Abstract
Thermoplastic starch (TPS) suffers from its intrinsic low mechanical strength and high brittleness due to its strong hydrogen bonding and low chain mobility. The conventional way to crosslink the TPS film can improve the strength and stiffness of the films, but usually reduces the flexibility of the film, and increases its brittleness. In this study, the incorporation of the hybrid nanofiller [1 wt% nanocellulose (C) and 4 wt% nano bentonite (B)] into the TPS proved to improve greatly the films’ strength and flexibility. The hybrid nanofillers with ratio 4B:1C was incorporated into the crosslinked thermoplastic corn starch (CR-TPCS) film to increase the its flexibility and toughness and produced a high mechanical strength fully biodegradable film. Two different aqueous carboxylic acids: citric acid (CA) and tartaric acid (TA) with different pH values (2,4,6) as the green crosslinker were employed. Substantial increase of tensile strength (3.98 to 9.17 MPa), Young’s modulus (9.10 to 46.30 MPa) and elongation at break (55.2 to 135.7%) was observed for the CA- 4B1C/pH2 films compared to the CR-TPCS films. The melting temperature (Tm) of the CA-4B1C/pH2 improved compared to the TPCS/4B1C (un-crosslinked) film due to its crosslinking effect. Meanwhile, the CA-4B1C films exhibited the highest degree of substitution and di-esterification with the lowest swelling and water solubility properties due to the formation of a special “bridge” structure between the CA, nanocellulose and plasticizer. The “bridge” structure developed between the TPCS chains serves as the toughener to motivate higher chain stress relaxation and load endurance. The crosslinked “bridge structure” also proved to effectively reduce the retrogradation phenomenal in the TPCS films. This combination method of hybridization and crosslinking is an efficient, low cost, and environmentally friendly technique to overcome the low flexibility and brittleness problem of the TPS based packaging film.
Introduction
Plastic pollution is constantly the top priority issues of the scientific community, policymakers, and environmentalists worldwide. 1 Plastic packaging, particularly single-use plastics with a service life spanning from a few hours to weeks, contribute significantly to the enormous proportion of plastic garbage. Most plastic packaging wastes will generate plastic pollution as they accumulate in landfills or the ocean. The accumulated plastic waste in the ocean has become a threat to the ocean ecosystem and plastic marine debris will be developed into microplastic and adversely impacts the economy, marine environment, ecosystem, and human health. It was estimated that there would be one ton of ocean plastics for every three tons of fish by 2025 if no preventative measures were implemented.2,3 To overcome the plastic pollution problem, many environmental agencies and governments from various countries are working together to ban and encourage people to reduce the use of single-use plastic packaging. 4
Biopolymers such as thermoplastic starch (TPS) and cellulose derived from renewable sources have been extensively studied and investigated recently to solve the plastic pollution problem as they are biodegradable, low-cost, and promising raw materials. However, thermoplastic starch has poor mechanical properties, processability, long-term stability, and high-water sensitivity compared to synthetic polymers. 5 Various TPS-based biocomposites have been investigated and developed by incorporating various nanofillers into their structure to overcome the drawback of thermoplastic starch matrix and enhance its properties. 6 Nevertheless, TPS biocomposites tend to exhibit an intrinsic brittle behaviour, which raises integrity concerns in packaging applications as high toughness and flexibility are critical to film packaging applications. Therefore, exploring toughening strategies to overcome the brittle properties and low toughness has arisen as an important research topic. In our previous study, we have successfully developed a high tensile strength, Young’s modulus and elongation at break by solely incorporating natural hybrid nanofiller (nano bentonite and nanocellulose) into the thermoplastic corn starch (TPCS) films. The detailed toughening mechanism of the hybrid TPCS biocomposite films is being reported in our previous publication, showing the exfoliation of nano bentonite that responsible for the enhancement of the mechanical strength, whereas the small and evenly distributed aggregated nanocellulose responsible for triggering the accelerated chain relaxation of the TPS during straining. 7 Besides that, the hybrid nanofillers were also reported to effectively inhibit the retrogradation rate of the films and prolong the films’ stability. 8
Currently, non-toxic chemical crosslinking is in high demand as most of the common chemical crosslinkers have been raise the toxicity issues by certain researchers in their unreacted forms. Chemical crosslinkers such as glutaraldehyde, 9 formaldehyde,10,11 boric acid 11 and epichlorohydrin 12 are identify to be toxic to human cells which limiting its application in food packaging even though they were proved has high efficiency for stimulate high crosslinked density. To overcome the toxicity dilemma and maintain the high crosslinked efficiency, the green crosslinker such as citric acid and tartaric acid were used to produce crosslinked TPS films.13,14 TPS chains are highly susceptible to crosslinked reaction with carboxylic acid as they readily contain highly reactive hydroxyl group to react with carboxyl group from carboxylic acid for esterification reaction. 15 Several research showed that the citric acid and tartaric acid could form crosslinked TPS films in acidic conditions.16–18 Furthermore, the citric acid and tartaric also been recognized as safe material according to FDA and EFSA and thus, suitable for use in food packaging applications. 19 The incorporation of the carboxylic acid can enhance the TPS film’s mechanical and hydration properties by forming strong hydrogen bonding and ester chemical bonding with the TPS chains.20,21 The carboxylic acid can function as an external plasticizer and as an internal plasticizer in the TPS matrix. The dual function carboxylic acid is due to the partial esterification ability of carboxylic acid with the TPS chains, which interrupts the intra and inter TPS molecular chains by chemically attaching to them. The partial esterification of the TPS chains not only improved the mechanical properties of the TPS films but also opened the potential for TPS films to be incorporated with other biodegradable polyesters for modification purposes. Other than chemical crosslinking method, the additional of plasticizer could also enhance the elongation at break and flexibility of biopolymer. 22
Considering the carboxylic acid contains at least two carboxyl group per molecules, it is expected to react with more than one hydroxyl group present in the TPS biocomposite system. Many studies reported that the TPS biocomposite system components, such as plasticizer and nanocellulose, which contain the hydroxyl group in their structure would compete with the TPS chains to form ester linkage with a carboxylic acid such as citric acid.18,23,24 Some studies even showed that the citric acid has a higher esterification crosslinked reaction with the plasticizer such as glycerol and nanocellulose due to the primary hydroxyl group and the lower steric hindrance compared to the vast TPS chains.25–27 The competing crosslinked reaction between the carboxylic acid and the TPS chains reduces the crosslinked density and subsequently minimized the enhancement toward the films’ strength. The incorporation of carboxylic acid with glycerol was reported to enhance the plasticizer degree of the TPS films, however, with the loss of tensile strength. 23
It is interesting to note that, as the TPS chain interacted with a carboxylic acid in an acidic condition and excess water conditions with elevated temperature, a concurrent reaction to the crosslinked reaction, acid hydrolysis reaction was also taking place.16,28 The TPCS chains were hydrolysed at the glycosidic linkages as protonation of the glycosidic oxygen and the nucleophilic replacement reaction with the water molecules to yield the reducing sugar end group. The molecular weight of the TPS chains was significantly reduced due to the acid hydrolysis reaction. The acid hydrolysis reaction will also compromise the TPS films’ mechanical properties and reduce the films’ crosslinked density. Therefore, a novel method to prevent the acid hydrolysis of the TPS chains is vital when carboxylic acid is mixed with the TPS at elevated temperatures. From the previous studies in this area, it is known that the onset of acid hydrolysis is highly related to the low pH values and high content of carboxylic acid.19,29 Therefore, the adjustment of the pH values could be possible to eliminate the acid hydrolysis happening in the TPS films.
As reported in the previous studies, chemical crosslinking was effective to enhance the mechanical properties of TPS films by crosslinking the TPS chains. However, most of the chemical crosslinking procedures on the TPS films indicate a lack of capability in improving the tensile strength and elongation at break at the same time. The limited mobility of the crosslinked TPS chains was due to the low volume extension and low relaxation enthalpy during films straining. Therefore, to increase the flexibility of the crosslinked TPS chains, the idea of extending crosslinking chains within the TPS structure has been proposed to stimulate higher film’s toughness and flexibility. 30 Literature has discussed various reactive agents for crosslinking reactions, such as epichlorohydrin, 31 citric acid, 32 epoxidized palm oil (EPO), 33 N,N-Dimethylformamide 34 and tartaric acid. 35 Peidayesh et al. reported an effective way to improve the physicochemical properties of thermoplastic starch by in situ reaction of starch, glycerol, and epichlorohydrin (ECH). 36 The formation of both noncovalent and covalent bonds between ECH and glycerol in starch produced a long chain in the starch matrix and improved film physicochemical properties. Meanwhile, Jianlei et al. tailored the flexibility of the thermoplastic starch via citric acid-epoxidized palm oil (CEPO) oligomer using melt blending. 33 The strong esterification between the starch/fiber and the citric acid was reported to be responsible for the high toughness of the films. Meanwhile, Liu et al. also reported the significant enhancement in the toughness of the bamboo fibers-reinforced bio-composites upon the application of the tannic acid-crosslinked ESO oligomer via reactive extrusion. 37 Based on the literatures, we can the crosslinking between the reactive agent and the fillers or fibers could induce a long flexible chain and tough bridge, improving the tensile properties and flexibility of the films.
Therefore, in this studies, two natural carboxylic acids with different pH reactions medium were incorporated with our previous reported highest toughness TPCS hybrid biocomposites films to produce the optimal crosslinked TPCS hybrid biocomposites films and further enhanced the films’ mechanical. Based on our previous report, the nanocellulose greatly enhanced the films’ flexibility. However, the linkage between the nanocellulose and TPCS starch was merely depending on the noncovalent bonding: hydrogen bonding. The crosslinking reaction was proposed in this study to further enhance by forming a strong covalent bond between the linkage of nanocellulose and TPCS chain. Surprisingly, the incorporation of carboxylic acid not only strengthen the tensile strength but also enhanced the elongation at break which was not reported in any other studies. The outstanding films’ mechanical properties is explained well in this study and showing the carboxylic acid enhanced the compatibility between nanofillers and TPCS chain through the crosslinking reaction even further promoted the synergic effect the hybrid nanofiller. The chemical crosslinking between the nanofiller, plasticizer and the carboxylic acid could act as the “bridge” between the TPCS chain which result in higher mechanical strength and high flexibility. The extension of “bridge” concept to increase the polymer flexibility was well established in the reported journal studies.33–36,38
Material
Corn starch with 72% amylopectin and 28% amylose was purchased from Sigma Aldrich, Nanocellulose was chemically extracted from oil palm empty fruit bunch (OPEFB) and used as organic nanofiller. Nano bentonite, which was purchased from Sigma Aldrich, used as the hybrid nanofiller with nanocellulose to enhance the toughness of the films. The detail of the nano hybrid filler preparation was reported in our previous study. 39 Distilled water and glycerol purchased from HmbG Chemicals were used as the plasticizer for the TPCS film. Citric acid and Tartaric acid were used as the crosslinker, and each carboxylic acid was purchased from the Sigma Aldrich.
Preparation of crosslinked TPCS and TPCS/B/C films (CR-TPCS and CR-TPCS/B/C)
Corn starch was added with water in a ratio of 1:20 into a 250 mL beaker. Afterward, glycerol was added into the starch solution as the plasticizer for the starch in the ratio 1:50. The mixture was continuously stirred at a temperature of 75–85°C using a magnetic stirrer (500 r/min) for 45 min to obtain a viscous and transparent mixture. A gel form of starch was obtained.
From the previous research work in this area of studies,8,39 TPCS with 4 wt% of nano bentonite (B) and 1 wt% of nanocellulose (C) possessed the excellent homogeneity between the nanofiller interaction which enhance significantly the TPCS film’ toughness. Therefore, the ratio of 4B:1C were selected to promote flexibility of CR-TPCS films with different carboxylic acid. Hybrid nanofiller 4B:1C was added into the TPCS solution and continuously stirred at 75–85°C.
Acronym of CR-TPCS and TPCS hybrid biocomposites films (CR-4B1C).

Testing and characterization procedures
Fourier transform infrared
The chemical structure and interaction of the hybrid nanofiller, carboxylic acid with the TPCS were analyzed using a Perkin Elmer FTIR spectrometer with Attenuated Total Reflectance (ATR). The interaction of the chemical functional group in CR-TPCS and CR-4B1C films were identified using a Perkin Elmer spectrum 65 FTIR spectrometer with a wavelength range of 650–4500 cm−1 with 16 scans and resolution of 4 cm−1.
X-ray diffraction analysis
The crystalline structure of TPCS, TPCS/4B1C, CR-TPCS and CR-4B1C films was analyzed using Bruker D2 Phaser X-ray diffractometer with Cu Kα X-rays. The XRD data were analyzed using the high software score plus version 2.0 software (licensed to PANalytical B.V. Almelo). All the film samples were cut into 25 mm × 25 mm × 0.3 mm. The samples were tested using scan rate 0.1 s per step from 2θ = 5° – 40°.
Scanning electron microscope
The fracture surface structure and morphology of the films samples were analysed using a scanning electron microscope (SEM) (JEOL JSM-6460LA). The morphology of fracture surface of the films was studied under SEM for investigation of the fracture behaviour of the films. Before the micro-images were captured, the films’ fracture surface was coated with platinum using JFC-1600 Auto Fine Coater (JEOL Ltd, Japan) to get the optimal SEM image.
Tensile testing
The tensile test of the film samples was performed using Instron machine model-5582, according to ASTM D 638 Type V. The film was cut into dumbbell shapes with 10 mm × 3.2 mm cross-section area, and each film’s thickness is around 0.20–0.25 mm. The samples were stored in an airtight seal desiccator to control the film samples’ moisture content. The samples were tested with a crosshead speed of 10 mm/min, and the stress-strain curve was obtained. Ten samples were tested for each formulation, and the mean value and standard deviation for all the formulations were calculated. Statistical analysis was performed using two-tailed Student’s t-test for unpaired data to compare among materials. A significance level of 0.05 or less was accepted as statistically significant.
Differential scanning calorimetry
The thermal properties associated with melting retrograded crystalline structure of TPCS films were studied from temperature 25°C to 180°C, with a heating rate of 10°C/min. 3.0 mg of films were weighed, and an aluminium pan was used as the reference. All the films’ melting temperature (Tm), and the enthalpy of fusion (ΔHf) were obtained from the DSC equipped with the analysis of the thermal analysis software.
Degree of substitution of CR-TPCS and CR-4B1C hybrid biocomposites films
The degree of substitution of the hydroxyl group of the films was determined by the titration method applied in the study of Owi et al. and Zain et al.28,40 1.5 g of samples were stirred in the 50 mL of distilled water, and the mixture was saponified with 0.5 N of sodium hydroxide solution. The mixture was continuously stirred to dissolute the films for 6 h at 25°C with a speed of 500 r/min. A few drops of indicator phenolphthalein were dropped into the mixture. The excess NaOH was back-titrated with 0.5 N of H2SO4 solution until the phenolphthalein changed from pink to colourless. The volume of the H2SO4 solution was recorded. The degree of substitution was calculated using the equation (2).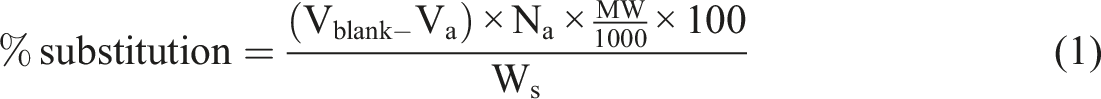
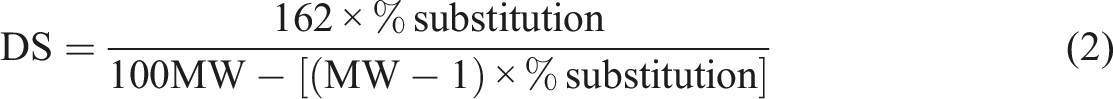
Where Vblank (ml) is the volume of H2SO4 used for non-crosslinked TPCS and TPCS hybrid biocomposites films. Va (ml) is the volume of H2SO4 used for CR-TPCS and CR-TPCS hybrid biocomposites films, Na a (mol/L) is the normality of H2SO4, MW (g/mol) is the molecular weight of ester group, and Ws is the weight of the samples.
The degree of di-esterification of CR-TPCS and CR-4B1C hybrid biocomposite films according to the complexometric titration of a carboxylic acid with copper (ii)-sulfate
The degree of esterification of carboxylic acid in the films was carried out using the complexometric titration method according to Olsson et al.
19
Due to the stable complex formation reaction between the mono-esterified carboxylic acid with copper (ii) ions, two independent titration process was carried out on all the crosslinked films. For the first titration, 0.3 g of films was hydrolyzed in the 50 mL, 0.1 M KOH solution in boiling form for 20 min. After that, the solution was cooled to room temperature, and the pH of the solution was adjusted to 8.5 by using a borax/boric acid buffer solution. The mixture was mixed with distilled water to form a total of 250 mL, and eriochrome Black T was added as an indicator. The mixture was then titrated with 0.02 M copper (ii) ions until the solution changed from purple to blue. The first titration was done to determine the total amount of free and esterification of the carboxylic acid. Meanwhile, the crosslinked films were repeated in the same process as the first titration for the second titration. However, the crosslinked films were not hydrolyzed in the KOH solution but only pre-swollen in the water for 20 min. The difference between the titration of the hydrolyzed and non-hydrolyzed films was able to calculate the degree of di-esterification by using the formula below.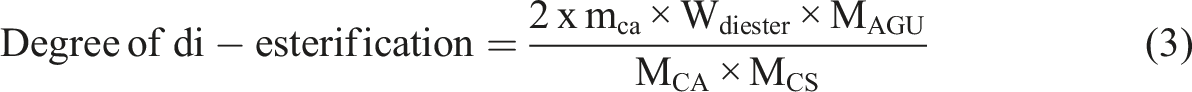
Where
Water vapor permeability
The water vapor permeability of the films was determined by using ASTM E 96 for the cup method, where the water vapor permeability of films was calculated by water vapor transmitted out from the container under constant temperature and humidity. CR-TPCS and CR-4B1C films were prepared in even thickness (0.20 mm). A constant amount of distilled water was filled into the container (19 mm depth), and samples films were attached and sealed with rubber to prevent water leakage around the container edges during the testing. The testing temperature and humidity were regulated with different within 1°C and 2%, respectively. Constant airflow condition was required to maintain good circulation of the environment for the test location. The container with films 80.0 g was weighed, and the reading was recorded as the initial weight. The specimen’s weight is weighed periodically (everyday) for 7 days. The water vapor permeability test was calculated according to the below formula:
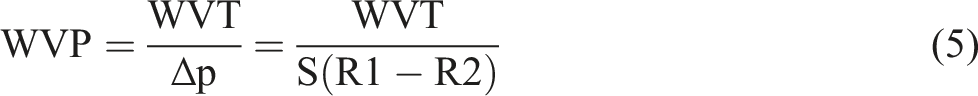
Where WVP defined as the rate of water vapor permeability and ∆p is the vapor pressure difference in the container and the environment, mmHg. Meanwhile S representing the saturation vapor pressure at the test temperature, R1 is the relative humidity in the container in a fraction and R2 = relative humidity of the environment in a fraction
The swelling test and solubility test of CR-TPCS and CR-4B1C hybrid biocomposites films
The swelling test was conducted by immersing the films into 20 mL dimethyl sulfoxide (DMSO) with 2 cm × 2 cm and the film’s thickness 0.015–0.020 cm at 25°C according to the method of Mohaiyiddin et al.
41
The films were taken out after 5 h, gently wiped off the excess DMSO, and weighed. The swelling percentage was calculated by equation (6). After that, the swelling samples were dried in the oven at 50°C for 1 day to remove the film’s moisture content altogether. The weight before and after the swelling was calculated to determine the solubility percentage of the crosslinked films.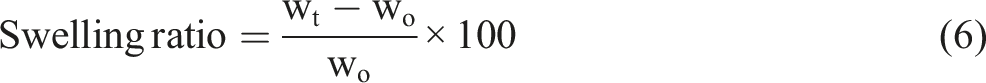
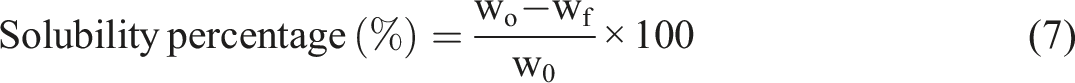
Result and discussion
Tensile properties for CR-TPCS and CR-4B1C films
Tensile strength, young’s modulus, and elongation at break of CR-TPCS and CR-4B1C.
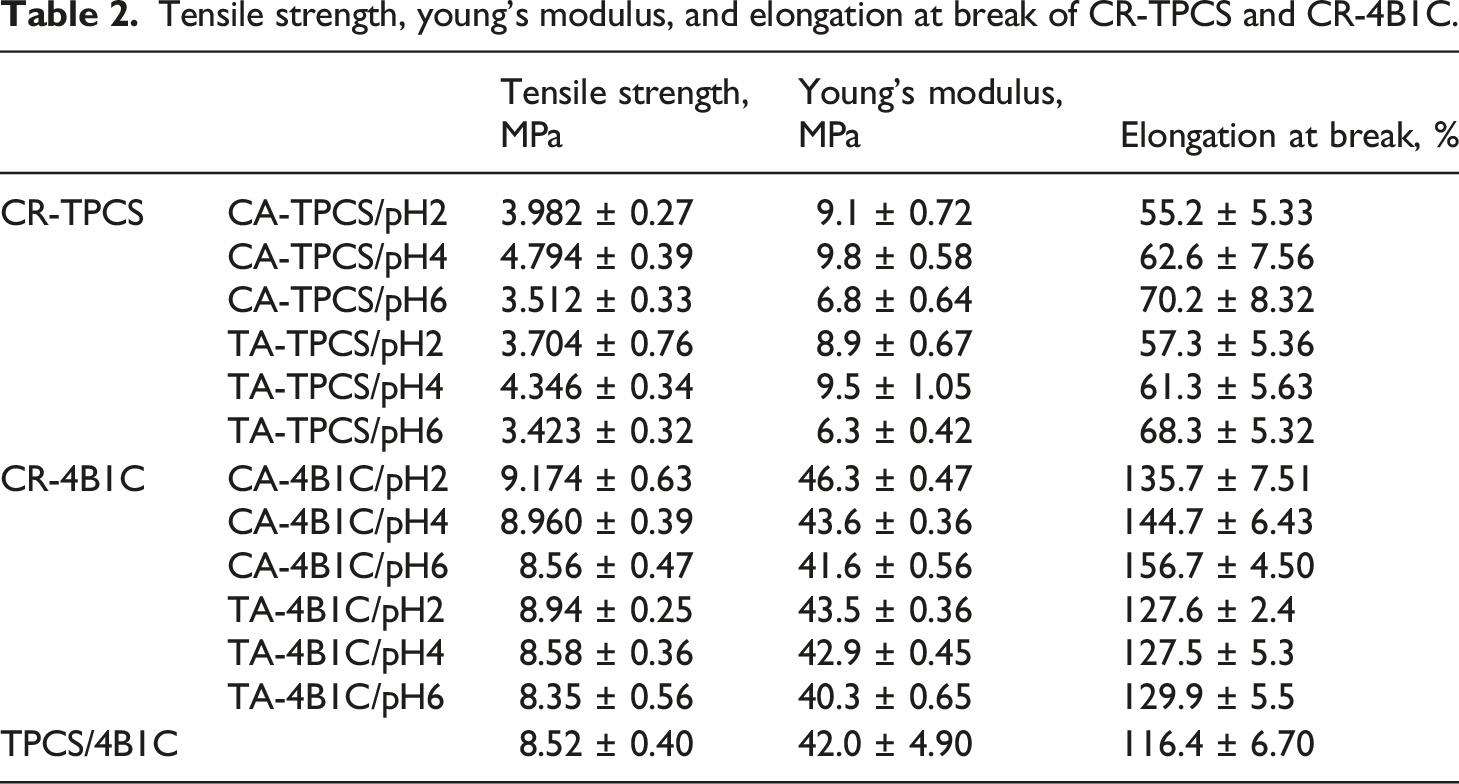
Generally, the CA-TPCS films showed marginally higher tensile strength and Young’s modulus than the TA-TPCS films in their respective pH values. The different mechanical properties between the CA-TPCS and TA-TPCS may be attributed to the different carboxyl groups in their chemical structure. CA has tri-carboxyl group in its structure, which exhibited a higher crosslink ability than the TA (di-carboxyl group) due to the extra one carboxyl group in the structure. The higher cross-linked ability resulted in higher crosslinking density in the TPCS matrix and exhibited higher tensile properties. 42 Besides that, the esterification reaction between carboxylic acid and the hydroxyl group was catalysed in the acidic medium. Therefore, the lower the pH values, the higher the tensile properties were expected as low pH values favoured the crosslinked reaction. However, the CA-TPCS/pH 2 and TA-TPCS/pH 2 exhibited a lower tensile strength and Young’s Modulus than CA-TPCS and TA-TPCS at pH 4. The lower tensile strength and Young’s modulus of the CR-TPCS at pH 2 could be attributed to the lower film cross-linked density compared to the CR-TPCS at pH 4.
Besides that, the CR-TPCS films at pH 2 also displayed the lowest elongation at break compared to other CR-TPCS films. The lower mechanical properties of the CR-TPCS films at pH2 could be due to the acid hydrolysis reaction of amylopectin chain at the low pH value, which reduces the molecular size of the TPCS chain. The reduced molecular length of the amylopectin chain causes lower interconnectivity between the amylose and amylopectin, resulting in the lowest elongation at break. The same result was also reported by Hassan et al., where the incorporation equal to or more than 5% of citric acid could induce hydrolysis of the TPCS chain, which results in lower elongation at break. 18 The acid hydrolysis reaction of amylose was proved through the result of XRD and where the CR-TPCS/pH2 has higher Vh crystallinity percentage compared to CR-TPCS/pH4 indicating increase of the single-helix amylose crystalline structure due to the hydrolysis reaction of the amylopectin. Besides that, the fractured surface morphology of the films further strengthening the acid hydrolysis reaction as hydrolysed agglomerated TPCS matrix was observed in Figure 8(a) and (b). Meanwhile, the hydrolysed amylopectin chains will result in a more open and less compact branch structure, which provides higher flexibility for the carboxylic acid to form ester bond linkage with the plasticizer and nanocellulose rather than the TPCS chains. The predominating chemically bonding between carboxylic acid, glycerol and nanocellulose promoting the competing reaction reduced the available reaction side of carboxylic acid with TPCS chain, resulting in lower crosslinked density. The crosslinking between the carboxylic acid, glycerol or either nanocellulose are accumulated as a relative long segments of chain without crosslinked and induce higher plastic flow and lead to lower load endurance. 36 Besides that, the shorter amylopectin in CR-TPCS at pH2 due to acid hydrolysis reaction could form a different extent of small crystalline structure in the TPCS matrix during retrogradation. The small crystalline structure in the films may act as the stress concentration point or the weakness, causing premature failure and low-stress transfer efficiency during the film straining.
Meanwhile, for CR-4B1C films, the tensile strength and Young’s modulus were decreased as the pH values increased. Different tensile properties trend was observed compared with CR-TPCS films. CR-4B1C films exhibited the highest tensile strength and Young’s modulus at pH2 rather than pH4. The highest tensile strength and Young’s modulus of CR-4B1C films was observed at pH2 indicated that the acid hydrolysis of the TPCS chains could significantly reduce by incorporating the hybrid nanofiller. The results also in good agreement with the XRD data, where the crystallinity percentage of Vh structure was seen to reduce and the agglomerated TPCS hydrolysed structure was diminished, as observed through the SEM morphology. Meanwhile, the tensile strength and Young’s modulus of the films decreased as the pH values increased, demonstrating that the cross-linked reaction's reactivity was reduced with the increase in pH values. This result was expected as the high cross-linked reaction was preferred at the low pH values. The explanation was supported by Olsson et al., where the esterification of citric acid decreased when the pH values increased from 2 to 5. 19 The lower esterification reaction was confirmed through the analysis of degree of di-esterification and degree of substitution as both results showed a decreasing trend when the pH values increased.
Besides that, the greater tensile properties of CA-4B1C/pH2 films compared to non-crosslinked TPCS/4B1C also suggested that the carboxylic acid can further enhance the synergic effect of the hybrid nanofiller through the cross-linked reaction. CR-4B1C films at pH6 exhibited the highest elongation at break compared to the films at pH 2 and 4, showing the highest chain mobility and least cross-linked density. As mentioned previously, for the CR-TPCS films, the pH values near the neutral pH may not favour the cross-link reaction, which results in less chemical bonding between the carboxylic acid and the TPCS, glycerol, and nanocellulose. In this case, the carboxylic acid acted as the external plasticizer, forming intra or inter-linking with all the components through hydrogen bonding. Even though the CR-4B1C/pH6 showed improved tensile strength and elongation at break compared to TPCS/4B1C, Young’s modulus of the films experienced a slight decrease. The decrease in Yong’s modulus showed that the plasticizing effect of a carboxylic acid is more dominant than the strengthening effect.
However, an interesting observation was that the CA-4B1C/pH2 and pH4 showed improvement in all tensile parameters compared to the TPCS/4B1C films. Meanwhile, for TA-4B1C/pH2 and pH4, the only improved tensile parameter was tensile strength and Young’s modulus. The elongation at break for TA-4B1C/pH2 and pH4 does not exhibit a significantly improved compared to the TPCS/4B1C films. The higher tensile parameter of CA-4B1C/pH2 tentatively attributed to the high crosslinked reactivity of citric acid, which chemically bonded the glycerol and nanocellulose to form an extended chemical “bridge” between the TPCS chain. The long and chemically bonded bridge formed in the CA-4B1C/pH2 provided a strong linkage and stimulated a higher chain relaxation reaction between all the components during straining.
A comparative study was done to compare our results with other TPS biocomposite crosslinked systems that other researchers investigated. Based on the literature analysis, the tensile strength of our crosslinked TPCS hybrid films is on par with the reported value from other studies. Meanwhile, some studies have reported a much higher tensile strength and Young’s modulus than our films. However, the high tensile strength and Young’s modulus films are reported with extremely low elongation at break, such as 5%, which was not viable in film packaging applications. Besides the on-par tensile strength and Young’s modulus, our crosslinked TPCS films also exhibited the highest elongation at break compared to the reported values. Limin et al. produced a crosslinked starch-based nanocomposite film with the crosslinked reaction between the starch nanocrystalline and sodium hexametaphosphate (SHMP). 43 The optimal tensile strength, 10.8 MPa, was achieved as 10% of starch nanocrystalline was crosslinked within the starch-based structure. However, the elongation at the nanocomposite’s break has decreased significantly from 150% to 52%. In another study, Babak et al. reinforced the starch films with the crosslinking reaction between citric acid and carboxymethyl cellulose (CMC). The tensile strength of the crosslinked starch films has increased from 6.57 MPa to 16.11 MPa when added with 20% of CMC, and the elongation at break was reduced to 63%. 44 For more recent work, WeiTieng et al. have unveiled the properties of crosslinked tapioca starch with the crosslinked lime juice and varying the loading of nanocellulose from 1 phrs to 5 phrs. 40 They have reported that adding ten parts of lime juice per hundred parts of starch and 1 phrs of nanocellulose could significantly enhance the tensile strength and flexibility of the bionanocomposite films by achieving the optimal elongation at break, which was 106%. Based on the reported tensile properties, especially elongation at break, the reported values for crosslinked TPS films in other studies were much lower than our optimal crosslinked TPCS hybrid filler films at pH2. From our previous study, 7 an impressive high tensile strength and elongation at break of the TPCS hybrid nanofillers films was reported and this trend is rarely reported and obtained. In this study, the high tensile strength, and the high elongation at break were further enhanced by adding carboxylic acid into the TPCS hybrid nanofiller films. The carboxylic acid has played a role in improving the tensile strength and elongation at break in this study by introducing the crosslinking reaction between the added components and the TPCS matrix.
Crystalline structure of CR-TPCS and CR-4B1C studies through the XRD analysis
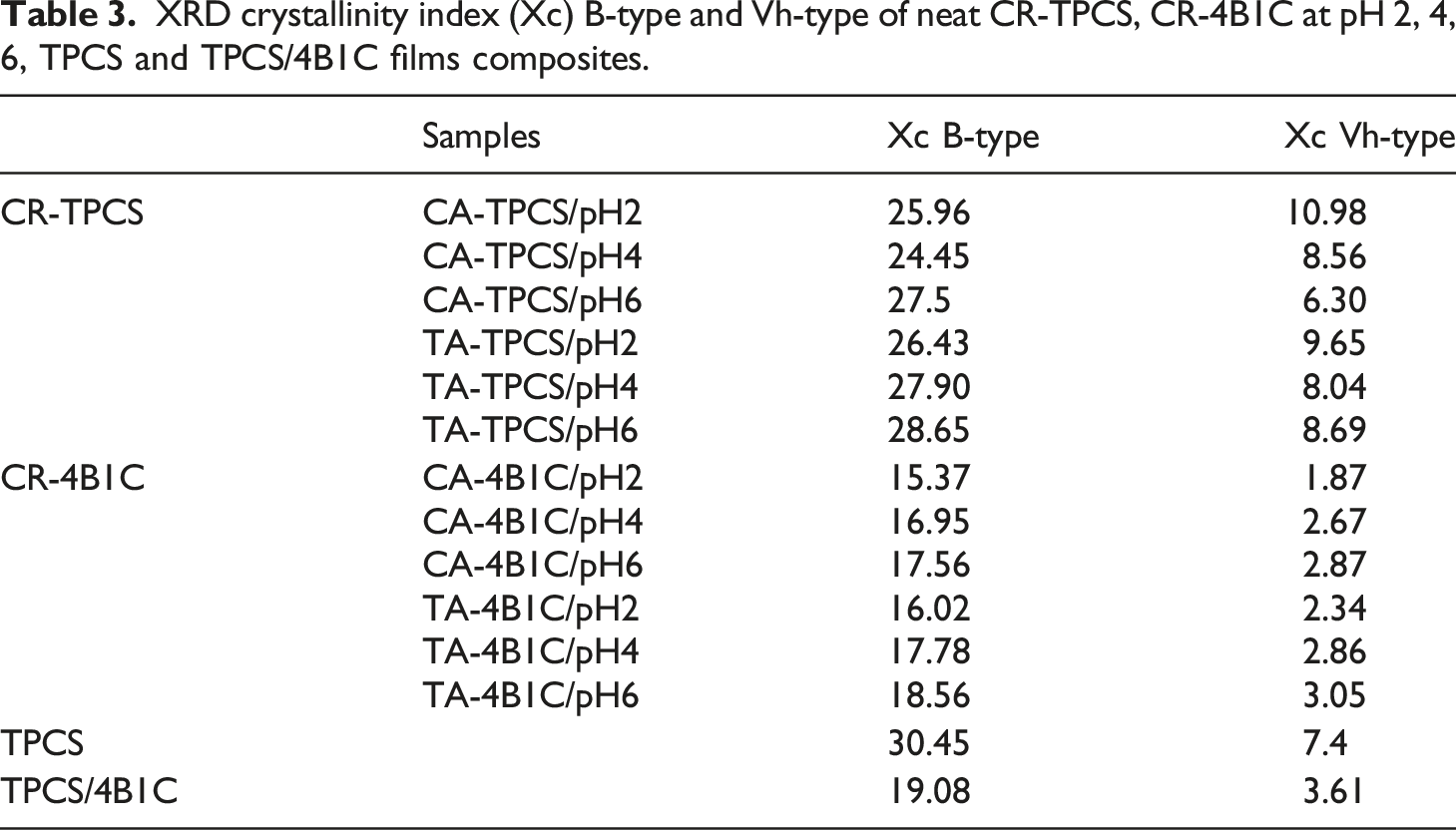
The crystalline structure of CR-TPCS and CR-4B1C was studied by XRD analysis. Figure 1(a) displays the crystalline structure of CR-TPCS, CR-4B1C, TPCS, and TPCS/4B1C films. The crystalline percentage of the CR-TPCS and CR-4B1C films was lower than the pure TPCS and TPCS/4B1C films indicating the retrogradation was effectively supressed by adding the carboxylic acid, as shown in Table 3. The lower crystallinity percentage showed that the carboxylic acid could further enhanced the TPCS hybrid biocomposites films stability during storage. Besides, the incorporation of carboxylic acid does not create a new crystalline peak in the XRD diagram showing the cross-linking reaction in the TPCS and the adding components does not alter the main TPCS structure. Carboxylic acid is expected to interact with the TPCS as internal plasticizer or external plasticizer by forming physical or chemical bonding between the TPCS chain, plasticizer, and nanocellulose.21,42,45 XRD crystallinity of CR-TPCS and CR-4B1C at (a) pH2 with TPCS and TPCS/4B1C, (b) pH 4 and (c) pH6. XRD crystallinity index (Xc) B-type and Vh-type of neat CR-TPCS, CR-4B1C at pH 2, 4, 6, TPCS and TPCS/4B1C films composites.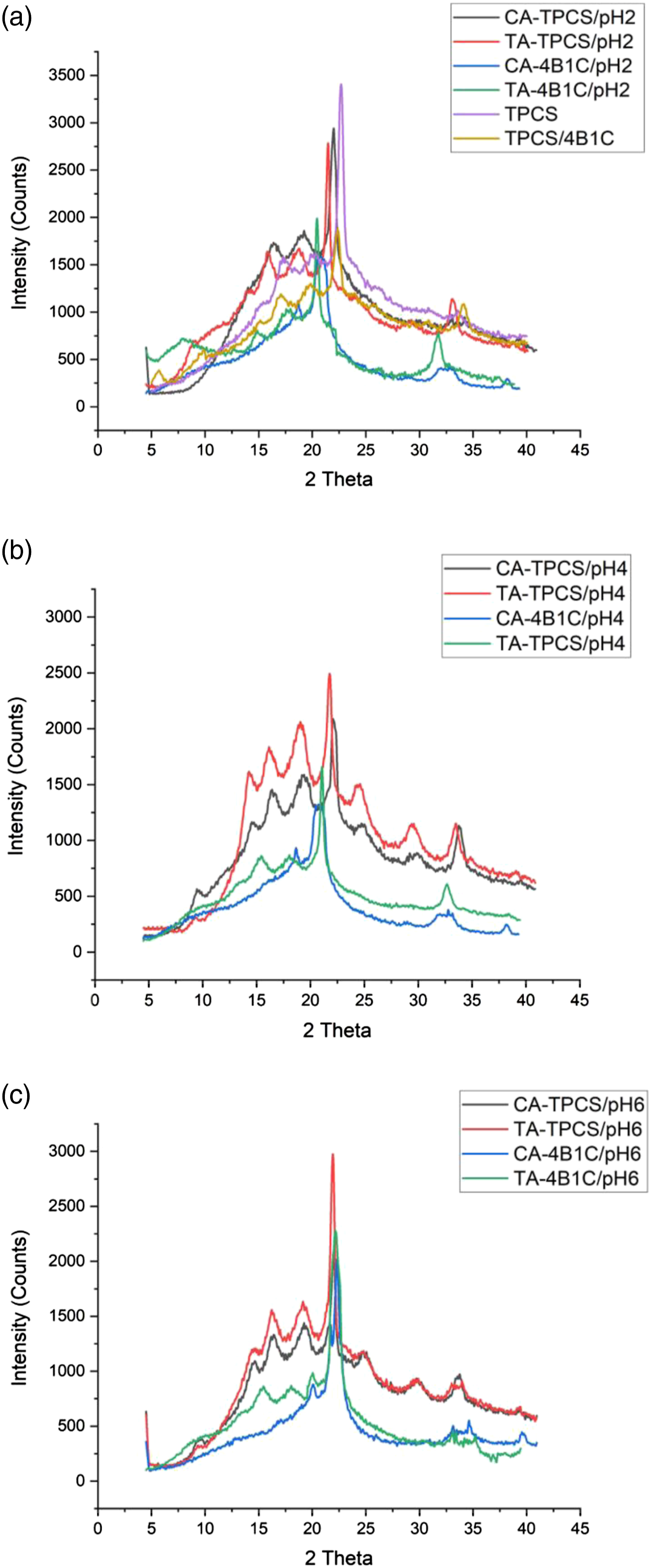
As reported in the literature, the carboxylic acid can act as internal plasticizer form covalent bonding with the TPCS chain through mono-esterification without cross-linking the intermolecular TPCS chains. The mono-esterification formed starch citrate in the TPCS chain, which acts as the internal plasticizer and interrupts the inter and intra-molecular hydrogen bond of the TPCS chain, as shown in Figure 2. The starch citrate can effectively prevent the close interaction between the TPCS chain and increase the free volume in the TPCS matrix. Meanwhile, the carboxylic acid that was not covalently bonded with the TPCS structure also acted as an external plasticizer, co-plasticization with glycerol to break down the granule structure by diffusing into the starch network. The presence of carboxylic acid between the TPCS matrix acted like a lubricant to facilitate the sliding of the TPCS chain during the straining and promote higher elongation at break.46,47 The mono-esterification of citric acid and TPCS chain and starch citrate structure.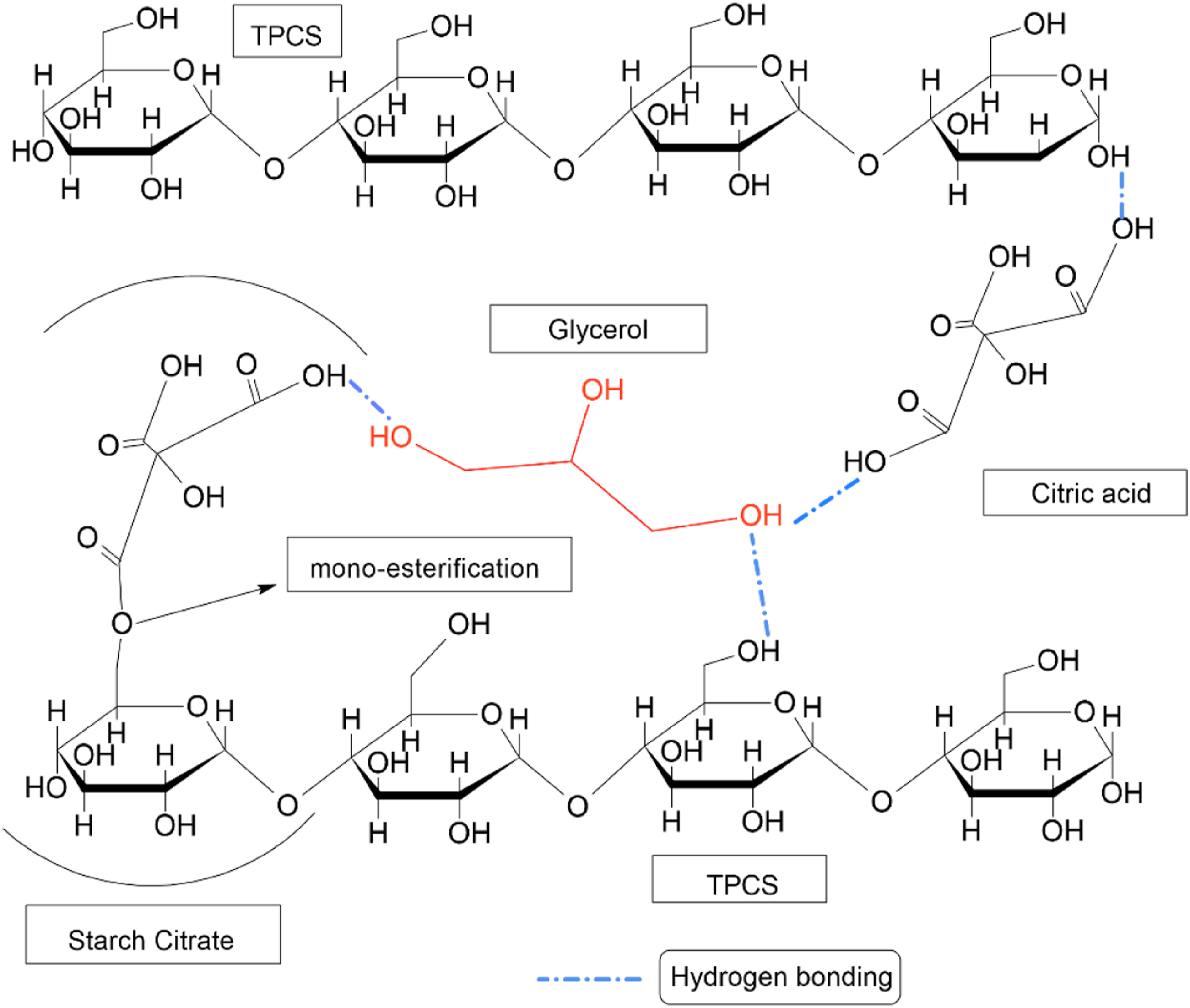
Based on the XRD result, the crystalline structure of TPCS was form with the combination two crystalline structure: type B and type Vh which was related to the double helix and single helix (amylose) crystalline structure, respectively in TPCS matrix. For CA-TPCS/pH2 and TA-TPCS/pH2, both films have a higher Vh crystallinity percentage than the CR-TPCS films at pH4 and pH6. All the CR-TPCS films were produced from the same batch of corn starch; therefore, the increase of the single helix crystalline structure in the CR-TPCS films was attributed to the acid hydrolysis properties of carboxylic acid, which hydrolysed the low molecular weight amylopectin chain into the short and linear amylose chain. The hydrolysed amylopectin chain behaves like the linear amylose chain, promoting stable single helix crystalline formation during drying. The same observation was also found in the study of Javidi et al. 21 where they found that the TPCS chain liberated from the amylopectin has a high potential to recrystallize into a single helix helical structure during the post-processing stage. Besides that, they also concluded that adding the carboxylic acids could effectively reduce the retrogradation in the corn starch films due to the cross-linking reaction between the TPCS chains.
The crystallinity percentage of the CR-TPCS films decreased as the pH increased to pH4 and decreased as the pH values increased to pH6. The different crystallinity percentages obtained in Table 3 suggesting different TPCS crystalline structure was expected by varying the pH values with the same amount of carboxylic acid. CA-TPCS/pH6 (33.8%) and TA-TPCS/pH6 (37.34%) showed the highest crystallinity percentage in their respective pH series. As the esterification reaction was catalysed in the acidic medium, both films were expected to have the lowest chemically cross-linked density compared to other films and displayed lower resistance toward retrogradation.
Meanwhile, the CA-TPCS/pH2 (36.94%) and TA-TPCS/pH2 (35.98%) have a higher crystalline percentage than CA-TPCS/pH4 (33.01%) and TA-TPCS/pH4 (35.01%), respectively. By analysing the films’ crystalline peaks for pH2 and pH4, the Vh peak of films at pH 2 exhibited much higher intensity than those at pH4. The higher Vh peak showed that the increase of single helix crystalline structure in the films for pH2 is higher than the pH4 due to the hydrolysis reaction of the TPCS chains. Meanwhile, the hydrolysis of the amylopectin chain results in a more open structure of the amylopectin, which promotes higher glycerol diffusing into the structure of amylopectin. The higher glycerol in the amylopectin promotes the cross-linked reaction between carboxylic acid with glycerol rather than the TPCS chain and reduce the cross-linked density between the TPCS chain as shown in Figure 3. The lower cross-linked density at pH2 promotes a higher amylose retrogradation rate and results in a higher crystalline percentage. The esterification of citric acid and glycerol in the open structure of TPCS chains.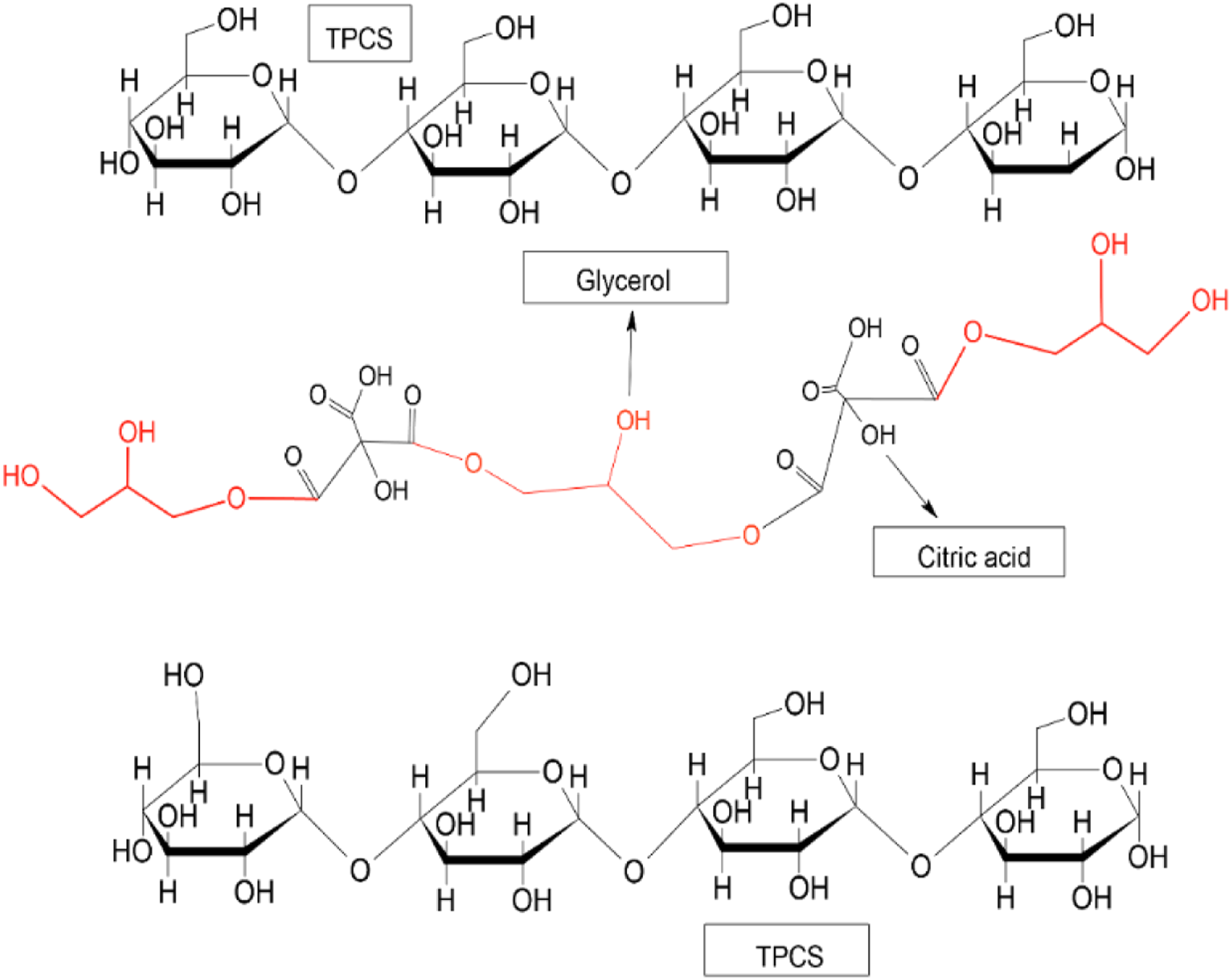
On the other hand, for the CR-4B1C films, a constant or slight alteration of Vh crystalline structure occurred as the pH increased from 2 to 4, associated the hydrolysis process was inhibited with the incorporation of the hybrid nanofiller. The increase of the Vh crystalline percentage with the increase in pH was not found in CR-4B1C. As expected, the CR-4B1C films at pH6 have the highest crystalline percentage among the CR-4B1C films due to the lowest cross-linked density in the film matrix. Meanwhile, CR-4B1C films exhibited the lowest crystalline percentage at pH2, and the crystalline percentage experienced a slight increase as the pH increased to 4. The slight increase in crystalline percentage may contribute to lower cross-linked density formed in the matrix as the pH values increase.
Thermal properties of the CR-TPCS and CR-4B1C films study by DSC
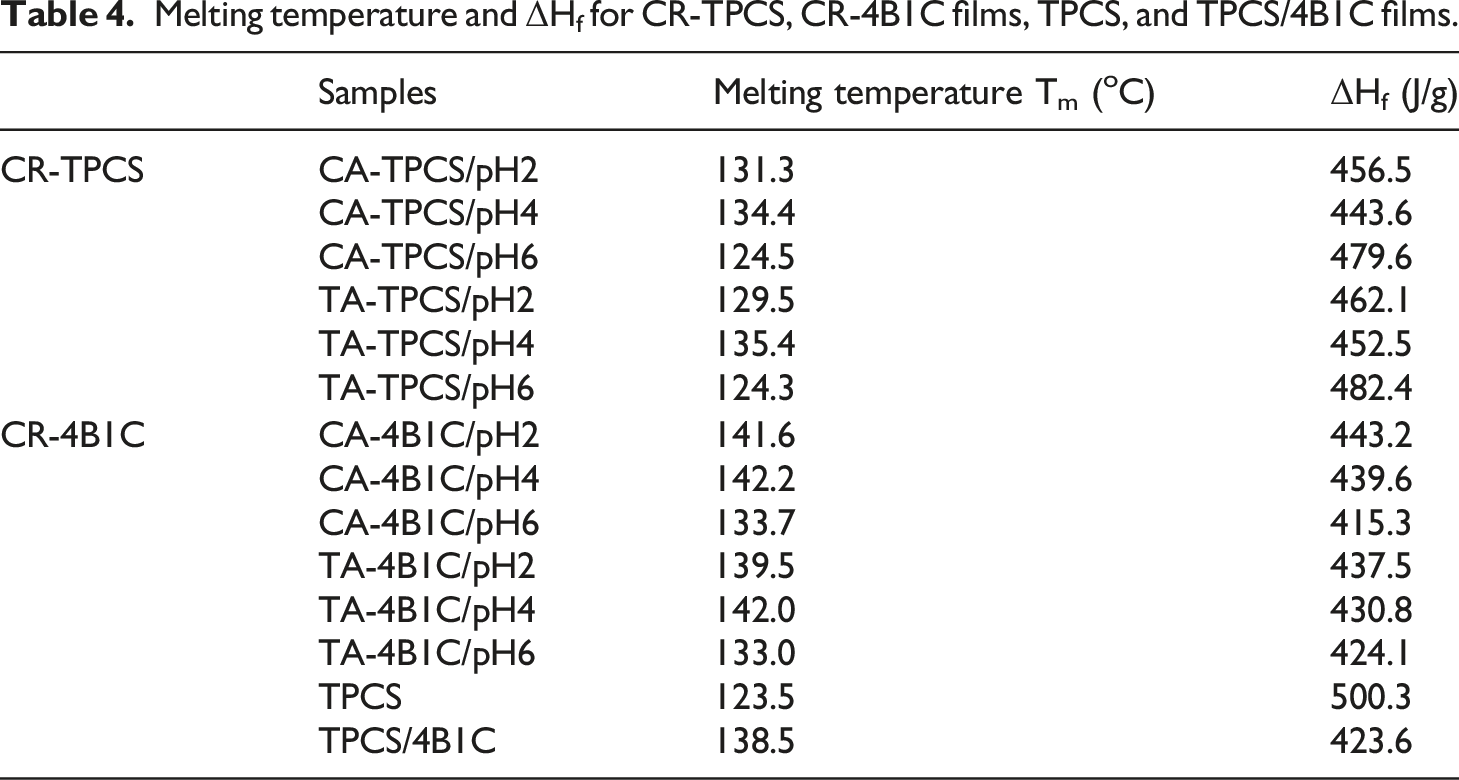
Melting temperature and ΔHf for CR-TPCS, CR-4B1C films, TPCS, and TPCS/4B1C films.
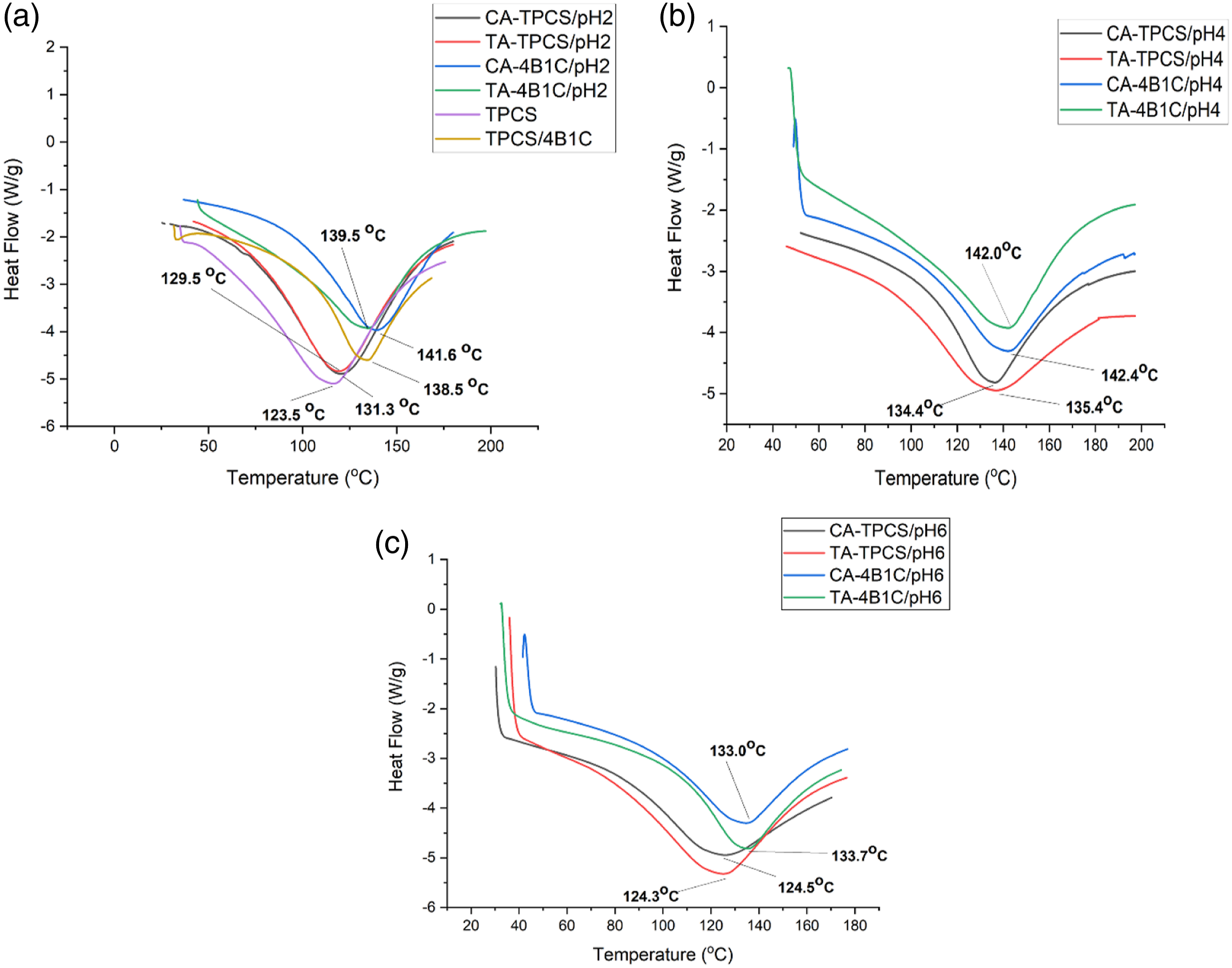
DSC heating curves for CR-TPCS, CR-4B1C films at (a) pH2 with TPCS and TPCS/4B1C films, (b) pH 4, and (c) pH6.
Meanwhile, the CA-TPCS films have narrower peak broadness and lower enthalpy of fusion compared to the TA-TPCS films in pH2, 4, 6, indicating citric acid has a higher efficiency than tartaric acid in reducing the retrogradation phenomenon. Generally, CA-TPCS and TA-TPCS films exhibited a decrease of ΔHf as the pH values increased to pH4, and as pH values increased to pH6, the CA-TPCS/pH6 and TA-TPCS/pH6 exhibited an increased broadness of melting peak. The broader melting peaks showed that the higher chain mobility due to the low cross-linked density and the formation of a small, unstable, and unsymmetrical double helix crystalline structure. The low cross-linking density of CA-TPCS/pH6 and TA-TPCS/pH6 also exhibited the lowest enthalpy of fusion in the CR-TPCS films series. The higher ΔHf exhibited by CR-TPCS films at pH2 and 4 indicates that cross-linked can improve the stability of the intercrystallite amorphous structure by forming a more compact structure. 48 Therefore, higher energy was required to break down the crystalline structure in the films. Besides that, the broader melting peak of CA-TPCS/pH6 and TA-TPCS/pH6 may also contribute by melting the residual carboxylic acid in the TPCS matrix. 49 CA-TPCS/pH2 and TA-TPCS/pH2 have a broader and higher ΔHf compared to CA-TPCS/pH4 and TA-TPCS/pH4, considered due to the increasing heterogeneity of the crystalline structure formed in the films. The higher heterogeneity crystalline structure observed in the films at pH2 may be due to the hydrolysis of amylopectin branches, which resulted in variation extent of the molecular weight, and the linear chain length contributed to a different degree of chain retrogradation in the matrix compared to other films. 47
Generally, the CR-4B1C films showed drastically increased melting temperature and a narrower melting peak than the CR-TPCS films. Meanwhile, CR-4B1C films also exhibited an increased melting temperature compared to TPCS/4B1C films. The increase of the melting parameter indicating the incorporation of the carboxylic acid can further improve the thermal stability of films and reduce the retrogradation of the TPCS chain.48,50 CA-4B1C/pH6 (133.7°C) and TA/4B1C-pH6 (133.0°C) exhibited a slight decrease in melting temperature compared to TPCS/4B1C (138.5°C). The slightly decreased melting temperature indicating the size and crystalline distribution structure of films at pH6 do not experience significant alterations, showing citric acid, and tartaric acid is a suitable plasticizer to co-plasticization with glycerol in TPCS films.
The ΔHf was an indicator to interpret the melting behavior of the crystalline structure of the TPCS chain. However, at acidic medium, pH2 and pH4, CA-4B1C and TA-4B1C films exhibited higher enthalpy of fusion compared to TPCS/4B1C films. As discussed in the XRD analysis, CA-4B1C and TA-4B1C films at pH2 and 4 have lower crystalline percentages than TPCS/4B1C films. This showed that increase in the ΔHf is not dependent on the crystalline structure formed in the matrix but attributed more toward the cross-linked phenomenon in the films. The increase of the ΔHf was due to a more compact structure formed in the matrix, which reduces the mobility of the amorphous region adjacent to the crystalline structure, and higher thermal energy was required to melt the crystalline structure. 51
The chemical interaction in CR-TPCS and CR-4B1C study through the FTIR
FTIR spectra for the CR-TPCS films and CR-4B1C films at pH 2, 4, and 6 were plotted in Figures 5(a), 6(a) and (b), respectively. The reaction between carboxylic acid and the TPCS films was studied through the peak assigned to the ester carbonyl (C=O) stretching vibrations bond, and the hydroxyl (OH) stretching bond was focused on for interpretation. The fingerprint peak for TPCS films was assigned between 800–1200 cm−1, representing the C-O bond stretching in the anhydro glucose ring vibration. Meanwhile, the 3300–3400 cm−1 corresponds to the O-H stretching bond related to the hydrogen bonding in the films. The other peak, such as 1600–1640 cm−1 assigned as the water absorbed, and the peak at 1300–1320 cm−1 and 2900–2930 cm−1 represent the C-H bond bending and stretching, respectively. FTIR Spectrum of CR-TPCS and CR-4B1C films at pH2 (a) 4000–500, (b) 1800–1000 cm−1. FTIR Spectrum of CR-TPCS and CR-4B1C films at pH4 (a) 4000–500 cm−1, (b) 1800–1000 cm−1.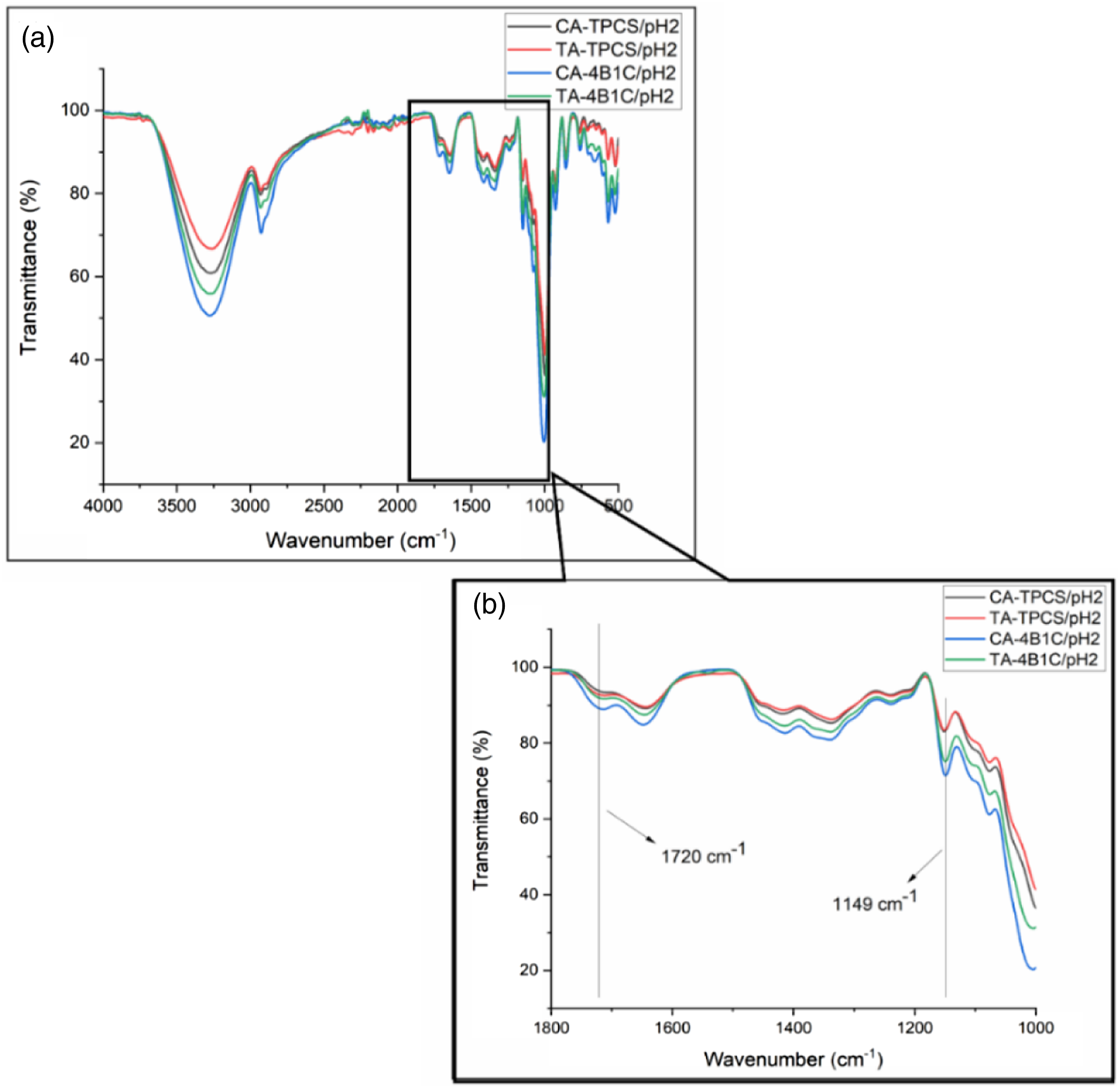
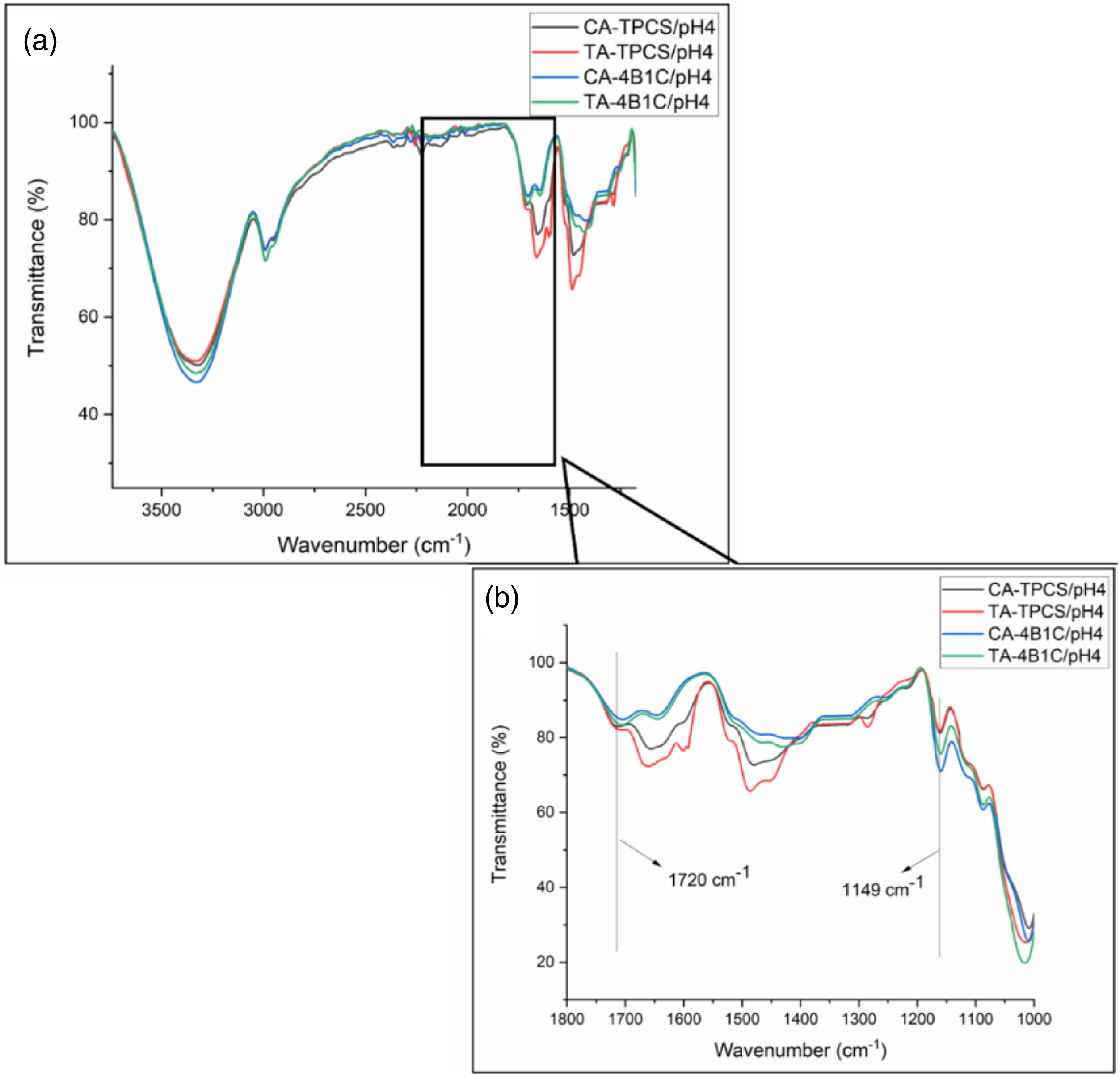
The previous study showed that the peak at 1700–1730 cm−1 could be attributed to the ester carbonyl bond linkage between the carboxylic and hydroxyl groups. This peak in the FTIR spectrum indicates an esterification reaction between TPCS, filler, and plasticizer with carboxylic acid. Meanwhile, the study of Castro-Cabado et al. showed that the unreacted and free carboxyl group would form a shoulder at the 1700–1730 cm−1 peak.
42
All the CR-TPCS and CR-4B1C films at pH6 exhibited a shoulder peak at the region 1700–1730 cm−1 as shown in Figure 7(b), indicating less crosslinked reaction was expected. The degree of esterification could affect the hydrogen bonding in the TPCS matrix, where the esterification reaction reduced the hydrogen bonding site in the films. The reduction of the hydrogen bonding result will minimize the intensity of the OH stretching band (3300–3500 cm−1). Therefore, the ratio between the C=O stretching band (FCO) intensity and the OH stretching band (FOH) intensity can be used as the indicator to study the progress of the esterification reaction in the films. The calculated ratio of FCO/FOH of CR-TPCS films and CR-4B1C films was tabulated in Table 5. FTIR Spectrum of CR-TPCS and CR-4B1C films at pH2 (a) 4000–500 cm−1 (b) 1800–1000 cm−1. Ratio of FCO/FOH and transmittance (%) at 1149 cm−1 for neat CR-TPCS, CR-4B1C at pH 2, 4, 6.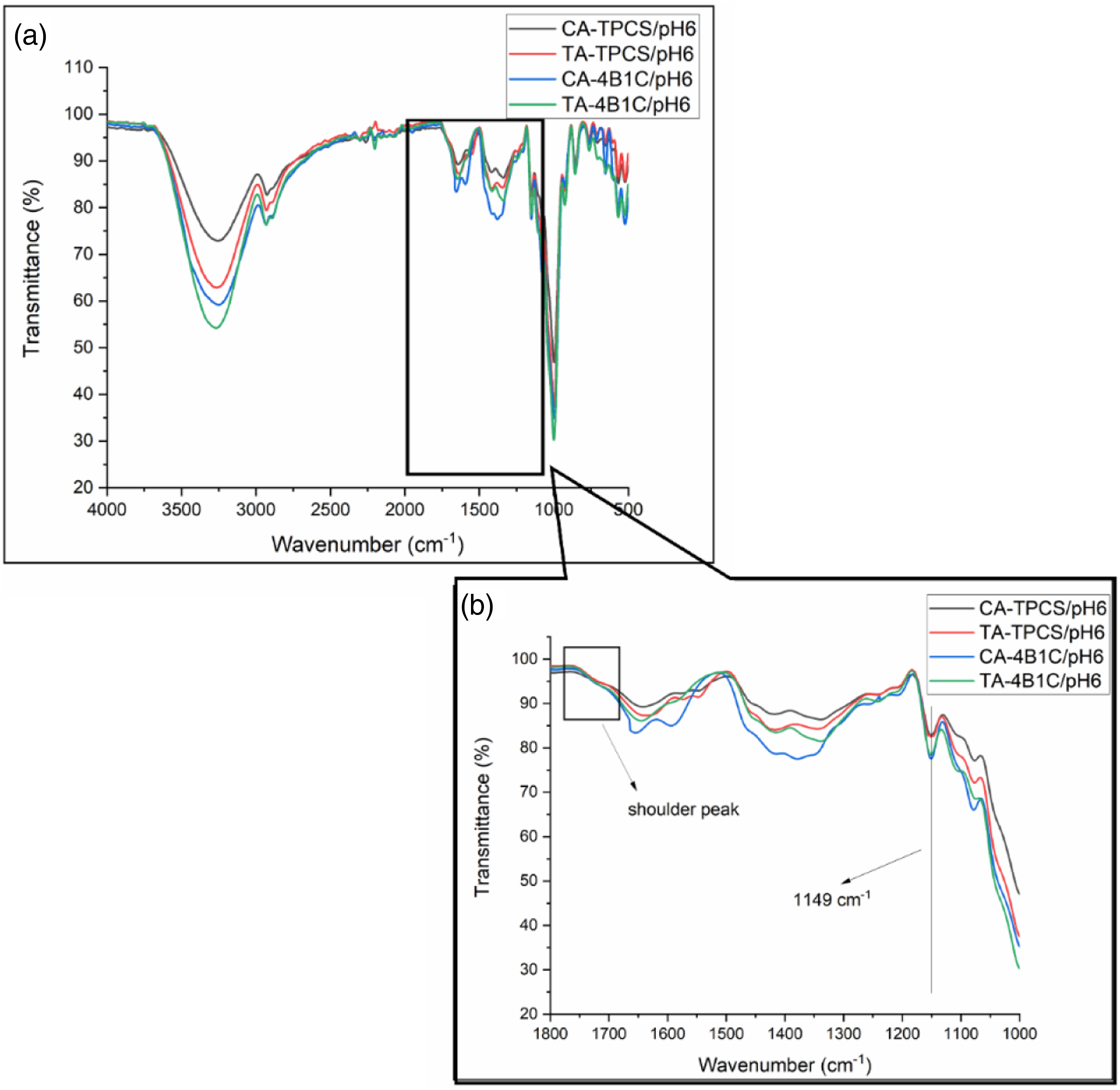
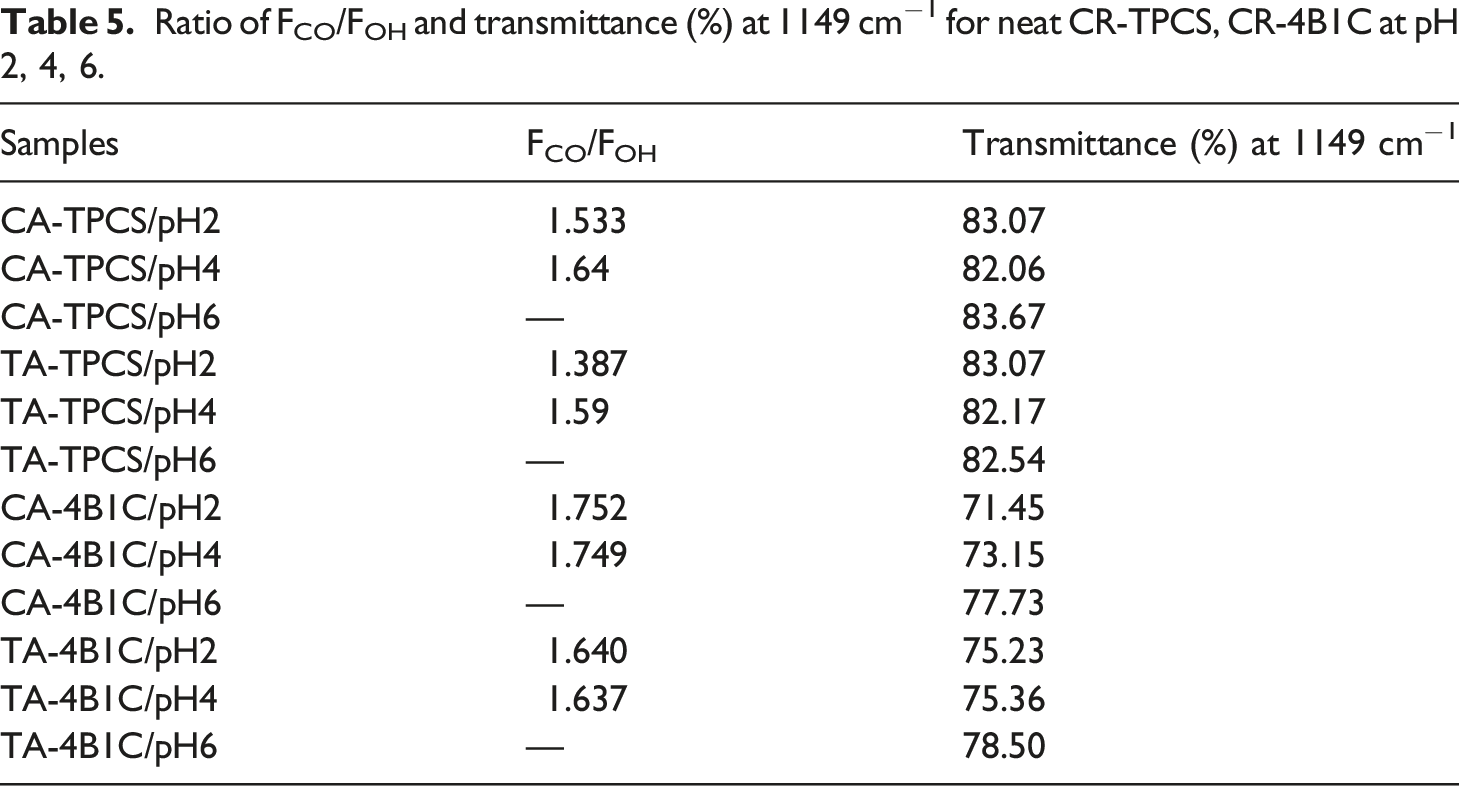
For CR-TPCS films, the ratio of FCO/FOH was increased from pH 2 to 4. Meanwhile, the ratio values were not calculated for pH6 as no defined peak was detected, indicating a less crosslinked reaction in the films. The increased ratio value from pH2 to pH4 shows that the esterification reaction in the films increased. Besides that, the films at pH6 also exhibited the highest intensity for the -OH group, which further verified that carboxylic acid prefers to form physical bonding in the TPCS matrix rather than cross-linking. Based on the esterification cross-linking catalyzed condition, the CR-TPCS films at the low pH should exhibit the highest value of FCO/FOH. However, films at pH 2 have the contradicting observation where they exhibited the second highest ratio of FCO/FOH was spotted. The possible explanation was due to the hydrolysis of the TPCS chain at pH2, causing the more short linear chain to be released from the branched structure and increasing the interaction of the starch chain. 46 These results showed that the acid hydrolysis reaction is more dominant than the cross-linking reaction at pH 2. This speculation was in line with Olsson et al., which showed that the hydrolysis of starch-carboxylic acid could be completely hindered as the solvent medium was maintained at pH ≥ 4. 19 Besides that, CA-TPCS films have a higher FCO/FOH than TA-TPCS films at pH4, indicating the cross-linked density is higher due to the higher CA reactivity than TA.
Interestingly for CR-4B1C films, the ratio of FCO/FOH was relatively constant from pH 2 to pH 4. The constant ratio of FCO/FOH shows that the acid hydrolysis of the carboxylic acid films in pure TPCS films does not occur in the CR-4B1C films. The constant ratio from pH2 to pH4 showed that the hybrid filler protects the TPCS chain from hydrolysis reaction. The study of Jeon and Nam showed that clay possesses the buffering capacity by allowing the H+ ions to be absorbed onto the interlayer negatively charged ions surface. The buffering capacity of clay slightly reduces the medium’s acidity and suppresses the films’ hydrolysis during the mixing process. 52 The absorption of H+ ions properties of clay at the low pH value also was found in different applications such as wastewater treatment 53 and modification of clay dispersion. 54
The FTIR spectra peak at 1149 cm−1 could be associated with the stretching vibration of C-O in the C-O-H group, which is always used as the reference band to study the amount of C-O in the matrix. The transmission percentage at peak 1149 cm−1 was recorded in Table 5. The intensity increased at 1149 cm−1, showing that the C-O group in the matrix was increased. Due to the homogenous distribution of the plasticizer and nanofiller with TPCS, the C-O group in the plasticizer or nanofiller was more likely to detect by FTIR, resulting in higher intensity of the 1149 cm−1 peaks. 25 The lower the transmittance percentage, the higher the intensity of the peak. Table 5 shows that the intensity of 1149 cm−1 CA-TPCS/pH2 composite films is the highest among the CR-4B1C films indicating an excellent distribution of citric acid with nanocellulose and plasticizer, resulting in higher peak intensity.
Fracture morphology of CR-TPCS and CR-4B1C with different pH values
The fracture morphology of CR-TPCS and CR-4B1C with different pH values and carboxylic acid is shown in Figures 8 and 9. For CA-TPCS and TA-TPCS film at pH2, the fracture surface film spotted some isolated and agglomerated matrix was observed (see Figure 8(a) and (b)). The agglomerated matrix formed on the fracture could be attributed to the hydrolyzed and fragmented crystalline structure, which acts as the stress concentration point and reduces the tensile properties, as discussed in the mechanical properties. The CA-TPCS/pH2 exhibited a 35-micron-sized agglomerated matrix in Figure 8(a), while TA-TPCS/pH2 exhibited a much smaller agglomerated matrix around 15-micron-sized in Figure 8(b). The more enormous, agglomerated structure formed in CA-TPCS/pH2 could be attributed to CA’s higher hydrolysis capacity as it contains three carboxyl groups compared to TA. Meanwhile, the higher crosslinking ability of CA also resulted in brittle fracture morphology observed in CA-TPCS/pH2 compared to TA-TPCS/pH2. The fracture surface of CR-TPCS films (a) CA-TPCS/pH2, (b) TA-TPCS/pH2, (c) CA-TPCS/pH4, (d) TA-TPCS/pH4, (e) CA-TPCS/pH6 and (f) TA-TPCS/pH6 at ×2000 magnification. The fracture surface of CR-4B1C films (a) CA-4B1C/pH2, (b) TA-4B1C/pH2, (c) CA-4B1C/pH4, (d) TA-4B1C/pH4, (e) CA-4B1C/pH6 and (f) TA-4B1C/pH6 at ×2000 magnification.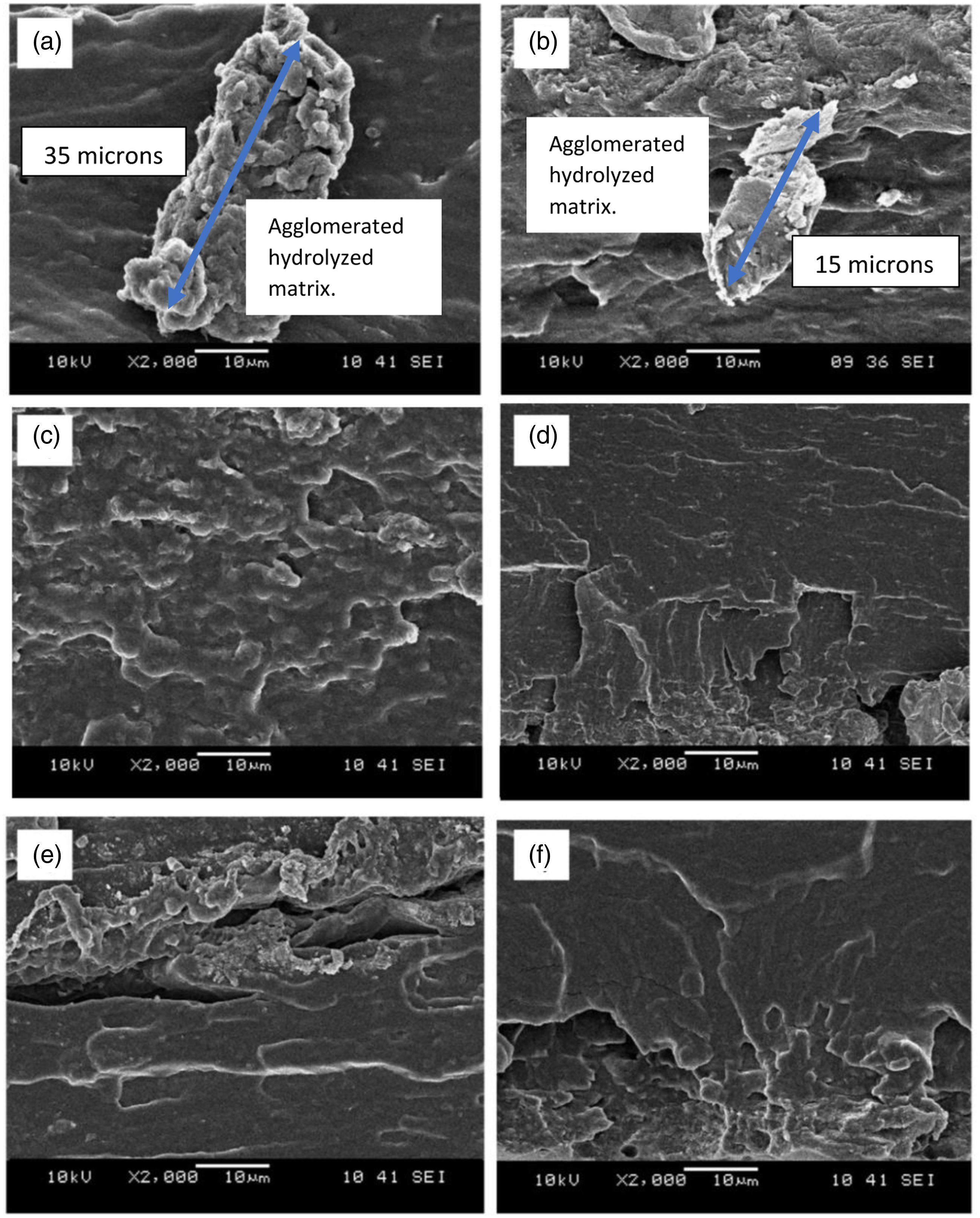
As the pH values increased to pH4, CA-TPCS/pH4 and TA-TPCS/pH4 exhibited a compact and smooth fracture surface in Figure 8(c) and (d), respectively, indicating the chemical cross-linked of carboxylic acid resulted in the brittle behaviour. However, comparing the surface's homogeneity, the CA-TPCS/pH4 exhibited a much higher homogenous fracture structure than the TA-TPCS/pH4 films. Meanwhile, for TA-TPCS/pH4, the smooth fracture surface was slowly transited to the rough fracture surface from the top of the films to the bottom. The rough fracture surface at the bottom of the films may be attributed to the accumulation of the unreacted TA in the matrix due to its less cross-linked reactivity than the CA. The unreacted TA in the matrix acted as a plasticizer and provided flexibility to the TPCS chain forming the rough fracture surface. As the pH increased to 6, CA-TPCS and TA-TPCS exhibited a ductile deformation area in Figure 8(e) and (f), respectively, indicating carboxylic acid co-plasticizing with glycerol could increase the ductility of pure TPCS films and promote higher plasticity for the films during elongation.
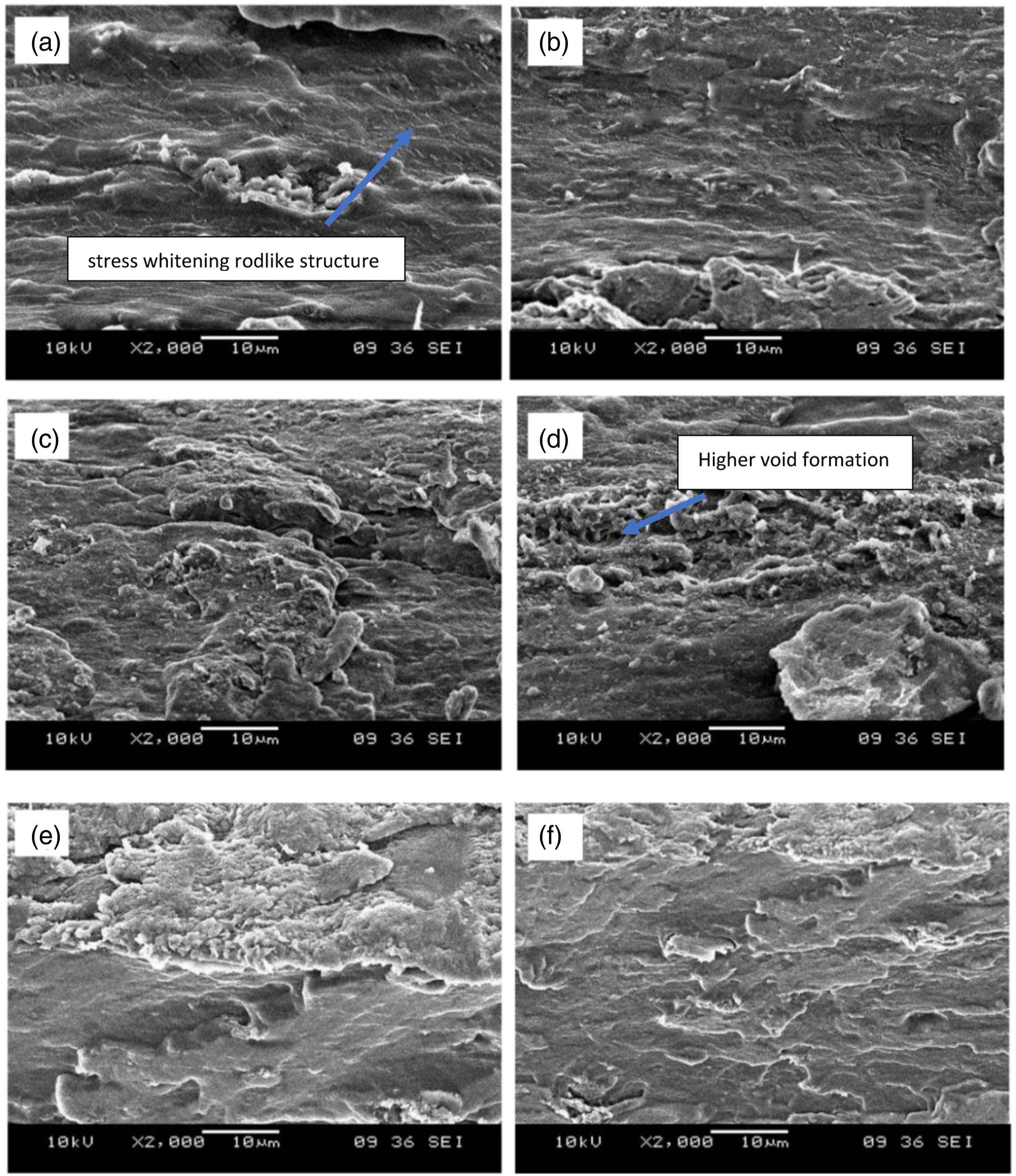
Besides that, for the CR-4B1C films, all the films display the ductile and crack shear zone on the fracture surface. The high roughness fracture surface shows that the crosslinking formed in the CR-4B1C films does not reduce the high strength and flexibility properties. For CA-4B1C/pH2 films (Figure 9(a)), the fracture surface of the films behaved like a sea wave, and a small, homogenous stress whitening rodlike structure was observed across the film. The crack-less stress whitening rodlike structure could be attributed to the formation of craze or shear ridges. The formation of the craze ridges on the fracture surface, indicating the localized deformation or yielding, was further enhanced before failure. The craze ridges formed like a large sink for the strain energy release and absorbed higher deformation energy leading to higher film toughness.
Meanwhile, the striation surface morphology was also observed, showing that the toughening mechanism triggered by the hybrid nanofillers was maintained and enhanced. The formation of a homogenous craze and the striation surface morphology indicates a more significant plastic deformation. However, this stress-whitening rodlike structure was not observed in the TA-4B1C/pH2 (Figure 9(b)), showing less enhancement effect by TA. The less efficiency enhancement of TA may be due to the lower cross-linked ability than CA, which results in lower cross-linked density.
Furthermore, the fracture surface of CA-4B1C films and TA-4B1C at pH4 and pH6 is shown in Figure 9(c)–(f), respectively. All the films exhibited a high roughness and plastic deformation fracture surface, showing that the hybrid nanofillers’ synergic effect is highly compatible with the carboxylic acid. Meanwhile, TA-4B1C/pH4 films (Figure 9(d)) showed higher void formation than CA-4B1C/pH4 films (Figure 9(c)). The higher compact structure of CA-4B1C/pH4 films may contribute to the higher reactivity of CA, which formed higher cross-linked with the TPCS chains and resulted in less void formation during deformation.
Solubility and swelling of CR-TPCS and CR-4B1C in DMSO
The swelling percentage (%), solubility percentage (%), and water vapor permeability of CR-TPCS and CR-4B1C at pH 2, pH 4, and pH 6.
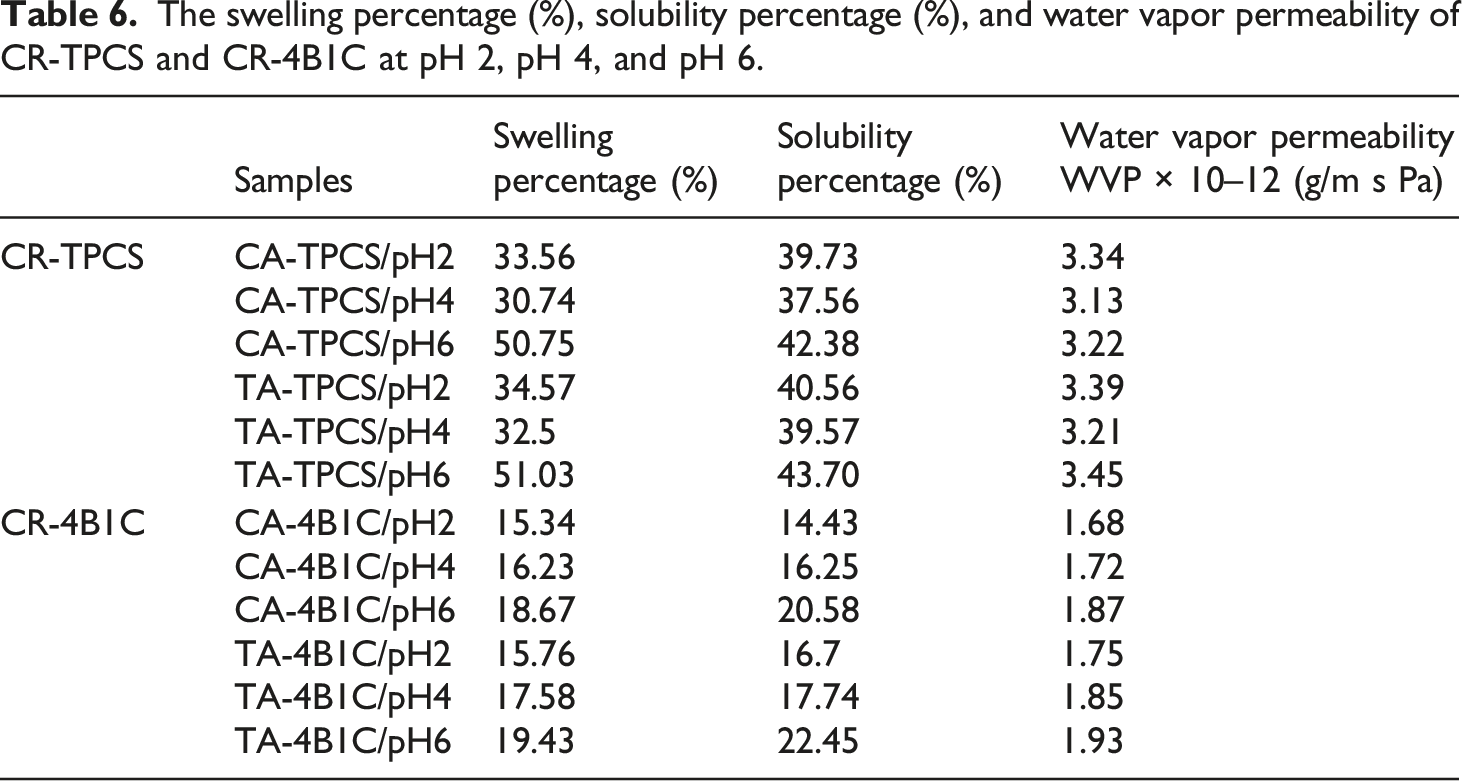
Apparently, the CR-TPCS at pH2 exhibited a higher swelling percentage and solubility than CR-TPCS at pH4, as observed in Table 6. The higher swelling and solubility percentage could be attributed to acid hydrolysis TPCS chain at low pH values. However, the amount of amylose in the TPCS matrix increased due to hydrolysis of the amylopectin chain should lower the swelling percentage as observed in the other amylose cross-linked films. However, the CR-TPCS/pH2 in our study showed that the increase in amylose structure resulted in a higher swelling percentage. The contradicted results may be explained by the significantly shorter and lower molecular weight of the linear amylose formed by the acid hydrolysis than the natural amylose-high films. 55 The low molecular structure of hydrolyzed amylose structure has a lower interaction with carboxylic acid, and it was more likely to form a single helix crystalline structure, as mentioned in the XRD result. The low molecular single helix crystalline amylose structure can easily leach out during the swelling test, resulting in higher solvent solubility than pH4.
The swelling properties are highly interrelated to the water solubility properties, which depend highly on the dense matrix structure and the cross-linked density. As the matrix structure is dense and highly cross-linked, the diffusion of the swelling agent was limited, and the solubility was delayed or inhibited. 56 The short branches in amylopectin were hydrolyzed in pH2, resulting in a less compacted structure than CR-TPCS films at pH4, contributing to a higher swelling percentage. Meanwhile, CA-TPCS/pH4 has a lower swelling percentage than TA-TPCS/pH4, indicating CA can form higher ester linkage density in the TPCS matrix and prevent the diffusion of the solvent into the TPCS matrix compared to TA.
Generally, the CR-4B1C films also displayed the same swelling trend as the CR-TPCS films, with a much lower swelling percentage. Meanwhile, the CR-4B1C films exhibited significantly lower solubility than the CR-TPCS films in all the pH values. The much lower solubility of the CR-4B1C films shows that the matrix components in the TPCS/4B1C, such as glycerol and nanocellulose, were chemically cross-linked with the TPCS chains through the carboxylic acid. The chemically cross-linked can effectively prevent the matrix components from diffusing out of the matrix. Besides that, the physical bonding between the TPCS chains and the adding components (nanocellulose, glycerol, and carboxylic acid) was more susceptible to the swelling mechanism, causing a much higher solubility. This explanation was in line with the swelling and solubility trend observed in CR-4B1C films at pH6, where the low films’ cross-linked density at pH6 has the highest swelling and solubility percentage in their respective film series.
The solubility percentage of the films is highly dependent on the swelling density and the number of added components or soluble parts leaching out from the films. All the films’ swelling percentage is directly proportional to the solubility percentage. The higher the swelling percentage, the higher the solubility percentage was expected. However, there is an interesting observation where CA/4B1C-pH2 exhibited almost the same swelling percentage compared to TA/4B1C-pH2 films, but it displayed a much lower solubility percentage, considering the same carboxylic acid content was incorporated into the films. The lowest solubility percentage exhibited by CA/4B1C-pH2 is highly suggestive that the matrix components in the films were highly chemically bonded with the TPCS chain compared to other CR-4B1C films. The strong chemical bond forming within the TPCS chain, and the matrix component drastically reduce the leaching of soluble parts or matrix components out of the films.
Water vapor permeability of CR-TPCS and CR-4B1C films
Table 6 presents the WVP of the CR-TPCS and CR-4B1C films. The WVP of the TPCS films can be reduced by forming the hydrophobic ester bond in the TPCS matrix and reducing the susceptibility of water to penetrate through the films. Many studies have reported that the incorporation of a carboxylic acid into the TPCS film could reduce the WVP of films17,44,57
CA-TPCS films have a lower WVP than the TA-TPCS films, indicating that higher ester bond linkage was formed in the matrix at pH 2 and 4. Water molecules were less likely to penetrate the hydrophobic ester structure than the hydroxyl group. The CA-TPCS/pH6 has the highest WVP compared to pH2 and pH4, showing that CA-TPCS/pH6 has the lowest ester linkage in the films, with a high TPCS chain mobility which is more favorable for the water molecules penetration. The physical bonding in the CA-TPCS/pH6 can be easily broken down as the film’s water content increases, leading to higher water permeability as the time storing increases. Meanwhile, CA-TPCS/pH4 exhibited the lowest WVP compared to CA-TPCS/pH2 and CA-TPCS/pH6 showing water molecules were more difficult to pass through the highly cross-linked and compact TPCS matrix. The denser chemically cross-linked amylopectin structure has less molecular movement and is less susceptible to water penetration. Besides that, the hydrolysis TPCS chain will increase the number of hydroxyl groups exposed to contact with water molecules and increase the WVP. 58
Meanwhile, for CR-4B1C films, the WVP experienced a decrease as the pH value increased. The different WVP trends observed in CR-4B1C films indicated that the hydrolysis of films was non-exists with the addition of the hybrid filler, and a different cross-linked structure was expected. The lowest WVP values were observed for the CA-4B1C/pH2 and TA-4B1C/pH2 films. The WVP of CR-4B1C films decreases with the increase of pH due to the lower cross-linked density, as proved in the FTIR spectra. Meanwhile, the TA-TPCS films have higher WVP than CA-TPCS films at pH 2 and 4. The CA-TPCS/pH2 films can form a much higher compact structure and low free matrix volume, reducing water penetration.
Degree of substitution of the hydroxyl group of the TPCS structure
Degree of substitution and di-esterification of CR-TPCS and CR-4B1C.
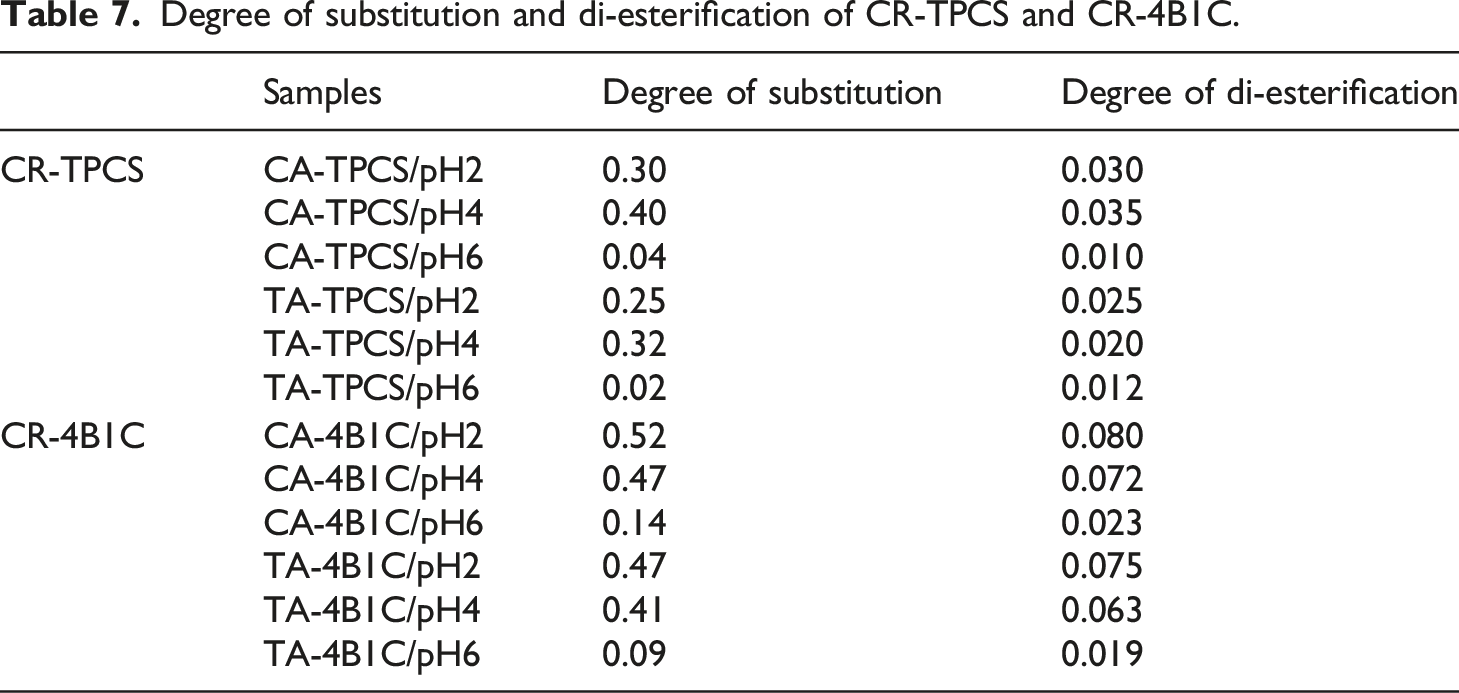
The DS value for CA-TPCS and TA-TPCS films at pH 6 showed the least DS at 0.04 and 0.02, respectively. The extremely low DS indicated that the chemically cross-linked reaction was not happening in the TPCS matrix. This showed that the esterification reaction between the carboxyl and hydroxyl groups could only happen in the acidic medium even though post-curing was carried out. Meanwhile, the CA-TPCS/pH4 films possess the highest DS (0.40) for CR-TPCS films which is much higher than CA-TPCS/pH2 films (0.30) even though CA-TPCS/pH2 films were post-cured in a much more acidic medium. The lower DS showed that acid hydrolysis happens in the CA-TPCS/pH2 and in the more open structure of amylopectin, which does not favor the chemical interaction between the CA and the TPCS chain.
The overall DS of CR-4B1C films is much higher than the CR-TPCS films, indicating that the film’s hybrid fillers promote the chemical interaction between the citric acid and the TPCS chain. The higher DS of CR-4B1C may be due to the evenly spread of glycerol content in the TPCS matrix, as explained in objective 3. According to various studies24,59 and reviews, 60 they showed that carboxylic acid is more likely to react with glycerol first due to the small molecular size and consists of two high reactive primary hydroxyl group in their structure.
For CR-TPCS films, the heterogenous accumulation of glycerol was observed in the amylopectin region. The high accumulation of glycerol in the amylopectin region caused all carboxylic acid to mainly form the chemical linkage with the glycerol and reduce the chemical interaction with the TPCS chain. Meanwhile, for CR-4B1C films, the glycerol in the films was homogenously distributed in the matrix, where the carboxylic acid only partially reacted with glycerol, and the remaining carboxyl group in the carboxylic could form a chemical linkage with the TPCS chain or nanocellulose. The DS for CR-TPCS films decreased as the pH values increased. CA-4B1C/pH2 films possess the highest degree of DS, indicating that the film's acidic medium does not cause acid hydrolysis of the TPCS chains. For CR-4B1C films, the DS was less affected by the low pH values, and the slight improvement of the DS could be based on the reactivity of the carboxylic acid.
Degree of di-esterification according to complexometric titration with copper (II)- sulfate
The degree of di-esterified in the CR-TPCS and CR-4B1C films is shown in Table 7. The determined values for di-esterification in all the films varied across the increase of pH values and the different types of carboxylic acid. Generally, the degree of di-ester content CR-TPCS and CR-4B1C at pH6 is relatively lower than the films at pH2 and 4. This result was in line with the study of Lipatova et al. and Olsson et al., where the esterification reaction was catalyzed at a low pH value.19,45 CA-TPCS films have higher di-esterification than TA-TPCS films due to the different esterification mechanisms during the curing process. When CA is heated and dehydrated, it forms anhydride, which has a lower activation energy to form ester linkage than the TA and promotes higher diester linkage in the films.
Besides that, the CA-TPCS/pH2 exhibited the same degree of di-esterification compared with CA-TPCS/pH4, indicating that the citric acid has the same activation energy for the esterification reaction in either pH4 or pH2. However, there is no clear indication of the location of the di-esterification reaction reacted in the TPCS matrix. Carboxylic acid could be forming the di-esterification either with one of the components, such as TPCS chain, glycerol, and nanocellulose, or reacted with more than one of the components. Therefore, the previous discussion was included to clarify the interaction between citric acid and the components in the TPCS matrix, given the same degree of di-esterification with the varying pH values. The CA-TPCS/pH2 exhibited high swelling, solubility percentage, and lower degree of substitution than CA-TPCS/pH4. The citric acid in CA-TPCS/pH2 was proposed to have a higher preference to form a chemical linkage with glycerol rather than the TPCS matrix. According to the study of Holser et al., citric acid prefers to interact with glycerol due to the higher activation primary hydroxyl group of glycerol compared to the starch chain. 24 Meanwhile, the hydrolysis of the amylopectin chain happens at CA-TPCS/pH2 and creates a more open structure of amylopectin, which provides a higher interaction volume for citric acid to interact with glycerol instead of with the TPCS chain freely.
Many studies have reported the possible interaction between nanocellulose and citric acid through the ester linkage. Meanwhile, for CR-4B1C films, the di-esterification degree of CR-4B1C films is much higher than the CR-TPCS films indicating di-esterification in CR-4B1C films involving the nanocellulose was expected. CA-4B1C/pH2 films exhibited the highest diesterification degree compared to other films. TA-4B1C/pH2 exhibited a lower di-esterification degree than CA-4B1C/pH2, indicating that tartaric acid was more likely to form monoester linkage than diester cross-linking. Besides that, CA-4B1C/pH2 films exhibited the highest degree of substitution and the lowest solubility, suggesting that citric acid is more likely to form ester linkage with the TPCS chain and connect the glycerol and nanocellulose in the matrix. The high degree of di-esterification of CA-4B1C/pH2 films showed that citric acid act as the bridge by forming a chemical bond between TPCS and the glycerol or nanocellulose.
Proposed possible structure of CR-4B1C films
As observed from the other studies, incorporating cross-linked between the biopolymer could further enhance the films’ tensile properties. However, the films’ flexibility and mobility will be reduced as cross-linking forms between the biopolymer chain. As reported in the last objective, high-strength and high-toughness films were successfully produced by incorporating the citric acid at ph2 with the hybrid nanofiller in the TPCS matrix.
From the past studies, the elongation at break of TPCS films with high cross-linked density was expected to experience a decrease due to the lower mobility of the TPCS chain. Surprisingly, the incorporation of citric acid in pH2 not only improved the tensile properties of the films, but CA-4B1C/pH2 also exhibited a 20% improvement in the elongation at break. This shows that the synergistic effect of the nanoclay and nanocellulose in the TPCS matrix as reported in our previous study (in which the nanoclay responsibles for the increase of strength and the nanocellulose responsibles for the film’s flexibility) is still exists, even after the crosslinking procedure. 7
The unexpected result may be explained by concluding all the discussion above, and the proposed possible structure of CA-4B1C/pH2 films is shown in Figure 10. CA-4B1C/pH2 displayed the lowest swelling and solubility percentage compared to other CR-4B1C films. The highly stable TPCS network structure of CA-4B1C/pH2 could be attributed to the highest cross-linked density in the matrix and well cross-linked structure formed between the citric acid, glycerol, and nanocellulose to the TPCS chain. The interlinking of glycerol and nanocellulose to the TPCS chain through the citric acid was further verified by the FTIR result and a higher degree of substitutional than others. The well-cross-linked structure also contributes to the low crystalline percentage shown in the XRD result. The proposed molecular structure of CA-4B1C/pH2 with high flexibility and strength.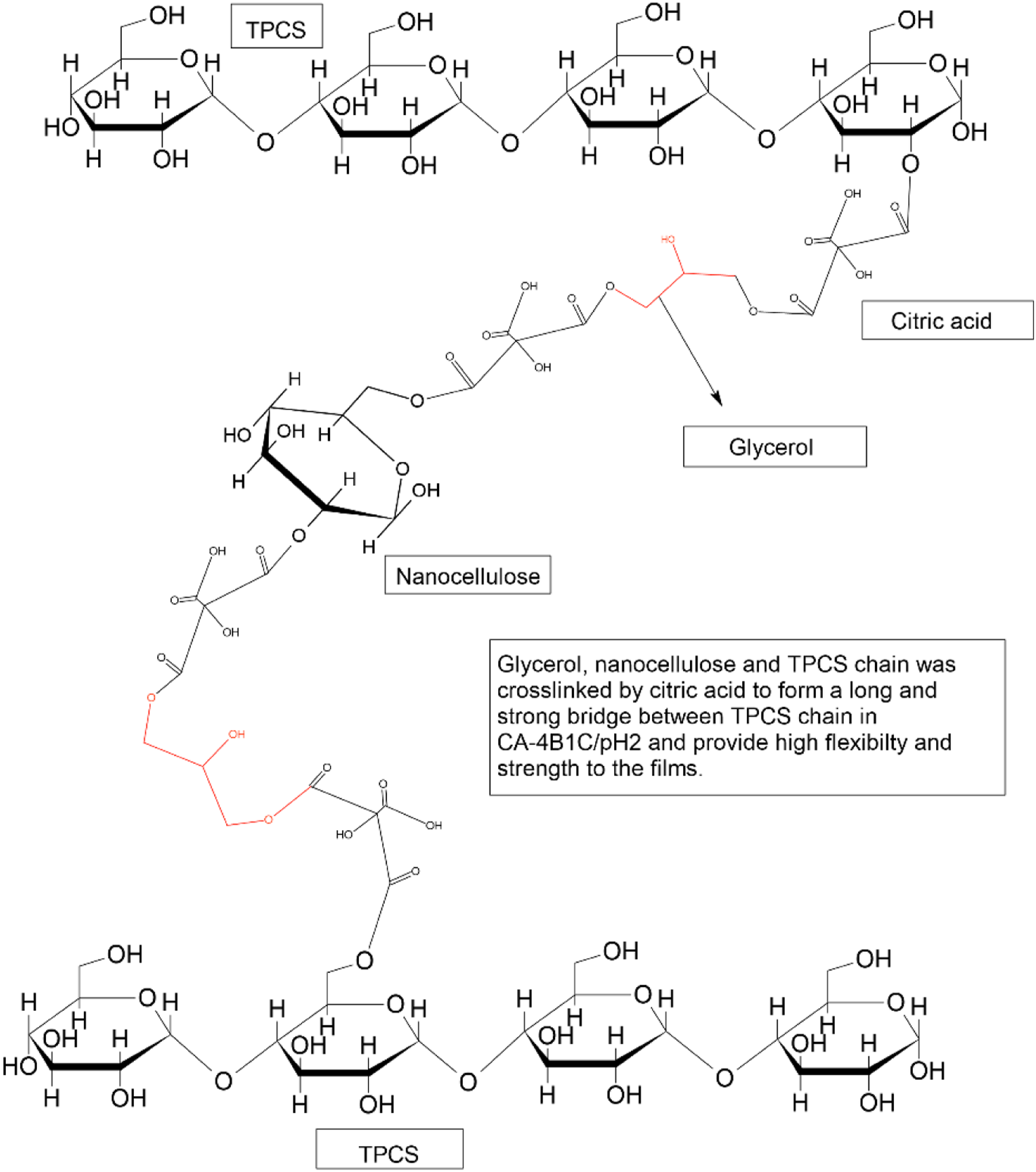
Besides that, the improvement of elongation at break showed that the cross-linking between citric acid, glycerol, and nanocellulose provided a much higher free volume and dynamic during the straining of the films compared to another cross-linked film. For most of the cross-linked films, the mobility of the chains is highly restricted due to the limited matrix volume. Many studies showed that the flexibility of the cross-linked films could be improved by increasing the length of the cross-linked linkage.33–36,38 Therefore, the proposed possible structure of CA-4B1C/pH2 films is shown in Figure 10. Figure 10 shows a long-cross-linked linkage involving citric acid, glycerol, and nanocellulose formed between the TPCS chain. The long-crosslinked linkage provided a more extensive volume for the mobility of the TPCS chain during strain than other cross-linked films. The more significant volume provides the free volume for the chain alignment and reduces the film’s stress build-up. Meanwhile, the chemical cross-linking of the components in CA-4B1C/pH2 provides a stronger load transfer mechanism and further improves the mechanical properties compared to TPCS/4B1C films. This proposed structure explained the high strength and flexibility of the CA-4B1C/pH2 and justified all the analysis explanations.
Conclusion
In this study, the hybrid nanofillers were incorporated into the crosslinked TPCS film to enhance the strength and reduce the stiffness of the CR-TPCS film through the developed toughening mechanism. The tensile properties of the CR-4B1C films are superior compared to the CR-TPCS films. Particularly, the CR-4B1C films exhibit higher tensile strength and elongation at break than the TPCS/4B1C films. The improvement of both tensile strength and elongation at break is rarely reported as the crosslinked films always experience an increase in tensile strength with drastic reduction in film’s flexibility. The exceptional improvement of the tensile properties showed that the toughening mechanism of the hybrid nanofillers was successfully incorporated into the CR-TPCS films. Interestingly, the toughening mechanism was further magnified through the crosslinked reaction and this has been detailed out in this article. Citric acid (CA) can induce higher crosslinked density than the tartaric acid (TA) due to a higher degree of substitution and di-esterification. This leads to the highest tensile strength achievement. Meanwhile, incorporating nano clay into the CR-4B1C films could also act as the buffer solution medium to diminish the acid hydrolyzing effect of the CA at low pH values. The hydrolysed structure of the TPCS was diminished in CR-4B1C films through the observation of SEM. Taken into account all the analysis and results obtained, a “bridge extension” formed between the TPCS chains, which crosslinked the plasticizer, nanocellulose, and TPCS chains through CA, was proposed in this study. The long “bridge extension” between the TPCS chains provided the high flexibility and more effective load transfer mechanism in the TPCS matrix during tensile loading. The crosslinked hybrid TPCS biocomposite films offered a novel perspective where the high stiffness/brittle crosslinked films could be overcame without went through the heavy and toxic chemical treatment and by only adding green hybrid nanofillers. This crosslinked hybrid TPCS biocomposite film can be a promising green film material to replace the fossil fuel-based plastic in packaging applications.
Footnotes
Author contributions
Conceptualization, A.F.O. and S.A.A.; data curation, D.S.L. and M.J.M; formal analysis, D.S.L. and M.J.M.; investigation, A.F.O. and D.S.L.; methodology, A.F.O., D.S.L., and I.I.; project administration, A.F.O., S.A.A., I.I., and M.N.A.S.; software, M.N.A.S. and M.J.M; supervision, A.F.O. and S.A.A.; writing—original draft, D.S.L.; writ-ing—review and editing, A.F.O. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author would like to acknowledge the support from the Fundamental Research Grant Scheme (FRGS) under a grant no: FRGS/1/2019/TK10/UNIMAP/03/2 from the Ministry of Education Malaysia.
