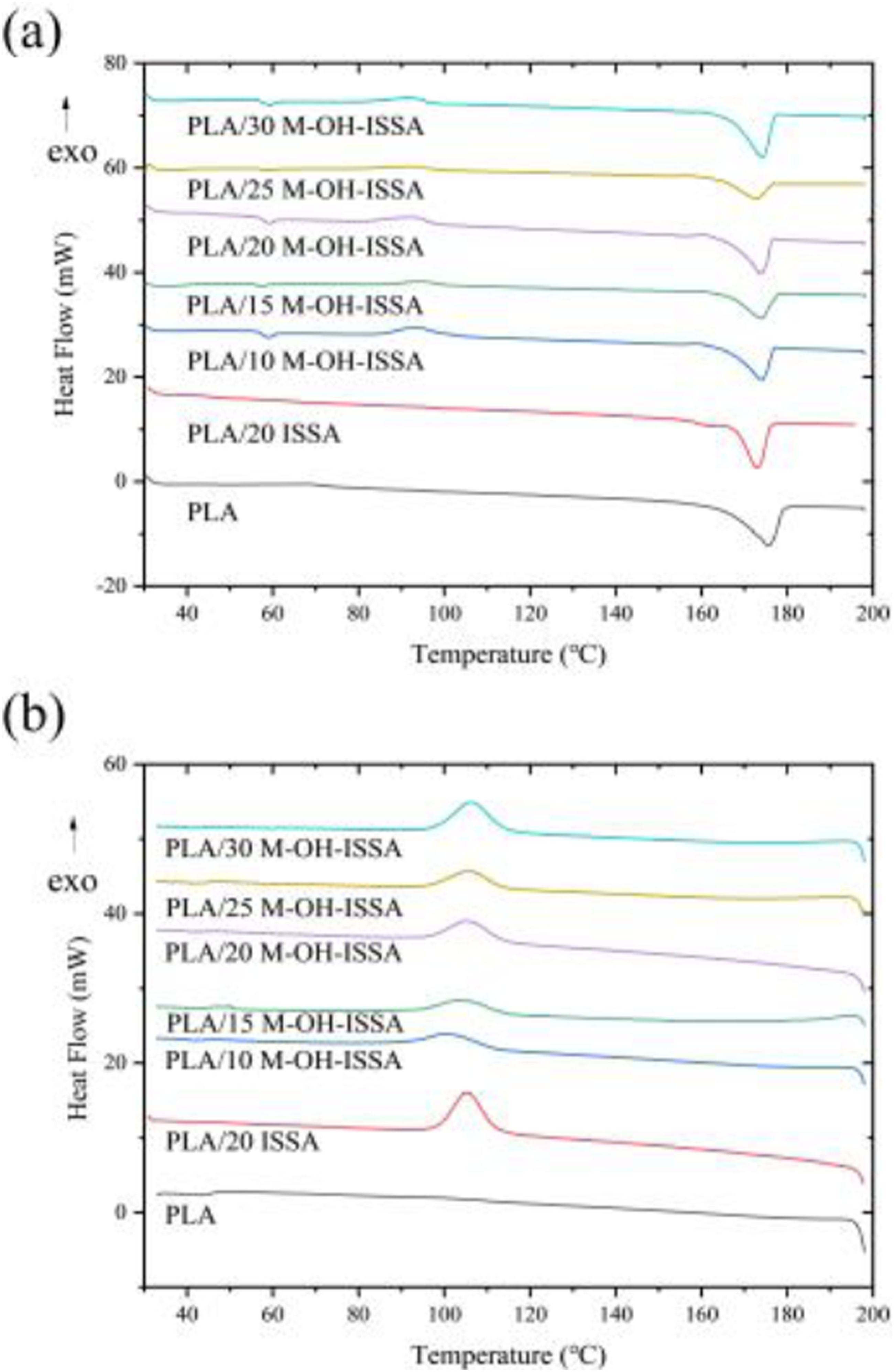
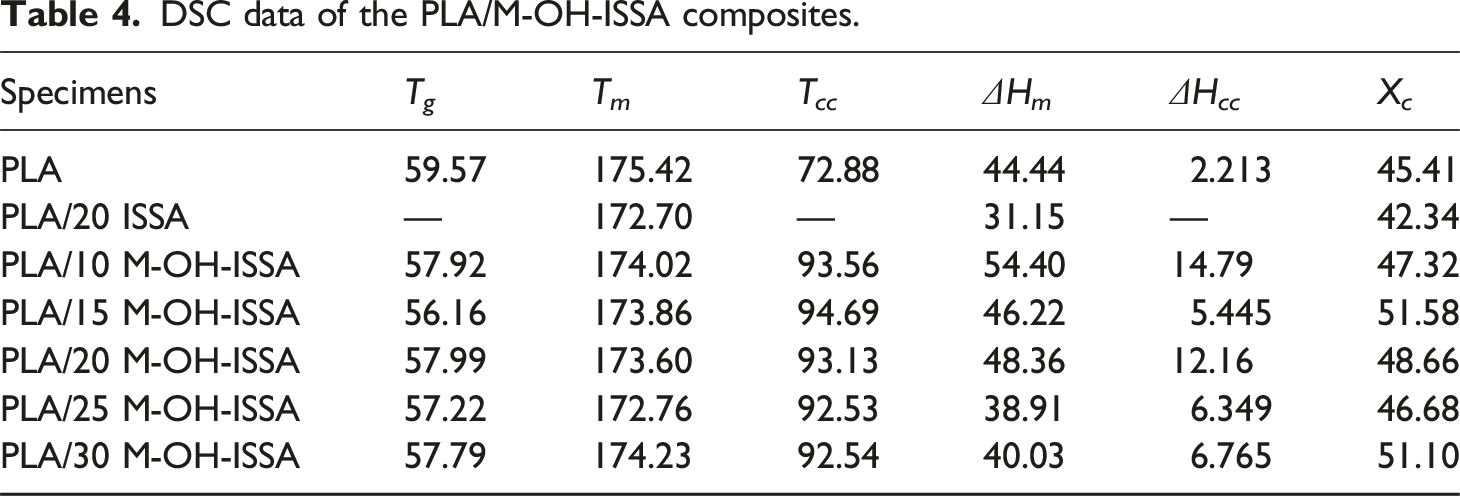
The crystallinity of the composites gradually increased with increasing M-OH-ISSA content. M-OH-ISSA exists as a non-homogeneous particle in the PLA matrix and undergoes heterogeneous nucleation to enhance the molecular chain segment, resulting in crystallinity after the addition of M-OH-ISSA. 48 After the addition of modified M-OH-ISSA, the T cc of the composites was increased compared to that of PLA, and the cold crystallization temperature of the composites increased, indicating that the addition of M-OH-ISSA weakened the chain segment movement ability of the composites, which made the composites crystallizable at higher temperatures. The reason for this is the difference that there are chemical bonds or entanglements between the silane-modified M-OH-ISSA and the PLA chains, therefore, the movement of the PLA chains is restricted and the cold crystallization of the composites becomes more difficult. 49 Moreover, the Tcc of the composites slightly increased when the content of M-OH-ISSA was less than 15 wt %, and gradually decreased when the content of M-OH-ISSA was more than 15 wt %. This is because the increase in the content of M-OH-ISSA, as a heterogeneous nucleating agent, triggers more nuclei formation and promotes the occurrence of cold crystallization. As concluded from the DSC cooling curves in Figure 10(b), neat PLA shows no melting‐crystallization peak during the cooling process. This crystallization behavior is attributed to too slow crystallization rate of pure PLA to organize its molecular chains. 50 Additionally, the glass transition temperature of the PLA/M-OH-ISSA composites was lower than that of pure PLA due to the incorporation of M-OH-ISSA. Moreover, the modified PLA/M-OH-ISSA composites showed increased T m and crystallinity compared to the unmodified PLA/ISSA composites, indicating that the modification altered the crystal structure within the composites and effectively improved the interfacial compatibility between M-OH-ISSA and PLA. 51
Thermogravimetric analysis (TG/DTG)
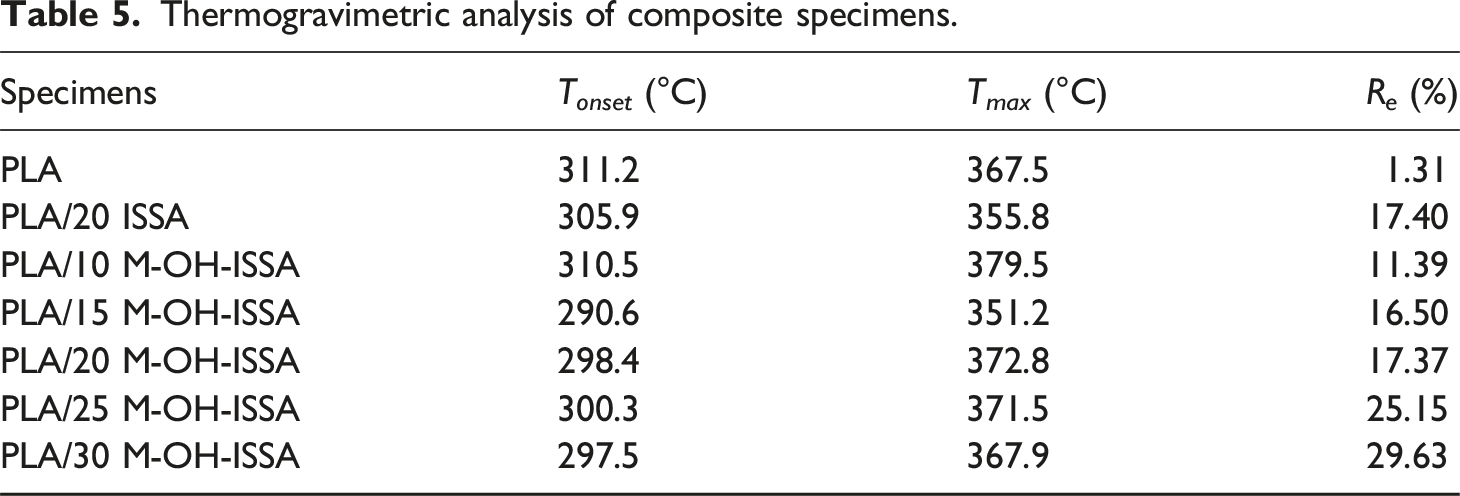
Figure 11 and Table 5 illustrate how the M-OH-ISSA content affects the thermal stability of PLA/M-OH-ISSA composites. The maximum weight loss temperature (T
max
) was taken from the peak of the derivative thermogravimetric (DTG) temperature plot, as shown in Figure 11(b), while the onset decomposition temperature (T
onset
) and the final residual weight % (R
e
) were derived from the TG plot, as shown in Figure 11(a). In the temperature range of 290 to 400, the T
onset
and T
max
both showed a decreasing trend as the M-OH-ISSA content increased, indicating that the addition of M-OH-ISSA reduced the thermal stability of the composites. This is because M-OH-ISSA is an inorganic mineral filler with greater thermal stability than PLA, but M-OH-ISSA also contains a significant amount of metal oxide, which most likely contributes to catalyzing the degradation of PLA.
52
Nevertheless, PLA/M-OH-ISSA composites still exhibited good thermal stability at approximately 200°C, and thermal degradation will not occur at this processing temperature. However, after the M-OH-ISSA content exceeded 15 wt%, T
onset
and T
max
began to increase with increasing M-OH-ISSA content. The most likely reason is that the aggregation of M-OH-ISSA particles causes the barrier effect of the M-OH-ISSA particles against the volatile pyrolyzed products. This result is consistent with what Xue et al. reported when they used fly ash as a reinforcing material for PLA composites.
52
Moreover, the initial degradation temperature of unmodified PLA/ISSA composites was higher than that of PLAM-OH-ISSA, while the maximum degradation temperature was lower than that of PLA/M-OH-ISSA, indicating that ISSA modification improved the thermal stability of the composites. TG of PLA/M-OH-ISSA composites (a) and DTG of PLA/M-OH-ISSA composites (b). Thermogravimetric analysis of composite specimens.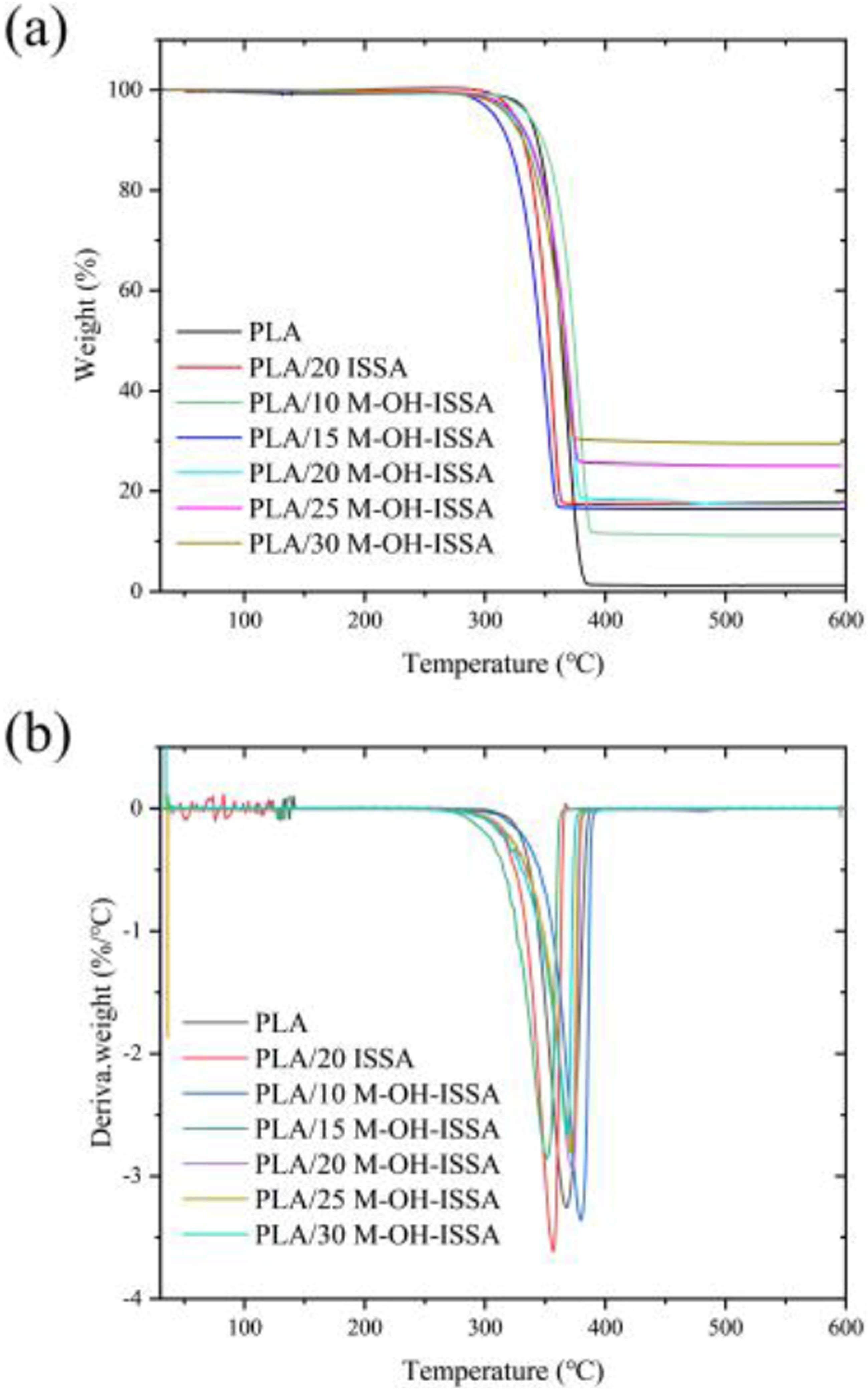
Rheological properties of PLA/M-OH-ISSA composites
The addition of M-OH-ISSA affects the rheological properties of the composites. The energy storage modulus (G′), loss modulus (G″), and complex viscosity (η*) of all the composites at 200°C are shown in Figure 12. Energy storage modulus (a), loss modulus (b), and complex viscosity (c) of composites with different M-OH-ISSA contents.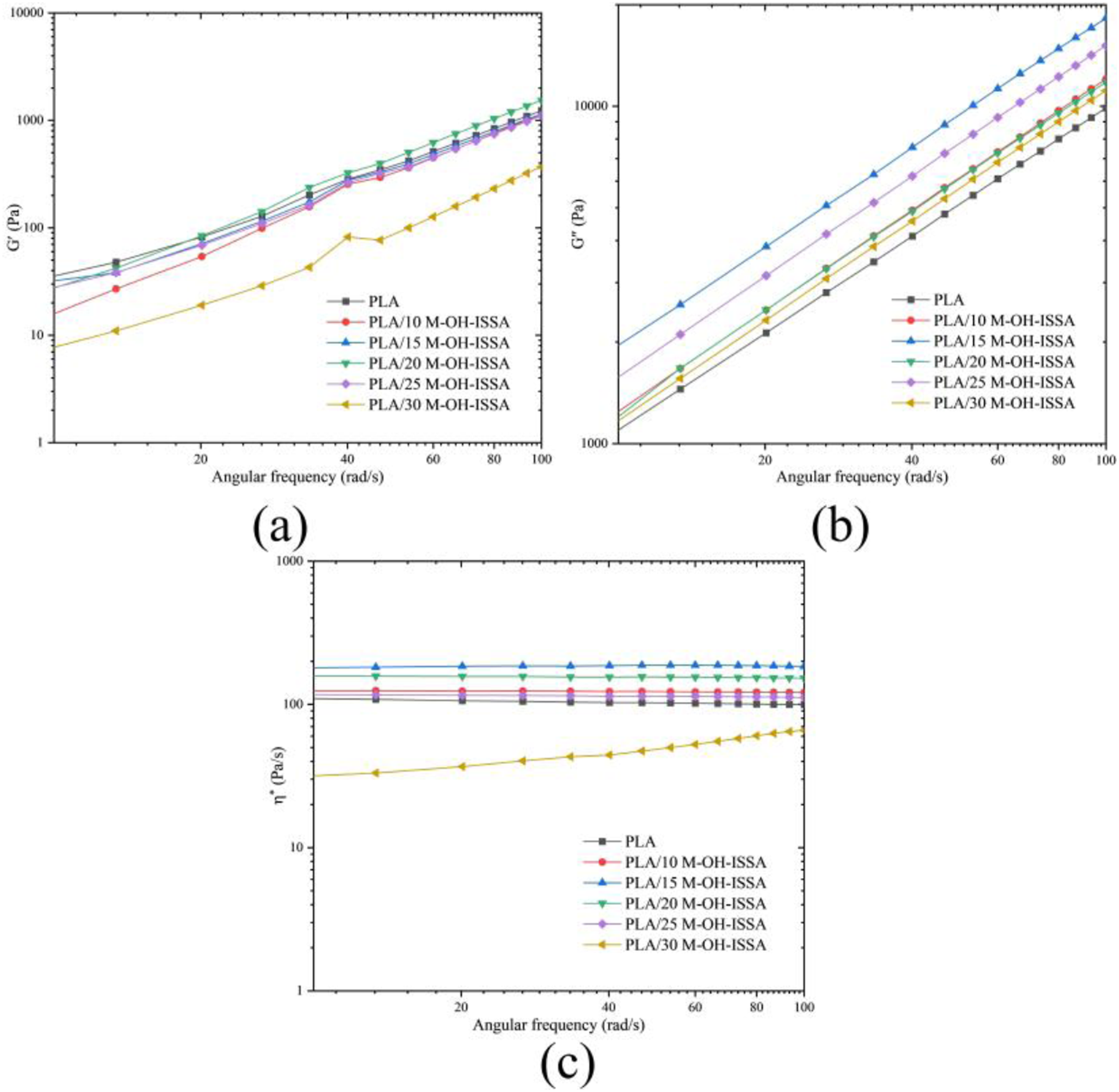
The PLA/M-OH-ISSA composite G′ was found to increase with increased frequency. At low frequencies, composite deformation was mainly caused by viscous flow, such that energy loss caused by deformation could not be recovered. At increased frequency, the viscous flow time was too short, such that elastic deformation occurred much faster than the viscous flow and composite deformation was mainly elastic deformation, with most of the elastic energy recoverable. 53
In the low-frequency region, the complex viscosity of PLA is almost constant. The loss modulus and complex viscosity of all composites are greater than that of pure PLA, and the complex viscosity of PLA/30 M-OH-ISSA gradually increases with increasing frequency. This may be related to the decrease in polymer chain mobility in the molten state, which leads to higher flow resistance due to lower chain mobility. 54 All the PLA/M-OH-ISSA composites showed strong shear thinning behavior. The disappearance of the Newtonian behavior of PLA and the increase in complex viscosity in the low-frequency region with increasing concentration of M-OH-ISSA could be attributed to its interaction with PLA. It was reported in a study by Ez-Zahraoui et al. that melt rheology can be used to predict the processing behavior of composites, and that pressure and temperature are key parameters in the processing of composites. It is important to note that high-viscosity composites require higher pressures or temperatures during the manufacturing process. 55 Therefore, the developed PLA/M-OH-ISSA composites will consume more energy during processing compared to PLA.
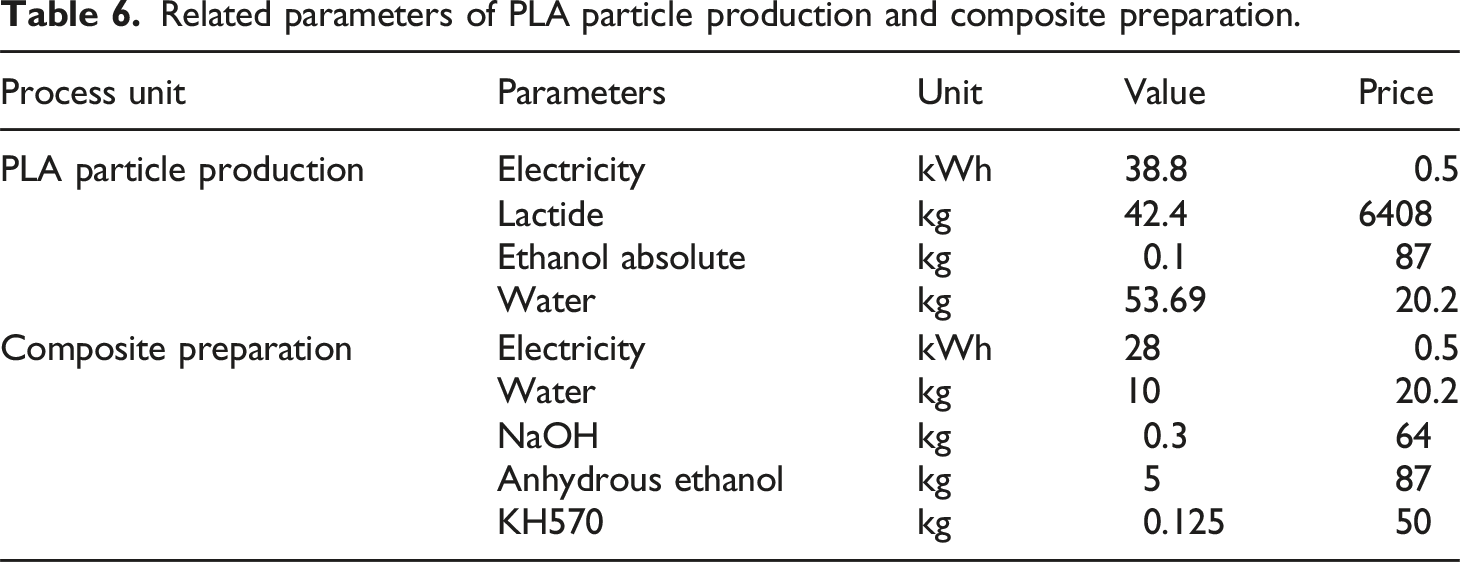
Cost analysis
Related parameters of PLA particle production and composite preparation.
Molding mechanism analysis
This study, proposed the reaction process and mechanism of alkali-silane modification of ISSA and preparation of PLA/M-OH-ISSA composites, as shown in Figure 13. The ISSA surface is rougher and more hydroxyl groups are exposed after alkali treatment with NaOH.
57
The more hydroxyl groups there are, the more silanes can be grafted onto the surface. KH570 typically has the following chemical structure: R-Si-R′-X, where R′ stands for the short alkyl bond CH2CH2CH2, R represents the hydrolysis group methoxy, and X stands for the organic functional group methacryloyloxy. The “chemical bonding theory” is the widely acknowledged mechanism underlying the modification of inorganic fillers by KH570.
58
When methoxy groups are hydrolyzed with water and hydrochloric acid acting as catalysts, their methoxyl groups hydrolyze to generate silanol groups (Si-OH), which are extremely reactive. Si-OH self-polymerizes into oligomers,
59
whereas reactive Si-OH oligomers are grafted to the ISSA surface and act as surface modifiers by physically adsorbing onto the surface through the formation of hydrogen bonds with the hydroxyl groups of the ISSA surface and covalent bonding through dehydration condensation. In addition, due to the presence of organic functional groups, the surface property after grafting surface modification converts from hydrophilic to hydrophobic. When the surface-modified ISSA was melt blended with PLA, mechanical interlocking caused intermolecular entanglement to form between M-OH-ISSA and PLA, which connected the two substances and improved interfacial compatibility, which improved the properties of the composites. Consistent with the conclusion made by Arslan et al.
60
The hydrophobicity of the filler and the increase in intermolecular entanglement could be responsible for the improvement of the interfacial strength between the M-OH-ISSA filler and PLA. Possible reaction process and mechanism of alkali-silane modification of ISSA and preparation of PLA/M-OH-ISSA composites.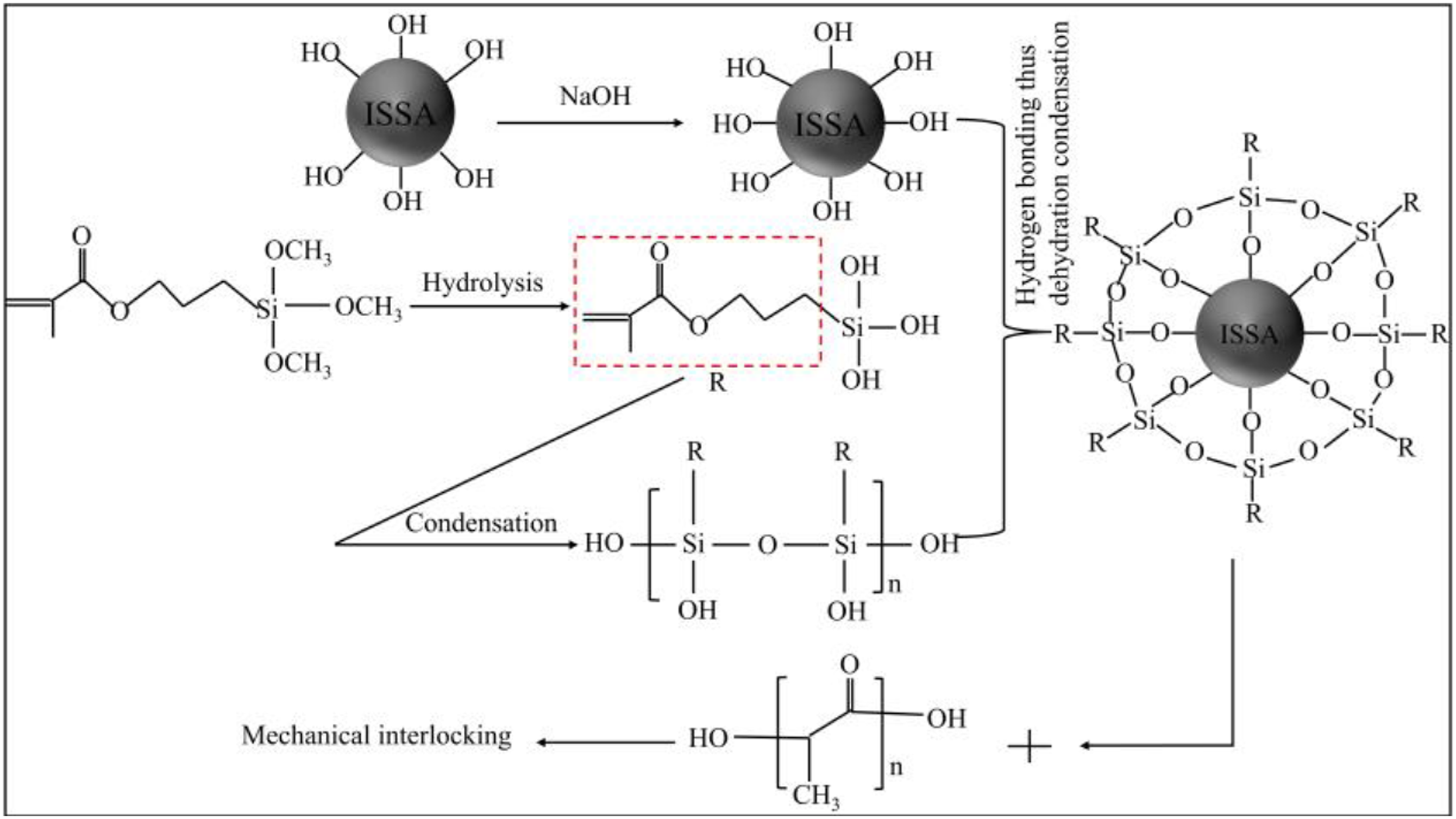
Conclusions
In this study, ISSA was modified by alkali-silane treatment, and a series of thermoplastic PLA/M-OH-ISSA composites were prepared using the melt-blending method. The following are the conclusions of the characterization and testing of M-OH-ISSA particulates and their composites: ISSA was successfully modified at the surface level to graft silane groups onto the surface of ISSA, which caused the surface to convert from hydrophilic to hydrophobic and significantly changed its micro-morphology, according to FTIR, contact angle, and SEM. The surface-modified ISSA can be uniformly dispersed in PLA, and the ISSA becomes tightly bonded to the PLA matrix. The mechanical test results showed that M-OH-ISSA-filled PLA composites improved the mechanical properties of PLA. Among a series of PLA/M-OH-ISSA composites, 15 wt% M-OH-ISSA-filled PLA composites showed the best mechanical properties. The tensile and flexural strengths of the 15 wt% M-OH-ISSA-filled PLA composites were increased by 59.3% and 61.8%, respectively, compared to pure PLA. Moreover, the surface modification induced better compatibility and adhesion between the filler and the matrix compared to the unmodified filled PLA composites, which improved the mechanical properties of the composites. The thermal performance of the composite test results showed that the addition of M-OH-ISSA had a promoting effect on the crystallization of PLA and improved the crystallinity of PLA. M-OH-ISSA resulted in a slight decrease in the initial thermal degradation temperature but an increase in the maximum weight loss temperature of the composites. The crystallinity of the composites increases with increasing M-OH-ISSA and the thermal stability between 290°C and 400°C decreases slightly with increasing M-OH-ISSA; however, the composites have good thermal stability near 200°C and do not undergo thermal degradation if processed at this temperature, according to DSC and TG/DTG. This was due to heterogeneous nucleation and catalysis of M-OH-ISSA. Moreover, The crystallinity and thermal stability of the modified PLA/M-OH-ISSA composites were superior to those of the unmodified composites. The addition of M-OH-ISSA affects the viscosity of PLA, and the viscosities of PLA/M-OH-ISSA composites are all higher than that of pure PLA. A cost analysis of the preparation of PLA/M-OH-ISSA composites confirms that the addition of M-OH-ISSA reduces the cost. It is known from the mechanism analysis that the interfacial strength of the composites increases due to the hydrophobicity of the fillers and the intermolecular interaction between them.
Therefore, surface modification of ISSA can effectively improve compatibility between polymer and inorganic materials. At the same time, the use of surface modification ISSA as a polymer filler not only improves the performance of the composites but also reduces the PLA preparation cost. These composites can be used in packaging applications and the 3D printing field. Moreover, this study can provide a solution for the reuse of ISSA. Future work will produce thermoplastic composites with higher ISSA content and better improve the brittleness of polylactic acid.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Jiangsu Provincial Key Laboratory of Food Advanced Manufacturing Equipment Technology (FMZ201905).
