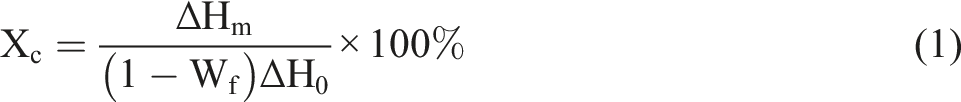Abstract
Inorganic particles dispersed in the polymer matrix can enhance the strength of polymers. In this paper, 3-Glycidyloxypropyltrimethoxysilane (KH560) and 2-Amino-1,3-propanediol were used to surface modify Zirconium dioxide (ZrO2) to prepare modified Zirconium dioxide (ZrO2), denoted as ZrO2-g. The modified ZrO2-g was characterized by Fourier transform infrared (FTIR) spectroscopy and nuclear magnetic hydrogen (1HNMR) spectroscopy, and the results showed that the modification was successful. Then the PBAT/ZrO2-g composites were synthesized by grafting ZrO2-g into poly (butylene adipate-co-terephthalate) (PBAT) molecular chain segments using in situ copolymerization. The results showed that ZrO2-g acted as a nucleating agent and improved the crystallinity of poly (butylene adipate-co-terephthalate) (PBAT). The rheological behavior analysis showed that the composite viscosity, storage modulus and loss modulus of PBAT/ZrO2-g composites were improved compared to PBAT. The ZrO2-g content had a significant effect on the tensile properties of the composites, and the composites had the highest tensile strength and elongation at break when the ZrO2-g content was 1% wt, reaching 23.27 MPa and 888.89%, respectively, which were increased by 60% and 38%, respectively, compared with PBAT. When the content of ZrO2-g exceeds 1.5 wt%, aggregation phenomenon occurs, which rather reduces the performance of the composites. Meanwhile, the thermal stability of the composites decreases with the increase of ZrO2-g incorporation.
Keywords
Introduction
In recent years, the severity of “white pollution” caused by non-biodegradable plastics has been increasing. As a result, there is a growing demand for the widespread use of biodegradable plastics.1,2 In the field of biodegradable plastics, PBAT is popular due to its excellent ductility, flexibility, biodegradability and biocompatibility. However, it does have certain limitations in its application scope due to its low mechanical thermal, and barrier properties.3–5 In order to compensate for the limited PBAT properties and broaden its application areas, a promising strategy is to enhance the properties of PBAT by creating PBAT nanocomposites through the integration of inorganic nanoparticles into the PBAT matrix.6–8
Dispersing inorganic nanoparticles into polymer matrix as reinforcement is an effective and relatively inexpensive method.9–11 The homogeneous dispersion of inorganic nanoparticles in the polymer matrix and the strong interfacial interactions between them are crucial for the study of polymer nanocomposites. Due to their large specific surface area and surface hydroxyl groups, inorganic nanoparticles are poorly compatible with the matrix, easily agglomerated and difficult to be uniformly dispersed. As a result, their ability to enhance properties is limited. 12 This requires graft modification of the surface of inorganic nanoparticles to introduce functional groups compatible with the polymer matrix.13,14 Modification of the surface of the nanoparticles15–18 will enhance the bonding effect and also increase the adhesion between the reinforcing material and the matrix for greater load transfer. 19 The addition of modified nanoparticles to the polymer matrix can greatly improve the mechanical properties. However, there seems to exist an optimal level beyond which the properties will no longer be improved or even the mechanical properties will be reduced.20,21 Several common methods for preparing polymer nanocomposites are in situ polymerization22–24 and melt blending.25,26
Nano-ZrO2 has high strength and good thermal stability. Incorporation of nano ZrO2 into PBAT matrix will improve the mechanical properties of the composites. 27 Here, ZrO2-g was prepared by modifying the surface of ZrO2 with a 3-Glycidyloxypropyltrimethoxysilane (KH560) and 2-Amino-1,3-propanediol. Then, ZrO2-g was mixed with p-Phthalic acid (PTA), Adipic acid (AA), and 1,4-Butanediol (BDO). ZrO2-g was grafted into the chain segments of PBAT molecules using in situ copolymerization, resulting in the synthesis of PBAT/ZrO2-g composites. The dispersion of ZrO2-g in the PBAT matrix was improved, and the study examined the impact of varying ZrO2-g contents on the mechanical properties of the composites.
Materials and Methods
Materials
p-Phthalic acid (PTA), Shanghai McLean Biochemical Technology Co., Ltd; Adipic acid (AA), Shanghai McLean Biochemical Technology Co., Ltd; 1,4-Butanediol (BDO), Shanghai McLean Biochemical Technology Co., Ltd; Zirconium dioxide (ZrO2), crystal system monoclinic, particle size 50 nm, density 5.89 g/cm3, melting point 2700°C, Mons’ hardness scale 8.5, thermal conductivity 2.09 W/(m•K), Shanghai McLean Biochemistry Technology Co.; 3-Glycidyloxypropyltrimethoxysilane (KH560), Shanghai McLean Biochemical Technology Co., Ltd; 2-Amino-1,3-propanediol, Shanghai McLean Biochemical Technology Co., Ltd; Titanium butoxide, analytically pure, Shanghai McLean Biochemical Technology Co., Ltd; Tin(II) 2-ethylhexanoate, analytically pure, Shanghai McLean Biochemical Technology Co., Ltd; Triphenyl phosphate, analytically pure, Shanghai McLean Biochemical Technology Co.
Preparation of Modified ZrO2-G
Deionized water and ethanol were mixed according to the volume ratio of 4:1 to produce a mixed solution. The mixed solution and 3-Glycidyloxypropyltrimethoxysilane (KH560) were mixed according to the volume ratio 4:1, and the pH of the reaction solution was adjusted to 3.5–5.5 using acetic acid, and the hydrolysis reaction was carried out in a water bath at 50°C for 2 h. Then ZrO2 was added, and the molar ratio of ZrO2 and KH560 was 1:1, and the reaction was stirred at a constant temperature of 60°C for 1 h. 28 By filtration, drying and pulverization, a white solid, denoted as ZrO2-KH560, was obtained. Filtering, drying, and pulverizing yielded a white solid labeled as ZrO2- KH560. ZrO2-KH560 was put into a three-necked flask with 2-Amino-1,3-propanediol at a molar ratio of 1:1, and an aqueous solution was prepared by adding deionized water. The reaction was carried out at 80°C in a water bath for 14 h. By filtration, drying and pulverization, a white solid, denoted as ZrO2-g, was obtained.
Preparation of PBAT/ZrO2-g Composites
ZrO2-g and BDO were weighed and poured into a three-necked flask, and then ultrasonically dispersed for 30 min, so that ZrO2-g was evenly dispersed in BDO. Then PTA was added into the flask according to the molar ratio of alcohol to acid 1.4:1, and then Titanium butoxide was added to react at 200°C under atmospheric pressure for 10 h. The reaction was considered to be finished when the water output of the esterification reaction reached 98% of the expected water output.
BDO and AA were added to a three-necked flask in accordance with the molar ratio of alcohol-acid 1.4:1, and then Titanium butoxide was added to react under atmospheric pressure at 180°C for 10 h. The reaction was considered to be finished when the water output of the esterification reaction reached 98% of the expected water output.
The two parts of the products at the end of the reaction were mixed into the same three-necked flask, adding Tin(II) 2-ethylhexanoate and Triphenyl phosphate, and the temperature was raised to 255°C. Then, a vacuum was drawn and the pressure was maintained at <50 Pa for 2 h for the polycondensation reaction. The grafting reaction of PBAT with ZrO2-g produces ZrO2-g-PBAT. when the viscosity of the reactants in the flask was no longer increased, it was regarded as the end of the reaction to obtain PBAT/ZrO2-g composites Figure 1. Flow chart for the preparation of PBAT/ZrO2-g composites.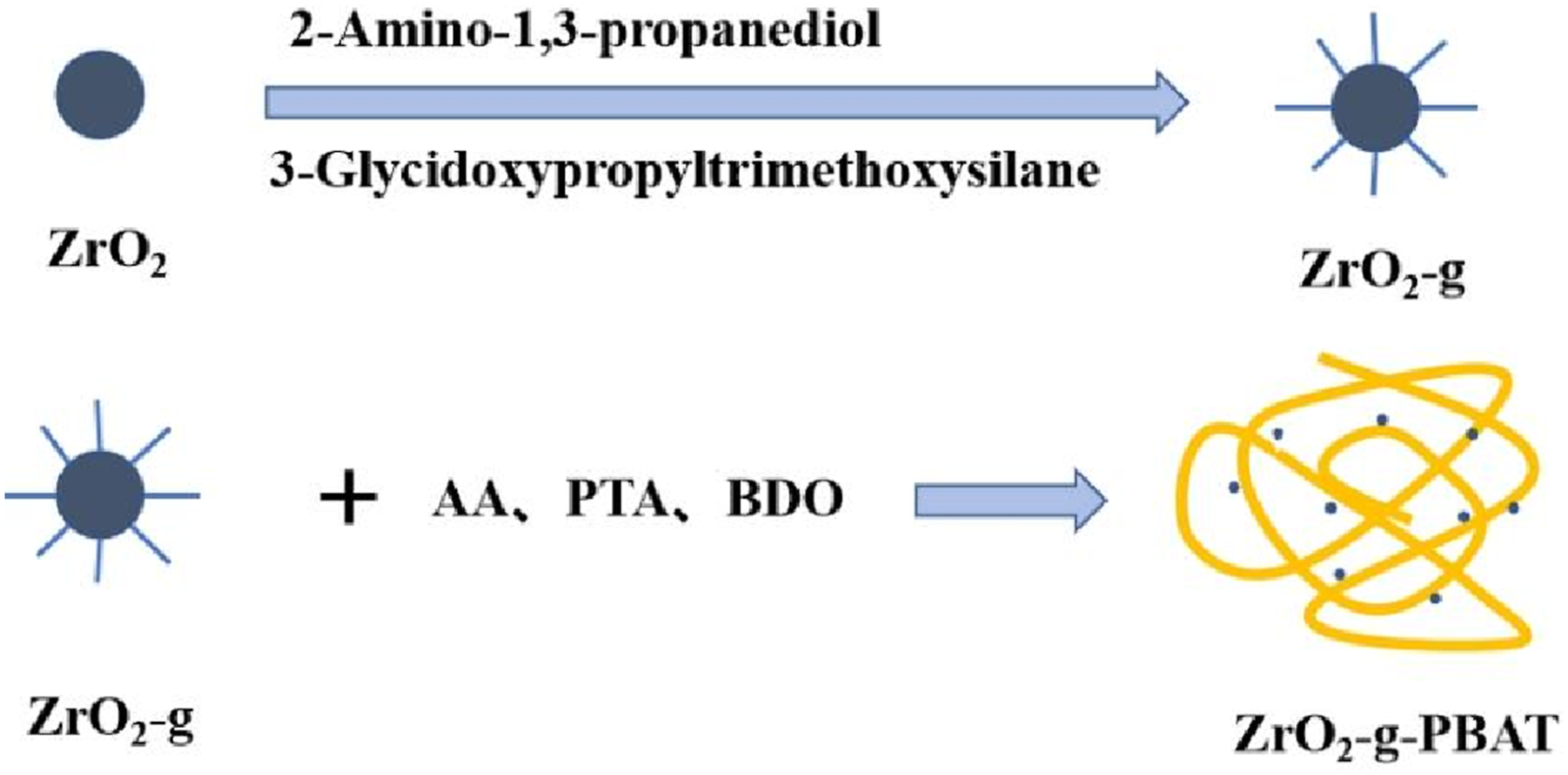
Testing and characterization
The structures of the synthesized substances were tested using 500 MHZv solid-liquid nuclear magnetic resonance (NMR) instrument manufactured by Bruker, Switzerland, with deuterated chloroform as the solvent and tetramethylsilane as the internal standard. The structures of ZrO2, ZrO2-g and ZrO2-g-PBAT were analyzed by FTIR. Thermal loss of weight (TG) analysis was performed on the composites by heating from room temperature to 800°C in N2 atmosphere at a heating rate of 10°C/min. The thermal properties of the composites were characterized using a DSC (TA Instruments Q-200). N2 atmosphere was used to increase the temperature from 40°C to 200°C with a constant temperature of 2 min; the temperature was decreased to −60°C with a cooling rate of 10°C/min, and the temperature was kept constant for 2 min, and then increased the temperature to 200°C at a rate of 10°C/min. Observe the crystallization curve and melting curve of the test sample. The crystallinity (Xc) was calculated by the following formula:
Results and discussion
Analysis of synthetic substances
ZrO2 and ZrO2-KH560 were characterized by FTIR as shown in Figure 2(a). At 2931 cm-1 and 1463 cm−1 from -CH2, and 1194 cm−1 and 1040 cm−1 from C-O-C specific to KH560. -NH generated by the reaction of KH560 with 2-Amino-1,3-propanediol led to the enhancement of 3411 cm−1 and 1584 cm−1, which proved both the success of the modification of ZrO2 by KH560 and 2-Amino-1,3-propanediol. The purified insoluble ZrO2-g-PBAT was characterized by FTIR. As shown in Figure 2(b), comparing the FTIR spectra of the modified ZrO2-g, it can be seen that the insoluble ZrO2-g-PBAT does not have the -OH group in the as-modified ZrO2-g (3430 cm−1), and at the same time, there is the appearance of a C = O stretching vibrational peak at 1713 cm−1. In addition, a benzene ring absorption peak at 731 cm−1 can be seen, which proves that ZrO2-g was successfully grafted to the PBAT chain segment. (a) FTIR spectra of ZrO2 and ZrO2-g. (b) FTIR spectra of ZrO2-g and ZrO2-g-PBAT.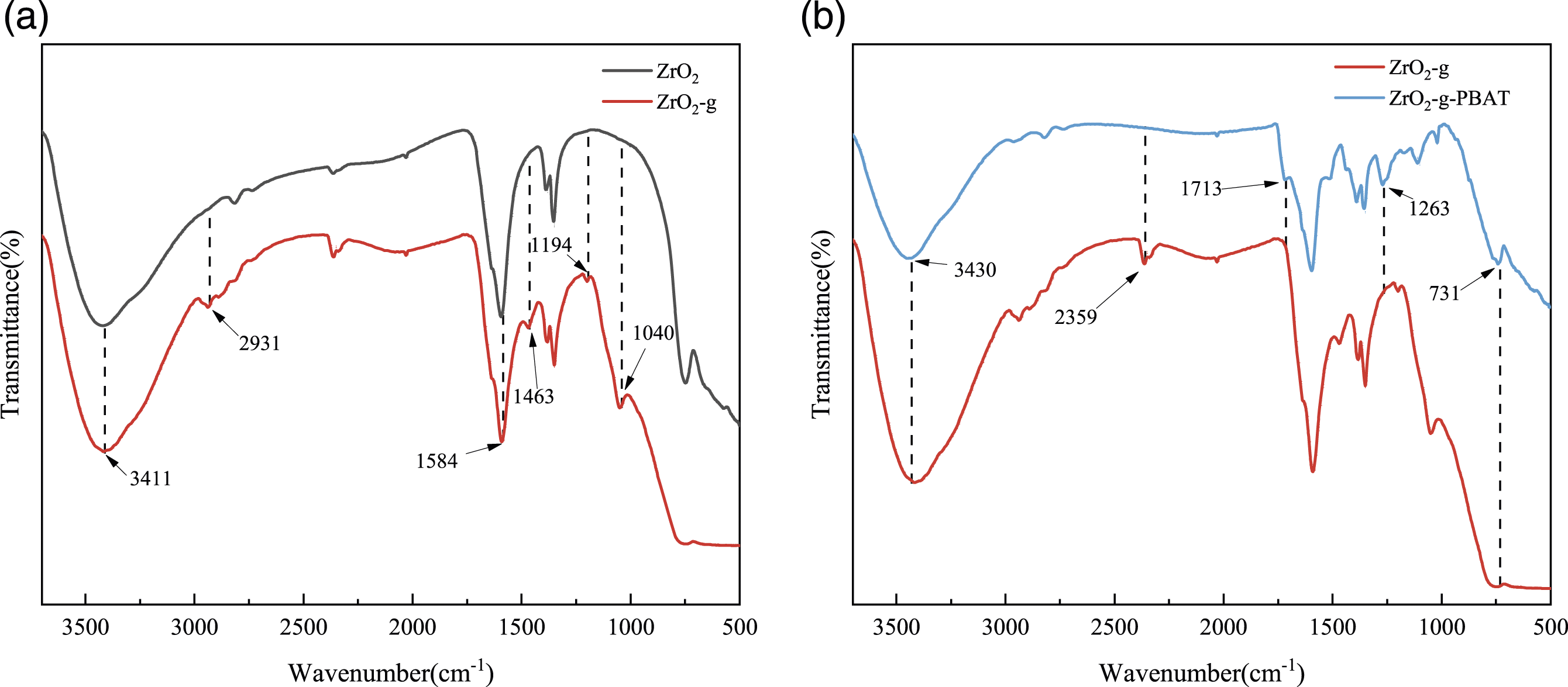
The insoluble substance was extracted by filtering the chloroform solution of the composites material. The insoluble substance should be ZrO2-g-PBAT. 1HNMR was used to determine the polymer chain structure of the soluble substance. The spectra and chemical shift peaks of each group were shown in Figure 3. -CH in BT unit was 8.10 ppm, -CH2 was 4.44, 4.36 and 1.98 ppm, respectively. The chemical shifts of -CH2 near the carbon group in butanediol adipate BA unit were 2.33 and 1.66 ppm, and -CH near the ether were 4.15, 4.09 and 1.84 ppm.29,30 Nuclear magnetic resonance hydrogen spectroscopy.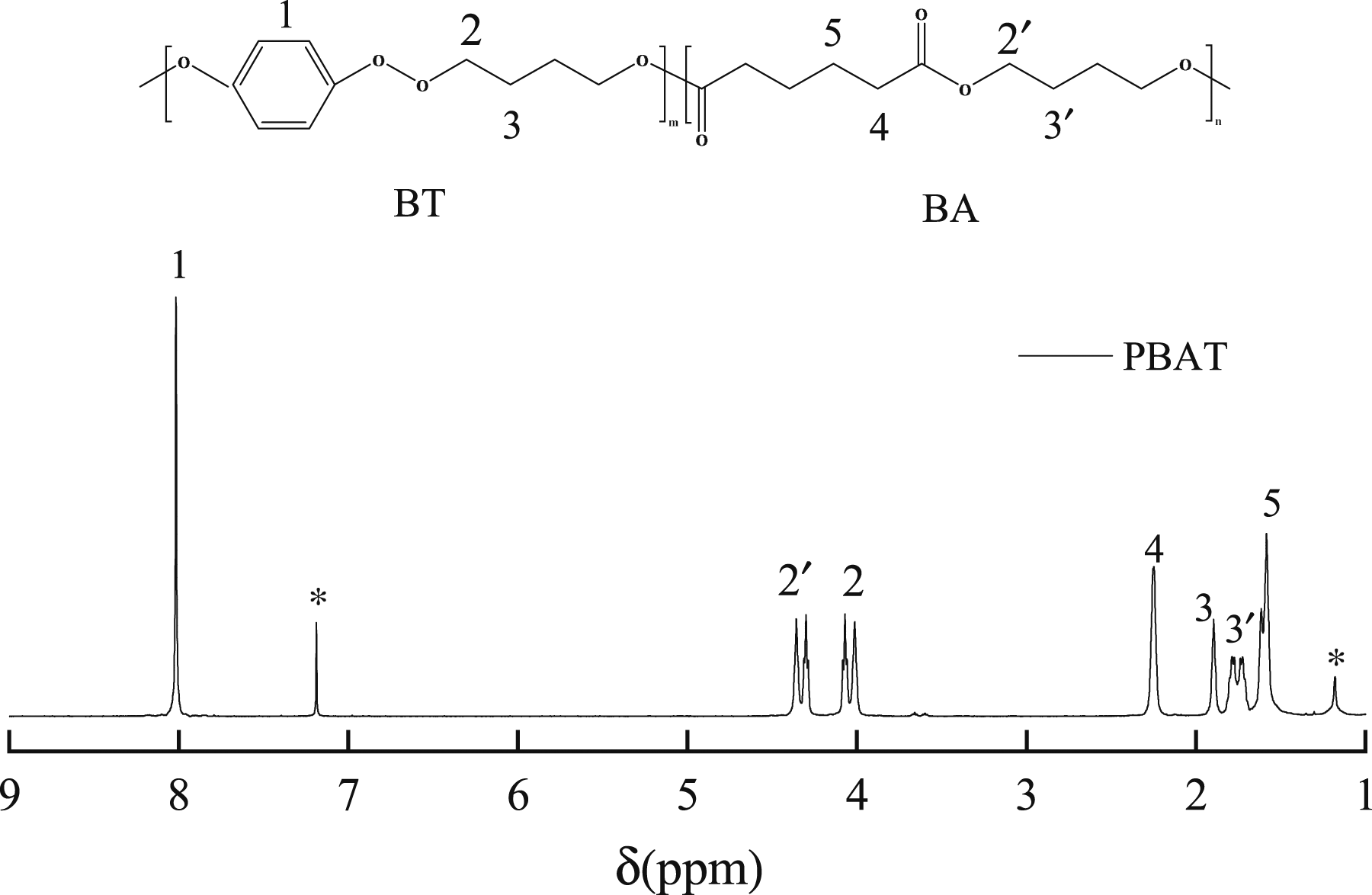
Thermal stability analysis of PBAT/ZrO2-G composites
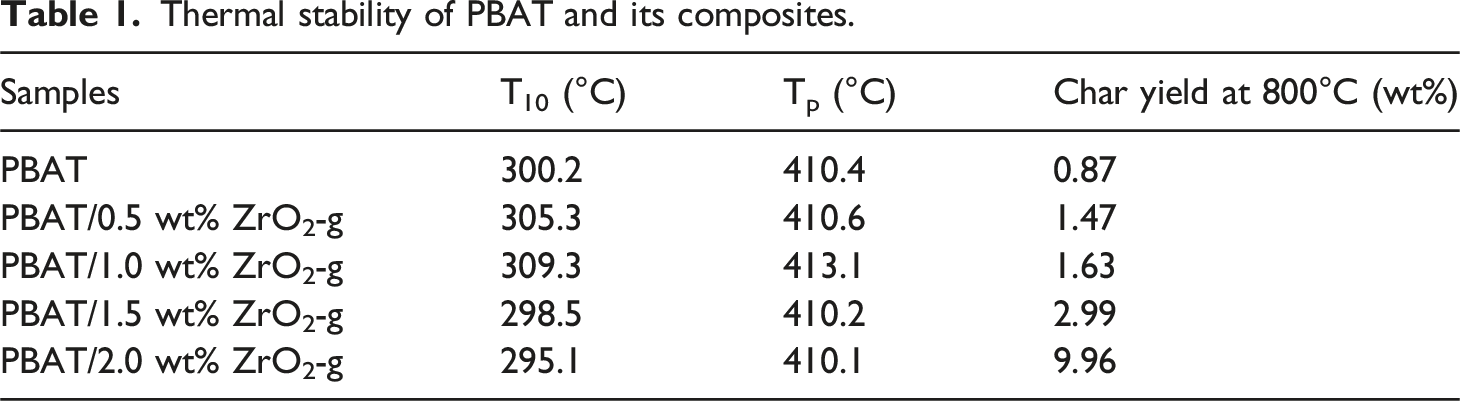
The effect of ZrO2-g on the thermal stability of PBAT was shown in Figure 4. The initial decomposition temperature (T10), peak decomposition temperature (Tp), and carbon yield at 800°C are listed in Table 1. In Figure 4, it could be seen that the values of T10 and Tp for the PBAT/ZrO2-g composites remain essentially unchanged compared to PBAT alone. This suggested that the addition of ZrO2-g had minimal impact on the thermal stability of the composites. With the increase in ZrO2-g content, the carbon yield of PBAT/ZrO2-g composites gradually increased at 800°C. This increase could be attributed to the addition of ZrO2-g. Heat loss analysis curves of PBAT composites with different contents of ZrO2-g. (a) TGA. (b) DTG. Thermal stability of PBAT and its composites.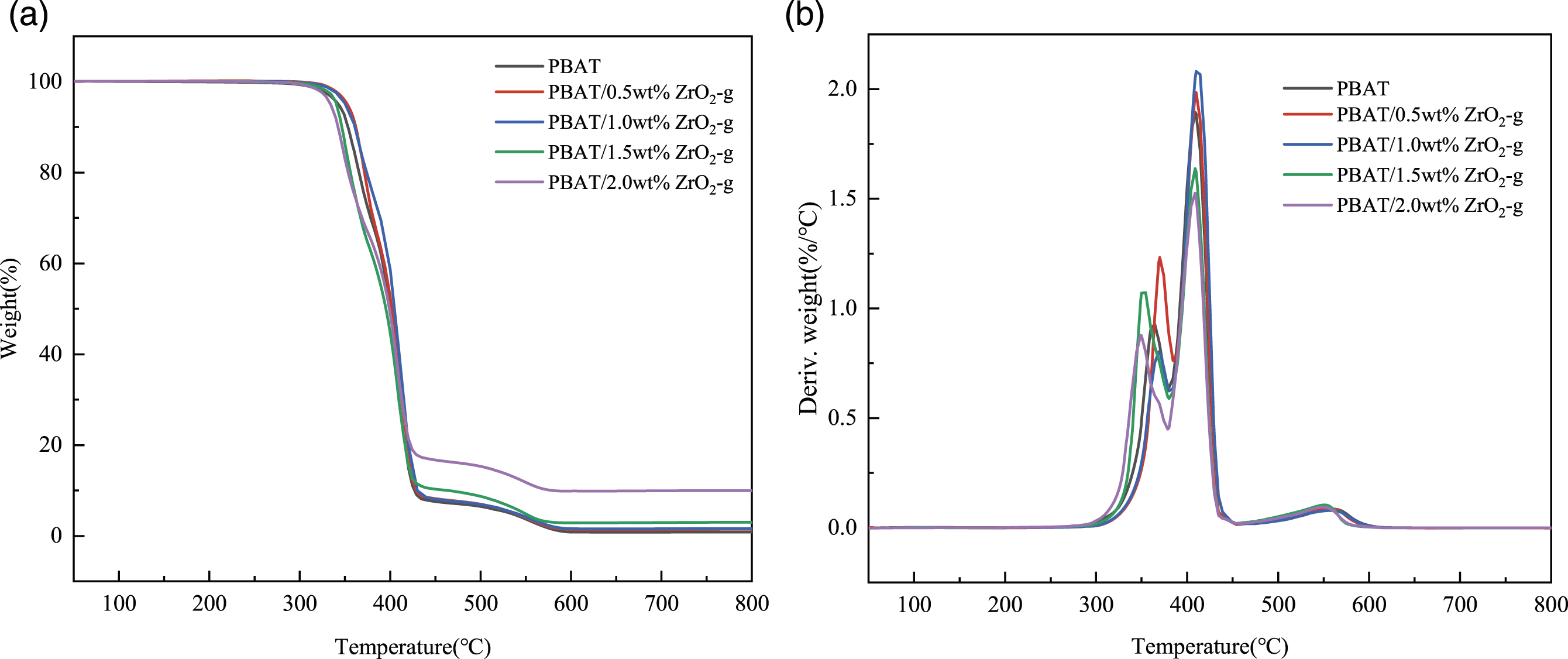
DSC analysis of PBAT/ZrO2-g composites
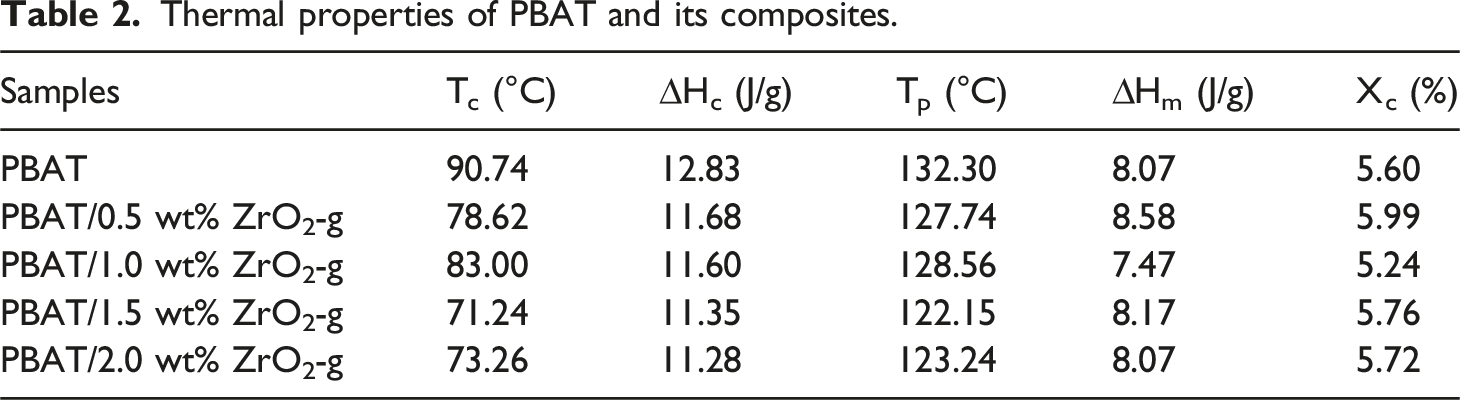
Based on the comparison of Figure 5 and Table 2, the trends of crystallization temperature (Tc), enthalpy of crystallization (ΔHc), melting temperature (Tp), enthalpy of melting (ΔHm), and degree of crystallinity (Xc) could be known. The ΔHm and ΔHc values of PBAT/ZrO2-g composites did not change significantly compared to PBAT. However, the Tc values were decreased. This was attributed to the nanoeffect of ZrO2-g nanoparticles, which acted as nucleating agents, thus increasing the crystallization rate and lowering the Tc,
31
and the Xc of the composites increased. The decrease in Tp with increasing ZrO2-g content was attributed to the fact that ZrO2-g acts as a thermally conductive material and could be used for heat transfer. The increase in ZrO2-g content made it easier for the PBAT polymer chains to absorb heat, which reduced the energy required for melting, and thus reduced the Tp.
32
Differential scanning calorimetry (DSC) thermal spectra of PBAT composites with different contents of ZrO2-g. (a) Crystallization curve. (b) Melting curve. Thermal properties of PBAT and its composites.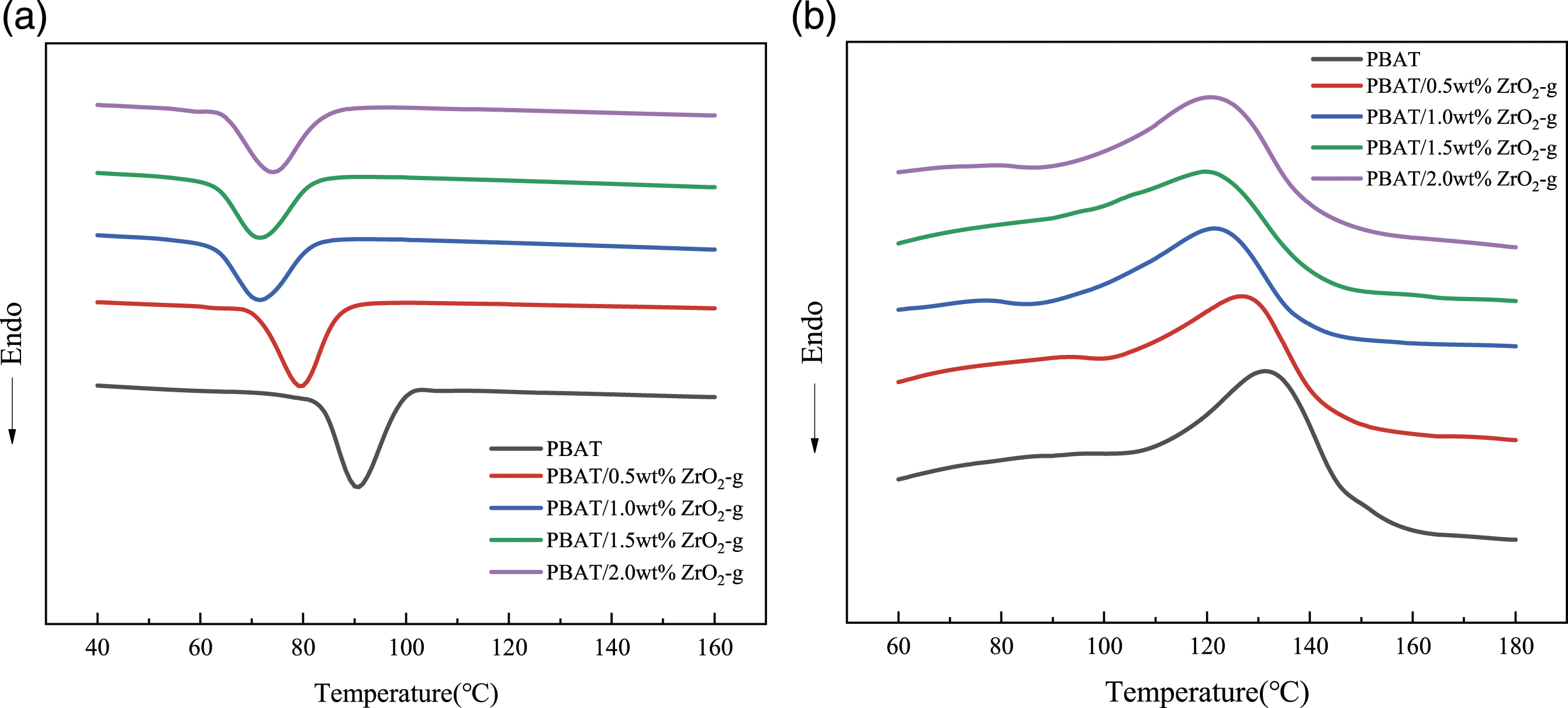
Rheological behavior analysis of PBAT/ZrO2-g composites
Figure 6 showed the results of dynamic rheological measurements of the complex viscosity (η*), storage modulus (G′) and loss modulus (G″) of PBAT and PBAT/ZrO2-g composites at 140°C as a function of frequency. From Figure 6(a), there was an increase in the complex viscosity of the composites as compared to PBAT. This is due to the incorporation of ZrO2-g due to the presence of inter-particle and inter-particle-polymer interactions, which hinders the chain segment movement, reduces the rate of molecular chain unraveling, and increases the relaxation time and hence the Newtonian plateau.
33
As shown in Figure 6(b), there was an increase in the energy storage modulus of the composites as compared to PBAT. With the increase of ZrO2-g content in the composites, there were interactions between ZrO2-g to form a permeable network structure, which inhibited the chain segment relaxation and increased the energy storage modulus.34–36 The results of Figure 6(c) showed an increase in loss modulus with the addition of ZrO2-g to the PBAT matrix, indicating an increase in viscous deformation, which acted as a tackifier. Dynamic rheological properties of PBAT composites with different contents of ZrO2-g. (a) Complex viscosity. (b) Storage modulus. (c) Loss modulus vs. frequency.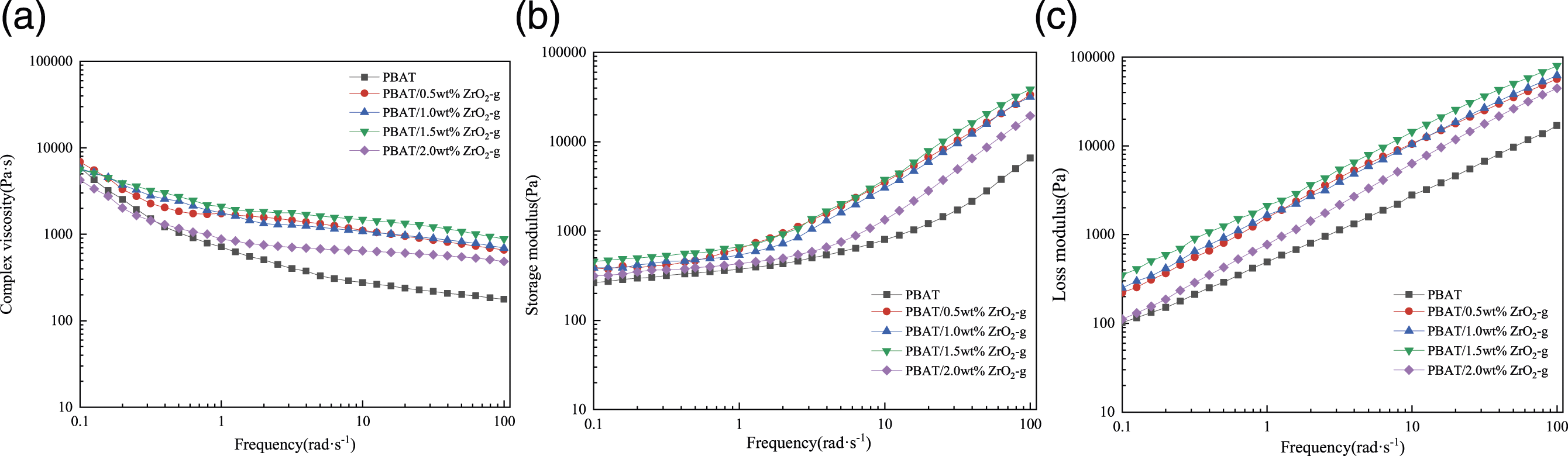
Mechanical properties of PBAT/ZrO2-g composites
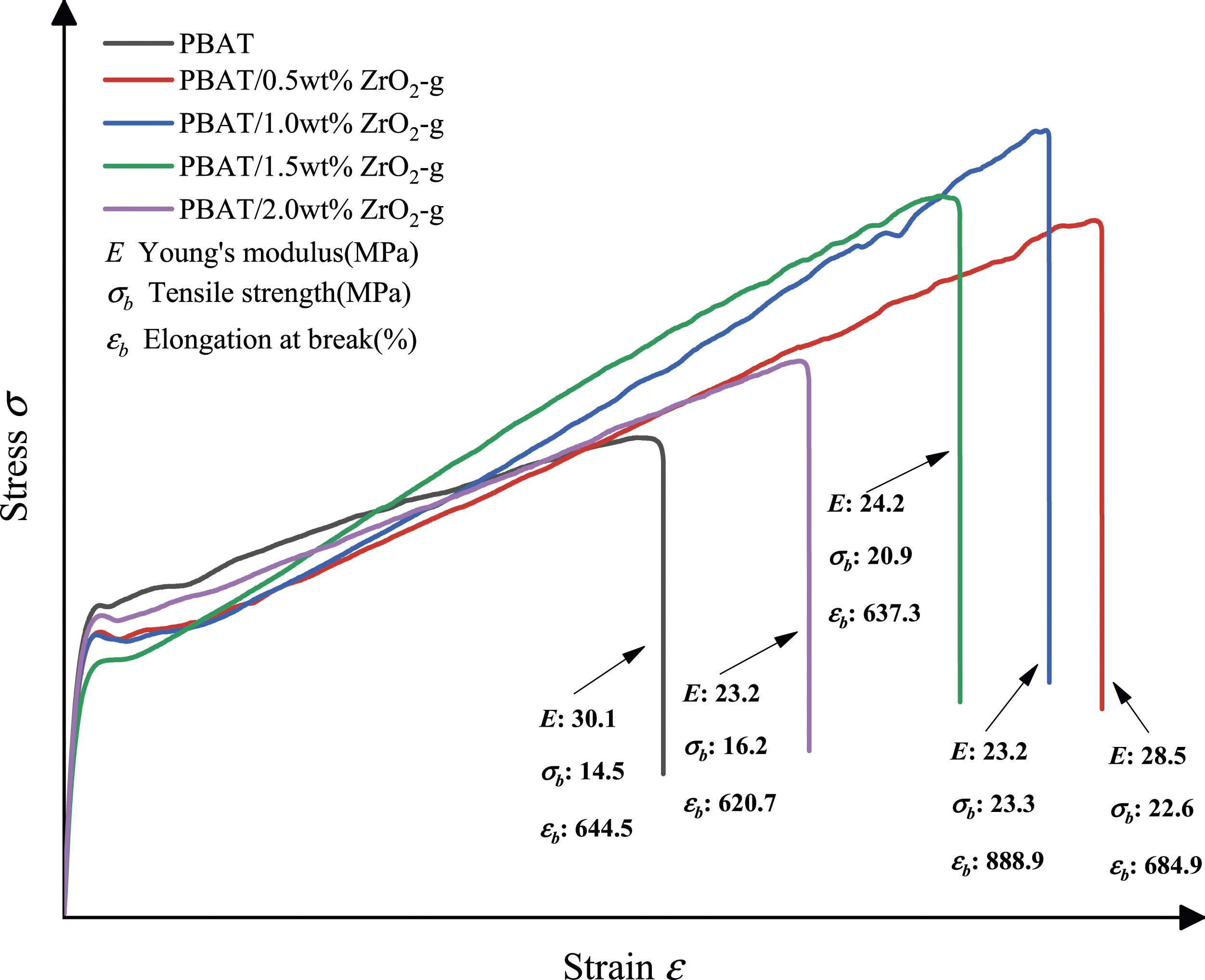
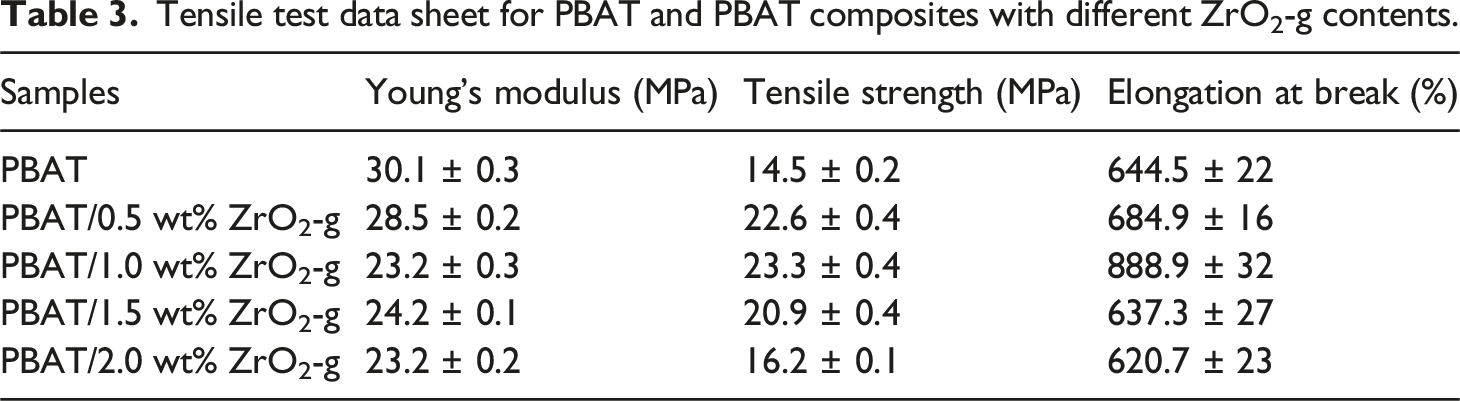
According to Figure 7, Figure 8 and Table 3, the tensile strength of PBAT/ZrO2-g composites increased with the increase of ZrO2-g content and the tensile modulus decreased with the increase of ZrO2-g content. This was because ZrO2-g particles, acting as rigid fillers, play a reinforcing role.
37
The tensile strength of PBAT composites reached a maximum value of 23.27 MPa when the ZrO2-g content was 1 wt%. However, with the increase of ZrO2-g content, the tensile strength began to decline, which was caused by the agglomeration of ZrO2-g, which was also verified by SEM. As shown in Figure 9(d), there was obvious aggregation of ZrO2-g particles, and the decrease in tensile strength was also significant. In addition, the elongation at break of PBAT composites increased slightly with the addition of ZrO2-g, but the elongation at break of PBAT decreased significantly with the further increase of nano-filler content. This was because of the concentration of stress caused by the accumulation of ZrO2-g, which reduced ductility. Tensile strength and elongation at break of PBAT composites with different contents of ZrO2-g. Stress-strain curves of PBAT composites with different ZrO2-g contents for tensile testing. Tensile test data sheet for PBAT and PBAT composites with different ZrO2-g contents. SEM surface topography of PBAT composites with different contents of ZrO2. (a) ZrO2 content 0.5 wt%. (b) ZrO2 content 1 wt%. (c) ZrO2 content 1.5 wt%. (d) ZrO2 content 2 wt%.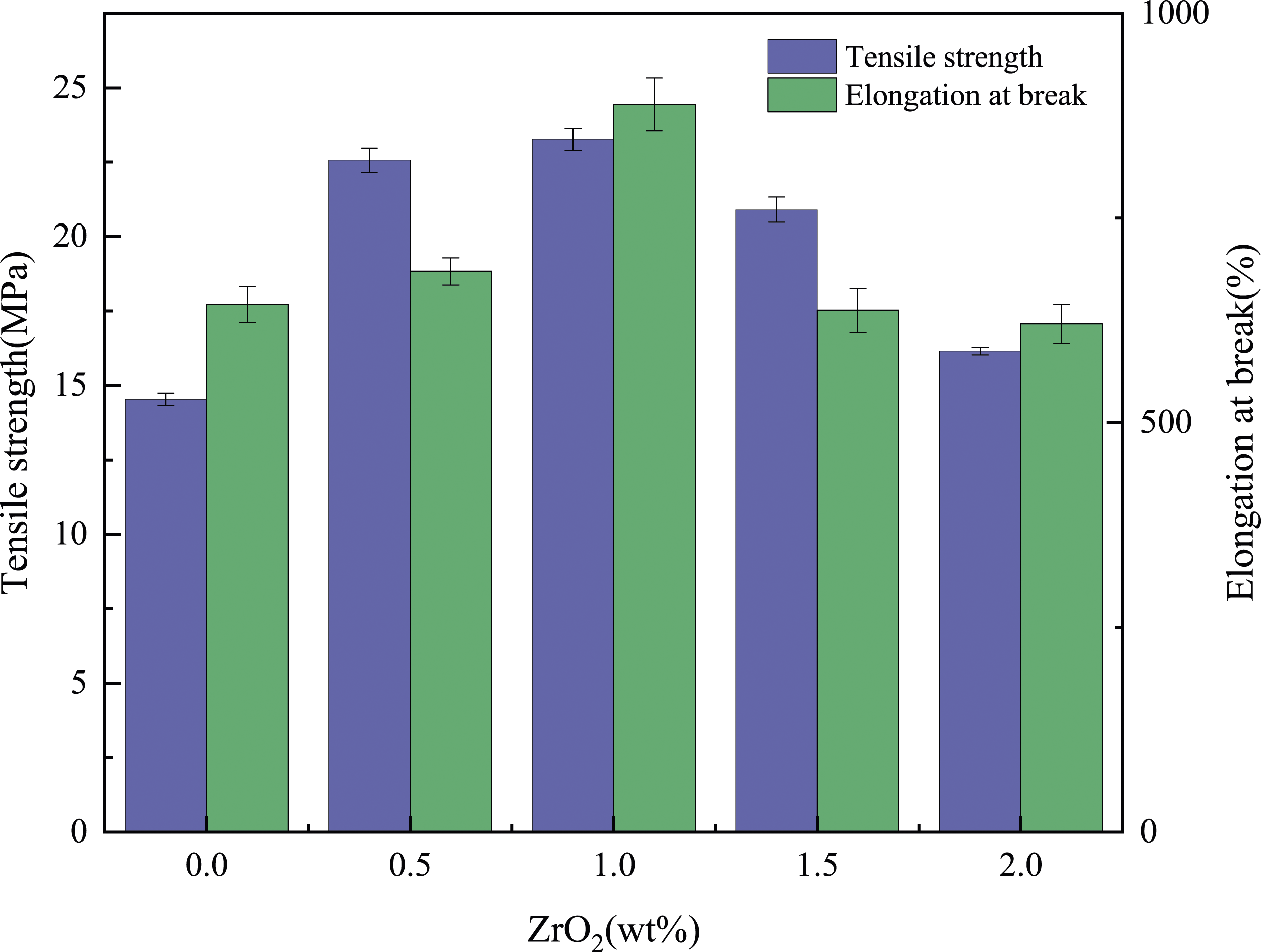
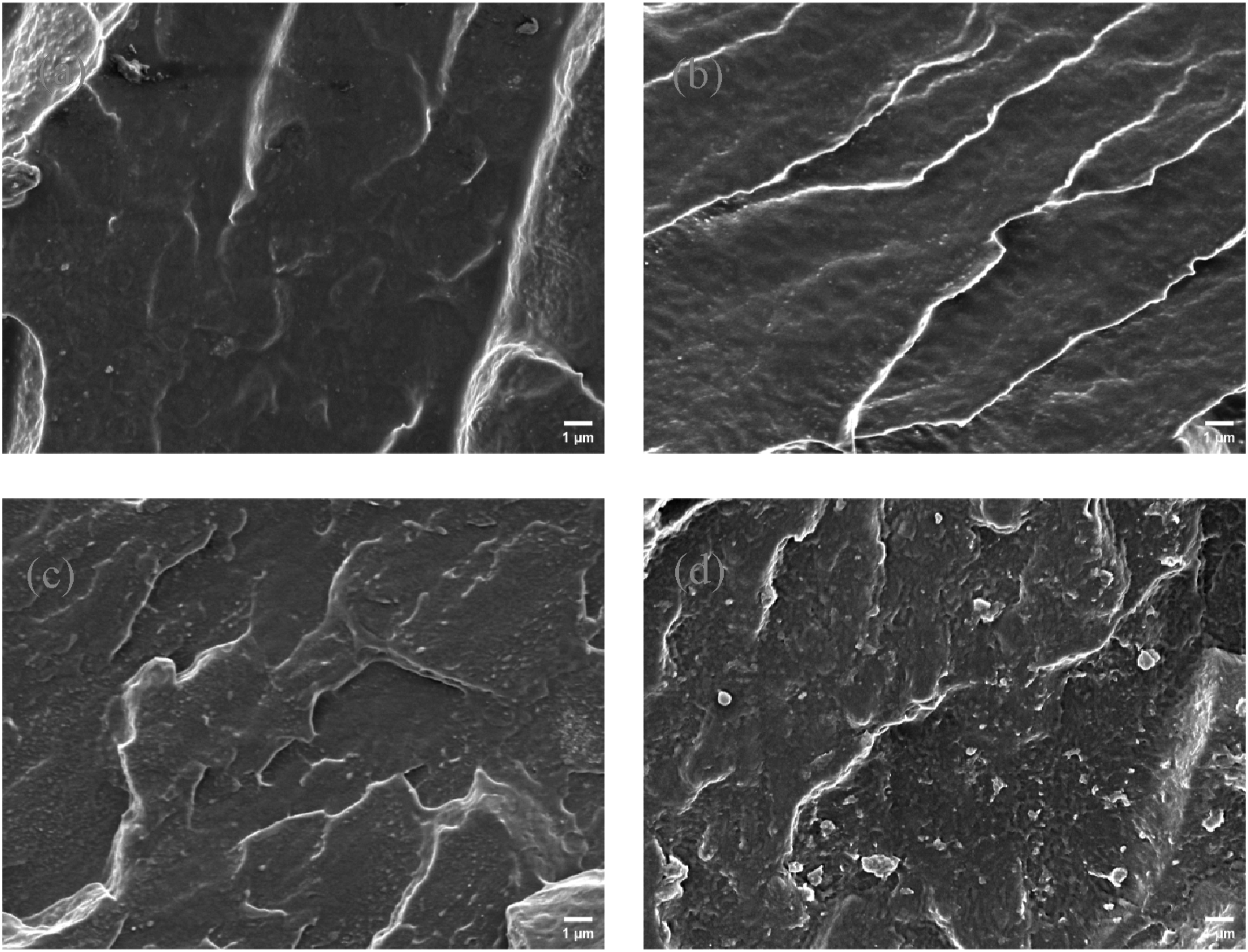
Surface phase analysis of PBAT/ZrO2-g composites
The scanning electron microscopy (SEM) images of PBAT/ZrO2-g composites were presented in Figure 9. As the ZrO2-g content increased, the fracture surface of PBAT/ZrO2-g composites becames rougher (Figure 9(b)–(c)). This suggested that the inclusion of rigid ZrO2-g nanoparticles contributed to the redistribution of load-bearing stress, 38 thereby enhancing the mechanical properties of the composites. This was consistent with the results of the previous test on mechanical properties. When the content of ZrO2-g increased to 1.5 wt%, the agglomeration of ZrO2-g appeared, which led to the degradation of the properties of PBAT/ZrO2-g composites.
Conclusion
In this paper, ZrO2 was surface modified using 3-Glycidyloxypropyltrimethoxysilane (KH560) and 2-Amino-1,3-propanediol, denoted as ZrO2-g. Subsequently, PBAT/ZrO2-g composites with varying contents were prepared through in situ polymerization. The thermal stability analysis of PBAT/ZrO2-g composites shows that the addition of ZrO2-g has little effect on the thermal stability of the material. Dynamic rheological measurements of the composite viscosity, storage modulus and loss modulus of PBAT and PBAT/ZrO2-g composites as a function of frequency showed an increase in the composite viscosity compared to PBAT. This is due to the fact that the incorporation of ZrO2-g causes inter-particle and particle-polymer interactions that hinder the movement of the chain segments, decrease the unlinking rate of the molecular chains, and increase the relaxation time, resulting in a Newtonian plateau. The energy storage modulus of the composites increased as compared to PBAT. With the increase of ZrO2-g content in the composites, the ZrO2-g interacts with each other to form a permeable network structure, which inhibits the chain segment relaxation and increases the energy storage modulus. With the addition of ZrO2-g to the PBAT matrix, the loss modulus increases, indicating an increase in viscous deformation, which acts as a viscosity builder. ZrO2-g nanoparticles have a nano effect. When they were added to PBAT, they acted as heterogeneous nucleating agents, promoting crystallization, lowering the crystallization temperature, and increasing the degree of crystallinity. ZrO2-g is a thermally conductive material that can be used for heat transfer, and the increase in ZrO2-g content makes it easier for the PBAT polymer chains to absorb heat, which reduces the amount of energy required for melting, and thus lowers the melting temperature. When the ZrO2-g content was less than 1 wt%, the tensile strength and elongation at break of PBAT composites increased with the increased in ZrO2-g content. When the ZrO2-g content was 1 wt%, the maximum tensile strength and elongation at break reach 23.27 MPa and 888.89%, respectively. These values were 60% and 38% higher than those of PBAT, respectively. However, with the increased in ZrO2-g content, the tensile strength began to decline. This was also supported by the SEM results, which indicated that the decrease in tensile strength was caused by the accumulation of ZrO2-g. The results also showed that adding a small amount of ZrO2-g to the PBAT matrix could increase its strength by 60%. This finding provided a way to enhance the strength of PBAT and broaden its range of applications.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Liaoning Provincial Key Technology Project for Synthesis of High Performance Biodegradable Material PBAT-X Based on Molecular Design; Liao Ke Fa (2022) No. 41.