Abstract
Biopolymer blend based nanocomposites have been interestingly investigated by researchers to achieve advanced composite functional materials that are environmentally amicable. Among the varieties of biopolymers, widespread attention has been given to polyvinyl alcohol (PVA) and polyvinyl pyrrolidone (PVP) owing to their interesting properties like water solubility, biodegradability, biocompatibility and non-toxicity. PVA and PVP form a completely miscible blend at all compositions as both the polymers are rich in a variety of functional groups that permit effective intermolecular interactions between the polymers as well as with filler. PVA/PVP blend exhibits excellent dopant sensitive properties also. Metal oxide nanoparticles (MONPs) have made a prominent place in the area of scientific and technological research due to their unique chemical and physical properties. Doping with MONPs enhances the properties of the PVA/PVP blend matrix and the resulting PVA/PVP/MONP polymer nanocomposites are suitable for multifunctional purposes. This review mainly focused on the influence of various MONPs such as ZnO, CuO, Al2O3, ZrO2, MnO, SnO, MgO, TiO2, etc. in the structural, morphological, thermal, optical and electrical properties of PVA/PVP blend as well as its applications in various fields.
Keywords
Introduction
Polymer nanocomposites (PNCs) are advanced classes of materials that offer great potential in various fields of applications. The properties of PNCs depend not only on the properties of individual polymers but also on the phase morphology of the polymer matrix and the interfacial interaction of nanofillers with the polymer matrix.1–3 PNCs have received much attention due to their large surface area and very high aspect ratio.4–8 Polymer blending is an efficient and modern technique to design novel polymeric materials with desired physical and chemical properties.9,10 If nanofillers are added to the polymer blend matrix, the resulting polymer blend nanocomposite will show enhanced properties.11,12 We can expect the advantages of inorganic nanosized fillers and organic polymer blend matrix in the final polymer blend nanocomposites. Research on polymer blend nanocomposites has been increasing as it holds great promise for present and future needs. The use of biopolymer-based polymeric materials has developed as a solution for many environmental issues caused by conventional plastic materials. It was a great challenge to the emerging researchers to develop novel eco-friendly polymeric materials with high performance and potential applications. The addition of suitable nanofillers to biopolymer blends has resulted in the development of novel polymeric materials with amazing properties. Such biodegradable polymer blend nanocomposites will offer high performance material with the power to ensure environmental safety along with several applications in biomedical, technological and engineering fields.13–17
Among the wide variety of biopolymers polyvinyl alcohol (PVA) and polyvinyl pyrrolidone (PVP) have received special attention because of their unique properties. Recently green nanocomposite functional materials based on them have been extensively reported. 10 PVA is a nontoxic synthetic biopolymer that exhibits excellent chemical resistance, water absorption, gas barrier properties, mechanical strength, high flexibility, elasticity, good charge storage capacity and film-forming properties. It can be blended with other polymers owing to the opportunity to form hydrogen bonding.18–23 PVP is one of the most popular water-soluble, biodegradable, biocompatible synthetic biopolymers with extremely low cytotoxicity.24–27 An important peculiarity of PVP is that it contains a lactam ring which helps in chelating with nanoparticles and other polymers and the protein-like structure makes it a researcher’s favorite polymer in a biological point of view. 28 Both the polymers are well known for their individual applications in various fields. Blending of PVA and PVP, and incorporation of various nanofillers to polymer blend matrix are considered to be effective method to improve their properties as well as to expand their applications. Recently PVA/PVP blends with various types of nanofillers have been studied extensively by researchers.23,29 PVA and PVP form a miscible blend for all compositions through the intermolecular hydrogen bonding between carbonyl and hydroxyl groups in the chain backbone of PVP and PVA respectively (Figure 1 and 2). These polar functional groups present in PVA and PVP are highly sensitive with various inorganic nanofillers and functional additives which results in the formation of advanced composite functional material.30–37 PVA/PVP blend with 50:50 compositions are found to exhibit optimum physicochemical properties. 38
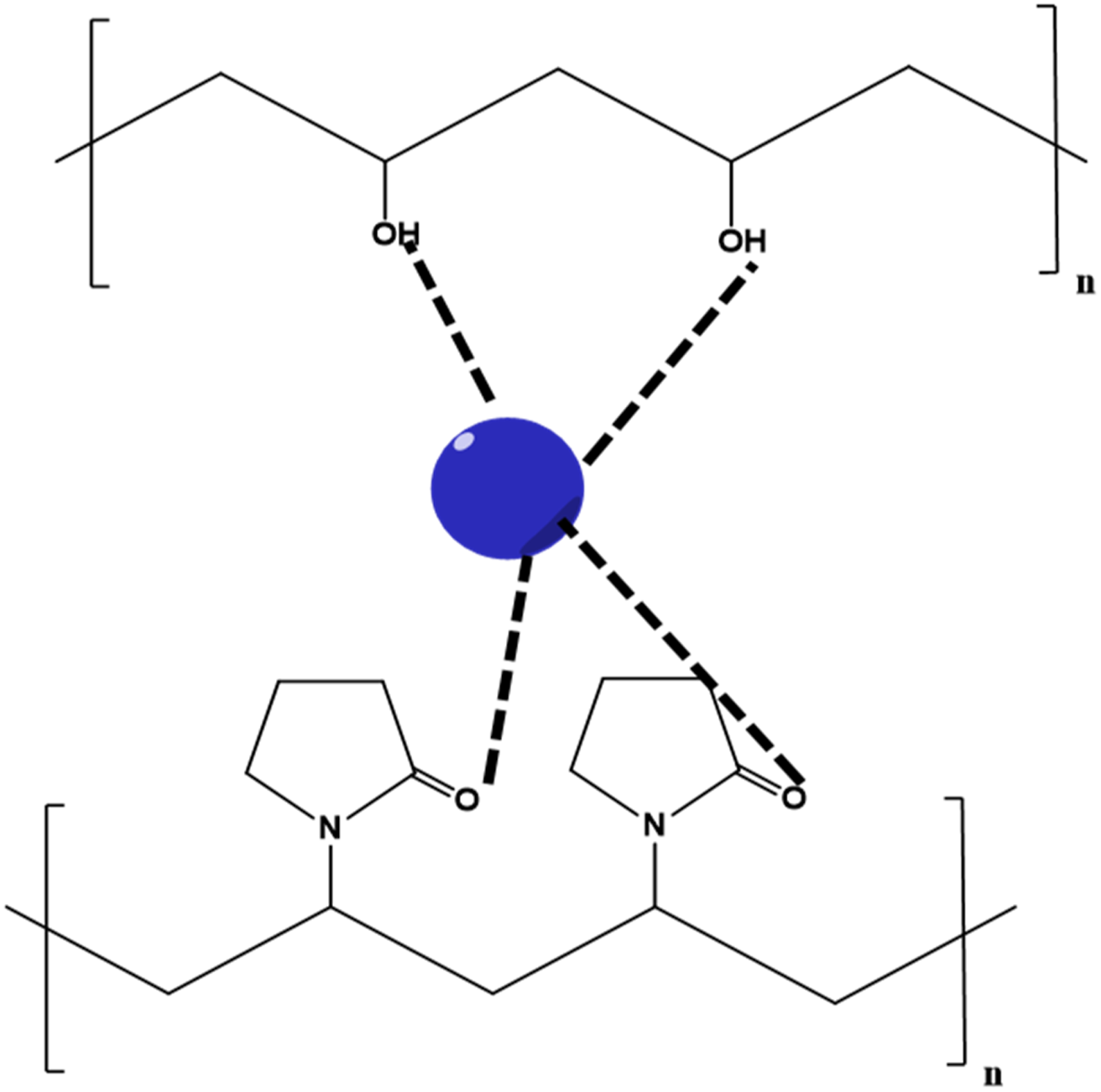
The field of nanotechnology has been growing explosively for the past few decades. Nanomaterials exhibit excellent features compared to their bulk counterparts. Metal oxide nanoparticles (MONPs) have made a prominent place in the area of scientific and technological research because of their unique characteristics and increasing interest for applications in the fields of physics, chemistry, biology, and medicine.39,40 The unique chemical and physical properties of MONPs are attributed to the high density and limited size of corners and edges on their surface.41,42 Proper dispersion of nanofiller in the polymer matrix is the major barrier associated with the development of PNC with good characteristics. In this scenario, PVA/PVP blend matrix is a boon to such MONPs as it can complex or coordinate with the functional group rich polymer back chain (CO and OH).
43
Poor dispersion of MONPs in the polymer matrix is mainly due to the hydrophilic surface and high surface energy which will lead to the aggregation of nanoparticles. So, surface modification of MONPs is also considered to be an appropriate method to enhance the dispersion and thereby improve the properties of final PNC materials. Among a large variety of modifiers, biosafe modifiers are largely preferred nowadays because of the growing environmental concern.44,45 Herein, we are reviewing the structural, morphological, thermal, optical and electrical properties of nanocomposite materials based on PVA/PVP blend incorporated with various metal oxide as well as modified metal oxide nanofillers. Potential applications of these PNC materials in various fields are also discussed here. Hydrogen bonding interactions in PVA/PVP blend. Interactions between nanofillers and PVA/PVP blend.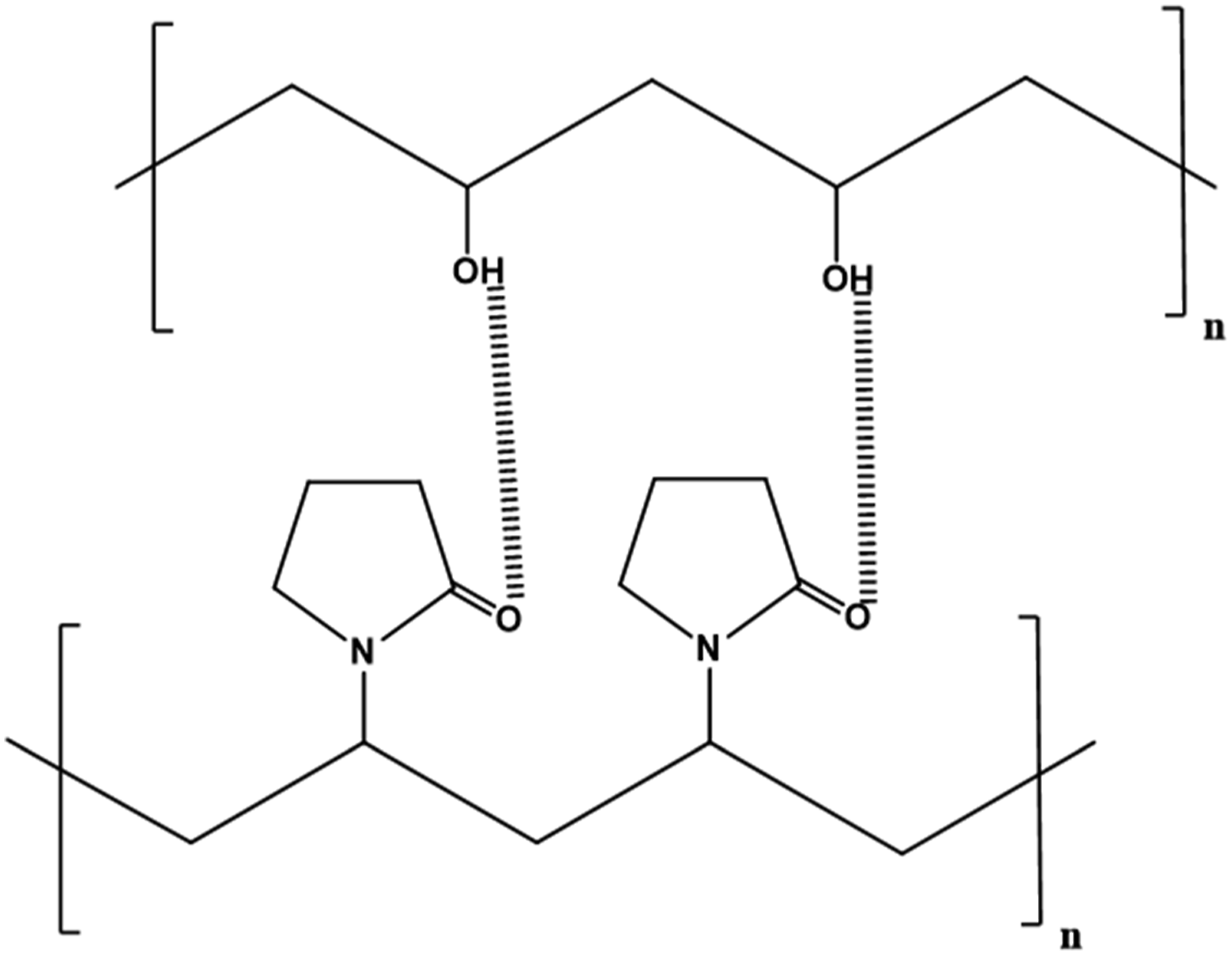
Structural features
Interaction between nanofillers and polymer blend matrix is one of the key factors in determining overall enhancement in the properties of polymer blend nanocomposite materials. These interactions and the structural features arise can be easily analyzed from the XRD and FTIR studies. PVA/PVP (50:50) blend matrix films incorporated with different contents of ZnO nanoparticles were prepared by Rajesh and coworkers
46
via a simple solution casting method. XRD of pristine blend showed a diffraction peak at about 19.6° attributed to the semicrystalline nature of PVA in the pure PVA/PVP blend. Several diffraction peaks appeared in the XRD of PNC in addition to the above one is attributed to the ZnO nanoparticles and its intensity is found to increase with the amount of nanoparticles in PVA/PVP blend increases. A similar study was carried out by Choudhary and Sengawa,
47
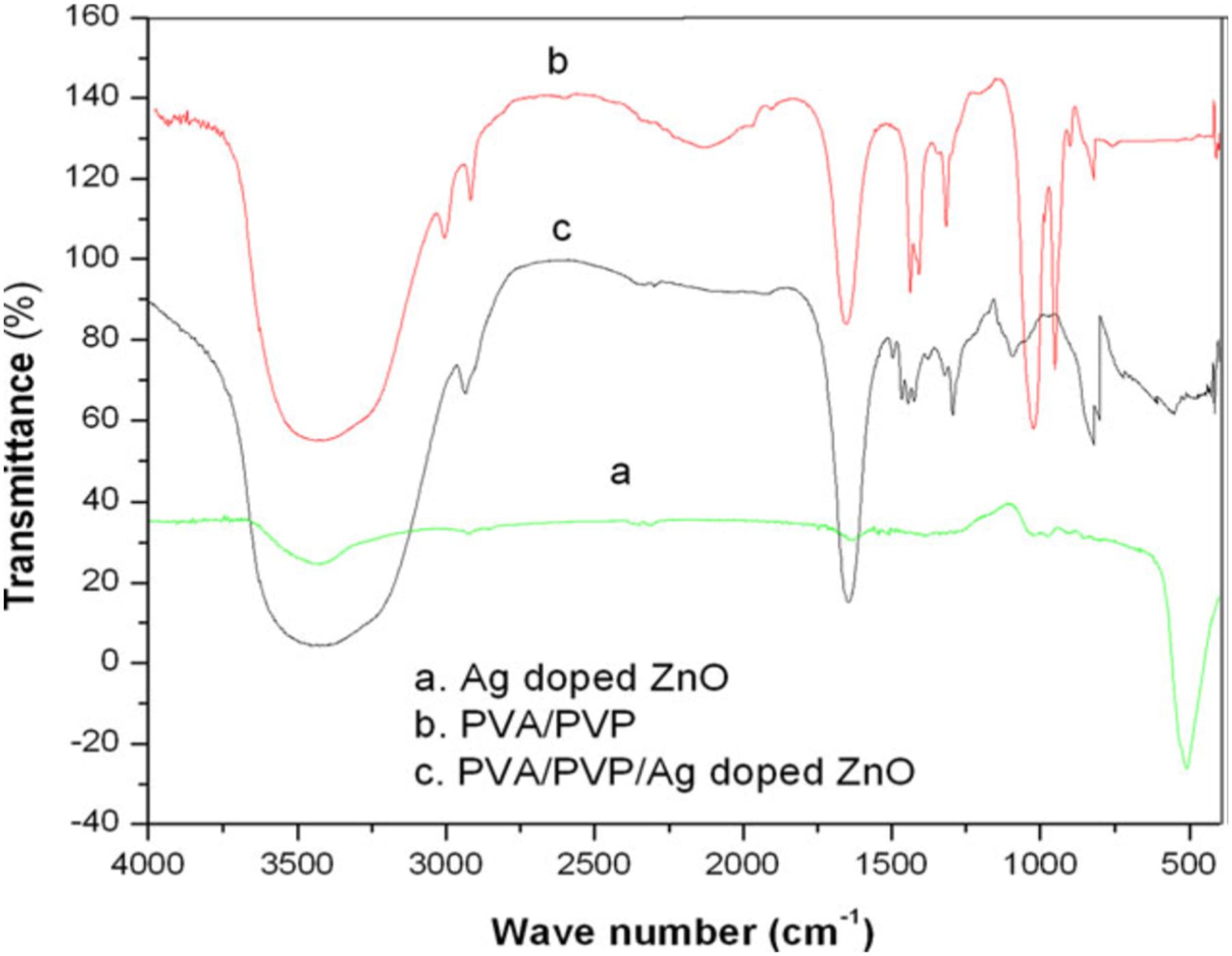
they have reported significant variation in the interplanar distance and crystallite size as the ZnO concentration increases in the PNC films which indicate the interpolymer and polymer-nanoparticles interactions. XRD peaks pertained to ZnO nanoparticles found to appear in the PNC films and its intensity is gradually increased as the concentration of ZnO changed from 1 to 5 wt. %. The interaction between nanoparticles and polymers was confirmed from the FTIR results where the PNC films reproduce all the characteristic peaks of PVA and PVP with slight variation in the intensities and no change in wavenumber. With the increase in the concentration of ZnO NPs, the intensity of these bands decreased and no prominent peaks corresponding to Zn-O stretching vibration are exhibited. XRD patterns of Ag-doped ZnO nanofillers dispersed PVA/PVP composites comprise all the characteristic bands of PVA/PVP blend as well as Ag-doped ZnO. An increase in intensities of all the peaks is attributed to the strong interaction between the polymer chain and nanoparticles (Figure 3). The addition of Ag-doped ZnO to the PVA/PVP blend has brought some changes in the FTIR of the pristine blend. The absorption band corresponding to the OH group shifted from 3431 cm−1 to 3442 cm−1 due to the strong H bonding interaction. A new band at 1084 cm−1 appeared in the FTIR of blend nanocomposite is considered as the evidence of interfacial interaction between polymer blend and nanoparticles.
48
The effect of CuO nanofiller on the structural properties of PVA/PVP blend was studied by Rajesh et al.
49
From the XRD FTIR spectra of Ag-doped ZnO, PVA/PVP, and PVA/PVP/Ag-doped ZnO composites. Reprinted with permission from Ramesan et al.
48
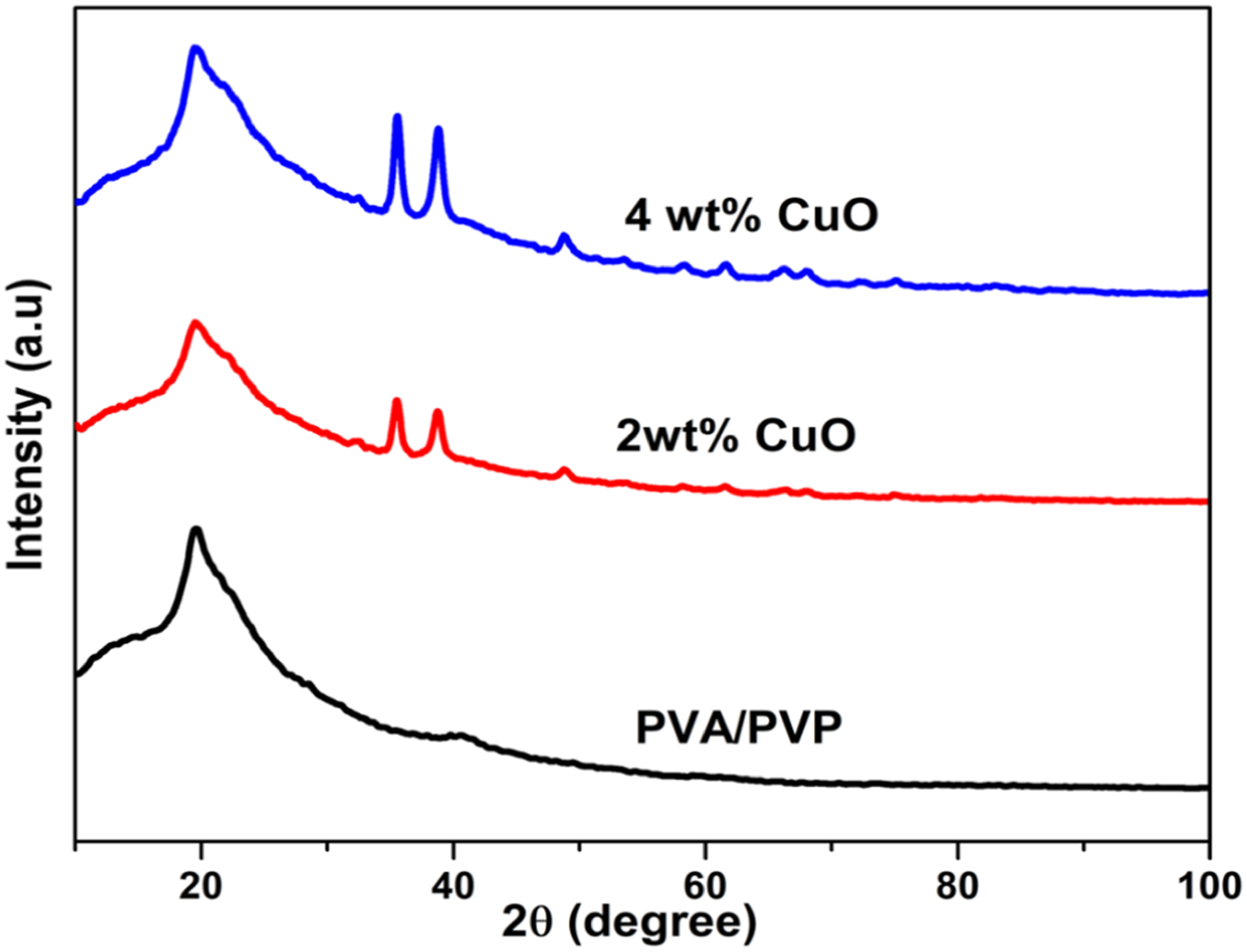
. XRD patterns of PVA/PVP and it's nanocomposites. Reprinted with Permission from Rajesh et al.
46
.
Low cost, biofriendly and easily available α-MnO2 nanorods having large surface area and structural flexibility is modified with stearic acid and used as nanofiller for the preparation of PVA/PVP/MnO2 PNC films by Mallakpour and Motirasoul. 51 A relatively easy and green ultrasonic irradiation method is employed for the preparation of these PNCs. In the XRD spectrum, peaks around 19–34° become more intense by the incorporation of a small increment of alpha MnO2-SA and no peak pertained to modified MnO2 is observed in the XRD. Interaction of alpha MnO2 with PVA/PVP blend is revealed from the shifts and changes of FTIR peaks. The peak at 1650 cm−1 pertained to the carbonyl stretching and the bands ascribed to OH group are shifted to a lower wavelength compared to pure PVP and PVA indicates the formation of H bonding. Additional peaks at 580 cm−1 and 650 cm−1 which could not be seen in the FTIR of pristine blend appear in the FTIR spectrum of alpha MnO2 dispersed PVA/PVP blend nanocomposite. The irregular shift in wavenumber, as well as variation in the intensity of FTIR peaks of TiO2 nanofiller, loaded PVA/PVP blend confirm the effective interaction among nanofiller and polymer chains through intra/intermolecular interaction. The shift of OH stretching vibration from 3292 cm−1 to lower wavenumber 3273 cm−1 upon adding 16 wt. % nanoTiO2 provides the evidence of strong hydrogen bonding. In spite of the fundamental XRD peak at 19.42° representing the semicrystalline nature of PVA/PVP blend with the additional peaks are appeared in the XRD of nanoTiO2 doped blend. As the TiO2 concentration in the blend matrix increases, the intensity of broad amorphous peaks declines and that of crystalline peaks increases. This confirms the presence of TiO2 NPs increases the crystallinity of PNC. 52 Madhloom et al. 53 studied and compared the effect of different size TiO2 nanoparticles on the physical properties of PVA/PVP blend films. XRD results revealed the small size TiO2 (15.7 nm) incorporated PVA/PVP PNC films became more semicrystalline and intensity of diffraction pattern also increased when compared with large-sized TiO2 (45.7 nm) added samples. Furthermore, the particle size of PVA/PVP/TiO2 (15.7 nm, 45.7 nm) films is observed to be 15.094 nm and 11.9613 nm respectively.
Siva et al. 43 and Guruswamy et al. 54 attempted the preparation of PVA/PVP/SnO nanocomposite where they synthesized SnO NPs by simple hydrothermal method 43 and chemical precipitation method 54 respectively. From XRD studies it was clear that the addition of nano SnO particles to the PVA/PVP blend increases its crystallinity. Crystalline peaks at about 25.91°, 33.77° and 51.64° are observed in the XRD of PNC and the intensity of these peaks increased with the concentration of SnO. Interaction of SnO nanoparticles with polymer blend is clear from the FTIR spectra of PNCs where the nano structured Sn-O stretching band at 598 cm−1 is shifted towards higher wavenumber 623 cm−1 with the raise in concentration. Mohammed et al. 55 prepared PVA/PVP (70:30)/MgO PNC via solution casting method. From the data obtained from XRD, the degree of crystallinity is determined and they have found the crystallinity of pure PVA decreased when blended with PVP. Furthermore, the addition of nano MgO to the aforementioned blend causes an increase in crystallinity and a maximum Xc was exhibited by 1 wt. % MgO loaded sample indicates the ordering characteristics of PVA/PVP blend at this ratio. No extra peak appears that signifies the nano MgO in the XRD spectrum of PNCs. Small shift of FTIR spectral bands at 2873 cm−1 and 2963 cm−1 representing symmetric and asymmetric stretching modes of the CH2 group respectively after loading nano MgO establishes the interaction among nanofiller and polymer blend matrix.
Surface morphology
Nanofillers significantly modify the surface morphology of polymers. Morphological features of PNCs have a major role in determining their properties. SEM, TEM and AFM techniques were commonly employed to study the morphology of PNCs in detail. Choudhary et al.
47
observed SEM images of 1 wt.% ZnO nanopowder incorporated PVA/PVP PNC films have unique and homogeneous beautiful 3D flower-like structures and the roughness of the surface increases with rise in ZnO concentration. Zone of agglomerated ZnO nanoparticles can be observed in the PNC with 3 and 5 wt. % of ZnO (Figure 5). Ramesan and coworkers
48
reported that there is a poor dispersion of Ag-doped ZnO nanoparticles in the blend matrix at 3 wt. % filler loading. But for 5 wt. % loading SEM images showed the spherical structure of particles with uniform morphology indicating the affinity of polar groups of polymers to the Ag-doped ZnO particles. At higher loading (10 wt. %) nanoparticles agglomerates are formed and irregular surface morphology with voids is observed (Figure 6). FESEM and TEM results confirm the better compatibility, homogeneous distribution with very less aggregation of modified CuO nanoparticles (CuO-VB1) in the PVA/PVP blend
50
and good dispersion of modified MnO2 (α- MnO2-SA) in the PVA/PVP blend matrix is evident from FESEM images, which is attributed to the increased interaction between the functional groups of α- MnO2-SA and PVA/PVP blend as well as the ultrasonication method of preparation.
45
SEM studies showed compact surface morphology for PVA/PVP/ZrO2-Th PNCs. Effective dispersion of modified ZrO2 in the PVA/PVP matrix was also evident from the TEM images. It is attributed to the ultrasonication process which prevents the aggregation of nanoparticles and decreases the size thereby leading to good dispersion.
56
Choudhari
57
prepared PVA/PVP-based PNC with different amounts of Al2O3 as nanofiller and SEM study revealed that addition of small amounts of Alumina nanoparticles can bring some modification in the morphology of PVA/PVP blend. It was observed that the roughness of the surface increases with the concentration of nano Al2O3 increases. In all the samples, uniform distribution of nanoparticles is observed with a low degree of agglomeration. Similarly, many studies are reported that the addition of metal oxide nanofillers to PVA/PVP blend changes the smooth and homogeneous surface of pristine blend to a rougher surface.38,52,54,55 Madhloom et al.
53
reported that SEM images of small sized TiO2 NPs added PVA/PVP films exhibits smooth surfaces in some regions and white spots of TiO2 NPs randomly oriented in the surface whereas in the case of large sized nanoTiO2 added films almost rough surface with segregated nanoparticles can be observed. SEM images of (PVA–PVP)–x wt. % ZnO polymer nanocomposite films (a) x = 0; (b) x = 1; (c) x = 3 and (d) x = 5 at the scales 10 μm and 2 μm, and magnifications 500× and 2.0 KX. Insets show the enlarged view of the segments of micrographs. Reprinted with permission from Choudhary et al.
47
. SEM photographs of (a) PVA/PVP (b) 3 wt.% (c) 5 wt.% and (d) 10 wt.% Ag-doped ZnO containing the PVA/PVP blend. Reprinted with permission from Ramesan et al.
48
.

Thermal properties
Thermal properties of PNCs can be analyzed using TGA, DSC, DTA, etc. Enhanced thermal stability is expected for metal oxide dispersed PVA/PVP polymer blend nanocomposites. Melting behavior and Glass transition temperature of polymer rely on molecular weight, crystallinity, the segmental motion of polymer chain, and the polar–polar interaction between polymer matrix and reinforcing filler. Choudhari and Sengwa 47 performed a thermal analysis of ZnO NPs doped PVA/PVP nanocomposite films. From the DSC thermograms, good compatibility between the two polymers in all the PNCs films was clear as it showed a single glass transition temperature (Tg) value centered around 100°C. Plasticizing effect of ZnO NPs on PVA/PVP blend was evident from the decrease in Tg values up to 3 wt. % ZnO. Tg of pure blend is observed to be 101.38°C and it decreased to 92.32°C for PVA/PVP/3 wt. % ZnO. A sharp rise in Tg of 5 wt. % ZnO doped PVA/PVP blend film is attributed to the agglomeration of nanoparticles. Whereas, Ramesan and coworkers 48 reported a different result that Tg of Ag-doped ZnO loaded PVA/PVP nanocomposites is smaller than the Tg of pristine blend and it is increasing with the metal oxide concentration. In a study by Guruswami et al., 54 all the SnO nanofiller incorporated PNC films showed higher Tg than the parent blend and with the increase of SnO concentration from 2 to 10 wt. %.
Figure 7 shows DSC thermograms of PVA/PVP (50:50) blend and 1, 3 and 5 wt.% Al2O3 added PNCs. Endothermic peaks located around 100°C and 210°C for PVA/PVP/x wt.% Al2O3 films were corresponding to Tg and Tm respectively. Tg value of 3 wt. % alumina dispersed PVA/PVP blend nanocomposite showed a significant decrease when compared with other samples, it is attributed to the destruction of intermolecular interactions between PVA and PVP in the presence of nanofillers. The Tm values are found to be higher than that of pure blend confirming the thermal stability. The highest thermal stability was observed for the sample with 1 wt. % nano alumina particle and whose crystallinity was found to be lowest indicating the effective interaction between blend and nanofiller.
57
Mohemmad and coworkers
55
investigated the thermal properties of PVA/PVP blend and MgO loaded PVA/PVP PNCs. From DTA curves the melting temperature (Tm) of PVA/PVP blend is determined which was found to be less than that of virgin PVA indicating the decrease in crystallinity. On adding MgO to the PVA/PVP blend, Tm is again increased and reached a maximum value of 214°C for 2 wt. % MgO loaded sample indicating the increased crystallinity as a result of nano MgO loading. The decomposition temperature of PVA/PVP blend was higher than that of pure PVA, whereas the presence of nano MgO decreases Tg to 270°C for PVA/PVP/2 wt.% MgO. From DSC analysis Tg of PNC films are found to be in the range of 82°C–86°C. According to Attaran et al.,
58
thermal stability of PVA/PVP blend is largely enhanced with BaZrO3 nanofiller is attributed to the excellent thermal stability of BaZrO3 nanoparticles and strong hydrogen bonding interaction present in the nanocomposite films. Many studies have reported the higher thermal stability of polymer nanocomposite than the pure blend. The composition of different PNC based on PVA/PVP with maximum melting temperature (Tm) and thermal stability reported by different researchers is given in Table 1. The DSC thermograms of (PVA–PVP)–x wt. % Al2O3 polymer nanocomposite films for (a) x = 0; (b) x = 1; (c) x = 3, and (d) x = 5. Reprinted with permission from Choudhary.
47
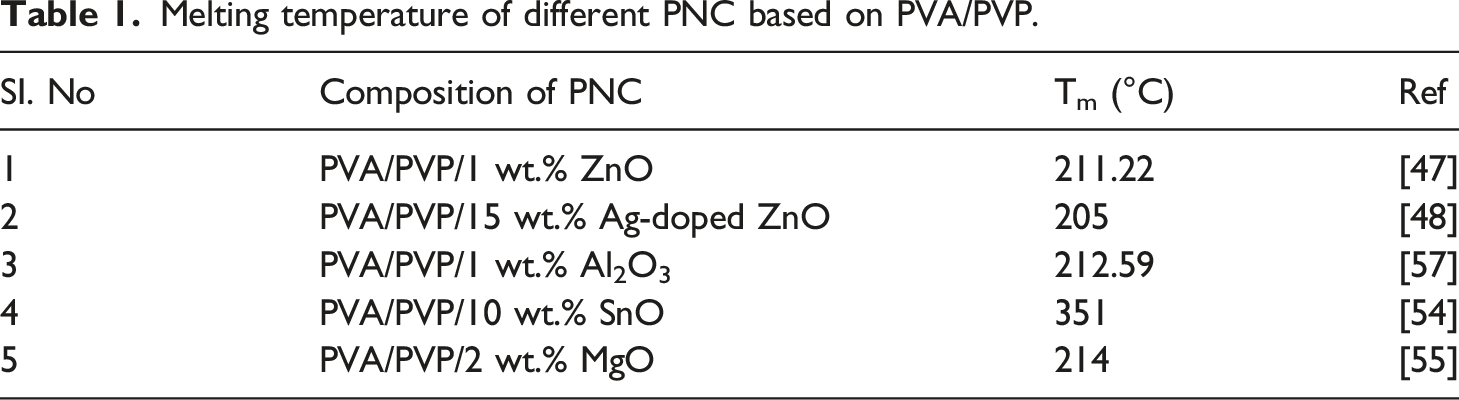
Melting temperature of different PNC based on PVA/PVP.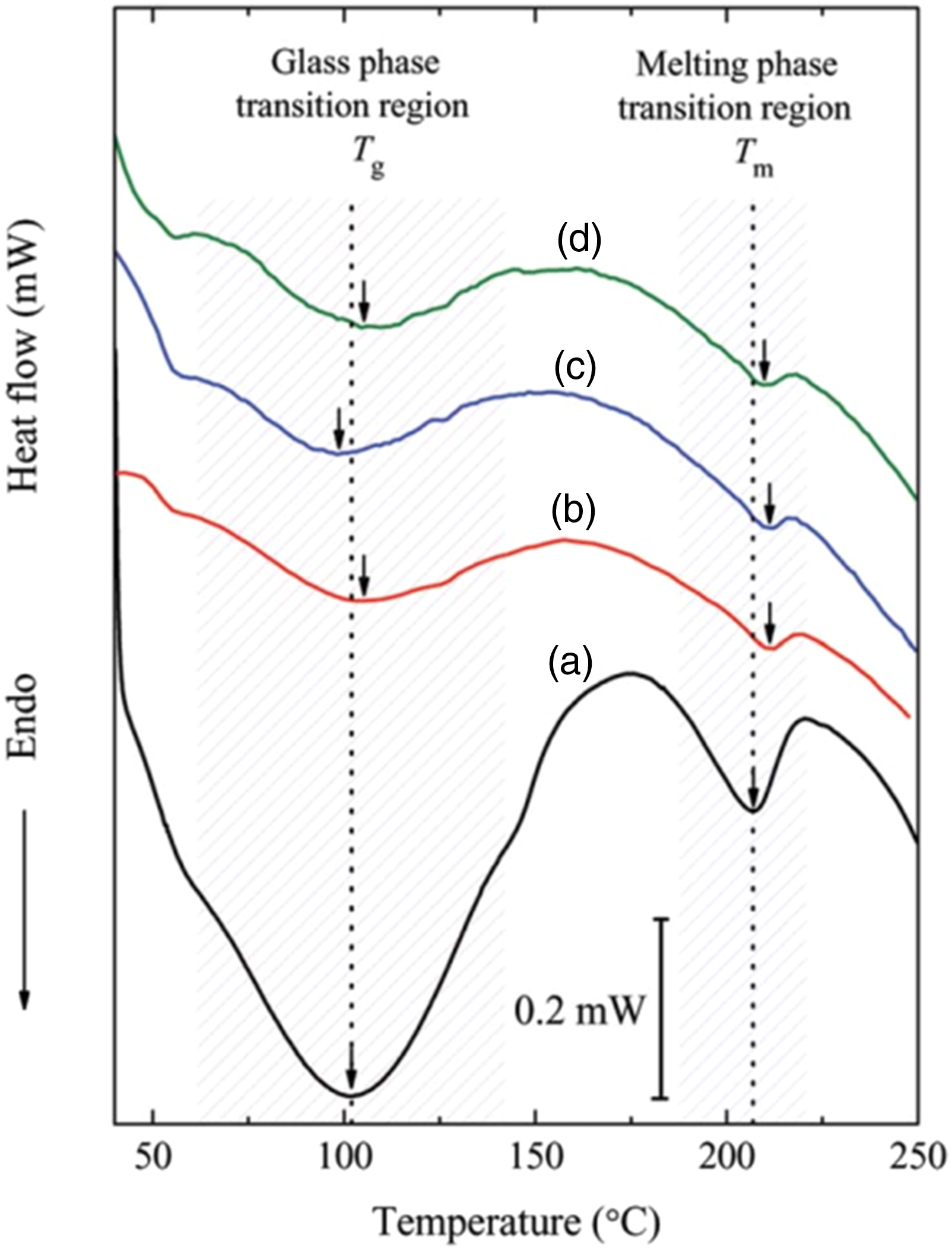
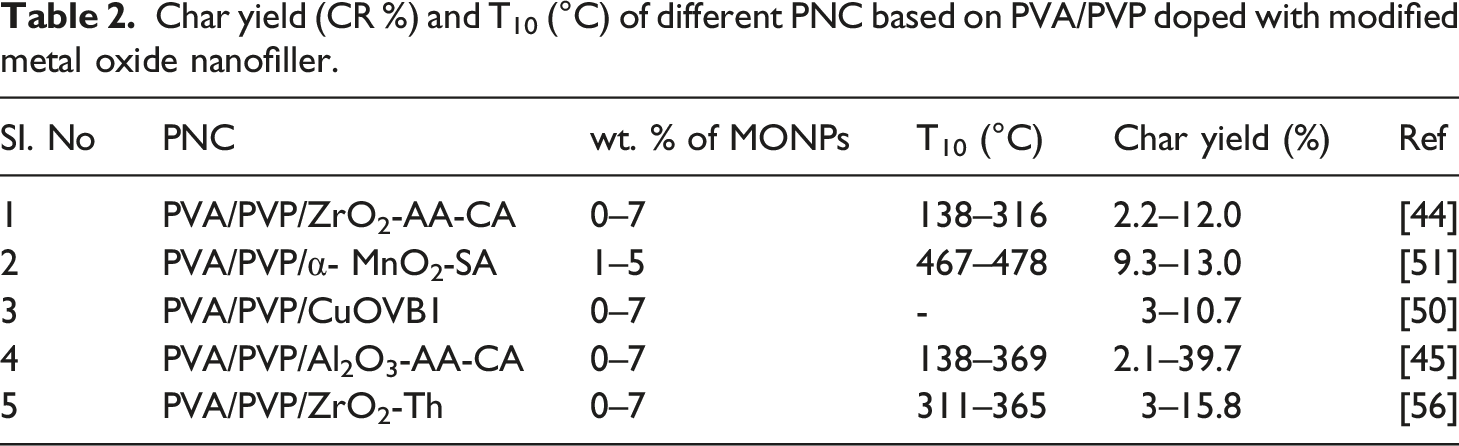
Char yield (CR %) and T10 (°C) of different PNC based on PVA/PVP doped with modified metal oxide nanofiller.
Optical properties
PVA/PVP blend exhibits dopant-dependent optical properties and these are extensively studied by researchers since it has potential applications in photonics, optoelectronic devices, etc. Choudhary and Sengwa
47
reported the effect of ZnO nanoparticles in the optical behavior of PVA/PVP blends. From UV- Visible absorbance spectra of PVA/PVP/ZnO films, they observed that the sharp absorption edge around 240 nm shifted towards a higher wavelength region and the absorbance also increased with the increase in ZnO concentration. The absorption band at 279 nm is appeared as such in pristine blend as well as in ZnO NPs doped blend films. A similar observation was made by Rajesh et al.
46
from the UV-Vis spectra, where they incorporated 2 to 16 wt.% ZnO nanofiller into PVA/PVP and the absorption edge is found to shift towards the longer wavelength side. In the former scenario, a ZnO nanoparticle with a size of 100 nm has utilized, and a band at 375 nm gets more intense and keeps its location as its concentration increases.
47
However, no such absorption bands emerged in this location where the nano ZnO size is 50 nm. The direct optical bandgap energy (Egd) of pure PVA/PVP is observed around 5 eV and the addition of ZnO nanoparticles (<50 nm) reduces the Egd to 2.90 eV for 8 wt% ZnO.
46
On the other hand, Egd did not show a significant decrease with particle size 100 nm.
47
Luminescence property of PVA/PVP/ZnO PNC films investigated using an excitation wavelength of 220 nm. A spectrum with three peaks is obtained. Maximum intense peak was observed around 440 nm for all the samples and its intensity raised to 8 wt. % nano ZnO concentrations and above which the intensity decreased due to agglomeration of nanoparticles.
46
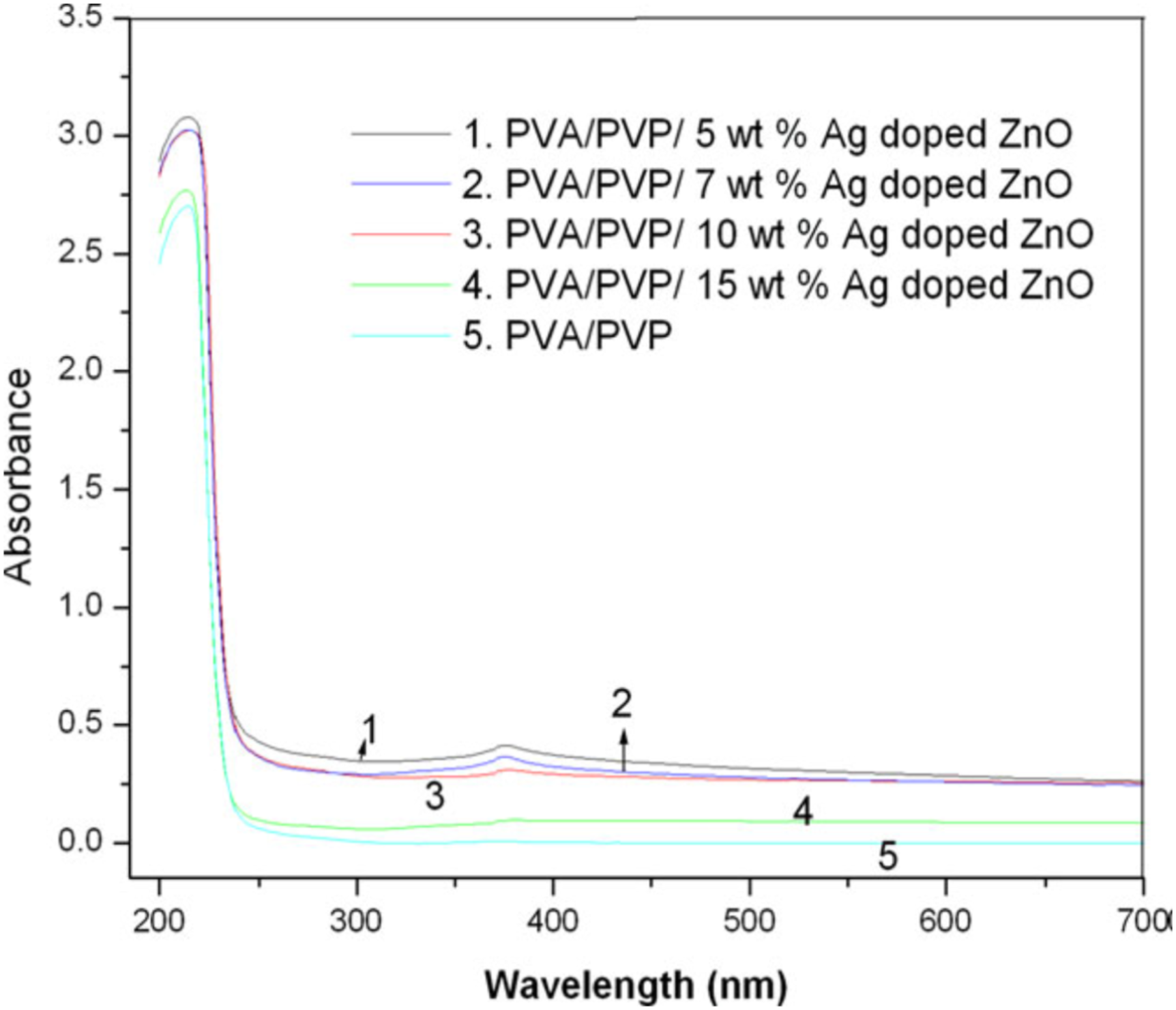
Figure 8 depicts the UV-Vis spectrum of Ag-doped ZnO loaded PVA/PVP PNC films. In addition to the absorption band of pure blend, a new absorption at 378 nm indicating the presence of nanoparticles in the blend has appeared in the spectra. Compared to the pure PVA/PVP blend all the Ag-doped ZnO added blend films showed higher absorption and a maximum absorbance is shown by 5 wt. % nanoparticles embedded polymer blend.
48
Efficient absorption of ZnO doped PVA/PVP in the UV region is attributed to the wide bandgap energy of ZnO (3.37 eV).47,48 UV spectrum of PVA/PVP blend and the blend with different contents of Ag-doped ZnO. Reprinted with permission from Ramesan et al.
48
.
TiO2, a semiconductor with a wide bandgap of 3.2 eV is used as a nanofiller by Rajesh et al. 52 and Madhloom et al. 53 UV -Visible spectral investigations have shown that, with the rise in doping concentration, absorption bands observed around 200–300 nm for pure PVA/PVP shifts toward higher wavelengths with varying intensity. Madhloom et al 53 reported an absorption band at 360 nm for TiO2 in the UV spectrum of PVA/PVP/TiO2 PNCs. The intensity of these absorption bands increased as the particle size of TiO2 NPs increased from 15.7 to 45.7 nm, which can be attributed to the ability of TiO2 nanoparticles to absorb more incident light. Moreover, the Egd of nanocomposites is observed at 2.80 eV and 2.30 eV for particle sizes of 15.7 and 45.7 nm, respectively.
Mallakpour and Mansourzadeh
50
prepared PVA/PVP doped with modified CuO (CuO-VB1) which were found to be highly transparent (Figure 9). Absorbance is increased as well as the absorption peak is found to shift slightly towards a longer wavelength with the nanofiller content increases. Pictures of the blend PVA-PVP (a), NC-3 wt.% (b), NC-5 wt.% (c) and NC-7 wt.% (d). Reprinted with permission from Mallakpour et al.
50
.
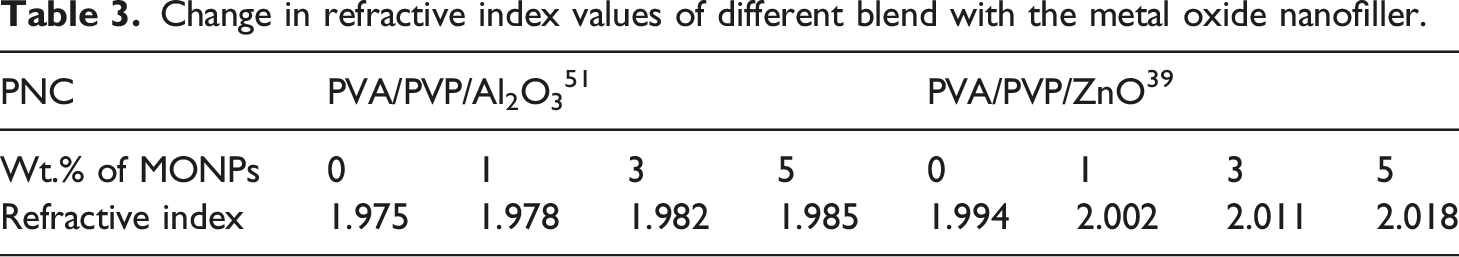
Change in refractive index values of different blend with the metal oxide nanofiller.
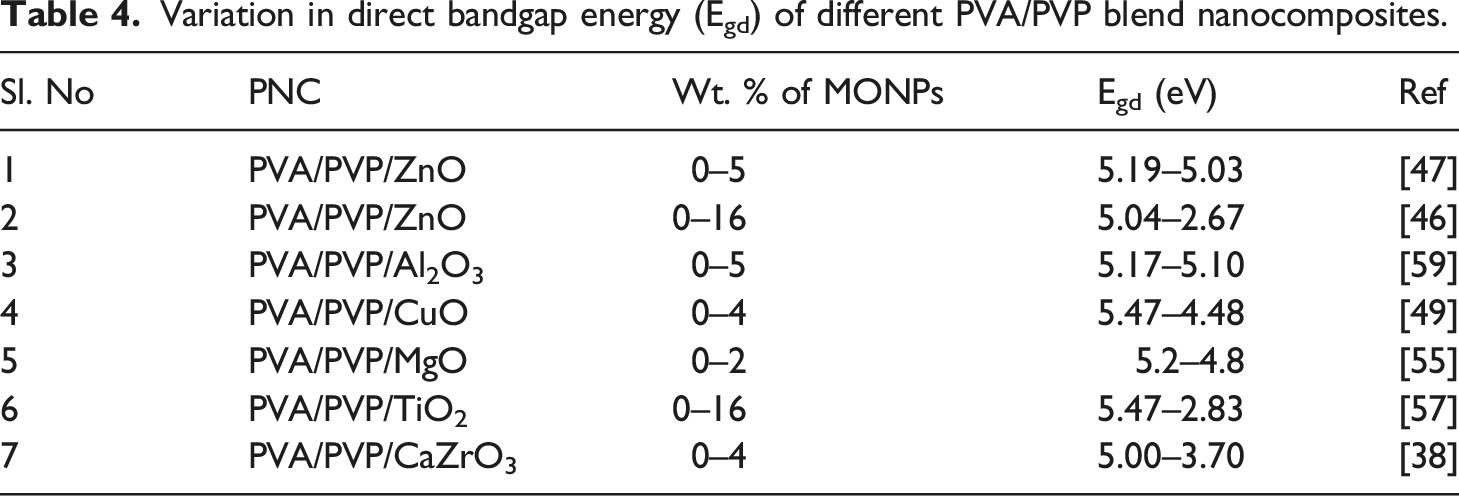
Variation in direct bandgap energy (Egd) of different PVA/PVP blend nanocomposites.
Electrical properties
Electrical properties of MONPs dispersed PVA/PVP blend nanocomposites are investigated by Ramesan and coworkers
48
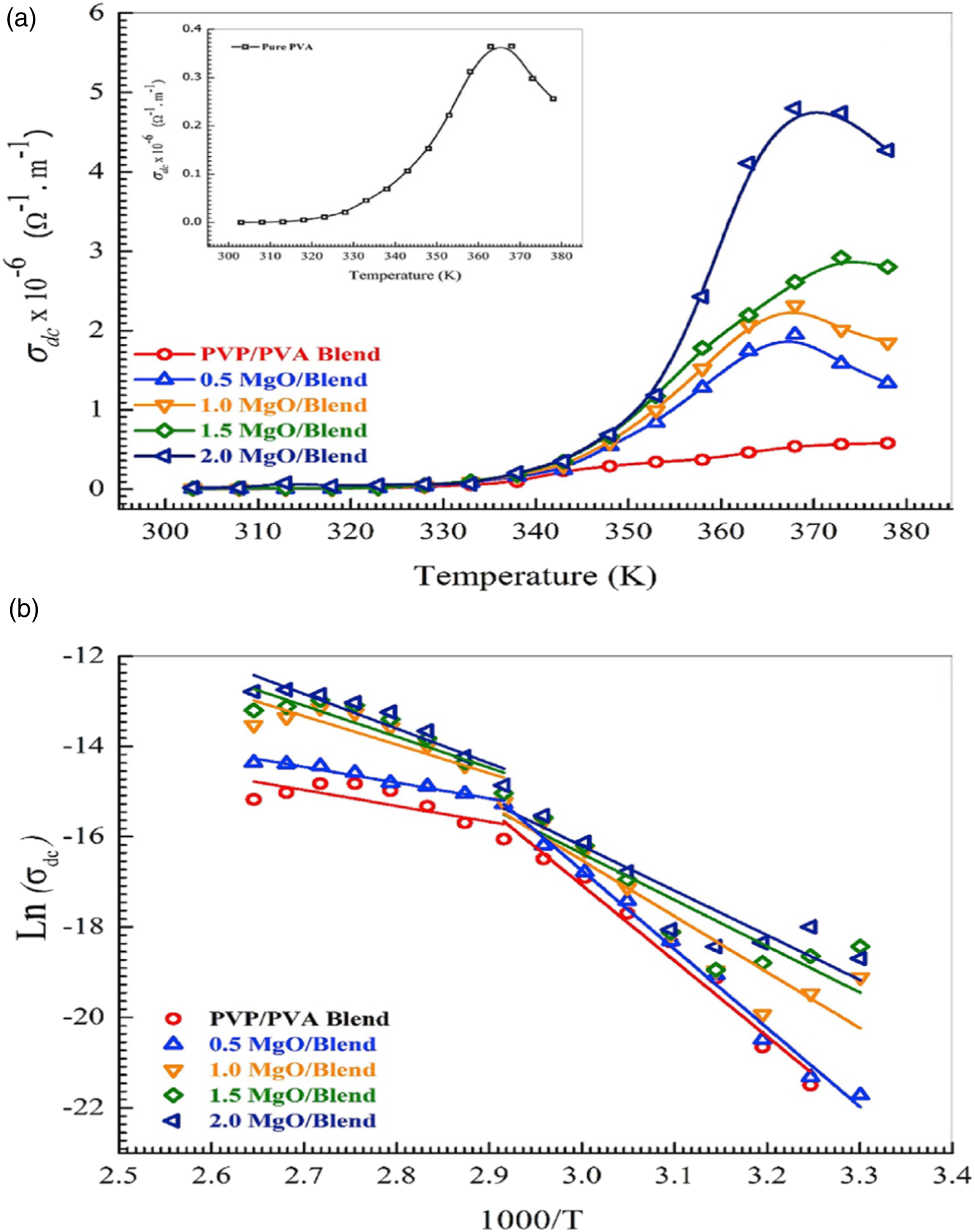
studied the influence of conductive metal (Ag) doped ZnO nanoparticles in the electrical properties of PVA/PVP blend films. AC conductive studies of PNC films showed higher conductivity values at a higher frequency which indicates the formation of excess charge carriers in the polymer matrix. The conductivity of PNC samples is found to be higher than that of pure blend and the conductivity of nanocomposites increased with the concentration of nanofiller in the blend matrix. PVA/PVP nanocomposite with 5 wt. % Ag-doped ZnO exhibits highest electrical conductivity owing to the uniform dispersion and the spatial arrangement of nanoparticles within the polymer matrix. Beyond that particular concentration, PNCs exhibit poor conductivity due to the agglomeration of NPs at higher loading. Mohemmad et al.
55
reported that blending with PVP and incorporation of MgO nanoribbons influences the DC conductivity of the PVA. They prepared PVA/PVP (70:30)/MgO PNC films with varying contents of nanofiller. DC conductivity is found to increase with temperatures and a maximum σdc value is shown by all the films at around 368°C. The DC conductivity increases with the MgO concentration can be attributed to the three-dimensional conducting pathway formed by MgO nanoribbons while they connect with each other. Figure 10 depicts the Arrhenius behavior followed by σdc values. As the concentration of nano MgO ribbons increases, more will be the conduction paths introduced in the blend matrix and as a result of this σdc are found to increase and Ea decreased. Rajesh et al.
52
investigated the DC conductivity of PVA/PVP/TiO2 films, which is found to increase with doping concentration up to 12 wt.%. This is attributed to the formation of charge transfer complex within the blend matrix which reduces barrier height between trapping sites. The highest conducting sample is one with 12 wt.% TiO2 loaded PVA/PVP and the conductivity is 1.86 × 10−10 Scm−1. Maximum AC conductivity is exhibited by the film with the same composition (PVA/PVP/12 wt.%). In a study by Attaran et al.,
50
they proposed the use of PVA/PVP/BaZrO3 nanocomposite films as proton exchange membrane fuel cells. PVA/PVP/1 wt. % BaZrO3 is observed to have the highest proton conductivity (3 × 10−2 Scm−1) at 25°C, which is much higher than pure PVA and PVA. On increasing temperature, the proton conductivity also increased and displayed 6.01 × 10−2 Scm−1 conductivity at 70°C. (a) Dependence of DC conductivity in PVP/PVA (blend) and blends loaded with MgO (b) Ln σdc against 1000/T for the investigated films. Reprinted with permission from Mohammed et al.
55
.
Applications
Metal oxide nanoparticles doped PVA/PVP blend nanocomposites have found applications in various fields (Figure 11). Choudhary and Sengwa 47 investigated the optical, electrical and thermal properties of ZnO NPs dispersed PVA/PVP PNC, which revealed the possibility of these PNC to be used in the fabrication of flexible nanodielectrics for multifunctional microelectronic devices such as high energy density capacitors, electronic packaging, photovoltaic devices, sensors, electronic shielding devices. Surface Plasmon Resonance (SPR) property of ZnO doped PVA/PVP blend nanocomposites confirmed its utility as tunable photosensors for optoelectronic devices. The detailed optical studies of PVA/PVP/Al2O3 PNC films assured its use as promising candidates for the partial protection of device surfaces from the incident visible photons and appreciable controllable shields for UV radiation. The optical band gap values obtained for PVA/PVP/Al2O3 PNC films confirmed its utility as bandgap regulated optical materials in designing transparent type advanced optoelectronic devices. 59 PVA/PVP incorporated with nano CuO modified with thiamine (CuO-VB1) showed antimicrobial properties and hence can be used in the antimicrobial wall coverings, hospital equipment and antimicrobial packaging materials as well as to control bacterial infections in medical, environmental and chemical fields.
Mallakpour et al.
50
explored PVA/PVP/5 wt. % α- MnO2-SA films as an effective tool for the removal of adsorbing CdII ions from water bodies via the adsorption method. They reported 81% removal percentage at 20 mgL−1 CdII ion concentration and 63% at 100 mgL−1 concentration. Rajesh et al.
52
confirmed the use of 4 wt. % nano TiO2 loaded PVA/PVP nanocomposite films for luminescence applications. For a material to be used for UV shielding purposes there should be a thoughtful balance of both greater transmittance in the visible region and higher optical absorption in UV regions. PVA/PVP PNC with 1 wt. % CaZrO3 exhibited an optimum UV shielding action and is evaluated by monitoring the UV irradiation induced photocatalytic degradation of IC solution. These studies revealed the suitability of 1 wt. % CaZrO3 nanofiller incorporated PVA/PVP films as mechanically flexible UV protective applications.
38
A study of optical characteristics of PVA/PVP incorporated with ZrO2, which was modified with Ascorbic acid and citric acid (ZrO2-AA-CA), revealed its application as an efficient candidate for UV shielding materials.
44
Hashim
60
investigated the optical and electronic properties of In2O3 and Cr2O3 nanoparticles doped PVA/PVP films to explore their use in flexible optoelectronic devices. Enhanced electrical and optical properties of these PNCs assured their use in huge optoelectronics fields like sensors, transistors, photovoltaic solar cells and electronic gates. Attaran et al.
50
prepared PVA/PVP/BaZrO3 nanocomposite films and investigated its fuel cell performance in various compositions. 1 wt. % BaZrO3 loaded sample is proved to be potential electrolytes in fuel cell device. Applications of PVA/PVP/MONP.
Conclusions
As there is a growing public interest in eco-safe polymeric materials for various applications, researchers have been focusing on biodegradable polymers. PVA and PVP have got great attention due to their several attractive properties including biodegradability. The presence of polar functional groups in these polymers facilitates the formation of miscible blends and provides an excellent opportunity to incorporate nanoparticles into it. Many studies have proved that structural, mechanical, dielectric, electrical and optical properties of PVA/PVP blend films can be effectively tailored by the addition of various nanofillers to it. Metal oxide nanofillers incorporated PVA/PVP blend nanocomposite materials are found to exhibit enhanced properties suitable for areas of biomedical, thermal conductors, optoelectronics, semiconductors and electrical insulators. Still, there is scope to employ various kinds of MONPs-based transition, inner transition metals and modified MONPs to bring fascinating properties in the resulting PVA/PVP blend nanocomposite materials.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
