Abstract
2,5-tetrahydrofurandimethanol (THFDM), which is an alicyclic diol in products of 5-hydroxymethylfurfural (HMF), is a sustainable monomer to protect environment for replacing the role of isophthalic acid (IPA) and 1,4-cyclohexanedimethanol (CHDM) which were optimized polyethylene terephthalate (PET) performance. By direct esterification method, poly (ethylene-co-tetrahydrofurandimethanol terephthalate) (PETT) was synthesized in 2.5 L reactor. The results showed that the contents of THFDM in copolyester were less than that feeding ratio attributed to its lower reaction reactivity. The color of PETT slightly turned yellow. PETT performed broader processing temperature window, while Tg maintained in good range because of the cyclic structure of THFDM. Especially, the cold crystallization occured in PETT but not in PET after finishing melt crystallization. Otherwise, the thermal stability and crystal structure of PETT remained the same as PET. The crystallization activity of PETT became significantly lower than PET. What proved THFDM was effective in reducing PET crystallinity was that crystallization kinetics using Jeziorny theory and the trend of activation energy of melt crystallization which was calculated by Friedman method. And PETT-2 fibre had comparable mechanical property to PET fibre. In the advancement of green chemistry, THFDM has added a choice of cyclic bio-based comonomers.
Keywords
Introduction
Polyethylene terephthalate (PET) was currently one of the widely used plastics, mainly in beverage bottles, packaging, and textiles. Because PET was semi-crystalline polymer, high crystallinity in productions reduced transparency and caused brittler. 1 Isophthalic acid (IPA), a comonomer, had been used in the production of PET since the beginning of the 21st century.2–4 2 mol% IPA was usually incorporated into PET to inhibit its crystallization activity for improving the transparency of the end products, 5 reducing the processing temperature, and making processing more easily in injection and blow moulding.6–8 As fibre, PET was improved moisture absorption and dyeing properties after polymerizing with IPA. 9 1,4-Cyclohexanedimethanol (CHDM) was another comonomer which gave the copolyesters better toughness and higher Tg because of its alicyclic structure.10,11 Poly (ethylene isophthalate-co-ethylene terephthalate) (PEIT) was accessed to market over 20 years, poly (ethylene glycol-co-1,4-cyclohexanedimethanol terephthalate) (PETG) was also successfully commercialised for over 10 years. Tg of PETG was the highest in this three copolyesters (PETG > PET > PEIT) with maintaining high transparency and great mechanical properties.12,13 PETG was mainly used in sheet, cosmetic packaging and heat shrinkable films. The reason that IPA and CHDM inhibited crystallization activity was that partially replacing the ethylene glycol (EG) unit in terephthalic acid (TPA) could reduce the unit-regularity of PET. 14 However, IPA is a structural isomer of TPA, and CHDM is produced by the hydrogenation of TPA, which is derived from non-renewable petroleum resources. It was contrary to the advocated concept of reducing anthropogenic greenhouse gas (GHG) and sustainable development. 15 On the other hand, rising price of crude oil day after day would be bound to affect the price of the final product. Therefore, how to transit from fossil energy to bio-resources is the focus of current research. 16
The mass production of 5-hydroxymethylfurfural (HMF), one of the 10-ten-most valuable bio-based platform compounds by the Department of Energy (DOE) in America, had brought the “furan family” into the mass market. 17 Bio-based Poly (ethylene furanoate) (PEF) considered as an ideal candidate for fossil-based PET due to the similarity of the furan ring to the benzene ring.18–21 2,5-dihydroxymethylfuran (DHMF) which is homologous diol of 2,5-furandicarboxylic acid (FDCA) is used for choosing a comonomer for PET. 19 But very poor thermal stability of BHMF didn’t allow synthesize copolyesters by direct esterification that was the method of industrial production for polyester.12,22,23 2,5-tetrahydrofurandimethanol (THFDM) that had better thermal stability than DHMF was produced by the hydrogenation of FDCA, just as the relationship between TPA to CHDM. 24 Today kilogram-Scale production THFDM has been successful from 5-HMF catalyzed with Ni/SiO2.25,26 There were some differences in ring size, polarity, and linearity between THFDM and CHDM though they were pretty similar. The angle and interatomic distance between two carboxylic acid groups in THFDM was closer to IPA, but not CHDM (THFDM: 129°, 4.830 Å. IPA: 120°, 4.4 Å. CHDM: 180°, 5.731 Å.).13,27 Moreover, the structure of THFDM was nonlinear molecules containing ether linkages. Ether linkages gave strong polarity, hydrophilicity, and biocompatibility to polyesters, and nonlinear structure was more rigid that produced high Tg and great mechanical properties.28–30
In this study, THFDM was introduced into the PET as a sustainable copolymer monomer to synthesize poly (ethylene-co-tetrahydrofurandimethanol terephthalate) (PETT) for replacing the previous role of CHDM or IPA. Due to the high price of THFDM, only a small amount (≤6 mol%) of THFDM was added during copolymerisation to design a new sustainable copolyester with similar properties to PET but with easier processing and less crystallization activity. In polymer processing, such as spinning, injection moulding and extrusion, was a non-isothermal crystallisation process that would impact on the properties of the final products.31–33 So the choice of non-isothermal crystallisation was more relevant to the actual production environment. Subsequently studying the effects of different cooling rates on the crystallisation, kinetics and activation energy of the melt crystallisation were investigated to find the optimum addition ratio of THFDM, with the aim of laying the foundation for future series production.
Experiment
Materials
Ethylene glycol, TPA, and Sb2O3 were provided by Wankai New Material Co., Ltd. THFDM was purchased from Zhejiang Sugar Energy Technology Co., Ltd. Triphenyl phosphate (TPP) was purchased from Shanghai Technology and Trade Co., Ltd. All the chemicals were used without further purification.
Synthesize PETT
Poly (ethylene-co-tetrahydrofurandimethanol terephthalate) was synthesized from TPA, EG and THFDM in two steps: pre-esterification and polycondensation in a 2.5 L reactor (Scheme 1). In the pre-esterification stage, the reactor was preheated to 150°C. TPA, EG, and THFDM (total acid/diol = 1/1.2, mol%), the stabilizer TPP (0.1 g), and the catalyst Sb2O3 (0.1 mol%, based on TPA) were fed. Then the temperature was risen to 250°C, at the same time, the pressure in the reactor was maintained at 200–400 kPa for 3 h. After removal of the by-product water, the temperature was increased to 275°C with vacuuming to 5–10 Pa for 1 h. The intrinsic viscosity ([η]) of the system reached above 0.6 dL/g and the power of the stirring paddle increased significantly could be also regarded as the destination of the reaction. Finally, Synthesis and chemical structures of PETTs. Characterization of PET and PETTs.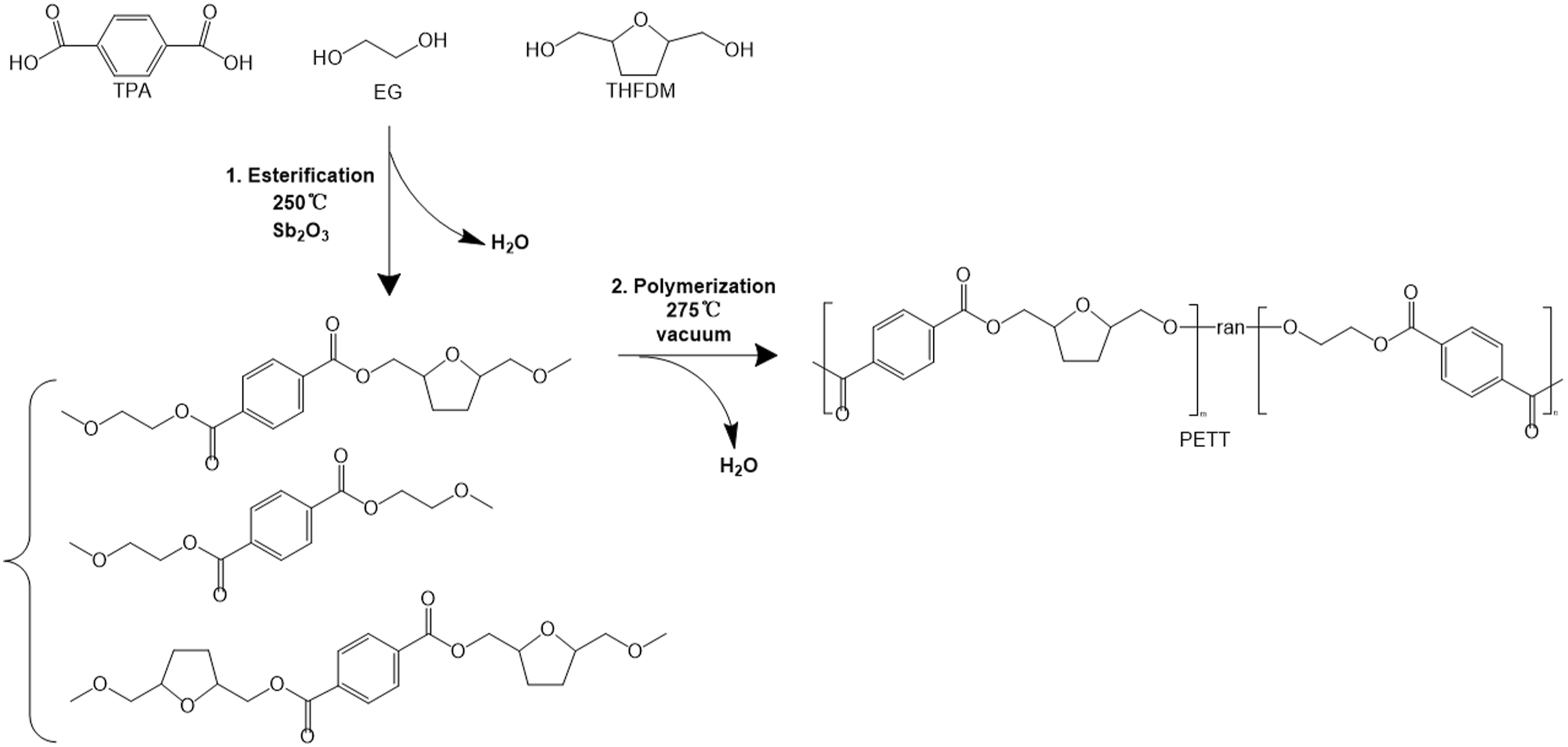
Melt spinning
Polyethylene terephthalate and PETTs were dried in drum vacuum dryer (JM-500ZGX) at 150°C during 12 h before melt spinning. For melt spinning, the temperatures were set up from 280 to 290°C. Then hot drawing ratio was 3.0, the temperatures were 90°C, 100°C and 140°C. Final speed of taken-up was 1000 m/min. The linear density of any one fibre was 155 dtex.
Characterization
Polyethylene terephthalate and PETTs were dissolved in CF3COOD (TFA, δ = 11.5 ppm). The composition of samples were confirmed by 1H NMR (AVANCE AV400 MHz, Bruker).
[η] were measured by an automatic viscometer (NCY-4, Shanghai S.R.D Scientific Instrument Co., Ltd) with a mixed solution of phenol and tetrachloroethane (60/40, w/w). The Mη was calculated from η using the Mark-Houwink equation34–36 (1) and the results were retained to three significant digits:
The samples were dissolved in the solution of phenol and trichloromethane (40/60, v/v). The carboxyl end-group concentration content (-COOH) of the copolyesters was obtained by titration with a standard potassium hydroxide-ethanol titration solution.
The samples were reacted transesterification with methanol at 210°C for 2 h. The diethylene glycol content (DEG) of the filtrate was then determined by gas chromatography (TRACE1310, ThermoFisher).
Colorimater (Color View, BYK) was used to obtain the average of colour value (Lab.) at 3 times spinning around 120° every test.
The water contact angles (WCAs) were measured by recording the lateral profiles of water droplets in three different areas on the same polyester flake using a contact angle meter (JY-82B, Chengdu Lingdu Instruments Co., Ltd) for image analysis. The process of preparing polyester flake was that samples were pressed at 255°C and 10 MPa for 5 min with a vulcameter (QLLHY-25Y, UNLONG), then cooled at room temperature to obtain polyester flakes.
Thermal properties of samples were measured with a differential scanning calorimeter (DSC) (DSC-400, Perkin Elmer) under
Thermal stability was performed with 3 mg for every sample heated from 25 to 550°C at a heating rate of 10°C/min under
The crystal structure of copolyesters was showed by X-ray diffraction (XRD) patterns. Using X-ray diffractometer (APL-XTRA, Thermo ARL) scanned flakes of samples by Cu-Kα rays (λ = 0.154 nm) that 40 kV and 200 mA was every step for 0.02 in 2θ range from 5° to 60°.
The mechanical properties of the fibres were carried out using the multifilament yarn strength tester (YG023C -III, shuanggudadun Co. Ltd, Changzhou, China). Here, the pre-tension was 0.05 ± 0.005 cN/dtex, the distance between two fixtures was 100 mm, and the tensile speed was set to 100 mm/min for mechanical testing. Each mechanical data was the average value after testing 10 groups of fibres to ensure accuracy and repeatability.
Non-isothermal melt crystallization
The non-isothermal melt crystallization behavior of PET and PETT copolyesters were studied with DSC (DSC400, Perkin Elmer). The samples with weight of about 5–10 mg was placed in an aluminum pan, and the measurement was taken under nitrogen atmosphere. This testing procedures were divided into 3 steps: (1) The samples were heated from 30 to 275°C at a heating rate of 10°C/min (the first heating) and isothermally maintain at 275°C for 5 min to eliminate thermal history. (2) Subsequently, they were cooled to 30°C at various φ (the cooling rate represented by φ) of 5, 10 and 20°C/min and isothermally maintain at 30°C for 5 min (the cooing). (3) Finally, the temperature was raised to 275°C (the second heating). The cooling and the second heating curves were respectively recorded to show the dependence of the melt crystallization behavior on the cooling rate. Second, the cooling curves were converted into those of the relative crystallinity as a function of time to study the non-isothermal melt crystallization kinetics. Meanwhile, the activation energy was calculated.
Results and discussion
THFDM contents in PETTs and characterization
To determine the chemical composition of PET and PETTs, 1H NMR was performed, and the results were shown in Figure 1 (a) 1H NMR spectra and characteristic peak assignment of PETTs, (b) enlarged region of 2.0–2.6 ppm and 4.0–4.8 ppm.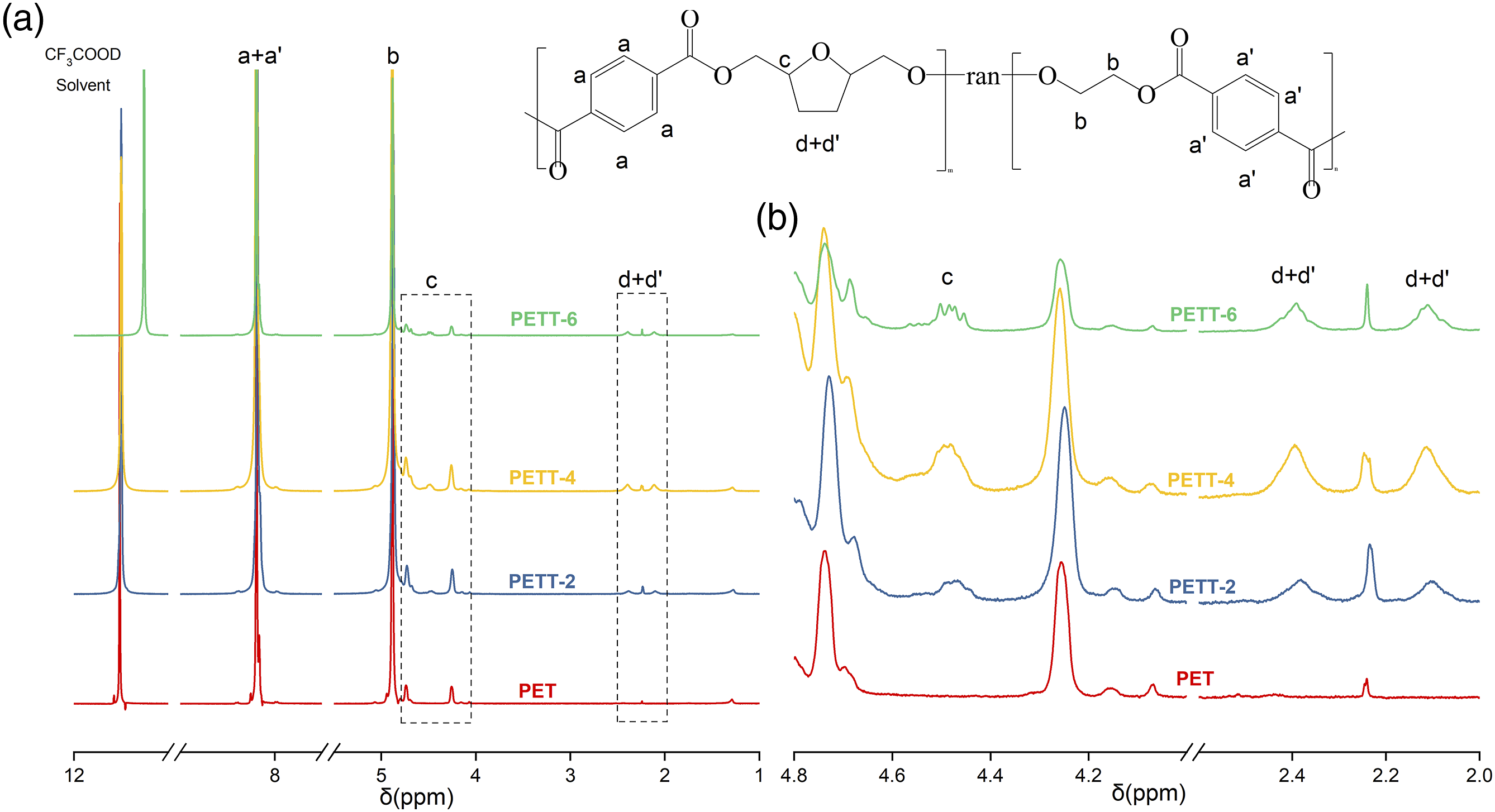


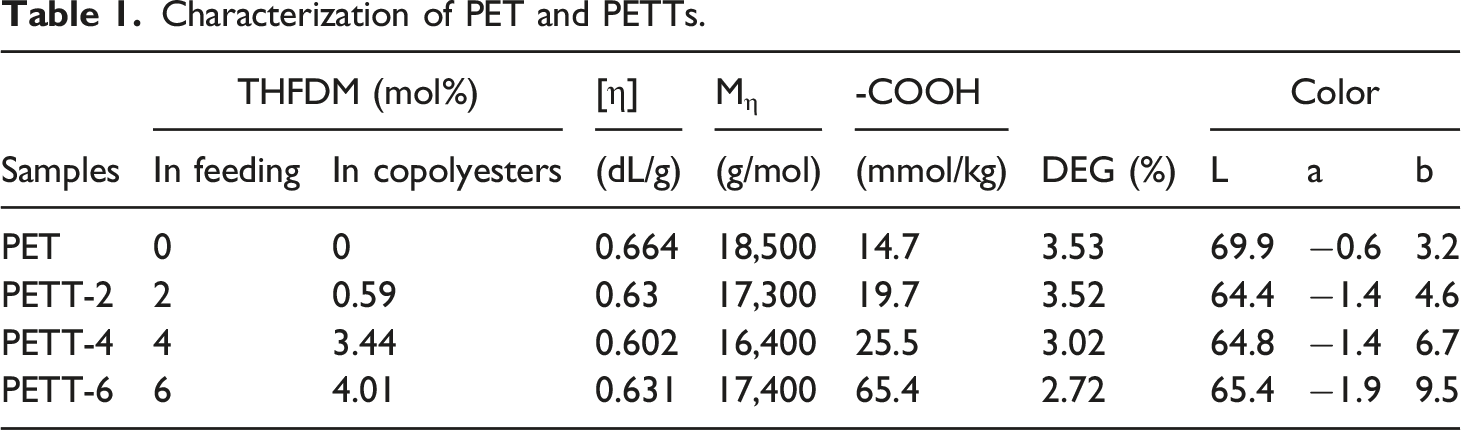
From Table 1, it was found that the content of THFDM in the copolymer was lower than the content in feeding, due to the more strengthening steric effect from THFDM. Although [η] of PETT was lower comparing with PET as the increase of THFDM in feeding ratio, all [η] exceeded 0.6 dL/g. High [η] would represent high molecular weight to meet requirements of follow-up productions.
-COOH is an important indicator of the quality of polyester melt, which affects the spinning stability to polyesters. Low -COOH will cause more fibre break in the spinning process. To PETT-4, its -COOH was the highest in four samples (25.5 mmol/kg) that also suggested the most complete alcohol reaction during production.
DEG that is a by-product in the polycondensation reflects the extent to the side reaction taking place. The ether linkage formed by DEG broke the chain structure of PETT, the cause that reduced the crystallinity and Tm, also enhanced the polarity. The DEG had turned less and less as more and more THFDM in feeding, but the sum of them gradually increased to lead the same effect.
In Table 1, the color of polyesters would be yellowish if b > 0. And photos were listed in Figure 2 The photos of polyesters (5 g for every sample, from left to right: PET PETT-2 PETT-4 PETT-6).
Wettability property
Determining wettability properties need compare their WCAs in Figure 3 The water contact angle of PET and PETTs, as well as the picture that water drops on the polymeric film surface.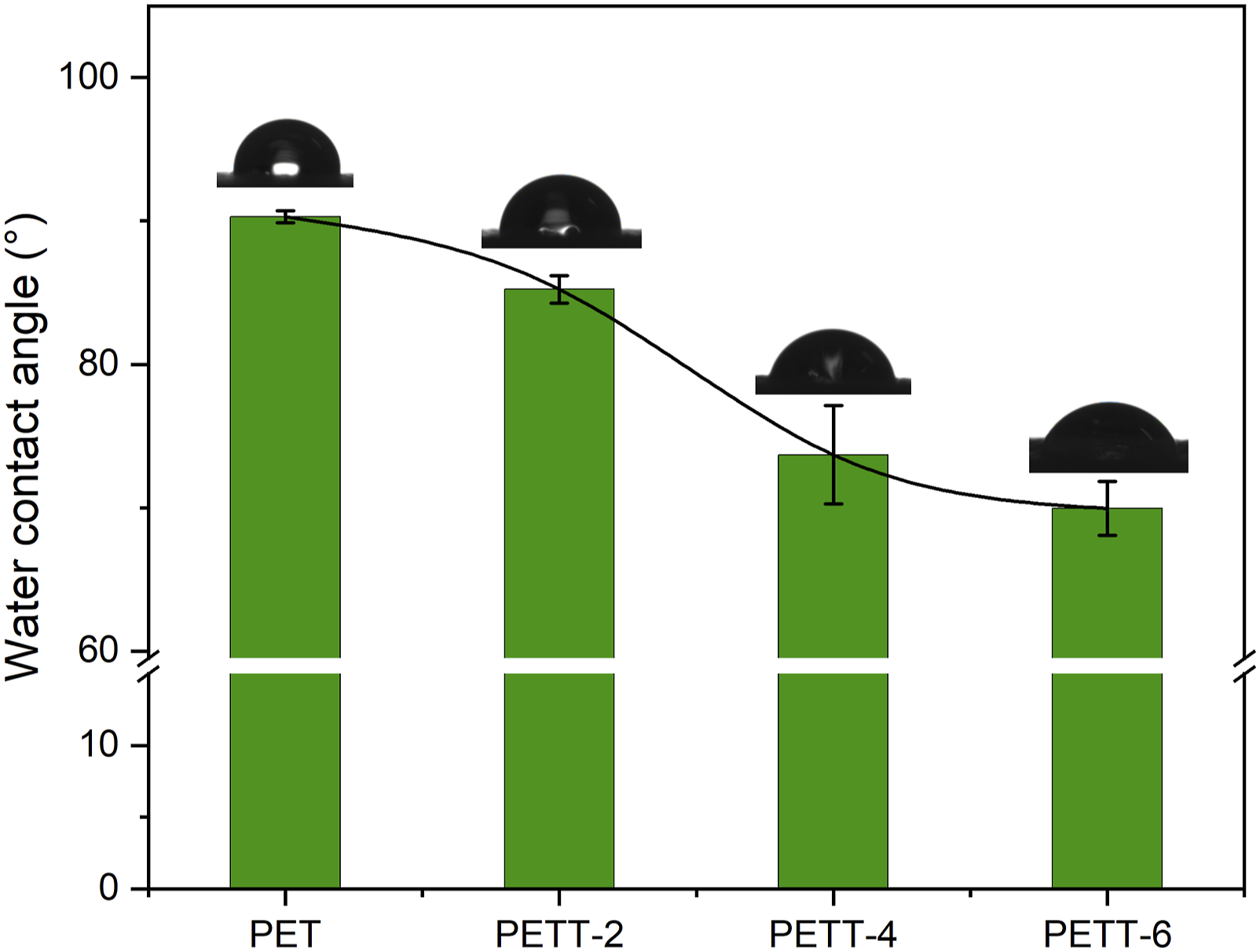
Thermal properties and crystalline structure
The melting and crystallisation of PET and PETTs were studied by DSC. The cooling and second heating curves were respectively shown in Figure 4(a) and (b) DSC curves for PET and PETTs: (a) cooling curves from 270 to 30°C at the cooling rate of 10°C/min, (b) second heating scans from 30 to 270°C at the heating rate of 10°C/min, (c) enlarged region of cold crystallization at the second. (d) XRD curves of PET and PETTs. (e) TGA and (f) DTG curves of PET and PETTs. Thermal properties of PET and PETTs.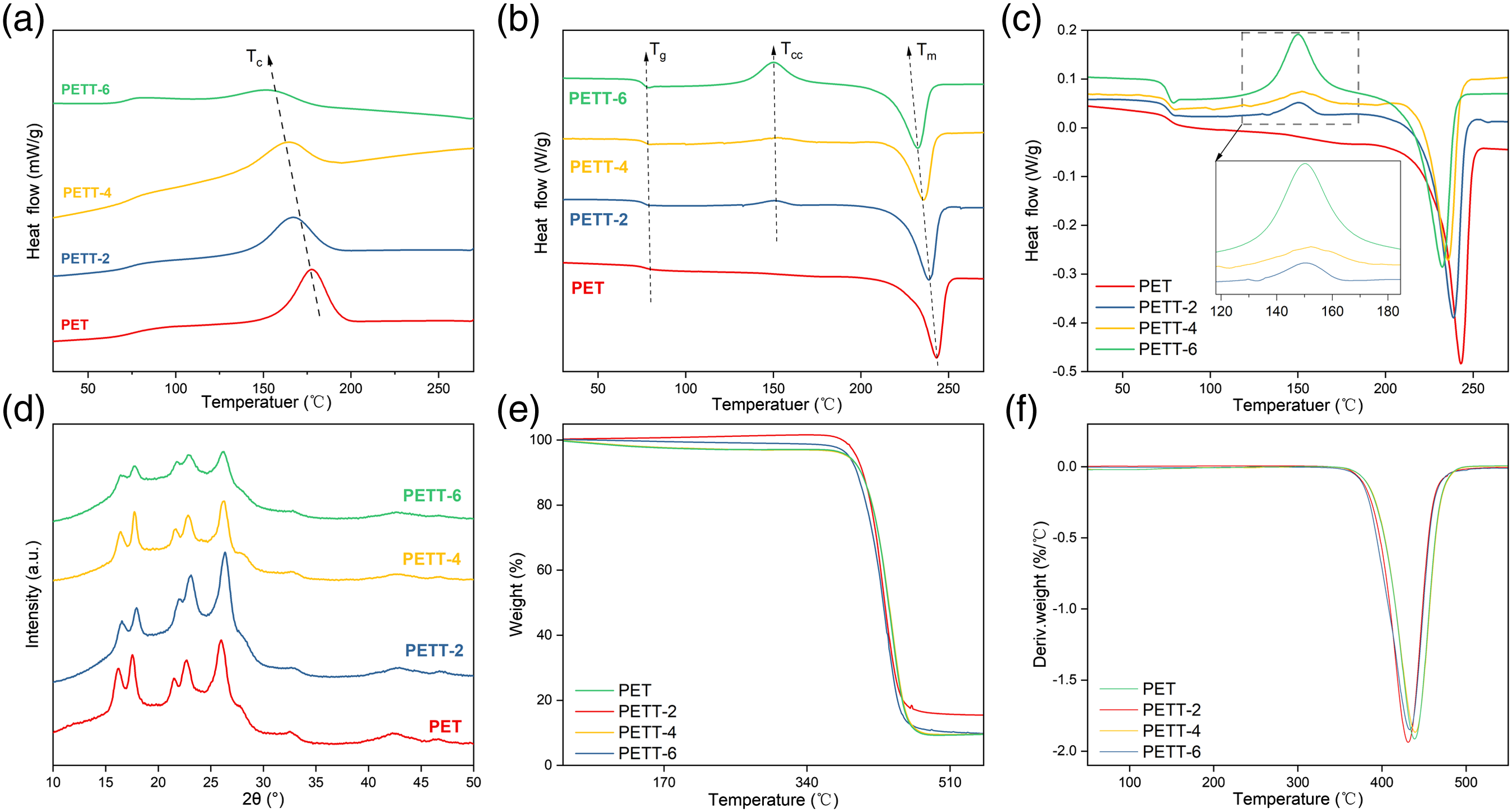
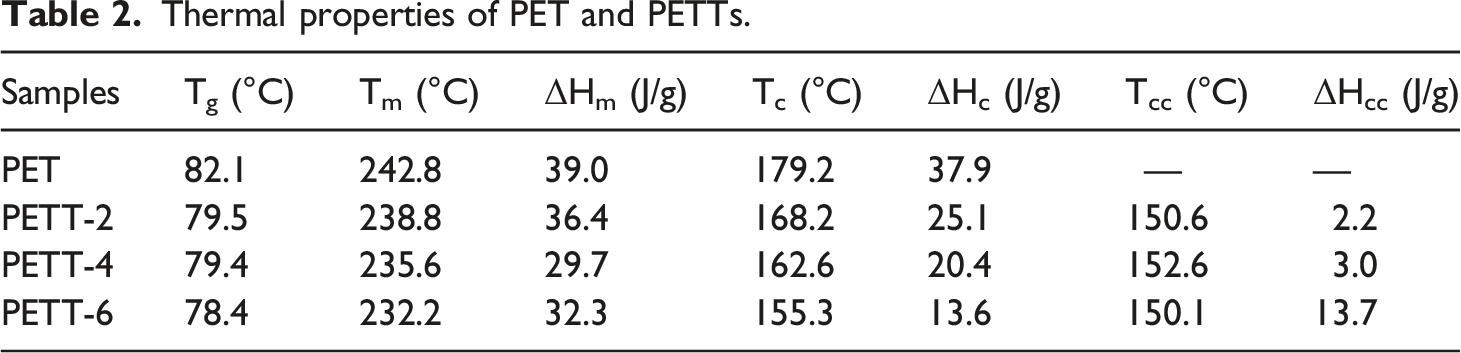
The cooling curves showed Tc and ΔHc decreased continuously with increasing THFDM content (179.2°C → 155.3°C, 39.0 J/g → 32.3 J/g). All polyesters in the second heating curves showed glass transition and melt peaks, with both Tg and Tm similarly decreasing. PETT-4 had the smallest ΔHm reaching 29.7 J/g. The Tg of PETT was lower than that of PET but only from 82.1°C to 78.4°C (max: PET vs min: PETT-6), which was also sufficient for daily use. That the Tm reduced from 242.8°C to 232.2°C illustrated the addition of THFDM diminished the processing temperature of PET. However, the maximum D-value in Tm was less pronounced than that in Tc (10.6°C vs 23.9°C), suggesting that THFDM greater effect on crystallisation, but not melt. The other evident supporting this point: PETT had a clear cold crystallisation peak compared to PET in Figure 4(c). The cold crystallization peak occurred because the segmental chain motion was so slow that it could not crystallize during cooling and start moving again after the second heating. It was no doubt that this chain with extremely slow motion were TT units. The bottom width of the cold crystallization peak became larger and ΔHcc became with the TT unit content increasing, while the melt crystallization ability of PETT decreased. Even ΔHcc and ΔHc were similar in PETT-6. Furthermore, the above conclusions could demonstrate that the crystallization ability of PETT can be regulated by controlling the ratio of THFDM in feeding.
Thermal stability evaluation and relative crystallinity of PET and PETTs.

Figure 4(d) showed the XRD patterns of PET and PETTs. The diffraction peaks of PET appeared at 2θ = 16° (010), 18° (010), 21° (011), 23° (100) and 26° (111). The diffraction peaks of the PETT were only slightly shifted towards the high angle region compared to the PET ones, due to the distortion of the unit cell caused after the addition of the TT unit and the decrease of the lattice constant α. For PETT-6, the intensity of the diffraction peaks was notably lower. It could be concluded that THFDM only reduces the crystallinity of PETT at low ratios without changing the crystal structure. In previous studies on the crystallinity of copolyesters, if the ratio of comonomers need to be over 30 mol%, the production will be obtained as amorphous morphologies.39–42
Thermogravimetric analyser and DTG curves were shown in Figure 4(e) and (f). The maximum decomposition rate temperature (Td,max), the temperature at which 5% of the weight was lost (Td,5%) and proportion of residual mass at 550°C (R550) were summarised in Table 2. The TGA curves show a single trend about decomposition. And the TT units didn’t change the thermal decomposition mechanism. The Td,max appear about 440°C due to the massive decomposition of the benzene ring. Td,max increased slightly with the addition of the TT unit, while Td,5% showed a decreasing trend (minimum: 388°C). PETT didn’t decompose until 300°C, which would ensure stability during melt processing. R550 decreased from 15.45% to 9.53%, but there was no guide to actual processing.
Non-isothermal melt and cold crystallization behavior of PET and PETTs at cooling and the second heating processes
The non-isothermal crystallisation behaviours of PET and PETTs were studied at cooling rates (φ) of 5, 10 and 20°C/min respectively, showing in Figure 5 Non-isothermal crystallization cureves at various cooling rates: (a) PET, (b) PETT-2, (c) PETT-4, and (d) PETT-6.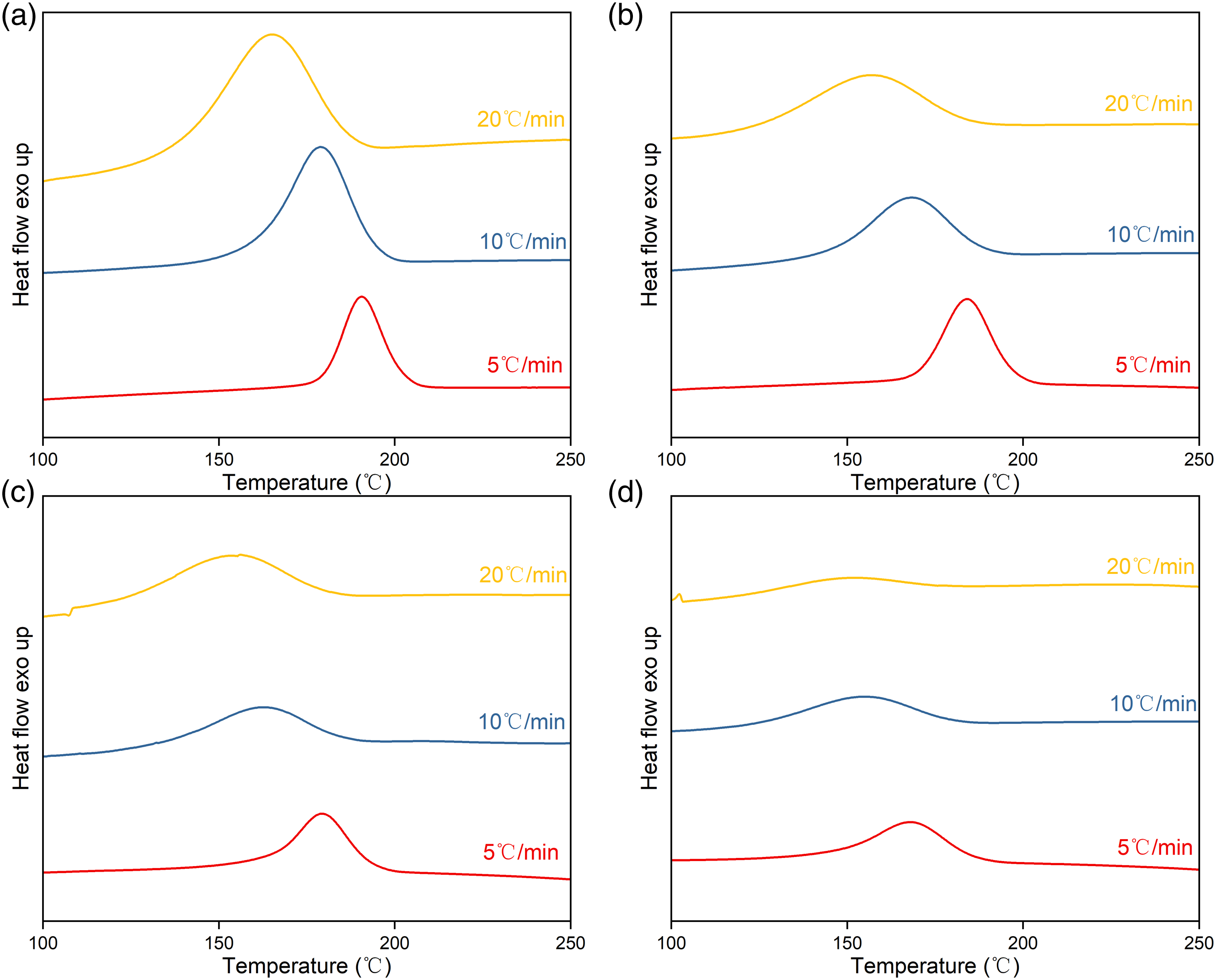
There were three excuses to explain. Firstly, the symmetry of chain was destroyed by TT units. Secondly, actual tetrahydrofuran ring-flipping in TT units was frustrated by the nonlinear axis of tetrahydrofuran ring rotation coupled with the ring polarity assuming larger intermolecular force. Thirdly, slower chain mobility required a larger ΔT to complete crystallisation.41,43,44
The samples were reheated to melt at φ = 10°C/min after non-isothermal crystallisation, shown in Figure 6 Melt curves after non-isothermal crystallization at the heating rates of 10°C/min: (a) PET, (b) PETT-2, (c) PETT-4, (d) PETT-6.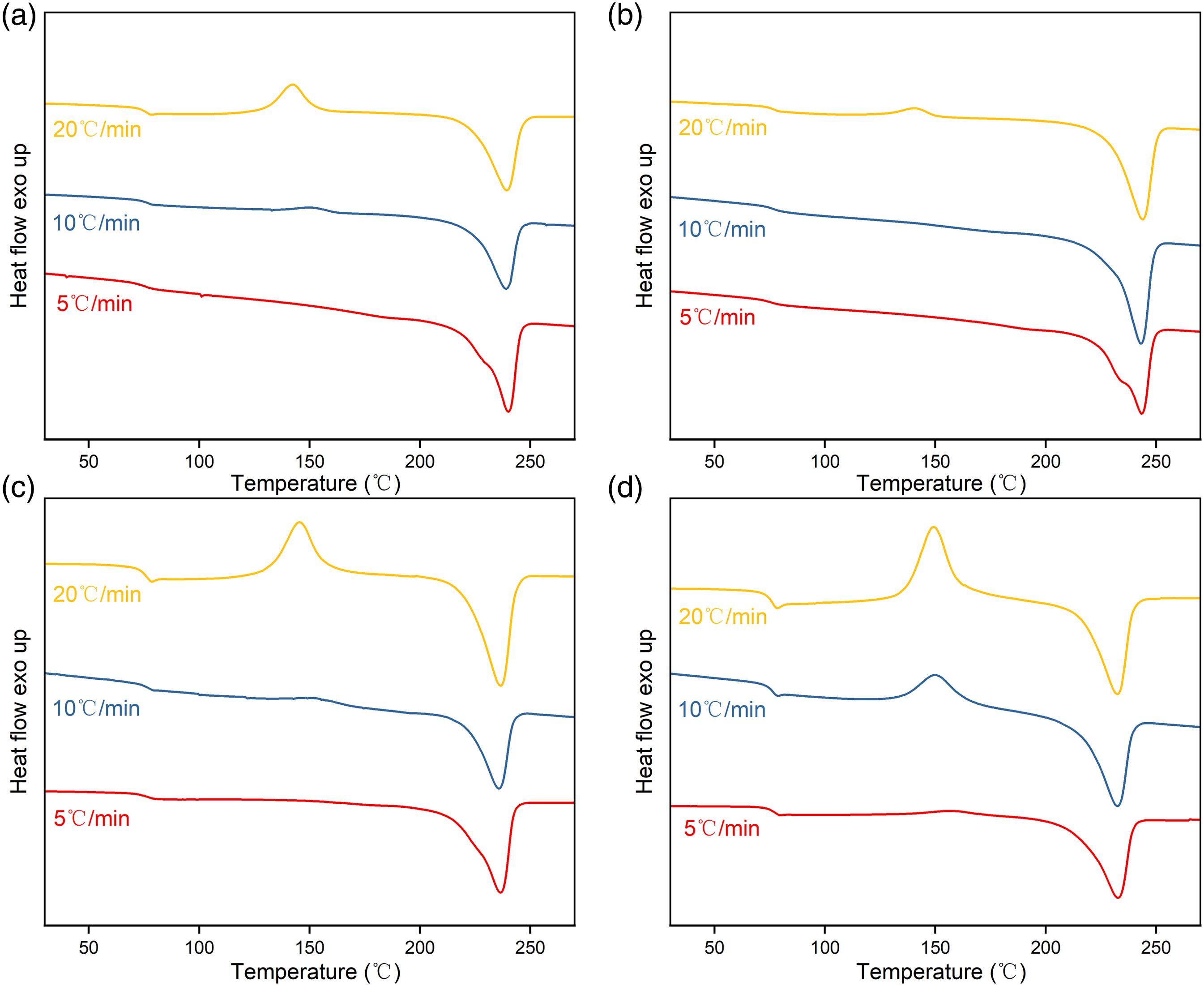
Xc,melt in the non-isothermal crystallisation were calculated again by equation (4). The trend of Xc,melt was in Figure 7(b) (a) ΔT in cooling; (b) crystallinity of melt.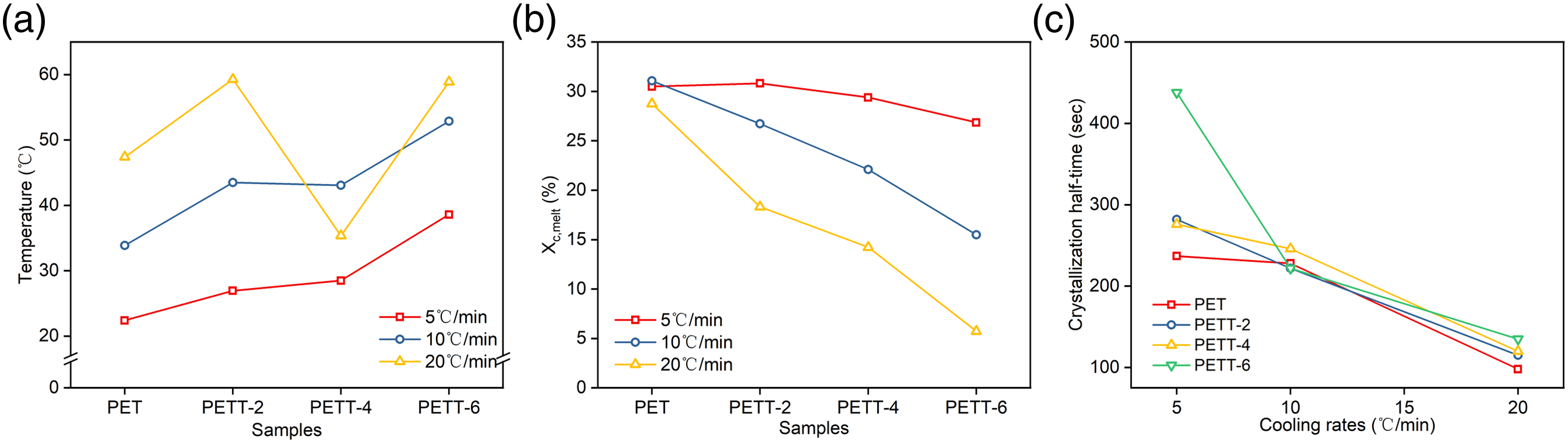
Kinetics of non-isothermal melt crystallization
Crystallization half-time
For a complete crystallization process, the relative crystallinity (Xt) as a function of time (t) can be expressed as in equation (5) and shown in Figure 7(c):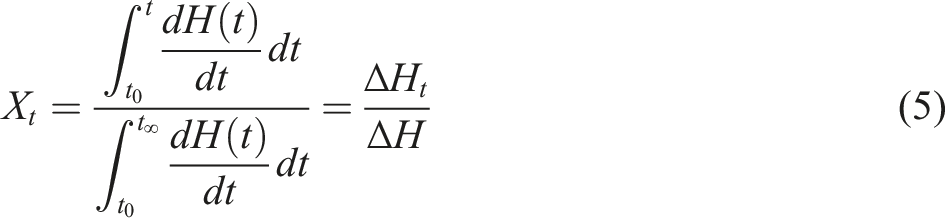

The semi-crystallisation time (t1/2), defined as the time taken from beginning crystallisation to Xt = 50%, is used to compare crystallisation rate. This value can be obtained directly from Figure 8 Plots of Xt versus time of samples for non-isothermal melt crystallization at various cooling rates: (a) 5 C/min, (b) 10 C/min, (c)20 C/min.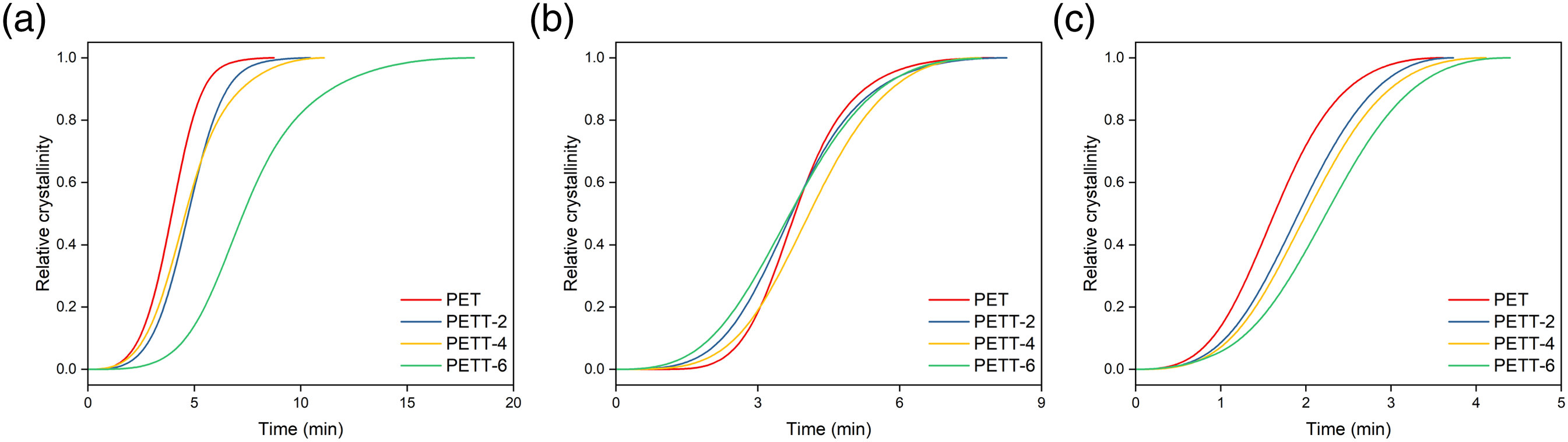
Jeziorny theory
The non-isothermal melt crystallisation kinetics of PET and PETT was described by Jeziorny theory45,46 based on the Avrami equation (7) which was based on Xc to calculate the crystallization rate (k) and the value of the Avrami index ( Relationship between of lg [-ln (1-Xt)] and lg(t) at different crystallization cooling rates for PET and PETTs: (a) 5°C/min, (b) 10°C/min, (c) 20°C/min.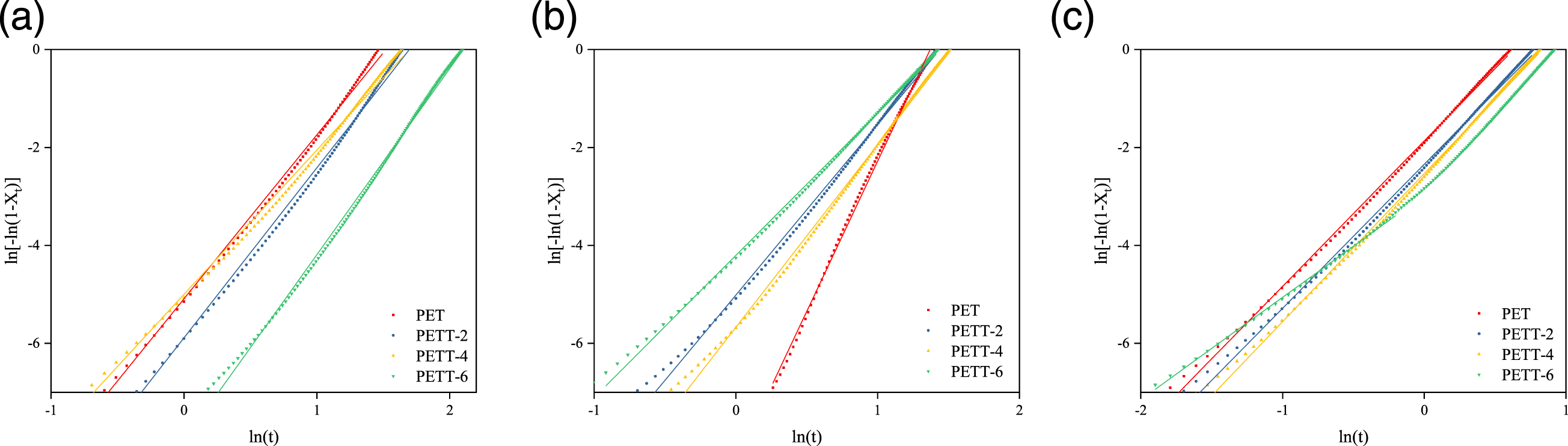

Taking the logarithm of both sides yields to equation (8):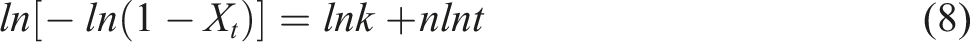
φ is introduced to recalculate the crystallisation rate k′ for non-isothermal crystallisation, as equation (9).
Avrami parameters of the PET and PETTs determined from Jeziorny’s theory.
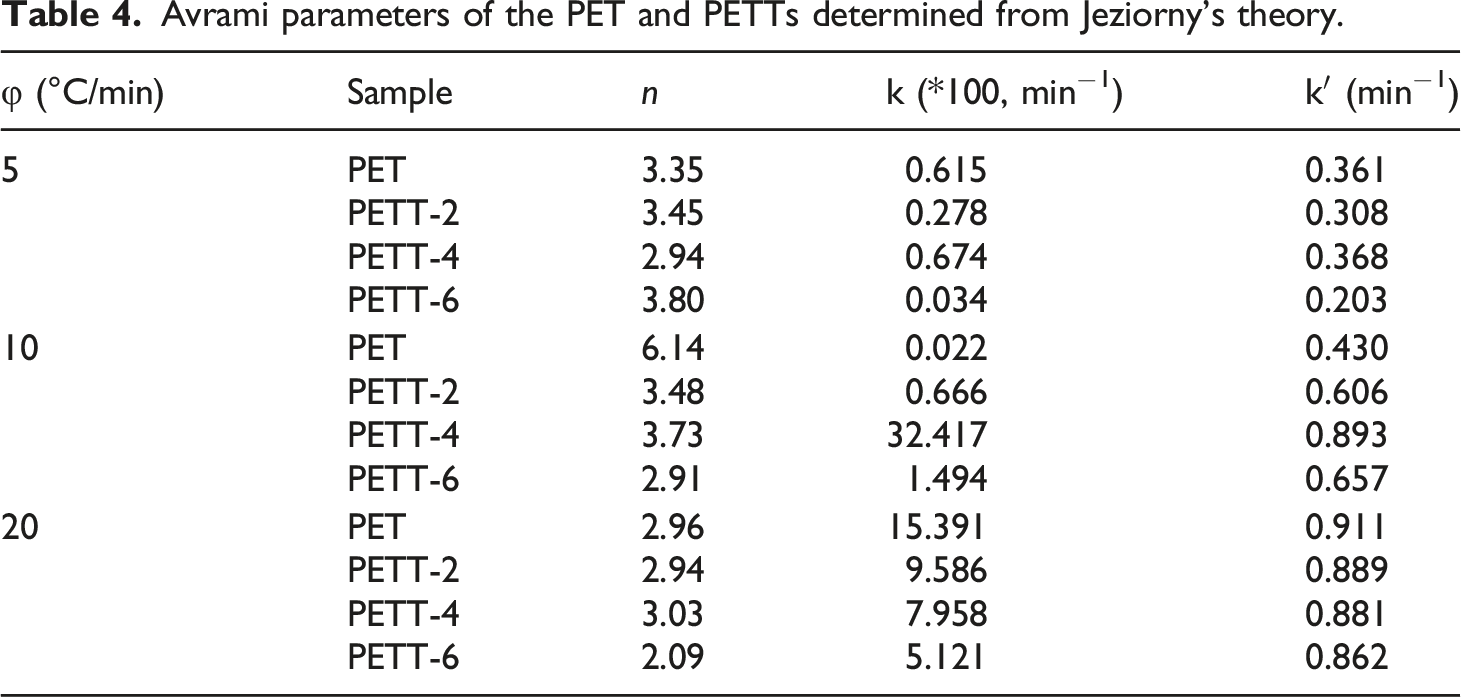
As we all know, copolyesters are homogeneous nucleation, at which point the time dimension is 1, therefore the crystal is in a one-dimensional growth mode, but this is counterfactual. What had been proved from the other side was that PETT-6 can barely crystallise under this condition φ = 20°C/min. At high crystallisation rates brought about by high cooling rates, the sphaerocrystal grew faster making them bump and crowd each other, or crystals tended to grow in two dimensions. k’ could compare to crystallization rate again. PETT-6 also was the slowest crystallising sample.
Activation energy of melt crystallization
The activation energy (ΔE) of melt crystallization is the energy required for the formation of nuclei during crystallisation and the phase change energy required for the chain segments to become regularly aligned into the crystalline phase from disorder. The commonly used methods for calculating ΔE are Kissinger method
47
and the Friedman method.48,49 Kissinger method is not accurate for cooling processes,
50
so Friedman method is a more appropriate choice, as following equation (10): Dependence of the activation energy on the relative crystallinity for PET and PETTs. Plots of ln (dXT/dt) versus 1/T: (a) PET, (b) PETT-2, (c) PETT-4, (d) PETT-6.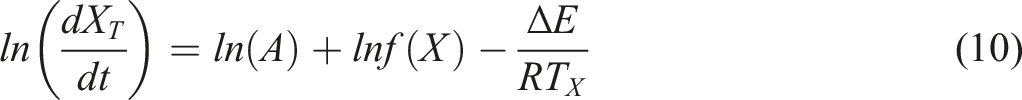
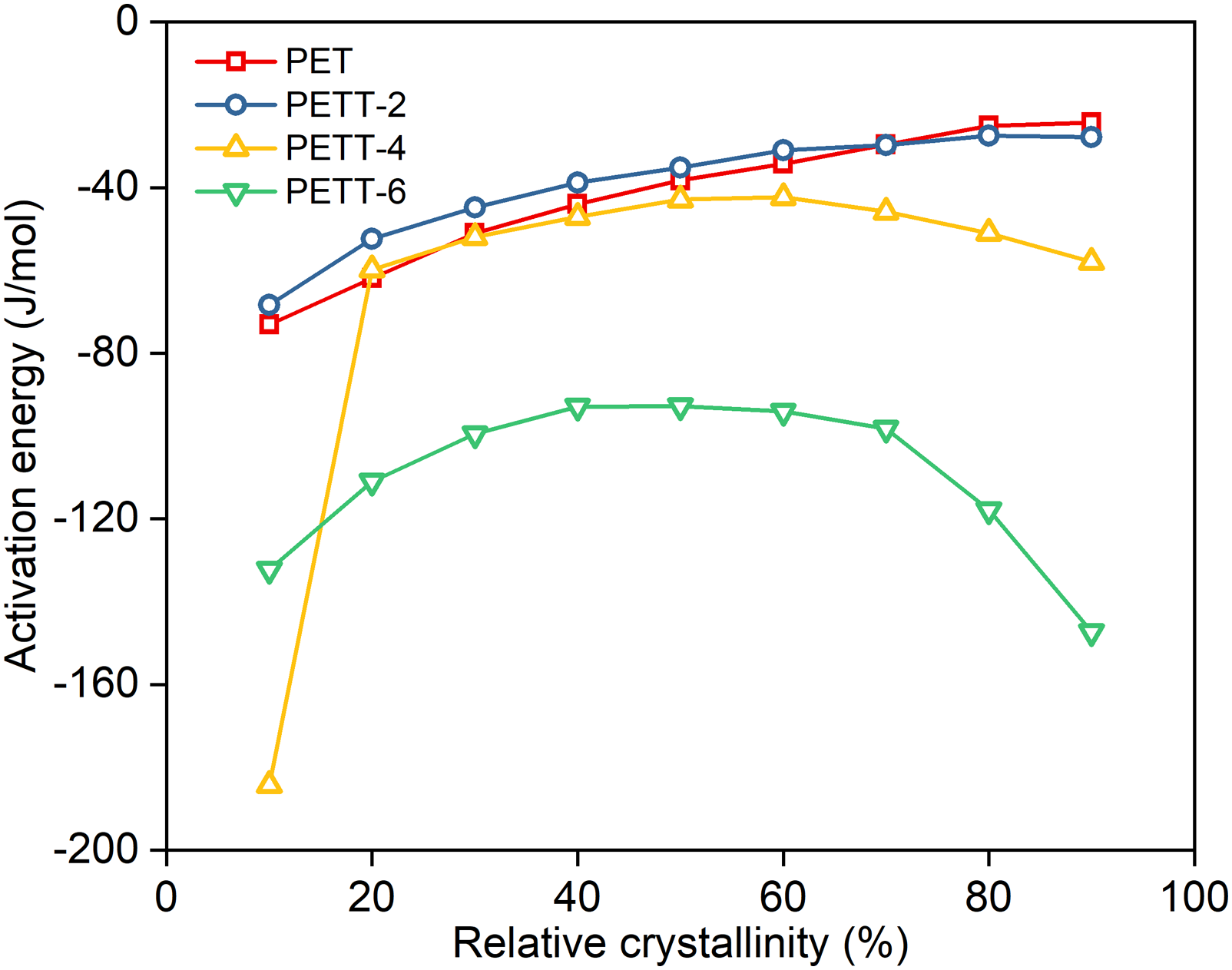
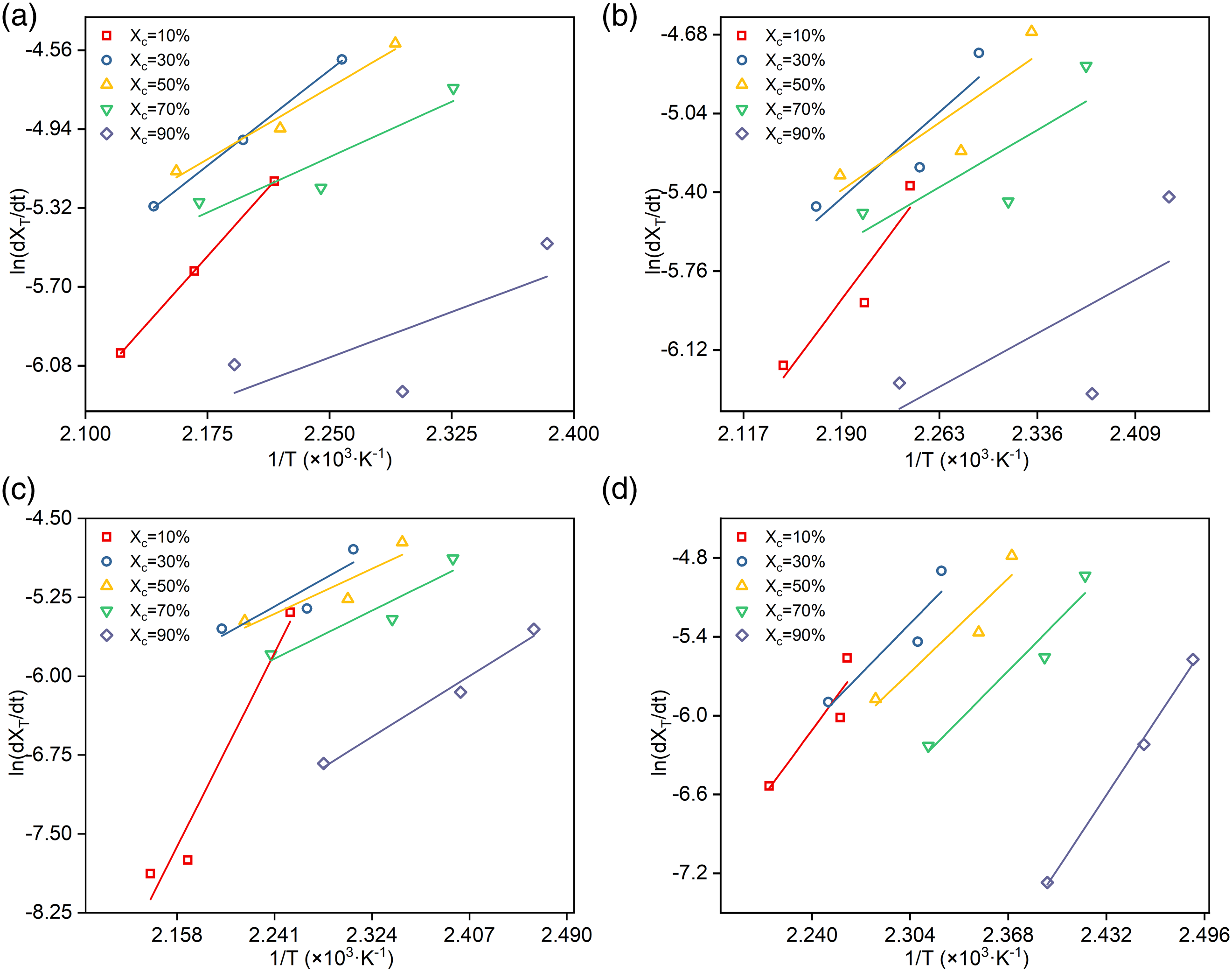
ΔE reflects the temperature dependence of the crystallisation, but not a direct assessment of crystallisation rate. The larger |ΔE| is, the more temperature sensitive crystallisation is. In Figure 10, ΔE increased monotonically with crystallisation for PET and PETT-2. Nevertheless, ΔE of PETT-4 and PETT-6 showed the different trend. They increased and then decreased, with reaching the maximum at Xc = 50 – 60%. Both|ΔE| were lower than the previous two in each stage. For semi-crystalline polymer, the decreasing |ΔE| in crystallisation indicates that the energy required to overcome the phase transition is decreasing and crystal growth becomes easier and less temperature dependent.51–54 From this, it could be concluded that the crystallisation mechanism of copolyesters had become more sensitive to temperature when THFDM additions exceeded 4%mol. In other words, only if temperatures reach Tc will their crystal grew. The other phenomenon could be seen from the Figure 10: the trend of PET was like a straight line, but others were obviously curved. And these lines became more and more curved with increased THFDM addition, especially the trend of PETT-6 showed a “∩”-shape. It also meant that PET crystal grew in a large temperature range, but PETT-6 crystal just grew at Tc. Thus PETT-6 crystal can’t grow with temperature passing fast Tc.
Mechanical property of PETT fibres
The breaking strength and elongation at break of PET and PETTs fibres were shown in Figure 12 The breaking strength and elongation at break of PET and PETTs fliers.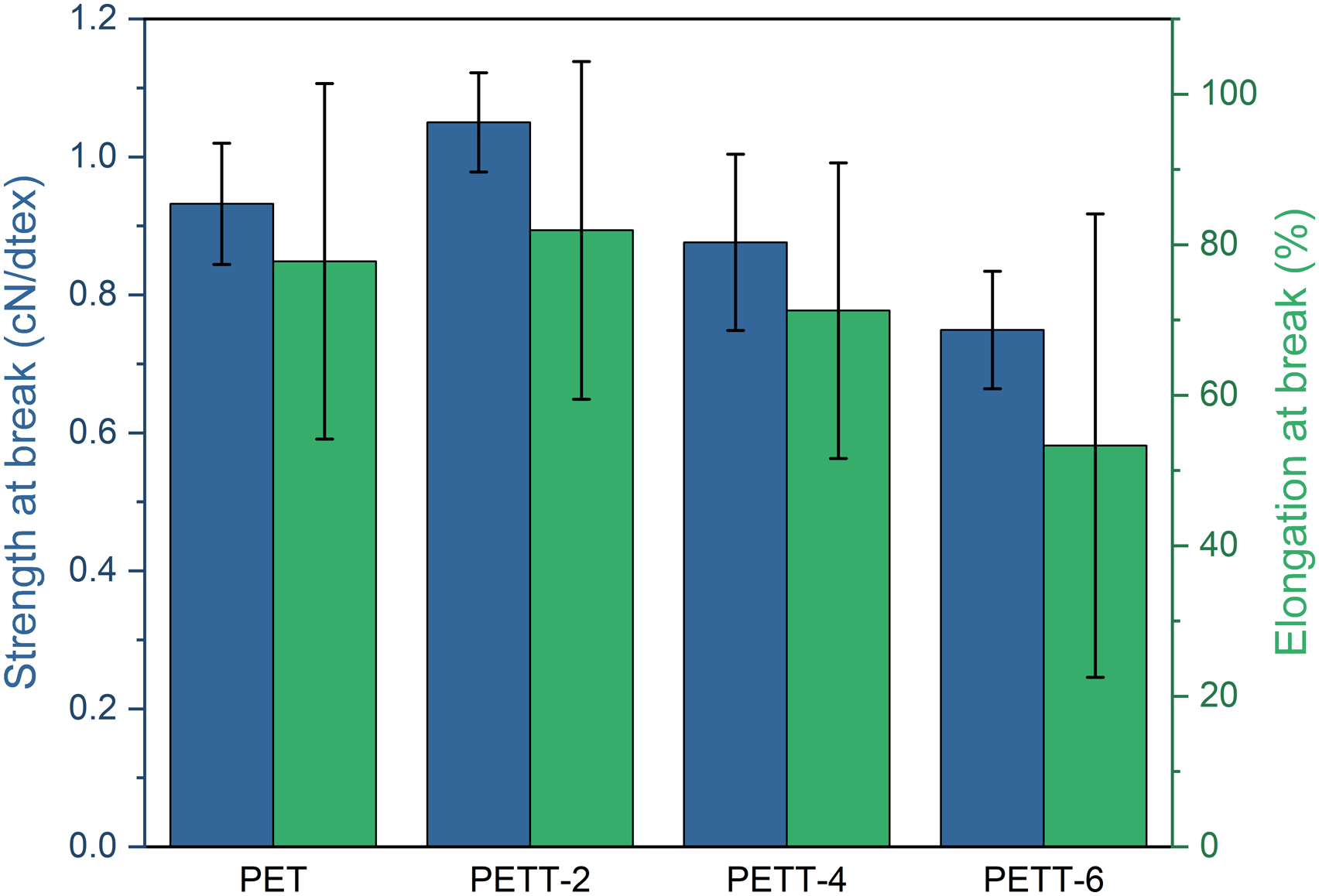
Conclusions
In this work, a bio-based alicyclic diol THFDM was introduced into PET, synthesizing PETT by direct esterification that is the mainstream method of Industrial producing PET nowadays. The influence by THFDM on copolyesters performance was clarified. All [η] exceeded 0.6 dL/g for satisfying the needs of daily use. The color of PETT became faint yellow. THFDM containing ether linkage increased the hydrophilicity. Tm, Tc, and corresponding entropy decreased with the contribution of THFDM so that PETT performed broader processing temperature window, while Tg maintained in good range (78.4°C ∼ 82.1°C) because the cyclic structure of THFDM kept rigidity of molecular chain. Besides, THFDM greater effected on crystallisation so the non-isothermal melt crystallisation of PETT had been focused research. The intensity (31.21% → 15.43%) and capacities of gradually decreased, due to PET chain regular arrangement destroyed by THFDM. The crystallization of PETT-6 was more strongly dependent on the heating rate than it of PET. The non-isothermal kinetics of samples were investigated by using Jeziorny theory. THFDM was effective in reducing the crystallinity (t1/2 at φ = 5°C/min: 438 s → 237 s). Calculations by Jeziorny theory also gave the same results. Activation energy of melt crystallization was fitted by Friedman mothed. |ΔE| trend suggested PETT-4 and PETT-6 crystallised most easily at Xc = 50 ∼ 60% that was around Tc, but PET and PETT-2 crystallised more and more easily with the crystallisation proceeding. Finally, the content of THFDM could regulate the crystallization of PETT and further realized the adjustment of the thermal. In the hand of mechanical property, PETT-2 had comparable to PET as fibre, but PETT-4 and PETT-6 were weaker than PET because of the reducing crystallinity. In a word, THFDM has added a choice in comonomers for the development of green chemistry.
Footnotes
Author contributions
Conceptualization: Shunhua Zhang; experimentation: Yang Liu and Zhiwei Chen; data analysis: Yang Liu; writing: Yang Liu; revision: Xinmin Pu and Shunhua Zhang; supervision: Bing Zheng and Shunhua Zhang.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Zhejiang Provincial Key Research and Development Project (No. 2021C01062).
