Abstract
This paper proposes a new approach for improving the compatibility of liquid crystalline polymers with engineering thermoplastics. To improve compatibility and mechanical performance of poly (phenylene oxide) (PPO)/liquid crystalline polymer (LCP) A950 blends, a third component compatibilizer is incorporated into the processing step. In this work, epoxy containing acrylate rubber (ACM) is used for the compatibilization of PPO/LCPA950 blends through the chemical reaction of epoxy functional groups of the acrylate rubber with terminated carboxylic acids or hydroxyl end groups of the two phases. The compatibilization effect on the properties of PPO/LCPA950 blends is investigated by adjusting the amount of acrylate rubber. Fourier transform infrared (FTIR) spectroscopy and melt-rheological analysis were used to study the compatibilization mechanism of acrylate rubber, and the results demonstrate that acrylate rubber is capable of reacting with PPO/LCPA950 to form a chemical bonding interface. The electron microscopic images revealed a well-compatibilized microstructure of PPO/LCPA950 blends in presence of acrylate rubber with submicron-sized liquid crystalline polymer domains in the continuous poly (phenylene oxide) matrix phase, which could not be detected for immiscible PPO/LCPA950 blend. The dynamic mechanical analysis shows that PPO/LCPA950 blend with acrylate rubber exhibit greater elastic storage moduli. It was observed that blends of PPO/LCPA950 compatible with acrylate rubber showed significant increases in tensile strength as well as notched impact strength. These blends can be used in automotive industry.
Introduction
The use of polymer mixes has significantly increased over the last few decades; they can be used to create materials with a variety of properties greater than those of the individual properties. However, the definitive properties of such mixes are vigorously impacted by the morphology of the systems involved as well as the interfacial adhesion between them. For general-purpose applications, liquid crystalline polymers (LCP) and poly (phenylene oxide (PPO) both work well as molding resins. Because of their superior chemical resistance, thermal stability, and other desirable qualities, they are particularly desirable in the electrical and electronics industries. However, each type of switch has a certain set of limitations.
Poly (phenylene oxide (PPO) is cost effective, possesses high impact tolerance and higher glass transition temperature, thus, making it a perfect material for high temperature stiffness. Yet, the performance of PPO is poor in typical thermoplastic fabrication equipment due to its viscosity. Therefore, in order to enhance the processibility, of this polymer, it was combined with other polymers such as polystyrene or copolymers with a styrene derived component. It has been reported that blending with these similar copolymers has reduced glass transition temperature, and lowered the stiffness by at least 40°C compared with pure PPO. It is necessary for a material with PPO's high temperature stiffness to process well and have good toughness at the same time in order to fulfill the needs of the industry. 1
Liquid crystalline polymers (LCPs) have been studied extensively in blends with thermoplastics.2–8 During processing, LCPs can form oriented fibrils in the blend, which can act as a reinforcing component. Therefore, they were also called in situ composites. The interest arises from two major advantages of blending LCPs with engineering thermoplastics. First, low melt viscosity LCPs can result in substantial reductions in melting viscosity of thermoplastics blends, improving the processability of engineering plastics when added to thermoplastics. Secondly, LCPs have a more rigid molecular structure, and under conditions of shear and extension during processing, they generally exhibit a high degree of order. As a result LCP can thus deform into fibrils, which serve as reinforcing elements in the in situ composite.9,10 Because of this, liquid crystal polymers (LCPs) possess extremely high mechanical properties when oriented molecularly. Many studies have been conducted on blending with LCPs and thermoplastic materials such as polycarbonate, polyester, polyethylene polyamide.11–18
Polymer blends that are immiscible exhibit morphologies with two phases as well as poor chemical and physical interactions across phase boundaries. Due to poor interfacial adhesion between the LCP domains and polymer matrix, polyblends suffer from low tensile strength and modulus. A phase morphology forms during processing depends on the process parameters, the intrinsic properties, and the interfacial properties of the component polymers. A third component, commonly known as a compatibilizer, can be used to modify the properties of such blend systems. A compatibilization technique can improve interfacial adhesion and reduce colloidal formation by introducing a third component into the system or by performing an in situ reaction between constituents.19–21 To improve the compatibility between PPO and LCP, a new type of compatibilizer i.e acrylic elastomer (ACM) was introduced. It will act as a bridge between the PPO matrix and LCP phase domains, and it will extend well beyond the PPO matrix into the LCP phase domain. Due to this, it is expected that this type of compatibilizer exhibits good interfacial activity and reinforces the interface. By improving PPO and LCP compatibility, the comprehensive properties of the blend system could be enhanced.
Numerous investigations on poly (phenylene oxide) (PPO/PS) alloys (NorylTM) and LCP mixtures have been conducted. Viswanathan et al. 22 investigated the mixes’ miscibility, rheology, mechanical characteristics, and morphology. It was discovered that a modest amount of LCP improved processibility by reducing the blend's viscosity. A LCP fibrous structure was revealed in injection-molded samples of the polymer blends, which had improved mechanical properties over pure PPO/PS alloys.
Thus, the aim of the work was to study the effect of ACM elastomer in PPO/LCP blend system. Where ACM elastomer can act as a compatibilizer which plays an effective role on reducing the Tg of the PPO matrix. Thus selection of ACM is vividly justified for this blend system. The compatibilization technique is used to improve the interfacial adhesion between the two phases giving rise to a stable morphology and enhanced properties of final composites such as high temperature capability, low temperature impact resistance, and excellent mechanical strength, which are essential for automotive industry.23,24
Experimental Section
Materials
A poly (phenylene oxide) powder (Mw = 45,000) with an intrinsic viscosity of 0.4 dlg−1 (measured in chloroform at 25°C) was purchased from General Electric Co.. It has a Tg of about 216°C. (LCPA) Vectra A950 produced by Ticona, USA was used as a thermotropic liquid-crystalline polymer. Vectra A950 is copolyester of 75 mol% P-hydroxybenzoic acid (HBA) and 25 mol% 2, 6-hydroxynaphthoic acid (HNA), which exhibits a nematic mesophase in the molten state. The elastomer used in this study was acrylic rubber (ACM) NIPOL AR 51 was obtained from Nippon Zeon Co. Ltd, Tokyo, Japan, which acts as a compatibilizer.
Blending and Extrusion
Materials designation and compositions.

Characterization
FTIR Measurements
The effect of acrylate rubber addition on the chemical structure of PPO/LCP blends was examined using Fourier transform infrared spectroscopy (FTIR). Thin-film specimens were prepared using a compression molding. FTIR measurements were conducted at room temperature between 400 and 4000 cm−1 using a NEXUS 870 FTIR (Thermo Nicolate). A total of 32 scans were averaged with a resolution of 4 cm−1.
Differential scanning calorimetry
Differential scanning calorimetry (DSC) analysis was conducted using Perkin Elmer DSC-7 machine. The weight of all samples was approximately 10 mg. The analysis was done by raising the temperature from 50 to 200°C at a rate of 10°C/min in inert atmosphere.
Dynamic mechanical analysis
A dynamic mechanical analyzer (DMA 2980) was used to measure the dynamic modulus and glass transition temperature of the composites. In a nitrogen atmosphere, the dynamic temperature profiles of the composites were determined in single cantilever mode at a constant vibration frequency of 1 Hz and with a heating rate of 5°C/min. The size of the testing specimen was 35 × 12.76 × 3.27 mm3.
Heat deflection temperature
Heat Deflection temperature (HDT) indicates how much a polymer or plastic sample deforms under a specified load. The use of thermoplastic components involves many aspects of product design, engineering, and manufacturing. Measurements of heat deflection temperatures (HDT) were performed in accordance with ASTM-D648, standard using 455.05 KPa of loading and a heating rate of 2.0°C per minute.
Rheological Properties
Torque versus time measurements was conducted in a Brabender Plastic-Corder mixer with a 30 gm capacity to verify the reaction between PPO and LCPA with ACM. The rotational speed was set at 30 r/min and the temperature was controlled at 300°C. Rheology measurements were performed using an Instron capillary rheometer model. Samples were loaded in pellet form at 300°C. After loading, the system reached thermal equilibrium after approximately 15 min. A capillary with a die diameter of 0.762 mm (L/D = 33.3) was used to cover a shear rate range of −50 to 4 × 102 S−1. Melt flow rates (MFRs) of matrices and blends were measured at 300°C using a 5-kg load.
PCOM Evidence for Interfacial Chemical Reaction
A sample of the polymer was placed on a cover glass and a sufficient amount of DMF solvent was poured over it, the PPO and ACM dissolved in the solvent and formed transparent amorphous thin films. The shape and size of dispersed LCPA particles in polymer blends could be maintained due to its insolubility in DMF. A contrast-phase optical microscope was used to observe the LCPA particles directly dispersed within a transparent matrix. In order to observe the optical texture of LCPA components, a polarized optical microscope Leica-DMPL (Leica Co.) was used.
Scanning electron microscope observation
A scanning electron microscope (SEM), Jeol JSM model, was used to investigate the morphology of cryogenically fractured surfaces of extruded strands. During the scanning of the composite samples, the vacuum was 10−4-10−6 mmHg. A weight-average diameter (Dw) of the LCPA droplets could be calculated using the following equation:
26
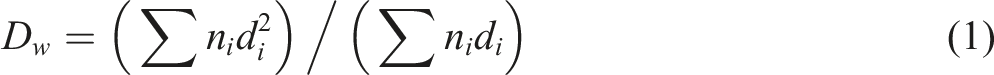
Mechanical properties
The samples’ toughness was evaluated using Notched Izod impact tests following the ASTM D 256 at room temperature. The samples were cut from tensile bars with dimensions of 64 × 12.7 × 3.2 mm. Each value reported represented the average of five samples. Tensile testing was conducted on dumb-bell-shaped samples after cooling the samples for at least 24 h after molding. A universal testing machine (Hounsfield HS 10 KS) was used to perform the tensile test at 5 mm/min extension speed. The initial gauge length measured was 35 mm. The standard deviations of the mechanical properties were reported.
AFM (Atomic force microscopy) study
AFM (Nanonics SPM 100) was used at room temperature to characterize the surface topography of the samples. All imaging was performed in Tapping-modeTM at its resonant frequency. In this study, a triangular cantilever with a fabricated QUARTZ tip (radius 5 to 10 nm) was used with a spring constant of 44 N/m in the imaging mode and scanning direction was perpendicular to the long axis of the cantilever. A Leica ultramicrotome cutter was used to smooth out the samples.
Results and discussion
FTIR studies
It is generally known that the epoxy group is capable of reacting with polymers' carboxylic (-COOH) or hydroxyl (-OH) end groups. It is necessary to understand that the acrylate rubber (ACM) used in this study contains epoxy groups as the reactive compatibilizer, which leads the ACM to interact with the hydroxyl groups or end-groups in PPO and LCPA at the interface during melt mixing, resulting in PPO-co-epoxy-co-LCPA graft copolymer. The PPO and LCPA employed in this study have end-groups of the carboxyl and hydroxyl form. The chemical structures of PPO, ACM and LCPA and related reactions involved between epoxy and end-groups of PPO and LCPA are shown in Scheme 1. The chemical structures and reactions of PPO, LCPA and ACM components.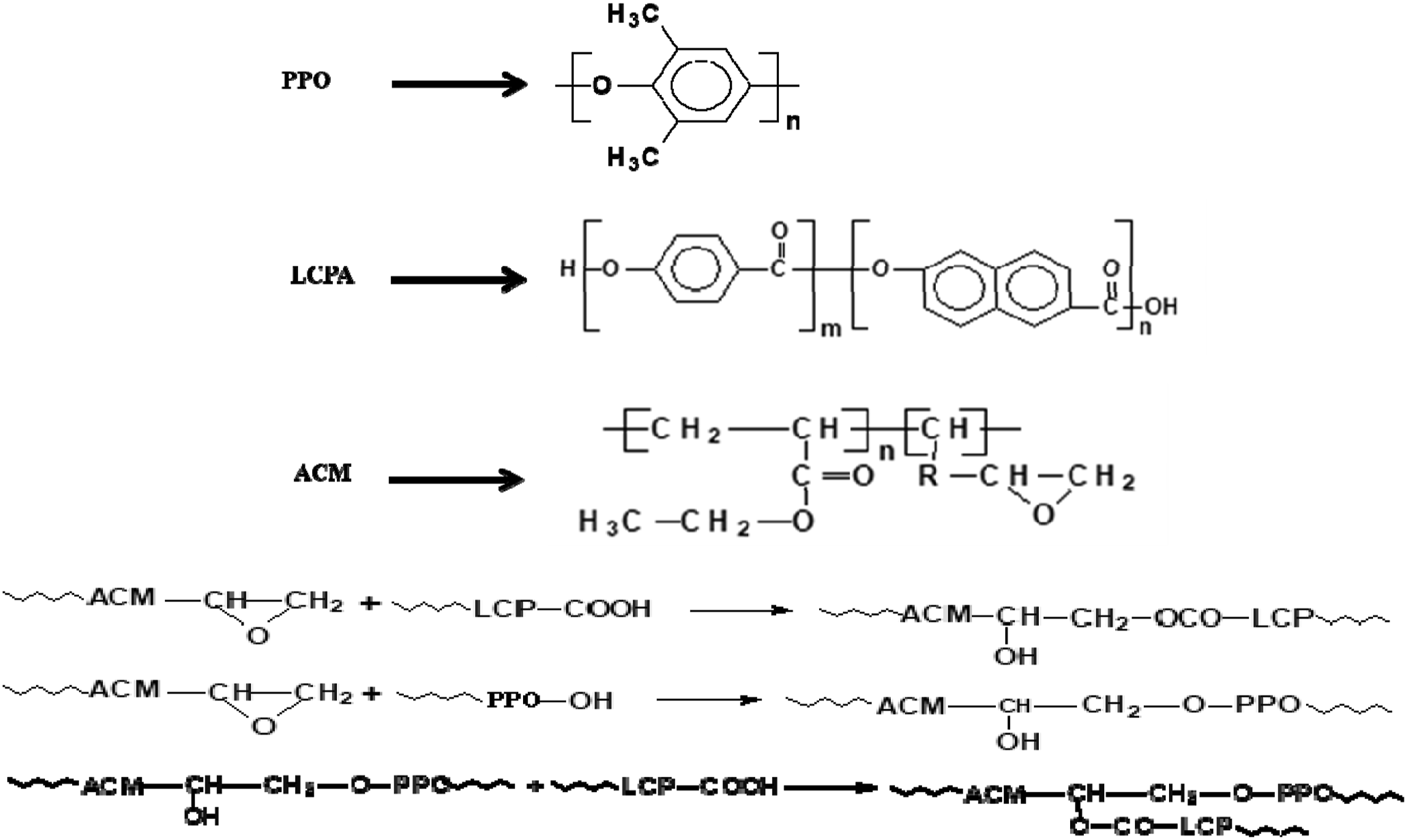
FTIR allows the study of molecular interactions within a blend by comparing the spectra of the blend and the component polymers. The spectral differences are normally manifested in shifting absorption frequencies, broadening bands, and changing absorption efficiencies. The IR peak at 908 cm−1 was a characteristic response of the epoxy group that has been used to monitor qualitatively the reaction between ACM and PPO terminal group. Figure 1 shows the FTIR spectra of neat ACM, PPO and LCPA respectively. Figure 1(a) shows that the epoxy group of ACM gives the characteristic absorption band at 908 cm−1. The PPO shows two characteristic bands of the ether at 1023 and 1184 cm−1, corresponding to the C-O Stretch as shown in Figure 1(b). The ring stretching of the PPO gives a band at 1632 cm−1. Figure 1(c) shows the characteristic absorption bands of the pure LCP at 1733 cm−1, corresponding to the carbonyl (νc = o) stretching frequency. The weak epoxy characteristic peak (908 cm−1) disappeared after melt blending of binary blends (PPO/LCPA). This result indicates that epoxy groups can react with the –OH and -COOH terminal groups of LCPA and result in the formation of LCPA-co-ACM Copolymer.
27
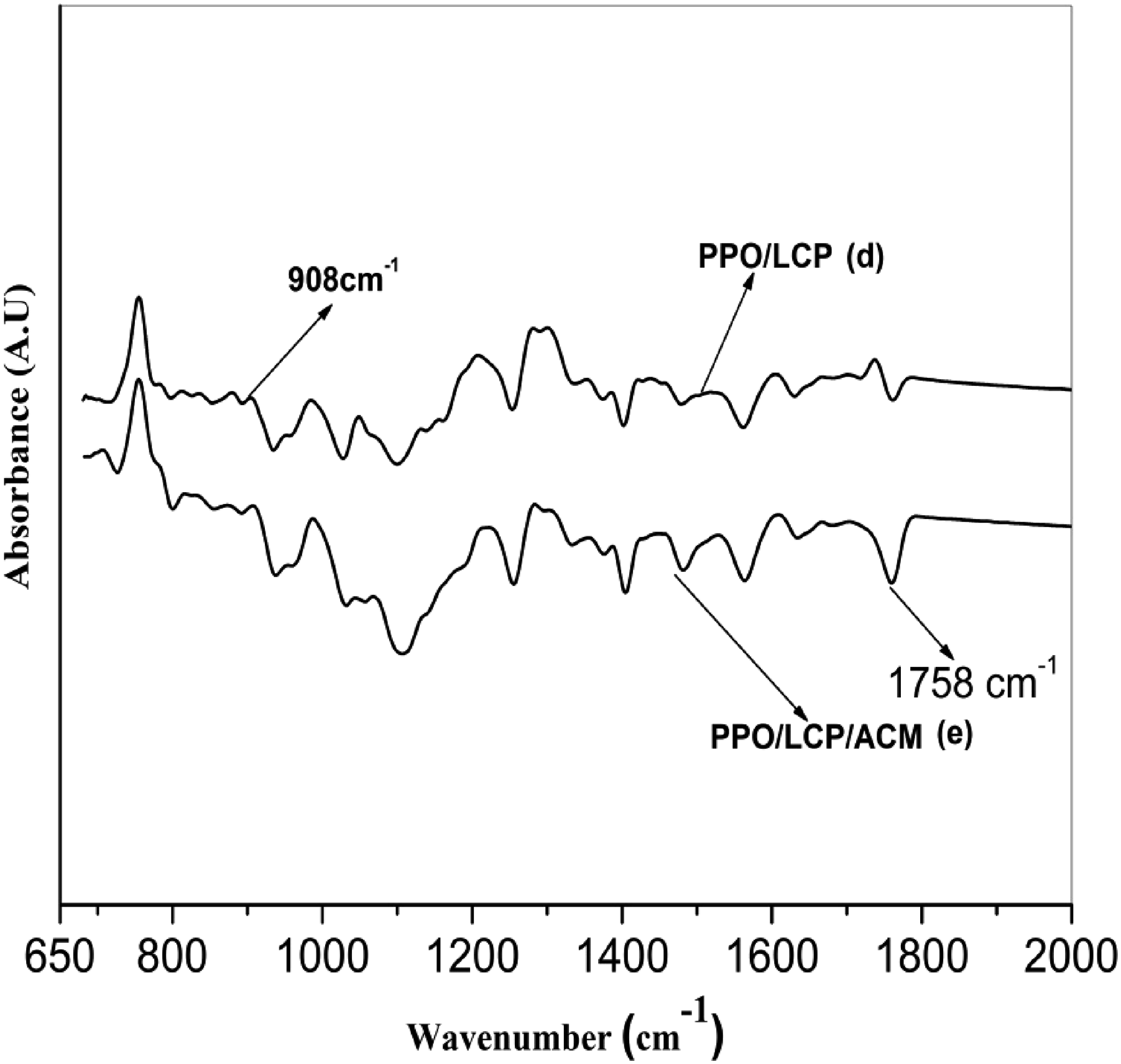
The acrylate rubber used in this study contains epoxy groups as the reactive curing site and hence, it can react with PPO and LCPA to produce the graft-block copolymer. The in-situ reaction among PPO, ACM and LCPA during melt mixing may form the PPO-co-ACM-co-LCPA copolymers. The FTIR spectrum of these copolymers (PPO-co-ACM-co-LCPA), is shown in Figure 2. Comparing the Figure 1 with the spectra of Figure 2, it was clear from the figure that the epoxy peak intensity at 908 cm−1 decreases and the carbonyl stretching frequency of LCPA (1733 cm−1) has been shifted to the higher frequency side. This suggests the reduction of the concentration of epoxy groups of ACM as the reaction proceeds. Based on the above information, the formation of the desirable PPO-co-ACM-co-LCPA copolymers during melt blending has been identified. FTIR spectra of neat components, (a) ACM, (b) PPO and (c) LCPA. FTIR spectra of the mixtures, (d) melt-blended PPO/LCPA mixture and (e) melt-blended PPO/ACM/LCPA mixture.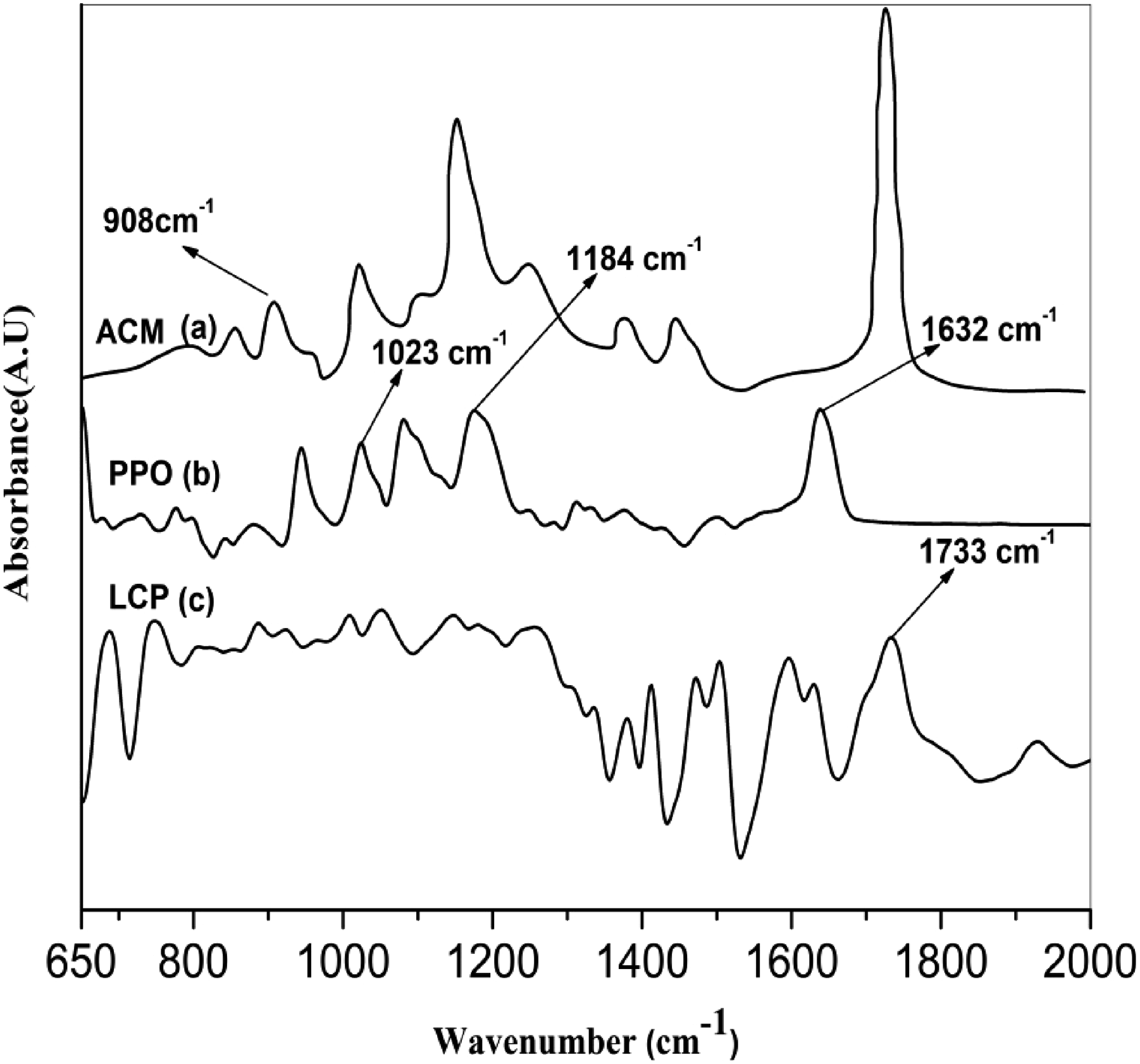
Thermal Properties
The DSC experiments were performed to determine the Tg of the blends. These measurements can be used to distinguish single-phase systems from phase-separated systems, as discussed by Utracki.
28
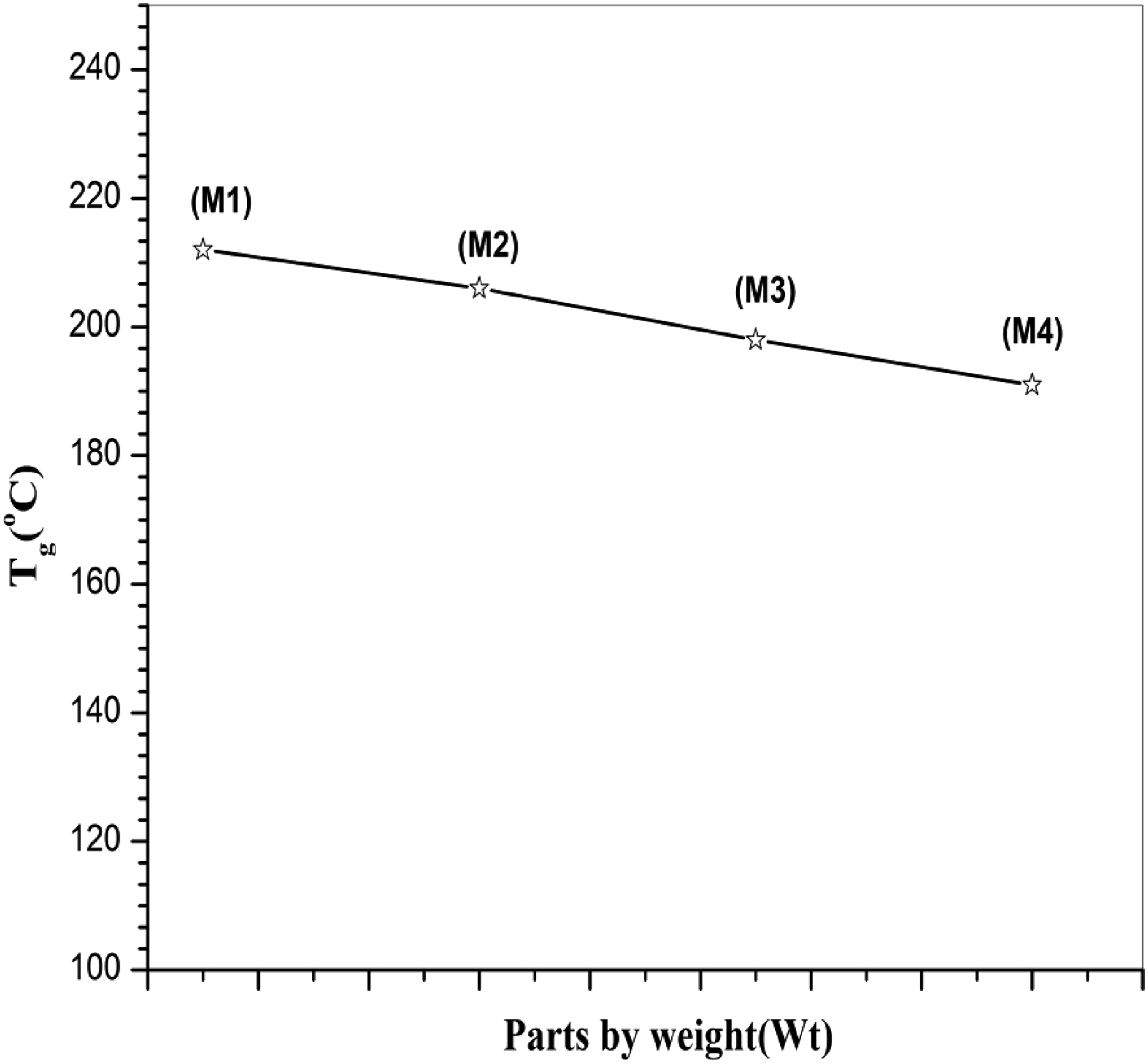
Several factors have been shown to affect the Tg of polymers and polymer blends, including chain structure and conformation,29,30 crystallinity, and dispersion. The Tg changes of the PPO phase in the PPO/LCPA blends as a function of the ACM content are summarized in Figures 3 and 4. In a PPO/LCPA (75/25) blend, a glass transition corresponding to the PPO phase located at about 206°C, which was near the glass transition of the neat PPO (212°C). The inability to resolve the Tg of LCPA in this blend, and even in neat LCPA, may be due to the rigidity and complicated structure of this LCPA. It was accepted that PPO and LCPA were completely immiscible. But with the addition of the compatibilizer the Tg was shifted to lower temperature side. It was observed that a five part by weight ACM in a PPO/LCPA (75/25) blend caused the Tg shift to be 8°C lower in PPO than the same blend of PPO/LCPA (75/25). The Tg shift of PPO/ACM/LCPA blends increased with addition of increasing amounts of ACM. When the ACM amount reached 10 parts by weight, the upper-temperature Tg of the blend was about 191°C, 15°C lower than that of PPO/LCPA (75/25). The stepwise shifts of Tg with increasing amounts of the compatibilizer, revealed by DSC and DMA measurements, confirm that the compatibilization in LCPA/PPO blends was accomplished by the addition of ACM. DSC thermograms of neat, binary and ternary polymer blends. The glass transition temperatures of the neat polymer and blends, determined by DSC.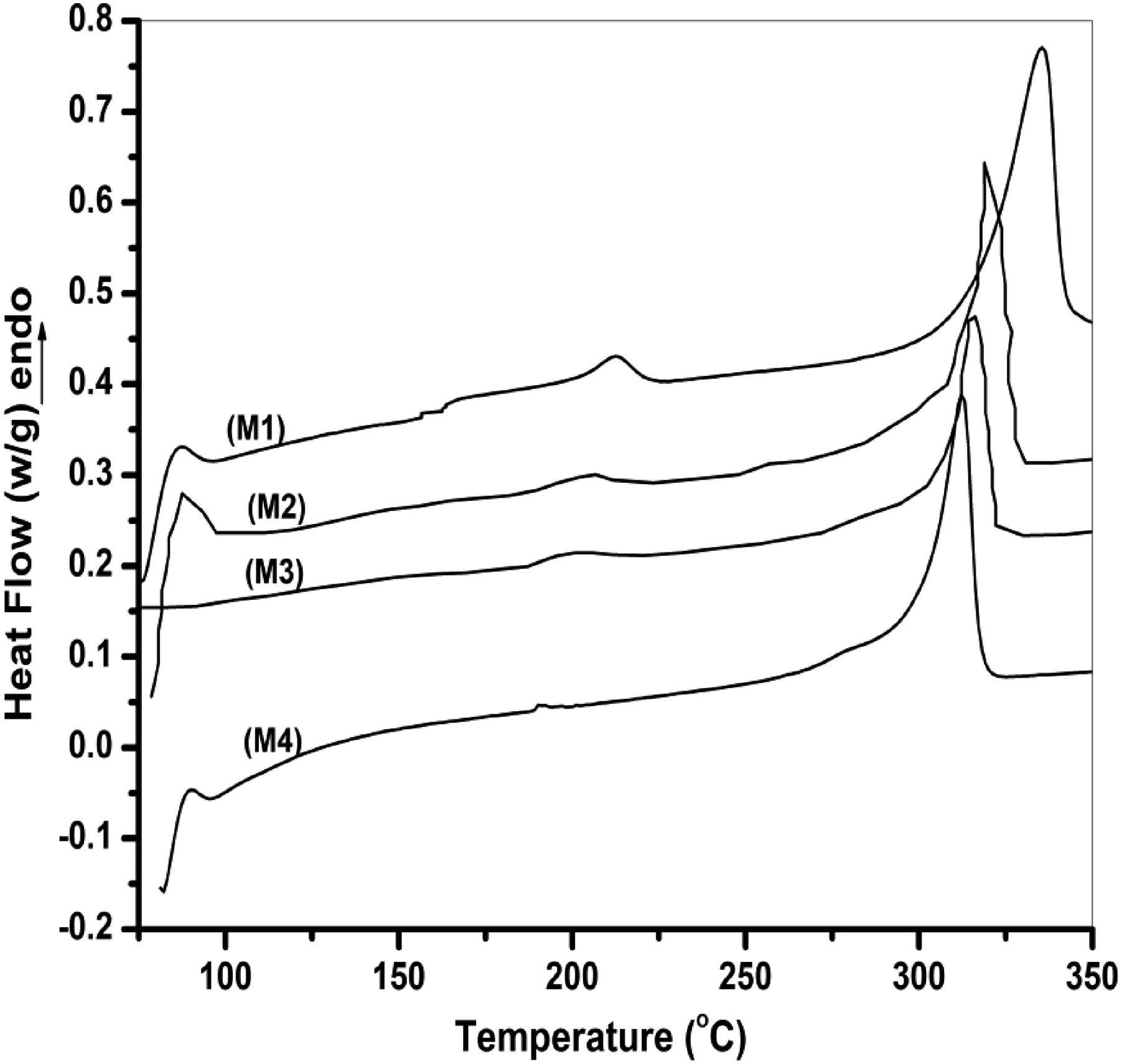
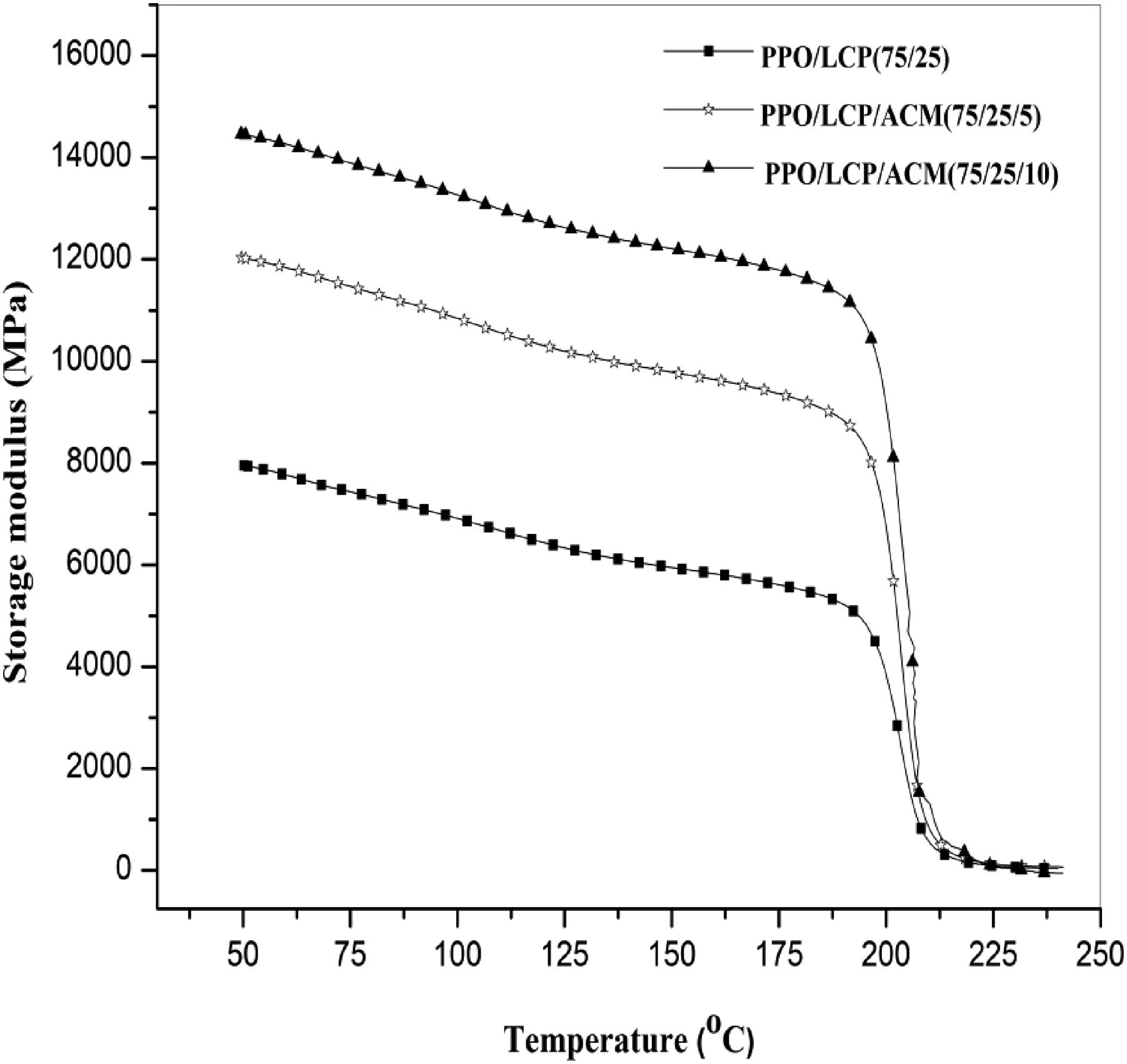
Phase behavior of polymer blends has been extensively analyzed using DMA methods. In Figure 5 Tanδ are plotted as a function of temperature (in the range of 50–250°C) for PPO/LCPA (75/25) blends without and with 5 and 10 (parts by weight) ACM. It can be found that, in Tanδ curves, all the materials analyzed show the presence of two transitions: the lower one associated with glass transition temperature of LCPA, and the higher one with that of PPO. As we can see, the Tg values obtained by DMA were slightly higher than those obtained by DSC. In comparison to a binary blend of PPO and LCPA, the Tg of a compatibilized blend shifted toward each other. The reduction caused by the addition of compatibilizer could be interpreted as progressive immobilization of polymer chains near the boundary between two phases. This was evidence that the compatibility of PPO and LCPA was partially improved. In Figure 6, storage modulus (E′) is shown as a function of temperature for blends with compatibilizers and for blends without compatibilizers. It was found that E′ was greater in the blends with ACM as compatibilizer over the temperature range than in the other blend. The reason for this could be attributed to the stronger interfacial interaction between matrix phase and dispersed phase (see Figure 6), which led to a more efficient stress transfer across interfaces. Normalized tanδ versus temperature for (■) PPO/LCPA blend and (□) PPO/LCPA/ACM blend. Storage modulus as a function of temperature at 1 HZ of (■) PPO/LCPA blend, (□) PPO/LCPA/ACM blend.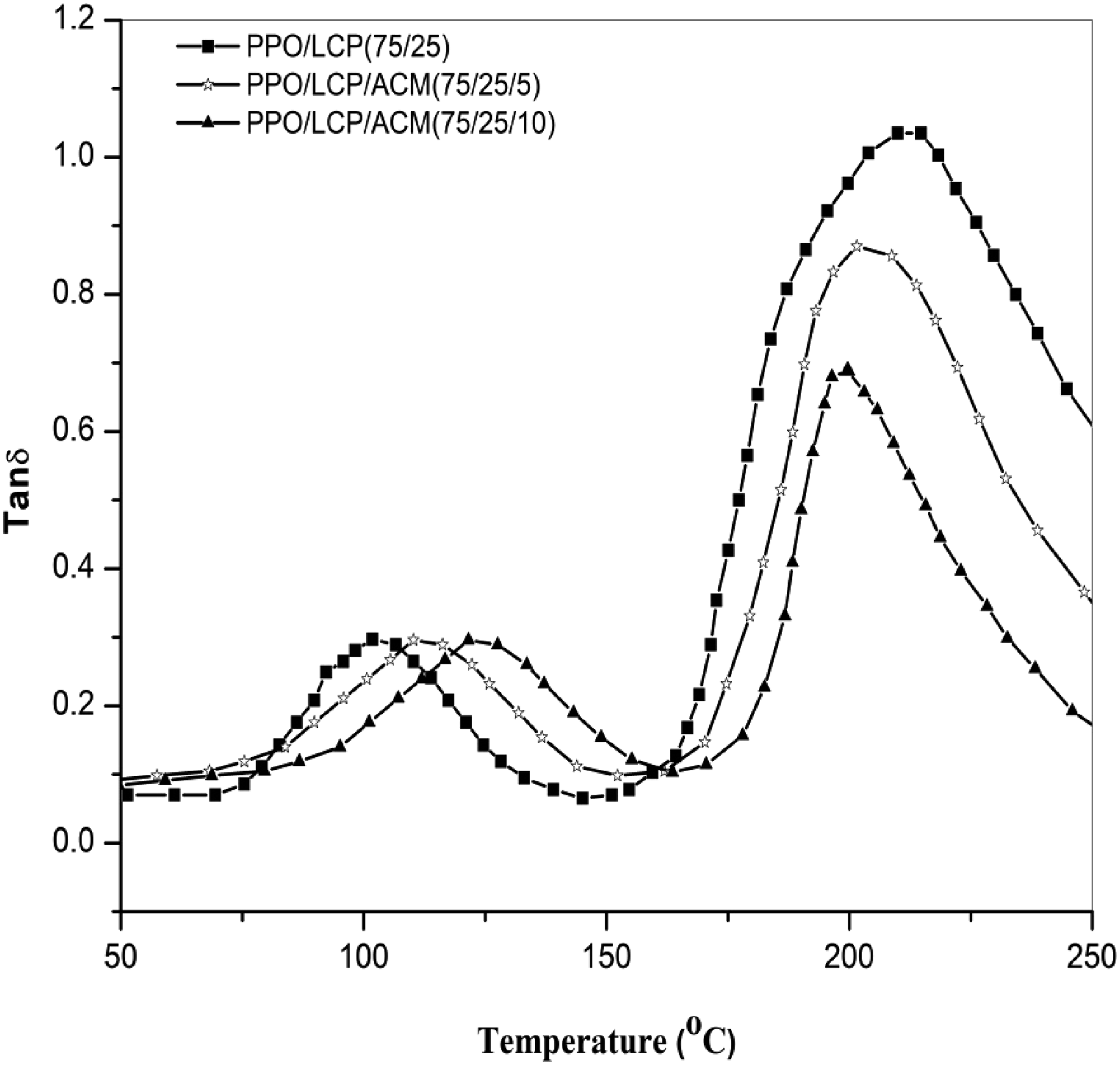
Heat deflection temperature
A comparison of the effects of elastomer content on the heat-deflection temperature (HDT) of compatibilized blends is presented in Figure 7. It has been observed that HDT of PPO increases with the addition of LCPA, while HDT of PPO/LCPA blends increases by approximately 6 – 10°C with the addition of ACM reactive compatibilizer. A PPO matrix reinforced with oriented LCPA fibers increases the material strength as well as the HDT of the composite, similar to composites made of glass or carbon fiber/polymer. The most common thermoplastic/LCP blends were immiscible with poor interfacial adhesion, and the presence of a compatibilizer was expected to increase interfacial adhesion, resulting in a higher HDT for the blends. This also indicates high structural strength capacity when simultaneously subjected to mechanical stress and temperature. Effect of compatibilizer on heat deflection temperature of PPO/LCPA blends.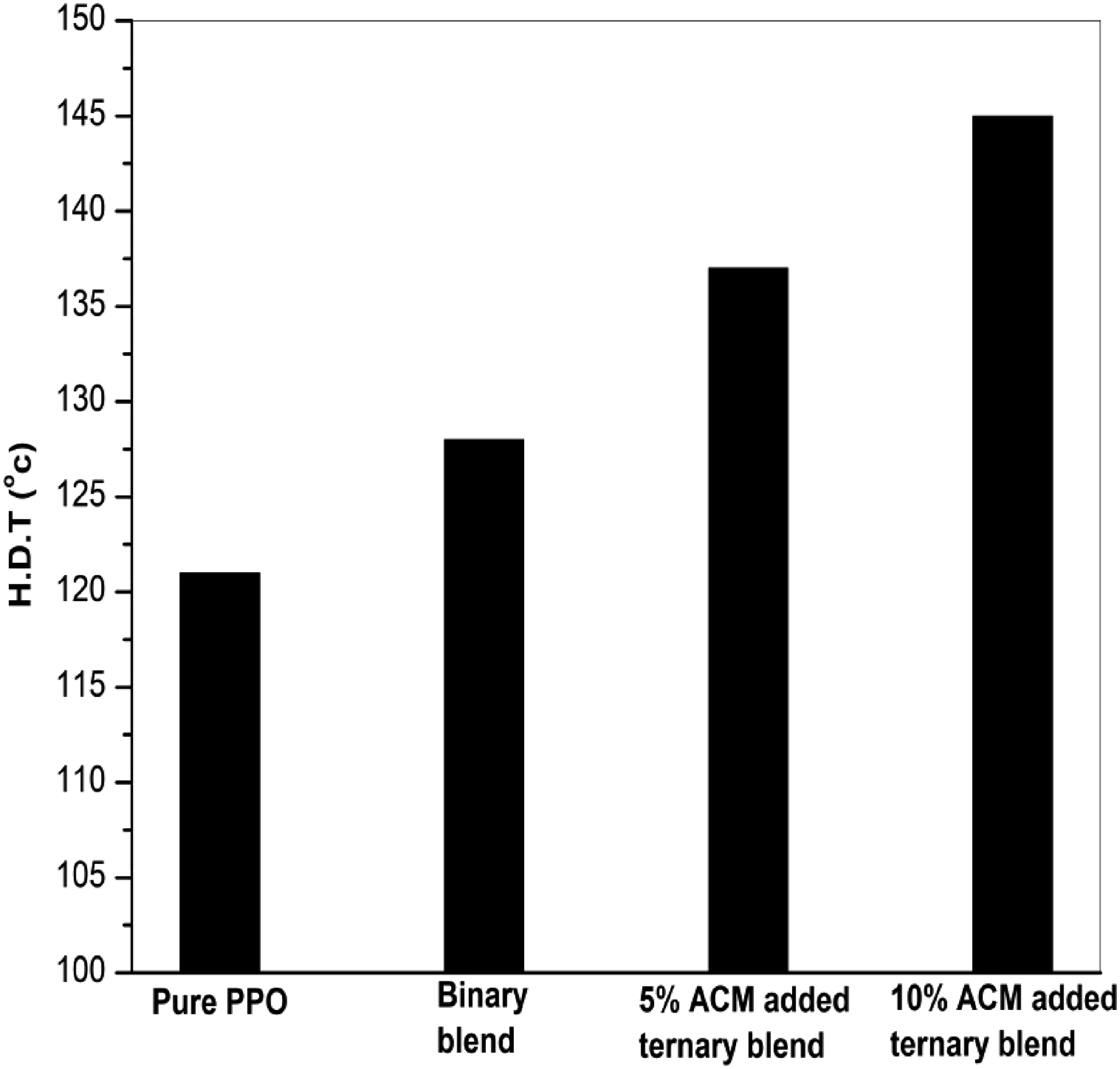
Rheological properties
One of the advanced engineering plastics, PPO has the drawback of having a high melt viscosity and poor processability. As a result of its processing, higher melt temperatures, higher molding pressures, and higher molding temperatures were required. In a sense, the final performances of PPO materials depend upon the processability of their formulations. The presence of LCPA facilitates the flow of PPO molecules and decreases the melt viscosity.
An understanding of the chemical reactivity and extent of reactions in a compatibilized blend has been gained by using torque measurements.
31
Figure 8 shows a torque experiment for the neat PPO, PPO/LCPA blends with and without acrylate rubber (ACM). Pure PPO exhibits about three times higher torque value than the pure LCPA under the same test conditions. The torque of PPO and its blends with LCPA containing 5 (parts by weight) ACM).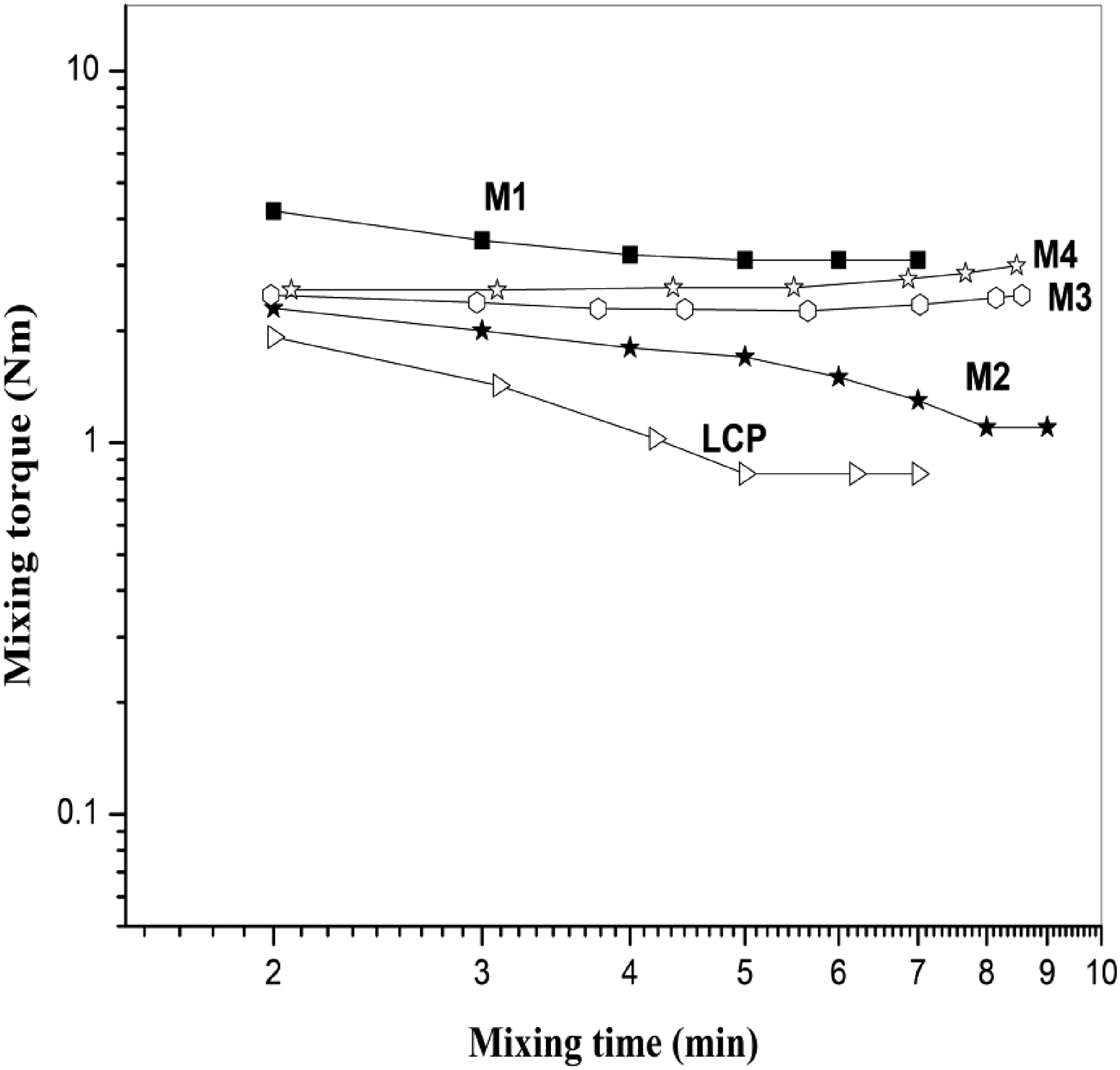
In both PPO and LCPA, torque values decrease gradually with mixing time, probably due to slight hydrolytic degradation caused by air moisture contact. Apparently, the melt torque of the PPO was reduced by adding 25 (parts by weight) LCPA. Due to a higher interfacial tension and a lower interfacial friction, two incompatible polymers can easily “slide” between phases during shear stress, resulting in a lower viscosity. The steady torques of the PPO/LCPA blends, however, seem to increase with the addition of ACM, indicating that ACM might react with the phenolic-OH end groups of PPO, LCPA and ACM, though at a slower rate. It would be expected that PPO molecules possess only one terminal OH, thus there would be less chain extension reaction, and viscosity increase would less than for PPO. Hence, the compatibilized PPO/LCPA (75/25 w/w) mixture exhibits a higher viscosity relative to the uncompatibilized one.
MFR of the PPO/LCPA blends.

Figure 9 shows the shear viscosity versus shear rate plots of pure PPO, LCPA, and uncompatibilized and compatibilized blends of PPO/LCPA at 290°C. As a result of the same test conditions, it became clear that PPO has a significantly higher viscosity than LCPA. It has generally been believed that LCPA components should have a lower viscosity than host matrix components to facilitate the formation of LCPA fibrils in blends. The uncompatibilized blend has the lowest viscosity, as would be expected. The compatibilized blends result in substantial viscosity rise, especially pronounced at lower shear rates. Based on the data discussed previously, the molecular weight increase resulting from chain extensions and coupling reactions was believed to be the major contributor to the viscosity increase observed in these compatibilized blends. The variation of shear viscosity with shear rates for base polymer and blends.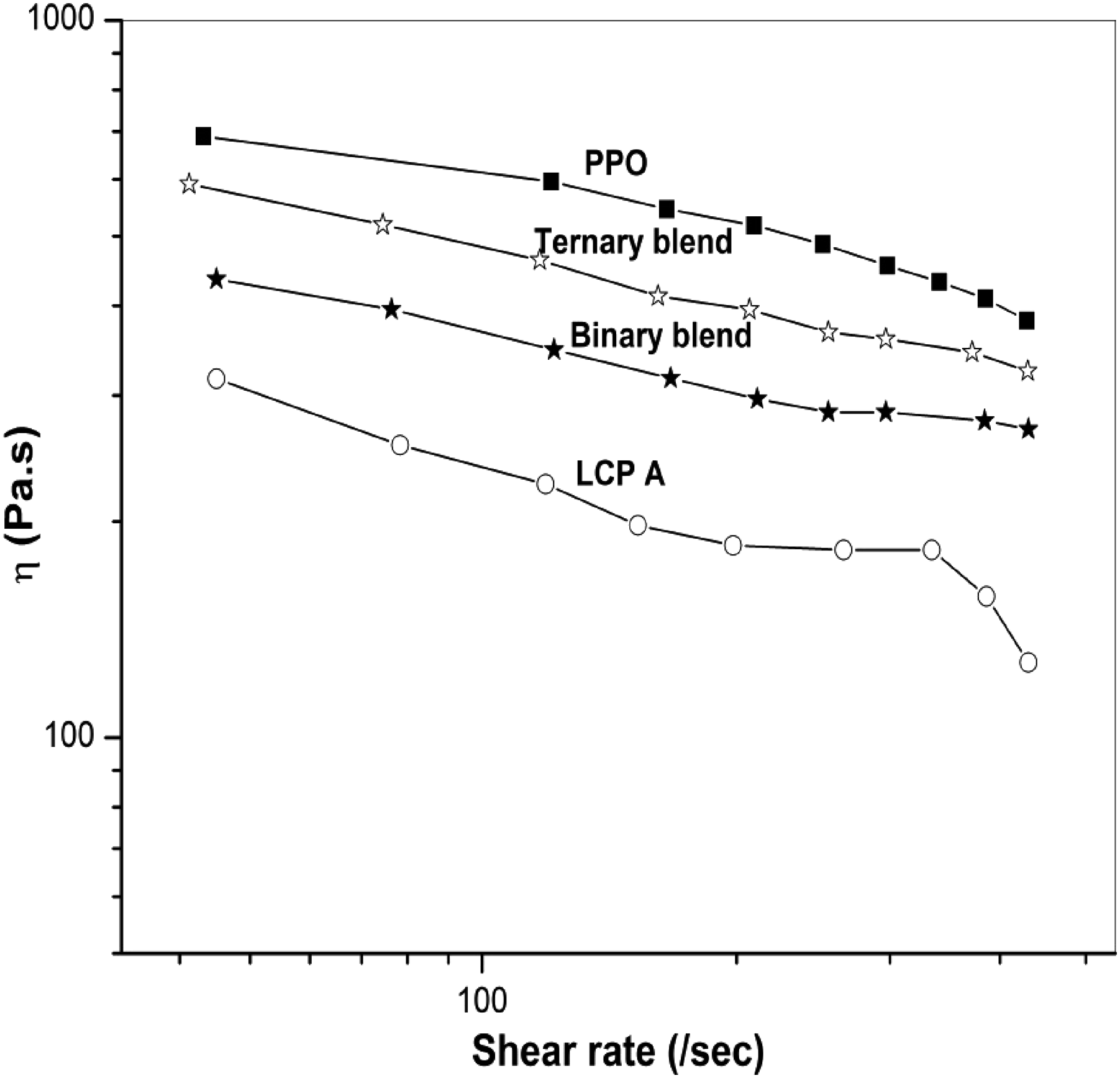
Morphological studies
The present study used phase contrast microscopy to observe the size and shape of dispersed LCPA phases in polymer blends. LCPA cannot be dissolved in most organic solvents, and the PPO has a good solubility in polar organic solvent, such as N, N-dimethyl formamide (DMF). Melt blending tends to cause the orientation and fibrillation of the LCPA phase, and the deformation of LCPA phase could affect the judgment about blend miscibility. Consequently, samples were prepared through the use of an internal mixer for optical microscopy observations. It is possible to observe the insoluble LCPA particles directly under optical microscopy after the extraction of PPO using DMF in PPO/LCPA blends. The dispersion of LCPA component of PPO/LCPA (75/25) without and with ACM (5 Parts by weight) is shown in Figure 10 Phase contrast optical micrographs of (a) PPO/LCPA (75/25) (b) PPO/LCPA/acrylate rubber (75/25/5).
Binarization of micrographs favors the quantitative size measurement. After counting LCPA particles and calculating the particle size of LCPA from equation (1), the curves for number frequency-size distribution of PPO/LCPA blends with various content of ACM (0,5,10 parts by weight) are obtained and shown in Figure 11 Distribution of diameter of LCPA particles in PPO/LCPA (75/25) blends without and with acrylic rubber.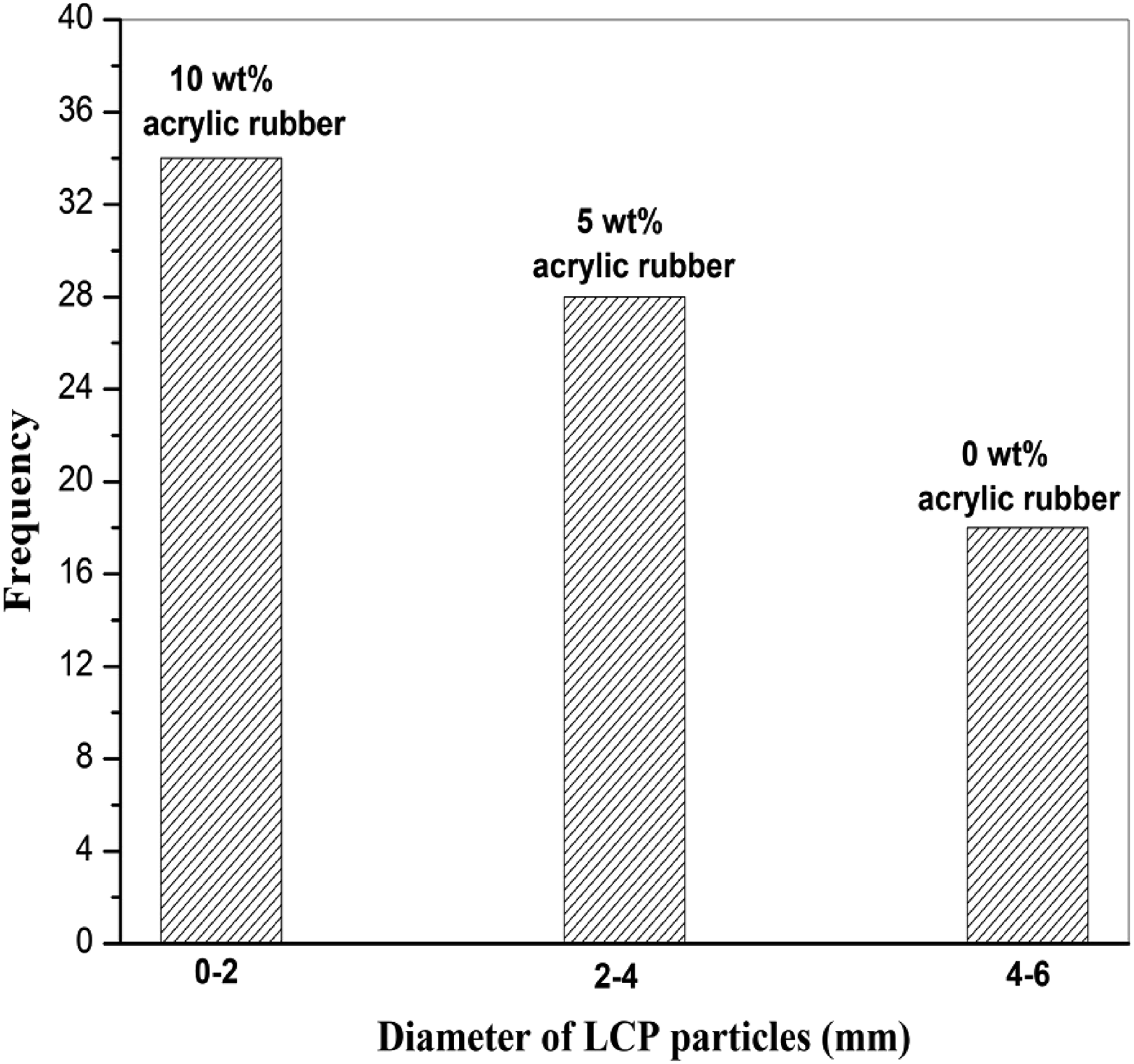
SEM micrographs of fractured blend surfaces are shown in Figure 12. The phase separation in a binary blend of PPO and LCPA (75/25) can be directly observed in Figure 12(a). This sample has a distinct two-phase morphology, i.e., a continuous PPO phase with a dispersed LCPA phase. The LCPA particles in the form of spheres have diameters in the range of 2 to 10 μm. All the spheres and holes have smooth surfaces, indicating poor interfacial adhesion between the LCPA and PPO phases. However, with the addition of the compatibilizer, the morphologies of fractured surfaces change dramatically. With five parts by weight ACM in the LCPA/PPO (25/75) blend, the domain size of the minor LCPA phase decreased significantly [Figure 12(b)]. The diameter of the largest particles was smaller than 1 μm. With 10 parts by weight ACM in the blend, the LCPA phase was well dispersed in the continuous PPO phase. The LCPA particles had uniform diameters below 0.5 μm [Figure 12(c)]. In addition to this, the fractured surface became indistinct. From these phenomena, it was evident that the compatibilization of the blends was achieved and the interfacial adhesion between two phases was improved.32,33 It is clear from these results that specific interactions between the phases can act as physical crosslinks along the interface and disperse polymer blends to a much finer degree. Morphologies of fractured surface of blends (a) LCPA/PPO (25/75); (b) LCPA/PPO/acrylate rubber (25/75/5) and (c) LCPA/PPO/acrylate rubber (25/75/10).
Impact and tensile properties
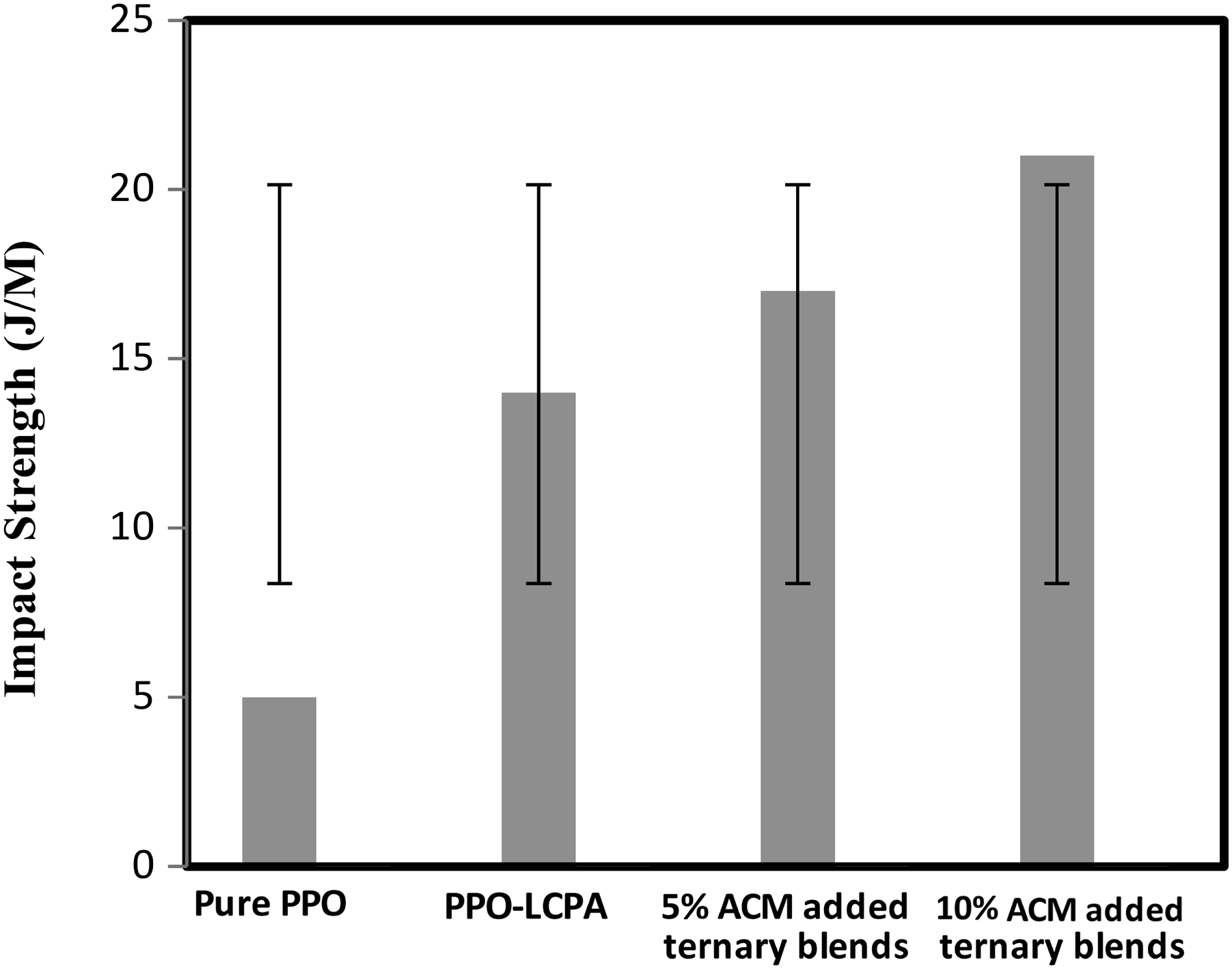
The notched impact and tensile strength, as a function of elastomer content for PPO/ACM/LCPA blends is shown in Figures 13 and 14. PPO/LCPA ratio was 75/25. When an immiscible blends is formed, the matrix component carries the majority of the impact force. There is a high degree of phase separation within the system, so energy cannot easily pass from one component to another. In addition, the compatibilizer on the interface allows better stress transfer across the phase boundary, allowing the impact energy to be dispersed on both surfaces and in the bulk. A compatibilized blend diffused the same amount of energy more efficiently than a uncompatibilized blend when both received exactly the same amount of energy in an impact event. The improvements of the compatibilized blends over the uncompatibilized counterparts in tensile and Izod impact properties were substantial and consistent. It has been reported that the degree of local segment mutual diffusion across the interface between the blend constituents profoundly affects the mechanical strength of the adhesive bond.
34
A better interfacial adhesion may contribute to the improved properties consistently observed for compatibilized blends over their uncompatibilized counterparts in this study. The LCPA fiber structural difference between the uncompatibilized and compatibilized blends was also believed to be responsible for the observed difference. The LCPA fibers of the compatibilized blends were relatively finer (greater orientation) and tend to be in the form of lamellae. The stiffness and strength of the formed LCPA fibers increase with the increase of LCPA orientation. Therefore, the LCPA fibers in the compatibilized blends have relatively higher strength and greater interfacial contact was a (higher aspect ratio) with the PPO matrix that the corresponding uncompatibilized blends. Tensile strength of pure PPO, uncompatibilized and compatibilized PPO/LCPA blends with standard deviation. Impact strength of pure PPO, uncompatibilized and compatibilized PPO/LCPA blends with standard deviation.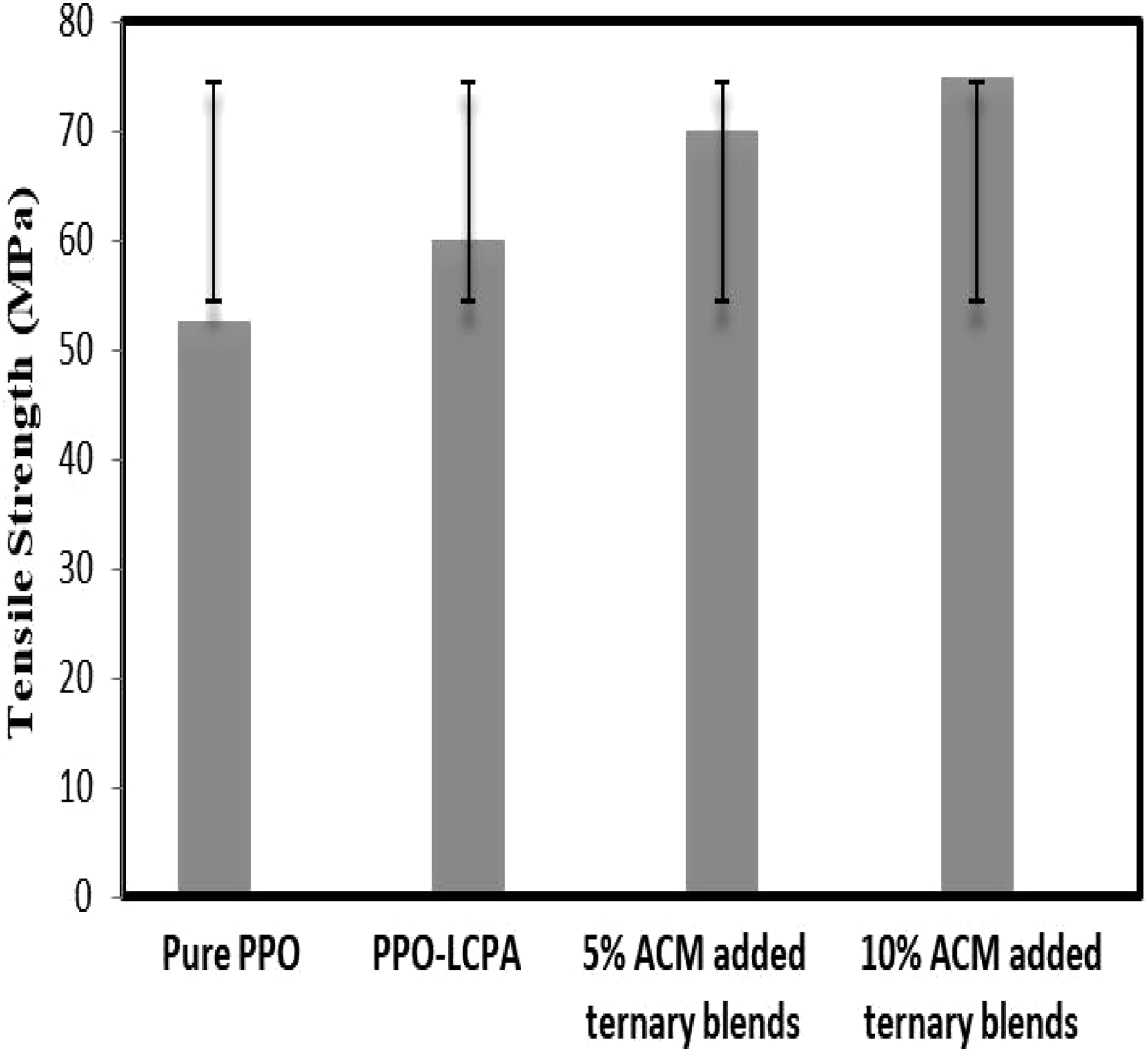
Surface roughness via atomic force microscopy
Atomic Force Microscopy (AFM) analysis was utilized to study the topography of the in-situ composite samples produced with and without the compatibilizer. Figures 15(a) and 15(b) illustrate the two-dimensional and three-dimensional AFM images of binary and ternary composites, respectively. However, the AFM topographies of composites and data on surface roughness indicate that the orientation of the LCPA during sample preparation might influence surface morphology more strongly. As compared to PPO/LCPA composites, the compatibilizer greatly increased the surface roughness of the composites. The surface roughness of composites made from PPO/LCPA and PPO/ACM/LCPA was evaluated with AFM in tapping mode, and the RMS of PPO/LCPA (75/25) and PPO/ACM/LCPA were 7.56 nm and 1.92 nm. A study by Wu et al.
35
demonstrated that an index of interface adhesion strength can be determined by the roughness of the interface. As a result, the roughness data of our samples strongly indicate that PPO/LCPA interface adhesion has been significantly enhanced in the presence of compatibilizer when compared to PPO/LCPA composite. This was due to fact that, during the addition of ACM to the binary PPO/LCPA blend, reinforcement was achieved by the miscibility of ACM with PPO and by the interactions between ACM and LCP-A. AFM images; (a) two dimensional AFM image of PPO/LCPA and (b) PPO/LCPA/ACM composites. (c) Three dimensional AFM image of PPO/LCPA and (d) PPO/LCPA/ACM composites.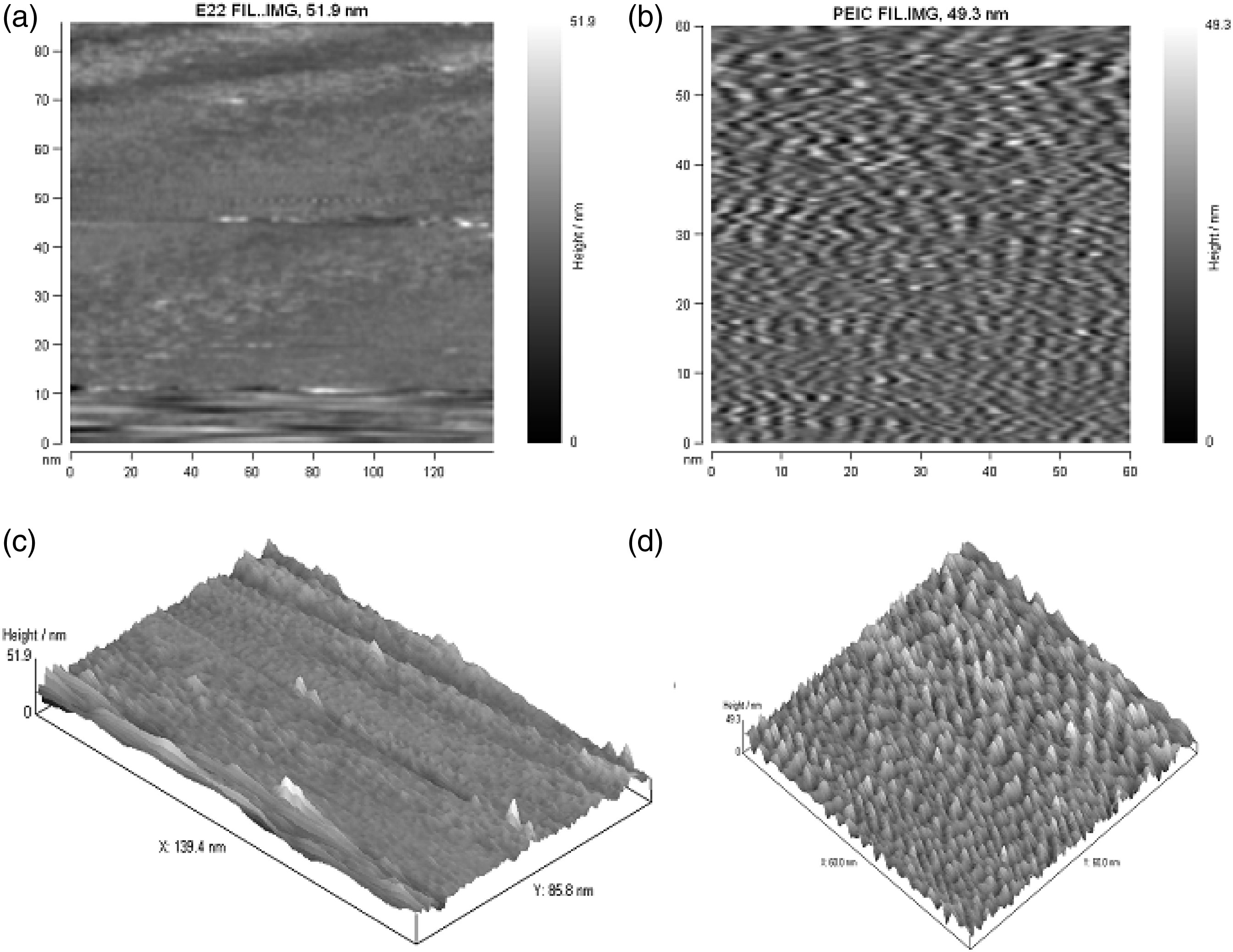
Conclusions
PPO and LCP were successfully blended by using epoxy containing acrylic rubber (ACM) as a reactive compatibilizer in the twin screw melt extrusion process. It is possible for ACM to form an ACM-g-LCP copolymer at the interface when its epoxy functional group reacts with the -COOH and/or -OH end groups of the LCP copolyester. It is likely that the in situ formed ACM-g-LCP copolymer will reside near the interface between PPO and LCP, and this will increase interfacial adhesion, which will produce finer dispersed LCP phases. The enhanced interfacial adhesion improved the ultimate tensile strength and impact strength of these ternary blends compared to their binary counterparts. The miscibility of polymer blends has been studied using FTIR spectroscopy. According to FTIR results, the ACM had been grafted onto PPO and the LCP and epoxy ring opening reactions had indeed taken place. An interfacial chemical interaction between ACM and LCP can also be confirmed by PCOM observation. The improvement of compatibility can therefore be attributed both to special interactions and to interfacial chemical reactions. The DMA data show that PPO and LCP damping peaks are broadened, causing inward migration, which is probably caused by the presence of an ACM at the interface. A SEM micrograph of fractured surface reveals that the addition of the compatibilizer increases adhesion between the matrix and the dispersed phase in the PPO/LCP blends. The domain size of the compatibilized PPO/LCP blend was drastically reduced by increasing epoxy content. During the torque test, it was found that compatibilized blends produced higher torque values than uncompatibilized blends.
Footnotes
Acknowledgements
The authors would like to acknowledge the Researchers Supporting Project number (RSP2023R373), King Saud University, Riyadh, Saudi Arabia.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The auhor(s) disclosed receipt of the following support for only the research work and not authorship and or publication of this article. Researchers supporting project number (RSP2023R373), King Saud University, Riyadh, Saudi Arabia.
