Abstract
This study investigates the impact of Titanium Suboxide (Ti4O7) on the flame retardancy, mechanical properties, thermal stability, and electrical and thermal conductivity of silicone rubber composites. The addition of 1.0 part of Ti4O7 to the silicone rubber composites results in a total heat release (THR) of 45 MJ/m2, which is 45.7% lower than the silicone rubber without Ti4O7, leading to a decrease in the peak of heat release rate (PHRR). Furthermore, Ti4O7 exhibits temperature stability at low temperatures and acts as a physical barrier, inhibiting the early decomposition of silicone rubber and enhancing the thermal stability of the composites. However, at temperatures exceeding 450°C, the catalytic activity of Ti4O7 is activated, promoting the decomposition of the silicone rubber composites and generating more residue. The addition of an appropriate amount of Ti4O7 can further improve the mechanical properties of the silicone rubber composites and enhance their electrical and thermal conductivity.
Introduction
Silicone rubber (SR) is a versatile synthetic elastomer that consists of silicon, oxygen, and organic side groups. It has become a valuable material for a variety of industries due to its unique combination of physical and chemical properties. 1 While its excellent temperature resistance, flexibility, and chemical stability make it a popular choice for coatings and adhesives applications, attention must be paid to its poor flame retardancy and potential release of toxic fumes. 2 To ensure the safety and durability of silicone rubber in practical applications, the incorporation of flame retardants is paramount. Zhang et al. 2 introduce ammonium polyphosphate (APP), aluminium diethylphosphonate (ADP), and octaphenylpolyhedral zwitterionic sesquioxane (OPS) into room temperature vulcanized silicone rubber insulation composites. The results show that the addition of the composite flame retardants significantly improves the ablative and flame-retardant properties of the Room Temperature Vulcanizing (RTV) silicone rubber composites.
In conjunction with its suboptimal flame retardancy, the comparatively modest electrical conductivity exhibited by silicone rubber imposes constraints on its viability across several applications necessitating favorable electrical attributes. Consequently, enhancing the electrical conductivity of silicone rubber has emerged as a noteworthy subject commanding attention within the realm of scientific inquiry.3,4 One of the main benefits of improving the conductivity of silicone rubber is that it can reduce the contact thermal resistance between the heat source surface and the silicone rubber contact surface. For example, conductive silicone rubber can be used as an insulator and electromagnetic interference (EMI) shield in electronic devices.5,6 In addition, conductive silicone rubber can be used in heating elements, sensors, and other applications where electrical properties are important. 7 Another benefit of increasing the conductivity of silicone rubber is its ability to dissipate static electricity. Static electricity can cause damage to sensitive electronic components, especially in manufacturing environments. 8 Wang et al. 6 choose carbon nanotubes (CNTs) and conductive sponges (CS) to construct a 1D-3D synergistic network in a silicone matrix. They prepare new silicone rubber composites with better thermal and electrical conductivity properties. Such silicone resin-based composites with high thermal and electrical conductivity have a wide range of applications in the thermal management of emerging electronic devices.
Titanium suboxide (Ti4O7), recognized as titanium suboxide, constitutes a ceramic compound characterized by the coexistence of titanium oxide (TiO) and titanium dioxide (TiO2) phases. Distinguished by its singular attributes encompassing catalytic prowess, elevated thermal stability, and a composite electrical conductivity profile, this material assumes a position of intrigue for a diverse array of application domains. 9 Ti4O7 exhibits high catalytic activity and selectivity for the oxidation of carbon monoxide (CO) and volatile organic compounds (VOCs), making it a promising material for use as a catalyst in the automotive industry for the removal of CO from exhaust gases. Sun et al. 10 synthesized micron-sized Ti4O7 powder using thermal reduction under H-2 atmosphere and successfully used it as a carrier material for Pt-based catalysts. Ti4O7 also exhibits mixed conductivity, making it useful in applications such as solid oxide fuel cells and oxygen sensors. Ti4O7 has been shown to have high ionic conductivity at high temperatures, which is important for efficient operation in these devices. Additionally, Ti4O7 has interesting electron transport properties that may have potential applications in electronics and optoelectronics. Tan et al. 11 proposed the preparation of Ti4O7-doped polypyrrole coatings on metal bipolar plates, resulting in conductive Ti4O7 particles that enhance the electrical conductivity and hydrophobicity of the composite coating while maintaining high chemical stability in simulated cathode proton exchange membrane fuel cell environments at 25°C and 70°C. In conclusion, Ti4O7 has unique properties that make it useful in a variety of applications, such as catalysis, high temperature environments, solid oxide fuel cells, and oxygen sensors. Further research may reveal even more potential applications for Ti4O7 due to its unique properties.12,13
Upon analyzing the properties of Ti4O7, we conclude that this ceramic material holds great potential in enhancing the properties of silicone rubber, making it a topic of interest. A key advantage of incorporating Ti4O7 into silicone rubber is its catalytic carbonization effect, which makes it a useful flame retardant. Silicone rubber is known to ignite and burn when exposed to heat, making it hazardous in certain applications. 14 Through the incorporation of Ti4O7 into silicone rubber, the material gains an elevated level of flame retardancy. This augmentation can be attributed to the catalytic attributes inherent to Ti4O7, which manifest when subjected to heat. This catalytic effect instigates the generation of a carbonaceous layer at the material's surface. Functioning as a safeguarding shield, this carbon layer effectively impedes the initiation of ignition and subsequent combustion processes. 15 Moreover, Ti4O7 has the potential to enhance the thermal and electrical conductivity of silicone rubber. It boasts a high thermal conductivity, which improves the heat dissipation properties of silicone rubber, making it ideal for thermal management applications such as electronics and high temperature environments. Additionally, Ti4O7 has hybrid conductivity, which enables it to make silicone rubber electrically conductive. This attribute is useful in applications that require electrical properties such as sensors or electromagnetic interference (EMI) shielding.16,17 Moreover, the efficacy of Ti4O7 in augmenting the thermal stability of silicone rubber is widely acknowledged. Prolonged exposure to elevated temperatures often prompts silicone rubber to undergo a decline in its desirable attributes, resulting in material degradation. Through the integration of Ti4O7, the composite material attains heightened resilience in high-temperature conditions. This fortification enables its application within industries demanding robust performance under extreme thermal circumstances, such as the automotive and aerospace sectors. 18 To investigate the effects of Ti4O7 on various properties of SR composites, this paper focuses on mixing Ti4O7 into SR in varying proportions, using fumed silica and silicone oil to enhance the mechanical properties of SR. The key properties of SR composites including flame retardancy, thermal and electrical conductivity, mechanical properties, and thermal stability were tested, and the catalytic carbonization effect of Ti4O7 was revealed through residue analysis.
Material and characterization
Materials
Silicone rubber (SR, 110-2, Relative molecular weight: 45–85, Vinyl content: 0.13–0.18) was brought from Nanjing Dongjue Silicone Co., Ltd. (China). Titanium suboxide (Ti4O7, the average size is about 5 μm, Figure 1) was offered by Hubei Xinghengye Technology Co., Ltd. (China). 2,5-dimethyl-2,5-di(tert-butylperoxy) hexane (C16H34O4, Molecular weight: 290.44) was attained from Sinopharm Chemical Reagents Co., Ltd. (China). Fumed silica (R9200) was offered by Evonik. (Germany). Silicone oil (JP-203, HO[(CH3)4SiO]nH) was brought from Shenzhen Ji Peng Silicone Fluorine Material Co., Ltd. The TEM images of Ti4O7 at low and high magnification.
Preparation of SR and its composites
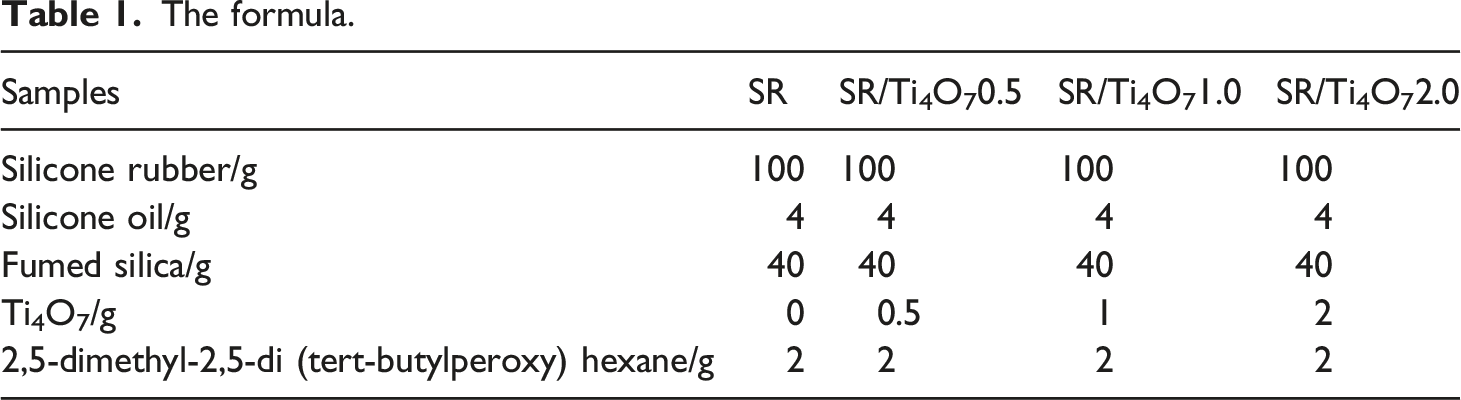
The formula.
Characterization
The morphologies of SR composites were investigated by Scanning electron microscopy (SEM, Phenom ProX, Phenom, China).
A Cone calorimeter test (CCT) was performed on an FTT cone calorimeter according to ISO 5660 at the heat flux of 50 kW/m2 (sample size: 100 mm × 100 mm × 3 mm).
The mechanical properties were obtained by using a universal testing machine (Rate: 250 mm/min. UTM4204, SUNS, China).
Thermal stability was performed using a Thermal Gravimetric Analyzer (TGA, Q50, TA, USA). About 10.0 mg of sample was put in an alumina crucible and heated from ambient temperature to 800°C and the heating rate was set as 10°C/min in nitrogen atmosphere or air.
The dielectric frequency domain spectrum test shall be conducted according to GB/T 1409-2006 Recommended Method for Measuring the Permittivity and Dielectric Loss Factor of Electrical Insulating Materials at Power Frequency, Audio Frequency and High Frequency. The test temperature is 25°C, and the frequency is 0.01–1000 Hz.
The sample of volume resistance is prepared into a round sheet with a diameter of 100 mm, and tested on a ZC-36 type impedance meter according to GB/T 1410-2006. The test temperature is 25°C and the test voltage is 1000 V.
The sample of dielectric strength is prepared into a round sheet with a suitable diameter and tested on an AC dielectric strength tester. The dielectric strength at breakdown is recorded and measured five times to obtain the average value.
The residues collected from cone calorimeter tests were evaluated by Raman spectra (RS, ATR3110-1064, OPTOSKY, China).
Results and discussion
Characterization of SR and its composites
SEM analysis (Figure 2) of the silicone rubber (SR) shows that in the absence of titanium black, only a few white particles are present in the cross-section of SR, indicating slight agglomeration of the silica. The majority of the silica combines with the silicone rubber, providing good reinforcement. Energy-dispersive X-ray spectroscopy (EDS) analysis of SR indicates that it mainly consists of silicon and oxygen, with a small amount of carbon, suggesting that silica is the primary filler in the silicone rubber without impurities mixed during the process. However, the fracture surface of SR/Ti4O7 presents a different phenomenon, with a large number of particles observed. EDS analysis has unequivocally identified the predominant composition of these particles to be Ti4O7. Concerning the SR, the majority of the silica content synergistically amalgamates with the polymer matrix, thereby engendering efficacious reinforcement. EDS analysis conducted on the SR reveals a predominant composition of silicon and oxygen, with a marginal occurrence of carbon, signifying that silica constitutes the principal filler in the silicone rubber, devoid of any impurities stemming from the fabrication process. However, a distinctive phenomenon unfolds upon scrutinizing the fracture surface of silicone rubber composites infused with Ti4O7 (SR/Ti4O7). This phenomenon is characterized by the conspicuous presence of numerous particles. EDS analysis corroborates that these particles are primarily comprised of Ti4O7. Nonetheless, a noteworthy observation arises: the majority of these particles display limited compatibility with the silicone rubber matrix. This lack of compatibility results in their segregation on the fracture surface, leading to an impaired affinity with the silicone rubber matrix.
19
The presence of large amounts of Ti4O7 promotes agglomeration, further diminishing the compatibility between Ti4O7 and silicone rubber. Therefore, a significant effect on the mechanical properties of the silicone rubber may result from filling content of Ti4O7 beyond a certain value. The SEM and EDS of SR and SR/Ti4O72.0 composite;(a) SR, (b) SR/Ti4O72.0.
Mechanical properties of SR and its composites
To evaluate the effect of Ti4O7 on the mechanical properties of silicone rubber, a series of tests are conducted and the results are presented in Figure 3. The initial tensile strength and elongation at break of the unfilled silicone rubber are measured to be 7.5 MPa and 317%, respectively. Upon the addition of 0.5 part of Ti4O7, both the tensile strength and elongation at break increase to 7.9 MPa and 374%, respectively, indicating that a small amount of Ti4O7 can have a positive complementary effect. However, as the amount of Ti4O7 increases, the elongation at break gradually decreases, possibly due to agglomeration of excessive Ti4O7, which impedes the transfer of internal forces within the silicone rubber.20,21 This agglomeration phenomenon can be observed in Figure 2(b). The agglomeration of Ti4O7 particles introduces the possibility of stress concentration when subjected to external stretching forces. This phenomenon can exacerbate crack formation and heighten the material's susceptibility to fracture. Interestingly, counter to this anticipated behavior, the inclusion of Ti4O7 paradoxically augments the tensile strength of the silicone rubber, albeit with a constrained improvement. This outcome can be ascribed to the entwining of a significant portion of Ti4O7 particles within the molecular chains of the silicone rubber.22,23 Consequently, Ti4O7 can act as physical cross-linking points, further reinforcing the tensile strength of the silicone rubber after cross-linking. The mechanical properties of SR and its composites.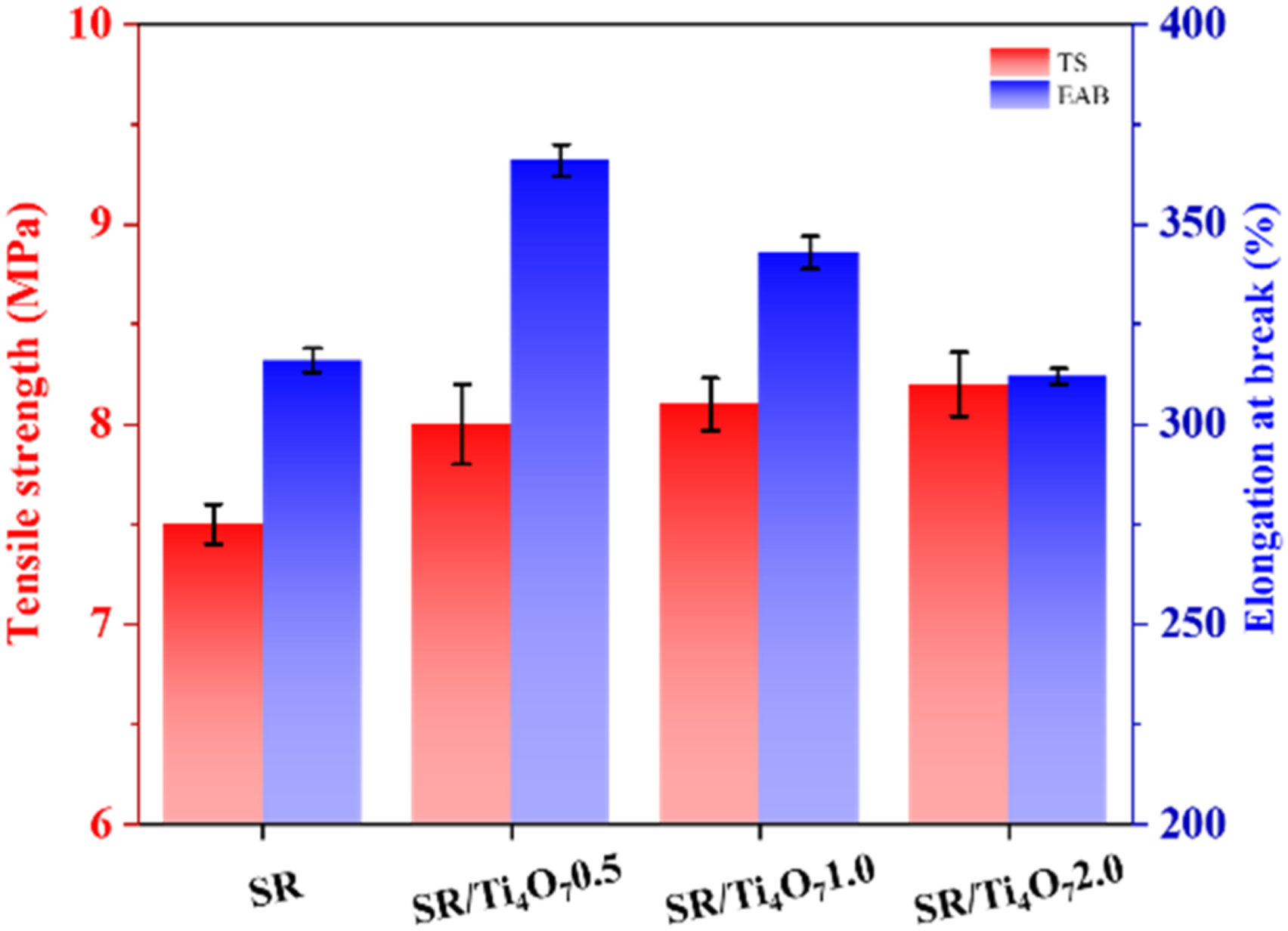
Thermal properties of SR and its composites
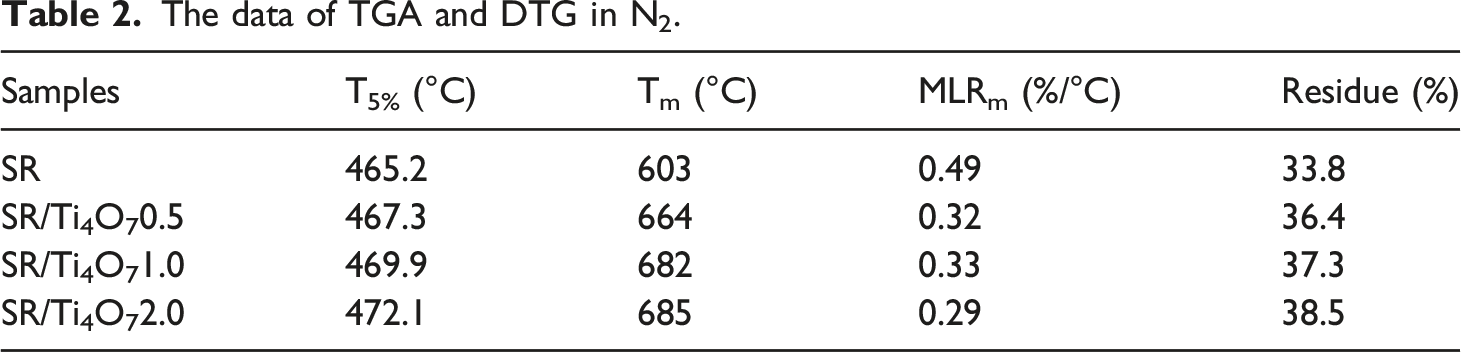
The data of TGA and DTG in N2.
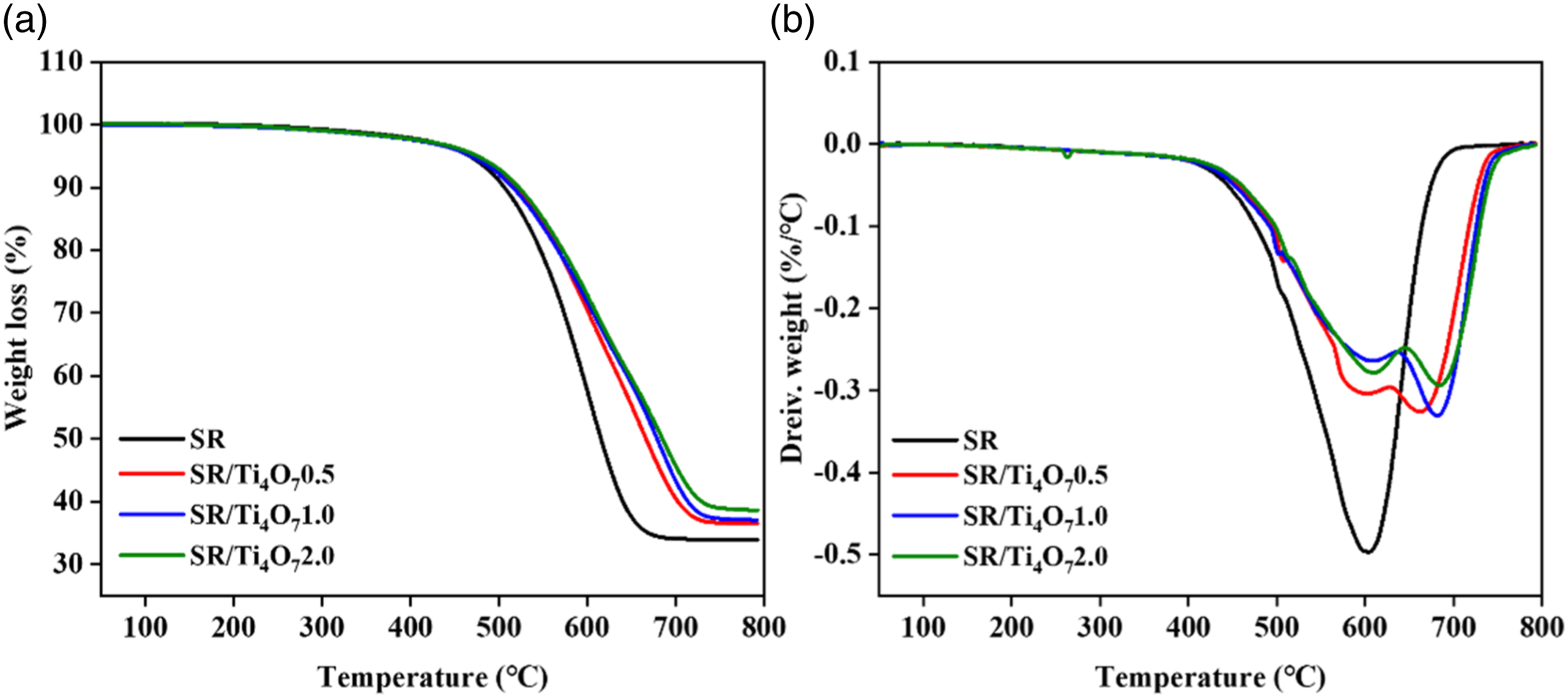
The TGA (a) and DTG (b) of SR and its composites in N2.
In addition to exploring the effect of Ti4O7 on the thermal decomposition stage of the silicone rubber composites in a nitrogen atmosphere, we are also conducting thermogravimetric tests on the silicone rubber composites in an air atmosphere to investigate the effect of oxygen on the thermal stability of the silicone rubber composites. From Figure 5(a) and Table 3, we can see that the T5% of SR without Ti4O7 addition is 424.2°C. As the Ti4O7 content continues to increase, the T5% of the silicone rubber composites gradually increases, and when the number of parts added reaches 2 phr, the T5% of SR/Ti4O72.0 increases to 436.1°C. This pattern is similar to the change in thermal weight loss in a nitrogen atmosphere, but the overall T5% is reduced compared to it because the presence of oxygen accelerates the thermal decomposition reaction of the silicone rubber composite, and the addition of Ti4O7 acts as a physical barrier to inhibit the early decomposition of the silicone rubber to some extent. In addition, the residue content of the silicone rubber composites gradually increases with the increase of Ti4O7 content, and when the addition of Ti4O7 reaches 1 part, the residue content increases to 44.2%, which is 49.0% compared to the residue content of SR.
28
Indeed, this phenomenon can be attributed to the catalytic activity of Ti4O7, which is activated at high temperatures. The elevated temperature stimulates the catalytic reaction between oxygen and the silicone rubber composite, leading to the formation of a solid-phase structure and ultimately promoting the generation of residues. Figure 5(b) shows the Tm and MLRm of the silicone rubber composites, from which we can see that the Tm of all the silicone rubber composites is around 550°C, which is different from the Tm pattern of the samples under nitrogen atmosphere, probably because Ti4O7 has more of a catalytic effect than an adsorption effect in the presence of oxygen, and therefore cannot inhibit the thermal oxidative decomposition of the silicone rubber at high temperatures, but instead acts as a promoter.
29
This can be explained by the change in MLRm. The MLRm of the silicone rubber composites under air atmosphere is generally higher compared to that of the silicone rubber composites under nitrogen atmosphere, which proves that the presence of oxygen promotes the decomposition of the silicone rubber composites, and the addition of Ti4O7 further increases the MLRm, which increases to 1.62%/°C when the number of Ti4O7 additions reaches 1.0 part, indicating that Ti4O7 promotes the degradation of SR composites. However, it should be noted that the MLRm decreases when the Ti4O7 addition fraction reaches 2 phr, due to the decomposition of SR/Ti4O72.0 as early as around 501°C and the unique decomposition peak in the graph. This phenomenon demonstrates the catalytic ability of Ti4O7 to be activated at high temperatures. The TGA (a) and DTG (b) of SR and its composites in air. The data of TGA and DTG in air.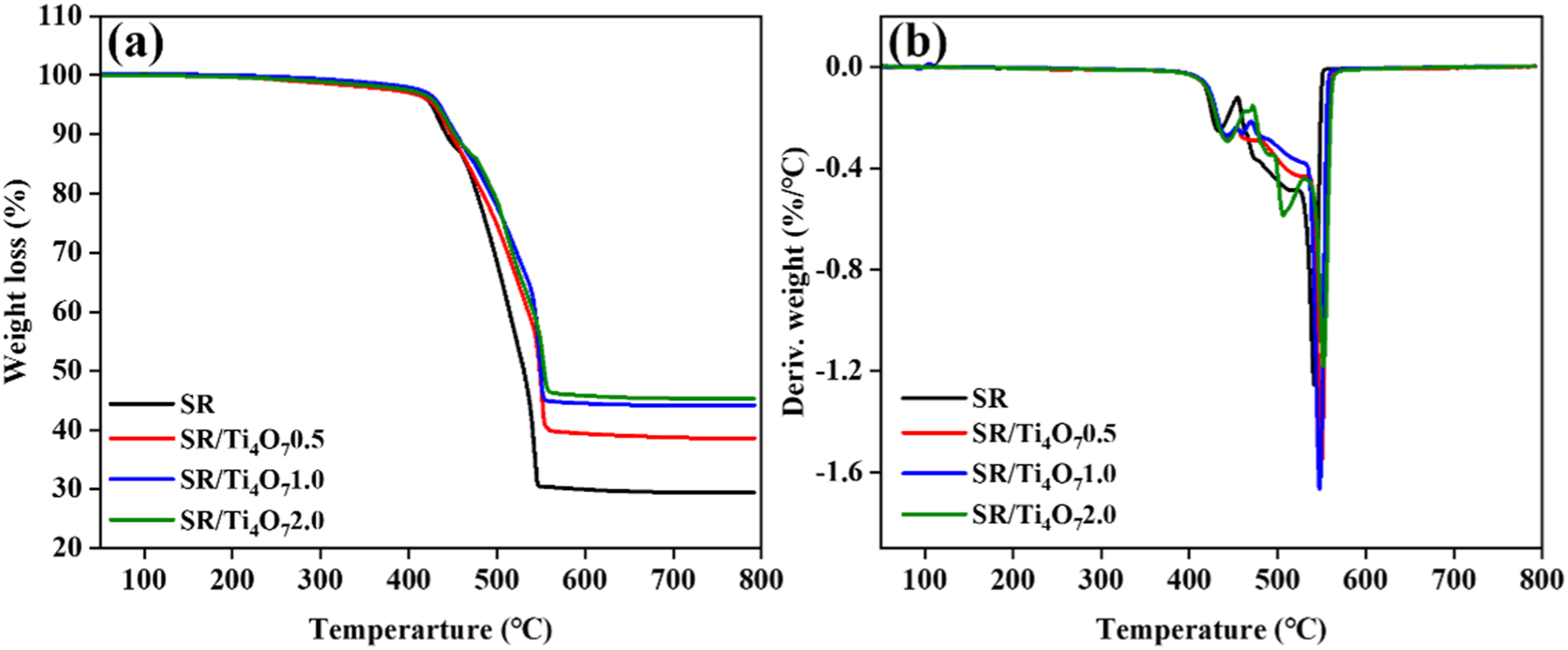

Fire properties of SR and its composites
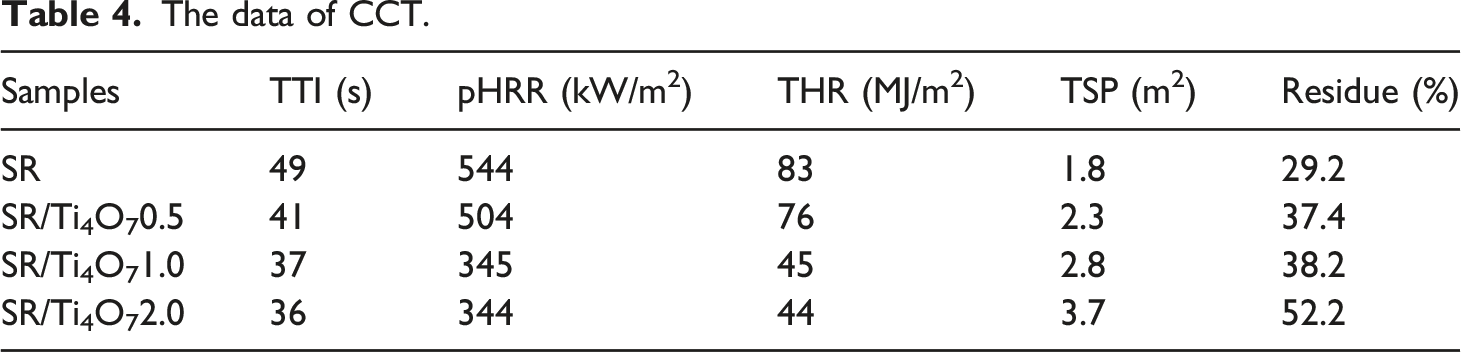
The data of CCT.
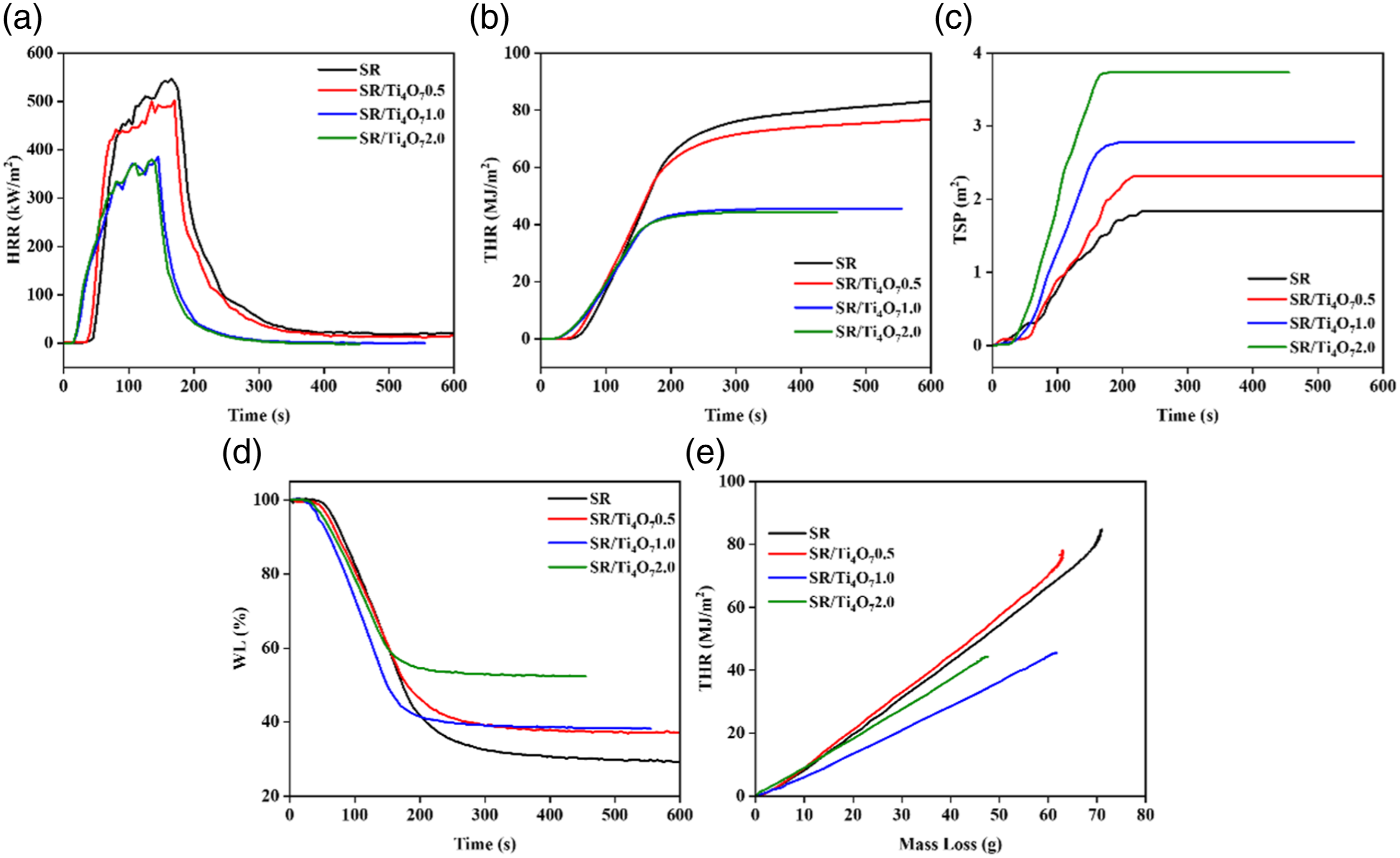
HRR (a), THR (b), TSP (c), WL (d) and THR/ML (e) of SR and its composites.
By exploring the co plotted curves of THR and ML, it can be found that the addition of Ti4O7 has an effect on the gas-phase and condensed phase flame retardancy of SR composite materials. The slope of the curve in Figure 6(e) represents the product of combustion efficiency and effective combustion heat in CCT. When the addition of Ti4O7 reaches 1 part, the flame-retardant effect of the condensed phase is significant, and the mass loss rate decreases while the total heat release also decreases. This indicates that the addition of Ti4O7 can effectively promote the formation of coke, increase its compactness, reduce the decomposition and volatilization of combustibles, and thereby reduce heat release. 33 Continuing to escalate the incorporation of Ti4O7 continues to decrease the rate of mass loss, albeit with marginal alterations in the overall heat release. This tendency might arise from the limitation of Ti4O7's capacity to yield further enhancements in the density of the resulting coke structure.
During the combustion of the silicone rubber composite, an important factor to consider is the changes in the content of various gases detected by the CCT. To further analyze the state of the silicone rubber composite during combustion, we can study these gas variations. Figure 7(a) depicts a graph of the oxygen concentration versus time. It is observed that the silicone rubber composites without Ti4O7 and those with 0.5 part added consume less oxygen during combustion. As the combustion becomes more intense, the oxygen concentration decreases to 19.3%, indicating that the action of oxygen promotes the oxidation and carbonization of the silicone rubber composites. However, the addition of Ti4O7 can increase the degree of incomplete combustion, leading to the production of more CO when it accelerates the generation of thermal insulation.22,23 It is also noteworthy that the peak COP of SR and SR/Ti4O70.5 is delayed by approximately 50 s, indicating that controlling the carbon formation effect can increase the combustibility of the combustible material.
34
This, in turn, can reduce the CO content and increase the CO2 content, thus improving the overall combustion performance of the silicone rubber composite. O2 content (a), COP (b) and CO
2
P (c) of SR and its composites.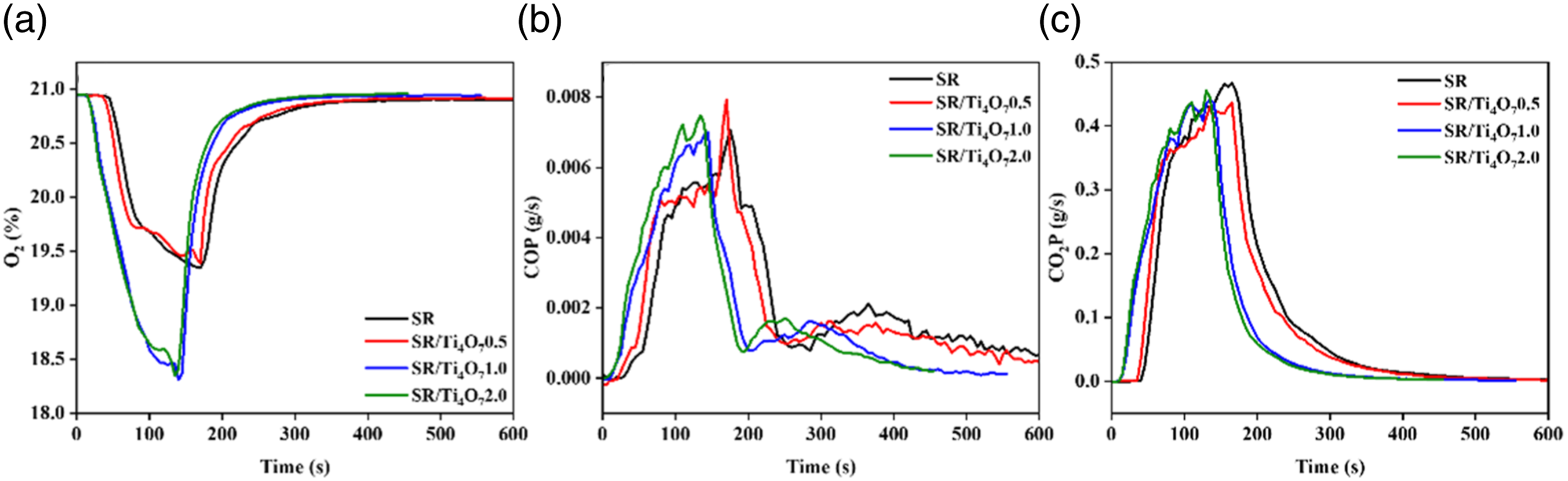
Characterization of char residues
To further understand the effect of Ti4O7 on the carbon formation properties of the silicone rubber composite, the CCT residue is examined. Figure 8 illustrates that the SR residue possesses a loose structure with numerous voids, which impedes the production of good thermal insulation, as both the flame and combustible gases can easily penetrate or diffuse into the interior, thus promoting the complete combustion of the combustible material within. This leads to the release of more heat and a significant rise in the flame hazard level. However, as the Ti4O7 content increases, the surface integrity of the insulation layer gradually improves, and the voids are reduced, thereby enhancing the insulation effect. However, when more than 1 part of Ti4O7 is added, the Ti4O7 tends to agglomerate, leading to a negative impact on the catalytic effect, thus producing more CO and smoke.
35
Therefore, it is essential to control the amount of Ti4O7 used to ensure that the total smoke release (TSP), carbon monoxide peak (COP), and total heat release (THR) are low at the same time. When the addition amount of Ti4O7 is controlled at 1–2 phr, TIO exhibits a significant catalytic carbonization effect during the combustion process of SR composite materials. At the same time as SR combustion produces silica flocs, Ti4O7 will transform the residual carbon in the combustible material into a more stable and dense carbon layer, which can effectively combine with silica flocs to form an effective insulation layer. On this basis, smoke particles and toxic gases generated internally during the combustion of SR composite materials can be blocked inside the insulation layer, making it difficult to release them externally. Therefore, the release of smoke and toxic gases from SR composite materials is reduced, and their flame retardancy is improved.
36
Digital photos of the char residues of (a) SR, (b) SR/Ti4O70.5, (c) SR/Ti4O71.0, (d) SR/Ti4O72.0.
The Raman spectra provide valuable insights into the degree of graphitization of the cone volume residues, which can be assessed by calculating the ID/IG ratio. As presented in Figure 9, the ID/IG values for SR, SR/Ti4O70.5, SR/Ti4O71.0, and SR/Ti4O72.0 are 0.91, 0.85, 0.79, and 0.82, respectively. This trend suggests that increasing the Ti4O7 content has a positive effect on the graphitization of the residue. In a specific context, the introduction of 1 part of Ti4O7 yields an optimal quantity that facilitates effective catalytic carbonization. This transformation induces a shift in amorphous carbon to a more stable configuration, rendering it less susceptible to secondary combustion. However, it’s noteworthy that when surpassing this point, the graphitization of the residual material displays a decline. This phenomenon implies that an excessive surplus of Ti4O7 imparts an adverse impact on the graphitization process. One plausible explanation for this observation is that heightened agglomeration of Ti4O7 particles curtails their catalytic efficacy, thereby impeding the efficient development of graphitic structures within the residual material.
37
These findings underscore the importance of precisely controlling the amount of Ti4O7 added to silicone rubber composites to optimize their graphitization and catalytic properties. Raman spectrum of the char residues of SR, (b) SR/Ti4O70.5, (c) SR/Ti4O71.0, (d) SR/Ti4O72.0.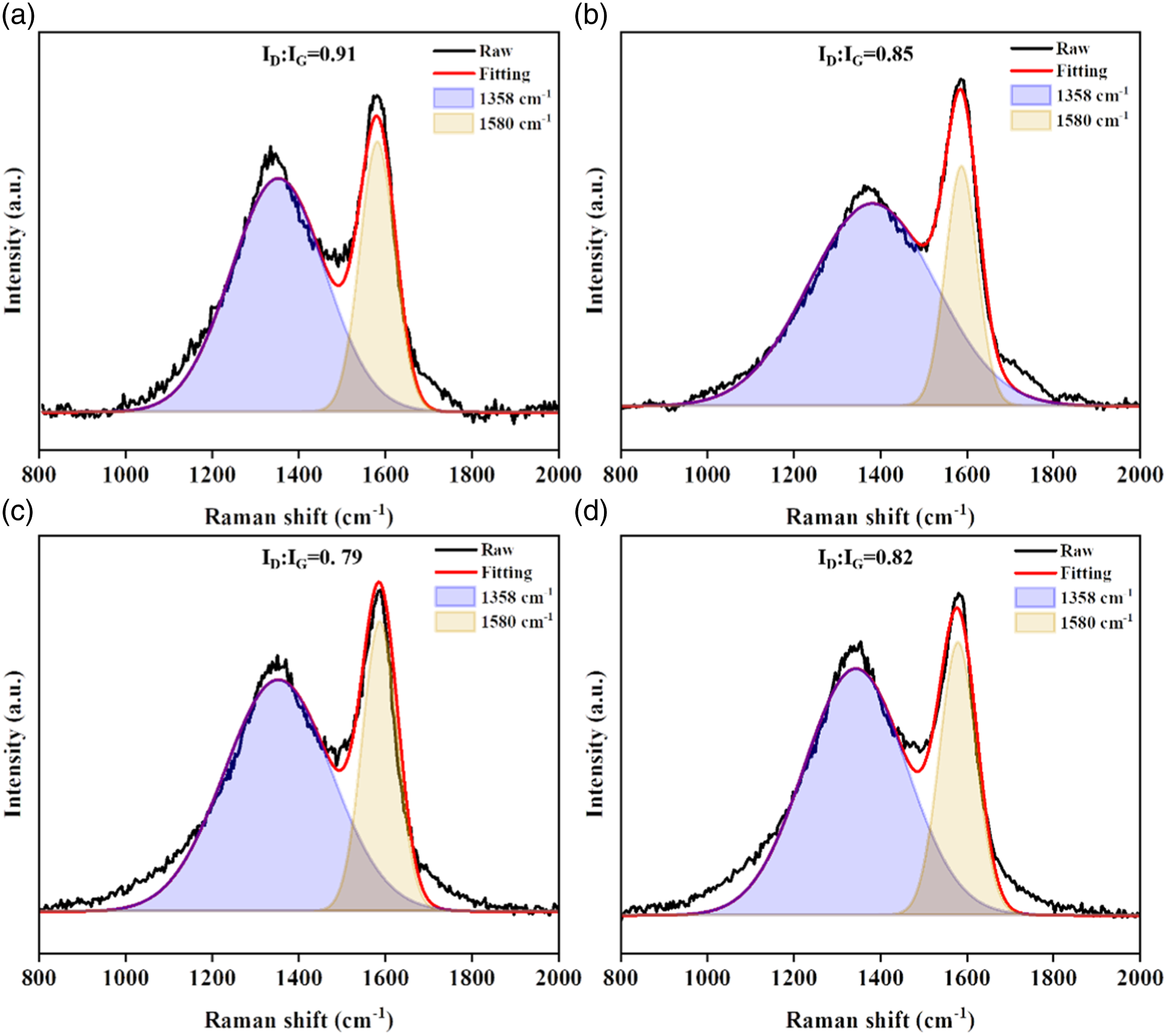
Electrical properties of SR and its composites
From the results presented in Figure 10, it is evident that the silicone rubber composite exhibits high breakdown strength and volume resistivity. However, with an increase in the Ti4O7 content, the breakdown strength and volume resistivity show a gradual decrease, indicating a significant reduction in the insulation properties of the silicone rubber composite, and an increase in its conductivity. The addition of Ti4O7 to the silicone rubber composite results in the formation of a conductive network, which becomes denser with an increase in Ti4O7 content. This increased density of the conductive network facilitates the migration of charges and shortens the breakdown path, leading to a reduction in insulation properties.
38
Moreover, the inclusion of Ti4O7 instigates the emergence of interfacial imperfections within the silicone rubber composite. This phenomenon elevates charge accumulation and induces perturbations in the electric field configuration. Consequently, these factors collectively precipitate a reduction in breakdown strength and volume resistivity. Dielectric strength (a) and volume resistivity (b) of SR and its composites.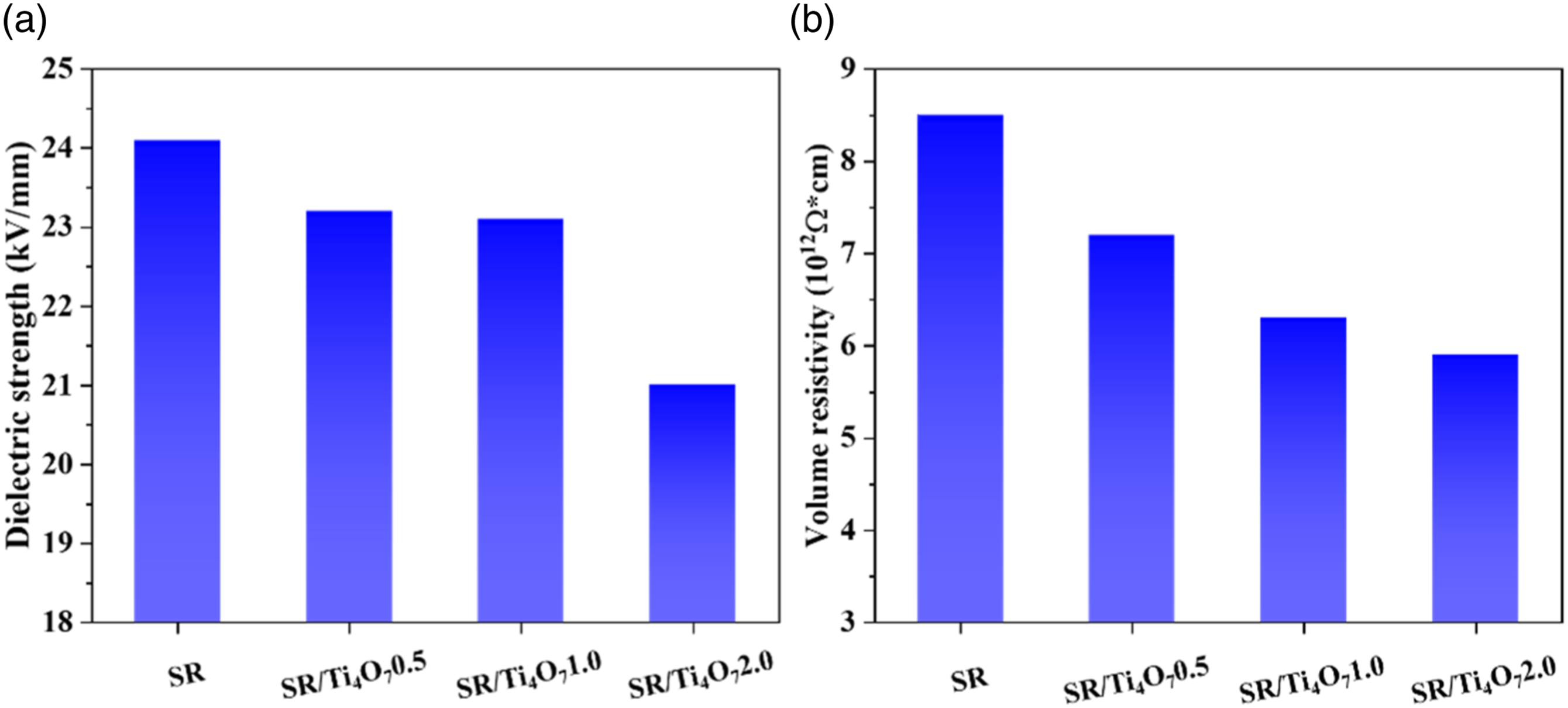
Figure 11(a) shows that the dielectric loss factor of each specimen is smooth in the 101–103 Hz band with no significant loss peaks. However, as the addition of Ti4O7 increases, the loss values of the specimens in the low frequency band (0.01–101 Hz) rise rapidly. This is because the weakly polar silicone rubber lacks polar groups and therefore does not experience high frequency relaxation losses. However, Ti4O7 increases the conductivity current and polarization rate within the silicone rubber, leading to an increase in conductivity losses.
39
In contrast, Figure 11(b) demonstrates that the relative permittivity of each specimen follows essentially the same pattern of variation. This can be attributed to the fact that the addition of Ti4O7 increases the degree of interfacial polarization, promotes the formation of a conductive network in the SR and further enhances the electrical losses. Consequently, these enhance the permittivity of the SR composite. TThe tanδ (a) and dielectric constant (b) of SR and its composites.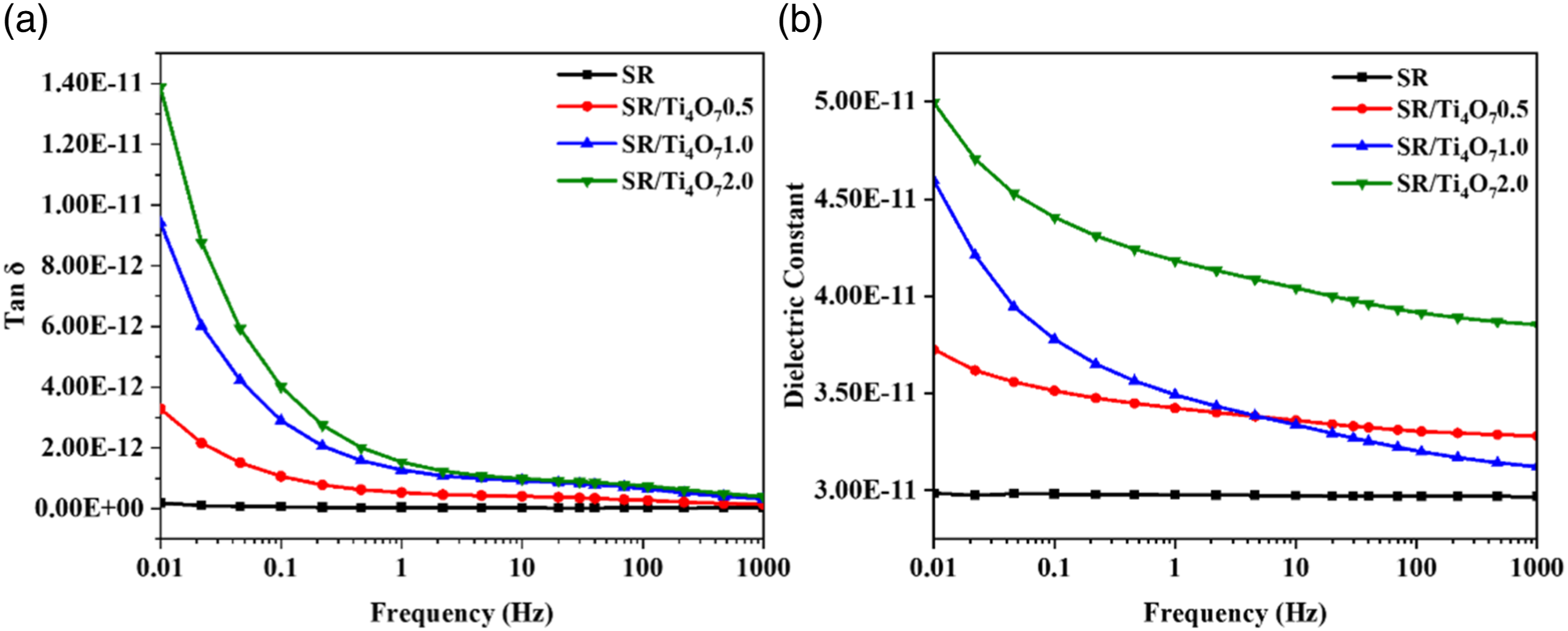
Thermal conduction of SR and its composites
It is important to consider the thermal conductivity of the SR composite, as the addition of Ti4O7 increases the electrical conductivity and therefore the heat inside the SR composite with the passage of electric current. As can be seen in Figure 12, the thermal conductivity of SR without the addition of Ti4O7 is 0.388 W/mK. However, when 0.5 part of Ti4O7 are added, the thermal conductivity of the SR/Ti4O70.5 composite increases to 0.447 W/mK, a significant increase of 15.2% compared to SR.
28
This observation highlights the favorable thermal conductivity properties of Ti4O7. The enhancement in thermal conductivity can be attributed to the creation of thermal conductivity channels within the silicone rubber (SR) composite by Ti4O7, which facilitate accelerated heat transfer. Moreover, as the amount of Ti4O7 added increases, the thermal conductivity further improves, indicating the potential of Ti4O7 to enhance the overall thermal conductivity of the SR composite. Thermal conduction of SR and its composites.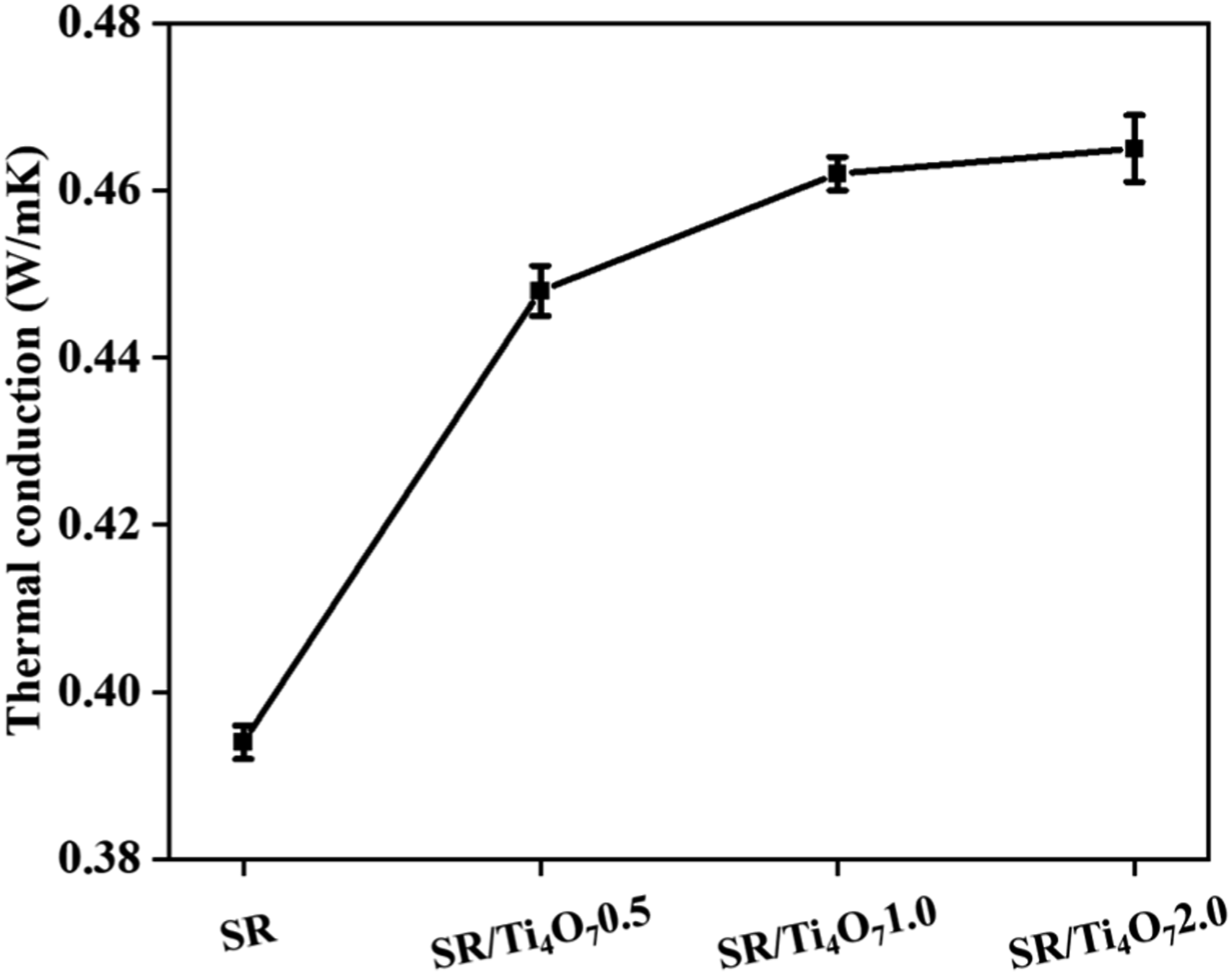
Conclusion
Ti4O7 imparts excellent flame retardancy, thermal stability, mechanical properties, and electrical and thermal conductivity to silicone rubber composites. The silicone rubber composites containing 1.0 part of Ti4O7 show a total heat release (THR) of 45 MJ/m2, which is 45.7% lower than the SR without Ti4O7 addition. The peak heat release rate (PHRR) and time to ignition (TTI) decrease gradually with an increase in Ti4O7 content, reaching 36.8% and 26.5% lower, respectively, at 1.0 part of Ti4O7 compared to SR. In addition, Ti4O7 is more effective at low temperatures and can act as a physical barrier to inhibit the early decomposition of silicone rubber and enhance the thermal stability of silicone rubber composites. When the temperature surpasses 450°C, the catalytic activity of Ti4O7 becomes active, leading to the stimulation of the decomposition process in silicone rubber composites and resulting in increased residue formation. Moreover, the addition of Ti4O7 in suitable proportions can additionally enhance the mechanical properties of the silicone rubber composites and improve their electrical and thermal conductivity.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
